Abstract
Fluopsin C (FlpC) is an organocupric secondary metabolite with low-molecular-weight, produced by some Pseudomonas and Streptomyces bacteria. The compound was identified in 1970 as prismatic dark-green crystals, with strong antimicrobial activity against several human and phytopathogens. Due to its high cytotoxicity, research on this compound decreased after the 1970s. During the early 2000s, FlpC gained more attention as a promising compound by which to develop new antimicrobials to control human, animal, and plant pathogens. This study provides an overview of the results pertaining to the in vitro and in vivo antimicrobial activity of pure FlpC, as well as semi-purified fractions containing FlpC, against phytopathogenic microorganisms. Furthermore, the bioprospection history of the extensively researched FlpC-producing Pseudomonas aeruginosa LV strain and the related molecular evidence regarding the compound’s biosynthesis are discussed. Overall, FlpC is proposed to be an important alternative to antimicrobial resistance in human and animal health, and in tackling the negative environmental impacts caused by the exacerbated use of pesticides against phytopathogens.
1. Introduction
Plant diseases are one of the main causes of productivity losses in crops, with an estimated impact of one billion dollars per year worldwide. These numbers are attained when the effects of phytopathogens and abiotic stresses are combined [1]. Currently, the use of pesticides is essential for maintaining the yield and quality of different crops. The negative effects of some of these chemicals on human, animal, and plant health have been well described, in addition to their potential to contaminate the soil, surface, and groundwater, affecting non-target vegetation and organisms. However, there is little information regarding the real magnitude of the global negative impacts of their use [2].
Secondary metabolites naturally produced by microorganisms are a major source of bioactive compounds, which are used in many biotechnological products produced in different industrial areas, and especially in new antimicrobial development [3,4]. These bioactive compounds could be a source of new molecules to control microorganisms that affect several crops, avoiding massive losses and presenting a lower environmental impact.
Fluopsin C (FlpC) is a secondary metabolite produced by Pseudomonas spp. or Streptomyces sp. grown in the presence of copper [5,6]. The compound has shown strong antimicrobial activity on a broad-spectrum of human pathogens [7,8,9,10] and phytopathogens [11,12,13,14,15]. Additionally, it has been described as a potential antitumoral agent [16,17].
The study of FlpC decreased after the 1970s due to its elevated cytotoxicity for clinical purposes [7,10], and has increased again over the last few decades with the rise of new technologies for cytotoxicity attenuation and new applications against plant pathogens. In the present review, both the past and present importance of FlpC, and the potential of this compound as a new antimicrobial product against phytopathogens, are discussed.
2. Fluopsin C
The first report of FlpC was published in 1970, in which it was described as a “dark green antibiotic”, purified from cultures of Pseudomonas MCRL 10107, called antibiotic YC 73 [7]. Other research groups called it antibiotic B1 [18,19], and organocopper antibiotic compound (OAC) [14], until the purified molecule was identified as FlpC. A timeline of the studies regarding FlpC is presented in Figure 1. Initially, the interest in FlpC was mainly concentrated on the treatment of human pathogens, focusing on the bioprospecting of FlpC-producing strains, methods of purification, screenings for its antimicrobial activity, cytotoxicity tests, and obtention of less toxic derivatives. This shifted after 2000, with the susceptibility of several plant pathogens determined, once its phytotoxic effects had been determined to be minimal [20,21,22,23,24,25,26,27,28,29,30,31,32,33,34].
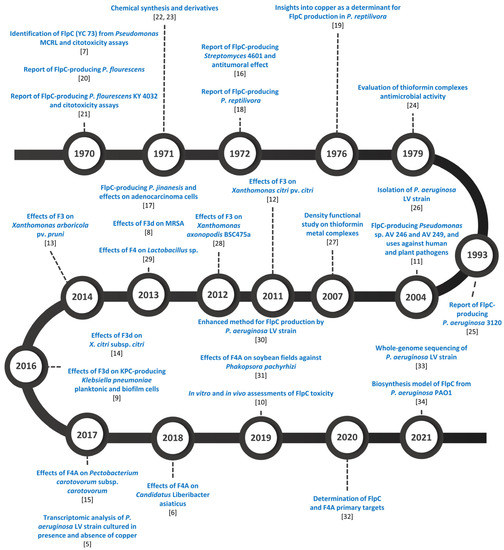
Figure 1.
The timeline of studies related to Fluopsin C research.
FlpC [bis(N-methylthioformohydroxamate)Cu(II)] is a compound with low-molecular-weight (~243,786 g·mol−1), produced in the secondary metabolism of a few species of Pseudomonas and one Streptomyces (Table 1) [6,7,16].

Table 1.
List of Fluopsin C-producing bacteria.
