Microwave-Assisted Green Synthesis and Antioxidant Activity of Selenium Nanoparticles Using Theobroma cacao L. Bean Shell Extract
Abstract
:1. Introduction
2. Results and Discussion
2.1. Quantitative Analysis of Cocoa Bean Shel Extract (CBSE)
2.2. Optimization of Synthesis Conditions of SeNPs
2.2.1. Effect of Synthesis Variables on SeNPs Crystalline Size
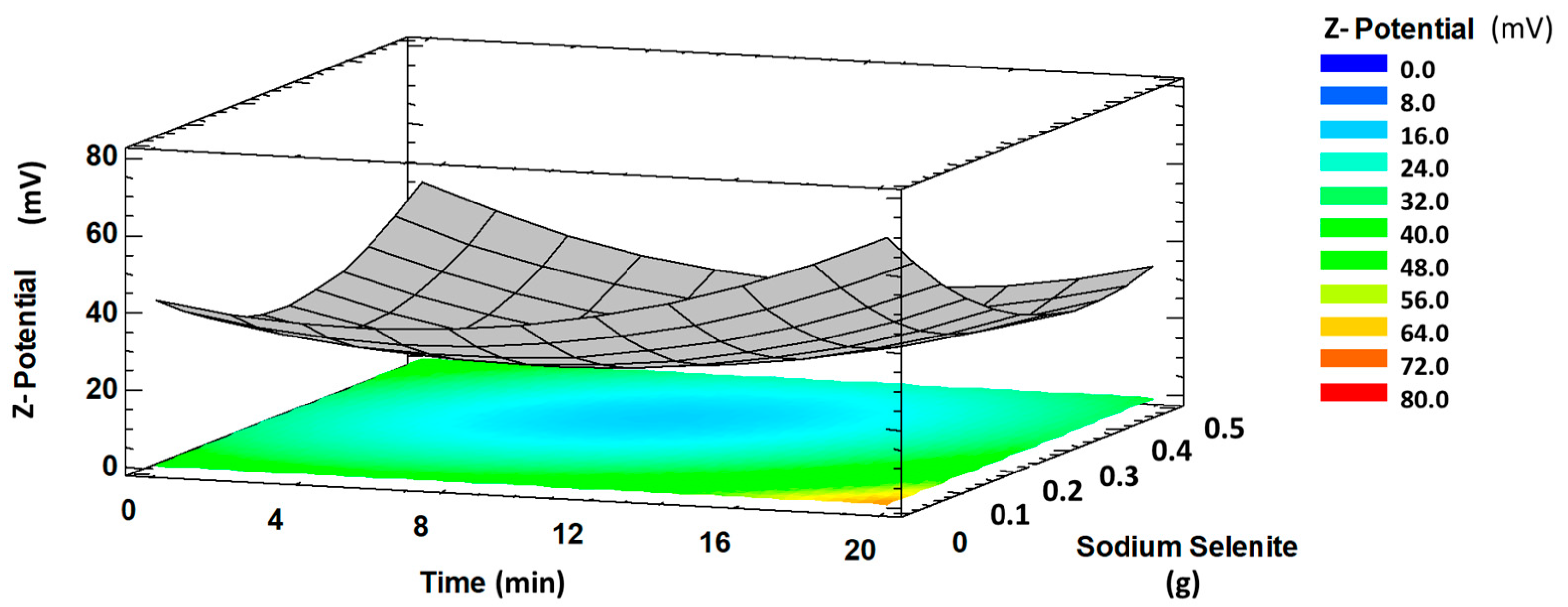
2.2.2. Effect of Synthesis Variables on Z-Potential Values
2.3. Characterization of SeNPs Obtained under Optimal Synthesis Conditions
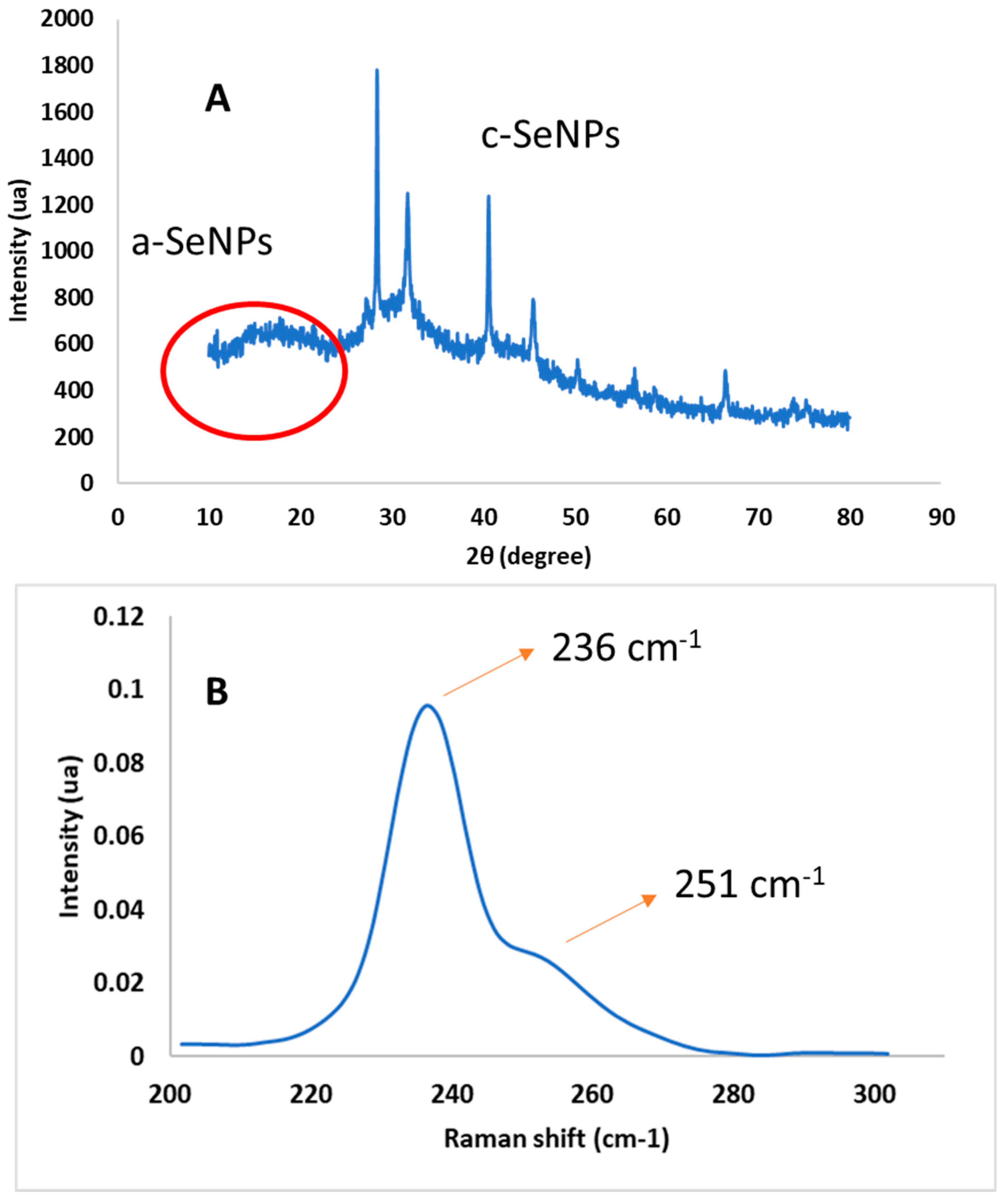
2.3.1. XRD Analysis
2.3.2. Raman Spectroscopy
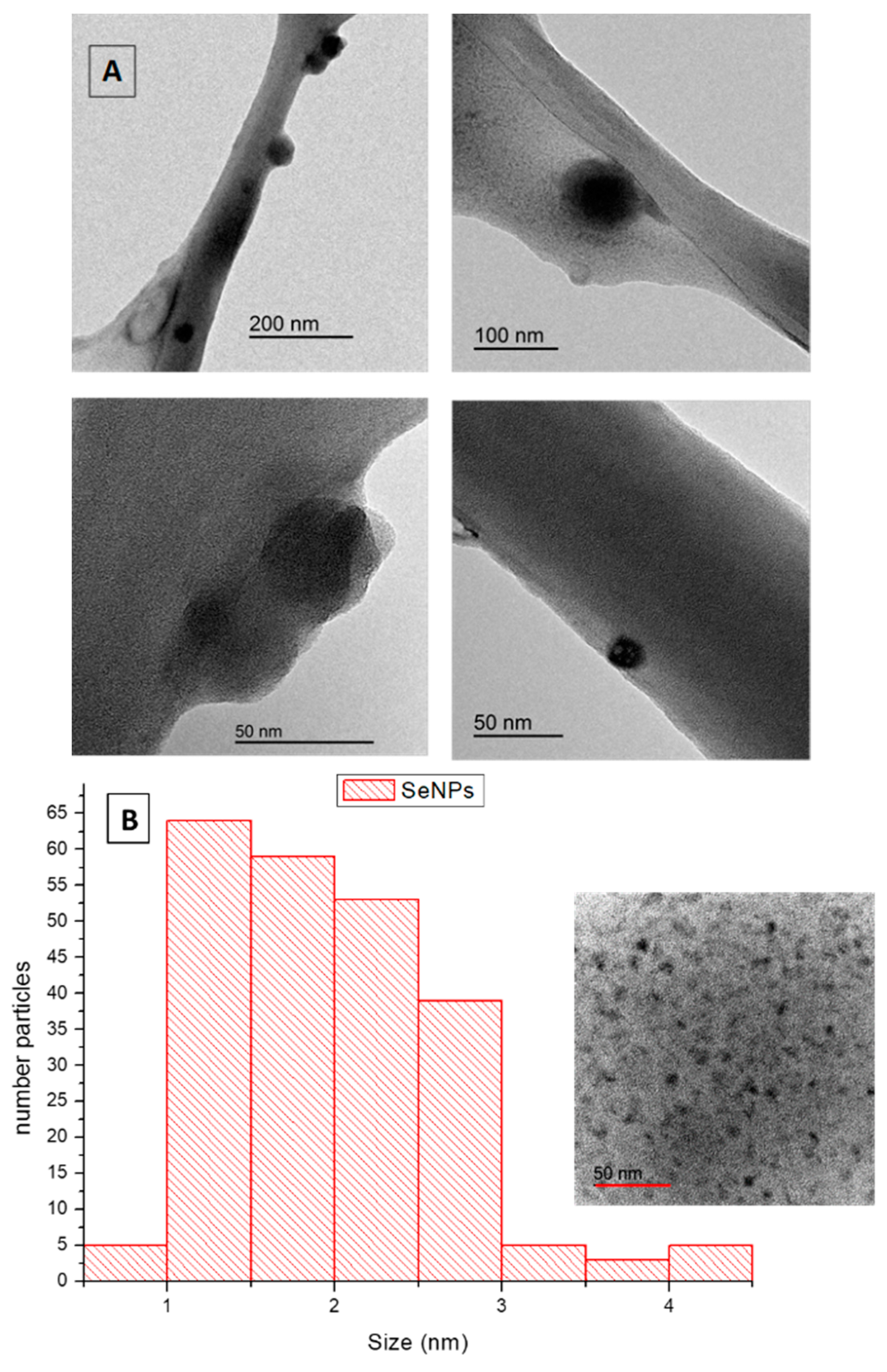
2.3.3. TEM
2.3.4. DLS
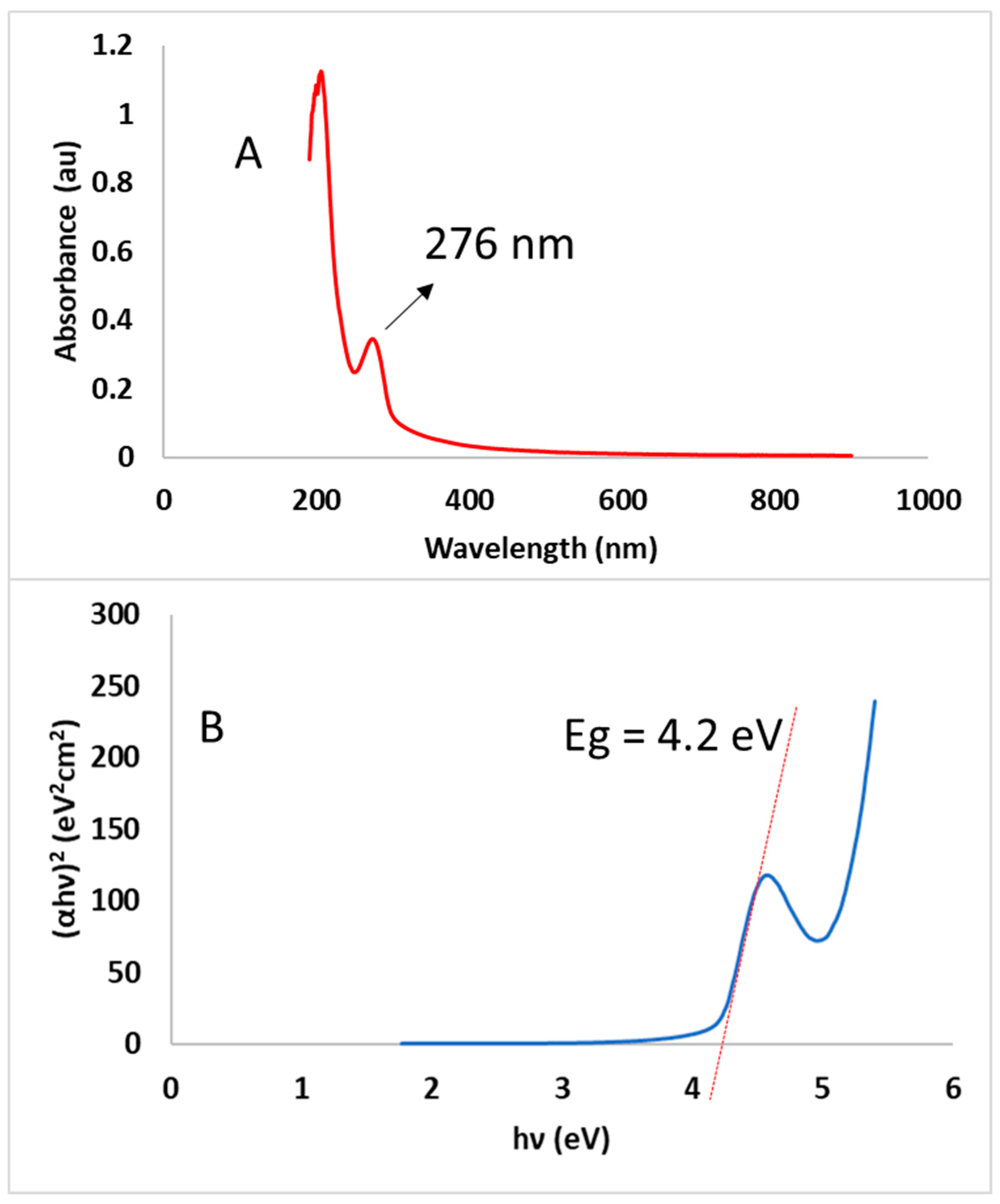
2.3.5. Optical Properties
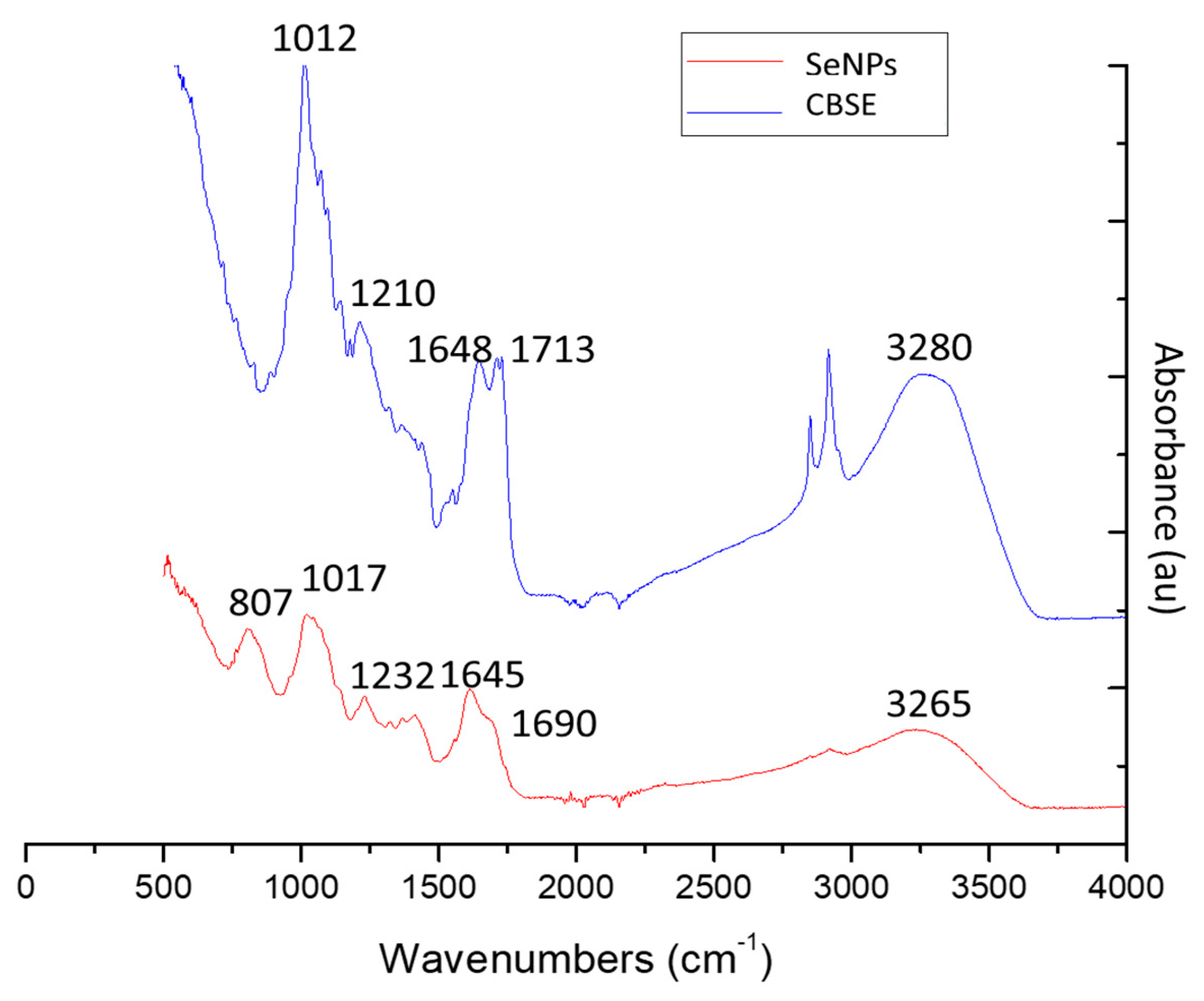
2.3.6. FTIR
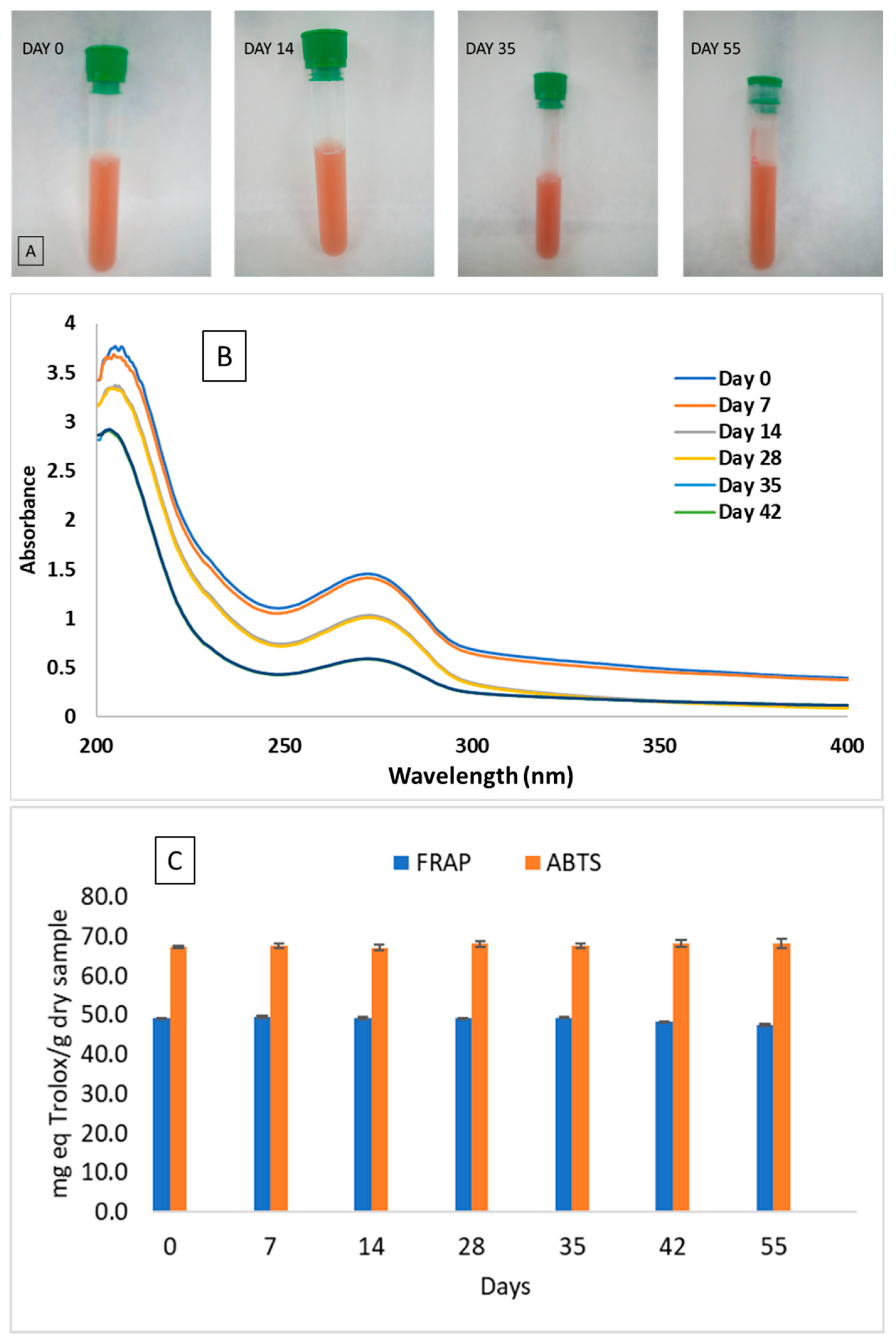
2.3.7. Stability of SeNPs and Antioxidant Activity
3. Materials and Methods
3.1. Materials and Reagents
3.2. CBS Extract Preparation
3.3. Quantitative Analysis of CBSE
3.4. Synthesis of SeNPs
3.5. Characterization of SeNPs
3.5.1. X-ray Diffraction (XRD)
3.5.2. Dynamic Light Scattering (DLS)
3.5.3. Optical Properties
3.5.4. FTIR Analysis
3.5.5. Raman Spectroscopy
3.5.6. Transmission Electron Microscopy (TEM)
3.5.7. Stability and Antioxidant Activity of SeNPs
3.6. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Li, J.; Shen, B.; Nie, S.; Duan, Z.; Chen, K. A combination of selenium and polysaccharides: Promising therapeutic potential. Carbohydr. Polym. 2019, 206, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Atkins, P.; Overton, T.; Rourke, J.; Weller, M.; Armstrong, F. Inorganic Chemistry, 5th ed.; Prentice Hall: Oxford, UK, 2010. [Google Scholar]
- Zhu, M.; Niu, G.; Tang, J. Elemental Se: Fundamentals and its optoelectronic applications. J. Mater. Chem. C 2019, 7, 2199–2206. [Google Scholar] [CrossRef]
- Shirsat, S.; Kadam, A.; Naushadc, M.; Mane, R.S. Selenium nanostructures: Microbial synthesis and applications. RSC Adv. 2018, 6. [Google Scholar]
- Basnet, P.; Chanu, T.I.; Samanta, D.; Chatterjee, S. A review on bio-synthesized zinc oxide nanoparticles using plant extracts as reductants and stabilizing agents. J. Photochem. Photobiol. B Biol. 2018, 183, 201–221. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, S.; Umar, A.; Mehta, S.K. Selenium nanomaterials: An overview of recent developments in synthesis, properties and potential applications. Prog. Mater. Sci. 2016, 83, 270–329. [Google Scholar] [CrossRef]
- Sirelkhatim, A.; Mahmud, S.; Seeni, A.; Kaus, N.H.M.; Ann, L.C.; Bakhori, S.K.M.; Hasan, H.; Mohamad, D. Review on zinc oxide nanoparticles: Antibacterial activity and toxicity mechanism. Nano-Micro Lett. 2015, 7, 219–242. [Google Scholar] [CrossRef] [PubMed]
- Alagesan, V.; Venugopal, S. Green synthesis of selenium nanoparticle using leaves extract of withania somnifera and its biological applications and photocatalytic activities. Bionanoscience 2019, 9, 105–116. [Google Scholar] [CrossRef]
- Adil, S.F.; Assal, M.E.; Khan, M.; Al-Warthan, A.; Siddiqui, M.R.H.; LiZ-Marzán, L.M. Biogenic synthesis of metallic nanoparticles and prospects toward green chemistry. Dalt. Trans. 2015, 44, 9709–9717. [Google Scholar] [CrossRef]
