Degradation of Anti-Nutritional Factors in Maize Gluten Feed by Fermentation with Bacillus subtilis: A Focused Study on Optimizing Fermentation Conditions
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Fermentation of Maize Gluten Feed
2.3. Determination of Phytic Acid
2.4. Determination of Water-Unextractable Arabinoxylan
2.5. Optimization Experimental Design
2.5.1. Single-Factor Test
2.5.2. Plackett–Burman Design
2.5.3. Central Composite Design
2.6. Enzymatic Activity Analysis
2.6.1. Phytase Activity Assay
2.6.2. Xylanase Activity Assay
2.6.3. Cellulase Activity Assay
2.6.4. Protease Activity Assay
2.7. Protein Nutritional Analysis
2.8. In Vitro Minerals Digestion
2.9. Statistical Analysis
3. Results and Discussion
3.1. Effects of Independent Factors on PA and WU-AX
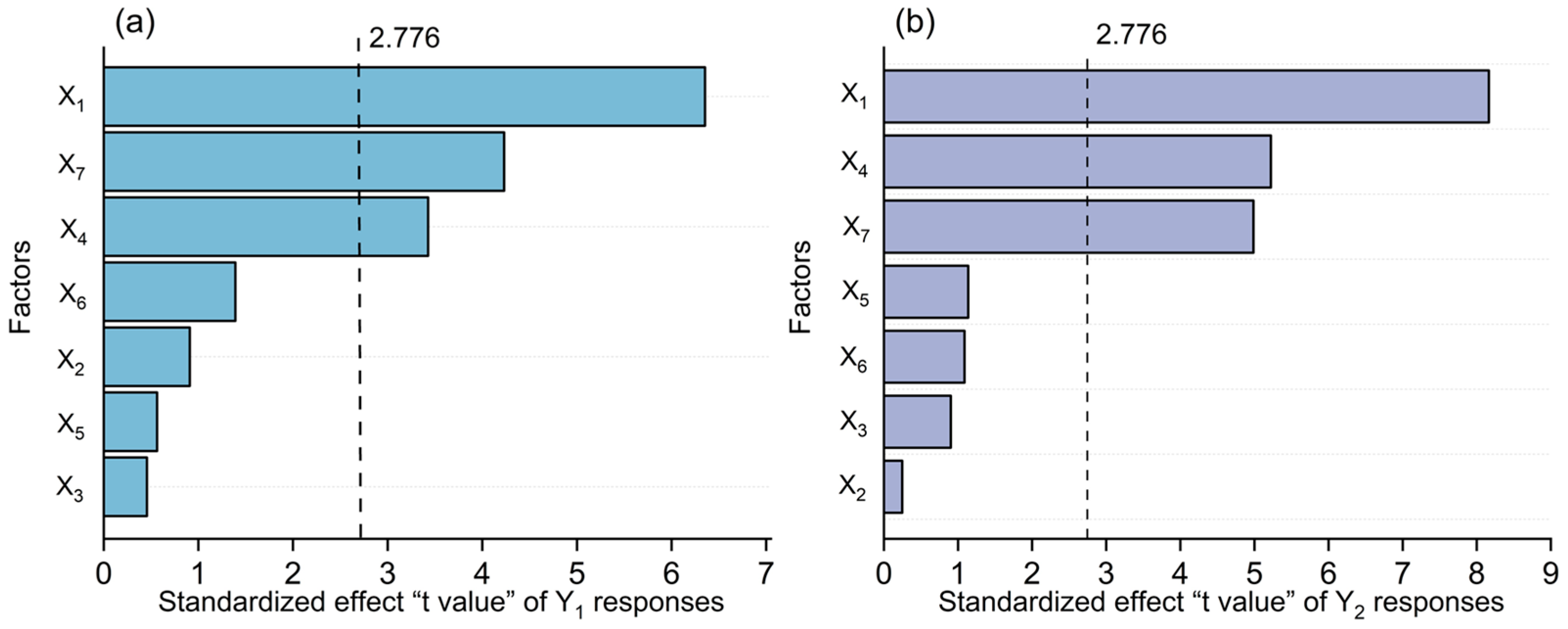
3.2. Screening of Significant Factors Using Plackett–Burman Design
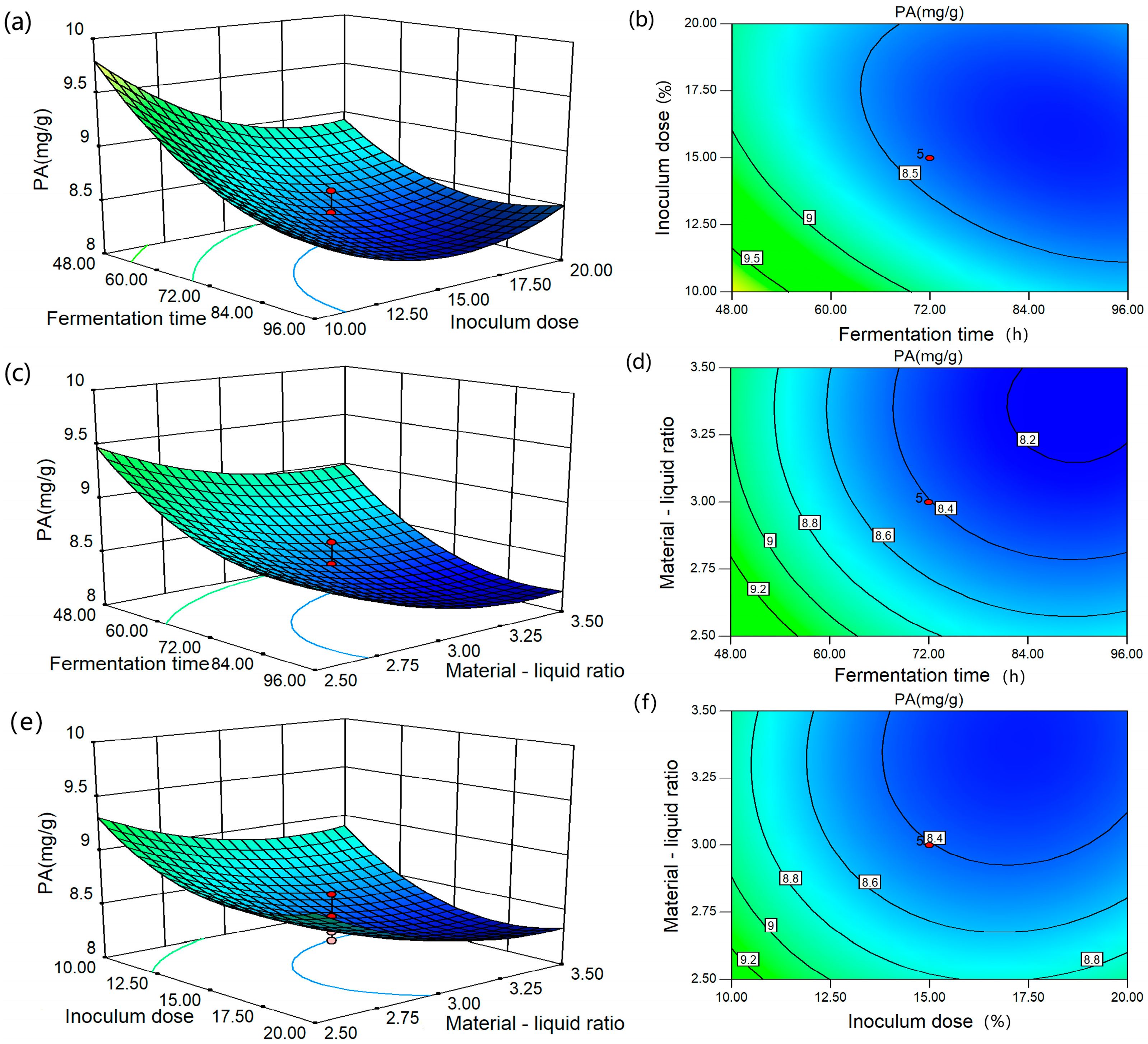
3.3. Statistical Analysis of Central Composite Design
3.4. Optimum Conditions and Authenticity of Predictive Model
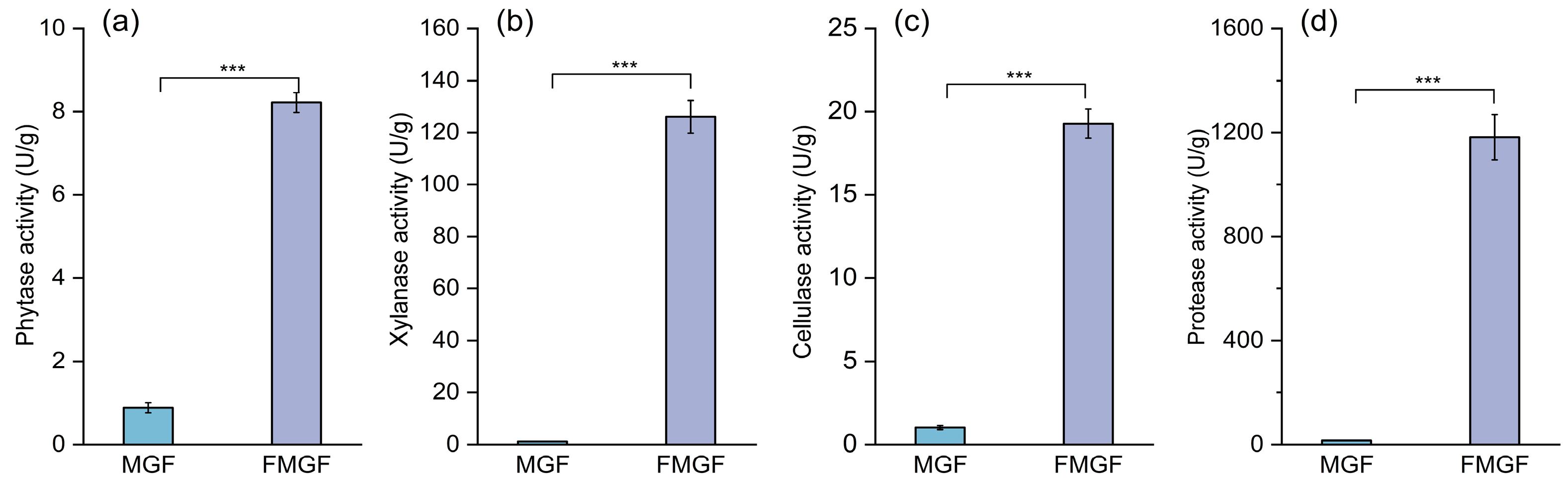
3.5. Changes in Enzymatic Activity Before and After Fermentation
3.6. Comparison of the Nutritional Values of MGF and FMGF
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, R.; Ma, S.; Li, L.; Zhang, M.; Tian, S.; Wang, D.; Liu, K.; Liu, H.; Zhu, W.; Wang, X. Comprehensive Utilization of Corn Starch Processing By-Products: A Review. Grain Oil Sci. Technol. 2021, 4, 89–107. [Google Scholar] [CrossRef]
- Guo, Y.; Wang, K.; Wu, B.; Wu, P.; Duan, Y.; Ma, H. Production of ACE Inhibitory Peptides from Corn Germ Meal by an Enzymatic Membrane Reactor with a Novel Gradient Diafiltration Feeding Working-Mode and in vivo Evaluation of Antihypertensive Effect. J. Funct. Foods 2020, 64, 103584. [Google Scholar] [CrossRef]
- Rocha-Villarreal, V.; Hoffmann, J.F.; Vanier, N.L.; Serna-Saldivar, S.O.; García-Lara, S. Hydrothermal Treatment of Maize: Changes in Physical, Chemical, and Functional Properties. Food Chem. 2018, 263, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Lyu, Z.; Li, Y.; Liu, H.; Li, E.; Li, P.; Zhang, S.; Wang, F.; Lai, C. Net Energy Content of Rice Bran, Defatted Rice Bran, Corn Gluten Feed, and Corn Germ Meal Fed to Growing Pigs Using Indirect Calorimetry. J. Anim. Sci. 2018, 96, 1877–1888. [Google Scholar] [CrossRef] [PubMed]
