Abstract
Ethnopharmacology, through the description of the beneficial effects of plants, has provided an early framework for the therapeutic use of natural compounds. Natural products, either in their native form or after crude extraction of their active ingredients, have long been used by different populations and explored as invaluable sources for drug design. The transition from traditional ethnopharmacology to drug discovery has followed a straightforward path, assisted by the evolution of isolation and characterization methods, the increase in computational power, and the development of specific chemoinformatic methods. The deriving extensive exploitation of the natural product chemical space has led to the discovery of novel compounds with pharmaceutical properties, although this was not followed by an analogous increase in novel drugs. In this work, we discuss the evolution of ideas and methods, from traditional ethnopharmacology to in silico drug discovery, applied to natural products. We point out that, in the past, the starting point was the plant itself, identified by sustained ethnopharmacological research, with the active compound deriving after extensive analysis and testing. In contrast, in recent years, the active substance has been pinpointed by computational methods (in silico docking and molecular dynamics, network pharmacology), followed by the identification of the plant(s) containing the active ingredient, identified by existing or putative ethnopharmacological information. We further stress the potential pitfalls of recent in silico methods and discuss the absolute need for in vitro and in vivo validation as an absolute requirement. Finally, we present our contribution to natural products’ drug discovery by discussing specific examples, applying the whole continuum of this rapidly evolving field. In detail, we report the isolation of novel antiviral compounds, based on natural products active against influenza and SARS-CoV-2 and novel substances active on a specific GPCR, OXER1.
1. Introduction
Humans possess discrete pharmacological knowledge of the therapeutic properties of plants from the beginning of their evolutionary history, leaving imprints in prehistoric and later cultural heritage [1,2]. However, this knowledge, accumulated in traditional medicine and ethnopharmacology, is declining. Nevertheless, elements of ethnopharmacological knowledge and practice, exercised either in parallel or supplementing the official treatment of diseases, create significant pressure on the scientific community to provide data related to the safety and effectiveness of the natural extracts. This element remains partially unfulfilled until now.
The use of plant extracts, containing hundreds of chemicals as pharmaceutical agents, is no longer a black box or the primary obstacle to understanding their mechanisms of action and/or the contained “active compound(s)”. This is powered by the significant increase in the detection and precision limits of analytical methods, the significant multiplication in computational power, and the construction of large public libraries of chemical (and natural) compounds (Figure 1, upper part). Indeed, many circulating drugs derive from natural products, and many emblematic drugs, such as taxol [3,4,5], vinblastine [6,7], quinine [8,9], and artemisinin [10], are rooted in traditional medicine and ethnopharmacology [11]; nevertheless, at a later stage, these drugs have been synthesized by modern chemistry and re-evaluated with modern analytical and pharmacological methods. Taxol (known as paclitaxel) is a nitrogen-containing diterpenoid isolated from the bark of Taxus brevifolia Nutt., which acts as a tubulin stabilizer and leads to cell cycle arrest, acting as an anticancer agent. Vinblastine and vincristine are closely related indole dihydroindole dimers (bisindole alkaloids), isolated from Catharanthus roseus (L.) G. Don (formerly known as Vinca rosea L.), the Madagascar periwinkle. Both of these anticancer agents, known as vinca alkaloids in the medical literature, are specific binders of tubulin, leading to tubulin depolymerization and cell cycle arrest in the metaphase stage. Quinine is an alkaloid obtained from Cinchona spp. It was the first antimalarial drug and served as an effective remedy for this potentially lethal infectious disease in colonial times, making possible European settlement in many tropical and subtropical parts of the world. Finally, artemisinin is a sesquiterpene lactone antimalarial compound with an endoperoxide group, discovered as a constituent of Artemisia annua L., with a unique mechanism of action on the heme complex.
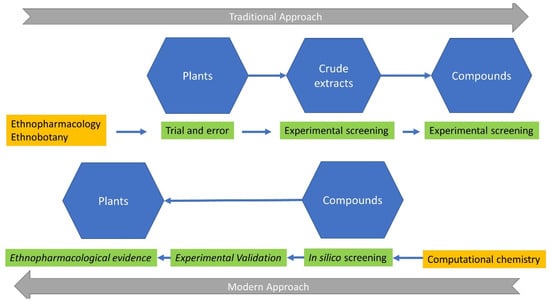
Figure 1.
Scientific disciplines (orange color), methodologies (green color), and compartments (blue color) from traditional to modern approaches in ethnopharmacology. Arrows denote the transition from the trial-and-error methodologies of the indigenous people to the in silico screening of computational chemistry. It is of note, as discussed in the text, that modern ethnopharmacology incorporates the computational and experimental validation of active natural compounds, prior to the detection of ethnopharmacological evidence. See text for further details.
In recent years, innovative extraction technologies, including semi-bionic extraction; supercritical fluid extraction; microwave-assisted, ultrasonic-assisted, and enzyme-assisted extraction; molecular distillation methods; membrane separation technology; and sophisticated new methodologies and instrumentation such as HPLC-MS, LC-MS, GC-MS, NMR, and crystallography, in parallel with the development of biology and clinical and experimental medicine, have allowed the re-evaluation of the corpus of traditional knowledge, the determination of chemical components of plant extracts, the identification of “active compound(s)”, and the development of novel drugs [12,13]. Galantamine [14], an Amaryllidaceae-type alkaloid from Galanthus woronowii Losinsk and other species of this genus, which has been recently approved for the treatment of early-onset Alzheimer’s disease, is an example of a recent achievement. A detailed presentation of plant-derived drug discovery in the last 30 years has been extensively reviewed and discussed in a recent series of publications [13,15,16,17].
Plants synthesize an immensely rich diversity of specialized secondary metabolites comprising an enormous number of active or complementary compounds [18,19]. This is due to several reasons, including (1) the high plant biodiversity in many areas of the world; (2) their significant ecological role in plant physiology, which is related to the high variability of problems that the plants have to face (protection from herbivores, pathogens, stress (including UV protection), other plant–plant and plant–animal interactions, etc); (3) the fact that, for the same problem, different evolutionary solutions have appeared in divergent plant lineages, with identical or similar pharmacological action [18,20]; and (4) the fact that different parts of the plant and different extraction methods of the same plant may result in a different collection of active compounds, with sometimes opposing biological effects [21]. Therefore, the validating process of ethnopharmacological knowledge is a laborious, and usually partially successful, enterprise [11], taking into account that only 1/10,000 tested compounds may lead to a successful drug in a time frame of almost ten years [22].
The exponential increase in computational power and data storage capabilities in recent decades has led to faster and, in some cases, economically sustainable solutions for drug discovery. The development of chemical libraries with billions of compounds and specific libraries of existing or putative natural compounds, with hundreds or thousands of molecules, together with the development of novel computational approaches (in silico docking methods, assisted by molecular dynamics, quantitative structure–activity relationships (QSARs), in silico evaluation of absorption–distribution–metabolism (ADMET), etc.), have been advanced as promising methods for the initial screening of the natural compound chemical space for a given disease (Figure 1, lower part) [23,24]. Computational high-throughput virtual screening has advanced as a cost-effective and less time-consuming method for drug discovery [22], as compounds from different chemical libraries have been subjected to high-throughput screening against a valid or presumed pathophysiological disease-related target. The first success of this approach was obtained in 1990, with the discovery of a dopamine D2 agonist [25]. Since then, the computational approach and virtual screening have been combined with network pharmacology (construction of signaling and interacting cellular networks, based on the observed or deducted interaction of compounds with cellular mechanisms). This has helped and accelerated drug discovery and development, positioning network pharmacology [26] as a paradigm shift in a newly emerged methodology, targeting all critical networks involved or perturbed in a disease. This approach complements the genomic, genetic, gene-related, and pathophysiological approach to disease. However, although computational chemistry has revolutionized the process of drug discovery, some limitations still exist, inherent to the accuracy of the computer programs used and the possible overfitting, induced by in silico methods, necessitating proper experimental validation.
Here, we discuss in brief the different steps in drug discovery, from ethnopharmacological observation to modern, high-throughput virtual screening, in an attempt to follow the rapid evolution of ideas in the field. Although a detailed review of all these topics is out of the scope of the current work, we review and discuss the progress and experimental approaches, applied to natural products, as depicted in Figure 1. We point out that in the past, the starting point was the plant itself, highlighted by sustained ethnopharmacological research, with the active compound deriving after extensive analysis and testing. In contrast, in the current state of scientific knowledge, the active substance is pinpointed by computational methods (in silico docking and molecular dynamics, quantitative structure–activity relationship (QSAR), network pharmacology, ADMET), followed by the identification of the plant(s) containing the active ingredient, seeking existing or possible ethnopharmacological information and relationships. We further stress the potential pitfalls of recent in silico methods and discuss the absolute need for in vitro and in vivo verification as a future recommendation. In addition, we present our contribution to this process, through specific examples of novel drug discovery. We further stress the potential pitfalls of recent in silico methods and discuss the absolute need for in vitro and in vivo verification of computer-generated data.