- Khan, M.; Shaik, M.R.; Adil, S.F.; Khan, S.T.; Al-Warthan, A.; Siddiqui, M.R.H.; Tahir, M.N.; Tremel, W. Plant extracts as green reductants for the synthesis of silver nanoparticles: Lessons from chemical synthesis. Dalt. Trans. 2018, 47, 11988–12010. [Google Scholar] [CrossRef]
- Tan, H.W.; Mo, H.Y.; Lau, A.T.Y.; Xu, Y.M. Selenium species: Current status and potentials in cancer prevention and therapy. Int. J. Mol. Sci. 2019, 20, 75. [Google Scholar] [CrossRef]
- Chaudhary, S.; Mehta, S.K. Selenium Nanomaterials: Applications in Electronics, Catalysis and Sensors. J. Nanosci. Nanotechnol. 2014, 14, 1658–1674. [Google Scholar] [CrossRef] [PubMed]
- Venkat Kumar, S.; Rajeshkumar, S. Plant-Based Synthesis of Nanoparticles and Their Impact. Nanomater. Plants Algae Microorg. 2018, 33–57. [Google Scholar]
- Palomo-Siguero, M.; Vera, P.; Echegoyen, Y.; Nerin, C.; Cámara, C.; Madrid, Y. Asymmetrical flow field-flow fractionation coupled to inductively coupled plasma mass spectrometry for sizing SeNPs for packaging applications. Spectrochim. Acta Part. B At. Spectrosc. 2017, 132, 19–25. [Google Scholar] [CrossRef]
- Srivastava, N.; Mukhopadhyay, M. Biosynthesis and structural characterization of selenium nanoparticles mediated by Zooglea ramigera. Powder Technol. 2013, 244, 26–29. [Google Scholar] [CrossRef]
- Tang, S.; Wang, T.; Jiang, M.; Huang, C.; Lai, C.; Fan, Y.; Yong, Q. Construction of arabinogalactans/selenium nanoparticles composites for enhancement of the antitumor activity. Int. J. Biol. Macromol. 2019, 128, 444–451. [Google Scholar] [CrossRef]
- Vieira, A.P.; Stein, E.M.; Andreguetti, D.X.; Cebrián-Torrejón, G.; Doménech-Carbó, A.; Colepicolo, P.; Ferreira, A.M.D.C. “Sweet chemistry”: A Green way for obtaining selenium nanoparticles active against cancer cells. J. Braz. Chem. Soc. 2017, 28, 2021–2027. [Google Scholar] [CrossRef]
- Fardsadegh, B.; Jafarizadeh-Malmiri, H. Aloe vera leaf extract mediated green synthesis of selenium nanoparticles and assessment of their in vitro antimicrobial activity against spoilage fungi and pathogenic bacteria strains. Green Process. Synth. 2019, 8, 399–407. [Google Scholar] [CrossRef]
- Fardsadegh, B.; Vaghari, H.; Mohammad-Jafari, R.; Najian, Y.; Jafarizadeh-Malmiri, H. Biosynthesis, characterization and antimicrobial activities assessment of fabricated selenium nanoparticles using Pelargonium zonale leaf extract. Green Process. Synth. 2019, 8, 191–198. [Google Scholar] [CrossRef]
- Mirzajani, R.; Karimi, S. Ultrasonic assisted synthesis of magnetic Ni-Ag bimetallic nanoparticles supported on reduced graphene oxide for sonochemical simultaneous removal of sunset yellow and tartrazine dyes by response surface optimization: Application of derivative spectrophotometry. Ultrason. Sonochem. 2019, 50, 239–250. [Google Scholar]
- Baruwati, B.; Polshettiwar Vivek, V.R.S. Microwave-assisted synthesis of nanomaterials in aqueous media. In Aqueous Microwave Assisted Chemistry; Polshettiwar, V., Varma, R.S., Eds.; Green Chemistry Series; The Royal Society of Chemistry: Cambridge, UK, 2010; pp. 176–216. [Google Scholar]
