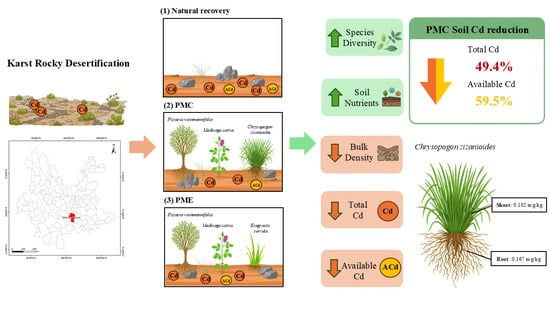
Vegetation Restoration in Karst Southwest China: Effects of Plant Community Diversity and Soil Physicochemical Properties on Soil Cadmium
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Analysis of Community Traits
2.3. Sample Collection and Preparation
2.4. Soil and Plant Chemical and Metal Analysis
2.5. Calculation Methods for Species Diversity and Accumulation Characteristics
2.6. Statistics Analysis
3. Results
3.1. Plant Community Characteristics
3.2. Soil Properties and Cd Concentrations
3.3. Biomass Cd Content and Accumulation of Dominant Plants
3.4. Correlations Between Environmental Variables and Cd Contents
4. Discussion
4.1. The Transport and Absorption of Cd by the Dominant Species
4.2. Response of Community Functional Diversity to Soil Chemical Properties and Cd Concentration
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Treatment | Species | IV (%) |
|---|---|---|
| BL | Imperata cylindrica | 65.89 ± 34.46 a |
| BL | Bidenspilosa | 30.24 ± 13.09 b |
| BL | Erigeron canadensis | 19.53 bc |
| BL | Miscanthus sinensis | 11.15 ± 0.36 c |
| PMC | Chrysopogon zizanioides | 58.11 ± 2.76 a |
| PMC | Medicago sativa | 30.69 ± 2.03 b |
| PMC | Miscanthus sinensis | 6.88 c |
| PMC | Imperata cylindrica | 4.99 ± 2.91 c |
| PMC | Oxalis corniculata | 5.01 c |
| PMC | Erigeron canadensis | 4.28 c |
| PMC | Praxelis clematidea | 4.13 c |
| PMC | Kummerowia striata | 2.00 c |
| PMC | Bidens pilosa | 1.39 c |
| PME | Medicago sativa | 59.66 ± 5.82 a |
| PME | Eragrostis curvula | 22.21 ± 2.39 b |
| PME | Heteropogon contortus | 17.44 c |
| PME | Inula cappa | 11.71 c |
| PME | Erigeron canadensis | 7.84 ± 0.47 cd |
| PME | Imperata cylindrica | 4.51 d |
| PME | Miscanthus sinensis | 3.75 d |
| PME | Bidens pilosa | 1.25 d |
References
- Rahim, H.U.; Akbar, W.A.; Alatalo, J.M. A Comprehensive Literature Review on Cadmium (Cd) Status in the Soil Environment and Its Immobilization by Biochar-Based Materials. Agronomy 2022, 12, 877. [Google Scholar] [CrossRef]
- Su, C.; Wang, J.; Chen, Z.; Meng, J.; Yin, G.; Zhou, Y.; Wang, T. Sources and Health Risks of Heavy Metals in Soils and Vegetables from Intensive Human Intervention Areas in South China. Sci. Total Environ. 2023, 857, 159389. [Google Scholar]
- Wu, Y.; Tian, X.; Wang, R.; Zhang, M.; Wang, S. Effects of Vegetation Restoration on Distribution Characteristics of Heavy Metals in Soil in Karst Plateau Area of Guizhou. PeerJ 2023, 3, e15044. [Google Scholar] [CrossRef]
- Firmani, G.; Chiavarini, M.; Dolcini, J.; Quarta, S.; D’Errico, M.M.; Barbadoro, P. The Association between Cadmium Exposure and Prostate Cancer: An Updated Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2024, 21, 1532. [Google Scholar] [CrossRef]
- Zhou, Z.; Shi, Z.; Yu, L.; Fan, H.; Wan, F. Soil Quality and Heavy Metal Source Analyses for Characteristic Agricultural Products in Luzuo Town, China. Agriculture 2025, 15, 1360. [Google Scholar] [CrossRef]
- Lin, G.; Zhang, C.; Yang, Z.; Li, Y.; Liu, C.; Ma, L.Q. High Geological Background Concentrations of As and Cd in Karstic Soils May Not Contribute to Greater Risks to Human Health via Rice Consumption. J. Hazard. Mater. 2024, 480, 135876. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chen, H.; Fu, Z.; Wang, K. Effects of Vegetation Restoration on Soil Properties along an Elevation Gradient in the Karst Region of Southwest China. Agric. Ecosyst. Environ. 2021, 320, 107572. [Google Scholar] [CrossRef]
