Periodontal and Peri-Implant Health Status in Traditional vs. Heat-Not-Burn Tobacco and Electronic Cigarettes Smokers: A Systematic Review
Abstract
:1. Introduction
2. Materials and Methods
2.1. Protocol Development
2.2. Search Strategy and Study Selection
2.3. Data Extraction and Synthesis
2.4. Risk of Bias Assessment
- Low risk of bias: The study is judged to be at low risk of bias for all domains;
- Moderate risk of bias: The study is judged to be at low or moderate risk of bias for all domains;
- Serious risk of bias: The study is judged to be at serious risk of bias in at least one domain, but not at critical risk of bias in any domain;
- Critical risk of bias: The study is judged to be at critical risk of bias in at least one domain.
3. Results
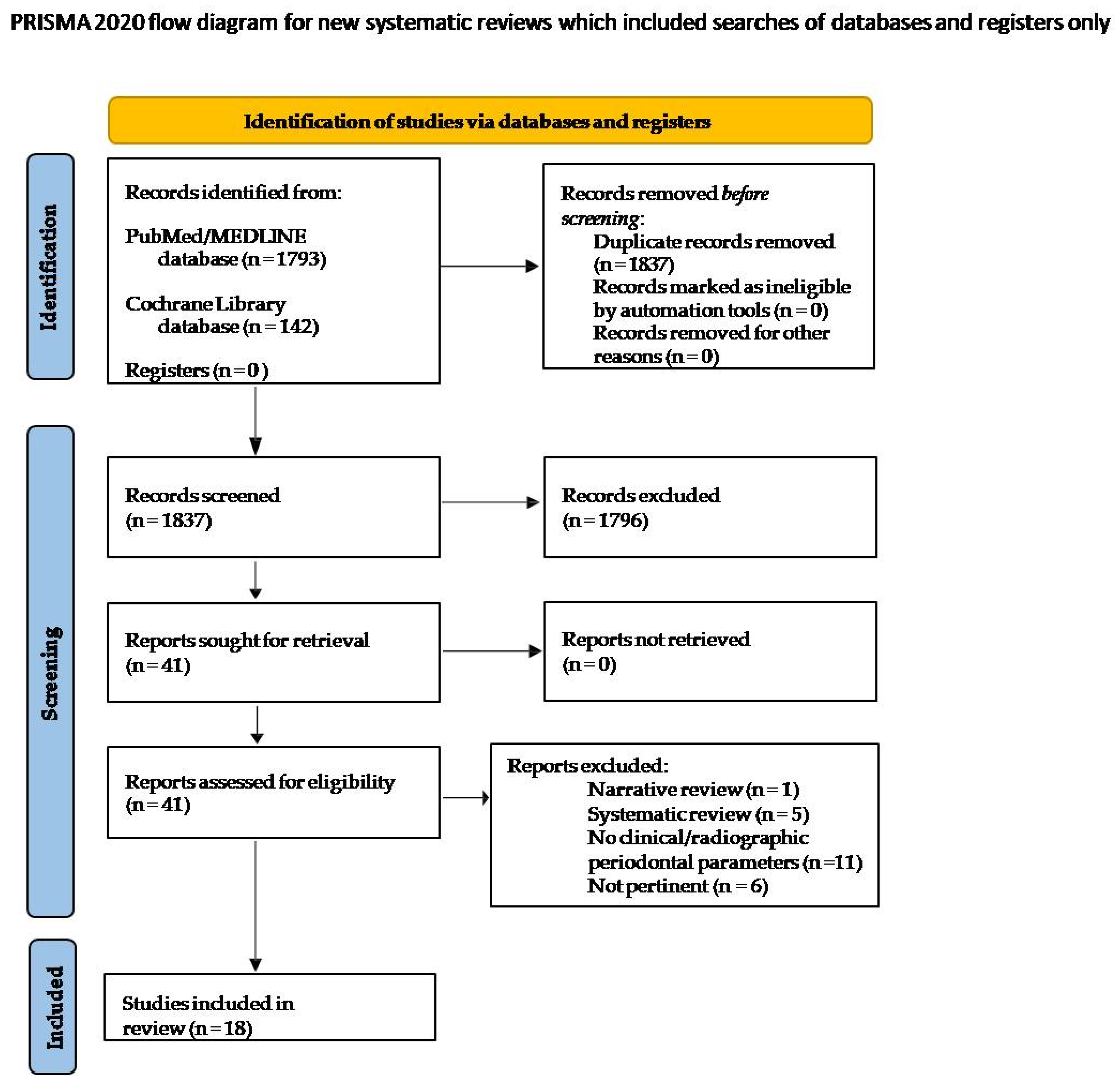
3.1. Search Strategy and Study Selection
3.2. Study Characteristics
3.3. Data Extraction and Synthesis
3.4. Quality Assessment of the Included Studies
4. Discussion
4.1. Clinical Periodontal and Peri-Implant Parameters in Traditional vs. HNB and E-Cigs Smokers
4.2. Radiographic Periodontal and Peri-Implant Parameters in Traditional vs. HNB and E-Cigs Smokers
4.3. Crevicular Inflammatory Periodontal and Peri-Implant Parameters in Traditional vs. HNB and E-Cigs Smokers
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tonetti, M.S.; Greenwell, H.; Kornman, K.S. Staging and grading of periodontitis: Framework and proposal of a new classification and case definition. J. Periodontol. 2018, 89, S159–S172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sbordone, C.; Toti, P.; Brevi, B.; Martuscelli, R.; Sbordone, L.; Di Spirito, F. Computed tomography-aided descriptive analysis of maxillary and mandibular atrophies. J. Stomatol. Oral Maxillofac. Surg. 2018, 120, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Di Spirito, F.; Toti, P.; Brevi, B.; Martuscelli, R.; Sbordone, L.; Sbordone, C. Computed tomography evaluation of jaw atrophies before and after surgical bone augmentation. Int. J. Clin. Dent. 2019, 12, 259–270. [Google Scholar]
- Checchi, V.; Gasparro, R.; Pistilli, R.; Canullo, L.; Felice, P. Clinical Classification of Bone Augmentation Procedure Failures in the Atrophic Anterior Maxillae: Esthetic Consequences and Treatment Options. BioMed Res. Int. 2019, 2019, 4386709. [Google Scholar] [CrossRef]
- Renvert, S.; Persson, G.R.; Pirih, F.Q.; Camargo, P.M. Peri-implant health, peri-implant mucositis, and peri-implantitis: Case definitions and diagnostic considerations. J. Periodontol. 2018, 89, S304–S312. [Google Scholar] [CrossRef]
- Di Spirito, F.; La Rocca, M.; De Bernardo, M.; Rosa, N.; Sbordone, C.; Sbordone, L. Possible Association of Periodontal Disease and Macular Degeneration: A Case-Control Study. Dent. J. 2020, 9, 1. [Google Scholar] [CrossRef]
- Chapple, I.L.C.; Genco, R. On behalf of working group 2 of the joint EFP/AAP workshop. Diabetes and periodontal diseases: Consensus report of the Joint EFP/AAPWorkshop on Periodontitis and Systemic Diseases. J. Periodontol. 2013, 84, 106–112. [Google Scholar] [CrossRef]
- Di Spirito, F.; Schiavo, L.; Pilone, V.; Lanza, A.; Sbordone, L.; D’Ambrosio, F. Periodontal and Peri-Implant Diseases and Systemically Administered Statins: A Systematic Review. Dent. J. 2021, 9, 100. [Google Scholar] [CrossRef]
- Genco, R.J.; Borgnakke, W.S. Risk factors for periodontal disease. Periodontology 2000 2013, 62, 59–94. [Google Scholar] [CrossRef]
- Di Spirito, F.; Sbordone, L.; Pilone, V.; D’Ambrosio, F. Obesity and Periodontal Disease: A Narrative Review on Current Evidence and Putative Molecular Links. Open Dent. J. 2019, 13, 526–536. [Google Scholar] [CrossRef]
- Di Spirito, F.; Toti, P.; Pilone, V.; Carinci, F.; Lauritano, D.; Sbordone, L. The Association between Periodontitis and Human Colorectal Cancer: Genetic and Pathogenic Linkage. Life 2020, 10, 211. [Google Scholar] [CrossRef] [PubMed]
- D’Ambrosio, F.; Caggiano, M.; Schiavo, L.; Savarese, G.; Carpinelli, L.; Amato, A.; Iandolo, A. Chronic Stress and Depression in Periodontitis and Peri-Implantitis: A Narrative Review on Neurobiological, Neurobehavioral and Immune–Microbiome Interplays and Clinical Management Implications. Dent. J. 2022, 10, 49. [Google Scholar] [CrossRef] [PubMed]
- Chrcanovic, B.R.; Albrektsson, T.; Wennerberg, A. Smoking and dental implants: A systematic review and meta-analysis. J. Dent. 2015, 43, 487–498. [Google Scholar] [CrossRef] [PubMed]
- Ramaglia, L.; Di Spirito, F.; Sirignano, M.; La Rocca, M.; Esposito, U.; Sbordone, L. A 5-year longitudinal cohort study on crown to implant ratio effect on marginal bone level in single implants. Clin. Implant Dent. Relat. Res. 2019, 21, 916–922. [Google Scholar] [CrossRef]
- Ramôa, C.P.; Eissenberg, T.; Sahingur, S.E. Increasing popularity of waterpipe tobacco smoking and electronic cigarette use: Implications for oral healthcare. J. Periodontal. Res. 2017, 52, 813–823. [Google Scholar] [CrossRef]
- Choi, K.; Forster, J.L. Beliefs and experimentation with electronic cigarettes: A prospective analysis among young adults. Am. J. Prev. Med. 2014, 46, 175–178. [Google Scholar] [CrossRef] [Green Version]
- Ratajczak, A.; Jankowski, P.; Strus, P.; Feleszko, W. Heat Not Burn Tobacco Product-A New Global Trend: Impact of Heat-Not-Burn Tobacco Products on Public Health, a Systematic Review. Int. J. Environ. Res. Public Health 2020, 17, 409. [Google Scholar] [CrossRef] [Green Version]
