Abstract
The preparation of complex architectures has inspired the search for new methods and new processes in organic synthesis. Multicomponent reactions have become an interesting approach to achieve such molecular diversity and complexity. This review intends to illustrate important gold-catalyzed examples for the past ten years leading to interesting skeletons involved in biologically active compounds.
1. Introduction
Multicomponent reactions (MCRs) have been defined as processes in which three or more reagents are added to a single vessel at the same time affording final products containing most of the atoms from the starting materials [1,2]. Therefore, more than one chemical transformation is involved in this approach without the necessity of changing the reaction media and without purification of the intermediates after each transformation. In the last years, MCRs have become powerful and efficient tools to afford molecular diversity, giving rise to libraries of small organic molecules while requiring less time and effort when compared with stop-and-go procedures [1,2]. These processes have also gained special interest in the pharmaceutical industry because of the easy formation of large libraries of compounds with potential biological activities [3].
This importance is reflected in the large number of publications reported in this field over the last decade [4,5]. Moreover, the utility and potential of multicomponent procedures have been confirmed by the development of a high number of molecules with remarkable biological activities [6,7,8,9,10].
Over the last two decades, gold catalysis has experienced a growing interest and an impressive development. Proof of that is the huge number of works published in this area of research [11,12,13]. The application of Au(I) in homogenous catalysis has been predominant [14,15,16,17], while reports based on Au(III) catalysis have been more scarce and have mainly focused on the application of simple salts such as AuCl3 or AuBr3 [18,19,20].
In relation with both fields of research, important contributions have been achieved with gold catalysts in the development of new multicomponent reactions, although many of these reactions have been only focused on the coupling of aldehydes, amines and alkynes known as A3-coupling-type reactions for the synthesis of propargylamines [21,22]. Since this process has already been compiled and revised in a previous pivotal review [23], we will give to the reader a broader vision of the scope of gold catalysts in the design and development of new MCRs beyond A3-coupling-type reactions.
In this context, we compile here some other representative pioneer multicomponent reactions for the construction of functionalized molecules such as those disclosed in Figure 1. Nevertheless, the field of multicomponent reactions using gold catalysts still needs a future progress and growth in comparison with other metals or approaches. Beyond the molecules depicted in Figure 1, other interesting protocols using gold catalysts have also been developed [24,25,26,27,28].
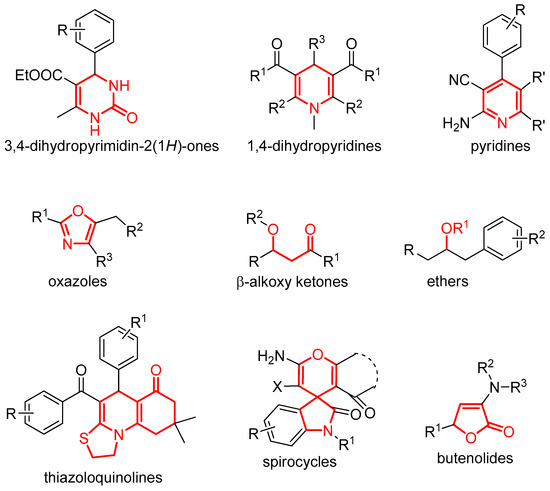
Figure 1.
Structural cores described in this review and synthesized by means of gold-catalyzed MCRs.
2. Gold-Catalyzed Multicomponent Reactions
2.1. Multicomponent Synthesis of 3,4-Dihydropyrimidin-2(1H)-ones. A Biginelli Approach
3,4-Dihydropyrimidin-2(1H)-ones 4 (DHPMs) have attracted considerable attention due to their antiviral, antitumor or anti-inflammatory activity, as well as their use as calcium channel blockers and as backbones of anticancer drugs [29,30,31]. The most common route to prepare DHPMs is the use of a classical Biginelli MCR [32,33,34].
Tran and co-workers reported the first use of unsupported Au nanorods to catalyze the Biginelli reaction [35]. The authors prepared gold nanoparticles in different shapes, such as nanospheres, nanorods and nanostars and developed the Biginelli MCR with the gold nanorods. After a preliminary screening of the reaction conditions, the reaction was utilized to prepare a variety of aromatic aldehydes using 1.0 mL of Au nanorod colloid at 80–120 °C (Scheme 1) [35].
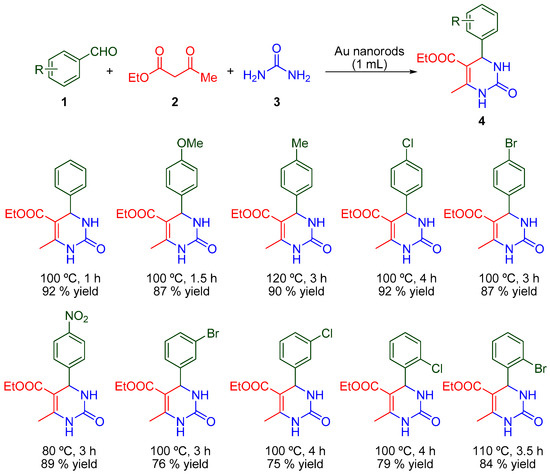
Scheme 1.
Biginelli reaction catalyzed by Au nanorods.
The final DHPMs were obtained in all cases with high yields, following a simple experimental procedure with a high catalyst efficiency. Moreover, the protocol allowed the elimination of volatile organic solvents and carrying out the reactions without an inert atmosphere Unfortunately, the authors did not propose a plausible reaction mechanism.
2.2. Multicomponent Synthesis of 1,4-Dihydropyridines (1,4-DHPs)
The 1,4-dihydropyridine (1,4-DHP) structural core, commonly synthesized following a classical Hantzsch condensation [36], has also attracted the attention of different research groups due to its appealing biological activity, including, among other properties, antioxidant, antidiabetic or antitumor effects (Figure 2) [37,38,39]. Therefore, their synthesis in racemic and chiral version is still an active area of research that includes multicomponent approaches [40,41,42,43,44].
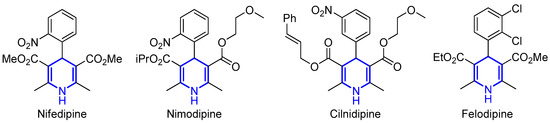
Figure 2.
Biologically active 1,4-DHPs.
In this domain of research Cao and co-workers pioneered a gold-catalyzed multicomponent example to synthesize N-substituted DHPs 8, through reaction of alkynes 5a,b, methanamine 6 and different substituted aldehydes 7a–r (Scheme 2). After an exploration of potential catalysts, base additives, solvent and temperature, the scope of the reaction was explored with the best reaction conditions (Scheme 2) [45].
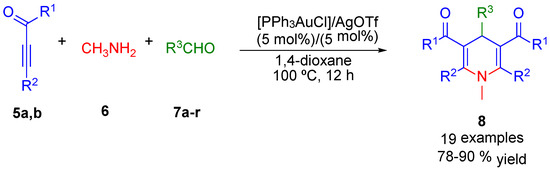
Scheme 2.
Au(I)-catalyzed multicomponent synthesis of 1,4-dihydropyridines 8.
This convergent approach, by generation of C–C and C–N bonds, affords N-substituted DHPs derivatives 8 in good yields. On the basis of the experimental results, a plausible mechanism was suggested by the authors to explain the synthesis of 1,4-DHPs 8 (Scheme 3).
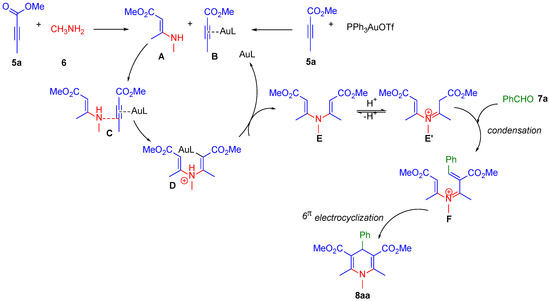
Scheme 3.
Proposed catalytic cycle for the synthesis of 8aa.
