Abstract
Brewers’ spent grain (BSG) represents the major byproduct of the brewing industry and remains largely underutilized. While BSG contains a rather high amount of protein, poor functional properties limit its use as a functional ingredient for foods without additional processing. In this work, we investigate emulsifying peptides embedded in the major BSG proteins based on a mass spectrometry-based proteomic analysis and subsequent bioinformatic prediction to explore the utilization of BSG as a raw material for the production of protein-based emulsifying ingredients. Forty-eight peptides were selected based on EmulsiPred score, amino acid sequence, and protein abundance for evaluation. All peptides effectively reduced the interfacial tension between oil–water, but only 15 could produce and stabilize emulsions with droplet sizes below 5 µm. Some peptides were able to produce stable emulsions with sub-micron droplet sizes, implying very promising emulsifying properties. This study demonstrated promising emulsifying properties of BSG peptides and suggested that the functionality could be predicted using bioinformatic tools. However, the used tool needs to be further optimized for higher success rate.
1. Introduction
Brewers’ spent grain (BSG) is the main byproduct of the brewing industry. During the brewing process, malt is mixed with water. Thereafter, the mixture is stirred under elevated temperature in a process called mashing. The mashing parameters, e.g., time and temperature, are dependent on the brewery and the type of beer. During the mashing step, different enzymes are activated in order to hydrolyze starch into fermentable and non-fermentable sugars like dextrans [1]. The sugar-rich liquid, now called wort, is separated and processed into beer. The remaining solid side stream constitutes the BSG [1]. BSG is a lignocellulosic material mainly composed of husk, pericarp, and seed coat of barley malt. Depending on the beer type, other cereals like rice, corn, or wheat are also added during mashing, and compounds from these ingredients will be present in the final BSG [1,2]. BSG consists of between 15 and 30 dw% protein, which makes it a very interesting raw material for protein extraction and utilization [3,4,5]. BSG constitutes around 85% of the total waste of the brewing process, and better utilization of it could help reduce the environmental impact and increase the sustainability of the brewing process [6]. Especially when considering that 20 kg of wet BSG, equal to roughly 6.2 kg of dry BSG, is generated for every 100 L of beer produced [7]. Utilization of BSG has gained increased attention lately for various materials, including flour replacement in baking, and as chips and snacks, and more recently, protein extraction has also been studied [3,6]. BSG could also be used as a raw material for the extraction of protein or peptide ingredients like emulsifiers.
Emulsifiers are amphiphilic molecules that are able to stabilize oil–water mixtures by minimizing the interfacial tension between the two phases and forming a protective layer around the droplets that prevents aggregation [8]. They are vital molecules in the food industry and are used in a wide range of foods such as bakery products, ice cream, drinks, and sauces. Many of the emulsifiers currently used are either synthetic surfactants, like Tweens, or animal-based emulsifiers such as egg proteins and lecithin, whey proteins, and caseinate. Lately, plant proteins and hydrolysates have gained increasing attention in the literature, industry, and from consumers [9]. The growing demand is due to increased awareness of sustainability and health impact [10]. Plant proteins normally have lower emulsification capabilities when compared with animal proteins, which can minimize their usage [11]. However, by hydrolyzing the proteins to produce peptides, the emulsifying properties and solubility can be improved compared to the native protein [12,13].
Identifying and testing new emulsifying ingredients can be a tedious and costly process. However, with the aid of proteomics and bioinformatics, the screening process could become more efficient. EmulsiPred (https://github.com/MarcatiliLab/EmulsiPred (accessed on 15 December 2025)) is an open-source bioinformatics tool used to predict the emulsifying potential of protein materials. EmulsiPred uses the amino acid (AA) sequence and its projection within a probable secondary structure to calculate an amphiphilic vector across AA sequences. Based on the assumption that peptides’ emulsifying activity is correlated to their amphiphilicity, peptide sequences with high amphiphilic vectors can be identified [14]. Furthermore, through targeted enzymatic hydrolysis, these peptides can be released in higher quantities [15]. By doing so, many costly trial-and-error experiments could be eliminated. EmulsiPred has previously been used with success to predict the emulsifying activity of peptides derived from seaweed, potato, and single-cell protein, and showed the potential of these as untapped natural emulsifiers [14,16,17].
Extractable protein from BSG has been applied as an emulsifier with varying results. Previously, we showed that BSG protein extracts obtained using different extraction techniques, either at neutral or at slightly alkaline conditions (pH 9), could form emulsions. However, the emulsion stability was poor [18]. Other than this, only a few studies have investigated the emulsifying activity of BSG proteins and protein hydrolysates [19,20,21]. Connolly et al. [20] tested alkaline-extracted and acid-precipitated protein isolates and their hydrolysates, and concluded that BSG-protein isolates and hydrolysates have potential as a techno-functional ingredient. A similar conclusion was made by Celus et al. [19], who showed that BSG-protein hydrolysates increased the emulsifying activity compared to native BSG. Nevertheless, these studies hydrolyzed BSG using a conventional trial-and-error approach, where the quantitative protein-level composition and sequence information thereof are not considered. While this approach is frequently used and considered the conventional process for protein hydrolysate production [22], the functionality of a hydrolysate can be improved by improving the understanding of the peptides that may be produced [15].
The aim of this study was to evaluate the potential of proteins extracted from BSG to produce a new emulsifying ingredient. To accomplish this, bioinformatic analysis was performed on the BSG proteome to identify and select specific peptide sequences with predicted emulsifying properties for evaluation and validation. Using synthesized peptides, the emulsifying properties were evaluated based on their ability to reduce interfacial tension between oil and water and their ability to provide physical stability for 5% oil-in-water emulsions during storage.
2. Materials and Methods
2.1. Materials
All peptides were purchased from SynPeptide Co., Ltd. (Shanghai, China), with a purity of at least 90%. Information about each peptide can be seen in Table 1. Rapeseed oil was purchased at the local supermarket. Sodium caseinate (Miprodan 30), with 93.5% protein (Nx6.38, calculated in % of dry matter), was provided by Arla Foods Ingredients amba (Viby J, Denmark) and used as a positive control for interfacial tension measurements. Medium-chain triglyceride (MCT) oil (WITARIX® MCT 60/40) was purchased from IOI Oleo GmbH (Hamburg, Germany) and stored at −20 °C until use. All chemicals were of analytical grade or higher.

Table 1.
Peptide name, amino acid sequence, amphiphilicity score by EmulsiPred, Protein ID, length, molecular weight, isoelectric point (pI), and net charge at pH 7 of potential emulsifying peptides tested in this study.
2.2. Bioinformatics Analysis and Peptide Selection
Proteins identified in BSG, based on previous work [23], with an abundance greater than 1% were analyzed using EmulsiPred [14,16]. The emulsifying potential of the peptides was computed based on their amphiphilicity, ascribed by the Kyte-Doolittle hydrophobicity scale [24], and their probable conformation at the oil–water interface. Peptides were predicted as either facial amphiphiles in an α-helix or β-strand conformation (α- and β-peptides, respectively) or as axial amphiphiles (i.e., one end predominantly hydrophobic and one end predominantly hydrophilic) independent of secondary structure (γ-peptides). Peptides are given a z-averaged score indicating their amphiphilicity, with a higher score correlating with more surface activity.