- Etter, J.-F.; Bullen, C.; Flouris, A.; Laugesen, M.; Eissenberg, T. Electronic nicotine delivery systems: A research agenda. Tob. Control 2011, 20, 243–248. [Google Scholar] [CrossRef] [Green Version]
- Cobb, N.; Abrams, D.B. E-Cigarette or Drug-Delivery Device? Regulating Novel Nicotine Products. N. Engl. J. Med. 2011, 365, 193–195. [Google Scholar] [CrossRef]
- Mishra, V.K.; Kim, K.-H.; Samaddar, P.; Kumar, S.; Aggarwal, M.; Chacko, K. Review on metallic components released due to the use of electronic cigarettes. Environ. Eng. Res. 2017, 22, 131–140. [Google Scholar] [CrossRef]
- Gaur, S.; Agnihotri, R. Health Effects of Trace Metals in Electronic Cigarette Aerosols-a Systematic Revew. Biol. Trace Elem. Res. 2019, 188, 295–315. [Google Scholar] [CrossRef] [PubMed]
- Ralho, A.; Coelho, A.; Ribeiro, M.; Paula, A.; Amaro, I.; Sousa, J.; Marto, C.; Ferreira, M.; Carrilho, E. Effects of Electronic Cigarettes on Oral Cavity: A Systematic Review. J. Evid. Based Dent. Pract. 2019, 19, 101318. [Google Scholar] [CrossRef] [PubMed]
- Figueredo, C.A.; Abdelhay, N.; Catunda, R.; Gibson, M.P. The impact of vaping on periodontitis: A systematic review. Clin. Exp. Dent. Res. 2020, 7, 376–384. [Google Scholar] [CrossRef] [PubMed]
- Jeong, W.; Choi, D.W.; Kim, Y.K.; Lee, H.J.; Lee, S.A.; Park, E.C.; Jang, S.I. Associations of electronic and conventional cigarette use with periodontal disease in South Korean adults. J. Periodontol. 2020, 91, 55–64. [Google Scholar] [CrossRef] [Green Version]
- Sundar, I.K.; Javed, F.; Romanos, G.E.; Rahman, I. E-cigarettes and flavorings induce inflammatory and pro-senescence responses in oral epithelial cells and periodontal fibroblasts. Oncotarget 2016, 7, 77196–77204. [Google Scholar] [CrossRef] [Green Version]
- Zarabadipour, M.; Hosseini, S.A.H.; Haghdoost-Yazdi, H.; Aali, E.; Yusefi, P.; Mirzadeh, M.; Piri, H. A study on the correlation between smoking and non-enzymatic antioxidant factors of the saliva of healthy smokers and non-smokers. Braz. Dent. Sci. 2022, 25, e2867. [Google Scholar] [CrossRef]
- Pouly, S.; Benzimra, W.T.; Soulan, M.; Blanc, A.; Zanetti, N.; Picavet, F.; Baker, P.; Haziza, G. Effect of Switching to the Tobacco Heating System Versus Continued Cigarette Smoking on Chronic Generalized Periodontitis Treatment Outcome: Protocol for a Randomized Controlled Multicenter Study. JMIR Res. Protoc. 2021, 10, e15350. [Google Scholar] [CrossRef]
- Pagano, S.; Negri, P.; Coniglio, M.; Bruscoli, S.; Di Michele, A.; Marchetti, M.C.; Valenti, C.; Gambelunghe, A.; Fanasca, L.; Billi, M.; et al. Heat-not-burn tobacco (IQOS), oral fibroblasts and keratinocytes: Cytotoxicity, morphological analysis, apoptosis and cellular cycle. An in vitro study. J. Periodontal. Res. 2021, 56, 917–928. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gotzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. PLoS Med. 2009, 6, e1000100. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [Green Version]
- Da Costa Santos, C.M.; Pimenta, C.A.M.; Nobre, M.R.C. ThePICOstrategyfor the research question construction and evidence search. Rev. Lat. Am. Enfermagem. 2007, 15, 508–511. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sterne, J.A.C.; Hernán, M.A.; Reeves, B.C.; Savović, J.; Berkman, N.D.; Viswanathan, M.; Henry, D.; Altman, D.G.; Ansari, M.T.; Boutron, I.; et al. ROBINS-I: A tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016, 355, i4919. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Javed, F.; Kellesarian, S.V.; Sundar, I.K.; Romanos, G.E.; Rahman, I. Recent updates on electronic cigarette aerosol and inhaled nicotine effects on periodontal and pulmonary tissues. Oral Dis. 2017, 23, 1052–1057. [Google Scholar] [CrossRef] [PubMed]
- Shaito, A.; Saliba, J.; Husari, A.; El-Harakeh, M.; Chhouri, H.; Hashem, Y.; Shihadeh, A.; El-Sabban, M. Electronic Cigarette Smoke Impairs Normal Mesenchymal Stem Cell Differentiation. Sci. Rep. 2017, 7, 14281. [Google Scholar] [CrossRef] [PubMed]
- Vyncke, T.; De Wolf, E.; Hoeksema, H.; Verbelen, J.; De Coninck, P.; Buncamper, M.; Monstrey, S.; Claes, K.E. Injuries associated with electronic nicotine delivery systems: A systematic review. J. Trauma Acute Care Surg. 2020, 89, 783–791. [Google Scholar] [CrossRef] [PubMed]
- Irusa, K.F.; Vence, B.; Donovan, T. Potential oral health effects of e-cigarettes and vaping: A review and case reports. J. Esthet. Restor. Dent. 2020, 32, 260–264. [Google Scholar] [CrossRef] [PubMed]
- Atuegwu, N.C.; Perez, M.F.; Oncken, C.; Thacker, S.; Mead, E.L.; Mortensen, E.M. Association between Regular Electronic Nicotine Product Use and Self-Reported Periodontal Disease Status: Population Assessment of Tobacco and Health Survey. Int. J. Environ. Res. Public Health 2019, 16, 1263. [Google Scholar] [CrossRef] [Green Version]
- Sancilio, S.; Gallorini, M.; Cataldi, A.; DI Giacomo, V. Cytotoxicity and apoptosis induction by e-cigarette fluids in human gingival fibroblasts. Clin. Oral Investig. 2015, 20, 477–483. [Google Scholar] [CrossRef]
- Javed, F.; Rahman, I.; Romanos, G.E. Tobacco-product usage as a risk factor for dental implants. Periodontology 2000 2019, 81, 48–56. [Google Scholar] [CrossRef]
- Ganesan, S.M.; Dabdoub, S.M.; Nagaraja, H.N.; Scott, M.L.; Pamulapati, S.; Berman, M.L.; Shields, P.G.; Wewers, M.E.; Kumar, P.S. Adverse effects of electronic cigarettes on the disease-naive oral microbiome. Sci. Adv. 2020, 6, eaaz0108. [Google Scholar] [CrossRef]
- Andrikopoulos, G.; Farsalinos, K.; Poulas, K. ElectronicNicotineDeliverySystems(ENDS) and TheirRelevanceinOralHealth. Toxics 2019, 7, 61. [Google Scholar]
