Abstract
Solubility studies are an essential requirement for the development of more efficient industrial processes. In this context, the use of cosolvents is a relevant strategy in pharmaceutical sciences, especially when dealing with green solvents such as water (W (2)) and Polyethylene glycol 400 (PEG 400 (1)). The objective of this study is to thermodynamically analyze the solubility of isoniazid in {PEG 400 (1) + W (2)} cosolvent mixtures at seven temperatures (288.15 to 318.15 K). The study was conducted by calculating thermodynamic functions from experimental solubility data determined using the flask shaking method, employing UV spectrophotometry as the quantification technique. The dissolution process was shown to be endothermic and entropy-driven. Although maximum solubility would be expected to be achieved in a cosolvent mixture, given that the solubility parameter of isoniazid (30.54 MPa1/2) has an intermediate value between the two pure solvents (PEG 400 ≈ 22.5 MPa1/2; Water MPa1/2), maximum solubility is achieved in pure PEG 400 and the lowest solubility is achieved in pure water.
1. Introduction
The solubility of active pharmaceutical ingredients (APIs) in mixed liquid systems is a physicochemical strategy of paramount importance in pharmaceutical sciences and liquid materials engineering [1,2]. Precise knowledge of phase equilibrium phenomena is crucial not only for designing stable and effective liquid dosage forms but also for optimizing purification and crystallization processes and developing theoretical models of liquid structure [3,4].
Isoniazid (Figure 1; IUPAC Name: pyridine-4-carbohydrazide; Molecular Formula: C6H7N3O; CAS: 54-85-3) is a first-line drug for the prevention and treatment of Tuberculosis, a disease that in 2024 caused the death of 1.23 million people, making it one of the top 10 diseases that cause the most deaths worldwide [5,6]. Although the aqueous solubility of isoniazid is not very low, the development of dosage strategies, especially pediatric and geriatric formulations, can be approached more rationally through the use of cosolvents [7,8,9]. In this context, cosolvency is one of the most versatile and simple strategies to modify drug solubility, in addition to allowing an analysis of possible solute–solvent molecular interactions [10]. One of the most important cosolvents in the pharmaceutical industry is PEG 400, due to its miscibility with water in all proportions because of its amphiphilic nature, and its properties such as non-toxicity, biocompatibility, and biodegradability, classifying it as a green solvent. Furthermore, PEG 400 is an economical solvent, safe to handle, recyclable, non-flammable, easy to use, and, most importantly, environmentally friendly and widely available, making it a solvent of great interest to the chemical–pharmaceutical industry [11,12].
Figure 1.
Molecular structure of isoniazid.
Thus, the {PEG 400 (1) + W (2)} cosolvent mixture is a practical system, and given its supramolecular structural complexity, it allows modulation of the physicochemical properties of this drug [13]. However, the physicochemical information generated exclusively through experimental solubility data is not sufficient to explain the mechanisms involved in the solution process, which makes it necessary to conduct a strict thermodynamic analysis to elucidate the possible molecular mechanisms involved in the solubility of the drug [14]. In this context, a thermodynamic analysis of the solubility of isonicid in {PEG 400 (1) + W (2)} cosolvent mixtures is developed at seven temperatures (288.15, 293.15, 298.15, 303.15, 308.15, 313.15, and 318.15 K). The apparent thermodynamic functions of the solution and mixture are calculated to evaluate the energetics of the dissolution process. Additionally, the enthalpy–entropy compensation is evaluated to analyze the possible thermodynamic mechanisms driving the process.
2. Materials and Methods
The solubility of isoniazid in {PEG 400 (1) + W (2)} cosolvent mixtures was quantified using UV/Vis spectrophotometry following the established protocol for the shake-flask method [15]. The shake-flask method consists of at least four steps. Initially, each sample is saturated in amber-colored flasks to minimize the likelihood of light degradation. These flasks are then placed in a recirculating bath at a constant temperature for approximately 48 h. Quantification is performed by preparing a validated calibration curve. The equilibrium solid phase obtained by recrystallization from a saturated solution at a temperature T is analyzed using DSC [7] (for more details, please refer to the Supplementary Files).