2.3. Reduction in Interfacial Tension
The dynamic interfacial tension at the oil–water interface was determined using an automated drop tensiometer OCA20 (Dataphysics Instruments GmbH, Filderstadt, Germany) at 25 °C as described in [13] with minor modifications. Peptides were dissolved in a 10 mM sodium acetate—10 mM imidazole buffer at pH 7 in a concentration of 0.1 wt%. The solutions were placed in a water bath at 50 °C and stirred with a magnetic stirrer for 2 h for solubilization. If the peptide had not solubilized, the solutions were stirred overnight at room temperature to solubilize it. The peptide solutions were filled into a syringe with a screwed-on needle (d = 1.83 mm). Droplets of 50 µL were generated using the automated system into a glass cuvette filled with MCT oil (WITARIX® MCT 60/40, IOI Oleo GmbH, Hamburg, Germany). Images of the drop were recorded, and the interfacial tension was calculated based on the Young-Laplace equation (Equation (1)) for 30 min, based on the drop shape:
where ∆P (mN/m2) is the pressure difference over the interface, γ (mN/m) is the interfacial tension, and R1 and R2 are the principal radii of curvature of the pendant drop. Measurements were performed in duplicates for each peptide. A solution of sodium caseinate was also measured for comparison.
2.4. Emulsion Production and Storage Experiment
To test the emulsifying properties of the peptides, 5% oil-in-water emulsions stabilized with 0.2 wt% peptide were produced as described by [11], with minor modifications. Peptides were dissolved in buffer containing 10 mM sodium acetate and 10 mM imidazole at pH 7 for a peptide concentration of 0.2 wt% in the final emulsion. The peptide solutions were continuously stirred in the fridge overnight for protein hydration and solubilization. Primary homogenization was performed by adding the rapeseed oil to the peptide solution and mixing at 18,000 rpm for 30 s with a POLYTRON® PT1200E (Kinematic Inc., New York, NY, USA). Secondary homogenization was performed using a Microson XL2000 sonicator equipped with a P1 probe (Misonix, Inc., New York, NY, USA). Emulsions were homogenized at an amplitude of 75% (max amplitude of 180 µm), running two rounds of 30 s with a 1 min break in between. During sonication, the emulsions were placed in iced water to minimize temperature increase. The emulsions were stored in the dark at room temperature for nine days. For each peptide, four emulsions were made. One emulsion was used for visual inspection of creaming, whereas the remaining three were used for physical stability tests.
2.5. Physical Stability of Emulsions
Creaming was observed by visual inspection over nine days of storage and ranked on a scale from 1 to 4, where 1 signifies no creaming, 2 visible creaming, 3 an oil layer on top, and 4 phase separation.
2.5.1. Droplet Size Determination
The droplet size distribution of the emulsions was measured on days 1, 5, and 9 by laser diffraction in a Mastersizer 2000 (Malvern Instruments, Ltd., Worcestershire, UK). The emulsions were diluted in circulating distilled water (3000 rpm) until an obscuration between 10 and 15% was reached. The refractive indices of sunflower oil (1.469) and water (1.330) were used as particles and dispersant, respectively. Results are given in volume mean diameter (D[4,3]). Each emulsion was measured twice.
2.5.2. Zeta Potential
The zeta potential of the emulsions was measured on day 1 using a Zetasizer Nano ZS (Malvern Instruments Ltd., Worcestershire, UK) with a DTS1070 cell. Emulsions were diluted (20 µL in 10 mL buffer) prior to measurements. Measurements were carried out at 25 °C, and the zeta potential range was set to −100 to + 50 mV. Each emulsion was measured twice.
2.6. Statistical Analysis
Statistical analysis was performed using Statgraphics 18 (Statgraphics Technologies, Inc., The Plains, VA, USA). Statistical significance was determined by a 95% confidence level using ANOVA and a post hoc Tukey test. Results are expressed as mean ± standard deviation. PCA was performed using Python 3.12.4, using the scikit-learn package. Numerical values for zeta-potential and charge were used. Only samples with full measurements were used in the PCA.
3. Results
Based on the proteomic data presented in the study by Gregersen Echers et al. [23] on BSG, malt, and barley, the proteins with relative intensity-based absolute quantification (riBAQ) above 1% (Table S1) were selected for bioinformatics analysis. From the EmulsiPred results files, a subset of peptides was selected based on sequence, score, length, and charge. The intention behind this approach was to include peptides with high EmulsiPred scores, but also to have peptides with different lengths and charges in order to evaluate the effect of these parameters, thereby adding to the fundamental understanding of peptides as emulsifiers. In a few cases, it was also possible to select peptides with overlapping sequences but different lengths and charges. In total, 48 peptides (15 α-peptides, 15 β-peptides, and 18 γ-peptides) were selected for commercial, certified synthesis (Table 1). To investigate the ability of EmulsiPed to predict emulsifying properties, four peptides with very low scores were included in the list of the 48 peptides (one α-peptide, one β-peptide, and two γ-peptides.
3.1. Screening of Surface Activity by Interfacial Tension
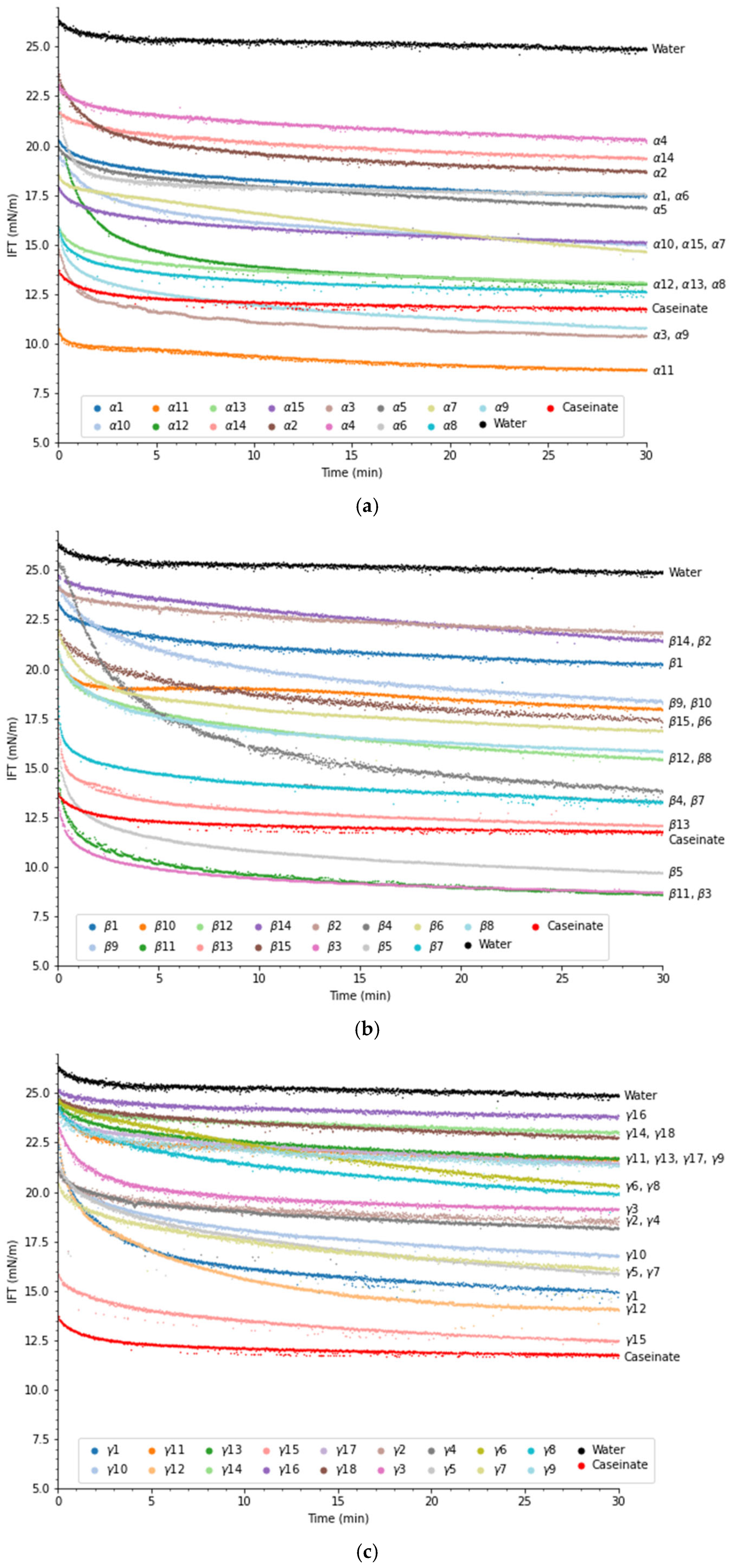
Peptides’ ability to reduce the interfacial tension between oil and water is an important factor for stabilization of emulsions, but this property does not guarantee that a peptide is a good emulsifier. Another important property that emulsifying peptides need is the ability to rearrange their secondary structure at the interface to maximize hydrophobic interactions and provide sufficient electrostatic repulsion between droplets [25]. In Figure 1, the evolution of the interfacial tension over 30 min can be viewed.
