Abstract
Background and objectives: Anterior cruciate ligament reconstruction (ACLR) is often associated with significant postoperative pain. Effective pain control is vital for early mobilization and reducing opioid use. While femoral nerve block (FNB) and adductor canal block (ACB) are common, they can cause motor weakness and incomplete analgesia. The genicular nerve block (GNB), typically used for chronic knee pain and arthroplasty, may offer a motor-sparing alternative for ACLR pain management. This review evaluates the evidence on GNB’s effectiveness for pain control, opioid reduction, and recovery after ACLR. Materials and Methods: A literature search (January 2014–May 2025) identified five studies involving adult ACLR patients receiving GNB. Data on demographics, techniques, pain scores, opioid use, and complications were analyzed. Results: Among 115 patients, GNB provided effective analgesia and reduced opioid needs. Randomized trials showed GNB was comparable to ACB and more effective when combined. Ultrasound, especially with Doppler, enhances precision and safety. No major motor deficits or adverse events were noted. Landmark-based approaches also showed utility in low-resource settings. Conclusions: GNB is a promising, motor-sparing option for postoperative pain in ACLR. Further high-quality trials are needed to confirm the benefits and standardize its use. The findings should be interpreted with caution, as the current evidence is of limited quality and lacks generalizability.
1. Introduction
The anterior cruciate ligament (ACL) plays a crucial role in maintaining the stability of the tibiofemoral joint, particularly in controlling anterior translation and rotational movements. ACL injuries commonly result from pivoting activities, and knees with ACL deficiency are predisposed to instability, pain, and long-term degenerative changes [1]. Among knee sports injuries, ACL rupture is the most frequent. In the United States alone, the annual incidence has reached approximately 200,000 cases and continues to rise [2].
ACL reconstruction (ACLR) appears to be a safe procedure, with a reported success rate of at least 95%. However, postoperative pain remains one of the most frequent complications, contributing to delayed discharge and increased healthcare costs [3]. Despite extensive research, the optimal pain management strategy after ACLR is still under debate. Commonly employed approaches include opioid and multimodal pharmacologic therapy, local infiltration analgesia (LIA), cryotherapy, compression, and femoral nerve block (FNB) [4,5].
LIA can be inherently painful, as the needle must traverse multiple sensitized tissues, including the synovium, fat pad, and joint capsule [6,7]. Following ACLR, tissue injury triggers the release of chemical mediators such as substance P and vasoactive intestinal peptide, which sensitize local nociceptors and contribute to postoperative pain [8,9].
Among analgesic strategies, FNB has been widely used. In addition to reducing pain, FNB shortens postoperative immobilization and lowers early postoperative opioid requirements [10,11]. Nevertheless, it carries risks such as quadriceps weakness, delayed mobilization, vascular or nerve injuries, and insufficient coverage during hamstring autograft harvest. FNB is also associated with an increased risk of falls due to isokinetic deficits in knee flexion and extension and requires preoperative time and additional equipment, such as ultrasonography [12,13,14,15].
In this regard, adductor canal block (ACB) has emerged as an alternative to FNB, providing similar analgesia while sparing the motor branch of the quadriceps [7]. However, recent studies indicate that ACB may still reduce knee flexor strength [16]. A direct comparison by Lynch et al. found that FNB and ACB provide comparable pain control in patients undergoing ACLR [6].
For chronic knee pain, ultrasound-guided (US-guided) genicular nerve block (GNB) and radiofrequency ablation have shown promising results [16,17,18,19]. The genicular nerves, which include the superior medial, middle, superior lateral, inferior medial, inferior lateral, and recurrent peroneal branches, are the primary sensory nerves of the knee joint. Compared with ACB, GNB offers advantages such as early ambulation and faster discharge, as it selectively blocks articular sensory branches while sparing motor function [16,17,20,21].
In patients with osteoarthritis (OA) experiencing acute or chronic knee pain, US-guided GNB is recommended. Color Doppler ultrasonography is used to identify the genicular arteries and prevent vascular injury, while the genicular nerves appear as a honeycomb structure. The procedure involves infiltrating a corticosteroid mixture around the nerves [22].
