Abstract
Bruxism is increasingly recognized as a psychosomatic behavior influenced by stress, anxiety, and sleep disturbances, which generates occlusal forces of non-physiological magnitude, potentially compromising the stability of dental implants. This narrative review examines the causal pathway linking psychological factors, bruxism, and implant complications. The literature from PubMed, Scopus, and Cochrane (2000–2025) was analyzed. Evidence consistently associates psychosocial stressors with increased mechanical complications, while biological outcomes remain heterogeneous. Digital occlusal analysis, finite element modeling, and AI-based monitoring show promise for early overload detection. An interdisciplinary model integrating dentistry, psychology, and sleep medicine is proposed to optimize implant outcomes and guide future research.
1. Introduction
Bruxism is defined as a repetitive jaw-muscle activity characterized by clenching or grinding of the teeth and bracing or thrusting of the mandible, which occurs during sleep (sleep bruxism) or wakefulness (awake bruxism) [1]. Contemporary consensus statements emphasize that bruxism should be understood as a behavior with multifactorial determinants rather than a purely mechanical or occlusal disorder, highlighting the central role of psychosocial stress, anxiety, and sleep-related factors in its pathophysiology [2]. This shift in perspective carries significant implications for implant dentistry, where sustained non-physiological occlusal forces may compromise prosthetic integrity and long-term outcomes.
Unlike natural teeth, dental implants lack a periodontal ligament and its associated proprioceptive and shock-absorbing functions, making implant-supported restorations particularly susceptible to biomechanical overload [3]. Clinical and biomechanical studies have associated bruxism with an increased incidence of mechanical complications, including screw loosening, ceramic chipping of veneers, and framework fractures [4]. However, evidence regarding biological complications such as marginal bone loss, peri-implantitis, and implant failure remains inconsistent, with some studies suggesting an elevated risk and others reporting survival rates comparable to those observed in non-bruxing patients [5,6,7]. This heterogeneity points to the complexity of the interaction between bruxism, host factors, and prosthetic design.
Advances in digital dentistry have introduced novel tools for assessing occlusal load and bruxism-related risk in implant patients. Digital occlusal analysis systems, finite element modeling, and emerging artificial intelligence-based approaches allow the objective evaluation of force distribution, stress concentration, and prosthetic vulnerability, enabling earlier detection of overload and more personalized preventive strategies [8,9,10]. At the same time, growing evidence supports the integration of psychological interventions and sleep medicine (particularly in patients with comorbid anxiety or obstructive sleep disorders) as part of a comprehensive approach to bruxism management.
Given the psychosomatic nature of bruxism and its potential impact on implant-supported rehabilitation, an interdisciplinary framework is increasingly warranted. The purpose of this narrative review is to synthesize current evidence linking psychological determinants, bruxism, and dental implant complications, and to propose a multidisciplinary, digitally assisted model for risk assessment, prevention, and long-term management. This approach aims to improve implant longevity and patient-centered outcomes while identifying priorities for future clinical research.
Despite the growing body of evidence linking bruxism to implant-related complications, the majority of published studies continue to conceptualize bruxism predominantly as a mechanical or occlusal risk factor. This reductionist approach has led to a fragmented understanding of implant overload, in which parafunctional forces are assessed largely in isolation from the psychological, neurophysiological, and sleep-related processes that generate and modulate them. As a consequence, clinical decision-making in implant dentistry often focuses on prosthetic or occlusal adjustments, while upstream psychosomatic drivers of bruxism remain insufficiently addressed. Concurrently, advances in neuroscience and behavioral medicine have increasingly characterized bruxism as a centrally mediated, biopsychosocial behavior associated with emotional regulation, stress responsiveness, and sleep-related arousal mechanisms. However, this paradigm shift has not been systematically translated into implant risk assessment or long-term maintenance strategies. Existing reviews frequently evaluate mechanical and biological outcomes independently, and few provide an integrative framework that connects psychological determinants, objective load assessment, prosthetic design, and interdisciplinary therapeutic interventions.
Therefore, a critical gap persists between contemporary conceptualizations of bruxism and their practical application in implant dentistry. This narrative review addresses this gap by synthesizing psychosocial, biomechanical, and emerging digital evidence to propose an interdisciplinary, clinically applicable model for the assessment, prevention, and long-term management of implant patients with bruxism. By reframing bruxism as a biopsychosocial load modifier rather than a purely mechanical parafunction, this work aims to support anticipatory, patient-centered strategies that move implant care beyond reactive management of complications.
