Abstract
Marine fungi produce many halogenated metabolites with a variety of structures, from acyclic entities with a simple linear chain to multifaceted polycyclic molecules. Over the past few decades, their pharmaceutical and medical application have been explored and still the door is kept open due to the need of new drugs from relatively underexplored sources. Biological properties of halogenated compounds such as anticancer, antiviral, antibacterial, anti-inflammatory, antifungal, antifouling, and insecticidal activity have been investigated. This review describes the chemical structures and biological activities of 217 halogenated compounds derived mainly from Penicillium and Aspergillus marine fungal strains reported from 1994 to 2019.
1. Introduction
Marine fungi are a treasure source of marine natural products. Marine-derived fungi are important providers of biologically prominent natural products due to their ability to produce secondary metabolites with novel structures and pharmacological activities. According to a paper on marine microbial natural products from 2010 to 2013 [1], natural products from marine fungi account for 63% of marine microorganisms. Due to the enormous amount of chloride and bromide ions available in seawater, many of these secondary metabolites are halogenated. Marine natural products cover a diverse assembly of molecules, including polyketides, peptides, terpenes, phenols, acetogenins, alkaloids, and volatile halogenated hydrocarbons [2]. The fungi isolated from the marine sources might also be found in the terrestrial region. However, marine derived fungi usually produce more halogenated compounds than their terrestrial counterparts due to the presence of high halogen concentrations in the Ocean. Halogenated natural products encompass many classes of compounds, ranging in complexity from halocarbons (mostly halomethanes and haloethanes) to higher molecular weight molecules, which often contain oxygen and/or nitrogen atoms in addition to halogens [3,4]. One of the major focal points of research undoubtedly has been the discovery and characterization of new halogenated compounds, along with a remarkable effort toward the assessment of their possible pharmacological activities and biomedical applications. Active compounds account for nearly 59.2% new halogenated natural products isolated from marine fungi. This paper provides an overview of the sources of marine-derived fungi, chemical structures, and biological activities of 217 halogenated compounds (Table S1) derived from marine fungi from 1994 to 2019.
2. Halogenated Compounds from Penicillium sp.
2.1. Sponges-Associated Penicillium sp.
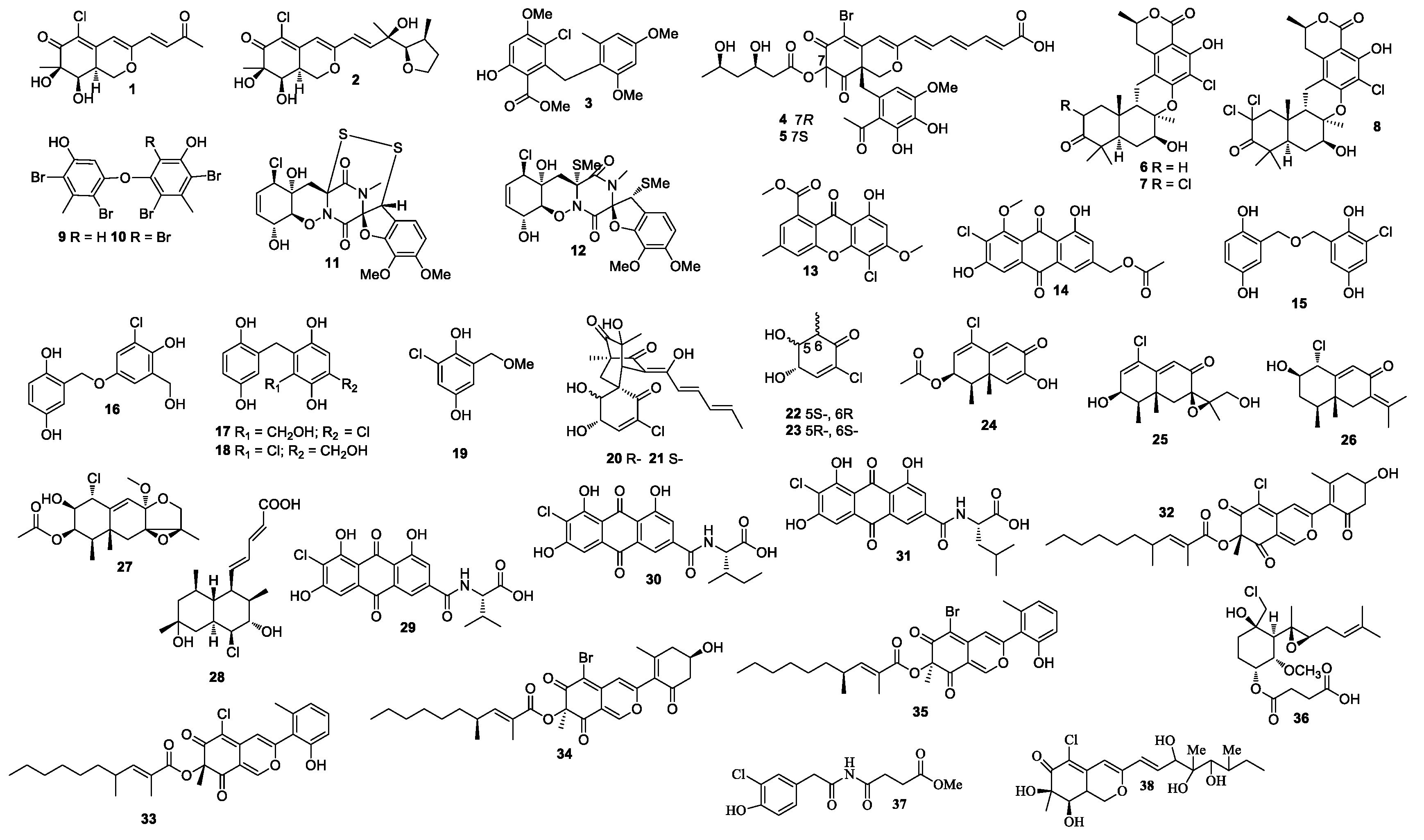
Two azaphilone derivatives penicilazaphilones D (1) and E (2) were isolated from a sponge-derived fungal strain Penicillium sclertiorum GDST-2013-0415 (Figure 1). Compound 2 was the first azaphilone with a tetrahydrofuran ring at C-3 [5]. A diphenyl ether methyl 3-chloro-2-(2,4-dimethoxy-6-methylphenoxy)-6-hydroxy-4-methoxybenzoate (3), bromophilones A (4) and B (5), were obtained from Penicillium canescens 4.14. 6a [6].
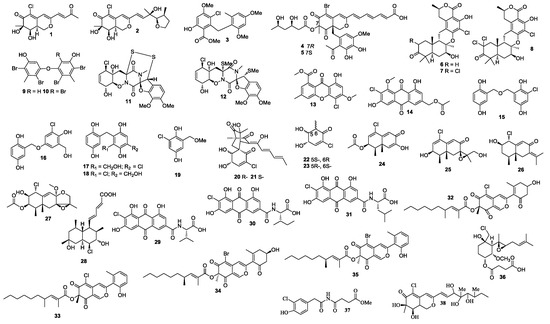
Figure 1.
Structures of compounds 1–38.
2.2. Other Marine Animals-Associated Penicillium sp.
New meroterpenoids chrodrimanins K and L (6 and 7) were separated from Penicillium sp. SCS-KFD09 (marine worm Sipunculus nudus), and 6 exhibited anti-H1N1 activity with an IC50 value of 74 μM [7]. A new meroterpenoid, named chrodrimanin O (8), was isolated from a fermentation of Penicillium sp. SCS-KFD09 (marine worm Sipunculus nudus). Compound 8 showed protein tyrosine phosphatase 1B inhibitory activity with an IC50 value of 71.6 μM [8].
2.3. Marine Algae-Associated Penicillium sp.
Diphenyl ethers 4,6,4′,6′-tetrabromo-3,3′-dihydroxy-5,5′-dimethyldiphenyl ether (9) and 4,6,2′,4′,6′-pentabromo-3,3′-dihydroxy-5,5′-dimethyldiphenyl ether (10) were obtained by feeding a culture of Penicillium chrysogenum with CaBr2. Compounds 9 and 10 showed 2,2-diphenyl-1-picrylhydrazyl (DPPH) activity with IC50 values of 18 and 15 μM, respectively [9].
2.4. Mangroves-Associated Penicillium sp.
Two new epipolythiodioxopiperazine alkaloids penicisulfuranols A (11) and D (12) with a rare spiro-furan ring, which were isolated from the mangrove endophytic fungus Penicillium janthinellum HDN13-309, showed cytotoxicity against Hela and HL-60 with IC50 values of 0.5 and 0.3, 0.1 and 1.2 μM, respectively [10]. In addition, 4-chloro-1-hydroxy-3-methoxy-6-methyl-8-methoxycarbonyl-xanthen-9-one (13) and 2′-acetoxy-7-chlorocitreorosein (14) were purified from the fungal strain Penicillium citrinum HL-5126, of which 14 showed activity against Vibrio parahaemolyticus with an MIC value of 10 μM [11].
2.5. Penicillium sp. from Marine Sediments
New gentisyl alcohol derivatives dimeric terrestrols B (15), D (16), F and G (17 and 18), and a monomer (19) were obtained from Penicillium terrestre and were cytotoxic toward HL-60, MOLT-4, A-549, and BEL-7402 with IC50 values in the range of 5.3 to 64.7 μM [12]. Compounds 15 and 16 exhibited tscavenging activity in a DPPH assay with IC50 values ranging from 4.1 to 5.2 μM. A study of the marine sediment derived fungus Penicillium terrestre resulted in the identification of chloctanspirones A (20), B (21), terrestrols K (22), and L (23). Compound 20 displayed inhibitory activity against HL-60 and A549 with IC50 values of 9.2 and 39.7 μM, respectively [13]. Compound 21 displayed inhibitory activity against HL-60 with an IC50 value of 37.8 μM. A chloro-trinoreremophilane sesquiterpene (24), and three chlorinated eremophilane-type sesquiterpenes (25–27) were purified from Penicillium sp. PR19N-1 isolated from the deep-sea sediment collected from Prydz Bay [14]. Compound 24 displayed inhibitory activity against HL-60 and A549 with IC50 values of 11.8 ± 0.2 and 12.2 ± 0.1 μM, respectively. Tanzawaic acid P (28) was isolated from a marine-derived fungal strain Penicillium sp. CF07370, and it was active against HeLa cell line with an IC50 value of 5.9 ± 0.8 µM after 72 h [15]. Emodacidamides C (29), F (30), and G (31) were obtained from a marine-derived fungal strain Penicillium sp. SCSIO sof101. Compound 29 inhibited interleukin-2 secretion with an IC50 value of 4.1 μM [16]. Penicilones C (32) and D (33) were purified from Penicillium janthinellum HK1-6, which were active against methicillin-resistant S. aureus (MRSA, ATCC 43300, ATCC 33591, ATCC 25923, ATCC 29213) and E. faecalis (ATCC 51299, ATCC 35667) with MIC values ranging from 3.13 to 12.5 μg/mL [17]. Penicillium janthinellum HK1-6 produced two azaphilones penicilones G (34) and H (35), which were active against MRSA (ATCC 43300, ATCC 33591, ATCC 25923, ATCC 29213) and E. faecalis (ATCC 51299, ATCC 35667) with MIC values in the range of 3.13–50 μg/mL [18].
2.6. Penicillium sp. from Other Marine Sources
Ligerin (36) was separated from Penicillium canescentia MMS351, which showed cytotoxicity against the POS1 cell with an IC50 value of 117 nM [19]. Ligerin was synthesized from fumagillin, and it showed good activity against SaOS2 [20]. The culture of Penicillium copticola TPU1270 (marine foam, Iriomote Island, Okinawa Prefecture, Japan) yielded penicillimide (37) [21]. A new azaphilone penicilazaphilone C (38), which was isolated from the fungus Penicillium sclerotiorum M-22, showed cytotoxicity against B-16 and SGC-7901 with IC50 values of 0.065 and 0.720 mM, respectively. Compound 38 also exhibited strong antibacterial activity against Pseudomonas aeruginosa, Staphylococcus aureus, Escherichia coli, and Klebsiella pneumonia with MIC values ranging from 0.037 to 0.150 mM [22].
3. Halogenated Compounds from Aspergillus sp.
3.1. Sponges-Associated Aspergillus sp.
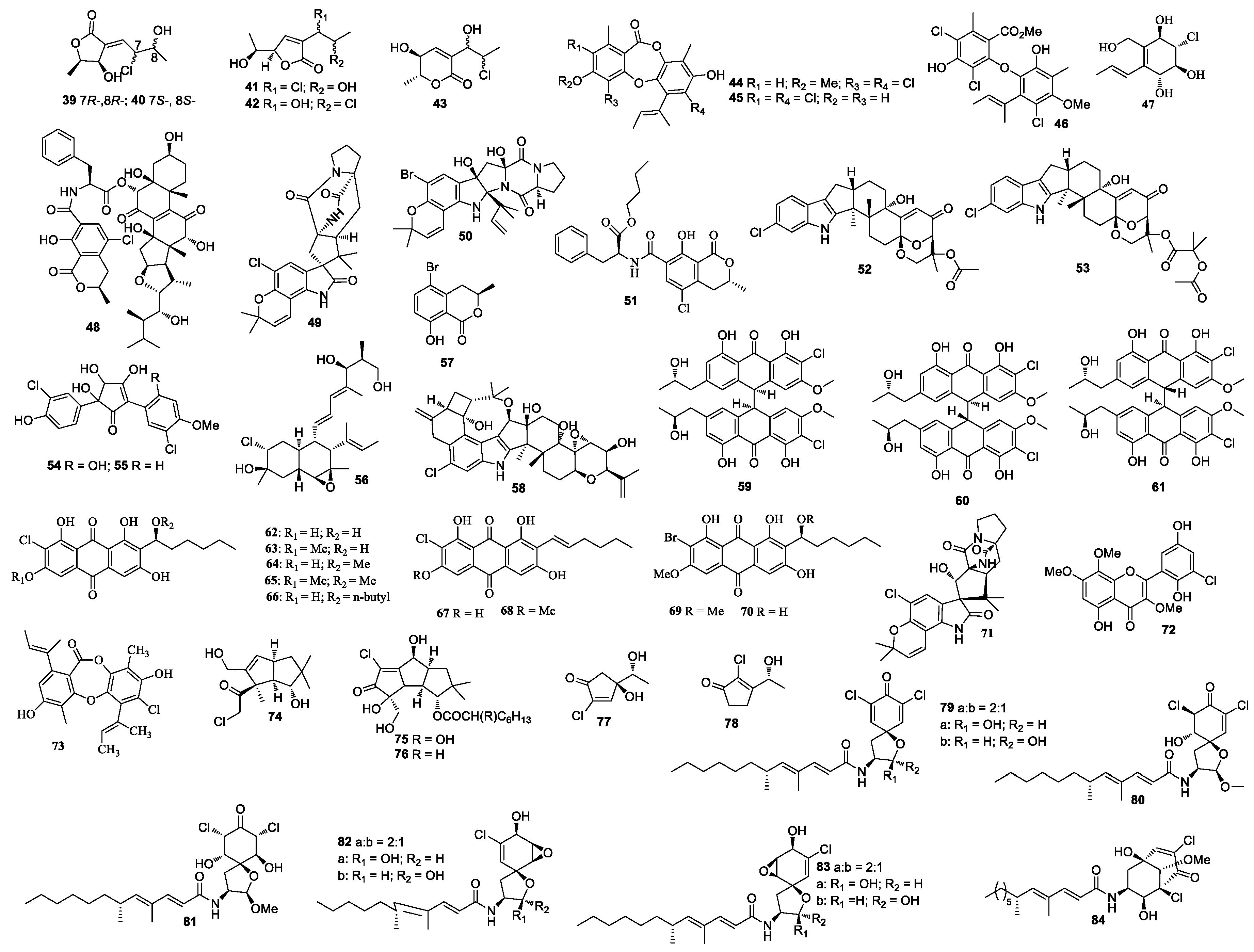
Two new polyketides chlorocarolides A (39) and B (40) were from Aspergillus cf. ochraceus 941,026 [23] (Figure 2). Aspergillus ostianus TUF 01F313 yielded 8-chloro-9-hydroxy-8,9-deoxyasperlactone (41), 9-chloro-8-hydroxy-8,9-deoxyasperlactone (42), and 9-chloro-8-hydroxy-8,9-deoxyaspyrone (43), of which compounds 42 and 43 inhibited the growth of Ruegeria atlantica at 25 μg/disc with an inhibition zone diameter of 10.1 and 10.5 mm, respectively [24]. Compound 41 was active against Regenia atlantica with an inhibition diameter of 12.7 mm at 5 μg/disc, and it was also active against S. aureus with an inhibition diameter of 10.2 mm at 25 μg/disc. Aspergillusidones B (44), C (45), and aspergillusether A (46) were separated from Aspergillus unguis CRI282-03 [25]. Compounds 44 and 45 inhibited aromatase with IC50 values of 4.1 and 0.7 μM, respectively. Compound 44 showed scavenging activity in a DPPH assay with an IC50 value less than 15.6 µM. Aspergillus sp. OUCMDZ-1583 (an unidentified marine sponge XD10410, Xisha Islands, China) produced a new metabolite, aspergone O (47), which inhibited α-glucosidase with an IC50 value of 1.54 mM [26]. Ochrasperfloroid (48) from Aspergillus flocculosus 16D-1 (the sponge Phakellia fusca, Yongxing Island, China) showed inhibitory activity towards THP-1 and NO production in LPS-activated RAW264.7, with IC50 values of 2.02 and 1.11 μM, respectively [27].
