Crithmum maritimum L.: Phytochemical Profile, Biological Activities, and Therapeutic Potential
Abstract
1. Introduction
2. Results
3. Discussion
3.1. Therapeutic Potential of the EO
3.2. Therapeutic Potential of the Extracts
4. Materials and Methods
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zengin, G.; Mahomoodally, M.F.; Paksoy, M.Y.; Picot-Allain, C.; Glamocilja, J.; Sokovic, M.; Diuzheva, A.; Jekő, J.; Cziáky, Z.; Rodrigues, M.J.; et al. Phytochemical Characterization and Bioactivities of Five Apiaceae Species: Natural Sources for Novel Ingredients. Ind. Crops Prod. 2019, 135, 107–121. [Google Scholar] [CrossRef]
- Thiviya, P.; Gunawardena, N.; Gamage, A.; Madhujith, T.; Merah, O. Apiaceae Family as a Valuable Source of Biocidal Components and Their Potential Uses in Agriculture. Horticulturae 2022, 8, 614. [Google Scholar] [CrossRef]
- Wang, X.-J.; Luo, Q.; Li, T.; Meng, P.-H.; Pu, Y.-T.; Liu, J.-X.; Zhang, J.; Liu, H.; Tan, G.-F.; Xiong, A.-S. Origin, Evolution, Breeding, and Omics of Apiaceae: A Family of Vegetables and Medicinal Plants. Hortic. Res. 2022, 9, uhac076. [Google Scholar] [CrossRef]
- Sayed-Ahmad, B.; Talou, T.; Saad, Z.; Hijazi, A.; Merah, O. The Apiaceae: Ethnomedicinal Family as Source for Industrial Uses. Ind. Crops Prod. 2017, 109, 661–671. [Google Scholar] [CrossRef]
- Atia, A.; Debez, A.; Barhoumi, Z.; Pacini, E.; Abdelly, C.; Smaoui, A. The Mericarp of the Halophyte Crithmum maritimum (Apiaceae): Structural Features, Germination, and Salt Distribution. Biologia 2010, 65, 489–495. [Google Scholar] [CrossRef]
- Dzhoglova, V.; Ivanov, K.; Benbassat, N.; Georgieva-Dimova, Y.; Ardasheva, R.; Karcheva-Bahchevanska, D.; Ivanova, S. Crithmum maritimum L.—Study on the Histochemical Localization of Essential Oil. Plants 2024, 13, 550. [Google Scholar] [CrossRef] [PubMed]
- Kraouia, M.; Nartea, A.; Maoloni, A.; Osimani, A.; Garofalo, C.; Fanesi, B.; Ismaiel, L.; Aquilanti, L.; Pacetti, D. Sea Fennel (Crithmum maritimum L.) as an Emerging Crop for the Manufacturing of Innovative Foods and Nutraceuticals. Molecules 2023, 28, 4741. [Google Scholar] [CrossRef]
- Renna, M. Reviewing the Prospects of Sea Fennel (Crithmum maritimum L.) as Emerging Vegetable Crop. Plants 2018, 7, 92. [Google Scholar] [CrossRef] [PubMed]
- Zenobi, S.; Fiorentini, M.; Zitti, S.; Aquilanti, L.; Foligni, R.; Mannozzi, C.; Mozzon, M.; Orsini, R. Crithmum maritimum L.: First Results on Phenological Development and Biomass Production in Mediterranean Areas. Agronomy 2021, 11, 773. [Google Scholar] [CrossRef]
- Glenn, E.P.; Brown, J.J.; Blumwald, E. Salt Tolerance and Crop Potential of Halophytes. Crit. Rev. Plant Sci. 1999, 18, 227–255. [Google Scholar] [CrossRef]
- Petropoulos, S.A.; Karkanis, A.; Martins, N.; Ferreira, I.C.F.R. Edible Halophytes of the Mediterranean Basin: Potential Candidates for Novel Food Products. Trends Food Sci. Technol. 2018, 74, 69–84. [Google Scholar] [CrossRef]
- Flowers, T.J.; Colmer, T.D. Plant Salt Tolerance: Adaptations in Halophytes. Ann. Bot. 2015, 115, 327–331. [Google Scholar] [CrossRef] [PubMed]
- Accogli, R.; Nutricati, E.; De Bellis, L.; Renna, M.; Luvisi, A.; Negro, C. Diversity of Crithmum maritimum L. from Salento Coastal Area: A Suitable Species for Domestication. Horticulturae 2024, 10, 81. [Google Scholar] [CrossRef]
- Flowers, T.J.; Galal, H.K.; Bromham, L. Evolution of Halophytes: Multiple Origins of Salt Tolerance in Land Plants. Funct. Plant Biol. 2010, 37, 604. [Google Scholar] [CrossRef]
- Ventura, Y.; Eshel, A.; Pasternak, D.; Sagi, M. The Development of Halophyte-Based Agriculture: Past and Present. Ann. Bot. 2015, 115, 529–540. [Google Scholar] [CrossRef]
- Nartea, A.; Orhotohwo, O.L.; Fanesi, B.; Lucci, P.; Loizzo, M.R.; Tundis, R.; Aquilanti, L.; Casavecchia, S.; Quattrini, G.; Pacetti, D. Sea Fennel (Crithmum maritimum L.) Leaves and Flowers: Bioactive Compounds, Antioxidant Activity and Hypoglycaemic Potential. Food Biosci. 2023, 56, 103417. [Google Scholar] [CrossRef]
- Franke, W. Vitamin C in Sea Fennel (Crithmum maritimum), an Edible Wild Plant. Econ. Bot. 1982, 36, 163–165. [Google Scholar] [CrossRef]
- Orhotohwo, O.L.; Nartea, A.; Lucci, P.; Jaiswal, A.K.; Jaiswal, S.; Pacetti, D. Application of Sea Fennel’s Bioactive Compounds in the Development of Edible Films and Coatings: A Review. Food Biosci. 2024, 61, 104843. [Google Scholar] [CrossRef]
- Karkanis, A.; Polyzos, N.; Kompocholi, M.; Petropoulos, S.A. Rock Samphire, a Candidate Crop for Saline Agriculture: Cropping Practices, Chemical Composition and Health Effects. Appl. Sci. 2022, 12, 737. [Google Scholar] [CrossRef]
- Senatore, F.; Napolitano, F.; Ozcan, M. Composition and Antibacterial Activity of the Essential Oil from Crithmum maritimum L. (Apiaceae) Growing Wild in Turkey. Flavour Fragr. J. 2000, 15, 186–189. [Google Scholar] [CrossRef]
