Effective Hydrogel Surfaces for Adsorption of Pharmaceutical and Organic Pollutants—A Mini Review
Abstract
1. Introduction
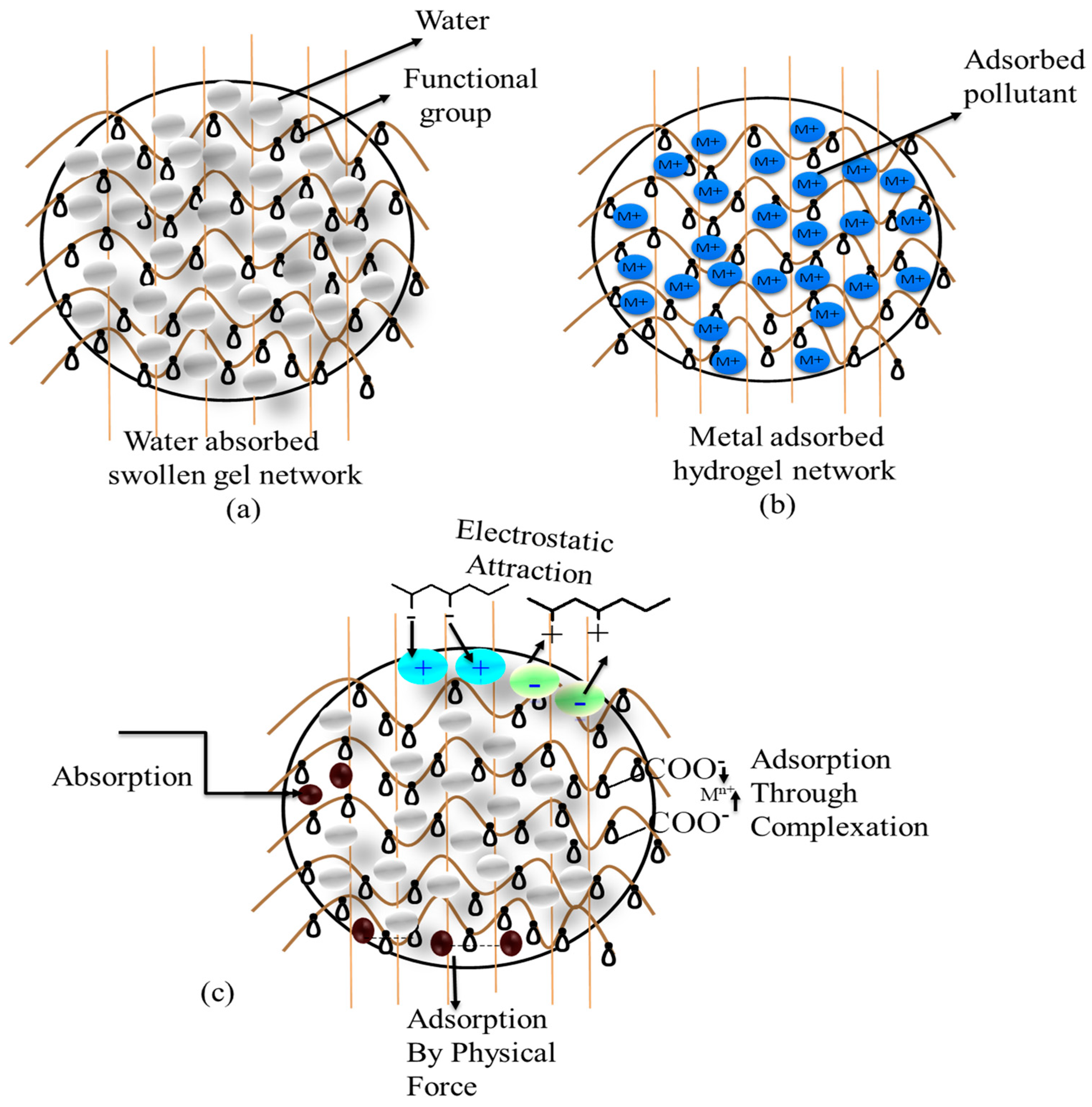
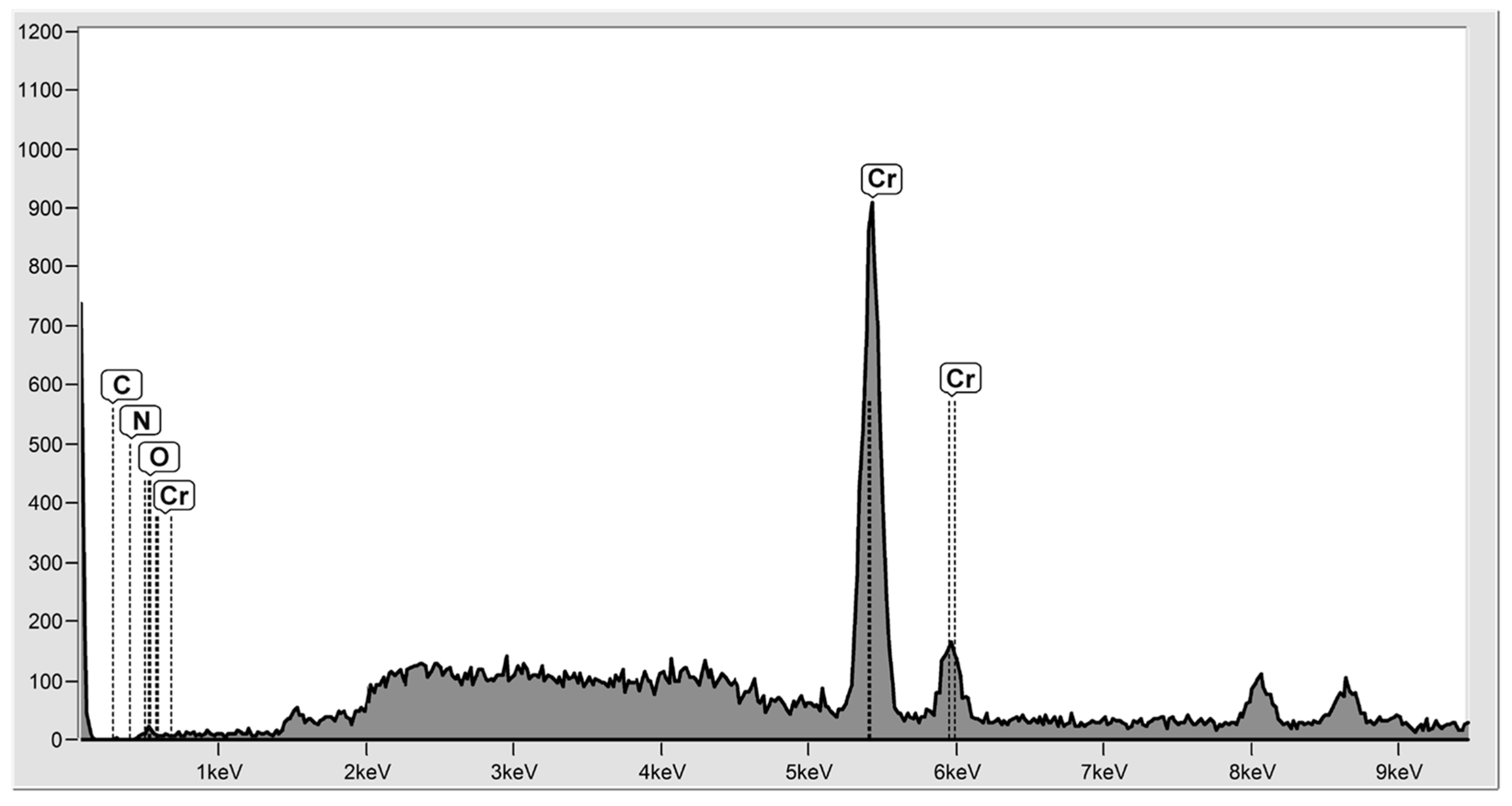
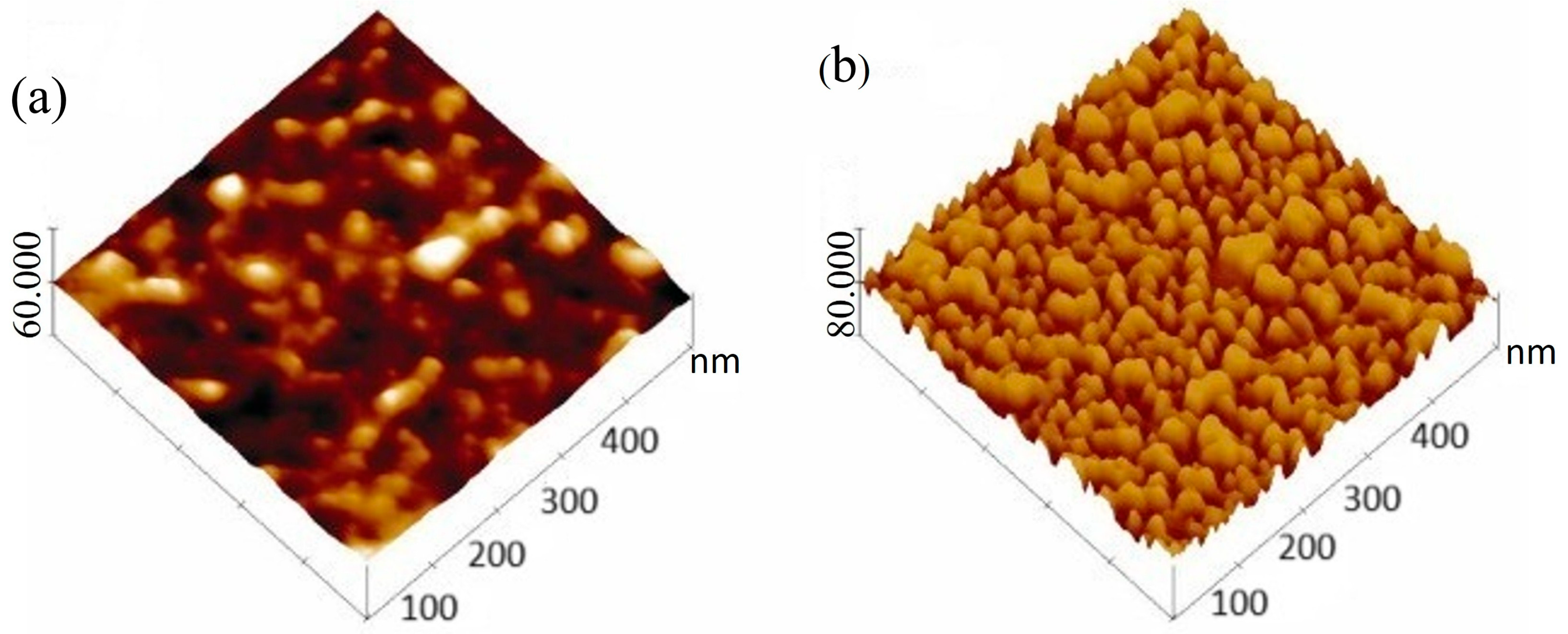
2. Hydrogel Surfaces for Adsorption
| Types of Hydrogels | Adsorbate | Adsorption Efficiency | Limitation | References |
|---|---|---|---|---|
| Graphene hydrogels | Ofloxacin | 105 mg/g gel | Lack of fast, cheap, and secure monitoring | [46] |
| Lignin sulfonate (LS)/acrylamide (AAm) | diclofenac | 50 mg/g gel | The inherent ‘hydrophobicity’ of lignin | [47] |
| Polyacrylamide-polyacrylic acid (PAA-PAM) | ciprofloxacin | 1401.53 mg/g gel | Aggregation of hydrogel reduces efficiency and recoverability | [48] |
| Polyvinyl alcohol-acrylic acid (PVA-AMA) | Streptomycin | 93.5 mg/g gel | Sensitivity to changes in ionic strength | [49] |
| Kappa carrageenan hydrogel | Oxytetracycline | 2.567 mg/g gels | Lower gel strength | [50] |
| Polyethyleneimine/poly(N-isopropylacrylamide) semi-IPN hydrogel | hydrophobic organic pollutants | p-nitro phenol—68.8 mmol/kg-dry gel 4-iodoaniline—28.7 mmol/kg-dry gel | Limited selectivity | [26] |
| β-cyclodextrin-polyacrylamide hydrogel | Organic Micropollutants | Phenolphthalein—34.97 mg/g Bisphenol A—29.59 mg/g propranolol hydrochloride—8.45 mg/g 2-napthol—55.56 mg/g | Costly and energy-consuming thermal regeneration | [51] |
| Polyvinyl alcohol/porous carbon composite hydrogels | Organic Pollutants | Congo red—27.39 mg/g Ibuprofen—15.48 mg/g Doxycycline hydrochloride—17.07 mg/g | Fragility of the hydrogel | [52] |
| Poly (N-isopropylacrylamide)-sodium carboxymethyl cellulose hydrogel | Organic nanoplastics | 199.64 mg/g | Mechanical fragility limits industrial applicability | [53] |
| Laccase-cellulose-DNA hydrogels | Micro-organic pollutants | The degradation rate of Flu (0.36 μg/h), 1-MFlu (0.38 μg/h) and 3-NFlu (0.30 μg/h) | Limited surface area and weak interaction forces | [54] |
| Immobilized laccase/chitosan/sodium alginate hydrogel | Organic Pollutant Bisphenol A | 34.92 µmol/L | Inadequate mechanical strength, poor stability, and low solubility | [55] |
| Graphite carbon nitride/calcium alginate | Rifampicin | The removal rate of 150 mg/L rifampicin | Limited selectivity hinders effectiveness | [56] |
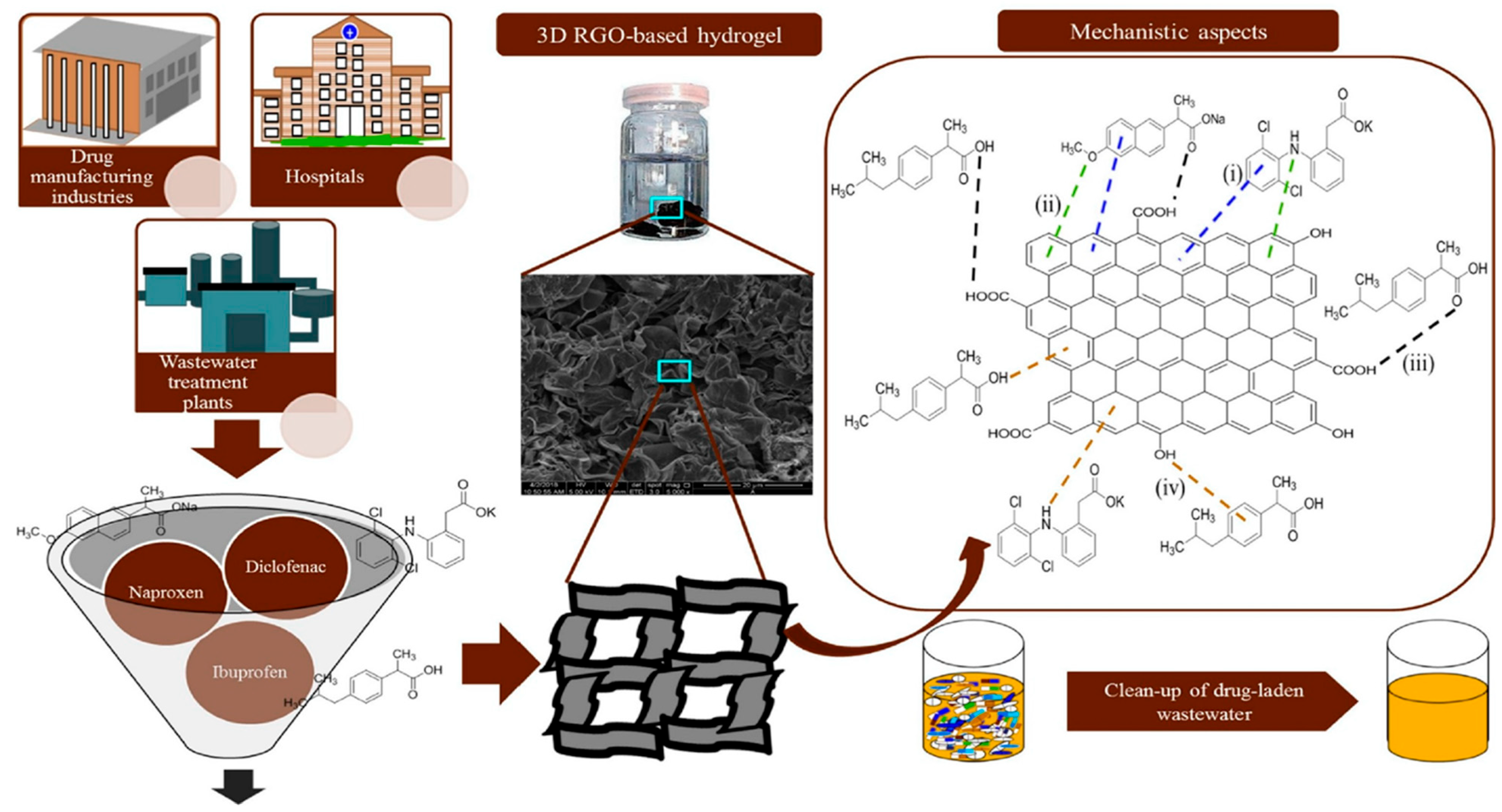
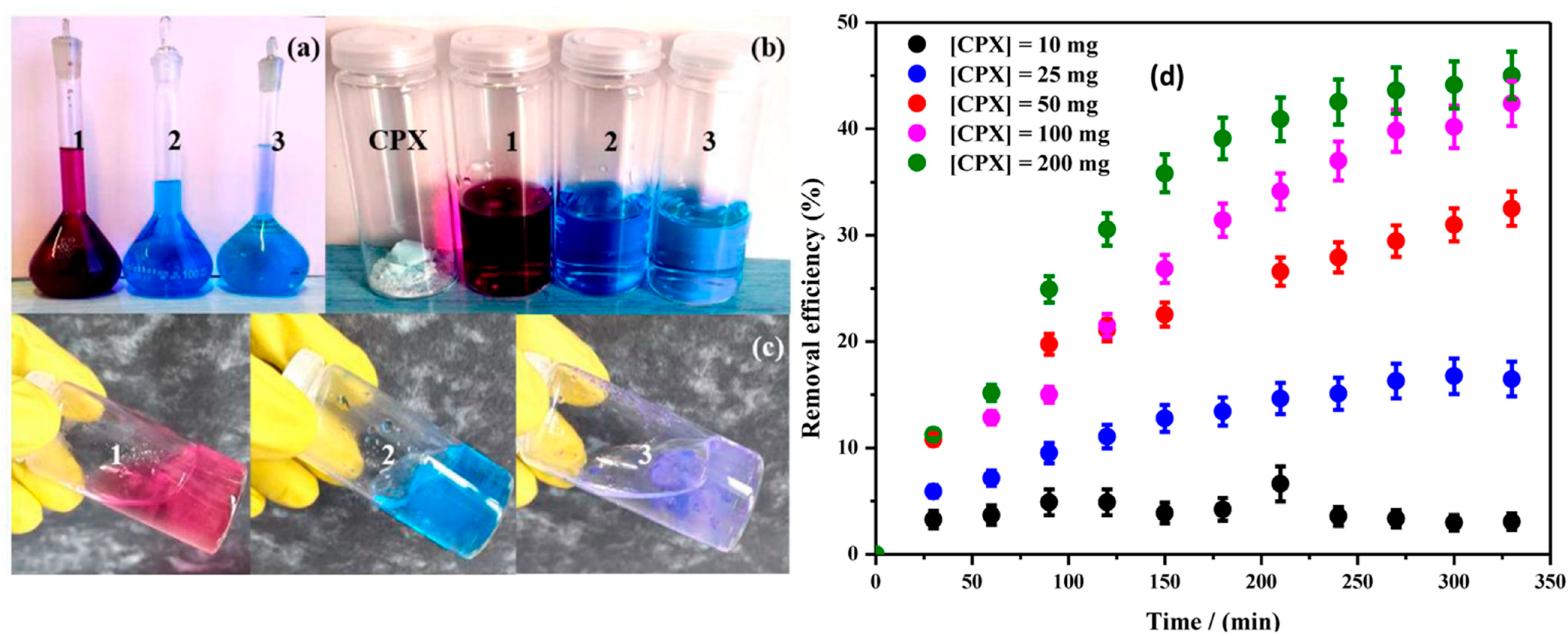
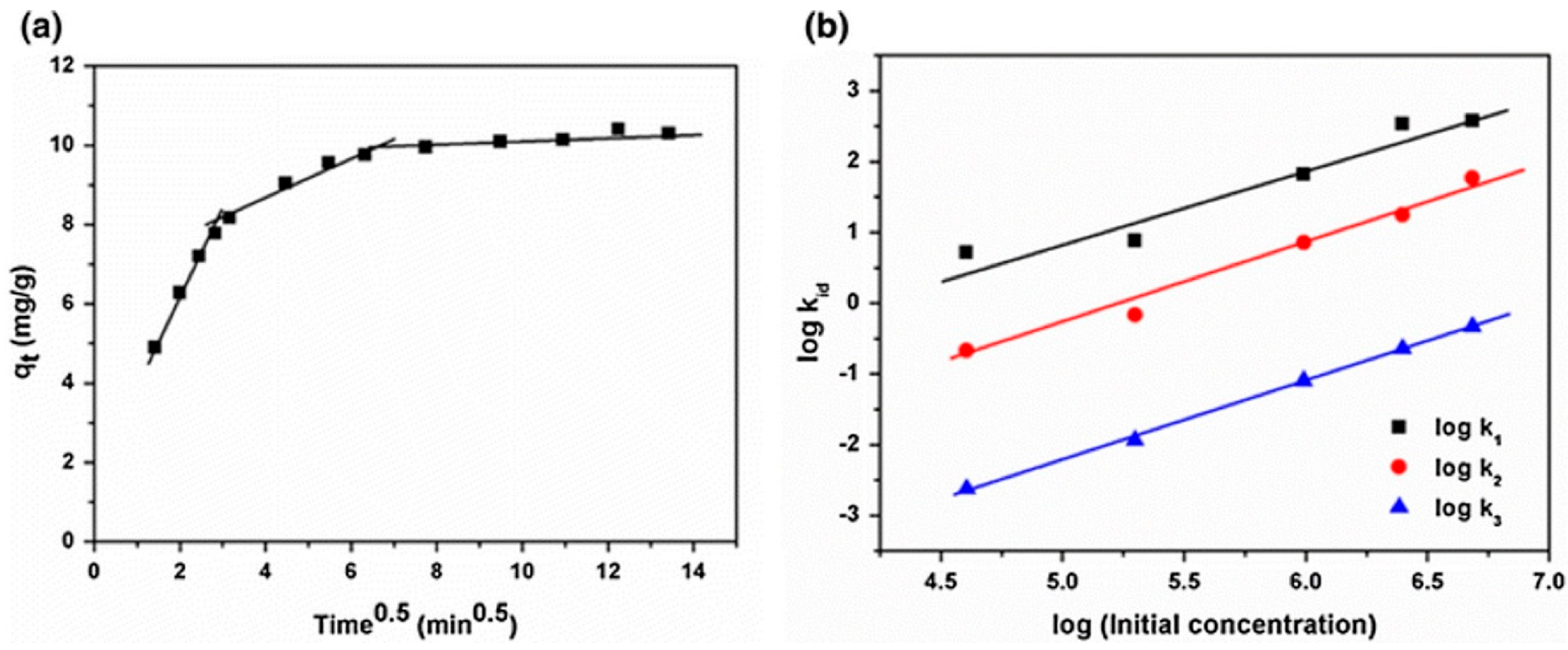
3. Adsorption of Pharmaceutical Pollutants
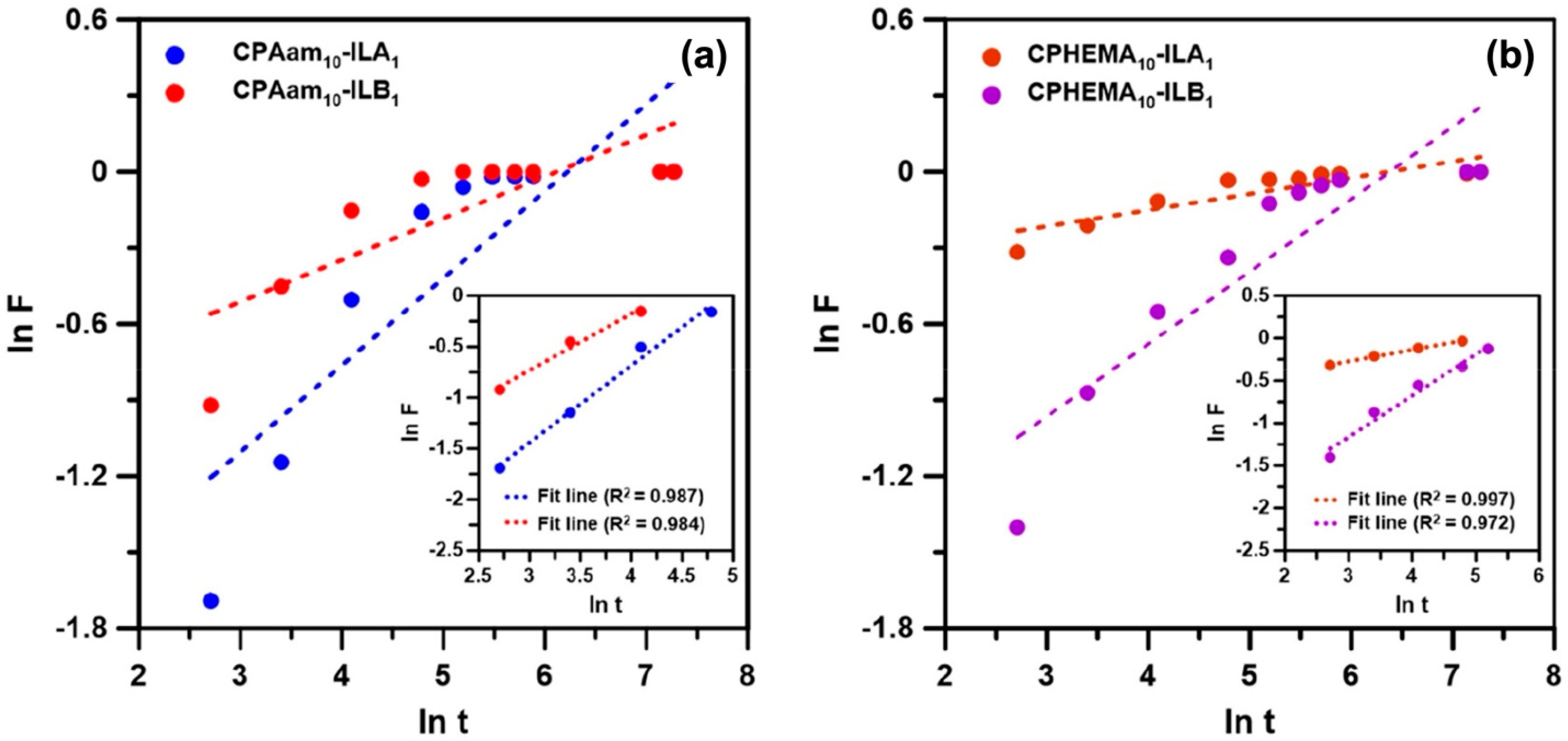
4. Adsorption of Organic Pollutants
5. Challenges and Scope of Improvements
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Correction Statement
References
- Godiya, C.B.; Martins Ruotolo, L.A.; Cai, W. Functional Biobased Hydrogels for the Removal of Aqueous Hazardous Pollutants: Current Status, Challenges, and Future Perspectives. J. Mater. Chem. A 2020, 8, 21585–21612. [Google Scholar] [CrossRef]
- Godiya, C.B.; Xiao, Y.; Lu, X. Amine Functionalized Sodium Alginate Hydrogel for Efficient and Rapid Removal of Methyl Blue in Water. Int. J. Biol. Macromol. 2020, 144, 671–681. [Google Scholar] [CrossRef]