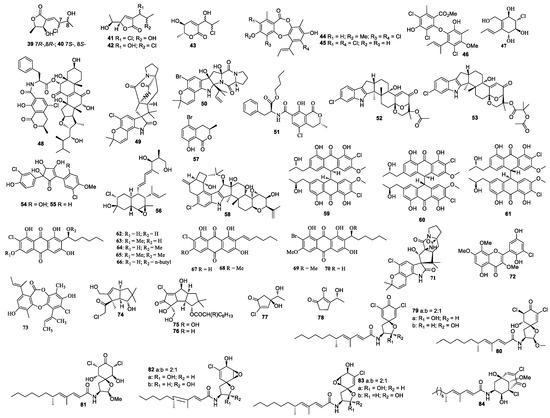
Figure 2.
Structures of compounds 39–84.
3.2. Other Marine Animals-Associated Aspergillus sp.
Notoamide N (49) and notoamide P (50) were isolated from the cultures of Aspergillus sp. MF297-2 [28,29]. A mycotoxin ochratoxin A n-butyl ester (51) was isolated from a marine-derived fungal strain Aspergillus sp. SCSGAF0093 from Melitodes squamata collected from the South China Sea. The bio-toxicity of compound 51 was determined by the brine shrimp lethality bioassay with a median lethal concentration (LC50) value of 4.14 μM [30]. Two new indole-diterpene alkaloids asperindoles A (52) and C (53) were isolated from the fermentation broth of Aspergillus sp. KMM 4676, of which compound 52 showed cytotoxicity toward PC-3 cells, LNCaP cells, and 22Rv1 cells, with IC50 values of 69.4, 47.8, and 4.86 µM, respectively [31].
3.3. Marine Algae-Associated Aspergillus sp.
Aspergillus sydowii produced sydowins A (54) and B (55) [32]. A polyoxygenated decalin derivative dehydroxychlorofusarielin B (56) was isolated from the culture of Aspergillus sp. MFB024, which exhibited antibacterial activities against S. aureus, MRSA, and multidrug-resistant S. aureus with an equal MIC of 62.5 μg/mL [33]. (R)-(–)-5-bromomellein (57), produced by Aspergillus ochraceus, exhibited radical scavenging activity against DPPH with an IC50 value of 24 μM [34]. Aspergillus nidulans EN-330 afforded a chlorinated indole-diterpenoid 19-hydroxypenitrem A (58), which inhibited cytotoxic activity against brine shrimp with a LD50 value of 3.2 μM and showed antibacterial activities [35]. Aspergillus alliaceus afforded allianthrones A–C (59–61), among which 59 displayed cytotoxic activity against the HCT-116 and SK-Mel-5 with IC50 values of 9.0 and 11.0 μM, respectively [36].
3.4. Aspergillus sp. from Marine Sediments
A study on the Aspergillus sp. SCSIO F063 derived from the marine sediment sample resulted in the discovery of chlorinated anthraquinones (1′S)-7-chloroaverantin (62), (1′S)-6-O-methyl-7-chloroaverantin (63), (1′S)-1′-O-methyl-7-chloroaverantin (64), (1′S)-6,1′-O,O-dimethyl-7-chloroaverantin (65), (1′S)-7-chloroaverantin-1′-butyl ether (66), 7-chloroaverythrin (67), 6-O-methyl-7-chloroaverythrin (68), brominated anthraquinones (1′S)-6,1′-O,O-dimethyl-7-bromoaverantin (69), and (1′S)-6-O-methyl-7-bromoaverantin (70) [37], of which compounds 63, 64, and 70 exhibited cytotoxic activities against SF-268 with MIC values of 7.11 ± 0.14, 34.06 ± 2.98, and 24.69 ± 0.72 µM, respectively. Compounds 63, 64, and 70 also showed cytotoxic activities against NCI-H460 with MIC values of 7.42 ± 0.14, 37.19 ± 1.95, and 18.91 ± 1.43 µM, respectively. Compounds 63, 64, and 70 further demonstrated cytotoxic activities against MCF-7 with MIC values of 6.64 ± 0.36 to 49.53 ± 0.72 µM, respectively. The deep-sea-derived fungal strain A. westerdijkiae DFFSCS013 afforded a new prenylated indole alkaloid 5-chlorosclerotiamide (71), which showed cytotoxicity against K562 with an MIC value of 44 μM [38].
3.5. Aspergillus sp. from Other Marine Sources
5’-Hydroxychlorflavonin (72) was purified from Aspergillus sp. AF119 [39]. A new depsidone 7-chlorofolipastatin (73) was isolated from Aspergillus ungui NKH-007 collected in the Suruga Bay, which inhibited SOAT1 and SOAT2 isozymes [40].
4. Halogenated Compounds from Other Marine Fungi
4.1. Other Sponges-Associated Fungi
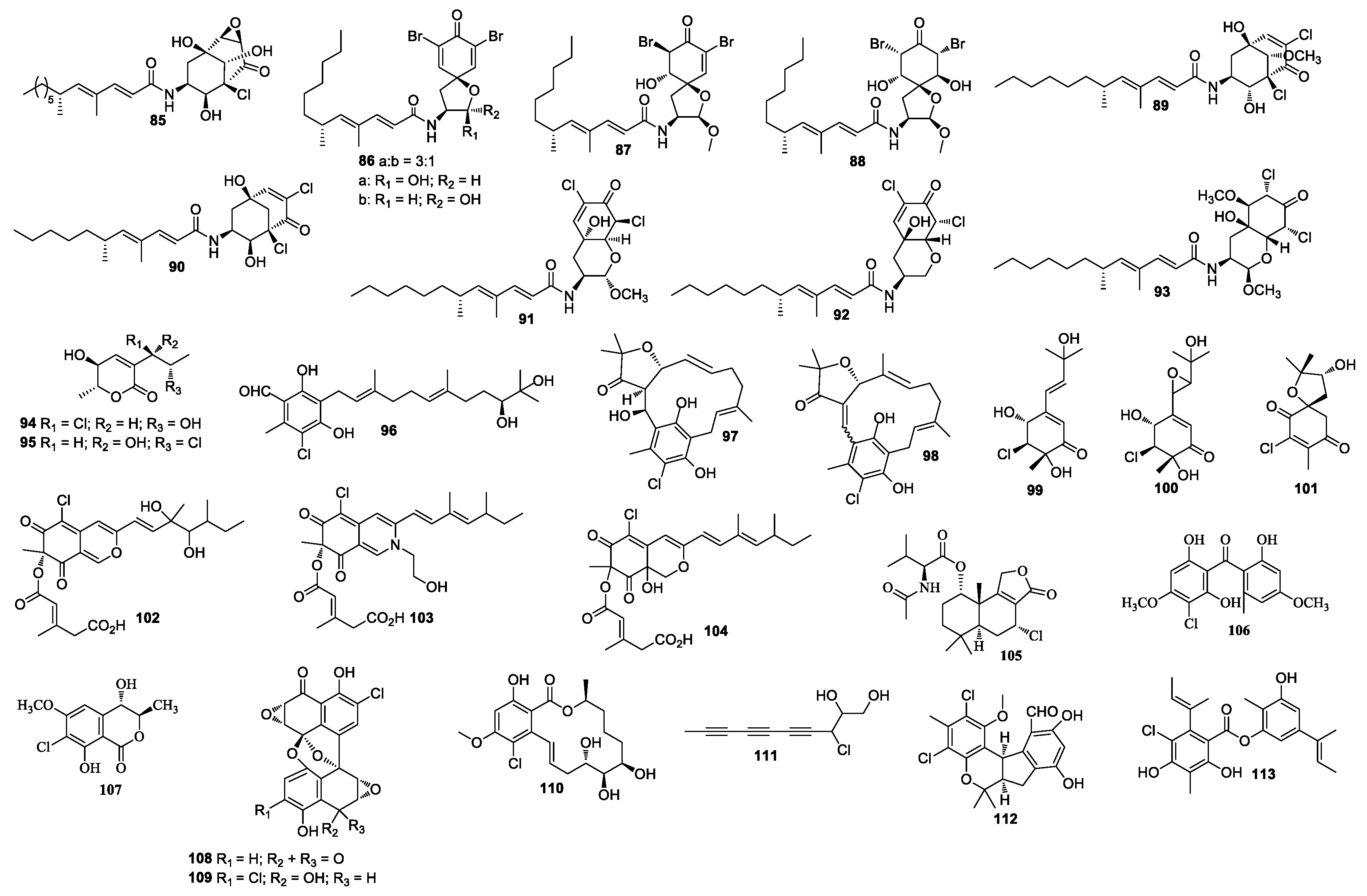
Cultivation of an unidentified fungal strain afforded three new chlorinated sesquiterpenes chloriolins A–C (74–76). Compound 74 inhibited human tumor cell lines T-47D and SNB-75 with IC50 values of 0.7 and 0.5 μM, respectively [41]. Trichodenone B (77), and trichodenone C (78) isolated from Trichoderma harzianum OUPS-N115 exhibited anticancer activity against P388 with ED50 values of 1.21 and 1.45 μg/mL, respectively [42]. Trichodenones B and C were synthesized by Usami et al. [43]. Gymnastatins A–G (79–85) [44,45,46], I–K (86–88) [47], Q (89) and R (90) [48], and dankastatins A–C (91–93) [48,49] were isolated from the cultures of Gymnascella dankaliensis. Gymnastatin A (79) was synthesized by anodic oxidation of the corresponding phenols [50]. Gymnastatins F (84) and Q (85) were synthesized by the tandem Michael and aldol reaction [51] (Figure 3). These compounds (79–93) showed cytotoxicity against P388, among which compounds 86 and 87 exhibited cytotoxicity against 39 human cancer cell lines with the average of log GI50 at −5.77 and −5.71, respectively. Compound 86 exhibited strong cytotoxic effect against HBC-5, NCI-H522, OVCAR-3, and MKN1, while compound 87 strongly inhibited SF-539, HCT-116, NCI-H522, OVCAR-3, and OVCAR-8. Compound 89 showed cytotoxicity against 39 human cancer cell lines with mean log GI50 values at −4.81, which also demonstrated cytotoxicity against BSY-1 and MKN7 with mean log GI50 values at −5.47 and −5.17, respectively. Compound 93 showed cytotoxicity against the P388 cell line with an ED50 value of 57 ng/mL. In a 2008 report, chlorohydroaspyrones A and B (94 and 95) obtained from Exophiala sp. showed antibacterial activity against S. aureus and multidrug-resistant S. aureus with an equal MIC value of 62.5 and 125 μg/mL [52]. Both compounds 94 and 95 demonstrated antimicrobial activity against MRSA with MIC values of 125 and 62.5 μg/mL, respectively. A culture of Acremonium sp. J05B-1-F-3 produced compounds 96–98 [53]. 5-Chloroacremines A and H (99 and 100), acremine O (101) were obtained from Acremonium persicinum [54]. New chloroazaphilone derivatives helicusin E (102), isochromophilone X (103), and isochromophilone XI (104) were isolated from Bartalinia robillardoides LF550. Compound 104 displayed antibacterial activity against Bacillus subtilis, Staphylococcus lentus, and Trichophyton rubrum with IC50 values of 55.6, 78.4, and 41.5 μM, respectively. Compounds 103 and 104 showed inhibitory activity against PDE4 with IC50 values of 11.7 and 8.30 μM [55], respectively. Minioluteumide A (105) was isolated from Talaromyces minioluteus, which showed weak cytotoxic activity [56]. Stachybogrisephenone B (106) was isolated from Stachybotry sp. HH1 ZSDS1F1-2, which displayed inhibitory activity against intestinal virus EV71 with an IC50 value of 30.1 μM and inhibited cyclooxygenase with an IC50 value of 8.9 μM [57]. One new isocoumarin derivative 107 was separated from Phoma sp. 135 [58], which was isolated from the sponge Ectyplasia perox collected in Dominica, Lauro Club Reef.
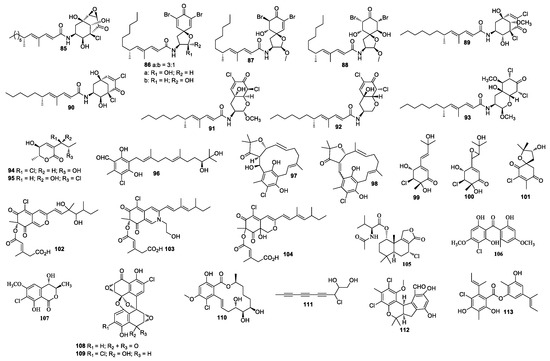
Figure 3.
Structures of compounds 85–113.
