Field Observations and Genetic Characterization of Sheep-Associated Malignant Catarrhal Fever in Egypt, 2018
Abstract
1. Introduction
2. Materials and Methods
2.1. Field Study
2.2. Nucleic Acid Extraction and Real-Time PCR
2.3. Sequencing, Nucleotide Sequence Alignments, and Phylogenetic Analysis
3. Results and Discussion
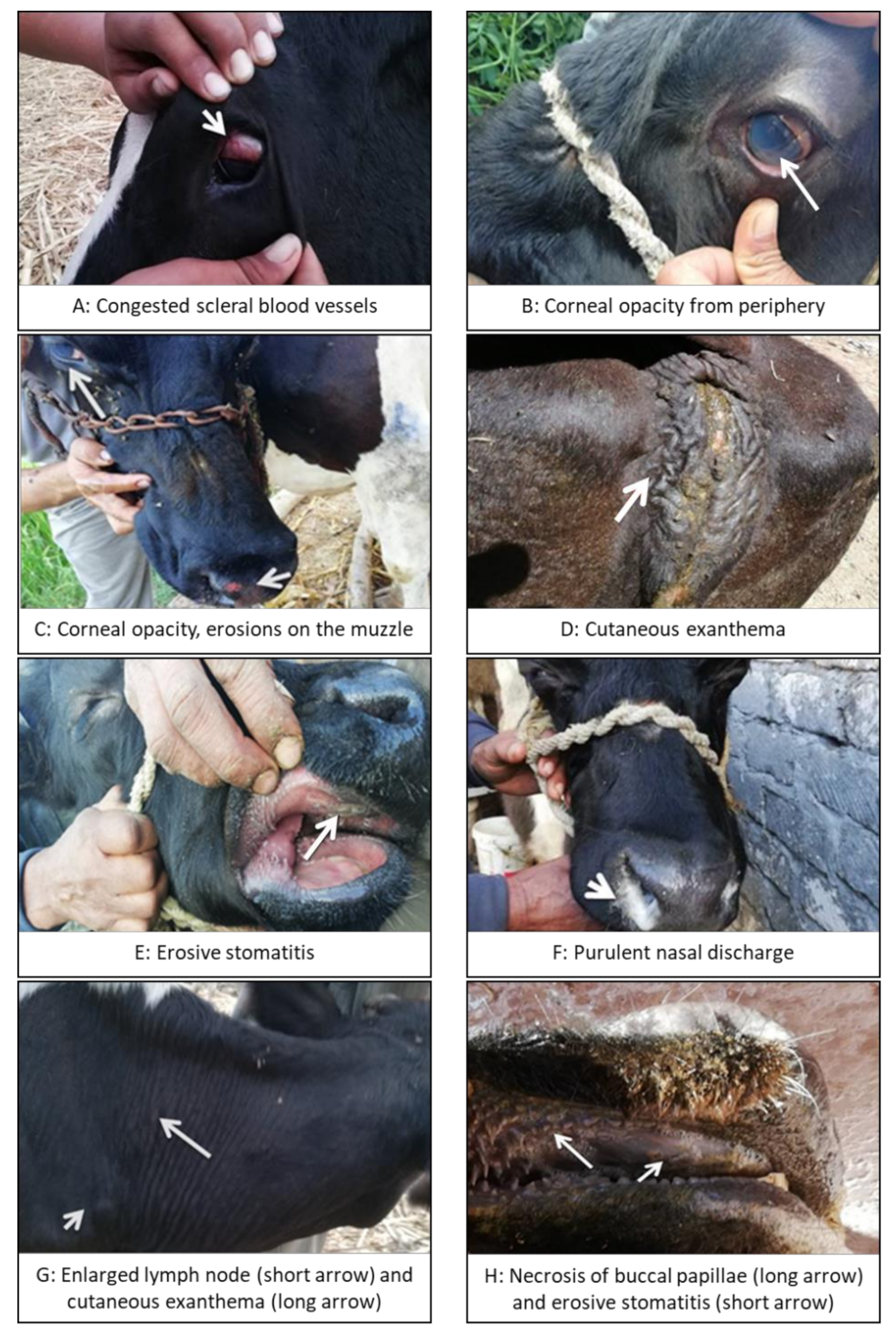
3.1. Clinical Disease
3.2. Virus Detection, Nucleotide Sequence Determination, and Phylogenetic Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Li, H.; Cunha, C.W.; Taus, N.S.; Knowles, D.P. Malignant catarrhal fever: Inching toward understanding. Annu. Rev. Anim. Biosci. 2014, 2, 209–233. [Google Scholar] [CrossRef] [PubMed]
- Mushi, E.Z.; Rurangirwa, F.R.; Karstad, L. Shedding of malignant catarrhal fever virus by wildebeest calves. Vet. Microbiol. 1981, 6, 281–286. [Google Scholar] [CrossRef]
- Li, H.; Snowder, G.; O’Toole, D.; Crawford, T.B. Transmission of ovine herpesvirus 2 in lambs. J. Clin. Microbiol. 1998, 36, 223–226. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Hua, Y.; Snowder, G.; Crawford, T.B. Levels of ovine herpesvirus 2 DNA in nasal secretions and blood of sheep: Implications for transmission. Vet. Microbiol. 2001, 79, 301–310. [Google Scholar] [CrossRef]
- OIE. Technical Disease Card: Malignant Catarrhal Fever. Available online: https://www.oie.int/fileadmin/Home/eng/Animal_Health_in_the_World/docs/pdf/Disease_cards/MALIGNANT_CATHARRAL_FEVER.pdf (accessed on 1 November 2020).
- Li, H.; Taus, N.S.; Lewis, G.S.; Kim, O.; Traul, D.L.; Crawford, T.B. Shedding of ovine herpesvirus 2 in sheep nasal secretions: The predominant mode for transmission. J. Clin. Microbiol. 2004, 42, 5558–5564. [Google Scholar] [CrossRef]
