-
 Extemporaneous Formulations for Pediatric Patients: Global Necessities, Challenges and Opportunities
Extemporaneous Formulations for Pediatric Patients: Global Necessities, Challenges and Opportunities -
 Delivery of siRNA and Chloroquine Through an Aspherical, Nanostructured Microparticle for Passive Targeting of Alveolar Macrophages and Inhibition of Local TNF-α Secretion
Delivery of siRNA and Chloroquine Through an Aspherical, Nanostructured Microparticle for Passive Targeting of Alveolar Macrophages and Inhibition of Local TNF-α Secretion -
 Inhalation-Based Nanoparticle Drug Delivery Targeting the Diseased Lower Airways in Idiopathic Pulmonary Fibrosis
Inhalation-Based Nanoparticle Drug Delivery Targeting the Diseased Lower Airways in Idiopathic Pulmonary Fibrosis
Journal Description
Pharmaceutics
Pharmaceutics
is a peer-reviewed, open access journal on the science and technology of pharmaceutics and biopharmaceutics, published monthly online by MDPI. The Spanish Society of Pharmaceutics and Pharmaceutical Technology (SEFIG), Pharmaceutical Solid State Research Cluster (PSSRC), Academy of Pharmaceutical Sciences (APS) and Korean Society of Pharmaceutical Sciences and Technology (KSPST) are affiliated with Pharmaceutics and their members receive a discount on the article processing charges.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), PubMed, PMC, Embase, CAPlus / SciFinder, and other databases.
- Journal Rank: JCR - Q1 (Pharmacology and Pharmacy) / CiteScore - Q1 (Pharmaceutical Science)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 15.7 days after submission; acceptance to publication is undertaken in 3.5 days (median values for papers published in this journal in the second half of 2025).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
- Companion journals for Pharmaceutics include: Future Pharmacology, Journal of Pharmaceutical and BioTech Industry and Medicines.
- Journal Clusters-Pharmaceutical Science: Scientia Pharmaceutica, Marine Drugs, Pharmaceuticals, Pharmaceutics, Pharmacy, Future Pharmacology, Pharmacoepidemiology, Drugs and Drug Candidates and Journal of Pharmaceutical and BioTech Industry.
Impact Factor:
5.5 (2024);
5-Year Impact Factor:
5.8 (2024)
Latest Articles
Andrographolide-Loaded Gold Carbon Quantum Dots and Their Doped Derivatives for Enhanced Hydrophilicity in a Drug Delivery System
Pharmaceutics 2026, 18(6), 647; https://doi.org/10.3390/pharmaceutics18060647 (registering DOI) - 24 May 2026
Abstract
Background/Objectives: Andrographolide (ADG) is a plant-derived compound with promising anticancer properties, but its medical use is limited due to poor water solubility and low bioavailability. This study proposes developing a gold-based nanocomposite drug delivery system, using a simplified synthesis method, to improve ADG’s
[...] Read more.
Background/Objectives: Andrographolide (ADG) is a plant-derived compound with promising anticancer properties, but its medical use is limited due to poor water solubility and low bioavailability. This study proposes developing a gold-based nanocomposite drug delivery system, using a simplified synthesis method, to improve ADG’s hydrophilicity and enhance its delivery efficiency. Methods: A one-step method was used to synthesize gold nanocomposites with carbon quantum dots (CBQDs) and doped CBQDs acting as reducing and stabilizing agents. These nanocomposites were then conjugated with ADG and thoroughly characterized using multiple structural and spectroscopic techniques such as X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FTIR), ultraviolet–visible spectroscopy (UV–Vis), transmission electron microscopy (TEM), Raman spectroscopy, and nuclear magnetic resonance (NMR) spectroscopy. Hydrophilicity enhancement was evaluated using NMR-based log P measurements. Biological assessment involved cell viability assays and confocal microscopy studies in PC3 prostate cancer cells, along with the morphological evaluation of human red blood cells. Results: XRD confirmed the formation of crystalline, face-centered cubic gold nanoparticles, while spectroscopic analyses verified successful nanocomposite formation and ADG conjugation. NMR results showed enhanced hydrophilicity of ADG. Biological tests demonstrated that the nanocomposites were compatible with cells. Conclusions: This study presents a straightforward strategy for synthesizing gold-based nanocomposites that enhance the hydrophilicity and delivery potential of andrographolide, supporting their applicability as nanocarrier platforms for anticancer drug delivery.
Full article
(This article belongs to the Special Issue Carbon-Based Nanomaterials for Pharmaceutical Applications)
►
Show Figures
Open AccessArticle
Optimized TELIP, an Echogenic Liposomal Nano-Carrier Loaded with Alteplase for Preclinical Studies
by
Maryam Ranjpour, Brion Frierson, Rebekah Lynn Emerine, Christian Jordan De Vera, Krishna Sarva, Melvin Earl Klegerman, David Dugald McPherson, Steven Idell, Galina Florova and Andrey Anatolievich Komissarov
Pharmaceutics 2026, 18(6), 646; https://doi.org/10.3390/pharmaceutics18060646 (registering DOI) - 24 May 2026
Abstract
Background: Pharmacological treatment under conditions of slow fibrinolysis/thrombolysis requires the targeted delivery of plasminogen-activating activity. Echogenic liposomal formulations (regular TELIP) of single-chain tissue plasminogen activator (sctPA), while possessing high affinity to fibrin, contain free/loosely bound sctPA. We hypothesized that removal of free sctPA,
[...] Read more.
Background: Pharmacological treatment under conditions of slow fibrinolysis/thrombolysis requires the targeted delivery of plasminogen-activating activity. Echogenic liposomal formulations (regular TELIP) of single-chain tissue plasminogen activator (sctPA), while possessing high affinity to fibrin, contain free/loosely bound sctPA. We hypothesized that removal of free sctPA, which competes with liposomes and plasmin for fibrin, enhances unique features of the TELIP. Methods: Optimized and regular TELIP were assessed for the distribution of active sctPA (loosely bound, tightly bound, encapsulated), stability, binding to fibrin, initiating fibrinolysis in vitro and ex vivo using a battery of biochemical methods. Results: One milligram of the regular TELIP consists of 2.0–5.0 × 109 echogenic liposomes (700–900 nm diameter). Non-specifically bound sctPA readily dissociates at the physiological ionic strength and pH. While up to 60% of sctPA in the regular TELIP is loosely bound with 6–15% encapsulated, and the rest is tightly bound to the liposomes; in the optimized TELIP, more than 80% of active sctPA is tightly bound with up to 40% of encapsulated. The latter is protected from high-molecular-weight ligands and could be released by an ultrasound pulse. Optimized TELIP shows low competition with plasmin for fibrin and effectively supports fibrinolysis in vitro and ex vivo. The optimized TELIP with maximal load of sctPA 3% (w/w) retains integrity at 37 °C for 5 h in vitro and up to 2 h ex vivo. Conclusions: The optimized TELIP is stable in vitro and ex vivo, does not interfere with fibrinolysis and retains a high level of encapsulated sctPA delivered precisely to the thrombus/fibrin clot.
