A 10-Year Single-Center Study of the Clinical Characteristics of Optic Neuritis-Related NMOSD, MS, and Double Seronegative Optic Neuritis, Together with Factors Predicting Visual Outcomes
Abstract
1. Introduction
2. Materials and Methods
2.1. Methods and Data Collection
2.2. Ethical Approval
2.3. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Beck, R.W.; Cleary, P.A.; Anderson Jr, M.M.; Keltner, J.L.; Shults, W.T.; Kaufman, D.I.; Buckley, E.G.; Corbett, J.J.; Kupersmith, M.J.; Miller, N.R.; et al. A randomized, controlled trial of corticosteroids in the treatment of acute optic neuritis. The Optic Neuritis Study Group. N. Engl. J. Med. 1992, 326, 581–588. [Google Scholar] [CrossRef]
- Wingerchuk, D.M.; Banwell, B.; Bennett, J.L.; Cabre, P.; Carroll, W.; Chitnis, T.; De Seze, J.; Fujihara, K.; Greenberg, B.; Jacob, A.; et al. International consensus diagnostic criteria for neuromyelitis optica spectrum disorders. Neurology 2015, 85, 177–189. [Google Scholar] [CrossRef] [PubMed]
- Polman, C.H.; Reingold, S.C.; Banwell, B.; Clanet, M.; Cohen, J.A.; Filippi, M.; Fujihara, K.; Havrdova, E.; Hutchinson, M.; Kappos, L.; et al. Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann. Neurol. 2011, 69, 292–302. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Jiang, H.; Jiang, L.; Peng, J.; Liu, H.; Wang, J.; Wei, W. Factors influencing intravenous methylprednisolone pulse therapy in Chinese patients with isolated optic neuritis associated with AQP4 antibody-seropositive neuromyelitis optica. Sci. Rep. 2021, 11, 22229. [Google Scholar] [CrossRef]
- Akaishi, T.; Takeshita, T.; Himori, N.; Takahashi, T.; Misu, T.; Ogawa, R.; Kaneko, K.; Fujimori, J.; Abe, M.; Ishii, T.; et al. Rapid Administration of High-Dose Intravenous Methylprednisolone Improves Visual Outcomes After Optic Neuritis in Patients With AQP4-IgG-Positive NMOSD. Front. Neurol. 2020, 11, 932. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Zhao, S.; Yin, D.; Chen, X.; Xu, Q.; Chen, T.; Li, X.; Wang, J.; Li, H.; Peng, C.; et al. Optic neuritis: A 5-year follow-up study of Chinese patients based on aquaporin-4 antibody status and ages. J. Neurol. 2016, 263, 1382–1389. [Google Scholar] [CrossRef]
- Stiebel-Kalish, H.; Hellmann, M.A.; Mimouni, M.; Paul, F.; Bialer, O.; Bach, M.; Lotan, I. Does time equal vision in the acute treatment of a cohort of AQP4 and MOG optic neuritis? Neurol. Neuroimmunol. Neuroinflamm. 2019, 6, e572. [Google Scholar] [CrossRef]
