Abstract
The increase in aesthetic demands has increased the use of ceramic dental restorations in dentistry. Ceramic restorations are bonded to the tooth structure using adhesives. There is a lack of standard guidelines in terms of post-bleaching time intervals and bond strengths of ceramic restorations. Bleaching products have also been stated to affect the morphology of enamel. Therefore, the purpose of this systematic review is to evaluate the bond strength between ceramic and enamel and the changes in the surface roughness of enamel post teeth bleaching. An electronic search was performed in the databases PubMed, OVID MEDLINE, Scopus, and Web of Science with MESH terms: “adhesion OR bonding”, “ceramic OR ceramics OR dental porcelain”, “tooth bleaching OR teeth bleaching OR tooth whitening OR teeth whitening” and “enamel OR dental enamel OR enamels OR dental enamels”. The articles were screened, and the final selection of articles was obtained by using the inclusion and exclusion criteria. Of the 170 studies identified from the search, only 12 studies met the inclusion criteria and were selected for full-text review. A further search by hand was performed, and additional 48 studies were selected. From the 60 full-text studies, 18 studies met the inclusion criteria and were included for data extraction. The results were based on a descriptive analysis of the effect on bond strength of ceramic to enamel after a bleaching protocol and the changes in the surface roughness of enamel post bleaching protocol. In conclusion, bleaching protocols alter the surface roughness of enamel and, thus, the shear bond strength between ceramic and enamel. Bleaching treatments with a higher concentration of hydrogen peroxide reduce the bond strength between ceramic and enamel. Delaying bonding after bleaching for up to 7 days increases the bond strength between ceramic and enamel.
1. Introduction
With increased knowledge of aesthetics and patient demand for a perfect smile, ceramic restorations and teeth whitening have become popular treatment options in aesthetic dentistry. Tooth whitening, especially, became popular with the increased number of at-home and in-office bleaching products available in the market [1,2,3]. Bleaching is a conventional, simple, and economical treatment option available for changing the colour of teeth and improving the harmony of the smile [4]. The first known article related to home bleaching techniques using carbamide peroxide was published in 1989 [5]. Since then, many novel systems have been introduced to the market with various concentrations of bleaching agents, including carbamide peroxide and hydrogen peroxide [6,7]. The mechanics of teeth whitening is that hydrogen peroxide (which is also produced when carbamide peroxide reacts with water) molecules penetrate through the enamel and oxidise the organic materials, reducing colour and bleaching the tooth [8]. Despite its clinical success and high patient acceptance, tooth bleaching obviously raises concerns among dental practitioners since it may not only weaken enamel and dentine but also alter the bond strength of both composite and ceramic restorations [9,10,11]. While more rapid bleaching of teeth is available with dental practitioners, with 25–40% concentrations of peroxide or carbamide, lower concentrations of 3–25% are available for use at home [2,12]. However, previous studies have shown that when a 35% concentration of hydrogen peroxide is used, it may alter enamel composition and structure [13,14].
Past studies have reported that weakened enamel due to bleaching decreases its bond strength to restorative materials and impacts the surface roughness [15], morphology [16,17] and microhardness [18,19,20] of human enamel [21]. Studies have also found alterations in enamel structure (with an increase in surface roughness) and in the depth of grooves after bleaching procedures [22,23,24]. However, some studies found minimal or no effect on the surface morphology of enamel after bleaching procedures [25,26,27]. The hydrogen peroxide in bleaching products alters the calcium-to-phosphate ratio within the enamel and forms porosity on superficial enamel and the loss of prismatic form [15,16,17,18]. The oxygen in hydrogen peroxide is absorbed into the tooth enamel and, as a result, into the dentine before being released through surface diffusion and accumulating within the enamel structure, reducing the bond strength of resin-based restorative materials [28,29].
