Abstract
In August 2023, a large forest fire burned more than 60% of the Dadia–Lefkimi–Soufli Forest National Park in northeastern Greece, following another large fire in 2022. To quantify the effects of these fires on local herpetofauna, we analyzed community composition, abundance, and diversity before and after the 2023 event. Standardized visual encounter surveys were conducted across 29 sites between 2015 and 2024, spanning burned and unburned areas. Species richness, abundance, and diversity metrics, together with Bray–Curtis community dissimilarities, were compared across sampling periods and fire-severity classes. Amphibian assemblages showed high postfire persistence, with 82% of regional species still detected and no significant changes in diversity indices, likely reflecting the buffering role of perennial streams and other hydrologically stable refugia. In contrast, reptile communities showed clear compositional shifts and experienced severe declines: overall reptile species richness decreased to 30% of prefire levels and diversity indices dropped significantly. Tortoises (i.e., Testudo graeca, T. hermanni) declined by nearly 90% relative to prefire estimates, indicating high vulnerability of low-mobility, long-lived species. Snakes were not detected in any burned sites, whereas only a few small-bodied lizards and the freshwater turtle Mauremys rivulata persisted locally. These findings demonstrate that extreme, landscape-scale fires can restructure reptile communities in Mediterranean forests, particularly where long-term habitat change and drought had already reduced population resilience. The study underscores the need for targeted postfire restoration, conservation planning for slow-dispersing taxa, and long-term biodiversity monitoring under increasingly frequent fire regimes.
1. Introduction
Fire is a major ecological process shaping the structure and functioning of ecosystems worldwide, influencing community composition, vegetation dynamics, nutrient cycling, and long-term landscape patterns [1,2,3]. In fire-prone regions such as the Mediterranean Basin, fire regimes, defined by their frequency, intensity, severity, seasonality, and spatial extent, create shifting habitat mosaics and landscape patches that may temporarily increase environmental heterogeneity [4], but can also fragment landscapes, simplify vegetation structure and reduce resource availability when fires are severe or recurrent [5,6]. Over recent decades, fire regimes in Mediterranean landscapes have changed markedly [6,7,8], driven by climate change, rural land abandonment, fuel accumulation, and fires suppression practices. These forces have contributed to larger, more frequent, and more intense fires, including megafires [7,9,10,11] now recognized as emerging threats to biodiversity worldwide. However, animal responses to these novel fire regimes remain highly variable and context dependent [12,13,14]. Understanding how wildlife communities reorganize after fire, and which ecological traits mediate persistence, is essential for predicting biodiversity trajectories and guiding effective conservation in fire-prone ecosystems [2].
Among vertebrates, reptiles and amphibians may be particularly sensitive to altered fire regimes due to their ectothermy, limited dispersal ability, and dependence on specific microhabitats for thermoregulation, shelter, and reproduction [13,15,16]. Fire can affect these taxa through multiple pathways: direct mortality during burns, loss of refuges such as leaf litter, coarse woody debris, and rock crevices, shifts in canopy structure and understory vegetation that modify thermal and hydric microclimates, and changes in prey availability or predation risk [17,18,19]. Yet empirical findings are mixed. Some reptiles, particularly small heliophilic lizards, may increase in abundance following low-severity burns that open habitat structure [16], whereas severe or recurrent fires often depress populations of habitat specialists, long-lived reptiles, and amphibians in landscapes with limited hydrological refugia [20,21]. These contrasting outcomes highlight strong trait- and context-dependent responses [13,22]. Despite growing interest in fire impacts on herpetofauna [23,24,25,26], community-level studies remain scarce in Mediterranean forest ecosystems, especially under extreme fire events [13]. This is especially true for amphibians, whose responses to fire remain comparatively poorly studied and often underrepresented in fire-ecology research [27,28,29]. In contrast, low-mobility reptiles such as tortoises have received more attention and are consistently identified as highly vulnerable to high-severity or frequent fires [30,31]. Such knowledge gaps underscore the need for integrated assessments of entire reptile and amphibian assemblages under contemporary fire regimes.
The Dadia–Lefkimi–Soufli Forest National Park (hereafter Dadia Forest) in northeastern Greece (Figure 1) is one of Europe’s most important biodiversity refuges and a core component of the NATURA 2000 network. Its heterogeneous landscape, comprising oak (Quercus spp.), pine (Pinus nigra, P. brutia) and mixed forests, shrublands and riparian ecosystems, supports a rich herpetofauna, including 11 amphibian and more than 25 reptile species [32,33,34,35,36]. Many of these taxa are of high conservation concern, being listed under Annexes II and IV of the European Union (EU) Habitats Directive (92/43/European Economic Community), protected by the Bern Convention and national legislation, and included in international agreements such as Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES). In the summers of 2022 and 2023, Dadia Forest experienced two consecutive large fires, with the 2023 event burning more than 60% of the Park and along with the fire flame front in South Evros region represent the largest fire ever recorded in the European Union [37]. These large fires resulted in extensive habitat loss, structural simplification, and widespread faunal mortality, raising urgent conservation concerns for low-mobility and habitat-specialist species. Although vegetation showed signs of early natural regeneration, full ecosystem recovery will likely require decades.
Figure 1.
Location of the Dadia–Lefkimi–Soufli Forest National Park in northeastern Greece. The main panel shows the regional context and the boundaries of the National Park within Greece. The inset map provides a global context highlighting the location of Greece within the Mediterranean Basin.
The ecological significance of Dadia Forest and the magnitude of these fires provided a unique opportunity to evaluate the postfire responses of its herpetofaunal communities. In this study, we assessed the effects of 2023 fire on reptile and amphibian assemblages in Dadia Forest. We analyzed pre- and postfire field data collected across burned and unburned sites and compared species richness, abundance, and diversity indices across fire-severity classes. Surveys overlapped previous sampling locations to ensure temporal comparability, and unburned sites served as reference conditions. Special focus was given to taxa of high conservation concern, including tortoises and amphibians with limited dispersal ability. By quantifying short-term community responses to consecutive large fires in a Mediterranean protected area, this study provides critical baseline information for assessing herpetofaunal vulnerability to extreme fire events and informing postfire restoration and management strategies in fire-affected landscapes.
2. Materials and Methods
2.1. Sampling Area
Fieldwork was carried out across an extensive area of the Dadia–Lefkimi–Soufli Forest National Park (Figure 1) during 2022 and 2023, prior to the August 2023 fire. Sampling stations were selected based on habitat suitability for local reptile and amphibian communities, and in total 29 locations (Supplementary Material Table S1 and Figure 2) representing a variety of habitat types were surveyed. Particular emphasis was placed on streams, springs, ponds, and other water bodies, which serve as key refuges for many amphibians and reptile species associated with humid environments. The study area included coniferous, deciduous, and mixed forests, as well as forest openings, rocky substrates, and shrublands.