3. Results
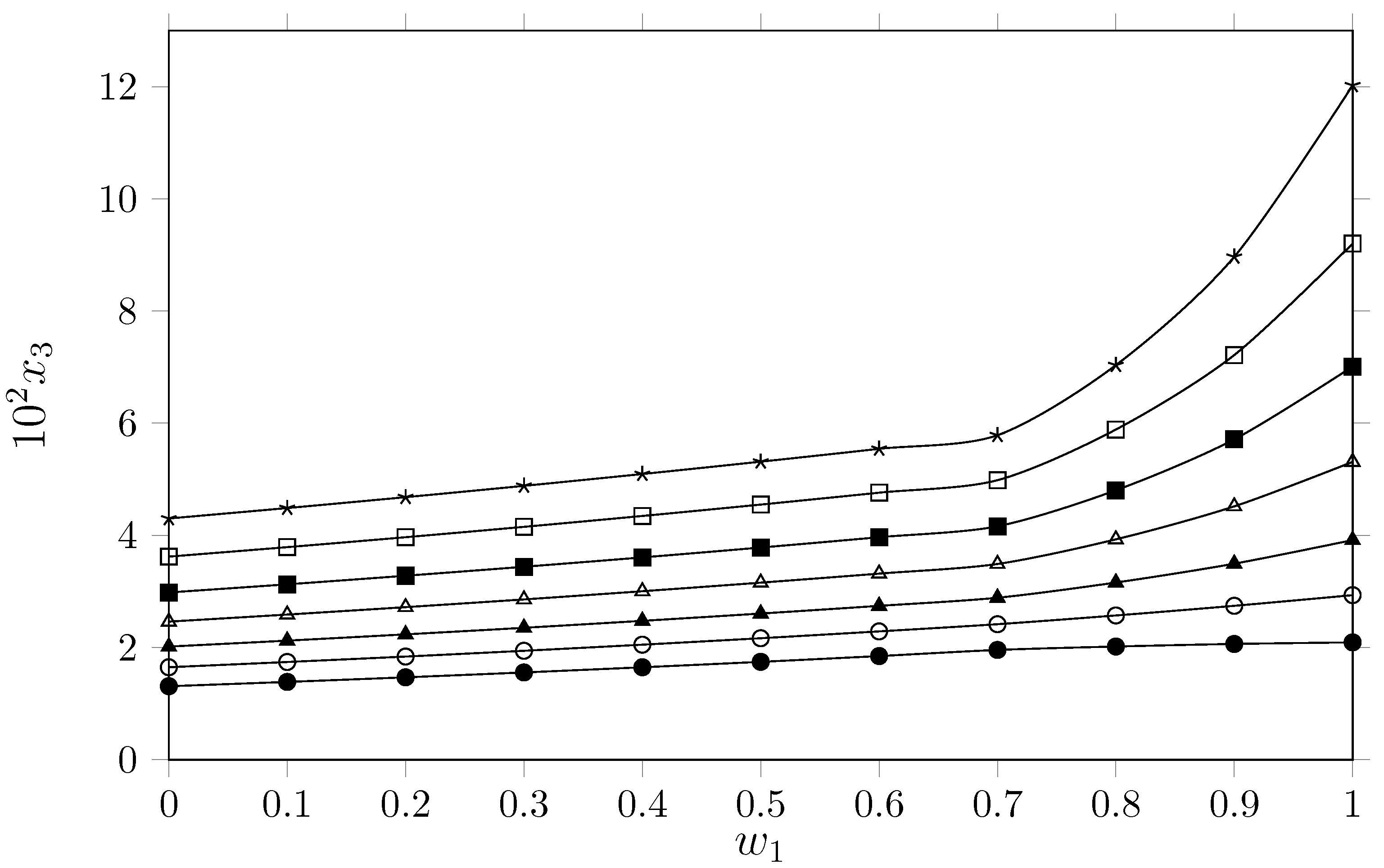
The analysis of isoniazid solubility in the binary PEG 400 + water system reveals a marked positive cosolvent effect and a significant temperature dependence. As shown in Table 1 and Figure 2, isoniazid solubility increases with the mass fraction of PEG 400 (), reaching maximum values in pure PEG 400 () and minimum values in pure water () across all studied isotherms (288.15 K to 318.15 K). It is important to indicate here that the isoniazid solubility in pure water has been discussed in our previous paper [7].

Table 1.
Experimental solubility of isoniazid (3) in (PEG 400 (1) + water (2)) cosolvent mixtures expressed in mole fraction () at different temperatures. Experimental pressure p: 0.1 MPa.
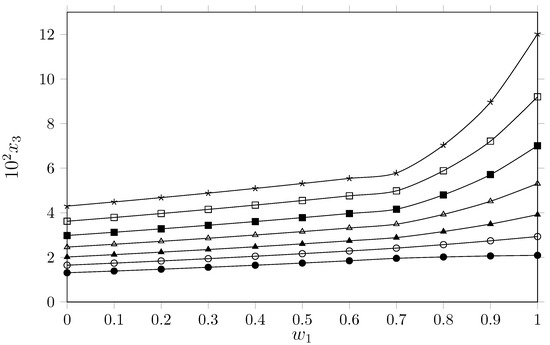
Figure 2.
