Abstract
Encapsulation in suitable nanostructures is a relevant strategy for the preservation and controlled release of bioactive compounds, allowing a safe application. In this work, dichloromethane extracts of Mentha suaveolens Ehrh. and Phytolacca americana L. were encapsulated in chitosan nanostructures and liposomes of soybean lecithin, considering their potential as biopesticides. Liposomes were prepared either by thin film hydration or ethanolic injection methods. For chitosan, the ionic gelation technique was used. The nanosystems were characterized regarding their size and polydispersity. Encapsulation efficiencies of the extracts were determined (being higher than 60%) and the release profiles were measured. Liposomes generally allowed a delayed release of the extracts, while chitosan nanosystems were suitable for a faster and complete release of the potential bioinsecticides.
1. Introduction
Besides the pharmaceutical potential of several plant extracts, their use as repellents and green insecticides has been described [1,2]. However, this application can be hampered by the high volatility and easy degradation of the active compounds. The encapsulation in suitable nanostructures is a relevant strategy for the preservation and controlled release of bioactive compounds, allowing a safe application [3].
Extracts from Mentha suaveolens Ehrh. have been described as potent antimicrobials, together with their analgesic, anti-inflammatory and antioxidant activities [4]. The insecticidal effect of extracts from Mentha suaveolens Ehrh. and Mentha pulegium L. towards Toxoptera aurantii, an insect pest of citrus, has also been reported [5]. On the other hand, Phytolacca americana L. components have been described to exhibit anti-inflammatory, antioxidant, anticancer, anthelmintic, antimicrobial, and insecticidal properties [6,7].
Following previous work [8], the extracts of Mentha suaveolens Ehrh. and Phytolacca americana L. in dichloromethane, after the removal of chlorophylls, have demonstrated a high potential as biopesticides in assays using the Sf9 (Spodoptera frugiperda) insect cell line. Encapsulation in chitosan nanostructures and lecithin liposomes were carried out because both nanosystems have been described as suitable carriers for plant extract components [9,10,11]. Preliminary release experiments were performed, keeping in mind future applications of these extracts as biopesticides.
2. Materials and Methods
2.1. Plant Material
The plant species were harvested in northern Portugal. After drying, the vegetable matter was grounded with a shredder, and the resulting powder passed through a sieve until a particle size ≤900 μm was obtained. The material was stored under vacuum for further use. The dried materials (8 g) were subjected to Soxhlet extraction with dichloromethane (50 mL) for 4 h. The organic solvent was removed by evaporation under reduced pressure at 40 °C. The evaporated extracts of dichloromethane were passed through a Chromabond C18 column, using methanol as eluent, for removal of chlorophylls. After the solvent evaporation under vacuum at 35 °C, the resulting extracts were stored at −20 °C until further use.
2.2. Biological Assays
The impact of the extracts in insect cell viability was evaluated using a resazurin-based assay, after 24 h of incubation. The cells used were Sf9 (Spodoptera frugiperda) and they were plated at a density of 3 × 104 cells/well.
2.3. Nanoencapsulation and Release Studies
For nanoencapsulation studies, liposomes were prepared using soybean lecithin [12] (Sternchemie), by ethanolic injection and thin film hydration, as previously described [13,14]. Four cycles of extrusion (Lipex™ Extruder, Northern Lipids, Burnaby, BC, Canada) through polycarbonate membranes (200 nm pore size) were carried out. Chitosan nanocapsules were obtained by ionic gelation, as reported by Chauhan et al. [15]. Hydrodynamic diameters were obtained by dynamic light scattering (DLS), with five independent measurements for each sample.
Encapsulation efficiencies were determined from the absorbance vs. concentration calibration curves. Absorption spectra were measured in a Shimadzu UV-3600 Plus UV–vis NIR spectrophotometer (Shimadzu Corporation, Kyoto, Japan). Three independent measurements were performed for each system. The extracts released from nanoencapsulation systems to phosphate buffer (pH = 7.3) was followed for 24 h at room temperature.
3. Results and Discussion
3.1. Insecticidal Activity
The activity of Phytolacca americana L. dichloromethane extracts against Sf9 cells has previously shown promising results for its application as a biopesticide [8], and we revisited it here for comparison purposes (Sample A). These studies were expanded to include the dichloromethane extract of Mentha Suaveolens Ehrh (B) (Figure 1).
Figure 1.
Viability of Sf9 cells when exposed to Phytolacca americana (A) or Mentha suaveolens (B) dichloromethane extracts. *** p < 0.001.
