Abstract
The present study investigates the potential of olive mill wastewater (OMW), supplemented with expired commercial glucose syrup, as a sustainable substrate for the submerged cultivation of Tuber spp. wild mushrooms. OMW contains considerable quantities of phenolic compounds, making it both a challenging pollutant and a promising nutrient source. To assess fungal performance under increasing phenolic stress, culture media were prepared with varying OMW concentrations (0–75% v/v on agar; 0–50% v/v in liquid media), while glucose was adjusted to ~30 g/L using expired glucose syrup. A sequential experimental approach was followed, beginning with Petri dish screenings on substrate/strain selection (measuring the mycelial growth rate; Kr, mm/day), progressing to 25-day shake flask fermentations and subsequently scaling up the most promising strain (Tuber mesentericum) in a controlled stirred-tank bioreactor. Throughout cultivation, substrate consumption (glucose, phenolics), pH evolution and decolorization were evaluated, while the resulting biomass was analyzed for polysaccharides, β-glucans, proteins, lipids, fatty acids, antioxidants, phenolic acids and triterpenoids content. Results showed that increasing OMW concentration enhanced tolerance and metabolic activity in selected Tuber species, with T. mesentericum exhibiting the highest resilience and achieving comparable or higher biomass yields in OMW-based media than in glucose (control). Phenolic removal exceeded 60% in flasks and 50% in the bioreactor, confirming simultaneous bioremediation capacity. Bioreactor cultivation demonstrated efficient substrate utilization and biomass production, while OMW-grown biomass presented high lipid content, enriched with unsaturated fatty acids, high β-glucan levels and increased antioxidant and phenolic profiles. Overall, this study demonstrates that OMW (supplemented with expired glucose syrup) can serve as a cost-effective and environmentally beneficial substrate for Tuber biomass production with dietary and antioxidant properties, offering an alternative source to mushroom carposomes, as well as supporting the circular bioeconomy strategies within olive oil processing industries.
1. Introduction
Truffles, belonging to the genus Tuber, are hypogeous Ascomycetes that establish ectomycorrhizal symbioses with the roots of a wide variety of woody hosts, including Quercus, Corylus and Pinus species [1,2]. Over 180 Tuber species have been described worldwide, many of which play key ecological roles in nutrient cycling and forest resilience while also supporting an economically valuable culinary industry [3,4]. Truffles are a valuable, unique resource, mostly for their organoleptic properties like aroma and taste and their market price ranges from 600 to 6000 €/kg [5]. However, the natural yield of truffles has drastically declined over the last century, mainly due to climate change, deforestation and soil degradation, which has stimulated efforts toward artificial and semi-controlled cultivation systems [6,7,8]. Natural truffle species also demonstrate substantial protein contributions, although their composition varies according to species and geographical origin. Generally, carbohydrates and proteins constitute the major macronutrient fractions of truffles [9], while minerals, dietary fiber, essential amino acids, fatty acids and lipids also comprise significant portions of their nutritional profile [10,11].
Submerged fermentation of edible and medicinal fungi has emerged as an efficient alternative to traditional fruiting-body cultivation, which is time-consuming, labor-intensive and highly dependent on environmental variables [12,13]. Compared with solid-state cultivation, submerged fermentation enables rapid production of mycelial biomass and bioactive metabolites, while requiring reduced space, offering shorter production cycles and lowering contamination risks. Given that truffles, unlike saprophytic mushrooms, are primarily dependent on symbiotic interactions, their cultivation on synthetic media remains challenging; nevertheless, liquid cultivation systems offer a useful biotechnological approach for the production of truffle mycelial biomass under controlled conditions. In this context, Tuber species have increasingly been examined under submerged cultivation, which allows the controlled production of mycelia and important metabolites such as polysaccharides, lipids and phenolic derivatives in significantly shorter timeframes [8,14]. Tuber sinensis and Tuber maculatum have been cultivated in liquid fermentations for polysaccharides extraction and enzyme production. The success of submerged cultivation is strongly dependent on the inoculum, medium composition, including carbon and nitrogen sources, aeration, pH and temperature. Most Tuber spp. exhibit optimal mycelial growth at 22–25 °C and pH between 5 and 7, with glucose- or malt-based media and yeast extract or peptone shown to promote abundant biomass and polysaccharide synthesis [1,4,7,8].
Another important environmental issue that could be addressed through biotechnological approaches is the management of the millions of liters of olive mill wastewater (OMW) generated annually by the Mediterranean olive oil industry. OMW is a phenolic-rich effluent with high organic load that contains noticeable quantities of phenolic compounds and (potentially) increased quantities of residual sugars, presenting low pH and considerable phytotoxicity and posing serious environmental challenges due to its high pollutant load and resistance to biodegradation [15,16,17]. However, these same characteristics (high sugar and organic acid content) render OMW a potentially valuable substrate for microbial bioconversion. Multiple macrofungi, including Pleurotus spp., Ganoderma spp., Schizophyllum and even Tuber spp., have demonstrated strong tolerance to OMW and the capacity to degrade phenolic compounds, reduce color and convert this waste into fungal biomass enriched with valuable metabolites [18,19,20,21,22]. Such findings highlight the dual nature of OMW; a harmful pollutant on one hand and a promising nutrient-rich substrate on the other. The valorization potential of OMW as cultivation substrate can be further enhanced by supplementation with additional low-cost carbon sources, specifically in sugar concentrations into the effluent that do not present excessively high values (i.e., in the case of the OMWs deriving from three-phase centrifugation systems [23,24,25,26]). Expired commercial glucose syrup, due to its high sugar concentration and low water activity, remains chemically stable and suitable for microbial fermentation, while its reuse supports circular-bioeconomy principles aimed at reducing food-industry waste [27,28]. Similar agro-industrial streams (including molasses, crude glycerol and other sugar-rich byproducts) have already been successfully utilized for fungal growth and metabolite production in species such as Morchella spp., Pleurotus spp., Lentinula edodes, Ganoderma resinaceum and Tuber aestivum [19,20,29,30,31].
Scaling up submerged fermentations from laboratory to industrial relevance requires a structured, gradual approach [32,33,34]. Shake flask cultures may serve as a small-scale process that provide enhanced aeration, mixing, and nutrient availability compared to static ones, enabling reliable assessment of growth kinetics and substrate utilization under rather moderate controlled conditions. However, process optimization and industrial feasibility can only be achieved in bioreactors [13,35,36]. Stirred-tank bioreactors allow precise control over aeration, pH, agitation and temperature, enabling reproducible growth and consistent metabolite production [8,37]. For Tuber spp., bioreactor cultivation is advantageous, as it enables real-time monitoring of glucose consumption, phenolic degradation and decolorization (key parameters when working with complex substrates such as OMWs). Consequently, integrating shake flask fermentation with controlled bioreactor studies is essential for evaluating the adaptation, scalability and biotechnological potential of Tuber species grown on agro-industrial waste streams [35,38].
The aim and scope of the present study was the investigation of whether OMW and expired glucose syrup can support the cultivation of several Tuber strains and stimulate the production of valuable metabolites. Initially, a screening stage was performed on agar media containing increasing 0–75% v/v OMW (measuring the mycelial growth rate in Kr, mm/day) concentrations to evaluate the tolerance and growth behavior of T. aestivum, Tuber mesentericum, Tuber uncinatum and Tuber macrosporum strains. Based on these results, the most resilient and high-performing strains (T. aestivum and T. mesentericum) were selected for further experimentation. Subsequently, these strains were cultivated in shake flasks using OMW-based media (addition of OMWs at 0–50%, v/v) with different dilution levels, allowing detailed assessment of growth kinetics, substrate consumption, phenolic compounds degradation, decolorization and biomass production under controlled but small-scale conditions. Finally, the process was scaled up with T. mesentericum into a stirred-tank 3.6 L bioreactor on glucose-based media with 50% v/v OMWs, enabling precise monitoring of glucose consumption, phenolic compounds removal and biomass production and investigating the strain’s capacity to grow and metabolize OMW at a large-scale level. Another objective of the study was to characterize the biochemical profile of the produced mycelial biomass, including dry mass, lipids, proteins, polysaccharides, fatty acids, antioxidants, triterpenoids, phenolic acids and glucans (total, α- and β-glucans). Investigation of the adaptation of Tuber mycelia on OMW-based media across the aforementioned cultivation processes could provide novel insights into the truffle production performance, contributing to the development of a sustainable fungal bioprocess aimed at the production of an alternative mushroom product capable for human nutrition and functional food development.
2. Materials and Methods
2.1. Fungal Strains
Four Tuber spp. mushroom strains were used in this study: T. aestivum (AMRL 364), T. mesentericum (AMRL 365), T. uncinatum (AMRL 367) and T. macrosporum (AMRL 368), obtained from the Laboratory of Edible Fungi, Institute of Technology of Agricultural Products, Hellenic Agricultural Organization—Dimitra. These strains are native fungal species originally collected from Boeotia and Northern Greece. They were stored on potato dextrose agar (PDA, Condalab, Madrid, Spain) at T = 4 ± 1 °C and were routinely sub-cultured to ensure their viability.
2.2. Measurement of Mycelial Linear Growth Rate
A preliminary screening of the four Tuber strains was performed by assessing their growth rate on Petri dishes containing a glucose medium (control, 0% v/v OMW) and glucose syrup media enriched with OMW at concentrations of 25, 50 and 75% (v/v) or 2.5, 5.0 and 7.5 g/L total phenolic compounds, respectively. The culture media were prepared using OMW obtained from a three-phase olive oil processing facility located in the industrial zone of the Holy City of Messolonghi, Western Greece and was collected during a single production period, thereby minimizing batch-to-batch variability. The OMW was stored at T = 4 ± 0.5 °C prior to use and the OMW solid particles were removed through filtration and subsequent centrifugation at 15,000 rpm, T = 4 °C for 30 min. Glucose syrup medium (GYSA) was prepared by supplementing all media with agar and a carbon source (expired commercial glucose syrup; 68 g sugars per 100 g product, produced by a Greek company in Larissa, Greece). The glucose syrup was added to achieve a final concentration of 30 g/L total sugars. Additionally, the GYSA contained a nitrogen source (yeast extract; Biokar, Beauvais, France) and essential mineral salts (KH2PO4, 7 g/L; Na2HPO4·2H2O, 2.5 g/L; MgSO4·7H2O, 1.5 g/L; (NH4)2SO4, 6 g/L; CaCl2·2H2O, 0.15 g/L; FeCl3·6H2O, 0.15 g/L; MnSO4·H2O, 0.06 g/L; ZnSO4·7H2O, 0.02 g/L; all from Merck, Darmstadt, Germany) and agar at 20 g/L.
The OMW used in the experiments had an initial sugar concentration of 23 g/L (DNS assay [39]), total phenolic compounds of 10 g/L [40] and a pH value of 4.84 (Hanna HI2002-02 pH-meter, Hanna Instruments, Nușfalău, Romania). The culture media were autoclaved at T = 121 ± 1 °C (1.1 atm) for at least 20 min. Each Petri dish was inoculated by placing a 9 mm mycelial plug at its center. After inoculation, the plates were incubated in a growth chamber (DRAWELL, Model DW-LBI-400, Shanghai, China) at T = 25 ± 1 °C. For each substrate and fungal strain, five replicates were prepared. Colony diameter was measured along two perpendicular axes once the mycelial margin had extended to 10 mm and the mycelial growth rate (Kr, mm/day) was then calculated.
2.3. Shake Flask Experiments: Broth Analyses and Biomass Production Determination
Based on the outcomes of the preliminary screening experiments, the fungal strains and OMW concentrations showing the most favorable growth rates were selected for subsequent submerged fermentation trials. Two strains, T. aestivum and T. mesentericum, were further examined in submerged fermentation to assess biomass production, substrate utilization and their ability to remove phenolic compounds and reduce color. Liquid GYS medium (prepared as previously described, without agar) was used as the control, along with GYS media supplemented with 25 and 50% (v/v) OMW. In all cases, the initial glucose concentration was adjusted to 30 g/L by appropriately diluting the expired glucose syrup into the medium. A total of 30 mL of medium was dispensed into 100 mL Erlenmeyer flasks and sterilized at T = 120 ± 0.5 °C for 20 min. After cooling to room temperature, flasks were inoculated with 9 mm mycelial plugs and incubated at T = 25 ± 1 °C under agitation (120 ± 5 rpm) for 25 days. pH was measured using a Hanna HI2002-02 meter (Hanna Instruments, Nușfalău, Romania). The initial pH of all media after sterilization (T = 121 ± 1 °C, 20 min) was 6.5 ± 0.1. Substrate utilization was assessed through glucose consumption (Glcc, g/L) and was quantified using the 3,5-dinitro-2-hydroxybenzoic acid (DNS) method [39]. Phenolic content was measured using the Folin–Ciocalteu (FC) assay, following the procedure of Slinkard and Singleton [40]. Total phenolic compounds (TPC, g/L) were expressed as gallic acid equivalents per liter of medium. Decolorization was determined by measuring the absorbance of OMW-based broth samples at 395 nm [41]. Mycelia biomass was collected by vacuum filtration using No. 2 Whatman filter papers (Kent, UK) and subsequently rinsed twice with distilled water. Dry biomass (X, g/L) was determined gravimetrically after drying the harvested and washed mycelia at T = 60 ± 5 °C in an Elvem drying oven (Spata, Attica, Greece) until constant weight was reached. Final dry weight measurements were carried out using a Kern AGB balance (Balingen, Germany).
