Abstract
The fortuitous modification of a quinoline-proline-piperazine side chain linked to a steroid in the presence of lithium (trimethylsilyl) acetylide has generated an unknown product that is more active than its precursor. After having characterized two β-enaminones (two-carbon homologation compounds) that were generated from a simplified model side chain, we have identified the unknown product as being the β-enaminone steroid derivative 1. NMR analysis, especially two-dimensional (2D) experiments (correlation spectroscopy (COSY), NOE spectroscopy (NOESY), heteronuclear single-quantum correlation (HSQC) and heteronuclear multiple-bond correlation (HMBC)) provided crucial information that was found essential in the characterization of enaminone 1. We also proposed a mechanism to rationalize the formation of this biologically active compound.
1. Introduction
Cancer is still a major cause of death worldwide [1,2]. It is therefore urgent to develop new molecules to counter the uncontrolled proliferation of cancer cells [3,4]. Steroid derivatives having a (1-quinolin-2-ylcarbonyl)-l-proline-piperazine side chain at position C2 of estra-1,3,5(10)-trien-3,17β-diol [5] or 5α-androstane-3α,17β-diol [6,7,8] represent a new family of anticancer agents. They were found to reduce the proliferation of various human cancer cell lines [8,9,10], as well as to block tumor growth when tested in mouse tumor (xenografts) models of human cancers (pancreas, ovary, breast, and leukemia) [7,11]. During our work to increase their metabolic stability, especially for oral administration, one of our strategies was to introduce an ethynyl group at position C17 of the steroid nucleus to generate a tertiary alcohol instead of a secondary alcohol [12]. In the field of steroid drugs, this kind of transformation is known to increase metabolic stability by avoiding the oxidation of 17β-OH into a ketone by the metabolism of phase-I enzymes of the CYP family [13,14].
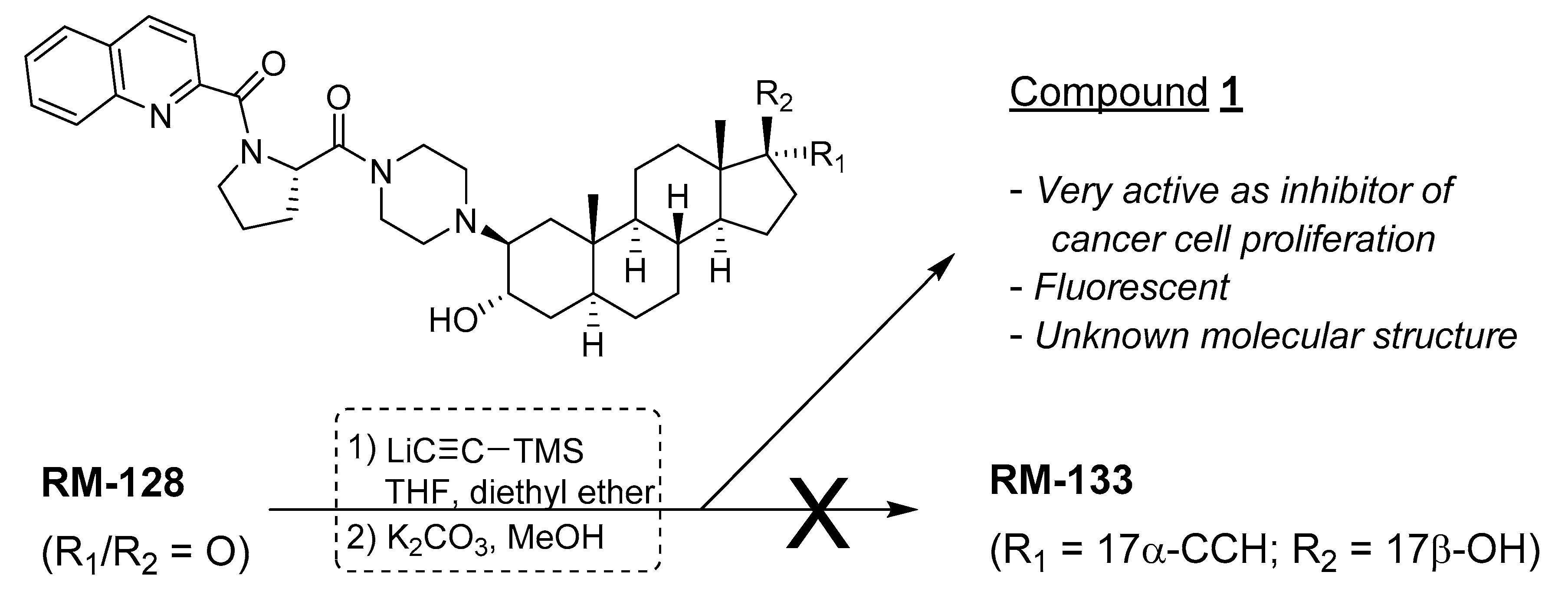
Trying to introduce the ethynyl group at the last step of the chemical synthesis gave unsatisfactory low yields (<30%) of the expected 17α-ethynyl/17β-OH derivative RM-133 from RM-128 (Figure 1), and the former was contaminated with unidentified side products. This more metabolic stable compound was alternatively obtained by another strategy that introduced the ethynyl group earlier in the reaction sequence, thus providing RM-133 in good yield and purity [7], but the negative results of the first chemical strategy provided an unexpected finding. In fact, the addition of lithium (trimethylsilyl)acetylide to the C17-carbonyl of RM-128 generated a new compound, which after purification was found to be very active as an antiproliferative agent on a variety of human cancer cell lines (OVCAR-3, PANC-1, MCF-7, T-47D), being about two times more potent than RM-133 in a comparative assay (unpublished results). The biological potential of this unknown compound prompted us to elucidate its chemical structure.
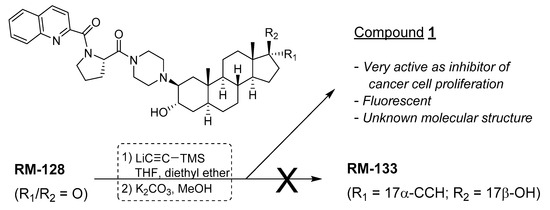
Figure 1.
