Regioselective Monobromination of Phenols with KBr and ZnAl–BrO3−–Layered Double Hydroxides
Abstract
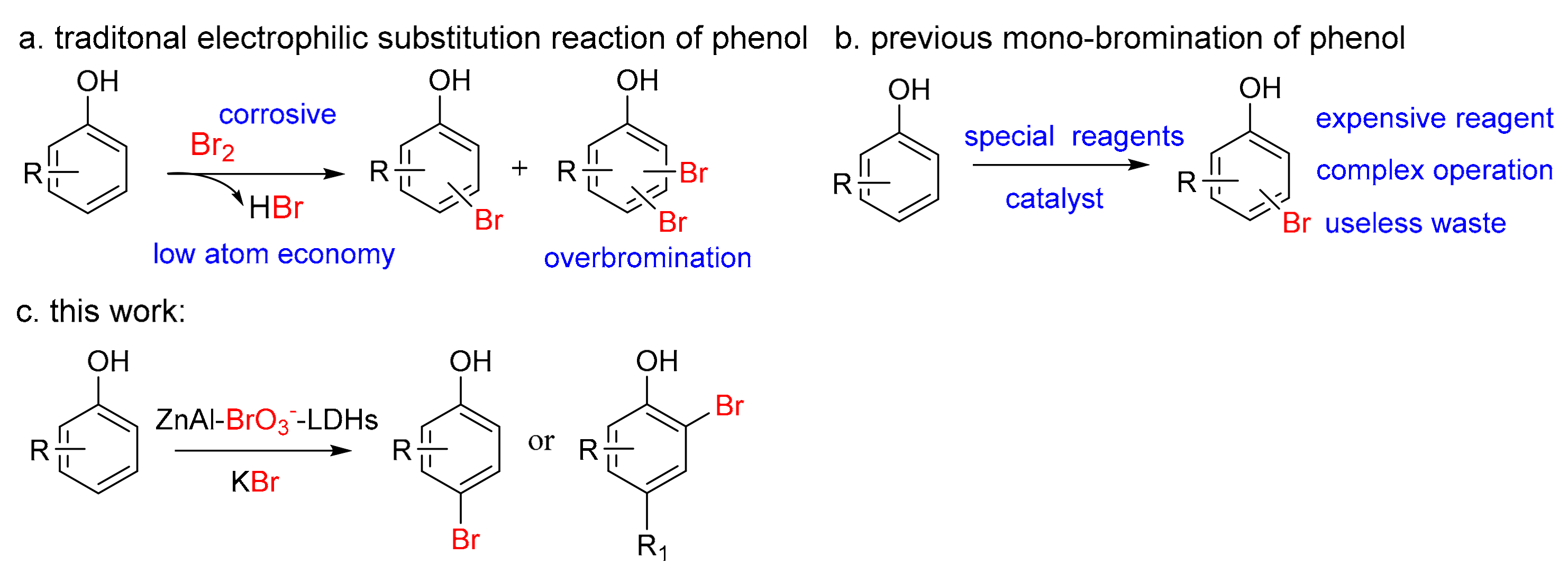
1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. General Information
4.2. General Procedure for the Bromination
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Du, F.; Zhou, Q.; Liu, D.; Fang, T.; Shi, Y.; Du, Y.; Chen, G. Dimerization of aromatic aompounds using Palladium-carbon-catalyzed Suzuki-Miyaura cross-coupling by one-pot synthesis. Synlett 2018, 29, 779–784. [Google Scholar] [CrossRef]
- Harada, K.; Arioka, C.; Miyakita, A.; Kubo, M.; Fukuyama, Y. Efficient synthesis of neurotrophic honokiol using Suzuki-Miyaura reactions. Tetrahedron Lett. 2014, 55, 6001–6003. [Google Scholar] [CrossRef]
- Miyaura, N.; Suzuki, A. Palladium-catalyzed cross-coupling reactions of organoboron compounds. Chem. Rev. 1995, 95, 2457–2483. [Google Scholar] [CrossRef]
- Schmidt, B.; Riemer, M. Suzuki-Miyaura coupling of halophenols and phenol boronic acids: Systematic investigation of positional isomer effects and conclusions for the synthesis of phytoalexins from pyrinae. J. Org. Chem. 2014, 79, 4104–4118. [Google Scholar] [CrossRef] [PubMed]
- Schmoger, C.; Szuppa, T.; Tied, A.; Schneider, F.; Stolle, A.; Ondruschka, B. Pd on porous glass: A versatile and easily recyclable catalyst for Suzuki and Heck reactions. Chemsuschem 2008, 1, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Lumb, J.-P. Phenol-directed C-H functionalization. Acs. Catal. 2019, 9, 521–555. [Google Scholar] [CrossRef]
- Jesus, A.; Correia-da-Silva, M.; Afonso, C.; Pinto, M.; Cidade, H. Isolation and potential biological applications of haloaryl secondary metabolites from macroalgae. Mar. Drugs 2019, 17. [Google Scholar] [CrossRef]
- Liu, M.; Hansen, P.E.; Lin, X. Bromophenols in marine algae and their bioactivities. Mar. Drugs 2011, 9, 1273–1292. [Google Scholar] [CrossRef]
- Oztaskin, N.; Cetinkaya, Y.; Taslimi, P.; Goksu, S.; Gulcin, I. Antioxidant and acetylcholinesterase inhibition properties of novel bromophenol derivatives. Bioorg. Chem. 2015, 60, 49–57. [Google Scholar] [CrossRef]
- Ghorpade, P.V.; Pethsangave, D.A.; Some, S.; Shankarling, G.S. Graphene oxide promoted oxidative bromination of anilines and phenols in water. J. Org. Chem. 2018, 83, 7388–7397. [Google Scholar] [CrossRef]
- Jiang, P.-P.; Yang, X.-J. A quick, mild and efficient bromination using a CFBSA/KBr system. Rsc Adv. 2016, 6, 90031–90034. [Google Scholar] [CrossRef]
- Su, P.; Fan, C.; Yu, H.; Wang, W.; Jia, X.; Rao, Q.; Fu, C.; Zhang, D.; Huang, B.; Pan, C.; et al. Synthesis of Ti-Al binary oxides and their catalytic application for C-H halogenation of phenols, aldehydes and ketones. Mol. Catal. 2019, 475. [Google Scholar] [CrossRef]
