Fermentation as a Promising Tool to Valorize Rice-Milling Waste into Bio-Products Active against Root-Rot-Associated Pathogens for Improved Horticultural Plant Growth
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Lactic Acid Bacteria and Enzymes
2.3. Pre-Treatment of Rice Bran
2.4. Plants and Potting Procedure
2.5. Evaluation of Plant Growth Performance
2.6. Indicatory Microorganisms
2.7. Determination of Antifungal Activity
2.8. UFLC Analysis of Amino Acids
2.9. Analysis of Phenolic Compounds
2.10. UHPLC Analysis of Organic Acids
2.11. Chlorophyll Determination
2.12. Statistical Analysis
3. Results
3.1. Characterization of Rice Bran Material
3.2. The Impact of Rice Bran Pre-Treatments on Amino Acid Profile
3.3. Antifungal Activity of Pre-Treated Rice Bran Extracts
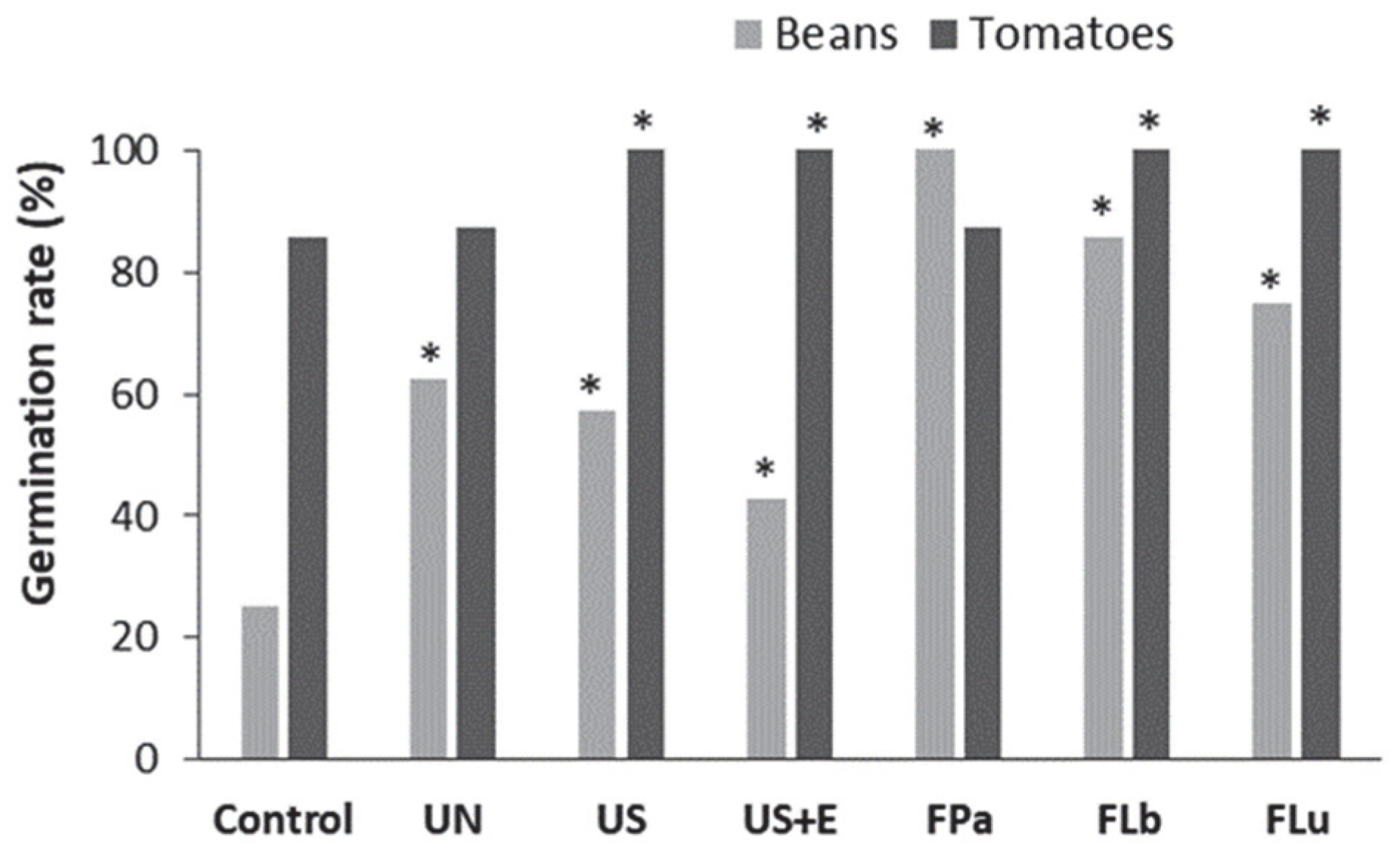
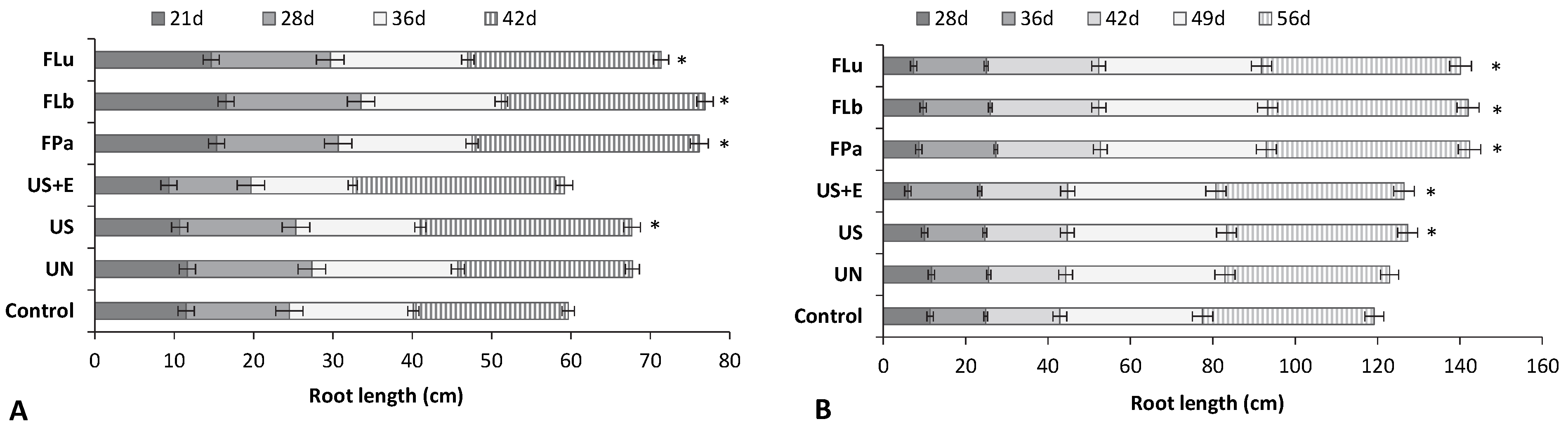
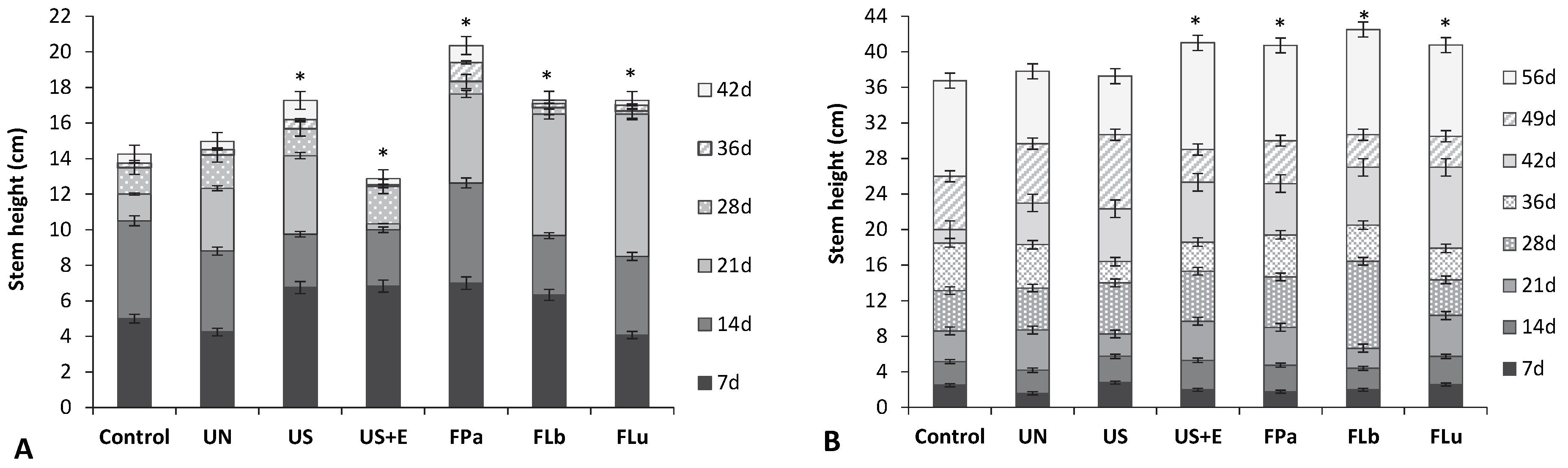
3.4. The Effect of Rice Bran Additives on the Germination Rate and the Growth of Beans and Tomatoes
3.5. The Effect of Rice Bran Additives on Chlorophyll Formation in Plants
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tilman, D.; Balzer, C.; Hill, J.; Befort, B.L. Global food demand and the sustainable intensification of agriculture. Proc. Natl. Acad. Sci. USA 2011, 108, 20260–20264. [Google Scholar] [CrossRef] [PubMed]
- Alauddin, M.; Mohsin, G.M.; Ali, A.H.M.Z.; Rahman, M.K. Interactive effects of rice bran compost and chemical fertilizers on macronutrients, oil and protein content in sunflower (Helianthus annuus L.). Int. J. Agric. Res. Innov. Technol. 2020, 10, 91–99. [Google Scholar] [CrossRef]
- Alauddin, M.; Jahidul, I.; Shirakawa, H.; Koseki, T.; Ardiansyah; Komai, M. Rice Bran as a Functional Food: An Overview of the Conversion of Rice Bran into a Superfood/Functional Food. In Superfood and Functional Food; Waisundara, V.Y., Shiomi, N., Eds.; Intech Open: London, UK, 2017; pp. 291–305. [Google Scholar]
- Vaitkeviciene, R.; Zadeike, D.; Gaizauskaite, Z.; Valentaviciute, K.; Marksa, M.; Mazdzieriene, R.; Bartkiene, E.; Lele, V.; Juodeikiene, G.; Jakstas, V. Functionalisation of rice bran assisted by ultrasonication and fermentation for the production of rice bran-lingonberry pulp-based probiotic nutraceutical. Int. J. Food Sci. Technol. 2022, 57, 1462–1472. [Google Scholar] [CrossRef]
- Vallabha, V.S.; Indira, T.N.; Lakshmi, A.J.; Radha, C.; Tiku, P.K. Enzymatic process of rice bran: A stabilized functional food with nutraceuticals and nutrients. J. Food Sci. Technol. 2015, 52, 8252–8259. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, A.C.; da Silva Graça, C.; Chiattoni, L.M.; Massarolo, K.C.; Duarte, F.A.; de Las Sallas Mellado, M.; de Souza Soares, L.A. Fermentation process in the availability of nutrients in rice bran. J. Microbiol. Biotechnol. 2017, 6, 45–52. [Google Scholar]