Although GNB has demonstrated efficacy in total knee arthroplasty (TKA), its role in arthroscopic knee surgery, including ACLR, remains underexplored. Although arthroscopic procedures are less invasive than TKA, tissue manipulation still occurs, and further research is needed to determine whether GNB improves postoperative pain control, reduces opioid use, and enhances functional recovery in this context.
Therefore, this review aims to evaluate the available literature on the clinical outcomes of genicular nerve block (GNB) for pain management in anterior cruciate ligament reconstruction (ACLR), focusing on its effectiveness in reducing postoperative pain, improving early mobilization, and decreasing opioid consumption.
2. Results
A comparative analysis of the five included studies highlights considerable variability in both the GNB administration protocols and the reported postoperative outcomes (Table 1). The results were presented narratively rather than through a quantitative meta-analysis due to the methodological heterogeneity of the studies and the limited availability of quantitative data. More information regarding the retrieved data from available literature can be seen in Table S1 from the Supplementary Materials.

Table 1.
Studies included in the review.
2.1. Sociodemographic Data
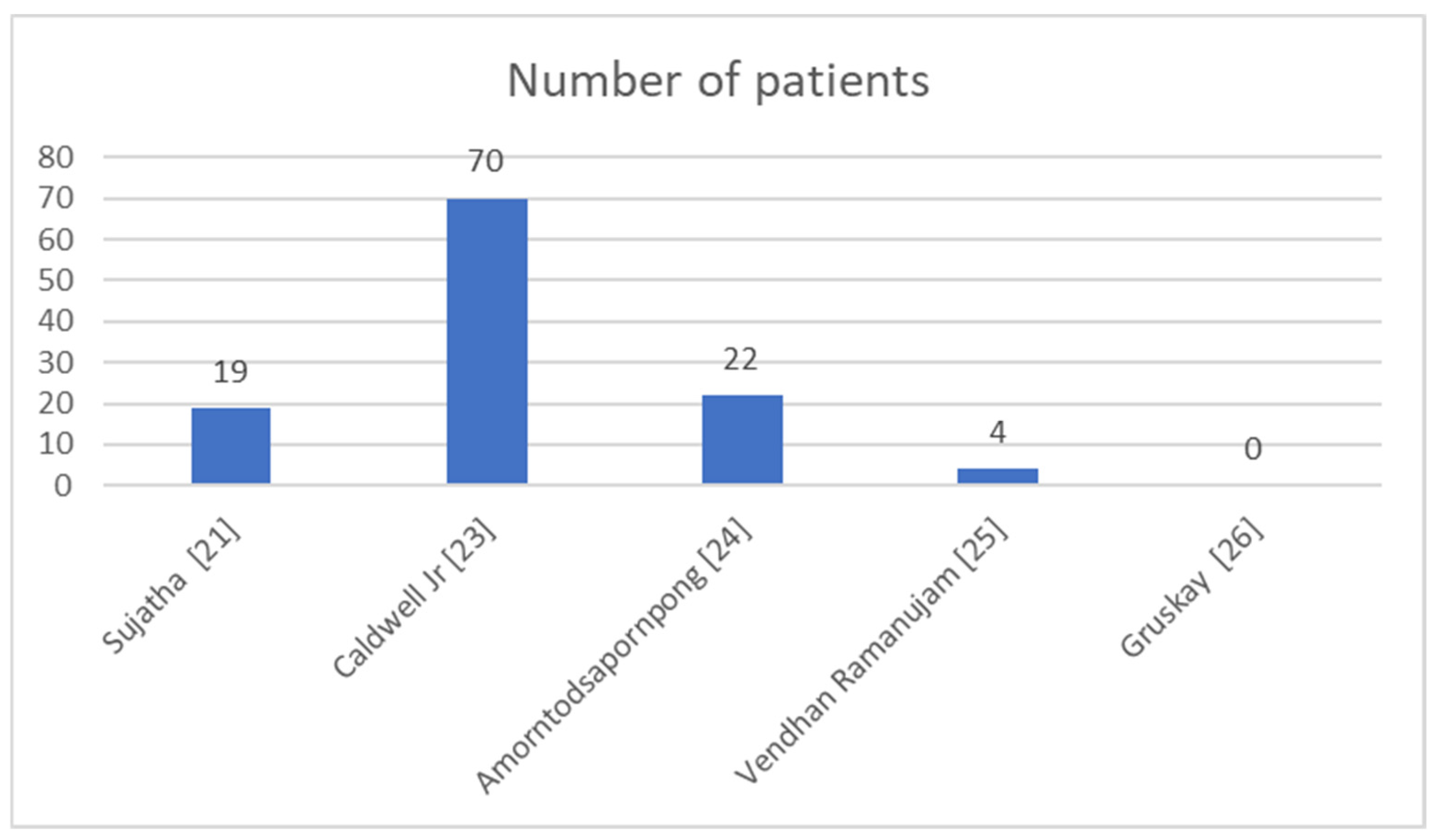
Of the five studies included in the review, four explicitly reported the group size, totaling 115 patients. The number of participants varied considerably between studies, from as few as 4 patients to as many as 70 patients (Figure 1). Thus, the study by Caldwell, Jr. et al. [23] had the largest cohort (n = 70), followed by Amorntodsapornpong et al. [24], n = 22, and Sujatha et al. [21] included 19 patients. Ramanujam et al. [25] included only four patients, while Gruskay et al. [26] did not report the number of participants or key clinical outcomes, limiting their contribution to the comparative analysis.