2. Evolution of Natural-Product-Derived Drug Development
2.1. Traditional Ethnopharmacology and Ethnobotany
Ethnopharmacology and ethnobotany are close neighboring fields. Ethnobotany is the study of complex relationships between cultures and their use of plants, focusing primarily on how plants are managed, used, and perceived across human societies. Ethnopharmacology, on the other hand, is defined as the interdisciplinary scientific exploration of traditionally employed indigenous drugs and biologically active agents [18,27,28,29]. Therefore, ethnopharmacology has a broader focus on exploring biologically active agents from plants, minerals, animals, fungi, and microbes. In both fields, a first step consists in the presentation of the use of extracts in a given disease, without investigating any potential causal relationship with contained ingredients/compounds (for a concrete example, see [30]). Ethnopharmacology has significantly contributed to the field explorations of indigenous and traditional medical knowledge and the biodiversity component to which such knowledge is linked.
In modern societies, this traditional use of plants as alternative pharmacological agents still persists. In China, traditional Chinese medicine is still serving many of the health needs of the population. It is practiced in parallel with modern medical treatment, due to the extensive recording of the plants’ medicinal properties. A series of institutions have been established to promote traditional Chinese medicine, such as the Academy of Traditional Chinese Medicine and training institutions. Almost all hospitals have an additional department of traditional medicine. Interestingly, the Chinese government proposes that both traditional Chinese and Western medicine can be combined to treat pneumonia caused by SARS-CoV-2, with promising results [31].
In contrast, western societies have mostly lost their traditional healing practice, although some elements of the plants’ medicinal properties have survived. Phytotherapists or naturopaths, operating within alternative and complementary health care systems, promote the ethnopharmacological use of plants. In some countries, they undergo training that is more or less regulated (in many cases by competent authorities) and have associations that recognize qualified members [32], making herbal medicine safe, effective, and standardized. In contrast, some other countries simply promote the exploitation of medicinal plants, without incorporating any regulatory policies, leading to the rapid loss of traditional practices. In the latter countries, the practice of ethnopharmacology and ethnobotany consists in the use of entire plant or crude plant extracts. It is worth mentioning, however, that the use of an entire plant, crude extract, or mixture of different plant extracts, with no isolation of components, results, in some cases, in a better therapeutic effect than the administration of individual compounds [33]. This is attributed to a synergy of active compounds included in the preparation (see [34] for a discussion and references therein). For this reason, teabags with the plant’s dried components, and bulk dried plant material both suggested as concoctions in use, are provided in the herbal markets [35].
Taking into account the above-discussed elements, an unbalanced perception of ethnopharmacology emerges, as this practice has the potential to contribute to the improvement of the health of indigenous people, supporting health care providers in the developing world, in addition to accelerating drug discovery [29,36]. In this respect, ethnopharmacology has, in our opinion, a characteristic of modernity, which ensembles particular sociocultural norms, attitudes, and practices.
Safety issues of herbal medicines is a global priority for national health authorities and the general public, both in their traditional use and in drug discovery [37,38]. At the European level, the Committee on Herbal Medicinal Products (HMPC), depending on the European Medicines Agency (EMA), is responsible for preparing the Agency’s opinions on the safety of herbal medicines. According to EMA regulations, clinical studies and tests on the safety and efficacy of herbal medicines are not required for their traditional use (local or per os administration), as long as this traditional use is well documented for at least 30 years, including at least 15 years within the EU (Directive 2004/24/EC of the European Parliament and of the Council of 31 March 2004).
In our contribution to the safety of herbal medicine [39], using partial-order analysis ([40], and the literature cited therein), we explored the reported undesirable effects of medicinal herbal substances, in their traditional/ethnopharmacological use. Data were retrieved from the European Union list of herbal substances for use in traditional medicinal products and the final European Union herbal monographs of the European Medicines Agency. Our analysis revealed that the reported undesirable effects were (in diminishing order) gastrointestinal disorders, allergic skin reactions, and allergic and hypersensitivity reactions. Variations in undesirable effects of herbal substances between plants of major phylogenetic groups of origin were also recorded, and an overall arrangement of medicinal herbal substances in rank order was obtained. This classification was proposed as a guide for the decision-making process for both healthcare providers and consumers. Moreover, taking into consideration that several data matrices have been published in many world areas, ranging from regional to continental scale, we attempted to demonstrate, for the first time, the implementation of partial-order techniques, processing ethnopharmacological information, with the purpose to reveal hidden inner structures and characteristics of reported raw data [41]. This methodology could potentially contribute to the conceptualization and management of ethnopharmacological knowledge [18].
2.2. Pharmacological Testing
Yeung et al. [42] analyzed the ethnopharmacology literature with regard to publication and citation data. They showed that research on recording medicinal plant species used by traditional medicine persists, but the evaluation of specific properties or treatment effects of extracts and compounds has increased enormously. Interestingly, the publications’ impact was directly related to the number of indigenous species in the authors’ countries. Currently, the trend of research has shifted from identifying and recording the medicinal plant species used in traditional medicine [30,43,44,45,46,47,48,49,50,51,52,53,54] to the evaluation of specific properties or treatment effects of crude plant extracts [55,56,57,58,59,60,61,62,63], or particular naturally derived products, such as flavonoids [64], alkaloids [65], tannins [66], saponins [67], phenols [68], and terpenoids [69] (an analysis of the provided bibliography together with applied methodology is presented in Table 1).

Table 1.
Indicative studies of the research shift from identifying and recording the medicinal plant species used in traditional medicine to the evaluation of specific properties or treatment effects of crude plant extracts, or particular naturally derived chemical substances.
However, the explosion of studies in pharmacological testing of plant extracts and compounds did not translate into an analogous increase in drug production. This was due to the fact that the majority of studies were mainly descriptive and did not integrate the next necessary step, which is the implementation of a state-of-the-art clinical trial. Millions of molecules are tested, and thousands have been produced, but most of them fail to progress in preclinical or clinical settings [70], mainly due to the lack of clinical studies in the field [42]. Indeed, the first edge of the translation chain, leading from a plant extract to a final product, relies on the choice of plants, as well as the choice of secondary metabolites, whose ethnopharmacological history is expected to ensure the success of the pharmacological testing and the desired health benefit (drug design). According to Pirintsos et al. [34], even the supply of plant raw material may become an obstacle influencing the possibility of new drug production, as several restrictions rule the natural collections (harvests) and trade of herbs and spices, especially within the framework of the EU environmental policy, as well as within the framework of the United Nations Convention on Biological Diversity. Therefore, in order to translate the results of the pharmacological testing to a new drug product, and in order to overcome the “valley of death”, a necessity of the bridging between successional steps, or successional links of the translational chain, should always be taken into consideration [34].
In our contribution to pharmacological testing, Lionis et al. [71] revealed low morbidity and mortality rates of coronary heart disease in Crete and the existence of an indigenous knowledge system in rural Crete with certain combinations of different aromatic plants, which have been used for the prevention and cure of the common cold and influenza. Origanum dictamnus (wild and cultivated dittany), Matricaria recutita (chamomile), Satureja thymbra (savory), Coridothymus capitatus (thyme), Mentha pulegium (penny royal), Salvia pomifera (wild sage), Salvia fruticosa (Greek sage), Origanum majorana (marjoram), and Mentha spicata (spearmint) were among the recorded medicinal plant species. Interestingly, the same plants’ concoction resulted in the prevention and cure of the common cold and influenza. At the same time, the exploration of the antioxidant activity of their extracts (without isolation and identification of the active compounds) was directed to detect a possible underlying mechanism of biological action.
2.3. Prospecting
2.3.1. Bioprospecting
The definition of bioprospecting involves the systematic search for genes, natural compounds, designs, and whole organisms in wildlife with potential for product development [72]. For the needs of this work, however, this term is restricted to the exploration, utilization, and exploitation of the plants’ biological diversity, either within the context of traditional medicinal knowledge or outside it. The collaboration between Merck Co. and Costa Rica’s National Institute of Biodiversity is a much-cited example of successful conventional bioprospecting, having identified novel compounds from fungi, such as arundifungin and durhamycin A, a novel antifungal compound and potent inhibitor of HIV Tat transactivation, respectively. Nevertheless, many other bioprospecting programs have not been as successful, impelling the exploration away from the assistance and primary knowledge of traditional healers [73].
Undoubtedly, the success of a commercial target necessitates a follow up across the translational chain, beyond the quality of the research product itself. Therefore, drug discovery and development is a long, costly, and high-risk process that takes over 10–15 years, while the attainment rate in translation, from R&D (preclinic) to the clinic stage, is less than 1% [70,74]. Harrison reported that in 174 drug development failures for the period 2013–2015, the majority of cases were due to a lack of either efficacy (52%) or safety (24%, including an insufficient therapeutic index). Strategic (15%), commercial (6%), and operational (3%) reasons were cited for the remainder of the failures [75]. Therefore, several efforts took place in order to facilitate future bioprospecting. Specific attention was given to the cross-cultural corroboration of medicinal usage of natural compounds or plants to guide bioprospecting. Roersch [76], in his ethnomedicinal review of Piper umbellatum, a species found in the Americas, Africa, and Asia, has recorded its use in 24 countries, stating that those with consensus across different cultures are more likely to be supported with scientific evidence and should be prioritized in pharmacological studies [76]. In this line, large-scale cross-cultural comparisons of ethnomedicinal floras were conducted, incorporating new phylogenetic and statistical methodologies [77,78,79,80], with promising results.
