Simple Summary
This study collected clinical data on 278 pancreatic cancer patients who underwent pancreatic resection and compared the patient-reported outcomes in 128 patients following pancreatic resection. We observed a survival difference between malignancy, pre-malignancy, and benign disease groups. Mental component scores seem to improve over time, whereas no changes were observed in the physical component scores.
Abstract
The aims of this study were to assess patient-reported outcomes and the survival of patients following curative resection for pancreas cancer. Adult patients undergoing curative pancreatic resection between April 2014 and April 2019 across six major hospitals in Sydney were invited to complete the Short-Form 36 (SF-36v2) and the Functional Assessment Cancer Therapy—Hepatobiliary (FACT-Hep) questionnaires. Time from surgery was categorised into four different time points: 3–11, 12–23, 24–35, and 36–62 months. Survival analyses were performed using Kaplan–Meier and log-rank tests. A total of 278 patients underwent curative resection. Mean (SD) age was 65.0 (13.2), and 50.7% (n = 141) were males. Out of the 205 (74%) alive patients, 128 (62%) completed the study surveys. The physical component score and total FACT-Hep scores showed no significant changes over time. The mental component score improved from 3–11 months to 12–23 months (p = 0.009) and from 3–11 months to 36–62 months (p = 0.007). Survivorship showed a significant difference between malignancy, pre-malignancy, and benign disease groups, with 45.8 months (95%CI: 42.4–49.1), 40.3 months (95%CI: 36.4–44.2), and 41.3 months (95%CI: 37.9–44.9), respectively. For patients undergoing curative resection for pancreatic cancer, mental component scores improved over time, whereas overall survival outcomes seem to be influenced according to cancer pathology.
1. Introduction
Pancreatic cancer ranks among the lowest survival rates of all cancers, with the 5-year survival in Australia at 11% and 9% worldwide [1,2]. This survival rate is due, in part, to the asymptomatic nature of the disease leading to a diagnosis of pancreatic cancer found at the locally advanced or metastatic stage, where a curative approach is not possible [2]. Patients who can undergo chemotherapy following a diagnosis of metastatic disease have a median survival of 12 months. Patients receiving best supportive care have a median survival of less than 6 months [3]. Patients undergoing curative resection have a median survival of 20–28 months [3].
Surgical intervention is the only potentially curative treatment for resectable pancreatic cancer. Treatment options include the following: pancreaticoduodenectomy (Whipple’s) or pancreatectomy (partial or total), with the surgery type dependent on the cancer type (adenocarcinoma or neuroendocrine tumour) and tumour location [4]. Despite resection being the only curative treatment, these operations are highly morbid with potentially lifelong problems such as delayed gastric emptying, malabsorption, and diabetes [4]. Palliative surgical treatments are sometimes employed for patients who have unresectable or metastatic disease to relieve jaundice or gastric outlet obstruction, although endoscopic options are increasingly utilised to improve the patients’ quality of life (QOL) [4].
The necessity of QOL measures has increased recently, as patients now often look to QOL research when deciding on treatment [5]. Improvements in QOL have allowed for the assessment of treatment effects, with general instruments able to assess effects beyond symptom relief, generally measured with disease-specific instruments [5]. Many studies investigating QOL in pancreatic cancer surgery use these measures to compare the success of different treatment options, with a systematic review published in 2010 looking into the two types of pancreaticoduodenectomies (pylorus-preserving or classic) [6]. Braga et al. (2015) investigated the difference in distal pancreatectomy method (open vs laparoscopic) using QOL as measure of treatment success [7]. In another study, Casadei et al. (2010) used QOL to measure treatment success on two different cancer types (ductal adenocarcinoma vs non-ductal adenocarcinoma) [8]. Prospective QOL research in this group is limited, with only a few studies looking exclusively at the long-term QOL. This study aimed to provide insight into the survival and QOL trajectories of pancreatic cancer patients undergoing curative resection. The main aim of this study was to examine QOL, pain, distress, and survival outcomes after pancreatic resection for benign or malignant disease.
