Abstract
The rapid increase in end-of-life lithium-ion batteries demands sustainable recycling routes for lithium recovery. This work presents a novel integrated hydrometallurgical–electrodialysis process designed specifically for recovering lithium from off-specification NMC cathode materials while enabling full reagent recyclability. Selective leaching with oxalic acid was optimised by setting the water-to-oxalic acid dihydrate ratio (H2O/OA·2H2O) to 7.3:1 w/w, achieving 81% lithium extraction at room temperature within 2 h while limiting the co-dissolution of Ni, Co and Mn to 0.2%, 1.6% and 1.7% by weight, respectively. The resulting leachate was processed in a four-chamber electrodialysis cell equipped with two Nafion 117 cation-exchange membranes and one Neosepta AMX-fmg anion-exchange membrane operating at −1.6 V versus Ag/AgCl, enabling 96% lithium recovery and 98% oxalic acid recovery. The regenerated oxalic acid stream (41.8 g L−1) was fully restored to its initial concentration and reused in successive cycles without performance loss. Subsequent precipitation of lithium with Na2CO3 yielded 99.3%-pure Li2CO3. This combined leaching–electrodialysis–precipitation presents a high selectivity, low-waste, circular recovery system, offering a scientifically original approach that integrates reagent regeneration with high-purity lithium production.
1. Introduction
The rapidly growing demand for lithium-ion batteries (LIBs), driven by the energy transition and electric mobility, will generate an accelerated accumulation of end-of-life waste (EoL). It is estimated that up to 4 million tons of LIB waste could be produced from electric vehicle batteries by 2040 [1]. These wastes contain critical metals such as lithium (Li), nickel (Ni), cobalt (Co), and manganese (Mn), whose recovery is essential to mitigate resource scarcity and to reduce the environmental impact associated with primary extraction. Conventional recycling routes—pyrometallurgy and hydrometallurgy—face significant challenges for a circular economy approach, where the substitution of toxic substances, the design of closed-loop processes that lower environmental impact, and the adoption of zero-waste strategies are key objectives [2]. Conventional hydrometallurgy employs inorganic acids such as sulphuric acid (H2SO4) [3], hydrochloric acid (HCl) [4], or nitric acid (HNO3) [5] for leaching the metals contained in LIB cathodes. These acids are attractive because of their high dissolution power and low cost, but they are highly corrosive and generate toxic fumes, large volumes of wastewater, and expensive neutralisation steps. In some studies, H2SO4 leaching is performed in the presence of reducing agents such as hydrogen peroxide (H2O2) [6]; the reducing agent converts Co(III) to the more soluble Co(II), improving extraction performance. Using 1 M H2SO4 at room temperature, a solid to liquid (S/L) ratio of 50 g L−1 and a 90 min reaction time, extraction yields above 99% wt. for Ni, Co, Mn, and Li have been achieved [7]. Other investigations avoid the reducer by applying more aggressive leaching conditions, achieving complete recovery of Ni, Co, and Li and 93% wt. of Mn using 1 M H2SO4, S/L = 1:10 at 90 °C for 1 h [8].
Hydrochloric acid leaching has been less explored because of its high corrosivity, which raises capital expenditure (CAPEX) due to the need for highly resistant materials such as graphite, titanium, or Hastelloy C, although coated vessels can reduce the investment. A notable advantage of HCl leaching is the subsequent separation of Co, Ni, and Mn by solvent extraction techniques [9]. Several studies have shown that hydrochloric acid can surpass sulphuric acid, achieving near-complete extraction of cobalt and lithium when 2 M HCl is employed at 60–80 °C, with an S/L ratio of 1:50 and a reaction time of 90 min [10].
Although effective, these acid-based routes are less sustainable than alternatives that employ other classes of chemicals, such as deep eutectic solvents (DESs) or organic acids. Consequently, the development of innovative, more sustainable, efficient, and cost-effective processes is a current challenge for the scientific community.
Deep eutectic solvents have attracted attention because of their low toxicity and their ability to dissolve metals from NMC (LiNixMnγCozO2) cathodes. A recent study using an equimolar mixture of choline chloride and oxalic acid (S/L = 1:100) achieved Li recoveries between 61% and 99% wt. depending on the temperature (60–120 °C) and leaching time (5–120 min). Mn leaching efficiencies ranged from 57% to 83% wt., while Ni and Co showed limited solubility in this DES [11]. DESs based on ethyl-ammonium or diethyl-ammonium chloride combined with ethylene glycol have also been investigated over molar ratios of 1:2 to 1:5. At a DES/cathode ratio of 20 g L−1, optimal leaching at 100 °C for 24 h yielded 84% wt. Li, 98% wt. Ni and Co, and 50% wt. Mn, underscoring the viability of DES as a greener alternative to volatile conventional acids [6].
Modifying DES composition with functional additives further improves performance. A DES comprising choline chloride, lactic acid, and a third component acting as a reductant (ascorbic acid) achieved >99% wt. extraction of Li and Mn from LiMn2O4 (LMO) cathodes at a molar ratio of 1:2:1, 90 °C, 1 h and an S/L of 20 g L−1 [12]. The reductant reduces Mn(III/IV) to Mn(II), facilitating complete leaching. Similarly, a DES of choline chloride, glycolic acid, and ascorbic acid (1:1.9:0.1) leached >95% wt. of Li, Co, Ni, and Mn from NMC batteries at S/L = 10 g L−1, 60 °C, and 1 h [13].
Nevertheless, DES-based methods face limitations that must be addressed for industrial implementation. High temperatures (60–120 °C) are often required to achieve sufficient metal solubility, increasing energy consumption and diminishing economic attractiveness. The sensitivity of the process to parameters such as DES component molar ratios or solid-to-liquid ratios hampers reproducibility and scalability. Moreover, the high viscosity of many DESs, especially at lower temperatures, can limit leaching kinetics.
Despite these challenges, DESs present substantial potential for sustainable recycling, and novel strategies can help overcome current drawbacks. For example, the application of ultrasound to a DES composed of β-alanine, citric acid, and water achieved recoveries of 97.8% wt. Li, 95.5% wt. Co, 97.9% wt. Mn and 96.7% wt. Ni within 1 h at 80 °C, compared with 3 h required without ultrasound. The DES could be reused up to five cycles without loss of efficiency, the improvement being attributed to cavitation-induced turbulence, which enhances mass transfer [14].
The use of organic acids—citric [15,16,17,18], oxalic [19,20,21,22,23], tartaric [24,25,26], lactic [27,28], levulinic [29,30] and fumaric [31]—has also been investigated for the recovery of metals from battery cathodes. These compounds are emerging as sustainable alternatives to mineral acids in LIB hydrometallurgy because they reduce the generation of saline effluents and toxic emissions, supporting circular economy goals and compliance with environmental regulations [32]. Their chelating ability enables the design of selective leaching processes that lower the number of downstream purification steps, thereby decreasing operating costs and improving product quality. Table 1 summarises data extracted from numerous studies on the recovery of lithium and transition metals (Co, Ni, Mn) from LIB cathodes of various chemistries using hydrometallurgical processes based on organic acids. As in the preceding examples, these leaching protocols incorporate strategies such as microwave (MW) heating or the addition of reductants to facilitate transition metal dissolution. High extraction efficiencies are achieved for all metals, indicating that organic acid leaching could become an industrially attractive solution once the economic limitations are resolved. The primary economic limitations arise from the high cost of reagents and the significant energy requirements associated with operating at elevated temperatures. Therefore, research focused on reducing these costs is essential for the practical implementation of the process. The selectivity of oxalic acid for lithium leaching is particularly noteworthy, as it can be exploited in diversified cathode valorisation pathways.

Table 1.
Extraction processes for Li using organic acids across different lithium-ion-battery chemistries.
In this context, the reuse of the organic acid employed becomes essential to make metal recovery processes more competitive. Various regeneration strategies have been reported, notably electrodialysis [33,34,35], solvent extraction [36,37,38], and fractional crystallisation [39,40]. Electrodialysis (ED) uses selective membranes to separate ions and to regenerate the acid, delivering high purity and relatively low energy consumption, and is thus a promising technology with high development potential in this field. Solvent extraction enables the simultaneous recovery of Ni, Co, and Mn, along with acid, by contacting the leach solution with liquid extractants. The technique can achieve recovery rates exceeding 90% wt. together with high selectivity, but it involves the use of organic solvents that must be managed to avoid environmental impact. Fractional crystallisation exploits differences in solubility to separate the acid from the leachate. It is effective for highly concentrated systems, although it is energy-intensive and less flexible than the other technologies when dealing with complex mixtures. These regeneration processes are pivotal for closing the leachate loop, reducing costs, and minimising waste, thereby enhancing the overall sustainability of battery recycling. Table 2 provides a comparative overview of the main characteristics of the different organic compound recovery techniques.

Table 2.
Properties of various organic compound recovery processes.
Aside from the black mass generated from EoL batteries, other waste streams of comparable composition are scarcely reported in the literature. Off-specification cathodes (OSC) are residues produced during battery manufacturing that fail to meet production standards. The reasons for rejection are varied but are generally linked to manufacturing and handling processes. The most common causes include deviations in chemical composition (incorrect ratios of Ni, Co, Mn, or Fe), contamination by metallic impurities during production, excess moisture in the active material, physical defects such as inappropriate particle size distribution or non-uniform density, and failures in quality control at critical stages such as mixing, coating, and drying [41]. These factors diminish electrochemical performance and present safety hazards, such as internal short-circuits and thermal runaway. At an industrial scale, some recovery schemes combine pyrometallurgical and hydrometallurgical techniques to retrieve the higher value metals (Ni and Co). However, when thermal recovery routes are employed, the lithium contained in these residues ends up incorporated in the slag, making its subsequent extraction considerably more difficult [42].
The present work proposes a novel solution for lithium valorisation based on an integrated hydrometallurgical process in which lithium is selectively leached, recovered, and converted into Li2CO3, while the oxalic acid lixiviant is simultaneously regenerated and reused through an electrodialysis step. This closed-loop configuration minimises reagent consumption, reduces waste generation, and enhances the circularity of the process, distinguishing it from previously reported approaches where leaching, acid recovery, and lithium precipitation are typically treated as independent operations. Although oxalic acid has been previously employed for lithium recovery from black mass obtained from various battery types, its behaviour in cathode manufacturing residues such as OSCs has not yet been evaluated. This makes the current study particularly relevant, as it explores the potential of this selective acid in a previously unassessed waste stream. Oxalic acid is particularly suitable for this application due to its high selectivity towards lithium leaching, minimising the co-extraction of transition metals such as Ni, Co, and Mn. This selectivity enables a more efficient and environmentally friendly recovery process, reducing downstream purification needs. Although oxalic acid presents certain handling risks—such as skin and eye irritation or burns, and adverse effects if ingested—its overall hazard profile is lower than that of strong mineral acids typically used in hydrometallurgical processes. Regarding flammability, oxalic acid poses only a minor risk; however, small amounts of toxic gases may be released if involved in a fire, making basic containment and protective measures advisable. Overall, its risks are moderate and readily manageable through standard safety practices, while also offering a reduced environmental and operational impact compared with more aggressive acids such as hydrochloric, sulfuric, or nitric acid. Moreover, electrodialysis—a technology with significant development potential in battery recycling but still underexplored in the valorisation of spent battery materials—offers a promising route for lithium recovery and acid regeneration. Notably, this technology has never been applied to the recovery of oxalic acid from leachates generated during the treatment of cathode residues such as OSCs, making its use in this work a clear and distinctive innovative contribution. In the proposed process, the electrodialysis stage extracts lithium from the leachate while regenerating the oxalic acid for reuse—without loss of efficacy—in subsequent leaching cycles. Finally, the lithium recovered in the electrodialysis stream is precipitated as Li2CO3 by reaction with Na2CO3, yielding a product of 99.3% purity—close to the ≥ 99.5% purity required for battery-grade material.
2. Materials and Methods
2.1. Materials and Reagents
All reagents were supplied by Scharlab (Barcelona, Spain) and were of analytical grade: 99% oxalic acid dihydrate (H2C2O4·2H2O), 98% sulphuric acid (H2SO4), 48% hydrofluoric acid (HF), 98% lithium hydroxide monohydrate (LiOH·H2O), 99.5% sodium carbonate (Na2CO3), 99.5% sodium hydrogen carbonate (NaHCO3) and 99% sodium chloride (NaCl). CPAChem-certified reference materials for Li, Co, Ni, Mn, and S, and Sigma-Aldrich oxalate standard for ion chromatography (1000 mg L−1 each) were supplied by Vidrafoc (Barcelona, Spain). The off-specification cathode (OSC) analysed herein was sourced from the LICORNE project and identified as NMC532 based on elemental composition. Table 3 summarises its chemical composition determined by ICP-OES.

Table 3.
ICP-OES characterisation of the off-specification cathode.
Lithium content (6.5%) was below the commercial NMC532 specification (7.0–7.6%), while mole fractions for Ni, Mn, and Co were 0.53, 0.33, and 0.27, respectively, exceeding the nominal values of 0.50 ± 0.01, 0.30 ± 0.01, and 0.20 ± 0.01 [43]. These deviations indicate a significant departure from the stoichiometric balance between lithium and transition metals required for optimal electrochemical performance [44]. Since sintering relies on the formation of a stable and homogeneous crystalline structure, the non-stoichiometric composition of the OSC material generates structural defects that lead to reduced capacity, cycle life, safety, and power output. Furthermore, the integration of this material into a sintering process generates low phase purity, compromising the performance of the final product. Therefore, alternative approaches, such as recycling, should be explored to recover the value of this material without incurring the risks associated with its use in conventional high-temperature manufacturing routes.
2.2. Characterisation Methods
2.2.1. Inductively Coupled Plasma Optic Emission Spectrometry (ICP-OES)
Solid samples were digested in a Milestone Ethos UP microwave system SK-15 rotor (Sorisole, Italy) at 200 °C with a 10 mL acid mixture comprising 2 mL 48% HF, 4 mL 37% HCl, and 4 mL 69% HNO3. Measurements were performed using an Agilent 5800 Vertical Dual View ICP-OES with Agilent SPS 4 autosampler (Santa Clara, CA, USA). The introduction system included a OneNeb nebuliser, an inert double-pass cyclonic spray chamber, and an Agilent Easy-fit semi-demountable VDV torch with a 1.8 mm alumina injector. Instrument control was via ICP Expert 7.6 software.