Full article
(This article belongs to the Special Issue Advanced Drug Delivery Systems for Natural Products)
►▼
Show Figures
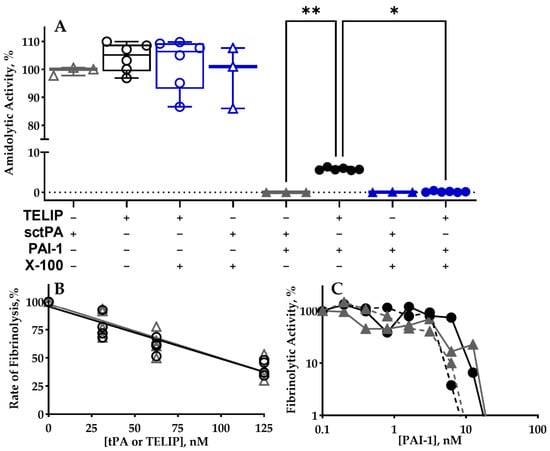
Figure 1
Open AccessArticle
Irinotecan Hydrochloride Administration Considering Dosing-Time Attenuates Delayed Diarrhea in Rats
by
Hideto To, Mari Tomonari, Makoto Myojin and Fumiyasu Okazaki
Pharmaceutics 2026, 18(6), 645; https://doi.org/10.3390/pharmaceutics18060645 (registering DOI) - 24 May 2026
Abstract
Background: Irinotecan hydrochloride (CPT-11) is an important anticancer drug used in a wide range of regimens to treat colorectal and gastric cancers, and one of its severe side effects is delayed diarrhea. Therefore, based on known circadian variations in intestinal function and drug
[...] Read more.
Background: Irinotecan hydrochloride (CPT-11) is an important anticancer drug used in a wide range of regimens to treat colorectal and gastric cancers, and one of its severe side effects is delayed diarrhea. Therefore, based on known circadian variations in intestinal function and drug metabolism, we investigated whether CPT-11-induced delayed diarrhea may be attenuated by the time of dosing. Methods: When CPT-11 was administered to rats at 9:00 or 21:00, CPT-11-induced delayed diarrhea was assessed, and concentrations of CPT-11, its active metabolite SN-38, and SN-38 glucuronide (SN-38GL) in blood, intestinal tissues, and intestinal contents were measured. Results: The severity of diarrhea was significantly less in the 21:00 dosing group compared with the 9:00 dosing group. Blood SN-38 concentrations 8 h after the administration of CPT-11 were significantly higher in the 9:00 dosing group than in the 21:00 dosing group. SN-38, which exerts potent cytotoxic effects, circulates enterohepatically. When SN-38 is absorbed from the intestinal mucosa, intestinal tissues may be injured, resulting in delayed diarrhea. CPT-11 and SN-38 concentrations in intestinal tissues and contents 8 h after the administration of CPT-11 were significantly higher in the 9:00 dosing group than in the 21:00 dosing group at all measurement points. This was consistent with more severe CPT-11-induced delayed diarrhea in the 9:00 dosing group. Conclusions: Chronotherapy with CPT-11 may reduce CPT-11-induced delayed diarrhea. These differences in SN-38 concentrations in the intestinal tract at different dosing times may contribute to the time-dependent reduction in CPT-11-induced delayed diarrhea.
Full article
(This article belongs to the Section Pharmacokinetics and Pharmacodynamics)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Chitosan-Coated Mesoporous Silica Nanoparticles Co-Loaded with Curcumin and Amphotericin B: A Drug Delivery Approach for Photodynamic Inhibition of Dual-Species Biofilms
by
Shima Afrasiabi, Mohammad Reza Karimi, Sepideh Khoee, Stefano Benedicenti and Antonio Signore
Pharmaceutics 2026, 18(6), 644; https://doi.org/10.3390/pharmaceutics18060644 (registering DOI) - 23 May 2026
Abstract
Background/Objectives: Metabolic dormancy in biofilms leads to reduced drug efficacy in these communities. Different pharmacokinetics and adverse side effects complicate the simultaneous delivery of multiple drugs at appropriate concentrations to the infection site. This study aimed to develop chitosan-coated mesoporous silica nanoparticles loaded
[...] Read more.
Background/Objectives: Metabolic dormancy in biofilms leads to reduced drug efficacy in these communities. Different pharmacokinetics and adverse side effects complicate the simultaneous delivery of multiple drugs at appropriate concentrations to the infection site. This study aimed to develop chitosan-coated mesoporous silica nanoparticles loaded with curcumin and amphotericin B (CS@MSNs-Cur-AmB) and to evaluate their antibiofilm activity combined with antimicrobial photodynamic therapy (PDT) against Streptococcus mutans and Candida albicans dual-species biofilms. Methods: CS@MSNs-Cur-AmB were developed. The structure and morphology of the nanoparticles were evaluated using Fourier transform-infrared spectroscopy (FTIR), zeta potential, field emission scanning electron microscopy (FESEM), and thermogravimetric analysis (TGA). Cytotoxicity toward human gingival fibroblasts was assessed. Colony-forming units per milliliter (CFU/mL) were determined. The metabolic activity of biofilm-forming cells was measured using the tetrazolium (MTT) assay. Results: Physicochemical analyses confirmed the synthesis of CS@MSNs-Cur-AmB, revealing a particle size of 228 nm and thermal stability up to 600 °C. Cytotoxicity assays showed that CS@MSNs-Cur-AmB exhibited good biocompatibility (> 90%). CS@MSNs-Cur-AmB improved antimicrobial activity, which was further enhanced by blue light-emitting diode (LED) irradiation. CS@MSNs-Cur-AmB under LED irradiation showed the strongest effect, reducing metabolic activity to 27.74 ± 4.08% (1 W/cm2, 1 min), p < 0.001). Conclusions: Formulating two drugs in nanocarrier systems may improve therapeutic efficacy by increasing local concentration and reducing systemic exposure. This offers an effective strategy for combating oral biofilms.
Full article
(This article belongs to the Special Issue Advanced Drug Delivery Systems for Natural Products)
Open AccessReview
The Effect of Anti-Inflammatory Drugs on the Incidence of Colorectal Cancer
by
Marek Misiak, Aleksandra Maciejowska, Maciej Pałęga, Rafał Burek, Anita Gołda, Michalina Dworak, Beata Pawuła-Prgomet, Karol Forysiński and Tomasz Miłek
Pharmaceutics 2026, 18(6), 643; https://doi.org/10.3390/pharmaceutics18060643 (registering DOI) - 23 May 2026
Abstract
Background/Objectives: Chronic inflammation is a key factor in the development and progression of colorectal cancer (CRC). When COX-2 levels and PGE2 production increase, nonsteroidal anti-inflammatory drugs (NSAIDs), including aspirin (ASA) and selective COX-2 inhibitors, such as celecoxib and rofecoxib, are commonly employed.
[...] Read more.
Background/Objectives: Chronic inflammation is a key factor in the development and progression of colorectal cancer (CRC). When COX-2 levels and PGE2 production increase, nonsteroidal anti-inflammatory drugs (NSAIDs), including aspirin (ASA) and selective COX-2 inhibitors, such as celecoxib and rofecoxib, are commonly employed. This paper presents the effect of anti-inflammatory drugs, primarilyNSAIDs, on the incidence of CRC. Methods: A comprehensive literature search (119 articles) was conducted with databases such as PubMed. During our research, we used keywords such as colorectal cancer (CRC), nonsteroidal anti-inflammatory drugs (NSAIDs), ASA, COX, precision oncology, and personalized medicine. Results: The development of CRC is primarily associated with chronic inflammation and the actions of COX-2 and prostaglandin E2 (PGE2), which promote cancer cell proliferation and angiogenesis. Anti-inflammatory drugs act by inhibiting the secretion of COX-1 and COX-2 enzymes, which leads to reduced PGE2 production and may limit tumor growth. Aspirin has the best-documented and studied anti-cancer effect; long-term use is associated with a reduced risk of CRC development and mortality through its anti-inflammatory and antiplatelet effects, thereby limiting metastasis. Particularly beneficial effects are observed in patients with mutations in the PIK3CA gene. Factors influencing the effectiveness of CRC treatment include molecular differences and tumor location. Conclusions: The future of CRC treatment and prevention lies in personalized medicine, which accounts for each patient’s genetic profile. Decisions regarding NSAIDs use and CRC prevention should consider the potential benefits and risks of side effects.