Mole fraction of isoniazid () depending on the cosolvent composition {PEG 400 (1) + W (2)} (mass fraction) free of solute. ●: 288.15 K; ○: 293.15 K; ▲: 298.15 K; △: 303.15 K; ◼: 308.15 K; ☐: 313.15 K, and ★: 318.15 K.
This behavior suggests that the PEG 400 molecular environment is thermodynamically more favorable for accommodating isoniazid molecules than the pure aqueous environment. From the perspective of molecular interactions, isoniazid is an amphipathic molecule comprising a hydrophobic pyridine ring and a hydrophilic hydrazide group capable of forming hydrogen bonds. Due to its high cohesive energy density and highly ordered hydrogen bond network, water incurs a high energetic cost to form the cavity required to house the hydrophobic moiety of the drug (the pyridine ring). The addition of PEG 400 decreases the solubility parameter of the cosolvent mixture due to the nonpolar groups (aliphatic chain) and the slightly polar groups such as the ether groups of PEG 400. It is possible that the increased mass fraction of PEG 400 generates a structural change that disrupts the aqueous network by disrupting the water around the nonpolar groups of the drug, favoring dipole–dipole interactions and London dispersion forces between the solvent medium and the aromatic ring of the isoniazid, while maintaining hydrogen bond interactions with the hydrazide group. The increased solubility in all cosolvent mixtures as a result of the temperature increase indicates that the dissolution process is endothermic. This suggests that the supplied energy breaks solute–solute and solvent–solvent interactions, increasing the drug’s solubility. Analyzing the solubility of isoniazid as a function of the solvents’ solubility parameters () shows a greater affinity of the drug for PEG 400. According to the comparative study by Baracaldo-Santamaría et al. [7], the solubility parameter of isoniazid is approximately 30.54 . Water exhibits a high solubility parameter ( [16]), making it highly polar relative to the drug. In contrast, polyethylene glycols present lower values closer to that of isoniazid (e.g., for PEG 200, and slightly lower for PEG 400 ( [17]) due to its longer hydrocarbon chain). As the proportion of PEG 400 increases, the solubility parameter of the solvent mixture decreases, approaching that of the solute (30.54 ). However, according to the regular solution theory [2], the maximum solubility of isoniazid would be expected in a cosolvent mixture with a similar to that of the drug rather than in a pure solvent with a lower polarity, such as PEG 400 () regardless the solubility magnitude; therefore, Hildebrand’s theory of regular solutions does not conclusively explain the solubility behavior of isoniazid in cosolvent mixtures {PEG 400 (1) + water 2}. A more reasonable explanation may be an increase in specific forces (hydrogen bonds) between isoniazid and PEG 400. Comparing these results with those reported by Baracaldo-Santamaría et al. [7], for the {PEG 200 (1) + W (2)} system, it reveals a qualitatively identical trend: in both cases, the cosolvent effect is positive, with maximum solubility occurring in the pure polyethylene glycol. Quantitative differences related to polymer chain length are evident; the higher polarity of PEG 200 compared to PEG 400 suggests that solubility is modulated by interactions with isoniazid’s hydrophobic moiety. In pure organic solvents, Heryanto et al. [18] established that isoniazid is most soluble in methanol, followed by acetone, ethanol, and ethyl acetate. The high solubility observed here in PEG 400 supports these data, as PEG 400 combines the hydroxyl functionality of lower alcohols with the amphiphilic nature of a linear polymer. While Heryanto attributed methanol’s efficacy to its polarity and hydrogen bonding capacity (), the results for PEG 400 (and Baracaldo’s PEG 200 data [7]) point to a requirement for thermodynamic balance. Isoniazid requires both proton donor/acceptor capabilities to stabilize the hydrazide group and apolar regions—specifically the ethylene moieties in PEG—to facilitate favorable interactions with the pyridine ring, a mechanism largely absent in pure water.
3.1. Activity Coefficients
The analysis of the activity coefficients () (Table 2) allows for the elucidation of molecular interaction behavior within the system, considering that is defined as the ratio of ideal solubility ( Equation (1)) to experimental solubility () [2].
where T and are in K, is the enthalpy of fusion (in kJ/mol) of the solute, R is the gas constant (in kJ/mol·K), and is the differential heat capacity of fusion (in kJ/mol·K) [19]. Some researchers such as Hildebrand et al. [2], Neau and Flynn [4], Neau et al. [20], and Opperhuizen et al. [3], assume as the entropy of fusion (), which is calculated as .

Table 2.