- Li, H.; Karney, G.; O’Toole, D.; Crawford, T.B. Long distance spread of malignant catarrhal fever virus from feedlot lambs to ranch bison. Can. Vet. J. 2008, 49, 183–185. [Google Scholar]
- Li, H.; Taus, N.S.; Jones, C.; Murphy, B.; Evermann, J.F.; Crawford, T.B. A devastating outbreak of malignant catarrhal fever in a bison feedlot. J. Vet. Diagn. Investig. 2006, 18, 119–123. [Google Scholar] [CrossRef]
- Berezowski, J.A.; Appleyard, G.D.; Crawford, T.B.; Haigh, J.; Li, H.; Middleton, D.M.; O’Connor, B.P.; West, K.; Woodbury, M. An outbreak of sheep-associated malignant catarrhal fever in bison (Bison bison) after exposure to sheep at a public auction sale. J. Vet. Diagn. Investig. 2005, 17, 55–58. [Google Scholar] [CrossRef]
- O’Toole, D.; Li, H. The pathology of malignant catarrhal fever, with an emphasis on ovine herpesvirus 2. Vet. Pathol. 2014, 51, 437–452. [Google Scholar] [CrossRef]
- Eschbaumer, M.; Vögtlin, A.; Paton, D.J.; Barnabei, J.L.; Sanchez-Vazquez, M.J.; Pituco, E.M.; Rivera, A.M.; O’Brien, D.; Nfon, C.; Brocchi, E.; et al. Non-discriminatory Exclusion Testing as a Tool for the Early Detection of Foot-and-Mouth Disease Incursions. Front. Vet. Sci. 2020, 7, 552670. [Google Scholar] [CrossRef]
- Schock, A.; Collins, R.A.; Reid, H.W. Phenotype, growth regulation and cytokine transcription in Ovine Herpesvirus-2 (OHV-2)-infected bovine T-cell lines. Vet. Immunol. Immunopathol. 1998, 66, 67–81. [Google Scholar] [CrossRef]
- OIE. Manual of Diagnostic Tests and Vaccines for Terrestrial Animals, Chapter 3.4.13: Malignant Catarrhal Fever. Available online: https://www.oie.int/fileadmin/Home/eng/Health_standards/tahm/3.04.13_MCF.pdf (accessed on 1 November 2020).