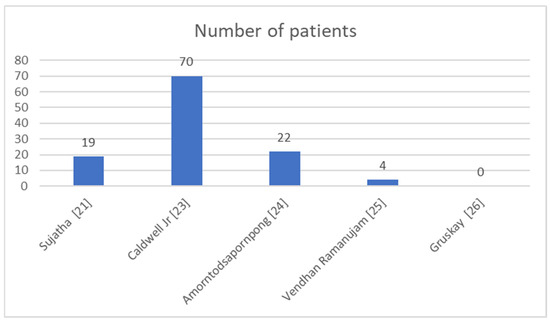
Figure 1.
Patients included in the studies [21,23,24,25,26].
The age of the participants ranged from 26 to 42 years, and body mass index (BMI) values were generally within the normal range (22.7–24.2 kg/m2), suggesting a relatively homogeneous demographic profile.
2.2. Postoperative Pain Management
In terms of postoperative pain intensity, the study by Sujatha et al. [21] provided specific numerical data on the Numeric Rating Scale (NRS), reporting scores of 1.0 at rest and during physical activity at 24 h postoperatively. These results indicate a notable analgesic effect of GNB, compared to that of the ACB. Amorntodsapornpong et al. and Ramanujam et al. [24] also evaluated pain using the Visual Analogue Scale (VAS) or numeric pain rating scores (NRS).
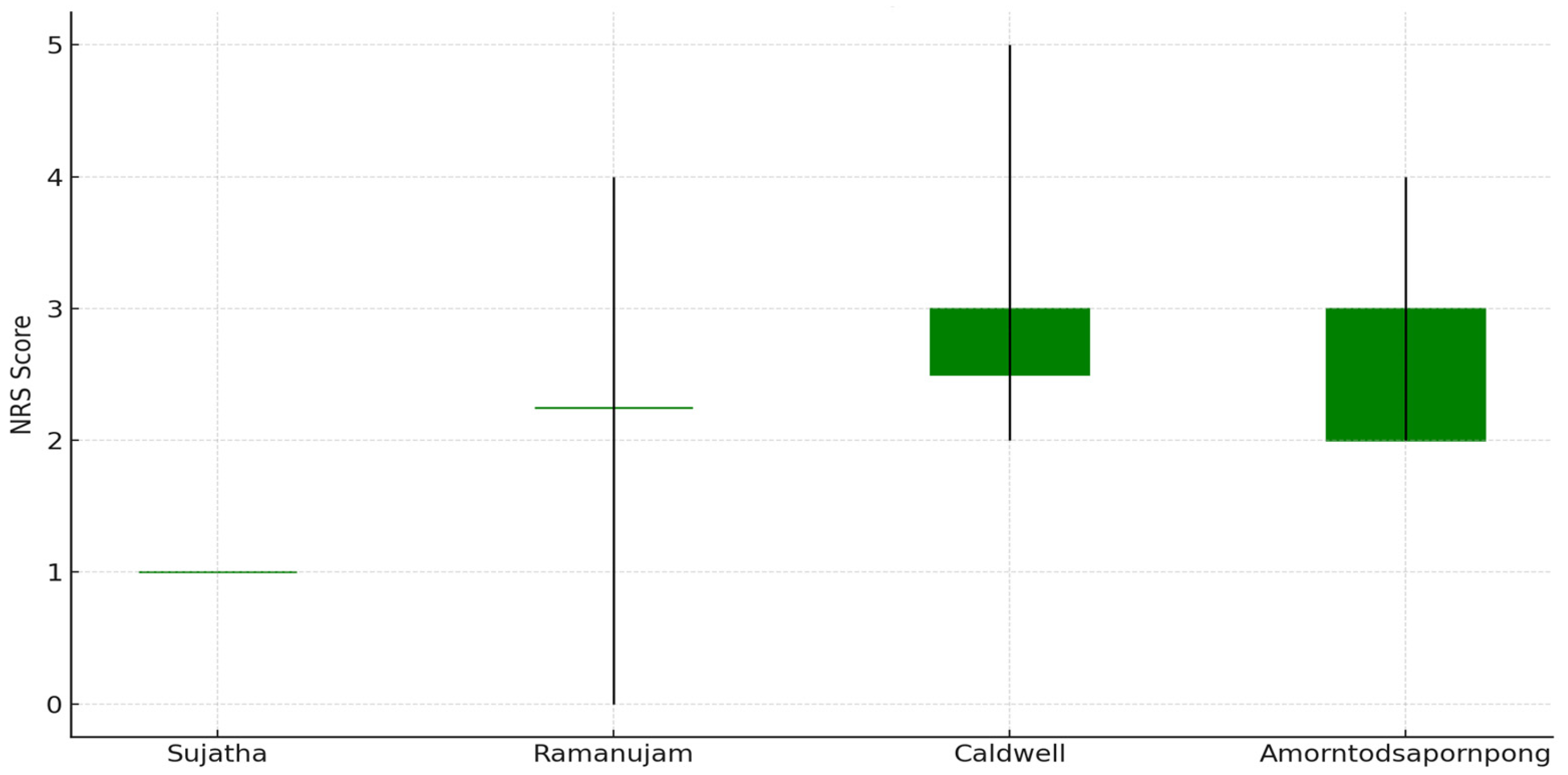
Figure 2 represents a candlestick chart illustrating the range and trend of postoperative NRS reported or estimated across four of the included studies. Each ‘candle’ displays the minimum and maximum pain scores (wicks) and the estimated or reported values for early and late postoperative pain (bodies).
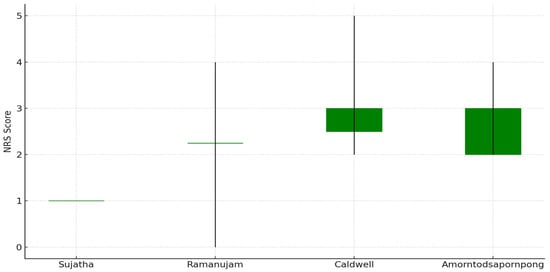
Figure 2.
Pain rating scale of the studies included in the review [21,23,24,25,26].