2.4. Bioreactor Experiments
Based on the favorable performance observed in the shake flask experiments (biomass production and substate consumption), T. mesentericum was selected for scale-up and its cultivation was subsequently carried out in a bioreactor system using previously described GYS medium, either as a glucose syrup medium (0%, v/v OMW) or as an OMW-based medium (addition of OMWs at a ratio of 50%, v/v). The fermentation was performed in a 3.6 L bench top bioreactor (Labfors 4, infors HT, Bottmingen, Switzerland) with a working volume of 2.16 L (60%, v/v) at an incubation temperature of T = 25 ± 1 °C. The agitation speed was 160 ± 5 rpm and the flow rate of the air supply was 2.16 L/min. The media were autoclaved and aseptically inoculated with fungal hyphae. During cultivation, sampling was conducted every 5 h to assess substrate components consumption and pH changes with complete glucose consumption occurring at approximately 70 h and 60 h of cultivation, respectively. In these cases, glucose concentration was quantified by HPLC analysis using a Waters Alliance 2695 HPLC apparatus (Waters Corporation, Milford, MA, USA) equipped with RI (2414 Refractive Index) detector and a 30.0 cm × 7.8 mm Aminex HPX-87H column (Bio-Rad, Hercules, CA, USA). The mobile phase used was H2SO4 at 0.005 M with a flow rate of 0.5 mL/min; the column temperature was 60 ± 0.5 °C and the injection volume 20 μL. Compounds were detected, identified and quantified according to standard curves. Before HPLC analysis, OMW-based samples were mixed 1:1 (v/v) with 5% (w/v) trichloroacetic acid (TCA) overnight to precipitate proteins prior to analysis. Phenolic content and decolorization were determined as previously described (Section 2.3). Afterwards, the biomass was subjected to several analyses after they were frozen (T = −20 ± 1 °C), dried by a Heto LyoLab 3000 freeze-dryer (Heto-Holten Als, Lillerod, Denmark) and milled.
2.5. Analyses of Biomass from Bioreactor Cultures
Intracellular polysaccharides (IPS, g/L) were quantified after acid hydrolysis following the methodology of Diamantopoulou et al. [42]. Dried mycelia (100 mg) were hydrolyzed with 2.5 M HCl (Merck, Darmstadt, Germany) at T = 100 ± 1 °C for 20 min, neutralized with 2.5 M NaOH (Merck, Darmstadt, Germany) and filtered. IPS were quantified using the DNS method [39]. Saccharide composition was determined by HPLC using extracts prepared as described by Diamantopoulou et al. [43]. Thus, filtered aliquots of the samples neutralized with NaOH were HPLC analyzed, as described previously. The α-glucan content of mycelia was quantified using a Yeast Beta-Glucan Assay Kit (Megazyme, Wicklow, Ireland) following the manufacturer’s protocol. β-Glucan content was calculated as the difference between total glucans and α-glucans. Each sample was analyzed in triplicate.
Total cellular lipids (L, w/w) were extracted from dried mycelial biomass using a chloroform/methanol solution (2:1, v/v; Merck, Darmstadt, Germany) and quantified gravimetrically, following Folch et al. [44]. Fatty acid composition was analyzed by gas chromatography (GC) using a Varian CP-3800 system equipped with a flame ionization detector (Agilent Technologies, Santa Clara, CA, USA) and an Agilent J&W DB-23 capillary column (30.0 m × 0.32 mm, 0.25 μm film thickness; model 123-2332). Helium served as the carrier gas at a flow rate of 2.0 mL/min. The oven program began at T = 150 ± 1 °C with an 18 min hold, followed by an increase to 185 ± 1 °C at 5 °C/min and a 2 min hold. The temperature was then raised to 210 ± 1 °C at 5 °C/min, held for 2 min and finally increased to 240 ± 1 °C at 10 °C/min. Injector and detector temperatures were set at T = 260 ± 1 °C and 270 ± 1 °C, respectively. Fatty acid methyl esters (FAMEs) were identified by comparing retention times with an external standard mixture (Supelco 37 Component FAME Mix, CRM47885, Merck KGaA, Darmstadt, Germany). The relative content of each fatty acid (%, w/w) is expressed as a percentage of the total fatty acid peak area detected in the chromatograms.
The total nitrogen content of the mycelial biomass was quantified using the total Kjeldahl nitrogen method. Protein content was then estimated by applying the standard nitrogen-to-protein conversion factor of 4.38.
Samples of mycelium were further analyzed regarding their chemical composition, total phenolic compounds and antioxidant activity. Methanolic extracts were prepared as follows: at a 1:5 (mg/mL) ratio using 80% methanol and sonicated (SKYMEN, JP-060S, Shenzhen, China) for 60 min at T = 25 ± 0.5 °C, followed by agitation and centrifugation (3500 rpm, 15 min, T = 25 ± 0.5 °C; Micro 22R, Hettich, Germany). Total phenolic compounds (TPCs) were determined using the Folin–Ciocalteu assay. Antioxidant activity was evaluated based on the sample’s ability to reduce Fe3+ to Fe2+ ions, according to the method described by Benzie et al. [45]. The results were expressed as mmol Trolox equivalents per 100 g of dry matter. Triterpenoid content was determined following the method of Fan and He [46], with minor modifications, and was expressed as milligrams of ursolic acid equivalents per gram of dry weight (mg UA/g d.w.). The identification and quantification of phenolic compounds in extracts were carried out using the Shimadzu Nexera HPLC instrument equipped with a pump (LC-40D), a diode-array detector (SPD-M40) detector and a Zorbax Eclipse XDB-C18 column (5 μm, 4.6 × 250 mm; Agilent Technologies, USA). The mobile phase consisted of 0.2% (v/v) orthophosphoric acid in DDW (solvent A), MeOH (solvent B) and ANC (solvent C). The gradient elution program was employed as follows: 2% B, 2% C initially, 25% B, 25% C 0–40 min, 30% B, 30% C 40–45 min, 50% B, 50% C 45–60 min, 2% B, 2% C 60–72 min, 2% B, 2% C 72–90 min (equilibration). The chromatographic analysis was conducted at a flow rate of 1 mL/min with an injection volume of 10 μL. Compound identification was based on a combination of retention time and spectral matching (200–600 nm). The quantification of each compound was performed by using external calibration curves constructed with analytical standards comparing sample peaks with those of analytical standards at 280 nm.
2.6. Data Analysis
All experiments were independently repeated twice (biological replicates). Within each biological replicate, multiple independent technical measurements were performed, including five Petri dishes, three shaken flasks and two bioreactor cultures per experimental condition. Results are reported as mean values, and standard deviations (<5%) were calculated across biological replicates, after averaging technical replicates. For experiments comparing two conditions (e.g., two substrates under identical operating conditions), Student’s t-test was used to compare means. For experiments involving multiple substrates and strains, one-way analysis of variance (ANOVA) was applied for each strain separately, followed by post hoc comparisons as appropriate. Statistical analyses were performed using JMP 7.0.1 (SAS Institute, Cary, NC, USA) with a significance level of α = 0.05.
3. Results and Discussion
3.1. Initial Screening with Mycelial Growth Response (Kr) to OMW-Enriched Medium
Initial screening of mycelial growth across increasing OMW concentrations revealed clear species-dependent differences in tolerance and metabolic adaptability. The Tuber strains exhibited distinct responses to increasing OMW concentrations, revealing clear interspecific differences in tolerance. Fungal growth (mycelial growth rate Kr, mm/day; Table 1) was monitored to evaluate the impact of OMW supplementation. T. mesentericum showed the strongest positive response to OMW, with Kr values increasing from 8.7 ± 0.1 mm/day in the control to 12.3 ± 0.3 mm/day at 25% OMW, followed by a slightly reduced but still enhanced growth rate at 50% (11.8 ± 0.26 mm/day) and sustained growth even at 75% OMW (10.2 ± 0.2 mm/day). This pattern indicates a high tolerance of the fungus to the phenolic load of OMW and suggests a potential detoxification and biotransformation capacity of the implicated strain. In contrast, T. aestivum displayed restricted growth tolerance to OMW concentrations, with growth stimulated only at 25%, v/v OMW added into the medium (8.6 ± 0.4 mm/day compared to 7.4 ± 0.1 mm/day in the control), followed by reduced growth (7.8 ± 0.1 g/L) at 50%, v/v and complete inhibition at 75%, v/v OMW. T. uncinatum and T. macrosporum were even more sensitive; both species showed reduced growth at 25%, v/v OMW and no detectable growth at 50 and 75%, v/v OMW, demonstrating low phenolic tolerance. The observed species-dependent growth rates are consistent with the findings of Diamantis et al. [30], who reported major differences among basidiomycetes exposed to OMW. In that study, species such as Agrocybe cylindracea and Lentinula edodes exhibited reduced growth at elevated OMW concentrations (up to 50%, v/v), whereas others (including Pleurotus sapidus, Ganoderma adspersum, T. aestivum and T. mesentericum) displayed moderate to high tolerance. Gwon et al. [4] measured the mycelium growth in malt extract agar and potato dextrose agar media at four pH conditions: the highest mycelial growth occurred at pH 6.0 in both PDA and MEA media, but they were still lower than the results of this study (under 6 mm/day). The strong tolerance and continuous growth presented by T. mesentericum and the moderate resilience of T. aestivum suggest that these Tuber strains possess unexplored metabolic capacities enabling survival under high phenolic and organic load conditions. Thus, they could serve as promising candidates for the development of advanced bioremediation strategies for OMW.

Table 1.
Linear growth rate (Kr, mm/day) of macrofungi T. aestivum, T. mesentericum, T. uncinatum and T. macrosporum on solid nutrient medium supplemented with OMW (0, 25, 50 and 75%, v/v) at T = 25 ± 1 °C. Each point is the mean value of at least five technical replicates per biological replicate, SD (<5%) across two independent biological replicates.
3.2. Kinetic Study of Fungal Growth in Shake Flask Cultures with OMW
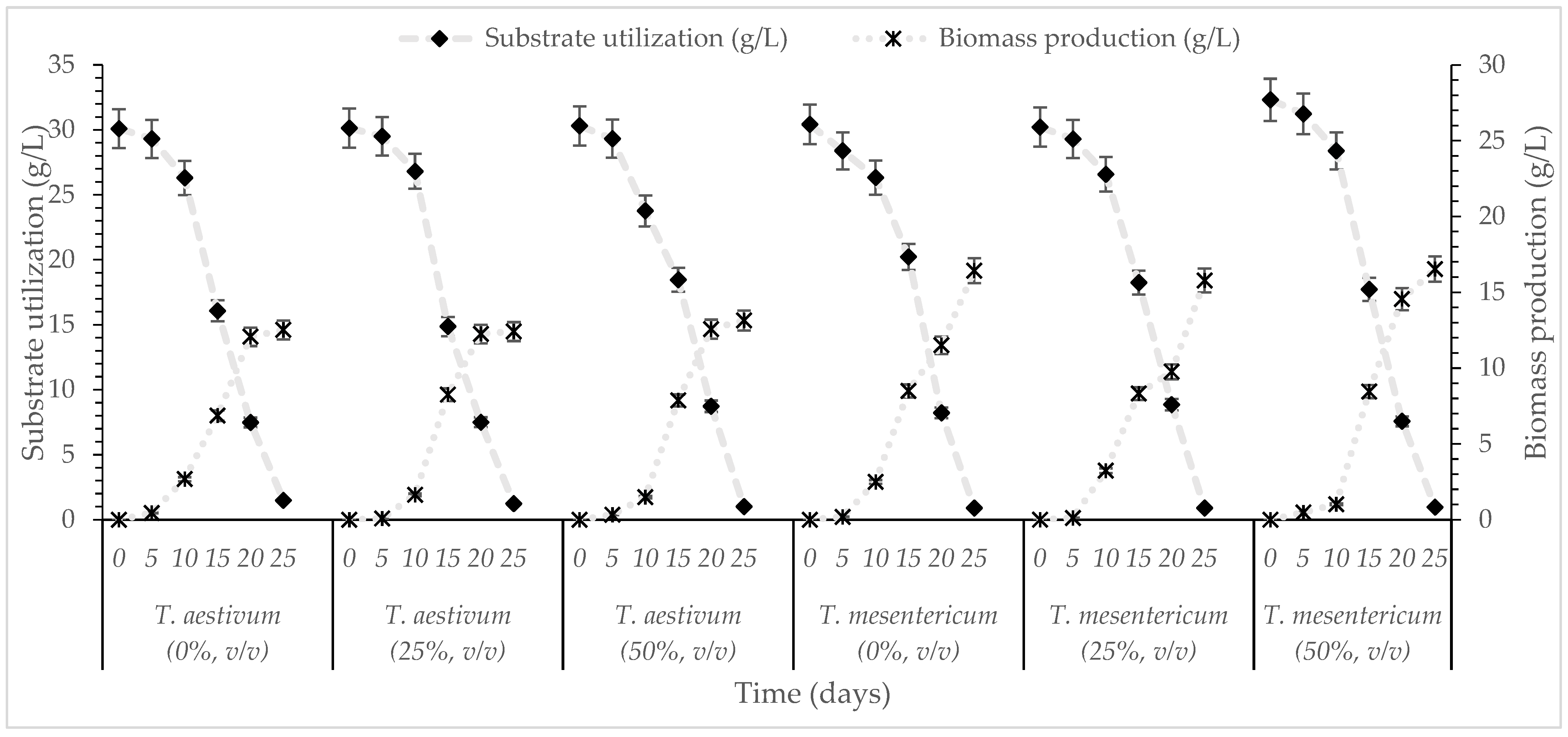
The scale-up of the bioprocess subsequently involved the batch shake flasks cultivation of the two selected fungal strains, T. aestivum and T. mesentericum, on OMW-based media supplemented with expired glucose syrup to a final glucose concentration of 30 g/L, under varying OMW concentrations (0, 25 and 50%, v/v) for a period of 25 days. The kinetics of dry biomass production of the fungal strains and glucose consumption for each distinct OMW concentration examined are illustrated in Figure 1.