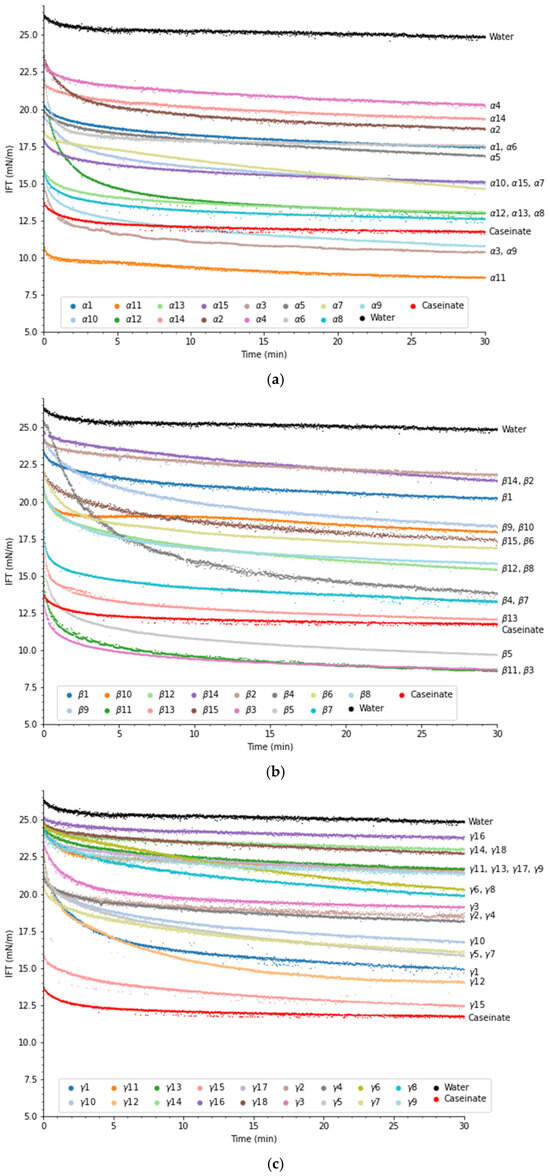
Figure 1.
Interfacial tension evolution of (a) α-peptides, (b) β-peptides, and (c) γ-peptides at a concentration of 0.1 wt% in MCT oil. Distilled water and 0.1 wt% sodium caseinate were used as a control and for comparison. Refer to Table 1 for more information regarding peptide sequences.
MCT and distilled water had an IFT of 25 mN/m, which is similar to findings in other studies [26]. All peptides were able to significantly (p < 0.05) lower the interfacial tension compared to pure MCT oil and water after 30 min. This suggests that all peptides tested, including the four peptides with low EmulsiPred scores, were surface active and potential emulsifiers, as expected based on results obtained from bioinformatics analysis.
Looking more closely at the α-peptides (Figure 1a), α3 (10.4 mN/m), α9 (10.8 mN/m), and α11 (8.7 mN/m) were able to lower the interfacial tension more than sodium caseinate (CAS) (11.8 mN/m). These three peptides had AA chain lengths of 23, 17, and 30 amino acids (AA), respectively. The reduction in interfacial tension was also larger than what García-Moreno et al. [14] observed for synthetic α-peptides derived from potato proteins, even though the peptide concentration was half of that reported in that study. α1 (NNLIKHGDRILDEANKVF) and α3 (LAWYSNNLIKHGDRILDEANKVF) share similar AA sequences, with α3 having LAWYS added to the N-terminal. However, α3 showed much better surface activity compared to α1. As L, A, W, and Y are hydrophobic amino acids, the N-terminal extension could promote anchoring of α3 to the oil phase, thereby improving the surface activity and stability.
García-Moreno et al. [14] determined that synthetic α-peptides derived from potato with chain lengths below 14 AA had poor ability to reduce interfacial tension as the chain was not long enough to form an amphiphilic α-helix. Similarly, Enser et al. [27] found superior emulsifying properties for peptides showing at least three helical turns. Although only one peptide (α14) with a length of 14 AAs was tested, our results do agree with this hypothesis, as α14 had the second-highest IFT after 30 min. However, α2, α4, and α5 had chain lengths between 28 and 30 AA, but were not as good as α3 or CAS to reduce interfacial tension, indicating that chain length itself cannot explain the surface activity of peptides, but the amphiphilicity of the helix also needs to be considered. Furthermore, peptides α2, α4, and α5 are all derived from the same proteins (Table 1), and their AA sequences are very similar, albeit with some differences at the termini. Yet they lower the IFT to different degrees, with α5 having the lowest IFT (17.63 mN/m) and α4 the highest (20.66 mN/m) (α2 = 18.72 mN/m). As the size and sequence are quite similar, the observed difference could be due to the charge difference. Especially the N-terminal of α2 (EDVDR) is highly charged, and depending on the helical conformation, could lead to disadvantageous interactions at the oil–water interface, which is not present in α5; however, this does not explain why α4 performed worse than α2, when that peptide has a less charged N-terminal. Instead, it could suggest that there is no simple link between sequence and function, as a small modification in sequence could change the secondary structure or how the AA overlaps in the helix. Further studies are needed to fully understand the mechanism.
α8, α12, and α13 were able to reduce interfacial tension to a degree close to that of sodium caseinate, which could indicate good surface activity and emulsifying properties. The low-scoring control peptide (α15), which had an EmulsiPred score of 2.0, was able to lower the interfacial tension similarly or to a higher degree than half of the α-peptides tested. Then, in theory, a score of 2.0 does not mean that the peptide should not be surface active, but peptides with higher scores should have a higher probability of being surface active.
For the β-peptides, β5, β11, and β3 were able to reduce the interfacial tension significantly (p < 0.05) more than CAS over 30 min (Figure 1b). These β-peptides had EmulsiPred scores ranging from 3.5 to 3.8. Higher scores were observed for other β-peptides (β1, β2, β8, β9, β10, β13, and β14). However, these performed very poorly in reducing interfacial tension, with the exception of β13. For peptides β1, β2, β8, β9, and β10, the results could be due to their size (below 9 AA). This limited length could hinder the formation of stable, amphiphilic β-strand structures with a sufficient surface area at the interface and hence reduce their emulsifying potential [14]. Furthermore, it has been hypothesized that medium-chain length β-peptides (13–15 AA) are better at reducing the interfacial tension compared to other β-peptides [14]. This also aligns with the chain length of the five best peptides in the present study, which ranged from 13 to 22 AA; however, more peptides need to be tested before any conclusion can be made.
