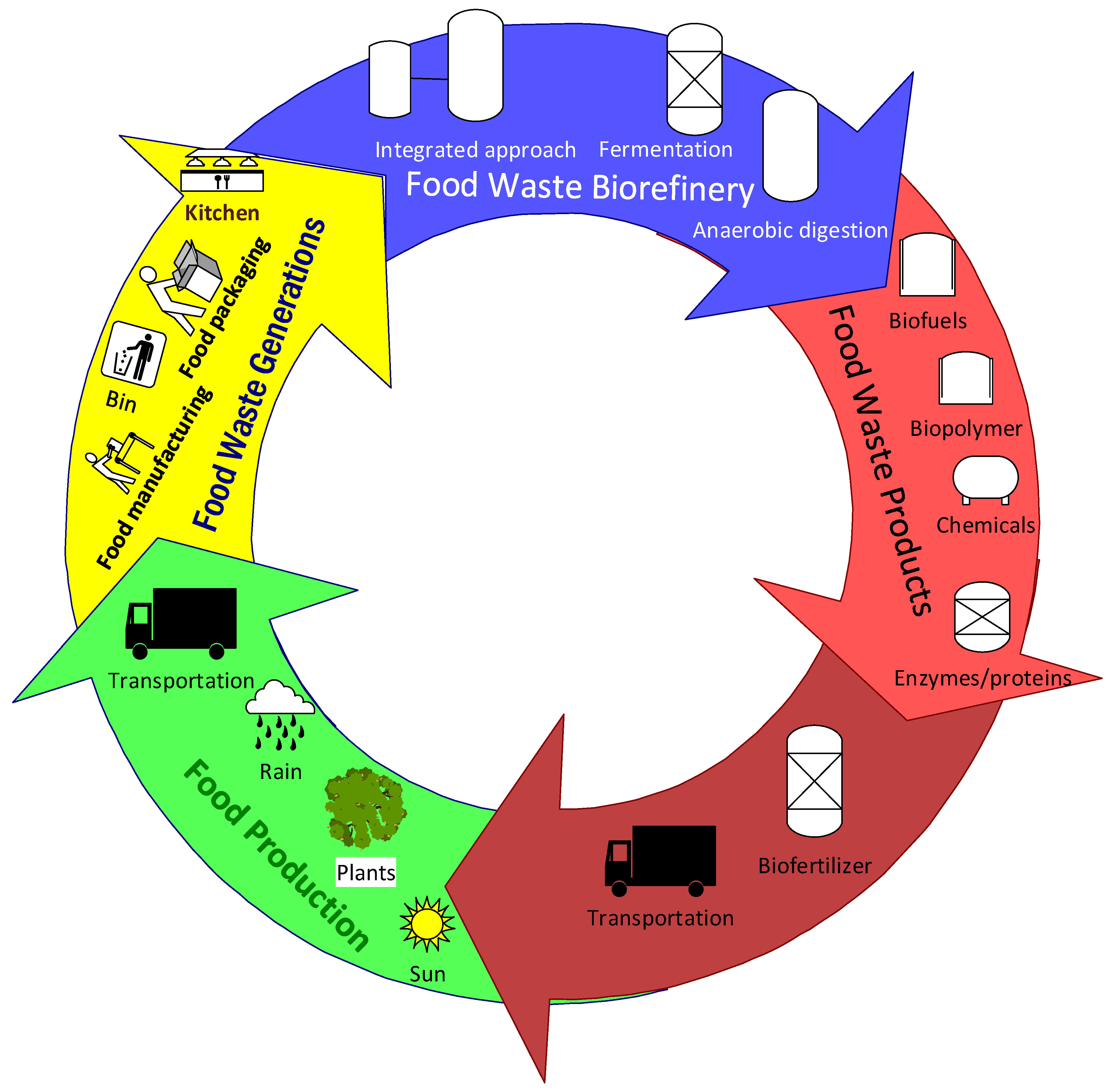
Food Waste Biorefinery: Pathway towards Circular Bioeconomy
Abstract
1. Introduction
2. Food Waste Generations
3. Impact of Food Waste on the Environment
4. Food Waste Biorefinery
4.1. Bioconversion Processes
4.1.1. Anaerobic Digestion
4.1.2. Dark Fermentation
4.1.3. Electro-Fermentation
4.1.4. Photofermentation
4.2. Integrated Approach
5. Food Waste Biorefinery Products
5.1. Biofuels
5.2. Platform Chemicals
5.3. Biopolymers
5.4. Bio-Based Proteins and Enzymes
5.5. Bio-Based Fertilizers
5.6. Other Bio-Based Compounds and Materials
6. Contributions of Food Wastes for Bioeconomy
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- FAO. Global Food Losses and Food Waste—Extent, Causes and Prevention; FAO: Rome, Italy, 2011; ISBN 9781788975391. [Google Scholar]
- Baiano, A. Recovery of biomolecules from food wastes—A review. Molecules 2014, 19, 14821–14842. [Google Scholar] [CrossRef] [PubMed]
- UN. Transforming Our World: The 2030 Agenda for Sustainable Development Preamble; United Nations: New York, NY, USA, 2015; ISBN 9781138029415. [Google Scholar]
- Ong, K.L.; Kaur, G.; Pensupa, N.; Uisan, K.; Lin, C.S.K. Trends in food waste valorization for the production of chemicals, materials and fuels: Case study South and Southeast Asia. Bioresour. Technol. 2018, 248, 100–112. [Google Scholar] [CrossRef] [PubMed]
- Matharu, A.S.; de Melo, E.M.; Houghton, J.A. Opportunity for high value-added chemicals from food supply chain wastes. Bioresour. Technol. 2016, 215, 123–130. [Google Scholar] [CrossRef] [PubMed]
- EU. A Sustainable Bioeconomy for Europe: Strengthening the Connection between Economy, Society and the Environment; European Commission: Brussels, Belgium, 2018. [Google Scholar]
- EU. Closing the Loop—An EU Action Plan for the Circular Economy; Europe Union: Brussels, Belgium, 2015. [Google Scholar]
- Cristóbal, J.; Caldeira, C.; Corrado, S.; Sala, S. Techno-economic and profitability analysis of food waste biorefineries at European level. Bioresour. Technol. 2018, 259, 244–252. [Google Scholar] [CrossRef] [PubMed]
- United Nations Environment Programme. Food Waste Index; United Nations: Nairobi, Kenya, 2021; ISBN 9789280738513. [Google Scholar]
- Chalak, A.; Abou-Daher, C.; Chaaban, J.; Abiad, M.G. The global economic and regulatory determinants of household food waste generation: A cross-country analysis. Waste Manag. 2016, 48, 418–422. [Google Scholar] [CrossRef] [PubMed]
- FAO. Food Wastage Footprint: Fool Cost–Accounting; FAO: Rome, Italy, 2014; ISBN 978-92-5-108512-7. [Google Scholar]
- United States Environmental Protection Agency. 2018 Wasted Food Report; EPA: Washington, DC, USA, 2018.