Colors indicate change: green for stable/increasing scores, red for decreasing scores (none observed here). Gruskay’s study [26] was excluded due to a lack of pain data. This chart supports the visual comparison of pain management effectiveness between techniques reported in the literature.
- -
- Sujatha: Constant score (1.0) throughout.
- -
- Ramanujam: Reported range 0–4, mean 2.25.
- -
- Caldwell: not reported.
- -
- Amorntodsapornpong: not reported.
2.3. Postoperative Analgesic Consumption
Regarding postoperative analgesic consumption, Caldwell, Jr. et al. reported the most substantial reduction, with 90% of patients discontinuing opioid use within seven days post-surgery following an anterior circumferential genicular nerve block performed without imaging guidance. Ramanujam et al. reported a morphine equivalent consumption of 2.5–7.5 mg at 24 h postoperatively, whereas Sujatha et al. found comparable analgesic use between the GNB and ACB groups, supporting the hypothesis that GNB can serve as an effective alternative.
2.4. Imaging Guidance
Concerning technique and imaging guidance, the majority of studies (Sujatha, Amorntodsapornpong, Ramanujam) employed ultrasound guidance for GNB administration. Notably, Sujatha et al. also used color Doppler to improve vascular identification. In contrast, Caldwell, Jr. et al. conducted the blocks without imaging assistance, which may influence accuracy and reproducibility.