- Mirzaei, A.; Neri, G. Microwave-assisted synthesis of metal oxide nanostructures for gas sensing application: A review. Sens. Actuators B Chem. 2016, 237, 749–775. [Google Scholar] [CrossRef]
- Jadhav, A.A.; Khanna, P.K. Impact of microwave irradiation on cyclo-octeno-1,2,3-selenadiazole: Formation of selenium nanoparticles and their polymorphs. RSC Adv. 2015, 5, 44756–44763. [Google Scholar] [CrossRef]
- Schütz, M.B.; Xiao, L.; Lehnen, T.; Fischer, T.; Mathur, S. Microwave-assisted synthesis of nanocrystalline binary and ternary metal oxides. Int. Mater. Rev. 2018, 63, 341–374. [Google Scholar] [CrossRef]
- Okiyama, D.C.G.; Navarro, S.L.B.; Rodrigues, C.E.C. Cocoa shell and its compounds: Applications in the food industry. Trends Food Sci. Technol. 2017, 63, 103–112. [Google Scholar] [CrossRef]
- Li, H.; Liu, D.; Li, S.; Xue, C. Synthesis and cytotoxicity of selenium nanoparticles stabilized by α-d-glucan from Castanea mollissima Blume. Int. J. Biol. Macromol. 2019, 129, 818–826. [Google Scholar] [CrossRef]
- Zhang, J.; Teng, Z.; Yuan, Y.; Zeng, Q.-Z.; Lou, Z.; Lee, S.-H.; Wang, Q. Development, physicochemical characterization and cytotoxicity of selenium nanoparticles stabilized by beta-lactoglobulin. Int. J. Biol. Macromol. 2018, 107, 1406–1413. [Google Scholar] [CrossRef]
- Pu, J.-B.; Xia, B.-H.; Hu, Y.-J.; Zhang, H.-J.; Chen, J.; Zhou, J.; Liang, W.-Q.; Xu, P. Multi-Optimization of ultrasonic-assisted enzymatic extraction of atratylodes macrocephala polysaccharides and antioxidants using response surface methodology and desirability function approach. Molecules 2015, 20, 22220–22235. [Google Scholar] [CrossRef]
- Menon, S.; KS, S.D.; R., S.; S., R.; S., V.K. Selenium nanoparticles: A potent chemotherapeutic agent and an elucidation of its mechanism. Colloids Surf. B Biointerfaces 2018, 170, 280–292. [Google Scholar] [CrossRef]
- Le Ching Tan, V.; Hinchman, A.; Williams, R.; Tran, P.A.; Fox, K. Nanostructured biomedical selenium at the biological interface (Review). Biointerphases 2018, 13, 06D301. [Google Scholar]
- Yan, J.-K.; Qiu, W.-Y.; Wang, Y.-Y.; Wang, W.-H.; Yang, Y.; Zhang, H.-N. Fabrication and stabilization of biocompatible selenium nanoparticles by carboxylic curdlans with various molecular properties. Carbohydr. Polym. 2018, 179, 19–27. [Google Scholar] [CrossRef]
- Karuppanan, K.; Nagaraj, E.; Sujatha, V. Diospyros montana leaf extract-mediated synthesis of selenium nanoparticles and their biological applications. New J. Chem. 2017, 41, 7481–7490. [Google Scholar]
- Clogston, J.D.; Patri, A.K. Zeta Potential Measurement. In Characterization of Nanoparticles Intended for Drug Delivery; McNeil, S.E., Ed.; Humana Press: Totowa, NJ, USA, 2011; pp. 63–70. [Google Scholar]
- Gabor, F. Characterization of Nanoparticles Intended for Drug Delivery. Sci. Pharm. 2011, 79, 701–702. [Google Scholar] [CrossRef]
- Qiu, W.-Y.; Wang, Y.-Y.; Wang, M.; Yan, J.-K. Construction, stability, and enhanced antioxidant activity of pectin-decorated selenium nanoparticles. Colloids Surfaces B Biointerfaces 2018, 170, 692–700. [Google Scholar] [CrossRef] [PubMed]