- Nelson, J.M.; Cuadra, G.A.; Palazzolo, D.L. A Comparison of Flavorless Electronic Cigarette-Generated Aerosol and Conventional Cigarette Smoke on the Planktonic Growth of Common Oral Commensal Streptococci. Int. J. Environ. Res. Public Health 2019, 16, 5004. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Willershausen, I.; Wolf, T.; Weyer, V.; Sader, R.; Ghanaati, S.; Willershausen, B. Influence of E-smoking liquids on human periodontal ligament fibroblasts. Head Face Med. 2014, 10, 39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rouabhia, M.; Alanazi, H.; Park, H.J.; Gonçalves, R.B. Cigarette Smoke and E-Cigarette Vapor Dysregulate Osteoblast Interaction with Titanium Dental Implant Surface. J. Oral Implant. 2019, 45, 2–11. [Google Scholar] [CrossRef]
- Holliday, R.; Preshaw, P.M.; Ryan, V.; Sniehotta, F.F.; McDonald, S.; Bauld, L.; McColl, E. A feasibility study with embedded pilot randomised controlled trial and process evaluation of electronic cigarettes for smoking cessation in patients with periodontitis. Pilot Feasibility Stud. 2019, 5, 74. [Google Scholar] [CrossRef]
- Zanetti, F.; Titz, B.; Sewer, A.; Sasso, G.L.; Scotti, E.; Schlage, W.K.; Mathis, C.; Leroy, P.; Majeed, S.; Torres, L.O.; et al. Comparative systems toxicology analysis of cigarette smoke and aerosol from a candidate modified risk tobacco product in organotypic human gingival epithelial cultures: A 3-day repeated exposure study. Food Chem. Toxicol. 2017, 101, 15–35. [Google Scholar] [CrossRef]
- Tatullo, M.; Gentile, S.; Paduano, F.; Santacroce, L.; Marrelli, M. Crosstalk between oral and general health status in e-smokers. Medicine 2016, 95, e5589. [Google Scholar] [CrossRef]
- Alqahtani, S.; Cooper, B.; Spears, C.A.; Wright, C.; Shannahan, J. Electronic nicotine delivery system-induced alterations in oral health via saliva assessment. Exp. Biol. Med. 2020, 245, 1319–1325. [Google Scholar] [CrossRef]
- Al Rifaiy, M.Q.; Qutub, O.A.; Alasqah, M.N.; Al-Sowygh, Z.H.; Mokeem, S.A.; Alrahlah, A. Effectiveness of adjunctive antimicrobial photodynamic therapy in reducing peri -implant inflammatory response in individuals vaping electronic cigarettes: A randomized controlled clinical trial. Photodiagn. Photodyn. Ther. 2018, 22, 132–136. [Google Scholar] [CrossRef]
- Mokeem, S.A.; Alasqah, M.N.; Michelogiannakis, D.; Al-Kheraif, A.A.; Romanos, G.E.; Javed, F. Clinical and radiographic periodontal status and whole salivary cotinine, IL-1β and IL-6 levels in cigarette- and waterpipe- smokers and E-cig users. Environ. Toxicol. Pharmacol. 2018, 61, 38–43. [Google Scholar] [CrossRef]
- Al-Aali, K.A.; Alrabiah, M.; ArRejaie, A.S.; Abduljabbar, T.; Vohra, F.; Akram, Z. Peri-implant parameters, tumor necrosis factor- alpha, and interleukin-1 beta levels in vaping individuals. Clin. Implant. Dent. Relat. Res. 2018, 20, 410–415. [Google Scholar] [CrossRef] [PubMed]
- Karaaslan, F.; Dikilitaş, A.; Yiğit, U. The effects of vaping electronic cigarettes on periodontitis. Aust. Dent. J. 2020, 65, 143–149. [Google Scholar] [CrossRef] [PubMed]
- AlQahtani, M.A.; Alayad, A.S.; Al-Shihri, A.; Oliveira Bello Correa, F.; Akram, Z. Clinical peri-implant parameters and inflammatory cyto- kine profile among smokers of cigarette, e-cigarette, and waterpipe. Clin. Implant. Dent. Relat. Res. 2018, 20, 1016–1021. [Google Scholar] [CrossRef] [PubMed]
- Binshabaib, M.; Alharthi, S.S.; Akram, Z.; Khan, J.; Rahman, I.; Romanos, G.E.; Javed, F. Clinical periodontal status and gingival crevicular fluid cytokine profile among cigarette-smokers, electronic-cigarette users and never-smokers. Arch. Oral Biol. 2019, 102, 212–217. [Google Scholar] [CrossRef]
- Vohra, F.; Bukhari, I.A.; Sheikh, S.A.; Albaijan, R.; Naseem, M. Comparison of self-rated oral symptoms and periodontal status among cigarette smokers and individuals using electronic nicotine delivery systems. J. Am. Coll. Health 2020, 68, 788–793. [Google Scholar] [CrossRef]
- Javed, F.; Abduljabbar, T.; Vohra, F.; Malmstrom, H.; Rahman, I.; Romanos, G.E. Comparison of Periodontal Parameters and Self-Perceived Oral Symptoms Among Cigarette Smokers, Individuals Vaping Electronic Cigarettes, and Never-Smokers. J. Periodontol. 2017, 88, 1059–1065. [Google Scholar] [CrossRef]
- Aldakheel, F.M.; Alduraywish, S.A.; Jhugroo, P.; Jhugroo, C.; Divakar, D.D. Quantification of pathogenic bacteria in the subgingival oral biofilm samples collected from cigarette-smokers, individuals using electronic nicotine delivery systems and non-smokers with and without periodontitis. Arch. Oral Biol. 2020, 117, 104793. [Google Scholar] [CrossRef]
- Ibraheem, W.I.; Fageeh, H.I.; Preethanath, R.S.; Alzahrani, F.A.; Al-Zawawi, A.S.; Divakar, D.D.; Al-Kheraif, A.A. Comparison of RANKL and osteoprottegerin levels in the gingival crevicular fluid of young cigarette- and waterpipe- smokers and individuals using electronic nicotine delivery systems. Arch. Oral Biol. 2020, 115, 104714. [Google Scholar] [CrossRef]
- Alharthi, S.S.; BinShabaib, M.; Akram, Z.; Rahman, I.; Romanos, G.E.; Javed, F. Impact of cigarette smoking and vaping on the outcome of full-mouth ultrasonic scaling among patients with gingival inflammation: A prospective study. Clin. Oral Investig. 2018, 23, 2751–2758. [Google Scholar] [CrossRef]