- Banerjee, A.; Ng, J.; Coleman, J.; Ospina, J.P.; Mealy, M.; Levy, M. Outcomes from acute attacks of neuromyelitis optica spectrum disorder correlate with severity of attack, age and delay to treatment. Mult. Scler. Relat. Disord. 2019, 28, 60–63. [Google Scholar] [CrossRef]
- Kim, H.; Park, K.-A.; Oh, S.Y.; Min, J.-H.; Kim, B.J. Association of Optic Neuritis with Neuromyelitis Optica Spectrum Disorder and Multiple Sclerosis in Korea. Korean J. Ophthalmol. 2019, 33, 82–90. [Google Scholar] [CrossRef]
- Hansapinyo, L.; Vivattanaseth, C. Clinical Characteristics, Treatment Outcomes and Predictive Factors in Optic Neuritis. Open Ophthalmol. J. 2018, 12, 247–255. [Google Scholar] [CrossRef]
- Thongmee, W.; Padungkiatsagul, T.; Jindahra, P.; Khongkhatithum, C.; Thampratankul, L.; Vanikieti, K. Prognostic Factors for Visual Outcomes Following the First Episode of NMOSD-Related Optic Neuritis in Affected Eyes. Clin. Ophthalmol. 2020, 14, 4271–4278. [Google Scholar] [CrossRef] [PubMed]
- Durgapriyadarshini, S.; Ambika, S.; Padmalakshmi, K.; Noronha, V.; Arjundas, D. Clinical profile, imaging features and short term visual outcomes of Indian optic neuritis patients with and without seromarkers for myelin oligodendrocyte glycoprotein and neuromyelitis optica. Indian J. Ophthalmol. 2022, 70, 194–200. [Google Scholar] [CrossRef]
- Thompson, A.J.; Banwell, B.L.; Barkhof, F.; Carroll, W.M.; Coetzee, T.; Comi, G.; Correale, J.; Fazekas, F.; Filippi, M.; Freedman, M.S.; et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol. 2018, 17, 162–173. [Google Scholar] [CrossRef]
- Chen, J.J.; Tobin, W.O.; Majed, M.; Jitprapaikulsan, J.; Fryer, J.P.; Leavitt, J.A.; Flanagan, E.P.; McKeon, A.; Pittock, S.J. Prevalence of Myelin Oligodendrocyte Glycoprotein and Aquaporin-4-IgG in Patients in the Optic Neuritis Treatment Trial. JAMA Ophthalmol. 2018, 136, 419–422. [Google Scholar] [CrossRef] [PubMed]
- Dutt, M.; Tabuena, P.; Ventura, E.; Rostami, A.; Shindler, K.S. Timing of corticosteroid therapy is critical to prevent retinal ganglion cell loss in experimental optic neuritis. Investig. Opthalmology Vis. Sci. 2010, 51, 1439–1445. [Google Scholar] [CrossRef] [PubMed]