Full article
(This article belongs to the Topic The Tumor Microenvironment, Immuno-Oncology, and Immune Checkpoint: Implications for Current and Emergent Immunotherapies, 2nd Edition)
Open AccessReview
Exploring Copaiba and Andiroba Oils: A Comprehensive Review of Composition, Physicochemical Properties and Pharmacological Activities in Advanced Delivery Systems
by
Ana Luisa Pinto Magalhães, Nayara Santana Peixoto Moura, Janaína de Alcântara Lemos, Carolina de Aguiar Ferreira, Danyelle M. Townsend, Juliana de Oliveira Silva, Anna Eliza Maciel de Faria Mota Oliveira and André Luis Branco de Barros
Pharmaceutics 2026, 18(6), 642; https://doi.org/10.3390/pharmaceutics18060642 (registering DOI) - 23 May 2026
Abstract
Background/Objectives: The convergence of traditional medicinal practices in Brazil’s vast biodiversity has fueled pharmaceutical interest in advancing plant-derived formulation. Copaiba (Copaifera spp.) and andiroba (Carapa guianensis) are central to both the economic landscape and healing traditions of the Amazon
[...] Read more.
Background/Objectives: The convergence of traditional medicinal practices in Brazil’s vast biodiversity has fueled pharmaceutical interest in advancing plant-derived formulation. Copaiba (Copaifera spp.) and andiroba (Carapa guianensis) are central to both the economic landscape and healing traditions of the Amazon rainforest. Derivatives from these species have diverse applications, with their oils representing important raw materials for therapeutic use. However, the poor aqueous solubility of oils remains a major barrier to developing formulations with optimal bioavailability. Nanotechnology offers a strategic approach to address this limitation, as nanosystems improve stability, solubility, and biological performance. Methods: This narrative review compiles and analyzes contemporary literature on the chemical composition, physicochemical properties, and pharmacological activities of copaiba and andiroba oils, with emphasis on studies involving nanoformulations, aiming to overcome the solubility limitations of these oils. Results: Evidence from the literature indicates that nanoencapsulation enhances the anti-inflammatory, antimicrobial, and wound-healing activity of the oils’ main constituents, such as beta-caryophyllene and limonoids. However, inconsistencies in reported chemical composition and physicochemical properties across studies highlight the lack of standardized characterization and extraction methods, potentially hindering the development of reproducible nanosystems. Conclusions: Nanoencapsulation represents a promising strategy to improve the therapeutic potential of Amazonian oils. Nevertheless, further efforts are required to standardize methodologies and expand clinical studies to confirm the efficacy and safety of nanosystems derived from these natural products.
Full article
(This article belongs to the Special Issue Biomedical Applications of Nanocarriers in Targeted Delivery of Bioactive Compounds)
Open AccessReview
Toward an AI Era: Application of Artificial Intelligence in Inclusion Complex Screening
by
Naixuan Deng, Yeqi Huang, Yue Gao, Hongluo Li, Wenjing Wang, Minjing Cheng, Chuanbin Wu, Xin Pan, Ling Guo, Junhuang Jiang and Zhengwei Huang
Pharmaceutics 2026, 18(6), 641; https://doi.org/10.3390/pharmaceutics18060641 (registering DOI) - 23 May 2026
Abstract
Supramolecular inclusion complexes are widely used in drug delivery and other fields, with the advantages of controllable structures, high stability, excellent biocompatibility, and the ability to improve drug solubility and achieve controlled release. However, traditional screening methods rely on experimental trial and error,
[...] Read more.
Supramolecular inclusion complexes are widely used in drug delivery and other fields, with the advantages of controllable structures, high stability, excellent biocompatibility, and the ability to improve drug solubility and achieve controlled release. However, traditional screening methods rely on experimental trial and error, which suffer from long cycles, high costs, and low throughput, limiting research and development efficiency. In recent years, the development of artificial intelligence has provided new solutions for the screening of inclusion complexes. This paper systematically reviewed the core technological system of AI in the screening of inclusion complexes, focusing on two aspects: prediction and optimization of key properties and rational design of host molecules, summarizing their specific application progress. At the same time, we analyzed the current core challenges, including data scarcity, insufficient model interpretability, and limited generalization capabilities, and propose future development directions such as building standardized databases, integrating physicochemical principles (e.g., molecular mechanics and thermodynamics), and establishing closed-loop research and development platforms. This review aims to provide a systematic reference for the in-depth application of artificial intelligence in the field of supramolecular inclusion complexes.
Full article
(This article belongs to the Section Pharmaceutical Technology, Manufacturing and Devices)
Open AccessReview
Polypharmacy and Drug–Drug Interactions in Chronic Obstructive Pulmonary Disease: A Narrative Clinical Review
by
Maria-Medana Drăgoi, Florina-Diana Goldiș, Sabina-Oana Vasii, Daiana Colibășanu, Liana Suciu, Angela Caunii and Lucreția Udrescu
Pharmaceutics 2026, 18(6), 640; https://doi.org/10.3390/pharmaceutics18060640 (registering DOI) - 23 May 2026
Abstract
Background: Chronic obstructive pulmonary disease (COPD) is commonly managed alongside multimorbidity, polypharmacy, recurrent treatment escalation, and older age, all of which increase vulnerability to drug–drug interactions (DDIs). We aimed to synthesize the main DDI domains relevant to COPD pharmacotherapy and to distinguish
[...] Read more.
Background: Chronic obstructive pulmonary disease (COPD) is commonly managed alongside multimorbidity, polypharmacy, recurrent treatment escalation, and older age, all of which increase vulnerability to drug–drug interactions (DDIs). We aimed to synthesize the main DDI domains relevant to COPD pharmacotherapy and to distinguish harmful DDIs from beneficial combination therapy and formal compatibility findings. Methods: We performed a narrative review using structured literature searches and citation tracking to evaluate COPD-related studies. We prioritized direct COPD-specific DDI evidence, while also including mechanistic, class-specific, and contextual studies when direct evidence was lacking. Retained evidence included observational cohorts, prescribing studies, pharmacokinetic trials, case reports, and systematic reviews. Results: The reviewed literature indicates that DDI vulnerability in COPD is driven less by isolated drug pairs than by overall regimen complexity, multimorbidity, aging, fragmented prescribing, and high-intensity treatment periods such as exacerbations, hospitalization, and discharge. Key DDI domains included cardiopulmonary co-treatment, QT-related vulnerability, and potential or clinically relevant interactions amplified during exacerbations. Inhaled therapies are not universally interaction-free, particularly with strong metabolic inhibitors. Psychotropics, frailty, dementia, and palliative care further increase clinical complexity. However, beneficial bronchodilator combinations and formal compatibility studies demonstrate that not all multidrug COPD regimens are harmful. Conclusions: In COPD, DDI assessment should focus on the full treatment regimen and not be limited to a set of iconic drug pairs. Clinicians must focus on exacerbation-related prescribing, QT-active drugs, theophylline exposure, psychotropic co-medication, and vulnerable subgroups such as older, frail, and palliative patients. Pharmacist-supported drug review, drug reconciliation, and selective deprescribing are key strategies for reducing clinically relevant DDI burden in COPD.