- ArRejaie, A.S.; Al-Aali, K.A.; Alrabiah, M.; Vohra, F.; Mokeem, S.A.; Basunbul, G.; Alrahla, A.; Abduljabbar, T. Pro-inflammatory cytokine levels and peri-implant parameters among cigarette smokers, individuals vaping electronic cigarettes and non-smokers. J. Periodontol. 2018, 90, 367–374. [Google Scholar] [CrossRef]
- Al-Hamoudi, N.; Alsahhaf, A.; Al Deeb, M.; Alrabiah, M.; Vohra, F.; Abduljabbar, T. Effect of scaling and root planing on the expression of anti-inflammatory cytokines (IL-4, IL-9, IL-10, and IL-13) in the gingival crevicular fluid of electronic cigarette users and non-smokers with moderate chronic periodontitis. J. Periodontal. Implant Sci. 2020, 50, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Alqahtani, F.; Alqahtani, M.; Shafqat, S.S.; Akram, Z.; Al-Kheraif, A.A.; Javed, F. Efficacy of mechanical debridement with adjunctive probiotic therapy in the treatment of peri-implant mucositis in cigarette-smokers and never-smokers. Clin. Implant Dent. Relat. Res. 2019, 21, 734–740. [Google Scholar] [CrossRef] [PubMed]
- Sinha, D.K.; Kumar, A.; Khan, M.; Kumari, R.; Kesari, M. Vishal Evaluation of tumor necrosis factor-alpha (TNF-α) and interleukin (IL)-1β levels among subjects vaping e-cigarettes and nonsmokers. J. Fam. Med. Prim. Care 2020, 9, 1072–1075. [Google Scholar] [CrossRef] [PubMed]
- Al Deeb, M.; Alsahhaf, A.; Alhamoudi, N.; Al-Aali, K.A.; Abduljabbar, T. Clinical and microbiological outcomes of photodynamic and systemic antimicrobial therapy in smokers with peri-implant inflammation. Photodiagn. Photodyn. Ther. 2020, 29, 101587. [Google Scholar] [CrossRef] [PubMed]
- AlJasser, R.; Zahid, M.; AlSarhan, M.; AlOtaibi, D.; AlOraini, S. The effect of conventional versus electronic cigarette use on treatment outcomes of peri-implant disease. BMC Oral Health 2021, 21, 480. [Google Scholar] [CrossRef]
- Xu, F.; Aboseria, E.; Janal, M.N.; Pushalkar, S.; Bederoff, M.V.; Vasconcelos, R.; Sapru, S.; Paul, B.; Queiroz, E.; Makwana, S.; et al. Comparative Effects of E-Cigarette Aerosol on Periodontium of Periodontitis Patients. Front. Oral Health 2021, 2, 58. [Google Scholar] [CrossRef]
- Bergström, J. Tobaccosmokingand chronic destructive periodontal disease. Odontology 2004, 92, 1–8. [Google Scholar] [CrossRef]
- Dietrich, T.; Bernimoulin, J.P.; Glynn, R.J. The effect of cigarette smoking on gingival bleeding. J. Periodontol. 2004, 75, 16–22. [Google Scholar] [CrossRef]
- Knight, E.T.; Liu, J.; Seymour, G.; Faggion, C.M.; Cullinan, M. Risk factors that may modify the innate and adaptive immune responses in periodontal diseases. Periodontology 2000 2016, 71, 22–51. [Google Scholar] [CrossRef]
- Rakic, M.; Struillou, X.; Petkovic-Curcin, A.; Matic, S.; Canullo, L.; Sanz, M.; Vojvodic, D. Estimation of Bone Loss Biomarkers as a Diagnostic Tool for Peri-Implantitis. J. Periodontol. 2014, 85, 1566–1574. [Google Scholar] [CrossRef]
- Petkovic, A.B.; Matic, S.M.; Stamatovic, N.V.; Vojvodic, D.V.; Todorovic, T.M.; Lazic, Z.R.; Kozomara, R.J. Pro-inflammatory cytokines (IL-1beta and TNF-alpha) and chemokines (IL-8 and MIP-1alpha) as markers of peri-implant tissue condition. Int. J. Oral Maxillofac. Surg. 2010, 39, 478–485. [Google Scholar] [CrossRef] [PubMed]
- Graves, D.T.; Li, J.; Cochran, D.L. Inflammation and Uncoupling as Mechanisms of Periodontal Bone Loss. J. Dent. Res. 2010, 90, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Abduljabbar, T.; Vohra, F.; Javed, F.; Akram, Z. Antimicrobial photodynamic therapy adjuvant to non-surgical periodontal therapy in patients with diabetes mellitus: A meta-analysis. Photodiagn. Photodyn. Ther. 2017, 17, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Bostanci, N.; Ilgenli, T.; Emingil, G.; Afacan, B.; Han, B.; Töz, H.; Atilla, G.; Hughes, F.J.; Belibasakis, G.N. Gingival crevicular fluid levels of RANKL and OPG in periodontal diseases: Implications of their relative ratio. J. Clin. Periodontol. 2007, 34, 370–376. [Google Scholar] [CrossRef]
- Teodorescu, A.C.; Martu, I.; Teslaru, S.; Kappenberg-Nitescu, D.C.; Goriuc, A.; Luchian, I.; Martu, M.A.; Solomon, S.M.; Mârțu, S. Assessment of Salivary Levels of RANKL and OPG in Aggressive versus Chronic Periodontitis. J. Immunol. Res. 2019, 2019, 6195258. [Google Scholar] [CrossRef] [Green Version]
- Algate, K.; Haynes, D.R.; Bartold, P.M.; Crotti, T.N.; Cantley, M.D. The effects of tumour necrosis factor-α on bone cells involved in periodontal alveolar bone loss; osteoclasts, osteoblasts and osteocytes. J. Periodontal. Res. 2016, 51, 549–566. [Google Scholar] [CrossRef]
- Reznick, A.Z.; ECross, C.; Hu, M.L.; Suzuki, Y.J.; Khwaja, S.; Safadi, A.; AMotchnik, P.; Packer, L.; Halliwell, B. Modification of plasma proteins by cigarette smoke as measured by protein carbonyl formation. Biochem. J. 1992, 286, 607–611. [Google Scholar] [CrossRef] [Green Version]
- Epstein, F.H.; Weiss, S.J. Tissue Destruction by Neutrophils. N. Engl. J. Med. 1989, 320, 365–376. [Google Scholar] [CrossRef]
- Di Spirito, F.; Iacono, V.J.; Iandolo, A.; Amato, A.; Sbordone, L. Evidence-based Recommendations on Periodontal Practice and the Management of Periodontal Patients during and after the COVID-19 Era: Challenging Infectious Diseases Spread by Air-borne Transmission. Open Dent. 2021, 15, 325–336. [Google Scholar] [CrossRef]
- Graziani, F.; Karapetsa, D.; Alonso, B.; Herrera, D. Nonsurgical and surgical treatment of periodontitis: How many options for one disease? Periodontology 2000 2017, 75, 152–188. [Google Scholar] [CrossRef]