- Akaishi, T.; Nakashima, I.; Takeshita, T.; Mugikura, S.; Sato, D.K.; Takahashi, T.; Nishiyama, S.; Kurosawa, K.; Misu, T.; Nakazawa, T.; et al. Lesion length of optic neuritis impacts visual prognosis in neuromyelitis optica. J. Neuroimmunol. 2016, 293, 28–33. [Google Scholar] [CrossRef] [PubMed]
- Zhu, B.; Moore, G.; Zwimpfer, T.J.; Kastrukoff, L.F.; Dyer, J.K.; Steeves, J.D.; Paty, D.W.; Cynader, M.S. Axonal cytoskeleton changes in experimental optic neuritis. Brain Res. 1999, 824, 204–217. [Google Scholar] [CrossRef]
- Soelberg, K.; Specovius, S.; Zimmermann, H.G.; Grauslund, J.; Mehlsen, J.J.; Olesen, C.; Neve, A.S.B.; Paul, F.; Brandt, A.U.; Asgari, N. Optical coherence tomography in acute optic neuritis: A population-based study. Acta Neurol. Scand. 2018, 138, 566–573. [Google Scholar] [CrossRef] [PubMed]
- Mealy, M.A.; Wingerchuk, D.M.; Greenberg, B.M.; Levy, M. Epidemiology of neuromyelitis optica in the United States: A multicenter analysis. Arch. Neurol. 2012, 69, 1176–1180. [Google Scholar] [CrossRef]
- Lin, C.-W.; Lin, I.-H.; Chen, T.-C.; Jou, J.-R.; Woung, L.-C.; Lee, S.S.; Yazar, S.; Pasquale, L.R.; Sanfilippo, P.G.; Hewitt, A.W.; et al. Clinical Course and Treatment Response of Neuromyelitis Optica Spectrum Disease: An 8-Year Experience. Asia Pac. J. Ophthalmol. 2019, 8, 206–210. [Google Scholar]
- Merle, H.; Olindo, S.; Bonnan, M.; Donnio, A.; Richer, R.; Smadja, D.; Cabre, P. Natural history of the visual impairment of relapsing neuromyelitis optica. Ophthalmology 2007, 114, 810–815. [Google Scholar] [CrossRef]
- Papais-Alvarenga, R.M.; Carellos, S.C.; Alvarenga, M.P.; Holander, C.; Bichara, R.P.; Thuler, L.C. Clinical course of optic neuritis in patients with relapsing neuromyelitis optica. Arch. Ophthalmol. 2008, 126, 12–16. [Google Scholar] [CrossRef]
- Kim, S.-M.; Waters, P.; Woodhall, M.; Kim, Y.-J.; Kim, J.-A.; Cheon, S.Y.; Lee, S.; Jo, S.R.; Kim, D.G.; Jung, K.C.; et al. Gender effect on neuromyelitis optica spectrum disorder with aquaporin4-immunoglobulin G. Mult. Scler. 2017, 23, 1104–1111. [Google Scholar] [CrossRef]
- Contentti, E.C.; De Virgiliis, M.; Hryb, J.P.; Gomez, A.; Morales, S.; Celso, J.; Leguizamón, F.; Chiganer, E.; Di Pace, J.L.; Lessa, C.; et al. Aquaporin-4 Serostatus and Visual Outcomes in Clinically Isolated Acute Optic Neuritis. J. Neuroophthalmol. 2019, 39, 165–169. [Google Scholar] [CrossRef]