The FlpC crystals (Figure 2A) are thin, prismatic, and are dark-green or brownish in color (Table 2) [7,20]. The compound is formed during a complexation reaction between cupric ions (Cu2+) and two molecules of the siderophore thioformin (N-methylthioformohydroxamic acid) (Figure 2B), an important component in the transport system of ferric ions (Fe3+) through the membrane of algae, fungi, and bacteria [27,35].
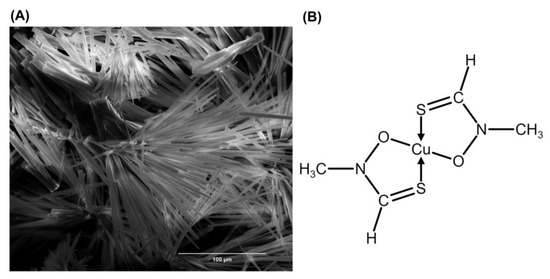
Figure 2.
Fluopsin C. (A) scanning electron microscopy micrography of Fluopsin C crystals; (B) molecular structure of Fluopsin C (C4H8N2O2S2Cu), a coordination compound formed by two molecules of the thiohydroxamic siderophore thioformin (C2H5NOS) bonded with one central metal ion of copper.

Table 2.
Main organometallic complexes in the Fluopsin group (thioformin complexes).
Several metallic ions are able to complex with thioformin, forming Fluopsins that present antimicrobial activity [27], as shown in Table 2. These complexes are obtained by microbial biosynthesis or chemical synthesis, including derivative molecules [23,24,36,37].
3. Fluopsin C-Producing Microorganisms
Until now, a few bacteria have been reported as FlpC producers, and almost all strains are from the Pseudomonas genus, especially the P. aeruginosa LV strain, which is the most studied strain.
The P. aeruginosa LV strain was isolated from old leaf lesions of orange (Citrus sinensis cv. Valencia) with citrus canker disease at Astorga city (Paraná State, Brazil) in 2004 [26]. Recently, its genome has been sequenced (GenBank CP058323) [33].
Curiously, in the orchard where LV strain and other bacterial strains were isolated, the control of citrus canker disease was carried out by spraying copper chloride solution on leaves and fruits. Most likely, the presence of a large amount of copper chloride selected tolerant bacteria strains, co-existing with Xanthomonas citri pv. citri inside the citrus canker lesions. The mechanisms of the LV strain to protect itself against copper chloride are proposed and are discussed here.
Molecular Insights on Fluopsin C Biosynthesis
The process of FlpC production by bacteria has not been completely elucidated, and efforts have been made using molecular biology to identify the genes involved in the biosynthesis pathway.
It is known that the presence of copper (Cu2+) in the culture media is a trigger for FlpC production. Transcriptome analysis of the P. aeruginosa LV strain, cultivated in the presence and absence of CuCl2, indicated that nine genes were differentially expressed (hyperexpressed) when compared with bacteria cultivated in the absence of CuCl2 [5]. The analysis also showed that the genes should be functionally grouped as metallic ions transporters (PA3574a; PA3920; PA3523; PA3251); transcriptional regulators (PA4878); phenazines biosynthesis (phzA2); and hypothetical proteins (PA2691; PA4141; PA4782).
Most of the detected hyperexpressed genes were related to metallic ions transport systems, strongly indicating that FlpC biosynthesis occurred as a bacterial bioremediation mechanism, to protect itself against high intracellular levels of copper [5]. The ions cause high cytotoxicity, disrupting iron-sulfur enzyme complexes and generating reactive oxygen species (ROS) [38].
The phzA2 gene belongs to phz2 operon, which is responsible for basal production of phenazine-1-carboxylic acid (PCA). The PCA acts as a signaling molecule, inducing phz2 operon transcription and the expression of phz1 operon, increasing PCA biosynthesis [39].
Based on the experimental data described above, the current hypothesis considers that PCA should be a metabolic precursor of thioformin. However, it is still unclear which precursors participate in FlpC biosynthesis. The synthesis of FlpC remains under investigation, and future studies comprising genetic silencing will indicate the action of those hyperexpressed genes detected in the FlpC pathway.
On the other hand, in the presence of elevated concentrations of copper, the transcriptome analysis of P. aeruginosa PAO1 revealed the hyperexpression of different operons, such as PA3515-3519 (which encodes five proteins with unknown functions) [40], copZ1 (which encodes a copper chaperone), and PA3521-PA3523 (which encodes an efflux pump) [38]. The presence of copper was detected in the cytoplasm by the Cu-sensing transcriptional regulator CueR, which activates these genes to re-establish copper homeostasis [34,38,41].
Patteson et al. [34] investigated if PA3515-3519 operon proteins could be responsible for the synthesis of FlpC. By the heterologous expression of the operon in P. fluorescens SBW25 cultured with CuSO4, it was possible to confirm that it contained all the necessary genes for FlpC production, being named as the flc gene cluster (PA3515-3519 as flcA to flcE).