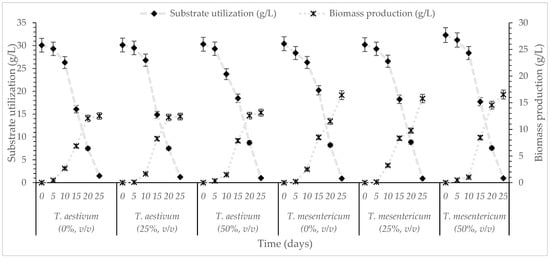
Figure 1.
Kinetics of substrate utilization and biomass production during cultivation of T. aestivum and T. mesentericum in liquid cultures with 0, 25, or 50% (v/v) OMW and initial concentration of glucose 30 g/L. Each point is the mean value of at least three independent measurements per biological replicate, SD (<5%) across two independent biological replicates.
According to the results, T. aestivum and T. mesentericum exhibited their highest dry biomass production on day 25 under these agitation-enhanced aerated conditions, reaching 12.5 and 16.0 g/L, respectively, regardless of the OMW concentration, while residual glucose levels were below 2 g/L. Although OMW is characterized by high phytotoxicity due to elevated polyphenolic content [47], numerous studies suggest that several fungi are capable not only of tolerating but also of growing effectively in OMW-enriched cultures. For instance, Pleurotus ostreatus achieved 5.7 g/L dry biomass in undiluted OMW and 5.0 g/L biomass in 50% (v/v) diluted OMW [48]. G. resinaceum showed enhanced biomass accumulation in medium containing 0.8 g/L of phenolic compounds [20], Ganoderma applanatum grew efficiently in 20% OMW [49] and Ganoderma carnosum demonstrated strong growth in 25% OMW [50]. In comparison, earlier studies reported 8.2 g/L of biomass for Ganoderma grown on distillery wastewater in shake flask cultures [51]. Similarly, P. ostreatus produced less than 13 g/L of mycelial in shake flask fermentations when cultivated on various carbon sources (including fructose, glucose, mannose and trehalose) [52], highlighting the moderate biomass yields typically obtained in submerged cultures of edible fungi. Our findings are similar or higher than those mentioned in the international literature and consistent with previous studies reporting enhanced biomass production under agitated conditions compared to static cultures [42]. Specifically, Flammulina velutipes reached up to 20.0 g/L and G. applanatum produced between 12.0 and 15.0 g/L under agitated conditions. Sarris et al. [31] investigated the growth of several ascomycetes (Morchella vulgaris, Morchella elata, T. aestivum) and basidiomycetes (L. edodes, Agaricus bisporus) in media containing glucose (30 g/L), glycerol (30 g/L), or various industrial waste-based substrates and observed that cultures grown with glycerol as the principal carbon source exhibited reduced biomass yields (1.5–4.2 g/L), compared with glucose-containing media (4.5–9.6 g/L) and especially with waste-derived substrates, which supported the highest biomass production (10.0–14.0 g/L). Yeasts were also shown to produce noticeable biomass production in phenolic-rich environments despite the presence of these recalcitrant compounds in the medium [53,54], while, interestingly, the biosynthesis and production of several metabolites can be stimulated during growth on (phenol-rich) OMW-based media, in comparison to the growth on the “blank” experiments (no presence of OMWs into the medium) [54,55]. Overall, the findings of this study demonstrate the strong growth capacity and substrate utilization efficiency of Tuber spp., indicating their strong potential for OMW bioconversion processes. In addition, the submerged cultivation of truffles has the advantage that the liquid media provides a more sterile, pure culture compared to the carposomes cultivated outdoors that are exposed to contaminants, providing a mycelium that can be used for the production of high-value ingredients available for functional foods, nutraceuticals and flavorings.
3.3. Fungal Efficiency in Phenolics Compounds Removal and Decolorization
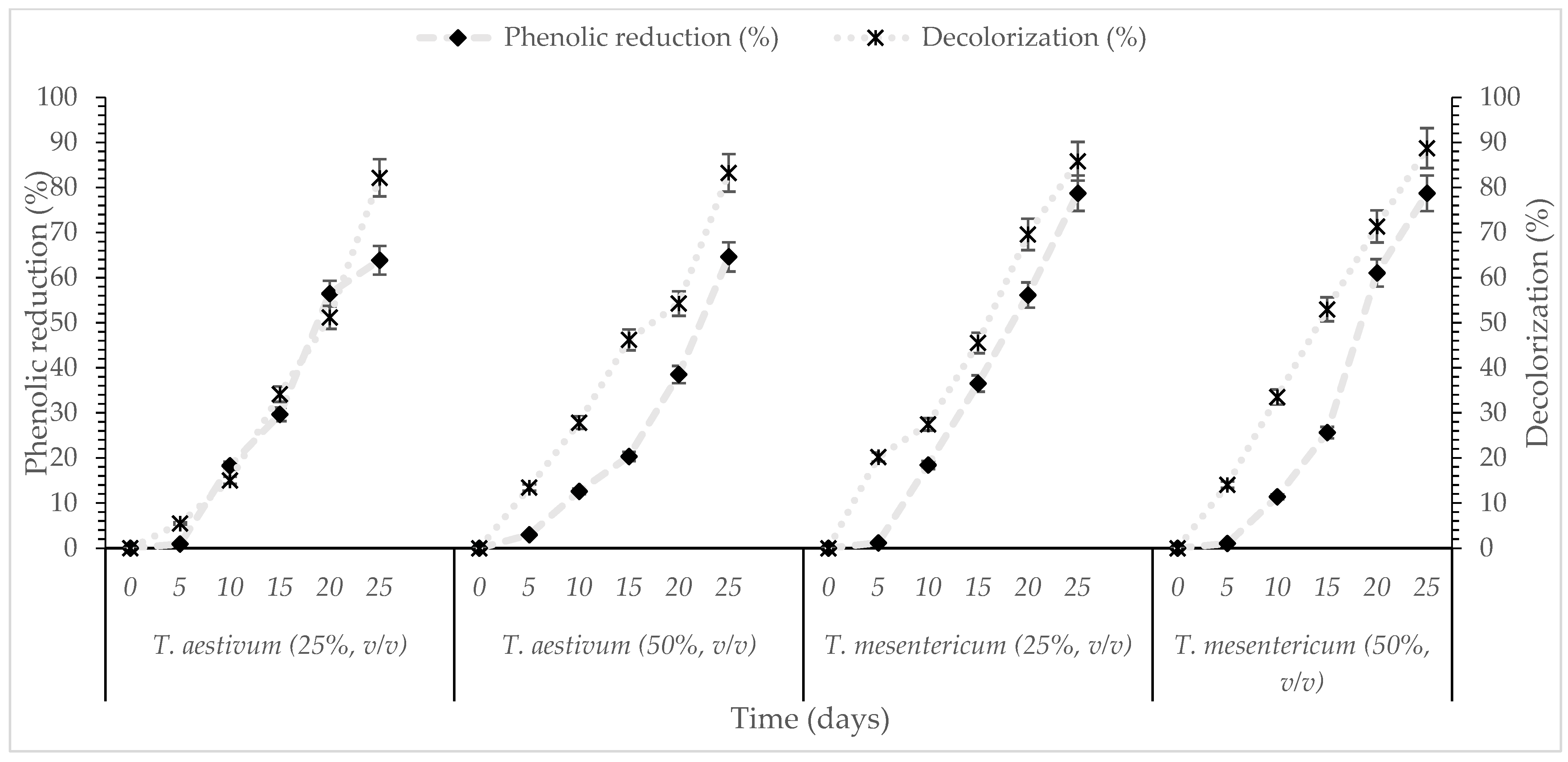
T. mesentericum and T. aestivum exhibited a strong ability to degrade phenolic compounds and substantially decolorize OMW under both concentration conditions, even at the highest (50%, v/v) phenolic concentrations. As shown in Figure 2, the results demonstrate a consistent pattern of phenolic removal (>63%) by both fungal strains, regardless of the initial phenolic concentration. More specifically, at the end of the fermentation period T. mesentericum achieved ~78% and T. aestivum ~64% phenolic compounds removal in both concentrations of OMW (25 and 50%, v/v). Increasing the concentration of initial phenolics from 2.5 to 5 g/L had no impact on removal efficiency, indicating that the fungal enzymes remained active and stable despite the higher OMW concentrations. In contrast, the decolorization reached higher efficiencies (>80%) (Figure 2). These observations are consistent with previous findings: G. applanatum has been shown to effectively degrade phenolic compounds (up to 94%) while removing 73% of the color in OMW [49]. Pleurotus pulmonarius has also demonstrated remarkable detoxification potential, achieving as much as 95% phenolic reduction and 85% decolorization, particularly when cultivated under carbon-rich conditions that stimulated ligninolytic enzyme production [19]. Earlier work further reports that Pleurotus sajor-caju is capable of degrading both phenolic and non-phenolic constituents of OMW, achieving removal efficiencies between 64.6 and 88.4% [56,57]. Strong detoxification activity has also been observed for G. resinaceum, which achieved 94.5% phenolic reduction and 76.5% decolorization in static cultures supplemented with glucose [20]. Consistent patterns have been reported for Pleurotus citrinopileatus, which reached 86.4% phenolic removal and 79.1% decolorization after 22 days of fermentation in 25% (v/v) OMW [22]. Significant removal of phenolic compounds from OMW has been reported for multiple mushroom strains (A. cylindracea, P. sajor-caju, P. sapidus, L. edodes, F. velutipes, G. adspersum, T. aestivum and T. mesentericum), with reductions approaching 80% at 15 and 35% v/v OMW [30]), while overall, higher (edible/medicinal) fungi are very efficient microbial cell factories amenable to be used in the treatment, valorization, and control of intensively colored and toxic abundant agro-industrial effluents (i.e., olive mill wastewaters—as is the present study, table olive-processing wastewaters, winery and other alcoholic beverage production process wastewaters, pulp bleach plant effluents, paper-processing wastewaters, etc.) due to their rich ligninolytic enzymatic arsenal [58].
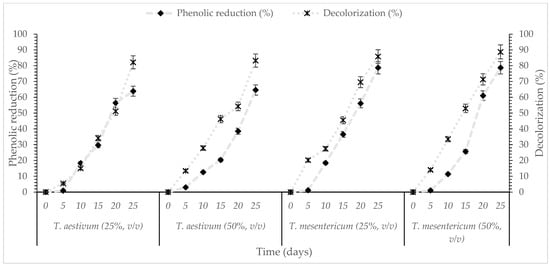
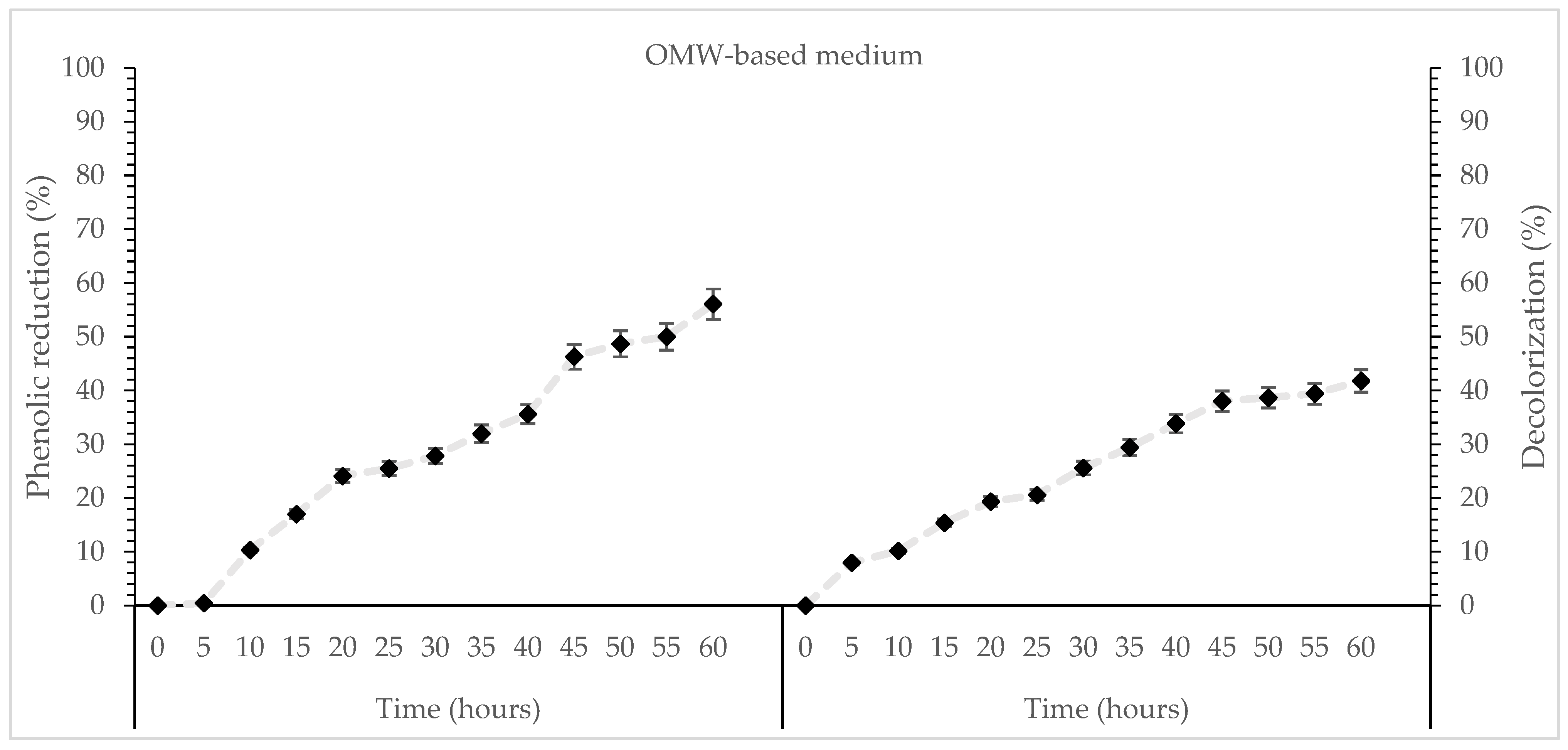
Figure 2.