Full article
(This article belongs to the Special Issue Drug–Drug Interactions—New Perspectives)
Open AccessArticle
Prediction of Tacrolimus–Posaconazole Interactions in Renal Transplant Patients with Different CYP3A5 Genotypes, Based on Physiological Pharmacokinetic Models
by
Mengmeng Guan, Wanyi Zhou, Haoran Qin, Yi Xu, Di Zhao, Hui Xue and Nan Hu
Pharmaceutics 2026, 18(6), 639; https://doi.org/10.3390/pharmaceutics18060639 - 22 May 2026
Abstract
Objective: Posaconazole, a second-generation triazole antifungal used for the prevention or treatment of invasive fungal infections, has been shown to markedly increase tacrolimus exposure in vivo when co-administered, potentially leading to clinically significant adverse events. A physiologically based pharmacokinetic (PBPK) model was developed
[...] Read more.
Objective: Posaconazole, a second-generation triazole antifungal used for the prevention or treatment of invasive fungal infections, has been shown to markedly increase tacrolimus exposure in vivo when co-administered, potentially leading to clinically significant adverse events. A physiologically based pharmacokinetic (PBPK) model was developed to predict tacrolimus–posaconazole interactions in renal transplant recipients with different CYP3A5 genotypes, to inform tacrolimus dose adjustment in clinical practice. Methods: First, to obtain the critical inhibition parameters, in vitro enzyme kinetic studies were conducted. Based on these data, a whole-body physiologically based pharmacokinetic (PBPK) model for TAC was developed and validated in PK-Sim. A published, validated posaconazole PBPK model was applied concurrently. Model performance was evaluated against published pharmacokinetic data in healthy volunteers receiving tacrolimus with posaconazole. A virtual Chinese renal transplant recipient was generated by incorporating population-specific physiological parameters, including CYP3A5 genotype-dependent enzyme expression. Results: In vitro experimental results demonstrated that POSA acts as a potent reversible competitive inhibitor of CYP3A4/5-mediated TAC metabolism. The tacrolimus PBPK model adequately captured pharmacokinetics across CYP3A5 genotypes, and tacrolimus pharmacokinetics during co-administration with posaconazole were also predicted. Compared with CYP3A5 expressers, nonexpressers showed greater variability in tacrolimus whole-blood concentrations and greater susceptibility to posaconazole-mediated interactions. The CYP3A5*3*3 genotype was associated with higher Cmax and AUC. Dose optimization simulations predicted that after 6–7 days of posaconazole co-administration, nonexpressers would require the reduction of tacrolimus dosing frequency from every 12 h to every 24 h to maintain trough concentrations within 8–15 ng/mL, whereas a 50% dose reduction was predicted to be optimal for expressers. Conclusions: A tacrolimus–posaconazole PBPK drug–drug interaction model was developed for the population of renal transplant recipients and used to simulate tacrolimus trough concentrations across CYP3A5 genotypes and dosing regimens, supporting genotype-informed co-administration in clinical practice.
Full article
(This article belongs to the Section Clinical Pharmaceutics)
►▼
Show Figures

Figure 1
Open AccessReview
Cannabidiol for Mucosal Diseases: Therapeutic Potential and Advanced Delivery Strategies
by
Bo Han, Yue Zhang, Yangmin Wang, Yue Shen, Jinping Niu, Shipo Li, Yuxi Li, Jingyu Wang, Xingyuan Ma and Wenyun Zheng
Pharmaceutics 2026, 18(6), 638; https://doi.org/10.3390/pharmaceutics18060638 - 22 May 2026
Abstract
Cannabidiol (CBD), a major non-psychoactive phytocannabinoid, has attracted considerable attention owing to its broad therapeutic potential. Its anti-inflammatory, antimicrobial, and antitumor properties make it a promising candidate for the treatment of mucosa-associated diseases. However, the clinical translation of CBD is significantly hindered by
[...] Read more.
Cannabidiol (CBD), a major non-psychoactive phytocannabinoid, has attracted considerable attention owing to its broad therapeutic potential. Its anti-inflammatory, antimicrobial, and antitumor properties make it a promising candidate for the treatment of mucosa-associated diseases. However, the clinical translation of CBD is significantly hindered by its unfavorable physicochemical properties, particularly high lipophilicity and poor aqueous solubility, which result in low bioavailability. To overcome these limitations, the rational selection of administration routes in combination with advanced drug delivery systems tailored to disease pathophysiology is essential. Such strategies are critical for improving the stability of CBD, enhancing mucosal permeation, and enabling controlled and targeted release at diseased sites. Nevertheless, a systematic review focusing on these aspects is still lacking. This review first summarizes the relationship between CBD and the mucosal endocannabinoid system, together with its pharmacological effects. It then discusses the therapeutic potential of CBD in mucosal disorders of the digestive and respiratory systems. In addition, current administration routes and advanced delivery systems for CBD are reviewed to provide insights for future research and clinical translation. Finally, the remaining challenges associated with the clinical application of CBD and future development directions are discussed.
Full article
(This article belongs to the Special Issue Advanced Drug Delivery Systems for Natural Products)
►▼
Show Figures
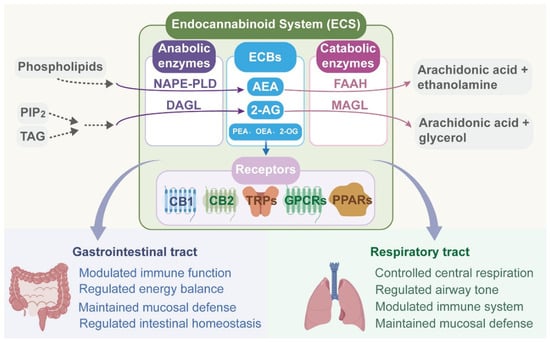
Figure 1
Open AccessArticle
Formulation Characteristics of Solid-Dispersible Self-Emulsifying Drug Delivery Systems for Dual Drug Delivery
by
Shailvi Soni and Terrick Andey
Pharmaceutics 2026, 18(6), 637; https://doi.org/10.3390/pharmaceutics18060637 - 22 May 2026
Abstract
Background: Oral delivery of chemotherapeutic agents remains challenging due to gastrointestinal degradation, poor intestinal permeability, and extensive first-pass metabolism, which collectively limit bioavailability. Lipid-based drug delivery systems offer a promising strategy to overcome these barriers. This study aimed to develop a freeze-dried,
[...] Read more.
Background: Oral delivery of chemotherapeutic agents remains challenging due to gastrointestinal degradation, poor intestinal permeability, and extensive first-pass metabolism, which collectively limit bioavailability. Lipid-based drug delivery systems offer a promising strategy to overcome these barriers. This study aimed to develop a freeze-dried, solid-dispersible self-emulsifying drug delivery system (SEDDS) using a water-in-oil-in-water (w/o/w) double emulsion approach for the co-encapsulation of hydrophilic (doxorubicin) and lipophilic (ellipticine) agents to enhance oral delivery. Methods: Double-emulsion SEDDS were prepared via a two-stage emulsification process to enable compartmentalized drug loading within aqueous and oil phases. The formulations were freeze-dried to improve stability and storage. Physicochemical properties were characterized using dynamic light scattering for droplet size and polydispersity index (PDI), zeta potential analysis for colloidal stability, and differential scanning calorimetry for thermal behavior. Drug encapsulation efficiency was determined, and cellular uptake was evaluated in breast cancer cells using fluorescence microscopy. Results: Optimized SEDDS exhibited droplet sizes of 90–347 nm with low PDI values (0.005–0.336), indicating uniform and stable dispersions. Zeta potential values (−10.64 to 2.38 mV) supported colloidal stability, while freeze-dried formulations retained dispersion characteristics upon reconstitution over extended storage. Both drugs demonstrated high encapsulation efficiency (>97%), and thermal analysis confirmed the formation of stable amorphous systems. Fluorescence imaging revealed enhanced intracellular uptake of both agents. Conclusions: This study demonstrates that freeze-dried double-emulsion SEDDS enable efficient co-delivery of hydrophilic and lipophilic drugs, improving stability and cellular uptake. This platform shows strong potential for overcoming key barriers in oral chemotherapy and provides a promising strategy for combination drug delivery.