- Menon, S.; K.S., S.D.; Agarwal, H.; Shanmugam, V.K. Efficacy of Biogenic Selenium Nanoparticles from an extract of ginger towards evaluation on anti-microbial and anti-oxidant activities. Colloid Interface Sci. Commun. 2019, 29, 1–8. [Google Scholar] [CrossRef]
- Ondari Nyakundi, E.; Padmanabhan, M.N. Green chemistry focus on optimization of silver nanoparticles using response surface methodology (RSM) and mosquitocidal activity: Anopheles stephensi (Diptera: Culicidae). Spectrochim. Acta Part. A Mol. Biomol. Spectrosc. 2015, 149, 978–984. [Google Scholar] [CrossRef] [PubMed]
- Dananjaya, S.H.S.; Kumar, R.S.; Yang, M.; Nikapitiya, C.; Lee, J.; De Zoysa, M. Synthesis, characterization of ZnO-chitosan nanocomposites and evaluation of its antifungal activity against pathogenic Candida albicans. Int. J. Biol. Macromol. 2018, 108, 1281–1288. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Wang, X.; Xie, A.; Huang, L.; Zhu, J.; Chen, L. Synthesis of dextran/Se nanocomposites for nanomedicine application. Mater. Chem. Phys. 2008, 109, 534–540. [Google Scholar] [CrossRef]
- Cui, D.; Yan, C.; Miao, J.; Zhang, X.; Chen, J.; Sun, L.; Meng, L.; Liang, T.; Li, Q. Synthesis, characterization and antitumor properties of selenium nanoparticles coupling with ferulic acid. Mater. Sci. Eng. C 2018, 90, 104–112. [Google Scholar] [CrossRef]
- Wang, L.; Li, X.; Wang, B. Synthesis, characterization and antioxidant activity of selenium modified polysaccharides from Hohenbuehelia serotina. Int. J. Biol. Macromol. 2018, 120, 1362–1368. [Google Scholar] [CrossRef]
- Tugarova, A.V.; Mamchenkova, P.V.; Dyatlova, Y.A.; Kamnev, A.A. FTIR and Raman spectroscopic studies of selenium nanoparticles synthesised by the bacterium Azospirillum thiophilum. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2018, 192, 458–463. [Google Scholar] [CrossRef]
- Larkin, P. Chapter 1 - Introduction: Infrared and Raman Spectroscopy. In Infrared and Raman Spectroscopy; Larkin, P., Ed.; Elsevier: Oxford, UK, 2011; pp. 1–5. ISBN 978-0-12-386984-5. [Google Scholar]
- Pilot, R.; Signorini, R.; Fabris, L. Surface-Enhanced Raman Spectroscopy: Principles, Substrates, and Applications. In Metal Nanoparticles and Clusters: Advances in Synthesis, Properties and Applications; Cham, S.L., Ed.; Springer: Berlin/Heidelberg, Germany, 2018; pp. 89–164. ISBN 978-3-319-68053-8. [Google Scholar]
- Oremland, R.S.; Herbel, M.J.; Blum, J.S.; Langley, S.; Beveridge, T.J.; Ajayan, P.M.; Sutto, T.; Ellis, A.V.; Curran, S. Structural and spectral features of selenium nanospheres produced by se-respiring bacteria. Appl. Environ. Microbiol. 2004, 70, 52–60. [Google Scholar] [CrossRef]
- Ruiz Fresneda, M.A.; Delgado Martín, J.; Gómez Bolívar, J.; Fernández Cantos, M.V.; Bosch-Estévez, G.; Martínez Moreno, M.F.; Merroun, M.L. Green synthesis and biotransformation of amorphous Se nanospheres to trigonal 1D Se nanostructures: Impact on Se mobility within the concept of radioactive waste disposal. Environ. Sci. Nano 2018, 5, 2103–2116. [Google Scholar] [CrossRef]