- Islam, I.U.; Qurashi, A.N.; Adnan, A.; Ali, A.; Malik, S.; Younas, F.; Akhtar, H.T.; Farishta, F.; Janiad, S.; Ali, F.; et al. Bioremediation and Adsorption: Strategies for Managing Pharmaceutical Pollution in Aquatic Environment; Springer: Berlin/Heidelberg, Germany, 2025; Volume 236, ISBN 0123456789. [Google Scholar]
- Georgin, J.; Ramos, C.G.; de Oliveira, J.S.; Dehmani, Y.; El Messaoudi, N.; Meili, L.; Franco, D.S.P. A Critical Review of the Advances and Current Status of the Application of Adsorption in the Remediation of Micropollutants and Dyes Through the Use of Emerging Bio-Based Nanocomposites. Sustainability 2025, 17, 2012. [Google Scholar] [CrossRef]
- Li, W.C. Occurrence, Sources, and Fate of Pharmaceuticals in Aquatic Environment and Soil. Environ. Pollut. 2014, 187, 193–201. [Google Scholar] [CrossRef]
- Schwarzenbach, R.P.; Egli, T.; Hofstetter, T.B.; Von Gunten, U.; Wehrli, B. Global Water Pollution and Human Health. Annu. Rev. Environ. Resour. 2010, 35, 109–136. [Google Scholar] [CrossRef]
- Huang, X.; Wan, Y.; Shi, B.; Shi, J. Effects of Powdered Activated Carbon on the Coagulation-Flocculation Process in Humic Acid and Humic Acid-Kaolin Water Treatment. Chemosphere 2020, 238, 124637. [Google Scholar] [CrossRef]
- Mukhopadhyay, A.; Duttagupta, S.; Mukherjee, A. Emerging Organic Contaminants in Global Community Drinking Water Sources and Supply: A Review of Occurrence, Processes and Remediation. J. Environ. Chem. Eng. 2022, 10, 107560. [Google Scholar] [CrossRef]
- Shan, R.; He, Y.; Zi, T.; Wang, G.; Liu, X.; Han, Z.; Zhang, T.; Zhu, Y. Immobilization of Calcined Layered Double Hydroxide into Alginate Hydrogel Beads for PNP and PAP Removal: Kinetics, Isotherms, Thermodynamics, and Mechanism. Water Air Soil Pollut. 2018, 229, 329. [Google Scholar] [CrossRef]
- Yuan, Z.; Liu, H.; Wu, H.; Wang, Y.; Liu, Q.; Wang, Y.; Lincoln, S.F.; Guo, X.; Wang, J. Cyclodextrin Hydrogels: Rapid Removal of Aromatic Micropollutants and Adsorption Mechanisms. J. Chem. Eng. Data 2020, 65, 678–689. [Google Scholar] [CrossRef]
- Osuoha, J.O.; Anyanwu, B.O.; Ejileugha, C. Pharmaceuticals and Personal Care Products as Emerging Contaminants: Need for Combined Treatment Strategy. J. Hazard. Mater. Adv. 2023, 9, 100206. [Google Scholar] [CrossRef]
- Loganathan, P.; Vigneswaran, S.; Kandasamy, J.; Cuprys, A.K.; Maletskyi, Z.; Ratnaweera, H. Treatment Trends and Combined Methods in Removing Pharmaceuticals and Personal Care Products from Wastewater—A Review. Membranes 2023, 13, 158. [Google Scholar] [CrossRef]
- Hanafi, M.F.; Sapawe, N. A Review on the Current Techniques and Technologies of Organic Pollutants Removal from Water/Wastewater. Mater. Today Proc. 2020, 31, A158–A165. [Google Scholar] [CrossRef]
- Ribeiro, A.R.; Nunes, O.C.; Pereira, M.F.R.; Silva, A.M.T. An Overview on the Advanced Oxidation Processes Applied for the Treatment of Water Pollutants Defined in the Recently Launched Directive 2013/39/EU. Environ. Int. 2015, 75, 33–51. [Google Scholar] [CrossRef]
- Ma, J.; Dai, R.; Chen, M.; Khan, S.J.; Wang, Z. Applications of Membrane Bioreactors for Water Reclamation: Micropollutant Removal, Mechanisms and Perspectives. Bioresour. Technol. 2018, 269, 532–543. [Google Scholar] [CrossRef]