After cluster determination, P. aeruginosa PAO1 was cultured in M9 minimal medium with CuSO4 and [15N,13C5] L-methionine or [15N,13C3] L-cysteine, in order to evaluate the incorporation of the amino acids into FlpC, using Liquid Chromatography-High Resolution Mass Spectrometry (LC-HRMS) analysis. The results indicated the incorporation of one carbon from L-Met (possibly for the N-methyl group), and then one carbon and one nitrogen from L-Cys (possibly to form the thiohydroximate backbone) to the antibiotic [34]. These findings enabled the investigation of each reaction involved in FlpC production and the proposition of a biosynthesis pathway model.
In the proposed model by Patteson et al. [34] (Figure 3), the biosynthesis of FlpC starts by FlcB catalyzing the conjugation of L-cysteine with fumarate, forming the precursor 2 (C7H12NO6S+; 238.040 m/z). Then, the heme oxygenase FlcE catalyzes oxidative decarboxylation and N-hydroxylation reactions converting 2 to 3 (C6H10NO5S+; 208.028 m/z). FlcD, another heme oxygenase protein, catalyzes a methylene excision in the 2-C of 3, forming 4 (C5H8NO5S+; 194.012 m/z) and formate. The lyase FlcC catalyzes the cleavage of the C-S bond of 4, forming 5 (thiohydroxamate) and fumarate, the latter being recycled for the first step of the pathway. Finally, FlcA methylates 5, in the presence of SAM (S-adenosyl-L-methionine), generates 6 (Cu-free fluopsin), that binds to the surrounding copper ions. Additionally, the omission of any Flc enzyme fully depletes the production of FlpC.
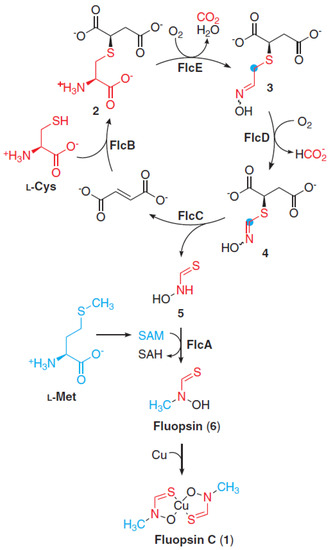
Figure 3.
Fluopsin C biosynthetic pathway model highlighting the reactions catalyzed by the Flc enzymes. Reprinted with permission from AAAS. Copyright 2021 from ref. [34]. The structural contribution of L−Cys and L−Met are highlighted by the colors red and blue, respectively. Each molecular potential precursor was named as molecules 1-6.SAM: S−adenosyl−L−methionine; SAH: S−adenosyl−L−homocysteine.
When comparing genomes using a Pseudomonas genome database, the flc cluster was present in 99.6% of the 4955 annotated genomes from environmental and clinical isolates; additionally, the cluster was also present in Acinetobacter baumannii, Enterobacter cloacae, Streptococcus dysgalactiae (clinical isolates), Lysobacter enzymogenes, and Lysobacter capsica (bacteria colonizing roots and leaves) [34].
Different patterns of genetic components were obtained by transcriptomic analysis between LV and PAO1 strains of P. aeruginosa, generating two current hypotheses for the synthesis of FlpC. Further studies should allow for the determination of which one is the most accurate, or if the synthesis of the compound comprises different metabolic pathways depending on the bacterial strain.
4. Fluopsin C In Vitro and In Vivo Inhibitory Effects on Phytopathogens
Fluopsins are promising antimicrobial molecules [22], especially FlpC, which controls important clinical and phytopathogenic microorganisms.
Recent studies have used semi-purified fractions containing FlpC, obtained by organic solvent extraction of P. aeruginosa LV strain culture supernatant, followed by column chromatography, vacuum liquid chromatography (VLC), and flash chromatography (FC).
Each step generated specific fractions, containing different amounts of FlpC and other compounds. FlpC was found in fractions named as F3 [8,12,13], F3d (30% FlpC) [9,14], and F4A (25% FlpC) [6,15,32].
The use of FlpC against phytopathogens is a more recent approach, and a restricted number of susceptible bacteria, fungi, and oomycetes have been reported to date (Table 3).

Table 3.
Susceptibility of phytopathogens to Fluopsin C and to semi-purified fractions (F3, F3d, and F4A).
X. citri pv. citri is the causal agent of citrus canker disease that affects fruit, leaves, and young stems of citrus plants, promoting lesions that reduce fruit quality and production. Scanning electron microscopy (SEM) analysis of X. citri pv. citri cultures treated for 1 h with F3 MIC (minimum inhibitory concentration) revealed reduced exopolysaccharides (EPS) content, without cellular disruption. In 3–6 h treatments, the EPS decreased, and cells were shrunken and rough [12].