2. Results
2.1. Characteristics of the Included Participants
The baseline demographics of the entire study population (N = 278) are described in Table 1. Overall, the patients’ mean age was 65 years, with an even distribution between males (50.7%) and females (49.3%). The most common procedures undertaken were pancreaticoduodenectomy (Whipple’s) (n = 169, 60.8%), followed by pancreatectomy (distal/partial) (n = 85, 30.6%). More than half (n = 174, 62.6%) of the study population had no in-hospital post-operative complications. Of those who had complications (n = 104), 76.9% (n = 80) of patients had minor postoperative complications.

Table 1.
Demographics and surgical outcomes of included participants.
Most of the baseline characteristics were similar across the four time points, except for age (p = 0.009), deceased status (p < 0.001), cancer type (p = 0.02), and postoperative complications (p = 0.02).
2.2. Questionnaire Response Rate
Of the 278 patients who underwent curative resection, 205 patients were alive and were sent a questionnaire. Of the 205 patients that were sent a questionnaire, 128 (62.4%) patients responded and were included in the study, 22 patients opted out of the study, and 55 patients did not respond at all (Figure 1). The characteristics of responders (n = 128) and non-responders (n = 55) were similar, with no significant difference between groups, except for age (p = 0.001). Overall, most of the respondents had some form of health insurance (87.5%, n = 112), most were married or living with a partner (76.6%, n = 98), 70.3% of responders were not in paid work (n = 90), and more than half had finished high school or a university degree (30.5%, n = 39 and 33.6%, n = 43, respectively). None of the socioeconomic characteristics were significantly different across the study time points.
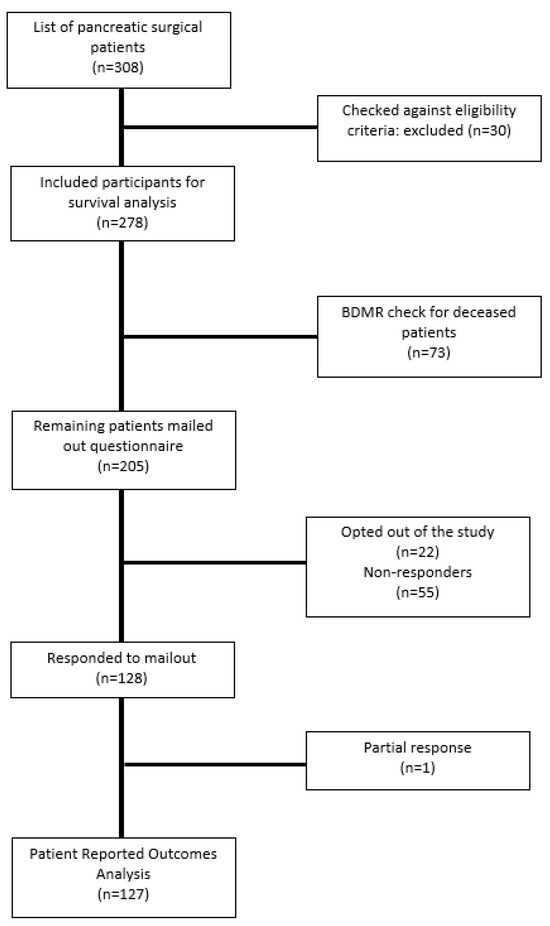
Figure 1.
Flowchart of participant inclusion.
2.3. Patient-Reported Outcomes
Out of the patients who responded to the self-reported questionnaire (n = 128), one patient returned an incomplete questionnaire. Therefore, 127 patients were reported in the patient-reported outcomes analyses.