- Sim, J.; Jo, H.; Viji, M.; Choi, M.; Jung, J.-A.; Lee, H.; Jung, J.-K. Rapid, Operationally simple, and metal-free NBS mediated one-pot synthesis of 1,2-Naphthoquinone from 2-naphthol. Adv. Synth. Catal. 2018, 360, 852–858. [Google Scholar] [CrossRef]
- Sengupta, G.; Pandey, P.; De, S.; Ramapanicker, R.; Bera, J.K. A bromo-capped diruthenium(I, I) N-heterocyclic carbene compound for in situ bromine generation with NBS: catalytic olefin aziridination reactions. Dalton T. 2018, 47, 11917–11924. [Google Scholar] [CrossRef] [PubMed]
- Anjaiah, B.; Prameela, K.; Srinivas, P.; Rajanna, K.C. Synthesis, kinetics, and mechanism of bromophenols by N-bromophthalimide in aqueous acetic acid. Int. J. Chem. Kinet. 2018, 50, 804–812. [Google Scholar] [CrossRef]
- Jereb, M.; Gosak, K. Acid-promoted direct electrophilic trifluoromethylthiolation of phenols. Org. Biomol. Chem. 2015, 13, 3103–3115. [Google Scholar] [CrossRef]
- Ishihara, K. Development of Highly selective organic transformation reactions using halogen lewis acids. J. Syn. Org. Chem. Jpn. 2014, 72, 137–148. [Google Scholar] [CrossRef]
- Andersh, B.; Murphy, D.L.; Olson, R.J. Hydrochloric acid catalysis of N-bromosuccinimide (NBS) mediated nuclear aromatic brominations in acetone. Synth. Commun. 2000, 30, 2091–2098. [Google Scholar] [CrossRef]
- Oberhauser, T. A new bromination method for phenols and anisoles: NBS/HBF4 center dot Et2O in CH3CN. J. Org. Chem. 1997, 62, 4504–4506. [Google Scholar] [CrossRef]
- Zysman-Colman, E.; Arias, K.; Siegel, J.S. Synthesis of arylbromides from arenes and N-bromosuccinimide (NBS) in acetonitrile - A convenient method for aromatic bromination. Can. J. Chem. 2009, 87, 440–447. [Google Scholar] [CrossRef]
- Khazaei, A.; Rostami, A.; Raiatzadeh, A. N-bromosuccinimide (NBS): a mild and efficient catalyst for tetrahydropyranylation of alcohols and Phenols under solvent-free conditions. J. Chin. Chem. Soc. 2007, 54, 1029–1032. [Google Scholar] [CrossRef]
- Semwal, R.; Ravi, C.; Kumar, R.; Meena, R.; Adimurthy, S. Sodium salts (NaI/NaBr/NaCl) for the halogenation of imidazo-fused heterocycles. J. Org. Chem. 2019, 84, 792–805. [Google Scholar] [CrossRef] [PubMed]
- Song, S.; Sun, X.; Li, X.; Yuan, Y.; Jiao, N. Efficient and practical oxidative bromination and iodination of arenes and heteroarenes with DMSO and hydrogen halide: a mild protocol for late-Stage functionalization. Org. Lett. 2015, 17, 2886–2889. [Google Scholar] [CrossRef] [PubMed]