- Lauritano, D.; Moreo, G.; Carinci, F.; Della Vella, F.; Di Spirito, F.; Sbordone, L.; Petruzzi, M. Cleaning Efficacy of the XP-Endo® Finisher Instrument Compared to Other Irrigation Activation Procedures: A Systematic Review. Appl. Sci. 2019, 9, 5001. [Google Scholar] [CrossRef] [Green Version]
- Di Spirito, F.; Pelella, S.; Argentino, S.; Sisalli, L.; Sbordone, L. Oral manifestations and the role of the oral healthcare workers in COVID-19. Oral. Dis. 2022, 28, 1003–1004. [Google Scholar] [CrossRef] [Green Version]
- Di Spirito, F.; Scelza, G.; Fornara, R.; Giordano, F.; Rosa, D.; Amato, A. Post-operative endodontic pain management: An overview of systematic reviews on post-operatively administered oral medications and integrated evidence-based clinical recommendations. Healthcare 2022, 10, 760. [Google Scholar] [CrossRef] [PubMed]
- Barone, A.; Chatelain, S.; Derchi, G.; Di Spirito, F.; Martuscelli, R.; Porzio, M.; Sbordone, L. Effectiveness of antibiotics in preventing alveolitis after erupted tooth extraction: A retrospective study. Oral Dis. 2020, 26, 967–973. [Google Scholar] [CrossRef] [PubMed]
- Pisano, M.; Amato, A.; Sammartino, P.; Iandolo, A.; Martina, S.; Caggiano, M. Laser Therapy in the Treatment of Peri-Implantitis: State-of-the-Art, Literature Review and Meta-Analysis. Appl. Sci. 2021, 11, 5290. [Google Scholar] [CrossRef]
- Di Spirito, F.; Argentino, S.; Martuscelli, R.; Sbordone, L. MRONJ incidence after multiple teeth extractions in patients taking oral bisphosphonates without “drug holiday”: A retrospective chart review. Oral Implantol. 2019, 12, 105–110. [Google Scholar]
| Authors, Year | Reason for Exclusion |
|---|---|
| Yang, 2020 [23] | Systematic review |
| Ralho, 2019 [22] | Systematic review |
| Javed, 2017 [33] | No clinical/radiographic parameters |
| Shaito, 2017 [34] | No clinical/radiographic parameters |
| Vyncke, 2020 [35] | Systematic review |
| Karina, 2020 [36] | Review |
| Atuegwu, 2019 [37] | No clinical/radiographic parameters |
| Chrcanovic, 2015 [11] | Systematic review |
| Sancilio, 2015 [38] | No clinical/radiographic parameters |
| Ryder, 2018 [12] | Relationship between e-cigarettes and alcohol |
| Javed, 2019 [39] | No clinical/radiographic parameters |
| Ganesan, 2020 [40] | Relationship between e-cigarettes and oral microbiome |
| Andrikopoulos, 2019 [41] | No clinical/radiographic parameters |
| Nelson, 2019 [42] | Relationship between e-cigarettes and oral microbiome |
| Willershausen, 2014 [43] | No clinical/radiographic parameters |
| Rouabhia, 2018 [44] | No clinical/radiographic parameters |
| Holliday, 2019 [45] | Study on tobacco cessation and starting e-cigarettes |
| Zanetti, 2016 [46] | No clinical/radiographic parameters |
| Sandar, 2016 [27] | No clinical/radiographic parameters |
| Tatullo, 2016 [47] | Study on tobacco smokers which started to use E-cigarette |
| Alqahtani, 2020 [48] | No clinical/radiographic parameters |
| Al Rifaiy, 2018 [49] | Effect of the antimicrobial photodynamic therapy (aPDT) individuals vaping electronic cigarettes |
| Figueredo, 2021 [25] | Systematic review |
| Authors, Year Reference Study Design Aim/Objective | Population Sample Size Mean Age Gender Ratio | Periodontal and Peri-Implant Assessment Time Points Treatment Dental Implants (n.) Clinical Parameters Radiographic Parameters Crevicular Parameters Other Parameters | Main Results | Conclusions |
|---|---|---|---|---|
| Mokeem, 2018 [50] Case-control To compare PI, BOP, PD, CAL, MBL and cotinine, interleukin IL-1β and IL-6 levels among CS, WS, E-Cigs and NS | 39 CS (42.4 ± 5.6 y.o.) 40 E-Cigs (44.7 ± 4.5 y.o.) 37 E-Cigs (28.3 ± 3.5 y.o.) 38NS (40.6 ± 4.5 y.o.) All males | Periodontal and peri-implant assessment at baseline No periodontal treatment No dental implants PI BoP CAL PD MBL IL-1b IL-6 cotinine | PI was significantly higher among CS and WS than E-Cigs (p < 0.05) and NS (p < 0.05).PI was significantly higher among E-Cigs than NS (p < 0.05). BOP were significantly higher among NS compared with CS (p < 0.05) and WS (p < 0.05) and E-Cigs (p < 0.05) PD (p < 0.05), CAL (p < 0.05) and MBL (p < 0.05) were significantly higher among CS and WS compared with E-Cigs and NS. There was no difference in PD, CAL, MBL, IL-1β and IL-6 levels among E-Cigs and NS IL-1β (p < 0.01) and IL-6 (p < 0.01) levels were significantly higher among CS, WS than E-Cigs and NS. Cotinine levels were significantly higher among CS (p < 0.001) and WS (p < 0.001) and E-Cigs (p < 0.001) than NS. | CS and WS had worse radiographic parameters of periodontal inflammation than E-Cigs and NS. Cotinine levels were similar in all groups. Salivary IL-1β and IL-6 levels were higher in CS and WS than E-Cigs and NS |
| Al-Aali, 2018 [51] Case-control To compare clinical and radiographic peri-implant parameters and TNF-a and IL-1b levels among E-Cigs and NS | 47 E-Cigs (35.8 ± 6.2 y.o.) 45 NS (42.6 ± 2.7 y.o.) All males | Periodontal and peri-implant assessment at baseline No periodontal treatment Dental implants n. 125 (at least one positioned for ≥36 months) PI BoP PD (>/=4 mm) PIBL TNF-α IL-1β PISF volume |
BOP was statistically significantly higher in NS compared to E-Cigs (p < 0.01). PD (p < 0.05); PIBL (p < 0.05);TNF-a (p < 0.001) and IL-1b (p < 0.01) were statistically significantly higher in E-Cigs than NS. There was a significant positive relationship among TNF-a levels and BOP (P5.024) and PIBL (P5.016); and among IL-1b and PIBL (P5.018) in E-Cigs. |
Clinical and radiographic peri-implant parameters are worse among E-Cigs than NS. E-Cigs show higher levels of pro-inflammatory cytokines than NS. |