- Zhang, W.; Chen, Z.; Liu, H.; Zhang, L.; Gao, P.; Li, D. Biosynthesis and structural characteristics of selenium nanoparticles by Pseudomonas alcaliphila. Colloids Surfaces B Biointerfaces 2011, 88, 196–201. [Google Scholar] [CrossRef]
- Guleria, A.; Singh, A.K.; Neogy, S.; Adhikari, S. Probing the phase transformation of Selenium nanoparticles synthesized in the host matrix of neat room temperature ionic liquid via radiation route. Mater. Chem. Phys. 2017, 202, 204–214. [Google Scholar] [CrossRef]
- Hosnedlova, B.; Kepinska, M.; Skalickova, S.; Fernandez, C.; Ruttkay-Nedecky, B.; Peng, Q.; Baron, M.; Melcova, M.; Opatrilova, R.; Zidkova, J.; et al. Nano-selenium and its nanomedicine applications: A critical review. Int. J. Nanomed. 2018, 13, 2107–2128. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.-H.; Li, L.-L.; Zhou, N.-Q.; Liu, J.-H.; Huang, Q.; Wang, H.-J.; Tian, J.; Yu, H.-Q. In vivo synthesis of nano-selenium by Tetrahymena thermophila SB210. Enzyme Microb. Technol. 2016, 95, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-Y.; Qiu, W.-Y.; Sun, L.; Ding, Z.-C.; Yan, J.-K. Preparation, characterization, and antioxidant capacities of selenium nanoparticles stabilized using polysaccharide–protein complexes from Corbicula fluminea. Food Biosci. 2018, 26, 177–184. [Google Scholar] [CrossRef]
- Bartosiak, M.; Giersz, J.; Jankowski, K. Analytical monitoring of selenium nanoparticles green synthesis using photochemical vapor generation coupled with MIP-OES and UV–Vis spectrophotometry. Microchem. J. 2019, 145, 1169–1175. [Google Scholar] [CrossRef]
- Chen, W.; Yue, L.; Jiang, Q.; Liu, X.; Xia, W. Synthesis of varisized chitosan-selenium nanocomposites through heating treatment and evaluation of their antioxidant properties. Int. J. Biol. Macromol. 2018, 114, 751–758. [Google Scholar] [CrossRef]
- Liu, Y.; Zeng, S.; Liu, Y.; Wu, W.; Shen, Y.; Zhang, L.; Li, C.; Chen, H.; Liu, A.; Shen, L.; et al. Synthesis and antidiabetic activity of selenium nanoparticles in the presence of polysaccharides from Catathelasma ventricosum. Int. J. Biol. Macromol. 2018, 114, 632–639. [Google Scholar] [CrossRef]
- Sharma, G.; Sharma, A.R.; Bhavesh, R.; Park, J.; Ganbold, B.; Nam, J.S.; Lee, S.S. Biomolecule-mediated synthesis of selenium nanoparticles using dried vitis vinifera (raisin) extract. Molecules 2014, 19, 2761–2770. [Google Scholar] [CrossRef]
- Moreno-Martin, G.; Pescuma, M.; PéreZ-Corona, T.; Mozzi, F.; Madrid, Y. Determination of size and mass-and number-based concentration of biogenic SeNPs synthesized by lactic acid bacteria by using a multimethod approach. Anal. Chim. Acta 2017, 992, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Pouri, S.; Motamedi, H.; Honary, S.; Kazeminezhad, I. Biological Synthesis of Selenium Nanoparticles and Evaluation of their Bioavailability. Braz. Arch. Biol. Technol. 2018, 60, 1–12. [Google Scholar] [CrossRef]
- Tam, K.; Ho, C.T.; Lee, J.H.; Lai, M.; Chang, C.H.; Rheem, Y.; Chen, W.; Hur, H.G.; Myung, N.V. Growth mechanism of amorphous selenium nanoparticles synthesized by Shewanella sp. HN-41. Biosci. Biotechnol. Biochem. 2010, 74, 696–700. [Google Scholar] [CrossRef]