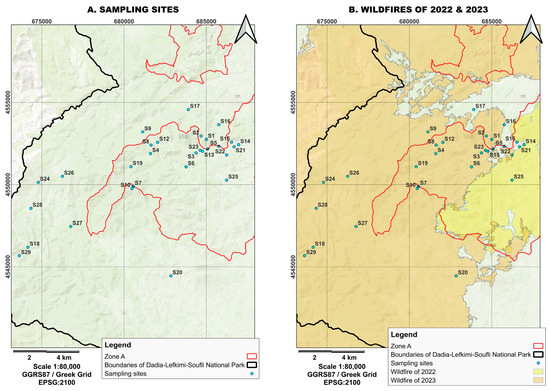
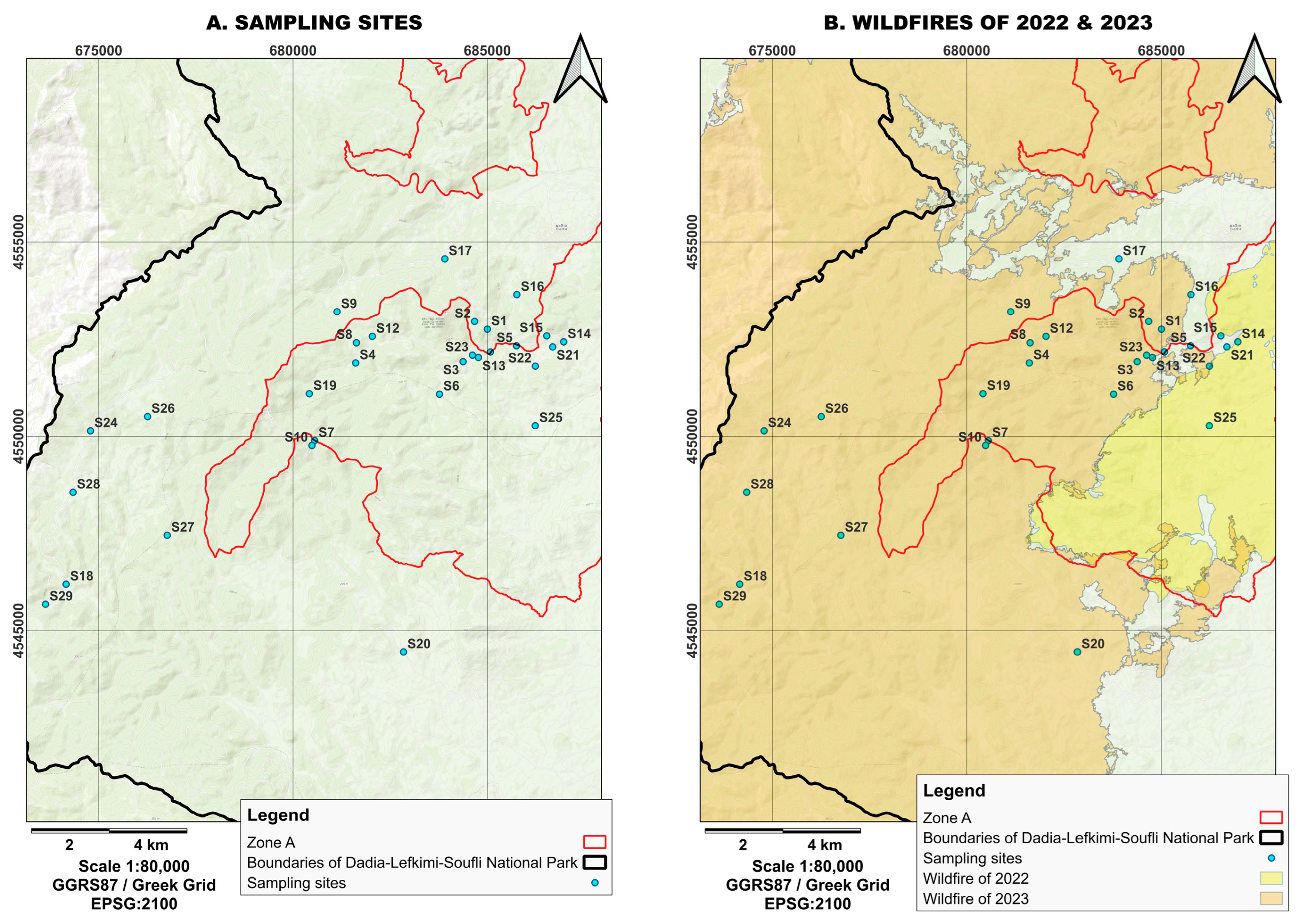
Figure 2.
(A) Sampling sites within the Dadia–Lefkimi–Soufli Forest National Park. The red line indicates the boundaries of Zone A. (B) Areas affected by the 2022 fire (yellow) and the 2023 fire (orange). (Source: WWF Greece, NECCA).
To collect postfire data, four seasonal field surveys were conducted from November 2023 to November 2024, each lasting four to five days. Survey timing was designed to account for seasonal variation in activity and detectability. In Mediterranean systems, amphibians are primarily active and detectable during spring breeding periods and around aquatic habitats, whereas reptiles show peak surface activity from spring to summer, with reduced detectability outside these periods [32]. Earlier access to the burned area was not permitted due to safety restrictions and persistent underground heat following the fire. Prefire datasets consisted of records collected at the same sites in April 2015 (provided by the Natural Environment and Climate Change Agency; NECCA), spring 2022, and July 2023; one month before the fire event (Supplementary Material Table S1). This sampling design enabled direct temporal comparisons of reptile and amphibian abundance, distribution, and community composition.
In addition, nine supplementary sampling locations situated outside or near the 2023 fire perimeter were surveyed to provide spatial reference conditions where fire impacts were absent or minor (Supplementary Materials Table S1). These unburned areas served as temporal and spatial controls, allowing us to distinguish fire-driven changes from natural interannual variability (Figure 3). After the fire, surveys were repeated both in previously sampled stations and in new locations within the burned area to document postfire assemblage responses. For evening- or nocturnally active species, such as Rana dalmatina, Salamandra salamandra, Bufotes viridis, Bufo bufo, and Vipera ammodytes, night surveys and targeted searches near water bodies were also conducted.
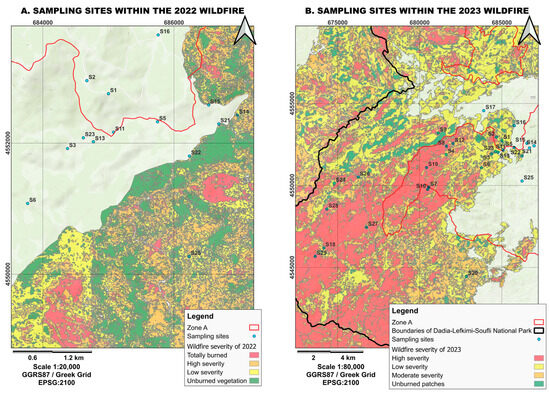
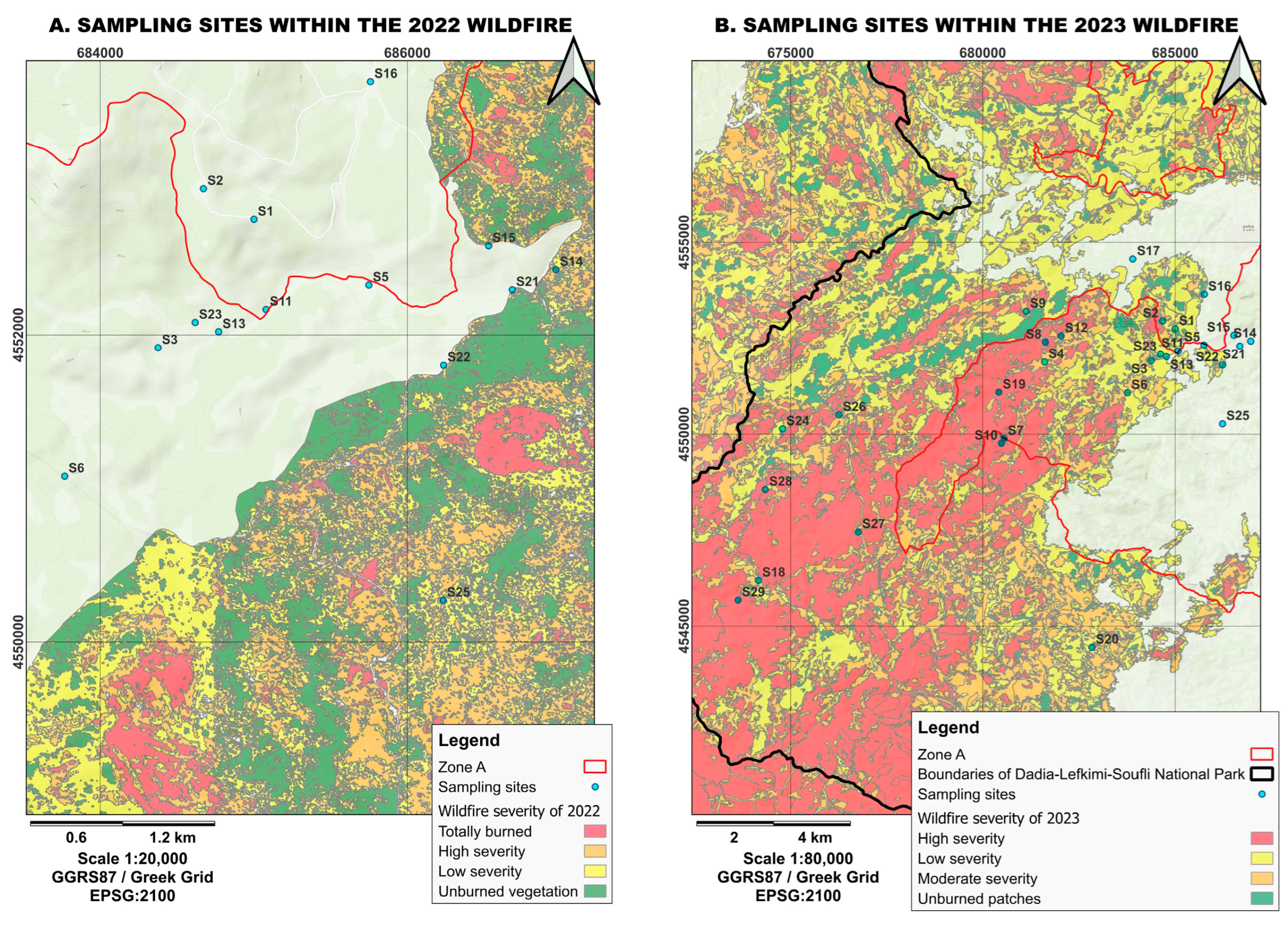
Figure 3.