- FUSIONS. Estimates of European Food Waste Levels; European Commission: Brussels, Belgium, 2016. [Google Scholar]
- Corrado, S.; Sala, S. Food waste accounting along global and European food supply chains: State of the art and outlook. Waste Manag. 2018, 79, 120–131. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Cheng, S.; Liu, X.; Cao, X.; Xue, L.; Liu, G. Plate waste in school lunch programs in Beijing, China. Sustainability 2016, 8, 1288. [Google Scholar] [CrossRef]
- Wang, L.E.; Liu, G.; Liu, X.; Liu, Y.; Gao, J.; Zhou, B.; Gao, S.; Cheng, S. The weight of unfinished plate: A survey based characterization of restaurant food waste in Chinese cities. Waste Manag. 2017, 66, 3–12. [Google Scholar] [CrossRef]
- De Clercq, D.; Wen, Z.; Gottfried, O.; Schmidt, F.; Fei, F. A review of global strategies promoting the conversion of food waste to bioenergy via anaerobic digestion. Renew. Sustain. Energy Rev. 2017, 79, 204–221. [Google Scholar] [CrossRef]
- Garot, G. Lutte Contre Le Gaspillage Alimentaire: Propositions Pour Une Politique Publique; Prime Minster Office: Paris, France, 2015. [Google Scholar]
- Buzby, J.C.; Wells, H.F.; Hyman, J. The Estimated Amount, Value, and Calories of Postharvest Food Losses at the Retail and Consumer Levels; USDA: Washington, DC, USA, 2014; Volume EIB-121.
- Venkat, K. ClimateChangeImpactofUSFoodWaste.pdf. Int. J. Food Syst. Dyn. 2012, 2, 431–446. [Google Scholar]
- Wharton, C.; Vizcaino, M.; Berardy, A.; Opejin, A. Waste watchers: A food waste reduction intervention among households in Arizona. Resour. Conserv. Recycl. 2021, 164, 105109. [Google Scholar] [CrossRef]
- Schmidt, K. Explaining and promoting household food waste-prevention by an environmental psychological based intervention study. Resour. Conserv. Recycl. 2016, 111, 53–66. [Google Scholar] [CrossRef]
- Carmona-Cabello, M.; Garcia, I.L.; Leiva-Candia, D.; Dorado, M.P. Valorization of food waste based on its composition through the concept of biorefinery. Curr. Opin. Green Sustain. Chem. 2018, 14, 67–79. [Google Scholar] [CrossRef]
- Xu, Y.; Lu, Y.; Zheng, L.; Wang, Z.; Dai, X. Perspective on enhancing the anaerobic digestion of waste activated sludge. J. Hazard. Mater. 2020, 389, 121847. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Samadder, S.R. Performance evaluation of anaerobic digestion technology for energy recovery from organic fraction of municipal solid waste: A review. Energy 2020, 197, 117253. [Google Scholar] [CrossRef]
- Feng, L.; Chen, Y.; Zheng, X. Enhancement of waste activated sludge protein conversion and volatile fatty acids accumulation during waste activated sludge anaerobic fermentation by carbohydrate substrate addition: The effect of pH. Environ. Sci. Technol. 2009, 43, 4373–4380. [Google Scholar] [CrossRef]
- Chernicharo, C.A.L.; van Lier, J.B.; Noyola, A.; Bressani Ribeiro, T. Anaerobic sewage treatment: State of the art, constraints and challenges. Rev. Environ. Sci. Biotechnol. 2015, 14, 649–679. [Google Scholar] [CrossRef]
- Carneiro, R.B.; Gonzalez-Gil, L.; Londoño, Y.A.; Zaiat, M.; Carballa, M.; Lema, J.M. Acidogenesis is a key step in the anaerobic biotransformation of organic micropollutants. J. Hazard. Mater. 2020, 389, 121888. [Google Scholar] [CrossRef] [PubMed]
- Mari, A.G.; Andreani, C.L.; Tonello, T.U.; Leite, L.C.C.; Fernandes, J.R.; Lopes, D.D.; Rodrigues, J.A.D.; Gomes, S.D. Biohydrogen and biomethane production from cassava wastewater in a two-stage anaerobic sequencing batch biofilm reactor. Int. J. Hydrogen Energy 2020, 45, 5165–5174. [Google Scholar] [CrossRef]
- Feng, K.; Wang, Q.; Li, H.; Zhang, Y.; Deng, Z.; Liu, J.; Du, X. Effect of fermentation type regulation using alkaline addition on two-phase anaerobic digestion of food waste at different organic load rates. Renew. Energy 2020, 154, 385–393. [Google Scholar] [CrossRef]
- Srisowmeya, G.; Chakravarthy, M.; Nandhini Devi, G. Critical considerations in two-stage anaerobic digestion of food waste—A review. Renew. Sustain. Energy Rev. 2020, 119, 109587. [Google Scholar] [CrossRef]
- Lohani, S.P.; Shakya, S.; Gurung, P.; Dhungana, B.; Paudel, D.; Mainali, B. Anaerobic co-digestion of food waste, poultry litter and sewage sludge: Seasonal performance under ambient condition and model evaluation. Energy Sources Part A Recover. Util. Environ. Eff. 2021. [Google Scholar] [CrossRef]
- Ghimire, A.; Luongo, V.; Frunzo, L.; Lens, P.N.; Pirozzi, F.; Esposito, G. Biohythane production from food waste in a two-stage process: Assessing the energy recovery potential. Environ. Technol. 2021. [Google Scholar] [CrossRef]
- Patinvoh, R.J.; Millati, R.; Sárvári-horváth, I.; Taherzadeh, M.J. Factors influencing volatile fatty acids production from food wastes via anaerobic digestion production. Bioengineered 2020, 11, 39–52. [Google Scholar] [CrossRef]
- Sarkar, O.; Santhosh, J.; Dhar, A.; Mohan, S.V. Green hythane production from food waste: Integration of dark-fermentation and methanogenic process towards biogas up- gradation. Int. J. Hydrogen Energy 2021. [Google Scholar] [CrossRef]
- Kuo, J.; Dow, J. Biogas production from anaerobic digestion of food waste and relevant air quality implications. J. Air Waste Manag. Assoc. 2017, 67, 1000–1011. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.; Srivastava, S.; Rai, P.; Duke, M. Cheese whey to biohydrogen and useful organic acids: A non-pathogenic microbial treatment by L. acidophilus. Sci. Rep. 2019, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Ghimire, A.; Frunzo, L.; Pirozzi, F.; Trably, E.; Escudie, R.; Lens, P.N.L.; Esposito, G. A review on dark fermentative biohydrogen production from organic biomass: Process parameters and use of by-products. Appl. Energy 2015, 144, 73–95. [Google Scholar] [CrossRef]