- Collongues, N.; Patte-Mensah, C.; De Seze, J.; Mensah-Nyagan, A.-G.; Derfuss, T. Testosterone and estrogen in multiple sclerosis: From pathophysiology to therapeutics. Expert Rev. Neurother. 2018, 18, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Kitley, J.; Leite, M.I.; Nakashima, I.; Waters, P.; McNeillis, B.; Brown, R.; Takai, Y.; Takahashi, T.; Misu, T.; Elsone, L.; et al. Prognostic factors and disease course in aquaporin-4 antibody-positive patients with neuromyelitis optica spectrum disorder from the United Kingdom and Japan. Brain 2012, 135 Pt 6, 1834–1849. [Google Scholar] [CrossRef] [PubMed]
- Vanikieti, K.; Poonyathalang, A.; Jindahra, P.; Bouzika, P.; Rizzo, J.F., 3rd; Cestari, D.M. Clinical characteristics and long-term visual outcome of optic neuritis in neuromyelitis optica spectrum disorder: A comparison between Thai and American-Caucasian cohorts. Mult. Scler. Relat. Disord. 2017, 17, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhou, H.; Qin, L.; Peng, C.; Zhao, J.; Teng, D.; Lin, D.; Ai, N.; Xu, Q.; Wei, S. Optic Neuritis in the Older Chinese Population: A 5-Year Follow-Up Study. J. Ophthalmol. 2017, 2017, 3458356. [Google Scholar] [CrossRef] [PubMed]
- Feng, C.; Chen, Q.; Zhao, G.; Li, Z.; Chen, W.; Sha, Y.; Sun, X.; Wang, M.; Tian, G. Clinical characteristics of optic neuritis phenotypes in a 3-year follow-up Chinese cohort. Sci. Rep. 2021, 11, 14603. [Google Scholar] [CrossRef]
- Ishikawa, H.; Kezuka, T.; Shikishima, K.; Yamagami, A.; Hiraoka, M.; Chuman, H.; Nakamura, M.; Hoshi, K.; Goseki, T.; Mashimo, K.; et al. Epidemiologic and Clinical Characteristics of Optic Neuritis in Japan. Ophthalmology 2019, 126, 1385–1398. [Google Scholar] [CrossRef]
- Hussain, R.; Ghoumari, A.M.; Bielecki, B.; Steibel, J.; Boehm, N.; Liere, P.; Macklin, W.B.; Kumar, N.; Habert, R.; Mhaouty-Kodja, S.; et al. The neural androgen receptor: A therapeutic target for myelin repair in chronic demyelination. Brain 2013, 136 Pt 1, 132–146. [Google Scholar] [CrossRef] [PubMed]
- Denis, M.; Woillez, J.-P.; Smirnov, V.M.; Drumez, E.; Lannoy, J.; Boucher, J.; Zedet, M.; Pruvo, J.-P.; Labreuche, J.; Zephir, H.; et al. Optic Nerve Lesion Length at the Acute Phase of Optic Neuritis Is Predictive of Retinal Neuronal Loss. Neurol. Neuroimmunol. Neuroinflamm. 2022, 9, e1135. [Google Scholar] [CrossRef] [PubMed]