| Karaaslan, 2020 [52] Case-control To evaluate the effects of vaping, cigarettes smoke and smoking cessation on periodontal clinical parameters, oxidative stress markers and pro-inflammatory cytokines in patients with periodontal disease | 19 CS (35.26 ± 2.31 y.o.) 19 E-Cigs (34.74 ± 2.38 y.o.) Ex- cigarettes smokers: 19 individuals (35.58 ± 2.04 y.o.) 39M/18F | Periodontal and peri-implant assessment at baseline No periodontal treatment No dental implants PI GI CAL IL-8 TNF-a GsH-Px (Glutathione peroxidase) 8-OHdG (8-hydroxydeoxyguanosine) | No significant differences were found between the groups for PD, PI, CAL. GI was significantly higher in group 2e 3 than group 1 and GI was significantly lower in group 2 than group 3. IL-8 level of Group I (70.47 ± 2.76) was significantly lower than in Groups II and III. TNF-a level of Group I (4.20 ± 0.14) was significantly higher than in Groups II and III. | Cigarette smoke and vaping have negative effects on the markers of oxidative stress and inflammatory cytokines. |
| AlQahtani, 2018 [53] Case-control To compare clinical and radiographic peri-implant parameters and cytokines among CS, WS, E-Cigs, and NS | 40 CS 40 WS 40 E-Cigs 40 NS Mean age: 41.8 y.o. All males | Periodontal and peri-implant assessment at baseline No periodontal treatment Dental implants n. 253 (at least one in service for >/=36 months) PI BoP PD (≥4 mm) MBL TNF-a IL-6 IL-1B PISF | Peri-implant PI (p < 0.05), PD (p < 0.05) and RBL (p < 0.01) was significantly higher among CS, WS and E-Cigs compared to NS. BOP in CS, WS, and E-Cigs showed statistical differences (p < 0.01) compared to NS CS and WS showed significantly higher PD ≥ 4 mm and RBL compared with E-Cigs (p < 0.05). TNF-α, IL-6, and IL-1β were significantly higher in CS, WS, and E-Cigs than NS. No statistical differences for cytokines were observed among CS and WS. | Tobacco smoking is associated with poor peri-implant health. |
| BinShabaib, 2019 [54] Case-control To compare clinical periodontal status and gingival crevicular fluid cytokine profile among CS, E-Cigs and NS | 46 CS (29.5 ± 5.8 y.o.) 43M:3F 44 E-Cigs (27.8 ± 3.1 y.o.) 42M:2F 45NS (30.2 ± 4.4 y.o.) 39M:6F | Periodontal and peri-implant assessment at baseline No periodontal treatment No dental implants PI BoP PD CAL MT MBL IL-1b IL-6 TNF-a MMP-8 IFN-g | PI (p < 0.05), PD (p < 0.05) and clinical AL (p < 0.05) were significantly higher among individuals in CS than NS. BOP was manifested more among NS than in CS (p < 0.05) and E-Cigs (p < 0.05). MBL was significantly higher in CS (p < 0.01) and E-Cigs (p < 0.01) than NS The concentrations of IL-1β, IL-6, IFN-γ, TNF-α and MMP-8 were significantly higher in the GCF samples of CS (p < 0.05) than E-Cigs and NS. | Periodontal status is worse and GCF levels of pro-inflammatory cytokines are higher in CS compared with E-Cigs and NS. |
| Vohra, 2020 [55] Case-control To compare self-rated oral symptoms and clinical and radiographic periodontal status among CS, E-Cigs, and NS | 28 CS (33.3 ± 2.2 y.o.) 26 E-Cigs (31.8 ± 2.05 y.o.) 26 NS (33.5 ± 1.4 y.o.) All males | Periodontal and peri-implant assessment at baseline No periodontal treatment No dental implants PI BoP CAL PD (>/=4 mm) MT MBL | PI (p < 0.05) and PD (p < 0.05) were increased in CS than E-Cigs and NS. There was no statistically significant difference in BOP, CAL, MBL and MT among the four groups. | PI and PD are increased in CS than other groups. Pain in teeth and gums are more often perceived by CS than other groups. |
| Javed, 2017 [56] Cohort To compare clinical periodontal parameters among CS, E-Cigs and NS | 33 CS (41.3 ± 2.8 y.o.) 31 E-Cigs (37.6 ± 2.1 y.o.) 30 NS (40.7 ± 1.6 y.o.) All males | Periodontal and peri-implant assessment at baseline No periodontal treatment No dental implants PI BoP CAL PD (>/=4 mm) MT MBL | PI (p < 0.01), and PD ≥ 4 mm (p < 0.01) were significantly higher in CS and in E-Cigs compared to NS. BOP was significantly higher in NS than CS (p < 0.01) and E-Cigs (p < 0.01). There was no difference in MT, CAL and MBL among the groups. | CS show worse clinical periodontal parameters compared with E-Cigs and NS. |
| Jeong, 2020 [26] Case-control To evaluate the association of CS and E-Cigs with periodontal disease | Total number: 13,551 With and without periodontal disease CS E-Cigs Ex-CS NS ≥18 y.o. 5715M/7836F | Periodontal and peri-implant assessment at baseline (data between 2013 and 2015) No periodontal treatment No dental implants CPI | Periodontal disease was more prevalent in E-Cigs and CS than NS. E-Cigs and CS had higher dental disease (dental caries, toothache and dental damages) than NS. | E-Cigs and CS were significantly associated with increased periodontal disease. So, vaping may not be a healthy alternative to cigarette smoke. |
| Aldakheel, 2020 [57] Case-control To compare pathogenic bacteria, count from the subgingival biofilm of CS and E-Cigs with periodontitis with that of NS with and without periodontitis | 15 CS (40.5 ± 2.1 y.o.) 5 E-Cigs (38.6 ± 3.3 y.o.) 15NS with periodontitis (39.4 ± 1.6 y.o.) 15 NS without periodontitis (39.5 ± 0.8 y.o.) M/F: MD | Periodontal and peri-implant assessment: MD No periodontal treatment No dental implants PI GI PD CAL MBL | The scores of PI (p < 0.001), GI (p < 0.001), CAL (p < 0.001), PD (p < 0.001) and mesial (p < 0.001) and distal (p < 0.001) MBL were significantly higher among CS, E-Cigs, and NS with periodontitis compared with NS without periodontitis. There was no statistically significant difference in GI, PD, CAL, mesial and distal MBL and PI among CS, E-Cigs, and NS with periodontitis. | E-Cigs and CS have the same number of pathogenic bacteria in the oral-biofilm. |