Based on the efficacy of combinations of aromatic plants reported previously [71] for the prevention of common cold and influenza, we prepared a combination of Thymbra capitata (thyme), Origanum dictamnus (Creta dittany), and Salvia fruticosa (Greek sage). The preparation was found to be safe in animals and humans [81] and efficient in vitro against a variety of upper respiratory tract viruses, including strains of influenza [82]. Interestingly, in vitro assays revealed that the preparation inhibited the nuclear translocation of the viral nucleoprotein, providing evidence for a specific mechanism of action [82] (see also the next paragraph). A clinical trial performed with the preparation reported its potency in treating influenza infections [83], and a post market analysis, after commercialization of the product as a dietary supplement, confirmed these data [84].
Performing detailed computational studies with ingredients of our preparation and taking into account our results of potentially active compounds [85] (see Section 2.4), we tested our preparation in vitro against SARS-CoV-2—infected cells. We found (Figure 2A) that it can promote the survival of cells after infection, reducing viral replication, both after pre- or coincubation with CAPeo, while, in a proof-of-principle study, in ambulatory COVID-19 patients, it induces a rapid elimination of disease-related symptoms (Figure 2B). A clinical trial is currently in progress.
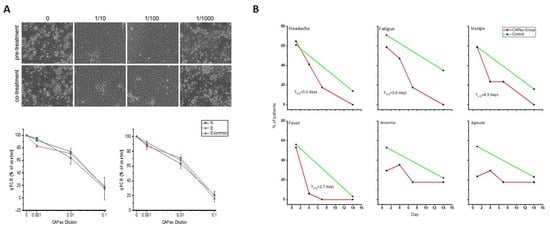
Figure 2.
Effect of a mixture of three aromatic plants ((thyme, Greek sage, and Cretan dittany) in SARS-CoV-2 infection. (A). In vitro testing. Left panel: light microscopy photographs of CPE in control (0, DMSO) and SARS-CoV-2–infected Vero cells (strain B.1, 0.1 m.o.i), pretreated or cotreated with different concentrations of CAPeo, in DMSO. Lower panel: Curves representing relative abundance (% of untreated control) of SARS-CoV-2 RNA after pretreatment (left curves) or cotreatment (right curves) with different concentrations of CAPeo, using real-time quantitative RT-PCR, targeting N and E regions of SARS-CoV-2 genome and E-common region shared by SARS-CoV and SARS-CoV-2 viruses. Values are shown as mean ± SD of three separate measurements. No significant differences in pretreated or cotreated cells were found. In both cases, CAPeo was efficient in concentrations almost 100 times lower than those administered per os and compatible with the estimated circulating concentration of the product. (B). Evolution of selected symptoms in our CAPeo-treated group (red curves), in a proof-of-principle trial (NCT04705753). T1/2 for the resolution of symptoms was calculated with a logistic regression fit. For comparison, the frequency of symptoms in the reference populations is also presented (green curves).
2.3.2. Mass Bioprospecting
Large-scale explorations, largely guided by the so-called “biodiversity” or “random” collection approach, where ethnobotanical or ethnopharmacological information plays a minimal or no role, are known as “mass bioprospecting” [27]. The most cited mass bioprospecting example concerns the efforts of the United States National Cancer Institute (NCI) in searching for plant-derived anticancer agents. About 114,000 extracts from an estimated 35,000 plant samples (representing 12,000–13,000 species), collected mostly from temperate regions of the world, had been screened against a number of tumor systems between 1960 and 1982 [27,86,87]. Despite the random collection approach and high-throughput methods of screening, mass bioprospecting of the NCI was also characterized by the fundamental feature of traditional pharmacology, which is the use of herbal formulae as the typical treatment. The herbal formula contains hundreds of chemical compounds. This complexity makes this approach complicated, time consuming, and challenging in understanding the mechanisms of action and bioactive ingredients. In 1983, the NCI’s mass bioprospecting effort was extended through the establishment of a National Cooperative Drug Discovery Group (NCDDG) program by the Developmental Therapeutics Program (DTP), Division of Cancer Treatment and Diagnosis (DCT).
The high failure rates, both in bioprospecting and mass bioprospecting, have raised the question of whether certain aspects of drug development are overlooked. Despite unprecedented investment in drug development and the major advances in many of the scientific and technological inputs into drug research and development (R&D), the number of new drugs approved by the US Food and Drug Administration (FDA) for the period 1950 to 2010 was low [86,87]. The number of new drugs approved per billion US dollars spent on R&D in this period has halved roughly every 9 years since 1950, falling around 80-fold in inflation-adjusted terms [86]. Therefore, a number of elements have been advanced, such as whether the new drug output may simply reflect the limitations of the current R&D model [87]. For example, given that most of the costs of new drug development are related to the costs of failed projects, the idea that high-affinity binding to a single biological target linked to a disease will lead to medical benefit in humans should be reevaluated. This has led to the conclusion that if the causal link between single targets and disease states is weaker than commonly thought, or if drugs rarely act on a single target, then the molecules that have been delivered by this research strategy into clinical development may not necessarily be more likely to succeed than those in earlier periods [86]. Finally, an interesting hypothesis has been advanced by Firn [88]. The author suggests that, even in the case of a positive hit in bioprospecting, the cost of synthesis of identified chemicals is high, given the complexity of natural molecules, an element that discourages pharmaceutical companies from investing in such molecules, investing in R&D, and subsequently decreasing the interest of investors in ethnopharmacology. In contrast, living organisms, having the correct enzymatic system, are capable of an efficient synthesis of such complex molecules. This situation is actually reversing, in view of the progress in molecular biology and ex vivo synthetic capabilities, increasing again the interest in natural product research.
2.4. Computational Chemistry
Computational drug discovery has over the past few decades become very relevant mainly due to the reduced risks, time, cost-effectiveness, and resources as compared with the traditional experimental approaches [89]. The substantial increase in computational power, the development and implementation of artificial intelligence methods, and the availability of huge freely available data collections were the main reasons for the development of computational methods with a special impact on novel drug compound identification. The development of novel powerful analytical techniques, as described above, has permitted the implementation of computational methods in the field of natural product derivatives (see [90] for a critical review and presentation of available resources). Computational methods include, among others: (1) the 3D resolution of the conformation of a large number of noncrystallized proteins [91,92], using modern artificial intelligence methods, with AlphaFold and AlphaFold2 being the more successful; (2) molecular docking developments, permitting the fully flexible association of (druggable) compounds to their putative targets (see [93] for a discussion). This was made possible with the recent increase in computational power. Indeed, older solutions considered the macromolecular target of drugs as a rigid molecule and tried to associate a rigid or flexible micromolecule/drug/natural product at a given binding pocket. However, it is known that the conformation of the macromolecule is equally modified by the binding of the ligand. With the increase in computational power, fully flexible solutions have been developed, in which all atoms of the macromolecule are also modified by the binding, providing a more accurate determination of the binding affinity [93]. These methods have been enhanced by molecular dynamics simulations, permitting the calculation of the movements of all atoms in the micromolecule for short (fsec) to very long (μsec) periods of time and the resulting conformational poses of the complex macromolecule–ligand, along with enhanced sampling techniques and elaborate methods of analysis, which have allowed unprecedented insight into complex phenomena in biology at extreme efficiency and accuracy. The combination of these methods, especially for the conformational changes of proteins, has permitted the simultaneous detection of movements and conformational states of thousands or millions of atoms and molecules in a given structure [94,95,96]; (3) the development of solutions mimicking the molecular mechanisms triggered by the activation of the (druggable) micromolecule to its (protein) target (see [85,97,98] for a discussion and a concrete example); (4) the development of “network pharmacology” methods [26,99,100,101,102,103,104,105], in which the exploitation of experimental and/or bibliographic evidence of signaling pathways and specific effects is explored, with increasingly sophisticated methods (see for example [99,106]), lead to a prediction of potential effects of a given substance, including natural products (see the thematic issue in ref. [100]) for a recent discussion on the subject); (5) the development of QSAR (quantitative structure–activity relationship) methods, in which, through sophisticated statistical methods, “active” parts of the ligand/drug/natural product, at a given conformation are extracted and predictions about a large number of putative or potential novel ligands can be drawn (see [107,108,109] for a critical analysis, pitfalls, and solutions of this methodology); (6) the development of bioinformatic methods for the prediction of absorption–distribution–metabolism and excretion of druggable molecules (ADMET), integrating artificial intelligence methods (see [110,111,112,113] for concrete examples and solutions).
With the help of computational chemistry, thousands of molecules have been evaluated for potential efficacy and safety at a small cost in a very short interval of time [114], overcoming the limitations of the experimental approach, helped by progress in artificial intelligence [115]. The lead compounds should have high-affinity prospective binding and specificity for a target associated with a disease and favorable pharmacodynamic and pharmacokinetic properties [22]. Of course, being a cheaper and less time-consuming process, when compared to experimental high-throughput screening, computational chemistry and pharmacology are expected to increase the output of the drug development process. Indeed, the median number of new molecules and biological license application approvals from 2010 to 2019 has increased by 60%, compared to the prior decade. However, the productivity of the pharmaceutical industry may be also linked to regulatory incentives, such as breakthrough therapy, fast-track designation, and Orphan Drug and GAIN acts, through which a large proportion of new drugs have been approved. As a concrete example, increased investments in basic neuroscience research by regulatory research authorities, such as the NIH or the European Union, have recently been linked to an increase in CNS startup investments [116].
