Early Performance of Recently Released Rootstocks with Grapefruit, Navel Orange, and Mandarin Scions under Endemic Huanglongbing Conditions in Florida
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Site Description
2.3. Experimental Design
2.4. Data Collection and Analysis
3. Results and Discussions
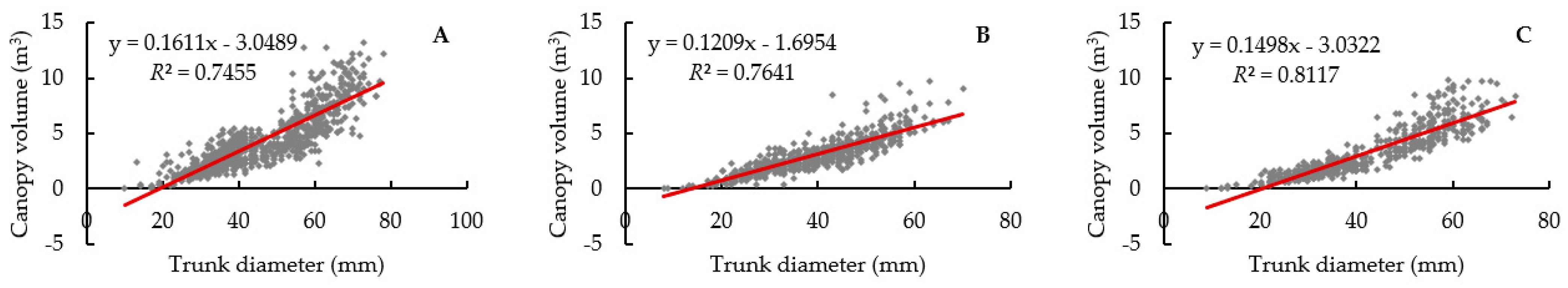
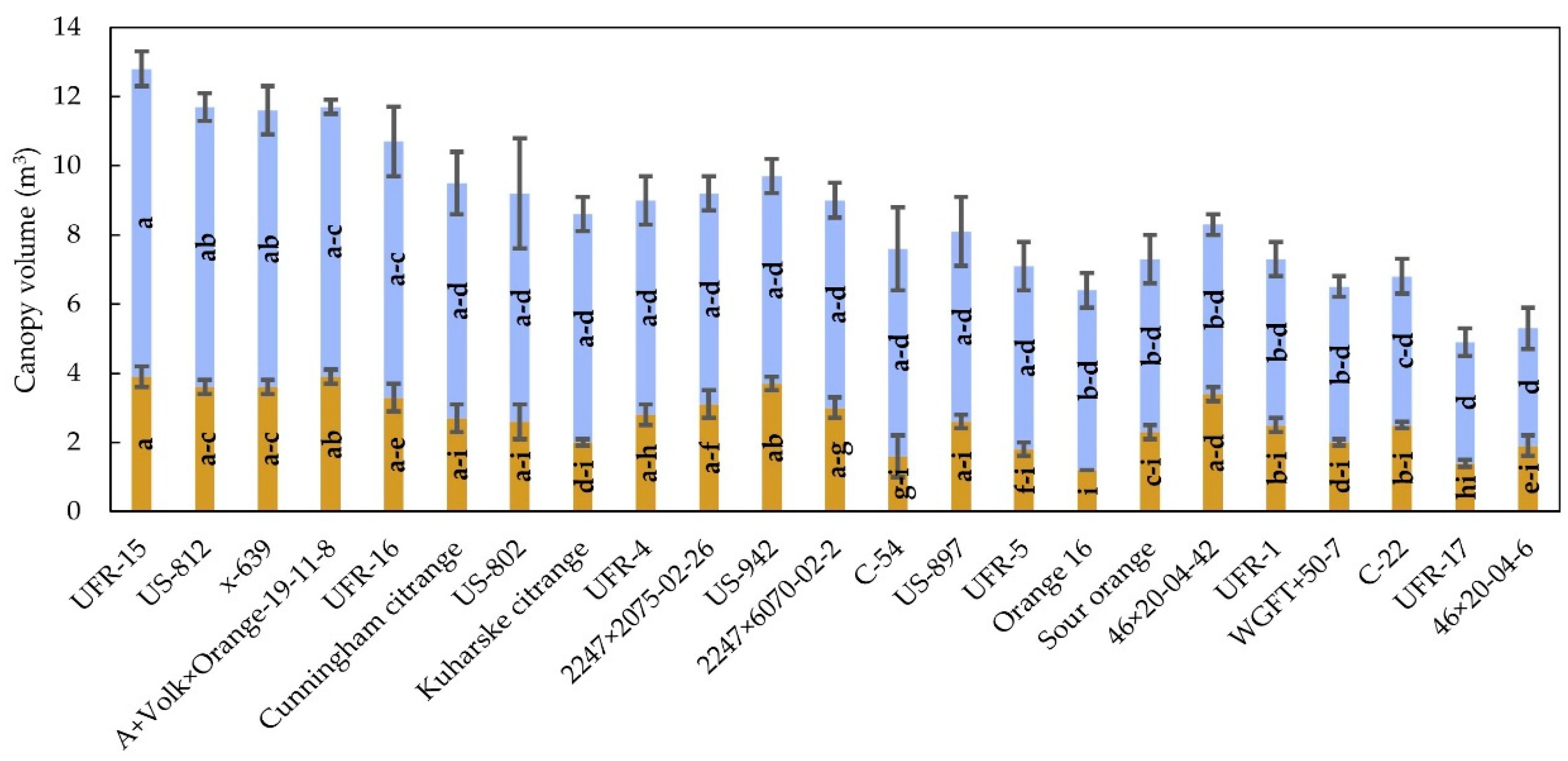
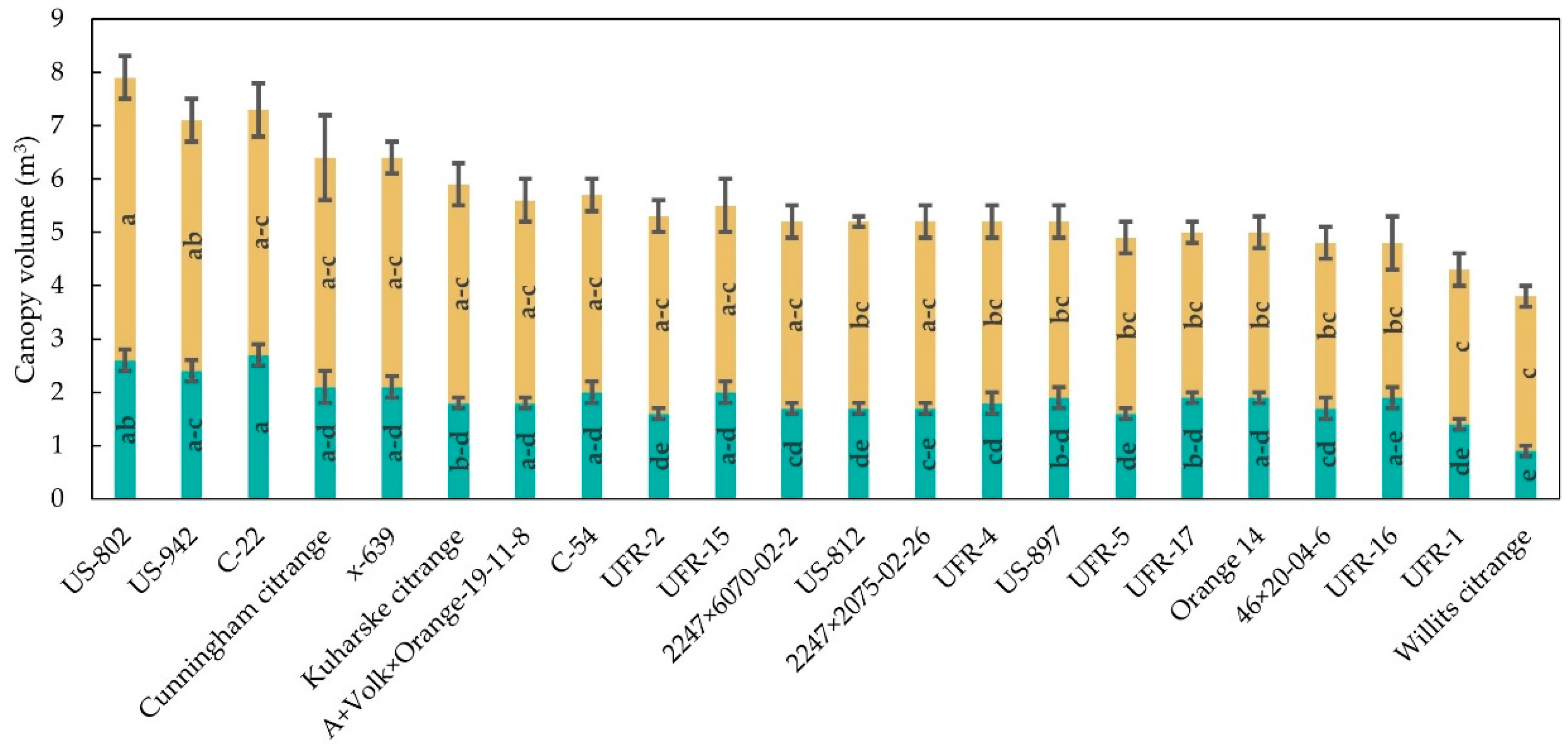
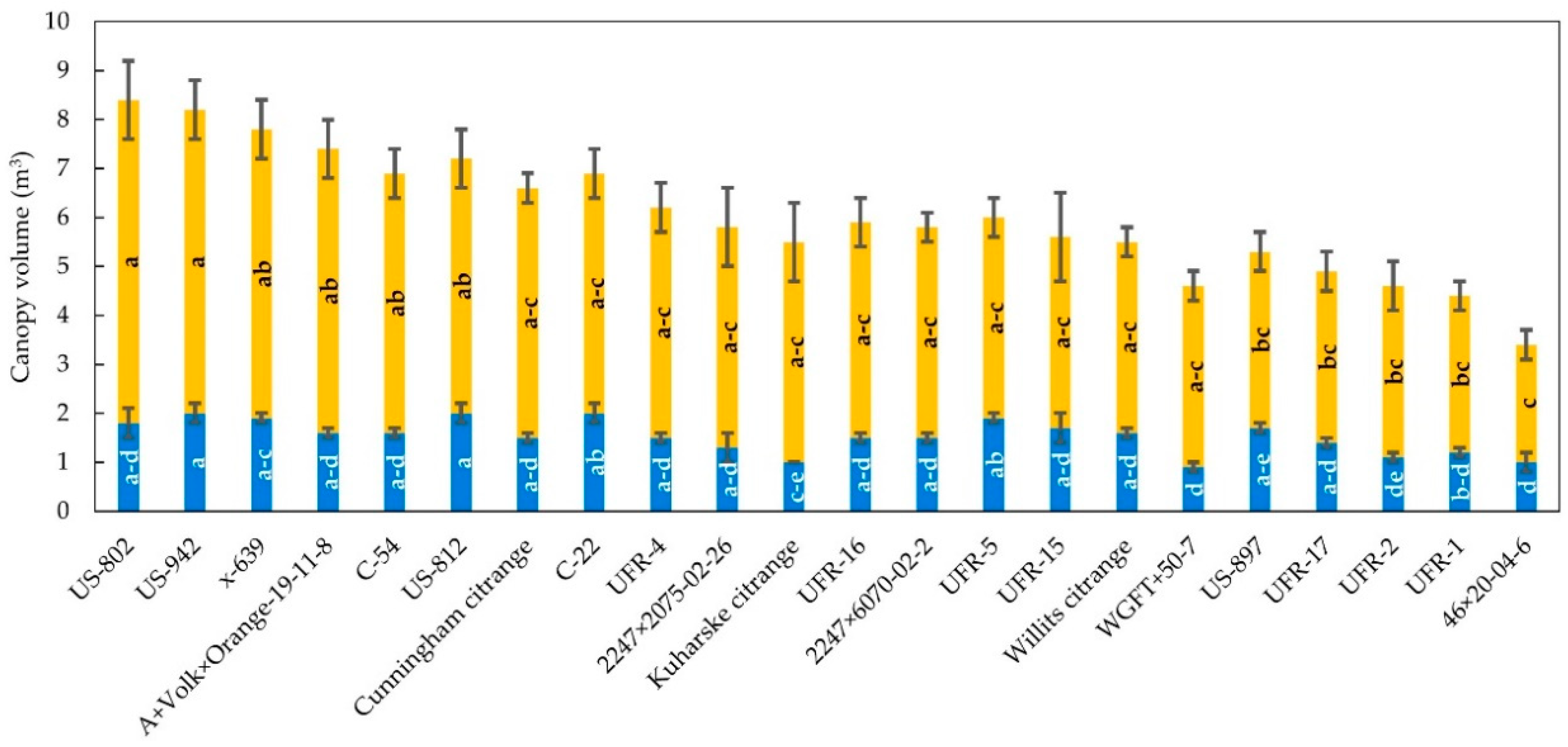
3.1. Tree Size
3.2. CLas Titer in Plant Leaf Tissue and HLB Severity Index
3.3. Leaf Nutrient Concentrations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Graham, J.; Gottwald, T.; Setamou, M. Status of Huanglongbing (HLB) outbreaks in Florida, California and Texas. Trop. Plant Pathol. 2020, 45, 265–278. [Google Scholar] [CrossRef]