Full article
(This article belongs to the Special Issue Advances in Nanoemulsion for Drug Delivery)
Open AccessArticle
Model-Based Virtual Clinical Trial Reveals Renal Impairment and Body Size as Key Determinants of Pharmacokinetic Variability and Drug-Drug Interaction Risk in Propranolol Therapy
by
Lara Marques and Nuno Vale
Pharmaceutics 2026, 18(6), 636; https://doi.org/10.3390/pharmaceutics18060636 - 22 May 2026
Abstract
Background/Objectives: Propranolol (PROP) is a non-selective β-blocker widely prescribed for cardiovascular and neurological disorders. Its pharmacokinetics (PK) are highly variable, and co-administration with omeprazole (OME), a CYP2C19 substrate and inhibitor, may alter systemic exposure. Herein, this study aimed to investigate factors influencing PROP
[...] Read more.
Background/Objectives: Propranolol (PROP) is a non-selective β-blocker widely prescribed for cardiovascular and neurological disorders. Its pharmacokinetics (PK) are highly variable, and co-administration with omeprazole (OME), a CYP2C19 substrate and inhibitor, may alter systemic exposure. Herein, this study aimed to investigate factors influencing PROP PK variability and evaluate the effect of OME coadministration using physiologically based pharmacokinetic (PBPK) modeling and population PK (popPK) analysis. Methods: PBPK models for PROP and OME were developed and validated against published data. DDI simulations were conducted across clinically relevant dosing regimens. A two-period fixed-sequence virtual trial of 125 subjects was simulated with PROP alone and PROP combined with OME. Population PK (popPK) analysis was performed on simulated plasma concentration data to identify covariates affecting PROP disposition and quantify DDI magnitude. Results: PBPK models were successfully developed and validated. PROP disposition was best described by a two-compartment model with linear elimination. Health status was found to influence clearance, and body surface area (BSA) affected the central volume of distribution. Co-administration with OME increased PROP exposure, with larger effects in patients with renal impairment. Simulated plasma concentrations remained below established toxicity thresholds. Conclusions: Virtual clinical trials integrating PBPK and popPK modeling provide a robust approach to identifying key determinants of PK variability and DDI risk. Although these findings were not directly translated to clinical observations, this helps identify sources of PK variability in PROP treatment settings and factors that may intensify its interaction with OME, thereby supporting model-informed precision dosing to enhance safety and efficacy.
Full article
(This article belongs to the Special Issue Pathophysiological Influences on Pharmacokinetics and Pharmacodynamics, 2nd Edition)
►▼
Show Figures

Figure 1
Open AccessArticle
An Interactive Constraint-Based Decision-Support Tool for Pharmaceutical Formulation Development
by
Reihaneh Manteghi and Eduardo Veas
Pharmaceutics 2026, 18(6), 635; https://doi.org/10.3390/pharmaceutics18060635 - 22 May 2026
Abstract
Background/Objectives: Pharmaceutical formulation involves designing a drug product by combining the properties of an active pharmaceutical ingredient (API) with suitable excipients and processing strategies to produce a safe, effective, and manufacturable dosage form. However, data in formulation science are often limited, expensive to
[...] Read more.
Background/Objectives: Pharmaceutical formulation involves designing a drug product by combining the properties of an active pharmaceutical ingredient (API) with suitable excipients and processing strategies to produce a safe, effective, and manufacturable dosage form. However, data in formulation science are often limited, expensive to generate, and frequently restricted by proprietary and confidentiality constraints. Interactive digital tools can support formulators during early drug product development by improving the structure, transparency, and efficiency of formulation decision-making. While the current system focuses on structured decision support, future extensions may incorporate machine-learning methods for recommendation and knowledge extraction. Methods: In this work, we developed the Formulation tool, an interactive decision-support and visualization system for formulation development based on a hierarchical formulation-strategy framework commonly used in pharmaceutical practice. The tool is designed to prioritize suitable formulation principles and associated processing routes, with oral solid formulation as the initial application domain. The evaluated scenarios also include pathway regions relevant to oral liquid formulations. Its modular architecture also makes it adaptable to other formulation scenarios. To assess practical applicability, the tool was evaluated in a structured expert study involving five expert users across six predefined formulation scenarios (n = 30 runs) , covering three drugs under adult and pediatric conditions. Results: The tool showed agreement with the expected dosage-form families and overall formulation properties, with adult scenarios converging to oral solid regions and pediatric scenarios converging to oral liquid regions. At the downstream formulation-profile level, users converged either to the dominant expected pathway or to alternative feasible pathways within the same formulation region. Variability in full pathway completion was observed and was primarily associated with differences in user interaction behavior and exploratory usage patterns. The median task completion time was 113.5 s. Conclusions: In addition to organizing formulation knowledge, the Formulation tool records user interactions in a structured manner, which may support future learning from usage patterns. Because detailed downstream formulation constraints are often institution-specific and are typically not available in the public domain, the present evaluation focused on agreement at the dosage-form-family level and on overall formulation properties rather than on highly specialized constraint logic. The system is based on a constraint satisfaction problem (CSP) framework, which is well suited for modeling complex decision processes under explicit constraints. CSP has also been widely applied in intelligent scheduling systems, supporting its suitability for structured, constraint-rich decision-making tasks such as pharmaceutical formulation strategy development.
Full article
(This article belongs to the Special Issue Advanced Computational Tools in Development and Assessment of Dosage Form)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Elucidating the Critical Role of Excipients in Gastric Emptying and Oral Absorption of a Rapidly Eliminated BCS I Drug: Implications from Zidovudine Bioequivalence
by
Yan Lin, Xian Zhang, Fulin Bi, Guangji Wang and Jin Yang
Pharmaceutics 2026, 18(6), 634; https://doi.org/10.3390/pharmaceutics18060634 - 22 May 2026
Abstract
Background/Objectives: Despite the presumption of bioequivalence for BCS Class I drugs due to their high solubility and permeability, recent evidence indicates that those with rapid systemic elimination exhibit heightened vulnerability to Cmax non-equivalence, primarily attributable to intrasubject variability in gastrointestinal transit and
[...] Read more.
Background/Objectives: Despite the presumption of bioequivalence for BCS Class I drugs due to their high solubility and permeability, recent evidence indicates that those with rapid systemic elimination exhibit heightened vulnerability to Cmax non-equivalence, primarily attributable to intrasubject variability in gastrointestinal transit and absorption kinetics. It is well known that gastric emptying is a significant physiological-dependent factor. But, does the formulation affect gastric emptying? Methods: Using zidovudine as a model drug, formulations containing sodium carboxymethyl starch (CMS-Na), pregelatinized starch, hydroxypropyl methylcellulose (HPMC), and lactose were investigated for their effects on gastric emptying kinetics, and the impact of excipient-mediated gastric emptying prolongation on pharmacokinetic parameters was also evaluated. Results: Relative to AZT alone (Cmax = 13,350 ng/mL; gastric %ID = 11.3%), co-administration with CMS-Na, pregelatinized starch, or HPMC significantly prolonged gastric retention (%ID: 23.4%, 30.5%, and 40.8% at 22.5 min) and reduced Cmax in rats by 47.8%, 34.4%, and 35.1%, respectively, with no effect on intestinal permeability. Viscosity positively correlated with gastric emptying delay. Conclusions: Our rat findings provide new possible mechanistic evidence that certain viscosity-modifying excipients can delay gastric emptying and reduce Cmax of zidovudine, a rapidly eliminated BCS Class I drug, with potential implications for biowaiver risk assessment. Gastric emptying is not only a physiological-dependent variation but also, in cases where common excipients may significantly delay gastric emptying, a formulation-dependent rate-limiting step. For such drugs, excipient-induced gastric emptying delay poses an underappreciated risk to the biowaiver approach, necessitating more prudent regulatory assessment that encompasses the dynamic interplay among sequential rate processes governing drug disposition.