In a transmission electron microscopy (TEM) examination, after 1 h of treatment with F3, X. citri pv. citri cells were elongated, and membrane alterations were observed when compared to the control. After 3 h, the cell wall and cytoplasmatic membrane were disrupted, evolving to cell lysis after 6 h.
The same study also reported that, in the orange tree (C. sinensis cv. Valencia) infection model, the application of 10 μg·mL−1 of F3 solution reduced leaf lesions by around 93.5% in preventive and curative treatments based on F3 aspersion.
In addition, the F3 fraction showed antibiotic activity against leaf blight disease caused by Xanthomonas axonopodis in Eucalyptus spp. (MIC of 100 μg·mL−1), which causes lesions on leaves that evolve necrosis until the leaves fall, reducing the photosynthetic area and plant growth [28]. In SEM, the authors observed that after 1 h of treatment with 200 μg·mL−1 F3d, no EPS was observed in cultures of X. axonopodis, and after 3 h, the cells were completely disrupted. The treatment of Eucalyptus urograndis 1404 seedlings with 1000 μg·mL−1 of F3, previous to or after pathogen inoculation, reduced foliar lesions by 93.9% and 89.7%, respectively, 30 days after treatment.
Similar studies were carried out using X. arboricola pv. pruni, one of the major phytopathogens of peach orchards, which causes bacterial spot disease [13]. Cultures treated with 200 μg·mL−1 of F3 showed the same positive effects observed in other studies.
Regarding the reduction of bacterial spot leaf lesions promoted by F3 on peach trees (Prunus persica cv. Maciel), no significant differences were found by spraying leaves with 150 or 450 μg·mL−1, as a post- or pre-treatment, and both concentrations reduced the lesions by >70.0%. However, the higher concentrations used in the study promoted phytotoxic effects on peach leaves.
Once it was determined that the F3 fraction was promising against phytopathogens, a new system of purification was developed to achieve a higher resolution in the purification of antimicrobial molecules. Oliveira et al. [14] partitioned F3 into nine new fractions, in which F3d presented increased antimicrobial activity; it was composed by two molecules, phenazine-1-carboxamide (PCN) and OAC. The OAC was determined later as FlpC, being 30% of F3d mass.
Using F3d, preventive and curative regimens against X. citri pv. citri were reassessed. In a preventive regimen, 1 μg·mL−1 of F3d led to a lesion-reducing effect of 90.0%, 10 μg·mL−1 reduced 93.0%, and 100 μg·mL−1 reduced 97.0%, with the preventive approach being more effective than curative treatments.
After 120 h of preventive treatment, just a few cells of the phytopathogen were found in the mesophyll, presenting morphological changes, cell lysis, and reduced EPS. Mesophyll cells seemed intact during the entire treatment. Additionally, the activity of the compound was maintained for weeks on the phylloplane and inside the leaves, reducing the potential inoculum and the phytopathogen transmission, without phytotoxic effects.
F4A is a semi-purified fraction obtained by a new system of VLC purification method, which provides a fraction with 25% FlpC (similar to F3d content). The antibiotic activity of the F4A fraction was evaluated against Pectobacterium carotovorum subsp. carotovorum, and F4A promoted bacterial cell lysis after 3 h of treatment, verified by SEM. In tomato plants (Solanum lycopersicum cv. Santa Clara), the preventive regimen provided a survival rate of 100.0% of plants using 7, 70, and 700 μg·mL−1, without displaying any signs of cytotoxicity. In addition, plants preventively treated with 70 μg·mL−1 recorded the highest productivity, both in terms of weight and number of fruits. The curative regimen promoted 20.0% plant survival with 70 and 700 μg·mL−1, and 100.0% with 7 μg·mL−1. Plants treated with lower doses reached the highest growth when compared with other treatments [15].
Pistori et al. [6] investigated the effect of F4A foliar aspersions on the non-axenic culturable Candidatus Liberibacter asiaticus (causal agent of Huanglongbing) in C. sinensis cv. Valencia infection. Preventive treatments with 100 μg·mL−1 of F4A led to only 14.2% of the citrus trees being positive for pathogen presence after 120 days of bud-grafting inoculation; after 240 days, 57.4% of the trees were positive. Curative treatments at the same concentration demonstrated that 0.0% and 42.8% of the trees were positive for the pathogen after 120 and 240 days, respectively.
The fraction was also indicated as a potential elicitor for the plant systemic acquired resistance (SAR) in orange plants against Huanglongbing, based on the increase of PR-2 gene expression (codifies β-1,3 glucanase). This increase was associated with the presence of the phenazines PCA and PCN in F4A, indicating that the different molecules contained in the semi-purified fraction may synergically act in producing multiple defensive responses against phytopathogens.