Visualization of fire severity for (A) the 2022 and (B) 2023 fires and the locations of the sampling stations within the Dadia–Lefkimi–Soufli Forest National Park. (Source: WWF Hellas, N.E.C.C.A.).
2.2. Sampling Methodology
Amphibian and reptile populations were surveyed using visual encounter surveys along random line transects [38,39]. This method is effective across a variety of habitat types, has minimal impact on site conditions, and poses no risk to the welfare of individual animals. Each transect was 200 m in length, allowing coverage of multiple microhabitats within each sampling site. Observers recorded all individuals detected within 2.5 m on either side of the transect line, resulting in a total surveyed area of 1000 m2 per transect. During surveys, good- and high-quality microhabitats up to 5 m from either side of the line were also searched. The number of transects per site varied according to habitat suitability, microhabitat complexity, and geomorphology, resulting in an average survey effort of 2.8 ± 0.8 transects per location. In addition, random quadrat sampling points were established along transects to increase detection probability. Time-constrained searches (~30 min per observer) were conducted at each site to detect species likely to be overlooked during transect walks, particularly cryptic reptiles. For amphibians, targeted searches were conducted at sampling stations near aquatic habitats. Population density was estimated using the formula:
where N is the total number of observed individuals across all transects and A is the total surveyed area (in m2).
Density per hectare = (N × 10,000)/A,
Visual encounter surveys along line transects were used as a standardized and widely applied method for surveying reptile and amphibian assemblages across heterogeneous habitats. Although this approach does not explicitly account for imperfect detection neither allow estimation of absolute population size, it is well suited for comparative assessments of community-level patterns [38,39]. In this study, we focused on evaluating relative changes in species richness, diversity, and community composition across pre- and postfire periods and between burned and unburned sites. More robust occupancy- or detection-based models [40,41] were not applied because the historical prefire datasets were not structured to provide the repeated, temporally independent surveys required for reliable estimation of detection probability.
2.3. Diversity Indices
For each sampling station, we calculated two widely used ecological diversity metrics: the Shannon-Wiener index (H′) [42,43] and the Simpson dominance index (Simpson’s 1–D) [44]. These metrics were chosen because they capture different ecological aspects of the communities. The Simpson index incorporates species richness and dominance and was calculated as:
where represents the relative abundance of species i. Values close to 0 indicate high diversity, whereas values approaching 1 indicate dominance by few taxa. Shannon diversity, more sensitive to rare species and richness patterns, was calculated as follows. Higher H′ values represent greater community diversity [45]:
2.4. Data Analyses
To evaluate changes in herpetofaunal abundance, diversity, and community structure before and after the 2023 fire, we analyzed species-level abundance data from all sampling sites and years. Shannon (H′) and Simpson (1–D) indices were calculated for each site by year combination. Because preliminary normality assessments indicated non-normal distributions, differences between prefire (2015, 2022, July 2023) and postfire (late 2023–2024) conditions were tested using non-parametric Kruskal–Wallis tests, followed by pairwise Wilcoxon rank-sum comparisons with Benjamini–Hochberg correction when appropriate. To examine how fire affected community composition, we applied Analysis of Similarities (ANOSIM) using the vegan v.2.8-0 package in R 4.3 [46]. Species abundance data were square-root transformed, and Bray–Curtis dissimilarities were calculated among all samples. Treatment (prefire vs. postfire) served as the grouping variable, and significance was assessed using 999 permutations. ANOSIM was conducted for the entire herpetofaunal assemblage and separately for reptiles and amphibians. To account for potential temporal variation unrelated to fire, we additionally fitted generalized linear mixed models (GLMMs) with Treatment (prefire vs. postfire) as a fixed effect and Year as a random effect. Sites with zero detections were retained and assigned diversity values of zero to avoid inflating postfire diversity estimates. All statistical analyses were conducted in R 4.3 [47].
3. Results
Across the prefire surveys conducted in April 2015, spring 2022, and July 2023, 10 of the 11 amphibian species (91%) known from Dadia Forest and 19 of the 27 reptiles (70%) were recorded (Table 1). Specifically, prefire surveys detected 8 amphibian and 15 reptile species in 2015, 8 amphibian and 17 reptile species in 2022, and 10 amphibian and 9 reptile species in July 2023. On the other hand, the postfire sampling conducted between late 2023 and 2024 revealed a significant decline in reptile richness, with only 8 reptiles (30% of the regional fauna) and 9 amphibian (82%) species detected. This shift in species detections was statistically significant (chi-square test; χ2 = 19.31, p < 0.001). Despite the more intensive postfire sampling effort, comprising four field surveys between September 2023 and November 2024, both reptile abundance and species richness were substantially lower than in any prefire year. These patterns were consistent even when pre- and post- fired datasets pooled together.

Table 1.
Summary table of amphibian and reptile species and the number of individuals recorded at the sampling sites during the pre-fire surveys (2015 and 2022), prior to the 2023 fire. H′ refers to the Shannon diversity index and 1–D to the Simpson diversity index. Gray-shaded cells indicate sites located within the burned area.
Shannon diversity index (H′; F = 21.163, p < 0.001) and the Simpson index (F = 24.598, p < 0.001) showed statistically significant declines following the 2023 fire (Table 2). Shannon values decreased from 1.18 ± 0.51 before the fire to 0.55 ± 0.49 afterwards, while Simpson values declined from 0.59 ± 0.17 to 0.32 ± 0.26 (see Table 1 and Table 2). We found no significant differences between prefire years (i.e., 2015 and 2022) for either Shannon (t = 1.28, df = 11.14, p = 0.228) or Simpson (t = 1.10, df = 10.54, p = 0.297) indicating that overall herpetofauna diversity remained relatively stable prior to the fire. However, the lack of change in composite diversity indices during 2015–2022 masks substantial population declines in certain species, particularly tortoises, whose populations had already decreased markedly prior to the fire.

Table 2.