- USDA. Florida Citrus Statistics 2020–2021; Florida Department of Agriculture and Consumer Services: Maitland, FL, USA, 2022; pp. 14–31. Available online: https://www.nass.usda.gov/Statistics_by_State/Florida/Publications/Citrus/Citrus_Statistics/2020-21/fcs2021b.pdf (accessed on 1 September 2022).
- Bové, J.M. Huanglongbing: A destructive, newly-emerging, century-old disease of citrus. J. Plant Pathol. 2006, 88, 7–37. [Google Scholar]
- Hall, D.G.; Richardson, M.L.; Ammar, E.D.; Halbert, S.E. Asian citrus psyllid, Diaphorina citri, vector of citrus huanglongbing disease. Entomol. Exp. Appl. 2013, 146, 207–223. [Google Scholar] [CrossRef]
- Ferrarezi, R.S.; Vincent, C.I.; Urbaneja, A.; Machado, M.A. Unravelling citrus Huanglongbing disease. Front. Plant Sci. 2020, 11, 609655. [Google Scholar] [CrossRef] [PubMed]
- Gottwald, T.R. Current epidemiological understanding of citrus huanglongbing. Annu. Rev. Phytopathol. 2010, 48, 119–139. [Google Scholar] [CrossRef]
- Albrecht, U.; Bowman, K.D. Tolerance of trifoliate citrus rootstock hybrids to Candidatus Liberibacter asiaticus. Sci. Hortic. 2012, 147, 71–80. [Google Scholar] [CrossRef]
- Tang, L.; Vashisth, T. New insight in Huanglongbing-associated mature fruit drop in citrus and its link to oxidative stress. Sci. Hortic. 2020, 265, 109246. [Google Scholar] [CrossRef]
- USDA. Citrus Fruits Summary 2021; National Agricultural Statistics Service: Maitland, FL, USA, 2021; p. 28. Available online: https://downloads.usda.library.cornell.edu/usda-esmis/files/j9602060k/kp78hg05n/1544cn77s/cfrt0921.pdf (accessed on 1 October 2021).