- Sun, R.; Wu, X. Extremes of Diurnal Temperature Range in Karst Regions: Definition, Trends and Interaction with Wind Speed. Atmos. Res. 2026, 327, 108379. [Google Scholar]
- Chai, Y.; Miao, C.; Slater, L.; Ciais, P.; Berghuijs, W.R.; Chen, T.; Huntingford, C. Underestimating Global Land Greening: Future Vegetation Changes and Their Impacts on Terrestrial Water Loss. One Earth 2025, 8, 101176. [Google Scholar] [CrossRef]
- Wen, Y.; Wang, Y.; Ji, W.; Wei, N.; Liao, Q.; Huang, D.; Meng, X.; Song, Y. Influencing Factors of Elevated Levels of Potentially Toxic Elements in Agricultural Soils from Typical Karst Regions of China. Agronomy 2023, 13, 2230. [Google Scholar] [CrossRef]
- Tan, S.; Zhang, Z.; Zhou, L.; Li, Y.; Lu, S.; Tang, C.; Yu, L. Natural Vegetation Restoration Promotes Soil Quality Improvement in Rocky Desertification Areas of Southwestern China. Plant Soil 2025. [Google Scholar] [CrossRef]
- Zhu, G.; Zhao, J.; Chen, Q.; Guo, Q.; Cheng, D.; Bijaya, G.C.D.; Li, W. The Comparative Potential of Four Compositae Plants for Phytoremediation of Karst Lead/Zinc Mine Tailings Contaminated Soil. BioResources 2022, 17, 2997–3013. [Google Scholar] [CrossRef]
- Wang, W.; Xue, J.; Zhang, L.; He, M.; Cai, R.; You, J. Phytoremediation as a Sustainable Tool to Rehabilitate Land Contaminated by High-Density Sludge Sediment: From Waste to Green. Plant Soil 2025, 513, 2847–2867. [Google Scholar] [CrossRef]
- Han, Y.; Wang, H.; Zhang, G.; Zhang, S.; Liu, X.; Liu, L. Distribution, Ecological Risk Assessment and Source Identification of Pollutants in Soils of Different Land-Use Types in Degraded Wetlands. PeerJ 2022, 10, e12885. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Zhu, S.; Zhao, B.; Li, W.; Gao, X.; Wang, X. Effect of Land Use Conversion on Surface Soil Heavy Metal Contamination in a Typical Karst Plateau Lakeshore Wetland of Southwest China. Int. J. Environ. Res. Public Health 2020, 17, 84. [Google Scholar] [CrossRef]
- Zhang, B.; Hu, G.; Xu, C.; Hu, C.; Zhong, C.; Chen, S.; Zhang, Z. Effects of Natural Vegetation Restoration on Soil Physicochemical Properties in Tropical Karst Areas, Southwestern China. Forests 2024, 15, 1270. [Google Scholar] [CrossRef]
- Jia, P.; Liang, J.; Yang, S.; Zhang, S.; Liu, J.; Liang, Z.; Li, F.; Zeng, Q.; Fang, Z.; Liao, B.; et al. Plant Diversity Enhances the Reclamation of Degraded Lands by Stimulating Plant–Soil Feedbacks. J. Appl. Ecol. 2020, 57, 1258–1270. [Google Scholar] [CrossRef]
- Sun, C.; Li, F.; He, X.; Qian, Z.; Qin, Y. Positive Effects of Plant Species Diversity on Organic Carbon Accumulation in Soil Aggregates Driven by Mineral Protection in a Subtropical Forest in Southwest China. J. Soils Sediments 2024, 24, 1082–1091. [Google Scholar] [CrossRef]
- Zhu, G.; Shangguan, Z.; Deng, L. Dynamics of Water-Stable Aggregates Associated Organic Carbon Assessed from Delta C-13 Changes Following Temperate Natural Forest Development in China. Soil Tillage Res. 2021, 205, 104782. [Google Scholar] [CrossRef]
- Wang, J.; Lu, X.; Zhang, J.; Ouyang, Y.; Wei, G.; Xiong, Y. Rice Intercropping with Alligator Flag (Thalia dealbata): A Novel Model to Produce Safe Cereal Grains While Remediating Cadmium Contaminated Paddy Soil. J. Hazard. Mater. 2020, 394, 122505. [Google Scholar] [CrossRef]