It is estimated that computational chemistry has increased the quality of filtering and selection and improved the filtering efficiency by several orders of magnitude but without increasing the output substantially, mainly due to the bottlenecks of the R&D chain, as the necessity for experimental and clinical trials remains [115]. Indeed, despite the increasing number of successful applications in prospective computational chemistry, the computational methods are still limited to reliably predicting biological activity from chemical structure [117].
The discrepancies between preclinical research, with the use of computational chemical methods and clinical results, need a thorough evaluation, as only a small part of the computationally suggested compounds in the literature are experimentally tested, and negative results are not published. Moreover, many computational screening applications in industrial R&D departments are kept confidential, and compounds of interest are not disclosed, before a successful intellectual property filing. The reverse is also true, as disclosing candidate active compounds for drug development without patent protection discourages possible investments from the pharmaceutical industry (see [118] for a recent overview and discussion). Therefore, integrating computational and preclinical experimental screening (see [119] for a discussion) is necessary for a successful prospective screening, both for the strong patent protection and for further efficacy improvement of clinical trials, influencing the output of the drug production process. However, a number of successes have been acknowledged, with a number of novel drugs already approved by regulatory authorities and being on the market (Table 2), while an extensive list of other, potential druggable natural product candidates is under investigation [120,121]. The interested reader should consult Issues 7 and 8 of the journal Drug Discovery Today (2022) for a series of articles on the subject).

Table 2.
Recent successful stories of computational drug discovery approved by FDA or in a clinical trial. For an extensive Table of commercial drugs that made use of computer-aided drug design during the discovery process see [121], while for a detailed list of proteins and phytocompounds for computational docking along with therapeutic potential see [120].
Our group has extensive and long-standing experience in exploring in silico, in vitro, and in vivo the effect of natural compounds as potential pharmacological agents. Since 2000, we have provided evidence that red wine polyphenols act as antiproliferative agents in breast and prostate cancer cells [127,128]. Later on, we reported that proanthocyanidins [129,130], and especially proanthocyanidin B2, was the most potent compound, acting on a cell membrane androgen receptor [131], which was later characterized as the receptor binding oxo-eikosanoids (OXER1) [132], on which testosterone and polyphenols act as antagonists, modifying cAMP production [132], actin cytoskeleton [130,131,132], and intracellular Ca2+ [133,134]. Proanthocyanidins were repeating the effects of testosterone, modifying actin cytoskeleton and cAMP production in vitro [131,133], and inducing apoptosis in vitro and the regression of tumors in vivo in BalbC−/− mice breast and prostate xenografts [131]. Using testosterone and polyphenols as baits, we explored the ZINC database of natural products, taking advantage of a developed bioinformatic resource, permitting the classification of compounds as agonists or antagonists, through the simulation of Gα protein binding to the ligand-bound G-protein coupled receptors [98]. Using quantitative structure–activity relationships (QSAR), followed by in silico binding simulations and extensive in vitro validation, we identified ZINC15957997 as a specific OXER1 Gαi antagonist [135], ZINC8589130 as a specific OXER1 Gβγ antagonist and ZINC4017374 as an OXER1 pan-G-protein antagonist (Panagiotopoulos et al., in preparation). In vivo experiments with these compounds in BalbC−/− mice xenografts of prostate cancer are programmed.
In another field, we interrogated the natural products database for potential inhibitors of SARS-CoV-2 infections. We retained fortunellin as an allosteric inhibitor of the main viral protease dimerization (an absolute requirement for its action). This identification was made through in silico binding simulations, followed by molecular dynamics in long simulation times (10 μs) and validated in vitro, in SARS-CoV-2–infected Vero cells [97]. Fortunellin is found in kumquat, while its homolog apiin was found in parsley and celery. Finally, we identified p-cymene, a main constituent of the essential oil mixture described in Section 2.3.1, as a potent anti-influenza and anti-SARS-CoV-2 agent, acting in viral nucleoprotein and nucleocapsin viral proteins, respectively. Initial in silico studies have also been verified in vitro in infected cells [85].
3. Conclusions
The development of new drugs from herbal plant ingredients has been the basic agenda in the R&D of the drug industry for many decades. Either random or knowledge-based selection of plants could potentially reveal valuable compounds for the drug industry. In this brief review, we provided evidence on the evolution of concepts and processes used in the field of natural products’ use for drug development, sustained by our experience in the field. We provided evidence about the development of concepts leading from the ethnopharmacological or ethnobotanical observations of the beneficial effects of plant concoctions or essential oils and end up with the isolation and characterization of specific compound(s) for use as drugs or food supplements. Many emblematic drugs (Taxol [3,4,5], Vinblastine [6,7], quinine [8,9], and artemisinin [10]) have their origin in traditional medicine and ethnopharmacology, although later, they have been synthesized and re-evaluated with modern analytical and pharmacological methods.
However, the large expansion of scientific analytical and detection methods, the tremendous increase in computational power, and the construction of large public natural compounds libraries have led to a paradigm shift in the pharmacological use of plants and plant extracts. Analysis, isolation, and combinatorial use of single or multiple “active” ingredients have been used for the treatment of specific diseases and conditions (see [34] for a discussion and a concrete example). The development of biology and clinical and experimental medicine has facilitated this transition by providing specific target molecules or molecular pathways in which active plant ingredients have a specific effect. This bottom-up approach has led to the detection of specific compounds, available for experimental testing, and redefining the flow direction of traditional natural product use in pharmacology. Indeed, in spite of the traditional plant initiation point to the isolation and exploitation of (an) active compound(s), now, the reverse is also possible. Indeed, plant selection, in many cases, may result after the detection, characterization, and biological evaluation of a specific compound. Ranking compounds according to pharmaceutical relevance has been made possible due to their ability to predict the putative binding affinities between small molecules and biological counteractors, with potential therapeutic traits. Computational tools have helped to define and elaborate the strength of interaction between ligands and targets and have been instrumental in the identification of lead molecules from databases. However, despite the high expectations that computational chemistry would translate into increased production of new drugs, the result showed limited success. As we stressed and showed with concreter paradigms, the computational identification and the combinatorial in silico identification of compounds is only the first step in successful drug discovery. This should be supported with at least an in vitro validation of results. The whole process of drug development (in silico, in vitro, in vivo, clinical trials) should be respected for the novel or repurposed use of natural compounds for successful drug development, an element that maintains a high cost and sustained effort. This is supported by the explosion of publications during the current COVID-19 pandemic, in which, in spite of the in silico prediction of active compounds or the repurposing of existing drugs (with the notable examples of colchicine and nicotine), few novel drugs have been advanced and made available.
It is now clear that beyond the one-dimensional explanations such as the continuous need for further improvements in prediction algorithms, all the R&D operations, which are the core aspect of drug discovery and development, are extensively affected and controlled by a broad socioeconomic context that has been defined as a “pharmaceutical ecosystem”. The pharmaceutical ecosystem refers to the interdependent relationships among levels of interacting stakeholder networks, in connection with processes, tools, and infrastructures that are controlled by policies, laws, and opinions [136], which influence the production rate of new medicines. In this context, the accelerated research of natural compounds based on ethnopharmacological observations keeps a prominent role.
4. Patents
This work mentions the following patents (and those derived from them) in which one or multiple authors are named as inventors: US2008227853A1, WO2004006966A1, WO2011045557A1, WO2007123682A3, WO2021160768A1, and WO2012038694A1.
Author Contributions
Writing—original draft preparation, S.P. and E.C.; review and editing, all authors (S.P., A.P., M.B., V.D., C.L., G.S., I.K., M.K. and E.C.). All authors have read and agreed to the published version of the manuscript.
Funding
This research was partially funded by grants by the Hellenic Foundation for Research and Innovation (H.F.R.I.) under the “First Call for H.F.R.I. Research Projects to support Faculty members and Researchers and the procurement of high-cost research equipment grant” (Project Number: 3725 to M.K.), by Greece and the European Union (European Social Fund (ESF)) through the Operational Programme “Human Resources Development, Education and Lifelong Learning” in the context of the project “Strengthening Human Resources Research Potential via Doctorate Research” (MIS-5000432), implemented by the State Scholarships Foundation (ΙΚΥ)” to A.A.P. (Ph.D. scholarship), the “PRACE support to mitigate impact of COVID-19 pandemic” call and the project “Epitope vaccines based on the dynamics of mutated SARS-CoV-2 proteins at all atom resolution” (to V.D.) and private funds from OLVOS Science SA.
Institutional Review Board Statement
The studies involving human participants were reviewed and approved by the University of Crete Bioethics Committee (No 78/01.04.2020).
Informed Consent Statement
The patients/participants provided their written informed consent to participate in this study. No potentially identifiable human images or data is presented in this study.
Data Availability Statement
All data are included in the text and the Figures.
Conflicts of Interest
The authors declare no further conflicts of interest different from their participation, as inventors, in the patents stated above. The funders had no role in the design, the collection, analyses, or interpretation of data, the writing of the manuscript, or in the decision to publish the results.