2.2.2. X-Ray Diffraction (XRD)
Diffraction patterns were recorded on a Bruker D8 Advance diffractometer (Karlsruhe, Germany) using Cu Kα radiation (λ = 1.5406 Å) and a scintillation detector. Scans ranged from 2θ = 15° to 70°, with 0.03° steps and 2 s dwell time. Phase analysis was performed using X’Pert HighScore 1.0 software.
2.2.3. Ion Chromatography
Oxalates were quantified by Metrohm 930 Compact IC Flex (Herisau, Switzerland) with chemical suppression and conductivity detection. Sample introduction was automated via Metrohm 919 Autosampler Plus. Separation was conducted on Metrosep A Supp 19 (250 × 4.0 mm).
2.3. Off-Specification Cathode Leaching
Leaching experiments were conducted in electrically heated reactors (250 mL and 1000 mL, max capacity) with an aluminium block and temperature-controlled hot-plate (IKA C Mag HS7 Controller, Staufen, Germany, or Scharlau Premium Reactor, Barcelona, Spain). The pH was monitored with an Endress + Hauser Liquiline CM442 transmitter equipped with a Memosens CPS16D pH/ORP probe (Reinach, Switzerland), which includes a temperature sensor accurate to ± 1 °C. Once the target temperature was reached, the leaching agent was contacted with the OSC sample under defined conditions. The slurry was vacuum-filtered using a Büchner funnel with a sintered glass disc, collection flask, and vacuum pump, employing 47 mm glass microfibre filters (Scharlab). The resulting filter cake was rinsed with deionised water and dried at 105 ± 5 °C for 24 h in a drying oven. The leachate, the washing solution, and the dried solid were all quantified gravimetrically to ensure accurate mass balance calculations. Variables studied included pH, stoichiometry, temperature and leaching time. Lithium, nickel, cobalt, and manganese were quantified in initial OSC, precipitates, leachates, and rinse solutions by ICP-OES (see Section 2.2). All experimental runs were performed at least in duplicate, and each liquid and solid sample was analysed by ICP-OES with four independent measurements. Considering the uncertainties associated with the calibration standards (1%), the instrumental variability (<3%), and the dispersion observed between experimental replicates (<5%), a combined uncertainty of 6% was used to contextualise the reliability of the mass balance results. Mass balance closures ranged from 92 to 103%, which is fully consistent with the expected cumulative analytical and experimental variability. These small deviations can be attributed not only to analytical dispersion but also to minor losses occurring during sample handling, such as transfers, filtration, and washing steps, which are inherent to multistep hydrometallurgical processes.
Extraction yields were calculated as
where mp,E and mi,E are masses (g) of element E in leachate and initial OSC, respectively.
2.4. Organic-Based Membrane Electrodialysis
Electrodialysis experiments were conducted using a BIOLOGIC VSP 3e potentiostat (Seyssinet-Pariset, France) coupled with a Micro Flow Cell MFC30010, active electrode area: 5 × 2 cm, supplied by ElectroCell (Tarm, Denmark). The working electrode (WE) was a stainless-steel cathode, the counter electrode (CE) a dimensionally stable anode (DSA), and Ag/AgCl served as the reference electrode. Membrane selection (both anion and cation exchange, Ø 16 mm) was performed in a 25 mL electrochemical H-cell using stainless-steel cathodes as WE, a platinum anode as CE, and Ag/AgCl as the reference electrode. Chronoamperometry was employed as the electrochemical technique in all tests.
The electrodialysis performance was assessed based on recovery yield for lithium and oxalic acid (OA). Lithium and OA recovery yields (Yx, %) were calculated as the ratio between the mass of Li or OA transferred to the catholyte or anolyte and the initial Li or OA content in the Li-bearing solution at the start of the experiment:
where Xcath./anol. and Xws represent the masses (g) of lithium or OA transferred to the catholyte or anolyte and lithium or OA contained in the working solution, respectively.
2.5. Lithium Carbonate Precipitation
Precipitation experiments were conducted in electrically heated reactors (100 mL) with an aluminium block and a temperature-controlled hot-plate (IKA C Mag HS7 Controller). The pH was monitored using an Endress+Hauser Liquiline CM442 transmitter with a Memosens CPS16D pH/ORP probe, which includes a temperature sensor (accuracy ± 1 °C).
The feed solution consisted of the catholyte obtained from the electrodialysis step, which had an initial pH of 12.6. Prior to precipitation, the solution was concentrated by evaporation until a lithium concentration of at least 10 g L−1 was achieved. Lithium carbonate was precipitated at 90 °C by adding a Na2CO3 solution. After completion of the precipitation step, the suspension was filtered using a Büchner funnel equipped with a sintered glass disc, a collection flask, and a vacuum pump. Filtration was performed using 47 mm nylon membrane filters (Scharlab). The recovered solid was subsequently washed to remove residual impurities.
3. Results and Discussion
3.1. Selective Extraction of Lithium by Oxalic Acid Leaching
3.1.1. Effect of Water-to-Oxalic Acid Ratio
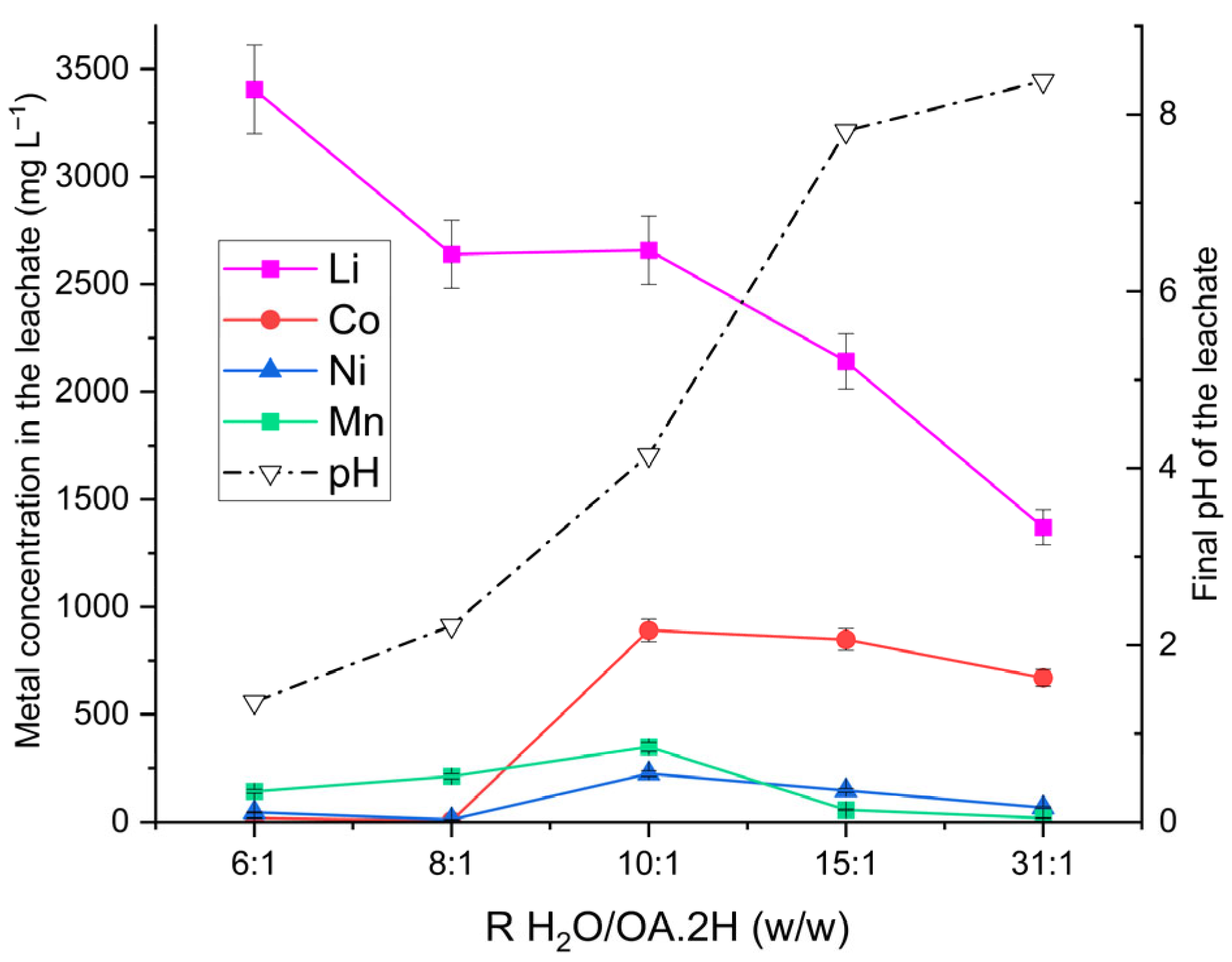
The effect of the water-to-oxalic acid dihydrate (H2O/OA·2H) mass ratio on lithium selectivity was investigated through a series of experiments using 100 g of leaching solution at ratios of 6:1, 8:1, 10:1, 15:1, and 31:1 (w/w). An OSC-to-leaching solution mass ratio (S/L) of 1:20 was maintained, with temperature fixed at 55 °C, reaction time of 1 h, and stirring at 320 rpm. After each run, the final pH of the slurry was measured to assess the impact of solution conditions on metal selectivity. ICP-OES analysis of the leachates is presented in Figure 1.
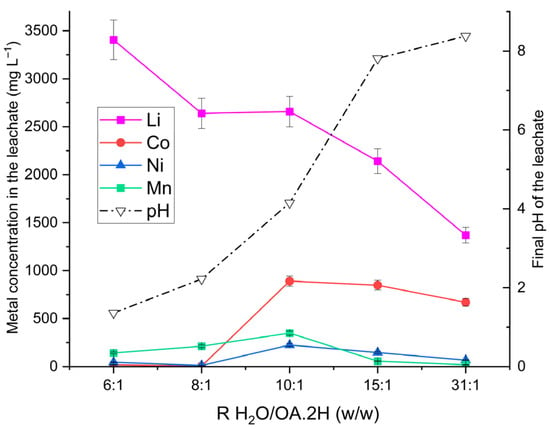
Figure 1.
Characterisation of leachates as a function of leaching solution formulation (H2O/OA·2H ratio, w/w). Operating conditions: S/L = 1:20 w/w (100 g solution and 5 g OSC containing 325 mg Li, 1460 mg Ni, 745 mg Co and 855 mg Mn), 55 °C, 325 rpm stirring, 1 h reaction time. The right-hand y-axis shows the final pH measured for each formulation.
The results demonstrate the high lithium selectivity of oxalic acid leaching, aligning with earlier findings [19,22,23]. Lithium concentrations between 2658 mg L−1 and 3405 mg L−1 were obtained for H2O/OA·2H ratios of 6:1 to 10:1 w/w, corresponding to extraction yields of approximately 77% wt. Increasing the water proportion sharply reduced the yield to 40% for the 31:1 formulation, with Li concentration falling to 1359 mg L−1. For Ni and Co, concentrations remained below 50 mg L−1 at ratios of 6:1 and 8:1, while Mn concentrations were 142 mg L−1 and 212 mg L−1, respectively.
Oxalic acid is a diprotic acid, and its degree of deprotonation depends on solution pH. The dissociation constants are
All formulated solutions had initial pH values below 1.27; thus, the dominant species was fully protonated H2C2O4, which does not readily form complexes. Under these conditions, transition metals tend to precipitate as insoluble oxalates. The overall leaching reaction can be expressed as [45]:
As oxalic acid is consumed during leaching, the solution pH rises. For highly aqueous formulations (H2O/OA·2H ratios of 10:1, 15:1, and 31:1 w/w), final pH values ranged from 4.2 to 8.4, favouring complex formation with transition metals. Literature [46] reports indicate that weak Ni, Co, and Mn complexes with HC2O4− appear at pH ≈ 2, while at pH ≈ 4, the dianion C2O42− predominates, forming more stable complexes and increasing metal oxalate solubility.
Figure 1 clearly illustrates the effect of the H2O/OA·2H mass ratio on the final pH of the leachate and, consequently, on the dissolution of transition metal: Ni and Co concentrations remain very low at pH < 2 but increase markedly with rising pH. Mn concentrations increase with pH, reaching their maximum around pH 4, which reflects the higher solubility of manganese oxalate under these mildly acidic conditions [47].
From these observations, it can be concluded that to achieve high lithium selectivity during oxalic acid leaching, the leaching agent should have an H2O/OA·2H ratio not exceeding 8:1 by weight, which allows the pH to be maintained below 2 under the studied S/L conditions.
3.1.2. Effect of Oxalic Acid Stoichiometry
Leaching experiments were conducted using 100 g of an H2O/OA·2H2O = 6:1 (w/w) solution. Five S/L ratios (1:5, 1:10, 1:17, 1:20, and 1:50, w/w) were evaluated at 55 °C for 1 h under magnetic stirring (320 rpm). The amount of oxalic acid used in each experiment was defined based on the stoichiometry of the reaction described in Equation (5). This value was calculated according to Equation (6):
where OA stoichiometry is the mass of oxalic acid used expressed as a percentage, OA is the mass of oxalic acid used in the assay (g), NMC represents the number of moles of the mixed metal oxide constituting the cathode material, and PmOA is the molar mass of oxalic acid (g mol−1). The factor of 2 accounts for the stoichiometric coefficient of oxalic acid in the considered reaction.
The effect of “oxalic acid stoichiometry” was investigated to assess its influence on lithium extraction and transition metal dissolution. This value indicates whether the system is deficient (<100%) or in excess (>100%) relative to the metal load.