β6 (28 AA) and β14 (29AA) were the longest tested β-peptides but were not able to lower interfacial tension to a level similar to CAS. Furthermore, β13 (IRLSTDVRISFRA) and β14 (IRLSTDVRISFRAYTTCLQSTEWHIDSEL) have similarities in their AA sequence, but β14 is 16 AA longer. Their ability to reduce interfacial tension differed significantly (p < 0.05), with β13 being able to reduce it similarly to CAS and β14 being one of the worst-performing peptides. Looking more closely at the AA sequence, the alternating hydrophobic–hydrophilic AA aligns very well with β13 whereas the extension on β14 does not align with the alternation. Furthermore, longer β-peptides can have a tendency to form β-sheet fibrils instead of moving to the interface [28].
Reduction in interfacial tension of the γ-peptides can be seen in Figure 1c. The majority of the γ-peptides exhibit high scores, typically around five or above (see Table 1). However, none of these peptides were able to reduce the interfacial tension to a level comparable to CAS. The γ-peptides, in general, performed worse than the α- and β-peptides (Figure 1a,b), even though their EmulsiPred scores were higher. It is, however, misleading to compare scores across conformations (α, β, and γ) and projections (facial vs. axial) due to the different methods for computing the amphiphilicity-based score [14]. γ15 was the best peptide for reducing interfacial tension (12.5 mN/m), followed by γ12 (15.1 mN/m) and γ1 (15.34 mN/m). As these three peptides have very different chain lengths (21, 11 and 30, respectively), the chain length seems not to be the main factor determining the surface activity for the gamma peptides. In Table 2 information regarding solubility of the peptides in the buffer is provided. Insoluble particles can influence the evolution of interfacial tension and can mask the actual contribution of the peptides [29]. It can be seen that 9 out of the 18 tested γ-peptides had solubility issues. This could be due to a specific region of the peptide being too hydrophobic, meaning that the peptides aggregate together to minimize the exposure of hydrophobic patches in aqueous solution. This could also result in the peptides failing to reach the interface, causing them to form clusters. This could explain why γ9 (LLLAAVLVSIFAAVAAVGSEDCTPW), γ11 (ILLVVASQRR), γ14 (LVLFVAVIVALVALTTAEREINGNN), γ16 (VLFVAVIVALVALTTAERE), and γ17 (FLIFALLAIVATSTIAQQQPYPQQPQ), although having high EmulsiPred scores (4.95, 4.88, 5.98, 5.83, and 5.93), were not able to decrease the IFT considerably. Peptide γ10 (AAHLALKALGRPNDIDEQRYSRESRRYTR) and the low-scoring peptide γ13 (ALGRPNDIDEQRYSRESRR) share overlapping AA sequences. While γ13 had a low EmulsiPred score (2.70) and was selected as a negative peptide, γ10 had a very high score (4.93). In agreement with the scores, γ10 was able to reduce interfacial tension significantly more than γ13 (p < 0.05).

Table 2.
Physical stability of 5% O/W emulsions stabilized with 0.2 wt% peptide during 9 days of storage. For peptide sequence refers to Table 1. n.m = not measured. For creaming: 1 = no creaming, 2 = creaming, 3 = oil layer on top, and 4 = phase separation. α1, α5, and α14 are not shown, as phase separation occurred within one day.
Importantly, α11, the best-performing α-peptide, and γ15, the best-performing γ-peptide, were derived from the same protein. HORVU2Hr1G023590.3 was previously predicted to be of lysosome/vacuole origin and identified as a 4-hydroxy-7-methoxy-3-oxo-3,4-dihydro-2H-1,4-benzoxazin-2-yl glucoside beta-D-glucosidase (DIMBOA-βG) by BLAST analysis [23]. It was found in moderate abundance (0.32%) in unprocessed barley but was enriched during malting (0.46%) and remained enriched (0.50%) in BSG. This is interesting in the context of future attempts to produce emulsifying peptides through enzymatic hydrolysis, as both peptides could be produced from the protein and therefore have an added effect in increasing surface activity within a hydrolysate originating from a protein extract enriched for this particular protein.
3.2. Physical Stability of 5% Oil-in-Water Emulsions
All peptides were further tested in 5% oil-in-water emulsion with 0.2 wt% peptide added. The evolution of physical properties of the emulsions can be seen in Table 2. Some of the peptides were unable to stabilize the emulsion over 9 days of storage; therefore, droplet sizes were not measured for these emulsions after phase separation occurred. At day 1, D[4,3] ranged from 0.39 µm (β2) to 127 µm (α12). Four peptides (α1, α14, α5, and γ13) could not stabilize the emulsion for more than 1 day of storage. After five days, nine additional emulsions (α12, α13, α15, β1, β9, β14, γ2, γ8, and γ9) had separated, and prior to day 9, further nine emulsions had also undergone phase separation (α6, α7, β2, β6, γ3, γ6, γ10, γ16, and γ18). Thus, after 9 days of storage, 26 out of 48 peptides were able to stabilize the emulsions. This finding is in contrast to results from the pendant drop analysis, in which all peptides were able to lower IFT. This could suggest that some of the peptides are surface active but might not provide the necessary electrostatic or steric stability to minimize coalescence. To be an effective emulsifier, both a reduction in IFT and sufficient repulsive forces are mandatory [8].
As shown in Figure 1a, α3, α9, and α11 were the most effective in lowering interfacial tension among the α-peptides. All three were also able to form and stabilize emulsions, with α11 forming the smallest droplets (1.33 µm on day 1), see Table 2. However, creaming was observed for α3 and α9 over the 9 days of storage. The creaming observed could be due to the large droplet size (13.65 µm and 10.22 µm, respectively), which would increase the gravitational separation [30]. The droplet size in the α11 emulsion was small at day 1 (1.33 µm), but increased significantly over the 9 days of storage to 5.07 µm. Among the α-peptides, five (α2, α4, α6, α8, and α11) were able to create emulsions with droplet sizes below 8 µm in diameter, but only four of those (α2, α4, α8, and α11) were stable over 9 days of storage.
α2 produced the smallest droplets of all the tested α-peptides (D[4,3] = 0.69 µm) without any observable creaming and no significant (p < 0.05) increase in droplet size over the 9 days of storage. This peptide showed a very poor ability to reduce interfacial tension (Figure 1a), indicating that interfacial tension measurements do not fully describe emulsifying abilities.
Similar to α2, α8 also produced small droplet sizes (D[4,3] = 1.21 µm at day 1) without showing any creaming. The emulsions with these peptides also had very high zeta potentials (−58.2 mV and −56.4 mV, respectively), suggesting high electrostatic repulsion between the droplets, which would increase the stability of the emulsions [31]. No significant (p < 0.05) difference was observed for the droplet size of α2, α8, and α11 on day 9. However, for α2 and α8, the droplet size did not significantly increase over the 9-day storage period, indicating better emulsifying stability compared to α11. α4 also showed a small droplet size initially (5.8 µm); however, severe creaming and phase separation occurred during storage. To form a stable emulsion, not only does the peptide need to lower the interfacial tension, but it must also form an interfacial structure protecting the droplets from aggregation and coalescence [9]. Notably, α2 (EDVDRVFKRNSRGEGEISEASEEQIRELSR), α4 (VDRVFKRNSRGEGEISEASEEQIRELSR), and α5 (RVFKRNSRGEGEISEASEEQIRELSRSCSR) have similar peptide sequence and length (28–30 AA), but the results showed big differences in emulsifying activity, especially α5, which could not stabilize an emulsion over 1 day. As the core sequence of the peptides is the same, the difference might be due to changes at the two terminals. The increased charge at the N-terminal of α2 could increase the electrostatic repulsion between droplets and thereby increase the stability of the emulsion. This is also supported by the higher zeta potential of α2 compared to α4. On the other hand, α5 completely lacks the negative charge, which could result in low electrostatic repulsion between the droplets and lower stability. Notably, α5 also contains a cysteine at the C-terminal, which is hydrophilic and could result in bridging flocculation through disulfide bonds between emulsion droplets [32].