- Ersanli, C.; Tzora, A.; Skoufos, I.; Fotou, K.; Maloupa, E.; Grigoriadou, K.; Voidarou, C.; Zeugolis, D.I. The Assessment of Antimicrobial and Anti-Biofilm Activity of Essential Oils against Staphylococcus Aureus Strains. Antibiotics 2023, 12, 384. [Google Scholar] [CrossRef] [PubMed]
- Marongiu, B.; Maxia, A.; Piras, A.; Porcedda, S.; Tuveri, E.; Gonçalves, M.J.; Cavaleiro, C.; Salgueiro, L. Isolation of Crithmum maritimum L. Volatile Oil by Supercritical Carbon Dioxide Extraction and Biological Assays. Nat. Prod. Res. 2007, 21, 1145–1150. [Google Scholar] [CrossRef] [PubMed]
- Houta, O.; Akrout, A.; Najja, H.; Neffati, M.; Amri, H. Chemical Composition, Antioxidant and Antimicrobial Activities of Essential Oil from Crithmum maritimum Cultivated in Tunisia. J. Essent. Oil Bear. Plants 2015, 18, 1459–1466. [Google Scholar] [CrossRef]
- Generalić Mekinić, I.; Blažević, I.; Mudnić, I.; Burčul, F.; Grga, M.; Skroza, D.; Jerčić, I.; Ljubenkov, I.; Boban, M.; Miloš, M.; et al. Sea Fennel (Crithmum maritimum L.): Phytochemical Profile, Antioxidative, Cholinesterase Inhibitory and Vasodilatory Activity. J. Food Sci. Technol. 2016, 53, 3104–3112. [Google Scholar] [CrossRef]
- Alves-Silva, J.M.; Guerra, I.; Gonçalves, M.J.; Cavaleiro, C.; Cruz, M.T.; Figueirinha, A.; Salgueiro, L. Chemical Composition of Crithmum maritimum L. Essential Oil and Hydrodistillation Residual Water by GC-MS and HPLC-DAD-MS/MS, and Their Biological Activities. Ind. Crops Prod. 2020, 149, 112329. [Google Scholar] [CrossRef]
- Ismail, A.; El-Shibani, F.A.; Mohammed, H.A.; Al-Najjar, B.O.; Korkor, A.M.; Abdulkarim, A.K.; Said, R.; Almahmoud, S.A.; Sulaiman, G.M. Chemical Composition, Antioxidant, and Enzyme Inhibition Activities of Crithmum maritimum Essential Oils: The First Chemo-Biological Study for Species Grown in North Africa. Sci. Rep. 2024, 14, 25318. [Google Scholar] [CrossRef]
- Beeby, E.; Magalhães, M.; Poças, J.; Collins, T.; Lemos, M.F.L.; Barros, L.; Ferreira, I.C.F.R.; Cabral, C.; Pires, I.M. Secondary Metabolites (Essential Oils) from Sand-Dune Plants Induce Cytotoxic Effects in Cancer Cells. J. Ethnopharmacol. 2020, 258, 112803. [Google Scholar] [CrossRef]
- Souid, A.; Della Croce, C.M.; Frassinetti, S.; Gabriele, M.; Pozzo, L.; Ciardi, M.; Abdelly, C.; Hamed, K.B.; Magné, C.; Longo, V. Nutraceutical Potential of Leaf Hydro-Ethanolic Extract of the Edible Halophyte Crithmum maritimum L. Molecules 2021, 26, 5380. Molecules 2021, 26, 5380. [Google Scholar] [CrossRef]
- Meot-Duros, L.; Cérantola, S.; Talarmin, H.; Le Meur, C.; Le Floch, G.; Magné, C. New Antibacterial and Cytotoxic Activities of Falcarindiol Isolated in Crithmum maritimum L. Leaf Extract. Food Chem. Toxicol. 2010, 48, 553–557. [Google Scholar] [CrossRef]
- Correia, I.; Antunes, M.; Tecelão, C.; Neves, M.; Pires, C.L.; Cruz, P.F.; Rodrigues, M.; Peralta, C.C.; Pereira, C.D.; Reboredo, F.; et al. Nutritive Value and Bioactivities of a Halophyte Edible Plant: Crithmum maritimum L. (Sea Fennel). Plants 2024, 13, 427. [Google Scholar] [CrossRef]
- Nabet, N.; Boudries, H.; Chougui, N.; Loupassaki, S.; Souagui, S.; Burló, F.; Hernández, F.; Carbonell-Barrachina, Á.A.; Madani, K.; Larbat, R. Biological Activities and Secondary Compound Composition from Crithmum maritimum Aerial Parts. Int. J. Food Prop. 2017, 20, 1843–1855. [Google Scholar] [CrossRef]
- Houta, O.; Akrout, A.; Neffati, M.; Amri, H. Phenolic Contents, Antioxidant and Antimicrobial Potentials of Crithmum maritimum Cultivated in Tunisia Arid Zones. J. Biol. Act. Prod. Nat. 2011, 1, 138–143. [Google Scholar] [CrossRef]
- Alemán, A.; Marín-Peñalver, D.; De Palencia, P.F.; Gómez-Guillén, M.D.C.; Montero, P. Anti-Inflammatory Properties, Bioaccessibility and Intestinal Absorption of Sea Fennel (Crithmum maritimum) Extract Encapsulated in Soy Phosphatidylcholine Liposomes. Nutrients 2022, 14, 210. [Google Scholar] [CrossRef]
- Gnocchi, D.; Cesari, G.; Calabrese, G.J.; Capone, R.; Sabbà, C.; Mazzocca, A. Inhibition of Hepatocellular Carcinoma Growth by Ethyl Acetate Extracts of Apulian Brassica Oleracea L. and Crithmum maritimum L. Plant Foods Hum. Nutr. 2020, 75, 33–40. [Google Scholar] [CrossRef]
- Huang, H.; Qian, J.; Zeng, L.; Zhang, J.; Peng, X.; Tang, X.; Tang, Y.; Fu, M.; Liu, Q.; Song, F.; et al. Full-Component Extract of Crithmum maritimum and Its Effect on Epidermal Regeneration. RSC Adv. 2025, 15, 1713–1720. [Google Scholar] [CrossRef]
- Pereira, C.G.; Moraes, C.B.; Franco, C.H.; Feltrin, C.; Grougnet, R.; Barbosa, E.G.; Panciera, M.; Correia, C.R.D.; Rodrigues, M.J.; Custódio, L. In Vitro Anti-Trypanosoma Cruzi Activity of Halophytes from Southern Portugal Reloaded: A Special Focus on Sea Fennel (Crithmum maritimum L.). Plants 2021, 10, 2235. [Google Scholar] [CrossRef]