Unexpected formation of an unknown biologically active steroid derivative.
2. Results and Discussion
2.1. Characteristic of Unknown Compound and Hypothesis
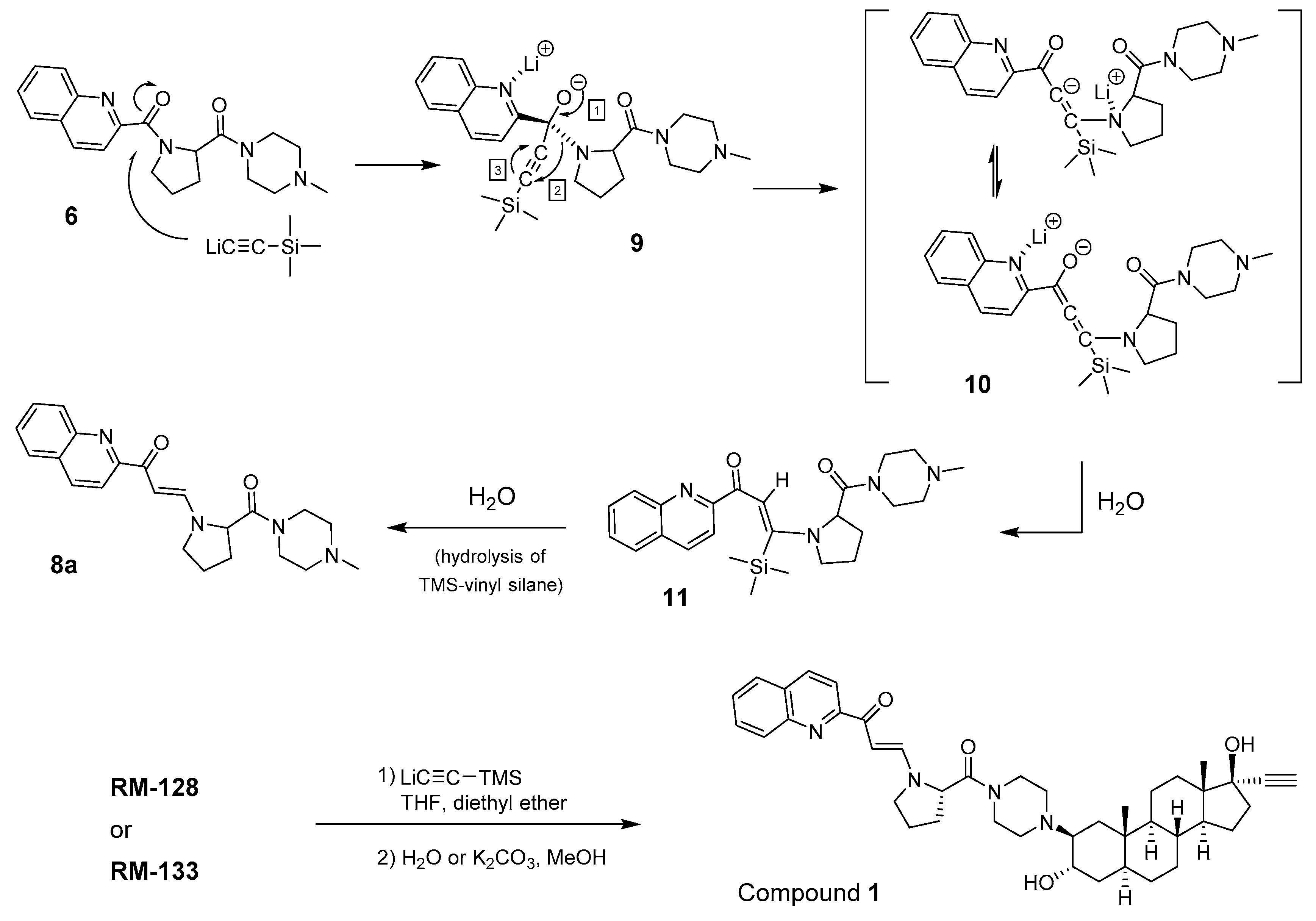
The major difficulty for the characterization of unknown compound 1 was the complex nature of the starting aminosteroid derivative RM-128 [8], which is well-known to exist as a mixture of two conformers in different proportions according to the solvent used for NMR analysis [7]. In fact, the presence of two amide bonds linking a piperazine, a proline, and a quinoline explain these two conformers, but the combinaison of this side chain and a steroid backbone with 19 carbons greatly complexifies the identification of such steroid derivatives by NMR. Moreover, as observed with other aminosteroids of similar structure, it is not possible to obtain a crystal for X-ray analysis. However, some conclusions can be drawn by the comparison of 1H NMR and 13C NMR spectra of the starting ketone (RM-128) and the unknown compound 1. In fact, the steroid backbone was not modified, except at C17, where an ethynyl group seems present, and the quinoline moiety was not altered either, thus suggesting a modification of the side chain close to the proline, although the two amide groups also seem to be present. Mass spectra analysis showed a peak at 679.5 m/z (M+H) instead of the expected peak at 652.4 m/z, suggesting the presence of another group in addition to the expected ethynyl group. The infrared (IR) analysis only confirmed the presence of amide and alcohol, the C≡C acetylenic signal being too small to be significant. Finally, an interesting observation is the strong fluorescence of this unknown compound (excitation λmax = 410 nm; emission λmax = 494 nm), which supports a modification of the side chain that is linked to the steroid nucleus.