A larger number of β-peptides created emulsions with small droplet sizes compared to the α-peptides (Table 2). Notably, β7 could stabilize emulsions with a droplet size (D[4,3]) of 0.5 µm and a zeta potential of −69.1 mV over 9 days of storage. Other β-peptides that could stabilize emulsions with small droplet sizes (D[4,3] < 8 µm) include β3, β4, β5, β8, β10, β12, and β13. These peptides had AA chain lengths between 8 AA and 16AA, which is lower than that required for the α-peptides. These findings are in line with previous findings for β-peptides [14,25]. The droplet size of β7 and β12 was smaller than what was observed for synthetic β-peptides from potatoes and methanotrophic bacteria, and similar to that of whey protein isolate, soy protein isolate, and casein in 5% O/W emulsions with 0.2wt% protein [14,17,33].
β2, which had the smallest droplet size on day 1 (0.39 µm), showed a large droplet size at day 5 (35 µm) and had separated at day 9. This could indicate that β2 did not provide sufficient repulsive forces to prevent coalescence. Similar observations were made for β1. β3 produced a stable emulsion with a droplet size of 2.76 µm after 9 days while displaying a large, positive zeta potential (60.35 mV). The positive zeta potential might be beneficial, as this could mean that transition metal ions, commonly present in food emulsion systems, would be repelled from the oil droplets. This may provide improved oxidative stability, as the initiation of lipid oxidation and the decomposition of lipid hydroperoxides into secondary volatiles could be accelerated by the presence of metal ions at the interface [34].
In accordance with the interfacial tension results, the related peptides, β13 and β14, showed clear differences in their emulsifying properties. β13 was able to create an emulsion with very small droplets (D[4,3] = 0.58 µm at day 1), while the longer β14 was not (D[4,3] = 22.7 µm at day 1). This peptide also had a chain length of 29 AA, and the extension possesses a highly hydrophilic (YTTCLQSTE) part, which could disrupt the amphiphilic β-strand structure at the interface. Furthermore, the extension carries a cysteine, which could lead to the formation of inter-droplet disulfide bonds. Furthermore, this could be related to the fact that longer β-strand peptides can assemble into β-sheet fibril structures, which reduces the solubility and the diffusion rate to the interface [28]. The droplet size of β11 (MKFSVSPVVRVAVQCKVASDLP) after 9 days of storage was significantly bigger (p < 0.05) compared to β8 (VRVAVQCKV), even though they share a familiar AA sequence, and even though β11 provided the lowest IFT among the β-peptides, see Figure 1b. As β11 had a solubility issue, possibly due to β-sheet fibril formation, the interfacial concentration might not have been enough to fully coat the droplets, leading to coalescence. Furthermore, the emulsion had very low absolute zeta potential, which would limit the electrostatic repulsions between droplets.
The γ-peptides have their amphiphilicity split axially across the peptide sequence instead of facially as α- and β-peptides. The droplet size of the γ-peptides ranged from 0.4 µm (γ12) to 58 µm (γ2) at day 1. Notably, many of the peptides had solubility issues in the buffer but were still able to create stable emulsions. γ7 and γ12 were the only γ-peptides able to form droplets below 1 µm in D[4,3] at day 1 and only γ12 was able to stabilize this droplet size over 9 days of storage. γ7 still showed small droplet size after 9 days of storage (3.09 µm), however the droplet size significantly increased.
γ1 formed droplets below 2 µm in diameter; however, creaming was observed in this emulsion. Most of the γ-peptides did show creaming or phase separation during storage, and only γ12 did not show any signs of instability, which further suggests its higher emulsifying activity compared to the other γ-peptides. This was in agreement with the good ability of this peptide to lower the IFT (Figure 1c). Similarly, γ14, γ15, and γ17 were able to stabilize emulsion with droplet sizes below 5 µm after day 9, with no significant increase in droplet size observed during storage. The AA chain length of γ1, γ12, γ14, γ15, and γ17 ranged from 30 to 11, which suggests that the chain length might not be a significant factor. γ12 (REDIAGVVVFV) is composed mainly of hydrophobic AAs (central region and C-terminus), enabling it to penetrate deep into the oil phase and stabilize the droplet. This could also result in higher affinity for the oil phase, meaning that dissociation of the peptides from the interface to the water phase is minimized and the peptide binds more strongly to each droplet. γ12 had solubility issues, which could be the result of the hydrophobic C-terminus. The short hydrophilic N-terminus (RED) is highly charged, which could increase emulsion stability due to electrostatic repulsions. γ1 (LAIVLSLCLSLSFASWDAEDEGRGSRRWQE) and γ3 (LALAIVLSLCLSLSFASWDAEDEGRGSRR) have mostly overlapping AA sequences, where γ3 has an N-terminal LA extension and a C-terminal WQE truncation compared to γ1; however, γ1 showed stable emulsions with small droplets (1.93 µm) during 9 days of storage, whereas complete phase separation was observed for γ3. As γ1 showed a smaller droplet size and stable emulsion compared to γ3, it is hypothesized that the addition of charged hydrophilic AA results in enough repulsion between droplets, which minimizes droplet interactions that could otherwise lead to coalescence. Similar results were shown with γ14 (LVLFVAVIVALVALTTAEREINGNN) and γ16 (VLFVAVIVALVALTTAERE). By the addition of the 4 hydrophilic AAs at the C-terminal of γ14, compared to γ16, a longer hydrophilic chain is generated. We hypothesize that this can explain the increased stability by giving higher steric repulsion. This is also the case for γ7 and γ15, which have longer hydrophilic chains.
EmulsiPred was employed to identify emulsifying peptides derived from BSG proteins. Of the 48 peptides predicted, only 26 could stabilize emulsions over a 9-day storage period, corresponding to a predictive success rate of 54%. While this level of accuracy can be considered promising for an in silico tool, further evaluations of the experimental results gave another insight. Specifically, when only emulsions exhibiting stable droplet sizes (<5 µm) were considered, the success rate dropped to 33%. These findings indicate that, although EmulsiPred holds potential as a first screening tool to narrow down potential candidate peptides, its current performance underscores the need for further optimization before it can reliably reduce the screening experiments necessary.
The findings suggest that BSG could be a potentially untapped resource for emulsifying peptides, especially if the peptides α2, α8, α11, β3, β4, β4, β5, β7, β8, β12, β13, γ1, γ7, γ12, γ14, γ15, and γ17 could be released by enzymatic hydrolysis. Notably, it is important to highlight that α2, γ1, and γ15 were derived from the same protein, HORVU5Hr1G104630.1. This protein was previously identified as a Cupin type-1 embryo globulin (Cupin-1) and is present in a relative abundance of 0.61% in unprocessed barley [23]. While depleted to 0.35% during malting, Cupin-1 is enriched in BSG up to around 1% of the total protein. This implies that purification and hydrolysis of single proteins could also lead to elevated emulsifying properties, but enriching the protein in extraction may also be beneficial to produce a hydrolysate with good emulsifying properties.