- Wieczorek, J.; Baran, A.; Bubak, A. Mobility, Bioaccumulation in Plants, and Risk Assessment of Metals in Soils. Sci. Total Environ. 2023, 882, 163574. [Google Scholar] [CrossRef]
- Wu, Q.; Zheng, W.; Rao, C.; Wang, E.; Yan, W. Soil Quality Assessment and Management in Karst Rocky Desertification Ecosystem of Southwest China. Forests 2022, 13, 1513. [Google Scholar] [CrossRef]
- Zheng, W.; Wu, Q.; Guo, X.; Zhou, P.; Wu, J.; Yan, W. Rocky Desertification Succession Alters Soil Microbial Communities and Survival Strategies in the Karst Context. Sci. Total Environ. 2024, 927, 172171. [Google Scholar] [CrossRef]
- Hu, Z.; Wu, Z.; Luo, W.; Liu, S.; Tu, C. Spatial Distribution, Risk Assessment, and Source Apportionment of Soil Heavy Metals in a Karst County Based on Grid Survey. Sci. Total Environ. 2024, 953, 176049. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.; Xiao, H.; Guo, Q.; Song, B.; Zheng, G.; Zhang, Z.; Zhao, J.; Okoli, C.P. Heavy Metal Contents and Enrichment Characteristics of Dominant Plants in Wasteland of the Downstream of a Lead-Zinc Mining Area in Guangxi, Southwest China. Ecotoxicol. Environ. Saf. 2018, 151, 266–271. [Google Scholar] [CrossRef] [PubMed]
- Sharma, J.K.; Kumar, N.; Singh, N.P.; Santal, A.R. Phytoremediation Technologies and Their Mechanism for Removal of Heavy Metal from Contaminated Soil: An Approach for a Sustainable Environment. Front. Plant Sci. 2023, 14, 1076876. [Google Scholar] [CrossRef]
- Xu, T.; Wu, X.; Tian, Y.; Li, Y.; Zhang, W.; Zhang, C. Soil Property Plays a Vital Role in Vegetation Drought Recovery in Karst Region of Southwest China. J. Geophys. Res. Biogeosci. 2021, 126, e2021JG006544. [Google Scholar] [CrossRef]
- Zou, X.; Yao, K.; Zeng, Z.; Zeng, F.; Lu, L.; Zhang, H. Effect of Different Vegetation Restoration Patterns on Community Structure and Co-Occurrence Networks of Soil Fungi in the Karst Region. Front. Plant Sci. 2024, 15, 1440951. [Google Scholar] [CrossRef]
- Huang, Y.; Ma, R.; Shi, H.; Li, J.; Tu, S. Centennial Lake Environmental Evolution Reflected by Diatoms in Yilong Lake, Yunnan Province, China. Appl. Sci. 2023, 13, 5288. [Google Scholar] [CrossRef]
- Xiao, C.; You, R.; Zhu, N.; Mi, X.; Gao, L.; Zhou, X.; Zhou, G. Variation of Soil Physicochemical Properties of Different Vegetation Restoration Types on Subtropical Karst Area in Southern China. PLoS ONE 2023, 18, e0282620. [Google Scholar] [CrossRef]
- Choi, M.K.; Azeez, B.S.; Lee, S.W.; Li, W.Y.; Choi, S.; Choi, I.-Y.; Choi, K.-Y.; Na, J.-K. Transcriptome Analysis of a Tropical Medicinal Plant, Pistacia weinmannifolia. J. Plant Biotechnol. 2024, 51, 4. [Google Scholar] [CrossRef]
- Song, X.; Yuan, Z.-Q.; Fang, C.; Hu, Z.-H.; Li, F.-M.; Sardans, J.; Penuelas, J. The Formation of Humic Acid and Micro-Aggregates Facilitated Long-Time Soil Organic Carbon Sequestration after Medicago Sativa L. Introduction on Abandoned Farmlands. Geoderma 2024, 445, 116889. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, L. Advances in Basic Biology of Alfalfa (Medicago sativa L.): A Comprehensive Overview. Hortic. Res. 2025, 12, uhaf081. [Google Scholar] [CrossRef]
- Chen, X.W.; Wong, J.T.F.; Wang, J.-J.; Wong, M.H. Vetiver Grass-Microbe Interactions for Soil Remediation. Crit. Rev. Environ. Sci. Technol. 2021, 51, 897–938. [Google Scholar] [CrossRef]
- Motsomane, N.; Magadlela, A. Eragrostis Curvula Cultivars Improve Soil Bacterial Diversity, Extracellular Enzyme Activities, and Nutrition in Grassland Ecosystem Soils. Sci. Rep. 2025, 15, 35898. [Google Scholar] [CrossRef]