- McCollum, G.; Bowman, K.D. Rootstock effects on fruit quality among ‘Ray Ruby’grapefruit trees grown in the Indian River District of Florida. HortScience 2017, 52, 541–546. [Google Scholar] [CrossRef]
- Barry, G.H.; Caruso, M.; Gmitter, F.G., Jr. Commercial scion varieties. In The Genus Citrus; Woodhead Publishing: Cambridge, UK, 2020; pp. 83–104. [Google Scholar]
- FDACS. Citrus Budwood: Annual Report 2020–2021; Florida Department of Agriculture and Consumer Services: Winter Haven, FL, USA, 2021; p. 33. Available online: https://www.fdacs.gov/content/download/101636/file/CitrusBudwoodAnnualReport2021v3.pdf (accessed on 1 September 2022).
- Spyke, P.; Castle, W.S. Navel Oranges: Current status and Future in Florida. Citrus Ind. 2021, 8, 1–4. [Google Scholar]
- Stover, E.; Inch, S.; Richardson, M.L.; Hall, D.G. Conventional citrus of some scion/rootstock combinations show field tolerance under high Huanglongbing disease pressure. HortScience 2016, 51, 127–132. [Google Scholar] [CrossRef]
- Deng, H.; Achor, D.; Exteberria, E.; Yu, Q.; Du, D.; Stanton, D.; Liang, G.; Gmitter, F.G., Jr. Phloem regeneration is a mechanism for Huanglongbing-tolerance of “Bearss” lemon and “LB8-9” Sugar Belle® mandarin. Front. Plant Sci. 2019, 10, 277. [Google Scholar] [CrossRef] [PubMed]
- Castle, W.S. A career perspective on citrus rootstocks, their development, and commercialization. HortScience 2010, 45, 11–15. [Google Scholar] [CrossRef]
- Castle, W.S. HLB and ‘Tolerance’ as a Practical Matter in Choosing Rootstocks. Citrus Ind. 2016, 8, 1–3. [Google Scholar]
- Bowman, K.D.; Joubert, J. Citrus rootstocks. In The Genus Citrus; Woodhead Publishing: Cambridge, UK, 2020; pp. 105–127. [Google Scholar]
- Bowman, K.D.; Albrecht, U. Rootstock influences on health and growth following Candidatus Liberibacter asiaticus infection in young sweet orange trees. Agronomy 2020, 10, 1907. [Google Scholar] [CrossRef]
- Grosser, J.W.; Ollitrault, P.; Olivares-Fuster, O. Somatic hybridization in citrus: An effective tool to facilitate variety improvement. Vitr. Cell. Dev. Biol. Plant 2000, 36, 434–449. [Google Scholar] [CrossRef]
- Grosser, J.W.; Medina-Urrutia, V.; Ananthakrishnan, G.; Serrano, P. Building a replacement sour orange rootstock: Somatic hybridization of selected mandarin+ pummelo combinations. J. Am. Soc. Hortic. Sci. 2004, 129, 530–534. [Google Scholar] [CrossRef]
- Castle, W.S.; Bowman, K.D.; Grosser, J.; Ferrarezi, R.S.; Futch, S.H.; Rogers, S. Florida Citrus Rootstock Selection Guide, 4th ed.; Univeristy of Florida Cooperative Extension: Gainesville, FL, USA, 2019; p. 4. [Google Scholar]
- Allen, L.; Calvert, D.-R.N.A.; Cohen, M.; Pelosi, R.R.; Rogers, J.-R.A.; Stewart, E.H. The Florida citrus soil water atmosphere plant (SWAP) project: Final summary of cumulative yields and tree health. Proc. Fla. State Hortic. Soc. 2014, 127, 45–52. [Google Scholar]
- USDA. Web Soil Survey. Natural Resources Conservation Service. Available online: https://websoilsurvey.sc.egov.usda.gov/App/WebSoilSurvey.aspx. (accessed on 1 September 2022).