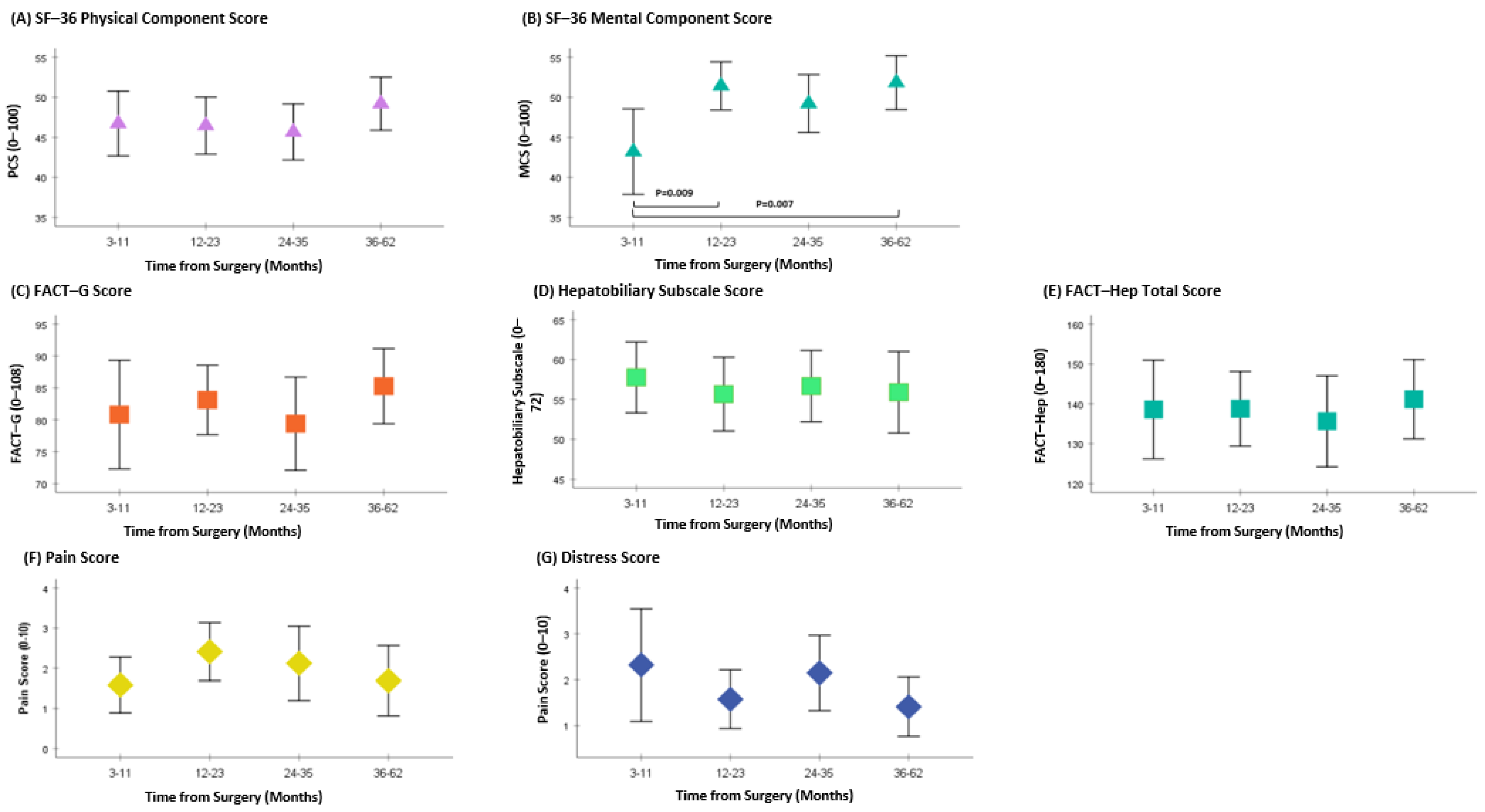
The MCS showed a significant difference between 3–11 months to 12–23 months (p = 0.009) and between 3–11 months to 36–62 months (p = 0.007) from surgery. No differences in PCS, FACT-Hep (including FACT-G and Hepatobiliary sub-scores), pain, and distress scores were found between time points (Figure 2A–G).