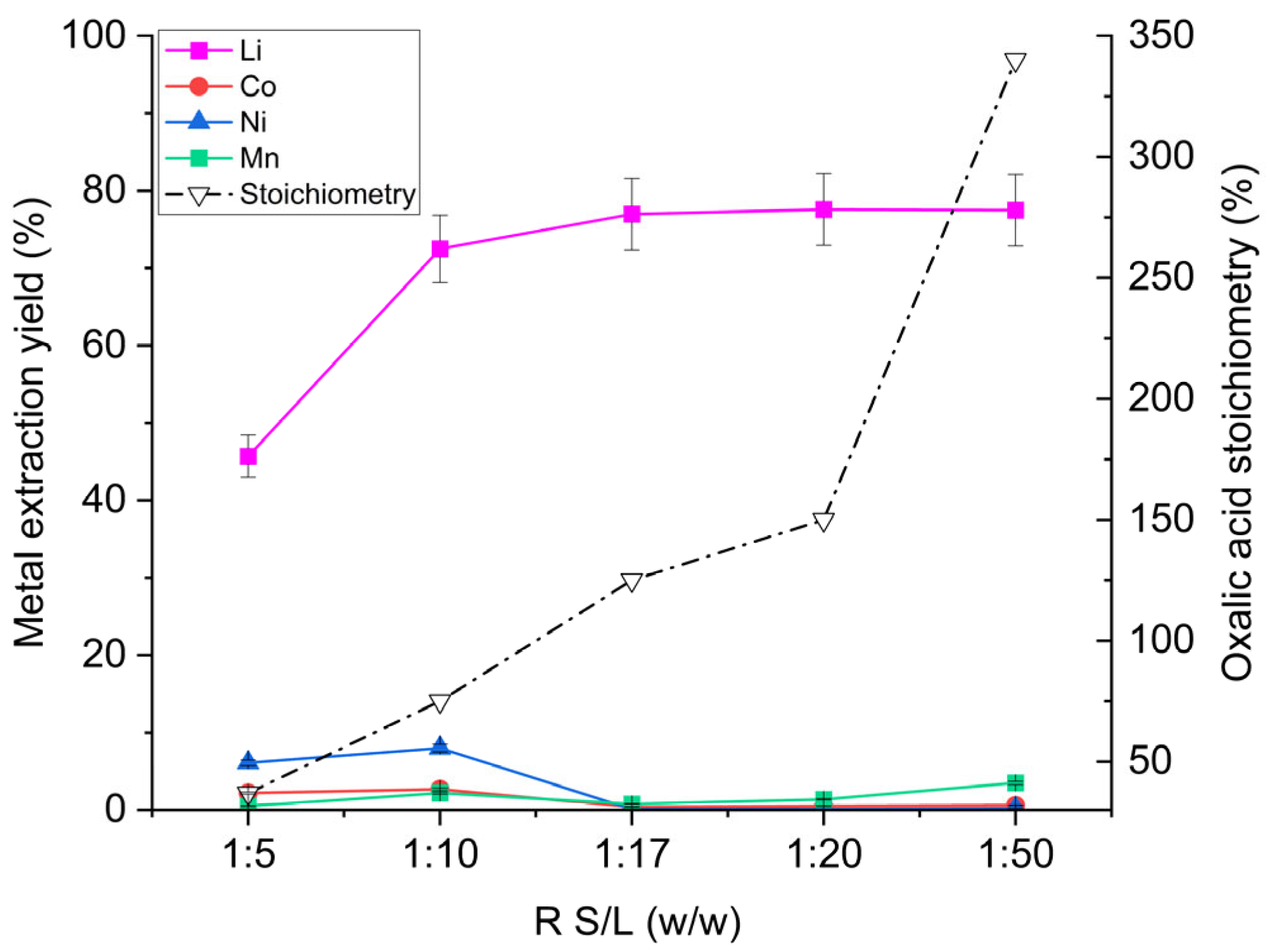
Figure 2 illustrates the effect of S/L ratio on extraction performance. At an S/L ratio of 1:5 w/w, lithium extraction was very low (46%). Increasing the liquid proportion to 1:10 raised lithium recovery to 73%. Under these two conditions, nickel and cobalt extraction values were approximately 2.5% and 7%, respectively, indicating substantial formation of Ni and Co oxalate complexes (final leachate pH was 8.6 for S/L 1:5 and 8.9 for S/L 1:10 w/w). When the leaching solution volume was further increased relative to the cathode material (S/L = 1:17 w/w), the trend changed completely: lithium extraction reached its maximum, while the remaining metals remained almost entirely in the solid phase (≈ 100% retention). Beyond this ratio, lithium extraction remained essentially constant; however, the extraction of Ni, Co, and Mn progressively increased owing to the solubility of their respective oxalates. Transition metal dissolution rose because a larger solution volume was available per unit mass of solid. Consequently, nickel and cobalt leaching increased from 0.1% and 0.4% by weight at S/L = 1:17 w/w to 0.2% and 0.6% at 1:50 w/w, respectively, while manganese leaching rose from 0.8% to 3.5%.
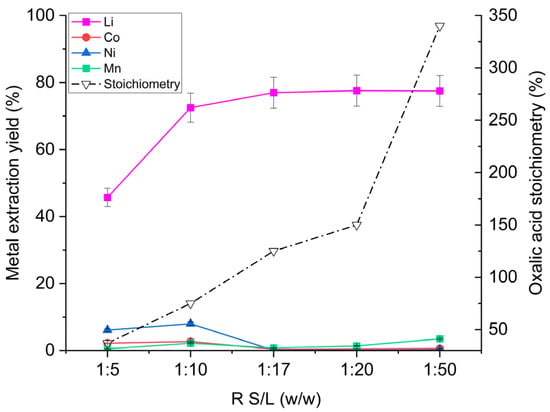
Figure 2.
Metal extraction yields from the off-specification cathode (OSC) as a function of the S/L mass ratio. Operating conditions: 100 g of leaching solution (H2O/OA·2H2O = 6:1 w/w), OSC masses 20–10–5.9–5.0–2.0 g, 55 °C, 325 rpm stirring, 1 h reaction time. The right-hand y-axis indicates the stoichiometric amount of oxalic acid present in solution, expressed as a percentage of the theoretical quantity required to react with all metals in the solid phase.
Figure 2 also plots the stoichiometric amount of oxalic acid present in each leaching solution. An excess of oxalic acid is required to maintain pH levels that suppress complexation, as discussed in Section 3.1.1. For the formulation used (H2O/OA·2H2O = 6:1 w/w), an S/L ratio of 1:17 w/w minimised Co, Ni, and Mn extraction, yielding a final leachate pH of 1.5. The figure further shows that an oxalic acid stoichiometry between 125% and 150% provides the best selectivity, maintaining extraction yields of 77–78% wt. for Li, 0.4–0.5% wt. for Co, 0.1% wt. for Ni, and 0.8–1.4% wt. for Mn.
To improve process economics, the H2O/OA·2H2O ratio and the S/L ratio were optimised. By maintaining constant OA stoichiometry, a higher oxalic acid concentration in the solution allows treatment of a greater mass of cathode waste. Accordingly, a new series of experiments was conducted in which both parameters were varied so that the leaching step was performed with 125% of the oxalic acid amount required to satisfy Equation 5. The extraction yields obtained for each experiment are listed in Table 4, confirming high lithium selectivity across all investigated ratios.

Table 4.
Extraction yields obtained at 125% stoichiometry for different leaching agent formulations. Operating conditions: 100 g of leaching solution, OSC masses 3.8–5.0–5.9–8.3–13.9 g, 55 °C, 325 rpm stirring, 1 h reaction time.
The leachate obtained with H2O/OA·2H2O = 2:1 w/w and S/L = 1:7.2 w/w crystallised oxalic acid upon cooling to room temperature. This behaviour is explained by the strong temperature dependence of oxalic acid solubility, which decreases from 376 g L−1 at 55 °C to 95.5 g L−1 at 20 °C [48]. Therefore, a higher water-to-acid ratio is required to maintain leachate stability at room temperature.
For optimal process economics, the H2O/OA·2H2O = 4:1 w/w formulation was selected for operation at 55 °C. Although this ratio initially contains more oxalic acid than its solubility limit at room temperature, the acid is partially consumed during the reaction, reducing its concentration below the solubility threshold by the end of the leaching step and preventing precipitation. A further cost-reduction strategy involves lowering the reaction temperature to ambient conditions. However, this affects the amount of oxalic acid that can remain in solution and, consequently, the mass of OSC that can be processed. When operating at 20 °C, the minimum H2O/OA·2H2O ratio must be calculated based on oxalic acid solubility at that temperature (95 g L−1). The S/L ratio is then adjusted to maintain overall stoichiometry at 125% of the theoretical requirement. Considering these factors, the process was evaluated at room temperature, and a leaching solution was prepared with H2O/OA·2H2O = 7.3:1 w/w and S/L = 1:20 w/w. These adjustments are intended to achieve high lithium selectivity, minimise reagent consumption, and ensure leachate stability throughout the process.
3.1.3. Effect of Temperature and Reaction Time
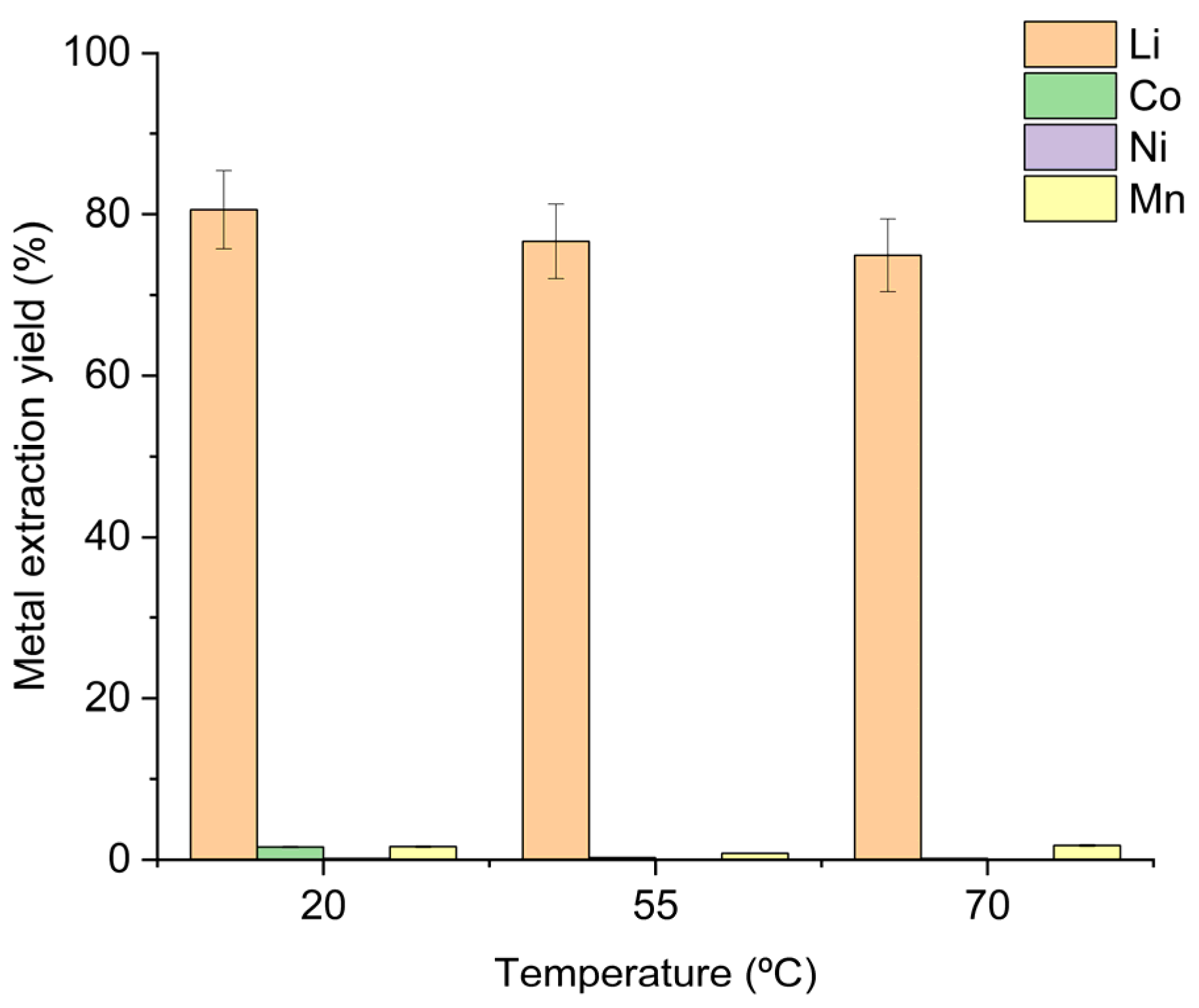
To evaluate the effect of lowering the temperature, the same leaching formulation described above (H2O/OA·2H2O = 7.3:1 w/w and S/L = 1:20 w/w) was used, providing a 125% stoichiometric excess of oxalic acid relative to the metal content. Leaching experiments were then performed at 20 °C for 2 h with magnetic stirring at 320 rpm. The results were compared with those obtained using the formulation selected in Section 3.1.2. (H2O/OA·2H2O = 4:1 w/w and S/L = 1:12 w/w) at 55 °C. To increase the number of evaluation points for temperature effect, an additional test was performed at 70 °C with H2O/OA·2H2O = 6:1 w/w and S/L= 1:17.3 w/w.
Leaching experiments performed over a 2 h period showed a slight improvement in lithium recovery as the temperature was lowered: 75% wt. at 70 °C, 77% wt. at 55 °C and 81% wt. at room temperature (Figure 3).
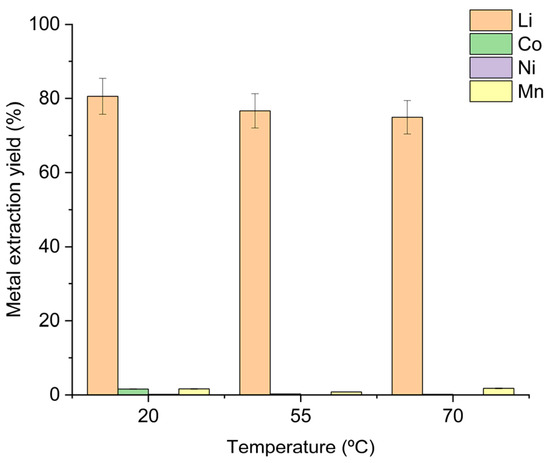
Figure 3.
Extraction yields obtained at different temperatures with 125% oxalic acid-to-metals stoichiometry. Operating conditions: 20 °C—100 g leaching solution (H2O/OA·2H2O = 7.3:1 w/w), OSC 5.0 g, 325 rpm stirring, 2 h reaction time; 55 °C—100 g leaching solution (H2O/OA·2H2O = 4:1 w/w), OSC 8.3 g, 325 rpm stirring, 2 h reaction time; 70 °C—100 g leaching solution (H2O/OA·2H2O = 6:1 w/w), OSC 5.8 g, 325 rpm stirring, 2 h reaction time.