The preferential use of aesthetic dental materials for restorations due to the increase in the aesthetic demands of patients has led to an increase in ceramic systems, especially ceramic crowns [30,31,32]. The bonding strength of resin to enamel is dependent on the time of bonding after bleaching [33]. The low shear bond strength between resin and enamel is shown in the first two weeks after bleaching [33]; however, the reduction of bond strength between resin and enamel is more significant in the first 24 h post bleaching [11]. The study by Cavalli et al., (2001) [33] showed that after vital bleaching with hydrogen-peroxide- and carbide-peroxide-based bleaching agents, it takes almost three weeks for the resin–enamel bond strength values to return to unbleached enamel values. This was also investigated by Basting et al., (2003) [34]. The reduction in bonding between ceramic restoration and enamel post bleaching is a concern in aesthetic dentistry, where vital bleaching is required to improve the appearance of teeth preceding the bonded restoration [34].
The study by Rahul et al., (2017) [35] evaluated the shear bond strength of at-home and in-office bleaching agents and found that using an in-office bleaching technique 24 h prior to the bonding procedure significantly reduced the shear bond strength between ceramic and enamel, whereas at-home bleaching agents did not have any significant effect on bond strength [34]. Similar findings were also reported by Nilgum et al., (2011), who found a significant reduction in the micro tensile bond strength between enamel and ceramic laminate veneers [36]. The investigators further stated that an important factor in the effectiveness of the bonding of ceramics is the bleaching procedure [37]. However, Oztas et al., (2012) did not find any significant difference within 24 h or 14 days after bleaching in the shear bond strength of ceramic orthodontic brackets within two groups of chemically and light-cured composite resins attached to enamel [37]. The study conducted by Firoozmand et al., (2013) further reported that in addition to the type of bleaching treatment, the type of bonding resin also affects the bonding strength of restorations [38].
The increase in aesthetic demands has increased the use of ceramic dental restorations in dentistry. Ceramic restorations are bonded to the tooth structure using adhesives. There is a lack of standard guidelines in terms of post-bleaching time intervals and the bond strength of ceramic restorations. Bleaching products have also been stated to affect the surface morphology of enamel. Therefore, the purpose of this systematic review is to evaluate the bond strength between ceramic and enamel and the changes in surface roughness of enamel post teeth bleaching.
2. Materials and Methods
2.1. Search Strategy
A search was performed in the PubMed, OVID MEDLINE, Scopus, and Web of Science databases with MESH (Medical Subject Headings) terms: “adhesion OR bonding”, “ceramic OR ceramics OR dental porcelain”, “tooth bleaching OR teeth bleaching OR tooth whitening OR teeth whitening” and “enamel OR dental enamel OR enamels OR dental enamels”. The search strategy focused on patient, intervention, comparison, and outcome PICO criteria, and the focus questions are presented in Table 1.

Table 1.
Focus questions and PICO criteria.
Database searches were restricted to the English language, in vitro studies, in vivo studies, randomised control trials, case–control studies, and studies with commercially available products. The search includes all available scientific evidence in PubMed, OVID Medline, Scopus, Web of Sciences databases up to February 2022. Further hand searches of studies identified from the bibliographies of relevant studies were conducted. Following the inclusion and exclusion criteria, the studies were then included or excluded from the total studies identified (Table 2).

Table 2.
Inclusion and exclusion criteria.
2.2. Selection of Studies
Potential studies were assessed independently by three reviewers (I.S., A.G., J.J.E.C.) following the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines [39]. After applying the exclusion and inclusion criteria, full-text studies were elected for both reading and final selection. All differences in the selection of the articles between the three authors were analysed, and agreement was established through discussion, as per the PRISMA guidelines.
3. Results
Of the 170 studies identified from the search, only 12 studies met the inclusion criteria and were selected for full-text review based on the information given in the abstract. A further hand search was performed from the citations of the 12 studies, and 48 additional studies were selected for full-text review. Only 18 of the 60 full-text studies met the inclusion criteria and were included in the data extraction process (Figure 1).