- Ortiz de Erive, M.; Wang, T.; He, F.; Chen, G. Development of High-Fiber Wheat Bread Using Microfluidized Corn Bran. Food Chem. 2020, 310, 125921. [Google Scholar] [CrossRef]
- Bloot, A.P.M.; Kalschne, D.L.; Amaral, J.A.S.; Baraldi, I.J.; Canan, C. A Review of Phytic Acid Sources, Obtention, and Applications. Food Rev. Int. 2023, 39, 73–92. [Google Scholar] [CrossRef]
- Shi, C.; Zhang, Y.; Lu, Z.; Wang, Y. Solid-State Fermentation of Corn-Soybean Meal Mixed Feed with Bacillus subtilis and Enterococcus faecium for Degrading Antinutritional Factors and Enhancing Nutritional Value. J. Anim. Sci. Biotechnol. 2017, 8, 50. [Google Scholar] [CrossRef]
- Sun, X.; Ma, L.; Lux, P.E.; Wang, X.; Stuetz, W.; Frank, J.; Liang, J. The Distribution of Phosphorus, Carotenoids and Tocochromanols in Grains of Four Chinese Maize (Zea mays L.) Varieties. Food Chem. 2022, 367, 130725. [Google Scholar] [CrossRef]
- Noureddini, H.; Malik, M.; Byun, J.; Ankeny, A.J. Distribution of Phosphorus Compounds in Corn Processing. Bioresour. Technol. 2009, 100, 731–736. [Google Scholar] [CrossRef]
- Sun, H.; Cozannet, P.; Ma, R.; Zhang, L.; Huang, Y.K.; Preynat, A.; Sun, L.-h. Effect of Concentration of Arabinoxylans and a Carbohydrase Mixture on Energy, Amino Acids and Nutrients Total Tract and Ileal Digestibility in Wheat and Wheat by-Product-Based Diet for Pigs. Anim. Feed Sci. Technol. 2020, 262, 114380. [Google Scholar] [CrossRef]
- Huang, M.; Bai, J.; Buccato, D.G.; Zhang, J.; He, Y.; Zhu, Y.; Yang, Z.; Xiao, X.; Daglia, M. Cereal-Derived Water-Unextractable Arabinoxylans: Structure Feature, Effects on Baking Products and Human Health. Foods 2024, 13, 2369. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Bai, J.; Fan, M.; Li, T.; Li, Y.; Qian, H.; Wang, L.; Zhang, H.; Qi, X.; Rao, Z. Cereal-Derived Arabinoxylans: Structural Features and Structure–Activity Correlations. Trends Food Sci. Technol. 2020, 96, 157–165. [Google Scholar] [CrossRef]
- Rosicka-Kaczmarek, J.; Komisarczyk, A.; Nebesny, E.; Makowski, B. The Influence of Arabinoxylans on the Quality of Grain Industry Products. Eur. Food Res. Technol. 2016, 242, 295–303. [Google Scholar] [CrossRef]
- Bautil, A.; Verspreet, J.; Buyse, J.; Goos, P.; Bedford, M.R.; Courtin, C.M. Age-Related Arabinoxylan Hydrolysis and Fermentation in the Gastrointestinal Tract of Broilers Fed Wheat-Based Diets. Poult. Sci. 2019, 98, 4606–4621. [Google Scholar] [CrossRef] [PubMed]