Full article
(This article belongs to the Special Issue Drug Product Performance: Bioavailability, Relative Bioavailability and Bioequivalence, 3rd Edition)
►▼
Show Figures
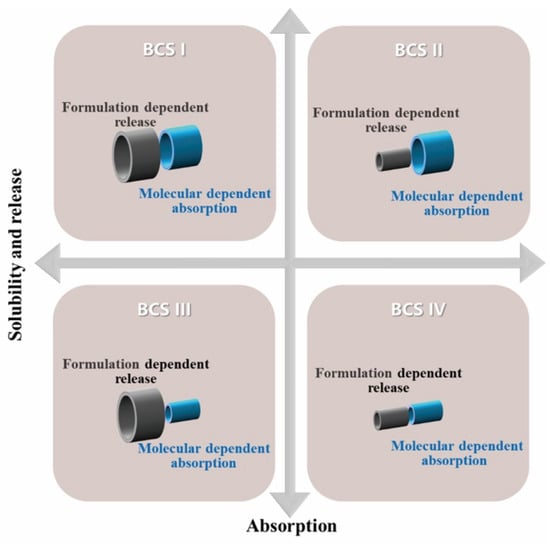
Figure 1
Open AccessArticle
Single-Nucleotide Polymorphisms in Capecitabine Bioactivation Genes and Their Contribution to Breast Cancer Therapy
by
Andrea Fernández, Yasmín Cura-Cuevas, Susana Rojo-Tolosa, José María Gálvez-Navas, Encarnación González-Flores, Cristina Pérez-Ramírez and Alberto Jiménez-Morales
Pharmaceutics 2026, 18(6), 633; https://doi.org/10.3390/pharmaceutics18060633 - 22 May 2026
Abstract
Background/Objectives: Breast cancer (BC) is a highly prevalent neoplasm worldwide. Despite the wide range of therapeutic options currently available, it remains the leading cause of cancer-related mortality among women. Capecitabine, a prodrug of 5-fluorouracil (5-FU), is widely used in the treatment of advanced
[...] Read more.
Background/Objectives: Breast cancer (BC) is a highly prevalent neoplasm worldwide. Despite the wide range of therapeutic options currently available, it remains the leading cause of cancer-related mortality among women. Capecitabine, a prodrug of 5-fluorouracil (5-FU), is widely used in the treatment of advanced BC. However, despite its efficacy, capecitabine exhibits considerable interindividual variability in therapeutic response. This study aimed to evaluate the effect of single-nucleotide polymorphisms (SNPs) in genes involved in capecitabine bioactivation on progression-free survival (PFS) in patients with BC. Methods: An ambispective cohort study was conducted. Four relevant SNPs in the CES1, CDA, and TYMP genes were analyzed in 85 Caucasian patients with BC using real-time polymerase chain reaction (PCR) with TaqMan® probes. Results: A significant association was observed between shorter PFS and the GA genotype of the CES1 rs71647871 SNP (p = 0.010; HR = 7.46; 95% CI = 1.24–122.52), as well as with the TT genotype of the CDA rs602950 SNP (p = 0.009; HR = 3.50; 95% CI = 1.36–9.03). Conclusions: These findings suggest that CES1 rs71647871 and CDA rs602950 may serve as predictive biomarkers of capecitabine effectiveness in patients with BC. Further studies involving larger cohorts are needed to validate these findings and generate additional evidence to support their potential implementation in clinical practice.
Full article
(This article belongs to the Section Gene and Cell Therapy)
►▼
Show Figures

Figure 1
Open AccessReview
Carbon Dots in Nanomedicine: Advanced Fabrication, Biomedical Applications, and Future Clinical Perspectives
by
Muhammad Sohail Khan, Imran Zafar, Dayeon Ham, Ki Sung Kang and Il-Ho Park
Pharmaceutics 2026, 18(5), 632; https://doi.org/10.3390/pharmaceutics18050632 - 21 May 2026
Abstract
Carbon dots (CDs), including carbon quantum dots (CQDs), are ultra-small carbon-based nanomaterials, typically below 10 nm, with tunable photoluminescence, high aqueous dispersibility, favorable biocompatibility, low toxicity, and abundant surface functional groups. These properties make CDs promising multifunctional platforms for nanomedicine, particularly in bioimaging,
[...] Read more.
Carbon dots (CDs), including carbon quantum dots (CQDs), are ultra-small carbon-based nanomaterials, typically below 10 nm, with tunable photoluminescence, high aqueous dispersibility, favorable biocompatibility, low toxicity, and abundant surface functional groups. These properties make CDs promising multifunctional platforms for nanomedicine, particularly in bioimaging, biosensing, targeted drug/gene delivery, photodynamic therapy (PDT), photothermal therapy (PTT), antimicrobial treatment, and theranostic applications. This review critically examines recent advances in CD fabrication, including top-down, bottom-up, green biomass-derived, microwave-assisted, hydrothermal, and emerging hybrid strategies, with emphasis on how precursor selection, heteroatom doping, surface passivation, and polymer/ligand functionalization regulate optical performance, biological interaction, and therapeutic efficiency. The review discusses structural classification, including CQDs, graphene quantum dots (GQDs), carbon nanodots, and carbonized polymer dots (CPDs), together with major characterization approaches such as ultraviolet–visible (UV–Vis) spectroscopy, Fourier-transform infrared (FTIR) spectroscopy, X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), Raman spectroscopy, and high-resolution transmission electron microscopy (HRTEM). Particular attention is given to red/near-infrared (NIR) emission, renal clearance, drug-loading behavior, reactive oxygen species (ROS) generation, toxicity mechanisms, biodistribution, and long-term biosafety. This review also highlights key translational barriers, including batch-to-batch variability, limited standardization, scalable manufacturing, regulatory uncertainty, and incomplete pharmacokinetic evaluation. It considers artificial intelligence (AI) and machine learning (ML) as emerging tools for reproducible CD design. CDs represent versatile and clinically promising nanoplatforms, but their translation requires standardized synthesis, rigorous safety assessment, and application-specific regulatory validation.
Full article
(This article belongs to the Special Issue Nanomaterials for Cell Biological and Biomedical Applications)
►▼
Show Figures
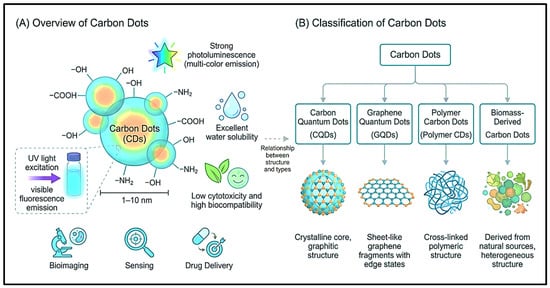
Figure 1
Open AccessArticle
Crosslinking-Dependent Design of Hyaluronic Acid Matrices for Enhanced Bioadhesion and Cellular Response
by
Alina Diana Panainte, Cătălina Anișoara Peptu, Andreea Crețeanu, Nela Bibire, Isabella Nacu, Liliana Vereștiuc, Eliza Grațiela Popa, Larisa Păduraru, Liliana Mititelu Tartau, Radu Dănilă, Tudor Bibire and Catalina Natalia Yilmaz
Pharmaceutics 2026, 18(5), 631; https://doi.org/10.3390/pharmaceutics18050631 - 21 May 2026
Abstract
Hyaluronic acid (HA) hydrogels have attracted increasing interest for biomedical applications due to their tunable properties and biocompatibility. Methods: In this study, hyaluronic acid HA-based hydrogels were developed using two distinct crosslinking strategies: physical crosslinking through poly(vinyl alcohol) (PVA) incorporation and covalent crosslinking
[...] Read more.