Kinetics of % phenol reduction and % decolorization during cultivation of T. aestivum and T. mesentericum in liquid cultures with 0, 25, or 50% (v/v) OMW and initial concentration of glucose 30 g/L at T = 25 ± 1 °C per biological replicate, SD (<5%) across two independent biological replicates.
The differing rates of phenolic reduction and decolorization indicate that the various pollutant components of OMW differ significantly in their susceptibility to biodegradation. However, reduction in decolorization efficiency was observed at higher initial phenolic concentrations, potentially being attributed to substrate inhibition, elevated phenolic toxicity, or partial loss of activity of oxidative enzymes under heightened chemical stress. Submerged fungal fermentation is increasingly suggested as an environmentally friendly approach capable of efficiently converting phenolic wastewater into less toxic forms [58]. Although species like T. aestivum and T. mesentericum have only rarely been investigated for this purpose, they indicate promising potential pending further experimental validation under submerged fermentation conditions.
3.4. Scale-Up Cultivation in Bioreactors Using OMW-Based Medium
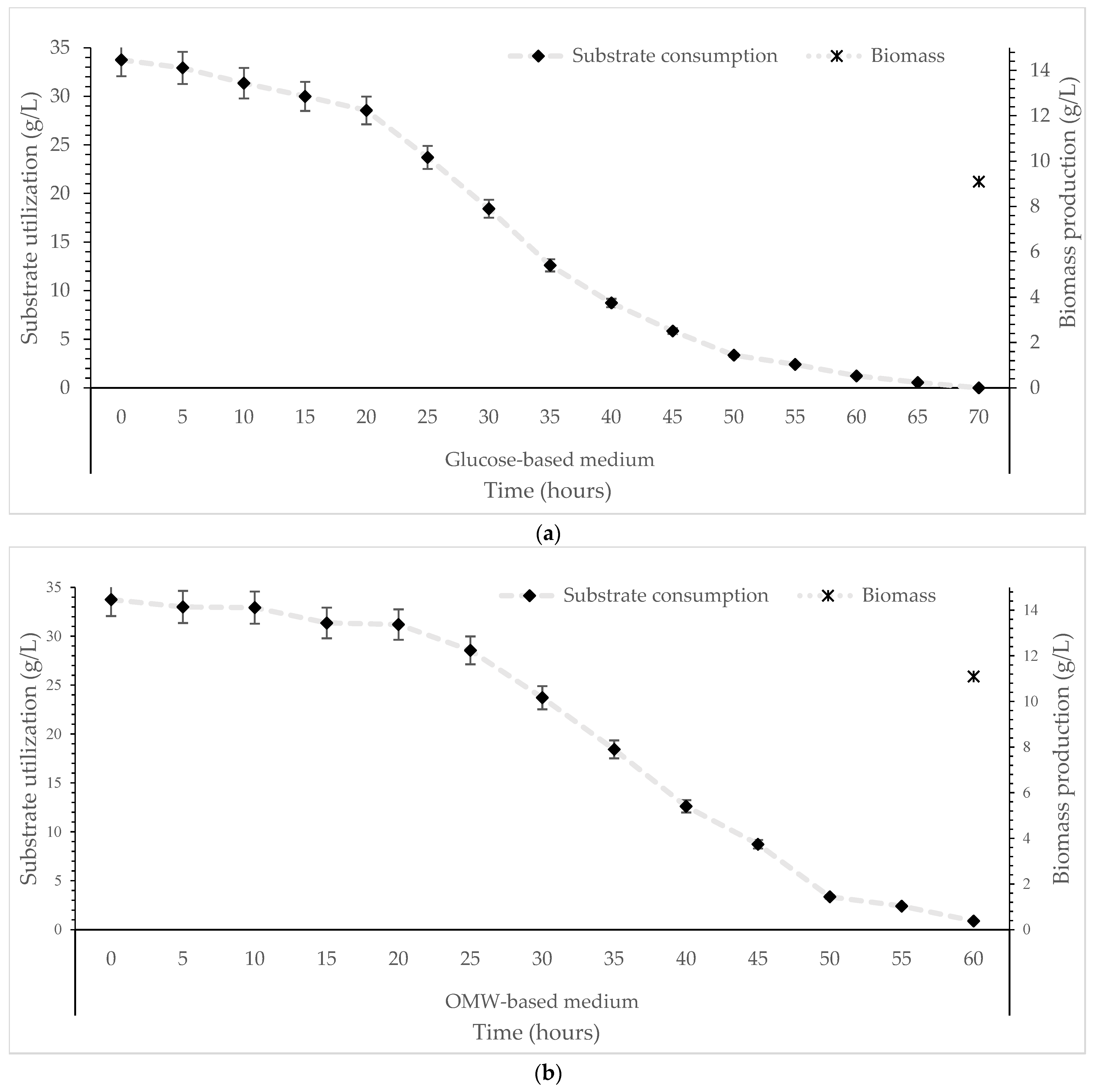
The scale-up of the bioprocess was subsequently carried out in a stirred-tank bioreactor (active volume 2.16 L), where the mushroom strain T. mesentericum was batch-cultivated using OMW-media (50%, v/v), supplemented with expired glucose syrup (initial glucose at 30 g/L). A glucose-based medium (0%, v/v OMW) was used as a control experiment (no phenolic compounds into the medium). The kinetics of glucose consumption under both conditions tested are presented in Figure 3a,b. The OMW-based media supported biomass production at levels comparable to the control experiment, producing approximately 11.1 g/L of mycelial biomass (Figure 3b), whereas the glucose syrup-media yielded around 9.1 g/L (Figure 3a) (Table 2). Residual substrate levels decreased progressively throughout the process, which was terminated when glucose concentration reached nearly 0.5 g/L (Figure 3a,b), reflecting efficient substrate utilization by T. mesentericum.

Figure 3.
Kinetics of substrate utilization and final biomass production during cultivation of T. mesentericum in bioreactor on solid nutrient medium supplemented with (a) 0% or (b) 50% (v/v) OMW and initial concentration of glucose 30 g/L at T = 25 ± 1 °C. Each point is the mean value of at least three independent measurements per biological replicate, SD (<5%) across two independent biological replicates.

Table 2.
Chemical composition of T. mesentericum biomass from bioreactor on nutrient medium supplemented with OMW (50%, v/v) at T = 25 ± 1 °C. Each point is the mean value of at least three independent measurements (mean ± SD).
These results confirm that glucose-enriched OMW-based substrates can function effectively as an alternative carbon source, as their complex mixture of phenolic compounds and residual carbohydrates provides sufficient nutrients to support biomass synthesis. The comparable biomass obtained after cultivation on both OMW-based and glucose-based media (control) further demonstrates that, despite its chemical complexity, OMW does not impose a growth-limiting effect on the fungus. Comparisons with other Tuber-related culture systems highlight the variability in biomass formation across species, substrates, and cultivation strategies. In Tuber sinense, Tang et al. [8] reported a biomass amount of 24.1 g/L when cultured in flasks with 125 g/L of initial sucrose, while Liu et al. [7] achieved 23.9 g/L using an optimized medium containing glucose, yeast extract and peptone. Higher yields have also been documented in fed-batch systems; Liu et al. [59] reported 41.6 ± 2.4 g/L biomass at 80 g/L sucrose, underscoring the impact of carbon abundance and feeding strategies on fungal growth. In contrast, Tuber borchii generally exhibits lower biomass concentrations under submerged conditions: Chen et al. [60] reported a maximum biomass of 5.4 g/L using sucrose (80 g/L) with yeast extract (20 g/L), while Fu et al. [61] observed biomass values of 2.1 ± 0.1 g/L, which increased to 3.0 ± 0.2 g/L upon medium optimization. These comparisons indicate that the biomass yield of T. mesentericum grown on OMW-based medium (despite the complexity and potential inhibitory nature of the substrate) is well within the range reported for submerged truffle cultures. The ability of T. mesentericum to achieve biomass levels comparable to glucose controls while utilizing an agro-industrial by-product highlights its robustness and positions OMW as a promising low-cost substrate for sustainable truffle mycelial biomass production. As observed in the present study, the final biomass obtained in the bioreactor was lower compared to the shake flask cultures, a trend that is also reflected in previous reports. For instance, Kim et al. [37] reported 11.6 g/L of Agrocybe aegerita biomass in a stirred-tank bioreactor, whereas shake flask cultures reached 25.0 g/L. These findings collectively suggest that although bioreactor cultures often result in lower final biomass concentrations, likely due to losses during the downstream filtration processes, they offer significantly higher productivity rate and process efficiency due to their accelerated growth dynamics and controlled operating conditions. In similar types of experimental designs, scale-up in 3.0, 10, and 20 L bioreactors of other filamentous fungi (Zygomycetes species of Mucor hiemalis, Cunninghamella elegans and Thamnidium elegans) did not reveal significantly higher biomass productions in the bioreactors compared to the shake flask experiments [62,63], suggesting flask fermentations provide a reliable, quick and inexpensive culture configuration very useful for the study of the fungal physiology and the process and medium optimization.