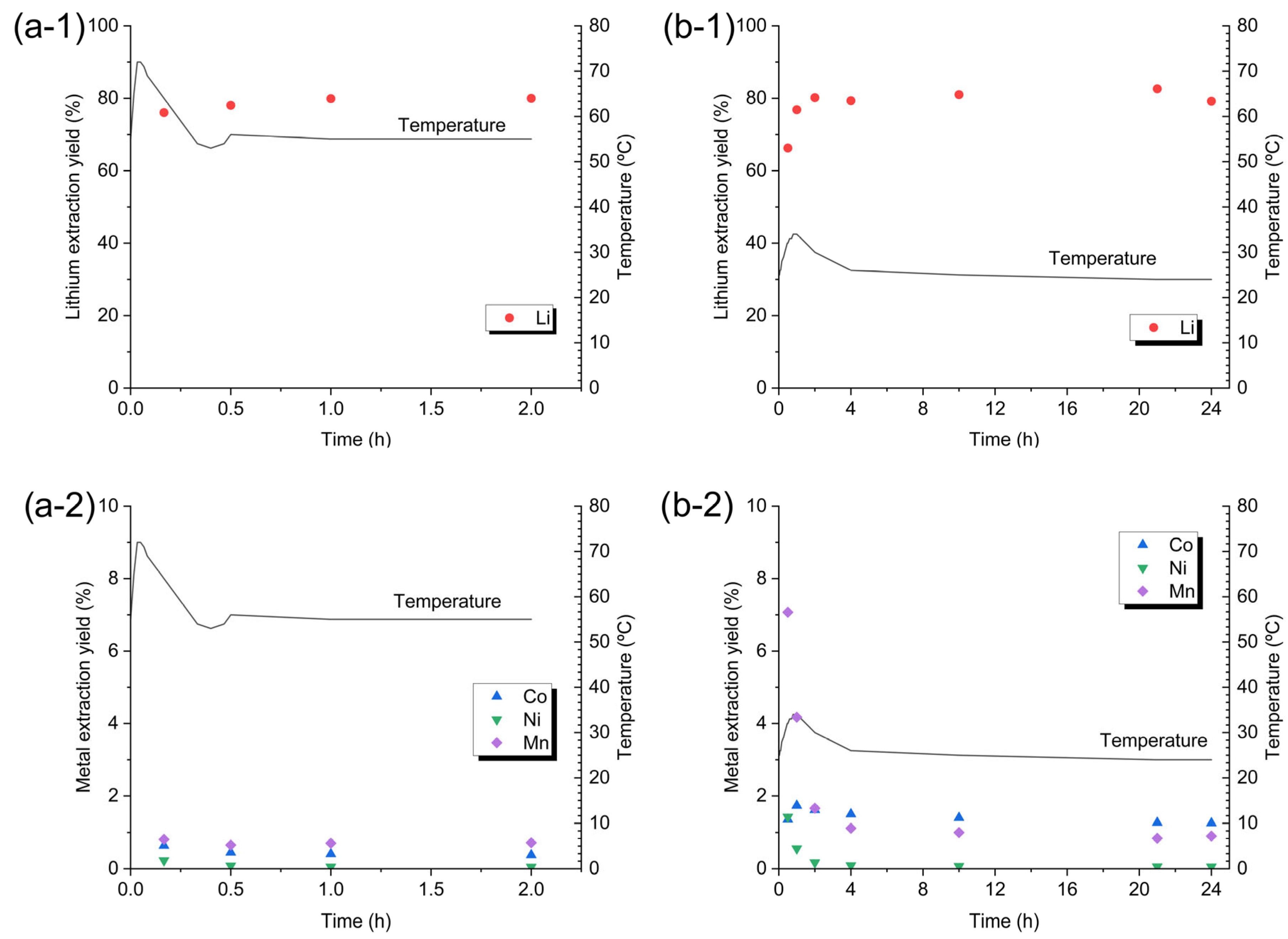
The corresponding lithium concentrations measured in the leachates were 3026 mg L−1 (70 °C), 4701 mg L−1 (55 °C), and 2798 mg L−1 (20 °C). Throughout the temperature range, the co-extraction of nickel, cobalt and manganese remained negligible, confirming that lithium selectivity is preserved irrespective of the operating temperature. Figure 4(a-1,a-2) shows the evolution of lithium and transition metal leaching, respectively, during a 2 h reaction at 55 °C, whereas Figure 4(b-1,b-2) presents the corresponding evolution at room temperature over 24 h. At 55 °C, the reaction is extremely fast and is complete within the first ten minutes; a temperature rise of ≈17 °C is observed after only two minutes of adding 8.3 g of OSC. This exothermic process releases CO2, which can cause reactor overflow. The substantial heat generation and rapid gas evolution represent a challenge for scale-up, requiring additional temperature control and reactor fill-level measures to prevent overflow. At room temperature, the rise in leachate temperature is considerably smaller (≈9 °C), and the reaction progresses over a longer period, indicating markedly slower kinetics.
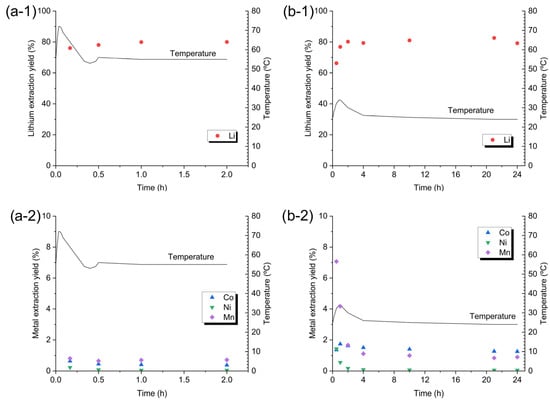
Figure 4.
Effect of reaction time on metal extraction from OSC and temperature evolution. In each plot, the right-hand y-axis displays temperature evolution as a function of time, and the left-hand y-axis displays metal extraction yields as % by weight. Operating conditions: (a) 55 °C, 100 g of leaching solution (H2O/OA·2H2O = 4:1 w/w), OSC 8.3 g, 325 rpm stirring; extraction yield: (a-1) Li and (a-2) transition metal. (b) Room temperature, 100 g of leaching solution (H2O/OA·2H2O = 7.3:1 w/w), OSC 5.0 g, 325 rpm stirring; extraction yield: (b-1) Li and (b-2) transition metal.
Under 55 °C conditions, lithium extraction and subsequent precipitation of transition metal oxalates are essentially complete within ten minutes. By contrast, the precipitation pathway at room temperature proceeds more gradually, with manganese requiring a substantially extended residence time to reach comparable extraction levels.
The decrease in transition metal extraction with increasing temperature is consistent with the van ’t Hoff equation. The formation of Ni, Co and Mn oxalate complexes is exothermic (ΔH° < 0); therefore, the stability constant (Kx) diminishes as temperature rises, leading to lower complex formation at higher temperatures.
The results demonstrate that lithium can be recovered efficiently at room temperature with excellent selectivity and low reaction time. On this basis, a larger-scale test (1000 g of leaching solution) was performed under the optimal room temperature conditions (H2O/OA·2H2O = 7.3:1 w/w, S/L = 1:20 w/w, 2 h, 325 rpm). The mass balance for this run is shown in Table 5. The resulting leachate was subsequently used as feed for lithium extraction by membrane electrodialysis in a four-chamber cell.

Table 5.
Mass balance for the selected room temperature leaching conditions (used as feed for membrane electrodialysis). Operating conditions: 1000 g of leaching solution (H2O/OA·2H2O = 7.3:1 w/w), S/L = 1:20 w/w, room temperature, 2 h reaction time, 325 rpm stirring.
The filter cake obtained after oxalic acid leaching consists primarily of insoluble oxalates of Ni, Co, and Mn, along with minor amounts of OSC unreacted. These compounds are widely recognised as suitable precursors for the synthesis of layered oxide cathode materials for lithium-ion batteries.
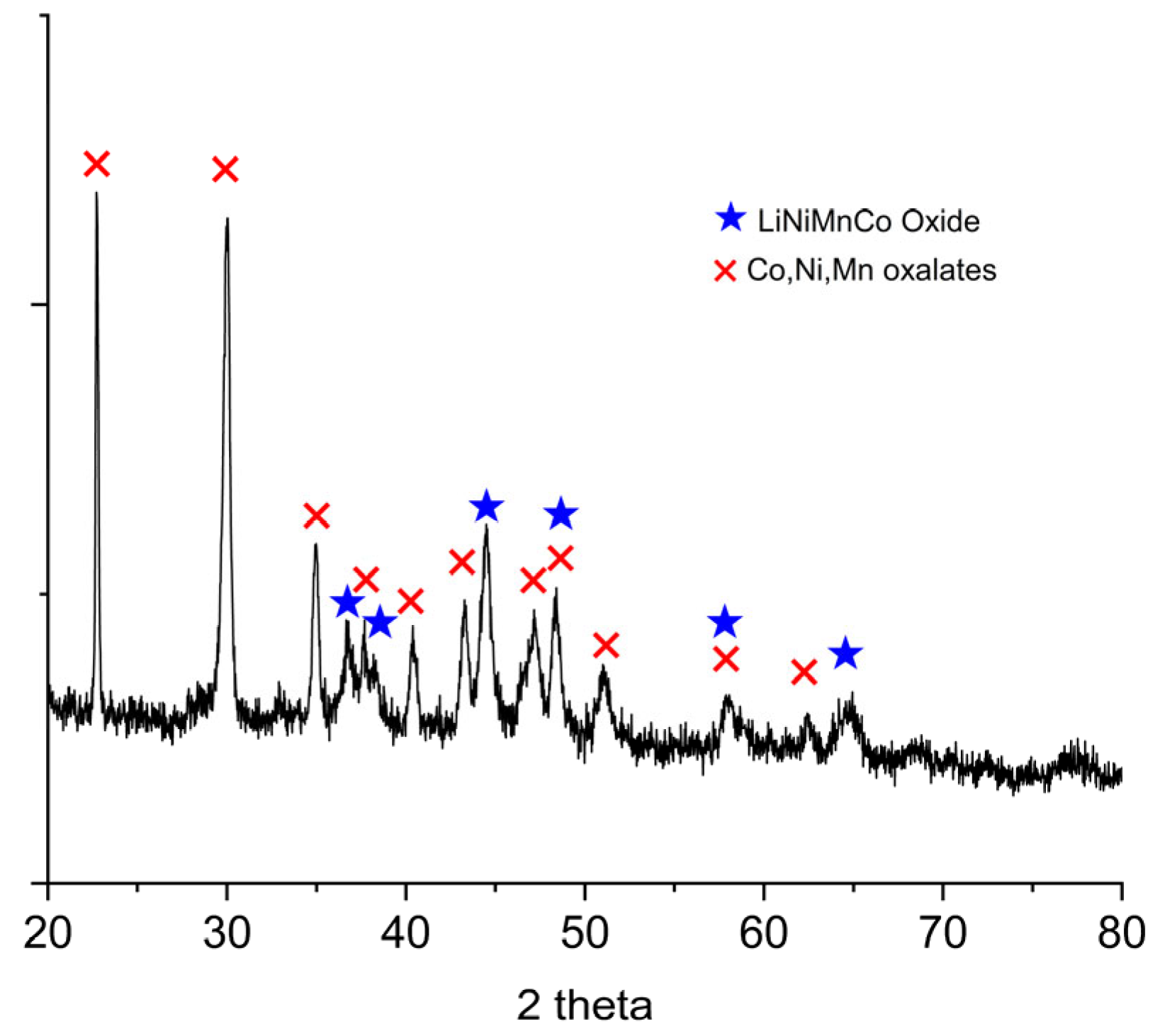
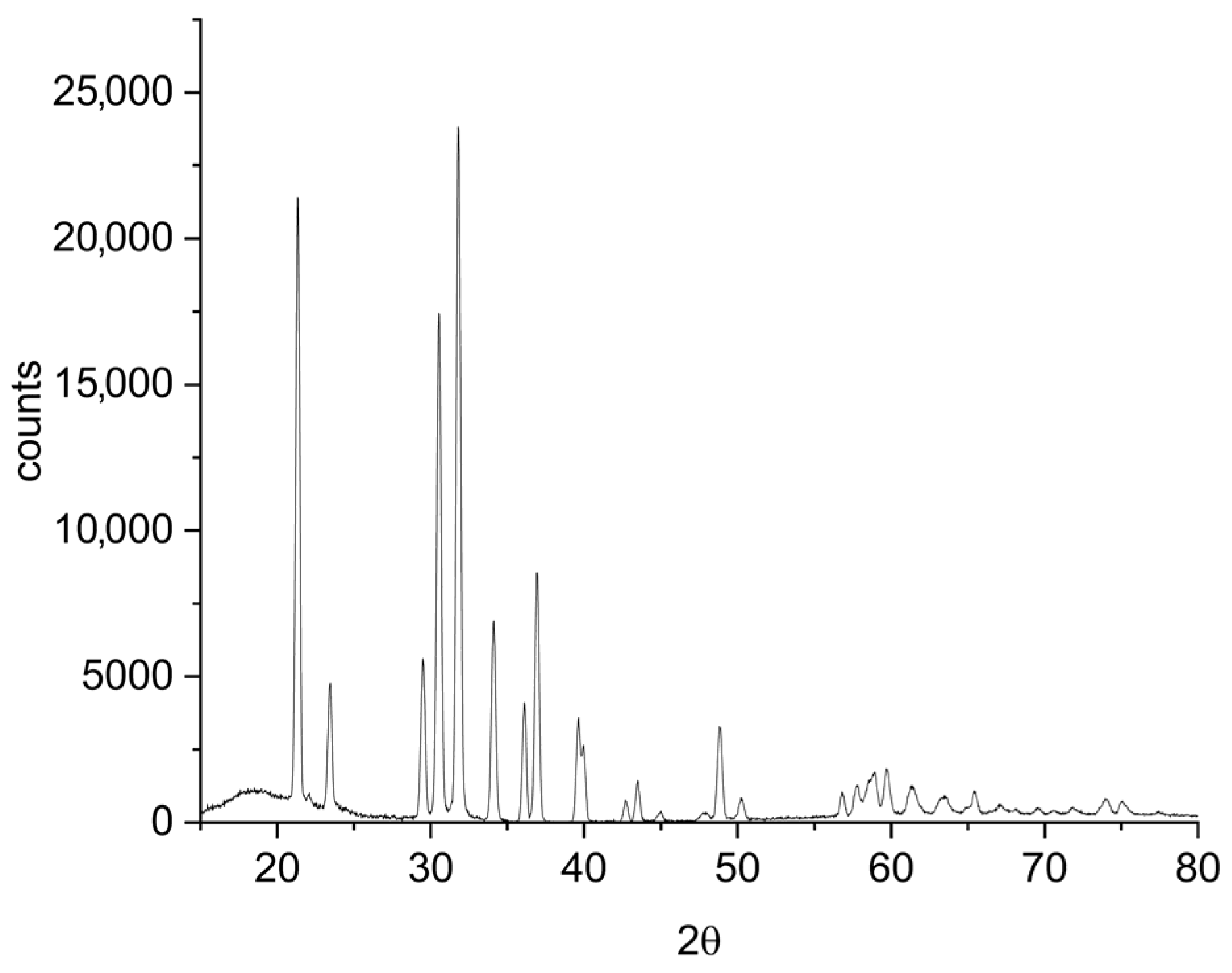
Figure 5 shows the XRD pattern of the filter cake. The spectrum is consistent with the presence of simple or mixed oxalates of Ni, Co, and Mn. In addition, the presence of residual mixed oxide phase of Li, Ni, Co, and Mn is confirmed.