2.2. Synthesis of Side Chain 6 as a Model for the Formation of Unknown Compounds
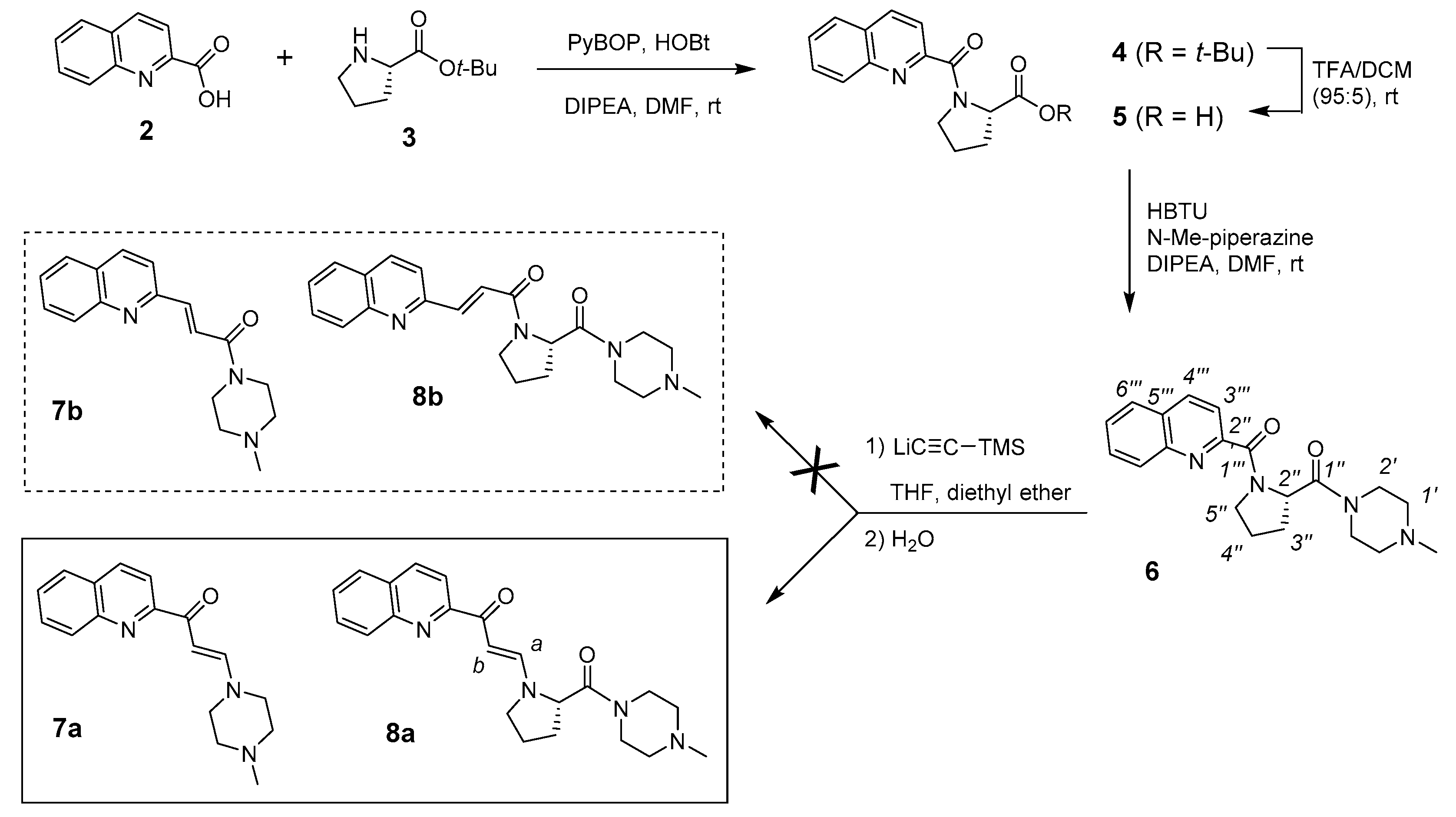
Given the limited information that is generated by the analysis of the spectral data of the unknown compound 1, as well as the complex structure of this type of aminosteroid derivative, we decided to try the same kind of reaction on the side chain model, as represented by compound 6. This later was easily prepared in three steps (Figure 2), by 1) a coupling of 2-quinaldic acid (2) with proline-t-butylester (3) using benzotriazol-1-yl-oxytripyrrolidinophosphonium hexafluorophosphate (PyBOP), 2) a hydrolysis of the ester group to the corresponding acid, and 3) a coupling of this acid with N-methyl-piperazine using 2-(1H-benzotriazol-1-yl)-1,1,3,3-tetramethyluronium hexafluorophosphate (HBTU) as reagent. When reacting compound 6 with lithium (trimethylsilyl)acetylide, we obtained a mixture of products that was purified by flash chromatography, leading to two unknown compounds in 15 and 30% yields, respectively.
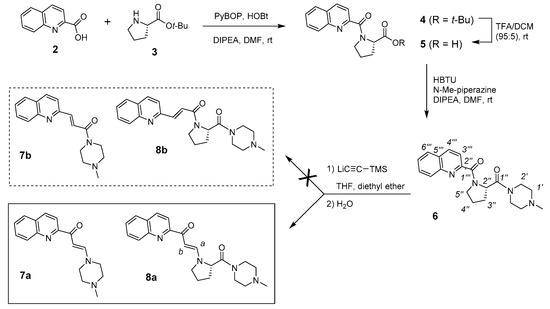
Figure 2.
Synthesis of model side chain (compound 6) and two rearranged side chains (compounds 7a and 8a) resulting from the ethynylation reaction.