- Souid, A.; Croce, C.M.D.; Pozzo, L.; Ciardi, M.; Giorgetti, L.; Gervasi, P.G.; Abdelly, C.; Magné, C.; Hamed, K.B.; Longo, V. Antioxidant Properties and Hepatoprotective Effect of the Edible Halophyte Crithmum maritimum L. against Carbon Tetrachloride-Induced Liver Injury in Rats. Eur. Food Res. Technol. 2020, 246, 1393–1403. [Google Scholar] [CrossRef]
- Caucanas, M.; Montastier, C.; Piérard, G.E.; Quatresooz, P. Dynamics of Skin Barrier Repair Following Preconditioning by a Biotechnology-Driven Extract from Samphire (Crithmum maritimum) Stem Cells: Samphire and Skin Barrier Repair. J. Cosmet. Dermatol. 2011, 10, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Simos, Y.V.; Zerikiotis, S.; Lekkas, P.; Athinodorou, A.-M.; Zachariou, C.; Tzima, C.; Assariotakis, A.; Peschos, D.; Tsamis, K.; Halabalaki, M.; et al. Oral Supplementation with Hydroxytyrosol Synthesized Using Genetically Modified Escherichia Coli Strains and Essential Oils Mixture: A Pilot Study on the Safety and Biological Activity. Microorganisms 2023, 11, 770. [Google Scholar] [CrossRef]
- Politeo, O.; Ćurlin, P.; Brzović, P.; Auzende, K.; Magné, C.; Generalić Mekinić, I. Volatiles from French and Croatian Sea Fennel Ecotypes: Chemical Profiles and the Antioxidant, Antimicrobial and Antiageing Activity of Essential Oils and Hydrolates. Foods 2024, 13, 695. [Google Scholar] [CrossRef]
- Gnocchi, D.; Nikolic, D.; Paparella, R.R.; Sabbà, C.; Mazzocca, A. Crithmum maritimum Extract Restores Lipid Homeostasis and Metabolic Profile of Liver Cancer Cells to a Normal Phenotype. Plant Foods Hum. Nutr. 2024, 79, 417–424. [Google Scholar] [CrossRef] [PubMed]
- Guil-Guerrero, J.L.; Rodríguez-García, I. Lipids Classes, Fatty Acids and Carotenes of the Leaves of Six Edible Wild Plants. Eur. Food Res. Technol. 1999, 209, 313–316. [Google Scholar] [CrossRef]
- Pereira, C.G.; Barreira, L.; Da Rosa Neng, N.; Nogueira, J.M.F.; Marques, C.; Santos, T.F.; Varela, J.; Custódio, L. Searching for New Sources of Innovative Products for the Food Industry within Halophyte Aromatic Plants: In Vitro Antioxidant Activity and Phenolic and Mineral Contents of Infusions and Decoctions of Crithmum maritimum L. Food Chem. Toxicol. 2017, 107, 581–589. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Upadhyay, S.; Erdogan Orhan, I.; Kumar Jugran, A.; Jayaweera, S.L.D.; Dias, D.A.; Sharopov, F.; Taheri, Y.; Martins, N.; Baghalpour, N.; et al. Therapeutic Potential of α- and β-Pinene: A Miracle Gift of Nature. Biomolecules 2019, 9, 738. [Google Scholar] [CrossRef] [PubMed]
- Vespermann, K.A.C.; Paulino, B.N.; Barcelos, M.C.S.; Pessôa, M.G.; Pastore, G.M.; Molina, G. Biotransformation of α- and β-Pinene into Flavor Compounds. Appl. Microbiol. Biotechnol. 2017, 101, 1805–1817. [Google Scholar] [CrossRef]
- Silva, A.C.R.D.; Lopes, P.M.; Azevedo, M.M.B.D.; Costa, D.C.M.; Alviano, C.S.; Alviano, D.S. Biological Activities of A-Pinene and β-Pinene Enantiomers. Molecules 2012, 17, 6305–6316. [Google Scholar] [CrossRef]
- Guzmán-Gutiérrez, S.L.; Bonilla-Jaime, H.; Gómez-Cansino, R.; Reyes-Chilpa, R. Linalool and β-Pinene Exert Their Antidepressant-like Activity through the Monoaminergic Pathway. Life Sci. 2015, 128, 24–29. [Google Scholar] [CrossRef]
- Weston-Green, K.; Clunas, H.; Jimenez Naranjo, C. A Review of the Potential Use of Pinene and Linalool as Terpene-Based Medicines for Brain Health: Discovering Novel Therapeutics in the Flavours and Fragrances of Cannabis. Front. Psychiatry 2021, 12, 583211. [Google Scholar] [CrossRef]
- Rozza, A.L.; Moraes, T.D.M.; Kushima, H.; Tanimoto, A.; Marques, M.O.M.; Bauab, T.M.; Hiruma-Lima, C.A.; Pellizzon, C.H. Gastroprotective Mechanisms of Citrus lemon (Rutaceae) Essential Oil and Its Majority Compounds Limonene and β-Pinene: Involvement of Heat-Shock Protein-70, Vasoactive Intestinal Peptide, Glutathione, Sulfhydryl Compounds, Nitric Oxide and Prostaglandin E2. Chem.-Biol. Interact. 2011, 189, 82–89. [Google Scholar] [CrossRef]
- Ancuceanu, R.; Anghel, A.I.; Hovaneț, M.V.; Ciobanu, A.-M.; Lascu, B.E.; Dinu, M. Antioxidant Activity of Essential Oils from Pinaceae Species. Antioxidants 2024, 13, 286. [Google Scholar] [CrossRef]
- Bond, A.D.; Davies, J.E. γ-Terpinene. Acta Crystallogr. E Struct. Rep. Online 2001, 57, o1032–o1033. [Google Scholar] [CrossRef]
- Geron, C.; Rasmussen, R.; Arnts, R.R.; Guenther, A. A Review and Synthesis of Monoterpene Speciation from Forests in the United States. Atmos. Environ. 2000, 34, 1761–1781. [Google Scholar] [CrossRef]
- Ramalho, T.; Pacheco De Oliveira, M.; Lima, A.; Bezerra-Santos, C.; Piuvezam, M. Gamma-Terpinene Modulates Acute Inflammatory Response in Mice. Planta Med. 2015, 81, 1248–1254. [Google Scholar] [CrossRef] [PubMed]