- Endalew, H.W.; Atlabachew, M.; Karavoltsos, S.; Sakellari, A.; Aslam, M.F.; Allen, L.; Griffiths, H.; Zoumpoulakis, P.; Kanellou, A.; Yehuala, T.F.; et al. Effect of Fermentation on Nutrient Composition, Antinutrients, and Mineral Bioaccessibility of Finger Millet Based Injera: A Traditional Ethiopian Food. Food Res. Int. 2024, 190, 114635. [Google Scholar] [CrossRef] [PubMed]
- Watanakij, N.; Visessanguan, W.; Petchkongkaew, A. Aflatoxin B1-Degrading Activity from Bacillus subtilis BCC 42005 Isolated from Fermented Cereal Products. Food Addit. Contam. Part A 2020, 37, 1579–1589. [Google Scholar] [CrossRef]
- Iqbal, S.; Begum, F.; Rabaan, A.A.; Aljeldah, M.; Al Shammari, B.R.; Alawfi, A.; Alshengeti, A.; Sulaiman, T.; Khan, A. Classification and Multifaceted Potential of Secondary Metabolites Produced by Bacillus subtilis Group: A Comprehensive Review. Molecules 2023, 28, 927. [Google Scholar] [CrossRef]
- Suprayogi, W.P.S.; Ratriyanto, A.; Akhirini, N.; Hadi, R.F.; Setyono, W.; Irawan, A. Changes in Nutritional and Antinutritional Aspects of Soybean Meals by Mechanical and Solid-State Fermentation Treatments with Bacillus subtilis and Aspergillus oryzae. Bioresour. Technol. Rep. 2022, 17, 100925. [Google Scholar] [CrossRef]
- Bruno Siewe, F.; Kudre, T.G.; Narayan, B. Optimisation of Ultrasound-Assisted Enzymatic Extraction Conditions of Umami Compounds from Fish by-Products Using the Combination of Fractional Factorial Design and Central Composite Design. Food Chem. 2021, 334, 127498. [Google Scholar] [CrossRef]
- Buddrick, O.; Jones, O.A.H.; Cornell, H.J.; Small, D.M. The Influence of Fermentation Processes and Cereal Grains in Wholegrain Bread on Reducing Phytate Content. J. Cereal Sci. 2014, 59, 3–8. [Google Scholar] [CrossRef]
- Douglas, S.G. A Rapid Method for the Determination of Pentosans in Wheat Flour. Food Chem. 1981, 7, 139–145. [Google Scholar] [CrossRef]
- Rouau, X.; Surget, A. A Rapid Semi-Automated Method for the Determination of Total and Water-Extractable Pentosans in Wheat Flours. Carbohydr. Polym. 1994, 24, 123–132. [Google Scholar] [CrossRef]