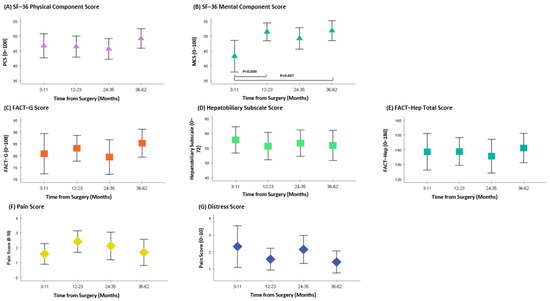
Figure 2.
Patient reported outcomes following pancreatic resection. Higher SF–36 PCS and MCS scores indicate better QOL; higher FACT–G and FACT–Hep scores indicate better QOL; higher hepatobiliary subscale scores indicate fewer symptoms; lower scores on the pain and distress scales indicate better outcomes.
2.4. Survival
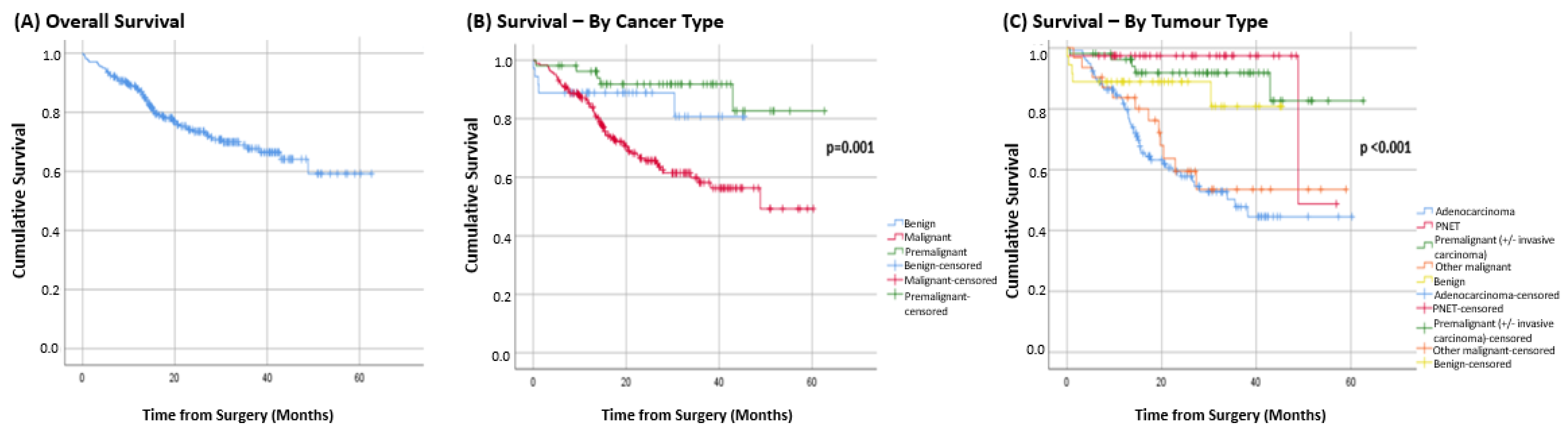
Mean overall survival was 45.8 months (95%CI = 42.4 to 49.1) (Figure 3A). When analysed by pathology type, the mean survival of malignant tumours, premalignant tumours, and benign tumours was 39.2 months (95%CI = 34.2 to 44.2), 40.3 months (95%CI = 36.4 to 44.2), and 56.5 months (95%CI = 51.2 to 61.6), respectively (p = 0.001; Figure 3B).
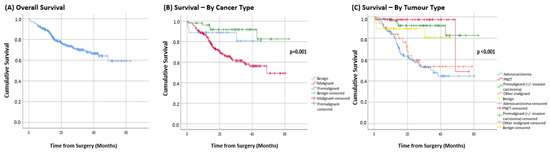
Figure 3.
Survival curves.