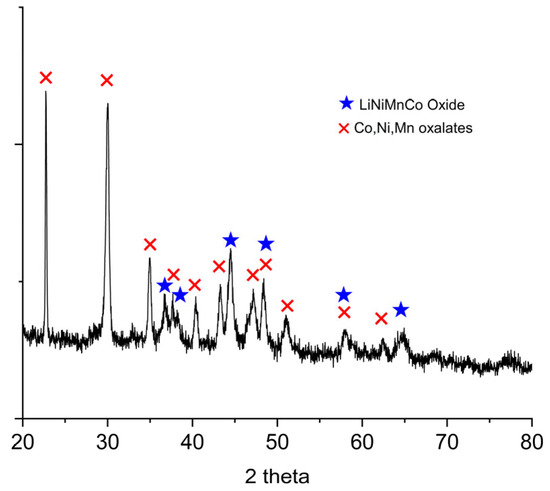
Figure 5.
X-ray diffraction (XRD) pattern of the filter cake obtained after oxalic acid leaching of the off-specification cathode.
The filtration cake obtained retains lithium at a concentration of 0.75% w/w (dry basis), corresponding to approximately 19% of the lithium initially present in the off-specification cathode material. Previous studies have shown that, during oxalic acid leaching of NMC cathodes, a superficial layer composed of mixed oxides and metal oxalates forms on the solid residue [23]. This layer acts as a passivating film that inhibits the diffusion of leaching species—namely hydrogen ions and oxalate—into the bulk of the NMC particles, thereby slowing the dissolution of the active material and ultimately limiting lithium extraction. The slower formation of oxalates at room temperature explains why higher lithium recoveries were achieved under these milder conditions, as the passivating layer develops less rapidly and allows longer access to the reactive interfaces. Therefore, the unrecovered lithium remains predominantly in the solid residue, either structurally embedded within the partially leached NMC matrix or trapped within the passivating mixed oxide/oxalate layer that inhibits complete dissolution.
Several studies have demonstrated the successful use of mixed oxalates of Ni, Co, and Mn in the preparation of NMC-type cathodes [49,50,51]. Building on this, it would be highly valuable to further improve the leaching process in order to reduce the lithium content remaining in the filtration cake. Preliminary studies performed by this research group have demonstrated that mechanical activation of the filtration residue via ball milling, followed by an additional leaching step, effectively reduces the lithium concentration in the final solid, confirming the liberation of unrecovered lithium through enhanced solid-state reactivity, but further investigation is needed to reach a balance between the technical efficiency and the economics of the process.
3.1.4. Reusability of the Leaching Agent
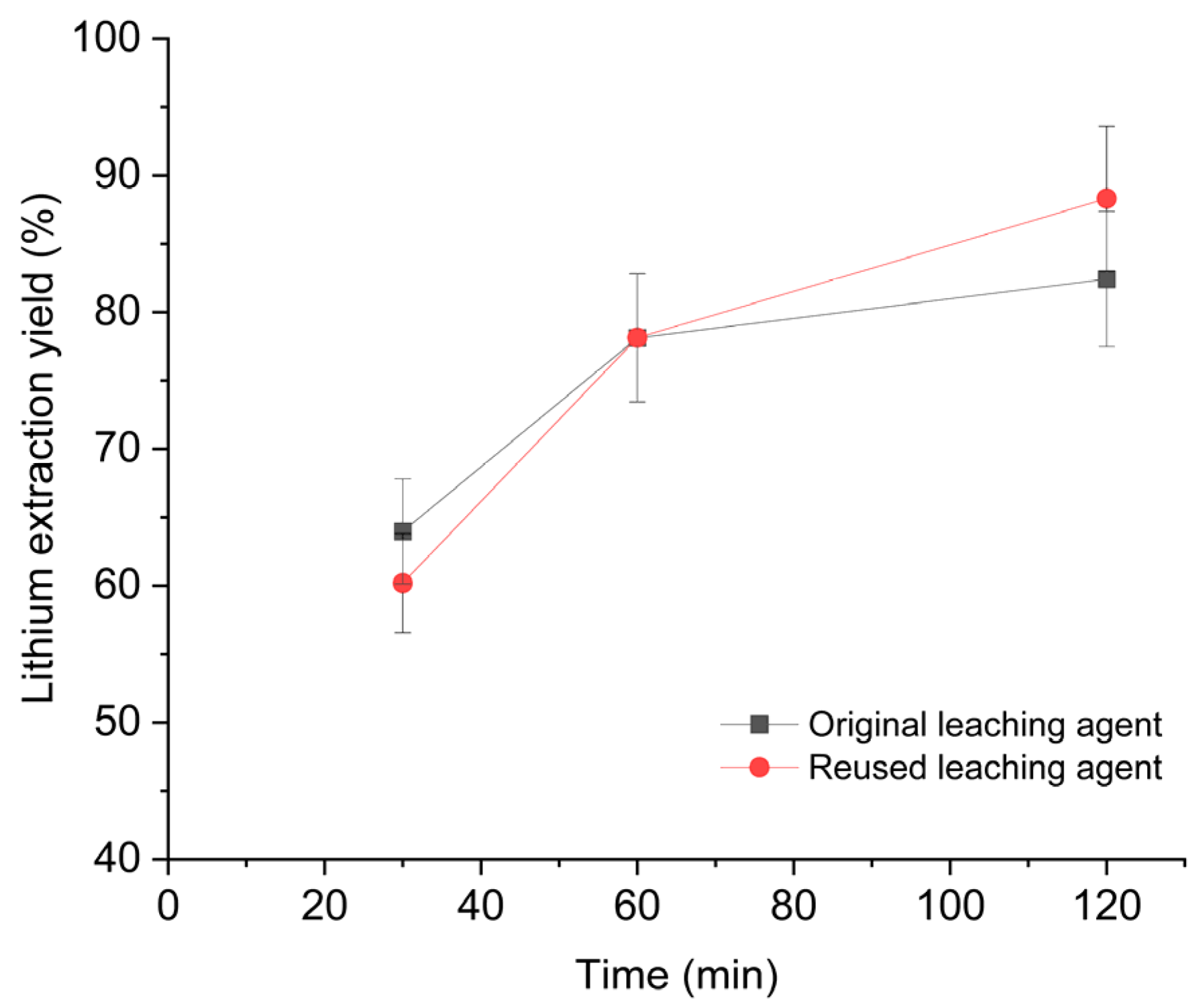
The electrolyte obtained from the membrane electrodialysis step (Section 3.2.2) contains regenerated oxalic acid, but at a lower concentration than the fresh leaching agent because some of the acid was consumed during the precipitation of nickel, cobalt, and manganese oxalates. Ion chromatographic analysis of the exhausted electrolyte revealed an oxalic acid concentration of 41.8 g L−1. To restore the initial H2O/OA·2H2O ratio of 7.3:1 w/w, additional oxalic acid dihydrate, equivalent to 55% wt. of the original oxalic acid load, was added to the electrolyte, thereby reestablishing the target ratio. Leaching experiments were then repeated using the regenerated leachate under the same operating conditions as those applied to the fresh solution (H2O/OA·2H2O = 7.3:1 w/w, S/L 1:20 w/w, 20 °C, 325 rpm, 2 h). The results, shown in Figure 6, demonstrate that both the lithium extraction yield and the kinetic profile are comparable to those obtained with the fresh leaching agent.
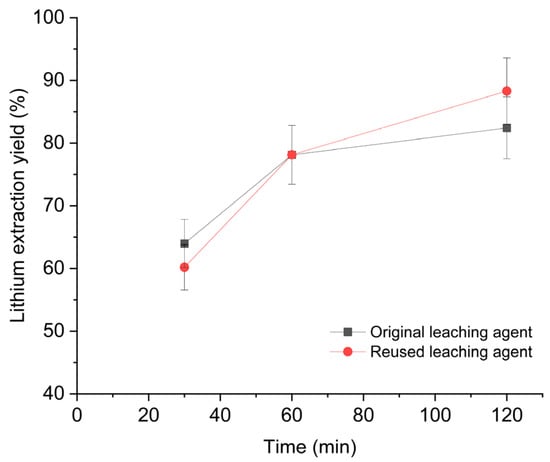
Figure 6.
Comparison of Li extraction performance with original and reused leaching agent. Operating conditions: 100 g of leaching solution (H2O/OA·2H2O = 7.3:1 w/w), S/L ratio = 1:20 w/w, room temperature, 2 h reaction time, 325 rpm stirring.
These findings indicate that the reutilisation of the electrolyte is not only technically viable but also provides a strategic opportunity to promote circular economy principles within the lithium value chain. By restoring the optimal H2O/OA·2H2O ratio through the addition of the exact amount of oxalic acid consumed during transition metal precipitation, lithium extraction efficiency is maintained while avoiding residual organic effluent.
3.2. Recovery of Lithium and Oxalic Acid by Membrane-Based Electrodialysis
3.2.1. Membrane Selection
Three cation-exchange membranes (CEM)—Fumasep FKB-PK-130, Nafion 117, and Neosepta CMX-fg—were tested to evaluate their performance in promoting lithium migration towards the catholyte. Experiments were conducted in a two-chamber H-cell using stainless steel (SS) as the cathode and platinum (Pt) as the anode. The anolyte consisted of 15 mL of a solution containing 2.76 g L−1 Li and 61.1 g L−1 oxalic acid, obtained from OSC leaching. The catholyte was 15 mL of a 0.5 M NaCl solution. The potential was controlled at the cathode (WE) at −1.1 V vs. Ag/AgCl, and residence times of 1 and 3 h were studied. Initial conductivities of the anolyte and catholyte were 36.7 and 39.0 mS, respectively.
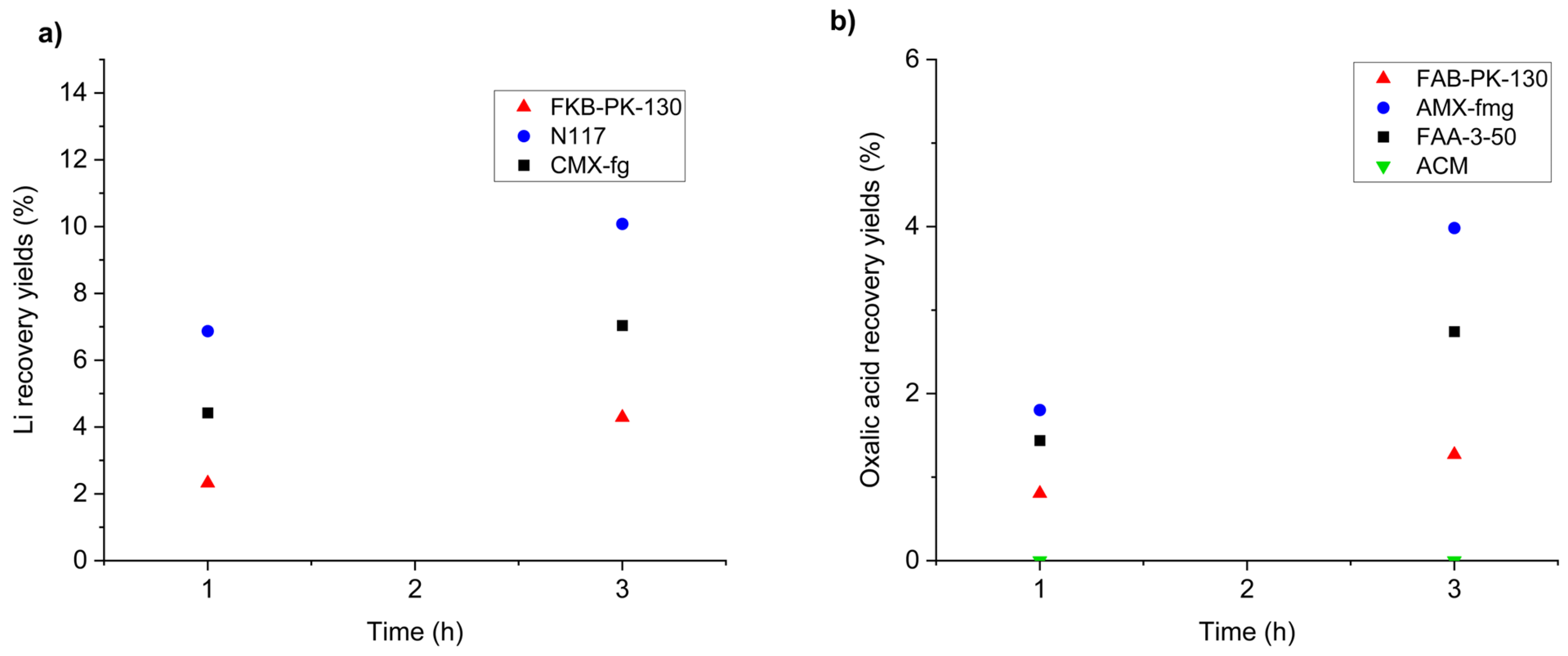
Figure 7a shows the recovery yields of lithium in the catholyte for the three membranes tested. Recovery was calculated based on the lithium concentration in the catholyte relative to its initial concentration in the working solution (anolyte).