- Hernández-Espinosa, N.; Posadas-Romano, G.; Dreisigacker, S.; Crossa, J.; Crespo, L.; Ibba, M.I. Efficient Arabinoxylan Assay for Wheat: Exploring Variability and Molecular Marker Associations in Wholemeal and Refined Flour. J. Cereal Sci. 2024, 117, 103897. [Google Scholar] [CrossRef] [PubMed]
- Akpoilih, B.U.; Adeshina, I.; Chukwudi, C.F.; Abdel-Tawwab, M. Evaluating the Inclusion of Phytase Sources to Phosphorus-Free Diets for GIFT Tilapia (Oreochromis niloticus): Growth Performance, Intestinal Morphometry, Immune-Antioxidant Responses, and Phosphorus Utilization. Anim. Feed Sci. Technol. 2023, 303, 115678. [Google Scholar] [CrossRef]
- Dhaver, P.; Pletschke, B.; Sithole, B.; Govinden, R. Optimization, Purification, and Characterization of Xylanase Production by a Newly Isolated Trichoderma Harzianum Strain by a Two-Step Statistical Experimental Design Strategy. Sci. Rep. 2022, 12, 17791. [Google Scholar] [CrossRef]
- Al Talebi, Z.A.; Al-Kawaz, H.S.; Mahdi, R.K.; Al-Hassnawi, A.T.; Alta’ee, A.H.; Hadwan, A.M.; Khudhair, D.A.; Hadwan, M.H. An Optimized Protocol for Estimating Cellulase Activity in Biological Samples. Anal. Biochem. 2022, 655, 114860. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, K.; Lu, F.; Wang, Y.; Ouyang, N.; Ma, H. Application of Ultrasound Technology in the Field of Solid-State Fermentation: Increasing Peptide Yield through Ultrasound-Treated Bacterial Strain. J. Sci. Food Agric. 2021, 101, 5348–5358. [Google Scholar] [CrossRef]
- Zhang, Y.; Ishikawa, M.; Koshio, S.; Yokoyama, S.; Dossou, S.; Wang, W.; Zhang, X.; Shadrack, R.S.; Mzengereza, K.; Zhu, K.; et al. Optimization of Soybean Meal Fermentation for Aqua-Feed with Bacillus Subtilis Natto Using the Response Surface Methodology. Fermentation 2021, 7, 306. [Google Scholar] [CrossRef]
- Pearce, K.N.; Karahalios, D.; Friedman, M. Ninhydrin Assay For Proteolysis in Ripening Cheese. J. Food Sci. 1988, 53, 432–435. [Google Scholar] [CrossRef]
- Kamble, D.B.; Singh, R.; Rani, S.; Kaur, B.P.; Upadhyay, A.; Kumar, N. Optimization and Characterization of Antioxidant Potential, in vitro Protein Digestion and Structural Attributes of Microwave Processed Multigrain Pasta. J. Food Process. Preserv. 2019, 43, e14125. [Google Scholar] [CrossRef]
- Kumar, A.; Lal, M.K.; Kar, S.S.; Nayak, L.; Ngangkham, U.; Samantaray, S.; Sharma, S.G. Bioavailability of Iron and Zinc as Affected by Phytic Acid Content in Rice Grain. J. Food Biochem. 2017, 41, e12413. [Google Scholar] [CrossRef]