First, adduct A would be obtained after nucleophilic addition of 5a with 6, while the activation of 5a by PPh3AuOTf would afford Au(I)–alkyne complex B. The interaction of species A and B could produce intermediate C, which can undergo C–N bond formation to generate polarized intermediate D prior to the formation of intermediate E. This process is facilitated by the electrophilicity enhancement of the C-C bond promoted by the previous coordination of the gold complex to the species 5a. The iminium ion intermediate E′ can be obtained through an isomerization pathway from E, which can condense with aldehyde 7a affording intermediate F by the attack from E′ on the carbonyl carbon atom of the benzaldehyde. Finally, intermediate F can evolve to product 8aa through a 6π electrocyclization followed by a deprotonation process.
2.3. Multicomponent Synthesis of Pyridines
Polysubstituted pyridines are compounds attracting great attention mostly because they constitute the skeleton of many natural products and organic functional materials, and also because they can be found in many synthetic compounds with pharmaceutical applications [46,47,48]. For example, 2-amino-3-cyanopyridine derivatives (Figure 3) have gained considerable attention due to their use as potent HIV-1 inhibitors [49,50].
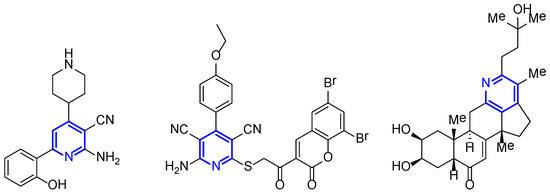
Figure 3.
Biologically active pyridine derivatives.
In this field, Jonnalagadda and co-workers developed an efficient and reusable Au/MgO catalyst to promote the multicomponent synthesis of highly substituted pyridines 13 and 15 in high yields and in short reaction times (Scheme 4) [51].
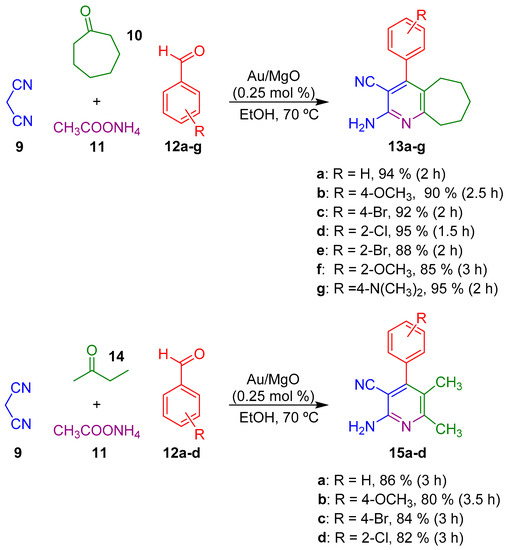
Scheme 4.
Multicomponent synthesis of highly substituted pyridines 13 and 15.
A plausible reaction mechanism for the preparation of these highly substituted pyridines has been proposed by Jonnalagadda and co-workers. The first step consists on the formation of the arylidenemalononitrile intermediate 16 through a standard Knoevenagel condensation. The subsequent Michael-type addition of ketone 10 to the activated double bond of the arylidene affords species 17. Finally, condensation with ammonia, followed by the cyclization of the resulting enamine 18 and the corresponding oxidation step forms compounds 13a–g (Scheme 5) [51].
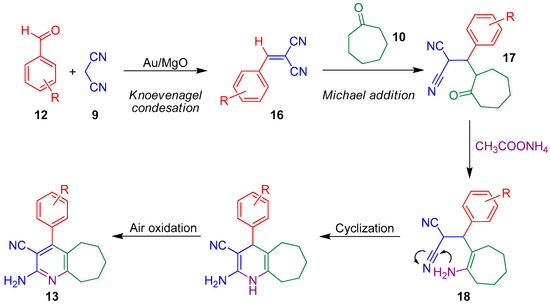
Scheme 5.
Proposed stepwise mechanism for the preparation of pyridines 13.
It should be noted that the catalyst was easily recovered and reused after the reaction without any loss of its catalytic activity during at least 5 runs.
2.4. Multicomponent Synthesis of Oxazoles
The oxazole motif is found in many compounds with useful applications and their synthesis is an active task in different areas of research [52,53,54,55,56,57,58,59,60]. In 2013, Strand and co-workers pioneered the first efficient gold-catalyzed three-component domino reaction to generate trisubstituted oxazoles directly from imines, alkynes, and acid chlorides (Scheme 6, Scheme 7 and Scheme 8) [61]. The use of MCRs had already been used for the synthesis of this family of products [62,63,64] and more recently Cai and co-workers have also pioneered a gold-catalyzed example [65]. The scope of the reaction was investigated by varying the corresponding alkyne compound 21 (Scheme 6), the acyl chloride 20 (Scheme 7) or the N-benzylimines 19 (Scheme 8), which provides variation on three substituted positions in the final oxazole ring [61].
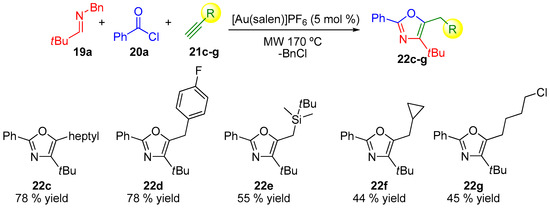
Scheme 6.
Structural variations in the alkyne compound 21 (salen: N,N′-ethylenebis(salicylimine)).
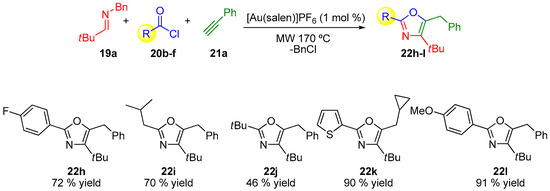
Scheme 7.
Structural variations in the acyl chlorides 20.
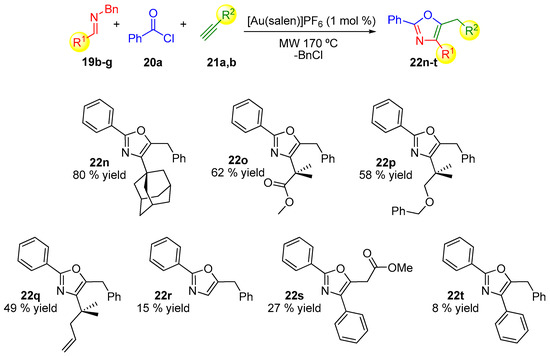
Scheme 8.
Structural variations in the imines 19.
Several qualitative kinetic experiments confirmed that gold was not significantly involved in the cyclization step but did contribute to the isomeric transformation of IX into X (Scheme 9). Those observations led to propose the addition of metal acetylide II to the activated N-acyl iminium salt V to give propargyl amide as the initial step for the formation of the oxazole derivative. The acylation of amine (IV) induces the release of a chlorine anion that can subsequently react with VII to produce intermediate IX and the respective benzyl chloride (VIII) release. Finally, the isomerization of IX into the desired oxazole XI is achieved by a synergistic process between a Brønsted acid and a metal-mediated catalysis [66]. The same research group has also reported a more recent example of a gold-catalyzed multicomponent reaction for the synthesis of oxazoles [67].
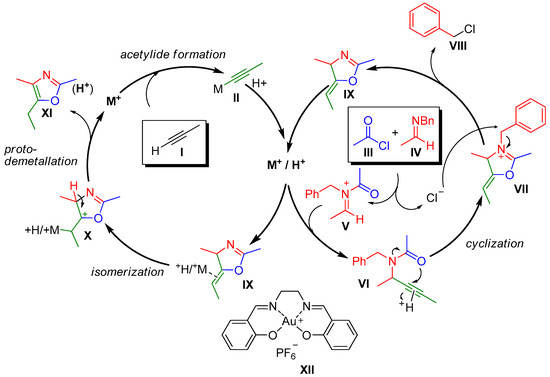
Scheme 9.
Proposed mechanism for the gold-catalyzed formation of oxazoles XI.