- Ren, L.; Huo, J.; Xiang, X.; Pan, Y.; Li, Y.; Wang, Y.; Meng, D.; Yu, C.; Chen, Y.; Xu, Z. Environmental Conditions Are the Dominant Factor Influencing Stability of Terrestrial Ecosystems on the Tibetan Plateau. Commun. Earth Environ. 2023, 4, 196. [Google Scholar] [CrossRef]
- Chen, X.; Hein, P.P.; Shi, M.; Yang, F.; Yang, J.; Fu, Y.; Yang, X. Diversity and Traditional Knowledge Concerning Fodder Plants Are Invaluable Assets for Enhancing the Sustainable Management of Crop-Livestock System of Zhaotong City in the Mountainous Southwest China. Plant Divers. 2024, 47, 311–322. [Google Scholar] [CrossRef]
- Xu, G.; Mo, Q.; Li, Z.; Qin, W.; Dong, R.; Zhao, X.; Chen, C.; Gunina, A.; Shurpali, N.; Thentu, T.L.; et al. Integrated Physical–Chemical Mechanisms Drive Carbon Stabilization under Conservation Tillage in Karst Agroecosystems. CATENA 2025, 261, 109582. [Google Scholar] [CrossRef]
- Chang, H.; Zhang, C.; Zang, S.; Zhu, Y.; Zhao, W.; Qu, Y.; Wang, H. Study on the Influencing Mechanism of Biochar and Ethylenediaminetetraacetic Acid Combination on the Remediation of Cd Polluted Soil by Sedum alfredii Hance. Environ. Technol. Innov. 2024, 36, 103875. [Google Scholar] [CrossRef]
- Ou, H.-B.; Liu, X.-S.; Wei, S.-X.; Jiang, Y.; Gao, F.; Wang, Z.-H.; Fu, W.; Du, H. The Effects of Different Vegetation Restoration Models on Soil Quality in Karst Areas of Southwest China. Forests 2024, 15, 1061. [Google Scholar] [CrossRef]
- Kama, R.; Li, S.; Nabi, F.; Aidara, M.; Huang, P.; Li, Z.; Diatta, S.; Ma, C.; Li, H. Hyperaccumulators’ Diversity Enhances Cd-Contaminated Soil Restoration and Reduces Rice Cd Uptake under an Intercropping System. ACS Omega 2024, 9, 28784–28790. [Google Scholar] [CrossRef]
- Zeng, P.; Guo, Z.; Xiao, X.; Peng, C. Dynamic Response of Enzymatic Activity and Microbial Community Structure in Metal(Loid)-Contaminated Soil with Tree-Herb Intercropping. Geoderma 2019, 345, 5–16. [Google Scholar] [CrossRef]
- Guo, N.; Xie, M.; Fang, Z.; Jiao, F.; Han, X. Divergent Responses of Plant Biomass and Diversity to Short-Term Nitrogen and Phosphorus Addition in Three Types of Steppe in Inner Mongolia, China. Ecol. Process. 2022, 11, 32. [Google Scholar] [CrossRef]
- Cai, Q.Y.; Li, B.; Cai, M.T.; Liu, Y.Z.; Wu, L.; Ge, G. Diversity and Distribution of Bryophytes in Ion-Type Rare Earth Mines in Southern Jiangxi Province, Southeast China, and the Implications for Vegetation Restoration. Plant Soil 2023, 484, 79–94. [Google Scholar]
- Marabesi, A.O.; Lessl, J.T.; Coolong, T.W. Cadmium Bioconcentration and Translocation Potential in Day Neutral and Photoperiod Sensitive Hemp Grown Hydroponically for the Medicinal Market. Water 2023, 15, 2176. [Google Scholar] [CrossRef]
- Roebuck, C.J.; Klink, M.J. Phytoremediation Potential of Hemp in Metal-Contaminated Soils: Soil Analysis, Metal Uptake, and Growth Dynamics. Processes 2025, 13, 3384. [Google Scholar] [CrossRef]
- Wang, C.; Wang, X.; Zhang, Y.; Morrissey, E.; Liu, Y.; Sun, L.; Qu, L.; Sang, C.; Zhang, H.; Li, G.; et al. Integrating Microbial Community Properties, Biomass and Necromass to Predict Cropland Soil Organic Carbon. ISME Commun. 2023, 3, 86. [Google Scholar] [CrossRef]
- Xiong, P.; He, C.; Oh, K.; Chen, X.; Liang, X.; Liu, X.; Cheng, X.; Wu, C.; Shi, Z. Medicago sativa L. Enhances the Phytoextraction of Cadmium and Zinc by Ricinus communis L. on Contaminated Land in Situ. Ecol. Eng. 2018, 116, 61–66. [Google Scholar] [CrossRef]