- Zhang, L.; Yang, Y.; Sun, J.; Shen, Y.; Wei, D.; Zhu, J.; Chu, J. Microbial Production of 2,3-Butanediol by a Mutagenized Strain of Serratia Marcescens H30. Bioresour. Technol. 2010, 101, 1961–1967. [Google Scholar] [CrossRef] [PubMed]
- Terlabie, N.N.; Sakyi-Dawson, E.; Amoa-Awua, W.K. The Comparative Ability of Four Isolates of Bacillus subtilis to Ferment Soybeans into Dawadawa. Int. J. Food Microbiol. 2006, 106, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Dayana Priyadharshini, S.; Bakthavatsalam, A.K. Optimization of Phenol Degradation by the Microalga Chlorella Pyrenoidosa Using Plackett-Burman Design and Response Surface Methodology. Bioresour. Technol. 2016, 207, 150–156. [Google Scholar] [CrossRef]
- Chen, F.; Zhang, Q.; Fei, S.; Gu, H.; Yang, L. Optimization of Ultrasonic Circulating Extraction of Samara Oil from Acer Saccharum Using Combination of Plackett–Burman Design and Box–Behnken Design. Ultrason. Sonochem. 2017, 35, 161–175. [Google Scholar] [CrossRef]
- Xi, J.; Xiang, B.; Deng, Y. Comparison of Batch and Circulating Processes for Polyphenols Extraction from Pomelo Peels by Liquid-Phase Pulsed Discharge. Food Chem. 2021, 340, 127918. [Google Scholar] [CrossRef]
- Chen, W.; Xu, D. Phytic Acid and Its Interactions in Food Components, Health Benefits, and Applications: A Comprehensive Review. Trends Food Sci. Technol. 2023, 141, 104201. [Google Scholar] [CrossRef]
- Dahiya, S.; Kumar, A.; Singh, B. Enhanced Endoxylanase Production by Myceliophthora thermophila Using Rice Straw and Its Synergism with Phytase in Improving Nutrition. Process Biochem. 2020, 94, 235–242. [Google Scholar] [CrossRef]
- Tse, T.; Schendel, R.R. Cereal Grain Arabinoxylans: Processing Effects and Structural Changes during Food and Beverage Fermentations. Fermentation 2023, 9, 914. [Google Scholar] [CrossRef]
- Liu, Y.; Li, H.; Liu, W.; Ren, K.; Li, X.; Zhang, Z.; Huang, R.; Han, S.; Hou, J.; Pan, C. Bioturbation Analysis of Microbial Communities and Flavor Metabolism in a High-Yielding Cellulase Bacillus subtilis Biofortified Daqu. Food Chem X 2024, 22, 101382. [Google Scholar] [CrossRef]