Activity coefficient of isoniazid (3) in {PEG 400 (1) + water (2)} cosolvent mixtures at different temperatures and a pressure p = 0.096 MPa.
Thus, the ideal solubility () at the study temperature is calculated based on the melting temperature (445.1 K) and enthalpy (28.1 kJ/mol) data [7] (namely, 288.15 K = 0.0368 ± 0.0022; 293.15 K = 0.042 ± 0.0024; 298.15 K = 0.0477 ± 0.0027; 303.15 K = 0.0541 ± 0.0029; 308.15 K = 0.0613 ± 0.0032; 313.15 K = 0.0693 ± 0.0034; 318.15 K = 0.0781 ± 0.0037; 318.15 K = 0.0781 ± 0.0037).
In water-rich mixtures ( to ), values remain significantly above unity—observed, for instance, at 2.81 at 288.15 K in pure water—indicating a positive deviation from ideality. From a molecular standpoint, this suggests that solvent–solvent interactions (, dominated by water’s structured hydrogen bond network) and solute–solute interactions () are energetically favored over solute–solvent interactions (). Such energetic preferences impede the incorporation of isoniazid into the aqueous phase. Conversely, increasing the PEG 400 mass fraction () consistently reduces across all isotherms. Notably, at 298.15 K, the coefficient decreases from 2.37 in pure water to 1.22 in pure PEG 400, eventually dropping below unity (0.65) at 318.15 K in the pure cosolvent. In the context of molecular interaction analysis () [19,20,21], this trend demonstrates that adding PEG 400 substantially enhances solute–solvent affinity (), thereby lowering the energy barrier for cavity formation and solvation. Consequently, the tendency of to approach or fall below unity in regions rich in cosolvent (PEG 400) implies nearly-ideal behavior. This behavior could be attributed to a greater affinity between PEG 400 and isoniazid. Finally, the effect of temperature on activity coefficients exhibits an inverse correlation; as the temperature rises from 288.15 K to 318.15 K, decreases across all cosolvent compositions. This decrease with increasing thermal energy confirms an enhancement in favorable molecular interactions between isoniazid and the solvent system (), facilitating the dissolution process. Furthermore, this validates that at higher thermal energies, the system more effectively overcomes the cohesive energy barriers of both the solute and the solvent, approximating more closely the calculated ideal solubility.
3.2. Thermodynamic Functions of Solution and Mixing Processes
The apparent thermodynamic solute functions are calculated according to the Gibbs-van’t Hoff–Krug model [22,23], according to Expressions (2)–(6).
where (in kJ/mol), (in kJ/mol), and (in kJ/mol·K) are the thermodynamic functions: enthalpy, Gibbs energy, and entropy of solution. T is the study temperature (in K), is the harmonic mean temperature (in K), R is the gas constant (kJ/mol·K), and and are the enthalpic and entropic contributions to the solution process.
The harmonic temperature is calculated as follows:
where n is the number of study temperatures (in this case 9).
From the van’t Hoff–Krug equation
The and are calculated, according to Equations (2) and (3) using the values of m (slope) and a (intercept) of Equation (8).
The thermodynamic mixing functions are calculated as follows:
In this research, is replaced by the thermodynamic functions relative to the ideal solution process, which are calculated from the ideal solubility at .
Thus, the behavior of the standard Gibbs energy of solution (), presented in Table 3, must be primarily interpreted as a measure of the chemical affinity between the solute and the solvent medium, given that the system is at saturation equilibrium.

Table 3.
Thermodynamic functions of the solution process of isoniazid (3) in {PEG 400 (1) + water (2)} cosolvent mixtures at .
The positive values of across the entire composition range (decreasing from 9.4 kJ/mol in pure water to 7.46 kJ/mol in pure PEG 400) are a mathematical consequence of calculating from the mole fraction, which is inherently less than unity [24]. However, the decreasing trend of this function as the proportion of PEG 400 () increases reflects a reduction in the net energy required to transfer isoniazid molecules from the solid state to the solution state. This evidences a higher thermodynamic affinity of the drug for polymer-rich mixtures compared to the pure aqueous medium. Therefore, the energy required to overcome the crystal lattice forces of isoniazid and form a cavity in the solvent matrix exceeds the energy released by solute–solvent molecular interactions. Increasing the concentration of PEG 400 resulted in a decrease in enthalpy from 30.1 kJ/mol in pure water to 27.6 kJ/mol in the cosolvent mixture with a mass fraction of 0.7 of PEG 400. From this cosolvent mixture, with the addition of PEG 400, the standard enthalpy of solution increased to 44.2 kJ/mol in pure PEG 400. The above suggests that in water-rich mixtures, the addition of PG 400 to the cosolvent mixture could disrupt the water structure, reducing the energy required to form the solute-occupying cavity. However, starting with the cosolvent mixture = 0.7, the increase in the standard solution entropy is possibly due to the difficulty in separating the polymer chains, which are likely entangled, as well as some solute–solvent interactions, making it less exothermic than the cosolvent mixture.
