- Review
Non-coding RNAs (ncRNAs) are conserved in the genome of cells across the three domains of life. They comprise a diverse group that are particularly prominent in metazoans where they provide a crucial interface between genes and proteins, participating in key cellular processes at different levels: from control of DNA transcription to modulation of messenger RNA stability to modification of protein activity. The interactions of ncRNAs with one another as well as with other RNAs, DNA and proteins form the basis of a genome-wide regulatory network (GRN). Because of the mutual influence of its components on each other, the GRN is a dynamic system. Further, the GRN imposes constraints on which genes are expressed and when, leading to specific gene-expression patterns or transcriptomes. The configurations of the activities of all gene loci represent self-stabilizing cell states, referred to as “attractor” states, each of which corresponds to a distinct cell type. The cancer cell is also an attractor state that arises from a change in the topography of the epigenetic landscape caused by dysregulation of the GRN. It is proposed that the transition to a neoplastic attractor state is caused by ncRNA alterations, while subsequent somatic mutations of oncogenes and tumor suppressor genes drive cell proliferation and clonal expansion.
2 March 2026



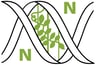
![Pathways without (A) and with (B) feedback. Both systems are modeled in power-law format. Equations for pathway A: Ẋ = Input − E·X0.5; Ẏ = E·X0.5 − Y0.5; Ż = Y0.5 − Z0.5. For pathway B, the following equations are added: TḞ = Zp − TF0.5; Ġ = TF0.5 − G0.5; Ė = G0.5 − E0.5. (For further details, refer to Reference [60].)](https://mdpi-res.com/cdn-cgi/image/w=470,h=317/https://mdpi-res.com/genes/genes-17-00304/article_deploy/html/images/genes-17-00304-g001-550.jpg)