2. Materials and Methods
We conducted a narrative review of the scientific literature with the objective of synthesizing evidence on the relationship between psychosomatic bruxism and complications in dental implant therapy. The review was designed to integrate clinical, psychological, biomechanical, and technological perspectives relevant to implant dentistry. A narrative review design was chosen to allow an integrative synthesis of heterogeneous evidence and to support the development of a conceptual, interdisciplinary clinical model, which would not be feasible within the constraints of a strict systematic review.
A comprehensive literature search was carried out using PubMed, Scopus, and the Cochrane Library. Preference was given to studies published between January 2000 and December 2025 to reflect contemporary diagnostic frameworks, biomechanical concepts, and digital technologies; however, seminal and clinically relevant articles published prior to 2000 were also included when necessary to contextualize foundational concepts. Search terms and Medical Subject Headings (MeSH), used alone or in combination, included bruxism, sleep bruxism, awake bruxism, dental implants, implant-supported prostheses, psychosomatic factors, stress, anxiety, sleep disorders, biomechanics, occlusal overload, implant failure, mechanical complications, digital dentistry, finite element analysis, artificial intelligence, and interdisciplinary management. Reference lists of selected articles were manually screened to identify additional relevant studies.
Study selection and synthesis process.
Eligible studies were selected based on predefined inclusion and exclusion criteria. Inclusion criteria comprised peer-reviewed clinical studies (observational or interventional), systematic or narrative reviews, biomechanical analyses with direct clinical relevance, and consensus statements addressing bruxism, psychosocial determinants, occlusal load, and dental implant outcomes. Exclusion criteria included non-peer-reviewed publications, conference abstracts without full text, animal or purely in vitro studies lacking clinical correlation, articles not written in English, and studies not directly related to implant-supported rehabilitations or bruxism-related loading.
Title and abstract screenings were independently performed by two authors, followed by full-text assessment of potentially eligible articles. Discrepancies were resolved by discussion and consensus among the authors.
Data synthesis was conducted using a thematic qualitative approach. Included studies were grouped into predefined conceptual domains: (1) psychosomatic and neurophysiological determinants of bruxism; (2) biomechanical implications for implant-supported restorations; (3) mechanical and biological implant complications; (4) digital and AI-assisted diagnostic tools; and (5) interdisciplinary management strategies. This narrative synthesis aimed to integrate heterogeneous evidence and support the development of a conceptual clinical model rather than to perform quantitative pooling or formal risk-of-bias assessment.
The initial literature search across PubMed, Scopus, and the Cochrane Library identified 214 records. After removal of duplicates and screening of titles and abstracts, 107 articles underwent full-text assessment. Following the eligibility evaluation, these publications were included in the final qualitative thematic synthesis. Given the narrative and model-building aim of this review, a formal PRISMA flow diagram was not generated.
Evidence was synthesized using a thematic qualitative approach. Extracted data were grouped into predefined conceptual domains, including psychosomatic determinants of bruxism, biomechanical implications for implant-supported restorations, implant-related complications, AI- and digitally assisted diagnostic tools, and interdisciplinary management strategies.
3. Bruxism as a Psychosomatic Behavior
Bruxism is currently conceptualized as a biopsychosocial behavior rather than a purely mechanical or occlusal disorder [2,11,12]. International consensus statements emphasize that bruxism is not inherently pathological but represents a behavior that may become clinically relevant when associated with negative consequences, such as dental wear, pain, or prosthetic complications [1,13]. This reconceptualization has shifted the focus from occlusal factors toward central nervous system mechanisms and psychosocial determinants, which are increasingly recognized as primary drivers of bruxism expression.
From a psychosomatic perspective, psychosocial stress and anxiety play a central role in the initiation and maintenance of bruxism [11]. Elevated stress levels activate autonomic and hypothalamic–pituitary–adrenal axis responses, leading to increased muscle tone and parafunctional jaw activity. Both sleep and awake bruxism have been associated with emotional dysregulation, heightened anxiety traits, and maladaptive coping strategies [12,14]. Awake bruxism, in particular, is strongly linked to conscious or semi-conscious stress-related behaviors, such as sustained clenching during periods of cognitive or emotional demand [15,16]. These findings support the interpretation of bruxism as an external motor manifestation of internal psychological load rather than a local occlusal phenomenon.