Hyaluronic acid (HA) hydrogels have attracted increasing interest for biomedical applications due to their tunable properties and biocompatibility. Methods: In this study, hyaluronic acid HA-based hydrogels were developed using two distinct crosslinking strategies: physical crosslinking through poly(vinyl alcohol) (PVA) incorporation and covalent crosslinking via DCC/NHS-mediated reactions. Piroxicam (Px) was included as a model drug to evaluate the drug delivery potential of the resulting systems. The hydrogels were characterized in terms of morphology, swelling behaviour, adhesion, enzymatic degradation, drug release, and in vitro cytocompatibility. Results: The results indicate that formulation parameters significantly influence the overall performance of the systems. PVA-containing hydrogels exhibited higher swelling capacity and improved adhesive properties, while covalently crosslinked networks showed reduced swelling and enhanced structural stability and resistance to enzymatic degradation. Drug release profiles were dependent on network structure, with more compact systems displaying slower release behaviour. In vitro assays suggested that the developed hydrogels are cytocompatible and that drug incorporation influences both release kinetics and cellular response. However, it should be noted that the biological evaluation was performed under simplified in vitro conditions, which primarily reflect specific aspects such as cell viability and migration. Conclusions: This study provides a comparative analysis of physical and covalent crosslinking strategies within a HA platform and highlights how formulation variables influence key physicochemical and biological properties. These findings contribute to the rational design of HA-based hydrogels, although further studies are required to establish their performance in more complex biological environments.
Full article
(This article belongs to the Special Issue Application of Stimuli-Sensitive Hydrogel for the Treatment of Human and Animal Diseases)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Pharmacokinetic and Pharmacodynamic Assessments of the Ivermectin and Levamisole Combination to Control Resistant Nematodes in Cattle
by
Candela Canton, Laura Ceballos, Lucila Canton, Laura Moreno, Paula Domínguez, Luis Alvarez and Carlos Lanusse
Pharmaceutics 2026, 18(5), 630; https://doi.org/10.3390/pharmaceutics18050630 - 21 May 2026
Abstract
Background/Objectives: Combination of antiparasitic drugs with different mechanisms of action has been suggested as an effective strategy to delay the development of parasite resistance. Considering the need to understand the pharmacological basis of drug combinations, the current study evaluated the potential pharmacokinetic (PK)
[...] Read more.
Background/Objectives: Combination of antiparasitic drugs with different mechanisms of action has been suggested as an effective strategy to delay the development of parasite resistance. Considering the need to understand the pharmacological basis of drug combinations, the current study evaluated the potential pharmacokinetic (PK) interactions and the clinical efficacy (pharmacodynamic response) occurring after the subcutaneous administration of ivermectin (IVM) and levamisole (LEV), administered either as single treatments or concurrently to different groups of parasitized calves on three commercial farms (A, B and C). Methods: Forty-five (45) male calves naturally infected with gastrointestinal nematodes were randomly allocated into three groups (n = 15): IVM, treated with IVM by subcutaneous injection (0.2 mg/kg); LEV, treated subcutaneously with LEV (8 mg/kg); IVM + LEV, simultaneously treated with IVM and LEV (two subcutaneous injections at the same dose rates). Seven animals from each treated group (farm C) were randomly selected to perform the PK study. Drug concentrations were measured by HPLC. The therapeutic response (efficacy) was determined at 14 days after treatment by the fecal egg reduction test. Results: The mean area under the concentration vs time curve (AUC) for IVM obtained after administration of IVM alone (274 ± 65.1 ng.d/mL) was similar to that obtained when IVM was co-administered with LEV (295 ± 111 ng.d/mL). Likewise, mean LEV AUC values were similar after LEV administration alone (8.90 ± 2.69 µg.h/mL) or combined with IVM (9.11 ± 1.82 µg.h/mL). No adverse PK interactions were observed after the combined treatment, with similar PK parameters (p > 0.05) obtained between the single-drug and the combination-based strategies. On farm A, the overall fecal egg reductions were 38% (IVM), 99% (LEV) and 100% (IVM + LEV). While Cooperia spp. and Haemonchus spp. showed reduced susceptibility to IVM treatment, LEV demonstrated high efficacy against both genera, with only a minimal proportion of Haemonchus spp. remaining after treatment. Similarly, total fecal egg reductions were 42% (IVM), 99% (LEV) and 100% (IVM + LEV) on farm B, and 54% (IVM), 99% (LEV) and 100% (IVM + LEV) on farm C. On those farms, IVM was ineffective against Cooperia spp. and/or Haemonchus spp., while LEV failed to control Ostertagia spp. Remarkably, the combination of both molecules was the only treatment that achieved 100% efficacy against all nematode genera (Cooperia, Ostertagia, Haemonchus and Oesophagostomum spp.). Conclusions: Based on the described PK and pharmacodynamic (PD) assessments, the IVM + LEV combination appears to be a promising pharmacological option for controlling resistant gastrointestinal nematodes in cattle, with the additional potential to delay the progression of nematode anthelmintic resistance. Overall, the study provides original and robust pharmacokinetic and efficacy data that contribute to the optimization of parasite control strategies in cattle. This drug combination strategy may enhance treatment efficacy and contribute to improved parasite control in cattle production systems.
Full article
(This article belongs to the Section Pharmacokinetics and Pharmacodynamics)
►▼
Show Figures

Figure 1
Open AccessArticle
Expanding the Gene Expression Profiling of Drug Transporters and Drug-Metabolizing Enzymes to Include the Upper Female Reproductive Tract
by
An Le, Guru R. Valicherla, Junmei Zhang, Lin Wang, Mark K. Donnelly, Robert Bies and Lisa C. Rohan
Pharmaceutics 2026, 18(5), 629; https://doi.org/10.3390/pharmaceutics18050629 - 21 May 2026
Abstract
Background/Objectives: With the ongoing efforts in supporting the discovery of novel targeted drug delivery systems for the upper region of the female reproductive tract (FRT), it is imperative to understand the local drug disposition pathways. We aim to obtain a comprehensive profile
[...] Read more.
Background/Objectives: With the ongoing efforts in supporting the discovery of novel targeted drug delivery systems for the upper region of the female reproductive tract (FRT), it is imperative to understand the local drug disposition pathways. We aim to obtain a comprehensive profile of the drug transporters and drug-metabolizing enzymes in the human ectocervix, uterus, and fallopian tubes, as these factors may substantially influence mucosal penetration, tissue exposure, drug disposition, and the risk of drug–drug interactions. Methods: Gene expression of 12 drug transporters and 21 drug-metabolizing enzymes was quantified using RT-qPCR. Protein expression of highly expressed transporters was assessed using immunohistochemistry (IHC). Results: Among the 12 transporters analyzed, the efflux transporters P-gp, BCRP, and MRP4 exhibited the highest expression across the ectocervix, endometrium, myometrium, and fallopian tubes, with P-gp consistently showing the greatest abundance in all evaluated FRT tissues. Expression of these transporters was significantly higher (6–17×) in myometrium compared with ectocervix. IHC demonstrated strong localization of P-gp, BCRP, and MRP4 to epithelial layers facing the lumen, as well as to stromal and vascular endothelial cells. For drug-metabolizing enzymes, all 21 phase I and II enzymes were detectable across the FRT, and 15 were expressed at comparatively higher levels across all tissue types. These included CYP1A1, CYP1B1, CYP2B6, CYP2C8, CYP2C19, CYP3A4, UGT1A1, UGT1A3, UGT1A4, UGT1A7, UGT1A8, UGT1A10, UGT2B4, UGT2B15, and UGT2B17. Conclusions: The gene expression and localization data obtained from this work may improve our understanding of drug disposition in the FRT, which will inform selection, design, and optimization of drugs intended for targeted delivery within the FRT.