- Sinha, P.; Pandey, A. An evaluative report and challenges for fermentative biohydrogen production. Int. J. Hydrogen Energy 2011, 36, 7460–7478. [Google Scholar] [CrossRef]
- Zong, W.; Yu, R.; Zhang, P.; Fan, M.; Zhou, Z. Efficient hydrogen gas production from cassava and food waste by a two-step process of dark fermentation and. Biomass Bioenergy 2009, 33, 1458–1463. [Google Scholar] [CrossRef]
- Nguyen, M.T.; Hung, P.; Vo, T. Effect of food to microorganisms (F/M) ratio on biohythane production via single-stage dark fermentation. Int. J. Hydrogen Energy 2020. [Google Scholar] [CrossRef]
- Cabrol, L.; Marone, A.; Tapia-Venegas, E.; Steyer, J.P.; Ruiz-Filippi, G.; Trably, E. Microbial ecology of fermentative hydrogen producing bioprocesses: Useful insights for driving the ecosystem function. FEMS Microbiol. Rev. 2017, 41, 158–181. [Google Scholar] [CrossRef] [PubMed]
- Saady, N.M.C. Homoacetogenesis during hydrogen production by mixed cultures dark fermentation: Unresolved challenge. Int. J. Hydrogen Energy 2013, 38, 13172–13191. [Google Scholar] [CrossRef]
- Liu, C.G.; Xue, C.; Lin, Y.H.; Bai, F.W. Redox potential control and applications in microaerobic and anaerobic fermentations. Biotechnol. Adv. 2013, 31, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Toledo-Alarcón, J.; Fuentes, L.; Etchebehere, C.; Bernet, N.; Trably, E. Glucose electro-fermentation with mixed cultures: A key role of the Clostridiaceae family. Int. J. Hydrogen Energy 2021, 46, 1694–1704. [Google Scholar] [CrossRef]
- Toledo-Alarcón, J.; Moscoviz, R.; Trably, E.; Bernet, N. Glucose electro-fermentation as main driver for efficient H2-producing bacteria selection in mixed cultures. Int. J. Hydrogen Energy 2019, 2230–2238. [Google Scholar] [CrossRef]
- Moscoviz, R.; Toledo-Alarcón, J.; Trably, E.; Bernet, N. Electro-Fermentation: How to Drive Fermentation Using Electrochemical Systems. Trends Biotechnol. 2016, 34, 856–865. [Google Scholar] [CrossRef]
- Creasey, R.C.G.; Mostert, A.B.; Nguyen, T.A.H.; Virdis, B.; Freguia, S.; Laycock, B. Microbial nanowires—Electron transport and the role of synthetic analogues. Acta Biomater. 2018, 69, 1–30. [Google Scholar] [CrossRef]
- Thrash, J.C.; Coates, J.D. Review: Direct and indirect electrical stimulation of microbial metabolism. Environ. Sci. Technol. 2008, 42, 3921–3931. [Google Scholar] [CrossRef] [PubMed]
- Hirose, A.; Kouzuma, A.; Watanabe, K. Towards development of electrogenetics using electrochemically active bacteria. Biotechnol. Adv. 2019, 37, 107351. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, J.; Meng, J.; Wang, X. A cathodic electro-fermentation system for enhancing butyric acid production from rice straw with a mixed culture. Sci. Total Environ. 2021, 767, 145011. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Deng, Z.; Li, H.; Feng, K. Contribution of electrodes and electric current to process stability and methane production during the electro-fermentation of food waste. Bioresour. Technol. 2019, 288, 121536. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.; Li, M.; Wang, Y.; Wu, Y.; Zhu, L.; Wang, X.; Zhao, Y. Enhancement of hydrogen production and energy recovery through electro-fermentation from the dark fermentation effluent of food waste. Environ. Sci. Ecotechnology 2020, 1, 100006. [Google Scholar] [CrossRef]
- Hanipa, M.A.F.; Abdul, P.M.; Jahim, J.M.; Takriff, M.S.; Reungsang, A.; Wu, S.Y. Biotechnological approach to generate green biohydrogen through the utilization of succinate-rich fermentation wastewater. Int. J. Hydrogen Energy 2020, 45, 22246–22259. [Google Scholar] [CrossRef]
- Basak, N.; Jana, A.K.; Das, D.; Saikia, D. Photofermentative molecular biohydrogen production by purple-non-sulfur (PNS) bacteria in various modes: The present progress and future perspective. Int. J. Hydrogen Energy 2014, 39, 6853–6871. [Google Scholar] [CrossRef]
- Seifert, K.; Waligorska, M.; Laniecki, M. Hydrogen generation in photobiological process from dairy wastewater. Int. J. Hydrogen Energy 2010, 35, 9624–9629. [Google Scholar] [CrossRef]
- Laurinavichene, T.; Tekucheva, D.; Laurinavichius, K.; Tsygankov, A. Utilization of distillery wastewater for hydrogen production in one-stage and two-stage processes involving photofermentation. Enzyme Microb. Technol. 2018, 110, 1–7. [Google Scholar] [CrossRef]
- Lu, H.; Zhang, G.; He, S.; Peng, C.; Ren, Z. Production of photosynthetic bacteria using organic wastewater in photobioreactors in lieu of a culture medium in fermenters: From lab to pilot scale. J. Clean. Prod. 2020, 259, 120871. [Google Scholar] [CrossRef]
- Assawamongkholsiri, T.; Reungsang, A.; Plangkang, P.; Sittijunda, S. Repeated batch fermentation for photo-hydrogen and lipid production from wastewater of a sugar manufacturing plant. Int. J. Hydrogen Energy 2018, 43, 3605–3617. [Google Scholar] [CrossRef]
- Sinigaglia, T.; Lewiski, F.; Santos Martins, M.E.; Mairesse Siluk, J.C. Production, storage, fuel stations of hydrogen and its utilization in automotive applications-a review. Int. J. Hydrogen Energy 2017, 42, 24597–24611. [Google Scholar] [CrossRef]
- Hay, J.X.W.; Wu, T.Y.; Juan, J.C.; Jahim, J.M. Effect of adding brewery wastewater to pulp and paper mill effluent to enhance the photofermentation process: Wastewater characteristics, biohydrogen production, overall performance, and kinetic modeling. Environ. Sci. Pollut. Res. 2017, 24, 10354–10363. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Dairkee, U.K.; Chowdhury, R.; Bhattacharya, P. Hydrogen from food processing wastes via photofermentation using Purple Non-sulfur Bacteria (PNSB)—A review. Energy Convers. Manag. 2017, 141, 299–314. [Google Scholar] [CrossRef]