- Karki, M.; Magolan, J. Bromination of olefins with HBr and DMSO. J. Org. Chem. 2015, 80, 3701–3707. [Google Scholar] [CrossRef] [PubMed]
- Barhate, N.B.; Gajare, A.S.; Wakharkar, R.D.; Bedekar, A.V. Simple and practical halogenation of arenes, alkenes and alkynes with hydrohalic acid/H2O2 (or TBHP). Tetrahedron 1999, 55, 11127–11142. [Google Scholar] [CrossRef]
- Bora, U.; Bose, G.; Chaudhuri, M.K.; Dhar, S.S.; Gopinath, R.; Khan, A.T.; Patel, B.K. Regioselective bromination of organic substrates by tetrabutylammonium bromide promoted by V2O5-H2O2: An environmentally favorable synthetic protocol. Org. Lett. 2000, 2, 247–249. [Google Scholar] [CrossRef] [PubMed]
- Pandit, P.; Gayen, K.S.; Khamarui, S.; Chatterjee, N.; Maiti, D.K. Addition of halide to π-bond directly from aqueous NaX solution: A general strategy for installation of two different functional groups. Chem. Commun. 2011, 47, 6933–6935. [Google Scholar] [CrossRef]
- Truchan, N.; Jandl, C.; Pothig, A.; Breitenlechner, S.; Bach, T. Access to biphenyls by Palladium-catalyzed oxidative coupling of phenyl carbamates and phenols. Synthesis 2019, 51, 3060–3076. [Google Scholar] [CrossRef]
- Khatun, R.; Biswas, S.; Ghosh, S.; Islam, S.M. Polymer-anchored Fe(III)Azo complex: An efficient reusable catalyst for oxidative bromination and multi-components reaction for the synthesis of spiropiperidine derivatives. J. Organomet. Chem. 2018, 858, 37–46. [Google Scholar] [CrossRef]
- Song, S.; Huang, X.; Liang, Y.-F.; Tang, C.; Li, X.; Jiao, N. From simple organobromides or olefins to highly value-added bromohydrins: a versatile performance of dimethyl sulfoxide. Green Chem. 2015, 17, 2727–2731. [Google Scholar] [CrossRef]
- Huang, Z.; Li, F.; Chen, B.; Lu, T.; Yuan, Y.; Yuan, G. A sustainable process for catalytic oxidative bromination with molecular oxygen. Chemsuschem 2013, 6, 1337–1340. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.A.; Rohitha, C.N.; Kulkarni, S.J.; Narender, N. Bromination of aromatic aompounds using ammonium bromide and oxone (R). Synthesis 2010, 1629–1632. [Google Scholar] [CrossRef]
- Adimurthy, S.; Ramachandraiah, G.; Bedekar, A.V.; Ghosh, S.; Ranu, B.C.; Ghosh, P.K. Eco-friendly and versatile brominating reagent prepared from a liquid bromine precursor. Green Chem. 2006, 8, 916–922. [Google Scholar] [CrossRef]
- Terent’ev, A.O.; Khodykin, S.V.; Krylov, I.B.; Ogibin, Y.N.; Nikishin, G.I. A convenient synthesis of 2,2-dibromo-1-arylethanones by bromination of 1-arylethanones with the H2O2-HBr system. Synthesis 2006, 1087–1092. [Google Scholar] [CrossRef]