- Öpik, H.; Rolfe, S. Plant Growth Hormones. In The Physiology of Flowering Plants; Öpik, H., Rolfe, S., Willis, A., Eds.; Cambridge University Press: Cambridge, UK; London, UK, 2005; pp. 177–194. [Google Scholar]
- Zadeike, D.; Vaitkeviciene, R.; Degutyte, R.; Bendoraitiene, J.; Rukuiziene, Z.; Cernauskas, D.; Svazas, M.; Juodeikiene, G. A comparative study on the structural and functional properties of water-soluble and alkali-soluble dietary fibres from rice bran after hot-water, ultrasound, cellulase hydrolysis and combined pre-treatments. Int. Food Sci. Technol. 2022, 57, 1137–1149. [Google Scholar] [CrossRef]
- Wang, T.; Chen, X.; Wang, W.; Wang, L.; Jiang, L.; Yu, D.; Xie, F. Effect of ultrasound on the properties of rice bran protein and its chlorogenic acid complex. Ultrason. Sonochem. 2021, 79, 105758. [Google Scholar] [CrossRef]
- Digaitiene, A.; Hansen, A.S.; Juodeikiene, G.; Eidukonyte, D.; Josephsen, J. Lactic acid bacteria isolated from rye sourdoughs produce bacteriocin-like inhibitory substances active against Bacillus subtilis and fungi. J. Appl. Microbiol. 2012, 112, 732–742. [Google Scholar] [CrossRef]
- Vaitkeviciene, R.; Bendoraitiene, J.; Degutyte, R.; Svazas, M.; Zadeike, D. Optimization of the sustainable production of resistant starch in rice bran and evaluation of its physicochemical and technological properties. Polymers 2022, 14, 3662. [Google Scholar] [CrossRef]
- Li, H.; Xu, M.; Yao, X.; Wen, Y.; Lu, S.; Wang, J.; Sun, B. The promoted hydrolysis effect of cellulase with ultrasound treatment is reflected on the sonicated rather than native brown rice. Ultrason. Sonochem. 2022, 83, 105920. [Google Scholar] [CrossRef]
- Singh, V.K.; Singh, A.K.; Kumar, A. Disease management of tomato through PGPB: Current trends and future perspective. 3 Biotech 2017, 7, 255. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.K.; Singh, H.B.; Upadhyay, R.S. Role of fusaric acid in the development of ‘Fusarium wilt’ symptoms in tomato: Physiological, biochemical and proteomic perspectives. Plant Physiol. Biochem. 2017, 118, 320–332. [Google Scholar] [CrossRef] [PubMed]
- Taheri, A.E.; Chatterton, S.; Foroud, N.A.; Gossen, B.D.; McLaren, D.L. Identification and community dynamics of fungi associated with root, crown, and foot rot of field pea in western. Can. Eur. J. Plant Pathol. 2017, 147, 489–500. [Google Scholar] [CrossRef]
- Lamont, J.R.; Wilkins, O.; Bywater-Ekegärd, M.; Smith, D.L. From yogurt to yield: Potential applications of lactic acid bacteria in plant production. Soil Biol. Biochem. 2017, 111, 1–9. [Google Scholar] [CrossRef]
- Garsa, A.K.; Kumariya, R.; Sood, S.K.; Kumar, A.; Kapila, S. Bacteriocin production and different strategies for their recovery and purification. Probiotics Antimicrob. Proteins 2014, 6, 47–58. [Google Scholar] [CrossRef]