When analyzing the entropic behavior, the process is favored by the standard entropy of solution across the entire range of cosolvent compositions, since entropy always has positive values (). The entropy trend is analogous to that of enthalpy, showing a marked increase in mixtures rich in PEG 400, where it reaches a maximum value of 116.8 J/mol·K in pure PEG 400. This significant gain in entropy compensates for the enthalpic cost of pure PEG 400, boosting solubility in PEG 400-rich media. Furthermore, when analyzing the contribution of the enthalpy and entropy of solution to the standard Gibbs free energy of solution, in all cases the enthalpy contribution () is greater than 0.5, which implies that the Gibbs free energy value depends more heavily on the energy component.
Regarding the thermodynamic functions of mixing (Table 4), the Gibbs energy of mixing () exhibits positive values in water-rich regions, progressively decreasing to slightly negative values in pure PEG 400 (−0.1 kJ/mol). Positive values indicate non-ideal deviations where the experimental solubility is lower than that predicted by the ideal model, except in the pure cosolvent, where the system approaches or slightly exceeds ideality. Thermodynamically, the mixing process is endothermic () and entropy-driven (). The positive enthalpy of mixing (ranging from 11.4 to 25.5 kJ/mol) suggests that the net isoniazid–solvent interaction is energetically weaker than the homotactic interactions (solvent–solvent and solute–solute) that must be disrupted. However, the entropy of mixing is highly positive, particularly in pure PEG 400 (84 J/mol·K), indicating that mixing the solute with the flexible polymer chains generates a significantly higher number of configurational microstates than in the more rigid structure of water or in an ideal mixture. In conclusion, the higher solubility (affinity) of isoniazid in PEG 400 is not due to stronger energetic interactions (enthalpy), but rather to a dominant entropic effect derived from the destructuring of the system and the greater conformational freedom within the polymeric environment.

Table 4.
Thermodynamic functions of mixing isoniazid (3) in {PEG 400 (1) + water (2)} cosolvent mixtures at 302.82 K and a pressure p = 0.096 MPa.
3.3. Enthalpy–Entropy Compensation (EEC) Analysis
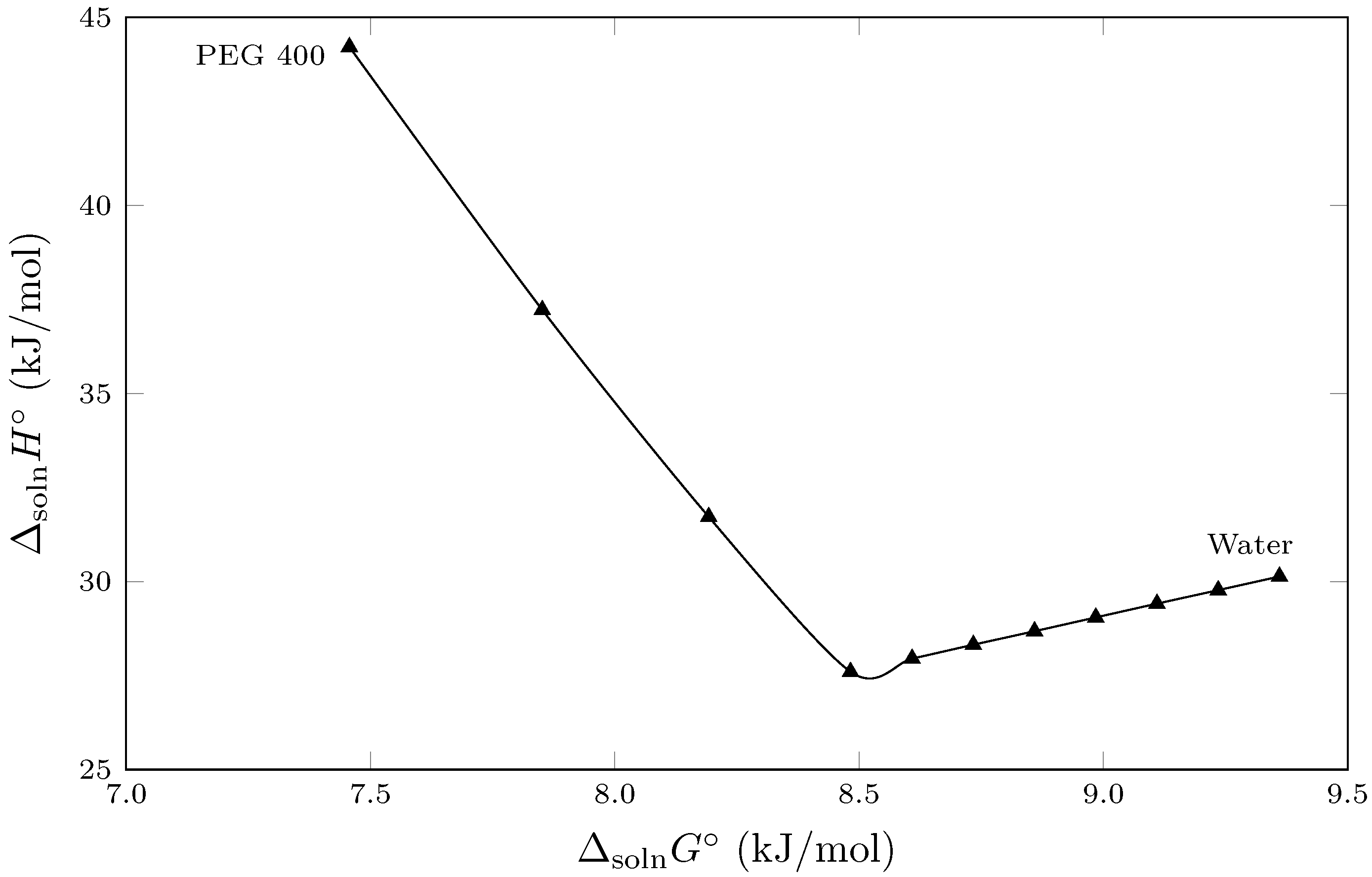