For patients presenting with malignant tumours, adenocarcinoma had a mean survival of 36.2 months (95%CI = 31.3 to 41.1) and pancreatic neuroendocrine tumours had a survival of 51.6 months (95%CI = 47.5 to 61.0). While the mean survival for patients presenting with other malignancies was 38.7 months (95%CI = 30.0 to 47.4) (p < 0.001) (Figure 3C).
3. Materials and Methods
3.1. Study Design and Setting
This cross-sectional study included patients who underwent pancreatic surgery in six major hospitals in Sydney, Australia. This study was approved by the Sydney Local Health District Ethics Review Board (SLHD-RPA Zone) (X21-0224-2019/STE16037).
3.2. Participants
Patients were deemed eligible if they met the following inclusion criteria: adults aged ≥18 years old, undergoing curative pancreatic resection between 1 April 2014 to 1 April 2019 for benign or malignant disease.
3.3. Clinical Data Collection
The data collected for this study were a combination of clinical records and patient-reported outcomes. Clinical information included the following: demographic (age and gender), surgical procedure performed, disease indicator (malignant, premalignant, benign), pathology type, in-hospital perioperative complications (Clavien Dindo Score: minor (I–II) and major (IIIa–V), survival outcomes, and hospital discharge destination (home, other). All clinical information was provided by the consulting surgeon from their rooms’ database and was complemented with a clinical auditing database.
3.4. Patient-Reported Outcomes Data Collection
The patient-reported outcome measures included the Short Form Version 2 (SF36v2), [9]. Functional Assessment of Cancer Therapy Hepatobiliary Cancer (FACT-Hep) [10]—which includes the Functional Assessment of Cancer Therapy General Cancer (FACT-G), Numeric Pain Rating Score (NPRS), and the Distress thermometer [11,12].
The SF-36v2 provides a general measure of physical functioning, role-physical, bodily pain, general health, vitality, social functioning, role-emotional, and mental health. These domains are ultimately summarised into 2 health component scores, which are scored 0–100: The Physical and Mental Component Scores (PCS and MCS). The FACT-G provides a general measure of physical (score range 0–28), social (score range 0–28), emotional (score range 0–24), and functional well-being (score range 0–28) in cancer patients. These scores are combined to provide the FACT-G score (score range 0–108). The FACT-Hep is the disease-specific component, items are scored on a 0–4 scale, which is combined to provide a Hepatobiliary Cancer Subscale Score with a score range 0–72. The cancer subscale score is combined with the FACT-G score to provide the Total FACT-Hep (score range 0–180). The NPRS provides a pain score on a scale of 0–10, with 0 representing no pain and 10 representing extreme pain, and the distress thermometer provides a distress score on a scale of 0–10, with 0 representing no distress and 10 representing extreme distress.
All questionnaires were scored using the associated scoring software and/or scoring formulas. The participant reported socioeconomic information (education, employment status, household demographics, GP details, and health cover), along with the QOL questionnaires.
3.5. Statistical Analysis
All data were entered into a Record Electronic Capture Application (REDCap) database. All analyses were determined prior in the study protocol by the study investigators. Descriptive statistics were used to summarise patient’s characteristics and clinical and self-reported outcomes. The time of analysis of patient-reported outcomes was categorised into four groups. These groups were based on time from surgery and collected patients by month (3–11; 12–23; 24–35; and 36–62). A comparison of the patient’s characteristics and surgical outcomes over the timepoints was conducted using chi-square tests or Fisher’s Exact Test when appropriate, and for continuous variables (age), ANOVA test was utilised. Patient-reported outcomes (PCS, MCS, FACT-G, Hepatobiliary subscale, FACT-Hep Total, distress, and pain) scores were summarised using the mean and 95% confidence intervals. Statistical significance was tested using the independent samples t-test, given that the data were normally distributed.
Overall survival was analysed using Kaplan–Meier and log-rank tests. Time from surgery was used as the censored point for the survival analysis, where the participant was either deceased or returned the questionnaire. For the non-responders, the last date seen in the surgeon’s clinical rooms was taken as the censored point.