More recently, a study dedicated to the investigation of ultrastructural effects promoted by pure FlpC and F4A indicated that they are similar (Table 4) [32]. Additionally, damage to the cytoplasmatic membrane was also assessed by live/dead staining after 15 min exposure to FlpC and F4A, indicating severe damage to the structure by considerably increasing its permeability. The results corroborated the current hypothesis that the main action site of FlpC on Gram-negative and Gram-positive bacteria is the cytoplasmatic membrane, which becomes more permeable [32].

Table 4.
Ultrastructural effects in microbial cells exposed to Fluopsin C and F4A fraction analyzed by scanning (SEM) and transmission electron microscopy (TEM).
A secondary effect observed in X. citri subsp. citri 306 was the dissolution of the Z-ring, impairing cell septation and, consequently, the cell division process. This may occur due to the breakdown of the membrane potential, which is essential for positioning FtsZ proteins on the cell division axis. Still, no effects were observed regarding the genetic content or the chromosomal segregation process [32].
Under field conditions, F4A-treated soybeans presented decreased infection by Phakopsora pachyrhizi (Asian soybean rust), and when an association of 10 μg·mL−1 F4A plus Sphere Max® (Bayer, São Paulo, Brazil) (a commercial fungicide) was used, the arbuscular mycorrhizal fungi colonization was higher in treated plants (77.84%) compared to other treatments. The nitrogen and phosphorous foliar content increased in treated plants, as well as in terms of seed proteins, plant height, and yield (4.45 ton.ha−1). The combination of F4A (10 μg·mL−1) and Sphere Max® promoted higher fungicide activity on P. pachyrhizi, with more dead spores and hyphae. However, treatments consisting of F4A alone were unable to control the disease [31].
5. Conclusions
The development of new antimicrobial agents derived from natural sources is crucial to enhance the control of human, animal, and plant pathogens that are resistant to currently used compounds. In crops, they can also represent new products to control different plant diseases, reducing productivity losses and the negative environmental impacts of chemical pesticides.
FlpC is a promising antimicrobial bio-compound produced by bacteria. However, the number of studies on this molecule is still relatively low, especially considering its cytotoxicity. This is probably the main reason why pioneering research groups neglected investigations on this compound for so long. Research regarding FlpC has reemerged during the last decade, with several studies focusing on the antimicrobial activity of different human and phytopathogens under in vitro and in vivo conditions.
To date, results have indicated that FlpC applications in plant protection are promising and require a low concentration of the compound to be effective. Furthermore, when combined with other secondary metabolites or commercial products, it may synergistically act in promoting multiple beneficial responses.
Still, no attempts have been made to evaluate the environmental changes promoted by the introduction of FlpC in crops. We hypothesize that, due to the molecule’s natural occurrence, which may be frequent in areas where copper solutions are used for phytopathogens management, its biocompatibility is suitable, since these areas operate without reported damage. However, as a broad-spectrum antimicrobial, inappropriate and excessive use of the compound could alter the composition of soil microbiota.
In the future, FlpC should have its environmental impacts determined, especially regarding depletory effects on soil communities, its degradation time and accumulation, its runoff potential towards superficial and underground water, and its effect on non-targeted organisms. Regarding FlpC cytotoxicity, the use in formulations with addition of other compounds could be a useful tool against plant diseases.
There is still a lot of research to be conducted regarding FlpC, but the great potential of this compound is undeniable. In the last few decades, many studies have been conducted, but these only represent a beginning in explorations of the potential of FlpC.
Author Contributions
L.A., S.F.A., A.P.d.N., J.V.F.R.d.S., G.d.S.G. and L.D.d.S.A. researched data for the article; L.A. wrote the article; M.F.d.L.A., A.S.S. and M.V.T.C. substantially contributed to discussion and organization of content; A.L.C. and G.A. reviewed/edited the manuscript before submission. All authors have read and agreed to the published version of the manuscript.