- Du Toit, J.P.; Pott, R.W.M. Transparent polyvinyl-alcohol cryogel as immobilisation matrix for continuous biohydrogen production by phototrophic bacteria. Biotechnol. Biofuels 2020, 13, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Radhakrishnan, R.; Banerjee, S.; Banerjee, S.; Singh, V.; Das, D. Sustainable approach for the treatment of poultry manure and starchy wastewater by integrating dark fermentation and microalgal cultivation. J. Mater. Cycles Waste Manag. 2021. [Google Scholar] [CrossRef]
- Mahata, C.; Dhar, S.; Ray, S.; Das, D. Effect of thermal pretreated organic wastes on the dark fermentative hydrogen production using mixed microbial consortia. Fuel 2021, 284, 119062. [Google Scholar] [CrossRef]
- Rambabu, K.; Bharath, G.; Thanigaivelan, A.; Das, D.B.; Show, P.L.; Banat, F. Augmented biohydrogen production from rice mill wastewater through nano-metal oxides assisted dark fermentation. Bioresour. Technol. 2021, 319, 124243. [Google Scholar] [CrossRef] [PubMed]
- Ventura, J.R.S.; Rojas, S.M.; Ventura, R.L.G.; Nayve, F.R.P.; Lantican, N.B. Potential for biohydrogen production from organic wastes with focus on sequential dark- and photofermentation: The Philippine setting. Biomass Convers. Biorefinery 2021. [Google Scholar] [CrossRef]
- Mishra, P.; Krishnan, S.; Rana, S.; Singh, L.; Sakinah, M.; Ab Wahid, Z. Outlook of fermentative hydrogen production techniques: An overview of dark, photo and integrated dark-photo fermentative approach to biomass. Energy Strateg. Rev. 2019, 24, 27–37. [Google Scholar] [CrossRef]
- Rai, P.K.; Singh, S.P. Integrated dark- and photo-fermentation: Recent advances and provisions for improvement. Int. J. Hydrogen Energy 2016, 41, 19957–19971. [Google Scholar] [CrossRef]
- Shanthi Sravan, J.; Butti, S.K.; Sarkar, O.; Vamshi Krishna, K.; Venkata Mohan, S. Electrofermentation of food waste—Regulating acidogenesis towards enhanced volatile fatty acids production. Chem. Eng. J. 2018, 334, 1709–1718. [Google Scholar] [CrossRef]
- Sarkar, O.; Kiran Katari, J.; Chatterjee, S.; Venkata Mohan, S. Salinity induced acidogenic fermentation of food waste regulates biohydrogen production and volatile fatty acids profile. Fuel 2020, 276, 117794. [Google Scholar] [CrossRef]
- Yan, B.H.; Selvam, A.; Wong, J.W.C. Bio-hydrogen and methane production from two-phase anaerobic digestion of food waste under the scheme of acidogenic off-gas reuse. Bioresour. Technol. 2020, 297, 122400. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.K.S.; Gupta, R.K.; Das, D.; Lee, J.K.; Kalia, V.C. Continuous biohydrogen production from poplar biomass hydrolysate by a defined bacterial mixture immobilized on lignocellulosic materials under non-sterile conditions. J. Clean. Prod. 2021, 287, 125037. [Google Scholar] [CrossRef]
- Ben Yahmed, N.; Dauptain, K.; Lajnef, I.; Carrere, H.; Trably, E.; Smaali, I. New sustainable bioconversion concept of date by-products (Phoenix dactylifera L.) to biohydrogen, biogas and date-syrup. Int. J. Hydrogen Energy 2021, 46, 297–305. [Google Scholar] [CrossRef]
- RamKumar, N.; Anupama, P.D.; Nayak, T.; Subudhi, S. Scale up of biohydrogen production by a pure strain; Clostridium butyricum TM-9A at regulated pH under decreased partial pressure. Renew. Energy 2021, 170, 1178–1185. [Google Scholar] [CrossRef]
- Jung, J.H.; Sim, Y.B.; Baik, J.H.; Park, J.H.; Kim, S.H. High-rate mesophilic hydrogen production from food waste using hybrid immobilized microbiome. Bioresour. Technol. 2021, 320, 124279. [Google Scholar] [CrossRef]
- Yeshanew, M.M.; Frunzo, L.; Pirozzi, F.; Lens, P.N.L.; Esposito, G. Production of biohythane from food waste via an integrated system of continuously stirred tank and anaerobic fixed bed reactors. Bioresour. Technol. 2016, 220, 312–322. [Google Scholar] [CrossRef]
- Kumar, G.; Sivagurunathan, P.; Sen, B.; Kim, S.H.; Lin, C.Y. Mesophilic continuous fermentative hydrogen production from acid pretreated de-oiled jatropha waste hydrolysate using immobilized microorganisms. Bioresour. Technol. 2017, 240, 137–143. [Google Scholar] [CrossRef]
- Fazzino, F.; Mauriello, F.; Paone, E.; Sidari, R.; Calabrò, P.S. Integral valorization of orange peel waste through optimized ensiling: Lactic acid and bioethanol production. Chemosphere 2021, 271. [Google Scholar] [CrossRef] [PubMed]
- Clementz, A.L.; Manuale, D.; Sanchez, E.; Vera, C.; Yori, J.C. Use of discards of bovine bone, yeast and carrots for producing second generation bio-ethanol. Biocatal. Agric. Biotechnol. 2019, 22, 101392. [Google Scholar] [CrossRef]
- Kastner, V.; Somitsch, W.; Schnitzhofer, W. The anaerobic fermentation of food waste: A comparison of two bioreactor systems. J. Clean. Prod. 2012, 34, 82–90. [Google Scholar] [CrossRef]
- Bolzonella, D.; Battista, F.; Cavinato, C.; Gottardo, M.; Micolucci, F.; Lyberatos, G.; Pavan, P. Recent developments in biohythane production from household food wastes: A review. Bioresour. Technol. 2018, 257, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.Z.; Xing, Y.; Yu, M.; Wang, Q. Feasibility of converting lactic acid to ethanol in food waste fermentation by immobilized lactate oxidase. Appl. Energy 2014, 129, 89–93. [Google Scholar] [CrossRef]
- Vescovi, V.; Rojas, M.J.; Baraldo, A.; Botta, D.C.; Santana, F.A.M.; Costa, J.P.; Machado, M.S.; Honda, V.K.; de Lima Camargo Giordano, R.; Tardioli, P.W. Lipase-Catalyzed Production of Biodiesel by Hydrolysis of Waste Cooking Oil Followed by Esterification of Free Fatty Acids. JAOCS J. Am. Oil Chem. Soc. 2016, 93, 1615–1624. [Google Scholar] [CrossRef]
- Moscoviz, R.; Trably, E.; Bernet, N.; Carrère, H. The environmental biorefinery: State-of-the-art on the production of hydrogen and value-added biomolecules in mixed-culture fermentation. Green Chem. 2018, 20, 3159–3179. [Google Scholar] [CrossRef]
- Iglesias, J.; Martínez-Salazar, I.; Maireles-Torres, P.; Martin Alonso, D.; Mariscal, R.; López Granados, M. Advances in catalytic routes for the production of carboxylic acids from biomass: A step forward for sustainable polymers. Chem. Soc. Rev. 2020, 49, 5704–5771. [Google Scholar] [CrossRef]