| Ibraheem, 2020 [58] Case-control To compare the levels of Receptor activator of NF-kappa B ligand (RANKL) and osteoprotegerin in the gingival crevicular fluid of CS, WS, E-Cigs, NS | 30 CS (46.5 ±5.3 y.o.) 30 WS (45.5 ±4.4 y.o.) 30 E-Cigs (45.6 ± 3.6 y.o.) 30 NS (3.8 + 1.7 y.o.) All males | Periodontal and peri-implant assessment: MD No periodontal treatment No dental implants PI BoP PD CAL MBL RANKL OPG | PI (p < 0.01) and PD (p < 0.01) were significantly higher among CS, WS, E-Cigs than NS. The GCF volume was significantly higher among CS (0.92 ± 0.05 μL) (p < 0.01) and WS (0.66 ± 0.08 μL) (p < 0.001) and E-Cigs (0.62 ± 0.03 μL) (p < 0.01) than NS (0.21 ± 0.007 μL). The RANKL levels were significantly higher among CS (14.9 ± 8.2 pg/mL) (p < 0.001) and WS (12.6 ± 8.8 pg/mL) (p < 0.01) and E-Cigs (11.5 ± 8.4 pg/mL) (p < 0.01) than NS (3.5 ± 0.7 pg/mL). The OPG levels were significantly higher among CS (95.9 ± 7.2 pg/mL) (p < 0.001) and WS (86.6 ± 5.8 pg/mL) (p < 0.01) and E-Cigs (77.5 ± 3.4 pg/mL) (p < 0.05) than NS (21.5 ± 10.7 pg/mL). There was no significant difference in RANKL and OPG levels among CS, WS and E-Cigs. | CS, WS and E-Cigs have higher levels of RANKL and OPG in the GCF than NS. |
| ALHarthi, 2019 [59] Prospective To investigate the impact of cigarette smoking and vaping on periodontal tissues after a full-mouth ultrasonic scaling | 30 CS (36.4 ± 2.8 y.o.) 28 E-Cigs (32.5 ± 4.8 y.o.) 31 NS (32.6 ± 3.5 y.o.) All males | Periodontal and peri-implant assessment at baseline and after 3 and 6 months of follow-up Scaling No dental implants PI BoP PD >/= 4 mm CAL MT | At baseline, BOP was significantly higher in NS than CS and E-Cigs (p < 0.01). In CS, there was no statistically significant difference in mean PI and PD at 6 months’ follow-up compared with baseline and 3 months follow-up. In E-Cigs and NS, there was no significant difference in PI, BOP, and PD at 3 months’ (p > 0.05) and 6 months’ (p > 0.05) follow-up. | GI is worse in CS compared with E-Cigs and NS after FMUS |
| ArRejaie, 2018 [60] Case-control To compare clinical and radiographic peri-implant parameters and levels of MMP-9 and IL-1b among CS, E-Cigs and NS | 32 CS (40.4 ± 3.5) 31 E-Cigs (35.8 ± 6.2 y.o.) 32 NS (42.6 ± 2.7 y.o.) All males | Periodontal and peri-implant assessment at baseline No periodontal treatment Dental implants n. 159 (at least one positioned for >= 36 months) PI BoP PD >/= 4 mm MBL IL-1b MMP-9 | BOP was significantly higher in NS compared with CS and E-Cigs (p < 0.01). PI (p < 0.01), PD (p < 0.01),MMP-9 (p < 0.001) and IL-1b levels (p < 0.01) were significantly higher in CS and E-Cigs than NS. MBL was significantly higher in CS compared with E-Cigs and NS (p < 0.01). Significant positive associations were found between MMP-9 (p = 0.0198) and IL-1b (p = 0.0047) levels and MBL in CS; and a significant positive association between IL-1b and MBL in E-Cigs (p = 0.0031). | Higher levels of cytokines in CS and E-Cigs suggest greater peri-implant inflammatory response and so worse clinical and radiographic peri-implant parameters. |
| Al-Hamoudi, 2020 [61] Cross-sectional To investigate the effect of scaling and root planing on inflammatory cytokines IL-4, IL-9, IL-10, and IL-13 in E-Cigs and NS with periodontitis | 36 E-Cigs (47.7± 5.8 y.o.) 35NS (46.5 ± 3.4 y.o.) 62M/9F | Periodontal and peri-implant assessment at baseline and after 3 months of follow-up Scaling and root planing No dental implants PI GI PD CAL MBL IL-4 IL-9 IL-10 IL-13 Crevicular fluid volume | At baseline, there were no differences in PI, PD, CAL, MBL, and GCF IL-4, IL-9, IL-10, and IL-13 among E-Cigs and NS. At the 3-month follow-up, there were no significant differences in PI, GI, PD, CAL and MBL in E-Cigs compared to baseline, while there were significant reductions in PI, GI, and PD among NS. At the 3-month follow-up, GCF IL-4, IL-9, IL-10, and IL-13 levels were significantly elevated in E-Cigs and in NS (p < 0.05) compared to baseline.After3-month, GCF IL-4, IL-9, IL-10, and IL-13 levels were significantly higher in NS (p < 0.05) than in E-Cigs | Levels of GCF IL-4, IL-9, IL-10, and IL-13 increased after SRP in E-Cigs and NS with CP. |
| Alqahtani, 2019 [62] Cross-sectional To compare cotinine levels in the PISF among CS, WS, E-Cigs and NS | 35 CS (36.3 ± 1.2 y.o.) 33 WS (34.1 ± 1.4 y.o.) 34 E-Cigs (33.5 ± 0.7 y.o.) 35 NS (32.2 ± 0.6 y.o.) All males | Periodontal and peri-implant assessment at baseline No periodontal treatment Dental implants n.137 PI PD BoP Cotinine in the PISF | PI (p < 0.05) and PD (p < 0.05) were significantly higher in CS, WS and E-Cigs than NS BoP was higher in NS compared with CS (p < 0.05), WS (p < 0.05) and E-Cigs (p < 0.05). PISF and cotinine levels were significantly higher among CS (p < 0.05) and WS (p < 0.05) and E-Cigs (p < 0.05) than NS. | Nicotine increases the expression of cotinine in the PISF. |
| Sinha, 2020 [63] Case-control To evaluate PI, BoP, PD, TNF-α and IL-1b levels among E-Cigs and NS | 47 E-Cigs (34.6 ± 6.1 y.o.) 45 NS (44.8 ± 2.5 y.o.) All males | Periodontal and peri-implant assessment at baseline No periodontal treatment Dental implants n. 66 for E-Cigs and 55 for NS PI BoP PD PIBL IL1b TNF-a PISF volume | BOP was significantly higher in NS than E-Cigs. PD and PIBL were significantly higher in E-Cigs than NS. TNF- α levels and IL-1β levels were significantly higher in E-Cigs than NS PISF concentrations were also found relatively higher in E-Cigs than NS. | E-Cigs show PD, PIBL and TNF-α levels and IL-1β levels worse than NS |