Neurophysiological mechanisms further support the psychosomatic model of bruxism. Sleep bruxism is considered a centrally mediated motor activity closely associated with sleep micro-arousals and fluctuations in autonomic nervous system activity [16]. Electromyographic and polysomnographic studies suggest that rhythmic masticatory muscle activity often occurs in temporal proximity to cortical arousals, heart rate acceleration, and sympathetic activation [17]. This pattern suggests that sleep bruxism reflects a broader arousal-related phenomenon rather than a response to peripheral dental stimuli. Neurotransmitter systems involved in stress and arousal regulation, including dopaminergic and serotonergic pathways, have also been implicated, reinforcing the link between emotional processing, sleep regulation, and motor output [18].
Sleep-related disorders, particularly obstructive sleep apnea, further illustrate the complex interaction between bruxism, neurophysiology, and psychosomatic stress [2,11]. In a subset of patients, sleep bruxism co-occurs with respiratory events and arousals, suggesting that jaw-muscle activity may represent a protective or reactive motor response to upper airway instability [19,20]. Although this association is not universal, it reinforces the importance of considering sleep quality, respiratory disturbances, and central arousal mechanisms when evaluating bruxism. Together, psychosocial stressors, neurophysiological arousal, and sleep disturbances form an integrated framework in which bruxism emerges as a multidimensional behavior with significant clinical implications for implant dentistry.
4. Biomechanical Implications of Bruxism in Implant Dentistry
Dental implants differ from natural teeth in their biomechanical behavior due to the absence of the periodontal ligament, which normally provides proprioception and shock absorption; consequently, occlusal forces applied to implant-supported restorations are transmitted directly to the implant–bone interface, making implants more susceptible to non-physiological loading [21]. Bruxism generates sustained, high-magnitude, and often non-axial forces that exceed normal masticatory loads, increasing mechanical stress on implant components and surrounding bone [6,22].
These parafunctional forces promote micro-movements at the implant–abutment interface and repeated loss of screw preload, leading to fatigue-related complications over time [23,24]. Finite element analysis studies consistently demonstrate stress concentration at the cervical implant region, abutment screw, and prosthetic framework under bruxism-like loading, particularly in the presence of increased crown height, steep occlusal morphology, and cantilevered designs [22,25].
Material properties and prosthetic design further influence load transmission [26]. Rigid restorative materials may amplify stress at the implant–bone interface, whereas more compliant materials may reduce stress transmission but increase the risk of veneering fractures [27]. Therefore, biomechanical optimization in patients with bruxism requires an integrated approach that considers implant positioning, framework design, occlusal scheme, and material selection. Overall, bruxism represents a significant biomechanical modifier in implant dentistry and warrants preventive and protective strategies in at-risk patients.
5. Implant-Related Complications Associated with Bruxism
5.1. Mechanical Complications
Mechanical complications represent the most consistently reported adverse outcomes associated with bruxism in implant-supported rehabilitations [4,23,28]. The sustained, repetitive, and frequently non-axial forces generated during bruxism episodes promote fatigue of prosthetic components, leading primarily to screw loosening and loss of preload at the implant–abutment interface [29]. This phenomenon is commonly reported as an early sign of overload and may predispose to secondary complications if not addressed.
In addition, ceramic chipping or fracture of veneering materials is frequently observed, particularly in implant-supported fixed restorations with extensive occlusal surfaces or parafunctional contacts [23]. Framework fractures, although less common, tend to occur in long-span prostheses or cantilevered designs, where stress concentration is amplified. Overall, mechanical complications are more prevalent than biological failures and often precede them, making their early recognition clinically relevant [4,28].
5.2. Biological Complications
The association between bruxism and biological complications remains less clear. Some studies suggest that excessive occlusal loading may contribute to marginal bone loss, potentially by exceeding the adaptive capacity of peri-implant bone [7,30]. However, findings across clinical studies are inconsistent, and bruxism alone does not appear to be a sufficient cause of peri-implant bone breakdown [31].
Similarly, evidence linking bruxism to peri-implantitis or implant failure is heterogeneous [32]. While certain meta-analyses report an increased risk of implant failure in patients with probable bruxism, multiple cohort studies substantiate implant survival rates comparable to those of non-bruxing patients, particularly when implants are properly planned and maintained [6,21,28]. These observations suggest that biological complications are influenced by multiple interacting factors beyond occlusal overload alone.
5.3. Evidence Consistency and Controversies
The variability in reported outcomes reflects substantial heterogeneity in study design, bruxism definitions, diagnostic methods, follow-up duration, and confounding variables such as smoking, implant design, prosthetic configuration, and comorbid sleep disorders [1,13]. Bruxism is often diagnosed based on patient self-report or clinical signs rather than instrumental assessment, limiting comparability across studies [33,34].