- Vidal-Antich, C.; Perez-Esteban, N.; Astals, S.; Peces, M.; Mata-Alvarez, J.; Dosta, J. Assessing the potential of waste activated sludge and food waste co-fermentation for carboxylic acids production. Sci. Total Environ. 2021, 757, 143763. [Google Scholar] [CrossRef]
- Jones, R.J.; Fernández-feito, R.; Massanet-nicolau, J.; Dinsdale, R.; Guwy, A. Continuous recovery and enhanced yields of volatile fatty acids from a continually-fed 100 L food waste bioreactor by filtration and electrodialysis. Waste Manag. 2021, 122, 81–88. [Google Scholar] [CrossRef]
- Wainaina, S.; Parchami, M.; Mahboubi, A.; Horváth, I.S. Food waste-derived volatile fatty acids platform using an immersed membrane bioreactor. Bioresour. Technol. 2019, 274, 329–334. [Google Scholar] [CrossRef]
- Yousuf, A.; Schmidt, J.E. Effect of total solid content and pretreatment on the production of lactic acid from mixed culture dark fermentation of food waste. Waste Manag. 2018, 77, 516–521. [Google Scholar] [CrossRef]
- Wu, Q.; Feng, X.; Chen, Y.; Liu, M.; Bao, X. Continuous medium chain carboxylic acids production from excess sludge by granular chain-elongation process. J. Hazard. Mater. 2021, 402, 123471. [Google Scholar] [CrossRef] [PubMed]
- Maciel, M.; Coelho, H.; Wagner, N.; Morais, S.; Jorge, T.; Ferreira, T.; Schiavon, F.; Silva, S.; Lopes, E. Carboxylic acids production using residual glycerol as a substrate in anaerobic fermentation: A kinetic modeling study. Biomass Bioenergy 2020, 143. [Google Scholar] [CrossRef]
- Ma, H.; Lin, Y.; Jin, Y.; Gao, M.; Li, H.; Wang, Q.; Ge, S.; Cai, L.; Huang, Z.; Van Le, Q.; et al. Effect of ultrasonic pretreatment on chain elongation of sacchari fi ed residue from food waste by anaerobic fermentation. Environ. Pollut. 2021, 268, 115936. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Cao, J.; Zhang, T.; Zhao, J.; Xu, R.; Zhang, Q. A novel approach of synchronously recovering phosphorus as vivianite and volatile fatty acids during waste activated sludge and food waste co- fermentation: Performance and mechanisms. Bioresour. Technol. 2020, 305, 123078. [Google Scholar] [CrossRef]
- Leite, P.; Silva, C.; Salgado, J.M.; Belo, I. Simultaneous production of lignocellulolytic enzymes and extraction of antioxidant compounds by solid-state fermentation of agro-industrial wastes. Ind. Crops Prod. 2019, 137, 315–322. [Google Scholar] [CrossRef]
- Zhou, Y.; Xu, X.Y.; Gan, R.Y.; Zheng, J.; Li, Y.; Zhang, J.J.; Xu, D.P.; Li, H. Bin Optimization of ultrasound-assisted extraction of antioxidant polyphenols from the seed coats of red sword bean (Canavalia gladiate (Jacq.) DC.). Antioxidants 2019, 8, 200. [Google Scholar] [CrossRef]
- Zhou, Y.; Zheng, J.; Gan, R.Y.; Zhou, T.; Xu, D.P.; Li, H. Bin Optimization of ultrasound-assisted extraction of antioxidants from the mung bean coat. Molecules 2017, 22, 638. [Google Scholar] [CrossRef]
- Chuyen, H.V.; Nguyen, M.H.; Roach, P.D.; Golding, J.B.; Parks, S.E. Microwave-assisted extraction and ultrasound-assisted extraction for recovering carotenoids from Gac peel and their effects on antioxidant capacity of the extracts. Food Sci. Nutr. 2018, 6, 189–196. [Google Scholar] [CrossRef]
- Moorthy, I.G.; Maran, J.P.; Ilakya, S.; Anitha, S.L.; Sabarima, S.P.; Priya, B. Ultrasound assisted extraction of pectin from waste Artocarpus heterophyllus fruit peel. Ultrason. Sonochem. 2017, 34, 525–530. [Google Scholar] [CrossRef]
- Zhang, A.Y.; Sun, Z.; Leung, C.C.J.; Han, W.; Lau, K.Y.; Li, M.; Lin, C.S.K. Valorisation of bakery waste for succinic acid production 2. Green Chem. 2013, 15, 690–695. [Google Scholar] [CrossRef]
- Sengar, A.S.; Rawson, A.; Muthiah, M.; Kalakandan, S.K. Comparison of different ultrasound assisted extraction techniques for pectin from tomato processing waste. Ultrason. Sonochem. 2020, 61, 104812. [Google Scholar] [CrossRef] [PubMed]
- Pataro, G.; Bobinaitė, R.; Bobinas, Č.; Šatkauskas, S.; Raudonis, R.; Visockis, M.; Ferrari, G.; Viškelis, P. Improving the Extraction of Juice and Anthocyanins from Blueberry Fruits and Their By-products by Application of Pulsed Electric Fields. Food Bioprocess Technol. 2017, 10, 1595–1605. [Google Scholar] [CrossRef]
- Garrido, T.; Gizdavic-Nikolaidis, M.; Leceta, I.; Urdanpilleta, M.; Guerrero, P.; de la Caba, K.; Kilmartin, P.A. Optimizing the extraction process of natural antioxidants from chardonnay grape marc using microwave-assisted extraction. Waste Manag. 2019, 88, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Koller, M. Recycling of Waste Streams of the Biotechnological Poly(hydroxyalkanoate) Production by Haloferax mediterranei on Whey. Int. J. Polym. Sci. 2015, 2015, 370164. [Google Scholar] [CrossRef]
- Poomipuk, N.; Reungsang, A.; Plangklang, P. Poly- b -hydroxyalkanoates production from cassava starch hydrolysate by Cupriavidus sp. KKU38. Int. J. Biol. Macromol. 2014, 65, 51–64. [Google Scholar] [CrossRef]
- Uranga, J.; Etxabide, A.; Guerrero, P.; Caba, K. De Development of active fi sh gelatin fi lms with anthocyanins by compression molding. Food Hydrocoll. 2018, 84, 313–320. [Google Scholar] [CrossRef]
- Araújo, C.S.; Rodrigues, A.M.C.; Joele, M.R.S.P.; Araújo, E.A.F.; Lourenço, L.F.H. Optimizing process parameters to obtain a bioplastic using proteins from fi sh byproducts through the response surface methodology. Food Packag. Shelf Life 2018, 16, 23–30. [Google Scholar] [CrossRef]
- Prieto, C.V.G.; Ramos, F.D.; Estrada, V.; Villar, M.A.; Diaz, M.S. Optimization of an integrated algae-based biore fi nery for the production of biodiesel, astaxanthin and PHB. Energy 2017, 139, 1159–1172. [Google Scholar] [CrossRef]
- Novak, M.; Koller, M.; Braunegg, G.; Horvat, P. Mathematical Modelling as a Tool for Optimized PHA Production. Chem. Biochem. Eng. Q. 2015, 29, 183–220. [Google Scholar] [CrossRef]