- Verma, P.; Maheshwari, S.K. Preparation of Sliver and Selenium Nanoparticles and Its Characterization by Dynamic Light Scattering and Scanning Electron Microscopy. J. Microsc. Ultrastruct. 2018, 6, 182–187. [Google Scholar] [PubMed]
- Sun, D.; Kang, S.; Liu, C.; Lu, Q.; Cui, L.; Hu, B. Effect of zeta potential and particle size on the stability of SiO2 nanospheres as carrier for ultrasound imaging contrast agents. Int. J. Electrochem. Sci. 2016, 11, 8520–8529. [Google Scholar] [CrossRef]
- Meng, Y.; Zhang, Y.; Jia, N.; Qiao, H.; Zhu, M.; Meng, Q.; Lu, Q.; Zu, Y. Synthesis and evaluation of a novel water-soluble high Se-enriched Astragalus polysaccharide nanoparticles. Int. J. Biol. Macromol. 2018, 118, 1438–1448. [Google Scholar] [CrossRef]
- Luesakul, U.; Komenek, S.; Puthong, S.; Muangsin, N. Shape-controlled synthesis of cubic-like selenium nanoparticles via the self-assembly method. Carbohydr. Polym. 2016, 153, 435–444. [Google Scholar] [CrossRef]
- Petryayeva, E.; Krull, U.J. Localized surface plasmon resonance: Nanostructures, bioassays and biosensing—A review. Anal. Chim. Acta 2011, 706, 8–24. [Google Scholar] [CrossRef]
- Roy, R.; Choudhary, V.S.; Patra, M.K.; Pandya, A. Effect of annealing temperature on the electrical and optical properties of nanocrystalline selenium thin films. J. Optoelectron. Adv. Mater. 2006, 8, 1352–1355. [Google Scholar]
- Panahi-Kalamuei, M.; Salavati-Niasari, M.; Hosseinpour-Mashkani, S.M. Facile microwave synthesis, characterization, and solar cell application of selenium nanoparticles. J. Alloys Compd. 2014, 617, 627–632. [Google Scholar] [CrossRef]
- Jiang, F.; Cai, W.; Tan, G. Facile Synthesis and Optical Properties of Small Selenium Nanocrystals and Nanorods. Nanoscale Res. Lett. 2017, 12, 401. [Google Scholar] [CrossRef] [PubMed]
- Guleria, A.; Neogy, S.; Adhikari, S. Room temperature ionic liquid assisted synthesis of highly stable amorphous Se nanoparticles: A rapid and facile methodology. Mater. Lett. 2018, 217, 198–201. [Google Scholar] [CrossRef]
- Zhang, H.; Zhou, H.; Bai, J.; Li, Y.; Yang, J.; Ma, Q.; Qu, Y. Biosynthesis of selenium nanoparticles mediated by fungus Mariannaea sp. HJ and their characterization. Colloids Surfaces A Physicochem. Eng. Asp. 2019, 571, 9–16. [Google Scholar] [CrossRef]
- Kong, H.; Yang, J.; Zhang, Y.; Fang, Y.; Nishinari, K.; Phillips, G.O. Synthesis and antioxidant properties of gum arabic-stabilized selenium nanoparticles. Int. J. Biol. Macromol. 2014, 65, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Gunti, L.; Dass, R.S.; Kalagatur, N.K. Phytofabrication of selenium nanoparticles from emblica officinalis fruit extract and exploring its biopotential applications: Antioxidant, antimicrobial, and biocompatibility. Front. Microbiol. 2019, 10, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Cruz, L.Y.; Wang, D.; Liu, J. Biosynthesis of selenium nanoparticles, characterization and X-ray induced radiotherapy for the treatment of lung cancer with interstitial lung disease. J. Photochem. Photobiol. B Biol. 2019, 191, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Huang, Q.; Zheng, Z.; Guan, H.; Liu, S. Construction of a Cordyceps sinensis exopolysaccharide-conjugated selenium nanoparticles and enhancement of their antioxidant activities. Int. J. Biol. Macromol. 2017, 99, 483–491. [Google Scholar] [CrossRef]