Figure 3 shows the enthalpy–entropic compensation behavior ( vs. ) of the isoniazid solution process in cosolvent mixtures {PEG 400 (1) + W (2)} cosolvent mixtures. In this context, the EEC graph exhibits nonlinear behavior, with two trends having opposite slopes, implying that two solvation mechanisms are present in the solution process. Thus, from pure water to the cosolvent mixture (), both the standard enthalpy of solution () and the standard Gibbs energy of solution () decrease as the mass fraction of PEG 400 increases. This behavior suggests that the addition of PEG 400 affects the ordered network of hydrogen bonds in water (structured water), decreasing the energetics associated with cavity formation to accommodate the solute. Thus, from pure water up to , the process is driven by the enthalpy of solution [25,26].
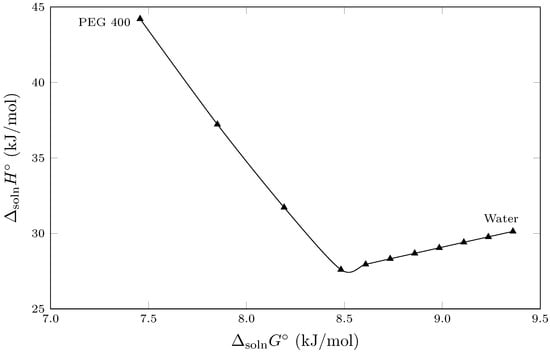
Figure 3.
Enthalpy–entropy compensation plot for the solubility of isoniazid (3) in {PEG 400 (1) + water (2)} mixtures at = 302.82 K.
The other trend described in Figure 3 occurs from cosolvent mixtures with to pure PEG 400. In this range, the standard enthalpy of solution () increases from its lowest value at (23.24±0.18 kJ/mol) to PEG, where it reaches its highest value (42.96±0.16 kJ/mol), changing the solvation mechanism observed in water-rich mixtures. Thus, as the mass fraction of PEG 400 increases, the standard enthalpy of solution () increases; However, the standard Gibbs energy of solution () continues to decrease, demonstrating that the solution process is driven by the standard entropy of solution () [25,27], indicating further that in mixtures rich in PEG 400 the cosolvent presents various conformational probabilities given the flexibility of the polymer chains of PEG 400 that favor the solution process of the isoniazid.
4. Conclusions
The dissolution process of isoniazid is endothermic, entropically favored in all cosolvent mixtures. Although the activity coefficients are greater than one, they are very low in all cases, exhibiting an overall quasi-ideal behavior. The enthalpy–entropy compensation analysis shows nonlinear behavior with an inflection point at the cosolvent mixture 0.6 in the mass fraction of PEG 400, confirming the existence of two distinct solvation mechanisms. In water-rich mixtures, the process is enthalpy-driven, possibly due to hydrogen bond breaking, while in cosolvent-rich mixtures, the process is entropy-driven, possibly due to the flexibility and conformation of the polymer chains and the increase in possible microstate configurations. In conclusion, the greater affinity of isoniazid for PEG 400 is not purely energetic, but predominantly entropic due to the structuring of the system.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/liquids6010005/s1, Table S1: Source and purities of the compounds used in this research.