References
- Halberstein, R.A. Medicinal plants: Historical and cross-cultural usage patterns. Ann. Epidemiol. 2005, 15, 686–699. [Google Scholar] [CrossRef] [PubMed]
- Hardy, K. Paleomedicine and the Evolutionary Context of Medicinal Plant Use. Rev. Bras. Farmacogn. 2021, 31, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Malik, S.; Rosa, M.; Cusidó, R.M.; Mirjalili, M.; Moyano, E.; Palazón, J.; Bonfill, M. Production of the anticancer drug taxol in Taxus baccata suspension cultures: A review. Broc. Biochem. 2011, 46, 23–34. [Google Scholar] [CrossRef]
- Gallego-Jara, J.; Lozano-Terol, G.; Sola-Martinez, R.A.; Canovas-Diaz, M.; de Diego Puente, T. A Compressive Review about Taxol((R)): History and Future Challenges. Molecules 2020, 25, 5986. [Google Scholar] [CrossRef]
- Rose, W.C. Taxol: A review of its preclinical in vivo antitumor activity. Anticancer Drugs 1992, 3, 311–321. [Google Scholar] [CrossRef]
- Brade, W. Critical Review of Pharmacology, Toxicology, Pharmacokinetics of Vincristine, Vindesine, Vinblastine. In International Vinca Alkaloid Symposium—Vindesine; Brade, W., Nagel, G., Seeber, S., Eds.; Oncology Karger: Basel, Switzerland, 1981; Volume 6, pp. 95–123. [Google Scholar]
- Nelson, R.L. The comparative clinical pharmacology and pharmacokinetics of vindesine, vincristine, and vinblastine in human patients with cancer. Med. Pediatr. Oncol. 1982, 10, 115–127. [Google Scholar] [CrossRef]
- Achan, J.; Talisuna, A.O.; Erhart, A.; Yeka, A.; Tibenderana, J.K.; Baliraine, F.N.; Rosenthal, P.J.; D’Alessandro, U. Quinine, an old anti-malarial drug in a modern world: Role in the treatment of malaria. Malar J. 2011, 10, 144. [Google Scholar] [CrossRef]
- Liles, N.W.; Page, E.E.; Liles, A.L.; Vesely, S.K.; Raskob, G.E.; George, J.N. Diversity and severity of adverse reactions to quinine: A systematic review. Am. J. Hematol. 2016, 91, 461–466. [Google Scholar] [CrossRef]
- Dai, Y.; Zhou, W.; Meng, J.; Du, X.-L.; Sui, Y.-P.; Dai, L.; Wang, P.-Q.; Huo, H.-R.; Sui, F. The pharmacological activities and mechanisms of artemisinin and its derivatives: A systematic review. Med. Chem. Res. 2017, 26, 867–880. [Google Scholar] [CrossRef]
- Fabricant, D.S.; Farnsworth, N.R. The value of plants used in traditional medicine for drug discovery. Environ. Health Perspect. 2001, 109 (Suppl. S1), 69–75. [Google Scholar] [CrossRef]
- Dias, D.A.; Urban, S.; Roessner, U. A historical overview of natural products in drug discovery. Metabolites 2012, 2, 303–336. [Google Scholar] [CrossRef] [PubMed]
- Najmi, A.; Javed, S.A.; Al Bratty, M.; Alhazmi, H.A. Modern Approaches in the Discovery and Development of Plant-Based Natural Products and Their Analogues as Potential Therapeutic Agents. Molecules 2022, 27, 349. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M.; Snader, K.M. Natural products as sources of new drugs over the period 1981–2002. J. Nat. Prod. 2003, 66, 1022–1037. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs from 1981 to 2014. J. Nat. Prod. 2016, 79, 629–661. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the 30 years from 1981 to 2010. J. Nat. Prod. 2012, 75, 311–335. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the last 25 years. J. Nat. Prod. 2007, 70, 461–477. [Google Scholar] [CrossRef]
- Pirintsos, S.; Bariotakis, M.; Laina, D.; Lionis, C.; Castanas, E.; Bruggemann, R. Analyzing Ethnopharmacological Data Matrices on Traditional Uses of Medicinal Plants with the Contribution of Partial Order Techniques. In Partial Order Concepts in Applied Sciences; Fattore, M., Bruggemann, R., Eds.; Springer: Cham, Switzerland, 2017. [Google Scholar] [CrossRef]
- Hartmann, T. From waste products to ecochemicals: Fifty years research of plant secondary metabolism. Phytochemistry 2007, 68, 2831–2846. [Google Scholar] [CrossRef]
- Cowan, M.M. Plant products as antimicrobial agents. Clin. Microbiol. Rev. 1999, 12, 564–582. [Google Scholar] [CrossRef]
- Karali, D.; Georgescu, L.; Pirintsos, S.; Athanassakis, I. T cell regulation by Phlomis lanata protein extracts in mice. Pharm. Biol. 2016, 54, 207–214. [Google Scholar] [CrossRef]
- Murugan, N.A.; Podobas, A.; Gadioli, D.; Vitali, E.; Palermo, G.; Markidis, S. A Review on Parallel Virtual Screening Softwares for High-Performance Computers. Pharmaceuticals 2022, 15, 63. [Google Scholar] [CrossRef]
- Zaman, W.; Ye, J.; Saqib, S.; Liu, Y.; Shan, Z.; Hao, D.; Chen, Z.; Xiao, P. Predicting potential medicinal plants with phylogenetic topology: Inspiration from the research of traditional Chinese medicine. J. Ethnopharmacol. 2021, 281, 114515. [Google Scholar] [CrossRef]
- Zaman, W.; Ye, J.; Ahmad, M.; Saqib, S.; Shinwari, Z.; Chen, Z. Phylogenetic exploration of traditional chinese medicinal plants: A case study of Lamiaceae. Pak. J. Bot. 2022, 54, 1033–1040. [Google Scholar] [CrossRef]
- Martin, Y. Accomplishments and challenges in integrating software for computer-aided ligand design in drug discovery. Persp. Drug Discov. Des. 1995, 3, 139–150. [Google Scholar] [CrossRef]
- Boezio, B.; Audouze, K.; Ducrot, P.; Taboureau, O. Network-based Approaches in Pharmacology. Mol. Inform. 2017, 36, 1700048. [Google Scholar] [CrossRef] [PubMed]
- Soejarto, D.D.; Fong, H.H.; Tan, G.T.; Zhang, H.J.; Ma, C.Y.; Franzblau, S.G.; Gyllenhaal, C.; Riley, M.C.; Kadushin, M.R.; Pezzuto, J.M.; et al. Ethnobotany/ethnopharmacology and mass bioprospecting: Issues on intellectual property and benefit-sharing. J. Ethnopharmacol. 2005, 100, 15–22. [Google Scholar] [CrossRef]
- Mukherjee, P.K.; Venkatesh, P.; Ponnusankar, S. Ethnopharmacology and integrative medicine—Let the history tell the future. J. Ayurveda Integr. Med. 2010, 1, 100–109. [Google Scholar] [CrossRef]
- Heinrich, M.; Gibbons, S. Ethnopharmacology in drug discovery: An analysis of its role and potential contribution. J. Pharm. Pharmacol. 2001, 53, 425–432. [Google Scholar] [CrossRef]
- Ahmet Sargin, S. Ethnobotanical survey of medicinal plants in Bozyazi district of Mersin, Turkey. J. Ethnopharmacol. 2015, 173, 105–126. [Google Scholar] [CrossRef]
- Zhao, Z.; Li, Y.; Zhou, L.; Zhou, X.; Xie, B.; Zhang, W.; Sun, J. Prevention and treatment of COVID-19 using Traditional Chinese Medicine: A review. Phytomedicine 2021, 85, 153308. [Google Scholar] [CrossRef]
- Jäger, A. Medicinal Plant Research: A Reflection on Translational Tasks. In Ethnopharmacology; Heinrich, M., Jäger, A., Eds.; Karger: Basel, Switzerland, 2015. [Google Scholar] [CrossRef]
- Thomford, N.E.; Senthebane, D.A.; Rowe, A.; Munro, D.; Seele, P.; Maroyi, A.; Dzobo, K. Natural Products for Drug Discovery in the 21st Century: Innovations for Novel Drug Discovery. Int. J. Mol. Sci. 2018, 19, 1578. [Google Scholar] [CrossRef]
- Pirintsos, S.A.; Bariotakis, M.; Kampa, M.; Sourvinos, G.; Lionis, C.; Castanas, E. The Therapeutic Potential of the Essential Oil of Thymbra capitata (L.) Cav., Origanum dictamnus L. and Salvia fruticosa Mill. and a Case of Plant-Based Pharmaceutical Development. Front. Pharmacol. 2020, 11, 522213. [Google Scholar] [CrossRef] [PubMed]
- Hanlidou, E.; Karousou, R.; Kleftoyanni, V.; Kokkini, S. The herbal market of Thessaloniki (N Greece) and its relation to the ethnobotanical tradition. J. Ethnopharmacol. 2004, 91, 281–299. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Garcia, V. The relevance of traditional knowledge systems for ethnopharmacological research: Theoretical and methodological contributions. J. Ethnobiol. Ethnomed. 2010, 6, 32. [Google Scholar] [CrossRef]
- World Health Organization. WHO Guidelines on Safety Monitoring of Herbal Medicines in Pharmacovigilance Systems; World Health Organization (WHO): Geneva, Switzerland, 2004; Available online: https://apps.who.int/iris/handle/10665/43034 (accessed on 24 May 2022).