3.3. Principal Component Analysis
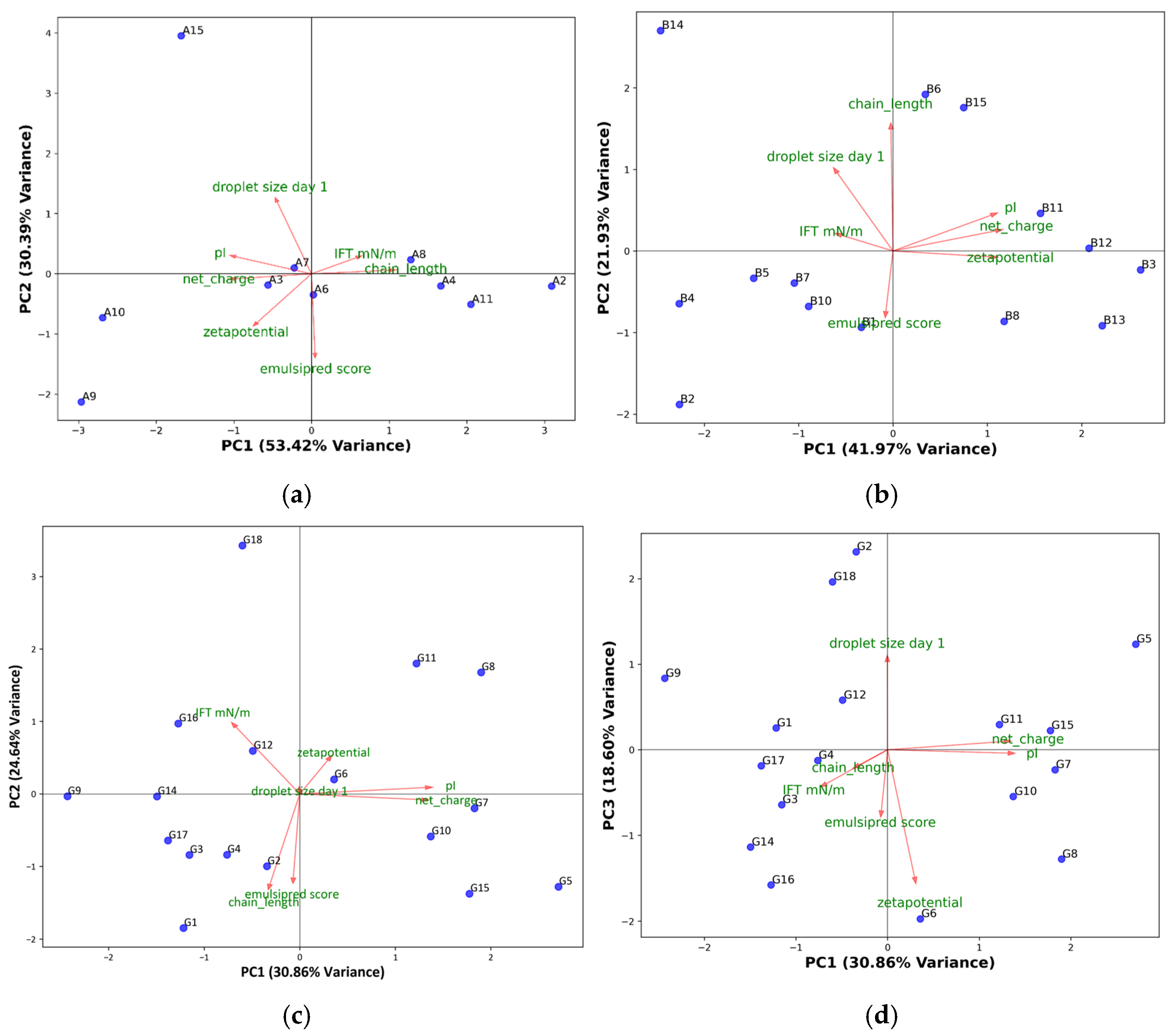
Principal component analysis (PCA) was performed on the results from interfacial tension and droplet size on day 1, together with physicochemical properties of the peptides and the EmulsiPred score (Figure 2).
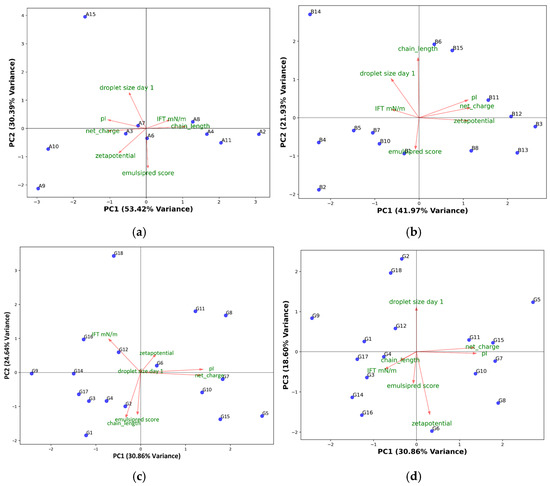
Figure 2.
PC1 vs. PC2 of (a) α-peptides, (b) β-peptides, and (c) γ-peptides, or (d) PC1 vs. PC3 of γ-peptides, based on peptide chain length, EmulsiPred score, pI, net charge at pH 7, and results from interfacial tension measurements, and 5% O/W emulsions storage. Arrows and green labels refer to the loading directions, and descriptions respectively. For peptide sequences, see Table 1.
For the α-peptides (Figure 2a), PC1 and PC2 explained 84% of the variation in the data. Most of the variation in the data was found in PC1, which mainly explained the variance in IFT, zeta potential, chain length, and net charge. From the loadings in the α-peptide PCA, there is no correlation between droplet size at day 1 and IFT values, and only a weak negative correlation with zeta potential. EmulsiPred score showed a negative correlation with droplet size, suggesting that a higher score was related to smaller droplets, as expected. The α-peptides showed clustering based on the chain length along PC1, very close to the center of PC2, except for α15, which also showed a very large droplet size. PCA was also performed without considering α15, which resulted in changes in the loadings (Figure S1). However, a negative correlation between EmulsiPred score and droplet size was still observed.
PC1 and PC2 explained 64% of the variance in the data for the β-peptides (Figure 2b). The PCA shows some of the same trends as for α-peptides: a negative correlation between droplet size and the EmulsiPred score and zeta potential. However, the chain length showed correlation with droplet size at day 1, suggesting that an increase in chain length could result in larger droplet sizes. This was also evident from the emulsion study (Table 2). This suggests that for β-peptides, longer chain length may be unfavorable for emulsion stability and also result in lower EmulsiPred scores for β-peptides. Moreover, there was a weak correlation between IFT and the droplet size on day 1, whereas none of the other parameters showed good correlation with droplet size.
For the γ-peptides, PC1 and PC2 explained 55% of the variance in the data. While PC1 and PC2 explain the majority of the variance in the dataset, the droplet size contributes minimally to these components (see Figure 2c), indicating it is not aligned with the dominant patterns of variation. Instead, droplet size shows a strong loading on PC3 (see Figure 2d), which accounts for 18.60% of the variance. This suggests that droplet size follows a distinct pattern in this dataset, orthogonal to those captured by PC1 and PC2. Therefore, understanding droplet size requires considering PC3, as its variation is not well represented in the first two principal components. The need to consider PC3 to explain droplet size underscores the complexity of emulsifying properties in γ-peptides and suggests that droplet formation may be driven by different physicochemical factors than those influencing the dominant variance patterns.
PC1 and PC3 explained 49% of the variability of the γ-peptides (Figure 2d), with droplet size on day 1 being highly correlated with PC3. Similar to the two other groups of peptides, a strong negative correlation between droplet size on day 1 and EmulsiPred score was observed. Interestingly, there was also a negative correlation between droplet size and zeta potential, which was not observed in PC3 for the other peptide classes, suggesting that zeta potential may have a more pronounced role in producing smaller droplets in γ-peptides compared to other peptide types.