With respect to the type of graft used in ACLR, only Caldwell, Jr. et al. differentiated between autografts (n = 48) and allografts (n = 22), while other studies either did not report graft type or relied exclusively on allografts (e.g., Ramanujam).
3. Discussion
The five studies included in this review collectively explored the role of genicular nerve blocks (GNBs) in improving postoperative analgesia and reducing opioid consumption following anterior cruciate ligament reconstruction (ACLR). These studies employed a variety of approaches, with GNB used either as a standalone technique, in combination with adductor canal block (ACB), or as part of a surgeon-administered intraoperative strategy. Although the methodologies were heterogeneous, several consistent and clinically relevant findings emerged across the literature.
In the randomized controlled trial by Sujatha et al., GNB was directly compared with ACB. Both interventions demonstrated similar postoperative pain scores and opioid consumption within the first 24 h, and the mean time to the first analgesic request was comparable (approximately 12–14 h). These results indicate that GNB provides an analgesic effect and duration equivalent to ACB while preserving motor function, suggesting its potential as a viable alternative when ACB is contraindicated or unavailable [21].
Amorntodsapornpong et al. conducted a randomized trial assessing the combined use of GNB and ACB. Although visual analogue scale (VAS) pain scores were not significantly different between groups, the GNB + ACB group exhibited a marked reduction in fentanyl consumption at 12, 24, and 48 h postoperatively (p ≤ 0.001). This finding indicates an opioid-sparing advantage of GNB when used as an adjunct to ACB, possibly due to improved blockade of posterior and capsular nociceptive fibers that are not adequately targeted by ACB alone [24].
In an observational study, Caldwell et al. evaluated a landmark-based, intraoperative GNB performed by the surgeon without ultrasound guidance. The results demonstrated notably low postoperative opioid use, averaging 5.5 hydrocodone tablets over 2.6 days, with 21% of patients requiring no opioids and 90% discontinuing them by postoperative day eight. No adverse events or delayed recoveries were reported. These results underscore the practicality, safety, and efficacy of a simplified, surgeon-delivered GNB approach in opioid-naïve ACLR patients [23].
Similarly, the case series by Ramanujam et al. reported favorable outcomes with ultrasound-guided GNB targeting the superomedial, superolateral, and inferomedial genicular nerves. Patients demonstrated low postoperative pain scores (NRS 0–4) in the post-anesthesia care unit (PACU) and required minimal opioid doses (2.5–7.5 MME). No motor deficits or complications were observed, and all patients were discharged on the same day. When analyzed by graft type, autograft recipients exhibited slightly higher pain and opioid use than allograft recipients, likely due to additional donor site morbidity; however, the analgesic efficacy of the block remained consistent across both groups [25].
Gruskay et al. described a technical modification involving a surgeon-administered anterolateral GNB designed to supplement ACB coverage by targeting the lateral femoral condyle and anterior capsule through infiltration near the vastus lateralis and lateral retinacular nerve. Although outcome data were not reported, the anatomical rationale and ease of intraoperative administration support its potential utility, particularly for addressing sensory territories that ACB may not adequately cover [26].
In addition, a recent literature review highlights that graft selection in ACL reconstruction should be individualized and integrated within the overall clinical strategy rather than viewed as a standalone technical choice. Bone–patellar tendon–bone grafts continue to offer excellent biomechanical stability and low revision rates, particularly in high-demand athletes, but are limited by donor-site morbidity and sport-specific constraints. Hamstring tendon autografts provide reliable outcomes with reduced anterior knee pain, yet concerns remain regarding graft size variability, flexor strength deficits, and reinjury risk. The growing evidence supporting quadriceps tendon autografts suggests they represent a balanced alternative, combining favorable biomechanical properties with lower donor-site morbidity. Emerging graft options, such as the peroneus longus tendon, appear promising in selected cases but require further long-term validation. Overall, optimal outcomes depend not only on graft choice but also on surgical technique, fixation, infection prevention, and individualized rehabilitation and return-to-sport strategies [27].