Funding
This article was funded by the Brazilian National Council for Scientific and Technological Development (CNPq-AMR MCTIC process 439754/2018-6).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Martins, P.M.M.; Merfa, M.V.; Takita, M.A.; De Souza, A.A. Persistence in Phytopathogenic Bacteria: Do we Know Enough? Front. Microbiol. 2018, 9, 1099. [Google Scholar] [CrossRef] [PubMed]
- Hough, R.L. A World View of Pesticides. Nat. Geosci. 2021, 14, 183–184. [Google Scholar] [CrossRef]
- Gokulan, K.; Khare, S.; Cerniglia, C. Metabolic Pathways: Production of Secondary Metabolites of Bacteria. In Encyclopedia of Food Microbiology, 2nd ed.; Batt, C.A., Tortorello, M.L., Eds.; Academic Press: Cambridge, MA, USA, 2014; Volume 2, pp. 561–569. ISBN 978-0-19-969739-7. [Google Scholar]
- Patrick, G.L. An Introduction to Medical Chemistry, 5th ed.; Oxford University Press: Oxford, UK, 2013; pp. 199–213. ISBN 978-0-19-969739-7. [Google Scholar]
- Gionco, B.; Tavares, E.R.; Oliveira, A.G.; Yamada-Ogatta, S.F.; Carmo, A.O.; Pereira, U.P.; Chideroli, R.T.; Simionato, A.S.; Navarro, M.O.P.; Chryssafidis, A.L.; et al. New Insights about Antibiotic Production by Pseudomonas aeruginosa: A Gene Expression Analysis. Front. Chem. 2017, 5, 66. [Google Scholar] [CrossRef] [PubMed]
- Pistori, J.F.; Simionato, A.S.; Navarro, M.O.P.; Andreata, M.F.L.; Santos, I.M.O.; Meneguim, L.; Leite, R.P., Jr.; Oliveira, A.G.; Andrade, G. Low-molecular-weight Metabolites Produced by Pseudomonas aeruginosa as an Alternative to Control Huanglongbing in Citrus sinensis cv. Valencia. Trop. Plant Pathol. 2018, 43, 289–296. [Google Scholar] [CrossRef]
- Egawa, Y.; Umino, K.; Awataguchi, S.; Kawano, Y.; Okuda, T. Antibiotic YC 73 of Pseudomonas origin. I. Production, Isolation and Properties. J. Antibiot. 1970, 23, 267–270. [Google Scholar] [CrossRef]
- Cardozo, V.F.; Oliveira, A.G.; Nishio, E.K.; Perugini, M.R.; Andrade, C.G.; Silveira, W.D.; Durán, N.; Andrade, G.; Kobayashi, R.K.; Nakazato, G. Antibacterial Activity of Extracellular Compounds Produced by a Pseudomonas Strain against Methicillin-resistant Staphylococcus aureus (MRSA) strains. Ann. Clin. Microbiol. Antimicrob. 2013, 12, 12. [Google Scholar] [CrossRef]
- Kerbauy, G.; Vivan, A.C.P.; Simões, G.C.; Simionato, A.S.; Pelisson, M.; Vespero, E.C.; Costa, S.F.; Andrade, C.G.T.J.; Barbieri, D.M.; Mello, J.C.P.; et al. Effect of a Metalloantibiotic Produced by Pseudomonas aeruginosa on Klebsiella pneumoniae Carbapenemase (KPC)-producing K. pneumoniae. Curr. Pharm. Biotechnol. 2016, 17, 389–397. [Google Scholar] [CrossRef]
- Navarro, M.O.P.; Simionato, A.S.; Pérez, J.C.B.; Barazetti, A.R.; Emiliano, J.; Niekawa, E.T.G.; Andreata, M.F.L.; Modolon, F.; Dealis, M.L.; Araújo, E.J.A.; et al. Fluopsin C for Treating Multidrug-resistant Infections: In vitro Activity against Clinically Important Strains and in vivo Efficacy Against Carbapenemase-producing Klebsiella pneumoniae. Front. Microbiol. 2019, 10, 2431. [Google Scholar] [CrossRef]
- Hedman, R.; Levenfors, J.; Stanislaw, P.; Welch, C.; Daugherty, S. Use of a Known Substance for the Treatment of Cancer and Bacterial and Fungal Infections. WO 2004050095 A1. 2004. Available online: https://patents.google.com/patent/WO2004050095A1/en (accessed on 21 October 2022).