- Huang, Z.; Guo, B.J.; Wong, R.N.S.; Jiang, Y. Characterization and antioxidant activity of selenium-containing phycocyanin isolated from Spirulina platensis. Food Chem. 2007, 100, 1137–1143. [Google Scholar] [CrossRef]
- Yu, B.; You, P.; Song, M.; Zhou, Y.; Yu, F.; Zheng, W. A facile and fast synthetic approach to create selenium nanoparticles with diverse shapes and their antioxidation ability. New J. Chem. 2016, 40, 1118–1123. [Google Scholar] [CrossRef]
- Vyas, J.; Rana, S. Antioxidant activity and green synthesis of selenium nanoparticles using allium sativum extract. Int. J. Phytomedicine 2017, 9, 634. [Google Scholar] [CrossRef]
- Martínez, R.; Torres, P.; Meneses, M.A.; Figueroa, J.G.; PéreZ-Álvarez, J.A.; Viuda-Martos, M. Chemical, technological and in vitro antioxidant properties of cocoa (Theobroma cacao L.) co-products. Food Res. Int. 2012, 49, 39–45. [Google Scholar] [CrossRef]
- Filisetti-Cozzi, T.M.C.C.; Carpita, N.C. Measurement of uronic acids without interference from neutral sugars. Anal. Biochem. 1991, 197, 157–162. [Google Scholar] [CrossRef]
- Albalasmeh, A.A.; Berhe, A.A.; Ghezzehei, T.A. A new method for rapid determination of carbohydrate and total carbon concentrations using UV spectrophotometry. Carbohydr. Polym. 2013, 97, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Awe, F.B.; Fagbemi, T.N.; Ifesan, B.O.T.; Badejo, A.A. Antioxidant properties of cold and hot water extracts of cocoa, Hibiscus flower extract, and ginger beverage blends. Food Res. Int. 2013, 52, 490–495. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Bezerra, M.A.; Ferreira, S.L.C.; Novaes, C.G.; dos Santos, A.M.P.; Valasques, G.S.; da Mata Cerqueira, U.M.F.; dos Santos Alves, J.P. Simultaneous optimization of multiple responses and its application in Analytical Chemistry–review. Talanta 2019, 194, 941–959. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.; Arshad, M.A.; Ijaz, S.; Khurshid, U.; Sofi, F.; Azam, R. Review on methods used to determine Antioxidant activity. Int. J. Multidiscip. Res. Dev. 2014, 1, 41–46. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |
| Constituents | Content |
|---|---|
| Uronic acid (mg GlcA/g) | 82.7 ± 6.3 |
| Total phenolics (mg GAE/g) | 23.2 ± 0.4 |
| Total polysaccharides (mg Glu/g) | 300.8 ± 2.5 |
| Proteins (mg BSA/g) | 170.3 ± 3.1 |
| Run | Time (min) | Power (W) | Na2SeO3 (g) | Average Crystalline Size (nm) * | Z-Potential (mV) ** |
|---|---|---|---|---|---|
| 1 | 10.0 | 600.0 | 0.275 | 53.1 | −19.27 |
| 2 | 15.0 | 400.0 | 0.150 | 43.8 | −33.60 |
| 3 | 1.6 | 600.0 | 0.275 | 46.9 | −26.07 |
| 4 | 10.0 | 263.6 | 0.275 | 48.5 | −24.67 |
| 5 | 10.0 | 600.0 | 0.065 | 48.9 | −31.67 |
| 6 | 18.4 | 600.0 | 0.275 | 46.5 | −25.63 |
| 7 | 15.0 | 800.0 | 0.150 | 40.9 | −36.47 |
| 8 | 10.0 | 600.0 | 0.275 | 45.9 | −14.57 |