Summary table of amphibian and reptile species and the number of individuals recorded at the sampling sites of the study area during the post-fire surveys (2023–2024). H′ refers to the Shannon diversity index and 1–D to the Simpson diversity index. Gray-shaded cells indicate sites located within the burned area.
Multivariate analyses further confirmed a shift in community structure associated with the fire. ANOSIM based on Bray–Curtis dissimilarities revealed a statistically significant difference in herpetofaunal community composition between prefire and postfire samples (R = 0.132, p = 0.001). Although the effect size was modest, suggesting partial overlaps between pre- and postfire assemblages, between-group dissimilarities (median rank; 1443.5) were consistently higher than within-group dissimilarities. In addition, postfire communities (median rank; 1391.0) exhibited greater internal variability than prefire communities (median rank; 1128.3), suggesting increased spatial heterogeneity in species composition across burned areas. Together, these findings indicate that while overall postfire diversity was sharply declined, the fire also generated shifts in community structure, with different taxonomic groups responding in distinct ways.
3.1. Effects of Fire on Reptilian Populations
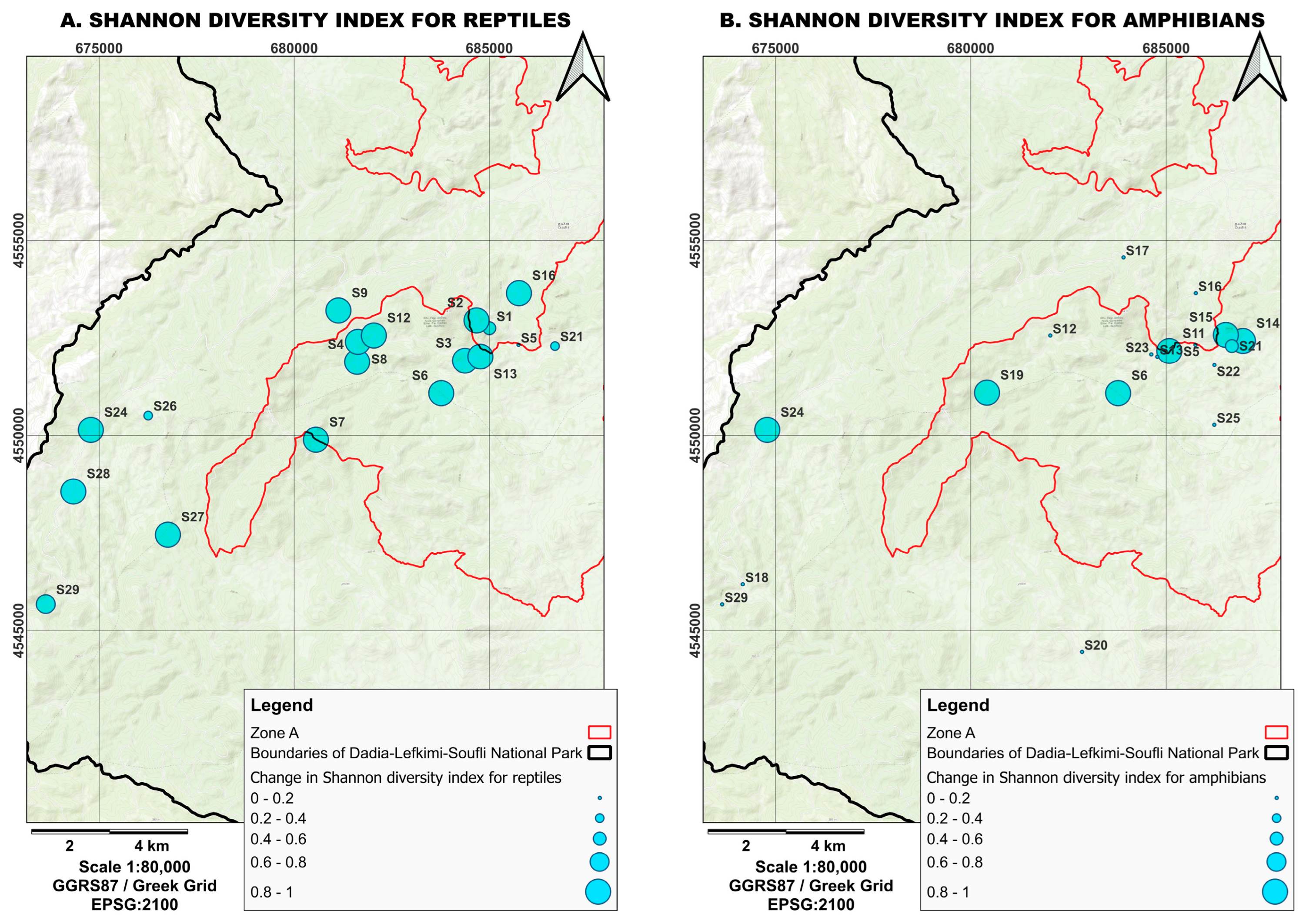
We found substantial declines in reptiles’ abundance and diversity following the 2023 fire. Shannon diversity decreased from 1.08 ± 0.54 before the fire to 0.30 ± 0.38 afterwards (Figure 4A), while Simpson diversity declined from 0.55 ± 0.24 to 0.19 ± 0.23 (Table 1 and Table 2). Both indices showed statistically significant reductions (Kruskal–Wallis test; Shannon: U = 11.64, p < 0.001; Simpson: U = 12.16, p < 0.001). These differences were driven exclusively by sites located within burned areas, where diversity decreased sharply between prefire and postfire surveys (Shannon: U = 10.07, p = 0.001; Simpson: U = 10.07, p = 0.001). In contrast, unburned reference sites showed no significant changes in either index (both p > 0.10). Reptile species richness also declined markedly after the fire, with only eight species detected in the postfire period compared to nineteen species documented across prefire surveys. This reduction was reflected in ANOSIM where reptile community composition differed significantly between pre- and postfire assemblages (ANOSIM; R = 0.294, p < 0.001), indicating a clear shift in reptile assemblages associated with the 2023 fire. Postfire reptile communities displayed increased internal dissimilarity compared to prefire communities, suggesting greater spatial variability in species occurrence across burned sites. The magnitude of the observed decline is consistent with the seasonal life-history and activity patterns of reptiles, which are most surface-active during spring and summer for mating and reproduction and retreat to underground refuges later in the year [32]; notably, the August 2023 fire coincided with this period of peak activity.
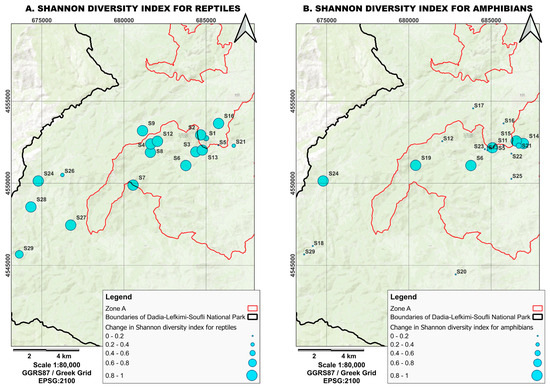
Figure 4.
Shifts in Shannon diversity indices values for (A) reptiles and (B) amphibian populations before and after the 2023 fire, based on the data presented in Table 1 and Table 2. The size of the circle size reflects the magnitude of the change. A transition from larger to smaller circles indicates a greater reduction in diversity.
Declines in population abundance were most pronounced for low-mobility, long-lived reptiles. Tortoise populations experienced severe reductions: observations of Testudo graeca declined from 23 individuals in spring 2022 to 11 individuals in July 2023 (prefire), and to only 2 individuals during postfire surveys. Testudo hermanni showed a similar pattern, with counts decreasing from 17 (2022) and 7 (July 2023) to only 1 individual after the fire. Generalized linear models confirmed substantial declines in tortoise counts (GLM: χ2 = 9.14, p = 0.002). Estimated population densities fell from 13.8 individuals/ha in 2015 and 6.9 individuals/ha in 2023 to only 1.03 individuals/ha after the fire, representing an overall reduction of 92.5% since 2015 and 85.1% since 2022. Notably, an approximately 50% decline had already occurred between 2015 and 2023, prior to the fire, indicating ongoing population deterioration. Finally, no snakes were recorded inside burned areas during any of the four postfire surveys (from November 2023 to November 2024), despite extensive sampling effort.