- Ketaubon, P.; Ritthikasem, N.; Tanheng, P.; Prapagdee, B. Enhancing Heavy Metal Phytoremediation in Landfill Soil by Chrysopogon zizanioides (L.) Roberty through the Application of Bacterial-Biochar Pellets. Environ. Technol. Innov. 2024, 35, 103738. [Google Scholar] [CrossRef]
- Li, S.; Wan, L.; Nie, Z.; Li, X. Fractal and Topological Analyses and Antioxidant Defense Systems of Alfalfa (Medicago sativa L.) Root System under Drought and Rehydration Regimes. Agronomy 2020, 10, 805. [Google Scholar] [CrossRef]
- Holanda, F.S.R.; Santos, L.D.V.; Pedrotti, A.; De Araújo Filho, R.N.; Sartor, L.R.; Santos-Sobrinho, V.R.A.; De Jesus, R.J.S.; De Oliveira Silva, P.A.; Andrade, K.M.A. Evaluation of the Root System of Vetiver Grass (Chrysopogon zizanioides L. Roberty) Using Different Sampling Methods. Environ. Syst. Res. 2022, 11, 16. [Google Scholar] [CrossRef]
- Gomes, P.I.A.; Asaeda, T. Spatial and Temporal Heterogeneity of Eragrostis curvula in the Downstream Flood Meadow of a Regulated River. Ann. Limnol. Int. J. Limnol. 2009, 45, 181–193. [Google Scholar] [CrossRef]
- Williams, L.J.; Butler, E.E.; Cavender-Bares, J.; Stefanski, A.; Rice, K.E.; Messier, C.; Paquette, A.; Reich, P.B. Enhanced Light Interception and Light Use Efficiency Explain Overyielding in Young Tree Communities. Ecol. Lett. 2021, 24, 996–1006. [Google Scholar] [CrossRef]
- Pacheco-Insausti, M.C.; Ponce, I.T.; Quiñones, M.A.; Pedranzani, H.E.; Pueyo, J.J. Effects of Inoculation with Stress-Tolerant Rhizobia on the Response of Alfalfa (Medicago sativa L.) to Combined Salinity and Cadmium Stress. Plants 2023, 12, 3972. [Google Scholar] [CrossRef]
- Phusantisampan, T.; Meeinkuirt, W.; Saengwilai, P.; Pichtel, J.; Chaiyarat, R. Phytostabilization Potential of Two Ecotypes of Vetiveria Zizanioides in Cadmium-Contaminated Soils: Greenhouse and Field Experiments. Environ. Sci. Pollut. Res. 2016, 23, 20027–20038. [Google Scholar] [CrossRef]
- Abaga, N.O.Z.; Dousset, S.; Munier-Lamy, C. Phytoremediation Potential of Vetiver Grass (Vetiveria zizanioides) in Two Mixed Heavy Metal Contaminated Soils from the Zoundweogo and Boulkiemde Regions of Burkina Faso (West Africa). J. Geosci. Environ. Prot. 2021, 9, 73–88. [Google Scholar] [CrossRef]
- Wu, B.; Li, J.; Peng, D.; Wang, Z.; Xu, H. Cadmium Exposure Alters Rhizospheric Microbial Community and Transcriptional Expression of Vetiver Grass. Front. Plant Sci. 2022, 13, 808844. [Google Scholar] [CrossRef] [PubMed]
- Ng, C.C.; Abas, M.R. Tolerance Threshold and Phyto-Assessment of Cadmium and Lead in Vetiver Grass, Vetiveria zizanioides (Linn.) Nash. Chiang Mai J. Sci. 2017, 44, 1367–1378. [Google Scholar]
- Cheng, L.; Li, Z.; Zhou, L.; Xie, J.; Zhou, Q.; Ding, M.; Wang, P.; Zhang, H.; Nie, M.; Huang, G. Rhizosphere Microbiota Modulate Cadmium Mobility Dynamics and Phytotoxicity in Rice under Differential Cd Stress. Plant Soil 2025, 516, 1455–1469. [Google Scholar] [CrossRef]
- Lan, J.; Jiang, Y.; Huang, M. Vegetation Restoration Enhances Soil Organic Carbon Accumulation in Southwest China’s Karst Region: The Role of Aggregation, Calcium, and Microbes. CATENA 2025, 257, 109184. [Google Scholar] [CrossRef]
- Chen, J.; Hao, W.; Shi, Y.; Chen, L.; Li, H.; Zhao, Z.; Mo, M.; Li, T. Phytomanagement with Forage Grasses for Sustainable Remediation of Contaminated Tailings Soil: Enhancing Soil Functionality and Addressing Forage Safety Risks. Environ. Technol. Innov. 2025, 40, 104443. [Google Scholar] [CrossRef]