- Hemati, R.; Shahvelayati, A.S.; Yadollahzadeh, K. o-Xylylene Bis(Triethyl Ammonium Tribromide) as a mild and ecyclable reagent for rapid and regioselective bromination of anilines and phenols. Lett. Org. Chem. 2018, 15, 682–687. [Google Scholar] [CrossRef]
- Bovonsombat, P.; Teecomegaet, P.; Kulvaranon, P.; Pandey, A.; Chobtumskul, K.; Tungsirisurp, S.; Sophanpanichkul, P.; Losuwanakul, S.; Soimaneewan, D.; Kanjanwongpaisan, P.; et al. Regioselective monobromination of aromatics via a halogen bond acceptor-donor interaction of catalytic thioamide and N -bromosuccinimide. Tetrahedron 2017, 73, 6564–6572. [Google Scholar] [CrossRef]
- Ma, X.; Yu, J.; Jiang, M.; Wang, M.; Tang, L.; Wei, M.; Zhou, Q. Mild and regioselective bromination of phenols with TMSBr. Eur. J. Org. Chem. 2019, 2019, 4593–4596. [Google Scholar] [CrossRef]
- Satkar, Y.; Ramadoss, V.; Nahide, P.D.; García-Medina, E.; Juárez-Ornelas, K.A.; Alonso-Castro, A.J.; Chávez-Rivera, R.; Jiménez-Halla, J.O.C.; Solorio-Alvarado, C.R. Practical, mild and efficient electrophilic bromination of phenols by a new I(iii)-based reagent: the PIDA–AlBr3 system. RSC Adv. 2018, 8, 17806–17812. [Google Scholar] [CrossRef]
- Georgiev, D.; Saes, B.W.H.; Johnston, H.J.; Boys, S.K.; Healy, A.; Hulme, A.N. Selective and efficient generation of ortho-brominated para-substituted phenols in ACS-grade methanol. Molecules 2016, 21, 8. [Google Scholar] [CrossRef] [PubMed]
- Bovonsombat, P.; Ali, R.; Khan, C.; Leykajarakul, J.; Pla-on, K.; Aphimanchindakul, S.; Pungcharoenpong, N.; Timsuea, N.; Arunrat, A.; Punpongjareorn, N. Facile p-toluenesulfonic acid-promoted para-selective monobromination and chlorination of phenol and analogues. Tetrahedron 2010, 66, 6928–6935. [Google Scholar] [CrossRef]
- Alinezhad, H.; Tavakkoli, S.M.; Salehian, F. Efficient, rapid, and regioselective bromination of phenols and anilines with N-bromosaccharin using tungstophosphoric acid as a heterogeneous recyclable catalyst. Synth. Commun. 2010, 40, 3226–3232. [Google Scholar] [CrossRef]
- Narender, N.; Naresh, M.; Arun Kumar, M.; Mahender Reddy, M.; Swamy, P.; Nanubolu, J. Fast and efficient bromination of aromatic compounds with ammonium bromide and oxone. Synthesis 2013, 45, 1497–1504. [Google Scholar] [CrossRef]
- Mokhtary, M.; Lakouraj, M.M. Polyvinylpolypyrrolidone–bromine complex: Mild and efficient polymeric reagent for bromination of activated aromatic compounds. Chinese Chem. Lett. 2011, 22, 13–17. [Google Scholar] [CrossRef]
- Borikar, S.P.; Daniel, T.; Paul, V. Mild, Efficient, and regioselective monobromination of arylamines and phenols using [BBIm]Br3 as a new reagent. Synth. Commun. 2010, 40, 647–653. [Google Scholar] [CrossRef]