As shown in Figure 1, both extracts were capable of eliciting loss of cell viability in the cells under study, when compared to control conditions. Among the two samples, B was the most active, resulting in cell viability around 40%, while A resulted in approximately 75%.
3.2. Encapsulation Systems
Encapsulation assays in liposomes and chitosan nanocapsules were carried out. Chitosan has been reported for the encapsulation of natural compounds and essential oils [9,15]. Regarding the lipid-based systems, ethanolic injection method has been described to be suitable for compounds with a very low solubility in water [13]. On the other hand, the thin film hydration technique allows obtaining homogeneous liposomes after extrusion and is adequate for hydrophilic substances [14]. Table 1 shows the hydrodynamic diameters of the several nanosystems and the encapsulation efficiencies for both plant extracts. The nanosystems present sizes around or below 200 nm, with low standard deviation values.

Table 1.
Hydrodynamic sizes of nanosystems and encapsulation efficiencies, EE (%) ± SD (%), of plant extracts (SD: standard deviation).
The encapsulation efficiencies were higher for liposomes than for chitosan nanocapsules. The lipid nanocarriers prepared by ethanolic injection presented the highest encapsulation efficiency for Mentha suaveolens extract (EE (%) = 84%), while thin film hydration was better for the encapsulation of Phytolacca americana L. extract. Nevertheless, the encapsulation efficiency in chitosan nanostructures was higher than 60% for both extracts.
Preliminary assays of extract release from the nanoencapsulation systems towards phosphate buffer (pH near 7) were performed, and the release profiles are displayed in Figure 2 and Figure 3. It can be observed that the release of encapsulated extracts from chitosan nanocapsules is faster and complete, while liposomes allowed a more delayed release. Extension of the release assays to 40 h in the case of Mentha suaveolens encapsulated in liposomes prepared by ethanolic injection evidenced that the release is continuous, attaining almost 70% at 40 h.
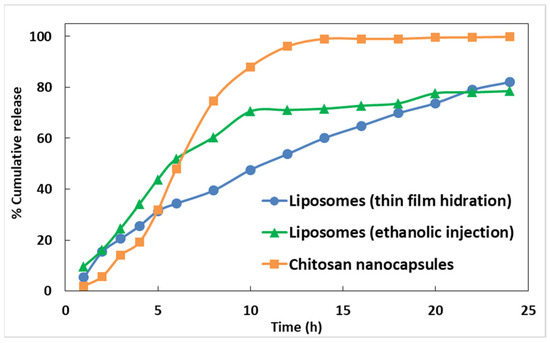
Figure 2.
Release profiles of Phytolacca americana L. extract from liposomes and chitosan nanostructures, for 24 h.
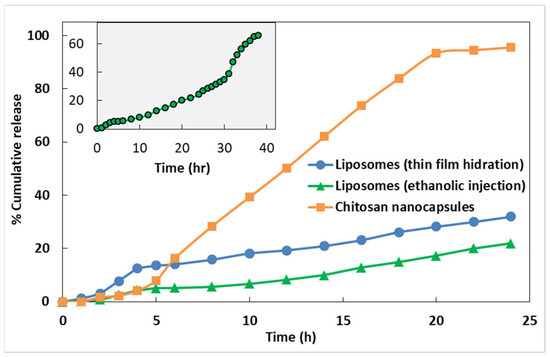
Figure 3.
Release profiles of Mentha suaveolens Ehrh. extract from liposomes and chitosan nanostructures, for 24 h. Inset: Release profile from liposomes prepared by ethanolic injection up to 40 h.
4. Conclusions
Extracts of Mentha suaveolens Ehrh. and Phytolacca americana L. exhibited promising insecticidal activity against Spodoptera frugiperda cells. The extracts were successfully encapsulated in liposomes and chitosan nanocapsules. Liposomes prepared by ethanolic injection presented the highest encapsulation efficiency for Mentha suaveolens extract (EE(%) = 84%), while for Phytolacca americana the thin film hydration method is advantageous. Chitosan nanocapsules displayed lower efficiencies of encapsulation and are suitable for a faster release of the extracts. These results anticipate a promising use of these plant extracts in biopesticide nanoformulations.