- Marco, M.L.; Heeney, D.; Binda, S.; Cifelli, C.J.; Cotter, P.D.; Foligné, B.; Gänzle, M.; Kort, R.; Pasin, G.; Pihlanto, A.; et al. Health benefits of fermented foods: Microbiota and beyond. Curr. Opin. Biotechnol. 2017, 44, 94–102. [Google Scholar] [CrossRef]
- Lutz, M.P.; Michel, V.; Martinez, C.; Camps, C. Lactic acid bacteria as biocontrol agents of soil-borne pathogens. IOBC-WPRS Bull. 2012, 78, 285–288. [Google Scholar]
- Vaitkevičienė, R.; Žadeikė, D.; Bartkienė, E.; Krunglevičiūtė, V.; Baliukoniene, V.; Supronienė, S.; Juodeikienė, G. The use of rice polish medium for the evaluation of antifungal activity of lactic acid bacteria. Zemdirb. Agric. 2019, 106, 59–64. [Google Scholar] [CrossRef]
- Jukonyte, R.; Zadeike, D.; Bartkiene, E.; Lele, V.; Juodeikiene, G.; Cernauskas, D.; Suproniene, S. A potential of brown rice polish as a substrate for the lactic acid and bioactive compounds production by the lactic acid bacteria newly isolated from cereal-based fermented products. LWT Food Sci. Technol. 2018, 97, 323–331. [Google Scholar] [CrossRef]
- Ranal, M.A.; de Santana, D.G. How and why to measure the germination process? Braz. J. Bot. 2006, 29, 1–11. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods Enzymol. 1999, 299, 152–178. [Google Scholar]
- Costache, M.A.; Campeanu, G.; Neata, G. Studies concerning the extraction of chlorophyll and total carotenoids from vegetables. Rom. Biotechnol. Lett. 2012, 17, 7702–7708. [Google Scholar]
- Jarboui, R.; Dhouib, B.; Ammar, E. Effect of food waste compost (FWC) and its non-aerated fermented extract (NFCE) on seeds germination and plant growth. Open J. Soil Sci. 2021, 11, 122–138. [Google Scholar] [CrossRef]
- Nazari, B.; Mohammadifar, M.A.; Shojaee-Aliabadi, S.; Feizollahi, E.; Mirmoghtadaie, L. Effect of ultrasound treatments on functional properties and structure of millet protein concentrate. Ultrason. Sonochem. 2018, 41, 382–388. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, M.; Hoshina, M.; Tanabe, S.; Higuchi, Y.; Sakai, K.; Ohtsubo, S.; Hoshino, K.; Tanaka, T. Production of L-lactic acid by simultaneous saccharification and fermentation using unsterilized defatted rice bran as a carbon source and nutrient components. Food Sci. Technol. Res. 2005, 11, 400–406. [Google Scholar] [CrossRef][Green Version]
- Wang, Y.; Yang, Z.; Qin, P.; Tan, T. Fermentative L-(+)-lactic acid production from defatted rice bran. RSC Adv. 2014, 4, 8907–8913. [Google Scholar] [CrossRef]
- Nnam, N.M.; Obiakor, P.N. Effect of fermentation on the nutrient and antinutrient composition of baobab (Adansonia digitata) seeds and rice (Oryza sativa) grains. Ecol. Food Nutr. 2003, 42, 265–277. [Google Scholar] [CrossRef]