- Bauer, M.; Castle, W.; Boman, B.; Obreza, T. Field Guide to Soil Identification for Florida’s Citrus-Growing Regions; University of Florida, IFAS Cooperative Extension: Gainesville, FL, USA, 2007; pp. 1–24. [Google Scholar]
- Dewdney, M.M.; Vashisth, T.; Diepenbrock, L.M. 2019–2020 Florida Citrus Production Guide: Huanglongbing (Citrus Greening); University of Florida, IFAS Cooperative Extension: Gainesville, FL, USA, 2019. [Google Scholar]
- Kanissery, R.; McAvoy, C.E.; Burrow, J.D.; Futch, S.H.; Sellers, B.A.; Teems, S.S. Quick Reference Guide to Postemergence Herbicides for Citrus Weed Control: HS1410; University of Florida: Gainesville, FL, USA, 2021. [Google Scholar]
- Obreza, T.A.; Rouse, R.E. Fertilizer Effects on Early Growth and Yield of ‘Hamlin’ Orange Trees. HortScience 1993, 28, 111–114. [Google Scholar] [CrossRef]
- Slinski, S. Grower Field Trial Methods for the Evaluation of Bactericides against Huanglongbing (Citrus Greening); Citrus Research and Development Foundation: Lake Alfred, FL, USA, 2016. [Google Scholar]
- CoreTeam, R. R: A Language and Environment for Statistical Computing; R Core Team: Viena, Austria, 2013. [Google Scholar]
- Grosser, J.; Karp, D. Citrus rootstocks. Register of New Fruit and Nut Cultivars List 50. HortScience 2020, 55, 1181–1182. [Google Scholar] [CrossRef]
- Kunwar, S.; Grosser, J.; Gmitter, F.G.; Castle, W.S.; Albrecht, U. Field performance of ‘Hamlin’orange trees grown on various rootstocks in huanglongbing-endemic conditions. HortScience 2021, 56, 244–253. [Google Scholar] [CrossRef]
- Siebert, T.; Krueger, R.; Kahn, T.; Bash, J.; Vidalakis, G. Descriptions of new varieties recently distributed from the Citrus Clonal Protection Program. Citrograph 2010, 1, 20–26. [Google Scholar]
- Castle, W.S.; Phillips, R.L. Potentially Dwarfing Rootstocks for Florida Citrus. In Proceedings of the International Society of Citriculture, Orlando, FL, USA, 1–8 May 1977; Volume 2, pp. 558–561. [Google Scholar]
- Bowman, K.D.; Faulkner, L.; Kesinger, M. New citrus rootstocks released by USDA 2001–2010: Field performance and nursery characteristics. HortScience 2016, 51, 1208–1214. [Google Scholar] [CrossRef]
- Huang, M.; Roose, M.L.; Yu, Q.; Du, D.; Yu, Y.; Zhang, Y.; Deng, Z.; Stover, E.; Gmitter, F.G., Jr. Construction of high-density genetic maps and detection of QTLs associated with Huanglongbing tolerance in citrus. Front. Plant Sci. 2018, 9, 1694. [Google Scholar] [CrossRef] [PubMed]
- Folimonova, S.Y.; Achor, D.S. Early events of citrus greening (Huanglongbing) disease development at the ultrastructural level. Phytopathology 2010, 100, 949–958. [Google Scholar] [CrossRef]
- Canale, M.C.; Komada, K.; Lopes, J.R. Latency and incubation of ‘Candidatus Liberibacter asiaticus’ in citrus after vector inoculation. Trop. Plant Pathol. 2020, 45, 320–326. [Google Scholar] [CrossRef]
- Obreza, T.A.; Zekri, M.; Hanlon, E.A. Chapter 4. Soil and Leaf Tissue Testing: SL458/SS671, 2/2020. In Nutrition of Florida Citrus Trees; EDIS: Gainesville, FL, USA, 2020; pp. 13–22. [Google Scholar] [CrossRef]
- Obreza, T.; Zekri, M.; Futch, S.H. Chapter 3. General Soil Fertility and Citrus Tree Nutrition: SL457/SS670, 3/2020. In Nutrition of Florida Citrus Trees; EDIS: Gainesville, FL, USA, 2020; pp. 23–32. [Google Scholar] [CrossRef]