2.3. NMR Characterization of Unknown Side Chain (Enaminones 7 and 8)
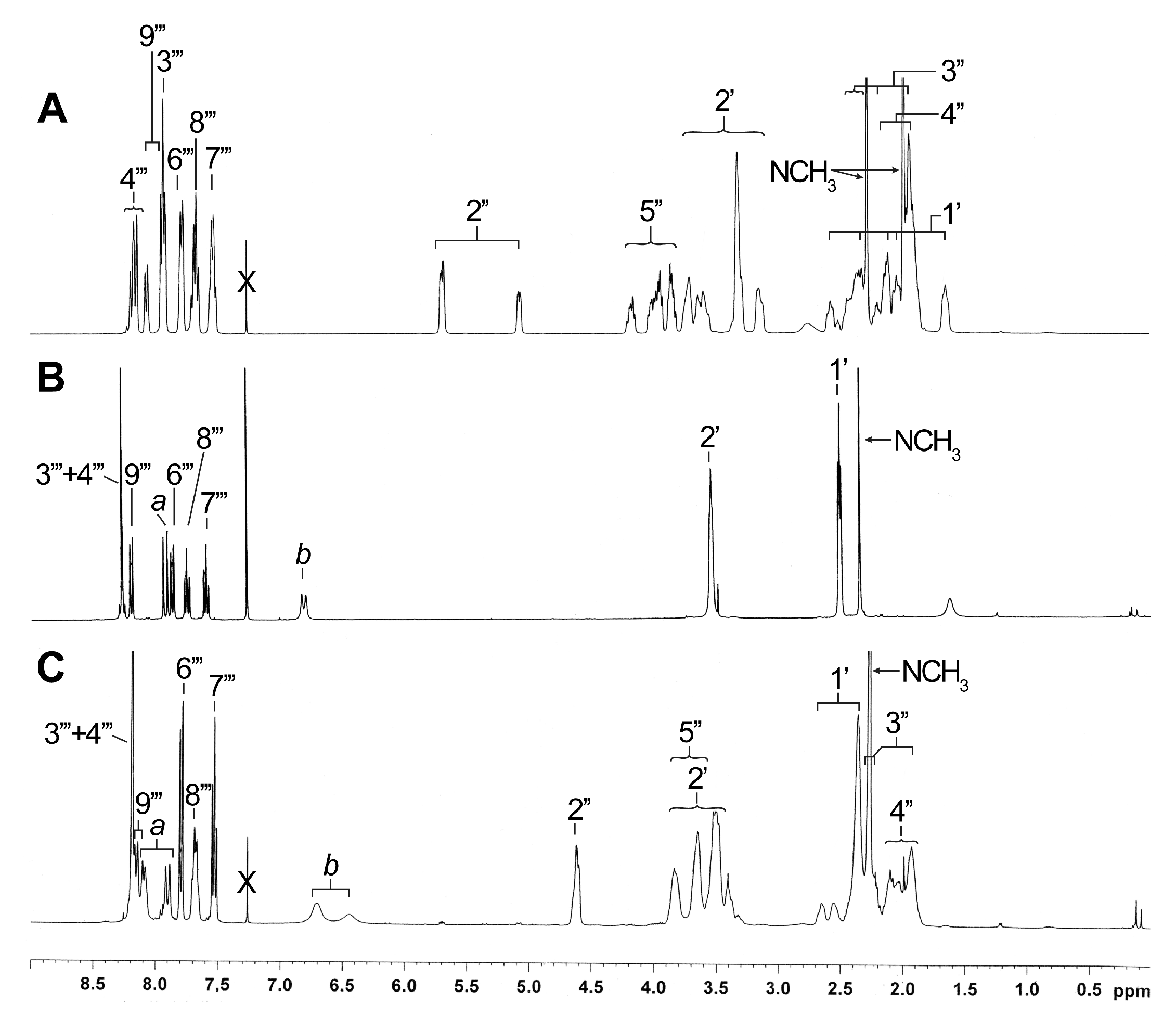
Comparison of the NMR data of compound 6 (Figure 3A) with those of the two unknown side chains generated from 6 provided interesting information. In fact, the 1H NMR analysis of the minor compound (Figure 3B) demonstrated the presence of N-methylpiperazine and quinoline groups, but surprisingly, the absence of the proline moiety. These observations were also confirmed by mass analysis ([M+H]+ = 282.1 m/z). For the major compound, however, the proline was clearly present in NMR spectra (Figure 3C) and mass analysis ([M+H]+ = 379.1 m/z). In NMR spectra of both compounds, there is no evidence of peak splitting for the N-CH3 of piperazine, the CH of proline and some quinoline signals. This duplication is typical of the presence of the two rotamers observed for a side chain like 6 [7], especially obvious for the CH-2′′ (Figure 3A). New signals (a and b) integrating for 2H were also detected in the aromatic or vinylic region (6.80 and 7.91 ppm for 7 and 6.45/6.70 and 7.90/8.08 ppm for 8; correlations in correlation spectroscopy (COSY) spectra).
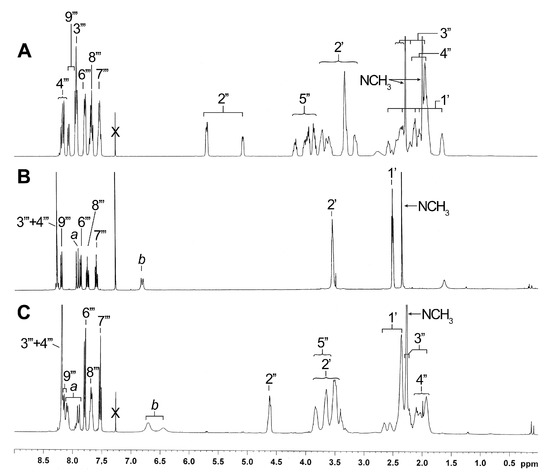
Figure 3.
Comparison of 1H NMR spectra in CDCl3 of known side chain 6 (A) and unknown side chains 7a (B) and 8a (C).
In 13C NMR, the carbonyl peaks at 166.1 and 166.6 ppm (C-1′′′) disappeared and a weak signal at 187.6 or 187.4 ppm for 7 or 8, respectively. Moreover, two new weak peaks (CH) at 91.0/153.0 or 93.7/150.0 ppm appeared for 7 or 8, respectively (Table 1). Two CH-aromatic signals were also deshielded, while the CH-3′′′ signal was slightly shielded, but the other signals were roughly the same. Taking into account NMR and mass data, especially an additional 26-mass units that were observed for the major compound, we can therefore assume the presence of a CH=CHCO group between the quinoline and proline moieties, which compounds could be represented by 7a or 7b, for the minor compound, and 8a or 8b, for the major compound.

Table 1.
Chemical shifts (δ in ppm) and assignation of carbons from compounds 6, 7a, 8a, and 1.