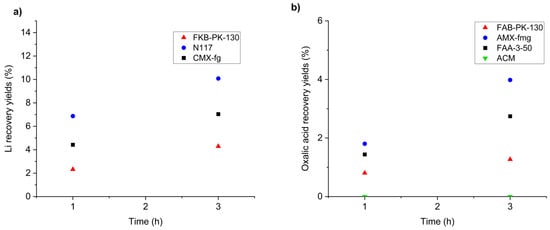
Figure 7.
Recovery yields of lithium and oxalic acid during electrodialysis as a function of membrane type. (a) Effect of cation-exchange membranes (FKB-PK-130, N117, CMX-fg) on lithium recovery. Conditions: two-chamber H-cell, stainless steel cathode, platinum anode, Ag/AgCl reference, −1.1 V vs. Ag/AgCl applied potential, residence times of 1 and 3 h, catholyte 0.5 M NaCl, anolyte with Li (2.76 g L−1) and oxalic acid (61.1 g L−1). (b) Effect of anion-exchange membranes (FAB-PK-130, AMX-fmg, FAA-3-50, ACM) on oxalic acid recovery. Same conditions as (a), with reversed electrolytes.
Similarly, four anion-exchange membranes (AEM)—Fumasep FAB-PK-130, Neosepta AMX-fmg, Fumasep FAA-3-50, and Neosepta ACM—were tested to evaluate oxalic acid migration towards the anolyte. Experiments were performed under the same conditions as for CEM selection, but in this case, the catholyte contained the oxalic acid solution obtained from OSC leaching, and the anolyte was the 0.5 M NaCl solution.
Figure 7b shows the recovery yields of oxalic acid, calculated based on its concentration in the anolyte relative to its initial content in the working solution (catholyte).
The selection of suitable ion-exchange membranes is critical for achieving efficient separation and recovery of lithium and oxalic acid. Among the cation-exchange membranes tested (FKB-PK-130, Nafion 117, and CMX-fg), Nafion 117 exhibited the highest lithium recovery yield, reaching 10.1% wt. after 3 h, compared to 4.3% wt. and 7.0% wt. for FKB-PK-130 and CMX-fg, respectively. This superior performance can be attributed to the high ionic conductivity and chemical stability of lithium forms of Nafion 117 [52], which facilitates Li+ migration under the applied potential while maintaining low resistance.
For anion-exchange membranes, AMX-fmg demonstrated the best performance for oxalic acid recovery, achieving 4.0% wt. after 3 h, whereas FAB-PK-130 and FAA-3-50 yielded 1.2% wt. and 2.7% wt., respectively, and ACM showed no recovery. Notably, Neosepta AMX membranes have previously been successfully employed in electrodialysis processes for carboxylic acids such as lactic acid, supporting their suitability for similar applications [53]. The superior performance of AMX-fmg can be attributed to its optimised fixed charge density and enhanced permeability for oxalate anions, which promote selective migration and minimise co-ion transport.
Although the recovery yields remain modest, the results open the possibility of studying the feasibility of membrane-based electrodialysis for the simultaneous recovery of lithium and oxalic acid from leachates. The use of Nafion 117 as the cation-exchange membrane and AMX-fmg as the anion-exchange membrane establishes the basis for designing an efficient flow cell configuration. This work focuses on defining the cell architecture.
3.2.2. Flow Cell Design
The objective is to define the configuration of an electrochemical flow cell that enables the simultaneous recovery of lithium in the catholyte and oxalic acid in the anolyte.
Three-Chamber Flow Cell
The experiment was conducted using a three-chamber flow cell under controlled conditions. Nafion 117 was employed as the CEM with a catholyte of 0.5 M NaHCO3, while Neosepta AMX-fmg served as the AEM with an anolyte of 0.5 M NaCl. The central compartment was filled with a working solution, obtained by mixing leachates from OSC processed under various conditions, which contained 2.78 g L−1 Li and 67.5 g L−1 oxalic acid. Initial conductivities of the catholyte, anolyte, and OSC leachate were 25.4, 38.0, and 44.5 mS, respectively. The WE was a stainless-steel cathode operated at an applied potential of −1.6 V versus Ag/AgCl, and the CE was a DSA-O2 anode. An Ag/AgCl reference electrode was used to maintain accurate potential control throughout the experiment. Each compartment was operated in batch mode with 100 mL of solution, supplied from external reservoirs and recirculated at 120 mL min−1 to ensure stable hydrodynamic conditions.
Table 6 summarises the results obtained from the analysis of the working solution at different residence times. Lithium and oxalic acid recovery yields increased progressively, reaching values close to 100%, indicating that nearly all Li and OA migrated from the feed compartment to the catholyte and anolyte, respectively.

Table 6.
Lithium and oxalic acid recovery yields in a three-chamber flow cell (CEM: N117; AEM: AMX-fmg; catholyte: 0.5 M NaHCO3; anolyte: 0.5 M NaCl; feed chamber: OSC leachate with 2.78 g L−1 Li and 67.5 g L−1 OA; applied potential: −1.6 V vs. Ag/AgCl; flow rate: 120 mL min−1; room temperature).
Analysis of the catholyte confirmed Li concentrations consistent with the calculated migration from the working solution; however, only trace amounts of OA were detected in the anolyte, which is likely due to oxalic acid oxidation at the anode. The oxidation of oxalic acid is well documented in the literature [54] and can be represented by the following reaction:
Given that the oxidation potential of oxalic acid is −0.49 V, and the applied cell potential ranged from −3.68 V to −4.64 V, oxidation is thermodynamically expected throughout the entire operating range, as the applied potentials greatly exceed the oxidation threshold. This makes the oxidative degradation of oxalic acid highly favourable whenever the solution is in direct contact with the anode. To address this limitation, a four-chamber flow cell design is proposed, incorporating a cation-exchange membrane to isolate the oxalic acid solution from the anolyte, thereby preventing anodic oxidation and ensuring effective recovery.
Four-Chamber Flow Cell
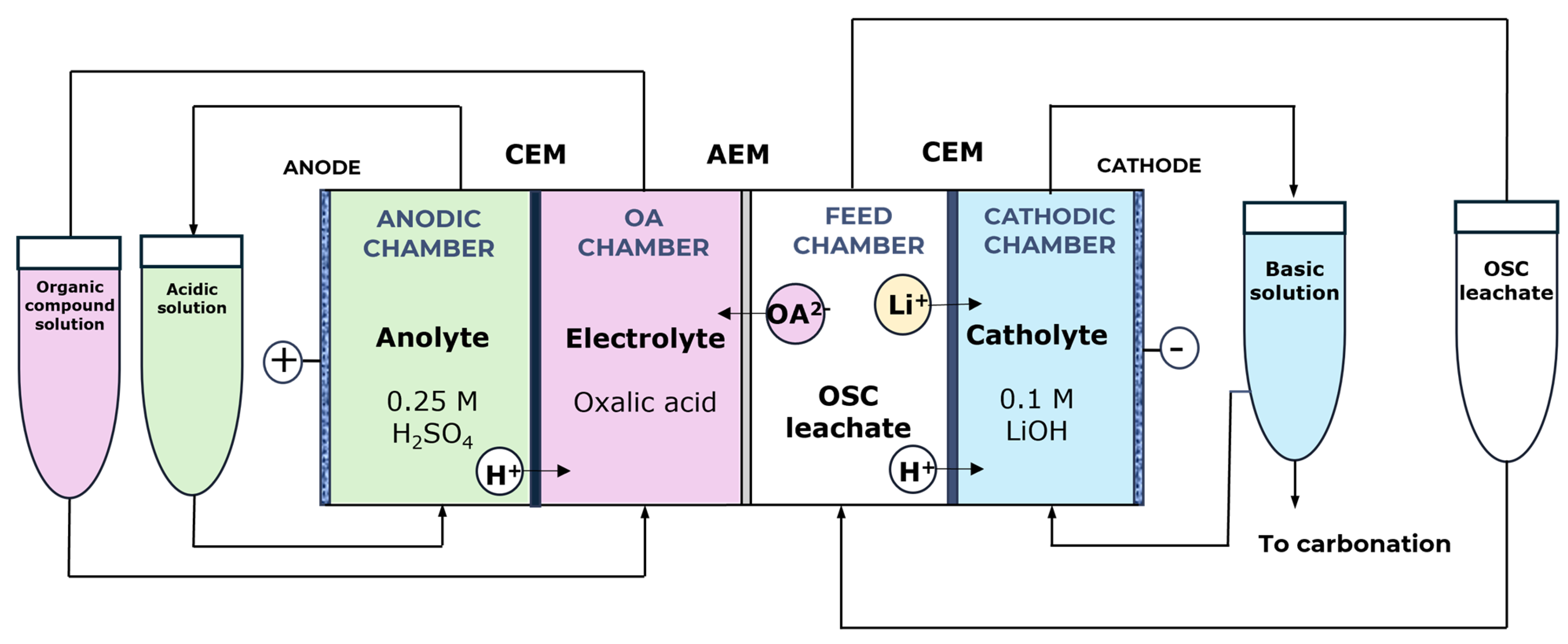
To overcome the limitations observed in the three-chamber configuration, a four-chamber flow cell was developed to enable the simultaneous and selective recovery of lithium and oxalic acid. This type of configuration has been previously reported in the literature as an effective alternative for the concentration and regeneration of oxalic acid from aqueous solutions [55]. In the proposed design, oxalic acid is recovered in a dedicated compartment that is physically isolated from the anolyte by a cation-exchange membrane, thereby preventing its exposure to oxidising conditions and minimising oxidative degradation (Figure 8).
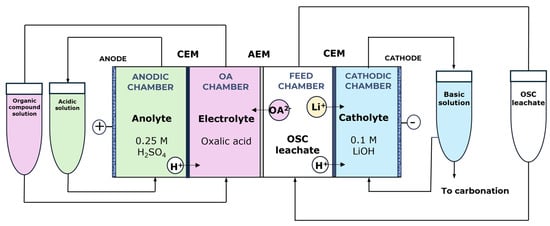
Figure 8.
Proposed four-chamber flow cell for the recovery of Li and oxalic acid from the leachate generated during OSC leaching. From left to right: anolyte chamber (0.25 M H2SO4); cation-exchange membrane (CEM, N117); oxalic acid (OA) chamber; anion-exchange membrane (AEM, AMX-fmg); feed chamber containing the OSC leachate; cation-exchange membrane (CEM, N117); and catholyte chamber (0.1 M LiOH).
Additional modifications were implemented to enhance lithium recovery. The catholyte was replaced with a 0.1 M LiOH solution, which increases lithium concentration and facilitates subsequent precipitation as Li2CO3. Similarly, the anolyte was substituted with a 0.25 M H2SO4 solution to provide the required acidity for optimal electrochemical performance. This configuration ensures efficient separation of species and minimises undesired side reactions, establishing a robust basis for selective recovery processes.
The experimental work was conducted using a four-chamber flow cell configuration with an active membrane area of 10 cm2. In this design, Nafion 117 served as the CEM separating the anodic compartment from the oxalic acid (OA) chamber, which contained a 0.1 M oxalic acid solution, while the anolyte consisted of 0.25 M H2SO4. A Neosepta AMX-fmg (AEM) was positioned between the OA chamber and the feed compartment, which was filled with the OSC leachate containing 2.75 g L−1 of Li and 40.4 g L−1 of oxalic acid. A second Nafion 117 membrane was placed between the feed chamber and the cathodic compartment, the latter containing a 0.1 M LiOH solution. The initial electrical conductivities of the anolyte, oxalic acid, OSC leachate, and catholyte solutions were 88.6, 19.3, 21.8, and 17.3 mS, respectively, while the corresponding initial pH values were 0.5, 1.1, 1.7, and 12.8.
A stainless-steel electrode operated as the WE at an applied potential of −1.6 V versus Ag/AgCl, while a DSA-O2 electrode functioned as the CE. An Ag/AgCl reference electrode ensured accurate potential control throughout the test. All compartments were supplied in batch mode (100 mL each) from external reservoirs, circulated at a flow rate of 120 mL min−1 to maintain stable hydrodynamic conditions.
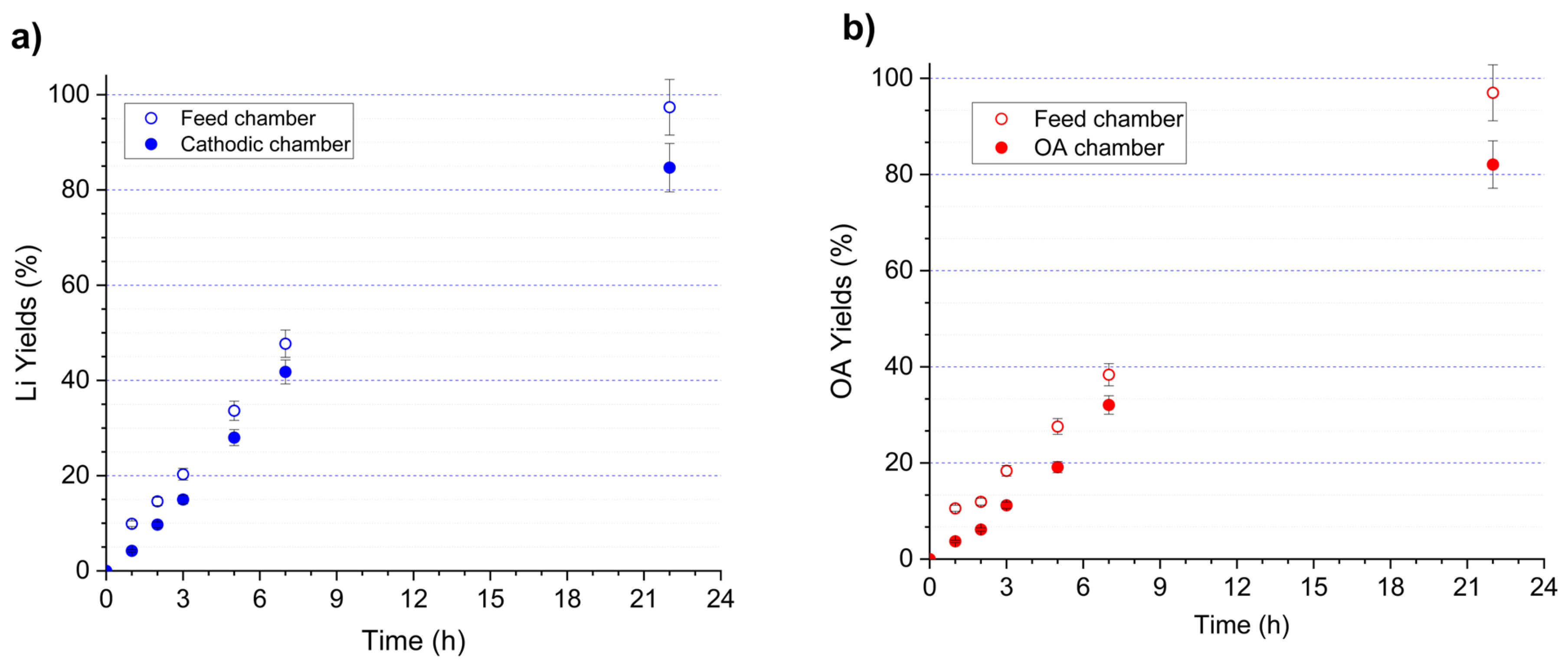
Figure 9 displays the recovery profiles of lithium (a) and oxalic acid (b) during batch operation of the four-chamber electrochemical flow cell over a period of 1 to 22 h. Recovery was evaluated using two complementary criteria: (i) depletion of each species in the feed chamber and (ii) accumulation in their respective product chambers (cathodic chamber for Li+ and OA chamber for oxalate). Both Li and OA exhibited a continuous increase in recovery, reaching 97.4% wt. and 97.0% wt., respectively, after 22 h based on feed-chamber depletion. Enrichment in the product chambers followed the same trend, although reaching slightly lower values (84.7% wt. for Li and 82.1% wt. for OA).