- Bueno, L.; Toro, C.; Martín, M. Techno-economic evaluation of the production of polyesters from glycerol and adipic acid. Chem. Eng. Res. Des. 2014, 93, 432–440. [Google Scholar] [CrossRef]
- Zepka, L.Q.; Jacob-Lopes, E.; Goldbeck, R.; Queiroz, M.I. Production and biochemical profile of the microalgae Aphanothece microscopica Nägeli submitted to different drying conditions. Chem. Eng. Process. Process Intensif. 2008, 47, 1305–1310. [Google Scholar] [CrossRef]
- Kadim, I.T.; Mahgoub, O.; Baqir, S.; Faye, B.; Purchas, R. Cultured meat from muscle stem cells: A review of challenges and prospects. J. Integr. Agric. 2015, 14, 222–233. [Google Scholar] [CrossRef]
- Aggelopoulos, T.; Katsieris, K.; Bekatorou, A.; Pandey, A.; Banat, I.M.; Koutinas, A.A. Solid state fermentation of food waste mixtures for single cell protein, aroma volatiles and fat production. Food Chem. 2014, 145, 710–716. [Google Scholar] [CrossRef] [PubMed]
- Yunus, F.U.N.; Nadeem, M.; Rashid, F. Single-cell protein production through microbial conversion of lignocellulosic residue (wheat bran) for animal feed. J. Inst. Brew. 2015, 121, 553–557. [Google Scholar] [CrossRef]
- Aruna, T.E.; Aworh, O.C.; Raji, A.O.; Olagunju, A.I. Protein enrichment of yam peels by fermentation with Saccharomyces cerevisiae (BY4743). Ann. Agric. Sci. 2017, 62, 33–37. [Google Scholar] [CrossRef]
- Sharif, M.; Zafar, M.H.; Aqib, A.I.; Saeed, M.; Farag, M.R.; Alagawany, M. Single cell protein: Sources, mechanism of production, nutritional value and its uses in aquaculture nutrition. Aquaculture 2021, 531, 735885. [Google Scholar] [CrossRef]
- Akyüz, A.; Ersus, S. Optimization of enzyme assisted extraction of protein from the sugar beet (Beta vulgaris L.) leaves for alternative plant protein concentrate production. Food Chem. 2021, 335, 127673. [Google Scholar] [CrossRef]
- Mg, G.-; Gaonkar, S.K.; Furtado, I.J. Valorization of low-cost agro-wastes residues for the maximum production of protease and lipase haloextremozymes by Haloferax lucentensis. Process Biochem. 2021, 101, 72–88. [Google Scholar] [CrossRef]
- Guan, Y.; Wang, Q.; Lv, C.; Wang, D.; Ye, X. Fermentation time-dependent pectinase activity is associated with metabolomics variation in Bacillus licheniformis DY2. Process Biochem. 2021, 101, 147–155. [Google Scholar] [CrossRef]
- Saleh, F.; Hussain, A.; Younis, T.; Ali, S.; Rashid, M.; Ali, A.; Mustafa, G.; Jabeen, F.; Al-surhanee, A.A.; Alnoman, M.M.; et al. Comparative growth potential of thermophilic amylolytic Bacillus sp. on unconventional media food wastes and its industrial application. Saudi J. Biol. Sci. 2020, 27, 3499–3504. [Google Scholar] [CrossRef]
- Debosz, K.; Petersen, S.O.; Kure, L.K.; Ambus, P. Evaluating effects of sewage sludge and household compost on soil physical, chemical and microbiological properties. Appl. Soil Ecol. 2002, 19, 237–248. [Google Scholar] [CrossRef]
- Tsai, S.; Liu, C.; Yang, S. Microbial conversion of food wastes for biofertilizer production with thermophilic lipolytic microbes. Renew. Energy 2007, 32, 904–915. [Google Scholar] [CrossRef]
- Gao, S.; Lu, D.; Qian, T.; Zhou, Y. Thermal hydrolyzed food waste liquor as liquid organic fertilizer. Sci. Total Environ. 2021, 775, 145786. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, V.; Dias-ferreira, C.; González-garcía, I.; Labrincha, J.; Horta, C. A novel approach for nutrients recovery from municipal waste as biofertilizers by combining electrodialytic and gas permeable membrane technologies. Waste Manag. 2021, 125, 293–302. [Google Scholar] [CrossRef]
- Chakravarty, I.; Mandavgane, S.A. Valorization of fruit and vegetable waste for biofertilizer and biogas. Food Process Eng. 2020, 1–8. [Google Scholar] [CrossRef]
- dos Santos Mathias, T.R.; de Aguiar, P.F.; de Almeida e Silva, J.B.; de Mello, P.P.M.; Camporese Sérvulo, E.F. Brewery waste reuse for protease production by lactic acid fermentation. Food Technol. Biotechnol. 2017, 55, 218–224. [Google Scholar] [CrossRef]
- Javed, U.; Ansari, A.; Aman, A.; Ul Qader, S.A. Fermentation and saccharification of agro-industrial wastes: A cost-effective approach for dual use of plant biomass wastes for xylose production. Biocatal. Agric. Biotechnol. 2019, 21, 101341. [Google Scholar] [CrossRef]
- Javed, U.; Aman, A.; Qader, S.A.U. Utilization of corncob xylan as a sole carbon source for the biosynthesis of endo-1,4-β xylanase from Aspergillus niger KIBGE-IB36. Bioresour. Bioprocess. 2017, 4, 1–7. [Google Scholar] [CrossRef]
- Naik, B.; Goyal, S.K.; Tripathi, A.D.; Kumar, V. Screening of agro-industrial waste and physical factors for the optimum production of pullulanase in solid-state fermentation from endophytic Aspergillus sp. Biocatal. Agric. Biotechnol. 2019, 22, 101423. [Google Scholar] [CrossRef]
- Campos-vega, R.; Oomah, B.D. Spent coffee grounds: A review on current research and future prospects. Trends Food Sci. Technol. 2015, 45, 24–36. [Google Scholar] [CrossRef]
- Azmir, J.; Zaidul, I.S.M.; Rahman, M.M.; Sharif, K.M.; Mohamed, A.; Sahena, F.; Jahurul, M.H.A.; Ghafoor, K.; Norulaini, N.A.N.; Omar, A.K.M. Techniques for extraction of bioactive compounds from plant materials: A review. J. Food Eng. 2013, 117, 426–436. [Google Scholar] [CrossRef]
- González-Rivera, J.; Spepi, A.; Ferrari, C.; Duce, C.; Longo, I.; Falconieri, D.; Piras, A.; Tinè, M.R. Innovative Novel configurations for a citrus waste based biorefinery: From solventless to simultaneous ultrasound and microwave assisted extraction. Green Chem. 2016, 18, 6482–6492. [Google Scholar] [CrossRef]
- Pavlovic, M.D.; Buntic, A.V.; Šiler-Marinkovic, S.S.; Suzana, I. Dimitrijevic’-Brankovic Ethanol influenced fast microwave-assisted extraction for natural antioxidants obtaining from spent filter coffee. Sep. Purif. Technol. 2013, 118, 503–510. [Google Scholar] [CrossRef]