4.2. Other Marine Animals-Associated Fungi
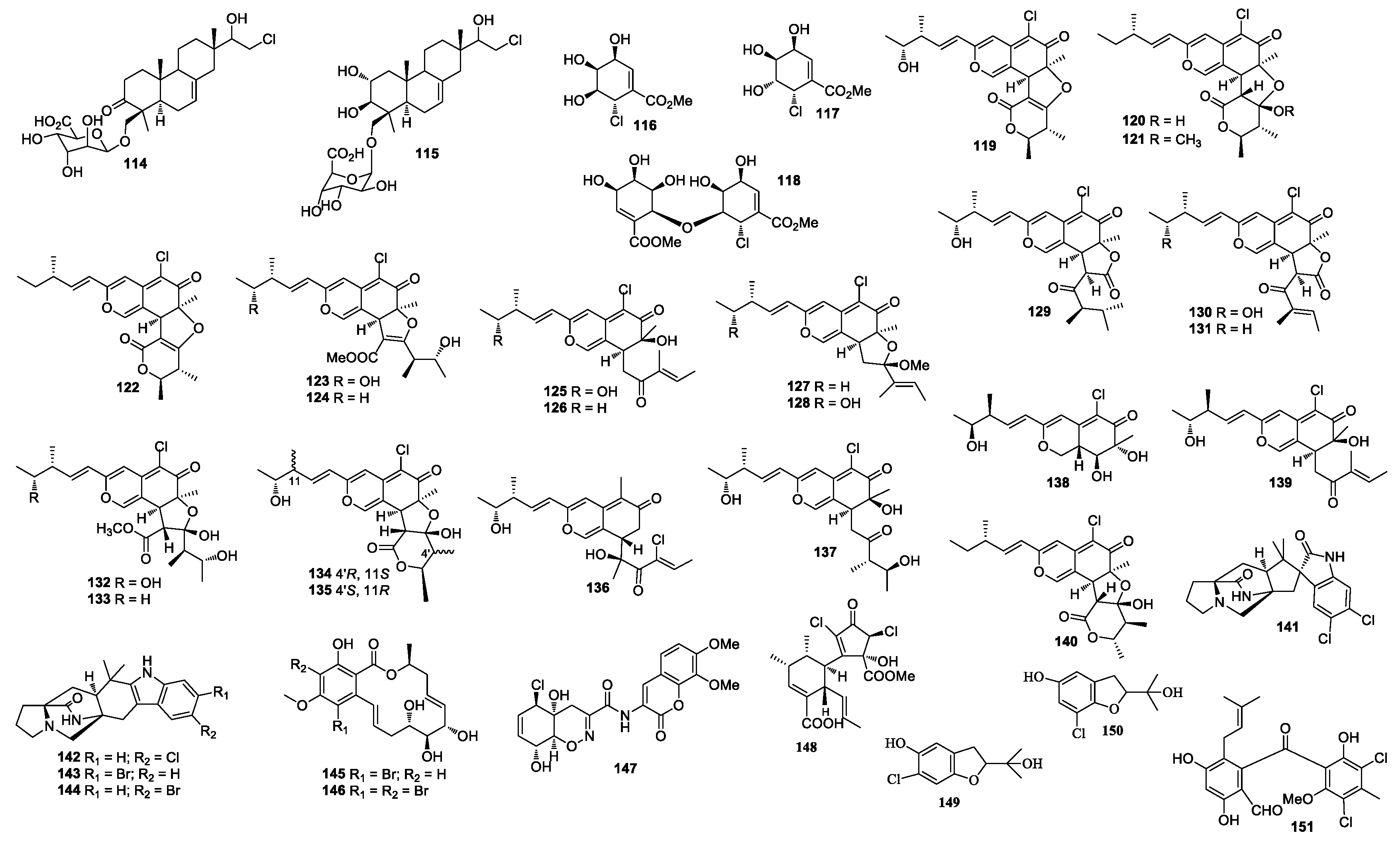
A marine-derived fungus LL-37H248 produced spiroxins A (108) and E (109). Compound 108 showed growth inhibition against 25 cancer cell lines with a mean IC50 value of 0.09 μg/mL [59]. The total synthesis of spiroxin A has been achieved in two competing cascade processes [60]. Cochliomycin C (110) was obtained from Cochliobolus lunatus, which was isolated from the gorgonian coral D. gemmacea [61]. Cochliomycin C (110) was synthesized from sugar D-lyxose [62]. Chondrosterin H (111) was purified from Chondrostereum sp. nov. SF002. The fungal strain SF002 was isolated from the coral Sarcophyton tortuosum [63]. A new chlorinated benzophenone derivative named (±)-pestalachloride D (112) was obtained from Pestalotiopsis sp. ZJ-2009-7-6 [64], which showed inhibitory activity against Escherichia coli, Vibrio anguillarum, and V. parahaemoly-ticus with MIC values of 5, 10, and 20 μM, respectively. (±)-Pestalachloride D was synthesized by way of a biomimetic Knoevenagel/Hetero-Diels–Alder Cascade reaction [65]. One new depside guisinol (113), which was active against S. aureus (5 mg/mL DMSO, 15 μL added), was identified from the metabolites of Emericella unguis M87-2 isolated from the cannonball jellyfish Stomolopus meliagris [66]. The chemical investigation of Acremonium striatisporum KMM 4401 from the sea cucumber Eupentacta fraudatrix [67] yielded two compounds, virescenosides Z5 and Z7 (114 and 115) (Figure 4). Pericosines A (116), D (117), and E (118) [68] were obtained from Periconia byssaides OUPS-N133, which was isolated from the sea hare Aplysia kurodai. Pericosine A (116) was synthesized from diverse aromatic cis-dihydrodiol precursors by the chemoenzymatic synthesis [69]. Compound 118 was synthesized by Mizuki et al. in 2014 [70]. Chaetomium globosum OUPS-T106B-6 isolated from the flathead grey mullet Mugil cephalus (Japan) yielded chaetomugilins C (119) [71,72], D-F (120–122) [73], G (123), H (124) [74], and I–O (125–131) [75], seco-chaetomugilins A (132) and D (133) [76], 11-epi-chaetomugilin A (134), 4’-epi-chaetomugilin A (135) [77], chaeto-mugilins P–R (136–138), 11-epi-chaetomugilin I (139) [78], chaetomugilin S (140), of which 119–122 were cytotoxic against P388 and HL-60 cell lines with IC50 values of 3.3–15.7 and 1.3–13.2 μM [71,72], respectively. Compounds 123–128, 130–131, and 134 showed growth inhibition against many cancer cell lines. (−)-Spiromalbramide (141), (+)-isomalbrancheamide B (142), (+)-malbrancheamide C (143), and isomalbrancheamide B (144) were produced by Malbranchea graminicola 086937A [79]. Two new brominated resorcylic acid lactones, 5-bromozeaenol (145) and 3,5-dibromozeaenol (146) [80] were produced by Cochliobolus lunatus TA26-46 induced by inhibitors of histone deacetylase. C. lunatus TA26–46 was isolated from the Zoanthid Palythoa haddoni. Trichodermamide B (147) was obtained from Trichoderma virens CNL910, which displayed cytotoxicity against HCT-116 with IC50 values of 0.32 μg/mL [81]. Compound 147 also showed inhibitory activity against C. albicans, vancomycin-resistant E. faecium, and MRSA with an equal MIC value of 15 μg/mL. The synthesis of 147 was reported by Lu and Zakarian in 2008 [82]. An unprecedented polyketide carbon skeleton roussoellatide (148) was obtained from the marine-derived fungus Roussoella sp. DLM33 [83]. Two benzofuran derivatives, 6-chloro-2-(2-hydroxypropan-2-yl)-2,3-dihydro-5-hydroxybenzofuran and 7-chloro-2-(2-hydroxypropan-2-yl)-2,3-dihydro-5-hydroxybenzofuran (149 and 150) were separated from Pseudallescheria boydii, which was isolated from the crown-of-thorns starfish Acanthaster planci (Hainan Sanya National Coral Reef Reserve, Hainan) [84].
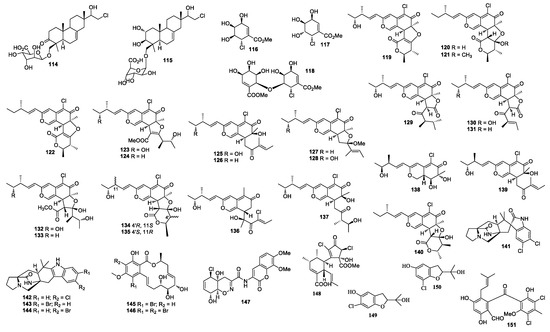
Figure 4.
Structures of compounds 114–151.
4.3. Other marine Algae-Associated Fungi
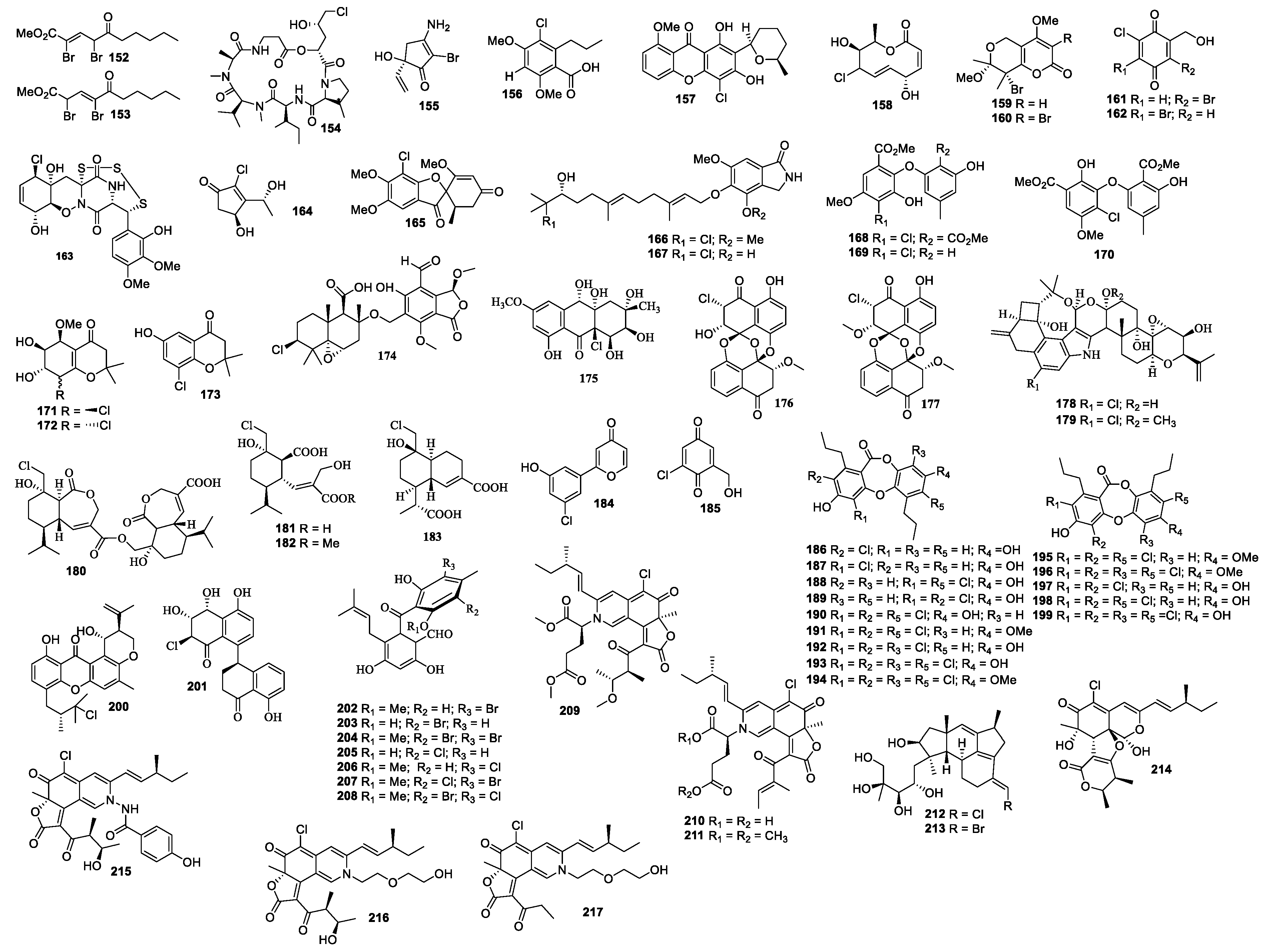
A new benzophenone pestalone (151) was isolated from a coculture broth of Pestalotia sp. CNL-365 and bacterium strain CNJ-328A. Compound 151 exhibited inhibitory activity against MRSA and vancomycin-resistant Enterococcus faecium with MIC values of 37 and 78 ng/mL, respectively. Compound 151 was cytotoxic against the NCI 60 human cancer cell lines with a mean GI50 value of 6.0 μM [85]. Compound 151 was synthesized with orcinol as the starting material [86]. Two new alkenoates, methyl 2,4-dibromo-5-oxo-2-decenoate (152) and methyl 2,4-dibromo-5-oxo-3-decenoate (153) were discovered from an unidentified fungus from the seaweed Gracillaria verrucose [87] (Figure 5). The chemical investigation of a culture of Beauveria felina yielded [β-MePro] destruxin E (154) [88]. A study of Botrytis sp. led to the identification of bromomyrothenone B (155) [89]. Acremonisol A (156) was obtained from Acremonium sp. [90]. Chaetoxanthone C (157) was separated from Chaetomium sp., which was active against Trypanosoma cruzi with an IC50 value of 1.5 μg/mL [91]. A 10-membered lactone (158) was isolated from a culture of Curvularia sp. 768 associated with the marine red algae Acanthophora spicifera [92]. Two new pyranopyranones, bromomethylchlamydosporols A (159) and B (160) were obtained from Fusarium tricinctum, which was active against SA, MRSA, and MDRSA with an equal MIC value of 15.6 μg/mL [93]. Bromochlorogentisylquinones A (161) and B (162) were isolated from Phoma herbarum and showed scavenging activity in a DPPH assay with IC50 values of 3.8 and 3.9 μM, respectively [94]. Trichoderma sp. (cf. T. brevicompactum) TPU199 in natural seawater medium supplemented with dimethyl sulfoxide afforded an unprecedented trithio-derivative of epidiketopiperazine, chlorotrithiobrevamide (163) [95]. One new trichodenone 3-hydroxytrichodenone C (164) was isolated from Trichoderma asperellum cf44-2 and showed antibacterial activities against four Vibrio strains with the inhibitory zone of 6.5–8.5 mm at 20 μg/disk. Compound 164 was active against Prorocentrum donghaiense, Karlodinium veneficum, Heterosigma akashiwo, and Chattonella marina with IC50 values of 37, 39, 35, and 30 μg/mL, respectively [96].
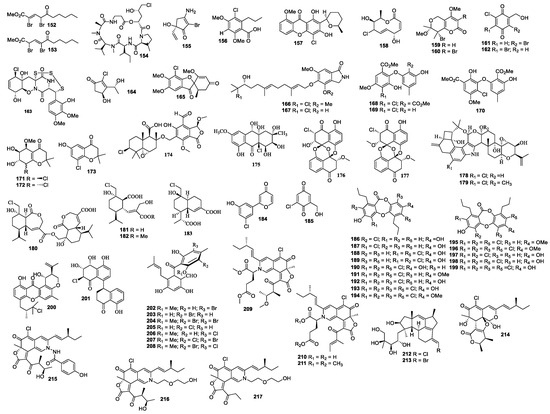
Figure 5.
Structures of compounds 152–217.
4.4. Other Mangroves-Associated Fungi
A new griseofulvin derivative 7-chloro-2’,5,6-trimethoxy-6’-methylspiro(benzofuran-2(3H),1’-(2) cyclohexene)3,4’-dione (165) was produced by Sporothrix sp. 4335 [97]. Emeriphenolicins A (166) and B (167) were produced by Emericella sp. HK-ZJ and were found to show antiviral activity with IC50 values of 42.1 and 62.0 μg/mL, respectively [98]. Pestalotethers A-C (168–170) and pestalochromones A-C (171–173) were purified from Pestalotiopsis sp. PSU-MA69, which was isolated from a branch of a mangrove plant Rhizophora apiculata [99]. Pestalotiopene C (174), a polyketide derivative, was obtained from Acremonium strictum, collected from the mangrove tree Rhizophora apiculate Blume [100]. Paradictyoarthrinium diffractum BCC 8704 produced a new hydroanthraquinone, paradictyoarthrin A (175), which showed cytotoxicity against KB, MCF-7, NCI-H187, and Vero with IC50 values of 26, 24, 23, and 31 μg/mL, respectively [101]. The marine mangrove A. ilicifolius provided Lasiodiplodia theobromae ZJ-HQ1, which produced chloropreussomerins A (176) and B (177). Compounds 176 and 177 showed antimicrobial activity against S. aureus and B. subtili with MIC values of 6.2, 50, 3.2, and 25 μg/mL, respectively. Compounds 176 and 177 also showed cytotoxicity against A549, HepG2, HeLa, MCF-7, and HEK293T with IC50 values ranging from 5.9 to 27 μM [102]. Rhizovarins A and B (178 and 179) were separated from a fermentation of Mucor irregularis QEN-189 isolated from Rhizophora stylosa (Hainan Island) and were cytotoxic against A-549 with IC50 values of 11.5 and 9.6 μM, respectively. Both compouds 178 and 179 were also cytotoxic to HL-60 cells with IC50 values of 6.3 and 5.0 μM, respectively [103]. Sesquiterpenoid derivatives, rhinomilisins A–C (180–182) and I (183) were isolated from Rhinocladiella similis, of which 180 showed cytotoxicity against L5178Y with an IC50 value of 5.0 μM [104].
4.5. Other Marine Plants-Associated Fungi
Polyporapyranone D (184) with a 2-phenylpyranon-4-one derivative skeleton was isolated from an extract of Polyporales sp. PSU-ES44 [105].