4. Discussion
The QOL results from the SF-36 have shown an improvement in MCS between time points, with a significant difference between the 3–11-month group when compared to the 12–24-month group, and the 3–1-month group when compared to the 36–62-month group. The PCS were relatively stable over time, with all scores between 45–50. Similarly, no statistical difference was observed in the FACT-Hep total scores.
This study demonstrated that the overall survival of resected pancreatic cancer patients in this study was 45.8 months, with the pre-malignant group having a mean survival of 40.3 and those with adenocarcinoma having a mean survival of 36.2 months.
4.1. Strengths and Weaknesses of This Study
While using patient information from local surgeons at our institution, given their specialisation and practice at other institutions around Sydney, we were able to capture a large population of pancreatic resection patients mostly from Sydney, which may provide limited generalisability. Despite this, the results reflect a smaller population of pancreatic resection patients within NSW and Australia. A larger population of pancreatic surgical patients would provide a more in-depth view of the survival of surgical pancreatic resected patients.
The design for this study provides important information about participation in a QOL study and the feasibility of patients returning the questionnaire in a patient group that is usually overlooked in QOL research due to the aggressive nature of their disease and poor outcomes. An overall response rate of 62% for a mail-out study, where patients were unaware of the study prior to receiving the questionnaire, is encouraging for future studies with this population. If they were to be recruited prospectively prior to treatment, response rates might be higher. This design does have some limitations, as the population is taken at a cross-section from 1 April 2014 to 1 April 2019, and most patients were between 1–2 years following surgery. Due to this study design and questionnaire completion rate, the analyses of clinical outcomes such as the survival rate are not representative of the population who answered surveys. Therefore, the survival rate presented in this study is not representative of those who answered surveys, and the PROMS is not representative of all the patients who were included in all other analyses.
The QOL scores of this study show a stable PCS following surgery. When the PCS of this study were compared to normative data, 61.4% of the overall study population had a physical score the same or better than the average of the normal population. More than half of the study population at each of the time points was the same or better when compared to the general population (62.5% at 3–11 months, 64.7% at 12–23 months, 73.5% at 36–42 months), with the exception of 24–35 months, where only 45.7% scored the same or better than the general population.
When the MCS of this study were compared to normative data, 67.7% of the overall study population had a mental health score the same or better than the general population. The percentage of the study population who scored the same or better than general population was higher at the various time points, with a small decrease in the 24–35 months group (68.6%). In the 3–11 months group, 45.8% of the study population were the same or better than the general population, 76.5% at 12–23 months, and 73.5% at 36–42 months.
Another limitation is that the study population that underwent analysis for clinical outcomes had undergone one of four pancreatic procedures, as seen in Table 1. While different pancreatic resection types have different levels of severity, it is possible that each type might have different PROMs and survival outcomes when compared to each other.
Having a cross-section of post-surgical patients has its limitations for measuring the disease burden with the FACT-Hep; without baseline scores, it is challenging to ascertain if the procedure has caused any changes (improvement or decline) in the study population in general but also concerning disease-specific complaints. As this was a cross-sectional study, patients were only surveyed at a single point in time. Future research into the QOL of pancreatic cancer patients should strive to conduct more longitudinal studies in QOL to provide a more accurate picture of QOL after pancreatic surgery. Another limitation of this study is that of the healthy responder bias. While measures were taken to alleviate this through the recruitment of benign and premalignant cases, the healthy responder bias should be considered in the interpretation of these results. There were 55 non-responders (26.8%) to our questionnaire, and while their characteristics (gender and age) were similar to those who responded, there is still the potential for bias in our responder group.