- Li, G.-X.; Liu, Z.-Q. Unusual Antioxidant Behavior of α- and γ-Terpinene in Protecting Methyl Linoleate, DNA, and Erythrocyte. J. Agric. Food Chem. 2009, 57, 3943–3948. [Google Scholar] [CrossRef] [PubMed]
- Pina, L.T.S.; Rabelo, T.K.; Borges, L.P.; Gonçalves, V.S.S.; Silva, A.S.; Oliveira, M.A.; Quintans, J.S.S.; Quintans Júnior, L.J.; Scotti, L.; Scotti, M.T.; et al. Antihyperalgesic Effect of γ-Terpinene Complexed in β-Cyclodextrin on Neuropathic Pain Model Induced by Tumor Cells. Int. J. Pharm. 2024, 662, 124538. [Google Scholar] [CrossRef]
- Cristani, M.; D’Arrigo, M.; Mandalari, G.; Castelli, F.; Sarpietro, M.G.; Micieli, D.; Venuti, V.; Bisignano, G.; Saija, A.; Trombetta, D. Interaction of Four Monoterpenes Contained in Essential Oils with Model Membranes: Implications for Their Antibacterial Activity. J. Agric. Food Chem. 2007, 55, 6300–6308. [Google Scholar] [CrossRef]
- Waller, S.B.; Cleff, M.B.; Serra, E.F.; Silva, A.L.; Gomes, A.D.R.; De Mello, J.R.B.; De Faria, R.O.; Meireles, M.C.A. Plants from Lamiaceae Family as Source of Antifungal Molecules in Humane and Veterinary Medicine. Microb. Pathog. 2017, 104, 232–237. [Google Scholar] [CrossRef]
- Russo, R.; Corasaniti, M.T.; Bagetta, G.; Morrone, L.A. Exploitation of Cytotoxicity of Some Essential Oils for Translation in Cancer Therapy. Evid.-Based Complement. Altern. Med. 2015, 2015, 397821. [Google Scholar] [CrossRef]
- Corzo-Gómez, J.C.; Espinosa-Juárez, J.V.; Ovando-Zambrano, J.C.; Briones-Aranda, A.; Cruz-Salomón, A.; Esquinca-Avilés, H.A. A Review of Botanical Extracts with Repellent and Insecticidal Activity and Their Suitability for Managing Mosquito-Borne Disease Risk in Mexico. Pathogens 2024, 13, 737. [Google Scholar] [CrossRef]
- Eddin, L.B.; Jha, N.K.; Meeran, M.F.N.; Kesari, K.K.; Beiram, R.; Ojha, S. Neuroprotective Potential of Limonene and Limonene Containing Natural Products. Molecules 2021, 26, 4535. [Google Scholar] [CrossRef]
- Gupta, A.; Jeyakumar, E.; Lawrence, R. Strategic Approach of Multifaceted Antibacterial Mechanism of Limonene Traced in Escherichia Coli. Sci. Rep. 2021, 11, 13816. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Sun, Z.; Chen, W. Antimicrobial Susceptibility and Antibacterial Mechanism of Limonene against Listeria Monocytogenes. Molecules 2019, 25, 33. [Google Scholar] [CrossRef]
- Koeduka, T.; Watanabe, B.; Shirahama, K.; Nakayasu, M.; Suzuki, S.; Furuta, T.; Suzuki, H.; Matsui, K.; Kosaka, T.; Ozaki, S. Biosynthesis of Dillapiole/Apiole in Dill (Anethum graveolens): Characterization of Regioselective Phenylpropene O -methyltransferase. Plant J. 2023, 113, 562–575. [Google Scholar] [CrossRef]
- de Almeida, R.R.P.; Souto, R.N.P.; Bastos, C.N.; da Silva, M.H.L.; Maia, J.G.S. Chemical Variation in Piper aduncum and Biological Properties of Its Dillapiole-Rich Essential Oil. Chem. Biodivers. 2009, 6, 1427–1434. [Google Scholar] [CrossRef] [PubMed]
- Belzile, A.-S. Synergistic Activity of the Monolignol Dillapiol and Variation of Its Production in Two Plants, Anethum Graveolens and Piper aduncum. Master’ Thesis, University of Ottawa, Ottawa, ON, Canada, 1998. [Google Scholar] [CrossRef]
- Schepetkin, I.A.; Özek, G.; Özek, T.; Kirpotina, L.N.; Klein, R.A.; Khlebnikov, A.I.; Quinn, M.T. Composition and Biological Activity of the Essential Oils from Wild Horsemint, Yarrow, and Yampah from Subalpine Meadows in Southwestern Montana: Immunomodulatory Activity of Dillapiole. Plants 2023, 12, 2643. [Google Scholar] [CrossRef]
- Pereira Filho, A.A.; Do Vale, V.F.; De Oliveira Monteiro, C.M.; Barrozo, M.M.; Stanton, M.A.; Yamaguchi, L.F.; Kato, M.J.; Araújo, R.N. Effects of Piper aduncum (Piperales: Piperaceae) Essential Oil and Its Main Component Dillapiole on Detoxifying Enzymes and Acetylcholinesterase Activity of Amblyomma sculptum (Acari: Ixodidae). Int. J. Mol. Sci. 2024, 25, 5420. [Google Scholar] [CrossRef]
- Ferreira, A.K.; de-Sá-Júnior, P.L.; Pasqualoto, K.F.M.; De Azevedo, R.A.; Câmara, D.A.D.; Costa, A.S.; Figueiredo, C.R.; Matsuo, A.L.; Massaoka, M.H.; Auada, A.V.V.; et al. Cytotoxic Effects of Dillapiole on MDA-MB-231 Cells Involve the Induction of Apoptosis through the Mitochondrial Pathway by Inducing an Oxidative Stress While Altering the Cytoskeleton Network. Biochimie 2014, 99, 195–207. [Google Scholar] [CrossRef]
- Parise-Filho, R.; Pastrello, M.; Pereira Camerlingo, C.E.; Silva, G.J.; Agostinho, L.A.; De Souza, T.; Motter Magri, F.M.; Ribeiro, R.R.; Brandt, C.A.; Polli, M.C. The Anti-Inflammatory Activity of Dillapiole and Some Semisynthetic Analogues. Pharm. Biol. 2011, 49, 1173–1179. [Google Scholar] [CrossRef] [PubMed]
- Shafi, S.; Khurana, N.; Gupta, J. PPAR Gamma Agonistic Activity of Dillapiole: Protective Effects against Diabetic Nephropathy. Nat. Prod. Res. 2025, 39, 3581–3586. [Google Scholar] [CrossRef]