Full article
(This article belongs to the Special Issue Understanding Drug Disposition: The Interplay of Absorption, Distribution, and Excretion Processes, and Transport Mechanisms)
►▼
Show Figures
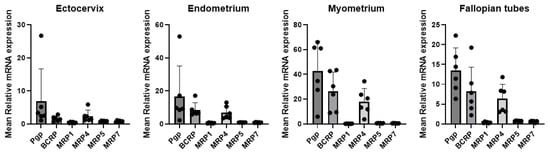
Figure 1
Open AccessArticle
Mucoadhesive Chitosan–Gellan Gum Nanoparticles for Rifampicin Delivery: Taguchi Optimization and In Vitro Release Behavior
by
Siu-Yin Cheung, Aldana Galiyeva, Yerkeblan Tazhbayev, Tolkyn Zhumagaliyeva, Yuliia Bardadym and Vladimir Aseyev
Pharmaceutics 2026, 18(5), 627; https://doi.org/10.3390/pharmaceutics18050627 - 21 May 2026
Abstract
Background/Objectives: Tuberculosis treatment remains challenging due to the limited stability and side effects of conventional rifampicin formulations. This study aimed to synthesize and optimize mucoadhesive chitosan–gellan gum nanoparticles for improved rifampicin delivery. The novelty of this work was the introduction of ethanol into
[...] Read more.
Background/Objectives: Tuberculosis treatment remains challenging due to the limited stability and side effects of conventional rifampicin formulations. This study aimed to synthesize and optimize mucoadhesive chitosan–gellan gum nanoparticles for improved rifampicin delivery. The novelty of this work was the introduction of ethanol into the synthesis process, which improved the solubility of rifampicin and contributed to the formation of nanoparticles with the desired physicochemical characteristics. Methods: Rifampicin-loaded chitosan–gellan gum nanoparticles were produced using the polyelectrolyte complex coacervation method. The polymer ratios, drug-to-polymer ratio, temperature and ethanol volume were the main factors that were optimized using the Taguchi method. The physicochemical properties, such as TGA, DSC and FTIR spectroscopy, were investigated. In addition, drug release, mucoadhesive properties and mycobacterial activity against the H37Rv strain of Mycobacterium tuberculosis were examined. Results: Optimization using the Taguchi method produced nanoparticles with a narrow particle distribution (PDI: 0.212 ± 0.021), a satisfactory average size (153 ± 3 nm) and stability against aggregation (zeta potential: 22.94 ± 1.30 mV). A study of the degree of rifampicin release from nanoparticles showed that the drug release is influenced by pH and has a prolonged effect. Drug-loaded nanoparticles exhibited increased mucoadhesion compared with the pure drug. The minimum inhibitory concentration of rifampicin in chitosan–gellan gum nanoparticles for the suppression of the H37RV strain of Mycobacterium tuberculosis was determined. Spectroscopic and thermal analyses confirmed the incorporation of rifampicin in the polymer matrix. Conclusions: The developed chitosan–gellan gum nanoparticles represent a promising mucoadhesive delivery system for rifampicin. The incorporation of ethanol and the use of Taguchi optimization provide an effective strategy for controlling nanoparticle properties and improving drug delivery performance.
Full article
(This article belongs to the Section Drug Delivery and Controlled Release)
►▼
Show Figures

Figure 1

Journal Menu
► ▼ Journal Menu-
- Pharmaceutics Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
27 January 2026
Meet Us at the 5th Molecules Medicinal Chemistry Symposium, 14–17 May 2026, Beijing, China
Meet Us at the 5th Molecules Medicinal Chemistry Symposium, 14–17 May 2026, Beijing, China

19 May 2026
Interview with Dr. João Carlos Fernandes da Silva—Winner of the Pharmaceutics Young Investigator Award
Interview with Dr. João Carlos Fernandes da Silva—Winner of the Pharmaceutics Young Investigator Award

Topics
Topic in
Biomedicines, Cancers, JCM, Nanomaterials, Pharmaceutics, JNT
Application of Nanomaterials and Nanobiotechnology in Cancer
Topic Editors: Ayan Kumar Barui, Susheel Kumar NethiDeadline: 31 May 2026
Topic in
CIMB, Molecules, Pharmaceuticals, Pharmaceutics, Sci. Pharm.
Challenges and Opportunities in Drug Delivery Research, 2nd Edition
Topic Editors: Lenuta Profire, Ioana Mirela VasincuDeadline: 30 June 2026
Topic in
Bioengineering, Molecules, Pharmaceuticals, Pharmaceutics, Processes, Sci. Pharm.
Complementary Strategies in Drug Delivery: From Particle Engineering to System Optimization
Topic Editors: Barbara R. Conway, Hisham Al-ObaidiDeadline: 30 October 2026
Topic in
Antibiotics, IJMS, Microbiology Research, Pharmaceuticals, Pharmaceutics, Nanomaterials, Microorganisms
Challenges and Future Prospects of Antibacterial Therapy, 2nd Edition
Topic Editors: Kwang-Sun Kim, Zehra EdisDeadline: 30 November 2026

Conferences
10–13 November 2026
The 2nd International Conference on Bioengineering: Bioengineering in an Era of AI

27–29 September 2027
Meet Us Virtually at the 1st International Online Conference on Molecules (IOCMO 2027), 27–29 September 2027

Special Issues
Special Issue in
Pharmaceutics
Innovations in Advanced Drug Delivery Systems: Overcoming Challenges for Personalized Medicine
Guest Editor: Hélcio Silva Dos SantosDeadline: 28 May 2026
Special Issue in
Pharmaceutics
Biomaterials for Drug Delivery and Advanced In Vitro Models in Pharmaceutical Research
Guest Editors: Lorenza Draghi, Clara Mattu, Viola SgarminatoDeadline: 30 May 2026
Special Issue in
Pharmaceutics
Dosage Forms in Drug Delivery: State of the Art and Future Perspectives, 2nd Edition
Guest Editor: Cristina Martín-SabrosoDeadline: 30 May 2026
Special Issue in
Pharmaceutics
Cutting-Edge Liposome Innovations for Precision Delivery and Cancer Treatment
Guest Editors: Sued Eustáquio Mendes Miranda, Anna Eliza Maciel de Faria Mota OliveiraDeadline: 30 May 2026
Topical Collections
Topical Collection in
Pharmaceutics
Women in Pharmaceutics
Collection Editors: Donatella Paolino, Cinzia Anna Ventura
Topical Collection in
Pharmaceutics
Advanced Drug Delivery Systems and Technology in Hungary
Collection Editors: Romána Zelkó, Istvan Antal
Topical Collection in
Pharmaceutics
Advanced Pharmaceutical Research in the Czech Republic
Collection Editors: Jarmila Zbytovska, Jan Gajdziok, Jakub Vysloužil
Topical Collection in
Pharmaceutics
Advanced Pharmaceutical Science and Technology in Portugal
Collection Editors: João Sousa, Carla Vitorino, Alberto A. C. C. Pais