Taken together, current evidence supports the interpretation of bruxism as a risk indicator, particularly for mechanical complications and increased maintenance burden, rather than as an absolute predictor of implant failure [6,21]. This distinction is critical for clinical decision-making and underscores the importance of individualized risk assessment and long-term monitoring in implant patients with suspected bruxism.
6. Proposed Interdisciplinary Model for Implant Patients with Bruxism
Bruxism in the context of implant dentistry is best managed not as a single-domain problem but as a complex, multidimensional condition in which psychological stressors, sleep physiology, biomechanics, prosthetic design and materials, and long-term maintenance interact. Based on the preceding synthesis, we propose a structured, interdisciplinary model that (1) identifies at-risk patients early; (2) quantifies and characterizes the load environment objectively; (3) applies prosthetic and clinical measures to minimize overload; (4) treats upstream psychosomatic and sleep drivers; and (5) monitors outcomes with scheduled surveillance and adaptive interventions.
The model rests on four operational pillars:
- Systematic screening and risk stratification: detect psychosocial and sleep drivers and clinical signs of bruxism prior to definitive prosthetic rehabilitation.
- Digital and instrumental load assessment: obtain objective baseline measures of occlusal forces, timing, and bruxism activity to guide design and to enable longitudinal monitoring.
- Prosthetic and biological protection: apply surgical, prosthetic, material and occlusal strategies that reduce stress concentration and increase redundancy.
- Interdisciplinary therapeutic intervention: concurrent psychological, behavioral and sleep interventions to reduce the frequency/intensity of parafunction and thereby the cumulative mechanical load.
The primary clinical goals are to (a) reduce mechanical complication rates, (b) prevent progression to biological damage where possible, (c) minimize maintenance burden and emergency repairs, and (d) preserve patient function and quality of life.
6.1. Proposed Protocol
Following initial screening, objective diagnostic assessment plays a critical role in refining risk evaluation. Whenever feasible, instrumental confirmation of bruxism should be pursued, particularly for suspected sleep bruxism, using polysomnography, electromyography, or validated wearable devices. In parallel, baseline digital occlusal analysis should be performed at the time of prosthesis delivery to quantify contact timing and force distribution across implant-supported restorations. These measurements provide an individualized biomechanical profile that serves as a reference for future comparisons and facilitates early detection of overload patterns that may not be clinically evident.
The next phase focuses on prosthetic planning and surgical considerations tailored to the patient’s biomechanical risk. Implant number, distribution, and positioning should be optimized to minimize non-axial loading and reduce cantilever effects. Prosthetic designs should favor passive-fit frameworks, controlled crown height, and occlusal schemes that limit lateral forces on implants. Material selection should balance rigidity for structural stability with the capacity to mitigate stress concentration. In patients with active or poorly controlled bruxism, more conservative loading protocols may be considered to ensure adequate osseointegration before functional demands increase.
At the time of prosthesis delivery, protective and compensatory strategies should be implemented. Occlusal adjustment is essential to eliminate premature contacts and reduce excursion overload. In moderate- to high-risk patients, the provision of a full-coverage hard occlusal splint is recommended to redistribute forces and protect prosthetic components during parafunctional activity. Patient education at this stage is critical, emphasizing the behavioral nature of bruxism, the importance of adhering to splint use, and the role of stress and sleep quality in symptom modulation.
Interdisciplinary therapeutic interventions represent a core element of the pathway, as shown in Figure 1. Referral to psychological care enables the use of cognitive-behavioral therapy and stress-management strategies, which are particularly effective in reducing awake bruxism and improving emotional regulation. Concurrently, collaboration with sleep medicine specialists is essential for patients with suspected or confirmed sleep disorders. Diagnosis and treatment of obstructive sleep apnea, when present, may reduce sleep fragmentation and bruxism episodes in selected individuals. Adjunctive medical interventions, such as botulinum toxin injections into masticatory muscles, may be considered in refractory cases, although their use should be individualized and approached cautiously, given unresolved long-term implications.
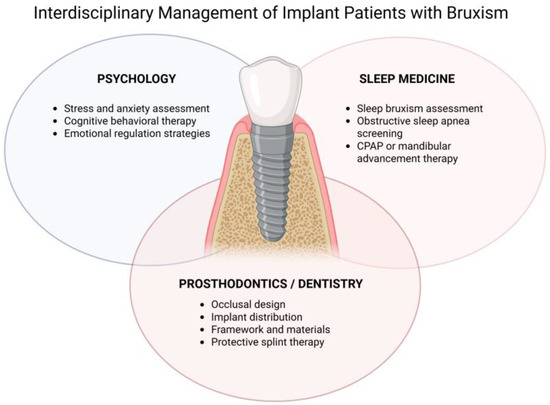
Figure 1.