- Demirbas, A. Heavy Metal Adsorption onto Agro-Based Waste Materials: A Review. J. Hazard. Mater. 2008, 157, 220–229. [Google Scholar] [CrossRef]
- Akhtar, M.S.; Ali, S.; Zaman, W. Innovative Adsorbents for Pollutant Removal: Exploring the Latest Research and Applications. Molecules 2024, 29, 4317. [Google Scholar] [CrossRef]
- Bhuyan, M.M.; Hasan, N.; Jeong, J.-H. Single- and Multi-Network Hydrogels for Soft Electronics—A Review. Gels 2025, 11, 480. [Google Scholar] [CrossRef]
- Bhuyan, M.M.; Islam, M.; Jeong, J.-H. The Preparation and Characterization of N,N-Dimethyl Acrylamide-Diallyl Maleate Gel/Hydrogel in a Non-Aqueous Solution. Gels 2023, 9, 598. [Google Scholar] [CrossRef]
- Bhuyan, M.M.; Jeong, J.H. Synthesis and Characterization of Gamma Radiation Induced Diallyldimethylammonium Chloride-Acrylic Acid-(3-Acrylamidopropyl) Trimethylammonium Chloride Superabsorbent Hydrogel. Gels 2023, 9, 159. [Google Scholar] [CrossRef]
- Wu, Y.; Joseph, S.; Aluru, N.R. Effect of Cross-Linking on the Diffusion of Water, Ions, and Small Molecules in Hydrogels. J. Phys. Chem. B 2009, 113, 3512–3520. [Google Scholar] [CrossRef]
- Yuan, Y.; Zhang, Q.; Lin, S.; Li, J. Water: The Soul of Hydrogels. Prog. Mater. Sci. 2025, 148, 101378. [Google Scholar] [CrossRef]
- Ahmed, M.S.; Yun, S.; Kim, H.Y.; Ko, S.; Islam, M.; Nam, K.W. Hydrogels and Microgels: Driving Revolutionary Innovations in Targeted Drug Delivery, Strengthening Infection Management, and Advancing Tissue Repair and Regeneration. Gels 2025, 11, 179. [Google Scholar] [CrossRef]
- Fortunato, A.; Mba, M. A Peptide-Based Hydrogel for Adsorption of Dyes and Pharmaceuticals in Water Remediation. Gels 2022, 8, 672. [Google Scholar] [CrossRef]
- Ahmed, M.S.; Islam, M.; Hasan, M.K.; Nam, K.W. A Comprehensive Review of Radiation-Induced Hydrogels: Synthesis, properties, and multidimensional applications. Gels 2024, 10, 381. [Google Scholar] [CrossRef]
- Kunori, M.; Tokuyama, H. Development of a Polyethyleneimine/Poly(N-Isopropylacrylamide) Semi-IPN Hydrogel for Use in the Temperature-Swing Adsorption and Selective Desorption of Hydrophobic Organic Compounds. J. Taiwan Inst. Chem. Eng. 2024, 156, 105331. [Google Scholar] [CrossRef]
- Van Tran, V.; Park, D.; Lee, Y.C. Hydrogel Applications for Adsorption of Contaminants in Water and Wastewater Treatment. Environ. Sci. Pollut. Res. 2018, 25, 24569–24599. [Google Scholar] [CrossRef]
- Baruah, U.; Konwar, A.; Chowdhury, D. A Sulphonated Carbon Dot-Chitosan Hybrid Hydrogel Nanocomposite as an Efficient Ion-Exchange Film for Ca2+ and Mg2+ Removal. Nanoscale 2016, 8, 8542–8546. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, L.; Fu, S.; Zheng, L.; Zhan, H. Adsorption Behavior of Cd2+, Pb2+, and Ni2+ from Aqueous Solutions on Cellulose-Based Hydrogels. BioResources 2012, 7, 2752–2765. [Google Scholar] [CrossRef]
- Yang, Z.; Peng, H.; Wang, W.; Liu, T. Crystallization Behavior of Poly(ε-Caprolactone)/Layered Double Hydroxide Nanocomposites. J. Appl. Polym. Sci. 2010, 116, 2658–2667. [Google Scholar] [CrossRef]
- Seida, Y.; Tokuyama, H. Hydrogel Adsorbents for the Removal of Hazardous Pollutants—Requirements and Available Functions as Adsorbent. Gels 2022, 8, 220. [Google Scholar] [CrossRef]