- Oladeji, B.S.; Irinkoyenikan, O.A.; Akanbi, C.T.; Gbadamosi, S.O. Effect of fermentation on the physicochemical properties, pasting profile and sensory scores of normal endosperm maize and quality protein maize flours. Int. Food Res. J. 2018, 25, 1100–1108. [Google Scholar]
- dos Oliveira, M.S.; Feddern, V.; Kupski, L.; Cipolatti, E.P.; Badiale-Furlong, E.; de Souza-Soares, L.A. Physico-chemical characterization of fermented rice bran biomass. CyTA-J. Food 2010, 8, 229–236. [Google Scholar] [CrossRef]
- Aguirre, M.; Kiegle, E.; Leo, G.; Ezquer, I. Carbohydrate reserves and seed development: An overview. Plant Reprod. 2018, 3, 263–290. [Google Scholar] [CrossRef]
- Tanaka, T.; Hoshina, M.; Tanabe, S.; Sakai, K.; Ohtsubo, S.; Taniguchi, M. Production of D-lactic acid from defatted rice bran by simultaneous saccharification and fermentation. Bioresour. Technol. 2006, 97, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Shao, Y.; Xu, F.; Sun, X.; Bao, J.; Beta, T. Phenolic acids, anthocyanins, and antioxidant capacity in rice (Oryza sativa L.) grains at four stages of development after flowering. Food Chem. 2014, 143, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Iftikhar, M.; Zhang, H.; Iftikhar, A.; Raza, A.; Begum, N.; Tahamina, A.; Syed, H.; Khan, M.; Wang, J. Study on optimization of ultrasonic assisted extraction of phenolic compounds from rye bran. LWT Food Sci. Technol. 2020, 134, 110243. [Google Scholar] [CrossRef]
- Nisa, K.; Rosyida, V.; Nurhayati, S.; Indrianingsih, A.; Darsih, C.; Apriyana, W. Total phenolic contents and antioxidant activity of rice bran fermented with lactic acid bacteria. IOP Conf. Ser. Earth Environ. Sci. 2019, 251, 0120202019. [Google Scholar] [CrossRef]
- Razak, D.L.A.; Rashid, N.Y.A.; Jamaluddin, A.; Sharifudin, S.A.; Long, K. Enhancement of phenolic acid content and antioxidant activity of rice bran fermented with Rhizopus oligosporus and Monascus purpureus. Biocatal. Agric. Biotechnol. 2015, 4, 33–38. [Google Scholar] [CrossRef]
- Borges, A.; Ferreira, C.; Saavedra, M.J.; Simões, M. Antibacterial activity and mode of action of ferulic and gallic acids against pathogenic bacteria. Microb. Drug Resist. 2013, 19, 256–265. [Google Scholar] [CrossRef]
- Padayachee, A.; Netzel, G.; Netzel, M.; Day, L.; Zabaras, D.; Mikkelsen, D.; Gidley, M. Binding of polyphenols to plant cell wall analogues-part 2: Phenolic acids. Food Chem. 2012, 135, 2287–2292. [Google Scholar] [CrossRef]
- Coniglio, R.O.; Díaz, G.V.; Barua, R.C.; Albertó, E.; Zapata, P.D. Enzyme-assisted extraction of phenolic compounds and proteins from sugarcane bagasse using a low-cost cocktail from Auricularia fuscosuccinea. Int. J. Food Sci. Technol. 2022, 57, 1114–1121. [Google Scholar] [CrossRef]