4.2. Strengths and Weaknesses Compared to Other Studies
Our QOL results were not different to those of other cross-sectional studies and those using the same QOL instruments as this study. Our cross-sectional study findings present similar results to Cloyd et al. (2017) with respect to their FACT-G, Hepatobiliary Scale, and FACT-Hep scores after surgery [13]. The trajectory of the cross-section reported a low severity of symptoms and generally favourable QOL across domains [13]. Petzel et al. (2012) also used the FACT-Hep measure in a cross-sectional study, and their changes are similar to this study and that of Cloyd. et al. [13,14]. It is much harder to infer the effects of their disease on this change as we do not have a baseline score to compare it to.
Other studies show that mental health improves over time, and while there are many factors which could contribute to this, it is thought that after surgery, the burden of a death sentence has been removed. Yeo et al. (2012) show that the MCS improved after surgery, as do Balyaev et al., who showed that those patients who underwent resection had an improved MCS 3 months after surgery [15,16]. These studies are in line with the findings of the current study. The MCS scores reported in the current study were higher than previous studies, and this could be due, in part, to the extended time period, the study design, and, potentially, the healthy responder bias.
The hepatobiliary symptom scale is limited again because we are unable to measure this against a baseline score in this study. Patients had already undergone surgery at the time of the study; unsurprisingly, their symptoms were better and not burdensome. This illustrates the effect of surgery on the symptoms caused by pancreatic disease. Most patients seek treatment to manage symptoms such as jaundice, which improves greatly post-surgery. The hepatobiliary subscale score, overall, for Cloyd et al. (2017) was 3 points more than found in this study [13]. This is interesting to note as the population in Cloyd et al.’s study were recurrence-free survivors, and this study has no such limitation surrounding our eligibility criteria.
Further research investigating the survival of pancreatic cancer surgical patients is needed wherein variables such as stage of disease at diagnosis and the use of neoadjuvant chemoradiotherapy are explored. There is also potential to investigate the disease-free interval of pancreatic surgical patients. This type of research would also be very interesting in those patients who have survived 5 years or beyond from surgery to see if there are any characteristics which would be predictors for survival.
QOL investigations should be carried out in a prospective manner to allow for analysis of the change in QOL over time. It would be recommended that a multicentre/national study be undertaken in QOL for pancreatic cancer.
5. Conclusions
This study showed a significant increase in MCS between the 3–11 months and 12–23 months, and 3–11 months and 36–62 months, with no further significant changes in QOL outcomes observed. A significant difference in mean survival curves was observed for malignant, premalignant, and benign diseases.
Author Contributions
Conceptualization, C.T., D.S., D.Y., C.P. and C.S.; methodology, C.T., D.S., and C.S.; formal analysis, C.T. and D.S.; investigation, C.T., D.S., D.Y., C.P. and C.S.; resources, D.S. and C.S.; data curation, C.T.; writing—original draft preparation, C.T. and D.S.; writing—review and editing, C.T., D.S., J.M. and C.S.; supervision, D.S. and C.S.; project administration, C.T. and J.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no funding or grants.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by Royal Prince Alfred Hospital Research Ethics and Governance Offices (X21-0224-2019/STE16037).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study. Written informed consent has been obtained from the patient(s) to publish this paper.
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to our ethics not allowing for data to be shared.
Acknowledgments
The authors would like to acknowledge the surgeons who contributed their patients to this study. They would also like to acknowledge the staff within the Department of Upper Gastrointestinal and Hepatobiliary Surgery and the Surgical Outcomes Research Centre (SOuRCe) at Royal Prince Alfred Hospital, Australia for their support of this study.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Pancreatic Cancer Statistics. 2020. Available online: https://www.canceraustralia.gov.au/affected-cancer/cancer-types/pancreatic-cancer/pancreatic-cancer-australia-statistics (accessed on 15 November 2020).
- Ilic, M.; Ilic, I. Epidemiology of pancreatic cancer. World J. Gastroenterol. 2016, 22, 9694–9705. [Google Scholar] [CrossRef]
- Loveday, B.P.T.; Lipton, L.; Thomson, B.N. Pancreatic cancer: An update on diagnosis and management. Aust. J. Gen. Pract. 2019, 48, 826–831. [Google Scholar] [CrossRef]
- Treatment Options. 2020. Available online: https://www.canceraustralia.gov.au/cancer-types/pancreatic-cancer/treatment (accessed on 15 November 2020).