Moreover, it has been shown that antigravity treadmill training represents a promising adjunct to postoperative rehabilitation after knee surgery, particularly in patients with early weight-bearing restrictions following procedures such as ACL reconstruction and knee arthroplasty. The currently available evidence suggests potential benefits in facilitating early gait training while minimizing joint loading; however, the limited number of studies and their generally low methodological quality preclude definitive conclusions regarding superiority over conventional physiotherapy. As highlighted by existing reviews, further high-quality randomized controlled trials are necessary to clarify clinical effectiveness, optimal indications, and long-term outcomes, as well as to evaluate the cost-effectiveness of antigravity treadmill training within standard postoperative rehabilitation protocols [28].
Collectively, these studies somehow support the use of GNB as a safe, motor-sparing, and clinically effective component of multimodal analgesia in ACLR. The existing evidence may suggest that GNB—especially when performed under ultrasound guidance using a standardized technique—can appear to meaningfully reduce postoperative pain and opioid requirements. GNB alone appears comparable in efficacy to ACB, while the combination of GNB and ACB provides superior opioid-sparing effects. Both ultrasound-guided and landmark-based GNBs have demonstrated feasibility in outpatient and resource-limited settings, with minimal risk of motor impairment or delayed recovery.
However, the conclusions drawn from this review should be interpreted with caution. The small sample size—only five studies, comprising a total of 115 patients—substantially limits the strength of the evidence. Moreover, methodological heterogeneity, variations in block techniques, differences in perioperative analgesic regimens, and inconsistent outcome reporting restrict direct comparisons across studies and reduce the generalizability of findings. The lack of blinding in most studies further introduces potential bias.
Additionally, direct comparisons between GNB and other established analgesic modalities such as ACB, femoral nerve block (FNB), and local infiltration analgesia (LIA) remain scarce. Further high-quality randomized controlled trials are needed to establish the relative efficacy, safety, and long-term functional outcomes associated with GNB in ACLR. Future research should prioritize standardized protocols for block administration, consistent outcome measures, and extended follow-up to assess the durability of pain control, rehabilitation progress, and overall patient satisfaction.
4. Conclusions
Based on the currently available literature, genicular nerve block (GNB) has been investigated as a motor-sparing analgesic technique for anterior cruciate ligament reconstruction (ACLR); however, the certainty of evidence supporting its safety and effectiveness remains low. Existing studies suggest that GNB may provide postoperative analgesia comparable to adductor canal block (ACB) and may be associated with reduced opioid consumption when used as an adjunct, but these findings are derived from small, methodologically heterogeneous studies with limited external validity. Both ultrasound-guided and landmark-based approaches have been reported as feasible, with few complications, although these observations cannot be considered definitive. Consequently, conclusions regarding the clinical efficacy and safety of GNB should be interpreted with caution. Adequately powered, high-quality randomized controlled trials are required to confirm these preliminary findings, standardize technical approaches, and more clearly define the role of GNB within multimodal analgesic strategies for ACLR.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/surgeries7010017/s1, Table S1: Studies included in the review.
Author Contributions