Interdisciplinary Management of Implant Patients with Bruxism. Conceptual model illustrating the integration of psychological assessment and intervention, sleep medicine evaluation, and prosthodontic/implant-related strategies for the prevention and management of bruxism-related overload in implant-supported rehabilitations. The model emphasizes a dynamic and iterative approach centered on long-term monitoring and adaptive management.
Finally, long-term monitoring and adaptive management are fundamental to the success of the model. Patients should be enrolled in a structured follow-up program with more frequent recalls during the first year after prosthesis delivery, focusing on detection of mechanical complications, occlusal changes, and peri-implant tissue health. Digital occlusal assessments and radiographic evaluations should be repeated periodically to identify evolving overload trends. When complications or early warning signs are detected, the pathway allows for timely reassessment of psychosomatic factors, prosthetic design, and interdisciplinary interventions, ensuring that management strategies remain responsive to changes in patient status over time.
6.2. Integration of Digital and AI Tools into the Model
Digital technologies augment every pillar. Baseline and longitudinal digital occlusal analyses provide objective metrics that remove subjective ambiguity; wearable EMG/force sensors add nocturnal data otherwise missed in clinic; FEA supports design choices in complex cases; and AI algorithms—when validated—can synthesize longitudinal T-Scan, sensor, radiographic and clinical data to predict impending mechanical failure and prioritize patients for intervention [8,9,10,35]. In practice, begin with feasible digital steps (T-Scan and intraoral scans) and progressively incorporate sensor/AI tools as resources and evidence accrue.
7. Clinical Implications and Future Directions
The findings of this review emphasize the need to move beyond a purely mechanical interpretation of bruxism in implant dentistry and adopt a comprehensive, interdisciplinary approach to patient management. Clinically, bruxism should be considered a relevant risk indicator that warrants systematic screening for psychosocial stressors and sleep-related disorders prior to implant rehabilitation [1,2,11,34]. Incorporating digital occlusal analysis and structured follow-up protocols can facilitate early detection of overload and reduce the incidence of mechanical complications [9]. From a therapeutic perspective, collaboration with psychology and sleep medicine enhances preventive strategies and supports long-term implant stability [36,37,38]. Future research should prioritize prospective and randomized studies evaluating interdisciplinary interventions, define clinically meaningful thresholds for overload detection, and validate artificial intelligence-based tools for risk prediction and personalized maintenance strategies in implant patients with bruxism.
8. Limitations of the Review
This review has several limitations inherent to its narrative design. The heterogeneity of the included studies, variability in bruxism definitions and diagnostic methods, and differences in follow-up duration limit direct comparison of outcomes. Many studies rely on self-reported or clinically inferred bruxism rather than instrumental confirmation, which may introduce misclassification bias. These limitations illustrate the need for standardized diagnostic criteria, objective monitoring tools, and well-designed longitudinal studies to strengthen the evidence base.
9. Conclusions
Bruxism represents a complex psychosomatic behavior in which psychological stress, emotional regulation, sleep physiology, and neuromuscular activity interact to generate non-physiological occlusal loads. In the context of implant dentistry, these forces primarily compromise the mechanical integrity of implant-supported restorations, increasing the incidence of technical complications while exerting a more variable influence on biological outcomes. Current evidence supports considering bruxism as a clinically relevant risk indicator rather than an absolute contraindication for implant therapy.
This review indicates the limitations of traditional, purely mechanical approaches and emphasizes the need for an interdisciplinary model that integrates dentistry, psychology, and sleep medicine. Systematic screening, objective digital assessment of occlusal load, and individualized prosthetic design are essential components for mitigating overload-related risks. When combined with behavioral and sleep-focused interventions, these strategies allow for proactive management rather than reactive repair.
The proposed interdisciplinary, digitally assisted framework offers a structured pathway for improving implant longevity and patient-centered outcomes in individuals with bruxism. Its implementation in clinical practice, alongside prospective data collection and validation of emerging digital and artificial intelligence-based tools, has the potential to refine risk stratification, reduce complication rates, and advance evidence-based care in implant dentistry.
Ultimately, the challenge posed by bruxism in implant dentistry is not purely mechanical but epistemological: it forces clinicians to reconsider how psychological and neurophysiological processes translate into structural failure. Recognizing bruxism as a biopsychosocial load modifier rather than a simple parafunction reframes implant care from reactive repair to anticipatory prevention. This shift has the potential to redefine risk assessment and long-term success in modern implantology.