2.5. Multicomponent Synthesis of β-Alkoxy Ketones
β-Alkoxyketones are valuable building blocks in organic synthesis [68,69], and this structural core is present in many biologically active compounds and natural products [70,71,72], such as those disclosed in Figure 4. They are commonly synthesized either via aldol reaction or sequential epoxidation and reduction of enones.
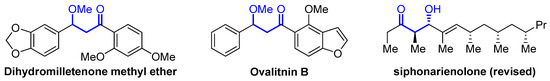
Figure 4.
Biologically active β-alkoxy ketones.
In this field, a gold(I)-catalyzed multicomponent synthesis of β-alkoxyketones from readily available precursors such as aldehydes 23, alcohols 25, and alkynes 24 was described in 2012 by Schultz and co-workers. The authors used the gold complex SPhosAuNTf2 as a catalyst to prepare diverse β-alkoxyketone products 26 (Scheme 10) [73].
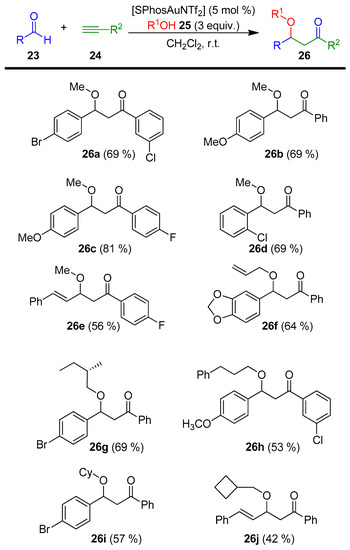
Scheme 10.
Gold(I)-catalyzed multicomponent synthesis of β-alkoxyketones 26.
With the best reaction conditions, the scope of this approach was explored by varying the aldehyde 23, alkyne 24, and alcohol 25 reaction partners. The resulting β-alkoxyketones 26 were obtained from moderate to good yields.
The authors proposed that an intermolecular oxyauration reaction between the alkyne 24 and the hemiacetal 27, generated in situ by the reaction of the aldehyde with one equivalent of the alcohol 25, could provide 28, which could subsequent split into gold(I) enol 29 and oxocarbenium ion 30 (Scheme 11). Both intermediates could then undergo an intermolecular addition to furnish the β-alkoxyketone product 26 [74].
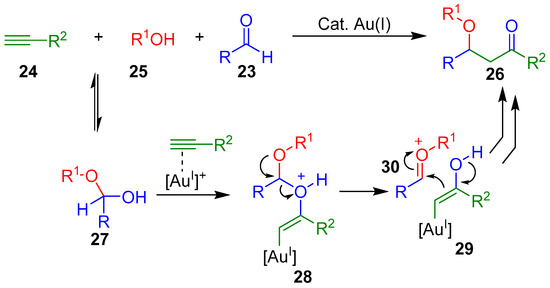
Scheme 11.
Previously proposed mechanism for Au(I)-catalyzed synthesis of β-alkoxyketones 26 via oxyauration of hemiacetals.
2.6. Multicomponent Oxidative Oxyarylation of Alkenes
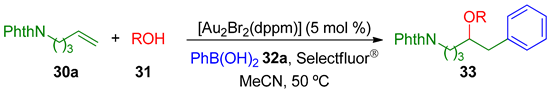
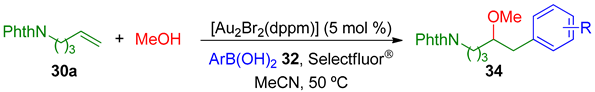
Toste’s group reported the first example of a three-component gold-catalyzed oxidative oxyarylation of alkenes, in which alcohols, carboxylic acids, and even water were employed as nucleophiles in this pioneering protocol [75].
The desired ethers 33 were obtained in moderate to high yield when using several primary and secondary alcohols and even acids 31, 5-phthalimidopentene 30a and phenylboronic acid 32a (Table 1).

Table 1.
Alkoxy- and acyloxyarylation using different alcohols 31.
With these results the authors also explored the variation of the arylboronic acid 32, maintaining the alkenylamine 30a and using MeOH as nucleophile. The final products 34 were obtained in good yields (Table 2).

Table 2.
Methoxyarylation with different ArB(OH)2 32.
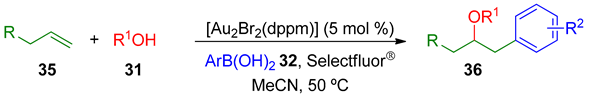
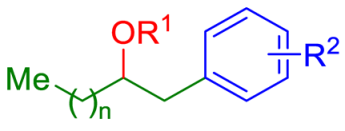
The authors also checked the viability of this protocol using simple alkenes 35, which were also successful as suitable substrates for alkoxy and acyloxyarylation under the optimized conditions (Table 3).

Table 3.
Alkoxy- and acyloxyarylation of simple alkenes 35.
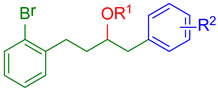
Interestingly, water was also employed as an appropriate nucleophile for the formation of alcohols 38 directly via hydroxyarylation, avoiding the deprotection of the previous alkoxy compounds (Scheme 12).
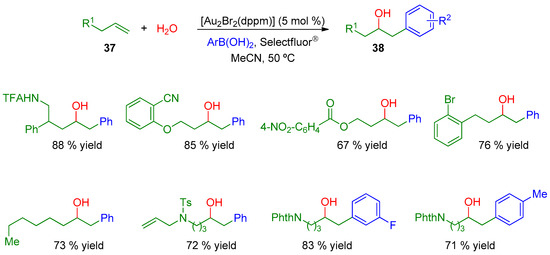
Scheme 12.
Use of water as nucleophile in the hydroxyarylation process (TFAHN: trifluoroacetamide; TsN: toluensulfonamide).
A plausible mechanism for the formation of the alcohols 38 was proposed (Scheme 13). The mechanism is similar to one previously reported by the same authors for a related intramolecular aminoarylation reaction [76]. Hence, first, oxidation of the gold(I) bromide could occur generating a cationic gold(III) species 39, which would activate the alkene toward the nucleophilic attack of H2O. Then, an oxyauration followed by C-C bond formation of 40 with the boronic acid could happen (41). Regeneration of the gold catalyst and release of the FB(OH)2 species would afford the final alcohol 38 (Scheme 13).
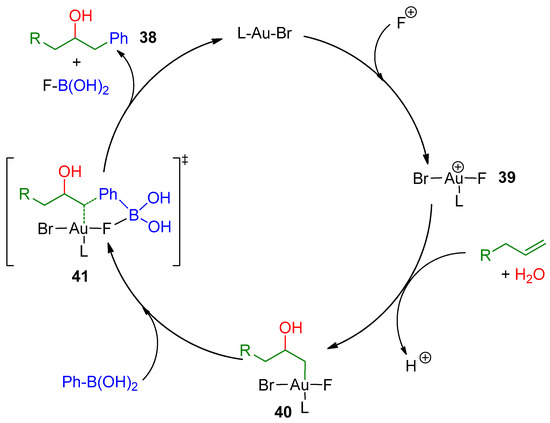
Scheme 13.
Plausible mechanism of the hydroxyarylation process.
2.7. Multicomponent Synthesis of Thiazoloquinolines
The use of microwave irradiation or sonication has also provided further possibilities to perform chemical reactions, tuning the selectivity in organic synthesis [77]. Sadeghzadeh developed a green approach for the four-component synthesis of thiazoloquinolines 46 using gold(III) dipyridine complex immobilized on SBA-15 as nano catalysts at room temperature (Scheme 14) [78], based on a previously reported work by Singh and co-workers [79].
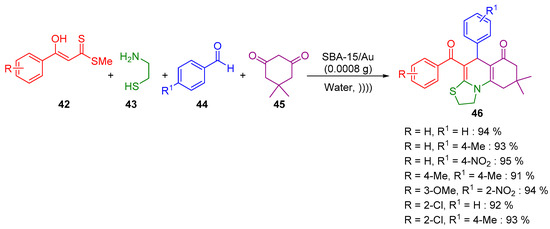
Scheme 14.
Synthesis of thiazoloquinolines 46.