4.6. Other Marine Sediments-Associated Fungi
Chlorogentisylquinone (185) was purified from a marine-derived fungus FOM-8108, which showed nSMase activity with an IC50 value of 1.2 μM [106]. Spiromastixones B-O (186–199) were isolated from Spiromastix sp. MCCC3A00308, which exhibited antibacterial activity against Staphylococcus aureus ATCC 29213, Bacillus thuringiensis SCSIO BT01, and Bacillus subtilis SCSIO BT01 with MIC values in the range of 0.125–8.0 μg/mL. Compounds 190–194 exhibited activity against MRSA and S. epidermidis (MRSE) with the same inhibitory activity as levofloxacin. Compound 194 displayed inhibitory activity against VREF and VRE with an equal IC50 value of 4 μM [107]. Emerixanthone A (200) was isolated from Emericella sp. SCSIO 05240, which exhibited weak antibacterial activity against Klebsiella pneumonia (ATCC 13883), Escherichia coli (ATCC 29922), Staphylococcus aureus (ATCC 29213), Aeromonas hydrophila (ATCC 7966), Acineto bacterbaumannii (ATCC 19606), and Enterococcus faecalis (ATCC 29212) [108]. Cladosporol G (201) was purified from a fermentation of Cladosporium cladosporioides HDN14-342, which was isolated from a sediment sample (Indian Ocean). Compound 201 was cytotoxic against HeLa cell line with an IC50 value of 3.9 μM [109]. Pestalotiopsis neglecta yielded pestalones B–H (202–208), which were cytotoxic against PANC-1, A549, HCT116, MCFM, DU145, and HepG2 tumor cell lines with IC50 values in the range of 4.8–37 μM [110]. Chaetomium globosum HDN151398 yielded azaphilone alkaloids N-glutarylchaetoviridins A–C (209–211). Compound 211 exhibited cytotoxicity against HO8910 and MGC-803 with IC50 values of 6.6 and 9.7 µM, respectively [111].
4.7. Other Marine Source-Associated Fungi
A culture of F. heterosporum CNC-477 produced neomangicols A and B (212 and 213). Compound 212 was cytotoxic against MCF-7 and CACO-2 cells with IC50 values of 4.9 and 5.7 μM, respectively, and compound 213 showed antibacterial activity against B. subtilus at 50 μg/disc with an inhibition zone diameter of 10 mm [112]. Chaephilone C (214) and chaetoviridides A–C (215–217) were isolated from Chaetomium sp. NA-S01-R1. These compounds (214–217) showed antimicrobial activity and cytotoxicity [113].
5. Conclusions
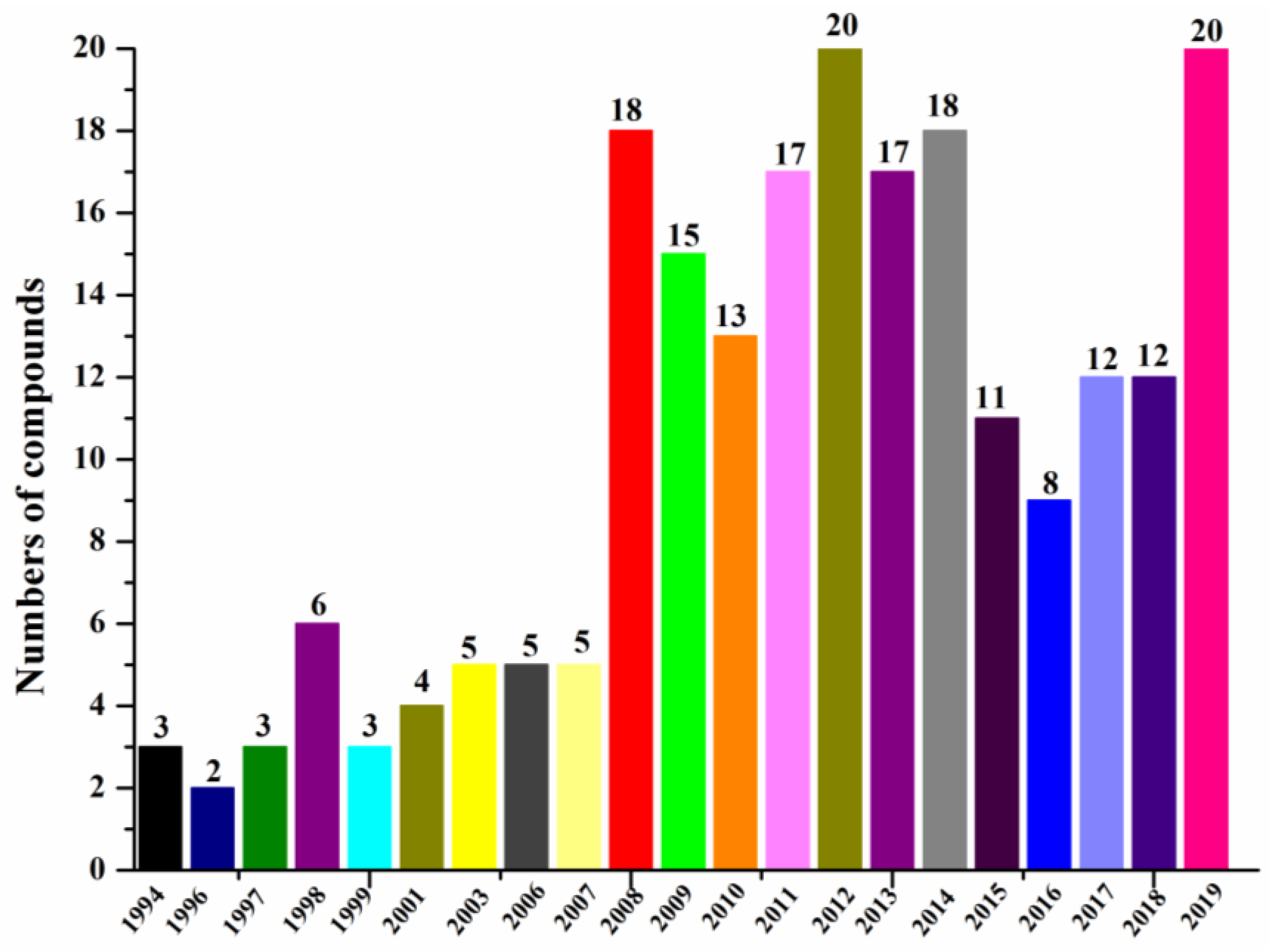
According to our summary of halogenated compounds identified from 1994 to 2019 (Figure 6, Table 1), the research on halogenated compounds from marine fungi was traced back to 1994 when chloriolins A–C (74–76) were discovered from an unidentified fungus isolated from the Indo-Pacific sponge Jaspis aff. johnstoni (Table 2) [41]. Since 2008, more new halogenated compounds than ever from marine fungi were isolated annually except before 2016. By the end of 2019, 217 new halogenated compounds from marine fungi have been reported. We have done our best to include as many new halogenated compounds isolated from marine fungi as possible, but the list may still not be complete.
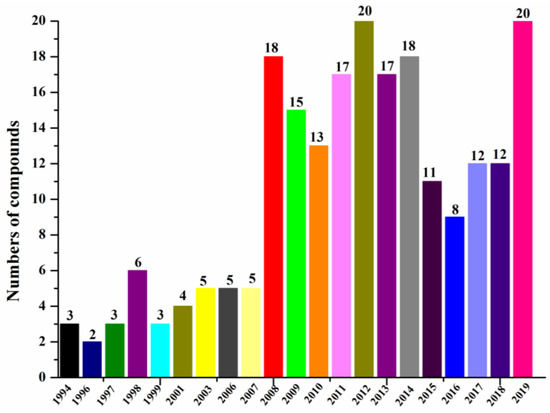
Figure 6.
Numbers of new halogenated compounds reported annually from 1994–2019.

Table 1.
The initial research on antimicrobial active compounds from fungi.

Table 2.
Halogenated compounds isolated from marine fungi (1994–2019).
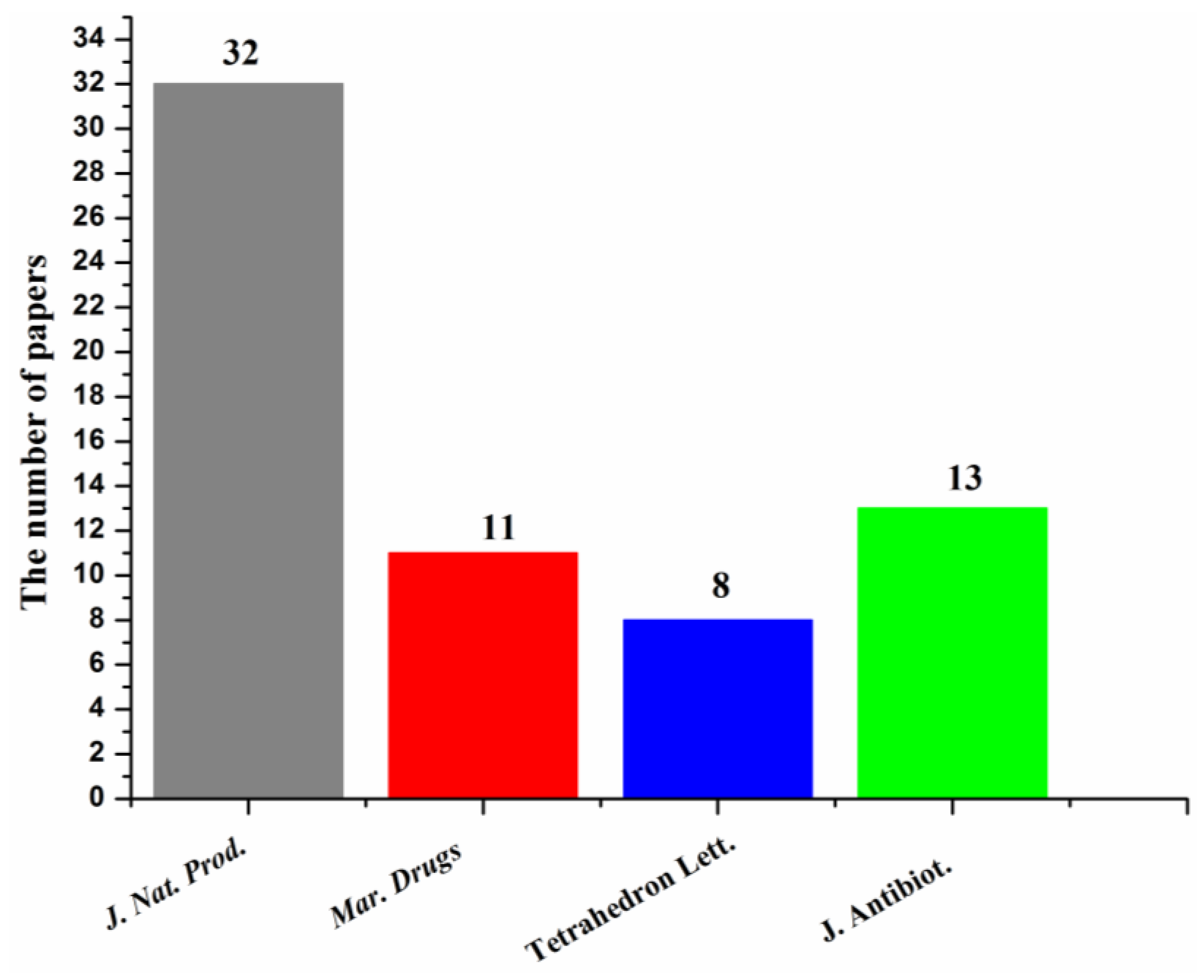
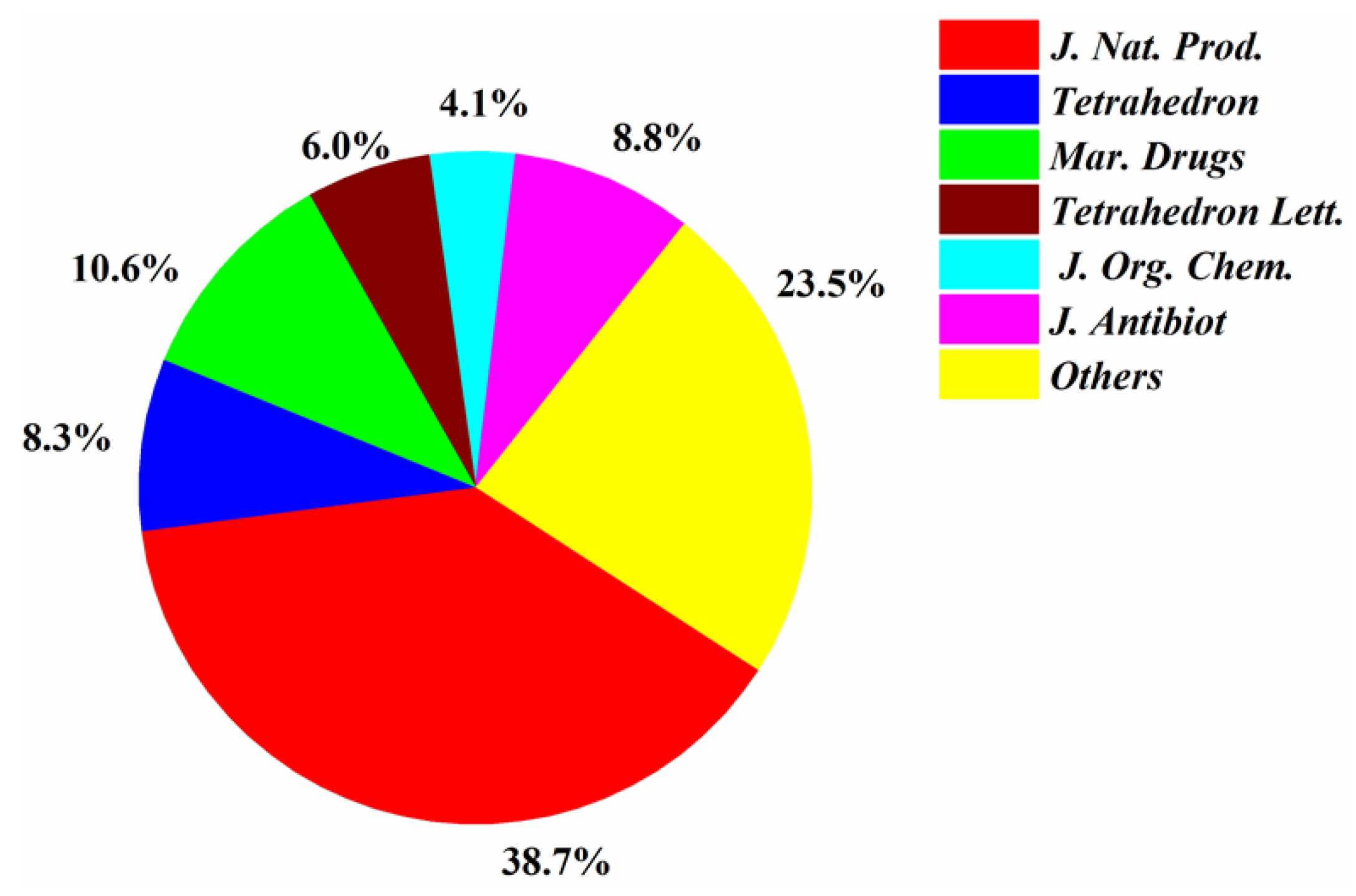
Most of the papers that reported new halogenated compounds in this period of time (1994–2019) were published in J. Nat. Prod. (32), J. Antibiot. (13), Marine Drugs (11), and Tetrahedron Letters (8) (Figure 7). The main journals that reported new halogenated compounds from marine fungi were J. Nat. Prod. (38.7%), J. Antibiot. (8.8%), Tetrahedron (8.3%), Mar. Drugs (10.6%), Tetrahedron Lett. (6.0%), and J. Org. Chem. (4.1%) (Figure 8). J. Nat. Prod. is the most preeminent journal that published more articles and more new halogenated compounds than any other journal.
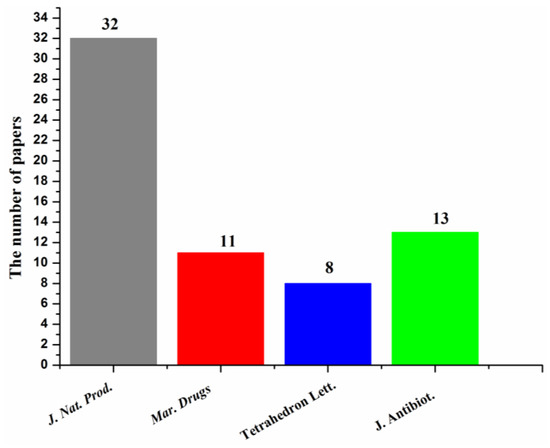
Figure 7.