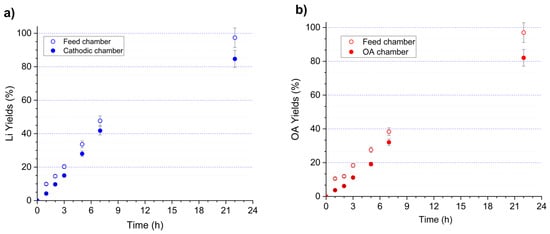
Figure 9.
Recovery yields of (a) lithium and (b) oxalic acid during electrodialysis in a four-chamber flow electrochemical cell. The EC configuration (left to right) consisted of the following: anodic chamber containing 0.25 M H2SO4; cation-exchange membrane (CEM, N117); oxalic acid chamber containing 0.1 M OA; anion-exchange membrane (AEM, AMX-fmg); feed chamber containing the OSC leachate (2.75 g L−1 Li and 40.4 g L−1 oxalic acid); cation-exchange membrane (CEM, N117); and cathodic chamber containing 0.1 M LiOH. Operating conditions: applied potential of −1.6 V vs. Ag/AgCl, flow rate of 120 mL min−1 and room temperature.
Table 7 presents the mass balance for lithium and oxalate ion obtained after 22 h. The mass balance performed at t = 22 h confirms the overall consistency of the electrochemical experiment conducted in the four-chamber cell. For lithium, a total of 339 mg was introduced into the system, of which 306 mg were recovered in the outlet streams, corresponding to a closure of approximately 90%. Most of the lithium was recovered in the cathodic compartment (297 mg), consistent with the selective migration of Li+ across the CEM. For oxalic acid, the total input was 5064 mg, and 4546 mg were recovered in the outlet streams, yielding a closure also close to 90%, comparable to that of lithium. The regenerated OA chamber accounted for the largest fraction of recovered oxalate (4322 mg), reflecting efficient anion transport through the AEM and minimal distribution across the remaining compartments. Minor discrepancies between input and output masses for both Li and OA can be attributed to experimental uncertainties such as sampling losses, temporary retention within membrane domains, or transient accumulation in boundary layers. Overall, the mass balances for Li and oxalic acid show good agreement between inputs and outputs and align with the recovery trends observed, reinforcing the reliability of the experimental results and the effectiveness of the four-chamber electrochemical configuration.

Table 7.
Mass balance of lithium and oxalate at t = 22 h in the electrochemical experiment conducted using the four-chamber electrochemical cell.
In the oxalic acid chamber, acid regeneration was achieved, resulting in a final oxalic acid concentration of 41.8 g L−1. This regenerated solution can be reused for subsequent leaching operations, as described in Section 3.1.4, after adjusting its concentration to the required level for OSC leaching. In the case of lithium, the use of a Li-containing catholyte enabled an increase in concentration from 2.75 g L−1 in the initial OSC leachate to 2.91 g L−1 in the catholyte. This solution is suitable for downstream lithium carbonate production via the precipitation process, as described in Section 3.3.
3.3. Lithium Carbonate Valorisation
Lithium precipitation as lithium carbonate is widely reported in the literature [56,57,58,59], both by using CO2 and by employing Na2CO3. The solution obtained by electrodialysis has a pH of 12.6 and is essentially free of impurities. Consequently, the lithium carbonate process was simplified to a single evaporation step to concentrate the lithium, followed by a precipitation step via the addition of Na2CO3.
A measure of 70 mL of catholyte produced by electrodialysis—containing 2.80 g L−1 Li—was evaporated to a final volume of 10 mL. Subsequently, 5 mL of a 400 g L−1 Na2CO3 solution—corresponding to a 25% excess over the stoichiometric amount required to precipitate all lithium present—was added. The mixture was held at 90 °C with agitation at 100 rpm for 2 h, with the volume maintained at 10 mL by the addition of hot water. The lithium carbonate formed was filtered and washed with 40 mL of boiling water, yielding 0.57 g of dry Li2CO3. ICP-OES analysis of the solid product confirmed a lithium carbonate purity of 99.3%. The recovery yield was 54.7% wt., with losses primarily attributable to the partial solubility of Li2CO3 and its redissolution during the washing step. These quantitative results are summarised in Table 8, and the product purity is further confirmed by the XRD pattern shown in Figure 10, which closely matches the characteristic diffraction spectrum of lithium carbonate.

Table 8.
Lithium carbonate characterisation by ICP-OES.
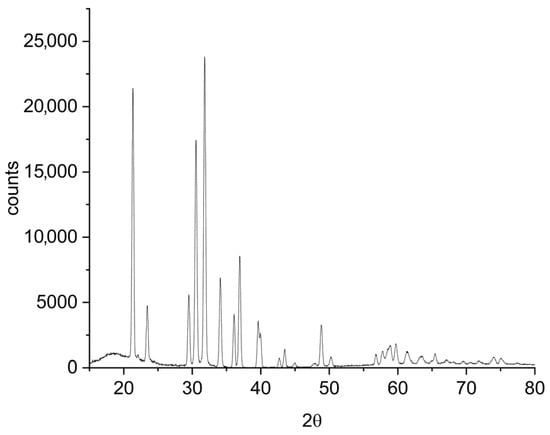
Figure 10.
XRD pattern of the obtained lithium carbonate.
The proposed process generates only Na and Li containing effluents, which can be used to prepare the catholyte required for electrodialysis, as these aqueous streams still contain enough dissolved lithium suitable for this purpose. This strategy not only reduces global water consumption but also improves the overall lithium recovery efficiency by reincorporating lithium-bearing streams back into the process. The continuous reuse of this water will increase sodium levels over time, requiring periodic purges. These purges must be adequately treated to remove impurities and limit liquid discharge.
4. Conclusions
This study demonstrates the viability of a fully integrated and low-impact hydrometallurgical process for the valorisation of lithium from OSC residues. The central contribution lies in the implementation—for the first time—of a closed-loop oxalic acid → electrodialysis → Li2CO3 route, in which lithium is selectively leached, the lixiviant is electrochemically regenerated and reused without loss of efficiency, and the recovered lithium is ultimately converted into a high-purity product. This unified approach contrasts with previous studies, where leaching, acid recovery, and lithium carbonate precipitation have been treated as separate, non-integrated stages, without demonstrating the feasibility of a fully closed cycle.
The careful optimisation of the oxalic acid leaching stage was crucial in attaining a highly selective recovery of lithium from the OSC. By setting the water to oxalic acid (H2O/OA·2H2O) ratio to 7.3:1 w/w, operating at room temperature for 2 h and employing a 125% stoichiometric excess of oxalic acid (S/L = 1:20 w/w), lithium recoveries of 81% wt. were obtained while co-extraction of nickel, cobalt and manganese remained at only 0.2% wt., 1.6% wt. and 1.7% wt., respectively. These conditions prevent unwanted oxalic acid precipitation, moderate CO2 evolution, and enable efficient operation at room temperature. This is particularly noteworthy because oxalic acid leaching of EoL battery black mass is generally inefficient at 20 °C, often requiring temperatures above 60 °C to achieve acceptable yields. The superior performance observed for OSC suggests higher intrinsic chemical reactivity, likely linked to its non-stoichiometric composition and structural defects arising from manufacturing, making it especially suitable for selective organic-acid-based valorisation routes.
A key advancement of this work is the integration of a four-chamber electrodialysis flow cell, equipped with two Nafion 117 cation-exchange membranes and one Neosepta AMX-fmg anion-exchange membrane. This configuration enabled near-complete migration of lithium and oxalic acid (97% wt. after 22 h), with efficient transport from the leachate to the respective product streams while preventing the oxidative degradation of oxalic acid. The reusability of the lixiviant was confirmed by repeating the leaching step with the regenerated solution supplemented with 55% wt. fresh oxalic acid, achieving lithium recoveries comparable to those obtained using a fresh leaching reagent.
Although the precipitation stage was not explored in depth, it was demonstrated that lithium carbonate can be obtained with a purity of 99.3%, as confirmed by ICP-OES and XRD. The recovery yield (54.7%) was limited by the solubility of Li2CO3 and its partial redissolution during washing; nonetheless, the resulting product approaches battery-grade purity requirements. Importantly, the precipitation step generates aqueous effluents containing only Li and Na, which can be reused to prepare the catholyte required for electrodialysis, thereby reducing water consumption and improving overall recovery efficiency. An industrial process operating in a closed loop, integrating electrodialysis and carbonation with the recirculation of the unprecipitated lithium solution as catholyte, could achieve higher overall yields. Preliminary calculations estimating a 5% purge rate predict an integrated electrodialysis–carbonation efficiency of 80.5% wt., suggesting that optimised operational conditions could lead to even higher performance.
In summary, the combination of selective oxalic acid leaching, lithium and oxalic acid recovery by electrodialysis, and lithium carbonate precipitation constitutes a low-impact and waste-free pathway for the valorisation of OSC. The process aligns with circular economy objectives for the battery sector. Future work should focus on (i) optimising the energy and time requirements of the electrochemical cell, (ii) refining the lithium carbonate precipitation step to maximise lithium recovery, and (iii) conducting techno-economic and environmental assessments to validate the process at an industrial scale.
Author Contributions
Conceptualization, L.Y.; methodology, L.Y., J.L.A. and J.A.; validation, J.L.A., J.A., J.N. and C.d.R.; investigation, J.L.A., J.A., J.N. and C.d.R.; writing—original draft preparation, J.L.A.; writing—review and editing, J.L.A. and L.Y.; supervision, L.Y. and J.A.; project administration, L.Y.; funding acquisition, L.Y. All authors have read and agreed to the published version of the manuscript.