- Alam, M.N.; Christopher, L.P. Natural Cellulose-Chitosan Cross-Linked Superabsorbent Hydrogels with Superior Swelling Properties. ACS Sustain. Chem. Eng. 2018, 6, 8736–8742. [Google Scholar] [CrossRef]
- Ahmed, E.M. Hydrogel: Preparation, Characterization, and Applications: A Review. J. Adv. Res. 2015, 6, 105–121. [Google Scholar] [CrossRef]
- De Andrade, J.R.; Oliveira, M.F.; Da Silva, M.G.C.; Vieira, M.G.A. Adsorption of Pharmaceuticals from Water and Wastewater Using Nonconventional Low-Cost Materials: A Review. Ind. Eng. Chem. Res. 2018, 57, 3103–3127. [Google Scholar] [CrossRef]
- Alsaka, L.; Alsaka, L.; Altaee, A.; Zaidi, S.J.; Zhou, J.; Kazwini, T. A Review of Hydrogel Application in Wastewater Purification. Separations 2025, 12, 51. [Google Scholar] [CrossRef]
- Qi, Y.-L.; Zhou, H.-Y.; Han, G.-Z. A Novel Photo-Regulated Self-Healing Hydrogel Based on Hollow SiO2@g-C3N4@TiO2 and PVA. RSC Adv. 2025, 15, 21300–21310. [Google Scholar] [CrossRef]
- Kim, S.J.; Baek, M.; Choe, J.; Shin, J.J. Reprocessible, Reusable, and Self-Healing Polymeric Adsorbent For Removing Perfluorinated Pollutants. Materials 2024, 17, 5170. [Google Scholar] [CrossRef]
- Gu, J.; Zhao, L.; Ala, U.M.; Zhao, K.; Liu, H.; Zhang, W.; Dai, J.; Ge, J.; Yan, T.; Cheng, Y.; et al. Dynamic Covalent Bonds Enabled Robust and Self-Healing Superhydrophobic Coatings with Multifunctions. Sep. Purif. Technol. 2025, 359, 130824. [Google Scholar] [CrossRef]
- Visan, A.I.; Negut, I. Environmental and Wastewater Treatment Applications of Stimulus-Responsive Hydrogels. Gels 2025, 11, 72. [Google Scholar] [CrossRef]
- Wei, M.; Serpe, M.J. Temperature–Light Dual-Responsive Au@PNIPAm Core-Shell Microgel-Based Optical Devices. Part. Part. Syst. Charact. 2019, 36, 1800326. [Google Scholar] [CrossRef]
- Kim, J.; Minh, N.H.; Kwon, D.I.; Kim, K.; Kang, D.H.; Yoo, Y.-E.; Yoon, J.S. Photonic Crystal Hydrogels Based on Highly Reproducible Molding Method. Sci. Rep. 2025, 15, 26044. [Google Scholar] [CrossRef]
- Liu, J.; Su, D.; Yao, J.; Huang, Y.; Shao, Z.; Chen, X. Soy Protein-Based Polyethylenimine Hydrogel and Its High Selectivity for Copper Ion Removal in Wastewater Treatment. J. Mater. Chem. A 2017, 5, 4163–4171. [Google Scholar] [CrossRef]
- Dwivedi, C.; Pathak, S.K.; Kumar, M.; Tripathi, S.C.; Bajaj, P.N. Preparation and Characterization of Potassium Nickel Hexacyanoferrate-Loaded Hydrogel Beads for the Removal of Cesium Ions. Environ. Sci. Water Res. Technol. 2015, 1, 153–160. [Google Scholar] [CrossRef]
- Al-Mubaddel, F.S.; Aijaz, M.O.; Haider, S.; Haider, A.; Almasry, W.A.; Al-Fatesh, A.S. Synthesis of Chitosan Based Semi-IPN Hydrogels Using Epichlorohydrine as Crosslinker to Study the Adsorption Kinetics of Rhodamine B. Desalin. Water Treat. 2016, 57, 17523–17536. [Google Scholar] [CrossRef]
- Heydari, A.; Sheibani, H. Fabrication of Poly(β-Cyclodextrin-Co-Citric Acid)/Bentonite Clay Nanocomposite Hydrogel: Thermal and Absorption Properties. RSC Adv. 2015, 5, 82438–82449. [Google Scholar] [CrossRef]
- Ehtesabi, H.; Bagheri, Z.; Yaghoubi-Avini, M. Application of Three-Dimensional Graphene Hydrogels for Removal of Ofloxacin from Aqueous Solutions. Environ. Nanotechnol. Monit. Manag. 2019, 12, 100274. [Google Scholar] [CrossRef]