Author Contributions
Conceptualization, B.S.-P., C.D.-V. and P.M.P.-L.; methodology, C.D.-V.; software, C.D.-V.; validation, B.S.-P., P.M.P.-L. and D.A.T.-R.; formal analysis, C.D.-V.; investigation, B.S.-P.; resources, C.D.-V.; data curation, C.D.-V.; writing—original draft preparation, B.S.-P.; writing—review and editing, C.D.-V.; visualization, C.D.-V.; supervision, C.D.-V.; project administration, B.S.-P., C.D.-V. and P.M.P.-L.;. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data was created.
Acknowledgments
The authors thank Emiliano Peña-Durán for his valuable advisory support during the development of this work.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| AI | Artificial Intelligence |
| EMG | Electromyography |
| FEA | Finite Element Analysis |
References
- Lobbezoo, F.; Ahlberg, J.; Raphael, K.G.; Wetselaar, P.; Glaros, A.G.; Kato, T.; Santiago, V.; Winocur, E.; De Laat, A.; De Leeuw, R.; et al. International Consensus on the Assessment of Bruxism: Report of a Work in Progress. J. Oral Rehabil. 2018, 45, 837–844. [Google Scholar] [CrossRef] [PubMed]
- Manfredini, D.; Lobbezoo, F. Role of Psychosocial Factors in the Etiology of Bruxism. J. Orofac. Pain. 2009, 23, 153–166. [Google Scholar] [PubMed]
- Jacobs, R.; van Steenberghe, D. Role of Periodontal Ligament Receptors in the Tactile Function of Teeth: A Review. J. Periodontal Res. 1994, 29, 153–167. [Google Scholar] [CrossRef]
- Häggman-Henrikson, B.; Ali, D.; Aljamal, M.; Chrcanovic, B.R. Bruxism and Dental Implants: A Systematic Review and Meta-Analysis. J. Oral Rehabil. 2024, 51, 202–217. [Google Scholar] [CrossRef]
- Naert, I.; Koutsikakis, G.; Duyck, J.; Quirynen, M.; Jacobs, R.; van Steenberghe, D. Biologic Outcome of Implant-Supported Restorations in the Treatment of Partial Edentulism. Part I: A Longitudinal Clinical Evaluation. Clin. Oral Implants Res. 2002, 13, 381–389. [Google Scholar] [CrossRef]
- Zhou, Y.; Gao, J.; Luo, L.; Wang, Y. Does Bruxism Contribute to Dental Implant Failure? A Systematic Review and Meta-Analysis. Clin. Implant. Dent. Relat. Res. 2016, 18, 410–420. [Google Scholar] [CrossRef] [PubMed]
- Chambrone, L.; Chambrone, L.A.; Lima, L.A. Effects of Occlusal Overload on Peri-Implant Tissue Health: A Systematic Review of Animal-Model Studies. J. Periodontol. 2010, 81, 1367–1378. [Google Scholar] [CrossRef]
- Proshchenko, A.; Terekhov, S.; Vesova, O.; Kaminskyy, V.; Kryvosheieva, A. UTILIZATION OF ARTIFICIAL INTELLIGENCE FOR PREDICTIVE MODELING IN DENTAL IMPLANTOLOGY. Georgian Med. News 2024, 6–15. [Google Scholar]
- Moraschini, V.; de Almeida, D.C.F.; Louro, R.S.; de Oliveira Silva, A.M.; Neto, M.P.C.; Dos Santos, G.O.; Granjeiro, J.M. Accuracy of Artificial Intelligence in Implant Dentistry: A Scoping Review with Systematic Evidence Mapping. J. Prosthet. Dent. 2025, 133, 1461.e1–1461.e10. [Google Scholar] [CrossRef]
- Revilla-León, M.; Gómez-Polo, M.; Vyas, S.; Barmak, B.A.; Galluci, G.O.; Att, W.; Krishnamurthy, V.R. Artificial Intelligence Applications in Implant Dentistry: A Systematic Review. J. Prosthet. Dent. 2023, 129, 293–300. [Google Scholar] [CrossRef]
- Kuhn, M.; Türp, J.C. Risk Factors for Bruxism. Swiss Dent. J. 2018, 128, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Flueraşu, M.I.; Bocşan, I.C.; Țig, I.-A.; Iacob, S.M.; Popa, D.; Buduru, S. The Epidemiology of Bruxism in Relation to Psychological Factors. Int. J. Environ. Res. Public Health 2022, 19, 691. [Google Scholar] [CrossRef] [PubMed]