- Meot-Duros, L.; Magné, C. Antioxidant Activity and Phenol Content of Crithmum maritimum L. Leaves. Plant Physiol. Biochem. 2009, 47, 37–41. [Google Scholar] [CrossRef]
- Maoloni, A.; Pirker, T.; Pferschy-Wenzig, E.-M.; Aquilanti, L.; Bauer, R. Characterization of Potentially Health-Promoting Constituents in Sea Fennel (Crithmum maritimum) Cultivated in the Conero Natural Park (Marche Region, Central Italy). Pharm. Biol. 2023, 61, 1030–1040. [Google Scholar] [CrossRef]
- Zengin, G.; Sinan, K.I.; Ak, G.; Mahomoodally, M.F.; Paksoy, M.Y.; Picot-Allain, C.; Glamocilja, J.; Sokovic, M.; Jekő, J.; Cziáky, Z.; et al. Chemical Profile, Antioxidant, Antimicrobial, Enzyme Inhibitory, and Cytotoxicity of Seven Apiaceae Species from Turkey: A Comparative Study. Ind. Crops Prod. 2020, 153, 112572. [Google Scholar] [CrossRef]
- Santana-Gálvez, J.; Cisneros-Zevallos, L.; Jacobo-Velázquez, D. Chlorogenic Acid: Recent Advances on Its Dual Role as a Food Additive and a Nutraceutical against Metabolic Syndrome. Molecules 2017, 22, 358. [Google Scholar] [CrossRef] [PubMed]
- Siracusa, L.; Kulisic-Bilusic, T.; Politeo, O.; Krause, I.; Dejanovic, B.; Ruberto, G. Phenolic Composition and Antioxidant Activity of Aqueous Infusions from Capparis spinosa L. and Crithmum maritimum L. before and after Submission to a Two-Step in Vitro Digestion Model. J. Agric. Food Chem. 2011, 59, 12453–12459. [Google Scholar] [CrossRef] [PubMed]
- Naveed, M.; Hejazi, V.; Abbas, M.; Kamboh, A.A.; Khan, G.J.; Shumzaid, M.; Ahmad, F.; Babazadeh, D.; FangFang, X.; Modarresi-Ghazani, F.; et al. Chlorogenic Acid (CGA): A Pharmacological Review and Call for Further Research. Biomed. Pharmacother. 2018, 97, 67–74. [Google Scholar] [CrossRef]
- Calvo, M.M.; Martín-Diana, A.B.; Rico, D.; López-Caballero, M.E.; Martínez-Álvarez, O. Antioxidant, Antihypertensive, Hypoglycaemic and Nootropic Activity of a Polyphenolic Extract from the Halophyte Ice Plant (Mesembryanthemum crystallinum). Foods 2022, 11, 1581. [Google Scholar] [CrossRef]
- Mubarak, A.; Bondonno, C.P.; Liu, A.H.; Considine, M.J.; Rich, L.; Mas, E.; Croft, K.D.; Hodgson, J.M. Acute Effects of Chlorogenic Acid on Nitric Oxide Status, Endothelial Function, and Blood Pressure in Healthy Volunteers: A Randomized Trial. J. Agric. Food Chem. 2012, 60, 9130–9136. [Google Scholar] [CrossRef]
- Taguchi, K.; Hida, M.; Matsumoto, T.; Ikeuchi-Takahashi, Y.; Onishi, H.; Kobayashi, T. Effect of Short-Term Polyphenol Treatment on Endothelial Dysfunction and Thromboxane A2 Levels in Streptozotocin-Induced Diabetic Mice. Biol. Pharm. Bull. 2014, 37, 1056–1061. [Google Scholar] [CrossRef]
- Heydenreuter, W.; Kunold, E.; Sieber, S.A. Alkynol Natural Products Target ALDH2 in Cancer Cells by Irreversible Binding to the Active Site. Chem. Commun. 2015, 51, 15784–15787. [Google Scholar] [CrossRef]
- Gnocchi, D.; Sabbà, C.; Mazzocca, A. The Edible Plant Crithmum maritimum Shows Nutraceutical Properties by Targeting Energy Metabolism in Hepatic Cancer. Plant Foods Hum. Nutr. 2022, 77, 481–483. [Google Scholar] [CrossRef]

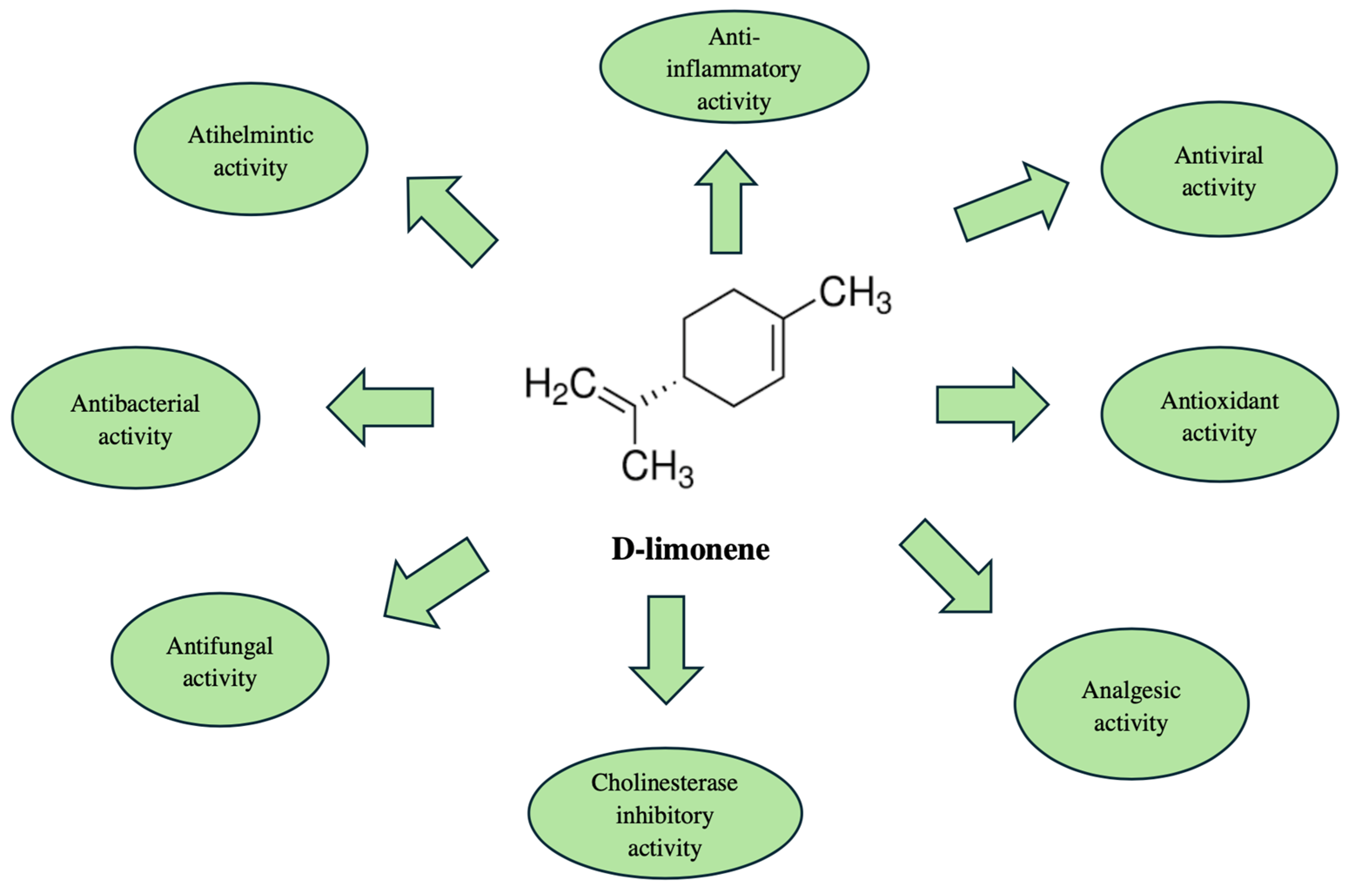
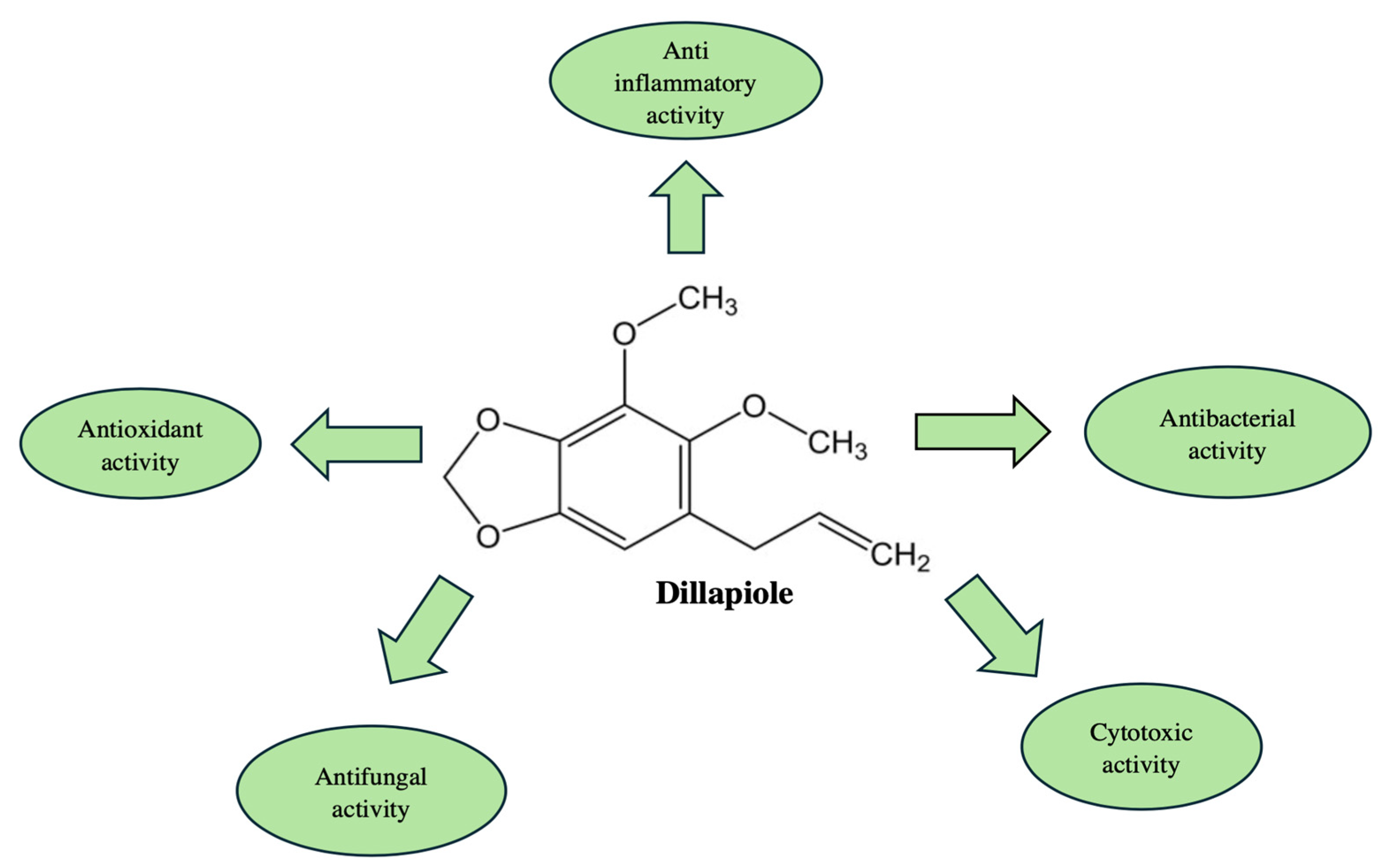
| Study Design | Country | Main Bioactive Compounds | Main Findings | Ref. |
|---|---|---|---|---|
| The antibacterial activity was evaluated by a paper-disk diffusion technique. The antibacterial activity study included eight bacteria species including S. aureus. Different studies evaluated the acetylcholinesterase, B. subtilis, B. cereus, P. aeruginosa, P. mirabilis, E. coli, and S. typhi. The concentrations of C. maritimum L. EO used were from 30 to 1.87 mg/mL. Positive controls—gentamycin and tetracycline. | Turkey | The major identified compounds were β-phellandrene (30%) and thymol methyl ether (25%). Other compounds, which were found, were γ-terpinene (24%) and dillapiole (21%). | The EO from C. maritimum L. exerted antibacterial activity, especially against Gram-positive bacteria. The most sensitive strain was B. cereus. P. mirabilis had no sensitivity to the EO. | [20] |
| The antimicrobial activity of the C. maritimum L. EO was evaluated by the disc diffusion method. The concentration of the tested EO was 5%. The EO was examined against methicillin-sensitive S. aureus and methicillin-resistant S. aureus. As positive controls were used penicillin, enrofloxacin, gentamicin sulfate, tetracycline hydrochloride, and cefaclor. | Greece | The main substances in the EO were 1,8-cineol (39.70%) and β-phellandrene (28.01%). | The EO did not demonstrate sufficient antimicrobial activity. | [21] |