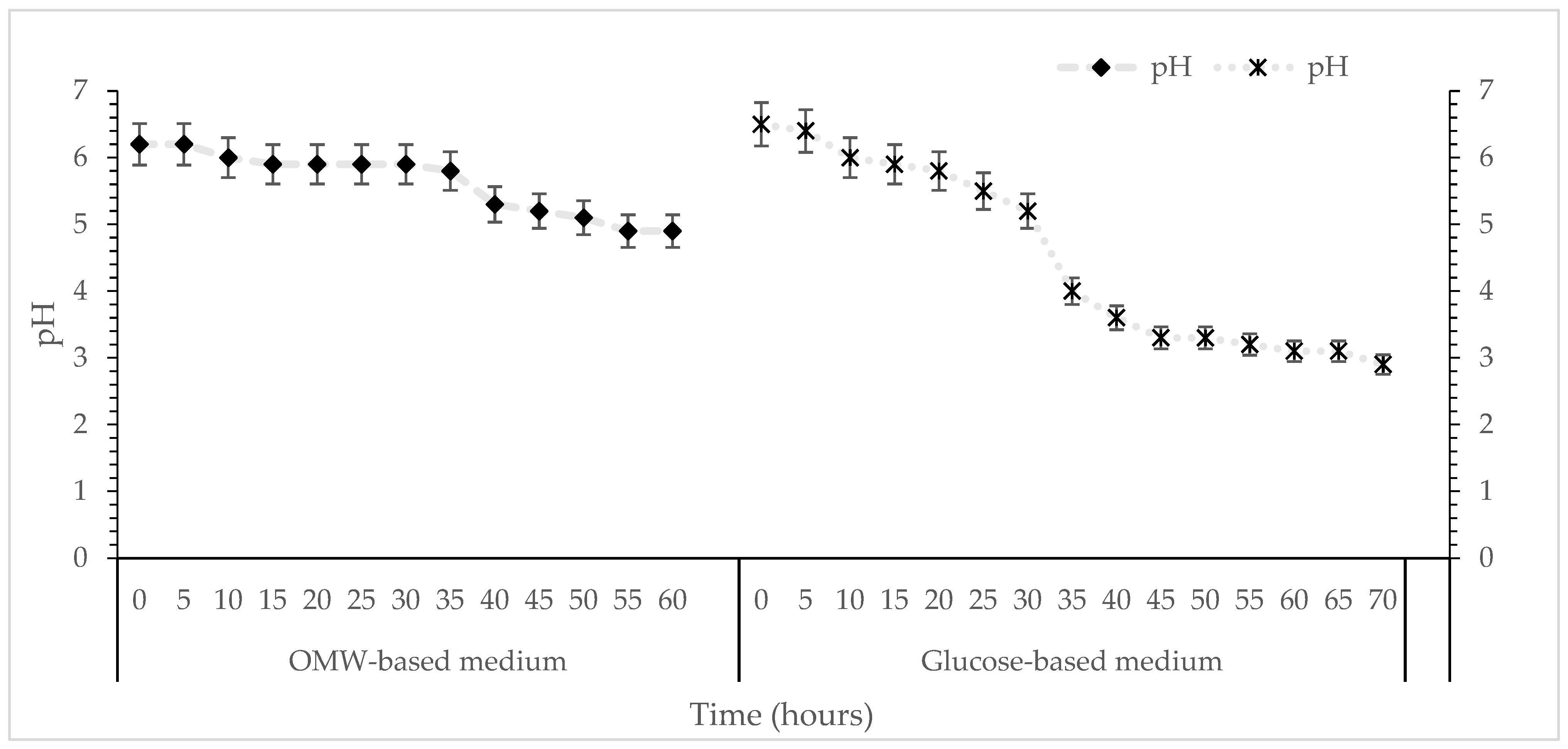
Continuous pH monitoring indicated a gradual decrease during cultivation from 6.0 to 4.9 for OMW-based media and 2.9 for glucose syrup media (control), reflecting metabolic activity (Figure 4). The pH of the culture medium can influence cell membrane function, cellular morphology and structure, nutrient uptake, as well as metabolite biosynthesis. These changes are consistent with carbohydrate catabolism and organic acid secretion, both of which are characteristic of truffle mycelial growth under submerged conditions. The milder acidification observed in the OMW-based medium may be attributed to its content of organic acids, phenolics and mineral ions, which may act as a natural buffering system, moderating pH fluctuations during fungal metabolism and thereby providing a more chemically stable growing environment compared to the glucose syrup medium. Tuber species are generally associated with alkaline or neutral soils; however, their preferred pH range varies considerably across species [64]. For instance, the optimal pH for T. borchii mycelial growth is reported as 6.5, even though its fruiting bodies most commonly develop in neutral soils [65]. T. japonicum shows optimal growth at pH 5.0–6.0, consistent with its occurrence in weakly acidic environments [66]. Similarly, T. magnatum exhibits optimal in vitro growth at pH 6.0, matching the pH typically observed in soils where its ascomata are found [67]. Recent work on T. koreanum indicates a preferred pH of 6.0 as well, with the best growth achieved at 25 °C [4]. More broadly, optimal temperature requirements also vary across the genus: T. melanosporum grows best at 25 °C [68], while T. magnatum prefers slightly lower temperatures of around 20 °C [69]. These studies collectively highlight that Tuber species exhibit distinct ecological and physiological preferences, particularly regarding pH and temperature. The moderate acidification observed in the OMW-based medium cultures of T. mesentericum is within the tolerance range documented for several Tuber species, supporting the observation that this species is capable of maintaining metabolic activity under mildly acidic conditions generated during submerged fermentation.
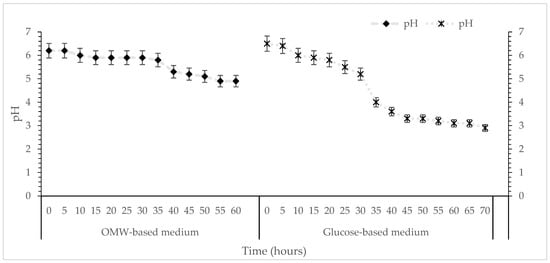
Figure 4.
Kinetics of pH during cultivation of T. mesentericum in bioreactor on solid nutrient medium supplemented with 0 or 50% (v/v) OMW and initial concentration of glucose 30 g/L at T = 25 ± 1 °C. Each point is the mean value of at least three independent measurements per biological replicate, SD (<5%) across two independent biological replicates.
The OMW medium also showed a decolorization of 42% and a phenolic compound reduction of 56% (Figure 5), which was lower than the values obtained in the shake flask cultures. This difference may be attributed to the shorter residence time in the bioreactor, resulting in reduced exposure of the fungus to phenolic substrates, as well as to the higher oxygen transfer and agitation rates, which can shift metabolism. The bioreactor operating conditions in the bioreactor may limit the production of oxidative enzymes responsible for phenolic degradation compared to the more static environment of shake flask cultures. Aggelis et al. [38] reported that P. ostreatus grown in bioreactor batch cultures using OMW achieved over 75% phenolic removal within 7 days, demonstrating the strong bioremediation potential of the species under controlled reactor conditions. In a similar study, Pleurotus flavido-alba cultivated in a laboratory-scale bioreactor on OMW achieved 70% decolorization and 51% reduction under controlled submerged culture conditions at room temperature (25 °C) after 19 days of fermentation [70].
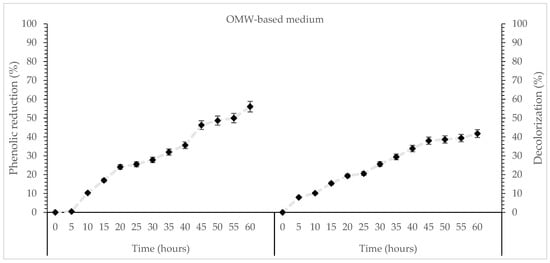
Figure 5.
Kinetics of % phenol reduction and % decolorization during cultivation of T. mesentericum in bioreactor on solid nutrient medium supplemented with 50% (v/v) OMW and initial concentration of glucose 30 g/L at T = 25 ± 1 °C. Each point is the mean value of at least three independent measurements per biological replicate, SD (<5%) across two independent biological replicates.
In summary of the above, from a production perspective, the biomass was lower than in long-term flask cultures, due primarily to the shorter cultivation time and metabolic adaptations to the dynamic conditions of large-scale cultivation. Nevertheless, the bioreactor offers significant advantages: precise control of pH, dissolved oxygen, temperature and agitation allow reproducible and scalable production. Maintaining aerobic conditions and optimizing nutrient delivery ensures consistent metabolic activity, critical for downstream applications and industrial-scale production. Moreover, bioreactor cultivation requires markedly less time, highlighting its substantially higher productivity (g/L/day) and efficiency in rapidly generating fungal biomass.
3.5. Mycelial Analyses
3.5.1. IPS Content, Saccharides Composition and Total Glucans
The IPS was quantified for both the mycelial biomasses derived from glucose syrup and OMW-based media, as IPS constitute an important group of bioactive metabolites with significant relevance to functional food/feed and pharmaceutical applications. Cultures grown in OMW exhibited an IPS content of 28.5%, w/w, not significantly higher than the 27.1%, w/w observed in glucose-based media (Table 2). This finding indicates that the phenolic load of OMW does not hinder carbohydrate metabolism; rather, it may stimulate metabolic routes linked to polysaccharide accumulation. The IPS levels obtained here are within the range commonly reported for truffle fermentation systems. For instance, T. magnatum cultured on soybean–red adlay substrates produced approximately 30.6% crude polysaccharides [71], whereas T. melanosporum grown under grain-based solid-state conditions yielded ~11.6% polysaccharides [72]. Such variability reflects the strong influence of substrate composition and cultivation mode on IPS biosynthesis. Glucose has repeatedly been identified as an efficient carbon source for IPS production in truffles, as demonstrated by Liu et al. [7] and the comparable yields observed here confirm that OMWs can support metabolic activity in the same way as conventional carbohydrate-rich media. Previous studies have shown that IPS production can be substantially enhanced through controlled nutrient strategies: for example, fed-batch sucrose supplementation increased T. sinense IPS to 2.9 ± 0.2 g/L [8], while G. lucidum responded to lactose feeding with a 40.4% increase in IPS yield [73]. Related work achieved IPS concentration of 4.0 ± 0.2 g/L (8.1%, w/w) in truffle species [59]. Comparable values have been reported in submerged cultures of other edible and medicinal fungi, including 4.4 g/L for P. pulmonarius [19] and 5.2 g/L for G. resinaceum cultivated on glucose-enriched OMW [20]. Overall, these comparisons highlight that T. mesentericum produces polysaccharides at levels equivalent to, or in some cases exceeding, those of established IPS-producing fungi.
The saccharide composition of the IPS fraction consisted primarily of glucose and mannitol (Table 2), with no qualitative differences between glucose-medium and OMW-medium that is of a saccharide composition similar to that of polysaccharides isolated/quantified in other edible/medicinal mushrooms [74]. Other saccharides/polyols identified in various concentrations in the mycelia, pilei and stipes of edible and medicinal fungi are trehalose, arabitol, myo-inositol and xylitol [74]. The consistent detection of mannitol in the IPS fraction across all cultivation conditions indicates that mannitol is a stable and recurrent component of the intracellular polysaccharide profile. As a major fungal polyol, mannitol functions both as a carbon-storage intermediate and as a redox-balancing molecule, supporting cellular homeostasis during polysaccharide biosynthesis [75]. The similarity observed between cultivation conditions suggests that the presence of OMW does not lead to major alterations in the overall polysaccharide profile under the conditions tested. These findings support the use of OMW as an alternative nutrient substrate based on comparable experimental outcomes rather than metabolic mechanisms. These results are in line with the observations of Diamantopoulou et al. [43], who reported that IPS composition varies with strain and imposed agitation, yet glucose remains the dominant sugar in most fungi due to the prevalence of glucan-type polysaccharides. Their study also demonstrated that mannitol and other polyols, such as xylitol, tend to accumulate mainly under static conditions and in specific species, functioning as metabolic intermediates linked to carbon storage and stress response. The detection of mannitol in our cultures, irrespective of medium, further supports the idea that polyol synthesis is a conserved metabolic feature rather than a medium-induced effect.
Beyond their quantitative significance, IPS represent one of the most important bioactive outputs of fungal metabolism due to their structural, nutritional and therapeutic roles. In particular, β-glucans are recognized for their antimicrobial, anticancer, hypoglycemic and immunomodulatory activities. They exert these functions through interactions with diverse innate and adaptive immune receptors [76]. Notably, in the present study, nearly 35–40% of the IPS fraction consisted of β-glucans (Table 2), a proportion that positions T. mesentericum among the more promising truffle species for bioactive glucan production. This observation is consistent with reports from T. maculatum and T. borchii, where stress-related metabolic responses in submerged culture enhanced antioxidant-associated polysaccharide synthesis [77]. The high β-glucan proportion, coupled with their structural features, highlights the potential of T. mesentericum IPS as a bioactive metabolite with immunomodulatory and therapeutic applications.
Together, these findings reveal that T. mesentericum not only maintains robust primary carbohydrate metabolism in OMW-based medium but also generates polysaccharides with high β-glucan content, underscoring its potential as a valuable producer of nutraceutical-grade fungal biomolecules. The ability of OMW to sustain or even stimulate IPS biosynthesis highlights an important synergy between fungal metabolism and waste-derived substrates, strengthening the case for integrating truffle mycelial cultivation into sustainable bioprocess frameworks.
3.5.2. Lipid and Fatty Acids
Lipid extraction and fatty acid profiling revealed distinct metabolic shifts between the two cultivation media. Total lipid content was substantially higher in OMW-grown mycelia (13.2%, w/w) compared with the glucose syrup medium (control) (9.5%, w/w) (Table 2), indicating that exposure to the phenolic load of OMW stimulates lipid biosynthesis in accordance with reports that have characterized the OMWs as “lipogenic” compounds; viz., media that increase the lipid in dry cell weight values during growth of various microorganisms in these substrates, compared to the “control” experiments (no OMWs added) [55]. Such an increase is consistent with reports showing that submerged fungal cultures often accumulate lipids when subjected to chemical or oxidative stress, as carbon flux is redirected toward membrane remodeling and storage lipid formation [31,36]. Such stress-induced lipid enrichment has also been reported in fungal cultures grown in phenolic-rich wastewaters [19,30], supporting the view that OMW triggers a coordinated metabolic response that includes increased lipid deposition [78]. Studies on specific species confirm this variability: Tuber texense contains approximately 2.8% (w/w) lipid [79], while T. melanosporum has been reported to reach around 8% (w/w) [80]. Even broader variability has been documented by Sancholle [81], who observed lipid contents ranging from 3.6 to 25.2% depending on growth stage. Among Asian truffles, T. latisporum, T. subglobosum, and T. pseudohimalayense contain 2.2–2.5 w/w total lipids [82]. More generally, reported lipid content in truffle fruiting bodies spans 1.8–7.4% (w/w) [83], with differences attributed to species, maturity, substrate availability and geographic origin. Taken together, these data highlight the inherently broad lipid variability in the genus Tuber, shaped by both environmental and developmental factors.