- Ranieri, S.; Pisani, M.; Luzi, F.; Parlapiano, M.; Giorgini, E.; Rallini, M.; Corinaldesi, V.; Astolfi, P. Development and Characterization of Lignin-Based Hydrogels for Efficient Adsorption of Diclofenac From Aqueous Environment. J. Polym. Sci. 2025, 1–14. [Google Scholar] [CrossRef]
- Gao, M.; Sun, M.; Bi, J.; Wang, S.; Guo, X.; Li, F.; Liu, J.; Zhao, Y. Removal of Ciprofloxacin by PAA-PAM Hydrogel: Adsorption Performance and Mechanism Studies. J. Water Process Eng. 2025, 71, 107361. [Google Scholar] [CrossRef]
- Radia, N.D.; Kamona, S.M.H.; Jasem, H.; Abass, R.R.; Izzat, S.E.; Ali, M.S.; Ghafel, S.T.; Aljeboree, A.M. Role of Hydrogel and Study of Its High-Efficiency to Removal Streptomycin Drug from Aqueous Solutions. Int. J. Pharm. Qual. Assur. 2022, 13, 160–163. [Google Scholar] [CrossRef]
- Afzaal, M.; Nawaz, R.; Hussain, S.; Nadeem, M.; Irshad, M.A.; Irfan, A.; Mannan, H.A.; Al-Mutairi, A.A.; Islam, A.; Al-Hussain, S.A.; et al. Removal of Oxytetracycline from Pharmaceutical Wastewater Using Kappa Carrageenan Hydrogel. Sci. Rep. 2024, 14, 19687. [Google Scholar] [CrossRef]
- Song, X.; Mensah, N.N.; Wen, Y.; Zhu, J.; Zhang, Z.; Tan, W.S.; Chen, X.; Li, J. β-Cyclodextrin-Polyacrylamide Hydrogel for Removal of Organic Micropollutants from Water. Molecules 2021, 26, 5031. [Google Scholar] [CrossRef]
- Zhang, M.; Yu, X.; Zhu, M.; Xiang, A.; Bai, Y.; Zhou, H. Adsorptive Behaviors and Mechanisms for Removing Three Organic Pollutants from Aqueous Solutions by Polyvinyl Alcohol/Porous Carbon Composite Hydrogels. J. Environ. Chem. Eng. 2023, 11, 111095. [Google Scholar] [CrossRef]
- Mondal, S.; Baghel, K.; Cho, S.; Zahra; Lim, H.; Lee, J. Efficient Removal of Amine-Modified Polystyrene Nanoplastics Utilizing Poly(N-Isopropylacrylamide)-Sodium Carboxymethyl Cellulose Hydrogel Beads: Parametric Optimization and Mechanistic Insights. Sep. Purif. Technol. 2025, 363, 132035. [Google Scholar] [CrossRef]
- Zhang, J.; White, J.C.; He, J.; Yu, X.; Yan, C.; Dong, L.; Tao, S.; Wang, X. Sustainable Bioactive Hydrogels for Organic Contaminant Elimination in Wastewater. Nat. Commun. 2025, 16, 2512. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, X.; Wang, L.; Wang, B.; Zeng, X.; Ren, B. Functionalized Chitosan and Alginate Composite Hydrogel-Immobilized Laccase with Sustainable Biocatalysts for the Effective Removal of Organic Pollutant Bisphenol A. Catalysts 2024, 14, 304. [Google Scholar] [CrossRef]
- Li, P.; Jiang, L.; Luan, J.; Zhang, Y.; Liu, W.; Yan, Z.; Ke, X. Construction of Highly Stable Multifunctional Hydrogels by Tungstenization Engineering: Built-in Electric Field-Driven Adsorption-Dark Degradation-Photocatalysis Triple Synergistic Mechanism for Efficient Removal of Organic Pollutants. J. Alloys Compd. 2025, 1037, 182328. [Google Scholar] [CrossRef]
- Milosavljević, N.B.; Ristić, M.D.; Perić-Grujić, A.A.; Filipović, J.M.; Štrbac, S.B.; Rakočević, Z.L.; Krušić, M.T.K. Removal of Cu2+ Ions Using Hydrogels of Chitosan, Itaconic and Methacrylic Acid: FTIR, SEM/EDX, AFM, Kinetic and Equilibrium Study. Colloids Surf. A Physicochem. Eng. Asp. 2011, 388, 59–69. [Google Scholar] [CrossRef]