| The antifungal activity was elevated against C. albicans, C. guillermondii, C. neoformans, E. floccosum, T. mentagrophytes, M. canis, and M. gypseum. The method used was macrodilution broth. Concentrations of 0.02–20 µL/mL of the EO were used. Fluconazole was used as a positive control. | Portugal | The major compound was dillapiole (64.2%). p-Cymene (11.8%), γ-terpinene (40.1%), and thymol methyl ether (23%) were also found in the EO. | The EO from C. maritimum L. has antifungal properties and it has the potential to be applied as a treatment of dermatophytosis. | [22] |
| The antioxidant activity was evaluated, using the 2,2’-azinobis-3-ethylbenzthiazoline-6-sulphonic acid (ABTS) radical-scavenging assay. The amount of EO used was 20 µL. The EO was mixed with ABTS. Ascorbic acid was used as a positive control. | Tunisia | The dominant compound was dillapiole (2.39–41.35%). Other components found in the EO were thymol methyl ether (20.13–34.75%), p-cymene (4.83–22.08%), and γ-terpinene (22.54–43.29%). | The tested EO from C. maritimum L. demonstrated antioxidant properties. | [23] |
| The antioxidant potential of C. maritimum L. EO was estimated as having ferric reducing/antioxidant power (FRAP), using DPPH assay. The concentration used was 0.1 mg/mL. The results were obtained after measurement with a spectrophotometer. | Croatia | Limonene was the major compound in the EO (57.5–74.2%). Other compounds at high levels were γ-terpinene (13.8–4.6%) and sabinene (13.4–8.1%). | The study revealed a low antioxidant capacity of the isolated EO. | [24] |
| The study investigated anti-inflammatory activity and involved cell line RAW 264.7 (ATCC TIB-71)—a mouse leukemic macrophage cell culture. This activity was estimated using the levels of nitrite production. For the cell stimulation, lipopolysaccharide was used. The concentrations of the applied EO from C. maritimum L. were 50–3.125 μg/mL. The results were obtained using an automatic microplate reader, measuring the absorbance. | Portugal | The main substances were γ-terpinene (33.6%), sabinene (32.0%), and thymol methyl ether (15.7%). | The EO demonstrated an anti-inflammatory activity and no toxicity. | [25] |
| The acetylcholinesterase inhibitory activity was evaluated by the presence of a yellow color by 2-nitrobenzoic acid and thiocholine, produced by acetylcholinesterase. The inhibitory activity was assessed based on the intensity of the yellow color obtained. The activity of the EO was compared to donepezil and tropolone. After incubation, the results from the study were obtained after absorbance measurement and calculations, which demonstrated acetylcholinesterase-inhibitory activity. | Libya | The major compounds found were thymyl methyl ether (56.86%), and γ-terpinene (16.17%). | The EO demonstrated a acetylcholinesterase-inhibitory effect. This finding revealed future application of the EO as an enhancer of cholinergic neurotransmission, improving the symptoms of Alzheimer’s disease. | [26] |
| The cholinesterase-inhibitory activity of C. maritimum L. EO was estimated, using a modified Ellman method. First, 10 µL of the sample was applied. Ethanol was used as a negative control. Non-enzymatic hydrolysis was also monitored. A spectrophotometer was used for all measurements. | Croatia | Limonene (57.5–74.2%) was the major compound in the EO. Other constituents in the EO were γ-terpinene (4.6–13.8%), β-pinene (0.1–4.9%), thymol methyl ether (0.4–0.2%), and terpinen-4-ol (6.9–2.0%). | The study showed that C. maritimum L. EO possessed cholinesterase inhibitory activity. | [24] |
| The estimation of the tyrosinase-inhibitory activity was performed using spectrophotometry. A mixture of a solution of tyrosine and a methanol solution of the enzyme inhibitor—arbutin—was prepared. After the addition of mushroom tyrosinase solution, the tyrosine was oxidized. The EO from C. maritimum L. was also prepared and tested. Methanol solution was used as a control. | Libya | The main constituents were thymyl methyl ether (56.86%), and γ-terpinene (16.17%). | The EO exerted tyrosinase-inhibitory activity. Consequently, the EO could be used in skincare, targeting melanin synthesis for the purpose of reducing hyperpigmentation. | [26] |
| A study of the anticancer activity of C. maritimum L. EO was conducted using the following cell lines: HEK293-T (human embryonic kidney), RKO (colorectal cancer), and MCF7 (breast cancer). The applied concentrations of the EO were 1 μL/mL to 10−5 μL/mL. Cell viability and growth inhibition were observed. Doxorubicin hydrochloride was used as a reference. | Portugal | The major bioactive compounds were γ-terpinene, thymol methyl ether, o-cymene, and β-phellandrene. Each of these compounds was found to be more than 10% of the EO. | The isolated EO did not demonstrate significant cytotoxic activity in the tested cell lines. | [27] |