Journals that reported new halogenated compounds and numbers of papers published (1994–2019).
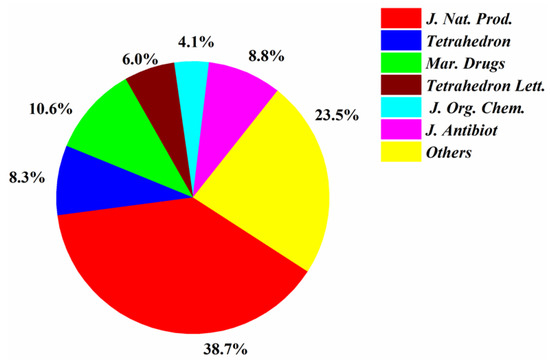
Figure 8.
Percentages of new halogenated compounds published in different journals (1994–2019).
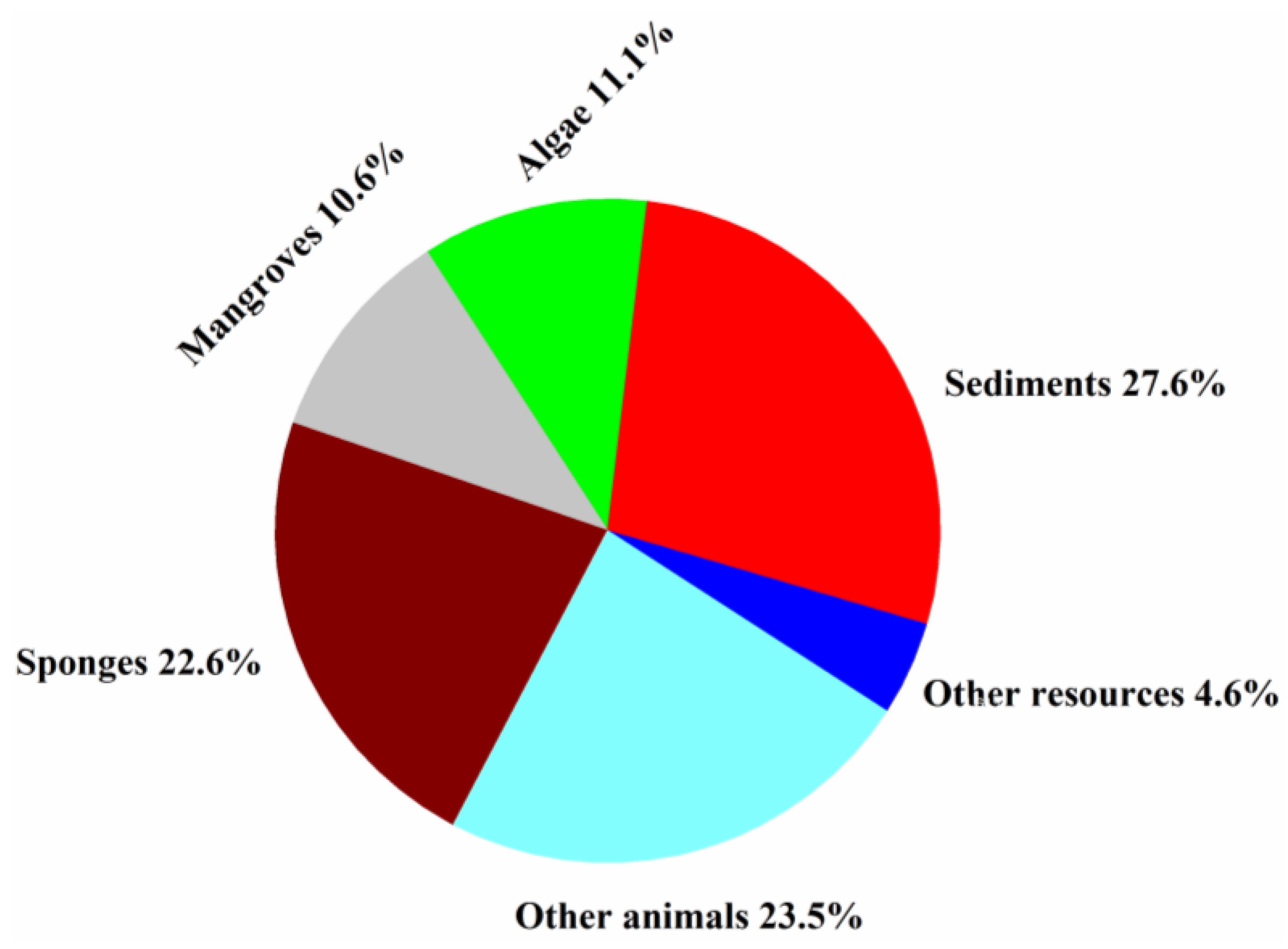
Fungi isolated from sponges, sediments, algae, and mangroves produced most of the new halogenated compounds (22.6, 27.6, 11.1, and 10.6%, respectively) (Figure 9). Marine animals hosted diverse fungal species and strains that produced more than 50% of the new halogenated compounds from 1994 to 2019, indicating that they are an excellent source for the discovery of new halogenated compounds.
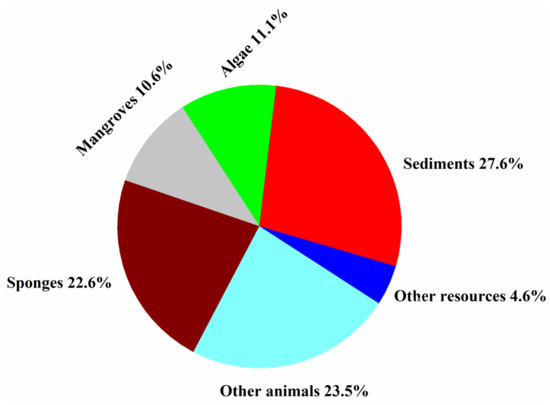
Figure 9.
Percentages of new halogenated compounds from different sources of marine origins (1994–2019).
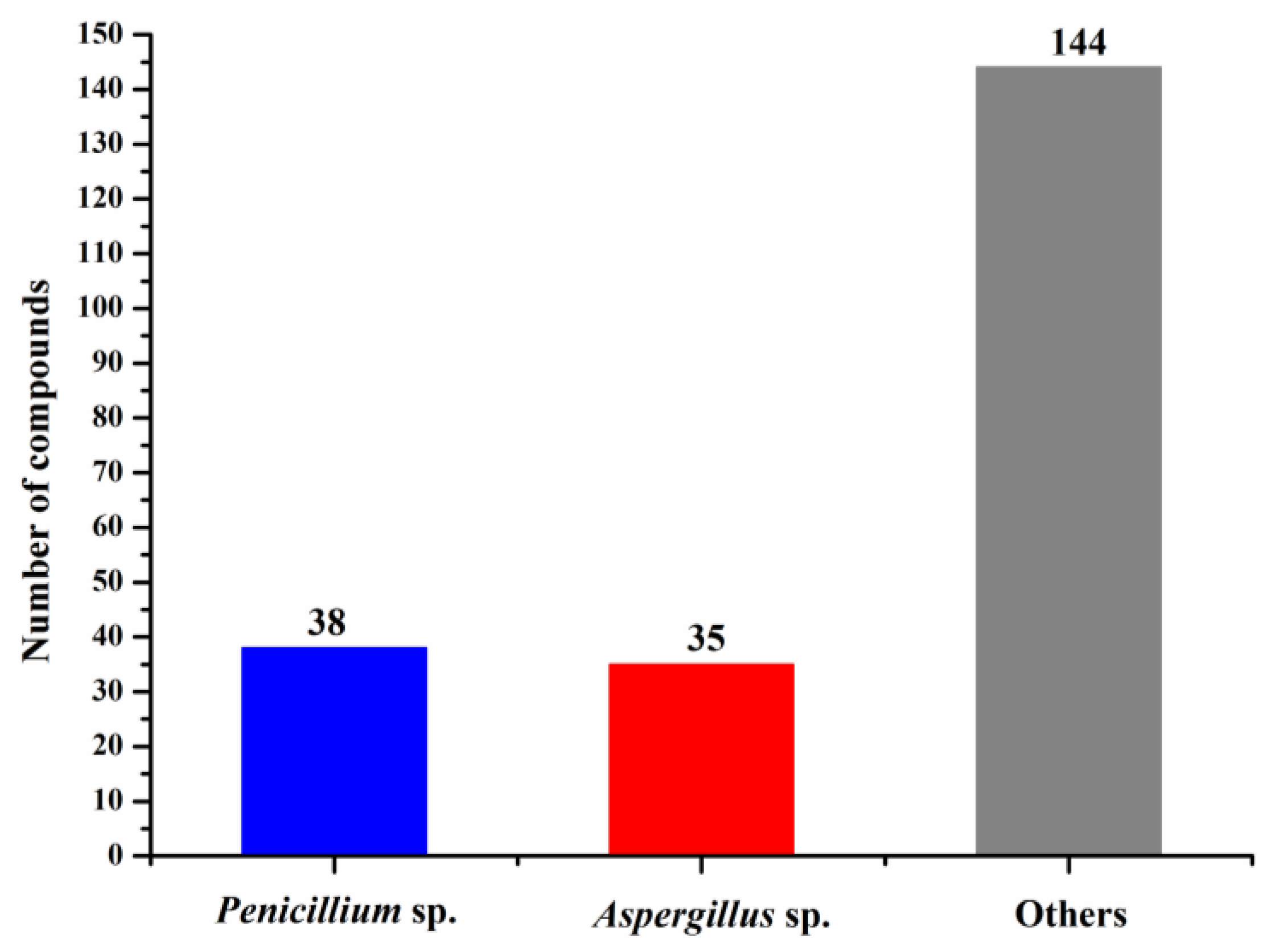
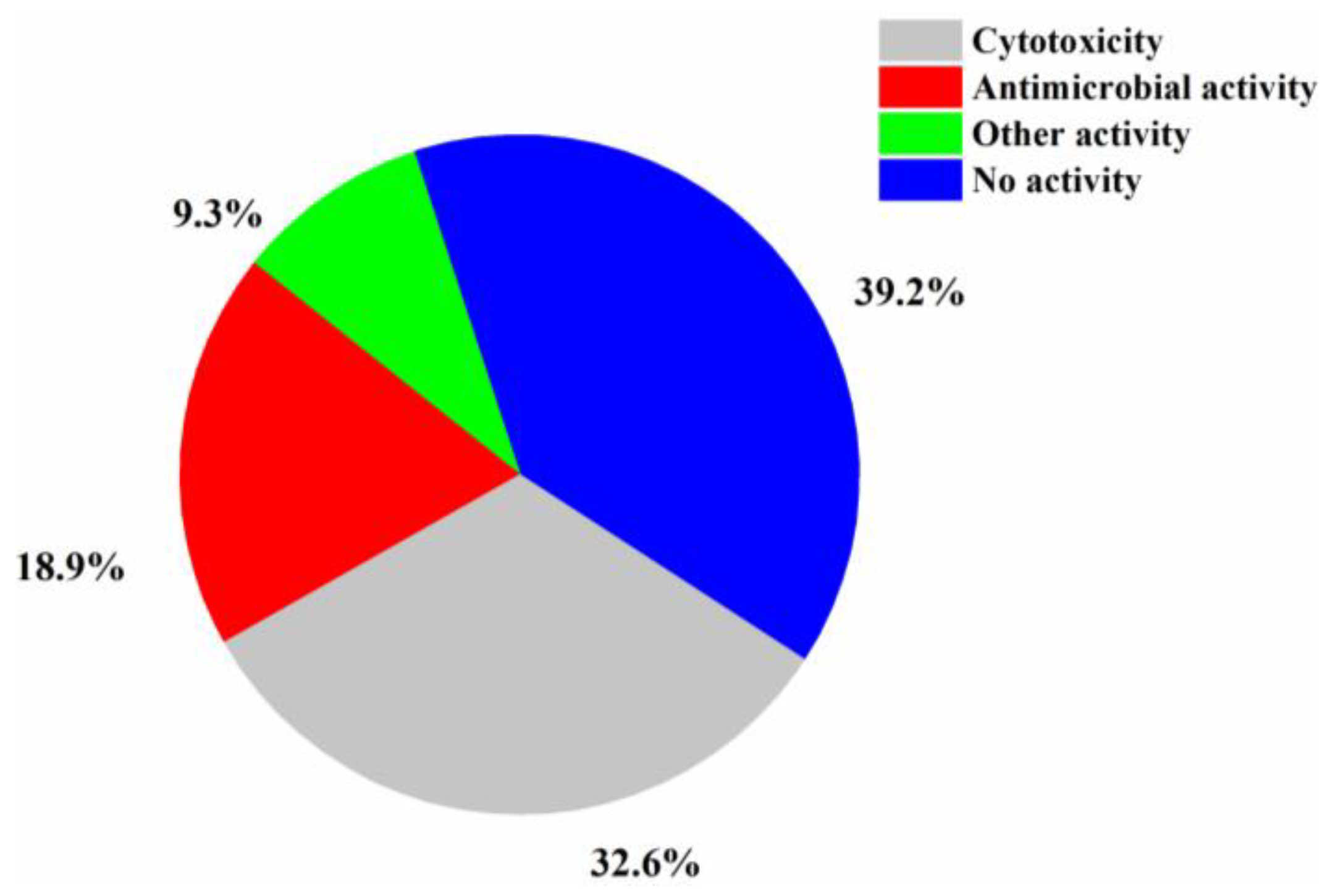
The numbers of halogenated compounds from marine Penicillium sp., Aspergillus sp., and the other fungi were 38, 35, and 144, respectively (Figure 10). It seems that halogenation in the marine environment is not specifically favorable to any fungal species or strains. Therefore, it would be interesting to investigate whether halogenations in marine fungi are enzymatic or nonenzymatic. The numbers of cytotoxic and antimicrobial halogenated compounds from marine fungi account for 32.6 and 18.9%, respectively (Figure 11). In addition, 39.2% of the halogenated compounds were tested as inactive in the reported assays, but it is worthy to evaluate these compounds in other biological settings.
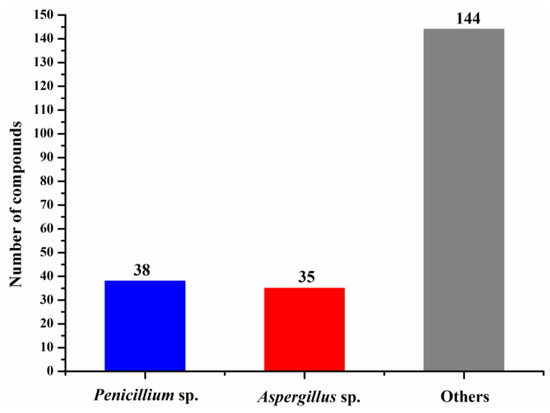
Figure 10.
Numbers of new halogenated compounds from different marine fungi (1994–2019).
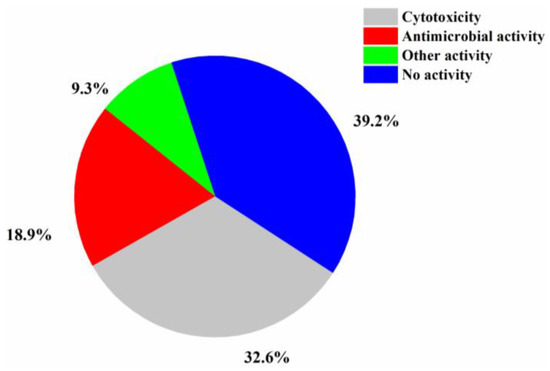
Figure 11.
Activity of new halogenated compounds from marine fungi (1994–2019).