- de la Rosa, L.A.; Moreno-Escamilla, J.O.; Rodrigo-García, J.; Alvarez-Parrilla, E. Phenolic Compounds. In Postharvest Physiology and Biochemistry of Fruits and Vegetables; Yahia, E.M., Ed.; Woodhead Publishing: London, UK, 2019; pp. 253–271. [Google Scholar]
- Liu, L.; Wen, W.; Zhang, R.; Wei, Z.; Deng, Y.; Xiao, J.; Zhang, M. Complex enzyme hydrolysis releases antioxidative phenolics from rice bran. Food Chem. 2017, 214, 1–8. [Google Scholar] [CrossRef]
- Lattanzio, V.; Veronica, M.; Lattanzio, T.; Cardinali, A. Role of Phenolics in the Resistance Mechanisms of Plants Against Fungal Pathogens and Insects. In Phytochemistry: Advances in Research; Imperato, F., Ed.; Research Signpost: Kerala, India, 2006; pp. 23–67. [Google Scholar]
- Li, Z.J.; Liu, M.; Dawuti, G.; Dou, Q.; Ma, Y.; Liu, H.G.; Aibai, S. Antifungal activity of gallic acid in vitro and in vivo. Phytother. Res. 2017, 19, 1039–1045. [Google Scholar] [CrossRef]
- Kan, C.-C.; Chung, T.-Y.; Juo, Y.-A.; Hsieh, M.-H. Glutamine rapidly induces the expression of key transcription factor genes involved in nitrogen and stress responses in rice roots. BMC Genom. 2015, 16, 731. [Google Scholar] [CrossRef]
- Gaufichon, L.; Reisdorf-Cren, M.; Rothstein, S.J.; Chardon, F.; Suzuki, A. Biological functions of asparagine synthetase in plants. Plant Sci. 2010, 179, 141–153. [Google Scholar] [CrossRef]
- Killiny, N.; Hijaz, F. Amino acids implicated in plant defense are higher in Candidatus Liberibacter asiaticus-tolerant citrus varieties. Plant Signal. Behav. 2016, 11, 1171449. [Google Scholar] [CrossRef]
- Adebiyi, J.A.; Obadina, A.O.; Adebo, O.A.; Kayitesi, E. Comparison of nutritional quality and sensory acceptability of biscuits obtained from native, fermented, and malted pearl millet (Pennisetum glaucum) flour. Food Chem. 2017, 233, 210–217. [Google Scholar] [CrossRef]
- Osman, M.A. Effect of traditional fermentation process on the nutrient and antinutrient contents of pearl millet during preparation of Lohoh. J. Saudi Soc. Agric. Sci. 2011, 10, 1–6. [Google Scholar] [CrossRef]
- Meinlschmidt, P.; Ueberham, E.; Lehmann, J.; Schweiggert-Weisz, U.; Eisner, P. Immunoreactivity, sensory and physicochemical properties of fermented soy protein isolate. Food Chem. 2016, 205, 229–238. [Google Scholar] [CrossRef]
- Aguirre, L.; Garro, M.S.; de Giori, G.S. Enzymatic hydrolysis of soybean protein using lactic acid bacteria. Food Chem. 2008, 111, 976–982. [Google Scholar] [CrossRef]
- Lavermicocca, P.; Valerio, F.; Evidente, A.; Lazzaroni, S.; Corsetti, A.; Gobbeti, M. Purifcation and characterization of novel antifungal compounds from the sourdough Lactobacillus plantarum Strain 21B. Appl. Environ. Microbiol. 2000, 66, 4084–4090. [Google Scholar] [CrossRef]