After a broad exploration of the synthesis of gold nanoparticles and the optimization of the reaction conditions, the authors successfully developed an efficient synthesis of thiazoloquinolines 46 in very good yields using ultrasound.
It is worth noting that the gold catalyst could be efficiently recovered from the reaction mixtures. The activity of the recycled catalyst was also evaluated under optimized conditions of temperature, reaction time and the amount of catalyst. The gold catalyst was reused up to seven times. After each catalytic cycle, ICP technique was used to estimate whether the reaction took place at the surface of the SBA-15/Au catalyst. They found that the amount of Au(III) remaining after the seventh cycle was of about 2.1%. This value suggested good stability of the heterogeneous gold catalyst under the reaction conditions employed.
2.8. Multicomponent Synthesis of Spirocycles
A series of compounds containing an indole [80,81,82,83] or isatin [84,85,86] core have been described to possess important biological activities. Therefore, the effect of these structures in the generation of spirocycles could highly enhance biological activity of the resulting compounds [87]. Moreover, the spirooxindole ring is involved in many pharmaceuticals and natural products as shown in Figure 5 [88].
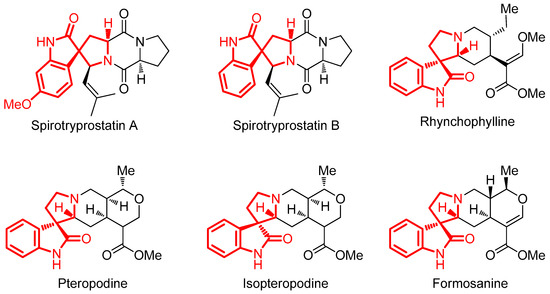
Figure 5.
Spirotryprostatins A and B [89], pteropodine and isopteropodine [90], rhynchophylline [91] and formosanine [92].
The Kidwai group developed a protocol for the efficient synthesis of functionalized spirochromenes 50 and 51 mediated by a gold(III) catalyst [93]. They used PEG 400 as it is an eco-friendly solvent medium, being inexpensive, thermally stable, recyclable and biodegradable (Scheme 15 and Scheme 16) [94]. Several parameters were explored in order to optimize the processes. Thus, the effect of catalyst loading, the presence of various Lewis acids as catalysts, a screening of solvents and the study of temperature was also analyzed. With the optimal reaction conditions, the methodology was firstly evaluated by using different isatins 47, activated methylene compounds 48 and cyclic 1,3-diketones 49 for the synthesis of a series of tetrahydrospiro[chromene-4,3′-indoline] derivatives 50 and 51 (Scheme 14).
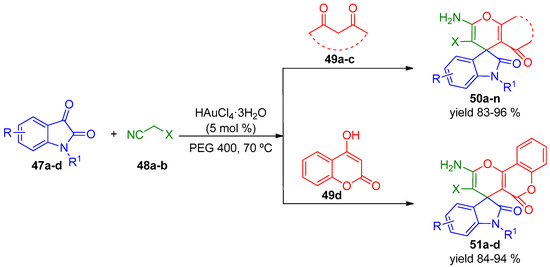
Scheme 15.
HAuCl4·3H2O catalyzed multicomponent synthesis of spirocycles 50 and 51.
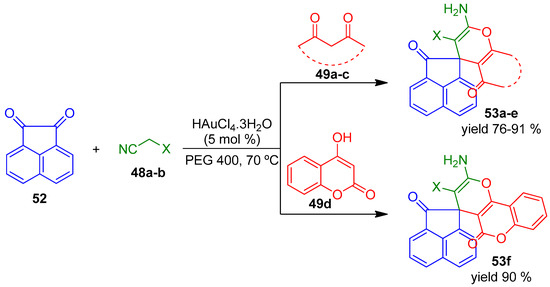
Scheme 16.
HAuCl4·3H2O catalyzed multicomponent synthesis of spirocycles 53.
All compounds 50 and 51 were obtained with very good results (up to 96% yield). The authors extended the protocol to another reactant acenaphthoquinone 52, instead of isatins 47, under the same reaction conditions to prepare a variety of spiroacenaphthyleneone derivatives 53a–f with excellent yields (Scheme 16).
To explain the synthesis of spirooxindole derivatives 50 and 51 two plausible pathways were proposed based on previous studies [95] (Scheme 17).
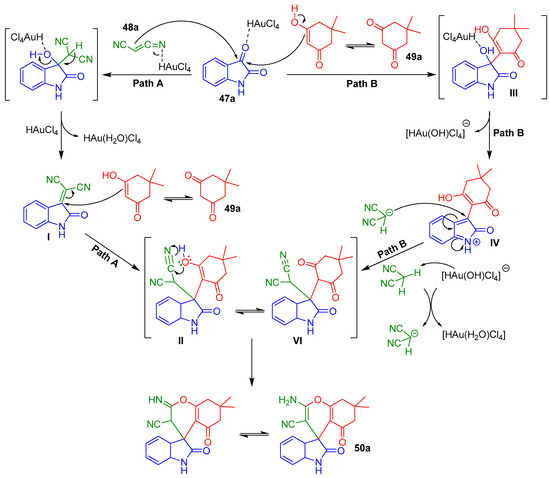
Scheme 17.
Plausible pathways for the synthesis of spirooxindole 50a.
Path A would start with a Knoevenagel condensation between isatin 47a and malononitrile 48a catalyzed by HAuCl4. This step would give rise to isatylidene malononitrile I. In a second step dimedone 49a would attack to Knoevenagel adduct I via Michael addition producing intermediate II (path A). In contrast, path B would start with the attack of the dimedone 49a to the isatin 47a coordinated with HAuCl4 to afford the aldol adduct III followed by a subsequent dehydration of III and nucleophilic attack of 48a to afford the intermediate VI. In both reaction pathways, the intermediate II would involve the cycloaddition between the hydroxyl group and the cyano moiety to form the desired spirooxindole 50a (Scheme 17, path A and path B).
The recyclability of PEG 400 was also explored to proof the sustainability of the solvent. The solvent could be recycled with minimum loss and decomposition during three runs. The recycled PEG does not change in its reactivity but approximately 5% weight loss of PEG was observed after each completed cycle. However, the recyclability of the catalyst after the extraction of the product was accompanied by considerable leaching of the gold(III) chloride catalyst, avoiding its recyclability.
2.9. Multicomponent Synthesis of Butenolides
The butenolide scaffold is also found in many natural products [96]. Moreover, they can exhibit interesting biological activities as antibiotic, antifungal, antifouling, or anticancer, among other properties [97,98,99,100]. In 2010, Ji and co-workers developed a gold-catalyzed three-component tandem approach between commercially available alkynes, amines and glyoxylic acid for the generation of two types of butenolides 57 and 59 (Scheme 18 and Scheme 19) [101].
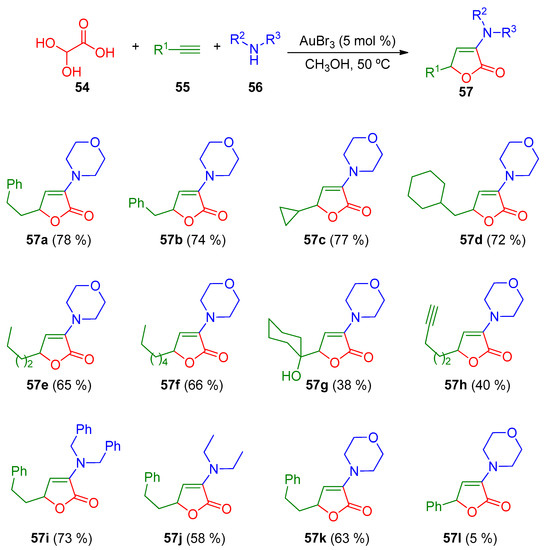
Scheme 18.
Gold-catalyzed multicomponent synthesis of butenolides 57.