The PCA unsurprisingly revealed a positive correlation between pI, net charge, and zeta potential, and the three parameters generally showed a negative correlation with IFT. This indicates that electrostatic forces are a key driving factor in IFT reduction. This pattern is more evident for α- and β-peptides, suggesting a different mode of action for these compared to γ-peptides. For γ-peptides, the observed zeta potential is only weakly correlated with pI and net charge, indicating a difference in the way charged AAs are presented at the interface, as some charges may be shielded, thereby not contributing to the observed interfacial zeta potential. The difference in mode of action is also reflected by the lack of correlation between IFT and zeta potential.
The PCA also shows that the influence of peptide length on the prediction is not trivial and generalizable but depends on the class/conformation of the peptides, as no correlation is observed for α-peptides, while the correlation is negative and positive for β- and γ-peptides, respectively. Overall, the PCA highlights the multidimensional nature of the physicochemical properties governing peptide behavior in emulsions. This entails that the physicochemical properties that influence the emulsifying activity depend on the secondary structure of the peptides. Nevertheless, the PCA suggests that the magnitude of the EmulsiPred score can be used as an in silico method for screening of peptides in relation to their ability to form emulsions with small droplet sizes, as these variables generally show a negative correlation.
4. Conclusions
BSG constitutes a major and underutilized industrial byproduct, but the limited functionality of extracted protein constitutes a barrier to its use as a high-value, functional food ingredient. Based on the protein-level composition of BSG, bioinformatic analysis was performed using EmulsiPred in order to predict potential emulsifying peptides embedded in abundant BSG proteins. Forty-eight peptides were selected for initial screening in small-scale emulsions and assessed for their ability to reduce interfacial tension (IFT) at the oil–water interface. The results showed that the EmulsiPred score did not fully reflect the emulsifying activity of peptides, as some of these peptides were not able to form stable emulsions. However, six of the tested peptides were able to reduce IFT to an extent greater than sodium caseinate. Moreover, peptides γ12 and β7 were able to stabilize 5% oil-in-water emulsions with D[4,3] below 0.5 µm over 9 days of storage without any signs of creaming, and a total of sixteen peptides, representing all three classes, produced droplet sizes below 5 µm at storage day 9. Through PCA, we found that length by itself is not a good indicator of the emulsifying potential of peptides; however, a negative correlation between EmulsiPred score and droplet size was observed. β-peptides that were too long were found to negatively affect the emulsification potential, in agreement with previous findings that β-peptides between 8 and 16 AA provide the best emulsion stability with smaller droplets. The best emulsion stability was observed for peptides α2, α8, α11, β3, β4, β4, β5, β7, β8, β12, β13, γ1, γ7, γ12, γ14, γ15, and γ17. This study validates the applicability of EmulsiPred as an in silico screening tool for novel peptide emulsifiers, as a higher score was found to correlate with smaller droplet sizes in small-scale emulsions. Nevertheless, the success rate of 33% for producing stable emulsions (sixteen of forty-eight peptides showed small droplets after 9 days of storage) underlines the necessity for developing new and more accurate predictive models. Another option could be to optimize the model so that, instead of predicting peptides based on protein composition, it could generate the optimal amino acid sequences for the different conformations with greater emulsifying properties, which could be tested. Ultimately, BSG peptides show promising potential as a new vegetarian emulsifying ingredient if these peptides can be released in high abundance through targeted hydrolysis of BSG or BSG protein extracts.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/colloids10010001/s1, Table S1; BSG abundant proteins, Figure S1; principal component analysis of α-peptides with α15 omitted.
Author Contributions
Conceptualization, R.K.M., S.G.E., M.T.O., C.J. and B.Y.; methodology, R.K.M., I.F., C.J. and B.Y.; formal analysis, R.K.M., S.G.E., N.A.-K., C.J. and B.Y.; investigation, R.K.M. and I.F.; data curation, R.K.M. and I.F.; writing—original draft preparation, R.K.M.; writing—review and editing, R.K.M., S.G.E., N.A.-K., M.T.O., C.J. and B.Y.; visualization, R.K.M.; supervision, M.T.O., C.J. and B.Y.; project administration, C.J. and B.Y.; funding acquisition, C.J. and B.Y. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Novo Nordisk fonden, under the project “GRAINPEP”, grant number: NNF21OC0071633.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors on request.
Acknowledgments
The work presented in the article is part of the PhD thesis of Rasmus Kranold Mikkelsen. The full thesis can be found at https://orbit.dtu.dk/en/publications/extraction-and-utilization-of-brewers-spent-grain-proteins/ (accessed on 5 December 2025).
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Mussatto, S.I. Brewer’s Spent Grain: A Valuable Feedstock for Industrial Applications. J. Sci. Food Agric. 2014, 94, 1264–1275. [Google Scholar] [CrossRef]
- Hejna, A. More than Just a Beer—The Potential Applications of by-Products from Beer Manufacturing in Polymer Technology. Emergent Mater. 2022, 5, 765–783. [Google Scholar] [CrossRef]
- Devnani, B.; Moran, G.C.; Grossmann, L. Extraction, Composition, Functionality, and Utilization of Brewer’s Spent Grain Protein in Food Formulations. Foods 2023, 12, 1543. [Google Scholar] [CrossRef] [PubMed]
- Lynch, K.M.; Steffen, E.J.; Arendt, E.K. Brewers’ Spent Grain: A Review with an Emphasis on Food and Health. J. Inst. Brew. 2016, 122, 553–568. [Google Scholar] [CrossRef]
- Lech, M.; Labus, K. The Methods of Brewers’ Spent Grain Treatment towards the Recovery of Valuable Ingredients Contained Therein and Comprehensive Management of Its Residues. Chem. Eng. Res. Des. 2022, 183, 494–511. [Google Scholar] [CrossRef]
- Ikram, S.; Huang, L.; Zhang, H.; Wang, J.; Yin, M. Composition and Nutrient Value Proposition of Brewers Spent Grain. J. Food Sci. 2017, 82, 2232–2242. [Google Scholar] [CrossRef] [PubMed]
- Mussatto, S.I.; Dragone, G.; Roberto, I.C. Brewers’ Spent Grain: Generation, Characteristics and Potential Applications. J. Cereal Sci. 2006, 43, 1–14. [Google Scholar] [CrossRef]