Author Contributions
Conceptualization, D.I.C.-R. and D.R.D.; methodology, D.I.C.-R. and C.P.O.; software, R.E.C.-T.; validation, F.M. and D.R.D.; formal analysis, D.I.C.-R.; investigation, D.I.C.-R. and C.P.O.; resources, F.M. and D.R.D.; data curation, D.R.D.; writing—original draft preparation, D.I.C.-R.; writing—review and editing, D.I.C.-R., D.R.D. and F.M.; visualization, D.R.D.; supervision, F.M.; project administration, D.R.D.; funding acquisition, D.R.D. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Universidad Cooperativa de Colombia grant number INV2976.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data are contained within the article.
Acknowledgments
We thank the National Directorate of Research and National Committee for Research Development of the Universidad Cooperativa de Colombia, for the financial support of the project “Análisis matemático y termodinámico de la solubilidad algunas sustancias antimicrobianas de uso industrial en mezclas cosolventes” with code INV2976. We also thank the Universidad Cooperativa de Colombia, Sede Neiva, for facilitating the use of their laboratories and equipment.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Fowles, D.J.; Connaughton, B.J.; Carter, J.W.; Mitchell, J.B.O.; Palmer, D.S. Physics-Based Solubility Prediction for Organic Molecules. Chem. Rev. 2025, 125, 7057–7098. [Google Scholar] [CrossRef] [PubMed]
- Hildebrand, J.H.; Prausnitz, J.M.; Scott, R.L. Regular and Related Solutions: The Solubility of Gases, Liquids, and Solids; Wiley: Hoboken, NJ, USA, 1970. [Google Scholar]
- Opperhuizen, A.; Gobas, F.A.P.C.; Van der Steen, J.M.D.; Hutzinger, O. Aqueous solubility of polychlorinated biphenyls related to molecular structure. Environ. Sci. Technol. 1988, 22, 638–646. [Google Scholar] [CrossRef] [PubMed]
- Neau, S.H.; Flynn, G.L. Solid and Liquid Heat Capacities of n-Alkyl Para-aminobenzoates Near the Melting Point. Pharm. Res. 1990, 7, 157–1162. [Google Scholar] [CrossRef] [PubMed]
- Erwin, E.R.; Addison, A.P.; John, S.F.; Olaleye, O.A.; Rosell, R.C. Pharmacokinetics of isoniazid: The good, the bad, and the alternatives. Tuberculosis 2019, 116, S66–S70. [Google Scholar] [CrossRef]
- Nahid, P.; Dorman, S.E.; Alipanah, N.; Barry, P.M.; Brozek, J.L.; Cattamanchi, A.; Chaisson, L.H.; Chaisson, R.E.; Daley, C.L.; Grzemska, M.; et al. Official American Thoracic Society/Centers for Disease Control and Prevention/Infectious Diseases Society of America Clinical Practice Guidelines: Treatment of Drug-Susceptible Tuberculosis. Clin. Infect. Dis. 2016, 63, e147–e195. [Google Scholar] [CrossRef]
- Baracaldo-Santamaría, D.; Calderon-Ospina, C.A.; Ortiz, C.P.; Cardenas-Torres, R.E.; Martinez, F.; Delgado, D.R. Thermodynamic Analysis of the Solubility of Isoniazid in (PEG 200 + Water) Cosolvent Mixtures from 278.15 K to 318.15 K. Int. J. Mol. Sci. 2022, 23, 190. [Google Scholar] [CrossRef] [PubMed]
- Strickley, R.G. Solubilizing excipients in oral and injectable formulations. Pharm. Res. 2004, 21, 201–230. [Google Scholar] [CrossRef]
- Mohammadian, E.; Barzegar-Jalali, M.; Rahimpour, E. Solubility prediction of lamotrigine in cosolvency systems using Abraham and Hansen solvation parameters. J. Mol. Liq. 2019, 276, 675–679. [Google Scholar] [CrossRef]
- Rubino, J.; Yalkowsky, S. Cosolvency and Cosolvent Polarity. Pharm. Res. 1987, 4, 220–230. [Google Scholar] [CrossRef]
- Johnson, D.M.; Taylor, W.F. Degradation of fenprostalene in polyethylene glycol 400 solution. J. Pharm. Sci. 1984, 73, 1414–1417. [Google Scholar] [CrossRef]