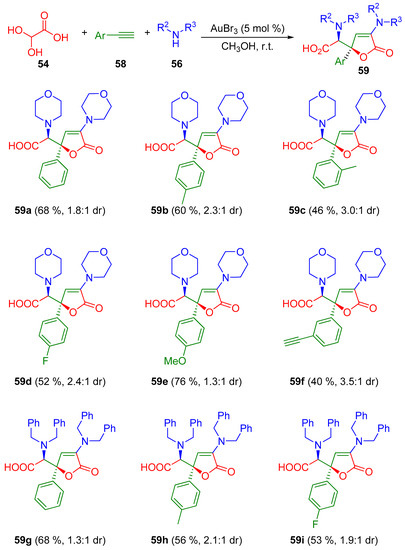
Scheme 19.
Gold-catalyzed tandem process for the synthesis of butenolides 59.
The authors proposed a plausible reaction mechanism based on their experimental results and previous studies (Scheme 20) [102,103,104]. The mechanism is suggested to be a tandem reaction involving two catalytic cycles (I and II). Firstly, in cycle I a gold-catalyzed three component coupling reaction between an alkyne, an amine, and glyoxylic acid occurs to give intermediate A. Next, gold-promoted endo-dig cyclization of A giving rise to intermediate B. Active intermediate B is subsequently transformed to butenolides 57 or 59 via electrophilic trapping along with a 1,2-hydride shift pathway or deprotonation-protonation sequence (Scheme 20). This gold-catalyzed multicomponent tandem protocol provides a valuable synthetic route to butenolides, expanding the area of gold catalysis.
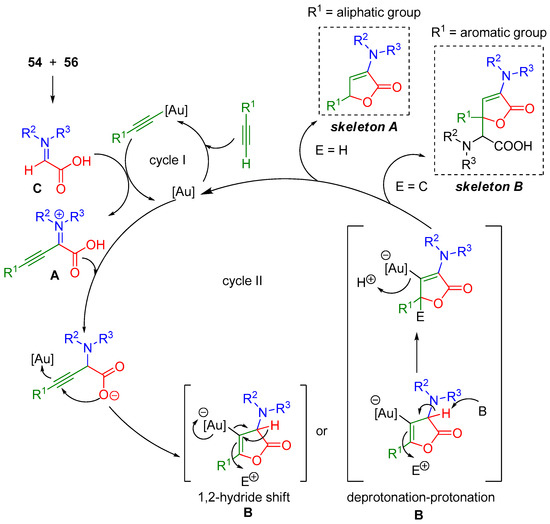
Scheme 20.
Catalytic cycle for the synthesis of butenolides 57 and 59.
3. Conclusions
The preparation of complex architectures has inspired the search for new methods and new processes in organic synthesis. In this sense, multicomponent reactions have become interesting protocols to achieve such molecular diversity and complexity. This objective, joined to the interest in the chemistry of gold, has allowed the synthesis of model structures using new efficient approaches. This review has illustrated these important and scarce examples in order to give a general vision related to the application and scope of gold catalysts in multicomponent reactions, until now dominated by the A3 coupling reaction. However, here it has been demonstrated that new processes have been developed, for which there is a preponderance of alkynes, carbonyls and amines as components for the coupling reactions. The plausible catalytic cycles to understand the role of gold catalyst in addition of each reagent involved have also been discussed in this work, and they clearly state the high Lewis acid capacity of gold catalysts to activate alkyne or carbonyl groups. The increasing interest in both gold catalysis and the development of more sustainable an economical procedures, make that a significant progress in the next few years is expected, with the use of gold catalysts for the discovery of new coupling reactions involving novel components that will allow the preparation of more complex scaffolds with potentially interesting properties.
Author Contributions
All authors wrote the paper; and all authors read and approved the final manuscript.
Funding
Ministerio de Economía y Competitividad (MINECO/FEDER CTQ2016-75816-C2-1-P and CTQ2017-88091-P). Gobierno de Aragón-Fondo Social Europeo (E07_17R).
Conflicts of Interest
The authors declare no conflict of interest.
References and Note
- Bienaymé, H.; Hulme, C.; Oddon, G.; Schmitt, P. Maximizing synthetic efficiency: Multi-component transformations lead the way. Chem. Eur. J. 2000, 6, 3321–3329. [Google Scholar] [CrossRef]
- Ruijter, E.; Scheffelaar, R.; Orru, R.V.A. Multicomponent reaction design in the quest for molecular complexity and diversity. Angew. Chem. Int. Ed. 2011, 50, 6234–6246. [Google Scholar] [CrossRef] [PubMed]
- Touré, B.B.; Hal, D.G. Natural product synthesis using multicomponent reaction strategies. Chem. Rev. 2009, 109, 4439–4486. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Bienaymé, H. (Eds.) Multicomponent Reactions; Wiley-VCH: Weinheim, Germany, 2005. [Google Scholar]
- Herrera, R.P.; Marqués-López, E. (Eds.) Multicomponent Reactions: Concepts and Applications for Design and Synthesis; John Wiley & Sons: Hoboken, NJ, USA, 2015. [Google Scholar]
- Weber, L. The application of multi-component reactions in drug discovery. Curr. Med. Chem. 2002, 9, 2085–2093. [Google Scholar] [CrossRef] [PubMed]
- Hulme, C.; Gore, V. Multi-component reactions: Emerging chemistry in drug discovery “from xylocain to crixivan”. Curr. Med. Chem. 2003, 10, 51–80. [Google Scholar] [CrossRef] [PubMed]
- Dömling, A. Recent developments in isocyanide based multicomponent reactions in applied chemistry. Chem. Rev. 2006, 106, 17–89. [Google Scholar] [CrossRef] [PubMed]
- Akritopoulou-Zanze, I. Isocyanide-based multicomponent reactions in drug discovery. Curr. Opin. Chem. Biol. 2008, 12, 324–331. [Google Scholar] [CrossRef] [PubMed]
- Dömling, A.; Wang, W.; Wang, K. Chemistry and biology of multicomponent reactions. Chem. Rev. 2012, 112, 3083–3135. [Google Scholar] [CrossRef] [PubMed]
- Hashmi, A.S.K. Homogeneous gold catalysis beyond assumptions and proposals-characterized intermediates. Angew. Chem. Int. Ed. 2010, 49, 5232–5241. [Google Scholar] [CrossRef] [PubMed]
- Rudolph, M.; Hashmi, A.S.K. Heterocycles from gold catalysis. Chem. Commun. 2011, 47, 6536–6544. [Google Scholar] [CrossRef] [PubMed]
- Hashmi, A.S.K. Gold-catalyzed synthesis of N,O-heterocycles. Pure Appl. Chem. 2010, 82, 657–668. [Google Scholar] [CrossRef]
- Shapiro, N.D.; Toste, F.D. A reactivity-driven approach to the discovery and development of gold-catalyzed organic reactions. Synlett 2010, 675–691. [Google Scholar]
- Gorin, D.J.; Toste, F.D. Relativistic effects in homogeneous gold catalysis. Nature 2007, 446, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Rudolph, M.; Hashmi, A.S.K. Gold catalysis in total synthesis. Chem. Soc. Rev. 2008, 37, 1766–1775. [Google Scholar]
- Rudolph, M.; Hashmi, A.S.K. Gold catalysis in total synthesis—An update. Chem. Soc. Rev. 2012, 41, 2448–2462. [Google Scholar] [CrossRef] [PubMed]
- Schmidbaur, H.; Schier, A. Gold(III) compounds for homogeneous catalysis: Preparation, reaction conditions, and scope of application. Arab. J. Sci. Eng. 2012, 37, 1187–1225. [Google Scholar] [CrossRef]
- Hashmi, A.S.K. Gold-catalyzed organic reactions. Chem. Rev. 2007, 107, 3180–3211. [Google Scholar] [CrossRef] [PubMed]
- Krause, N.; Winter, C. Gold-catalyzed nucleophilic cyclization of functionalized allenes: A powerful access to carbo- and heterocycles. Chem. Rev. 2011, 111, 1994–2009. [Google Scholar] [CrossRef] [PubMed]
- Montanel-Pérez, S.; Herrera, R.P.; Laguna, A.; Villacampa, M.D.; Gimeno, M.C. The fluxional amine gold(III) complex as an excellent catalyst and precursor of biologically active acyclic carbenes. Dalton Trans. 2015, 44, 9052–9062. [Google Scholar] [CrossRef] [PubMed]