Beyond total lipid content, the quality of the lipid fraction also differed between treatments. Glucose-grown mycelia presented a proportion of saturated fatty acids (SFAs) equal to 21.6%, w/w, while cultures grown in OMW accumulated unsaturated fatty acids (UFAs) at 80.1%, w/w, with linoleic acid (C18:2) remaining the predominant fatty acid across all conditions at 62.0%, w/w (OMW-based media) and 58.7%, w/w (control), respectively (Table 3). The SFAs identified included lauric, myristic, pentadecanoic, palmitic, stearic, arachidic, behenic and tricosanoic acids, with palmitic acid consistently among the most abundant (15.6–16.0% w/w). Monounsaturated fatty acids (MUFAs) consisted mainly of oleic and nervonic acids, while polyunsaturated fatty acids (PUFAs) included linoleic acid, eicosadienoic acid and cis-4,7,10,13,16,19-docosahexaenoic acid. The relative enrichment of PUFAs in OMW-grown biomass aligns with well-documented adaptive responses in Basidiomycetes, where oxidative or phenolic stress enhances fatty acid desaturation to maintain membrane fluidity and mitigate toxicity [77]. This remodeling of the lipid profile indicates that T. mesentericum may reflect an integrated metabolic response to the compositional complexity of OMW. Comparable patterns are well established in truffle carposomes. Linoleic, palmitic and oleic acids (dominant in the present study) are among the major fatty acids found in fruiting bodies of various Tuber species [84]. According to Yan et al. [82], Chinese truffle species such as T. latisporum, T. subglobosum and T. pseudohimalayense contain substantial proportions of MUFAs (96–265 mg/100 g) and PUFAs (249–368 mg/100 g), with comparatively lower levels of SFAs (70–121 mg/100 g). Across desert and European truffles (including T. melanosporum), UFAs typically constitute more than 60% of total fatty acids [85,86], conferring cardioprotective and anti-inflammatory properties [87]. Species-specific differences are also evident: T. aestivum is rich in palmitic and elaidic acids [88], while T. magnatum tends to accumulate a higher proportion of SFAs [89]. In a classic analysis, T. texense contained 45.9% oleic, 38.0% linoleic, and 11.4% palmitic acid [79]. Apart from the fact that unsaturated and essential fatty acids enhance the nutritional value of biomass, the lipid fractions of mushrooms play an important role in the improvement of their organoleptic properties. The oxidation of unsaturated fatty acids like palmitoleic, oleic, linoleic and linolenic creates various compounds that are responsible for the distinct taste and aroma of mushrooms, including truffles, the most important being 1-octen-3-ol [72].

Table 3.
Fatty acid composition of lipids synthesized at T. mesentericum biomass from bioreactor on nutrient medium supplemented with OMW (50%, v/v) and the control (glycose medium) at T = 25 ± 1 °C. Each point is the mean value of at least three independent measurements (mean ± SD).
Overall, the lipid profile of T. mesentericum mycelium not only simulates but often exceeds the unsaturation levels typically found in natural truffles. This correspondence further supports the conclusion that phenolic-rich OMW stimulates lipid accumulation, a response frequently associated with fungal adaptive mechanisms under chemical or oxidative stress and triggers a targeted lipid remodeling response (enhancing both total lipid accumulation and the proportion of unsaturated fatty acids). This further underscores its suitability for the biotechnological exploitation of phenolic agro-industrial waste streams and its capacity to generate nutritionally and functionally rich fungal biomass.
3.5.3. Protein
Truffles are recognized for their considerable protein content, although their high market value limits their use as a daily dietary protein source [90]. In the present study, protein analysis revealed that biomass cultivated in OMW exhibited protein levels comparable to those produced in glucose syrup medium (approximately 35%, w/w) (Table 2), indicating that cultivation on OMW does not adversely affect protein synthesis compared to a conventional carbohydrate-rich substrate. The maintenance of similar protein concentrations across treatments indicates that T. mesentericum preserves its nitrogen assimilation and amino acid biosynthetic pathways even under the phenolic stress imposed by OMW. This stability highlights the species’ robust metabolic adaptability to chemically complex environments. Comparable protein concentrations have been reported in other truffle fermentation systems. T. magnatum mycelial cultures grown on soybean–red adlay substrate contained 22.9% (w/w) [71], while natural truffle species of the genera Terfezia and Tirmania commonly display protein ranges of 32–35% (w/w) and 8–29% (w/w), respectively [91,92]. Similar levels have also been reported for several Chinese truffle species, including T. latisporum, T. subglobosum, and T. pseudohimalayense, which contained 11–15% w/w protein [82]. Earlier analyses likewise demonstrated notable protein contributions in T. texense (16.1%, w/w) [79] and T. melanosporum (27.6%, w/w) [80].
Concluding, these findings indicate that T. mesentericum maintains a protein content consistent with that of other truffle species and that the presence of phenolic compounds in OMW does not compromise nitrogen metabolism. The ability to sustain high protein levels despite the inhibitory potential of OMW underscores the species’ strong physiological resilience and reinforces its suitability for biotechnological applications involving phenolic agro-industrial effluents, while simultaneously highlighting the potential of the produced mycelial biomass as a protein-containing fungal product.
3.5.4. Phenolic Content, Antioxidant Activity and Phenolic Acids
Biomass was also evaluated for its total phenolic content, antioxidant capacity (FRAP) and total triterpenoids (Tr) (Table 4). The present study demonstrates that cultivation of fungal mycelia in OMW, a substrate naturally enriched in phenolic compounds, markedly enhances the production and accumulation of bioactive metabolites compared to growth in a standard glucose syrup medium. This observation aligns with previous findings in truffles and other fungi, showing that phenolic-rich substrates can function both as nutrient sources and as metabolic elicitors, stimulating secondary metabolite biosynthesis.

Table 4.
Content of different bioactive groups and phenolic acids of T. mesentericum biomass from bioreactor on nutrient medium supplemented with OMW (50%, v/v) at T = 25 ± 1 °C. Each point is the mean value of at least three independent measurements (mean ± SD).
The TC of mycelia grown in OMW reached 1.9 ± 0.2 mg GAE/g d.w., almost double that of biomass grown in glucose syrup medium (1.0 ± 0.1 mg GAE/g d.w.) (Table 4). This increase highlights the effect of the cultivation substrate on phenolic synthesis. Similar concentration ranges have been reported for natural truffle species, which exhibit varying phenolic concentrations depending on species and environmental conditions. Villares et al. [93] reported TC of 1.9 mg/g d.w. in T. aestivum, followed by T. indicum and T. melanosporum (1.5 and 1.2 mg/g d.w., respectively). Even higher values have been found in the desert truffle T. nivea, which contains nearly tenfold higher TC compared to Tuber species [94]. Shah et al. [77] reported TC ranging from 1.1 to 2.9 mg GAE/g d.w. in bioreactor-based truffle fermentations using sucrose-supplemented MS medium at different cultivation stages, whereas Hsu et al. [71] recorded slightly higher values (2.1–2.4 mg GAE/g d.w.) for T. magnatum mycelia produced under solid-state fermentation on soybean–red adlay substrates. Additional evidence from fruiting bodies reinforces the variability of phenolic accumulation among truffle species: according to Beara et al. [89], aqueous and methanolic extracts of T. aestivum and T. magnatum exhibit moderate antioxidant activity corresponding to TC of 12–18 and 13–19 mg GAE/g d.w., respectively. The OMW-grown biomass produced in the present study therefore approaches the upper limit of phenolic accumulation reported for truffle mycelia. Comparable antioxidant activity profiles have also been widely documented in edible and medicinal mushrooms, where TC contribute substantially to the reducing capacity of fruiting bodies, mycelia and culture filtrates [95,96].
Antioxidant activity followed a similar pattern. The FRAP values were 19.8 ± 1.2 mg trx/g in OMW-grown cultures, substantially higher than 4.2 ± 0.3 mg trx/g in glucose syrup medium (Table 4). The enhanced FRAP likely reflects an intensified oxidative stress response induced by the phenolic environment, an adaptive behavior widely documented in edible and medicinal fungi [22,97]. Comparable antioxidant capacities have been reported: ethanol extracts of T. magnatum mycelium exhibited FRAP values of approximately 1.5 mg trx/g [71], while the desert truffle T. nivea displayed substantially higher reducing power, reaching 45.3–46.6 mg trx/g in its fruiting bodies [94]. In contrast, Shah et al. [77] reported much lower FRAP activities (0.025–0.10 mg trx/g) in submerged fungal cultures, underscoring that the OMW-grown T. mesentericum biomass in the present study demonstrates markedly enhanced antioxidant potential compared with previously characterized fungal systems.
Triterpenoid production was also enhanced in OMW-grown biomass, which reached 18.5 ± 1.4 mg UA/g, compared with 8.0 ± 1.2 mg UA/g in the glucose syrup medium (Table 4). This observation indicates that cultivation on OMW is associated with increased triterpenoid accumulation; however, the underlying mechanisms cannot be elucidated without transcriptomic, proteomic, or enzymatic analyses. Comparable levels have been reported in T. magnatum fermentation products [71] (12.7–20.6 mg/g), further reinforcing that truffle mycelia naturally produce substantial amounts of triterpenoids under favorable conditions.
Analysis of individual phenolic acids revealed a distinct and enriched profile in OMW-grown biomass, including high concentrations of gallic acid (13.3 ± 0.5 ppm), catechin (31.6 ± 1.0 ppm), hydroxytyrosol (1.6 ± 0.1 ppm) and oleuropein (1.7 ± 0.2 ppm), along with detectable levels of tyrosol, caffeic acid and p-coumaric acid (Table 4). These compounds likely reflect both direct uptake and partial biotransformation by fungal enzymes. Similar biotransformation patterns have been observed in other OMW–fungus systems [36,38]. Phenolic fingerprints reported for natural truffles [89,93] revealed that truffle species possess highly distinct and species-specific phenolic profiles. Villares et al. [93] demonstrated that T. aestivum exhibits the highest total phenolic acid content among the species examined, with hydroxycinnamic acids occurring exclusively in this taxon. Moreover, gallic and protocatechuic acids were detected only in T. aestivum and T. indicum, while T. aestivum contained markedly elevated levels of homogentisic acid and 3,4-dihydroxybenzaldehyde. Further differentiation between species was highlighted by Beara et al. [89], who reported that T. aestivum is enriched in p-hydroxybenzoic acid, baicalein and kaempferol, whereas T. magnatum predominantly accumulates epicatechin and catechin. These documented interspecific differences parallel the substrate-driven variations observed in the present study, emphasizing the dynamic nature of phenolic biosynthesis in truffles.
Overall, the OMW-grown T. mesentericum biomass displayed markedly enhanced levels of phenolics, antioxidant activity and triterpenoids, together with a distinct profile of bioactive phenolic acids. The enriched biochemical profile observed (characterized by elevated phenolics, strong reducing capacity and increased triterpenoid accumulation) highlights the species’ notable detoxification ability and metabolic adaptability. In summary, this study demonstrates that OMW is a promising substrate for producing fungal biomass enriched in specific bioactive compounds through submerged fermentation.
4. Conclusions
T. mesentericum exhibits remarkable physiological resilience and metabolic versatility when cultivated on OMW, a phenolic-rich and chemically complex agro-industrial byproduct. Across all cultivation processes (Petri dish screening, shake flask and bioreactor cultures), the fungus maintained vigorous growth, efficient substrate utilization and strong detoxification capacity, achieving substantial reductions in phenolic load and decolorization while sustaining biomass production comparable to glucose syrup media. Beyond supporting growth, OMW markedly enhanced the accumulation of selected bioactive compounds, including polysaccharides, β-glucans, proteins, unsaturated fatty acids, phenolics, antioxidants and triterpenoids. These findings are valuable for potential industrial production of truffle mycelium.
Author Contributions
I.D.: methodology, data curation, writing of original draft; G.V.: methodology, data curation, writing of original draft.; N.G.S.: supervision; S.P.: resources, supervision; P.D.: conceptualization, supervision, writing—review. All authors have read and agreed to the published version of the manuscript.
Funding
The research work was supported by the Hellenic Foundation for Research and Innovation (HFRI) under the 4th Call for HFRI PhD Fellowships (Fellowship Number: 11399).
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors on request.