- Heinrich, M. Ethnopharmacology in the 21st century—Grand challenges. Front. Pharmacol. 2010, 1, 8. [Google Scholar] [CrossRef] [PubMed]
- Laina, D.; Bariotakis, M.; Lionis, C.; Castanas, E.; Bruggemann, R.; Pirintsos, S.A. Partial ordering of undesirable effects reported for traditionally used medicinal herbal substances. Toxicol. Environ. Chem. 2017, 99, 1230–1241. [Google Scholar] [CrossRef]
- Brüggemann, R.; Patil, G. Ranking and Prioritization for Multi-Indicator Systems: Introduction to Partial Order Applications; Springer: Basel, Switzerland, 2011. [Google Scholar] [CrossRef]
- Bariotakis, M.; Georgescu, L.; Laina, D.; Oikonomou, I.; Ntagounakis, G.; Koufaki, M.I.; Souma, M.; Choreftakis, M.; Zormpa, O.G.; Smykal, P.; et al. From wild harvest towards precision agriculture: Use of Ecological Niche Modelling to direct potential cultivation of wild medicinal plants in Crete. Sci. Total Environ. 2019, 694, 133681. [Google Scholar] [CrossRef]
- Yeung, A.W.K.; Heinrich, M.; Kijjoa, A.; Tzvetkov, N.T.; Atanasov, A.G. The ethnopharmacological literature: An analysis of the scientific landscape. J. Ethnopharmacol. 2020, 250, 112414. [Google Scholar] [CrossRef]
- Alzweiri, M.; Sarhan, A.A.; Mansi, K.; Hudaib, M.; Aburjai, T. Ethnopharmacological survey of medicinal herbs in Jordan, the Northern Badia region. J. Ethnopharmacol. 2011, 137, 27–35. [Google Scholar] [CrossRef]
- Karousou, R.; Deirmentzoglou, S. The herbal market of Cyprus: Traditional links and cultural exchanges. J. Ethnopharmacol. 2011, 133, 191–203. [Google Scholar] [CrossRef]
- Al-Qura’n, S. Ethnopharmacological survey of wild medicinal plants in Showbak, Jordan. J. Ethnopharmacol. 2009, 123, 45–50. [Google Scholar] [CrossRef]
- Hudaib, M.; Mohammad, M.; Bustanji, Y.; Tayyem, R.; Yousef, M.; Abuirjeie, M.; Aburjai, T. Ethnopharmacological survey of medicinal plants in Jordan, Mujib Nature Reserve and surrounding area. J. Ethnopharmacol. 2008, 120, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Lardos, A. The botanical materia medica of the Iatrosophikon—A collection of prescriptions from a monastery in Cyprus. J. Ethnopharmacol. 2006, 104, 387–406. [Google Scholar] [CrossRef]
- Said, O.; Khalil, K.; Fulder, S.; Azaizeh, H. Ethnopharmacological survey of medicinal herbs in Israel, the Golan Heights and the West Bank region. J. Ethnopharmacol. 2002, 83, 251–265. [Google Scholar] [CrossRef]
- Lev, E.; Amar, Z. Ethnopharmacological survey of traditional drugs sold in Israel at the end of the 20th century. J. Ethnopharmacol. 2000, 72, 191–205. [Google Scholar] [CrossRef]
- Ali-Shtayeh, M.; Yaghmour, R.M.-R.; Faidi, Y.R.; Salem, K.; Al-Nuri, M.-A. Antimicrobial activity of 20 plants used in folkloric medicine in the Palestinian area. J. Ethnopharmacol. 1998, 60, 265–271. [Google Scholar] [CrossRef]
- Vázquez, F.; Suarez, M.; Pérez, A. Medicinal plants used in the Barros Area, Badajoz Province (Spain). J. Ethnopharmacol. 1997, 55, 81–85. [Google Scholar] [CrossRef]
- Honda, G.; Yeşilada, E.; Tabata, M.; Sezik, E.; Fujita, T.; Takeda, Y.; Takaishi, Y.; Tanaka, T. Traditional medicine in Turkey VI. Folk medicine in West Anatolia: Afyon, Kütahya, Denizli, Muğla, Aydin provinces. J. Ethnopharmacol. 1996, 53, 75–87. [Google Scholar] [CrossRef]
- Al-Khalil, S. A Survey of Plants Used in Jordanian Traditional Medicine. Int. J. Pharmacogn. 1995, 33, 317–323. [Google Scholar] [CrossRef]
- Dafni, A.; Yaniv, Z.; Palevitch, D. Ethnobotanical survey of medicinal plants in northern Israel. J. Ethnopharmacol. 1984, 10, 295–310. [Google Scholar] [CrossRef]
- Qnais, E.; Elokda, A.; Abu Ghalyun, Y.; Abdulla, F. Antidiarrheal Activity of the Aqueous Extract of Punica granatum. (Pomegranate) Peels. Pharm. Biol. 2007, 45, 715–720. [Google Scholar] [CrossRef]
- Hage-Sleiman, R.; Mroueh, M.; Daher, C.F. Pharmacological evaluation of aqueous extract of Althaea officinalis flower grown in Lebanon. Pharm. Biol. 2011, 49, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.Y.; Chan, Y.P.; Chang, J. Antioxidant activity of extract from Polygonum cuspidatum. Biol. Res.Res. 2007, 40, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.Y. Antioxidant activity of extract from Polygonum aviculare L. Biol. Res. 2006, 39, 281–288. [Google Scholar] [CrossRef]
- Perianayagam, J.B.; Sharma, S.K.; Pillai, K.K. Anti-inflammatory activity of Trichodesma indicum root extract in experimental animals. J. Ethnopharmacol. 2006, 104, 410–414. [Google Scholar] [CrossRef]
- Garrido, G.; Gonzalez, D.; Lemus, Y.; Garcia, D.; Lodeiro, L.; Quintero, G.; Delporte, C.; Nunez-Selles, A.J.; Delgado, R. In vivo and in vitro anti-inflammatory activity of Mangifera indica L. extract (VIMANG). Pharmacol. Res. 2004, 50, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Rajeshkumar, N.; Joy, K.; Kuttan, G.; Ramsewak, R.; Nair, M.; Kuttan, R. Antitumour and anticarcinogenic activity of Phyllanthus amarus extract. J. Ethnopharmacol. 2002, 81, 17–22. [Google Scholar] [CrossRef]
- Olajide, O.; Makinde, J.; Okpako, D.; Awe, S. Studies on the anti-inflammatory and related pharmacological properties of the aqueous extract of Bridelia ferruginea stem bark. J. Ethnopharmacol. 2000, 71, 153–160. [Google Scholar] [CrossRef]
- Bhakta, T.; Mukherjee, P.; Saha, K.; Pal, M.; Saha, B. Studies on Antitussive Activity of Cassia fistula (Leguminosae) Leaf Extract. Pharm. Biol. 1998, 36, 140–143. [Google Scholar] [CrossRef]
- Górniak, I.; Bartoszewski, R.; Króliczewski, J. Comprehensive review of antimicrobial activities of plant flavonoids. Phytochem. Rev. 2019, 18, 241–272. [Google Scholar] [CrossRef]
- Adamski, Z.; Blythe, L.L.; Milella, L.; Bufo, S.A. Biological Activities of Alkaloids: From Toxicology to Pharmacology. Toxins 2020, 12, 210. [Google Scholar] [CrossRef]
- Pizzi, A. Tannins medical / pharmacological and related applications: A critical review. Sust. Chem. Pharm. 2021, 22, 100481. [Google Scholar] [CrossRef]
- Metwaly, A.M.; Lianlian, Z.; Luqi, H.; Deqiang, D. Black Ginseng and Its Saponins: Preparation, Phytochemistry and Pharmacological Effects. Molecules 2019, 24, 1856. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Foo, L. Polyphenolics of Salvia—A review. Phytochemistry 2002, 59, 117–140. [Google Scholar] [CrossRef]
- Yang, W.; Chen, X.; Li, Y.; Guo, S.; Wang, Z.; Yu, X. Advances in Pharmacological Activities of Terpenoids. Nat. Prod. Commun. 2020, 15, 1934578X20903555. [Google Scholar] [CrossRef]
- Hinkson, I.V.; Madej, B.; Stahlberg, E.A. Accelerating Therapeutics for Opportunities in Medicine: A Paradigm Shift in Drug Discovery. Front. Pharmacol. 2020, 11, 770. [Google Scholar] [CrossRef] [PubMed]
- Lionis, C.; Faresjo, A.; Skoula, M.; Kapsokefalou, M.; Faresjo, T. Antioxidant effects of herbs in Crete. Lancet 1998, 352, 1987–1988. [Google Scholar] [CrossRef]
- Castree, N. Bioprospecting: From theory to practice (and back again). Trans. Inst. Brit. Geograph. 2003, 28, 35–55. [Google Scholar] [CrossRef]
- Buenz, E.J.; Schnepple, D.J.; Bauer, B.A.; Elkin, P.L.; Riddle, J.M.; Motley, T.J. Techniques: Bioprospecting historical herbal texts by hunting for new leads in old tomes. Trends Pharmacol. Sci. 2004, 25, 494–498. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Gao, W.; Hu, H.; Zhou, S. Why 90% of clinical drug development fails and how to improve it? Acta Pharm. Sin. B 2022, in press. [Google Scholar] [CrossRef]