- Erdem, S.; Öztekin, M.; Sağ Açıkel, Y. Investigation of Tetracycline Removal from Aqueous Solutions Using Halloysite/Chitosan Nanocomposites and Halloysite Nanotubes/Alginate Hydrogel Beads. Environ. Nanotechnol. Monit. Manag. 2021, 16, 100576. [Google Scholar] [CrossRef]
- Milosavljević, N.B.; Milašinović, N.Z.; Popović, I.G.; Filipović, J.M.; Kalagasidis Krušić, M.T. Preparation and Characterization of PH-Sensitive Hydrogels Based on Chitosan, Itaconic Acid and Methacrylic Acid. Polym. Int. 2011, 60, 443–452. [Google Scholar] [CrossRef]
- Zdravković, A.; Nikolić, V.; Ilić-Stojanović, S.; Stojanović, S.; Dinić, A.; Urošević, M.; Gajić, I.; Nikolić, L. Synthesis and Application of Poly(N-Isopropylacrylamide-Co-Methacrylic Acid) Hydrogels as Sorbent Materials for Wastewater Treatment. Separations 2025, 12, 100. [Google Scholar] [CrossRef]
- Samal, K.; Mahapatra, S.; Hibzur Ali, M. Pharmaceutical Wastewater as Emerging Contaminants (EC): Treatment Technologies, Impact on Environment and Human Health. Energy Nexus 2022, 6, 100076. [Google Scholar] [CrossRef]
- Batt, A.L.; Kim, S.; Aga, D.S. Comparison of the Occurrence of Antibiotics in Four Full-Scale Wastewater Treatment Plants with Varying Designs and Operations. Chemosphere 2007, 68, 428–435. [Google Scholar] [CrossRef]
- Feng, L.; van Hullebusch, E.D.; Rodrigo, M.A.; Esposito, G.; Oturan, M.A. Removal of Residual Anti-Inflammatory and Analgesic Pharmaceuticals from Aqueous Systems by Electrochemical Advanced Oxidation Processes. A Review. Chem. Eng. J. 2013, 228, 944–964. [Google Scholar] [CrossRef]
- Tiwari, B.; Sellamuthu, B.; Ouarda, Y.; Drogui, P.; Tyagi, R.D.; Buelna, G. Review on Fate and Mechanism of Removal of Pharmaceutical Pollutants from Wastewater Using Biological Approach. Bioresour. Technol. 2017, 224, 1–12. [Google Scholar] [CrossRef]
- Dolliver, H.; Gupta, S. Antibiotic Losses in Leaching and Surface Runoff from Manure-Amended Agricultural Land. J. Environ. Qual. 2008, 37, 1227–1237. [Google Scholar] [CrossRef]
- Guo, W.Q.; Zheng, H.S.; Li, S.; Du, J.S.; Feng, X.C.; Yin, R.L.; Wu, Q.L.; Ren, N.Q.; Chang, J.S. Removal of Cephalosporin Antibiotics 7-ACA from Wastewater during the Cultivation of Lipid-Accumulating Microalgae. Bioresour. Technol. 2016, 221, 284–290. [Google Scholar] [CrossRef]
- Lajeunesse, A.; Smyth, S.A.; Barclay, K.; Sauvé, S.; Gagnon, C. Distribution of Antidepressant Residues in Wastewater and Biosolids Following Different Treatment Processes by Municipal Wastewater Treatment Plants in Canada. Water Res. 2012, 46, 5600–5612. [Google Scholar] [CrossRef]
- Daughton, C.G.; Ternes, T.A. Pharmaceuticals and Personal Care Products in the Environment: Agents of Subtle Change? Environ. Health Perspect. 1999, 107, 907–938. [Google Scholar] [CrossRef]
- Cleuvers, M. Aquatic Ecotoxicity of Pharmaceuticals Including the Assessment of Combination Effects. Toxicol. Lett. 2003, 142, 185–194. [Google Scholar] [CrossRef]
- Gaynor, M.; Mankin, A.S. Macrolide Antibiotics: Binding Site, Mechanism of Action, Resistance. Front. Med. Chem. 2012, 2, 21–35. [Google Scholar] [CrossRef]
- Ebele, A.J.; Abou-Elwafa Abdallah, M.; Harrad, S. Pharmaceuticals and Personal Care Products (PPCPs) in the Freshwater Aquatic Environment. Emerg. Contam. 2017, 3, 1–16. [Google Scholar] [CrossRef]