Acknowledgments
The authors would like to express our sincere gratitude to Stefanos Diamantis, for kindly providing some of the Tuber spp. used in this study.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Wilgan, R. High species diversity but low specificity to ectomycorrhizal tree partners exhibited by native truffle species (Tuber spp., pezizales) in Poland, central Europe. Forests 2023, 14, 2407. [Google Scholar] [CrossRef]
- Zambonelli, A.; Iotti, M.; Murat, C. (Eds.) True Truffle (Tuber spp.) in the World: Soil Ecology, Systematics and Biochemistry; Soil Biology; Springer International Publishing: Cham, Switzerland, 2016; Volume 47. [Google Scholar] [CrossRef]
- Bonito, G.M.; Smith, M.E. General systematic position of the truffles: Evolutionary theories. In True Truffle (Tuber spp.) in the World; Springer International Publishing: Cham, Switzerland, 2016; Volume 47, pp. 3–18. [Google Scholar] [CrossRef]
- Gwon, J.-H.; Park, H.; Eom, A.-H. Effect of temperature, pH, and media on the mycelial growth of Tuber koreanum. Mycobiology 2022, 50, 238–243. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.-J.; Liu, R.-S.; Li, H.-M. Current progress on truffle submerged fermentation: A promising alternative to its fruiting bodies. Appl. Microbiol. Biotechnol. 2015, 99, 2041–2053. [Google Scholar] [CrossRef] [PubMed]
- Hall, I.R.; Haslam, W. Truffle cultivation in the southern hemisphere. In Edible Ectomycorrhizal Mushrooms; Springer: Berlin/Heidelberg, Germany, 2013; Volume 34, pp. 191–208. [Google Scholar] [CrossRef]
- Liu, R.-S.; Li, D.-S.; Li, H.-M.; Tang, Y.-J. Response surface modeling the significance of nitrogen source on the cell growth and tuber polysaccharides production by submerged cultivation of chinese truffle Tuber sinense. Process Biochem. 2008, 43, 868–876. [Google Scholar] [CrossRef]
- Tang, Y.-J.; Zhu, L.-L.; Li, D.-S.; Mi, Z.-Y.; Li, H.-M. Significance of inoculation density and carbon source on the mycelial growth and tuber polysaccharides production by submerged fermentation of chinese truffle Tuber sinense. Process Biochem. 2008, 43, 576–586. [Google Scholar] [CrossRef]
- El Enshasy, H.; Elsayed, E.A.; Aziz, R.; Wadaan, M.A. Mushrooms and truffles: Historical biofactories for complementary medicine in Africa and in the Middle East. Evid. Based Complement. Alternat. Med. 2013, 2013, 620451. [Google Scholar] [CrossRef]
- Baldelli, S.; Aiello, G.; De Bruno, A.; Castelli, S.; Lombardo, M.; Stocchi, V.; Tripodi, G. Bioactive compounds and antioxidant potential of truffles: A comprehensive review. Antioxidants 2025, 14, 1341. [Google Scholar] [CrossRef]
- Liu, P.; Li, H.-M.; Tang, Y.-J. Comparison of free amino acids and 5′-nucleotides between Tuber fermentation mycelia and natural fruiting bodies. Food Chem. 2012, 132, 1413–1419. [Google Scholar] [CrossRef]
- Tang, Y.-J.; Zhu, L.-W.; Li, H.-M.; Li, D.-S. Submerged culture of mushrooms in bioreactors—Challenges, Current State-of-the-Art, and Future Prospects. Food Technol. Biotechnol. 2007, 45, 221–229. [Google Scholar]
- Zhong, J.-J.; Tang, Y.-J. Submerged Cultivation of medicinal mushrooms for production of valuable bioactive metabolites. In Biomanufacturing; Advances in Biochemical Engineering/Biotechnology; Springer: Berlin/Heidelberg, Germany, 2004; Volume 87, pp. 25–59. [Google Scholar] [CrossRef]
- Zhao, W.; Wang, X.-H.; Li, H.-M.; Wang, S.-H.; Chen, T.; Yuan, Z.-P.; Tang, Y.-J. Isolation and characterization of polysaccharides with the antitumor activity from Tuber fruiting bodies and fermentation system. Appl. Microbiol. Biotechnol. 2014, 98, 1991–2002. [Google Scholar] [CrossRef]
- Azzam, M.O.J.; Al-Gharabli, S.I.; Al-Harahsheh, M.S. Olive mills wastewater treatment using local natural jordanian clay. Desalin. Water Treat. 2015, 53, 627–636. [Google Scholar] [CrossRef]
- Davies, L.C.; Vilhena, A.M.; Novais, J.M.; Martins-Dias, S. Olive mill wastewater characteristics: Modelling and statistical analysis. Grasas Aceites 2004, 55, 233–241. [Google Scholar] [CrossRef]
- Ntougias, S.; Baldrian, P.; Ehaliotis, C.; Nerud, F.; Antoniou, T.; Merhautová, V.; Zervakis, G.I. Biodegradation and detoxification of olive mill wastewater by selected strains of the mushroom genera Ganoderma and Pleurotus. Chemosphere 2012, 88, 620–626. [Google Scholar] [CrossRef] [PubMed]
- Allı, H. Effects of olive mill wastewater on mycelial growth of some macrofungi. J. Fungus 2023, 14, 55–59. [Google Scholar] [CrossRef]
- Diamantis, I.; Melanouri, E.-M.; Dedousi, M.; Panagopoulou, I.; Papanikolaou, S.; Stoforos, N.G.; Diamantopoulou, P. Sustainable and eco-friendly conversions of olive mill wastewater-based media by Pleurotus pulmonarius cultures. Fermentation 2022, 8, 129. [Google Scholar] [CrossRef]
- Diamantopoulou, P.; Gardeli, C.; Papanikolaou, S. Impact of olive mill wastewaters on the physiological behavior of a wild-type new Ganoderma resinaceum isolate. Environ. Sci. Pollut. Res. 2021, 28, 20570–20585. [Google Scholar] [CrossRef]
- Dourou, M.; Kancelista, A.; Juszczyk, P.; Sarris, D.; Bellou, S.; Triantaphyllidou, I.-E.; Rywinska, A.; Papanikolaou, S.; Aggelis, G. Bioconversion of olive mill wastewater into high-added value products. J. Clean. Prod. 2016, 139, 957–969. [Google Scholar] [CrossRef]
- Zerva, A.; Papaspyridi, L.-M.; Christakopoulos, P.; Topakas, E. Valorization of olive mill wastewater for the production of β-glucans from selected basidiomycetes. Waste Biomass Valoriz. 2017, 8, 1721–1731. [Google Scholar] [CrossRef]
- Crognale, S.; D’Annibale, A.; Federici, F.; Fenice, M.; Quaratino, D.; Petruccioli, M. Olive oil mill wastewater valorisation by fungi. J. Chem. Technol. Biotechnol. 2006, 81, 1547–1555. [Google Scholar] [CrossRef]
- Paraskeva, P.; Diamadopoulos, E. Technologies for Olive mill wastewater (OMW) treatment: A review. J. Chem. Technol. Biotechnol. 2006, 81, 1475–1485. [Google Scholar] [CrossRef]
- Moglie, M.; Biancini, G.; Norici, A.; Mugnini, A.; Cioccolanti, L. Valorization of olive mill wastewater for Arthrospira platensis production. Cell Rep. Sustain. 2024, 1, 100017. [Google Scholar] [CrossRef]
- Pikuli, K.; Devolli, A. Characterization and environmental impact of olive mill wastewater generated from the three-phase extraction process. Int. J. Innov. Approaches Agric. Res. 2024, 8, 263–276. [Google Scholar] [CrossRef]
- Teigiserova, D.A.; Hamelin, L.; Thomsen, M. Towards transparent valorization of food surplus, waste and loss: Clarifying definitions, food waste hierarchy, and role in the circular economy. Sci. Total Environ. 2020, 706, 136033. [Google Scholar] [CrossRef] [PubMed]
- Russo, G.L.; Langellotti, A.L.; Martín-García, B.; Verardo, V.; Romano, R.; Sacchi, R.; Masi, P. New biotechnological production of epa by Pythium irregulare using alternative sustainable media obtained from food industry by-products and waste. Sustainability 2023, 15, 1147. [Google Scholar] [CrossRef]
- Dedousi, M.; Fourtaka, K.; Melanouri, E.-M.; Argyropoulos, D.; Psallida, C.; Diamantis, I.; Papanikolaou, S.; Diamantopoulou, P. Detoxification of molasses and production of mycelial mass and valuable metabolites by Morchella species. Appl. Sci. 2021, 11, 9481. [Google Scholar] [CrossRef]
- Diamantis, I.; Stamatiadis, S.; Melanouri, E.-M.; Papanikolaou, S.; Diamantopoulou, P. From Screening to laboratory scale-up: Bioremediation potential of mushroom strains grown on olive mill wastewater. Biomass 2025, 5, 50. [Google Scholar] [CrossRef]
- Sarris, D.; Philippoussis, A.; Mallouchos, A.; Diamantopoulou, P. Valorization of low-cost, carbon-rich substrates by edible ascomycetes and basidiomycetes grown on liquid cultures. FEMS Microbiol. Lett. 2020, 367, fnaa168. [Google Scholar] [CrossRef]
- Shen, K.; Liu, Y.; Liu, L.; Khan, A.W.; Normakhamatov, N.; Wang, Z. Characterization, optimization, and scaling-up of submerged Inonotus hispidus mycelial fermentation for enhanced biomass and polysaccharide production. Appl. Biochem. Biotechnol. 2025, 197, 1534–1555. [Google Scholar] [CrossRef]
- Venkatachalam, M.; Mares, G.; Dufossé, L.; Fouillaud, M. Scale-Up of pigment production by the marine-derived filamentous fungus, Talaromyces albobiverticillius 30548, from shake flask to stirred bioreactor. Fermentation 2023, 9, 77. [Google Scholar] [CrossRef]
- Xia, J.; Wang, G.; Fan, M.; Chen, M.; Wang, Z.; Zhuang, Y. Understanding the scale-up of fermentation processes from the viewpoint of the flow field in bioreactors and the physiological response of strains. Chin. J. Chem. Eng. 2021, 30, 178–184. [Google Scholar] [CrossRef]
- Bakratsas, G.; Polydera, A.; Katapodis, P.; Stamatis, H. Recent trends in submerged cultivation of mushrooms and their application as a source of nutraceuticals and food additives. Future Foods 2021, 4, 100086. [Google Scholar] [CrossRef]
- Papaspyridi, L.-M.; Aligiannis, N.; Topakas, E.; Christakopoulos, P.; Skaltsounis, A.-L.; Fokialakis, N. Submerged fermentation of the edible mushroom Pleurotus ostreatus in a batch stirred tank bioreactor as a promising alternative for the effective production of bioactive metabolites. Molecules 2012, 17, 2714–2724. [Google Scholar] [CrossRef]
- Kim, H.O.; Lim, J.M.; Joo, J.H.; Kim, S.W.; Hwang, H.J.; Choi, J.W.; Yun, J.W. Optimization of submerged culture condition for the production of mycelial biomass and exopolysaccharides by Agrocybe cylindracea. Bioresour. Technol. 2005, 96, 1175–1182. [Google Scholar] [CrossRef]
- Aggelis, G.; Iconomou, D.; Christou, M.; Bokas, D.; Kotzailias, S.; Christou, G.; Tsagou, V.; Papanikolaou, S. Phenolic removal in a model olive oil mill wastewater using Pleurotus ostreatus in bioreactor cultures and biological evaluation of the process. Water Res. 2003, 37, 3897–3904. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.L. Use of Dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Slinkard, K.; Singleton, V.L. Total phenol analysis: Automation and comparison with manual methods. Am. J. Enol. Vitic. 1977, 28, 49–55. [Google Scholar] [CrossRef]
- Tsioulpas, A. Phenolic removal in olive oil mill wastewater by strains of Pleurotus spp. in respect to their phenol oxidase (laccase) activity. Bioresour. Technol. 2002, 84, 251–257. [Google Scholar] [CrossRef]
- Diamantopoulou, P.; Papanikolaou, S.; Katsarou, E.; Komaitis, M.; Aggelis, G.; Philippoussis, A. Mushroom polysaccharides and lipids synthesized in liquid agitated and static cultures. Part II: Study of Volvariella volvacea. Appl. Biochem. Biotechnol. 2012, 167, 1890–1906. [Google Scholar] [CrossRef]
- Diamantopoulou, P.; Papanikolaou, S.; Komaitis, M.; Aggelis, G.; Philippoussis, A. Patterns of major metabolites biosynthesis by different mushroom fungi grown on glucose-based submerged cultures. Bioprocess Biosyst. Eng. 2014, 37, 1385–1400. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Stanley, G.H.S. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP Assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.-P.; He, C.-H. Simultaneous quantification of three major bioactive triterpene acids in the leaves of Diospyros kaki by high-performance liquid chromatography method. J. Pharm. Biomed. Anal. 2006, 41, 950–956. [Google Scholar] [CrossRef] [PubMed]
- Mulinacci, N.; Romani, A.; Galardi, C.; Pinelli, P.; Giaccherini, C.; Vincieri, F.F. Polyphenolic content in olive oil waste waters and related olive samples. J. Agric. Food Chem. 2001, 49, 3509–3514. [Google Scholar] [CrossRef] [PubMed]
- Fountoulakis, M.S.; Dokianakis, S.N.; Kornaros, M.E.; Aggelis, G.G.; Lyberatos, G. Removal of phenolics in olive mill wastewaters using the white-rot fungus Pleurotus ostreatus. Water Res. 2002, 36, 4735–4744. [Google Scholar] [CrossRef]