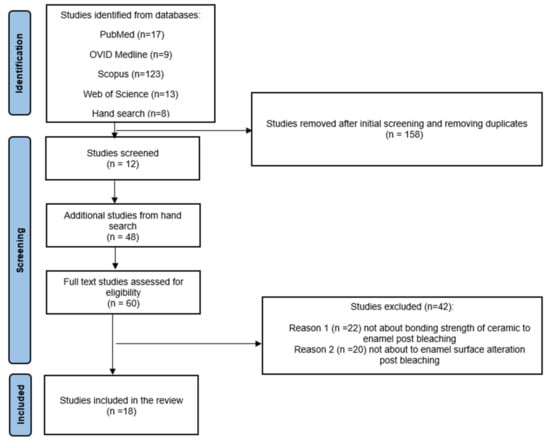
Figure 1.
PRISMA flow diagram summarising the selection process.
3.1. Effect on Bond Strength of Ceramic to Enamel after Bleaching Protocol
Of the 18 studies included in the review, a total of 9 studies evaluated the bond strength of ceramic to enamel after bleaching protocols [35,36,37,38,40,41,42,43,44] (Table 3). Three studies that used a 10% carbamide peroxide bleaching agent did not find any significant difference in shear mean bond strength values between enamel and ceramic after the application of the bleach for either 10, 20, or 30 s [41] when bonding was done directly after bleaching [42] and 24 h after the bleaching procedure [35]. Similarly, Öztas et al., (2011) did not find any significant difference in the shear bond strength of ceramic orthodontic brackets bonded with either chemically or light-cured composite resins to enamel both 24 h and 14 days after bleaching with 20% carbamide peroxide [37]. However, a study that used 35% hydrogen peroxide bleach recorded an increase in the shear bond strength of ceramic brackets bonded to the enamel with an application of desensitising agent after bleaching when compared to a specimen that had not undergone any bleaching protocol at all and also when compared to a specimen with three applications of 35% hydrogen peroxide bleach for 15 min [40]. Similarly, Gökce et al., (2008) also showed significantly higher bond strengths in samples with an antioxidant application (10% sodium ascorbate) after the bleaching procedure [44]. Delayed bonding (up to 7 days) was also found by two studies to increase the shear bonding strength of ceramic to enamel [40,44].

Table 3.
Main findings of studies on bond strength of ceramic/metal to enamel after bleaching protocol.
Two studies that used 35% hydrogen peroxide bleach found that the type of adhesive influences the bond strength of ceramic to enamel post bleaching procedure [36,38]. Interestingly, both studies showed that the type of luting cement affects the bond strength of ceramics to enamel post bleaching procedure. The study by Nilgun Ozturk et al., (2011) [36] recorded the highest bond strength in specimens with the Transbond XT resin adhesive, with an etching time of 30 s compared to specimens with Rely-X veneer cement adhesive; Firoozmand et al., (2013) showed higher bond strength values in specimens with Transbond XT resin adhesive compared to the Z250 composite resin adhesive [38]. The concentration of the bleach was also found to influence the bonding strength of ceramic to enamel [35,43]. The study by Dastijerdi et al., (2015) showed significantly lower bond strengths in 45% carbamide peroxide specimens than in 20% carbamide peroxide specimens when bonding was done 24 h after bleaching [43]. Similarly, Rahul et al., (2017) also found significantly lower bond strengths in specimens bleached with 40% hydrogen peroxide compared to specimens bleached with 10% carbamide peroxide when bonding was done 24 h after a bleaching procedure [35].