- Reynaud, Y.; Lopez, M.; Riaublanc, A.; Souchon, I.; Dupont, D. Hydrolysis of Plant Proteins at the Molecular and Supra-Molecular Scales during in vitro Digestion. Food Res. Int. 2020, 134, 109204. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Wang, L.; Zhang, Z.; Ding, L.; Hang, S. Combination of Fiber-Degrading Enzymatic Hydrolysis and Lactobacilli Fermentation Enhances Utilization of Fiber and Protein in Rapeseed Meal as Revealed in Simulated Pig Digestion and Fermentation in vitro. Anim. Feed Sci. Technol. 2021, 278, 115001. [Google Scholar] [CrossRef]


| Factors | Responses | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Run | X1 | X2 | X3 | X4 | X5 | X6 | X7 | Y1 | Y2 |
| 1 | 96 (+) | 28 (−) | 7 (+) | 20 (+) | 189 (−) | 3.42 (+) | 1:2.5 (−) | 8.6 ± 0.54 | 88.9 ± 0.94 |
| 2 | 96 (+) | 28 (−) | 6 (−) | 10 (−) | 30 (+) | 3.42 (+) | 1:3.5 (+) | 8.3 ± 0.26 | 90.0 ± 1.88 |
| 3 | 48 (−) | 34 (+) | 6 (−) | 10 (−) | 189 (−) | 3.42 (+) | 1:3.5 (+) | 9.1 ± 0.20 | 92.3 ± 1.01 |
| 4 | 48 (−) | 28 (−) | 6 (−) | 10 (−) | 189 (−) | 1.84 (−) | 1:2.5 (−) | 9.9 ± 0.36 | 97.9 ± 1.40 |
| 5 | 72 (0) | 31 (0) | 6.5 (0) | 15 (0) | 123 (0) | 2.63 (0) | 1:3 (0) | 8.3 ± 0.30 | 87.8 ± 1.24 |
| 6 | 48 (−) | 34 (+) | 7 (+) | 10 (−) | 30 (+) | 1.84 (−) | 1:2.5 (−) | 10.0 ± 0.38 | 98.3 ± 0.84 |
| 7 | 96 (+) | 34 (+) | 6 (−) | 20 (+) | 30 (+) | 1.84 (−) | 1:3.5 (+) | 8.2 ± 0.33 | 86.9 ± 0.96 |
| 8 | 48 (−) | 28 (−) | 7 (+) | 20 (+) | 30 (+) | 1.84 (−) | 1:3.5 (+) | 8.7 ± 0.31 | 91.8 ± 0.91 |
| 9 | 96 (+) | 28 (−) | 7 (+) | 10 (−) | 189 (−) | 1.84 (−) | 1:3.5 (+) | 8.4 ± 0.07 | 89.3 ± 0.57 |
| 10 | 96 (+) | 34 (+) | 6 (−) | 20 (+) | 189 (−) | 1.84 (−) | 1:2.5 (−) | 8.5 ± 0.31 | 88.8 ± 1.08 |
| 11 | 96 (+) | 34 (+) | 7 (+) | 10 (−) | 30 (+) | 3.42 (+) | 1:2.5 (−) | 8.8 ± 0.74 | 92.5 ± 0.59 |
| 12 | 48 (−) | 34 (+) | 7 (+) | 20 (+) | 189 (−) | 3.42 (+) | 1:3.5 (+) | 8.9 ± 0.40 | 91.8 ± 0.43 |
| 13 | 48 (−) | 28 (−) | 6 (−) | 20 (+) | 30 (+) | 3.42 (+) | 1:2.5 (−) | 9.0 ± 0.43 | 93.6 ± 0.92 |
| PA (Phytic Acid) | ||||||
| Source | Sum of Squares | Degree of Freedom | Mean Square | F-Value | p-Value | Significance |
| Model | 3.4036 | 7 | 0.4862 | 10.4564 | 0.0194 | * |
| X1 | 1.8723 | 1 | 1.8723 | 40.2645 | 0.0032 | ** |
| X2 | 0.0385 | 1 | 0.0385 | 0.8287 | 0.4142 | |
| X3 | 0.0096 | 1 | 0.0096 | 0.2072 | 0.6726 | |
| X4 | 0.5461 | 1 | 0.5461 | 11.7448 | 0.0266 | * |
| X5 | 0.0147 | 1 | 0.0147 | 0.3161 | 0.6040 | |
| X6 | 0.0901 | 1 | 0.0901 | 1.9384 | 0.2363 | |
| X7 | 0.8321 | 1 | 0.8321 | 17.8953 | 0.0134 | * |