Conceptualization, S.S. and J.M.P.J.; methodology, S.S.; software, D.N.; validation, S.F. and J.M.P.; formal analysis, D.N.; investigation, S.S.; resources, J.M.P.J.; data curation, D.N.; writing—original draft preparation, S.S.; writing—review and editing, S.F.; visualization, J.M.P.J.; supervision, J.M.P.; project administration, S.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by internal funding of the “Victor Babes” University of Medicine and Pharmacy, Eftimie Murgu Square 2, 300041 Timisoara, Romania.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new patient data were collected and therefore, ethics approval and informed consent were not required for this review article.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Domnick, C.; Raschke, M.J.; Herbort, M. Biomechanics of the anterior cruciate ligament: Physiology, rupture and reconstruction techniques. World J. Orthop. 2016, 7, 82–93. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.K.; Lee, D.K.; Shin, D.W.; Yum, T.H.; Kim, J.-H. Local infiltration analgesia versus femoral nerve block for pain control in anterior cruciate ligament reconstruction: A systematic review with meta-analysis. Orthop. J. Sports Med. 2021, 9, 23259671211050616. [Google Scholar] [CrossRef] [PubMed]
- Deviandri, R.; Yuliana, V.; Irawan, D.; Rahman, A. Genicular nerve radiofrequency ablation for pain control following anterior cruciate ligament reconstruction—A case report. Trauma Case Rep. 2022, 40, 100661. [Google Scholar] [CrossRef] [PubMed]
- Terai, S.; Zantop, C.; Zantop, T. The effect of combined cryotherapy/compression versus cryotherapy following ACL reconstruction in return-to-sport after 3 months. Orthop. J. Sports Med. 2020, 8, 2325967120S2325900548. [Google Scholar]
- Secrist, E.S.; Freedman, K.B.; Ciccotti, M.G.; Mazur, D.W.; Hammoud, S. Pain management after outpatient anterior cruciate ligament reconstruction: A systematic review of randomized controlled trials. Am. J. Sports Med. 2016, 44, 2435–2447. [Google Scholar] [CrossRef]
- Lynch, J.R.; Okoroha, K.R.; Lizzio, V.; Yu, C.C.; Jildeh, T.R.; Moutzouros, V. Adductor canal block versus femoral nerve block for pain control after anterior cruciate ligament reconstruction: A prospective randomized trial. Am. J. Sports Med. 2019, 47, 355–363. [Google Scholar] [CrossRef]
- Stebler, K.; Martin, R.; Kirkham, K.R.; Lambert, J.; De Sede, A.; Albrecht, E. Adductor canal block versus local infiltration analgesia for postoperative pain after anterior cruciate ligament reconstruction: A single-centre randomized controlled triple-blinded trial. Br. J. Anaesth. 2019, 123, e343–e349. [Google Scholar] [CrossRef]
- Hong, A.J.; Agarwalla, A.; Liu, J.N.; Gowd, A.K.; McMillan, S.; Sethi, P.M.; Amin, N.H. Neurological structures and mediators of pain sensation in anterior cruciate ligament reconstruction. Ann. Anat. 2019, 225, 28–32. [Google Scholar] [CrossRef]
- Fernandez, C.F.; Angeles, B.S. Comparison of analgesic efficiency between local infiltration of a long-acting analgesic and regional nerve block among patients undergoing arthroscopic anterior cruciate ligament reconstruction: Meta-analysis of randomized controlled trials. Clin. Orthop. Surg. 2025, 17, 228–237. [Google Scholar] [CrossRef]
- Choi, S.; McCartney, C.J.; van der Vyver, M. Femoral nerve block does provide significant analgesia after anterior cruciate ligament reconstruction. Arthroscopy 2010, 26, 1416–1417. [Google Scholar]
- Kurosaka, K.; Tsukada, S.; Nakayama, H.; Iseki, T.; Kanto, R.; Sugama, R.; Yoshiya, S. Periarticular injection versus femoral nerve block for pain relief after anterior cruciate ligament reconstruction: A randomized controlled trial. Arthroscopy 2018, 34, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Bushnell, B.D.; Sakryd, G.; Noonan, T.J. Hamstring donor-site block: Evaluation of pain control after anterior cruciate ligament reconstruction. Arthroscopy 2010, 26, 894–900. [Google Scholar] [CrossRef] [PubMed]
- Luo, T.D.; Ashraf, A.; Dahm, D.L.; Stuart, M.J.; McIntosh, A.L. Femoral nerve block is associated with persistent strength deficits at six months after anterior cruciate ligament reconstruction in pediatric and adolescent patients. Am. J. Sports Med. 2015, 43, 331–336. [Google Scholar] [CrossRef]