- Harrison, R.K. Phase II and phase III failures: 2013–2015. Nat. Rev. Drug Discov. 2016, 15, 817–818. [Google Scholar] [CrossRef]
- Roersch, C.M. Piper umbellatum L.: A comparative cross-cultural analysis of its medicinal uses and an ethnopharmacological evaluation. J. Ethnopharmacol. 2010, 131, 522–537. [Google Scholar] [CrossRef]
- Hao, D.-C.; Xiao, P.-G. Pharmaceutical resource discovery from traditional medicinal plants: Pharmacophylogeny and pharmacophylogenomics. Chin. Herb. Med. 2020, 12, 104–117. [Google Scholar] [CrossRef]
- Zhu, F.; Qin, C.; Tao, L.; Liu, X.; Shi, Z.; Ma, X.; Jia, J.; Tan, Y.; Cui, C.; Lin, J.; et al. Clustered patterns of species origins of nature-derived drugs and clues for future bioprospecting. Proc. Natl. Acad. Sci. USA 2011, 108, 12943–12948. [Google Scholar] [CrossRef] [PubMed]
- Saslis-Lagoudakis, C.H.; Williamson, E.M.; Savolainen, V.; Hawkins, J.A. Cross-cultural comparison of three medicinal floras and implications for bioprospecting strategies. J. Ethnopharmacol. 2011, 135, 476–487. [Google Scholar] [CrossRef]
- Saslis-Lagoudakis, C.H.; Savolainen, V.; Williamson, E.M.; Forest, F.; Wagstaff, S.J.; Baral, S.R.; Watson, M.F.; Pendry, C.A.; Hawkins, J.A. Phylogenies reveal predictive power of traditional medicine in bioprospecting. Proc. Natl. Acad. Sci. USA 2012, 109, 15835–15840. [Google Scholar] [CrossRef] [PubMed]
- Kalyvianaki, K.; Malamos, P.; Mastrodimou, N.; Manoura-Zonou, I.; Vamvoukaki, R.; Notas, G.; Malliaraki, N.; Moustou, E.; Tzardi, M.; Pirintsos, S.; et al. Toxicity evaluation of an essential oil mixture from the Cretan herbs thyme, Greek sage and Cretan dittany. NPJ Sci. Food 2020, 4, 20. [Google Scholar] [CrossRef] [PubMed]
- Tseliou, M.; Pirintsos, S.A.; Lionis, C.; Castanas, E.; Sourvinos, G. Antiviral effect of an essential oil combination derived from three aromatic plants (Coridothymus capitatus (L.) Rchb. f., Origanum dictamnus L. and Salvia fruticosa Mill.) against viruses causing infections of the upper respiratory tract. J. Herb. Med. 2019, 17–18, 100288. [Google Scholar] [CrossRef]
- Duijker, G.; Bertsias, A.; Symvoulakis, E.K.; Moschandreas, J.; Malliaraki, N.; Derdas, S.P.; Tsikalas, G.K.; Katerinopoulos, H.E.; Pirintsos, S.A.; Sourvinos, G.; et al. Reporting effectiveness of an extract of three traditional Cretan herbs on upper respiratory tract infection: Results from a double-blind randomized controlled trial. J. Ethnopharmacol. 2015, 163, 157–166. [Google Scholar] [CrossRef]
- Anastasaki, M.; Bertsias, A.; Pirintsos, S.A.; Castanas, E.; Lionis, C. Post-market outcome of an extract of traditional Cretan herbs on upper respiratory tract infections: A pragmatic, prospective observational study. BMC Complement. Altern. Med. 2017, 17, 466. [Google Scholar] [CrossRef]
- Panagiotopoulos, A.; Tseliou, M.; Karakasiliotis, I.; Kotzampasi, D.-M.; Daskalakis, V.; Kesesidis, N.; Notas, G.; Lionis, C.; Kampa, M.; Pirintsos, S.; et al. p-cymene impairs SARS-CoV-2 and Influenza A (H1N1) viral replication: In silico predicted interaction with SARS-CoV-2 nucleocapsid protein and H1N1 nucleoprotein. Pharmacol. Res.Res. Perspect 2021, 9, e00798. [Google Scholar] [CrossRef]
- Scannell, J.W.; Blanckley, A.; Boldon, H.; Warrington, B. Diagnosing the decline in pharmaceutical R&D efficiency. Nat. Rev. Drug Discov. 2012, 11, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Munos, B. Lessons from 60 years of pharmaceutical innovation. Nat. Rev. Drug Discov. 2009, 8, 959–968. [Google Scholar] [CrossRef]
- Firn, R. Bioprospecting—Why is it so unrewarding? Biodiv. Conserv. 2001, 12, 207–216. [Google Scholar] [CrossRef]
- Zhang, W.; Pei, J.; Lai, L. Computational Multitarget Drug Design. J. Chem. Inf. Model. 2017, 57, 403–412. [Google Scholar] [CrossRef] [PubMed]
- Prachayasittikul, V.; Worachartcheewan, A.; Shoombuatong, W.; Songtawee, N.; Simeon, S.; Prachayasittikul, V.; Nantasenamat, C. Computer-Aided Drug Design of Bioactive Natural Products. Curr. Top. Med. Chem. 2015, 15, 1780–1800. [Google Scholar] [CrossRef] [PubMed]
- Jumper, J.; Evans, R.; Pritzel, A.; Green, T.; Figurnov, M.; Ronneberger, O.; Tunyasuvunakool, K.; Bates, R.; Zidek, A.; Potapenko, A.; et al. Highly accurate protein structure prediction with AlphaFold. Nature 2021, 596, 583–589. [Google Scholar] [CrossRef]
- Callaway, E. What’s next for AlphaFold and the AI protein-folding revolution. Nature 2022, 604, 234–238. [Google Scholar] [CrossRef]
- Yuriev, E.; Holien, J.; Ramsland, P.A. Improvements, trends, and new ideas in molecular docking: 2012–2013 in review. J. Mol. Recognit. 2015, 28, 581–604. [Google Scholar] [CrossRef]
- Husic, B.E.; Pande, V.S. Markov State Models: From an Art to a Science. J. Am. Chem. Soc. 2018, 140, 2386–2396. [Google Scholar] [CrossRef]
- Kuzmanic, A.; Sutto, L.; Saladino, G.; Nebreda, A.R.; Gervasio, F.L.; Orozco, M. Changes in the free-energy landscape of p38 alpha MAP kinase through its canonical activation and binding events as studied by enhanced molecular dynamics simulations. eLife 2017, 6, e22175. [Google Scholar] [CrossRef]
- Daskalakis, V.; Papadatos, S.; Stergiannakos, T. The conformational phase space of the photoprotective switch in the major light harvesting complex II. Chem. Commun. 2020, 56, 11215–11218. [Google Scholar] [CrossRef] [PubMed]
- Panagiotopoulos, A.A.; Karakasiliotis, I.; Kotzampasi, D.M.; Dimitriou, M.; Sourvinos, G.; Kampa, M.; Pirintsos, S.; Castanas, E.; Daskalakis, V. Natural Polyphenols Inhibit the Dimerization of the SARS-CoV-2 Main Protease: The Case of Fortunellin and Its Structural Analogs. Molecules 2021, 26, 6068. [Google Scholar] [CrossRef] [PubMed]
- Panagiotopoulos, A.A.; Papachristofi, C.; Kalyvianaki, K.; Malamos, P.; Theodoropoulos, P.A.; Notas, G.; Calogeropoulou, T.; Castanas, E.; Kampa, M. A simple open source bioinformatic methodology for initial exploration of GPCR ligands’ agonistic/antagonistic properties. Pharmacol. Res.Res. Perspect 2020, 8, e00600. [Google Scholar] [CrossRef] [PubMed]
- Castresana-Aguirre, M.; Sonnhammer, E.L.L. Pathway-specific model estimation for improved pathway annotation by network crosstalk. Sci. Rep. 2020, 10, 13585. [Google Scholar] [CrossRef]
- Lai, X.; Wang, X.; Hu, Y.; Su, S.; Li, W.; Li, S. Editorial: Network Pharmacology and Traditional Medicine. Front. Pharmacol. 2020, 11, 1194. [Google Scholar] [CrossRef]
- Wu, Z.; Li, W.; Liu, G.; Tang, Y. Network-Based Methods for Prediction of Drug-Target Interactions. Front. Pharmacol. 2018, 9, 1134. [Google Scholar] [CrossRef]
- Muhammad, J.; Khan, A.; Ali, A.; Fang, L.; Yanjing, W.; Xu, Q.; Wei, D.Q. Network Pharmacology: Exploring the Resources and Methodologies. Curr. Top. Med. Chem. 2018, 18, 949–964. [Google Scholar] [CrossRef]
- Liu, C.; Ma, Y.; Zhao, J.; Nussinov, R.; Zhang, Y.-C.; Cheng, F.; Zhang, Z.-K. Computational network biology: Data, models, and applications. Phys. Rep. 2020, 846, 1–66. [Google Scholar] [CrossRef]
- Kibble, M.; Saarinen, N.; Tang, J.; Wennerberg, K.; Makela, S.; Aittokallio, T. Network pharmacology applications to map the unexplored target space and therapeutic potential of natural products. Nat. Prod. Rep. 2015, 32, 1249–1266. [Google Scholar] [CrossRef]