| WU-AX (Water-Unextractable Arabinoxylan) | ||||||
| Source | Sum of Squares | Degree of Freedom | Mean Square | F-Value | p-Value | Significance |
| Model | 129.6758 | 7 | 18.5251 | 17.4466 | 0.0075 | ** |
| X1 | 70.7616 | 1 | 70.7616 | 66.6420 | 0.0012 | ** |
| X2 | 0.0645 | 1 | 0.0645 | 0.0608 | 0.8174 | |
| X3 | 0.8640 | 1 | 0.8640 | 0.8137 | 0.4180 | |
| X4 | 28.9541 | 1 | 28.9541 | 27.2685 | 0.0064 | ** |
| X5 | 1.3736 | 1 | 1.3736 | 1.2937 | 0.3189 | |
| X6 | 1.2545 | 1 | 1.2545 | 1.1815 | 0.3382 | |
| X7 | 26.4033 | 1 | 26.4033 | 24.8662 | 0.0076 | ** |
| Factors | Responses | ||||
|---|---|---|---|---|---|
| Run | X1 | X4 | X7 | Y1 | Y2 |
| 5 | 48 (−1) | 10 (−1) | 1:3.5 (1) | 9.7 ± 0.27 | 94.8 ± 0.70 |
| 1 | 48 (−1) | 10 (−1) | 1:2.5 (−1) | 10.2 ± 0.46 | 102.2 ± 1.59 |
| 12 | 72 (0) | 25 (2) | 1:3 (0) | 8.9 ± 0.32 | 89.5 ± 1.25 |
| 13 | 72 (0) | 15 (0) | 1:2 (−2) | 9.4 ± 0.13 | 90.9 ± 1.13 |
| 4 | 96 (1) | 20 (1) | 1:2.5 (−1) | 9.3 ± 0.17 | 87.1 ± 0.32 |
| 16 (C) | 72 (0) | 15 (0) | 1:3 (0) | 8.3 ± 0.30 | 83.5 ± 1.46 |
| 10 | 120 (2) | 15 (0) | 1:3 (0) | 8.4 ± 0.30 | 89.7 ± 0.80 |
| 8 | 96 (1) | 20 (1) | 1:3.5 (1) | 8.6 ± 0.25 | 84.6 ± 0.65 |
| 3 | 48 (−1) | 20 (1) | 1:2.5 (−1) | 9.2 ± 0.70 | 97.0 ± 0.73 |
| 2 | 96 (1) | 10 (−1) | 1:2.5 (−1) | 9.2 ± 0.07 | 91.7 ± 0.97 |
| 7 | 48 (−1) | 20 (1) | 1:3.5 (1) | 9.0 ± 0.36 | 88.9 ± 0.99 |
| 6 | 96 (1) | 10 (−1) | 1:3.5 (1) | 8.7 ± 0.31 | 92.8 ± 1.05 |
| 19 (C) | 72 (0) | 15 (0) | 1:3 (0) | 8.3 ± 0.29 | 87.9 ± 0.56 |
| 17 (C) | 72 (0) | 15 (0) | 1:3 (0) | 8.2 ± 0.46 | 84.5 ± 0.72 |
| 14 | 72 (0) | 15 (0) | 1:4 (2) | 8.6 ± 0.25 | 86.8 ± 0.91 |
| 9 | 24 (−2) | 15 (0) | 1:3 (0) | 10.4 ± 0.44 | 105.5 ± 0.78 |
| 11 | 72 (0) | 5 (−2) | 1:3 (0) | 10.1 ± 0.19 | 99.9 ± 1.08 |
| 18 (C) | 72 (0) | 15 (0) | 1:3 (0) | 8.4 ± 0.28 | 85.8 ± 1.24 |
| 15 (C) | 72 (0) | 15 (0) | 1:3 (0) | 8.6 ± 0.61 | 85.3 ± 0.51 |
| Source | PA | WU-AX | ||||
|---|---|---|---|---|---|---|
| Sum of Squares | F-Value | p-Value | Sum of Squares | F-Value | p-Value | |
| Model | 7.8627 | 17.8433 | 0.0001 | 696.2109 | 33.6951 | <0.0001 |
| X1 | 2.5440 | 51.9596 | <0.0001 | 213.1600 | 92.4500 | <0.0001 |
| X4 | 0.9312 | 19.0195 | 0.0018 | 125.3280 | 54.3563 | <0.0001 |
| X7 | 0.9702 | 19.8160 | 0.0016 | 39.4384 | 17.1049 | 0.0025 |
| X1 × X4 | 0.2813 | 5.7443 | 0.0401 | 0.3200 | 0.1388 | 0.7181 |