Funding
The authors acknowledge the financial support provided by the LICORNE project, funded by the European Union’s Horizon Europe research and innovation programme under Grant Agreement No. 101069644.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
Authors Jose Luis Aldana, Lourdes Yurramendi, Javier Antoñanzas, Javier Nieto, and Carmen del Río were employed by the company TECNALIA, Basque Research & Technology Alliance (BRTA). The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- Or, T.; Gourley, S.W.D.; Kaliyappan, K.; Yu, A.; Chen, Z. Recycling of mixed cathode lithium-ion batteries for electric vehicles: Current status and future outlook. Carbon Energy 2020, 2, 6–43. [Google Scholar] [CrossRef]
- Dos Santos, M.P.; Garde, I.A.A.; Ronchini, C.M.B.; Filho, L.C.; de Souza, G.B.M.; Abbade, M.L.F.; Regone, N.N.; Jegatheesan, V.; de Oliveira, J.A. A technology for recycling lithium-ion batteries promoting the circular economy: The RecycLib. Resour. Conserv. Recycl. 2021, 175, 105863. [Google Scholar] [CrossRef]
- Li, C.; Dai, G.; Liu, R.; Wang, C.; Wang, S.; Ju, Y.; Jiang, H.; Jiao, S.; Duan, C. Separation and recovery of nickel cobalt manganese lithium from waste ternary lithium-ion batteries. Sep. Purif. Technol. 2023, 306, 122559. [Google Scholar] [CrossRef]
- Tran, T.T.; Moon, H.S.; Lee, M.S. Recovery of valuable metals from hydrochloric leaching of smelted alloys from spent lithium-ion batteries. J. Chem. Technol. Biotechnol. 2022, 97, 1247–1258. [Google Scholar] [CrossRef]
- Yuliusman; Fajaryanto, R.; Nurqomariah, A.; Silvia. Acid leaching and kinetics of cobalt recovery from spent lithium-ion batteries with nitric acid. E3S Web Conf. 2018, 67, 03025. [Google Scholar] [CrossRef]
- Fahmy, R.N.M.; Walvekar, R.; Yi, F.C.; Ler, L.Y.; Khalid, M. Sustainable battery recycling: Investigating ammonium-based DES solvents for NMC cathode material recovery. J. Environ. Chem. Eng. 2025, 13, 118599. [Google Scholar] [CrossRef]
- Hubert, P.; Vanderbruggen, A.; Jradi, S.; Chagnes, A. Selective metal recovery: Innovating leaching of LFP–NMC cathode mixtures from spent lithium-ion batteries. Sustain. Mater. Technol. 2025, 46, e01658. [Google Scholar] [CrossRef]
- Guimarães, L.F.; Botelho Junior, A.B.; Espinosa, D.C.R. Sulfuric acid leaching of metals from waste Li-ion batteries without using reducing agents. Miner. Eng. 2022, 183, 107597. [Google Scholar] [CrossRef]
- Xuan, W.; Otsuki, A.; Chagnes, A. Leaching mechanism of NMC811 by hydrochloric acid for recycling lithium-ion battery cathodes. RSC Adv. 2019, 9, 38612–38618. [Google Scholar] [CrossRef]
- Takacova, Z.; Havlik, T.; Kukurugya, F.; Orac, D. Cobalt and lithium recovery from active mass of spent Li-ion batteries: Theoretical and experimental approach. Hydrometallurgy 2016, 163, 9–17. [Google Scholar] [CrossRef]
- Güloğlu, E.; Orhan, G. Sustainable recycling of spent mixed LIB cathodes via organic acid-based DES: NMC532 regeneration and electrochemical performance. Sep. Purif. Technol. 2026, 382, 135817. [Google Scholar] [CrossRef]
- Singh, J.K.J.; Rana, M.; Khan, M.I.H.; Jo, Y.T.; Park, J.H. Choline chloride–lactic acid–ascorbic acid (ChCl–LA–AA) based green deep eutectic solvent for leaching LiMn2O4 cathode material of spent Li-ion batteries. Hydrometallurgy 2025, 236, 106529. [Google Scholar] [CrossRef]
- Nazlı, F.; Hasdemir, I.; Uysal, E.; Dursun, H.N.; Gezici, U.O.; Özçelik, D.Y.; Burat, F.; Gürmen, S. Eco-friendly leaching of spent lithium-ion battery black mass using a ternary deep eutectic solvent system based on choline chloride, glycolic acid and ascorbic acid. Minerals 2025, 15, 782. [Google Scholar] [CrossRef]
- Güloğlu, E.; Orhan, G. Recovery of NMC cathode powder using a novel NADES as β-alanine: Citric acid and electrochemical characterization. J. Environ. Chem. Eng. 2025, 13, 117448. [Google Scholar] [CrossRef]
- Punt, T.; van Wyk, A.P.; Bradshaw, S.M.; Akdogan, G. Citric acid leaching performance at high solid-to-liquid ratios for lithium-ion battery recycling. Min. Metall. Explor. 2024, 41, 3463–3474. [Google Scholar] [CrossRef]
- Bruno, M.; Francia, C.; Fiore, S. Selective leaching for the recycling of lithium, iron and phosphorous from lithium-ion battery cathodes’ production scraps. Batteries 2024, 10, 415. [Google Scholar] [CrossRef]
- Li, S.; Zhang, W.; Xia, Y.; Li, Q. Enhanced reducing capacity of citric acid for lithium-ion battery recycling under microwave-assisted leaching. Waste Manag. 2024, 189, 23–33. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, Y.; Han, Z.; Jin, M.; Li, X.; Li, Y.; Kong, X.; Zhang, Z.; Zhang, X. Kinetic optimization in green metal recovery: Citric acid vs glycine for sustainable Li-ion battery recycling. Ecotoxicol. Environ. Saf. 2025, 302, 118746. [Google Scholar] [CrossRef]
- Rouquette, L.M.J.; Petranikova, M.; Vieceli, N. Selective Li recovery with oxalic acid. Sep. Purif. Technol. 2023, 320, 124143. [Google Scholar] [CrossRef]
- Yu, H.; Wang, D.; Rao, S.; Duan, L.; Shao, C.; Tu, X.; Ma, Z.; Cao, H.; Liu, Z. Selective leaching of lithium from spent lithium-ion batteries using sulfuric acid and oxalic acid. Int. J. Miner. Metall. Mater. 2024, 31, 688–696. [Google Scholar] [CrossRef]
- Yang, C.; Wang, J.; Yang, P.; He, Y.; Wang, S.; Zhao, P.; Wang, H. Recovery of valuable metals from spent LiNi0.8Co0.1Mn0.1O2 cathode materials using compound leaching agents of sulfuric acid and oxalic acid. Sustainability 2022, 14, 4169. [Google Scholar] [CrossRef]
- Saleem, U.; Buvik, V.; Knuutila, H.K.; Bandyopadhyay, S. Recovery of lithium from oxalic acid leachate produced from black mass of spent electric vehicle Li-ion batteries. Chem. Eng. J. Adv. 2024, 20, 100648. [Google Scholar] [CrossRef]
- Rouquette, L.M.J.; Altenschmidt, L.; Culina, M.; Brant, W.R.; Ebin, B.; Petranikova, M. Kinetics study of the dissolution of black mass material using oxalic acid as a leaching agent. J. Hazard. Mater. Adv. 2025, 18, 100750. [Google Scholar] [CrossRef]
- Chen, X.; Kang, D.; Cao, L.; Li, J.; Zhou, T.; Ma, H. Separation and recovery of valuable metals from spent lithium-ion batteries: Simultaneous recovery of Li and Co in a single step. Sep. Purif. Technol. 2019, 210, 690–697. [Google Scholar] [CrossRef]
- Ma, C.; Mohamoud, M.; Punt, T.; Li, J.; Svärd, M.; Forsberg, K. Crystallization of cathode active material precursors from tartaric acid solution. ChemSusChem 2025, 18, 1523. [Google Scholar] [CrossRef]
- Wang, J.Z.; Tang, Y.C.; Shen, Y.H. Recovery of cobalt metal from waste lithium-ion batteries as sensor materials. Sens. Mater. 2022, 34, 2037–2043. [Google Scholar] [CrossRef]
- Sahu, S.; Devi, N. Effective leaching of spent lithium-ion batteries using DL-lactic acid as lixiviant and selective separation of metals through precipitation and solvent extraction. Environ. Sci. Pollut. Res. 2023, 30, 90152–90167. [Google Scholar] [CrossRef]
- Amarasekara, A.S.; Wang, D.; Shrestha, A.B. Efficient leaching of metal ions from spent Li-ion battery combined electrode coatings using hydroxy acid mixtures and regeneration of lithium nickel manganese cobalt oxide. Batteries 2024, 10, 170. [Google Scholar] [CrossRef]
- Charzewska, K.; Adamek, J.; Kateusz, F.; Piątek, J.; Zając, W. Levulinic acid as a sustainable and selective leaching agent for hydrometallurgical recycling of commercial Li-ion batteries: Towards greener critical metal recovery. Sep. Purif. Technol. 2026, 380, 135196. [Google Scholar] [CrossRef]
- Jiang, T.; Shi, Q.; Wei, Z.; Shah, K.; Efstathiadis, H.; Meng, X.; Liang, Y. Leaching of valuable metals from cathode active materials in spent lithium-ion batteries by levulinic acid and biological approaches. Heliyon 2023, 9, e15788. [Google Scholar] [CrossRef]
- Fu, Y.; He, Y.; Li, J.; Qu, L.; Yang, Y.; Guo, X.; Xie, W. Improved hydrometallurgical extraction of valuable metals from spent lithium-ion batteries via a closed-loop process. J. Alloys Compd. 2020, 847, 156489. [Google Scholar] [CrossRef]
- Gerold, E.; Schinnerl, C.; Antrekowitsch, H. Critical evaluation of the potential of organic acids for the environmentally friendly recycling of spent lithium-ion batteries. Recycling 2022, 7, 4. [Google Scholar] [CrossRef]
- Badre-Eddine, S.; Muhr, L.; Chagnes, A. Innovative integration of citric acid leaching and electrodialysis for selective lithium recovery from NMC cathode material. Metals 2025, 15, 598. [Google Scholar] [CrossRef]
- Cha, Y.Y.; Lee, C.; Kim, H.I.; Lee, G.G.; Nam, S.E.; Lee, J.S. Electrodialysis for rare metal recovery from battery waste: A brief review. Korean J. Chem. Eng. 2025, 42, 1599–1611. [Google Scholar] [CrossRef]
- Cournoyer, A.; Bazinet, L. Electrodialysis processes an answer to industrial sustainability: Toward the concept of eco-circular economy? Membranes 2023, 13, 205. [Google Scholar] [CrossRef]
- Tönjes, S.; Uitterhaegen, E.; de Winter, K.; Soetaert, W. Reactive extraction technologies for organic acids in industrial fermentation processes: A review. Sep. Purif. Technol. 2025, 356, 129881. [Google Scholar] [CrossRef]
- Almohasin, J.A.; Balag, J.; Miral, V.G.; Moreno, R.V.; Tongco, L.J.; Lopez, E.C.R. Green solvents for liquid–liquid extraction: Recent advances and future trends. Eng. Proc. 2023, 56, 16278. [Google Scholar] [CrossRef]
- Srishti, S.; Anilkumar, A.; Roy, Y. A review of advancements in solvent recovery from hazardous waste. J. Indian Inst. Sci. 2024, 104, 811–825. [Google Scholar] [CrossRef]
- Lee, J.K.; Lee, J.S.; Ahn, Y.S.; Kang, G.H. Restoring the reactivity of organic acid solution used for silver recovery from solar cells by fractional distillation. Sustainability 2019, 11, 3659. [Google Scholar] [CrossRef]
- Sun, Y.; Zhang, X.; Zheng, Y.; Yan, L.; Xiu, Z. Sugaring-out extraction combining crystallization for recovery of succinic acid. Sep. Purif. Technol. 2019, 209, 972–983. [Google Scholar] [CrossRef]
- Nasser, O.A.; Petranikova, M. Review of achieved purities after Li-ion batteries hydrometallurgical treatment and impurities effects on the cathode performance. Batteries 2021, 7, 60. [Google Scholar] [CrossRef]
- Liu, C.; Lin, J.; Cao, H.; Zhang, Y.; Sun, Z. Recycling of spent lithium-ion batteries in view of lithium recovery: A critical review. J. Clean. Prod. 2019, 228, 801–813. [Google Scholar] [CrossRef]
- MSE Supplies. Single Crystal NMC 532 Cathode Powder 500 g. Available online: https://www.msesupplies.com/products/single-crystal-nmc532-cathode-powder-lithium-nickel-manganese-cobalt-oxide-lini0-5mn0-3co0-2o2 (accessed on 12 December 2025).
- Bi, Y.; Yang, W.; Du, R.; Zhou, J.; Liu, M.; Liu, Y.; Wang, D. Correlation of oxygen non-stoichiometry to the instabilities and electrochemical performance of LiNi0.8Co0.1Mn0.1O2 utilized in lithium-ion battery. J. Power Sources 2015, 283, 211–218. [Google Scholar] [CrossRef]
- Tanriverdi Terzioğlu, S.; Ilhan, S. Investigation of leaching kinetics of spent lithium-ion battery cathode material in oxalic acid solution and production of NMC 111 cathode powder. J. Sustain. Metall. 2025, 11, 4049–4067. [Google Scholar] [CrossRef]
- Peñuela, J.; Martínez, J.D.; Araujo, M.L.; Brito, F.; Lubes, G.; Rodríguez, M.; Lubes, V. Speciation of the nickel(II) complexes with oxalic and malonic acids in 1.0 mol dm–3 NaCl at 25 °C. J. Coord. Chem. 2011, 64, 2698–2705. [Google Scholar] [CrossRef]
- Schmitz, D.; Prasetyo, H.; Birich, A.; Yeetsorn, R.; Friedrich, B. Co-precipitation of metal oxalates from organic leach solution derived from spent lithium-ion batteries. Metals 2024, 14, 80. [Google Scholar] [CrossRef]
- Apelblat, A.; Manzurola, E. Solubility of oxalic, malonic, succinic, adipic, maleic, malic, citric, and tartaric acids in water from 278.15 to 338.15 K. J. Chem. Thermodyn. 1987, 19, 317–320. [Google Scholar] [CrossRef]
- Zeng, T.; Zhang, C. An effective way of co-precipitating Ni2+, Mn2+ and Co2+ using ammonium oxalate as precipitant for Ni-rich Li-ion battery cathodes. J. Mater. Sci. 2020, 55, 11535–11544. [Google Scholar] [CrossRef]
- Wang, D.; Belharouak, I.; Zhou, G.; Amine, K. Synthesis of lithium- and manganese-rich cathode materials via an oxalate co-precipitation method. J. Electrochem. Soc. 2013, 160, A3108–A3112. [Google Scholar] [CrossRef]
- Nanda, R.D.; Kristianto, S.A.; Kartini, E.; Fakhrudin, M. The effect of stirring time in the synthesis of NMC-721 using oxalate co-precipitation. AIP Conf. Proc. 2023, 2932, 020008. [Google Scholar] [CrossRef]
- Stenina, I.A.; Sistat, P.; Rebrov, A.I.; Pourcelly, G.; Yaroslavtsev, A.B. Ion mobility in Nafion-117 membranes. Desalination 2004, 170, 49–57. [Google Scholar] [CrossRef]
- Ryu, H.W.; Kim, Y.M.; Wee, Y.J. Influence of operating parameters on concentration and purification of L-lactic acid using electrodialysis. Biotechnol. Bioprocess Eng. 2012, 17, 1261–1269. [Google Scholar] [CrossRef]
- Kosohin, O.; Mazanka, V. Anode material for oxidation of organic acids. Mater. Today Proc. 2019, 50, 518–520. [Google Scholar] [CrossRef]
- Richa, M.; García-Cervilla, R.; Lobato, J.; Cañizares, P.; Rodrigo, M.A. Carboxylic acids selective recovery from wastewater using electrodialysis. J. Environ. Chem. Eng. 2025, 13, 117752. [Google Scholar] [CrossRef]
- Torres, W.R.; Díaz Nieto, C.H.; Prévoteau, A.; Rabaey, K.; Flexer, V. Lithium carbonate recovery from brines using membrane electrolysis. J. Membr. Sci. 2020, 615, 118416. [Google Scholar] [CrossRef]
- Ma, Y.; Svärd, M.; Xiao, X.; Gardner, J.M.; Olsson, R.T.; Forsberg, K. Precipitation and crystallization used in the production of metal salts for Li-ion battery materials: A review. Metals 2020, 10, 1609. [Google Scholar] [CrossRef]
- Cipollina, A.; Winter, D.; Battaglia, G.; Berkemeyer, L.; Cortina, J.L.; de Labastida, M.F.; Rodriguez, J.L. Recovery of lithium carbonate from dilute Li-rich brine via homogeneous and heterogeneous precipitation. Ind. Eng. Chem. Res. 2022, 61, 13589–13602. [Google Scholar] [CrossRef]
- Shwan, K.; Yoon, H.; Min, T.; Han, B.; Lim, S.; Park, J. Carbon dioxide utilization in lithium carbonate precipitation: A short review. Environ. Eng. Res. 2024, 29, 230553. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.