- Matos, A.J.F.S.; Bezerra, R.M.F.; Dias, A.A. Screening of fungal isolates and properties of Ganoderma applanatum intended for olive mill wastewater decolourization and dephenolization. Lett. Appl. Microbiol. 2007, 45, 270–275. [Google Scholar] [CrossRef]
- Koutrotsios, G.; Zervakis, G.I. Comparative examination of the olive mill wastewater biodegradation process by various wood-rot macrofungi. BioMed Res. Int. 2014, 2014, 482937. [Google Scholar] [CrossRef]
- Campos, A.D.O.; Wahalathanthrige, H.J.; Russell, S.; Harrison, M.D.; Strong, P.J. Conversion of soluble compounds in distillery wastewater into fungal biomass and metabolites using australian Ganoderma isolates. J. Fungi 2025, 11, 432. [Google Scholar] [CrossRef]
- Papaspyridi, L.-M.; Katapodis, P.; Gonou-Zagou, Z.; Kapsanaki-Gotsi, E.; Christakopoulos, P. Optimization of biomass production with enhanced glucan and dietary fibres content by Pleurotus ostreatus ATHUM 4438 under submerged culture. Biochem. Eng. J. 2010, 50, 131–138. [Google Scholar] [CrossRef]
- Sarris, D.; Galiotou-Panayotou, M.; Koutinas, A.A.; Komaitis, M.; Papanikolaou, S. Citric acid, biomass and cellular lipid production by Yarrowia lipolytica strains cultivated on olive mill wastewater-based media. J. Chem. Technol. Biotechnol. 2011, 86, 1439–1448. [Google Scholar] [CrossRef]
- Sarris, D.; Tsouko, E.; Photiades, A.; Tchakouteu, S.S.; Diamantopoulou, P.; Papanikolaou, S. Growth response of non-conventional yeasts on sugar-rich media: Part 2: Citric acid production and circular-oriented valorization of glucose-enriched olive mill wastewaters using novel Yarrowia lipolytica strains. Microorganisms 2023, 11, 2243. [Google Scholar] [CrossRef]
- Sarris, D.; Giannakis, M.; Philippoussis, A.; Komaitis, M.; Koutinas, A.A.; Papanikolaou, S. Conversions of olive mill wastewater-based media by Saccharomyces cerevisiae through sterile and non-sterile bioprocesses. J. Chem. Technol. Biotechnol. 2013, 88, 958–969. [Google Scholar] [CrossRef]
- Duarte, K.R.; Justino, C.; Panteleitchouk, T.; Zrineh, A.; Freitas, A.C.; Duarte, A.C.; Rocha-Santos, T.A.P. Removal of phenolic compounds in olive mill wastewater by silica–alginate–fungi biocomposites. Int. J. Environ. Sci. Technol. 2014, 11, 589–596. [Google Scholar] [CrossRef]
- Fraij, A.; Massadeh, M.I. Use of Pleurotus sajor-caju for the biotreatment of olive mill wastewater. Rom. Biotechnol. Lett. 2015, 20, 10611–10617. [Google Scholar]
- Diamantopoulou, P.; Aggelis, G.; Papanikolaou, S. Renewable carbon sources as microbial substrates for the production of amylases and lignocellulases. Carbon Resour. Convers. 2026, 9, 100356. [Google Scholar] [CrossRef]
- Liu, Q.-N.; Liu, R.-S.; Wang, Y.-H.; Mi, Z.-Y.; Li, D.-S.; Zhong, J.-J.; Tang, Y.-J. Fed-Batch Fermentation of Tuber melanosporum for the hyperproduction of mycelia and bioactive Tuber polysaccharides. Bioresour. Technol. 2009, 100, 3644–3649. [Google Scholar] [CrossRef]
- Chen, C.-C.; Nargotra, P.; Kuo, C.-H.; Liu, Y.-C. High-molecular-weight exopolysaccharides production from Tuber borchii cultivated by submerged fermentation. Int. J. Mol. Sci. 2023, 24, 4875. [Google Scholar] [CrossRef]
- Fu, Y.-R.; Nargotra, P.; Kuo, C.-H.; Liu, Y.-C. Optimization of biomass cultivation from Tuber borchii and effect of additives on triterpenoid production. Fermentation 2023, 9, 735. [Google Scholar] [CrossRef]
- Kennedy, M.J.; Reader, S.L.; Davies, R.J.; Rhoades, D.A.; Silby, H.W. The scale up of mycelial shake flask fermentations: A case study of gamma linolenic acid production by Mucor hiemalis IRL 51. J. Ind. Microbiol. 1994, 13, 212–216. [Google Scholar] [CrossRef]
- Vasilakis, G.; Roidouli, C.; Karayannis, D.; Giannakis, N.; Rondags, E.; Chevalot, I.; Papanikolaou, S. Study of different parameters affecting production and productivity of polyunsaturated fatty acids (PUFAs) and γ-linolenic acid (GLA) by Cunninghamella elegans through glycerol conversion in shake flasks and bioreactors. Microorganisms 2024, 12, 2097. [Google Scholar] [CrossRef]
- Rosa-Gruszecka, A.; Hilszczańska, D.; Pacioni, G. Virtual truffle hunting—A new method of burgundy truffle (Tuber aestivum vittad.) site typing. Forests 2021, 12, 1239. [Google Scholar] [CrossRef]
- Mello, A.; Murat, C.; Bonfante, P. Truffles: Much more than a prized and local fungal delicacy. FEMS Microbiol. Lett. 2006, 260, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Nakano, S.; Kinoshita, A.; Obase, K.; Nakamura, N.; Furusawa, H.; Noguchi, K.; Yamanaka, T. Influence of pH on in vitro mycelial growth in three Japanese truffle species: Tuber Japonicum, T. Himalayense and T. Longispinosum. Mycoscience 2020, 61, 58–61. [Google Scholar] [CrossRef]
- Mischiati, P.; Fontana, A. In vitro culture of Tuber magnatum mycelium isolated from mycorrhizas. Mycol. Res. 1993, 97, 40–44. [Google Scholar] [CrossRef]
- Bustan, A.; Ventura, Y.; Kagan-Zur, V.; Roth-Bejerano, N. Optimized conditions for mycorrhiza formation between the pink rockrose (Cistus incanus) and the black périgord truffle (Tuber melanosporum). Isr. J. Plant Sci. 2006, 54, 87–96. [Google Scholar] [CrossRef]
- Iotti, M.; Leonardi, P.; Vitali, G.; Zambonelli, A. Effect of summer soil moisture and temperature on the vertical distribution of Tuber magnatum mycelium in soil. Biol. Fertil. Soils 2018, 54, 707–716. [Google Scholar] [CrossRef]
- Blánquez, P.; Caminal, G.; Sarrà, M.; Vicent, M.T.; Gabarrell, X. Olive oil mill waste waters decoloration and detoxification in a bioreactor by the white rot fungus Phanerochaete flavido-alba. Biotechnol. Prog. 2002, 18, 660–662. [Google Scholar] [CrossRef]
- Hsu, J.-Y.; Chen, M.-H.; Lai, Y.-S.; Chen, S.-D. Antioxidant profile and biosafety of white truffle mycelial products obtained by solid-state fermentation. Molecules 2021, 27, 109. [Google Scholar] [CrossRef]
- Sun, Q.; Zhang, Z.; Xu, L.; Shi, W.; Liu, X.; Wang, F. Increasing production of truffle polysaccharides in the solid-state fermentation of Tuber melanosporum by diosgenin based on orthogonal matrix and nonlinear regression analysis. Food Sci. Technol. Res. 2020, 26, 487–494. [Google Scholar] [CrossRef]
- Tang, Y.-J.; Zhong, J.-J. Fed-batch fermentation of Ganoderma lucidum for hyperproduction of polysaccharide and ganoderic acid. Enzym. Microb. Technol. 2002, 31, 20–28. [Google Scholar] [CrossRef]
- Diamantopoulou, P.; Papanikolaou, S. Biotechnological production of sugar-alcohols: Focus on Yarrowia lipolytica and edible/medicinal mushrooms. Process Biochem. 2023, 124, 113–131. [Google Scholar] [CrossRef]
- Solomon, P.S.; Waters, O.D.C.; Oliver, R.P. Decoding the mannitol enigma in filamentous fungi. Trends Microbiol. 2007, 15, 257–262. [Google Scholar] [CrossRef]
- Cateni, F.; Gargano, M.L.; Procida, G.; Venturella, G.; Cirlincione, F.; Ferraro, V. Mycochemicals in wild and cultivated mushrooms: Nutrition and health. Phytochem. Rev. 2022, 21, 339–383. [Google Scholar] [CrossRef]
- Shah, N.; Marathe, S.J.; Croce, D.; Ciardi, M.; Longo, V.; Juilus, A.; Shamekh, S. An investigation of the antioxidant potential and bioaccumulated minerals in Tuber borchii and Tuber maculatum mycelia obtained by submerged fermentation. Arch. Microbiol. 2022, 204, 64. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Li, Y.-Y.; Li, H.-M.; Wan, D.-J.; Tang, Y.-J. Comparison of lipid content and fatty acid composition between Tuber fermentation mycelia and natural fruiting bodies. J. Agric. Food Chem. 2011, 59, 4736–4742. [Google Scholar] [CrossRef] [PubMed]
- Beuchat, L.; Brenneman, T.; Dove, C. Composition of the pecan truffle (Tuber Texense). Food Chem. 1993, 46, 189–192. [Google Scholar] [CrossRef]
- Harki, E.; Bouya, D.; Dargent, R. Maturation-associated alterations of the biochemical characteristics of the black truffle Tuber melanosporum vitt. Food Chem. 2006, 99, 394–400. [Google Scholar] [CrossRef]
- Sancholle, M.; Weete, J.D.; Kulifaj, M.; Montant, C. Changes in lipid composition during ascocarp development of the truffle, Tuber melanosporum. Mycologia 1988, 80, 900–903. [Google Scholar] [CrossRef]
- Yan, X.; Wang, Y.; Sang, X.; Fan, L. Nutritional value, chemical composition and antioxidant activity of three Tuber species from China. AMB Express 2017, 7, 136. [Google Scholar] [CrossRef]
- Sawaya, W.N.; Al-Shalhat, A.; Al-Sogair, A.; Al-Mohammad, M. Chemical composition and nutritive value of truffles of Saudi Arabia. J. Food Sci. 1985, 50, 450–453. [Google Scholar] [CrossRef]
- Henkrar, F.; Khabar, L. Proximate analysis of lipid composition in moroccan truffles and desert truffles. Ital. J. Food Sci. 2022, 34, 67–73. [Google Scholar] [CrossRef]
- Lee, H.; Nam, K.; Zahra, Z.; Farooqi, M.Q.U. Potentials of truffles in nutritional and medicinal applications: A Review. Fungal Biol. Biotechnol. 2020, 7, 9. [Google Scholar] [CrossRef]
- Shah, N.N.; Hokkanen, S.; Pastinen, O.; Eljamil, A.; Shamekh, S. A study on the fatty acid composition of lipids in truffles selected from Europe and Africa. 3 Biotech 2020, 10, 415. [Google Scholar] [CrossRef]
- Tejedor-Calvo, E.; Amara, K.; Reis, F.S.; Barros, L.; Martins, A.; Calhelha, R.C.; Venturini, M.E.; Blanco, D.; Redondo, D.; Marco, P.; et al. Chemical composition and evaluation of antioxidant, antimicrobial and antiproliferative activities of Tuber and Terfezia truffles. Food Res. Int. 2021, 140, 110071. [Google Scholar] [CrossRef] [PubMed]
- Murcia, J.A.G. Ação de Reguladores Vegetais em Trigo (Triticum aestivum L.) e Cevada (Hordeum vulgare L.). Master’s Thesis, Universidade de São Paulo, Piracicaba, Brazil, 2016. [Google Scholar] [CrossRef]
- Beara, I.N.; Lesjak, M.M.; Četojević-Simin, D.D.; Marjanović, Ž.S.; Ristić, J.D.; Mrkonjić, Z.O.; Mimica-Dukić, N.M. Phenolic profile, antioxidant, anti-inflammatory and cytotoxic activities of black (Tuber aestivum vittad.) and white (Tuber magnatum pico) truffles. Food Chem. 2014, 165, 460–466. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-H.; Benucci, G.M.N.; Xie, X.-D.; Bonito, G.; Leisola, M.; Liu, P.-G.; Shamekh, S. Morphological, Mycorrhizal and molecular characterization of finnish truffles belonging to the Tuber anniae species-complex. Fungal Ecol. 2013, 6, 269–280. [Google Scholar] [CrossRef]
- Hamza, A.; Zouari, N.; Zouari, S.; Jdir, H.; Zaidi, S.; Gtari, M.; Neffati, M. Nutraceutical potential, antioxidant and antibacterial activities of Terfezia boudieri chatin, a wild edible desert truffle from Tunisia arid zone. Arab. J. Chem. 2016, 9, 383–389. [Google Scholar] [CrossRef]
- Kivrak, Ş.; Kivrak, İ. Investigation of chemical composition and nutritional value of truffle mushroom (Tuber nitidum vittad.). Süleyman Demirel Üniversitesi Fen Bilim. Enstitüsü Derg. 2018, 22, 339. [Google Scholar] [CrossRef]
- Villares, A.; García-Lafuente, A.; Guillamón, E.; Ramos, Á. Identification and quantification of ergosterol and phenolic compounds occurring in Tuber spp. truffles. J. Food Compos. Anal. 2012, 26, 177–182. [Google Scholar] [CrossRef]
- Al-Laith, A.A.A. Antioxidant components and antioxidant/antiradical activities of desert truffle (Tirmania nivea) from various middle eastern origins. J. Food Compos. Anal. 2010, 23, 15–22. [Google Scholar] [CrossRef]
- Cheung, L.M.; Cheung, P.C.K.; Ooi, V.E.C. Antioxidant activity and total phenolics of edible mushroom extracts. Food Chem. 2003, 81, 249–255. [Google Scholar] [CrossRef]
- Heleno, S.A.; Ferreira, R.C.; Antonio, A.L.; Queiroz, M.-J.R.P.; Barros, L.; Ferreira, I.C.F.R. Nutritional value, bioactive compounds and antioxidant properties of three edible mushrooms from Poland. Food Biosci. 2015, 11, 48–55. [Google Scholar] [CrossRef]
- Dutta, R.; Ghosal, S.; Roy, S.; Bhattacharyya, D.K.; Bhowal, J. Phytochemical screening and antioxidative property evaluation of lipid-producing fungi. Arch. Microbiol. 2023, 205, 65. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.