- Hasaninejad, A.; Beyrati, M. Eco-friendly polyethylene glycol (PEG-400): A green reaction medium for one-pot, four-component synthesis of novel asymmetrical bis-spirooxindole derivatives at room temperature. RSC Adv. 2018, 8, 1934–1939. [Google Scholar] [CrossRef]
- D’souza, A.A.; Shegokar, R. Polyethylene glycol (PEG): A versatile polymer for pharmaceutical applications. Expert Opin. Drug Deliv. 2016, 13, 1257–1275. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, C.P.; Cardenas-Torres, R.E.; Martínez, F.; Delgado, D.R. Solubility of Sulfamethazine in the Binary Mixture of Acetonitrile + Methanol from 278.15 to 318.15 K: Measurement, Dissolution Thermodynamics, Preferential Solvation, and Correlation. Molecules 2021, 26, 7588. [Google Scholar] [CrossRef]
- Dittert, L.W.; Higuchi, T.; Reese, D.R. Phase Solubility Technique in Studying the Formation of Complex Salts of Triamterene. J. Pharm. Sci. 1964, 53, 1325–1328. [Google Scholar] [CrossRef]
- Barton, A.F.M. Handbook of Solubility Parameters and Other Cohesion Parameters, 2nd ed.; CRC Press: Boca Raton, FL, USA, 1991. [Google Scholar]
- Shakeel, F.; Haq, N.; Alsarra, I.A.; Alshehri, S. Solubility, Hansen Solubility Parameters and Thermodynamic Behavior of Emtricitabine in Various (Polyethylene Glycol-400 + Water) Mixtures: Computational Modeling and Thermodynamics. Molecules 2020, 25, 1559. [Google Scholar] [CrossRef] [PubMed]
- Heryanto, R.; Hasan, M.; Abdullah, E.C. Solubility of Isoniazid in Various Organic Solvents from (301 to 313) K. J. Chem. Eng. Data 2008, 53, 1962–1964. [Google Scholar] [CrossRef]
- Yalkowsky, S.H.; Wu, M. Estimation of the ideal solubility (crystal-liquid fugacity ratio) of organic compounds. J. Pharm. Sci. 2010, 99, 1100–1106. [Google Scholar] [CrossRef] [PubMed]
- Neau, S.H.; Bhandarkar, S.V.; Hellmuth, E.W. Differential Molar Heat Capacities to Test Ideal Solubility Estimations. Pharm. Res. 1997, 14, 601–605. [Google Scholar] [CrossRef]
- Yalkowsky, S.H. Solubility and Solubilization in Aqueous Media; American Chemical Society: Washington, DC, USA, 1999. [Google Scholar]
- Krug, R.R.; Hunter, W.G.; Grieger, R.A. Enthalpy-entropy compensation. 1. Some fundamental statistical problems associated with the analysis of van’t Hoff and Arrhenius data. J. Phys. Chem. 1976, 80, 2335–2341. [Google Scholar] [CrossRef]
- Krug, R.R.; Hunter, W.G.; Grieger, R.A. Enthalpy-entropy compensation. 2. Separation of the chemical from the statistical effect. J. Phys. Chem. 1976, 80, 2341–2351. [Google Scholar] [CrossRef]
- Escobar Fiesco, G.F.; Caviedes-Rubio, D.I.; Ortiz, C.P.; Quintero Guerrero, Y.; Cerquera, N.E.; Rincón-Guio, C.; Cardenas-Torres, R.E.; Delgado, D.R. Thermodynamic analysis and preferential solvation of metronidazole solubility in methanol-water and ethanol-water cosolvent mixtures at different temperatures. Rev. Colomb. Cienc. Quim.-Farm. 2025, 54, 345–363. [Google Scholar]
- Bustamante, P.; Romero, S.; Peña, A.; Escalera, B.; Reillo, A. Enthalpy–entropy compensation for the solubility of drugs in solvent mixtures: Paracetamol, acetanilide, and nalidixic acid in dioxane–water. J. Pharm. Sci. 1998, 87, 1590–1596. [Google Scholar] [CrossRef] [PubMed]
- Di, L.; Fish, P.V.; Mano, T. Bridging solubility between drug discovery and development. Drug Discov. Today 2012, 17, 486–495. [Google Scholar] [CrossRef]
- Peña, M.; Escalera, B.; Reíllo, A.; Sánchez, A.; Bustamante, P. Thermodynamics of Cosolvent Action: Phenacetin, Salicylic Acid and Probenecid. J. Pharm. Sci. 2009, 98, 1129–1135. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.