- Doboro, F.A.; Njiro, S.; Sibeko-Matjila, K.; Van Vuuren, M. Molecular Analysis of South African Ovine Herpesvirus 2 Strains Based on Selected Glycoprotein and Tegument Genes. PLoS ONE 2016, 11, e0147019. [Google Scholar] [CrossRef] [PubMed]
- Bastawecy, I.M.; Abd El-Samee, A.A. First Isolation and Identification of Ovine Herpesvirus 2 Causing Malignant Catarrhal Fever Outbreak in Egypt. Life Sci. J. 2012, 9, 798–804. [Google Scholar]
- Azzam, R.A.; Elnesr, K.A.; Rouby, S.H.; Mahdy, E.M.; Hussein, H.A.; Menshawy, A.S. Clinical and molecular evidences of circulation of sheep-associated MCF in cattle and buffaloes in Egypt. Sadat Vet. Med. J. 2016, 10, 9–20. [Google Scholar]
- Zaki, A.A.M.; El-Said, H.M.; Abd El-Aziz, A.; Bastawecy, I.M.; Abd El-Wahab, S.A.; El-Sayed, M.M. Field Study on Malignant Catarrhal Fever (MCF) In Egypt. Life Sci. J. 2016, 13, 83–97. [Google Scholar]
- Jackson, P.G.G.; Cockcroft, P.D. Clinical Examination of Farm Animals; Blackwell Science Ltd.: Hoboken, NJ, USA, 2002. [Google Scholar]
- Traul, D.L.; Elias, S.; Taus, N.S.; Herrmann, L.M.; Oaks, J.L.; Li, H. A real-time PCR assay for measuring alcelaphine herpesvirus-1 DNA. J. Virol. Methods 2005, 129, 186–190. [Google Scholar] [CrossRef]
- Hüssy, D.; Stäuber, N.; Leutenegger, C.M.; Rieder, S.; Ackermann, M. Quantitative fluorogenic PCR assay for measuring ovine herpesvirus 2 replication in sheep. Clin. Diagn. Lab. Immunol. 2001, 8, 123–128. [Google Scholar] [CrossRef]
- Abd El Rahman, S.; Hoffmann, B.; Karam, R.; El-Beskawy, M.; Hamed, M.F.; Forth, L.F.; Höper, D.; Eschbaumer, M. Sequence Analysis of Egyptian Foot-and-Mouth Disease Virus Field and Vaccine Strains: Intertypic Recombination and Evidence for Accidental Release of Virulent Virus. Viruses 2020, 12, 990. [Google Scholar] [CrossRef]
- Hoffmann, B.; Depner, K.; Schirrmeier, H.; Beer, M. A universal heterologous internal control system for duplex real-time RT-PCR assays used in a detection system for pestiviruses. J. Virol. Methods 2006, 136, 200–209. [Google Scholar] [CrossRef]
- Wernike, K.; Hoffmann, B.; Kalthoff, D.; Konig, P.; Beer, M. Development and validation of a triplex real-time PCR assay for the rapid detection and differentiation of wild-type and glycoprotein E-deleted vaccine strains of Bovine herpesvirus type 1. J. Virol. Methods 2011, 174, 77–84. [Google Scholar] [CrossRef]
- Hart, J.; Ackermann, M.; Jayawardane, G.; Russell, G.; Haig, D.M.; Reid, H.; Stewart, J.P. Complete sequence and analysis of the ovine herpesvirus 2 genome. J. Gen. Virol. 2007, 88, 28–39. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef] [PubMed]