- Corsetti, A.; Gobbetti, M.; Rossi, J.; Damiani, P. Antimould activity of sourdough lactic acid bacteria: Identification of a mixture of organic acids produced by Lactobacillus sanfrancisco CB1. Appl. Microbiol. Biotechnol. 1998, 50, 253–256. [Google Scholar] [CrossRef]
- Ogunremi, O.R.; Banwo, K.; Sanni, A.I. Starter-culture to improve the quality of cereal-based fermented foods: Trends in selection and application. Curr. Opin. Food Sci. 2017, 13, 38–43. [Google Scholar] [CrossRef]
- Zadeike, D.; Vaitkeviciene, R.; Bartkevics, V.; Bogdanova, E.; Bartkiene, E.; Lele, V.; Juodeikiene, G.; Cernauskas, D.; Valatkeviciene, Z. The expedient application of microbial fermentation after whole-wheat milling and fractionation to mitigate mycotoxins in wheat-based products. LWT Food Sci. Technol. 2021, 137, 110440. [Google Scholar] [CrossRef]
- Mutale-Joan, C.; Redouane, B.; Najib, E.; Yassine, K.; Lyamlouli, K.; Laila, S.; Zeroual, Y.; Hicham, A. Screening of microalgae liquid extracts for their bio stimulant properties on plant growth, nutrient uptake and metabolite profile of Solanum lycopersicum L. Sci. Rep. 2020, 10, 2820. [Google Scholar] [CrossRef] [PubMed]
- Jędrczak, A. Composting and fermentation of biowaste—Advantages and disadvantages of processes. Civ. Environ. Eng. Reports 2018, 28, 71–87. [Google Scholar] [CrossRef]
- Popko, M.; Michalak, I.; Wilk, R.; Gramza, M.; Chojnacka, K.; Górecki, H. Effect of the new plant growth biostimulants based on amino acids on yield and grain quality of winter wheat. Molecules 2018, 23, 470. [Google Scholar] [CrossRef]
- Colla, G.; Rouphael, Y.; Canaguier, R.; Svecova, E.; Cardarelli, M. Biostimulant action of a plant-derived protein hydrolysate produced through enzymatic hydrolysis. Front. Plant Sci. 2014, 5, 448. [Google Scholar] [CrossRef]
- Kisiriko, M.; Anastasiadi, M.; Terry, L.A.; Yasri, A.; Beale, M.H.; Ward, J.L. Phenolics from medicinal and aromatic plants: Characterisation and potential as biostimulants and bioprotectants. Molecules 2021, 26, 6343. [Google Scholar] [CrossRef]
| Parameters | RB Samples | |||||
|---|---|---|---|---|---|---|
| UN | US | US+E | FPa | FLb | FLu | |
| pH | 6.48 ± 0.02 a | 6.50 ± 0.01 a | 6.70 ± 0.01 a | 3.67 ± 0.02 b | 3.58 ± 0.00 bc | 3.89 ± 0.02 b |
| LAB count | – | – | – | 8.21 ± 0.23 ab | 8.68 ± 0.19 a | 8.15 ± 0.11 b |
| Protein | 16.05 ± 0.09 a | 16.43 ± 0.29 a | 15.99 ± 0.02 a | 14.39 ± 0.27 b | 14.25 ± 0.20 b | 13.68 ± 0.03 b |
| Total sugars | 3.89 ± 0.01 e | 11.25 ± 0.02 d | 19.98 ± 0.03 a | 15.42 ± 0.02 c | 17.66 ± 0.03 b | 14.98 ± 0.02 c |
| Total lactic acid | – | – | – | 21.49 ± 0.37 b | 23.42 ± 0.27 a | 19.66 ± 0.36 c |
| L-lactic acid | – | – | – | 8.40 ± 0.27 c | 12.53 ± 0.25 a | 10.43 ± 0.21 b |
| D-lactic acid | – | – | – | 13.09 ± 0.15 a | 10.89 ± 0.17 b | 9.23 ± 0.22 c |
| L/D ratio | – | – | – | 0.64 ± 0.03 b | 1.15 ± 0.06 a | 1.13 ± 0.02 a |