3.2. Effects of Fire on Amphibian Populations
Amphibians showed comparatively limited responses to fire. Diversity indices did not differ significantly between prefire and postfire periods. Shannon diversity remained largely unchanged (prefire: 0.72 ± 0.53; postfire: 0.52 ± 0.45; U = 1.07, p = 0.301), as did Simpson diversity (prefire: 0.39 ± 0.27; postfire: 0.31 ± 0.26; U = 0.99, p = 0.318) (Figure 4B). All amphibian species detected before the fire were also detected afterward, and species richness did not differ significantly between years (χ2, p = 0.37). Further analyses evaluating burned versus unburned sites showed no significant changes in amphibian diversity within either category (all four ps > 0.05). ANOSIM results for amphibians likewise indicated no significant difference in community composition between prefire and postfire samples (ANOSIM; R = 0.067, p = 0.932). Amphibian assemblages thus remained compositionally stable across sampling periods.
This stability is consistent with the broadly shared life-history and activity patterns of amphibians in the study area, which reproduce in aquatic habitats during spring, with eggs and larvae developing in water, while adults remain closely associated with perennial or semi-permanent water bodies or reduce activity through aestivation during hot summer periods [32]; consequently, the August 2023 fire coincided with a phase when many individuals were either aquatic or sheltered in moist refugia.
4. Discussion
Our study examined how the fire in Dadia-Lefkimi-Soufli Forest National Park—one of the most extensive recent fires in southeastern Europe—affected amphibian and reptile assemblages. We hypothesized that a fire of such magnitude would impose pronounced negative effects across herpetofaunal diversity. Instead, our findings revealed a highly asymmetric response: reptiles underwent substantial declines in abundance, richness, and diversity, whereas amphibian assemblages remained largely unaffected and stable across the landscape. These divergent outcomes emphasize that fire magnitude and extent do not uniformly predict biological impacts ([48], but see [49]); rather, the ecological traits, behavioral patterns, and microhabitat associations of different taxa fundamentally shape their vulnerability or resilience [13,16,28,50].
Despite the extreme, landscape-scale extent of the 2023 fire [37], 82% of the regional amphibian fauna were still detected in the postfire surveys. As such, diversity metrics (Shannon, Simpson) and community-level analyses (ANOSIM) showed no significant departures from prefire assemblages. This relative stability aligns with recent global syntheses indicating that amphibian responses to fire are highly variable and strongly dependent on local environmental conditions [13,16,28,51]. In many systems, the postfire persistence of amphibian populations is facilitated by hydrologically buffered microrefugia, such as streams, ponds, and/or shaded riparian corridors, which can drastically reduce direct mortality, providing shelter during and after fire events. Renken [16] for instance, reported that moist microhabitats and the burrowing behavior of many amphibians can limit fire-related mortality in temperate forest ecosystems. A similar mechanism likely occurred in Dadia Forest. The combination of estivating underground during the hottest summer months and the continued flow of water in many stream sections within the burning sites, likely provided amphibians with cool, humid refuges during and after the fire, facilitating their high postfire detectability and contributed to the apparent resilience of the amphibian assemblage [52]. However, it is important to note that our assessment represents only the first year after the fire, capturing the initial, short-term phase of ecological response [28]. A growing body of literature cautions that early postfire surveys may not reflect longer-term outcomes, as delayed population declines, gradual shifts in community composition, and habitat-driven changes often emerge only after several years [28,53,54]. Studies reporting little or no immediate fire effects—particularly for amphibians—frequently attribute such patterns to the short temporal window of sampling, which can underestimate or entirely miss longer-term ecological trajectories [53,54,55]. Thus, while our findings describe the immediate postfire stage in Dadia Forest, continued monitoring will be critical to determine whether these patterns persist, intensify, or reverse over longer time scales.
Contrary to amphibians, reptile communities exhibited pronounced postfire declines across the burned sites of Dadia Forest. Only 30% of the region’s reptile fauna was detected after the fire, and both abundance and diversity indices decreased sharply relative to prefire years. These findings come in contrast to numerous studies reporting neutral or even positive reptile responses to fire [16,25,51,56,57], particularly in small, heliophilic lizard species that often increase in abundance following low-severity prescribed burns. Such burns typically create fine-scale habitat mosaics, preserve pockets of woody debris, maintain refugia, and increase ground-level temperatures, thereby enhancing basking and foraging opportunities for disturbance-tolerant reptiles [58]. The discrepancy between these outcomes and the severe declines observed in Dadia likely reflects fundamental differences in fire severity, spatial extent, and landscape context. The 2023 Dadia fire burned for nearly fourteen consecutive days, producing a landscape-scale event that consumed large tracts of forest with minimal unburned refuges [59]. Dadia fire was characterized by high-severity crowning which removed canopy cover, eliminated shrub and herb layers, and fully consumed the litter and coarse woody debris that many reptiles rely on for thermoregulation, predator avoidance, and shelter [17]. Under such conditions, even relatively mobile species face limited opportunities to escape advancing flames or recolonize burned areas.
But even within reptiles, these extreme fire characteristics affected different taxa disproportionally. Tortoises, i.e., Testudo graeca and T. hermanni, slow-moving, long-lived species with restricted dispersal abilities, experienced the most severe declines [25], with densities reduced to roughly one-tenth of prefire levels (Table 1 and Table 2). Although high fire-induced mortality in Mediterranean chelonians is well documented [30,31,60,61] our results show that the 2023 fire acted on populations that were already declining. A substantial decline of approximately 50% had already occurred between 2015 and July 2023, suggesting substantial demographic erosion predating the fire. We assume that long-term processes such as land abandonment, reduced grazing pressure, and resulting vegetation encroachment have progressively reduced habitat openness, creating cooler and more shaded microclimates that hinder thermoregulation, movement, and nesting [62]. These habitat changes, combined with multi-year drought and reduced precipitation between 2016 and 2022 (Standardized Precipitation Index; Hellenic National Meteorological Service), likely constrained foraging and reproductive output [62]. Τhese findings suggest that the 2023 fire did not initiate the tortoise collapse but accelerated and magnified ongoing negative demographic trends.
A similar pattern was observed for snakes, which were not detected at any burned site during the postfire surveys. Although this absence might suggest severe impacts, it should be interpreted with caution: snakes in Mediterranean forests typically occur at low densities, are highly cryptic, and rely heavily on shelter availability; characteristics that reduce detectability even when populations persist [32,63]. Thus, failure to record snakes does not necessarily indicate complete local extirpation. Nonetheless, their absence in postfire surveys aligns with the expectation that species with low mobility and strong dependence on structurally complex microhabitats are particularly vulnerable to high-severity, landscape-scale fires. Despite the latter, a few small-bodied lizard species (e.g., Lacerta trilineata, L. viridis, Podarcis muralis; Table 2) persisted in parts of the burned landscape. Their agility, small body size, and frequent use of microhabitats such as root systems, rock crevices, holes in the soil, and shrub bases may have offered partial protection during the fire, allowing some individuals to survive [17,64]. However, their persistence was limited in scope and did not offset the broader collapse observed across the reptile community. Another taxon that survived the fire was the freshwater turtle Mauremys rivulata, which remained present in several postfire sites but exclusively in close association with perennial streams. This pattern further supports our inference on amphibian results, where running water and riparian microhabitats functioned as critical fire refugia. The species’ strong aquatic dependence likely buffered it from direct fire mortality, while the thermal and hydric stability of streams provided shelter during the most intense phases of the burn. The survival of M. rivulata therefore reinforces the broader conclusion that hydrologically buffered habitats played a central role in structuring postfire vertebrate persistence across Dadia.