- Pataro, G.; Carullo, D.; Falcone, M.; Ferrari, G. Recovery of lycopene from industrially derived tomato processing by-products by pulsed electric fields-assisted extraction. Innov. Food Sci. Emerg. Technol. 2020, 63, 102369. [Google Scholar] [CrossRef]
- Frontuto, D.; Carullo, D.; Harrison, S.M.; Brunton, N.P.; Ferrari, G.; Lyng, J.G.; Pataro, G. Optimization of Pulsed Electric Fields-Assisted Extraction of Polyphenols from Potato Peels Using Response Surface Methodology. Food Bioprocess Technol. 2019, 12, 1708–1720. [Google Scholar] [CrossRef]
- Kehili, M.; Schmidt, L.M.; Reynolds, W.; Zammel, A.; Zetzl, C.; Smirnova, I.; Allouche, N.; Sayadi, S. Biorefinery cascade processing for creating added value on tomato industrial by-products from Tunisia. Biotechnol. Biofuels 2016, 9, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Abad, A.; Ramos, M.; Hamzaoui, M.; Kohnen, S.; Jiménez, A.; Garrigós, M.C. Optimisation of sequential microwave-assisted extraction of essential oil and pigment from lemon peels waste. Foods 2020, 9, 1493. [Google Scholar] [CrossRef]
- Lu, S.Y.; Chu, Y.L.; Sridhar, K.; Tsai, P.J. Effect of ultrasound, high-pressure processing, and enzymatic hydrolysis on carbohydrate hydrolyzing enzymes and antioxidant activity of lemon (Citrus limon) flavedo. LWT 2021, 138. [Google Scholar] [CrossRef]
- Pattnaik, M.; Pandey, P.; Martin, G.J.O.; Mishra, H.N.; Ashokkumar, M. Innovative technologies for extraction and microencapsulation of bioactives from plant-based food waste and their applications in functional food development. Foods 2021, 10, 279. [Google Scholar] [CrossRef] [PubMed]
- Mahato, N.; Sinha, M.; Sharma, K.; Koteswararao, R.; Cho, M.H. Modern Extraction and Purification Techniques for Obtaining High Purity Food-Grade Bioactive Compounds and Value-Added Co-Products from Citrus Wastes. Foods 2019, 8, 523. [Google Scholar] [CrossRef]
- Shahzad, K.; Narodoslawsky, M.; Sagir, M.; Ali, N.; Ali, S.; Rashid, M.I.; Ismail, I.M.I.; Koller, M. Techno-economic feasibility of waste biorefinery: Using slaughtering waste streams as starting material for biopolyester production. Waste Manag. 2017, 67, 73–85. [Google Scholar] [CrossRef] [PubMed]
- Sánchez Maldonado, A.F.; Mudge, E.; Gänzle, M.G.; Schieber, A. Extraction and fractionation of phenolic acids and glycoalkaloids from potato peels using acidified water/ethanol-based solvents. Food Res. Int. 2014, 65, 27–34. [Google Scholar] [CrossRef]
- Biddy, M.J.; Davis, R.; Humbird, D.; Tao, L.; Dowe, N.; Guarnieri, M.T.; Linger, J.G.; Karp, E.M.; Salvachúa, D.; Vardon, D.R.; et al. The Techno-Economic Basis for Coproduct Manufacturing to Enable Hydrocarbon Fuel Production from Lignocellulosic Biomass. ACS Sustain. Chem. Eng. 2016, 4, 3196–3211. [Google Scholar] [CrossRef]
- Yang, M.; Baral, N.R.; Simmons, B.A.; Mortimer, J.C.; Shih, P.M.; Scown, C.D. Accumulation of high-value bioproducts in planta can improve the economics of advanced biofuels. Proc. Natl. Acad. Sci. USA 2020, 117, 27061. [Google Scholar] [CrossRef] [PubMed]
- Clauser, N.M.; Felissia, F.E.; Area, M.C.; Vallejos, M.E. A framework for the design and analysis of integrated multi-product biorefineries from agricultural and forestry wastes. Renew. Sustain. Energy Rev. 2021, 139. [Google Scholar] [CrossRef]
- van Rijn, R.; Nieves, I.U.; Shanmugam, K.T.; Ingram, L.O.; Vermerris, W. Techno-Economic Evaluation of Cellulosic Ethanol Production Based on Pilot Biorefinery Data: A Case Study of Sweet Sorghum Bagasse Processed via L+SScF. Bioenergy Res. 2018, 11, 414–425. [Google Scholar] [CrossRef]
- Nitzsche, R.; Budzinski, M.; Gröngröft, A. Techno-economic assessment of a wood-based biorefinery concept for the production of polymer-grade ethylene, organosolv lignin and fuel. Bioresour. Technol. 2016, 200, 928–939. [Google Scholar] [CrossRef]
- Zetterholm, J.; Bryngemark, E.; Ahlström, J.; Söderholm, P.; Harvey, S.; Wetterlund, E. Economic evaluation of large-scale biorefinery deployment: A framework integrating dynamic biomass market and techno-economic models. Sustainability 2020, 12, 7126. [Google Scholar] [CrossRef]
| Region | Countries | Annual per Capita Food Wastage (kg/Capital/Year) | Estimated Amount of Total Food Waste Generated (Tons/Year) |
|---|---|---|---|
| Global | 121 | 931 million (17% of total produced) | |
| Africa | Egypt | 91 | 9,136,941 |
| Sudan | 97 | 4,162,396 | |
| Angola | 100 | 3,169,523 | |
| Burkina Faso | 103 | 2,086,893 | |
| Ethiopia | 92 | 10,327,236 | |
| Ghana | 84 | 2,555,332 | |
| Kenya | 99 | 5,217,367 | |
| Mali | 103 | 2,018,765 | |
| Nigeria | 189 | 37,941,470 | |
| Rwanda | 164 | 2,075,405 | |
| South Africa | 40 | 2,329,228 | |
| Uganda | 103 | 4,546,237 | |
| Zambia | 78 | 1,391,729 | |
| Asia | Uzbekistan | 91 | 3,001,868 |
| China | 64 | 91,646,213 | |
| Japan | 64 | 8,159,891 | |
| Indonesia | 77 | 20,938,252 | |
| Malaysia | 91 | 2,921,577 | |
| Vietnam | 76 | 7,346,717 | |
| Bangladesh | 65 | 10,618,233 | |
| India | 50 | 68,760,163 | |
| Pakistan | 74 | 15,947,645 | |
| Iraq | 120 | 4,734,434 | |
| Israel | 100 | 848,395 | |
| Jordan | 93 | 939,897 | |
| Saudi Arabia | 105 | 3,594,080 | |
| Australia | Australia | 102 | 2,563,110 |
| New Zealand | 61 | 291,759 | |
| Europe | Hungary | 94 | 908,669 |
| Poland | 56 | 2,119,455 | |
| Denmark | 81 | 469,449 | |
| Finland | 65 | 361,937 | |
| Ireland | 55 | 267,073 | |
| Norway | 79 | 423,857 | |
| Sweden | 81 | 812,948 | |
| UK | 77 | 5,199,825 | |
| Greece | 142 | 1,483,996 | |
| Italy | 67 | 4,059,806 | |
| Slovenia | 34 | 71,107 | |
| Spain | 77 | 3,613,954 | |
| Austria | 39 | 349,249 | |
| Belgium | 50 | 576,036 | |
| France | 85 | 5,522,358 | |
| Germany | 75 | 6,263,775 | |
| Netherland | 50 | 854,855 | |
| Switzerland | 72 | 616,037 | |
| North America | Canada | 79 | 2,938,321 |
| USA | 59 | 19,359,951 | |