- Subedi, B.; Du, B.; Chambliss, C.K.; Koschorreck, J.; Rüdel, H.; Quack, M.; Brooks, B.W.; Usenko, S. Occurrence of Pharmaceuticals and Personal Care Products in German Fish Tissue: A National Study. Environ. Sci. Technol. 2012, 46, 9047–9054. [Google Scholar] [CrossRef]
- Liu, J.; Lu, G.; Ding, J.; Zhang, Z.; Wang, Y. Tissue Distribution, Bioconcentration, Metabolism, and Effects of Erythromycin in Crucian Carp (Carassius auratus). Sci. Total Environ. 2014, 490, 914–920. [Google Scholar] [CrossRef]
- Gonzalez-Rey, M.; Bebianno, M.J. Does Non-Steroidal Anti-Inflammatory (NSAID) Ibuprofen Induce Antioxidant Stress and Endocrine Disruption in Mussel Mytilus Galloprovincialis? Environ. Toxicol. Pharmacol. 2012, 33, 361–371. [Google Scholar] [CrossRef]
- Rhodes, G.; Chuang, Y.H.; Hammerschmidt, R.; Zhang, W.; Boyd, S.A.; Li, H. Uptake of Cephalexin by Lettuce, Celery, and Radish from Water. Chemosphere 2021, 263, 127916. [Google Scholar] [CrossRef]
- Bu, Q.; Wang, B.; Huang, J.; Deng, S.; Yu, G. Pharmaceuticals and Personal Care Products in the Aquatic Environment in China: A Review. J. Hazard. Mater. 2013, 262, 189–211. [Google Scholar] [CrossRef]
- Houtman, C.J.; Van Oostveen, A.M.; Brouwer, A.; Lamoree, M.H.; Legler, J. Identification of Estrogenic Compounds in Fish Bile Using Bioassay-Directed Fractionation. Environ. Sci. Technol. 2004, 38, 6415–6423. [Google Scholar] [CrossRef]
- Clara, M.; Strenn, B.; Kreuzinger, N. Carbamazepine as a Possible Anthropogenic Marker in the Aquatic Environment: Investigations on the Behaviour of Carbamazepine in Wastewater Treatment and during Groundwater Infiltration. Water Res. 2004, 38, 947–954. [Google Scholar] [CrossRef]
- Amdany, R.; Chimuka, L.; Cukrowska, E. Determination of Naproxen, Ibuprofen and Triclosan in Wastewater Using the Polar Organic Chemical Integrative Sampler (POCIS): A Laboratory Calibration and Field Application. Water SA 2014, 40, 407–414. [Google Scholar] [CrossRef]
- Ma, J.; Sun, Y.; Zhang, M.; Yang, M.; Gong, X.; Yu, F.; Zheng, J. Comparative Study of Graphene Hydrogels and Aerogels Reveals the Important Role of Buried Water in Pollutant Adsorption. Environ. Sci. Technol. 2017, 51, 12283–12292. [Google Scholar] [CrossRef]
- Sun, Y.; Shao, D.; Chen, C.; Yang, S.; Wang, X. Highly Efficient Enrichment of Radionuclides on Graphene Oxide-Supported Polyaniline. Environ. Sci. Technol. 2013, 47, 9904–9910. [Google Scholar] [CrossRef]
- Sohni, S.; Gul, K.; Ahmad, F.; Ahmad, I.; Khan, A.; Khan, N.; Bahadar Khan, S. Highly Efficient Removal of Acid Red-17 and Bromophenol Blue Dyes from Industrial Wastewater Using Graphene Oxide Functionalized Magnetic Chitosan Composite. Polym. Compos. 2018, 39, 3317–3328. [Google Scholar] [CrossRef]
- Umbreen, N.; Sohni, S.; Ahmad, I.; Khattak, N.U.; Gul, K. Self-Assembled Three-Dimensional Reduced Graphene Oxide-Based Hydrogel for Highly Efficient and Facile Removal of Pharmaceutical Compounds from Aqueous Solution. J. Colloid Interface Sci. 2018, 527, 356–367. [Google Scholar] [CrossRef] [PubMed]
- Chelu, M.; Popa, M.; Calderon Moreno, J.; Leonties, A.R.; Ozon, E.A.; Pandele Cusu, J.; Surdu, V.A.; Aricov, L.; Musuc, A.M. Green Synthesis of Hydrogel-Based Adsorbent Material for the Effective Removal of Diclofenac Sodium from Wastewater. Gels 2023, 9, 454. [Google Scholar] [CrossRef]