- Lobbezoo, F.; Ahlberg, J.; Glaros, A.G.; Kato, T.; Koyano, K.; Lavigne, G.J.; de Leeuw, R.; Manfredini, D.; Svensson, P.; Winocur, E. Bruxism Defined and Graded: An International Consensus. J. Oral Rehabil. 2013, 40, 2–4. [Google Scholar] [CrossRef] [PubMed]
- Renner, A.C.; da Silva, A.A.M.; Rodriguez, J.D.M.; Simões, V.M.F.; Barbieri, M.A.; Bettiol, H.; Thomaz, E.B.A.F.; Saraiva, M.d.C. Are Mental Health Problems and Depression Associated with Bruxism in Children? Community Dent. Oral Epidemiol. 2012, 40, 277–287. [Google Scholar] [CrossRef]
- Przystańska, A.; Jasielska, A.; Ziarko, M.; Pobudek-Radzikowska, M.; Maciejewska-Szaniec, Z.; Prylińska-Czyżewska, A.; Wierzbik-Strońska, M.; Gorajska, M.; Czajka-Jakubowska, A. Psychosocial Predictors of Bruxism. BioMed Res. Int. 2019, 2019, 2069716. [Google Scholar] [CrossRef]
- Osses-Anguita, Á.E.; Sánchez-Sánchez, T.; Soto-Goñi, X.A.; García-González, M.; Alén Fariñas, F.; Cid-Verdejo, R.; Sánchez Romero, E.A.; Jiménez-Ortega, L. Awake and Sleep Bruxism Prevalence and Their Associated Psychological Factors in First-Year University Students: A Pre-Mid-Post COVID-19 Pandemic Comparison. Int. J. Environ. Res. Public Health 2023, 20, 2452. [Google Scholar] [CrossRef]
- Lan, K.-W.; Jiang, L.-L.; Yan, Y. Comparative Study of Surface Electromyography of Masticatory Muscles in Patients with Different Types of Bruxism. World J. Clin. Cases 2022, 10, 6876–6889. [Google Scholar] [CrossRef]
- Campello, C.P.; Moraes, S.L.D.; do E.Vasconcelos, B.C.; de Lima, E.L.S.; Pellizzer, E.P.; Lemos, C.A.A.; Muniz, M.T.C. Polymorphisms of the Serotonin Receptors Genes in Patients with Bruxism: A Systematic Review. J. Appl. Oral Sci. 2022, 29, e20210262. [Google Scholar] [CrossRef]
- Martynowicz, H.; Gac, P.; Brzecka, A.; Poreba, R.; Wojakowska, A.; Mazur, G.; Smardz, J.; Wieckiewicz, M. The Relationship between Sleep Bruxism and Obstructive Sleep Apnea Based on Polysomnographic Findings. J. Clin. Med. 2019, 8, 1653. [Google Scholar] [CrossRef]
- Miki, H.; Minakuchi, H.; Miyagi, M.; Hara, E.S.; Shigemoto, S.; Suzuki, Y.; Maekawa, K.; Matsuka, Y.; Clark, G.T.; Kuboki, T. Association of Masticatory Muscle Activity with Sleep Arousal and Other Concomitant Movements during Sleep. J. Oral Rehabil. 2020, 47, 281–288. [Google Scholar] [CrossRef]
- Ionfrida, J.A.; Stiller, H.L.; Kämmerer, P.W.; Walter, C. Dental Implant Failure Risk in Patients with Bruxism-A Systematic Review and Meta-Analysis of the Literature. Dent. J. 2024, 13, 11. [Google Scholar] [CrossRef]
- Shafiee, E.; Nourizadeh, A. Bruxism in Implant-Supported Rehabilitations: A Narrative Review of Clinical Complications and Management Strategies. BMC Oral Health 2025, 25, 1586. [Google Scholar] [CrossRef]
- Germán-Sandoval, R.; Ortiz-Magdaleno, M.; Sánchez-Robles, P.; Zavala-Alonso, N.; Fernando Romo-Ramírez, G.; Germán-Sandoval, R.; Ortiz-Magdaleno, M.; Sánchez-Robles, P.; Zavala-Alonso, N.; Fernando Romo-Ramírez, G. Analysis of the Mechanical Behavior and Effect of Cyclic Fatigue on the Implant-Abutment Interface. Odovtos Int. J. Dent. Sci. 2021, 23, 104–114. [Google Scholar] [CrossRef]