- Hopkins, A.L. Network pharmacology: The next paradigm in drug discovery. Nat. Chem. Biol. 2008, 4, 682–690. [Google Scholar] [CrossRef]
- Castresana-Aguirre, M.; Guala, D.; Sonnhammer, E. Benefits and Challenges of Pre-clustered Network-Based Pathway Analysis. Front. Genet. 2022, 13, 855766. [Google Scholar] [CrossRef] [PubMed]
- Gramatica, P.; Cassani, S.; Roy, P.P.; Kovarich, S.; Yap, C.W.; Papa, E. QSAR Modeling is not “Push a Button and Find a Correlation”: A Case Study of Toxicity of (Benzo-)triazoles on Algae. Mol. Inform. 2012, 31, 817–835. [Google Scholar] [CrossRef] [PubMed]
- Gramatica, P. Principles of QSAR Modeling: Comments and Suggestions from Personal Experience. Int. J. Quant. Struct.-Prop. Relatsh. 2020, 5, 61–97. [Google Scholar] [CrossRef]
- Gramatica, P. Principles of QSAR models validation: Internal and external. QSAR Comb. Sci. 2007, 26, 694–701. [Google Scholar] [CrossRef]
- Daina, A.; Michielin, O.; Zoete, V. SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci. Rep. 2017, 7, 42717. [Google Scholar] [CrossRef] [PubMed]
- Schyman, P.; Liu, R.; Desai, V.; Wallqvist, A. vNN Web Server for ADMET Predictions. Front. Pharmacol. 2017, 8, 889. [Google Scholar] [CrossRef]
- Xiong, G.; Wu, Z.; Yi, J.; Fu, L.; Yang, Z.; Hsieh, C.; Yin, M.; Zeng, X.; Wu, C.; Lu, A.; et al. ADMETlab 2.0: An integrated online platform for accurate and comprehensive predictions of ADMET properties. Nucleic Acids Res. 2021, 49, W5–W14. [Google Scholar] [CrossRef]
- Wei, Y.; Li, S.; Li, Z.; Wan, Z.; Lin, J. Interpretable-ADMET: A web service for ADMET prediction and optimization based on deep neural representation. Bioinformatics 2022, 38, 2863–2871. [Google Scholar] [CrossRef]
- Gupta, M.; Sharma, R.; Kumar, A. Docking techniques in pharmacology: How much promising? Comput. Biol. Chem. 2018, 76, 210–217. [Google Scholar] [CrossRef]
- Murugan, N.; Priya, G.; Sastry, G.; Markidis, S. Artificial intelligence in virtual screening: Models versus experiments. Drug Discov. Today 2022, 27, 1913–1923. [Google Scholar] [CrossRef]
- Brown, D.G.; Wobst, H.J. A Decade of FDA-Approved Drugs (2010–2019): Trends and Future Directions. J. Med. Chem. 2021, 64, 2312–2338. [Google Scholar] [CrossRef] [PubMed]
- Ripphausen, P.; Nisius, B.; Peltason, L.; Bajorath, J. Quo vadis, virtual screening? A comprehensive survey of prospective applications. J. Med. Chem. 2010, 53, 8461–8467. [Google Scholar] [CrossRef] [PubMed]
- Kunimoto, R.; Bajorath, J.; Aoki, K. From traditional to data-driven medicinal chemistry: A case study. Drug Discov. Today 2022, 27, 2065–2070. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; WWan, Z.-Y.; Zheng, G.-H.; Li, S. TCM network pharmacology: A new trend towards combining computational, experimental and clinical approaches. Chin. J. Nat. Med. 2021, 19, 1–11. [Google Scholar] [CrossRef]
- Singh, H.; Bharadvaja, N. Treasuring the computational approach in medicinal plant research. Prog. Biophys. Mol. Biol. 2021, 164, 19–32. [Google Scholar] [CrossRef]
- Sabe, V.T.; Ntombela, T.; Jhamba, L.A.; Maguire, G.E.M.; Govender, T.; Naicker, T.; Kruger, H.G. Current trends in computer aided drug design and a highlight of drugs discovered via computational techniques: A review. Eur. J. Med. Chem. 2021, 224, 113705. [Google Scholar] [CrossRef]
- Cui, J.; McTigue, M.; Kania, R.; Edwards, M. Chapter Twenty-Five—Case History: Xalkori™ (Crizotinib), a Potent and Selective Dual Inhibitor of Mesenchymal Epithelial Transition (MET) and Anaplastic Lymphoma Kinase (ALK) for Cancer Treatment. Ann. Rep. Med. Chem. 2013, 48, 421–434. [Google Scholar] [CrossRef]
- Cui, W.; Aouidate, A.; Wang, S.; Yu, Q.; Li, Y.; Yuan, S. Discovering Anti-Cancer Drugs via Computational Methods. Front. Pharmacol. 2020, 11, 733. [Google Scholar] [CrossRef]
- Kania, R. Structure-Based Design and Characterization of Axitinib. In Kinase Inhibitor Drugs; Li, R., Stafford, J., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2009; pp. 167–202. [Google Scholar] [CrossRef]
- Meadows, K.L.; Hurwitz, H.I. Anti-VEGF therapies in the clinic. Cold Spring Harb. Perspect. Med. 2012, 2, a006577. [Google Scholar] [CrossRef]
- Gervasio, F.; Spiwok, V.; Mannhold, R. Biomolecular Simulations in Structure-Based Drug Discovery; Willey: Hoboken, NJ, USA, 2019; Volume 75. [Google Scholar]
- Kampa, M.; Hatzoglou, A.; Notas, G.; Damianaki, A.; Bakogeorgou, E.; Gemetzi, C.; Kouroumalis, E.; Martin, P.M.; Castanas, E. Wine antioxidant polyphenols inhibit the proliferation of human prostate cancer cell lines. Nutr. Cancer 2000, 37, 223–233. [Google Scholar] [CrossRef]
- Damianaki, A.; Bakogeorgou, E.; Kampa, M.; Notas, G.; Hatzoglou, A.; Panagiotou, S.; Gemetzi, C.; Kouroumalis, E.; Martin, P.M.; Castanas, E. Potent inhibitory action of red wine polyphenols on human breast cancer cells. J. Cell Biochem. 2000, 78, 429–441. [Google Scholar] [CrossRef]
- Nifli, A.P.; Bosson-Kouame, A.; Papadopoulou, N.; Kogia, C.; Kampa, M.; Castagnino, C.; Stournaras, C.; Vercauteren, J.; Castanas, E. Monomeric and oligomeric flavanols are agonists of membrane androgen receptors. Exp. Cell Res. 2005, 309, 329–339. [Google Scholar] [CrossRef] [PubMed]
- Nifli, A.P.; Kampa, M.; Alexaki, V.I.; Notas, G.; Castanas, E. Polyphenol interaction with the T47D human breast cancer cell line. J. Dairy Res. 2005, 72, 44–50. [Google Scholar] [CrossRef]
- Kampa, M.; Theodoropoulou, K.; Mavromati, F.; Pelekanou, V.; Notas, G.; Lagoudaki, E.D.; Nifli, A.P.; Morel-Salmi, C.; Stathopoulos, E.N.; Vercauteren, J.; et al. Novel oligomeric proanthocyanidin derivatives interact with membrane androgen sites and induce regression of hormone-independent prostate cancer. J. Pharmacol. Exp. Ther. 2011, 337, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Kalyvianaki, K.; Gebhart, V.; Peroulis, N.; Panagiotopoulou, C.; Kiagiadaki, F.; Pediaditakis, I.; Aivaliotis, M.; Moustou, E.; Tzardi, M.; Notas, G.; et al. Antagonizing effects of membrane-acting androgens on the eicosanoid receptor OXER1 in prostate cancer. Sci. Rep. 2017, 7, 44418. [Google Scholar] [CrossRef] [PubMed]
- Panagiotopoulos, A.A.; Kalyvianaki, K.; Serifoglou, B.; Konstantinou, E.; Notas, G.; Castanas, E.; Kampa, M. OXER1 mediates testosterone-induced calcium responses in prostate cancer cells. Mol. Cell Endocrinol. 2022, 539, 111487. [Google Scholar] [CrossRef]
- Kampa, M.; Papakonstanti, E.A.; Alexaki, V.I.; Hatzoglou, A.; Stournaras, C.; Castanas, E. The opioid agonist ethylketocyclazocine reverts the rapid, non-genomic effects of membrane testosterone receptors in the human prostate LNCaP cell line. Exp. Cell Res. 2004, 294, 434–445. [Google Scholar] [CrossRef]
- Panagiotopoulos, A.A.; Kalyvianaki, K.; Notas, G.; Pirintsos, S.A.; Castanas, E.; Kampa, M. New Antagonists of the Membrane Androgen Receptor OXER1 from the ZINC Natural Product Database. ACS Omega 2021, 6, 29664–29674. [Google Scholar] [CrossRef]
- Osakwe, O.; Rizvi, S. Social Aspects of Drug Discovery, Development and Commercialization; Academic Press: New York, NY, USA, 2016. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).