- Yuan, X.; Guo, Z.; Wang, S.; Zhao, L.; Yuan, M.; Gao, Y.; Huang, L.; Liu, C.; Duan, C. Drivers and Mechanisms of Spontaneous Plant Community Succession in Abandoned Pb Zn Mining Areas in Yunnan, China. Sci. Total Environ. 2023, 904, 166871. [Google Scholar] [CrossRef]
- He, B.; Li, Q.; Zou, S.; Bai, X.; Li, W.; Chen, Y. Dynamic Changes of Soil Microbial Communities during the Afforestation of Pinus Armandii in a Karst Region of Southwest China. Microb. Ecol. 2024, 87, 36. [Google Scholar] [CrossRef]
- Peng, X.; Dai, Q.; Ding, G.; Shi, D.; Li, C. The Role of Soil Water Retention Functions of Near-Surface Fissures with Different Vegetation Types in a Rocky Desertification Area. Plant Soil 2019, 441, 587–599. [Google Scholar] [CrossRef]
- Su, Y.; Cui, Y.-J.; Dupla, J.-C.; Canou, J. Soil-Water Retention Behaviour of Fine/Coarse Soil Mixture with Varying Coarse Grain Contents and Fine Soil Dry Densities. Can. Geotech. J. 2022, 59, 291–299. [Google Scholar] [CrossRef]
- Dong, L.; Liu, Y.; Wu, J.; Liao, Y.; Li, J.; Yu, J.; Wang, S.; Yu, Z.; Shangguan, Z.; Deng, L. The Distribution of Soil C and N along the Slope Is Regulated by Vegetation Type on the Loess Plateau. CATENA 2023, 226, 107094. [Google Scholar] [CrossRef]
- Zhang, S.; Xiao, Z.; Huo, J.; Zhang, H. Key Factors Influencing on Vegetation Restoration in the Gullies of the Mollisols. J. Environ. Manag. 2021, 299, 113704. [Google Scholar] [CrossRef]
- Lu, Z.-X.; Wang, P.; Ou, H.-B.; Wei, S.-X.; Wu, L.-C.; Jiang, Y.; Wang, R.-J.; Liu, X.-S.; Wang, Z.-H.; Chen, L.-J.; et al. Effects of Different Vegetation Restoration on Soil Nutrients, Enzyme Activities, and Microbial Communities in Degraded Karst Landscapes in Southwest China. For. Ecol. Manag. 2022, 508, 120002. [Google Scholar] [CrossRef]
- Angst, Š.; Angst, G.; Mueller, K.E.; Lange, M.; Eisenhauer, N. Un(Der)Explored Links between Plant Diversity and Particulate and Mineral-Associated Organic Matter in Soil. Nat. Commun. 2025, 16, 5548. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Xing, X.; Bai, J.; Li, D. Soil Aggregate Structure, Stability, and Stoichiometric Characteristics in a Smelter-Impacted Soil under Phytoremediation. Front. Environ. Sci. 2022, 10, 900147. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, X.; Li, Z.; Liu, M.; Xu, C.; Zhang, R.; Luo, W. Effects of Vegetation Restoration on Soil Quality in Degraded Karst Landscapes of Southwest China. Sci. Total Environ. 2019, 650, 2657–2665. [Google Scholar] [CrossRef]
- Yan, Z.; Zhou, J.; Liu, C.; Jia, R.; Mganga, K.Z.; Yang, L.; Yang, Y.; Peixoto, L.; Zang, H.; Zeng, Z. Legume-Based Crop Diversification Reinforces Soil Health and Carbon Storage Driven by Microbial Biomass and Aggregates. Soil Tillage Res. 2023, 234, 105848. [Google Scholar] [CrossRef]
- Liu, L.; Zhu, Q.; Wen, D.; Yang, L.; Ni, K.; Xu, X.; Cao, J.; Meng, L.; Yang, J.; Zhou, J.; et al. Stimulation of Organic N Mineralization by N–acquiring Enzyme Activity Alleviates Soil Microbial N Limitation Following Afforestation in Subtropical Karst Areas. Plant Soil 2024, 504, 879–894. [Google Scholar] [CrossRef]
- Wang, S.; Li, T.; Yuan, X.; Yu, J.; Luan, Z.; Guo, Z.; Yu, Y.; Liu, C.; Duan, C. Biotic and Abiotic Drivers of Soil Carbon, Nitrogen and Phosphorus and Metal Dynamic Changes during Spontaneous Restoration of Pb–Zn Mining Wastelands. J. Hazard. Mater. 2025, 490, 137818. [Google Scholar] [CrossRef]