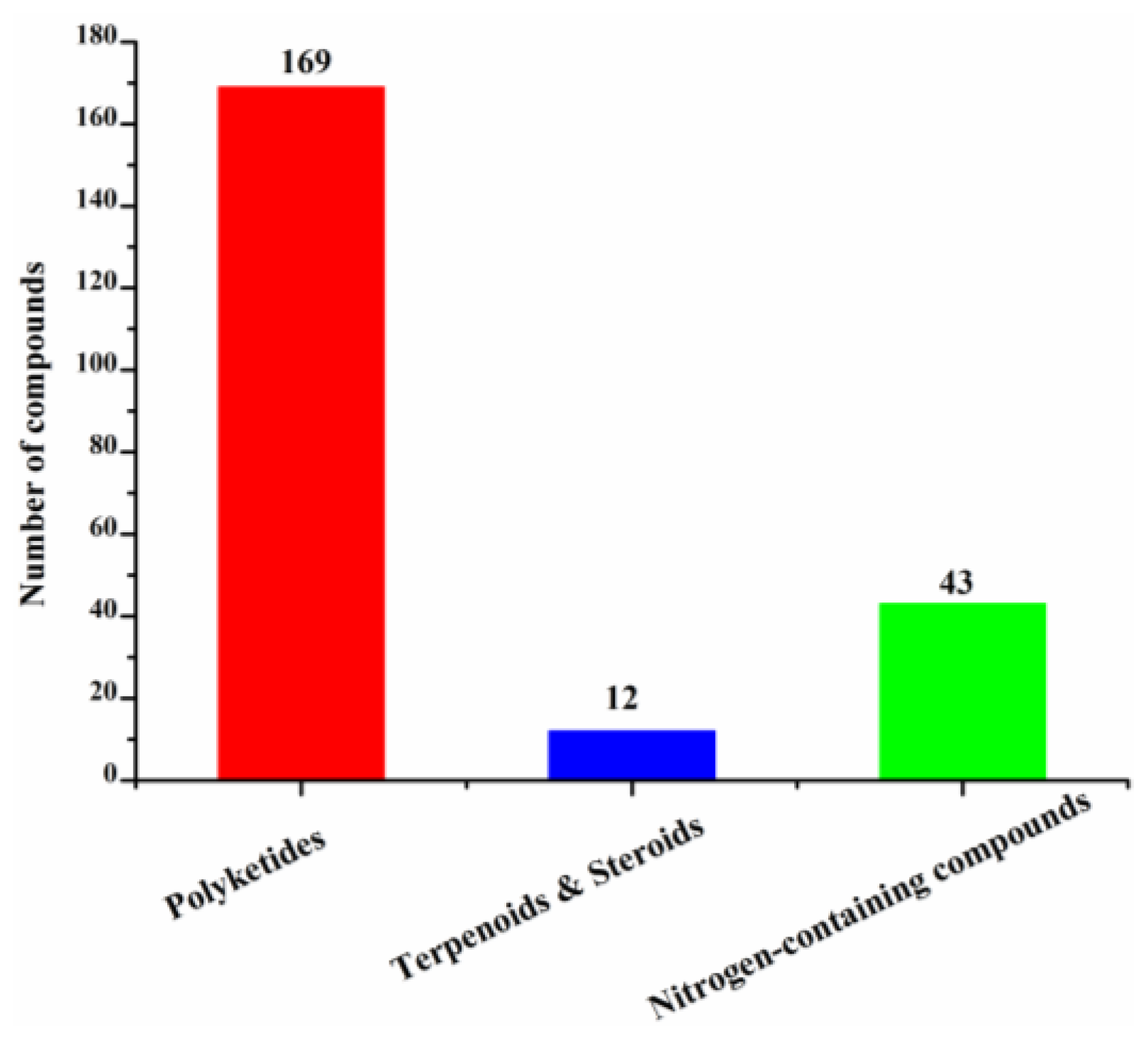
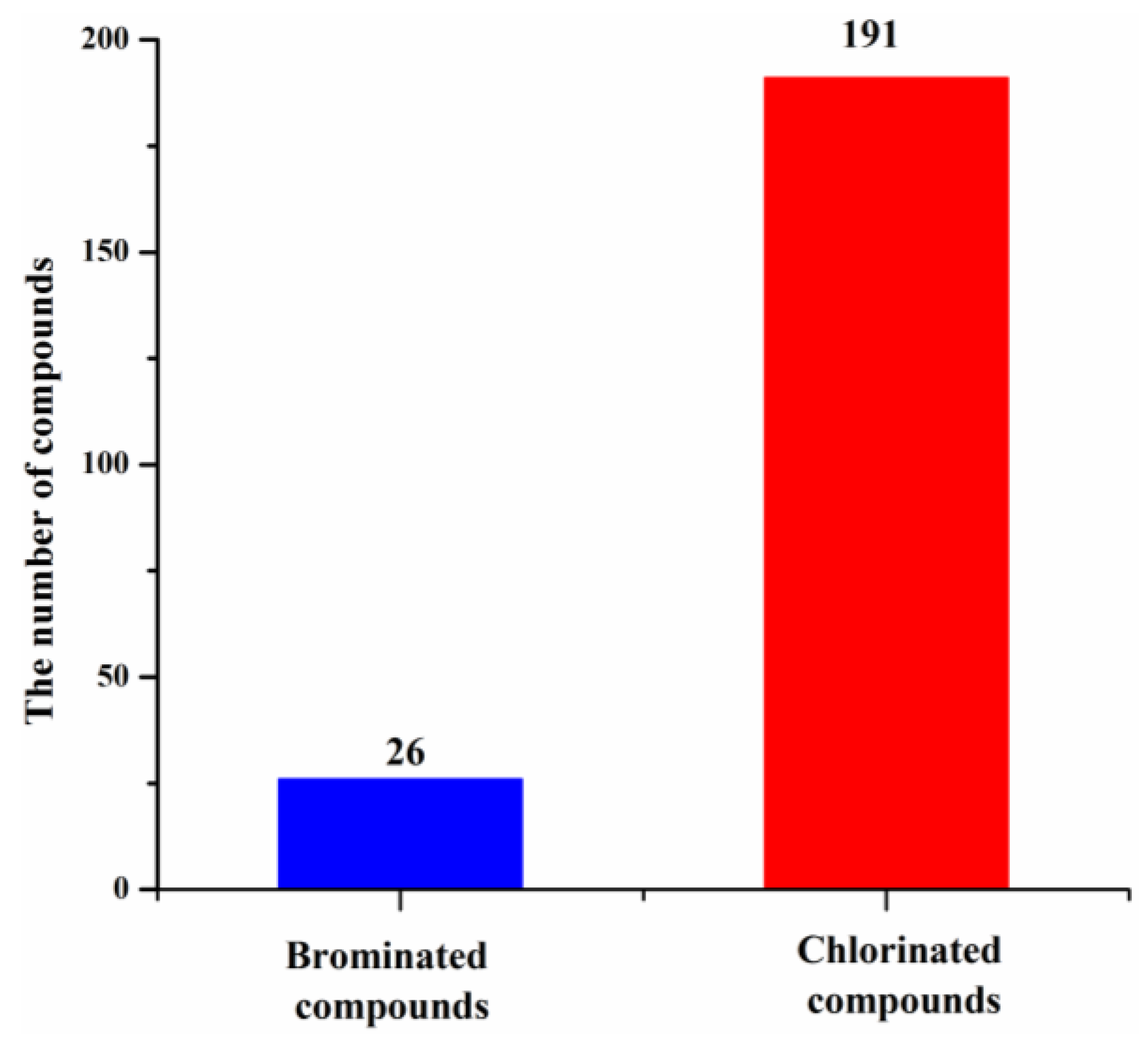
These new marine natural products from marine fungi have different structure skeletons including polyketides, nitrogen-containing compounds, sterols, and terpenoids (Figure 12). Polyketides account for the majority (169, 78%) of the new halogenated compounds (217) isolated from marine fungi (Figure 12). The number of chlorinated compounds is 191, which is far more than that of brominated compounds simply due to the fact that chloride/chlorine is dominant in the Ocean when compared with bromide/bromine (Figure 13).
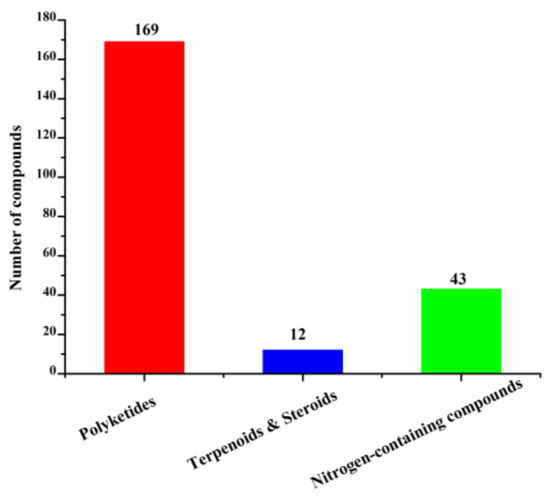
Figure 12.
Structural classes of new halogenated compounds (1994–2019).
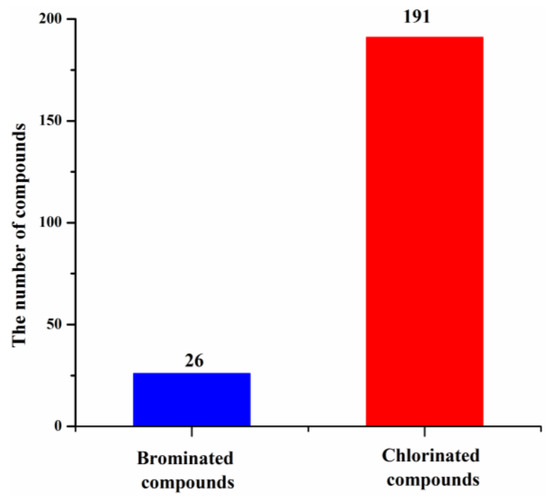
Figure 13.
Proportion of new halogenated compounds (1994–2019).
One of the challenges of discovering promising biologically active secondary metabolites from marine fungi is to mimic the culture environment as the marine. The surrounding environment such as oxygen, pressure, light, and salinity etc. significantly influence the growth of the marine fungi, as well as their ability to produce secondary metabolites. Although it is a challenge, investigating marine fungi for their halogenated secondary metabolites is worth it since more than 60% halogenated compounds isolated from marine fungi have some kind of significant biological activities. It is also worthy to assess halogenated compounds in a broader range of assays.
Supplementary Materials
The Supplementary Materials are available online.
Author Contributions
S.C. and C.W. conceived and designed the format of the paper; C.W. edited the article and analyzed the data; H.L. and J.L. drew the structures of the compounds; K.A.Z. reviewed the manuscript; S.C. revised the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by the Guangxi Natural Science Foundation under grant number 2019GXNSFBA185002, a specific research project of Guangxi for research bases and talents (AD18126005, AD18281066). Seed grants from the University of Hawaii at Hilo (UHH), start-up funding from the University of Hawaii Cancer Center (UHCC) and Daniel K. Inouye College of Pharmacy (DKICP), and the Victoria S. and Bradley L. Geist Foundation (15ADVC-74420, 17CON-86295, and 20CON-102163) to S.C. Funding for this work was also supported by the Hawaii IDeA Network for Biomedical Research Excellence III and IV (INBRE-III and INBRE-IV) project: NIGMS grant number 5P20GM103466.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available in this article and supplementary material.
Conflicts of Interest
The authors declare no conflict of interest.
Sample Availability
Samples of the compounds are not available from the authors.
References
- Zhao, C.; Zhu, T.; Zhu, W. New marine natural products of microbial origin from 2010 to 2013. Chin. J. Org. Chem. 2013, 33, 1195–1234. [Google Scholar] [CrossRef]
- Butler, A.; Sandy, M. Mechanistic considerations of halogenating enzymes. Nature 2009, 460, 848–854. [Google Scholar] [CrossRef] [PubMed]
- Ballschmiter, K. Review: Pattern and sources of naturally produced organohalogens in the marine environment: Biogenic formation of organohalogens. Chemosphere 2003, 52, 313–324. [Google Scholar] [CrossRef]
- Gribble, G.-W. The diversity of naturally produced organohalogens. Chemosphere 2003, 52, 289–297. [Google Scholar] [CrossRef]
- Wang, C.-Y.; Hao, J.-D.; Ning, X.-Y.; Wu, J.-S.; Zhao, D.-L.; Kong, C.-J.; Shao, C.-L.; Wang, C.-Y. Penicilazaphilones D and E: Two new azaphilones from a sponge-derived strain of the fungus Penicillium sclerotiorum. RSC Adv. 2018, 8, 4348–4353. [Google Scholar] [CrossRef]
- Frank, M.; Hartmann, R.; Plenker, M.; Mándi, A.; Kurtán, T.; Özkaya, F.-C.; Müller, W.-E.; Kassack, M.-U.; Hamacher, A.; Lin, W.; et al. Brominated azaphilones from the sponge-associated fungus Penicillium canescens strain 4.14. 6a. J. Nat. Prod. 2019, 82, 2159–2166. [Google Scholar] [CrossRef]
- Kong, F.-D.; Ma, Q.-Y.; Huang, S.-Z.; Wang, P.; Wang, J.-F.; Zhou, L.-M.; Yuan, J.-Z.; Dai, H.-F.; Zhao, Y.-X. Chrodrimanins K–N and related meroterpenoids from the fungus Penicillium sp. SCS-KFD09 isolated from a marine worm, Sipunculus nudus. J. Nat. Prod. 2017, 80, 1039–1047. [Google Scholar] [CrossRef]
- Kong, F.-D.; Zhang, R.-S.; Ma, Q.-Y.; Xie, Q.-Y.; Wang, P.; Chen, P.-W.; Zhou, L.-M.; Dai, H.-F.; Luo, D.-Q.; Zhao, Y.-X. Chrodrimanins O–S from the fungus Penicillium sp. SCS-KFD09 isolated from a marine worm, Sipunculus nudus. Fitoterapia 2017, 122, 1–6. [Google Scholar] [CrossRef]
- Yang, G.; Yun, K.; Nenkep, V.-N.; Choi, H.-D.; Kang, J.-S.; Son, B.-W. Induced production of halogenated diphenyl ethers from the marine-derived fungus Penicillium chrysogenum. Chem. Biodivers. 2010, 7, 2766–2770. [Google Scholar] [CrossRef]
- Zhu, M.; Zhang, X.; Feng, H.; Dai, J.; Li, J.; Che, Q.; Gu, Q.; Zhu, T.; Li, D. Penicisulfuranols A–F, alkaloids from the mangrove endophytic fungus Penicillium janthinellum HDN13-309. J. Nat. Prod. 2017, 80, 71–75. [Google Scholar] [CrossRef]
- He, K.-Y.; Zhang, C.; Duan, Y.-R.; Huang, G.-L.; Yang, C.-Y.; Lu, X.-R.; Zheng, C.-J.; Chen, G.-Y. New chlorinated xanthone and anthraquinone produced by a mangrove-derived fungus Penicillium citrinum HL-5126. J. Antibiot. 2017, 70, 823–827. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Fang, Y.; Zhu, T.; Gu, Q.; Zhu, W. Gentisyl alcohol derivatives from the marine-derived fungus Penicillium Terrestre. J. Nat. Prod. 2008, 71, 66–70. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Chen, L.; Zhu, T.; Kurtán, T.; Mándi, A.; Zhao, Z.; Li, J.; Gu, Q. Chloctanspirones A and B, novel chlorinated polyketides with an unprecedented skeleton, from marine sediment derived fungus Penicillium Terrestre. Tetrahedron 2011, 67, 7913–7918. [Google Scholar] [CrossRef]
- Wu, G.; Lin, A.; Gu, Q.; Zhu, T.; Li, D. Four new chloro-eremophilane sesquiterpenes from an Antarctic deep-sea derived fungus, Penicillium sp. PR19N-1. Mar. Drugs 2013, 11, 1399–1408. [Google Scholar] [CrossRef]
- Cardoso-Martínez, F.; José, M.; Díaz-Marrero, A.-R.; Darias, J.; Cerella, C.; Diederich, M.; Cueto, M. Tanzawaic acids isolated from a marine-derived fungus of the genus Penicillium with cytotoxic activities. Org. Biomol. Chem. 2015, 13, 7248–7256. [Google Scholar] [CrossRef]
- Luo, M.; Cui, Z.; Huang, H.; Song, X.; Sun, A.; Dang, Y.; Lu, L.; Ju, J. Amino acid conjugated anthraquinones from the marine-derived fungus Penicillium sp. SCSIO sof101. J. Nat. Prod. 2017, 80, 1668–1673. [Google Scholar] [CrossRef]
- Chen, M.; Shen, N.-X.; Chen, Z.-Q.; Zhang, F.-M.; Chen, Y. Penicilones A–D, anti-MRSA azaphilones from the marine-derived fungus Penicillium janthinellum HK1-6. J. Nat. Prod. 2017, 80, 1081–1086. [Google Scholar] [CrossRef]
- Chen, M.; Zheng, Y.-Y.; Chen, Z.-Q.; Shen, N.-X.; Shen, L.; Zhang, F.-M.; Zhou, X.-J.; Wang, C.-Y. NaBr-induced production of brominated azaphilones and related tricyclic polyketides by the marine-derived fungus Penicillium janthinellum HK1-6. J. Nat. Prod. 2019, 82, 368–374. [Google Scholar] [CrossRef]
- Vansteelandt, M.; Blanchet, E.; Egorov, M.; Petit, F.; Toupet, L.; Bondon, A.; Monteau, F.; Le Bizec, B.; Thomas, O.-P.; Pouchus, Y.-F. Ligerin, an antiproliferative chlorinated sesquiterpenoid from a marine-derived Penicillium strain. J. Nat. Prod. 2013, 76, 297–301. [Google Scholar] [CrossRef]
- Blanchet, E.; Vansteelandt, M.; Le Bot, R.; Egorov, M.; Guitton, Y.; Pouchus, Y.-F.; Grovel, O. Synthesis and antiproliferative activity of ligerin and new fumagillin analogs against osteosarcoma. Eur. J. Med. Chem. 2014, 79, 244–250. [Google Scholar] [CrossRef]
- Bu, Y.-Y.; Yamazaki, H.; Ukai, K.; Namikoshi, M. Penicillimide, an open-chain hemisuccinimide from Okinawan marine-derived Penicillium copticola. J. Antibiot. 2015, 68, 537–539. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.-L.; Wang, M.; Zhao, H.-G.; Huang, Y.-H.; Lin, Y.-Y.; Tan, G.-H.; Chen, S.-L. Penicilazaphilone C, a new antineoplastic and antibacterial azaphilone from the marine fungus Penicillium sclerotiorum. Arch. Pharm. Res. 2016, 39, 1621–1627. [Google Scholar] [CrossRef] [PubMed]
- Abrell, L.-M.; Borgeson, B.; Crews, P. Chloro polyketides from the cultured fungus (Aspergillus) separated from a marine sponge. Tetrahedron Lett. 1996, 37, 2331–2334. [Google Scholar] [CrossRef]
- Namikoshi, M.; Negishi, R.; Nagai, H.; Dmitrenok, A.; Kobayashi, H. Three new chlorine containing antibiotics from a marine-derived fungus Aspergillus ostianus collected in Pohnpei. J. Antibiot. 2003, 56, 755–761. [Google Scholar] [CrossRef] [PubMed]
- Sureram, S.; Wiyakrutta, S.; Ngamrojanavanich, N.; Mahidol, C.; Ruchirawat, S.; Kittakoop, P. Depsidones, Aromatase inhibitors and radical scavenging agents from the marine-derived fungus Aspergillus unguis CRI282-03. Planta Med. 2012, 78, 582–588. [Google Scholar] [CrossRef] [PubMed]
- Kong, F.; Zhao, C.; Hao, J.; Wang, C.; Wang, W.; Huang, X.; Zhu, W. New α-glucosidase inhibitors from a marine sponge-derived fungus, Aspergillus sp. OUCMDZ-1583. RSC Adv. 2015, 5, 68852–68863. [Google Scholar] [CrossRef]
- Gu, B.-B.; Jiao, F.-R.; Wu, W.; Liu, L.; Jiao, W.-H.; Sun, F.; Wang, S.-P.; Yang, F.; Lin, H.-W. Ochrasperfloroid, an ochratoxin–ergosteroid heterodimer with inhibition of IL-6 and NO production from Aspergillus flocculosus 16D-1. RSC Adv. 2019, 9, 7251–7256. [Google Scholar] [CrossRef]
- Tsukamoto, S.; Kawabata, T.; Kato, H.; Greshock, T.-J.; Hirota, H.; Ohta, T.; Williams, R.-M. Isolation of antipodal (−)-versicolamide B and notoamides L–N from a marine-derived Aspergillus sp. Org. Lett. 2009, 11, 1297–1300. [Google Scholar] [CrossRef]
- Tsukamoto, S.; Umaoka, H.; Yoshikawa, K.; Ikeda, T.; Hirota, H. Notoamide O, a structurally unprecedented prenylated indole alkaloid, and notoamides P-R from a marine-derived fungus, Aspergillus sp. J. Nat. Prod. 2010, 73, 1438–1440. [Google Scholar] [CrossRef]
- Xu, X.; He, F.; Zhang, X.; Bao, J.; Qi, S. New mycotoxins from marine-derived fungus Aspergillus sp. SCSGAF0093. Food Chem. Toxicol. 2013, 53, 46–51. [Google Scholar] [CrossRef]