| Propionic acid | – | – | – | 0.82 ± 0.13 b | 0.40 ± 0.22 c | 1.72 ± 0.23 a |
| Acetic acid | – | – | – | 11.49 ± 0.21 b | 14.24 ± 0.42 a | 10.54 ± 0.32 c |
| TPC | 0.61 ± 0.14 d | 1.17 ± 0.06 f | 1.28 ± 0.05 e | 0.82 ± 0.12 c | 1.04 ± 0.46 a | 0.96 ± 0.41 b |
| RB Sample | Essential Amino Acids | Total EAA | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| VAL | ILE | LEU | TRP | LYS | MET | PHE | THR | HIS | ||
| UN | 1.41 | 3.50 | 5.31 | 3.65 | 5.10 | 1.73 | 4.15 | 4.85 | 2.88 | 32.58 ± 0.42 a |
| US | 1.12 | 3.07 | 4.85 | 2.89 | 4.59 | 1.23 | 4.14 | 3.98 | 2.82 | 28.69 ± 0.74 b |
| US+E | 0.75 | 3.58 | 4.49 | 1.99 | 4.75 | 2.15 | 4.12 | 4.16 | 2.33 | 28.32 ± 0.76 b |
| FPa | 4.62 | 1.60 | 6.14 | 3.80 | 5.53 | 3.85 | 0.40 | 1.66 | – | 27.60 ± 0.23 c |
| FLb | 1.52 | 1.64 | 6.52 | 4.49 | 7.33 | 2.27 | 0.88 | 1.95 | 0.40 | 26.99 ± 0.84 c |
| FLu | 2.50 | 1.10 | 6.14 | 5.99 | 7.37 | – | 0.77 | 1.70 | 0.13 | 25.70 ± 0.42 d |
| Rb Sample | NEAA | Total NEAA | CEAA | Total CEAA | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| ALA | ASP | SER | GLU | CYS | PRO | GLY | TYR | ARG | |||
| UN | 4.71 | 9.20 | 5.40 | 8.42 | 27.73 ± 0.36 d | 5.71 | 3.60 | 2.32 | 2.47 | 10.08 | 24.18 ± 0.27 d |
| US | 3.30 | 3.34 | 4.94 | 6.31 | 17.89 ± 0.01 f | 5.20 | 3.38 | 1.60 | 0.66 | 8.80 | 19.64 ± 0.20 f |
| US+E | 3.96 | 3.28 | 4.54 | 7.50 | 19.28 ± 0.42 e | 5.51 | 3.31 | 1.81 | 0.93 | 9.49 | 21.05 ± 0.75 e |
| FPa | 13.14 | 8.53 | 7.23 | 5.03 | 33.93 ± 0.19 b | 23.98 | 0.95 | 3.12 | 0.68 | 1.38 | 30.11 ± 0.15 b |
| FLb | 6.49 | 12.87 | 11.34 | 14.87 | 45.57 ± 0.61 a | 19.12 | 2.03 | 4.80 | 1.28 | 1.36 | 28.59 ± 0.22 c |
| FLu | 9.87 | 6.09 | 9.90 | 3.38 | 29.24 ± 0.25 c | 21.91 | 1.89 | 5.31 | 1.90 | 2.17 | 33.18 ± 0.45 a |
| Pathogens | UN | US | US+E | FPa | FLb | FLu | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 24 h | 48 h | 24 h | 48 h | 24 h | 48 h | ||||||
| Altenaria alternata | – | – | 9 * | 9 * | 10 * | 8 * | 19 * | 9 * | 12 * | ||
| Aspergillus terreus | 8 | 8 | 9 | 13 * | 18 * | 8 | 9 | 12 * | 20 * | ||
| Aspergillus versicolor | – | – | – | – | 18 * | 22 * | 32 * | 9 * | 18 * | ||
| Cladosporium herbarum | – | – | 11 * | 10 * | 10 * | 8 * | 9 * | 8 * | 12 * | ||
| Fusarium avenaceum | 10 | 10 | 11 | – | – | 23 * | 29 * | 18 * | 20 * | ||
| Fusarium culmorum | 9 | 9 | 10 | – | – | 19 * | 20 * | – | 11 | ||
| Fusarium graminearum | 11 | 10 | 10 | – | – | 21 * | 33 * | 20 * | 27 * | ||
| Fusarium nivale | 9 | – | 9 | 8 | 19 * | 10 * | 11 * | 10 * | 18 * | ||
| Mucor mucedo | 8 | 19 * | 10 * | 9 | 9 | 9 | 18 * | 11 * | 19 * | ||
| Penicillium palitans | – | – | – | 10 * | 10 * | 19 * | 23 * | 11 * | 18 * | ||
| Penicillium spinulosum | 8 | 9 | 9 | 13 * | 14 * | 14 * | 19 * | 11 * | 19 * | ||