3.2. Changes in Surface Roughness of Enamel Post Bleaching Protocol
A total of 10 studies carried out qualitative analysis of the surface roughness of enamel after bleaching procedures using different concentrations of hydrogen peroxide (6–35%) and other commercially available bleaching systems, such as Proxigel (10% carbamide and cabapol), White and Bright (10% carbamide peroxide), Opalescence, Vital home bleaching, Rembrandt, Nupro Gold, and also placebo agents for control (Table 4). Four studies recorded minimal or no alteration to enamel surface morphology [22,25,29]. The hydrogen peroxide/carbamide peroxide contents of the bleaching agents used by these four studies were in the range of 6–22%. Alterations in enamel surface morphology were recorded by five studies using either SEM analysis, AFM analysis or light microscopic observation [13,26,45,46,47] and these changes included: ridged and scored, scattered depressions of various diameters and depths, a muted texture of rounded enamel rods with depressed rod boundaries, the unclear structure of dental enamel, seen by the blurring of the striae of Retzius and a darkening of the subsurface zone of enamel, indicating mineral loss in this region; a partially etched surface with many shallow depressions and an increase in surface porosity; white precipitate that could be seen in association with the enamel surface, giving it a frosted appearance. Three studies recorded greater enamel surface changes in specimens treated with 30% or more concentrations of hydrogen peroxide solution compared to the specimen treatment with bleaching agents with lower concentrations of hydrogen peroxide [45,46,47].

Table 4.
Main findings of studies on surface roughness of enamel after bleaching.
3.3. Risk of Bias
The results of the assessment of the 18 studies are presented in Table 5. None of the studies reported a method of determining the sample size or the specimen selection principle, and none of the in vitro studies applied the mechanism of randomisation, allocation, implementation, and blinding [6,7,8,9]. All included studies had reported background and objectives, intervention and outcomes accurately. However, one study did not include an abstract [29]. Several studies did not report specific objectives (n = 8) [10,13,25,29,40,43,45,47], sample size (n = 3) [10,35,47], statistical methods [10,22,25,29] and outcome and estimation [29,47]. Only five studies included had supporting funding for their research [10,22,37,38,47]. The risk of bias assessment showed that all included studies had a low risk of bias.

Table 5.
Risk of Bias.
4. Discussion
This systematic review has aimed to appraise the current literature on the bond strength of ceramic to bleached enamel and changes in the surface roughness of enamel post bleaching protocol. Ceramic restorations and tooth bleaching have become popular treatment options in response to modern aesthetic demands in dentistry. The bleaching protocol involves the penetration of hydrogen peroxide or carbamide peroxide molecules into the tooth structure and the subsequent oxidisation of the stain molecules [2]. Residual oxygen released during the bleaching process prevents resin penetration into the etched enamel and, therefore, reduces resin polymerisation, decreasing bond strength [10].
Two studies analysed the different methods of bleaching on bond strength between enamel and ceramic and found that bonding strength is influenced by the concentration of bleaching agents [35,43]. One study compared the bonding strength of specimens bleached with 45% carbamide peroxide with specimens bleached with 20% carbamide peroxide [43], while the other study [35] compared specimens bleached with 40% hydrogen peroxide with specimens bleached with 10% carbamide peroxide. Both studies found that specimens that were subjected to bleaching protocols containing a higher concentration of carbamide peroxide and hydrogen peroxide had significantly lower bonding strength. Three other studies that used 10–20% carbamide peroxide bleaching agents [37,41,42] did not report any statistically significant difference in bonding strength. One of the most commonly used active bleaching agents is hydrogen peroxide, and with the use of higher concentrations, there is the potential increase in hydrogen peroxide to create oxygen-free radicals that interact with coloured organic molecules and oxidise pigment stains and macromolecules, which change dental discolouration into smaller and lighter molecules [33,35]. Subsequently, more residual oxygen will be present on the etched enamel surface; this will interfere with the resin infiltration into the enamel structure and also the polymerisation of the resin.
A few studies found that in addition to delayed bonding (up to 7 days), applications of 10% sodium ascorbate (antioxidant) [44] and desensitising agents [40] after the bleaching treatment increased the bond strength between ceramic and enamel. The authors further stated that a possible reason for an increase in bonding strength in delayed bonding situations may be that the residual oxygen gets neutralised over time and, thus, does not interfere with the polymerisation of resin composites for bonding. Another study indicated that the enamel surface immediately after in-office or home bleaching is unsuitable for the adhesion of restorative materials, and it is essential to allow adequate time after bleaching to ensure the normal penetration of adhesives onto the enamel surface [36].