| X1 × X7 | 0.0008 | 0.0163 | 0.9011 | 24.7808 | 10.7477 | 0.0096 |
| X4 × X7 | 0.0145 | 0.2951 | 0.6001 | 2.2261 | 0.9655 | 0.3515 |
| X12 | 1.7536 | 35.8157 | 0.0002 | 229.7474 | 99.6442 | <0.0001 |
| X42 | 2.0730 | 42.3401 | 0.0001 | 134.6781 | 58.4115 | <0.0001 |
| X72 | 0.7016 | 14.3295 | 0.0043 | 20.0899 | 8.7132 | 0.0162 |
| Residual | 0.4407 | 20.7511 | ||||
| Lack of Fit | 0.3247 | 2.2411 | 0.2272 | 10.0901 | 0.7572 | 0.6234 |
| C.V.% | 2.45 | 1.67 | ||||
| Pure Error | 0.1159 | 10.6610 | ||||
| Cor Total | 8.3034 | 719.9620 | ||||
| R2 | 0.9469 | 0.9712 | ||||
| R2-adjusted | 0.8939 | 0.9424 | ||||
| Items | MGF | FMGF | p-Value | Change (%) |
|---|---|---|---|---|
| Protein (%) | 27.1 ± 0.13 | 28.63 ± 0.08 | <0.001 | 5.57 |
| PDI (%) | 37.9 ± 1.02 | 46.67 ± 0.58 | <0.001 | 23.17 |
| DH (%) | 2.3 ± 0.11 | 3.26 ± 0.12 | <0.001 | 43.61 |
| IVPD (%) | 44.3 ± 1.07 | 58.48 ± 0.78 | <0.001 | 31.92 |
| Minerals’ bioavailability | ||||
| Fe (%) | 22.8 ± 1.51 | 33.68 ± 2.15 | <0.001 | 47.72 |
| Mn (%) | 38.3 ± 1.53 | 53.23 ± 1.38 | <0.001 | 39.05 |
| Cu (%) | 46.0 ± 1.04 | 57.54 ± 1.06 | <0.001 | 25.20 |
| Zn (%) | 12.6 ± 1.23 | 16.67 ± 0.95 | <0.001 | 31.88 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, X.; Ma, L.; Xuan, Y.; Liang, J. Degradation of Anti-Nutritional Factors in Maize Gluten Feed by Fermentation with Bacillus subtilis: A Focused Study on Optimizing Fermentation Conditions. Fermentation 2024, 10, 555. https://doi.org/10.3390/fermentation10110555
Sun X, Ma L, Xuan Y, Liang J. Degradation of Anti-Nutritional Factors in Maize Gluten Feed by Fermentation with Bacillus subtilis: A Focused Study on Optimizing Fermentation Conditions. Fermentation. 2024; 10(11):555. https://doi.org/10.3390/fermentation10110555
Chicago/Turabian StyleSun, Xiaohong, Lei Ma, Yaoquan Xuan, and Jianfen Liang. 2024. "Degradation of Anti-Nutritional Factors in Maize Gluten Feed by Fermentation with Bacillus subtilis: A Focused Study on Optimizing Fermentation Conditions" Fermentation 10, no. 11: 555. https://doi.org/10.3390/fermentation10110555
APA StyleSun, X., Ma, L., Xuan, Y., & Liang, J. (2024). Degradation of Anti-Nutritional Factors in Maize Gluten Feed by Fermentation with Bacillus subtilis: A Focused Study on Optimizing Fermentation Conditions. Fermentation, 10(11), 555. https://doi.org/10.3390/fermentation10110555