| Penicillium viridicatum | 16 | – | – | 8 | 19 * | 9 | 19 * | 11 | 19 * | ||
| Pythium volutum | 9 | 9 | 9 | – | – | 22 * | 24 * | 24 * | 29 * | ||
| Rhizopus oryzae | 19 | – | – | 9 | 12 | – | – | 19 | 29 * | ||
| Sclerotinia sclerotiorum | – | 10 * | 10 * | – | – | 19 * | 22 * | – | – | ||
| Verticillium wilt | – | – | 14 * | 8 * | 18 * | 16 * | 20 * | – | 23 * | ||
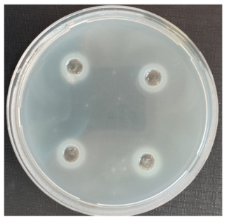 |  |  | |||||||||
| FLb against S. sclerotiorum | FLu against P. volutum | FLu against V. wilt | |||||||||
 |  | 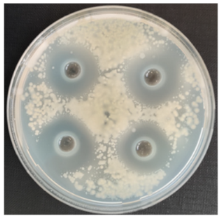 | |||||||||
| FLb against A. alternata | FLb against A. versicolor | FLu against R. oryzae | |||||||||
| Substrate | Bean | Tomato | ||
|---|---|---|---|---|
| Chl A | Chl B | Chl A | Chl B | |
| PGS | 17.14 ± 0.02 e | 6.46 ± 0.01 e | 18.23 ± 0.54 e | 7.00 ± 0.51 d |
| PGS with: | ||||
| RBUN | 22.19 ± 0.03 c | 9.26 ± 0.11 b | 21.27 ± 0.21 d | 8.31 ± 0.31 c |
| RBUS | 23.94 ± 0.09 b | 10.63 ± 0.24 a | 15.62 ± 0.01 f | 5.95 ± 0.26 e |
| RBUS+E | 14.76 ± 0.03 f | 7.32 ± 0.01 d | 27.12 ± 0.31 a | 10.92 ± 0.90 a |
| RBFPa | 25.49 ± 0.06 a | 8.18 ± 0.93 c | 26.07 ± 0.21 b | 9.37 ± 0.03 b |
| RBFLb | 19.19 ± 0.12 d | 6.69 ± 0.52 e | 23.93 ± 0.21 c | 9.10 ± 0.08 b |
| RBFLu | 24.10 ± 0.31 b | 10.47 ± 0.38 a | 19.05 ± 0.04 e | 6.84 ± 0.17 d |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vaitkeviciene, R.; Burbulis, N.; Masiene, R.; Zvirdauskiene, R.; Jakstas, V.; Damasius, J.; Zadeike, D. Fermentation as a Promising Tool to Valorize Rice-Milling Waste into Bio-Products Active against Root-Rot-Associated Pathogens for Improved Horticultural Plant Growth. Fermentation 2022, 8, 716. https://doi.org/10.3390/fermentation8120716
Vaitkeviciene R, Burbulis N, Masiene R, Zvirdauskiene R, Jakstas V, Damasius J, Zadeike D. Fermentation as a Promising Tool to Valorize Rice-Milling Waste into Bio-Products Active against Root-Rot-Associated Pathogens for Improved Horticultural Plant Growth. Fermentation. 2022; 8(12):716. https://doi.org/10.3390/fermentation8120716
Chicago/Turabian StyleVaitkeviciene, Ruta, Natalija Burbulis, Ramune Masiene, Renata Zvirdauskiene, Valdas Jakstas, Jonas Damasius, and Daiva Zadeike. 2022. "Fermentation as a Promising Tool to Valorize Rice-Milling Waste into Bio-Products Active against Root-Rot-Associated Pathogens for Improved Horticultural Plant Growth" Fermentation 8, no. 12: 716. https://doi.org/10.3390/fermentation8120716
APA StyleVaitkeviciene, R., Burbulis, N., Masiene, R., Zvirdauskiene, R., Jakstas, V., Damasius, J., & Zadeike, D. (2022). Fermentation as a Promising Tool to Valorize Rice-Milling Waste into Bio-Products Active against Root-Rot-Associated Pathogens for Improved Horticultural Plant Growth. Fermentation, 8(12), 716. https://doi.org/10.3390/fermentation8120716