- Ivanets, E.-V.; Yurchenko, A.-N.; Smetanina, O.-F.; Rasin, A.-B.; Zhuravleva, O.-I.; Pivkin, M.-V.; Popov, R.-S.; Von Amsberg, G.; Afiyatullov, S.-S.; Dyshlovoy, S.-A. Asperindoles A–D and a p-terphenyl derivative from the ascidian-derived fungus Aspergillus sp. KMM 4676. Mar. Drugs 2018, 16, 232. [Google Scholar] [CrossRef] [PubMed]
- Teuscher, F.; Lin, W.; Wray, V.; Edrada, R.; Padmakumar, K.; Proksch, P.; Ebel, R. Two new cyclopentanoids from the endophytic fungus Aspergillus sydowii associated with the marine alga Acanthophora spicifera. Nat. Prod. Commun. 2006, 1, 927–933. [Google Scholar] [CrossRef]
- Nguyen, H.-P.; Zhang, D.; Lee, U.; Kang, J.-S.; Choi, H.-D.; Son, B.-W. Dehydroxychlorofusarielin B, an antibacterial polyoxygenated decalin derivative from the marine-derived fungus Aspergillus sp. J. Nat. Prod. 2007, 70, 1188–1190. [Google Scholar] [CrossRef] [PubMed]
- Yun, K.; Feng, Z.; Choi, H.-D.; Kang, J.-S.; Son, B.-W. New production of (R)-(–)-5-bromomellein, a dihydroisocoumarin derivative from the marine-derived fungus Aspergillus ochraceus. Chem. Nat. Compd. 2013, 49, 24–26. [Google Scholar] [CrossRef]
- Zhang, P.; Li, X.-M.; Li, X.; Wang, B.-G. New indole-diterpenoids from the algal-associated fungus Aspergillus nidulans. Phytochem. Lett. 2015, 12, 182–185. [Google Scholar] [CrossRef]
- Mandelare, P.-E.; Adpressa, D.-A.; Kaweesa, E.-N.; Zakharov, L.-N.; Loesgen, S. Coculture of two developmental stages of a marine-derived Aspergillus alliaceus results in the production of the cytotoxic bianthrone allianthrone A. J. Nat. Prod. 2018, 81, 1014–1022. [Google Scholar] [CrossRef]
- Huang, H.; Wang, F.; Luo, M.; Chen, Y.; Song, Y.; Zhang, W.; Zhang, S.; Ju, J. Halogenated anthraquinones from the marine-derived fungus Aspergillus sp. SCSIO F063. J. Nat. Prod. 2012, 75, 1346–1352. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Zhang, X.-Y.; Tu, Z.-C.; Xu, X.-Y.; Qi, S.-H. Alkaloids from the deep-sea-derived fungus Aspergillus westerdijkiae DFFSCS013. J. Nat. Prod. 2013, 76, 983–987. [Google Scholar] [CrossRef]
- Liu, S.; Lu, C.; Huang, J.; Shen, Y. Three new compounds from the marine fungal strain Aspergillus sp. AF119. Rec. Nat. Prod. 2012, 6, 334–338. [Google Scholar]
- Uchida, R.; Nakajyo, K.; Kobayashi, K.; Ohshiro, T.; Terahara, T.; Imada, C.; Tomoda, H. 7-Chlorofolipastatin, an inhibitor of sterol O-acyltransferase, produced by marine-derived Aspergillus ungui NKH-007. J. Antibiot. 2016, 69, 647–651. [Google Scholar] [CrossRef]
- Cheng, X.-C.; Varoglu, M.; Abrell, L.; Crews, P.; Lobkovsky, E.; Clardy, J. Chloriolins A-C, chlorinated sesquiterpenes produced by fungal cultures separated from a Jaspis marine sponge. J. Org. Chem. 1994, 59, 6344–6348. [Google Scholar] [CrossRef]
- Amagata, T.; Usami, Y.; Minoura, K.; Ito, T.; Numata, A. Cytotoxic substances produced by a fungal strain from a sponge: Physico-chemical properties and structures. J. Antibiot. 1998, 51, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Usami, Y.; Ikura, T.; Amagata, T.; Numata, A. First total syntheses and configurational assignments of cytotoxic trichodenones A–C. Tetrahedron: Asymmetry 2000, 11, 3711–3725. [Google Scholar] [CrossRef]
- Numata, A.; Amagata, T.; Minoura, K.; Ito, T. Gymnastatins, novel cytotoxic metabolites produced by a fungal strain from a sponge. Tetrahedron Lett. 1997, 38, 5675–5678. [Google Scholar] [CrossRef]
- Amagata, T.; Doi, M.; Ohta, T.; Minoura, K.; Ito, T.; Numata, A. Absolute stereostructures of novel cytotoxic metabolites, gymnastatins A-E, from a Gymnascella species separated from a Halichondria sponge. J. Chem. Soc. Perkin Trans. 1 1998, 21, 3585–3600. [Google Scholar] [CrossRef]
- Amagata, T.; Minoura, K.; Numata, A. Gymnastatins F-H, cytostatic metabolites from the sponge-derived fungus Gymnascella dankaliensis. J. Nat. Prod. 2006, 69, 1384–1388. [Google Scholar] [CrossRef]
- Amagata, T.; Takigawa, K.; Minoura, K. Gymnastatins I-K, Cancer cell growth inhibitors from a sponge-derived Gymnascella dankaliensis. Heterocycles 2010, 81, 897–907. [Google Scholar] [CrossRef]
- Amagata, T.; Tanaka, M.; Yamada, T.; Minoura, K.; Numata, A. Gymnastatins and dankastatins, growth inhibitory metabolites of a Gymnascella species from a Halichondria Sponge. J. Nat. Prod. 2008, 71, 340–345. [Google Scholar] [CrossRef]
- Amagata, T.; Tanaka, M.; Yamada, T.; Chen, Y.-P.; Minoura, K.; Numata, A. Additional cytotoxic substances isolated from the sponge-derived Gymnascella dankaliensis. Tetrahedron Lett. 2013, 54, 5960–5962. [Google Scholar] [CrossRef]
- Ogamino, T.; Ohnishi, S.; Ishikawa, Y.; Sugai, T.; Obata, R.; Nishiyama, S. Synthesis and biological assessment of hemiacetal spiro derivatives towards development of efficient chemotherapeutic agent. Sci. Technol. Adv. Mater. 2006, 7, 175. [Google Scholar] [CrossRef]
- Murayama, K.; Tanabe, T.; Ishikawa, Y.; Nakamura, K.; Nishiyama, S. A synthetic study on gymnastatins F and Q: The tandem Michael and aldol reaction approach. Tetrahedron Lett. 2009, 50, 3191–3194. [Google Scholar] [CrossRef]
- Zhang, D.; Yang, X.; Kang, J.-S.; Choi, H.-D.; Son, B.-W. Chlorohydroaspyrones A and B, antibacterial aspyrone derivatives from the marine-derived fungus Exophiala sp. J. Nat. Prod. 2008, 71, 1458–1460. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Bao, B.; Dang, H.-T.; Hong, J.; Lee, H.-J.; Yoo, E.-S.; Bae, K.-S.; Jung, J.-H. Anti-inflammatory sesquiterpenoids from a sponge-derived fungus Acremonium sp. J. Nat. Prod. 2009, 72, 270–275. [Google Scholar] [CrossRef] [PubMed]
- Fraser, J.-A.; Lambert, L.-K.; Pierens, G.-K.; Bernhardt, P.-V.; Garson, M.-J. Secondary metabolites of the sponge-derived fungus Acremonium persicinum. J. Nat. Prod. 2013, 76, 1432–1440. [Google Scholar]
- Jansen, N.; Ohlendorf, B.; Erhard, A.; Bruhn, T.; Bringmann, G.; Imhoff, J.-F. Helicusin E, isochromophilone X and isochromophilone XI: New chloroazaphilones produced by the fungus Bartalinia robillardoides Strain LF550. Mar. Drugs 2013, 11, 800–816. [Google Scholar] [CrossRef]
- Ngokpol, S.; Suwakulsiri, W.; Sureram, S.; Lirdprapamongkol, K.; Aree, T.; Wiyakrutta, S.; Mahidol, C.; Ruchirawat, S.; Kittakoop, P. Drimane sesquiterpene-conjugated amino acids from a marine isolate of the fungus Talaromyces minioluteus (Penicillium minioluteum). Mar. Drugs 2015, 13, 3567–3580. [Google Scholar] [CrossRef]
- Qin, C.; Lin, X.; Lu, X.; Wan, J.; Zhou, X.; Liao, S.; Tu, Z.; Xu, S.; Liu, Y. Sesquiterpenoids and xanthones derivatives produced by sponge-derived fungus Stachybotry sp. HH1 ZSDS1F1-2. J. Antibiot. 2015, 68, 121–125. [Google Scholar] [CrossRef]
- Elsebai, M.-F.; Ghabbour, H.-A. Isocoumarin derivatives from the marine-derived fungus Phoma sp. 135. Tetrahedron Lett. 2016, 57, 354–356. [Google Scholar] [CrossRef]
- McDonald, L.-A.; Abbanat, D.-R.; Barbieri, L.-R.; Bernan, V.-S.; Discafani, C.-M.; Greenstein, M.; Janota, K.; Korshalla, J.-D.; Lassota, P.; Tischler, M.; et al. Spiroxins, DNA cleaving antitumor antibiotics from a marine-derived fungus. Tetrahedron Lett. 1999, 40, 2489–2492. [Google Scholar] [CrossRef]
- Ando, Y.; Tanaka, D.; Sasaki, R.; Ohmori, K.; Suzuki, K. Stereochemical dichotomy in two competing cascade processes: Total syntheses of both enantiomers of spiroxin A. Angew. Chem. 2019, 131, 12637–12643. [Google Scholar] [CrossRef]
- Shao, C.-L.; Wu, H.-X.; Wang, C.-Y.; Liu, Q.-A.; Xu, Y.; Wei, M.-Y.; Qian, P.-Y.; Gu, Y.-C.; Zheng, C.-J.; She, Z.-G.; et al. Potent antifouling resorcylic acid lactones from the gorgonian-derived fungus Cochliobolus lunatus. J. Nat. Prod. 2011, 74, 629–633. [Google Scholar] [CrossRef] [PubMed]
- Mahankali, B.; Srihari, P.-A. Carbohydrate approach for the first total synthesis of cochliomycin C: Stereoselective total synthesis of paecilomycin E, paecilomycin F and 6′-epi-Cochliomycin, C. Eur. J. Org. Chem. 2015, 18, 3983–3993. [Google Scholar] [CrossRef]
- Li, H.-J.; Chen, T.; Xie, Y.-L.; Chen, W.-D.; Zhu, X.-F.; Lan, W.-J. Isolation and structural elucidation of chondrosterins F-H from the marine fungus Chondrostereum sp. Mar. Drugs 2013, 11, 551–558. [Google Scholar] [CrossRef] [PubMed]
- Wei, M.-Y.; Li, D.; Shao, C.-L.; Deng, D.-S.; Wang, C.-Y. (±)-Pestalachloride D, an antibacterial racemate of chlorinated benzophenone derivative from a soft coral-derived fungus Pestalotiopsis sp. Mar. Drugs 2013, 11, 1050–1060. [Google Scholar] [CrossRef]
- Arredondo, V.; Roa, D.-E.; Yan, S.; Liu-Smith, F.; Van Vranken, D.-L. Total synthesis of (±)-pestalachloride C and (±)-pestalachloride D through a biomimetic knoevenagel/hetero-diels–alder cascade. Org. Lett. 2019, 21, 1755–1759. [Google Scholar] [CrossRef]
- Nielsen, J.; Nielsen, P.-H.; Frisvad, J.-C. Fungal depside, guisinol, from a marine derived strain of Emericella unguis. Phytochemistry 1999, 50, 263–265. [Google Scholar] [CrossRef]
- Afiyatullov, S.-S.; Kalinovsky, A.-I.; Antonov, A.-S. New virescenosides from the marine-derived fungus Acremonium striatisporum. Nat. Prod. Commun. 2011, 6, 1063–1068. [Google Scholar] [CrossRef]
- Yamada, T.; Iritani, M.; Ohishi, H.; Tanaka, K.; Minoura, K.; Doi, M.; Numata, A. Pericosines, antitumour metabolites from the sea hare-derived fungus Periconia byssoides. Structures and biological activities. Org. Biomol. Chem. 2007, 5, 3979–3986. [Google Scholar] [CrossRef]
- Boyd, D.-R.; Sharma, N.-D.; Acaru, C.-A.; Malone, J.-F.; O’Dowd, C.-R.; Allen, C.-C.; Stevenson, P.-J. Chemoenzymatic synthesis of carbasugars (+)-pericosines A–C from diverse aromatic cis-dihydrodiol precursors. Org. Lett. 2010, 12, 2206–2209. [Google Scholar] [CrossRef]
- Mizuki, K.; Iwahashi, K.; Murata, N.; Ikeda, M.; Nakai, Y.; Yoneyama, H.; Harusawa, S.; Usami, Y. Synthesis of marine natural product (−)-pericosine E. Org. Lett. 2014, 16, 3760–3763. [Google Scholar] [CrossRef]
- Yamada, T.; Doi, M.; Shigeta, H.; Muroga, Y.; Hosoe, S.; Numata, A.; Tanaka, R. Absolute stereostructures of cytotoxic metabolites, chaetomugilins A-C, produced by a Chaetomium species separated from a marine fish. Tetrahedron Lett. 2008, 49, 4192–4195. [Google Scholar] [CrossRef]
- Yasuhide, M.; Yamada, T.; Numata, A.; Tanaka, R. Chaetomugilins, new selectively cytotoxic metabolites, produced by a marine fish-derived Chaetomium species. J. Antibiot. 2008, 61, 615–622. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Yasuhide, M.; Shigeta, H.; Numata, A.; Tanaka, R. Absolute stereostructures of chaetomugilins G and H produced by a marine-fish-derived Chaetomium species. J. Antibiot. 2009, 62, 353–357. [Google Scholar] [CrossRef] [PubMed]