- Aliaga-Lavrijsen, M.; Herrera, R.P.; Villacampa, M.D.; Gimeno, M.C. Efficient Gold(I) Acyclic Diaminocarbenes for the Synthesis of Propargylamines and Indolizines. ACS Omega 2018, 3, 9805–9813. [Google Scholar] [CrossRef]
- Abbiati, G.; Rossi, E. Silver and gold-catalyzed multicomponent reactions. Beilstein J. Org. Chem. 2014, 10, 481–513. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Han, Z.-Y.; Luo, H.-W.; Gong, L.-Z. Highly enantioselective relay catalysis in the three-component reaction for direct construction of structurally complex heterocycles. Org. Lett. 2010, 12, 2266–2269. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.; Frey, G.D.; Kinjo, R.; Donnadieu, B.; Bertrand, G. Synthesis of a simplified version of stable bulky and rigid cyclic (alkyl)(amino)carbenes, and catalytic activity of the ensuing gold(I) complex in the three-component preparation of 1,2-dihydroquinoline derivatives. J. Am. Chem. Soc. 2009, 131, 8690–8696. [Google Scholar] [CrossRef] [PubMed]
- Tian, G.-Q.; Shi, M. Gold(I)-catalyzed three-component additions of 2-(arylmethylene)cyclopropylcarbinols, terminal arynes, and alcohols: An efficient access to 3-oxabicyclo[3.1.0]hexanes. Org. Lett. 2007, 9, 4917–4920. [Google Scholar] [CrossRef] [PubMed]
- Berrichi, A.; Bachir, R.; Benabdallah, M.; Choukchou-Braham, N. Supported nano gold catalyzed three-component coupling reactions of amines, dichloromethane and terminal alkynes (AHA). Tetrahedron Lett. 2015, 56, 1302–1306. [Google Scholar] [CrossRef]
- Shaabani, A.; Hezarkhani, Z.; Nejad, M.K. AuCu and AgCu bimetallic nanoparticles supported on guanidine-modified reduced graphene oxide nanosheets as catalysts in the reduction of nitroarenes: Tandem synthesis of benzo[b][1,4]diazepine derivatives. RSC Adv. 2016, 6, 30247–30257. [Google Scholar] [CrossRef]
- Ali, F.; Khan, K.M.; Salar, U.; Iqbal, S.; Taha, M.; Ismail, N.H.; Perveen, S.; Wadood, A.; Ghufrane, M.; Ali, B. Dihydropyrimidones: As novel class of β-glucuronidase inhibitors. Bioorg. Med. Chem. 2016, 24, 3624–3635. [Google Scholar] [CrossRef] [PubMed]
- Xuea, H.; Zhao, Y.; Wu, H.; Wang, Z.; Yang, B.; Wei, Y.; Wang, Z.; Tao, L. Multicomponent combinatorial polymerization via the Biginelli reaction. J. Am. Chem. Soc. 2016, 138, 8690–8693. [Google Scholar] [CrossRef] [PubMed]
- Dhumaskar, K.L.; Meena, S.N.; Ghadi, S.C.; Tilve, S.G. Graphite catalyzed solvent free synthesis of dihydropyrimidin-2(1H)-ones/thiones and their antidiabetic activity. Bioorg. Med. Chem. Lett. 2014, 24, 2897–2899. [Google Scholar] [CrossRef] [PubMed]
- Biginelli, P. Aldehyde-urea derivatives of aceto- and oxaloacetic acids. Gazz. Chim. Ital. 1893, 23, 360–413. [Google Scholar]
- Kappe, C.O. Recent advances in the Biginelli dihydropyrimidine synthesis. New tricks from an old dog. Acc. Chem. Res. 2000, 33, 879–888. [Google Scholar] [CrossRef] [PubMed]
- Marqués-López, E.; Herrera, R.P. Biginelli Multicomponent Reactions. In Multicomponent Reactions. Concepts and Applications for Design and Synthesis; Herrera, R.P., Marqués-López, E., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2015; pp. 306–330. [Google Scholar]
- Nguyen, N.-H.T.; Nguyen, P.-P.T.; Nguyen, T.-D.T.; Tran, M.-N.T.; Huynh, T.-N.T.; Tran, P.H. Au Nanorod: An efficient catalyst for one-pot synthesis of 3,4-dihydropyrimidin-2(1H)-ones via the multicomponent Biginelli reaction. ChemistrySelect 2017, 2, 3932–3936. [Google Scholar] [CrossRef]
- Hantzsch, A. Condensationprodukte aus Aldehydammoniak und ketonartigen Verbindungen. Ber. Dtsch. Chem. Ges. 1881, 14, 1637–1638. [Google Scholar] [CrossRef]
- Ioan, P.; Carosati, E.; Micucci, M.; Cruciani, G.; Broccatelli, F.; Zhorov, B.S.; Chiarini, A.; Budriesi, R. 1,4-Dihydropyridine scaffold in medicinal chemistry, the story so far and perspectives (part 1): Action in ion channels and GPCRs. Curr. Med. Chem. 2011, 18, 4901–4922. [Google Scholar] [CrossRef] [PubMed]
- Carosati, E.; Ioan, P.; Micucci, M.; Broccatelli, F.; Cruciani, G.; Zhorov, B.S.; Chiarini, A.; Budriesi, R. 1,4-Dihydropyridine scaffold in medicinal chemistry, the story so far and perspectives (part 2): Action in other targets and antitargets. Curr. Med. Chem. 2012, 19, 4306–4323. [Google Scholar] [CrossRef] [PubMed]
- Edraki, N.; Mehdipour, A.R.; Khoshneviszadeh, M.; Miri, R. Dihydropyridines: Evaluation of their current and future pharmacological applications. Drug. Discov. Today 2009, 14, 1058–1066. [Google Scholar] [CrossRef] [PubMed]
- Wan, J.-P.; Liu, Y. Recent advances in new multicomponent synthesis of structurally diversified 1,4-dihydropyridines. RSC Adv. 2012, 2, 9763–9777. [Google Scholar] [CrossRef]
- Pham, H.T.; Chataigner, I.; Renaud, J.-L. New approaches to nitrogen containing heterocycles: Enantioselective organocatalyzed synthesis of dihydropyridines (DHP’s), quinolizidine derivatives and dihydropyrimidines (DHPM’s). Curr. Org. Chem. 2012, 16, 1754–1775. [Google Scholar] [CrossRef]
- Auria-Luna, F.; Marqués-López, E.; Mohammadi, S.; Heiran, R.; Herrera, R.P. New Organocatalytic Asymmetric Synthesis of Highly Substituted Chiral 2-Oxospiro-[indole-3,4′-(1′,4′-dihydropyridine)] Derivatives. Molecules 2015, 20, 15807–15826. [Google Scholar] [CrossRef] [PubMed]
- Auria-Luna, F.; Marqués-López, E.; Herrera, R.P. Organocatalytic enantioselective synthesis of 1,4-dihydropyridines. Adv. Synth. Catal. 2017, 359, 2161–2175. [Google Scholar] [CrossRef]
- Auria-Luna, F.; Marqués-López, E.; Gimeno, M.C.; Heiran, R.; Mohammadi, S.; Herrera, R.P. Asymmetric organocatalytic synthesis of substituted chiral 1,4-dihydropyridine derivatives. J. Org. Chem. 2017, 82, 5516–5523. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Chen, H.; Zhao, H.; Cao, H.; Li, Y.; Liu, Q. Gold-catalyzed multicomponent reaction: Facile strategy for the synthesis of N-substituted 1,4-dihydropyridines by using activated alkynes, aldehydes, and methanamine. Eur. J. Org. Chem. 2013, 7300–7304. [Google Scholar] [CrossRef]
- Jayasinghe, L.; Jayasooriya, C.-P.; Hara, N.; Fujimoto, Y. A pyridine ring-containing ecdysteroid from Diploclisia glaucescens. Tetrahedron Lett. 2003, 44, 8769–8771. [Google Scholar] [CrossRef]