Two studies found higher bond strength in specimens with an application of either desensitising agent or antioxidant after bleaching compared to that of unbleached specimens [40,44]. The sodium ascorbate antioxidant has been stated to facilitate the free-radical polymerisation of adhesive resins without early cessation by re-establishing the altered redox potential of the oxidised bonding substrate and subsequently reversing the compromised bonding effect [44]. The use of desensitising agents directly after bleaching with hydrogen peroxide could be considered an alternative procedure to decrease undesired consequences of residual oxygen on bond strength. However, it is recommended to conduct further studies on the chemical and mechanical effects of bleaching gels containing calcium and desensitising agents that are applied to dental structures [40].
The concentration of hydrogen peroxide/carbamide peroxide has also been shown to influence the surface alteration of enamel. Studies recorded greater enamel surface changes in specimens treated with a 30% or higher concentration of hydrogen peroxide solution than specimen treatments with bleaching agents with lower concentrations (range of 6–22%) of hydrogen peroxide [45,46,47]. The study by Hegedüs et al., (1999) examined the effect of bleaching agents (Opalescence, Nite White, 30% hydrogen peroxide solution) on the enamel surface with atomic force microscopy and found that the depth of grooves present on the enamel surface post bleaching is directly proportional to the concentration of hydrogen peroxide [46]. The authors suggested that peroxide affects the inner structure and organic phase of enamel. The reaction between peroxide and the organic materials on either the surface or subsurface enamel causes alterations in the enamel surface structure [47].
The efficacy of any bleaching procedure depends on the concentration of bleaching agents and bleaching time [1]. The current literature indicates that both at-home and in-office bleaching can affect the surface roughness of enamel and, thus, the shear bond strength between ceramic and enamel [48,49,50]. Therefore, clinicians need to understand the effect of bleaching treatments on enamel and follow protocols shown by studies to improve the bonding strength between ceramic and enamel to increase the clinical success rate in aesthetic dentistry.
5. Conclusions
The findings of this systematic review have drawn the following conclusions:
- Bleaching treatments alter the surface roughness of enamel and, thus, the shear bond strength between ceramic and enamel.
- Bleaching treatments with a higher concentration of hydrogen peroxide (more than 35%) reduce the bond strength between ceramic and enamel.
- Delaying bonding after bleaching for up to 7 days increases the bond strength between ceramic and enamel.
Author Contributions
Conceptualization, J.J.E.C. and T.H.S.; methodology, J.J.E.C., A.G. and T.H.S.; formal analysis, J.J.E.C., A.G., V.M. and T.H.S.; investigation, J.J.E.C., A.G., V.M. and T.H.S.; writing—original draft preparation, J.J.E.C., A.G., V.M. and T.H.S.; writing—review and editing, J.J.E.C., A.G., V.M. and T.H.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Carey, C.M. Tooth whitening: What we now know. J. Evid. Based Dent. Pract. 2014, 14, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Kwon, S.R.; Wertz, P.W. Review of the mechanism of tooth whitening. J. Esthet. Restor. Dent. 2015, 27, 240–257. [Google Scholar] [CrossRef] [PubMed]