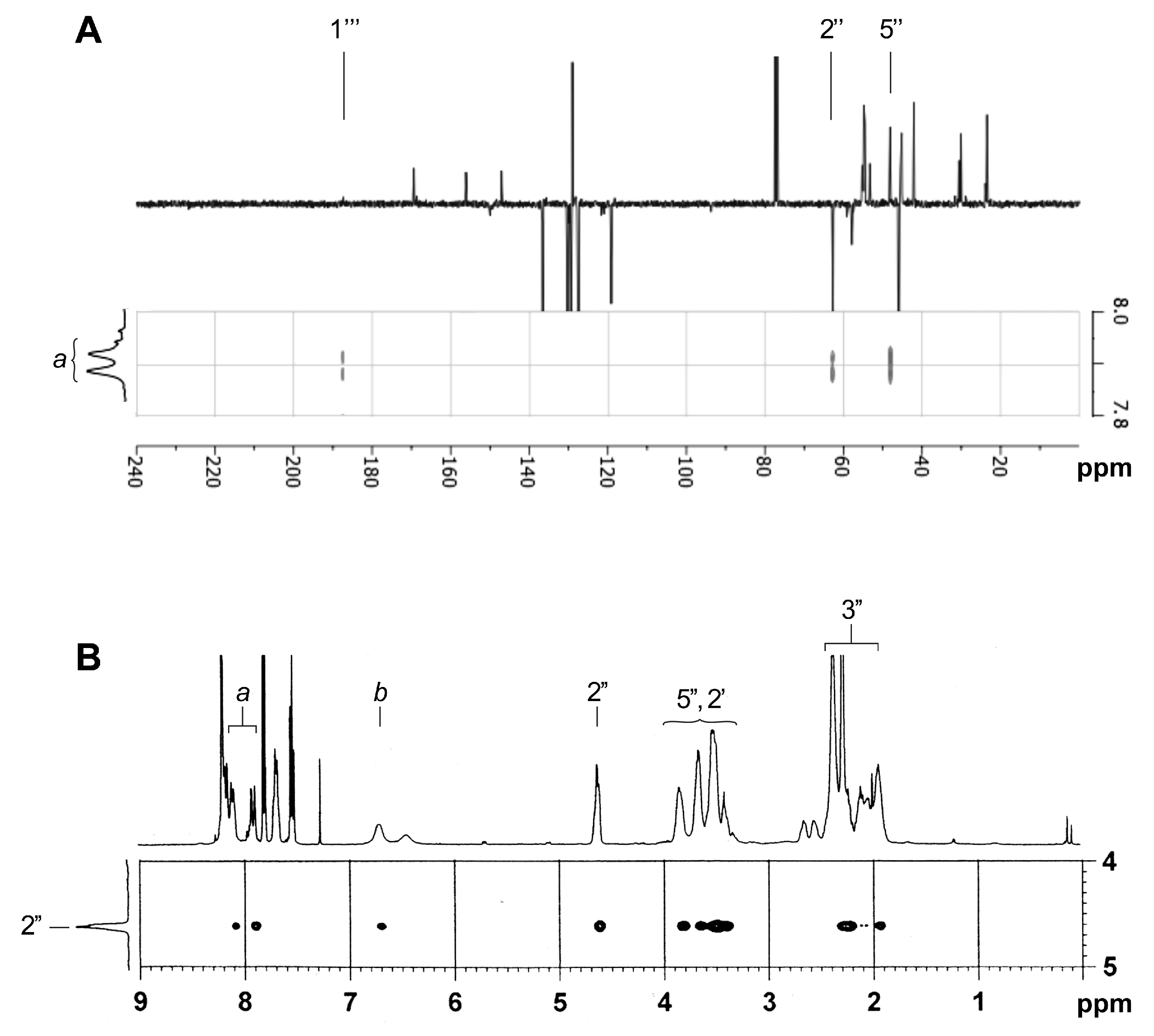
We next focused on the major compound 8 because its side chain is similar to the side chain that is present in the bioactive steroid compound 1. IR analysis does not make it possible to discriminate between the two possible regioisomers 8a and 8b (same carbonyl band at 1643 cm−1), but in 13C NMR, the peak at 187.4 ppm corresponds very well to a conjugated ketone rather than a conjugated amide. Moreover, the experimental and calculated (ChemDraw 14.0) chemical shifts for this conjugated ketone are identical (Table 1). The HMBC spectrum of 8 showed J3-coupling between the vinyl CH-a and the carbonyl C-1′′′ at 187.4 ppm, as well as with two proline signals (CH-2′′ and CH-5′′′) (Figure 4A). A J3-coupling between the CH-3′′′ of quinoline and the carbonyl at 187.4 ppm was also observed. The NOESY spectrum does not show correlations between a CH-2′′ of proline and any CH of the quinoline nucleus, but correlations between CH-2′′ and the vinylic CH-a and CH-b were found, thus supporting the β-enaminone structure of 8a (Figure 4B). The coupling constants (J = 13.0 Hz), as well as no NOE correlation between CH-a and CH-b confirm the trans-configuration of the alkene 8a. After the characterization of 8a, the minor unknown side chain was found to be the β-enaminone 7a. In addition to confirm the structures of 7a and 8a as the minor and major compounds resulting from the ethynylation of 6, two-dimensional (2D) NMR analyzes also allowed for the complete assignment of protons and carbons of 6, 7a, and 8a (Table 1). These data will be crucial for the elucidation of the unknown steroidal compound 1.
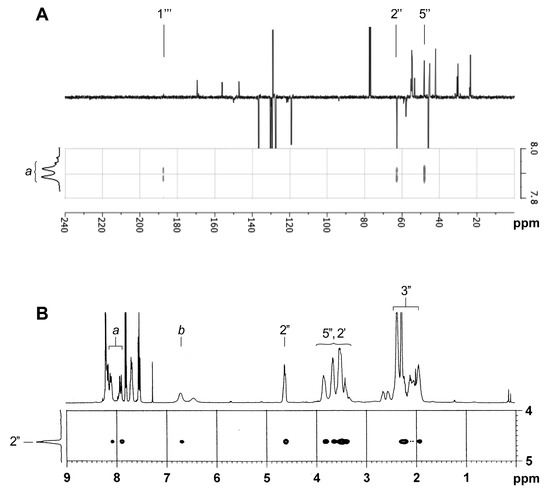
Figure 4.
Partial HMBC (A) and NOE spectroscopy (NOESY) (B) results supporting the structure of β-enaminone 8a.