- Henry, G.D. De novo synthesis of substituted pyridines. Tetrahedron 2004, 60, 6043–6061. [Google Scholar] [CrossRef]
- Kubota, T.; Nishi, T.; Fukushi, E.; Kawabata, J.; Fromont, J.; Kobayashi, J. Nakinadine A, a novel bis-pyridine alkaloid with a β-amino acid moiety from sponge Amphimedon sp. Tetrahedron Lett. 2007, 48, 4983–4985. [Google Scholar] [CrossRef]
- Murata, T.; Shimada, M.; Sakakibara, S.; Yoshino, T.; Kadono, H.; Masuda, T.; Shimazaki, M.; Shintani, T.; Fuchikami, K.; Sakai, K.; et al. Discovery of novel and selective IKK-β serine-threonine protein kinase inhibitors. Part 1. Bioorg. Med. Chem. Lett. 2003, 13, 913–918. [Google Scholar] [CrossRef]
- Deng, J.; Sanchez, T.; Al-Mawsawi, L.Q.; Dayam, R.; Yunes, R.A.; Garofalo, A.; Bolger, M.B.; Neamati, N. Discovery of structurally diverse HIV-1 integrase inhibitors based on a chalcone pharmacophore. Bioorg. Med. Chem. 2007, 15, 4985–5002. [Google Scholar] [CrossRef] [PubMed]
- Pagadala, R.; Maddila, S.; Moodley, V.; van Zyl, W.E.; Jonnalagadda, S.B. An efficient method for the multicomponent synthesis of multisubstituted pyridines, a rapid procedure using Au/MgO as the catalyst. Tetrahedron Lett. 2014, 55, 4006–4010. [Google Scholar] [CrossRef]
- Jin, Z.; Li, Z.; Huang, R. Muscarine, imidazole, oxazole, thiazole, Amaryllidaceae and Sceletium alkaloids. Nat. Prod. Rep. 2002, 19, 454–476. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z. Muscarine, imidaozle, oxazole and thiazole alkaloids. Nat. Prod. Rep. 2003, 20, 584–605. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z. Muscarine, imidaozle, oxazole and thiazole alkaloids. Nat. Prod. Rep. 2005, 22, 196–229. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z. Muscarine, imidaozle, oxazole and thiazole alkaloids. Nat. Prod. Rep. 2006, 23, 464–496. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z. Muscarine, imidaozle, oxazole and thiazole alkaloids. Nat. Prod. Rep. 2009, 26, 382–445. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z. Muscarine, imidaozle, oxazole and thiazole alkaloids. Nat. Prod. Rep. 2011, 28, 1143–1191. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z. Muscarine, imidaozle, oxazole and thiazole alkaloids. Nat. Prod. Rep. 2013, 30, 869–915. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z. Muscarine, imidaozle, oxazole and thiazole alkaloids. Nat. Prod. Rep. 2016, 33, 1268–1317. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.-Z.; Zhao, Z.-L.; Zhou, C.-H. Recent advance in oxazole-based medicinal chemistry. Eur. J. Med. Chem. 2018, 144, 444–492. [Google Scholar] [CrossRef] [PubMed]
- Wachenfeldt, Hv.; Röse, P.; Paulsen, F.; Loganathan, N.; Strand, D. Catalytic three-component domino reaction for the preparation of trisubstituted oxazoles. Chem. Eur. J. 2013, 19, 7982–7988. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Li, C.; Zhang, L. An efficient [2 + 2 + 1] synthesis of 2,5-disubstituted oxazoles via gold-catalyzed intermolecular alkyne oxidation. J. Am. Chem. Soc. 2011, 133, 8482–8485. [Google Scholar] [CrossRef] [PubMed]
- Rassadin, V.A.; Boyarskiy, V.P.; Kukushkin, V.Y. Facile gold-catalyzed heterocyclization of terminal alkynes and cyanamides leading to substituted 2-amino-1,3-oxazoles. Org. Lett. 2015, 17, 3502–3505. [Google Scholar] [CrossRef] [PubMed]
- Querard, P.; Girard, S.A.; Uhlig, N.; Li, C.-J. Gold-catalyzed tandem reactions of amide–aldehyde–alkyne coupling and cyclization synthesis of 2,4,5-trisubstituted oxazoles. Chem. Sci. 2015, 6, 7332–7335. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Zhang, R.; Yi, F.; Cai, M. A heterogeneous gold(I)-catalyzed [2 + 2 + 1] annulation of terminal alkynes, nitriles, and oxygen atoms leading to 2,5-disubstituted oxazoles. J. Org. Chem. 2017, 82, 5204–5211. [Google Scholar] [CrossRef] [PubMed]
- The authors could not exclude the possibility that an acid or gold species originating from thermal decomposition of the catalyst could catalyze the isomerization step. See Ref. [61].
- Von Wachenfeldt, H.; Polukeev, A.V.; Loganathan, N.; Paulsen, F.; Röse, P.; Garreau, M.; Wendt, O.F.; Strand, D. Cyclometallated gold(III) aryl-pyridine complexes as efficient catalysts for three-component synthesis of substituted oxazoles. Dalton Trans. 2015, 44, 5347–5353. [Google Scholar] [CrossRef] [PubMed]
- Denmark, S.E.; Stavanger, R.A. Asymmetric catalysis of aldol reactions with chiral Lewis bases. Acc. Chem. Res. 2000, 33, 432–440. [Google Scholar] [CrossRef] [PubMed]
- Saito, S.; Yamamoto, H. Design of acid-base catalysis for the asymmetric direct aldol reaction. Acc. Chem. Res. 2004, 37, 570–579. [Google Scholar] [CrossRef] [PubMed]
- Paterson, I.; Chen, D.Y.-K.; Coster, M.J.; Aceña, J.L.; Bach, J.; Gibson, K.R.; Keown, L.E.; Oballa, R.M.; Trieselmann, T.; Wallace, D.J.; et al. Stereocontrolled total synthesis of (+)-Altohyrtin A/Spongistatin 1. Angew. Chem. Int. Ed. 2001, 40, 4055–4060. [Google Scholar] [CrossRef]
- Calter, M.A.; Liao, W. First total synthesis of a natural product containing a chiral, β-diketone: Synthesis and stereochemical reassignment of siphonarienedione and siphonarienolone. J. Am. Chem. Soc. 2002, 124, 13127–13129. [Google Scholar] [CrossRef] [PubMed]
- Nicolaou, K.C.; Ritzen, A.; Namoto, K. Recent developments in the chemistry, biology and medicine of the epothilones. Chem. Commun. 2001, 17, 1523–1535. [Google Scholar] [CrossRef]
- Schultz, D.M.; Babij, N.R.; Wolfe, J.P. Intermolecular gold(I)-catalyzed alkyne carboalkoxylation reactions for the multicomponent assembly of β-alkoxy ketones. Adv. Synth. Catal. 2012, 354, 3451–3455. [Google Scholar] [CrossRef]
- Zhang, M.; Wang, Y.; Yang, Y.; Hu, X. An alternative approach to direct aldol reaction based on gold-catalyzed methoxyl transfer. Adv. Synth. Catal. 2012, 354, 981–985. [Google Scholar] [CrossRef]
- Melhado, A.D.; Brenzovich, W.E., Jr.; Lackner, A.D.; Toste, F.D. Gold-catalyzed three-component coupling: Oxidative oxyarylation of alkenes. J. Am. Chem. Soc. 2010, 132, 8885–8887. [Google Scholar] [CrossRef] [PubMed]
- Brenzovich, W.E. Jr.; Benitez, D.; Lackner, A.D.; Shunatona, H.P.; Tkatchouk, E.; Goddard, W.A., III; Toste, F.D. Gold-catalyzed intramolecular aminoarylation of alkenes: C-C bond formation through bimolecular reductive elimination. Angew. Chem. Int. Ed. 2010, 49, 5519–5522. [Google Scholar] [CrossRef] [PubMed]