- Kang, T.-Y.; Kim, J.-H.; Kwon, J.-S. In Vitro Effects of Cyclic Dislodgement on Retentive Properties of Various Titanium-Based Dental Implant Overdentures Attachment System. Materials 2019, 12, 3770. [Google Scholar] [CrossRef]
- Giannini, M.; Silva, A.P.; Cavalli, V.; Leme, A.F.P. Effect of carbamide peroxide-based bleaching agents containing fluoride or calcium on tensile strength of human enamel. J. Appl. Oral Sci. 2006, 14, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Haywood, V.B.; O Heymann, H. Nightguard vital bleaching. Quintessence Int. 1989, 20, 173–176. [Google Scholar] [PubMed]
- Kihn, P.W.; Barnes, D.M.; Romberg, E.; Peterson, K.A. Clinical evaluation of 10 percent vs. 15 percent carbamide peroxide tooth-whitening agents. J. Am. Dent. Assoc. 2000, 131, 1478–1484. [Google Scholar] [CrossRef] [PubMed]
- Christensen, G.J. Bleaching teeth: Practitioner trends. J. Am. Dent. Assoc. 1997, 128, 16S–18S. [Google Scholar] [CrossRef]
- Joiner, A. The bleaching of teeth: A review of the literature. J. Dent. 2006, 34, 412–419. [Google Scholar] [CrossRef]
- Türkün, M.; Kaya, A.D. Effect of 10% sodium ascorbate on the shear bond strength of composite resin to bleached bovine enamel. J. Oral Rehabil. 2004, 31, 1184–1191. [Google Scholar] [CrossRef]
- Titley, K.; Torneck, C.; Smith, D.; Chernecky, R.; Adibfar, A. Scanning electron microscopy observations on the penetration and structure of resin tags in bleached and unbleached bovine enamel. J. Endod. 1991, 17, 72–75. [Google Scholar] [CrossRef]
- García-Godoy, F.; Dodge, W.W.; Donohue, M.A.; O’Quinn, J. Composite resin bond strength after enamel bleaching. Oper. Dent. 1993, 18, 144. [Google Scholar]
- Silva, F.B.D.; Chisini, L.A.; Demarco, F.F.; Horta, B.L.; Correa, M.B. Desire for tooth bleaching and treatment performed in Brazilian adults: Findings from a birth cohort. Braz. Oral Res. 2018, 32, e12. [Google Scholar] [CrossRef]
- Josey, A.; Meyers, I.; Romaniuk, K.; Symons, A. The effect of a vital bleaching technique on enamel surface morphology and the bonding of composite resin to enamel. J. Oral Rehabil. 1996, 23, 244–250. [Google Scholar] [CrossRef]
- Ruse, N.D.; Smith, D.; Torneck, C.; Titley, K. Preliminary Surface Analysis of Etched, Bleached, and Normal Bovine Enamel. J. Dent. Res. 1990, 69, 1610–1613. [Google Scholar] [CrossRef]
- Rosa, E.; Soares, P. Effect of fluoride therapies on the surface roughness of human enamel exposed to bleaching agents. Quintessence Int. 2010, 41, 71–78. [Google Scholar]
- Al-Salehi, S.; Wood, D.; Hatton, P. The effect of 24h non-stop hydrogen peroxide concentration on bovine enamel and dentine mineral content and microhardness. J. Dent. 2007, 35, 845–850. [Google Scholar] [CrossRef]
- Grazioli, G.; Valente, L.L.; Isolan, C.P.; Pinheiro, H.A.; Duarte, C.G.; Münchow, E.A. Bleaching and enamel surface interactions resulting from the use of highly-concentrated bleaching gels. Arch. Oral Biol. 2018, 87, 157–162. [Google Scholar] [CrossRef]
- Attin, T.; Schmidlin, P.R.; Wegehaupt, F.; Wiegand, A. Influence of study design on the impact of bleaching agents on dental enamel microhardness: A review. Dent. Mater. 2009, 25, 143–157. [Google Scholar] [CrossRef]