| 9 | 5.0 | 800.0 | 0.150 | 43.4 | −28.53 |
| 10 | 10.0 | 600.0 | 0.275 | 49.9 | −18.50 |
| 11 | 10.0 | 600.0 | 0.275 | 47.5 | −15.67 |
| 12 | 5.0 | 800.0 | 0.400 | 58.9 | −25.03 |
| 13 | 15.0 | 400.0 | 0.400 | 66.6 | −25.70 |
| 14 | 5.0 | 400.0 | 0.400 | 58.0 | −27.30 |
| 15 | 10.0 | 600.0 | 0.275 | 48.6 | −14.77 |
| 16 | 10.0 | 600.0 | 0.275 | 51.5 | −14.73 |
| 17 | 10.0 | 936.4 | 0.275 | 45.6 | −26.97 |
| 18 | 15.0 | 800.0 | 0.400 | 58.1 | −26.77 |
| 19 | 5.0 | 400.0 | 0.150 | 43.8 | −22.27 |
| 20 | 10.0 | 600.0 | 0.275 | 49.1 | −16.30 |
| 21 | 10.0 | 600.0 | 0.485 | 66.2 | −22.50 |
| 22 | 10.0 | 600.0 | 0.275 | 49.2 | −17.90 |
| 23 | 10.0 | 600.0 | 0.275 | 48.9 | −15.13 |
| Source | Sum of Squares | DF | Mean Square | F-Value | p-Value |
|---|---|---|---|---|---|
| Size | |||||
| A | 1.57 | 1 | 1.57 | 0.35 | 0.5688 |
| B | 18.16 | 1 | 18.16 | 4.09 | 0.0777 |
| C | 715.34 | 1 | 715.34 | 161.25 | 0.0001 *** |
| AA | 7.17 | 1 | 7.17 | 1.62 | 0.2393 |
| AB | 17.73 | 1 | 17.73 | 4.00 | 0.0806 |
| AC | 13.03 | 1 | 13.03 | 2.94 | 0.1249 |
| BB | 4.77 | 1 | 4.77 | 1.08 | 0.3300 |
| BC | 2.48 | 1 | 2.48 | 0.56 | 0.4765 |
| CC | 158.74 | 1 | 158.74 | 35.78 | 0.0003 *** |
| Lack of fit | 55.06 | 5 | 11.01 | 2.48 | 0.1215 |
| Pure error | 35.49 | 8 | 4.44 | ||
| Cor. Total | 1030.31 | 22 | |||
| R2 | 0.9121 | ||||
| Adj R2 | 0.8512 | ||||
| CV (%) | 2.64 | ||||
| Z-Potential | |||||
| A | 25.52 | 1 | 25.52 | 7.92 | 0.0227 * |
| B | 10.19 | 1 | 10.19 | 3.16 | 0.1132 |
| C | 72.62 | 1 | 72.62 | 22.53 | 0.0015 ** |
| AA | 211.89 | 1 | 211.89 | 65.75 | 0.0001 *** |
| AB | 0.00 | 1 | 0.00 | 0.00 | 0.9924 |
| AC | 45.74 | 1 | 45.74 | 14.19 | 0.0055 ** |
| BB | 210.66 | 1 | 210.66 | 65.36 | 0.0001 *** |
| BC | 13.34 | 1 | 13.34 | 4.14 | 0.0763 |
| CC | 265.59 | 1 | 265.59 | 82.41 | 0.0001 *** |
| Lack of fit | 35.21 | 5 | 7.04 | 2.19 | 0.1558 |
| Pure error | 25.78 | 8 | 3.22 | ||
| Cor. Total | 907.21 | 22 | |||
| R2 | 0.9327 | ||||
| Adj R2 | 0.8862 | ||||
| CV (%) | 2.16 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mellinas, C.; Jiménez, A.; Garrigós, M.d.C. Microwave-Assisted Green Synthesis and Antioxidant Activity of Selenium Nanoparticles Using Theobroma cacao L. Bean Shell Extract. Molecules 2019, 24, 4048. https://doi.org/10.3390/molecules24224048
Mellinas C, Jiménez A, Garrigós MdC. Microwave-Assisted Green Synthesis and Antioxidant Activity of Selenium Nanoparticles Using Theobroma cacao L. Bean Shell Extract. Molecules. 2019; 24(22):4048. https://doi.org/10.3390/molecules24224048
Chicago/Turabian StyleMellinas, Cristina, Alfonso Jiménez, and María del Carmen Garrigós. 2019. "Microwave-Assisted Green Synthesis and Antioxidant Activity of Selenium Nanoparticles Using Theobroma cacao L. Bean Shell Extract" Molecules 24, no. 22: 4048. https://doi.org/10.3390/molecules24224048
APA StyleMellinas, C., Jiménez, A., & Garrigós, M. d. C. (2019). Microwave-Assisted Green Synthesis and Antioxidant Activity of Selenium Nanoparticles Using Theobroma cacao L. Bean Shell Extract. Molecules, 24(22), 4048. https://doi.org/10.3390/molecules24224048