- LaReau, J.M.; Robbins, C.E.; Talmo, C.T.; Mehio, A.K.; Puri, L.; Bono, J.V. Complications of femoral nerve blockade in total knee arthroplasty and strategies to reduce patient risk. J. Arthroplasty 2012, 27, 564–568. [Google Scholar] [CrossRef]
- Widmer, B.; Lustig, S.; Scholes, C.J.; Molloy, A.; Leo, S.P.; Coolican, M.R.; Parker, D.A. Incidence and severity of complications due to femoral nerve blocks performed for knee surgery. Knee 2013, 20, 181–185. [Google Scholar] [CrossRef]
- Sengoku, T.; Nakase, J.; Mizuno, Y.; Yoshimizu, R.; Kanayama, T.; Yanatori, Y.; Tsuchiya, H. Outcome comparison of femoral nerve block and adductor canal block during anterior cruciate ligament reconstruction: Adductor canal block may cause an unexpected decrease in knee flexor strength at six months postoperatively. Arch. Orthop. Trauma Surg. 2023, 143, 6305–6313. [Google Scholar] [CrossRef]
- Sotelo, V.G.; Maculé, F.; Minguell, J.; Bergé, R.; Franco, C.; Sala-Blanch, X. Ultrasound-guided genicular nerve block for pain control after total knee replacement: Preliminary case series and technical note. Rev. Esp. Anestesiol. Reanim. 2017, 64, 568–576. [Google Scholar]
- Yasar, E.; Kesikburun, S.; Kılıç, C.; Güzelküçük, Ü.; Yazar, F.; Tan, A.K. Accuracy of ultrasound-guided genicular nerve block: A cadaveric study. Pain Physician 2015, 18, E899–E904. [Google Scholar] [CrossRef]
- Sahoo, R.K.; Krishna, C.; Kumar, M.; Nair, A.S. Genicular nerve block for postoperative pain relief after total knee replacement. Saudi J. Anaesth. 2020, 14, 235–237. [Google Scholar] [CrossRef]
- Cuñat, T.; Mejía, J.; Tatjer, I.; Comino, O.; Nuevo-Gayoso, M.; Martín, N.; Tió, M.; Basora, M.; Sala-Blanch, X. Ultrasound-guided genicular nerves block vs. local infiltration analgesia for total knee arthroplasty: A randomized controlled non-inferiority trial. Anaesthesia 2023, 78, 188–196. [Google Scholar] [CrossRef]
- Sujatha, S.S.N.; Gupta, K.; Guria, S.; Chhabra, P.H. Comparison of genicular nerve block with adductor canal block for postoperative pain management in patients undergoing arthroscopic knee ligament reconstruction: A randomized controlled trial. Indian J. Anaesth. 2024, 68, 454–459. [Google Scholar] [CrossRef] [PubMed]
- Henry, S.; Best, T.M.; Jose, J.; Tiu, T. Procedural approach to ultrasound-guided geniculate nerve blockade for knee pain in patients with osteoarthritis. Curr. Sports Med. Rep. 2022, 21, 192–195. [Google Scholar]
- Caldwell, G.L., Jr.; Selepec, M.A. Reduced opioid use after surgeon-administered genicular nerve block for anterior cruciate ligament reconstruction in adults and adolescents. HSS J. 2019, 15, 42–50. [Google Scholar] [PubMed]
- Amorntodsapornpong, P.; Yuwapattanawong, K.; Sakura, S. Effects of genicular nerve blocks in combination with an adductor canal block in patients undergoing arthroscopic knee surgery: A randomized controlled trial. Cureus 2025, 17, e79215. [Google Scholar] [CrossRef]
- Ramanujam, V.; DiMaria, S.; Varma, V. Ultrasound-guided genicular nerve blocks for anterior cruciate ligament reconstruction surgery in an outpatient setting: A case series. Cureus 2023, 15, e44550. [Google Scholar] [CrossRef] [PubMed]
- Gruskay, J.A.; Pearce, S.S.; Ruttum, D.; Conrad, E.S.; Hackett, T.R. Surgeon-administered anterolateral geniculate nerve block as an adjunct to regional anesthetic for pain management following anterior cruciate ligament reconstruction. Arthrosc. Tech. 2022, 11, e1–e6. [Google Scholar] [CrossRef]
- Ostojic, M.; Indelli, P.F.; Lovrekovic, B.; Volcarenghi, J.; Juric, D.; Hakam, H.T.; Salzmann, M.; Ramadanov, N.; Królikowska, A.; Becker, R.; et al. Graft Selection in Anterior Cruciate Ligament Reconstruction: A Comprehensive Review of Current Trends. Medicina 2024, 60, 2090. [Google Scholar] [CrossRef]
- Hakam, H.T.; Kentel, M.; Kowal, M.; Królikowska, A.; Reichert, P.; Daszkiewicz, M.; Kentel, M.; Becker, R.; Ramadanov, N.; Prill, R. Antigravity treadmill training after knee surgery: A scoping review. Adv. Clin. Exp. Med. 2025, 34, 1011–1024. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.