- Zhu, H.; Chen, S.; Luo, Y. Adsorption Mechanisms of Hydrogels for Heavy Metal and Organic Dyes Removal: A Short Review. J. Agric. Food Res. 2023, 12, 100552. [Google Scholar] [CrossRef]
- Mittal, H.; Al Alili, A.; Morajkar, P.P.; Alhassan, S.M. GO Crosslinked Hydrogel Nanocomposites of Chitosan/Carboxymethyl Cellulose—A Versatile Adsorbent for the Treatment of Dyes Contaminated Wastewater. Int. J. Biol. Macromol. 2021, 167, 1248–1261. [Google Scholar] [CrossRef]
- Chen, J.F.; Lin, Q.; Yao, H.; Zhang, Y.M.; Wei, T.B. Pillar[5]arene-Based Multifunctional Supramolecular Hydrogel: Multistimuli Responsiveness, Self-Healing, Fluorescence Sensing, and Conductivity. Mater. Chem. Front. 2018, 2, 999–1003. [Google Scholar] [CrossRef]
- Mohammed, N.; Grishkewich, N.; Berry, R.M.; Tam, K.C. Cellulose Nanocrystal–Alginate Hydrogel Beads as Novel Adsorbents for Organic Dyes in Aqueous Solutions. Cellulose 2015, 22, 3725–3738. [Google Scholar] [CrossRef]
- Banerjee, P.; Dinda, P.; Kar, M.; Uchman, M.; Mandal, T.K. Ionic Liquid Cross-Linked High-Absorbent Polymer Hydrogels: Kinetics of Swelling and Dye Adsorption. Langmuir 2023, 39, 9757–9772. [Google Scholar] [CrossRef]
- Tiwari, J.N.; Mahesh, K.; Le, N.H.; Kemp, K.C.; Timilsina, R.; Tiwari, R.N.; Kim, K.S. Reduced Graphene Oxide-Based Hydrogels for the Efficient Capture of Dye Pollutants from Aqueous Solutions. Carbon 2013, 56, 173–182. [Google Scholar] [CrossRef]
- Thomas, M.; Naikoo, G.A.; Sheikh, M.U.D.; Bano, M.; Khan, F. Effective Photocatalytic Degradation of Congo Red Dye Using Alginate/Carboxymethyl Cellulose/TiO2 Nanocomposite Hydrogel under Direct Sunlight Irradiation. J. Photochem. Photobiol. A Chem. 2016, 327, 33–43. [Google Scholar] [CrossRef]
- Santoso, S.P.; Angkawijaya, A.E.; Bundjaja, V.; Hsieh, C.W.; Go, A.W.; Yuliana, M.; Hsu, H.Y.; Tran-Nguyen, P.L.; Soetaredjo, F.E.; Ismadji, S. TiO2/Guar Gum Hydrogel Composite for Adsorption and Photodegradation of Methylene Blue. Int. J. Biol. Macromol. 2021, 193, 721–733. [Google Scholar] [CrossRef]
- Hernandez Monroy, L.; Tavares, J.R.; Dumont, M.J. Application of Photocatalytic Hydrogels Used for the Degradation of Dyes, Pharmaceuticals and Other Contaminants in Water. Adsorption 2025, 31, 52. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bhuyan, M.M.; Ahmed, M. Effective Hydrogel Surfaces for Adsorption of Pharmaceutical and Organic Pollutants—A Mini Review. Surfaces 2025, 8, 61. https://doi.org/10.3390/surfaces8030061
Bhuyan MM, Ahmed M. Effective Hydrogel Surfaces for Adsorption of Pharmaceutical and Organic Pollutants—A Mini Review. Surfaces. 2025; 8(3):61. https://doi.org/10.3390/surfaces8030061
Chicago/Turabian StyleBhuyan, Md Murshed, and Mansur Ahmed. 2025. "Effective Hydrogel Surfaces for Adsorption of Pharmaceutical and Organic Pollutants—A Mini Review" Surfaces 8, no. 3: 61. https://doi.org/10.3390/surfaces8030061
APA StyleBhuyan, M. M., & Ahmed, M. (2025). Effective Hydrogel Surfaces for Adsorption of Pharmaceutical and Organic Pollutants—A Mini Review. Surfaces, 8(3), 61. https://doi.org/10.3390/surfaces8030061