2.4. NMR Characterization of Unknown Steroidal Compound 1
After we characterized the enaminone 8a from an experiment with a simplified model of side chain, it was possible to make a direct comparison of the NMR data of 1 and 8a. In 1H NMR, the quinoline and enaminone signals were found to be identical, while the CH-2′′ and CH2-5′′ signals of proline were at the same chemical shifts (4.6 and 3.4–3.9 ppm). However, a comparison of the other signals was not possible because of the presence of numerous protons of steroid backbone, but two methyl groups (CH3-18 and CH3-19) and an acetylenic proton (2.58 ppm) are also present. A comparison of the 13C NMR data clearly demonstrated the similarity of the chemical shifts for the β-enaminone 8a and the side chain at position 2β of the steroid. In fact, the only disparities are related to the presence of a CH3 instead of the steroid nucleus on a piperazine nitrogen, which affects the chemical shifts of the two CH2-1′. Analysis of the 13C NMR data also makes it possible to completely confirm the 5α-androstan-3β,17β-diol backbone, as well as the presence of an ethynyl group (73.9 and 87.6 ppm) at C-17α (79.8 ppm) [7]. Finally, mass analysis ([M+H]+ = 679.5 m/z) is in agreement with the proposed structure for compound 1.
2.5. Mechanism of β-Enaminone Formation
After an exhaustive research in literature for such transformation, we found that the preparation of an enaminone by a two-carbon homologation of amides with lithium(triphenylsilyl)acetylide was already described by Suzuki et al. in the eighties [15]. A brief explanation of the reaction mechanism was given, suggesting a sequence of events, including an initial formation of a silylalkynone, followed by a Michael-type addition of in situ-formed lithium amide, and a subsequent protiodesilylation [16]. Interestingly, a D2O-quenching experiment showed the double incorporation of deuterium in the generated double bond [15]. Inspired by the mechanistic explanations provided by Suzuki et al., and to better visualize this interesting transformation, we hereby suggest a stepwise mechanism to explain the β-enaminone formation from the addition of lithium (trimethylsilyl)acetylide to the quinoline-proline-piperazine side chain of compound 6 (Figure 5). In a first step, the lithium (trimethylsilyl)acetylide attacks the carbonyl of the amide that is located between the quinoline and proline. The resulting intermediate 9 undergoes a rearrangement following events 1–3, which is driven by the electrophilic nature of acetylenic carbons. The rearranged intermediate 10 is then protonated by a molecule of water to afford 11, which thereafter undergoes a protiodesilylation leading to the β-enaminone 8a. Using the proposed mechanism it is now possible to explain the synthesis of compound 1, from RM-128 (C17-ketone) or RM-133 (17α-ethynyl). In the first case, the carbonyls C-17 and C-1′′′ of RM-128 will react with lithium(trimethylsilyl)acetylide to introduce the 17α-ethynyl and to produce the β-enaminone side chain. In the second case, however, only the C-1′′′ of RM-133 is involved in the reaction producing 1.
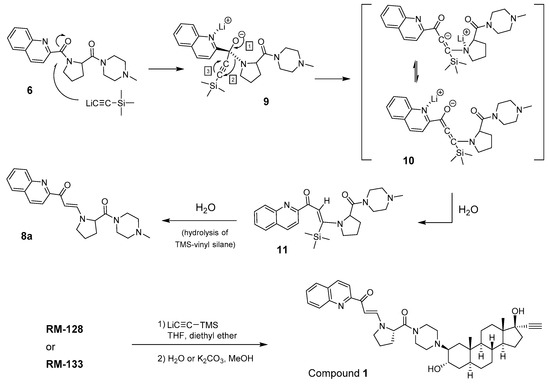
Figure 5.
Proposed mechanism of reaction leading to β-enaminones 8a and 1.
3. Materials and Methods
3.1. General
Chemical reagents and solvents were purchased from commercial suppliers and were used as received. Phase separator syringes were purchased from Biotage (Isolute phase separator, 6 mL). Thin-layer chromatography (TLC) and flash-column chromatography were performed on 0.20-mm silica gel 60 F254 plates (E. Merck; Darmstadt, Germany) and with 230–400 mesh ASTM silica gel 60 (Silicycle, Québec, QC, Canada), respectively. Infrared (IR) spectra were recorded with a Horizon MB 3000 ABB FTIR spectrometer (Québec, QC, Canada). Nuclear magnetic resonance (NMR) spectra were recorded at room temperature in CDCl3 with a 5-mm NMR tube on a Bruker AVANCE 400 spectrometer (Billerica, MA, USA). 1H and 13C NMR chemical shifts were referenced to the residual peak of CHCl3 (7.26 and 77.0 ppm, respectively). For characterization, we used the following experiments: attached proton test (APT), correlation spectroscopy (COSY), homonuclear two-dimensional NOE spectroscopy (NOESY), heteronuclear single-quantum correlation (HSQC), and heteronuclear multiple-bond correlation (HMBC). These NMR experiments were performed according to the manufacturer’s instructions. Low-resolution mass spectra (LRMS) were recorded on a Shimadzu Prominence apparatus (Kyoto, Japan) equipped with a Shimadzu LCMS-2020 mass spectrometer and an APCI probe.
3.2. Synthesis of Side Chain 6
The details of the chemical synthesis of 6 were previously published [6].
3.3. Synthesis of β-Enaminones 7a and 8a
To a solution of trimethylsilylacetylene (1.35 mL, 9.55 mmol) in anhydrous tetrahydrofuran (THF) (100 mL) at 0 °C under an atmosphere of argon was added dropwise methyl lithium (4.8 mL, 7.68 mmol; 1.6 M in diethyl ether) over a period of 10 min. This solution was left to return to room temperature and was stirred for 1 h. The solution was then cooled again at 0 °C and compound 6 (870 mg 1.91 mmol) was added in anhydrous THF (20 mL). The solution was stirred at room temperature for 1 h. The resulting solution was then poured into water (750 mL) and was extracted twice with dichloromethane (DCM). The organic layer was washed with water, dried over phase separator syringe, and evaporated under reduced pressure to give 750 mg of a brown amorphous solid. The crude compound was purified by flash chromatography using a gradient of DCM/MeOH (95:5) to DCM/MeOH (9:1) to give β-enaminones 7a (117 mg, 15%) and 8a (226 mg, 30%) as green and yellow amorphous solid, respectively.