| Study Design | Country | Main Bioactive Compounds | Main Findings | Ref. |
|---|---|---|---|---|
| The study of the antibacterial activity of the C. maritimum L. extract involved five bacteria species: S. aureus, E. faecalis, E. coli, E. aerogenes, and S. enterica ser. typhimurium. The extract was diluted with sterile water to the concentration of 0.05 mg/mL. Then 100 µL of the extract was added to different cultivated bacteria strains. The negative control was water. The antibacterial activity was evaluated by measuring the final optical density with a microplate reader. | France | The main compound in the extract was chlorogenic acid. Quinic acid, neochlorogenic acid, and trans ferulic acid were the other compounds found at high levels. | Hydro-ethanolic extract of C. maritimum L. leaves exerted strong antibacterial activity against Gram-positive and Gram-negative bacteria. | [28] |
| Purified falcarindiol, obtained from C. maritimum L. extract, was tested against Gram-positive and Gram-negative bacteria: M. luteus, B. cereus, S. enterica subsp. arizonae, P. fluorescens, P. marginalis, E. coli, and E. carotovara subsp. carotovora. The compound was studied for antifungal activity as well—against C. albicans. The applied concentrations of falcarindiol were 10, 20, and 50 µg/mL. Streptomycin and penicillin G were used as positive controls. | France | The test was performed using one of the main compounds from C. maritimum L. extract: falcarindiol. | It was found that falcarindiol affected the microorganisms differently. M. luteus and B. cereus were the most affected. | [29] |
| C. maritimum L. extract was studied for its activity against E. coli, K. pneumonia, S. aureus, and S. epidermidis. The concentrations of the test solutions were 2.34 to 600 µg/mL. Cefotaxime was used as a positive control. Minimal bactericidal concentrations and minimal inhibitory concentrations were measured. | Portugal | The study assessed the presence of minerals, Ω-6, and Ω-3 fatty acids, and polyphenols. | The extract revealed antibacterial activity, especially against the tested Gram-positive microorganisms: S. aureus and S. epidermidis. | [30] |
| The antioxidant activity of C. maritimum L. extract was evaluated using a purple-colored methanol solution of 2,2-diphenylpicrylhydrazyl (DPPH). To the 5 mL of a 0.004% methanol solution of DPPH was added 50 µL of the extract. Stable antioxidants were used as references. Hydrogen atom or electron donation abilities were assessed measuring the absorbance of the extract and the standards. | Algeria | Phenolic compounds were present at high levels in the compound, such as chlorogenic acid, 3-caffeoylquinic acid, 5-caffeoylquinic acid, and 4-caffeoylquinic acid. | Hydro-methanolic extract of aerial parts of the plant revealed antioxidant activity, and it was found that hydroxycinnamic acid derivatives were responsible for this activity. | [31] |
| The antioxidant activity of a methanolic extract of C. maritimum L. was assessed using the DPPH test. The prepared concentrations were 0.5, 1, 2, 5, and 10 mg/mL. Ascorbic acid, butylated hydroxytoluene, and quercitine were used as reference solutions. A solution of DPPH was prepared and mixed with the tested and the referent solutions. | Tunisia | The study presented the total polyphenol content, flavonoid content, and tannin content. | C. maritimum L. extract demonstrated antioxidant activity, due to the presence of phenolic compounds. | [32] |
| The anti-inflammatory activity of an aqueous extract from C. maritimum L. was tested using THP-1 cells. The tested cells were stimulated with lipopolysaccharide and treated with free aqueous extract at concentrations of 640 and 320 μg/mL. The aqueous extract was included in liposomes at concentrations of 1640 and 820 μg/mL. The levels of IL-10 and TNF-α were used to estimate the anti-inflammatory activity. | Spain | Chlorogenic acid. | Both extracts from C. maritimum L. demonstrated the induced release of IL-10 and reduced release of TNF-α, which is evidence for anti-inflammatory activity. | [33] |
| The study of anticancer activity was performed using two cell lines: Huh7 and HepG2. Ethyl acetate extracts of C. maritimum L. at concentrations of 2, 1, 0.5, 0.25 μM were used as test solutions. The proliferation of the cells was assessed with crystal violet and then the absorbance was measured by a plate reader. | Italy | No data provided | The plant extract showed a reduction in the growth of hepatocellular carcinoma cells. It also enhanced apoptosis and decreased cell cycle progression. | [34] |