- Oliveira, A.G.; Murate, L.S.; Spago, F.R.; Lopes, L.P.; Beranger, J.P.O.; Martin, J.A.B.S.; Nogueira, M.A.; Mello, J.C.P.; Andrade, C.G.T.J.; Andrade, G. Evaluation of the Antibiotic Activity of Extracellular Compounds Produced by the Pseudomonas Strain Against the Xanthomonas citri pv. citri 306 Strain. Biol. Control 2011, 56, 125–131. [Google Scholar] [CrossRef]
- Vasconcellos, F.C.S.; Oliveira, A.G.; Lopes-Santos, L.; Beranger, J.P.O.; Cely, M.V.T.; Simionato, A.S.; Pistori, J.F.; Spago, F.R.; Mello, J.C.P.; San Martin, J.A.B.; et al. Evaluation of Antibiotic Activity Produced by Pseudomonas aeruginosa LV Strain Against Xanthomonas arboricola pv. pruni. Agric. Sci. 2014, 5, 71–76. [Google Scholar] [CrossRef][Green Version]
- Oliveira, A.G.; Spago, F.R.; Simionato, A.S.; Navarro, M.O.P.; Silva, C.S.; Barazetti, A.R.; Cely, M.V.T.; Tischer, C.A.; San Martin, J.A.B.; Andrade, C.G.T.J.; et al. Bioactive Organocopper Compound from Pseudomonas aeruginosa Inhibits the Growth of Xanthomonas citri subsp. citri. Front. Microbiol. 2016, 7, 113. [Google Scholar] [CrossRef] [PubMed]
- Munhoz, L.D.; Fonteque, J.P.; Santos, I.M.O.; Navarro, M.O.P.; Simionato, A.S.; Goya, E.T.; Rezende, M.I.; Balbi-Peña, M.I.; Oliveira, A.G.; Andrade, G. Control of Bacterial Stem Rot on Tomato by Extracellular Bioactive Compounds Produced by Pseudomonas aeruginosa LV strain. Cogent Food Agric. 2017, 3, 1282592. [Google Scholar] [CrossRef]
- Miyamura, S.; Ogasawara, N.; Otsuka, H.; Niwayama, S.; Tanaka, H.; Take, T.; Uchiyama, T.; Nakazawa, H.; Abe, K.; Koizumi, K. An Antitumor Antibiotic, no. 4601 from Streptomyces, Identical with YC 73 of Pseudomonas origin. J. Antibiot. 1972, 25, 369–370. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Jiang, C.; Cui, M.; Lu, R.; Liu, S.; Zheng, B.; Li, L.; Li, X. Fluopsin C Induces Oncosis of Human Breast Adenocarcinoma Cells. Acta Pharmacol. Sin. 2013, 34, 1093–1100. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, L.A.; Gorgé, J.L.; Olivares, J.; Mayor, F. Antibiotics from Pseudomonas reptilivora II. Isolation, Purification, and Properties. Antimicrob. Agents Chemother. 1972, 2, 189–194. [Google Scholar] [CrossRef][Green Version]
- Martinez-Molina, E.; Del Rio, L.A.; Olivares, J. Copper and Iron as Determinant Factors of Antibiotic Production by Pseudomonas reptilivora. J. Appl. Bacteriol. 1976, 41, 69–74. [Google Scholar] [CrossRef]
- Shirahata, K.; Deguchi, T.; Hayashi, T.; Matsubara, I.; Suzuki, T. The Structures of Fluopsins C and F. J. Antibiot. 1970, 23, 546–550. [Google Scholar] [CrossRef]
- Itoh, S.; Inuzuka, K.; Suzuki, T. New Antibiotics Produced by Bacteria Grown on n-paraffin (Mixture of C12, C13 and C14 Fractions). J. Antibiot. 1970, 23, 542–545. [Google Scholar] [CrossRef]
- Egawa, Y.; Umino, K.; Ito, Y.; Okuda, T. Antibiotic YC 73 of Pseudomonas origin. II. Structure and Synthesis of Thioformin and its Cupric Complex (YC 73). J. Antibiot. 1971, 24, 124–130. [Google Scholar] [CrossRef][Green Version]
- Shirahata, K.; Hayashi, T.; Matsubara, I. Syntheses of Fluopsins. J. Antibiot. 1971, 24, 140–141. [Google Scholar] [CrossRef][Green Version]
- Bell, S.J.; Friedman, S.A.; Leong, J. Antibiotic Action of N -Methylthioformohydroxamate Metal Complexes. Antimicrob. Agents Chemother. 1979, 15, 384–391. [Google Scholar] [CrossRef] [PubMed]
- Ko, H.R.; Chun, H.K.; Kho, Y.H.; Sung, N.K. Production, Isolation and Characterization of the Antibiotic from Pseudomonas aeruginosa 3120. Appl. Biol. Chem. 1993, 36, 428–433. [Google Scholar]
- Rampazo, L.G.L. Evaluation of Biological Agents and their Products on the Incidence of Citrus Canker Foliar Lesions. Master’s Thesis, State University of Londrina, Londrina, Brazil, 2004. [Google Scholar]
- Kakkar, R.; Dua, A.; Gahlot, P. Metal Ion Complexes of Thioformin: A Density Functional Study. Polyhedron 2007, 26, 5301–5308. [Google Scholar] [CrossRef]