Overall, our findings highlight that the effects of large fires on herpetofauna are strongly taxa-specific and shaped by a combination of life-history traits, habitat associations, and the availability of refugia [22,65,66,67]. Reptiles, particularly low-mobility, habitat-specialist species, proved far more vulnerable to the extreme conditions of the 2023 Dadia fire than amphibians, whose persistence was supported by hydrologically buffered microhabitats. These patterns mirror emerging evidence from across the Mediterranean Basin showing that increasingly large, severe, and recurrent fires disproportionately impact ecological specialists and species reliant on structurally complex forest habitats, while favoring only a subset of disturbance-tolerant taxa [1,25]. Moreover, the Dadia Forest hosts a high diversity and density of raptor species [68,69], and the extensive postfire canopy openings likely increased prey detectability, potentially intensifying avian predation on reptiles. Such fire-induced structural simplification may therefore have indirectly contributed to reptile declines by facilitating more efficient foraging by avian predators. Although our conclusions are based on standardized visual encounter surveys that do not explicitly account for imperfect detection, our analyses focus on relative comparisons of species richness, diversity, and community composition across pre- and postfire periods and burned versus unburned sites. Occupancy-based approaches that incorporate detection probability [40,41] offer a powerful framework for inferring species occurrence and habitat use but require repeated, temporally independent surveys, which were not consistently available across the historical prefire datasets used here.
As climate warming, prolonged drought, and vegetation encroachment continue to intensify regional fire regimes, such large fire events are expected to become more frequent [70,71,72], further constraining recovery opportunities for slow-dispersing species such as Testudo spp. and many snakes. Future research and management in Dadia Forest and similar Mediterranean systems should integrate long-term, detection-corrected monitoring with assessments of fire severity, habitat structure, and climate trends, alongside management strategies that maintain habitat heterogeneity, protect riparian refugia, and prevent further loss of open habitats through targeted grazing or controlled vegetation management.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/conservation6010029/s1, Table S1: Sampling stations where field surveys were conducted before and after the fire of August 2023. The table also indicates whether each site was located inside or outside the burned area, as well as the corresponding fire severity level.
Author Contributions
Conceptualization, K.S. and P.M.; Methodology, K.S., T.D., D.I., N.G., V.Z. and P.M.; Field study, K.S., T.D., D.I., N.G. and V.Z.; Formal analysis, K.S. and V.Z.; Investigation, K.S., T.D., D.I., N.G., V.Z. and P.M.; Resources, P.M.; Data curation, K.S., I.M., V.Z. and P.M.; Writing—Original draft preparation, K.S., T.D., D.I., N.G., V.Z. and P.M.; Writing—Review and editing, K.S., T.D., D.I., N.G., I.M., V.Z. and P.M.; Visualization, K.S. and V.Z.; Supervision, K.S. and P.M.; Project administration, K.S. and P.M.; Funding acquisition, P.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by WWF International, grant number 76308 under the framework “Restoring key landscape values in the Dadia National Park”.
Institutional Review Board Statement
Ethical review and approval were not required for this study because no procedures falling under the scope of Directive 2010/63/EU on the protection of animals used for scientific purposes were performed. The study was conducted in accordance with Greek national legislation(Presidential Decree 67/1981) and under a valid permit, and consisted exclusively of non-invasive observational field surveys of wild animals without capture, handling, or experimental procedures. All necessary research and access permits were obtained from the competent national authorities.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Acknowledgments
We are grateful to Theodora Skartsi (Society for the Protection of Biodiversity of Thrace; Dadia–Lefkimi–Soufli Forest National Park) for her valuable support during the fieldwork, including assistance with access to sampling sites, coordination with local authorities, and facilitation of research permits and logistics. We also thank the Natural Environment and Climate Change Agency (N.E.C.C.A.) for access to pre-fire monitoring data. Fieldwork was conducted under the relevant research permits (ΥΠΕΝ/ΔΔΔ/79427/2022) issued by the competent authorities.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Santos, X.; Cheylan, M. Taxonomic and functional response of a Mediterranean reptile assemblage to a repeated fire regime. Biol. Conserv. 2013, 168, 90–98. [Google Scholar] [CrossRef]
- Pausas, J.G.; Keeley, J.E. Evolutionary fire ecology: An historical account and future directions. Bioscience 2023, 73, 602–608. [Google Scholar] [CrossRef] [PubMed]
- He, T.; Lamont, B.B.; Pausas, J.G. Fire as a key driver of Earth’s biodiversity. Biol. Rev. 2019, 94, 1983–2010. [Google Scholar] [CrossRef] [PubMed]
- Blanco-Rodríguez, M.Á.; Ameztegui, A.; Alday, J.G.; Lecina-Diaz, J.; Pineda-Zapata, S.; Coll, L. Fire severity shapes landscape heterogeneity in Mediterranean forest ecosystems. Landsc. Ecol. 2025, 40, 210. [Google Scholar] [CrossRef]
- Fernandes, P.M. Fire-smart management of forest landscapes in the Mediterranean basin under global change. Landsc. Urban Plan. 2013, 110, 175–182. [Google Scholar] [CrossRef]
- Vaz, P.G. Wildfire Resilience in Mediterranean Landscapes: A Review; Universidade de Lisboa: Lisboa, Portugal, 2009; p. 33. [Google Scholar]
- Brotons, L.; Aquilué, N.; de Cáceres, M.; Fortin, M.-J.; Fall, A. How Fire History, Fire Suppression Practices and Climate Change Affect Wildfire Regimes in Mediterranean Landscapes. PLoS ONE 2013, 8, e62392. [Google Scholar] [CrossRef]
- Mantero, G.; Morresi, D.; Marzano, R.; Motta, R.; Mladenoff, D.J.; Garbarino, M. The influence of land abandonment on forest disturbance regimes: A global review. Landsc. Ecol. 2020, 35, 2723–2744. [Google Scholar] [CrossRef]
- Dupuy, J.-l.; Fargeon, H.; Martin-StPaul, N.; Pimont, F.; Ruffault, J.; Guijarro, M.; Hernando, C.; Madrigal, J.; Fernandes, P. Climate change impact on future wildfire danger and activity in southern Europe: A review. Ann. For. Sci. 2020, 77, 35. [Google Scholar] [CrossRef]
- Salis, M.; Del Giudice, L.; Jahdi, R.; Alcasena-Urdiroz, F.; Scarpa, C.; Pellizzaro, G.; Bacciu, V.; Schirru, M.; Ventura, A.; Casula, M.; et al. Spatial Patterns and Intensity of Land Abandonment Drive Wildfire Hazard and Likelihood in Mediterranean Agropastoral Areas. Land 2022, 11, 1942. [Google Scholar] [CrossRef]
- Hantson, S.; Hamilton, D.S.; Burton, C. Changing fire regimes: Ecosystem impacts in a shifting climate. One Earth 2024, 7, 942–945. [Google Scholar] [CrossRef]
- Kelly, L.T.; Giljohann, K.M.; Duane, A.; Aquilué, N.; Archibald, S.; Batllori, E.; Bennett, A.F.; Buckland, S.T.; Canelles, Q.; Clarke, M.F.; et al. Fire and biodiversity in the Anthropocene. Science 2020, 370, eabb0355. [Google Scholar] [CrossRef] [PubMed]