- Castle, W.; Krezdorn, A. Effect of citrus rootstocks on root distribution and leaf mineral content of ‘Orlando’ tangelo trees. J. Am. Soc. Hortic. Sci. 1975, 100, 1–4. [Google Scholar] [CrossRef]
- Sétamou, M.; Alabi, O.J.; Kunta, M.; Jifon, J.L.; da Graça, J.V. Enhanced acquisition rates of ‘Candidatus Liberibacter asiaticus’ by the Asian citrus psyllid (Hemiptera: Liviidae) in the presence of vegetative flush growth in citrus. J. Econ. Entomol. 2016, 109, 1973–1978. [Google Scholar] [CrossRef]

| Rootstock | Trial 1 | Trial 2 | Trial 3 | Origin | Parentage |
|---|---|---|---|---|---|
| UFR-1 | ● | ● | ● | UF | C. reticulata ‘Nova’ + C. maxima ‘Hirado Buntan’ × C. reticulata ‘Cleopatra’ + P. trifoliata ‘Argentine’ |
| UFR-2 | ● | ● | UF | C. reticulata ‘Nova’ + C. maxima ‘Hirado Buntan’ × C. reticulata ‘Cleopatra’ + P. trifoliata ‘Argentine’ | |
| UFR-4 | ● | ● | ● | UF | C. reticulata ‘Nova’ + C. maxima ‘Hirado Buntan’ × C. reticulata ‘Cleopatra’ + P. trifoliata ‘Argentine’ |
| UFR-5 | ● | ● | ● | UF | C. reticulata ‘Nova’ + C. maxima ‘Hirado Buntan’ × C. sinensis ‘Succari’ + P. trifoliata |
| UFR-15 | ● | ● | ● | UF | C. maxima ‘Hirado Buntan’ × C. reticulata ‘Cleopatra’ |
| UFR-16 | ● | ● | ● | UF | C. maxima ‘Hirado Buntan’ × C. reticulata ‘Shekwasha’ |
| UFR-17 | ● | ● | ● | UF | C. reticulata ‘Nova’ + C. maxima ‘Hirado Buntan’ × C. aurantium + ‘Carrizo’ citrange |
| WGFT+50-7 | ● | ● | UF | C. paradisi ‘White’ + P. trifoliata ’50-7’ | |
| 2247×2075-02-26 | ● | ● | ● | UF | C. reticulata ‘Nova’ + C. maxima ‘Hirado Buntan’ × C. reticulata ‘Cleopatra’ + ‘Swingle’ citrumelo |
| A+Volk×Orange 19-11-8 | ● | ● | ● | UF | C. amblycarpa + C. volkameriana × Orange 19 (C. reticulata ‘Nova’ + C máxima ‘Hirado Buntan’ × P. trifoliata ‘Argentine’) |
| 46×20-04-6 | ● | ● | ● | UF | C. maxima ‘Hirado Buntan’ × C. reticulata ‘Cleopatra’ |
| 2247×6070-02-2 | ● | ● | ● | UF | C. reticulata ‘Nova’ + C. máxima ‘Hirado Buntan’ × C. aurantium + P. trifoliata ‘Flying Dragon’ |
| 46×20-04-42 | ● | UF | C. maxima ‘Hirado Buntan’ × C. reticulata ‘Cleopatra’ | ||
| Orange 14 | ● | UF | C. reticulata ‘Nova’ + C. maxima ‘Hirado Buntan’ × C. reticulata ‘Cleopatra’ + P. trifoliata ‘Argentine’ | ||
| Orange 16 | ● | UF | C. reticulata ‘Nova’ + C. maxima ‘Hirado Buntan’ × C. reticulata ‘Cleopatra’ + P. trifoliata ‘Argentine’ | ||
| Sour orange | ● | ● | NH | C. aurantium | |
| Willits citrange | ● | ● | USDA | P. trifoliata × C. sinensis ‘Ruby’ | |
| Kuharske citrange | ● | ● | ● | USDA | Natural seedling variant of Carrizo |
| US-802 | ● | ● | ● | USDA | C. grandis Osbeck ‘Siamese’ × P. trifoliata |
| US-812 | ● | ● | ● | USDA | C. reticulata ‘Sunki’ × P. trifoliata ‘Benecke’ |
| US-897 | ● | ● | ● | USDA | C. reticulata ‘Cleopatra’ × P. trifoliata ‘Flying Dragon’ |
| US-942 | ● | ● | ● | USDA | C. reticulata ‘Sunki’ × P. trifoliata ‘Flying Dragon’ |
| Cunningham citrange | ● | ● | ● | USDA | P. trifoliata × C. sinensis |
| x-639 | ● | ● | ● | ARC-ITSC | C. reticulata ‘Cleopatra’ × P. trifoliata ‘Rubidoux’ |
| C-22 (‘Bitters’) | ● | ● | ● | UCR | C. reticulata ‘Sunki’ × P. trifoliata ‘Swingle’ |