| South America | Argentina | 72 | 3,243,563 |
| Brazil | 60 | 12,578,308 | |
| Colombia | 70 | 3,545,499 | |
| Ecuador | 72 | 1,258,415 | |
| Mexico | 94 | 11,979,364 | |
| Peru | 72 | 2,354,806 | |
| Uruguay | 74 | 255,909 |
| Feedstock | Bioprocess Type | Reactor Type/Configuration | Products | Yields | Reference |
|---|---|---|---|---|---|
| Food waste | Dark fermentation | Lab-scale fermenter | H2 | 1.25 mol/mol of glucose | [76] |
| Fruit and vegetable waste | Dark fermentation and anaerobic digestion | Integrated CSTR + anaerobic fixed bed reactor | H2 and CH4 | 115.2 L H2/kg VS 334 L CH4/kg COD | [77] |
| De-oiled Jatropha waste | Acid pretreatment + fermentation | Lab-scale fermenter | H2 | 86 mL/g of reducing sugar | [78] |
| Orange peel waste | Ensiling + centrifugation | Freezing + thawing | Bioethanol | 120 g/kg TS | [79] |
| Date byproduct (Deglet-Nour) | Dark fermentation | 550 mL Plasma bottle | H2 | 292 mL H2/g VS | [74] |
| Date byproduct (Deglet-Nour) | Anaerobic digestion | 550 mL Plasma bottle | CH4 | 235 mL CH4/g VS | [74] |
| Carrot discard juices | Batch fermentation | 250 mL flask | Bioethanol | 11.98 g/L | [80] |
| Calcium alginate | Batch fermentation | 250 mL flask | Bioethanol | 29.9 g/L | [80] |
| Food waste (fruit and vegetable wastes, dairies waste, manure, blood, leftovers, animal feedstuff) | Anaerobic digestion | 45 L CSTR 40 °C, 53 HRT | Biogas (60% methane content) | 670 NL biogas/kg VS | [81] |
| Anaerobic digestion | 45 L Fluidized bed reactor 40 °C, 53 HRT | Biogas, (methane content of 60%) | 550 NL biogas/kg VS | [81] | |
| Various food waste | Dark fermentation and second stage anaerobic digestion | Fermenter | Biohythane | CH4 (70–90%, v/v) + H2 (10–30%, v/v | [82] |
| Kitchen waste | Immobilization of oxidase and glucoamylase | Simultaneous scarifications and fermentations, pH 6.2, 55 °C | ethanol | 30 g/L | [83] |
| Waste cooking oil | Immobilization of lipase | Hydrolysis and esterification | Biodiesel | 91.8% fatty acid | [84] |
| Feedstock | Bioprocess Type | Reactor Type/Conditions | Products | Yields | Reference |
|---|---|---|---|---|---|
| Orange peel waste | Ensiling + centrifugation | Freezing and thawing | Lactic acid | 55 g/kg TS | [79] |
| Orange peel waste | Ensiling + centrifugation | Freezing and thawing | Acetic acid | 26 g/kg TS | [79] |
| Grape stalk | Solvent extraction | Phenols | 4.44 g/kg dry solid | [95] | |
| Seed coat waste of red sword bean | Ultrasound treatment | 400 W L/S ratio (29.3 mL/g) 500 °C, 18.4 min | Polyphenols | 755.98 µmol Trolox/g | [96] |
| Mung seed waste | Ultrasound treatment | 500 W L/S ratio 35:1 700 °C, 46.1 min | Polyphenols | 178.28 µmol Trolox/g | [97] |
| Gac peel | Microwave assisted extraction | 120 W, 25 min | Carotenoid and Antioxidant | 262 mg/100 g and 716 µmol/L TE/100 g | [98] |
| Gac peel | Ultrasound assisted extraction | 200 W, 80 min | Carotenoid and Antioxidant | 268 mg/100 g and 820 µmol/L TE/100 g | [98] |
| Jackfruit peel | Ultrasound assisted extraction | 500 W S/L ratio 1:15, pH 1.6 60 °C, 24 min | Pectin | Yield, 14.5% | [99] |
| Pastry and cake waste | Hydrolysis and fermentation | Lab-scale fermenter | Succinic acid (96–98% purity) | 0.35–0.28 g/g of substrate | [100] |
| Tomato processing waste | Ultrasound assisted extraction | 600 W 60 °C, 8.61 min | Pectin | Yield, 15.21% | [101] |
| Tomato processing waste | Ultrasound assisted + microwave extraction | (600 W 60 °C, 8.61 min) + (450 W 85.1 °C, 8 min) | Pectin | Yield, 18% | [101] |
| Tomato processing waste | Ultrasound assisted + Ohmic heating extraction | (450 W, 10 min) + (60 V, 5 min) | Pectin | Yield, 14.6% | [101] |
| Blueberries waste (Juice waste) | Pulsed electric field | Energy input, 10 kJ/kg | Anthocyanin | 75% | [102] |
| Grape marc | Microwave assisted extraction | 48% ethanol, 1.77 g extract, 10 min | Flavanols | 1.21 mg GAE/mL | [103] |
| Feedstock | Bioprocess Type | Reactor Conditions | Products | Activity | Reference |
|---|---|---|---|---|---|
| Brewery waste | Lactic acid fermentation | Flask-500 mL, incubator 37 °C, pH 6.5, 100 rpm, Lactobacillus delbrueckii | Protease | 145 U/g | [126] |
| Brewery’s spent grain | Solid state fermentation | Glass petri dishes 25 °C, 6 days, A. nigerCECt2088 | β-glucosidase | 94 U/g | [95] |
| Brewery’s spent grain | Solid state fermentation | Glass petri dishes 25 °C, 6 days, A. ibericus | Xylanase | 300–313 U/g | [95] |
| Brewery’s spent grain | Solid state fermentation | Glass petri dishes 25 °C, 6 days, A. ibericus | Cellulase | 51–62 U/g | [95] |
| Wheat bran | Submerged fermentation | 30 °C, pH 8, 6 days, A. niger KIBGE-IB36 | Xylanase | 3071 U/mg | [127] |
| Corncob | Submerged fermentation | 30 °C, pH 8, 6 days, A. niger KIBGE-IB36 | Endo-1,4-β xylanase | 1523 U/mg | [128] |
| Wheat bran | Solid state fermentation | Aspergillus sp. 28.62 °C, 3 days, 69.92% moisture, 6.42 log inoculum size | Pullulanase | 396.2 U/g | [129] |
| Carrot discard juice | Batch fermentation | Flask 250 mL, S. cerevisiae 35 °C, 3 days | Single cell protein | [80] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsegaye, B.; Jaiswal, S.; Jaiswal, A.K. Food Waste Biorefinery: Pathway towards Circular Bioeconomy. Foods 2021, 10, 1174. https://doi.org/10.3390/foods10061174
Tsegaye B, Jaiswal S, Jaiswal AK. Food Waste Biorefinery: Pathway towards Circular Bioeconomy. Foods. 2021; 10(6):1174. https://doi.org/10.3390/foods10061174
Chicago/Turabian StyleTsegaye, Bahiru, Swarna Jaiswal, and Amit K. Jaiswal. 2021. "Food Waste Biorefinery: Pathway towards Circular Bioeconomy" Foods 10, no. 6: 1174. https://doi.org/10.3390/foods10061174
APA StyleTsegaye, B., Jaiswal, S., & Jaiswal, A. K. (2021). Food Waste Biorefinery: Pathway towards Circular Bioeconomy. Foods, 10(6), 1174. https://doi.org/10.3390/foods10061174