- Wang, Y.; Jing, J.; Li, Y.; Zhang, Y.; Liu, Y. Bioavailability and Speciation of Potentially Toxic Trace Metals in Limestone-Derived Soils in a Karst Region, Southwestern China. Water. Air. Soil Pollut. 2025, 236, 169. [Google Scholar] [CrossRef]
- Li, J.; Huang, C.; Huang, Z.; Wang, X.; Luo, J.; Feng, S.; Yang, Z. Exploring the Geochemical Characteristics, Sources, Influencing Factors, and Potential Remediation Strategies of Cd in a Typical Karst Region. Environ. Earth Sci. 2024, 83, 514. [Google Scholar] [CrossRef]
- Zheng, W.; Guo, X.; Zhou, P.; Tang, L.; Lai, J.; Dai, Y.; Yan, W.; Wu, J. Vegetation Restoration Enhancing Soil Carbon Sequestration in Karst Rocky Desertification Ecosystems: A Meta-Analysis. J. Environ. Manag. 2024, 370, 122530. [Google Scholar] [CrossRef]
- Liu, Y.; Qiu, H.; Wang, N.; Yang, D.; Zhao, K.; Yang, G.; Huangfu, W.; Luo, W. Thermokarst Disturbance Responses to Climate Change across the Circumpolar Permafrost Regions from 1990 to 2023. Geosci. Front. 2025, 16, 102147. [Google Scholar] [CrossRef]
- Yu, L.; Liu, Y.; Liu, T.; Yan, F. Impact of Recent Vegetation Greening on Temperature and Precipitation over China. Agric. For. Meteorol. 2020, 295, 108197. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhen, Q.; Ma, W.; Jia, J.; Li, P.; Zhang, X. Dynamic Responses of Soil Aggregate-Associated Organic Carbon and Nitrogen to Different Vegetation Restoration Patterns in an Agro-Pastoral Ecotone in Northern China. Ecol. Eng. 2023, 189, 106895. [Google Scholar] [CrossRef]
- Huo, W.; Zou, R.; Wang, L.; Guo, W.; Zhang, D.; Fan, H. Effect of Different Forms of N Fertilizers on the Hyperaccumulator Solanum Nigrum L. and Maize in Intercropping Mode under Cd Stress. RSC Adv. 2018, 8, 40210–40218. [Google Scholar] [CrossRef] [PubMed]
- Hu, P.; Yin, Y.-G.; Ishikawa, S.; Suzui, N.; Kawachi, N.; Fujimaki, S.; Igura, M.; Yuan, C.; Huang, J.; Li, Z.; et al. Nitrate Facilitates Cadmium Uptake, Transport and Accumulation in the Hyperaccumulator Sedum Plumbizincicola. Environ. Sci. Pollut. Res. 2013, 20, 6306–6316. [Google Scholar] [CrossRef] [PubMed]
- Bai, Z.; Li, D.; Zhu, L.; Tang, X.; Wang, Y.; Mao, R.; Wu, J. Nitrate Increases Cadmium Accumulation in Sweet Sorghum for Improving Phytoextraction Efficiency Rather than Ammonium. Front. Plant Sci. 2021, 12, 643116. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Zhang, J.; Hu, J.; Wang, H.; Zeng, Y.; Wang, Y.; Huang, P.; Deng, H.; Dahlgren, R.A.; Gao, H.; et al. Simultaneous Suppression of as Mobilization and N2O Emission from NH4+/as-Rich Paddy Soils by Combined Nitrate and Birnessite Amendment. J. Hazard. Mater. 2024, 465, 133451. [Google Scholar] [CrossRef]
- Liu, S.; Lin, Z.; Duan, X.; Deng, Y. Effects of Soil Microorganisms on Aggregate Stability during Vegetation Recovery in Degraded Granitic Red Soil Areas. Appl. Soil Ecol. 2024, 204, 105734. [Google Scholar] [CrossRef]







| Treatment | Code | Sample |
|---|---|---|
| Natural recover | NR | S1, S5, S8 |
| Pistacia weinmanniifolia J. Poiss. + Medicago sativa cv. WL525HQ + Chrysopogon zizanioides (L.) Roberty | PMC | S2, S4, S7 |
| Pistacia weinmanniifolia J. Poiss. + Medicago sativa cv. WL525HQ. + Eragrostis curvula (Schrad.) Nees | PME | S3, S6, S9 |
| Plant Species and Community Characteristics | NR | PMC | PME | ||||||
|---|---|---|---|---|---|---|---|---|---|