| C-54 (‘Carpenter’) | ● | ● | ● | UCR | C. reticulata ‘Sunki’ × P. trifoliata ‘Swingle’ |
| Rootstock | September 2020 | September 2021 | ||
|---|---|---|---|---|
| Ct Value of CLas DNA | HLB Severity Index y | Ct Value of CLas DNA | HLB Severity Index y | |
| 2247×2075-02-26 | 38.0 ± 1.3 z | 0.0 ± 0.0 | 36.7 ± 0.9 | 1.2 ± 0.3 |
| 2247×6070-02-2 | 39.3 ± 0.3 | 0.0 ± 0.0 | 38.9 ± 0.7 | 0.5 ± 0.2 |
| 46×20-04-42 | 37.0 ± 1.5 | 0.0 ± 0.0 | 38.2 ± 0.8 | 1.4 ± 0.4 |
| 46×20-04-6 | 38.3 ± 1.5 | 0.0 ± 0.0 | 39.8 ± 0.1 | 0.6 ± 0.2 |
| A+Volk×Orange-19-11-8 | 37.3 ± 1.3 | 0.0 ± 0.0 | 37.6 ± 0.9 | 1.5 ± 0.4 |
| C-22 | 36.1 ± 1.3 | 0.0 ± 0.0 | 35.9 ± 0.9 | 1.8 ± 0.4 |
| C-54 | 39.7 ± 0.3 | 0.0 ± 0.0 | 37.6 ± 0.9 | 1.0 ± 0.5 |
| Cunningham citrange | 35.4 ± 1.3 | 0.0 ± 0.0 | 39.5 ± 0.4 | 0.9 ± 0.1 |
| Kuharske citrange | 36.7 ± 0.8 | 0.0 ± 0.0 | 38.9 ± 0.5 | 0.9 ± 0.3 |
| Orange 16 | 37.1 ± 0.8 | 0.0 ± 0.0 | 36.6 ± 1.1 | 0.6 ± 0.2 |
| Sour orange | 39.1 ± 0.4 | 0.0 ± 0.0 | 36.7 ± 0.9 | 1.3 ± 0.4 |
| UFR-1 | 36.5 ± 0.7 | 0.0 ± 0.0 | 37.2 ± 1.0 | 0.8 ± 0.2 |
| UFR-15 | 35.8 ± 1.7 | 0.0 ± 0.0 | 36.8 ± 1.3 | 0.7 ± 0.2 |
| UFR-16 | 36.9 ± 2.0 | 0.0 ± 0.0 | 35.8 ± 0.9 | 1.7 ± 0.4 |
| UFR-17 | 36.9 ± 1.2 | 0.0 ± 0.0 | 37.5 ± 0.8 | 0.7 ± 0.2 |
| UFR-4 | 36.6 ± 1.9 | 0.0 ± 0.0 | 36.0 ± 1.0 | 0.8 ± 0.3 |
| UFR-5 | 35.9 ± 1.5 | 0.0 ± 0.0 | 38.2 ± 0.7 | 0.9 ± 0.2 |
| US-802 | 37.4 ± 1.5 | 0.0 ± 0.0 | 37.3 ± 1.2 | 1.1 ± 0.2 |
| US-812 | 36.8 ± 1.8 | 0.0 ± 0.0 | 38.4 ± 0.7 | 1.5 ± 0.3 |
| US-897 | 35.8 ± 1.3 | 0.0 ± 0.0 | 36.8 ± 0.9 | 1.3 ± 0.2 |
| US-942 | 37.3 ± 0.7 | 0.0 ± 0.0 | 37.7 ± 1.1 | 1.4 ± 0.4 |
| WGFT+50-7 | 38.0 ± 1.1 | 0.0 ± 0.0 | 39.7 ± 0.3 | 0.4 ± 0.2 |
| x-639 | 35.8 ± 1.5 | 0.0 ± 0.0 | 39.9 ± 0.1 | 1.4 ± 0.1 |
| F value | 1.11 ns | - | 1.96 ns | 1.81 ns |
| Rootstock | September 2020 | September 2021 | ||
|---|---|---|---|---|
| Ct Value of CLas DNA | HLB Severity Index y | Ct Value of CLas DNA | HLB Severity Index y | |
| 2247×2075-02-26 | 36.3 ± 2.2 z | 0.0 ± 0.0 | 35.0 ± 1.3 | 0.9 ± 0.3 |
| Cunningham citrange | 39.1 ± 0.1 | 0.0 ± 0.0 | 36.7 ± 1.3 | 0.3 ± 0.3 |
| Orange 14 | 38.1 ± 1.0 | 0.0 ± 0.0 | 34.1 ± 0.8 | 0.8 ± 0.3 |
| UFR-1 | 38.5 ± 0.2 | 0.0 ± 0.0 | 36.1 ± 1.1 | 0.7 ± 0.2 |
| UFR-15 | 36.9 ± 1.1 | 0.0 ± 0.0 | 33.7 ± 0.8 | 1.0 ± 0.3 |
| UFR-4 | 34.8 ± 1.4 | 0.0 ± 0.0 | 37.0 ± 1.1 | 0.8 ± 0.4 |
| UFR-5 | 38.5 ± 0.1 | 0.0 ± 0.0 | 39.4 ± 0.6 | 0.8 ± 0.2 |
| US-812 | 38.9 ± 0.3 | 0.0 ± 0.0 | 35.9 ± 0.9 | 0.9 ± 0.2 |
| Willits citrange | 37.4 ± 1.6 | 0.0 ± 0.0 | 36.3 ± 1.0 | 0.2 ± 0.1 |
| x-639 | 35.3 ± 1.0 | 0.0 ± 0.0 | 34.4 ± 0.6 | 1.2 ± 0.3 |
| US-897 | 36.6 ± 2.1 | 0.0 ± 0.0 | 36.0 ± 1.1 | 1.1 ± 0.2 |
| US-942 | 38.7 ± 0.2 | 0.0 ± 0.0 | 34.6 ± 0.7 | 0.6 ± 0.2 |
| UFR-17 | 38.3 ± 1.2 | 0.0 ± 0.0 | 37.1 ± 1.2 | 0.8 ± 0.2 |
| C-54 | 39.2 ± 0.3 | 0.0 ± 0.0 | 37.7 ± 0.9 | 1.2 ± 0.2 |
| Kuharske citrange | 37.3 ± 1.4 | 0.0 ± 0.0 | 37.6 ± 0.9 | 0.8 ± 0.3 |
| 46×20-04-6 | 35.7 ± 1.7 | 0.0 ± 0.0 | 35.8 ± 1.5 | 1.1 ± 0.1 |
| A+Volk×Orange-19-11-8 | 37.4 ± 1.0 | 0.0 ± 0.0 | 33.7 ± 0.6 | 1.0 ± 0.3 |
| US-802 | 36.9 ± 1.2 | 0.0 ± 0.0 | 33.4 ± 0.8 | 0.9 ± 0.3 |
| C-22 | 35.2 ± 1.8 | 0.0 ± 0.0 | 33.2 ± 0.6 | 1.3 ± 0.3 |
| UFR-2 | 38.3 ± 0.5 | 0.0 ± 0.0 | 34.1 ± 0.8 | 1.0 ± 0.2 |
| 2247×6070-02-2 | 37.7 ± 1.3 | 0.0 ± 0.0 | 37.7 ± 1.1 | 0.5 ± 0.2 |