- Hosseinzadeh, R.; Tajbakhsh, M.; Mohadjerani, M.; Lasemi, Z. Efficient and regioselective Bromination of aromatic compounds with ethylenebis(N-methylimidazolium) ditribromide (EBMIDTB). Synth. Commun. 2010, 40, 868–876. [Google Scholar] [CrossRef]
- Moghaddam, F.M.; Zargarani, D. Regioselective bromination of aromatic amines and phenols using N-benzyl-DABCO tribromide. Synth. Commun. 2009, 39, 4212–4220. [Google Scholar] [CrossRef]
- Borikar, S.P.; Daniel, T.; Paul, V. An efficient, rapid, and regioselective bromination of anilines and phenols with 1-butyl-3-methylpyridinium tribromide as a new reagent/solvent under mild conditions. Tetrahedron Lett. 2009, 50, 1007–1009. [Google Scholar] [CrossRef]
- Stropnik, T.; Bombek, S.; Kočevar, M.; Polanc, S. Regioselective bromination of activated aromatic substrates with a ZrBr4/diazene mixture. Tetrahedron Lett. 2008, 49, 1729–1733. [Google Scholar] [CrossRef]
- Suresh, P.; Annalakshmi, S.; Pitchumani, K. Regioselective monobromination of substituted phenols in the presence of β-cyclodextrin. Tetrahedron 2007, 63, 4959–4967. [Google Scholar] [CrossRef]
- Ghorbani-Vaghei, R.; Jalili, H. Mild and regioselective bromination of aromatic compounds with N,N,N′,N′-tetrabromobenzene-1,3-disulfonylamide and poly(N-bromobenzene-1,3-disulfonylamide). Synthesis 2005, 1099–1102. [Google Scholar] [CrossRef]
- Singh, P.P.; Thatikonda, T.; Kumar, K.A.A.; Sawant, S.D.; Singh, B.; Sharma, A.K.; Sharma, P.R.; Singh, D.; Vishwakarma, R.A. Cu-Mn Spinel oxide catalyzed regioselective halogenation of phenols and N-heteroarenes. J. Org. Chem. 2012, 77, 5823–5828. [Google Scholar] [CrossRef] [PubMed]
- Wischang, D.; Hartung, J. Bromination of phenols in bromoperoxidase-catalyzed oxidations. Tetrahedron 2012, 68, 9456–9463. [Google Scholar] [CrossRef]
- Chen, A.-J.; Wong, S.-T.; Hwang, C.-C.; Mou, C.-Y. Highly efficient and regioselective halogenation over well dispersed Rhenium-promoted mesoporous zirconia. ACS Catal. 2011, 1, 786–793. [Google Scholar] [CrossRef]
- Baharfar, R.; Alinezhad, H.; Azimi, S.; Salehian, F. Regioselective and high-yielding bromination of phenols and anilins using N-bromosaccharin and amberlyst-15. J. Chil. Chem. Soc. 2011, 56, 863–865. [Google Scholar] [CrossRef]
- Chen, A.-J.; Chen, X.-R.; Mou, C.-Y. Highly Regioselective oxybromination in an aqueous system using SBA-15 supported sulfated zirconia catalyst. J. Chin. Chem. Soc. 2010, 57, 820–828. [Google Scholar] [CrossRef]
- Wang, J.; Wang, W.; Li, J.-H. An efficient copper-catalysed aerobic oxybromination of arenes in water. Green Chem. 2010, 12, 2124–2126. [Google Scholar] [CrossRef]