- Kimura, M. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J. Mol. Evol. 1980, 16, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across Computing Platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Felsenstein, J. Confidence Limits on Phylogenies: An Approach Using the Bootstrap. Evolution 1985, 39, 783–791. [Google Scholar] [CrossRef] [PubMed]
- Turan, T.; Isidan, H.; Atasoy, M.O.; Sozdutmaz, I.; Bulut, H. Genetic Diversity of Ovine Herpesvirus 2 Strains Obtained From Malignant Catarrhal Fever Cases in Eastern Turkey. Virus Res. 2020, 276, 197801. [Google Scholar] [CrossRef]
- Taus, N.S.; Herndon, D.R.; Traul, D.L.; Stewart, J.P.; Ackermann, M.; Li, H.; Knowles, D.P.; Lewis, G.S.; Brayton, K.A. Comparison of ovine herpesvirus 2 genomes isolated from domestic sheep (Ovis aries) and a clinically affected cow (Bos bovis). J. Gen. Virol. 2007, 88, 40–45. [Google Scholar] [CrossRef]
- Dry, I.; Haig, D.M.; Inglis, N.F.; Imrie, L.; Stewart, J.P.; Russell, G.C. Proteomic analysis of pathogenic and attenuated alcelaphine herpesvirus 1. J. Virol. 2008, 82, 5390–5397. [Google Scholar] [CrossRef]
- May, J.S.; Walker, J.; Colaco, S.; Stevenson, P.G. The murine gammaherpesvirus 68 ORF27 gene product contributes to intercellular viral spread. J. Virol. 2005, 79, 5059–5068. [Google Scholar] [CrossRef]

| Cattle | Small Ruminants 1 | ||||||
|---|---|---|---|---|---|---|---|
| Sex | Age | ||||||
| Herd | Female | Male | <1 Year | 1–3 Years | 3–5 Years | Sheep | Goats |
| A | 33 | 2 | 10 | 6 | 19 | 6 | 4 |
| B | 64 | 3 | 15 | 18 | 34 | 25 | 13 |
| C | 137 | 4 | 20 | 32 | 89 | 21 | 8 |
| D | 132 | 3 | 38 | 30 | 67 | 14 | 9 |
| E | 47 | 2 | 12 | 10 | 27 | 7 | 4 |
| Total | 413 | 14 | 95 | 96 | 236 | 73 | 38 |
| Animal | Herd | Fever | Corneal Opacity | Erosions in Oral Cavity | Enlarged Lymph Nodes | Purulent Nasal Discharge | Hemorrhagic Diarrhea | Cutaneous Exanthema | Red-Tinged Urine | Peeling Skin on Muzzle |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | A | Yes | Yes | Yes | Yes | Yes | No | Yes | Yes | Yes |
| 2 | B | Yes | Yes | Yes | Yes | Yes | Yes | Yes | No | No |
| 3 | B | Yes | Yes | Yes | Yes | Yes | No | Yes | No | Yes |
| 4 | C | Yes | Yes | Yes | Yes | Yes | Yes | No | No | No |
| 5 | C | Yes | Yes | Yes | Yes | Yes | Yes | No | No | No |
| 6 | C | Yes | Yes | Yes | Yes | Yes | No | No | Yes | No |
| 7 | C | Yes | Yes | Yes | Yes | Yes | No | No | No | No |
| 8 | C | Yes | Yes | Yes | Yes | Yes | No | No | No | No |
| 9 | C | Yes | Yes | Yes | Yes | Yes | Yes | No | No | No |
| 10 | C | Yes | Yes | Yes | Yes | Yes | Yes | No | Yes | Yes |
| 11 | D | Yes | Yes | Yes | Yes | Yes | Yes | No | No | No |
| 12 | D | Yes | Yes | Yes | Yes | Yes | No | No | No | No |
| 13 | D | Yes | Yes | Yes | Yes | Yes | Yes | No | No | No |
| 14 | D | Yes | Yes | Yes | Yes | Yes | No | No | No | No |
| 15 | D | Yes | Yes | Yes | Yes | Yes | No | No | Yes | Yes |
| 16 | D | Yes | Yes | Yes | Yes | Yes | Yes | No | Yes | Yes |