(E)-3-(4-methylpiperazin-1-yl)-1-(quinolin-2-yl)prop-2-en-1-one (7a): 1H NMR (CDCl3) δ in ppm: 2.35 (s, 3H, NCH3), 2.51 (t, J = 5.0 Hz, 4H, CH2-1′), 3.54 (s broad, 4H, CH2-2′), 6.80 (d, J = 12.7 Hz, 1H, CH-b), 7.58 (t, J = 7.1 Hz, 1H, CH-7′′′), 7.74 (t, J = 8.4 Hz, 1H, CH-8′′′), 7.85 (d, J = 8.1 Hz, 1H, CH-6′′′), 7.91 (d, J = 12.9 Hz, 1H, CH-a), 8.18 (d, J = 8.5 Hz, 1H, CH-9′′′), 8.26 (s, 2H, CH-3′′′ and CH-4′′′). 13C NMR (CDCl3) δ in ppm: 46.6 (CH2-2′), 46.0 (NCH3), 54.6 (CH2-1′), 91.0 (CH-b), 119.0 (CH-3′′′), 127.4 (CH-7′′′), 127.6 (CH-6′′′), 129.0 (CH-5′′′), 129.4 (CH-9′′′), 130.1 (CH-8′′′), 136.6 (CH-4′′′), 147.0 (C-10′′′), 153.0 (CH-a), 155.9 (C-2′′′), 187.6 (C-1′′′). LRMS for C17H20N3O [M + H]+ 282.2 (calc), 282.1 (found).
(S,E)-3-(2-(4-methylpiperazine-1-carbonyl)pyrrolidin-1-yl)-1-(quinolin-2-yl)prop-2-en-1-one (8a): 1H NMR (CDCl3) δ in ppm: 1.90–2.70 (m, 8H, CH2-3′′, CH2-4′′, 2 x CH2-1′), 2.27 (s, 3H, NCH3), 3.41, 3.51, 3.65 and 3.84 (4m, 6H, CH2-2′ and CH2-5′′), 4.62 (s broad, 1H, CH-2′′), 6.45 and 6.70 (2s broad, 1H, CH-b), 7.53 (t, J = 7.1 Hz, 1H, CH-7′′′), 7.69 (t, J = 7.0 Hz, 1H, CH-8′′′), 7.79 (d, J = 8.1 Hz, 1H, CH-6′′′), 7.90 and 8.08 (2d, J = 13.0 Hz, 1H, CH-a), 8.14 (m, 1H, CH-9′′′), 8.19 (s, 2H, CH-3′′′ and CH-4′′′). 13C NMR (CDCl3) δ in ppm: 23.4 and 23.9 (CH2-4′′), 30.1 and 30.6 (CH2-3′′), 42.1, 45.1, 45.5 and 45.7 (CH2-2’), 46.0 (NCH3), 48.1 and 53.3 (CH2-5′′), 54.5 and 54.8 (CH2-1′), 57.9 and 62.7 (CH2-2′′), 93.7 (CH-b), 119.1 (CH-3′′′), 127.4 (CH-7′′′), 127.6 (CH-6′′′), 129.0 (CH-5′′′), 129.3 and 129.4 (CH-9′′′), 130.3 (CH-8′′′), 136.5 (CH-4′′′), 147.1 (C-10′′′), 150.0 (CH-a), 156.1 (C-2′′′), 168.6 and 169.4 (C-1′′), 187.4 (C-1′′′). LRMS for C22H27N4O2 [M + H]+ 379.2 (calc), 379.1 (found).
3.4. Synthesis of Enaminone 1
To a solution of trimethylsilylacetylene (216 µL, 1.52 mmol) in anhydrous THF (5 mL) at 0 °C under an atmosphere of argon, was added dropwise methyl lithium (860 µL, 1.38 mmol; 1.6 M in diethyl ether) over a period of 10 min. This solution was left to return to room temperature and was stirred for 1 h. The solution was then cooled again at 0 °C and compound RM-133 (100 mg 0.15 mmol) was added in anhydrous THF (20 mL). The solution was stirred at room temperature overnight. The resulting solution was then poured into water (200 mL) and extracted two times with EtOAc. The organic layer was washed with brine, dried over sodium sulfate, filtered, and evaporated under reduced pressure to give 110 mg of a yellow amorphous solid. The crude compound was purified by flash chromatography using a gradient of DCM/MeOH (97:3) to DCM/MeOH (9:1) to give β-enaminone 1 (31 mg, 30%) as fluorescent pale green amorphous solid.
(S,E)-3-(2-(4-[(3β,5α,17α)-17-hydroxypregn-20-yn-3-yl]piperazine-1-carbonyl)pyrrolidin-1-yl)-1-(quinolin-2-yl)prop-2-en-1-one (1): 1H NMR (CDCl3) δ in ppm: 0.75 (m, 1H, CH-9), 0.83 (s, 3H, CH3-18), 0.85 (s, 3H, CH3-19), 0.88 (m, 1H of CH2-7), 1.10–2.35 (residual CH and CH2), 2.40–2.75 (m, 5H, CH-2α and 2 x CH2-1′), 2.56 (s, 1H, CH-21), 3.35–3.40 (m, 6H, CH2-5′′ and 2 x CH2-2′), 3.86 (m, 1H, CH-3β), 4.66 (s broad, 1H, CH-2′′), 6.48 and 6.73 (2s broad, 1H, CH-b), 7.57 (t, J = 7.4 Hz, 1H, CH-7′′′), 7.73 (t, J = 7.2 Hz, 1H, CH-8′′′), 7.84 (d, J = 8.1 Hz, 1H, CH-6′′′), 7.94 and 8.10 (2d, J = 13.0 Hz, 1H, CH-a), 8.16 (m, 1H, CH-9′′′), 8.13 (s, 2H, CH-3′′′ and CH-4′′′). 13C NMR (CDCl3) δ in ppm: 23.6, 23.9 (CH2-4′′), 30.1 and 30.7 (CH2-3′′), 41.9 and 42.7, 45.8 and 46.2 (CH2-2’), 48.1 and 53.4 (CH2-5′′), 46.8, 48.0 and 48.6 (CH2-1′), 57.7 and 62.7 (CH2-2′′), 93.6 (CH-a), 119.1 (CH-3′′′), 127.0 (CH-7′′′), 127.4 (CH-6′′′), 129.1 (CH-5′′′), 129.9 and 130.3 (CH-9′′′), 129.4 (CH-8′′′), 136.5 (CH-4′′′), 147.1 (C-10′′′), 149.7 (CH-b), 156.1 (C-2′′′), 168.9 and 169.2 (C-1′′), 187.5 (C-1′′′). IR (film) ν in cm-1: 3480 (OH), 1643 (C=O), 1543 (C=C). LRMS for C42H54N4O4 [M + H]+ 679.4 (calc), 679.5 (found).