- Bhatt, S.; Nayak, S.K. Copper(II) Bromide: A simple and selective monobromination reagent for electron-rich aromatic compounds. Synth. Commun. 2007, 37, 1381–1388. [Google Scholar] [CrossRef]
- Chhattise, P.K.; Ramaswamy, A.V.; Waghmode, S.B. Regioselective, photochemical bromination of aromatic compounds using N-bromosuccinimide. Tetrahedron Lett. 2008, 49, 189–194. [Google Scholar] [CrossRef]
- Das, B.; Venkateswarlu, K.; Majhi, A.; Siddaiah, V.; Reddy, K.R. A facile nuclear bromination of phenols and anilines using NBS in the presence of ammonium acetate as a catalyst. J. Mol. Catal. A- Chem. 2007, 267, 30–33. [Google Scholar] [CrossRef]
- Sels, B.F.; De Vos, D.E.; Jacobs, P.A. Bromide-assisted oxidation of substituted phenols with hydrogen peroxide to the corresponding p-quinol and p-quinol ethers over WO42--exchanged layered double hydroxides. Angew. Chem. Int. Edit. 2005, 44, 310–313. [Google Scholar] [CrossRef]
- Zhang, L.; Gong, Z.; Jiang, B.; Sun, Y.; Chen, Z.; Gao, X.; Yang, N. Ni-Al layered double hydroxides (LDHs) coated superhydrophobic mesh with flower-like hierarchical structure for oil/water separation. Appl. Surf. Sci. 2019, 490, 145–156. [Google Scholar] [CrossRef]
- Wang, B.; Shang, J.; Guo, C.; Zhang, J.; Zhu, F.; Han, A.; Liu, J. A general method to ultrathin bimetal-MOF nanosheets arrays via in situ transformation of layered double hydroxides arrays. Small 2019, 15. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.; Na, W.; Kim, J.; Lee, S.; Jang, J. Improved electrochemical performances of MOF-derived Ni-Co layered double hydroxide complexes using distinctive hollow-in-hollow structures. J. Mater. Chem. A 2019, 7, 17637–17647. [Google Scholar] [CrossRef]
- Guo, D.; Song, X.; Tan, L.; Ma, H.; Sun, W.; Pang, H.; Zhang, L.; Wang, X. A facile dissolved and reassembled strategy towards sandwich-like rGO@NiCoAl-LDHs with excellent supercapacitor performance. Chem. Eng. J. 2019, 356, 955–963. [Google Scholar] [CrossRef]
- Chen, W.; Zhang, Y.; Huang, R.; Zhou, Y.; Wu, Y.; Hu, Y.; Ostrikov, K. Ni-Co hydroxide nanosheets on plasma-reduced Co-based metal-organic nanocages for electrocatalytic water oxidation. J. Mater. Chem. A 2019, 7, 4950–4959. [Google Scholar] [CrossRef]
- Wang, L.; Zheng, F.; Jiang, C.; Ni, Z. New reagent system for bromination built with Zn/Al-BrO3-- -LDHs as carrier. J. Chin. Cera. Soc. 2015, 43, 672–677. [Google Scholar]
- Wang, L.; Yu, Q.; Feng, C.; Zhang, Y.; Hu, J. Efficient synthesis of dibromoalkanes and iodoacetates from olefins using ZnAl-XO3(-)-LDHs/LiX (X= Br, I) as halogen sources. Chin. J. Org. Chem. 2019, 39, 1787–1793. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, H.; Yu, Q.; Feng, C.; Hu, J. Green synthesis of haloformates from olefins using formic acid as reactant, protonic acid, and solvent. Synlett 2018, 29, 1611–1616. [Google Scholar] [CrossRef]