- Mealy, M.A.; Whetstone, A.; Orman, G.; Izbudak, I.; Calabresi, P.A.; Levy, M. Longitudinally extensive optic neuritis as an MRI biomarker distinguishes neuromyelitis optica from multiple sclerosis. J. Neurol. Sci. 2015, 355, 59–63. [Google Scholar] [CrossRef] [PubMed]
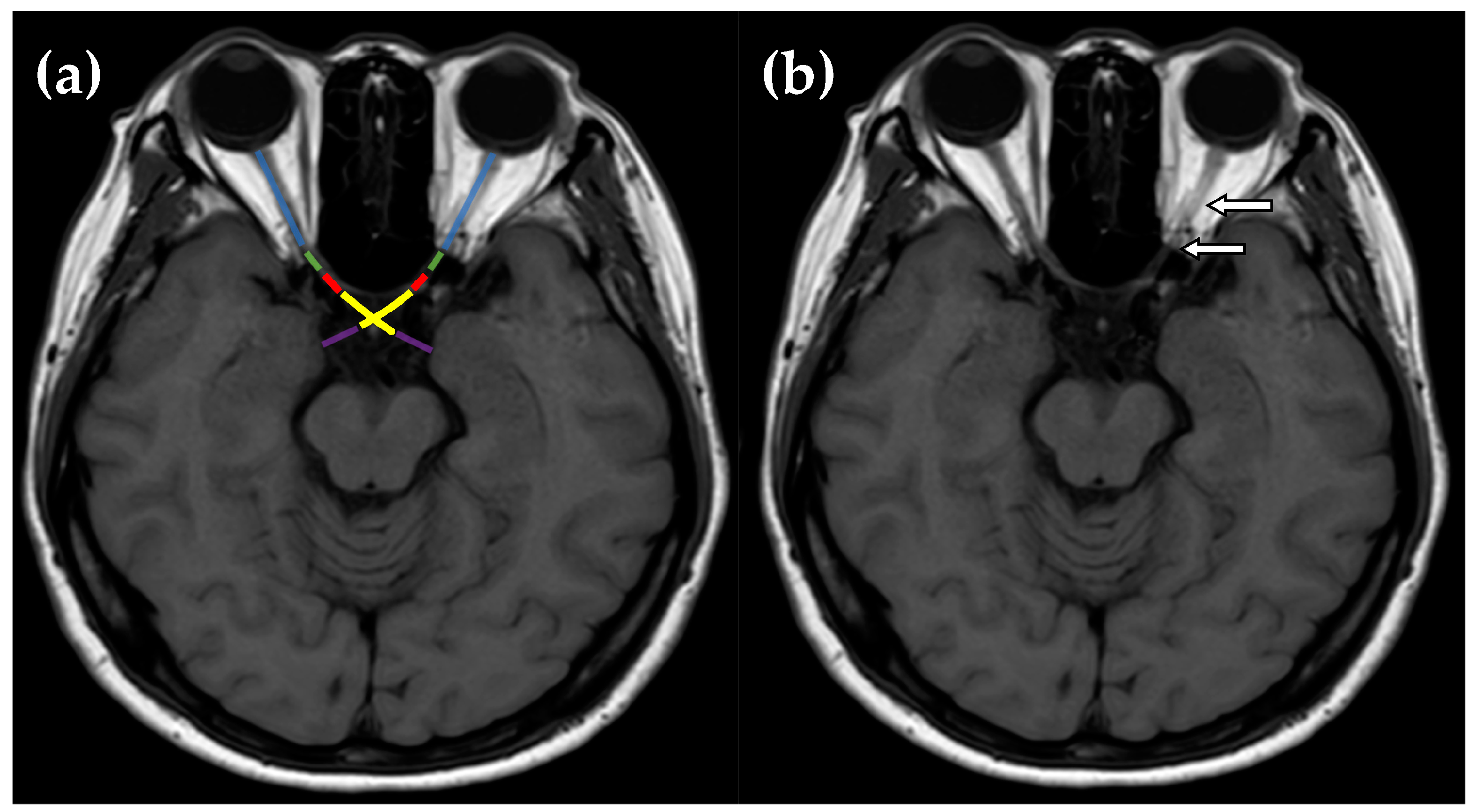

| Characteristic | Total Eyes (n = 76) | DN-ON (n = 40) | NMOSD-ON (n = 24) | MS-ON (n = 12) | p-Value | Subgroup Analysis (p-Value) | ||
|---|---|---|---|---|---|---|---|---|
| DN-ON vs. NMOSD-ON | DN-ON vs. MS-ON | NMOSD-ON vs. MS-ON | ||||||
| Number of patients | 61 (100) | 33 (54.1) | 18 (29.5) | 10 (16.4) | ||||
| Number of affected eyes | 76 (100) | 40 (52.6) | 24 (31.6) | 12 (15.8) | ||||
| Mean Age (SD)—yr | 40.6 (13.5) | 41.2 (13.4) | 46.2 (12.6) | 28.3 (6.6) | 0.002 | 0.505 | 0.016 | 0.002 |
| Female sex—no. | 50 (82.0) | 27 (81.8) | 17 (94.4) | 6 (60.0) | 0.076 | 0.209 | 0.153 | 0.041 |
| Laterality of first ON presentation—no. (%) Eyes | 43 (56.6) | 23 (57.5) | 14 (58.3) | 6 (50.0) | 0.88 | 0.579 | 0.447 | 0.451 |
| Bilateral simultaneous | 12 (19.7) | 6 (18.2) | 5 (27.8) | 1 (10.0) | 0.5 | 0.325 | 0.476 | 0.277 |
| Pain on eye movement—no. (%) | 25 (32.9) | 10 (25.0) | 9 (37.5) | 6 (50.0) | 0.229 | 0.218 | 0.101 | 0.358 |
| Median duration of onset (SD)—dys | 12.3 (13.4) | 9.8 (8.7) | 15.6 (20.4) | 14.8 (9.7) | 0.146 | 0.866 | 0.042 | 0.175 |
| Duration > 7 dys (%) | 21 (34.4) | 11 (33.3) | 5 (27.8) | 5 (50.0) | 0.486 | 0.468 | 0.277 | 0.221 |
| Co-existing autoimmune disease—no. (%) | 11 (18.0) | 4 (12.1) | 6 (33.3) | 1 (10.0) | 0.131 | 0.075 | 0.672 | 0.184 |
| Systemic lupus erythematosus | 9 (14.7) | 4 (12.1) | 4 (22.3) | 1 (10.0) | 0.56 | 0.287 | 0.672 | 0.389 |
| Graves’ disease | 2 (3.2) | 0 (0) | 2 (11.2) | 0 (0) | 0.085 | 0.12 | - | 0.405 |
| Visual parameter—no. (%) | ||||||||
| VA logMAR at nadir (SD) | 0.91 (0.68) | 0.58 (0.39) | 1.5 (0.73) | 0.84 (0.6) | <0.001 | <0.001 | 0.432 | 0.003 |
| VA Snellen at nadir (%) | <0.001 | <0.001 | 0.229 | <0.001 | ||||
| VA 20/20–20/60 | 13 (17.1) | 11 (27.5) | 0 (0) | 2 (16.7) | ||||
| VA 20/70–20/200 | 34 (44.7) | 26 (65.0) | 1 (4.2) | 7 (58.3) | ||||
| Poor baseline VA (20/200) | 29 (38.2) | 3 (7.5) | 23 (95.8) | 3 (25.0) | ||||
| Visual field at nadir (dB) | 15.4 (9.95) | 10.6 (7.1) | 25.3 (7.1) | 11.6 (8.9) | < 0.001 | <0.001 | 0.871 | <0.001 |