- González, T.M.; González-Trujillo, J.D.; Muñoz, A.; Armenteras, D. Effects of fire history on animal communities: A systematic review. Ecol. Process. 2022, 11, 11. [Google Scholar] [CrossRef]
- Plumanns-Pouton, E.; Santos, J.L.; Aponte, C.; Brotons, L.; Kelly, L.T.; Mason, S.C., Jr.; Parris, K.; Ponisio, L.; Keith, D.A. The Mechanisms Through Which Fire Drives Population Change in Terrestrial Biota. Glob. Change Biol. 2025, 31, e70479. [Google Scholar] [CrossRef]
- Ferreira, C.C.; Santos, X.; Carretero, M.A. Does ecophysiology mediate reptile responses to fire regimes? Evidence from Iberian lizards. PeerJ 2016, 4, e2107. [Google Scholar] [CrossRef]
- Renken, R.B. Does fire affect amphibians and reptiles in eastern U.S. oak forests? In Fire in Eastern Oak Forests: Delivering Science to Land Managers; Dickinson, M.B., Ed.; Department of Agriculture, Forest Service, Northern Research Station: Newtown Square, PA, USA, 2006; pp. 158–166. [Google Scholar]
- Vitt, L.J.; Caldwell, J.P. Herpetology: An Introductory Biology of Amphibians and Reptiles, 4th ed.; Academic Press: London, UK, 2014. [Google Scholar]
- Russell, K.R.; Van Lear, D.H.; Guynn, D.C. Prescribed fire effects on herpetofauna: Review and management implications. Wildl. Soc. Bull. 1999, 27, 374–384. [Google Scholar]
- Wells, K. The Ecology and Behavior of Amphibians; The University of Chicago Press: Chicago, IL, USA; London, UK, 2007. [Google Scholar]
- Hossack, B.R.; Lowe, W.H.; Corn, P.S. Rapid Increases and Time-Lagged Declines in Amphibian Occupancy after Wildfire. Conserv. Biol. 2013, 27, 219–228. [Google Scholar] [CrossRef]
- Dixon, K.M.; Cary, G.J.; Worboys, G.L.; Gibbons, P. The disproportionate importance of long-unburned forests and woodlands for reptiles. Ecol. Evol. 2018, 8, 10952–10963. [Google Scholar] [CrossRef]
- Pinto, T.; Moreira, B.; Freitas, H.; Santos, X. The role of fire history, land-use, and vegetation structure on the response of Mediterranean lizards to fire. For. Ecol. Manag. 2018, 419–420, 139–145. [Google Scholar] [CrossRef]
- Chergui, B.; Fahd, S.; Santos, X. Are reptile responses to fire shaped by forest type and vegetation structure? Insights from the Mediterranean basin. For. Ecol. Manag. 2019, 437, 340–347. [Google Scholar] [CrossRef]
- Nimmo, D.G.; Kelly, L.T.; Spence-Bailey, L.M.; Watson, S.J.; Taylor, R.S.; Clarke, M.F.; Bennett, A.F. Fire Mosaics and Reptile Conservation in a Fire-Prone Region. Conserv. Biol. 2013, 27, 345–353. [Google Scholar] [CrossRef]
- Santos, X.; Chergui, B.; Belliure, J.; Moreira, F.; Pausas, J.G. Reptile responses to fire across the western Mediterranean Basin. Conserv. Biol. 2025, 39, e14326. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.L.; Lim, A.S.Y. Hidden influence of fire on locally rare and cryptic reptile species. Ecology 2025, 106, e70121. [Google Scholar] [CrossRef] [PubMed]
- Schurbon, J.M.; Fauth, J.E. Effects of Prescribed Burning on Amphibian Diversity in a Southeastern U.S. National Forest. Conserv. Biol. 2003, 17, 1338–1349. [Google Scholar] [CrossRef]
- dos Anjos, A.G.; Solé, M.; Benchimol, M. Fire effects on anurans: What we know so far? For. Ecol. Manag. 2021, 495, 119338. [Google Scholar] [CrossRef]
- Eales, J.; Haddaway, N.R.; Bernes, C.; Cooke, S.J.; Jonsson, B.G.; Kouki, J.; Petrokofsky, G.; Taylor, J.J. What is the effect of prescribed burning in temperate and boreal forest on biodiversity, beyond pyrophilous and saproxylic species? A systematic review. Environ. Evid. 2018, 7, 19. [Google Scholar] [CrossRef]
- Sanz-Aguilar, A.; Anadón, J.D.; Giménez, A.; Ballestar, R.; Graciá, E.; Oro, D. Coexisting with fire: The case of the terrestrial tortoise Testudo graeca in mediterranean shrublands. Biol. Conserv. 2011, 144, 1040–1049. [Google Scholar] [CrossRef]
- Hailey, A. The effects of fire and mechanical habitat destruction on survival of the tortoise Testudo hermanni in northern Greece. Biol. Conserv. 2000, 92, 321–333. [Google Scholar] [CrossRef]
- Valakos, E.D.; Pafilis, P.; Sotiropoulos, K.; Lymberakis, P.; Maragou, P.; Foufopoulos, J. The Amphibians and Reptiles of Greece; Chimaira: Frankfurt am Main, Germany, 2008. [Google Scholar]
- Kati, V.; Foufopoulos, J.; Ioannidis, Y.; Papaioannou, H.; Poirazidis, K.; Lebrun, P. Diversity, ecological structure and conservation of herpetofauna in a Mediterranean area (Dadia National Park, Greece). Amphib.-Reptil. 2007, 28, 517–529. [Google Scholar] [CrossRef]
- Christopoulos, A. New record of Telescopus fallax (Squamata: Colubridae) in Rhodope Prefecture, Thrace, Greece. Herpetol. Notes 2022, 15, 873–876. [Google Scholar]
- Bakaloudis, D.E.; Vlachos, C.G.; Holloway, G.J. Habitat use by short-toed eagles Circaetus gallicus and their reptilian prey during the breeding season in Dadia Forest (north-eastern Greece). J. Appl. Ecol. 1998, 35, 821–828. [Google Scholar] [CrossRef]
- Bakaloudis, D. Spatial and temporal variation of reptiles. In The Dadia-Lefkimi-Soufli Forest National Park, Greece: Biodiversity, Management and Conservation; Catsadorakis, G., Kallander, H., Eds.; WWF Greece: Athens, Greece, 2010; pp. 139–150. [Google Scholar]
- Koukouli, M.-E.; Pseftogkas, A.; Karagkiozidis, D.; Mermigkas, M.; Panou, T.; Balis, D.; Bais, A. Extreme wildfires over Northern Greece during Summer 2023—Part B. Adverse effects on regional air quality. Atmos. Res. 2025, 320, 108034. [Google Scholar] [CrossRef]
- Buckland, S.T.; Anderson, D.R.; Burnham, K.P.; Laake, J.L.; Borchers, D.L.; Thomas, L. Introduction to Distance Sampling. Estimating Abundance of Biological Populations; Oxford University Press: Oxford, UK, 2001. [Google Scholar]
- Jaeger, R.G. Transect sampling. In Biological Diversity Handbook Series Measuring and Monitoring Biological Diversity; Heyer, W.R., Donnelly, M.A., McDiarmin, R.W., Hayek, L.C., Foster, M.C., Eds.; Smithsonian Institution Press: Washington, DC, USA; London, UK, 1994; pp. 103–107. [Google Scholar]
- MacKenzie, D.I.; Nichols, J.D.; Lachman, G.B.; Droege, S.; Andrew Royle, J.; Langtimm, C.A. Estimating site occupancy rates when detection probabilities are less than one. Ecology 2002, 83, 2248–2255. [Google Scholar] [CrossRef]
- MacKenzie, D.I.; Nichols, J.D.; Royle, J.A.; Pollock, K.H.; Bailey, L.; Hines, J.E. Occupancy Estimation and Modeling: Inferring Patterns and Dynamics of Species Occurrence, 2nd ed.; Academic Press: Boston, MA, USA, 2018. [Google Scholar]
- Shannon, C.E. A mathematical theory of communication. Bell Syst. Tech. 1948, 27, 379–423. [Google Scholar]
- Krebs, C.J. Ecological Methodology; Harper Collins: New York, NY, USA, 1998. [Google Scholar]
- Simpson, E.H. Measurement of diversity. Nature 1949, 163, 688. [Google Scholar] [CrossRef]
- Margalef, R. Homage to Evelyn Hutchinson, or why is there an upper limit to diversity. Trans. Conn. Acad. Arts Sci. 1972, 44, 211–235. [Google Scholar]
- Oksanen, J.; Simpson, G.; Blanchet, F.; Kindt, R.; Legendre, P.; Minchin, P.; O’Hara, R.; Solymos, P.; Stevens, M.; Szoecs, E.; et al. vegan: Community Ecology Package. R Package Version 2.8-0. Available online: https://vegandevs.github.io/vegan/ (accessed on 14 December 2025).