- Urbach, D.R. Measuring quality of life after surgery. Surg. Innov. 2005, 12, 161–165. [Google Scholar] [CrossRef]
- Fitzmaurice, C.; Seiler, C.M.; Buchler, M.W.; Diener, M.K. Survival, mortality and quality of life after pylorus-preserving or classical Whipple operation. A systematic review with meta-analysis. Der Chir. 2010, 81, 454–471. [Google Scholar] [CrossRef]
- Braga, M.; Pecorelli, N.; Ferrari, D.; Balzano, G.; Zuliani, W.; Castoldi, R. Results of 100 consecutive laparoscopic distal pancreatectomies: Postoperative outcome, cost-benefit analysis, and quality of life assessment. Surg. Endosc. 2015, 29, 1871–1878. [Google Scholar] [CrossRef]
- Casadei, R.; Monari, F.; Buscemi, S.; Laterza, M.; Ricci, C.; Rega, D.; D’Ambra, M.; Pezzilli, R.; Calculli, L.; Santini, D.; et al. Total pancreatectomy: Indications, operative technique, and results: A single centre experience and review of literature. Updates Surg. 2010, 62, 41–46. [Google Scholar] [CrossRef]
- Ware, J.E., Jr.; Sherbourne, C.D. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med. Care 1992, 30, 473–483. [Google Scholar] [CrossRef]
- Heffernan, N.; Cella, D.; Webster, K.; Odom, L.; Martone, M.; Passik, S.; Bookbinder, M.; Fong, Y.; Jarnagin, W.; Blumgart, L. Measuring health-related quality of life in patients with hepatobiliary cancers: The functional assessment of cancer therapy-hepatobiliary questionnaire. J. Clin. Oncol. 2002, 20, 2229–2239. [Google Scholar] [CrossRef]
- Haefeli, M.; Elfering, A. Pain assessment. Eur. Spine J. 2006, 15 (Suppl. S1), S17–S24. [Google Scholar] [CrossRef]
- Network, N.C.C. NCCN practice guidelines for the management of psychosocial distress. Oncology 1999, 13, 113–147. [Google Scholar]
- Cloyd, J.M.; Tran Cao, H.S.; Petzel, M.Q.; Denbo, J.W.; Parker, N.H.; Nogueras-González, G.M.; Liles, J.S.; Kim, M.P.; Lee, J.E.; Vauthey, J.N.; et al. Impact of pancreatectomy on long-term patient-reported symptoms and quality of life in recurrence-free survivors of pancreatic and periampullary neoplasms. J. Surg. Oncol. 2017, 115, 144–150. [Google Scholar] [CrossRef]
- Petzel, M.Q.; Parker, N.H.; Valentine, A.D.; Simard, S.; Nogueras-Gonzalez, G.M.; Lee, J.E.; Pisters, P.W.; Vauthey, J.N.; Fleming, J.B.; Katz, M.H. Fear of cancer recurrence after curative pancreatectomy: A cross-sectional study in survivors of pancreatic and periampullary tumors. Ann. Surg. Oncol. 2012, 19, 4078–4084. [Google Scholar] [CrossRef]
- Yeo, T.P.; Burrell, S.A.; Sauter, P.K.; Kennedy, E.P.; Lavu, H.; Leiby, B.E.; Yeo, C.J. A progressive postresection walking program significantly improves fatigue and health-related quality of life in pancreas and periampullary cancer patients. J. Am. Coll. Surg. 2012, 214, 463–475; discussion 475–477. [Google Scholar] [CrossRef]
- Belyaev, O.; Herzog, T.; Chromik, A.M.; Meurer, K.; Uhl, W. Early and late postoperative changes in the quality of life after pancreatic surgery. Langenbeck Arch. Surg. 2013, 398, 547–555. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).