| Abnormal color vision (%) | 49 (64.5) | 27 (67.5) | 16 (66.7) | 6 (50.0) | 0.035 | 0.008 | 0.365 | 0.038 |
| Presence of swollen optic disc (%) | 37 (48.7) | 18 (45.0) | 12 (50.0) | 7 (58.3) | 0.711 | 0.448 | 0.315 | 0.454 |
| Relapse rate (%) | 1 (1.6) | 0 (0) | 1 (5.6) | 0 (0) | 0.297 | 0.353 | - | 0.643 |
| Recurrent—no. (%) | 2 (3.3) | 1 (3.0) | 0 (0) | 1 (10.0) | 0.36 | 0.647 | 0.415 | 0.357 |
| Characteristic | Total Eyes (n = 76) | DN-ON (n = 40) | NMOSD-ON (n = 24) | MS-ON (n = 12) | p-Value | Subgroup Analysis (p-Value) | ||
|---|---|---|---|---|---|---|---|---|
| DN-ON vs. NMOSD-ON | DN-ON vs. MS-ON | NMOSD-ON vs. MS-ON | ||||||
| Segmental enhancement of AVP—no. (%) | ||||||||
| Orbital optic nerve | 40 (52.6) | 15 (37.5) | 17 (70.8) | 8 (66.7) | 0.02 | 0.01 | 0.073 | 0.544 |
| Intracanalicular optic nerve | 10 (13.2) | 2 (5.0) | 4 (16.7) | 4 (33.3) | 0.032 | 0.135 | 0.021 | 0.236 |
| Intracranial optic nerve | 6 (7.9) | 1 (2.5) | 0 (0) | 1 (8.3) | 0.126 | 0.061 | 0.412 | 0.451 |
| Optic chiasm | 0 (0) | 0 (0) | 0 (0) | 0 (0) | - | - | - | - |
| Optic tract | 0 (0) | 0 (0) | 0 (0) | 0 (0) | - | - | - | - |
| ≥2 consecutive segments | 10 (13.2) | 2 (5.0) | 4 (16.7) | 4 (33.3) | 0.032 | 0.135 | 0.021 | 0.236 |
| Characteristic | Total Eyes (n = 76) | DN-ON (n = 40) | NMOSD-ON (n = 24) | MS-ON (n = 12) | p-Value | Subgroup Analysis (p-Value) | ||
|---|---|---|---|---|---|---|---|---|
| DN-ON vs. NMOSD-ON | DN-ON vs. MS-ON | NMOSD-ON vs. MS-ON | ||||||
| Number of patients | 61 (100) | 33 (54.1) | 18 (29.5) | 10 (16.4) | ||||
| Final BCVA—no. (%) | ||||||||
| LogMAR—mean ± SD | 0.58 ± 0.72 | 0.26 ± 0.42 | 1.23 ± 0.78 | 0.36 ± 0.5 | <0.001 | <0.001 | 0.771 | <0.001 |
| VA 20/20–20/60 | 46 (60.5) | 35 (87.5) | 2 (8.3) | 9 (75.0) | <0.001 | <0.001 | 0.561 | <0.001 |
| VA 20/70–20/200 | 11 (14.5) | 2 (5.0) | 8 (33.3) | 1 (8.3) | ||||
| VA < 20/200 | 19 (25.0) | 3 (7.5) | 14 (58.3) | 2 (16.7) | ||||
| Mean difference of final VA vs. at nadir | 0.33 ± 0.34 | 0.32 ± 0.23 | 0.27 ± 0.38 | 0.48 ± 0.53 | 0.002 | 0.004 | 0.131 | 0.006 |
| Visual acuity outcome—no. (%) | 0.022 | 0.092 | 0.176 | 0.003 | ||||
| Improve (≥0.3 LogMAR) | 2 (2.6) | 1 (2.5) | 0 (0) | 1 (8.3) | - | - | - | - |
| Stable | 50 (65.8) | 25 (62.5) | 21 (87.5) | 4 (33.3) | - | - | - | - |
| Worse (≤−0.3 LogMAR) | 24 (31.6) | 14 (35.0) | 3 (12.5) | 7 (58.3) | - | - | - | - |