| S1 | S5 | S8 | S2 | S4 | S7 | S3 | S6 | S9 | |
| Imperata cylindrica (L.) Raeusch. | 17 | 19 | 6 | – | 8 | 4 | – | 5 | – |
| Miscanthus sinensis Anderss. | 5 | 4 | – | 10 | – | – | – | – | 6 |
| Bidens pilosa L. | 3 | – | 12 | 2 | – | – | 2 | – | – |
| Medicago sativa cv. WL525HQ | – | – | – | 36 | 29 | 43 | 44 | 40 | 47 |
| Eragrostis curvula (Schrad.) Nees | – | – | – | – | – | – | 24 | 18 | 20 |
| Chrysopogon zizanioides (L.) Roberty | – | – | – | 19 | 16 | 21 | – | – | – |
| Kummerowia striata (Thunb.) Schindl. | – | – | – | – | – | 3 | – | – | – |
| Erigeron canadensis L. | – | – | 6 | 6 | – | – | 13 | – | 11 |
| Praxelis clematidea | – | – | – | – | – | 5 | – | – | – |
| Pistacia weinmanniifolia J. Poiss. | – | – | – | 2 | 2 | 2 | 2 | 3 | 3 |
| Leucaena leucocephala (Lam.) de Wit | – | – | – | – | – | 1 | – | – | 1 |
| Photinia serratifolia (Desf.) Kalkman | – | – | – | – | 1 | – | 2 | – | – |
| Salix myrtillacea Andersson | – | – | – | 1 | – | – | – | – | – |
| Breynia fruticosa L. | – | – | – | – | – | – | – | – | – |
| Oxalis corniculata L. | – | – | – | – | 5 | – | – | – | – |
| Heteropogon contortus (L.) Beauv. ex Roem. & Schulz. | – | – | – | – | – | – | 6 | – | – |
| Inula cappa (Buch.-Ham. ex D.Don) DC. | – | – | – | – | – | – | – | 12 | – |
| Total numbers of individuals (n, n·m−2) | 25 | 23 | 24 | 76 | 61 | 80 | 93 | 78 | 88 |
| Factors | NR | PMC | PME |
|---|---|---|---|
| BD (g cm−3) | 1.64 ± 0.12 a | 1.27 ± 0.06 b | 1.35 ± 0.14 b |
| CP (%) | 16.6 ± 1.2 c | 23.5 ± 0.4 a | 21.1 ± 1.1 b |
| NCP (%) | 6.6 ± 0.9 a | 7.0 ± 0.7 a | 7.5 ± 0.7 a |
| TPO (%) | 23.1 ± 0.5 c | 30.5 ± 0.3 a | 28.6 ± 1.4 b |
| SOM (g·kg−1) | 10.21 ± 0.09 b | 13.59 ± 1.08 a | 10.05 ± 0.27 b |
| TN (g·kg−1) | 1.23 ± 0.04 b | 1.39 ± 0.02 a | 1.34 ± 0.07 a |
| TP (g·kg−1) | 0.444 ± 0.022 c | 0.561 ± 0.049 b | 0.735 ± 0.058 a |
| AN (mg kg−1) | 53.45 ± 6.63 c | 114.53 ± 12.13 a | 81.00 ± 5.08 b |
| AP (mg kg−1) | 22.55 ± 1.10 c | 23.75 ± 0.72 b | 25.56 ± 1.08 a |
| NH4+–N (mg kg−1) | 7.88 ± 0.48 c | 24.41 ± 1.95 a | 15.95 ± 1.82 b |
| NO3−–N (mg kg−1) | 27.66 ± 2.69 c | 47.88 ± 2.10 a | 41.77 ± 1.12 b |
| Total Cd (mg kg−1) | 0.156 ± 0.024 a | 0.079 ± 0.009 c | 0.101 ± 0.014 b |
| Available Cd (mg kg−1) | 0.042 ± 0.007 a | 0.017 ± 0.003 c | 0.027 ± 0.003 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Xing, Y.; Zhang, L.; Mei, Z.; Wang, X.; Li, C.; Li, Z.; Li, Y. Vegetation Restoration in Karst Southwest China: Effects of Plant Community Diversity and Soil Physicochemical Properties on Soil Cadmium. Toxics 2026, 14, 102. https://doi.org/10.3390/toxics14010102
Xing Y, Zhang L, Mei Z, Wang X, Li C, Li Z, Li Y. Vegetation Restoration in Karst Southwest China: Effects of Plant Community Diversity and Soil Physicochemical Properties on Soil Cadmium. Toxics. 2026; 14(1):102. https://doi.org/10.3390/toxics14010102
Chicago/Turabian StyleXing, Yun, Lin Zhang, Zhuoyi Mei, Xiuwen Wang, Chao Li, Zuran Li, and Yuan Li. 2026. "Vegetation Restoration in Karst Southwest China: Effects of Plant Community Diversity and Soil Physicochemical Properties on Soil Cadmium" Toxics 14, no. 1: 102. https://doi.org/10.3390/toxics14010102
APA StyleXing, Y., Zhang, L., Mei, Z., Wang, X., Li, C., Li, Z., & Li, Y. (2026). Vegetation Restoration in Karst Southwest China: Effects of Plant Community Diversity and Soil Physicochemical Properties on Soil Cadmium. Toxics, 14(1), 102. https://doi.org/10.3390/toxics14010102