- Rangert, B.; Jemt, T.; Jörneus, L. Forces and Moments on Branemark Implants. Int. J. Oral Maxillofac. Implants 1989, 4, 241–247. [Google Scholar]
- Song, J.-Y. Implant Complications in Bruxism Patients. J. Korean Assoc. Oral Maxillofac. Surg. 2021, 47, 149–150. [Google Scholar] [CrossRef] [PubMed]
- Martins, R.G.; de Castro, T.S.; Dib, L.L.; Gehrke, S.A.; Mesquita, A.M.M. Influence of Restorative Material on the Distribution of Loads to the Bone in Hybrid Abutment Crowns—In Vitro Study. Medicina 2023, 59, 1188. [Google Scholar] [CrossRef] [PubMed]
- Datte, C.-E.; Tribst, J.-P.-M.; Dal Piva, A.-M.O.; Nishioka, R.-S.; Bottino, M.-A.; Evangelhista, A.-D.M.; de M. Monteiro, F.M.; Borges, A.-L.-S. Influence of Different Restorative Materials on the Stress Distribution in Dental Implants. J. Clin. Exp. Dent. 2018, 10, e439–e444. [Google Scholar] [CrossRef]
- Vidal, R.A.; Martins, V.C.; Figueiredo, E.Z.; Roithmann, C.C.; Bertuzzi, D.; Grossi, M.L. Relationship between bruxism and different types of mechanical complications in implant-supported prosthesis: A systematic review with meta-analysis. J. Prosthodont. 2025, 1–21. [Google Scholar] [CrossRef]
- Gratton, D.G.; Aquilino, S.A.; Stanford, C.M. Micromotion and Dynamic Fatigue Properties of the Dental Implant-Abutment Interface. J. Prosthet. Dent. 2001, 85, 47–52. [Google Scholar] [CrossRef]
- Isidor, F. Influence of Forces on Peri-Implant Bone. Clin. Oral Implants Res. 2006, 17, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Oh, T.-J.; Yoon, J.; Misch, C.E.; Wang, H.-L. The Causes of Early Implant Bone Loss: Myth or Science? J. Periodontol. 2002, 73, 322–333. [Google Scholar] [CrossRef]
- Di Fiore, A.; Montagner, M.; Sivolella, S.; Stellini, E.; Yilmaz, B.; Brunello, G. Peri-Implant Bone Loss and Overload: A Systematic Review Focusing on Occlusal Analysis through Digital and Analogic Methods. J. Clin. Med. 2022, 11, 4812. [Google Scholar] [CrossRef]
- Bulanda, S.; Ilczuk-Rypuła, D.; Nitecka-Buchta, A.; Nowak, Z.; Baron, S.; Postek-Stefańska, L. Sleep Bruxism in Children: Etiology, Diagnosis, and Treatment-A Literature Review. Int. J. Environ. Res. Public Health 2021, 18, 9544. [Google Scholar] [CrossRef] [PubMed]
- Verhoeff, M.C.; Lobbezoo, F.; Ahlberg, J.; Bender, S.; Bracci, A.; Colonna, A.; Dal Fabbro, C.; Durham, J.; Glaros, A.G.; Häggman-Henrikson, B.; et al. Updating the Bruxism Definitions: Report of an International Consensus Meeting. J. Oral Rehabil. 2025, 52, 1335–1342. [Google Scholar] [CrossRef] [PubMed]
- Shopova, D. Digital Occlusion Tracking by T-Scan Novus System in Bruxism Patients Treated With 3D Printed Occlusal Splints. Int. J. Dent. 2025, 2025, 8842498. [Google Scholar] [CrossRef]
- Yap, A.U.; Chua, A.P. Sleep Bruxism: Current Knowledge and Contemporary Management. J. Conserv. Dent. 2016, 19, 383–389. [Google Scholar] [CrossRef]
- Lal, S.J.; Sankari, A.; Weber, D.D.S. Bruxism Management. In StatPearls; StatPearls Publishing: Treasure Island (FL), USA, 2025. Available online: https://www.ncbi.nlm.nih.gov/books/NBK482466/ (accessed on 7 September 2025).
- Minakuchi, H.; Fujisawa, M.; Abe, Y.; Iida, T.; Oki, K.; Okura, K.; Tanabe, N.; Nishiyama, A. Managements of Sleep Bruxism in Adult: A Systematic Review. Jpn. Dent. Sci. Rev. 2022, 58, 124–136. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.