| UFR-16 | 36.5 ± 1.2 | 0.0 ± 0.0 | 35.1 ± 1.2 | 1.1 ± 0.4 |
| F value | 1.01 ns | - | 2.43 ns | 0.99 ns |
| Rootstock | September 2020 | September 2021 | ||
|---|---|---|---|---|
| Ct Value of CLas DNA | HLB Severity Index y | Ct Value of CLas DNA | HLB Severity Index y | |
| 2247×2075-02-26 | 36.5 ± 0.6 z, ab | 0.0 ± 0.0 | 34.9 ± 4.4 | 0.3 ± 0.3 |
| 2247×6070-02-2 | 35.1 ± 0.2 ab | 0.0 ± 0.0 | 34.7 ± 3.6 | 0.0 ± 0.0 |
| 46×20-04-6 | 32.8 ± 2.3 ab | 0.2 ± 0.0 | 32.8 ± 1.7 | 0.7 ± 0.1 |
| A+Volk×Orange-19-11-8 | 36.1 ± 1.5 ab | 0.0 ± 0.0 | 37.8 ± 4.4 | 0.3 ± 0.2 |
| C-22 | 33.8 ± 2.3 ab | 0.4 ± 0.0 | 38.0 ± 4.0 | 0.1 ± 0.1 |
| C-54 | 36.1 ± 1.9 ab | 0.1 ± 0.0 | 37.9 ± 3.9 | 0.3 ± 0.1 |
| Cunningham citrange | 38.1 ± 0.9 ab | 0.0 ± 0.0 | 35.3 ± 4.7 | 0.3 ± 0.3 |
| Kuharske citrange | 36.3 ± 0.5 ab | 0.3 ± 0.0 | 36.1 ± 5.6 | 0.0 ± 0.0 |
| UFR-1 | 39.4 ± 0.3 a | 0.0 ± 0.0 | 34.5 ± 4.1 | 0.0 ± 0.0 |
| UFR-15 | 34.7 ± 0.3 ab | 0.0 ± 0.0 | 40.0 ± 0.0 | 0.0 ± 0.0 |
| UFR-17 | 35.9 ± 1.1 ab | 0.1 ± 0.0 | 36.3 ± 4.0 | 0.3 ± 0.3 |
| UFR-2 | 38.3 ± 0.2 a | 0.2 ± 0.0 | 38.8 ± 2.6 | 0.4 ± 0.4 |
| US-802 | 33.8 ± 1.7 ab | 0.0 ± 0.0 | 39.3 ± 1.4 | 0.2 ± 0.2 |
| US-812 | 33.9 ± 1.7 ab | 0.1 ± 0.0 | 38.5 ± 3.3 | 0.1 ± 0.1 |
| WGFT+50-7 | 34.4 ± 1.6 ab | 0.0 ± 0.0 | 36.3 ± 4.1 | 0.0 ± 0.0 |
| x-639 | 34.6 ± 2.7 ab | 0.0 ± 0.0 | 34.3 ± 3.5 | 0.2 ± 0.1 |
| UFR-4 | 32.4 ± 1.4 b | 0.2 ± 0.0 | 34.1 ± 3.7 | 0.1 ± 0.1 |
| UFR-5 | 33.0 ± 2.2 ab | 0.2 ± 0.0 | 37.8 ± 3.4 | 0.2 ± 0.1 |
| UFR-16 | 35.0 ± 0.2 ab | 0.1 ± 0.0 | 34.6 ± 3.8 | 0.0 ± 0.0 |
| US-897 | 36.0 ± 0.7 ab | 0.7 ± 0.0 | 36.3 ± 3.4 | 0.1 ± 0.1 |
| US-942 | 35.8 ± 1.4 ab | 0.0 ± 0.0 | 38.1 ± 3.4 | 0.1 ± 0.1 |
| F value | 2.2 ** | 1.60 ns | 1.20 ns | 1.29 ns |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zapien-Macias, J.M.; Ferrarezi, R.S.; Spyke, P.D.; Castle, W.S.; Gmitter, F.G., Jr.; Grosser, J.W.; Rossi, L. Early Performance of Recently Released Rootstocks with Grapefruit, Navel Orange, and Mandarin Scions under Endemic Huanglongbing Conditions in Florida. Horticulturae 2022, 8, 1027. https://doi.org/10.3390/horticulturae8111027
Zapien-Macias JM, Ferrarezi RS, Spyke PD, Castle WS, Gmitter FG Jr., Grosser JW, Rossi L. Early Performance of Recently Released Rootstocks with Grapefruit, Navel Orange, and Mandarin Scions under Endemic Huanglongbing Conditions in Florida. Horticulturae. 2022; 8(11):1027. https://doi.org/10.3390/horticulturae8111027
Chicago/Turabian StyleZapien-Macias, J. Martin, Rhuanito Soranz Ferrarezi, Peter D. Spyke, William S. Castle, Frederick G. Gmitter, Jr., Jude W. Grosser, and Lorenzo Rossi. 2022. "Early Performance of Recently Released Rootstocks with Grapefruit, Navel Orange, and Mandarin Scions under Endemic Huanglongbing Conditions in Florida" Horticulturae 8, no. 11: 1027. https://doi.org/10.3390/horticulturae8111027
APA StyleZapien-Macias, J. M., Ferrarezi, R. S., Spyke, P. D., Castle, W. S., Gmitter, F. G., Jr., Grosser, J. W., & Rossi, L. (2022). Early Performance of Recently Released Rootstocks with Grapefruit, Navel Orange, and Mandarin Scions under Endemic Huanglongbing Conditions in Florida. Horticulturae, 8(11), 1027. https://doi.org/10.3390/horticulturae8111027