| A study regarding the cytotoxic activity of falcarindiol, obtained from C. maritimum L. extract, was also performed. Small intestine cell lines from rats (IEC-6 cells) were used for the assay. The applied concentrations of the compound were 0.2 to 20 µM. The results were obtained after measuring the absorbance. | France | The assay was carried out using one of the main compounds from C. maritimum L. extract: falcarindiol. | Falcarindiol, isolated from C. maritimum L., exerted cytotoxic activity and induced a decrease in cell viability, but only at the highest concentration. | [29] |
| The cytotoxic activity of C. maritimum L. extract was estimated, using Caco-2 intestinal epithelial cell lines. 1, 2, 3, 4, 5% were the applied concentrations of the plant extract. | Portugal | The extract was rich in minerals, Ω-6, and Ω-3 fatty acids, and polyphenols. | The extract showed low toxicity concerning human intestinal epithelial Caco-2 model cells. | [30] |
| The assay was conducted using ethanol extracts from C. maritimum L. at a concentration of 30%. The cell line cultures cinsisted of keratinocytes and fibroblasts. The cell damage was induced by UVA radiation. The regenerative properties of the cells were evaluated by observing the histological morphology, loricrin content, collagen content, and collagen fiber content. | China | Chlorogenic acid | The study revealed better epidermal thickness and a notable increase in collagen fibers, collagen I, and loricrin. | [35] |
| Antiparasitic activity of hydro-ethanolic extracts from C. maritimum L. aerial parts was assessed on LLC-MK2 and U2OS cell lines. The cells were infected with Trypanosoma cruzi. The extract was diluted with DMSO in concentrations of 5, 10, and 20 mg/mL. Benznidazole was used as a positive control. | Portugal | Falcarindiol | The extract exerted antiparasitic activity against Trypanosoma cruzi. During the study, no cell line toxicity was demonstrated. | [36] |
| The hypoglycemic activity of the aqueous extracts from the leaves and flowers of C. maritimum L. was evaluated by measuring the inhibition of carbohydrate-hydrolyzing enzymes: α-amylase and α-glucosidase. In the α-amylase-inhibitory study, a starch solution with α-amylase was added to the extract in concentrations from 25 to 1000 μg/mL. For the α-glucosidase assay, the enzyme was added to maltose and o-dianisidine solutions and a peroxidase/glucose oxidase system color reagent. Then it was mixed with the extract of the plant at the same concentrations. Acarbose was used as a positive control. | Italy | The main compound in the extract was chlorogenic acid. Other identified compounds include cryptochlorogenic acid, neochlorogenic acid, rutin, and quercetin. | The plant extract possessed promising inhibitory activity against α-amylase and α-glucosidase and has the potential to be applied in the treatment of metabolic diseases. | [16] |
| The vasodilatory activity of C. maritimum L. extract was tested by estimating the isometric force of the isolated rat aortic rings. The rings were processed with ethanol extract in concentrations of 0.5‰ to 6‰ in organ baths. The relaxation of the vascular rings was presented as a percentage decrease in the stimulated vasoconstriction by noradrenaline. | Croatia | The study investigated the total phenol content, and the presence of chlorogenic acid in the plant extract is also reported. | C. maritimum L. extract revealed vasodilatatory activity, due to the presence of chlorogenic acid. | [24] |
| The prebiotic effect of C. maritimum L. extract was assessed, regarding Lactobacillus bulgaricus. The bacterial growth was assessed during aerobic conditions. The applied concentration of the extract was 0.1%. | Portugal | The extract was rich in minerals, Ω-6, and Ω-3 fatty acids, and polyphenols. | The results suggested accelerated growth of Lactobacillus bulgaricus after the exposure to C. maritimum L. extract. | [30] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dzhoglova, V.; Ivanova, S.; Shishmanova-Doseva, M.; Saracheva, K. Crithmum maritimum L.: Phytochemical Profile, Biological Activities, and Therapeutic Potential. Molecules 2025, 30, 2832. https://doi.org/10.3390/molecules30132832
Dzhoglova V, Ivanova S, Shishmanova-Doseva M, Saracheva K. Crithmum maritimum L.: Phytochemical Profile, Biological Activities, and Therapeutic Potential. Molecules. 2025; 30(13):2832. https://doi.org/10.3390/molecules30132832
Chicago/Turabian StyleDzhoglova, Velina, Stanislava Ivanova, Michaela Shishmanova-Doseva, and Kremena Saracheva. 2025. "Crithmum maritimum L.: Phytochemical Profile, Biological Activities, and Therapeutic Potential" Molecules 30, no. 13: 2832. https://doi.org/10.3390/molecules30132832
APA StyleDzhoglova, V., Ivanova, S., Shishmanova-Doseva, M., & Saracheva, K. (2025). Crithmum maritimum L.: Phytochemical Profile, Biological Activities, and Therapeutic Potential. Molecules, 30(13), 2832. https://doi.org/10.3390/molecules30132832