- Muroga, Y.; Yamada, T.; Numata, A.; Tanaka, R. Chaetomugilins I-O, new potent cytotoxic metabolites from a marine-fish-derived Chaetomium species. Stereochemistry and biological activities. Tetrahedron 2009, 65, 7580–7586. [Google Scholar] [CrossRef]
- Yamada, T.; Muroga, Y.; Tanaka, R. New azaphilones, seco-chaetomugilins A and D, produced by a marine-fish-derived Chaetomium globosum. Mar. Drugs 2009, 7, 249–257. [Google Scholar] [CrossRef]
- Muroga, Y.; Yamada, T.; Numata, A.; Tanaka, R. 11- and 4′-Epimers of chaetomugilin A, novel cytostatic metabolites from marine fish-derived fungus Chaetomium globosum. Helv. Chim. Acta 2010, 93, 542–549. [Google Scholar] [CrossRef]
- Yamada, T.; Muroga, Y.; Jinno, M.; Kajimoto, T.; Usami, Y.; Numata, A.; Tanaka, R. New class azaphilone produced by a marine fish-derived Chaetomium globosum. The stereochemistry and biological activities. Bioorg. Med. Chem. 2011, 19, 4106–4113. [Google Scholar] [CrossRef]
- Yamada, T.; Jinno, M.; Kikuchi, T.; Kajimoto, T.; Numata, A.; Tanaka, R. Three new azaphilones produced by a marine fish-derived chaetomium globosum. J. Antibiot. 2012, 65, 413–417. [Google Scholar] [CrossRef]
- Watts, K.-R.; Loveridge, S.-T.; Tenney, K.; Media, J.; Valeriote, F.-A.; Crews, P. Utilizing DART Mass Spectrometry to pinpoint halogenated metabolites from a marine invertebrate-derived fungus. J. Org. Chem. 2011, 76, 6201–6208. [Google Scholar] [CrossRef]
- Zhang, W.; Shao, C.-L.; Chen, M.; Liu, Q.-A.; Wang, C.-Y. Brominated resorcylic acid lactones from the marine-derived fungus Cochliobolus lunatus induced by histone deacetylase inhibitors. Tetrahedron Lett. 2014, 55, 4888–4891. [Google Scholar] [CrossRef]
- Garo, E.; Starks, C.-M.; Jensen, P.-R.; Fenical, W.; Lobkovsky, E.; Clardy, J. Trichodermamides A and B, cytotoxic modified dipeptides from the marine-derived fungus Trichoderma virens. J. Nat. Prod. 2003, 66, 423–426. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.-D.; Zakarian, A. Total synthesis of (±)-Trichodermamide B and of a putative biosynthetic precursor to Aspergillazine A using an oxaza-cope rearrangement. Angew. Chem. Int. Ed. 2008, 47, 6829–6831. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, E.-L.; Williams, D.-E.; Ioca, L.-P.; Morais-Urano, R.-P.; Santos, M.-F.; Patrick, B.-O.; Elias, L.-M.; Lira, S.-P.; Ferreira, A.-G.; Passarini, M.-R.; et al. Structure and biogenesis of roussoellatide, a dichlorinated polyketide from the marine-derived fungus Roussoella sp. DLM33. Org. Lett. 2015, 17, 5152–5155. [Google Scholar] [CrossRef] [PubMed]
- Yan, D.-F.; Lan, W.-J.; Wang, K.-T.; Huang, L.; Jiang, C.-W.; Li, H.-J. Two chlorinated benzofuran derivatives from the marine fungus Pseudallescheria boydii. Nat. Prod. Commun. 2015, 10, 621–622. [Google Scholar] [CrossRef] [PubMed]
- Cueto, M.; Jensen, P.-R.; Kauffman, C.; Fenical, W.; Lobkovsky, E.; Clardy, J. Pestalone, a new antibiotic produced by a marine fungus in response to bacterial challenge. J. Nat. Prod. 2001, 64, 1444–1446. [Google Scholar] [CrossRef]
- Slavov, N.; Cvengroš, J.; Neudörfl, J.-M.; Schmalz, H.-G. Total synthesis of the marine antibiotic pestalone and its surprisingly facile conversion into pestalalactone and pestalachloride A. Angew. Chem. Int. Ed. 2010, 49, 7588–7591. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Kim, S.-K.; Kang, J.-S.; Choi, H.-D.; Son, B.-W. Polyketide and sesquiterpenediol metabolites from a marine-derived fungus. Bull. Korean Chem. Soc. 2004, 25, 607–608. [Google Scholar] [CrossRef]
- Lira, S.-P.; Vita-Marques, A.-M.; Seleghim, M.-H.-R.; Bugni, T.-S.; LaBarbera, D.-V.; Sette, L.-D.; Sponchiado, S.-R.; Ireland, C.-M.; Berlinck, R.-G. New destruxins from the marine-derived fungus Beauveria felina. J. Antibiot. 2006, 59, 553–563. [Google Scholar] [CrossRef]
- Li, X.; Zhang, D.; Lee, U.; Li, X.; Cheng, J.; Zhu, W.; Jung, J.-H.; Choi, H.-D.; Son, B.-W. Bromomyrothenone B and botrytinone, cyclopentenone derivatives from a marine isolate of the fungus Botrytis. J. Nat. Prod. 2007, 70, 307–309. [Google Scholar] [CrossRef]
- Pontius, A.; Mohamed, I.; Krick, A.; Kehraus, S.; König, G.-M. Aromatic polyketides from marine algicolous fungi. J. Nat. Prod. 2008, 71, 272–274. [Google Scholar] [CrossRef]
- Pontius, A.; Krick, A.; Kehraus, S.; Brun, R.; König, G.-M. Antiprotozoal activities of heterocyclic- substituted xanthones from the marine-derived fungus Chaetomium sp. J. Nat. Prod. 2008, 71, 1579–1584. [Google Scholar] [CrossRef] [PubMed]
- Greve, H.; Schupp, P.-J.; Eguereva, E.; Kehraus, S.; König, G.-M. Ten-membered lactones from the marine-derived fungus Curvularia sp. J. Nat. Prod. 2008, 71, 1651–1653. [Google Scholar] [CrossRef] [PubMed]
- Nenkep, V.; Yun, K.; Zhang, D.; Choi, H.-D.; Kang, J.-S.; Son, B.-W. Induced production of bromomethylchlamydosporols A and B from the marine-derived fungus Fusarium tricinctum. J. Nat. Prod. 2010, 73, 2061–2063. [Google Scholar] [CrossRef] [PubMed]
- Nenkep, V.-N.; Yun, K.; Li, Y.; Choi, H.-D.; Kang, J.-S.; Son, B.-W. New production of haloquinones, bromochlorogentisylquinones A and B, by a halide salt from a marine isolate of the fungus Phoma herbarum. J. Antibiot. 2010, 63, 199–201. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, H.; Takahashi, O.; Murakami, K.; Namikoshi, M. Induced production of a new unprecedented epitrithiodiketopiperazine, chlorotrithiobrevamide, by a culture of the marine-derived Trichoderma cf. brevicompactum with dimethyl sulfoxide. Tetrahedron Lett. 2015, 56, 6262–6265. [Google Scholar] [CrossRef]
- Song, Y.-P.; Miao, F.-P.; Fang, S.-T.; Yin, X.-L.; Ji, N.-Y. Halogenated and nonhalogenated metabolites from the marine-alga-endophytic fungus Trichoderma asperellum cf44-2. Mar. Drugs 2018, 16, 266. [Google Scholar] [CrossRef]
- Wen, L.; Guo, Z.-Y.; Li, Q.; Zhang, D.; She, Z.; Vrijmoed, L.-L. A new griseofulvin derivative from the mangrove endophytic fungus Sporothrix sp. Chem. Nat. Compd. 2010, 46, 363–365. [Google Scholar] [CrossRef]
- Zhang, G.; Sun, S.; Zhu, T.; Lin, Z.; Gu, J.; Li, D.; Gu, Q. Antiviral isoindolone derivatives from an endophytic fungus Emericella sp. associated with Aegiceras corniculatum. Phytochemistry 2011, 72, 1436–1442. [Google Scholar] [CrossRef]
- Klaiklay, S.; Rukachaisirikul, V.; Tadpetch, K.; Sukpondma, Y.; Phongpaichit, S.; Buatong, J.; Sakayaroj, J. Chlorinated chromone and diphenyl ether derivatives from the mangrove-derived fungus Pestalotiopsis sp. PSU-MA69. Tetrahedron 2012, 68, 2299–2305. [Google Scholar] [CrossRef]
- Hammerschmidt, L.; Debbab, A.; Ngoc, T.-D.; Wray, V.; Hemphil, C.-P.; Lin, W.; Broetz-Oesterhelt, H.; Kassack, M.-U.; Proksch, P.; Aly, A.-H. Polyketides from the mangrove-derived endophytic fungus Acremonium strictum. Tetrahedron Lett. 2014, 55, 3463–3468. [Google Scholar] [CrossRef]
- Isaka, M.; Chinthanom, P.; Rachtawee, P.; Srichomthong, K.; Srikitikulchai, P.; Kongsaeree, P.; Prabpai, S. Cytotoxic hydroanthraquinones from the mangrove-derived fungus Paradictyoarthrinium diffractum BCC 8704. J. Antibiot. 2015, 68, 334–338. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Chen, D.; Cai, R.; Cui, H.; Long, Y.; Lu, Y.; Li, C.; She, Z. Cytotoxic and antibacterial preussomerins from the mangrove endophytic fungus Lasiodiplodia theobromae ZJ-HQ1. J. Nat. Prod. 2016, 79, 2397–2402. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.-S.; Li, X.-M.; Williams, K.; Proksch, P.; Ji, N.-Y.; Wang, B.-G. Rhizovarins A–F, indole-diterpenes from the mangrove-derived endophytic fungus Mucor irregularis QEN-189. J. Nat. Prod. 2016, 79, 2066–2074. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Zhao, Y.; Heering, C.; Janiak, C.; Müller, W.-E.; Akoné, S.-H.; Liu, Z.; Proksch, P. Sesquiterpenoids from the endophytic fungus Rhinocladiella similis. J. Nat. Prod. 2019, 82, 1055–1062. [Google Scholar] [CrossRef]
- Rukachaisirikul, V.; Kannai, S.; Klaiklay, S.; Phongpaichit, S.; Sakayaroj, J. Rare 2-phenylpyran-4-ones from the seagrass-derived fungi Polyporales PSU-ES44 and PSU-ES83. Tetrahedron 2013, 69, 6981–6986. [Google Scholar] [CrossRef]
- Uchida, R.; Tomoda, H.; Arai, M.; Omura, S. Chlorogentisylquinone, a new neutral sphingo-myelinase inhibitor, produced by a marine fungus. J. Antibiot. 2001, 54, 882–889. [Google Scholar] [CrossRef]
- Niu, S.; Liu, D.; Hu, X.; Proksch, P.; Shao, Z.; Lin, W. Spiromastixones A-O, antibacterial chlorodepsidones from a deep-sea-derived Spiromastix sp. fungus. J. Nat. Prod. 2014, 77, 1021–1030. [Google Scholar] [CrossRef]
- Fredimoses, M.; Zhou, X.; Lin, X.; Tian, X.; Ai, W.; Wang, J.; Liao, S.; Liu, J.; Yang, B.; Yang, X.; et al. New prenylxanthones from the deep-sea derived fungus Emericella sp. SCSIO 05240. Mar. Drugs 2014, 2, 3190–3202. [Google Scholar] [CrossRef]
- Zhang, Z.; He, X.; Liu, C.; Che, Q.; Zhu, T.; Gu, Q.; Li, D. Clindanones A and B and cladosporols F and G, polyketides from the deep-sea derived fungus Cladosporium cladosporioides HDN14-342. RSC Adv. 2016, 6, 76498–79504. [Google Scholar] [CrossRef]
- Wang, W.; Park, C.; Oh, E.; Sung, Y.; Lee, J.; Park, K.-H.; Kang, H. Benzophenone Compounds, from a Marine-Derived Strain of the Fungus Pestalotiopsis neglecta, Inhibit Proliferation of Pancreatic Cancer Cells by Targeting the MEK/ERK Pathway. J. Nat. Prod. 2019, 82, 3357–3365. [Google Scholar] [CrossRef]
- Sun, C.; Ge, X.; Mudassir, S.; Zhou, L.; Yu, G.; Che, Q.; Zhang, G.; Peng, J.; Gu, Q.; Zhu, T.; et al. New glutamine-containing azaphilone alkaloids from deep-sea-derived fungus Chaetomium globosum HDN151398. Mar. Drugs 2019, 17, 253. [Google Scholar] [CrossRef] [PubMed]
- Renner, M.-K.; Jensen, P.-R.; Fenical, W. Neomangicols: Structures and absolute stereochemistries of unprecedented halogenated sesterterpenes from a marine fungus of the genus Fusarium. J. Org. Chem. 1998, 63, 8346–8354. [Google Scholar] [CrossRef]
- Wang, W.; Liao, Y.; Chen, R.; Hou, Y.; Ke, W.; Zhang, B.; Gao, M.; Shao, Z.; Chen, J.; Li, F. Chlorinated azaphilone pigments with antimicrobial and cytotoxic activities isolated from the deep sea derived fungus Chaetomium sp. NA-S01-R1. Mar. Drugs 2018, 16, 61. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).