| Color vision improvement (%) | 29 (38.2) | 19 (47.5) | 7 (29.2) | 3 (25.0) | 0.204 | 0.118 | 0.147 | 0.56 |
| Recurrent—no. (%) | 3 (2.6) | 1 (2.5) | 0 (0) | 2 (16.6) | 0.103 | 0.625 | 0.129 | 0.105 |
| OR | 95% CI | p-Value | |
|---|---|---|---|
| Univariable model | |||
| Age ≥ 50 yrs | 1.33 | 0.468–3.781 | 0.59 |
| Female gender | 0.96 | 0.298–3.111 | 0.95 |
| Duration ≥ 7 dys | 2.65 | 1.000–7.007 | 0.05 |
| Disc swelling | 1.69 | 0.675–4.239 | 0.262 |
| MRI ≥ 2 segments | 2.31 | 0.593–8.974 | 0.227 |
| DN-ON | 0.16 | 0.060–0.449 | <0.001 |
| NMOSD-ON | 16.67 | 4.763–58.314 | <0.001 |
| MS-ON | 0.4 | 0.100–1.625 | 0.201 |
| Poor VA at nadir | 16.19 | 5.097–51.398 | <0.001 |
| Multivariable model | |||
| Duration ≥ 7 dys | 5.29 | 1.359–20.616 | 0.016 |
| DN-ON | 1.4 | 0.236–8.305 | 0.711 |
| NMOSD-ON | 10.47 | 1.095–99.993 | 0.041 |
| Poor VA at nadir | 4.7 | 0.767–28.78 | 0.094 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kemchoknatee, P.; Singhakul, C.; Arjkongharn, N.; Chainakul, M.; Tangon, D.; Srisombut, T. A 10-Year Single-Center Study of the Clinical Characteristics of Optic Neuritis-Related NMOSD, MS, and Double Seronegative Optic Neuritis, Together with Factors Predicting Visual Outcomes. Vision 2023, 7, 16. https://doi.org/10.3390/vision7010016
Kemchoknatee P, Singhakul C, Arjkongharn N, Chainakul M, Tangon D, Srisombut T. A 10-Year Single-Center Study of the Clinical Characteristics of Optic Neuritis-Related NMOSD, MS, and Double Seronegative Optic Neuritis, Together with Factors Predicting Visual Outcomes. Vision. 2023; 7(1):16. https://doi.org/10.3390/vision7010016
Chicago/Turabian StyleKemchoknatee, Parinee, Chotika Singhakul, Niracha Arjkongharn, Methaphon Chainakul, Duanghathai Tangon, and Thansit Srisombut. 2023. "A 10-Year Single-Center Study of the Clinical Characteristics of Optic Neuritis-Related NMOSD, MS, and Double Seronegative Optic Neuritis, Together with Factors Predicting Visual Outcomes" Vision 7, no. 1: 16. https://doi.org/10.3390/vision7010016
APA StyleKemchoknatee, P., Singhakul, C., Arjkongharn, N., Chainakul, M., Tangon, D., & Srisombut, T. (2023). A 10-Year Single-Center Study of the Clinical Characteristics of Optic Neuritis-Related NMOSD, MS, and Double Seronegative Optic Neuritis, Together with Factors Predicting Visual Outcomes. Vision, 7(1), 16. https://doi.org/10.3390/vision7010016