| 17 | E | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| 18 | E | Yes | Yes | Yes | Yes | Yes | Yes | No | No | Yes |
| Total (n) | 18 | 18 | 18 | 18 | 18 | 10 | 4 | 6 | 7 | |
| % of animals | 100 | 100 | 100 | 100 | 100 | 56 | 22 | 33 | 39 | |
| Herd | Animals Examined | Diseased Animals | Morbidity Rate | Deaths | Case Fatality Rate |
|---|---|---|---|---|---|
| A | 35 | 1 | 2.9% | 1 | 100.0% |
| B | 67 | 2 | 3.0% | 2 | 100.0% |
| C | 141 | 7 | 3.5% | 5 | 71.4% |
| D | 135 | 6 | 3.0% | 4 | 66.7% |
| E | 49 | 2 | 4.1% | 1 | 50.0% |
| Total | 427 | 18 | 4.2% | 13 | 72.2% |
| Animal | Herd | Buffy Coat | Whole Blood | Plasma | Serum | Saliva |
|---|---|---|---|---|---|---|
| 1 | A | No Cq | No Cq | n.s. | No Cq | n.s. |
| 2 | B | 31.03 | n.s. | No Cq | n.s. | n.s. |
| 3 | B | No Cq | n.s. | n.s. | n.s. | n.s. |
| 4 | C | No Cq | n.s. | No Cq | n.s. | n.s. |
| 5 | C | No Cq | n.s. | n.s. | n.s. | n.s. |
| 6 | C | No Cq | n.s. | n.s. | n.s. | No Cq |
| 7 | C | No Cq | n.s. | n.s. | n.s. | n.s. |
| 8 | C | No Cq | n.s. | n.s. | n.s. | n.s. |
| 9 | C | No Cq | n.s. | n.s. | n.s. | n.s. |
| 10 | C | 33.17 | n.s. | n.s. | 35.99 | n.s. |
| 11 | D | No Cq | n.s. | n.s. | n.s. | No Cq |
| 12 | D | No Cq | n.s. | n.s. | n.s. | n.s. |
| 13 | D | No Cq | n.s. | n.s. | n.s. | n.s. |
| 14 | D | No Cq | n.s. | n.s. | n.s. | n.s. |
| 15 | D | No Cq | n.s. | n.s. | n.s. | n.s. |
| 16 | D | 35.23 | n.s. | 35.34 | No Cq | n.s. |
| 17 | E | No Cq | n.s. | n.s. | n.s. | n.s. |
| 18 | E | No Cq | n.s. | n.s. | n.s. | n.s. |
| Samples tested (n) | 18 | 1 | 3 | 3 | 2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abd El Rahman, S.; Ateya, A.; El-Beskawy, M.; Wernike, K.; Hoffmann, B.; Eschbaumer, M. Field Observations and Genetic Characterization of Sheep-Associated Malignant Catarrhal Fever in Egypt, 2018. Vet. Sci. 2020, 7, 201. https://doi.org/10.3390/vetsci7040201
Abd El Rahman S, Ateya A, El-Beskawy M, Wernike K, Hoffmann B, Eschbaumer M. Field Observations and Genetic Characterization of Sheep-Associated Malignant Catarrhal Fever in Egypt, 2018. Veterinary Sciences. 2020; 7(4):201. https://doi.org/10.3390/vetsci7040201
Chicago/Turabian StyleAbd El Rahman, Sahar, Ahmed Ateya, Mohamed El-Beskawy, Kerstin Wernike, Bernd Hoffmann, and Michael Eschbaumer. 2020. "Field Observations and Genetic Characterization of Sheep-Associated Malignant Catarrhal Fever in Egypt, 2018" Veterinary Sciences 7, no. 4: 201. https://doi.org/10.3390/vetsci7040201
APA StyleAbd El Rahman, S., Ateya, A., El-Beskawy, M., Wernike, K., Hoffmann, B., & Eschbaumer, M. (2020). Field Observations and Genetic Characterization of Sheep-Associated Malignant Catarrhal Fever in Egypt, 2018. Veterinary Sciences, 7(4), 201. https://doi.org/10.3390/vetsci7040201