4. Conclusions
The combination of a steroid nucleus and a quinoline-proline-piperazine side chain has resulted in the discovery of a new family of anticancer drugs that induce the apoptosis of cancer cells via endoplasmic reticulum stress. The fortuitous modification of this chain in the presence of lithium (trimethylsilyl) acetylide having generated an unknown product that is more active than the precursor, it was important to determine its structure. After having determined the formation of two homologation products from a model side chain, thanks to NMR analysis, we have been able to identify the unknown product as being β-enaminone 1. We also proposed a mechanism to rationalize the formation of this biologically active compound. The optimization of the experimental conditions remains to be done, but this type of rearrangement could be favorably used in a diversity-oriented synthesis strategy to obtain structurally different substituted enaminones, such as 7a and 8a, from the same amide unit.
Acknowledgments
We are grateful to the Canadian Institutes of Health Research for financial support (POP-I program), to Jean-Yves Sancéau for helpful discussions, to Marie-Claude Trottier and Sophie Boutin for NMR analyses and to Micheline Harvey for careful reading of this manuscript.
Author Contributions
D.P. and R.M. conceived and designed the experiments; R.M. performed the experiments; D.P. analyzed the data; D.P. and R.M. wrote the paper.
Conflicts of Interest
The authors declare no conflict of interest. D.P. and R.M. have patent rights on US8653054 and CA2,744,369 (2-(N-Substituted piperazinyl) steroid derivatives) as well as on PCT/CA2017/000140 (Aminosteroid derivatives and process for producing same).
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2017. CA Cancer J. Clin. 2017, 67, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Gabriele, L.; Buoncervello, M.; Ascione, B.; Bellenghi, M.; Matarrese, P.; Care, A. The gender perspective in cancer research and therapy: Novel insights and on-going hypotheses. Ann. Ist. Super. Sanit. 2016, 52, 213–222. [Google Scholar]
- Gupta, A.; Kumar, B.S.; Negi, A.S. Current status on development of steroids as anticancer agents. J. Steroid Biochem. Mol. Biol. 2013, 137, 242–270. [Google Scholar] [CrossRef] [PubMed]
- Atkins, J.H.; Gershell, L.J. Selective anticancer drugs. Nat. Rev. Drug Discov. 2002, 1, 491–492. [Google Scholar] [CrossRef] [PubMed]
- Perreault, M.; Maltais, R.; Roy, J.; Dutour, R.; Poirier, D. Design of a mestranol 2-N-piperazino-substituted derivative showing potent and selective in vitro and in vivo activities in MCF-7 breast cancer models. ChemMedChem 2017, 12, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Perreault, M.; Maltais, R.; Dutour, R.; Poirier, D. Explorative study on the anticancer activity, selectivity and metabolic stability of related analogs of aminosteroid RM-133. Steroids 2016, 115, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Maltais, R.; Hospital, A.; Delhomme, A.; Roy, J.; Poirier, D. Chemical synthesis, NMR analysis and evaluation on a cancer xenograft model (HL-60) of the aminosteroid derivative RM-133. Steroids 2014, 82, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Ayan, D.; Maltais, R.; Hospital, A.; Poirier, D. Chemical synthesis, cytotoxicity, selectivity and bioavailability of 5alpha-androstane-3alpha,17beta-diol derivatives. Bioorg. Med. Chem. 2014, 22, 5847–5859. [Google Scholar] [CrossRef] [PubMed]
- Jegham, H.; Roy, J.; Maltais, R.; Desnoyers, S.; Poirier, D. A novel aminosteroid of the 5α-androstane-3α,17β-diol family induces cell cycle arrest and apoptosis in human promyelocytic leukemia HL-60 cells. Investig. New Drugs 2012, 30, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Jegham, H.; Maltais, R.; Roy, J.; Doillon, C.; Poirier, D. Biological evaluation of a new family of aminosteroids that display a selective toxicity for various malignant cell lines. Anti-Cancer Drugs 2012, 23, 803–814. [Google Scholar] [CrossRef] [PubMed]
- Kenmogne, L.C.; Ayan, D.; Roy, J.; Maltais, R.; Poirier, D. The aminosteroid derivative RM-133 shows in vitro and in vivo antitumor activity in human ovarian and pancreatic cancers. PLoS ONE 2015, 10, e0144890. [Google Scholar] [CrossRef] [PubMed]
- Talbot, A.; Maltais, R.; Poirier, D. New diethylsilylacetylenic linker for parallel solid-phase synthesis of libraries of hydroxy acetylenic steroid derivatives with improved metabolic stability. ACS Comb. Sci. 2012, 14, 347–351. [Google Scholar] [CrossRef] [PubMed]
- Huber, M.M.; Ternes, T.A.; von Gunten, U. Removal of estrogenic activity and formation of oxidation products during ozonation of 17alpha-ethinylestradiol. Environ. Sci. Technol. 2004, 38, 5177–5186. [Google Scholar] [CrossRef] [PubMed]
- Thorpe, K.L.; Cummings, R.I.; Hutchinson, T.H.; Scholze, M.; Brighty, G.; Sumpter, J.P.; Tyler, C.R. Relative potencies and combination effects of steroidal estrogens in fish. Environ. Sci. Technol. 2003, 37, 1142–1149. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.; Ohkuma, T.; Tsuchihashi, G. Preparation of enaminones by two-carbon homologation of amides with lithium (triphenylsilyl)acetylide. J. Org. Chem. 1987, 52, 2929–2930. [Google Scholar] [CrossRef]
- Newman, H. Preparation of α,β-unsaturated aldehydes from acid chlorides. J. Org. Chem. 1973, 38, 2254–2255. [Google Scholar] [CrossRef]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).