Funding
This research was funded by the Foundation for Science and Technology (FCT) for financial support to the research centers CQ/UM (UIDB/00686/2020), CF-UM-UP (UIDB/04650/2020) and REQUIMTE (UIDB/50006/2020). Funding by FCT, COMPETE2020 and FEDER through project PTDC/ASP-AGR/30154/2017 (POCI-01-0145-FEDER-030154) is also acknowledged. The NMR spectrometer Bruker Avance III 400 (part of the National NMR Network) was financed by FCT and FEDER.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Ebadollahi, A.; Ziaee, M.; Palla, F. Essential Oils Extracted from Different Species of the Lamiaceae Plant Family as Prospective Bioagents against Several Detrimental Pests. Molecules 2020, 25, 1556. [Google Scholar] [CrossRef] [PubMed]
- Magierowicz, K.; Górska-Dabrik, E.; Golan, K. Effects of plant extracts and essential oils on the behavior of Acrobasis advenella (Zinck.) caterpillars and females. J. Plant Dis. Prot. 2020, 127, 63–71. [Google Scholar] [CrossRef]
- Nuruzzaman, M.; Rahman, M.M.; Liu, Y.; Naidu, R. Nanoencapsulation, Nano-guard for Pesticides: A New Window for Safe Application. J. Agric. Food Chem. 2016, 64, 1447–1483. [Google Scholar] [CrossRef] [PubMed]
- Božović, M.; Pirolli, A.; Ragno, R. Mentha suaveolens Ehrh. (Lamiaceae) Essential Oil and Its Main Constituent Piperitenone Oxide: Biological Activities and Chemistry. Molecules 2015, 20, 8605–8633. [Google Scholar] [CrossRef] [PubMed]
- Zekri, N.; Handaq, N.; El Caidi, A.; Zair, T.; El Belghiti, M.A. Insecticidal effect of Mentha pulegium L. and Mentha suaveolens Ehrh. hydrosols against a pest of citrus, Toxoptera aurantii (Aphididae). Res. Chem. Intermed. 2016, 42, 1639–1649. [Google Scholar] [CrossRef]
- Patra, J.; Kim, E.; Oh, K.; Kim, H.; Kim, Y.; Baek, K. Antibacterial effect of crude extract and metabolites of Phytolacca americana on pathogens responsible for periodontal inflammatory diseases and dental caries. BMC Complement. Altern. Med. 2014, 14, 343. [Google Scholar] [CrossRef] [PubMed]
- Zheleva-Dimitrova, D.Z. Antioxidant and acetylcholinesterase inhibition properties of Amorpha fruticosa L. and Phytolacca americana L. Pharmacogn. Mag. 2013, 9, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Lopes, A.I.F.; Pereira, D.M.; Gonçalves, M.S.T.; Fortes, A.G.; Castanheira, E.M.S. Valorization of Plant Extracts by Encapsulation in Lipid Nanosystems for Application as Potential Insecticides. Proceedings 2019, 41, 6616. [Google Scholar] [CrossRef]
- Jamil, B.; Abbasi, R.; Abbasi, S.; Imran, M.; Khan, S.; Ihsan, A.; Javed, S.; Bokhari, H.; Imran, M. Encapsulation of Cardamom Essential Oil in Chitosan Nano-composites: In-vitro Efficacy on Antibiotic-Resistant Bacterial Pathogens and Cytotoxicity Studies. Front. Microbiol. 2016, 7, 1580. [Google Scholar] [CrossRef] [PubMed]
- El Asbahani, A.; Miladi, K.; Badri, W.; Sala, M.; Aït Addi, E.H.; Casabianca, H.; El Mousadik, A.; Hartmann, D.; Jilale, A.; Renaud, F.N.; et al. Essential oils: From extraction to encapsulation. Int. J. Pharm. 2015, 483, 220–243. [Google Scholar] [CrossRef] [PubMed]
- Ghayempour, S.; Montazer, M. Micro/nanoencapsulation of essential oils and fragrances: Focus on perfumed, antimicrobial, mosquito-repellent and medical textiles. J. Microencapsul. 2016, 33, 497–510. [Google Scholar] [CrossRef] [PubMed]
- Peters, S.E.; Brain, C.H. Benefits of a soy lecithin based nanotechnology for the animal and human food industry. ACS Symp. Ser. 2009, 1007, 183–197. [Google Scholar] [CrossRef]
- Jaafar-Maalej, C.; Diab, R.; Andrieu, V.; Elaissari, A.; Fessi, H. Ethanol injection method for hydrophilic and lipophilic drug-loaded liposome preparation. J. Liposome Res. 2010, 20, 228–243. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H. Thin-film hydration followed by extrusion method for liposome preparation. Methods Mol. Biol. 2017, 1522, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, N.; Dilbaghi, N.; Gopal, M.; Kumar, R.; Kim, K.H.; Kumar, S. Development of chitosan nanocapsules for the controlled release of hexaconazole. Int. J. Biol. Macromol. 2017, 97, 616–624. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).