- Attin, T.; Kocabiyik, M.; Buchalla, W.; Hannig, C.; Becker, K. Susceptibility of Enamel Surfaces to Demineralization after Application of Fluoridated Carbamide Peroxide Gels. Caries Res. 2003, 37, 93–99. [Google Scholar] [CrossRef]
- Chen, H.-P.; Chang, C.-H.; Liu, J.-K.; Chuang, S.-F.; Yang, J.-Y. Effect of fluoride containing bleaching agents on enamel surface properties. J. Dent. 2008, 36, 718–725. [Google Scholar] [CrossRef]
- Silva, N.R.; Coelho, P.G.; Valverde, G.B.; Becker, K.; Ihrke, R.; Quade, A.; Thompson, V.P. Surface characterization of Ti and Y-TZP following non-thermal plasma exposure. J. Biomed. Mater. Res. Part B Appl. Biomater. 2011, 99, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Leonard, R.H., Jr.; Eagle, J.C.; Garland, G.E.; Matthews, K.P.; Rudd, A.L.; Phillips, C. Nightguard vital bleaching and its effect on enamel surface morphology. J. Esthet. Restor. Dent. 2001, 13, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Titley, K.; Torneck, C.; Ruse, N.; Krmec, D. Adhesion of a resin composite to bleached and unbleached human enamel. J. Endod. 1993, 19, 112–115. [Google Scholar] [CrossRef]
- El-Din, A.N.; Miller, B.H.; Griggs, J.A.; Wakefield, C. Immediate bonding to bleached enamel. Oper. Dent. 2006, 31, 106–114. [Google Scholar] [CrossRef]
- Schemehorn, B.; González-Cabezas, C.; Joiner, A. A SEM evaluation of a 6% hydrogen peroxide tooth whitening gel on dental materials in vitro. J. Dent. 2004, 32, 35–39. [Google Scholar] [CrossRef]
- Turker, Ş.B.; Biskin, T. Effect of three bleaching agents on the surface properties of three different esthetic restorative materials. J. Prosthet. Dent. 2003, 89, 466–473. [Google Scholar] [CrossRef]
- Torneck, C.; Titley, K.; Smith, D.; Adibfar, A. The influence of time of hydrogen peroxide exposure on the adhesion of composite resin to bleached bovine enamel. J. Endod. 1990, 16, 123–128. [Google Scholar] [CrossRef]
- Swift, E.J., Jr. Reversal of Compromised Bonding in Bleached Enamel. J. Esthet. Restor. Dent. 2012, 24, 357–358. [Google Scholar] [CrossRef]
- Scherer, W.; Cooper, H.; Ziegler, B.; Vijayaraghavan, T. At-Home Bleaching System: Effects on Enamel and Cementum. J. Esthet. Restor. Dent. 1991, 3, 54–56. [Google Scholar] [CrossRef]
- Komine, F.; Tomic, M.; Gerds, T.; Strub, J.R. Influence of different adhesive resin cements on the fracture strength of aluminum oxide ceramic posterior crowns. J. Prosthet. Dent. 2004, 92, 359–364. [Google Scholar] [CrossRef]
- Raptis, N.V.; Michalakis, K.X.; Hirayama, H. Optical behavior of current ceramic systems. Int. J. Periodontics Restor. Dent. 2006, 26, 31–40. [Google Scholar]
- Li, Q.; Yu, H.; Wang, Y. Colour and surface analysis of carbamide peroxide bleaching effects on the dental restorative materials in situ. J. Dent. 2009, 37, 348–356. [Google Scholar] [CrossRef]
- Cavalli, V.; Reis, A.F.; Giannini, M.; Ambrosano, G.M. The effect of elapsed time following bleaching on enamel bond strength of resin composite. Oper. Dent. 2001, 26, 597–602. [Google Scholar]
- Basting, R.T.; Rodrigues, A.L., Jr.; Serra, M.C. The effects of seven carbamide peroxide bleaching agents on enamel microhardness over time. J. Am. Dent. Assoc. 2003, 134, 1335–1342. [Google Scholar] [CrossRef]
- Rahul, M.; Kumar, P.A.; Nair, A.S.; Mathew, S.; Amaladas, A.S.; Ommen, A. Effects of at-home and in-office bleaching agents on the shear bond strength of metal, ceramic, and composite brackets to enamel. Indian. J. Dent. Res. 2017, 28, 566–573. [Google Scholar]