- Lidström, P.; Tierney, J.; Wathey, B.; Westman, J. Microwave assisted organic synthesis–A review. Tetrahedron 2001, 57, 9225–9283. [Google Scholar] [CrossRef]
- Sadeghzadeh, S.M. Ultrasound-promoted green approach for the synthesis of thiazoloquinolines using gold(III) dipyridine complex immobilized on SBA-15 as nano catalysts at room temperature. RSC Adv. 2015, 5, 68947–68952. [Google Scholar] [CrossRef]
- Nagaraju, A.; Ramulu, B.J.; Shukla, G.; Srivastava, A.; Verma, G.K.; Raghuvanshi, K.; Singh, M.S. Catalyst-free one-pot four-component domino reactions in water–PEG-400: Highly efficient and convergent approach to thiazoloquinoline scaffolds. Green Chem. 2015, 17, 950–958. [Google Scholar] [CrossRef]
- Wu, Y.-J. New indole-containing medicinal compounds. Top. Heterocycl. Chem. 2010, 26, 1–29. [Google Scholar]
- Bronner, S.M.; Im, G.Y.J.; Garg, N.K. Heterocycles in Natural Product Synthesis; Majumdar, K.C., Chattopadhyay, S.K., Eds.; Wiley-VCH, Verlag GmbH & Co., KGaA: Weinheim, Germany, 2011; pp. 221–265. [Google Scholar]
- Kaushik, N.K.; Kaushik, N.; Attri, P.; Kumar, N.; Kim, C.H.; Verma, A.K.; Choi, E.H. Biomedical importance of indoles. Molecules 2013, 18, 6620–6662. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.-Z.; Chen, Q.; Yang, G.-F. A review on recent developments of indole-containing antiviral agents. Eur. J. Med. Chem. 2015, 89, 421–441. [Google Scholar] [CrossRef] [PubMed]
- Ohshima, T. Enantioselective total syntheses of several bioactive natural products based on the development of practical asymmetric catalysis. Chem. Pharm. Bull. 2004, 52, 1031–1052. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.; Kumar, P.; Pathak, D. Biological importance of the indole nucleus in recent years: A comprehensive review. J. Heterocyclic. Chem. 2010, 47, 491–502. [Google Scholar] [CrossRef]
- Biswal, S.; Sahoo, U.; Sethy, S.; Kumar, H.K.S.; Banerjee, M. Indole: The molecule of diverse biological activities. Asian J. Pharm. Clin. Res. 2012, 5, 1–6. [Google Scholar]
- Abdel-Rahman, A.H.; Keshk, E.M.; Hanna, M.A.; El-Bady, Sh.M. Synthesis and evaluation of some new spiro indoline-based heterocycles as potentially active antimicrobial agents. Bioorg. Med. Chem. 2004, 12, 2483–2488. [Google Scholar] [CrossRef] [PubMed]
- Williams, R.M.; Cox, R.J. Paraherquamides, Brevianamides, and Asperparalines: Laboratory synthesis and biosynthesis. An interim report. Acc. Chem. Res. 2003, 36, 127–139. [Google Scholar] [CrossRef] [PubMed]
- Cui, C.B.; Kakeya, H.; Okada, G.; Ubukata, M.; Takahashi, I.; Isono, K.; Osada, H. Tryprostatins A and B, novel mammalian cell cycle inhibitors produced by Aspergillus fumigatus. J. Antibiot. 1995, 48, 1382–1384. [Google Scholar] [CrossRef] [PubMed]
- Kang, T.-H.; Matsumoto, K.; Tohda, M.; Murakami, Y.; Takayama, H.; Kitajima, M.; Aimi, N.; Watanabe, H. Pteropodine and isopteropodine positively modulate the function of rat muscarinic M1 and 5-HT2 receptors expressed in Xenopus oocyte. Eur. J. Pharmacol. 2002, 444, 39–45. [Google Scholar] [CrossRef]
- Seaton, J.C.; Marion, L. The structure of rhyncophylline. Can. J. Chem. 1957, 35, 1102–1108. [Google Scholar] [CrossRef]
- Shamma, M.; Shine, R.J.; Kompiš, I.; Sticzay, T.; Morsingh, F.; Poisson, J.; Pousset, J.-L. The stereochemistry of the pentacyclic oxindole alkaloids. J. Am. Chem. Soc. 1967, 89, 1739–1740. [Google Scholar] [CrossRef] [PubMed]
- Bonsignore, L.; Loy, G.; Secci, D.; Calignano, A. Synthesis and pharmacological activity of 2-oxo-(2H) 1-benzopyran-3-carboxamide derivatives. Eur. J. Med. Chem. 1993, 28, 517–520. [Google Scholar] [CrossRef]
- Kidwai, M.; Jahan, A.; Mishra, N.K. Gold(III) chloride (HAuCl4·3H2O) in PEG: A new and efficient catalytic system for the synthesis of functionalized spirochromenes. App. Catal. A Gen. 2013, 425–426, 35–43. [Google Scholar] [CrossRef]
- Liang, B.; Kalidindi, S.; Porco, J.A. Jr.; Stephenson, C.R.J. Three-component synthesis of spirooxindoles. Org. Lett. 2010, 12, 572–575. [Google Scholar] [CrossRef] [PubMed]
- Slaughter, J.C. The naturally occurring furanones: Formation and function from pheromone to food. Biol. Rev. 1999, 74, 259–276. [Google Scholar] [CrossRef]
- Braun, M.; Hohmann, A.; Rahematpura, J.; Bühne, C.; Grimme, S. Synthesis and determination of the absolute configuration of Fugomycin and Desoxyfugomycin: CD spectroscopy and fungicidal activity of butenolides. Chem. Eur. J. 2004, 10, 4584–4593. [Google Scholar] [CrossRef] [PubMed]
- Mansoor, T.A.; Hong, J.; Lee, C.-O.; Sim, C.J.; Im, K.S.; Lee, D.S.; Jung, J.H. New cytotoxic metabolites from a marine sponge Homaxinella sp. J. Nat. Prod. 2004, 67, 721–724. [Google Scholar] [CrossRef] [PubMed]
- Husain, A.; Khan, M.S.Y.; Hasan, S.M.; Alam, M.M. 2-Arylidene-4-(4-phenoxy-phenyl)but-3-en-4-olides: Synthesis, reactions and biological activity. Eur. J. Med. Chem. 2005, 40, 1394–1404. [Google Scholar] [CrossRef] [PubMed]
- Wright, A.D.; de Nys, R.; Angerhofer, C.K.; Pezzuto, J.M.; Gurrath, M. Biological activities and 3D QSAR studies of a series of Delisea pulchra (cf. fimbriata) derived natural products. J. Nat. Prod. 2006, 69, 1180–1187. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Cheng, M.; Hu, X.; Li, B.-G.; Ji, J.-X. Gold-catalyzed three-component tandem process: An efficient and facile assembly of complex butenolides from alkynes, amines, and glyoxylic acid. J. Am. Chem. Soc. 2010, 132, 7256–7257. [Google Scholar] [CrossRef] [PubMed]
- Genin, E.; Tullec, P.Y.; Antoniotti, S.; Brancour, C.; Genêt, J.-P.; Michelet, V. Room temperature Au(I)-catalyzed exo-selective cycloisomerization of acetylenic acids: an entry to functionalized γ-lactones. J. Am. Chem. Soc. 2006, 128, 3112–3113. [Google Scholar] [CrossRef] [PubMed]
- Harkat, H.; Weibel, J.-M.; Pale, P. A mild access to γ- or δ-alkylidene lactones through gold catalysis. Tetrahedron Lett. 2006, 47, 6273–6276. [Google Scholar] [CrossRef]
- Genin, E.; Toullec, P.Y.; Marie, P.; Antoniotti, S.; Brancour, C.; Genêt, J.-P.; Michelet, V. Gold catalysis in organic synthesis: Efficient intramolecular cyclization of γ-acetylenic carboxylic acids to 5-exo-alkylidene-butyrolactones. ARKIVOC 2007, 2007, 67–78. [Google Scholar]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).