- Wang, L.; Chen, L.; Zhang, H.; Yu, Q. Selective oxidative bromination of anilines using potassium bromide and ZnAl-BrO3--LDHs. Chin. J. Org. Chem. 2017, 37, 3186–3190. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |


 | |||||
|---|---|---|---|---|---|
| Entry | Oxidant (equiv.) | Bromide (equiv.) | Solvent 2 | Temperature | Yield 3 |
| 1 | ZnAl-BrO3--LDHs (0.2) | LiBr (1.4) | AcOH | 25 °C | 71 |
| 2 | ZnAl-BrO3--LDHs (0.2) | LiBr (1.2) | AcOH | 25 °C | 73 |
| 3 | ZnAl-BrO3--LDHs (0.2) | LiBr (1.0) | AcOH | 25 °C | 76 |
| 4 | ZnAl-BrO3--LDHs (0.2) | LiBr (0.8) | AcOH | 25 °C | 68 |
| 5 | ZnAl-BrO3--LDHs (0.2) | NaBr (1.0) | AcOH | 25 °C | 80 |
| 6 | ZnAl-BrO3--LDHs (0.2) | KBr (1.0) | AcOH | 25 °C | 83 |
| 7 | ZnAl-BrO3--LDHs (0.2) | ZnBr2 (0.5) | AcOH | 25 °C | 73 |
| 8 | ZnAl-BrO3--LDHs (0.2) | KBr (1.0) | MeOH | 25 °C | - |
| 9 | ZnAl-BrO3--LDHs (0.2) | KBr (1.0) | EtOH | 25 °C | - |
| 10 | ZnAl-BrO3--LDHs (0.2) | KBr (1.0) | DCM | 25 °C | - |
| 11 | ZnAl-BrO3--LDHs (0.2) | KBr (1.0) | EA | 25 °C | - |
| 12 | ZnAl-BrO3--LDHs (0.2) | KBr (1.0) | AcOH/H2O | 25 °C | 86 |
| 13 | ZnAl-BrO3--LDHs (0.2) | KBr (1.0) | AcOH/H2O | 30 °C | 85 |
| 14 | ZnAl-BrO3--LDHs (0.2) | KBr (1.0) | AcOH/H2O | 35 °C | 91 |
| 15 | ZnAl-BrO3--LDHs (0.2) | KBr (1.0) | AcOH/H2O | 40 °C | 81 |
| 16 | ZnAl-BrO3--LDHs (0.2) | KBr (1.0) | AcOH/H2O | 45 °C | 77 |
| 17 | ZnAl-BrO3--LDHs (0.4) | KBr (2.0) | AcOH/H2O | 35°C | 76 (17) |
| 18 | KBrO3 (0.4) | KBr (2.0) | AcOH/H2O | 35 °C | 22 (61) |
| 19 | KBrO3 (0.2) | KBr (1.0) | AcOH/H2O | 35 °C | 35 (28) |
| 20 | - | KBr (1.0) | AcOH/H2O | 35 °C | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, L.; Feng, C.; Zhang, Y.; Hu, J. Regioselective Monobromination of Phenols with KBr and ZnAl–BrO3−–Layered Double Hydroxides. Molecules 2020, 25, 914. https://doi.org/10.3390/molecules25040914
Wang L, Feng C, Zhang Y, Hu J. Regioselective Monobromination of Phenols with KBr and ZnAl–BrO3−–Layered Double Hydroxides. Molecules. 2020; 25(4):914. https://doi.org/10.3390/molecules25040914
Chicago/Turabian StyleWang, Ligeng, Chun Feng, Yan Zhang, and Jun Hu. 2020. "Regioselective Monobromination of Phenols with KBr and ZnAl–BrO3−–Layered Double Hydroxides" Molecules 25, no. 4: 914. https://doi.org/10.3390/molecules25040914
APA StyleWang, L., Feng, C., Zhang, Y., & Hu, J. (2020). Regioselective Monobromination of Phenols with KBr and ZnAl–BrO3−–Layered Double Hydroxides. Molecules, 25(4), 914. https://doi.org/10.3390/molecules25040914