- Ozturk, A.N.; Oztürk, B.; Malkoc, M.A.; Buyukozer, T. Bond strengths of two ceramic laminate systems to bleached enamel. Mater. Res. Innov. 2011, 15, 63–65. [Google Scholar] [CrossRef]
- Öztaş, E.; Bağdelen, G.; Kılıçoğlu, H.; Ulukapı, H.; Aydin, I. The effect of enamel bleaching on the shear bond strengths of metal and ceramic brackets. Eur. J. Orthod. 2011, 34, 232–237. [Google Scholar] [CrossRef]
- Firoozmand, L.M.; Brandão, J.V.P.; Fialho, M.P.N. Influence of microhybrid resin and etching times on bleached enamel for the bonding of ceramic brackets. Braz. Oral Res. 2013, 27, 142–148. [Google Scholar] [CrossRef]
- Abouassi, T.; Wolkewitz, M.; Hahn, P. Effect of carbamide peroxide and hydrogen peroxide on enamel surface: An in vitro study. Clin. Oral Investig. 2010, 15, 673–680. [Google Scholar] [CrossRef]
- Britto, F.A.R.; Lucato, A.S.; Valdrighi, H.C.; Vedovello, S.A.S. Influence of bleaching and desensitizing gel on bond strength of orthodontic brackets. Dent. Press J. Orthod. 2015, 20, 49–54. [Google Scholar] [CrossRef][Green Version]
- Cooper, J.R.; Young, N.B.; Haywood, V.B.; Mettenburg, D.; Callan, R.S.; Rueggeberg, F.A. Effect of Short-Duration, Localized Carbamide Peroxide Application to Remove Enamel Staining on Bond Strength of Resin Cement to Enamel. J. Esthet. Restor. Dent. 2016, 28, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Miles, P.G.; Pontier, J.-P.; Bahiraei, D.; Close, J. The effect of carbamide peroxide bleach on the tensile bond strength of ceramic brackets: An in vitro study. Am. J. Orthod. Dentofac. Orthop. 1994, 106, 371–375. [Google Scholar] [CrossRef]
- Dastjerdi, E.V.; Khaloo, N.; Mojahedi, S.M.; Azarsina, M. Shear Bond Strength of Orthodontic Brackets to Tooth Enamel After Treatment with Different Tooth Bleaching Methods. Iran. Red Crescent Med. J. 2015, 17, e20618. [Google Scholar] [CrossRef] [PubMed]
- Gökçe, B.; Çömlekoğlu, E.; Özpinar, B.; Türkün, M.; Kaya, A.D. Effect of antioxidant treatment on bond strength of a luting resin to bleached enamel. J. Dent. 2008, 36, 780–785. [Google Scholar] [CrossRef] [PubMed]
- McGuckin, R.S.; Babin, J.; Meyer, B. Alterations in human enamel surface morphology following vital bleaching. J. Prosthet. Dent. 1992, 68, 754–760. [Google Scholar] [CrossRef]
- Hegedüs, C.; Bistey, T.; Flóra-Nagy, E.; Keszthelyi, G.; Jenei, A. An atomic force microscopy study on the effect of bleaching agents on enamel surface. J. Dent. 1999, 27, 509–515. [Google Scholar] [CrossRef]
- Titley, K.; Torneck, C.D.; Smith, D. The effect of concentrated hydrogen peroxide solutions on the surface morphology of human tooth enamel. J. Endod. 1988, 14, 69–74. [Google Scholar] [CrossRef]
- Pinto, C.F.; De Oliveira, R.; Cavalli, V.; Giannini, M. Peroxide bleaching agent effects on enamel surface microhardness, roughness and morphology. Braz. Oral Res. 2004, 18, 306–311. [Google Scholar] [CrossRef]
- Zuryati, A.-G.; Qian, O.Q.; Dasmawati, M. Effects of home bleaching on surface hardness and surface roughness of an experimental nanocomposite. J. Conserv. Dent. 2013, 16, 356–361. [Google Scholar] [CrossRef]
- Moraes, R.R.; Marimon, J.L.M.; Schneider, L.F.J.; Sobrinho, L.C.; Camacho, G.B.; Bueno, M. Carbamide peroxide bleaching agents: Effects on surface roughness of enamel, composite and porcelain. Clin. Oral Investig. 2006, 10, 23–28. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).