- R Development Core Team. R: A Language and Environment for Statistical Computing (Version 4.3); R Foundation for Statistical Computing: Vienna, Austria, 2024. [Google Scholar]
- Kelly, L.T.; Haslem, A.; Holland, G.J.; Leonard, S.W.J.; MacHunter, J.; Bassett, M.; Bennett, A.F.; Bruce, M.J.; Chia, E.K.; Christie, F.J.; et al. Fire regimes and environmental gradients shape vertebrate and plant distributions in temperate eucalypt forests. Ecosphere 2017, 8, e01781. [Google Scholar] [CrossRef]
- Kennedy, P.L.; Fontaine, J.B. Synthesis of Knowledge on the Effects of Fire and Fire Surrogates on Wildlife in U.S. Dry Forests; Oregon State University Agricultural Experiment Station: Hermiston, OR, USA, 2009. [Google Scholar]
- Klaus, J.M.; Noss, R.F. Specialist and generalist amphibians respond to wetland restoration treatments. J. Wildl. Manag. 2016, 80, 1106–1119. [Google Scholar] [CrossRef]
- Moseley, K.R.; Castleberry, S.B.; Schweitzer, S.H. Effects of prescribed fire on herpetofauna in bottomland hardwood forests. Southeast Nat. 2003, 2, 475–486. [Google Scholar]
- Crisafulli, C.M.; Trippe, L.S.; Hawkins, C.P.; MacMahon, J.A. Amphibian Responses to the 1980 Eruption of Mount St. Helens. In Ecological Responses to the 1980 Eruption of Mount St. Helens; Dale, V.H., Swanson, F.J., Crisafulli, C.M., Eds.; Springer: New York, NY, USA, 2005; pp. 183–197. [Google Scholar]
- Gorman, T.A.; Haas, C.A.; Himes, J.G. Evaluating Methods to Restore Amphibian Habitat in Fire-Suppressed Pine Flatwoods Wetlands. Fire Ecol. 2013, 9, 96–109. [Google Scholar] [CrossRef]
- Larson, D.M. Grassland Fire and Cattle Grazing Regulate Reptile and Amphibian Assembly Among Patches. Environ. Manag. 2014, 54, 1434–1444. [Google Scholar] [CrossRef]
- Ruthven, D.C.; Kazmaier, R.T.; Janis, M.W. Short-term response of herpetofauna to various burning regimes in the South Texas plains. Southeast Nat. 2008, 53, 480–487. [Google Scholar] [CrossRef]
- Keyser, P.D.; Sausville, D.J.; Ford, W.M.; Schwab, D.J.; Brose, P.H. Prescribed fire impacts to amphibians and reptiles in shelterwood-harvested oak-dominated forests. Va. J. Sci. 2004, 55, 159–168. [Google Scholar]
- Santos, X.; Belliure, J.; Gonçalves, J.F.; Pausas, J.G. Resilience of reptiles to megafires. Ecol. Appl. 2022, 32, e2518. [Google Scholar] [CrossRef] [PubMed]
- Mulhall, S.J.; Di Stefano, J.; Dorph, A.; Swan, M.; Sitters, H. Do reptile responses to habitat structure and time since fire depend on landscape structure? For. Ecol. Manag. 2024, 553, 121564. [Google Scholar] [CrossRef]
- WWF Hellas. Management Guidelines for the Restoration of Forest Ecosystems After the Evros Fire 2023; WWF Hellas: Athens, Greece, 2024; p. 28. [Google Scholar]
- Chergui, B.; Rodríguez-Caro, R.C.; Graciá, E.; Fahd, S.; Santos, X. Population density of the spur-thighed tortoise Testudo graeca declines after fire in north-western Africa. PLoS ONE 2019, 14, e0220969. [Google Scholar] [CrossRef]
- Lecq, S.; Ballouard, J.M.; Caron, S.; Livoreil, B.; Seynaeve, V.; Matthieu, L.A.; Bonnet, X. Body condition and habitat use by Hermann’s tortoises in burnt and intact habitats. Conserv. Physiol. 2014, 2, cou019. [Google Scholar] [CrossRef][Green Version]
- Anadón, J.D.; Giménez, A.; Martínez, M.; Martínez, J.; Pérez, I.; Esteve, M.A. Factors determining the distribution of the spur-thighed tortoise Testudo graeca in south-east spain: A hierarchical approach. Ecography 2006, 29, 339–346. [Google Scholar] [CrossRef]
- Dorcas, M.E.; Willson, J.D. Innovative Methods for Studies of Snake Ecology and Conservation. In Snakes; Mullin, S.J., Seigel, R.A., Eds.; Cornell University Press: Ithaca, NY, USA, 2011; pp. 5–37. [Google Scholar]
- Braithwaite, R.W. Effects of Fire Regimes on Lizards in the Wet-Dry Tropics of Australia. J. Trop. Ecol. 1987, 3, 265–275. [Google Scholar] [CrossRef]
- Pilliod, D.S.; Bury, R.B.; Hyde, E.J.; Pearl, C.A.; Corn, P.S. Fire and amphibians in North America. For. Ecol. Manag. 2003, 178, 163–181. [Google Scholar] [CrossRef]
- Driscoll, D.A.; Henderson, M.K. How many common reptile species are fire specialists? A replicated natural experiment highlights the predictive weakness of a fire succession model. Biol. Conserv. 2008, 141, 460–471. [Google Scholar] [CrossRef]
- Lindenmayer, D.B.; Wood, J.T.; MacGregor, C.; Michael, D.R.; Cunningham, R.B.; Crane, M.; Montague-Drake, R.; Brown, D.; Muntz, R.; Driscoll, D.A. How predictable are reptile responses to wildfire? Oikos 2008, 117, 1086–1097. [Google Scholar] [CrossRef]
- Catsadorakis, G.; Källander, H. The Dadia–Lefkimi–Soufli Forest National Park, Greece: Biodiversity, Management and Conservation; WWF Greece: Athens, Greece, 2010. [Google Scholar]
- Poirazidis, K. Systematic Raptor Monitoring as conservation tool: 12 year results in the light of landscape changes in Dadia-Lefkimi-Soufli National Park. Nat. Conserv. 2017, 22, 17–50. [Google Scholar] [CrossRef]
- Pausas, J.G.; Fernández-Muñoz, S. Fire regime changes in the Western Mediterranean Basin: From fuel-limited to drought-driven fire regime. Clim. Change 2012, 110, 215–226. [Google Scholar] [CrossRef]
- Abatzoglou, J.T.; Kolden, C.A.; Cullen, A.C.; Sadegh, M.; Williams, E.L.; Turco, M.; Jones, M.W. Climate change has increased the odds of extreme regional forest fire years globally. Nat. Comm. 2025, 16, 6390. [Google Scholar] [CrossRef]
- Moreira, F.; Ascoli, D.; Safford, H.; Adams, M.A.; Moreno, J.M.; Pereira, J.M.C.; Catry, F.X.; Armesto, J.; Bond, W.; González, M.E.; et al. Wildfire management in Mediterranean-type regions: Paradigm change needed. Environ. Res. Lett. 2020, 15, 011001. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.