- McClements, D.J.; Bai, L.; Chung, C. Recent Advances in the Utilization of Natural Emulsifiers to Form and Stabilize Emulsions. Annu. Rev. Food Sci. Technol. 2017, 8, 205–236. [Google Scholar] [CrossRef]
- McClements, D.J.; Gumus, C.E. Natural Emulsifiers—Biosurfactants, Phospholipids, Biopolymers, and Colloidal Particles: Molecular and Physicochemical Basis of Functional Performance. Adv. Colloid Interface Sci. 2017, 8, 205–236. [Google Scholar] [CrossRef]
- Lam, R.S.H.; Nickerson, M.T. Food Proteins: A Review on Their Emulsifying Properties Using a Structure-Function Approach. Food Chem. 2013, 141, 975–984. [Google Scholar] [CrossRef]
- Karaca, A.C.; Low, N.; Nickerson, M. Emulsifying Properties of Chickpea, Faba Bean, Lentil and Pea Proteins Produced by Isoelectric Precipitation and Salt Extraction. Food Res. Int. 2011, 44, 2742–2750. [Google Scholar] [CrossRef]
- Wang, Y.; Li, Z.; Li, H.; Selomulya, C. Effect of Hydrolysis on the Emulsification and Antioxidant Properties of Plant-Sourced Proteins. Curr. Opin. Food Sci. 2022, 48, 100949. [Google Scholar] [CrossRef]
- Wouters, A.G.B.; Rombouts, I.; Fierens, E.; Brijs, K.; Delcour, J.A. Relevance of the Functional Properties of Enzymatic Plant Protein Hydrolysates in Food Systems. Compr. Rev. Food Sci. Food Saf. 2016, 15, 786–800. [Google Scholar] [CrossRef]
- García-Moreno, P.J.; Gregersen, S.; Nedamani, E.R.; Olsen, T.H.; Marcatili, P.; Overgaard, M.T.; Andersen, M.L.; Hansen, E.B.; Jacobsen, C. Identification of Emulsifier Potato Peptides by Bioinformatics: Application to Omega-3 Delivery Emulsions and Release from Potato Industry Side Streams. Sci. Rep. 2020, 10, 690. [Google Scholar] [CrossRef] [PubMed]
- Gregersen Echers, S.; Jafarpour, A.; Yesiltas, B.; García-Moreno, P.J.; Greve-Poulsen, M.; Hansen, D.K.; Jacobsen, C.; Overgaard, M.T.; Hansen, E.B. Targeted Hydrolysis of Native Potato Protein: A Novel Workflow for Obtaining Hydrolysates with Improved Interfacial Properties. Food Hydrocoll. 2023, 137, 108299. [Google Scholar] [CrossRef]
- García-Moreno, P.J.; Jacobsen, C.; Marcatili, P.; Gregersen, S.; Overgaard, M.T.; Andersen, M.L.; Sørensen, A.-D.M.; Hansen, E.B. Emulsifying Peptides from Potato Protein Predicted by Bioinformatics: Stabilization of Fish Oil-in-Water Emulsions. Food Hydrocoll. 2020, 101, 105529. [Google Scholar] [CrossRef]
- Yesiltas, B.; Soria Caindec, A.M.; García-Moreno, P.J.; Echers, S.G.; Olsen, T.H.; Jones, N.C.; Hoffmann, S.V.; Marcatili, P.; Overgaard, M.T.; Hansen, E.B.; et al. Physical and Oxidative Stability of Fish Oil-in-Water Emulsions Stabilized with Emulsifier Peptides Derived from Seaweed, Methanotrophic Bacteria and Potato Proteins. Colloids Surf. A Physicochem. Eng. Asp. 2023, 663, 131069. [Google Scholar] [CrossRef]
- Mikkelsen, R.K.; Queiroz, L.; Echers, S.G.; Hobley, T.; Overgaard, M.; Jacobsen, C.; Yesiltas, B. Extracting Proteins from Brewers’ Spent Grain Using Emerging Technologies: Evaluating Efficiency and Use as Emulsifier. TechRxiv 2024. [Google Scholar] [CrossRef]
- Celus, I.; Brijs, K.; Delcour, J.A. Enzymatic Hydrolysis of Brewers’ Spent Grain Proteins and Technofunctional Properties of the Resulting Hydrolysates. J. Agric. Food Chem. 2007, 55, 8703–8710. [Google Scholar] [CrossRef]
- Connolly, A.; Piggott, C.O.; FitzGerald, R.J. Technofunctional Properties of a Brewers’ Spent Grain Protein-Enriched Isolate and Its Associated Enzymatic Hydrolysates. LWT-Food Sci. Technol. 2014, 59, 1061–1067. [Google Scholar] [CrossRef]
- Vieira, M.C.; Brandelli, A.; Thys, R.C.S. Evaluation of the Technological Functional Properties and Antioxidant Activity of Protein Hydrolysate Obtained from Brewers’ Spent Grain. J. Food Process. Preserv. 2022, 46, e16638. [Google Scholar] [CrossRef]
- García-Moreno, P.J.; Yesiltas, B.; Gregersen Echers, S.; Marcatili, P.; Overgaard, M.T.; Hansen, E.B.; Jacobsen, C. Recent Advances in the Production of Emulsifying Peptides with the Aid of Proteomics and Bioinformatics. Curr. Opin. Food Sci. 2023, 51, 101039. [Google Scholar] [CrossRef]
- Gregersen Echers, S.; Mikkelsen, R.K.; Abdul-Khalek, N.; Queiroz, L.S.; Hobley, T.J.; Schulz, B.L.; Overgaard, M.T.; Jacobsen, C.; Yesiltas, B. Residual Barley Proteins in Brewers’ Spent Grains: Quantitative Composition and Implications for Food Ingredient Applications. Innov. Food Sci. Emerg. Technol. 2025, 106, 104277. [Google Scholar] [CrossRef]
- Kyte, J.; Doolittle, R.F. A Simple Method for Displaying the Hydropathic Character of a Protein. J. Mol. Biol. 1982, 157, 105–132. [Google Scholar] [CrossRef] [PubMed]
- Saito, M.; Masashi, O.; Koichi, C.; Makoto, S. Synthesis of a Peptide Emulsifier with an Amphiphilic Structure. Biosci. Biotechnol. Biochem. 1995, 59, 388–392. [Google Scholar] [CrossRef]
- Yesiltas, B.; Gregersen, S.; Lægsgaard, L.; Brinch, M.L.; Olsen, T.H.; Marcatili, P.; Overgaard, M.T.; Hansen, E.B.; Jacobsen, C.; García-Moreno, P.J. Emulsifier Peptides Derived from Seaweed, Methanotrophic Bacteria, and Potato Proteins Identified by Quantitative Proteomics and Bioinformatics. Food Chem. 2021, 362, 130217. [Google Scholar] [CrossRef]
- Enser, M.; Bloomberg, G.B.; Brock, C.; Clark, D.C. De Novo Design and Structure-Activity Relationships of Peptide Emulsifiers and Foaming Agents. Int. J. Biol. Macromol. 1990, 12, 118–124. [Google Scholar] [CrossRef]
- Dexter, A.F. Interfacial and Emulsifying Properties of Designed β-Strand Peptides. Langmuir 2010, 26, 17997–18007. [Google Scholar] [CrossRef]
- Delahaije, R.J.B.M.; Sagis, L.M.C.; Yang, J. Impact of Particle Sedimentation in Pendant Drop Tensiometry. Langmuir 2022, 38, 10183–10191. [Google Scholar] [CrossRef] [PubMed]
- Piorkowski, D.T.; McClements, D.J. Beverage Emulsions: Recent Developments in Formulation, Production, and Applications. Food Hydrocoll. 2014, 42, 5–41. [Google Scholar] [CrossRef]
- Wang, Y.; Li, D.; Wang, L.-J.; Adhikari, B. The Effect of Addition of Flaxseed Gum on the Emulsion Properties of Soybean Protein Isolate (SPI). J. Food Eng. 2011, 104, 56–62. [Google Scholar] [CrossRef]
- Monahan, F.J.; McClements, D.J.; German, J.B. Disulfide-Mediated Polymerization of Whey Proteins in Whey Protein Isolate-Stabilized Emulsions. In Food Proteins and Lipids; Damodaran, S., Ed.; Springer: Boston, MA, USA, 1997; pp. 127–136. [Google Scholar] [CrossRef]
- Hu, M.; McClements, D.J.; Decker, E.A. Lipid Oxidation in Corn Oil-in-Water Emulsions Stabilized by Casein, Whey Protein Isolate, and Soy Protein Isolate. J. Agric. Food Chem. 2003, 51, 1696–1700. [Google Scholar] [CrossRef] [PubMed]
- Hennebelle, M.; Villeneuve, P.; Durand, E.; Lecomte, J.; van Duynhoven, J.; Meynier, A.; Yesiltas, B.; Jacobsen, C.; Berton-Carabin, C. Lipid Oxidation in Emulsions: New Insights from the Past Two Decades. Prog. Lipid Res. 2024, 94, 101275. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.