- Lopes, L.P.; Oliveira, A.G.; Beranger, J.P.O.; Góis, C.G.; Vasconcellos, F.C.S.; Martin, J.A.B.S.; Andrade, C.G.T.J.; Mello, J.C.P.; Andrade, G. Activity of Extracellular Compounds of Pseudomonas sp. against Xanthomonas axonopodis in vitro and Bacterial Leaf Blight in Eucalyptus. Trop. Plant Pathol. 2012, 37, 233–238. [Google Scholar] [CrossRef][Green Version]
- Góis, C.G.M.; Lopes-Santos, L.; Beranger, J.P.O.; Oliveira, A.G.; Spago, F.R.; Andrade, G. The Control of Lactobacillus sp. by Extracellular Compound Produced by Pseudomonas aeruginosa in the Fermentation Process of Fuel Ethanol Industry in Brazil. J. Sustain. Bioenergy Syst. 2013, 3, 194–201. [Google Scholar] [CrossRef]
- Bedoya, J.C.; Dealis, M.L.; Silva, C.S.; Niekawa, E.T.G.; Navarro, M.O.P.; Simionato, A.S.; Modolon, F.; Chryssafidis, A.L.; Andrade, G. Enhanced Production of Target Bioactive Metabolites Produced by Pseudomonas aeruginosa LV Strain. Biocatal. Agric. Biotechnol. 2019, 17, 545–556. [Google Scholar] [CrossRef]
- Barazetti, A.R.; Simionato, A.S.; Navarro, M.O.P.; Dealis, M.L.; Matos, J.M.S.; Modolon, F.; Andreata, M.F.L.; Liuti, G.; Andrade, G. Evaluation of Bioproducts and Mycorrhizal Inoculation in Asian Soybean Rust Control, Nutrient Leaf Contents and Yield Under Field Conditions. In Microbial Probiotics for Agricultural Systems, 1st ed.; Zúñiga-Dávila, D., González-Andrés, F., Ormeño-Orrillo, E., Eds.; Springer: Cham, Switzerland, 2019; Volume 1, pp. 193–204. ISBN 978-3-030-17597-9. [Google Scholar]
- Navarro, M.O.P.; Dilarri, G.; Simionato, A.S.; Grzegorczyk, K.; Dealis, M.L.; Cano, B.G.; Barazetti, A.R.; Afonso, L.; Chryssafidis, A.L.; Ferreira, H.; et al. Determining the Targets of Fluopsin C Action on Gram-negative and Gram-positive Bacteria. Front. Microbiol. 2020, 11, 1076. [Google Scholar] [CrossRef]
- Simionato, A.S.; Cano, B.G.; Navarro, M.O.P.; Tavares, E.R.; Ribeiro, R.A.; Hungria, M.; Yamauchi, L.M.; Yamada-Ogatta, S.F.; Andrade, G. Whole-genome Sequence of Bioactive Compound-producing Pseudomonas aeruginosa Strain LV. Microbiol. Resour. Announc. 2021, 10, e01120-20. [Google Scholar] [CrossRef]
- Patteson, J.B.; Putz, A.T.; Tao, L.; Simke, W.C.; Bryant, L.H.; Britt, R.D.; Li, B. Biosynthesis of Fluopsin C, a Copper-containing Antibiotic from Pseudomonas aeruginosa. Science 2021, 374, 1005–1009. [Google Scholar] [CrossRef] [PubMed]
- Leong, J.; Bell, S.J. Coordination Isomers of Antibiotic Thiohydroxamate-metal Complexes. Geometrical Isomers of tris(N-methylthioformohydroxamato)rhodium(III) and bis(N-methylthioformohydroxamato)platinum(II). Inorg. Chem. 1978, 17, 1886–1892. [Google Scholar] [CrossRef]
- Ito, Y.; Umino, K.; Sekiguchi, T.; Miyagishima, T.; Egawa, Y. Antibiotic YC 73 of Pseudomonas origin. III. Synthesis of Thioformin Analogues. J. Antibiot. 1971, 24, 131–134. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Miyagishima, T.; Yamaguchi, T.; Umino, K. Further Studies on Synthesis and Antimicrobial Activity of Thioformin Analogues. Chem. Pharm. Bull. 1974, 22, 2283–2287. [Google Scholar] [CrossRef][Green Version]
- Quintana, J.; Novoa-Aponte, L.; Argüello, J.M. Copper Homeostasis Networks in the Bacterium Pseudomonas aeruginosa. J. Biol. Chem. 2017, 292, 15691–15704. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Du, X.; Lu, Z.J.; Wu, D.; Zhao, Y.; Ren, B.; Huang, J.; Huang, X.; Xu, Y.; Xu, Y. Regulatory Feedback Loop of two phz Gene Clusters through 5′-untranslated Regions in Pseudomonas sp. M18. PLoS ONE 2011, 6, e19413. [Google Scholar] [CrossRef] [PubMed]
- Teitzel, G.M.; Geddie, A.; De Long, S.K.; Kirisits, M.J.; Whiteley, M.; Parsek, M.R. Survival and Growth in the Presence of Elevated Copper: Transcriptional Profiling of Copper-stressed Pseudomonas aeruginosa. J. Bacteriol. 2006, 188, 7242–7256. [Google Scholar] [CrossRef] [PubMed]
- Thaden, J.T.; Lory, S.; Gardner, T.S. Quorum-sensing Regulation of a Copper Toxicity System in Pseudomonas aeruginosa. J. Bacteriol. 2010, 192, 2557–2568. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).