| Al Deeb, 2020 [64] Case-control To assess the effectiveness of Photodynamic therapy in the treatment of peri-implant mucositis in CS, E-Cigs and NS | 25 CS (29.5 ± 5.8 y.o.) 21 E-Cigs (27.8 ± 3.1 y.o.) 25 NS (0.2 ± 4.4 y.o.) All males | Periodontal and peri-implant assessment at baseline and after 12 weeks Photodynamic therapy at baseline + mechanical debridement (MD) Dental implants n. 111 implants (at least one in service for ≥1.5 years) PI BoP PD TNF-a MMP-8 PISF volume | PI, PD, MMP-8 and TNF-a were higher in CS and E-Cigs than NS at baseline. BOP was higher in NS than other groups. A statistically significant reduction in PI and PD parameters was observed on baseline and at 12 weeks in all groups. BOP significantly increased in group 1 and 2 at 12 weeks. A statistically significant reduction from baseline to 12 weeks was reported in the biomarker levels for all the study groups. | PDT with adjunctive mechanical debridement reduced PI and PD and cytokines but increased BPO |
| AlJasser, 2021 [65] Case-control To evaluate the adverse effects of E-Cigs on periodontal health | 30 CS (46.9 M, 46.6 F y.o.) 32 E-Cigs (36 M, 46.8 F y.o.) 38 NS (28.6 M, 46.9 F y.o.) 70M/30F | Periodontal and peri-implant assessment at baseline, after1 month, 6 months and 1 year Surgical periodontal treatment No dental implants PD BOP CAL Cotinine levels saliva flow CO (carbon monoxide) | BOP and PD increased in all three groups, but CAL uniquely increased in E-Cigs. CS have higher carbon monoxide and salivary cotinine levels than other groups. | Among the recruited participants, CAL after 6 months was significantly worse only in the E-Cigs |
| Fangxi Xu, 2021 [66] Case-control To compare periodontal parameters among CS, E-Cigs and NS after peri-implantitis treatment | 20 CS (54.1 y.o.) 20 E-Cigs (46.8 y.o.) 20 NS (46.9 y.o.) 31M/29F | Periodontal and peri-implant assessment at baseline and after 6 months No periodontal treatment Dental implants n. 60 PD BOP PI IL-1b IL-6 MMP-8 TIMP-1 | The PI of 100% of NS changed to ‘0′ and 35% change in cigarettes and 30% change in E-Cigs which is statistically significant (p = 0.016). The mean values of PD have shown statistically significant change across the three groups over the four time intervals of observation. The comparison of mean values of IL-1 β, IL-6 and TIMP-1 showed statistically significant change across the three groups over the four intervals of observation (p < 0.0001). | Vaping was found to be the most prevalent risk indicator for peri-implantitis. |
| Periodontal Clinical and Radiographic Parameter | Author, Year Reference Study Design | Main Result(s) | Considerations |
|---|---|---|---|
| CAL | Mokeem, 2018 [50] Case-control | CAL (p < 0.05) was significantly higher among CS and WS compared to E-Cigs and NS. There was no difference in CAL, between E-Cigs and NS | CS and WS have worse CAL values compared to E-Cigs and NS; E-Cigs and NS have no significant difference in CAL values |
| CAL | BinShabaib, 2019 [54] Case-control | CAL (p < 0.05) was significantly higher among CS than NS. No differences were among E-Cigs and NS | Cigarette smoke negatively affects the CAL values |
| CAL | Vohra, 2020 [55] Case-control | There was no statistically significant difference in CAL, among the CS, E-Cigs and NS | CS, E-Cigs and NS have similar CAL values |
| CAL | Aldakheel, 2020 [57] Case-control | The scores of CAL (p < 0.001) was significantly higher among CS, E-Cigs, and NS with periodontitis compared with NS without periodontitis. There was no statistically significant difference in CAL among CS, E-Cigs, and NS with periodontitis | Periodontal bacteria negatively influence CAL values |
| CAL | Al-Hamoudi, 2020 [61] Cross-sectional | At baseline and after 3 months of follow up there were no significant differences in CAL between the E-Cigs and NS | E-Cigs do not have differences with NS for the CAL |
| CAL | ALHarthi, 2018 [59] Prospective | The levels of CAL remained unchanged in all groups | CAL remains unchanged among the groups |
| CAL | Javed, 2017 [56] Cohort | There was no statistically significant difference in CAL among individuals among the groups | Smoke does not negatively influence CAL |
| CAL | Karaaslan, 2020 [52] Case-control | There were No significant differences among the groups for mean AL | Smoke does not negatively influence CAL |
| CAL | Ibraheem, 2020 [58] Case-control | CAL was significantly higher in CS, WS and E-Cigs compared with NS | Smoke negatively affects CAL values |
| CAL | Fangxi Xu, 2021 [66] Case-control | CAL increased in E-Cigs | Vaping negatively affects CAL values |
| BoP | Mokeem, 2018 [50] Case-control | Percentage of sites with BOP were significantly higher among NS compared with CS and WS and E-Cigs. There was no statistically significant difference in BOP among CS and WS and E-Cigs | Smoke improves BoP |
| BoP | Vohra,2020 [55] Case-control | There was no statistically significant difference in BOP, among the four groups | Smoke does not influence BoP |
| BoP | Javed, 2017 [56] Cohort | BOP was significantly higher in NS than CS (p < 0.01) and E-Cigs (p < 0.01) | Smoke improves BoP |
| BoP | Ibraheem, 2020 [58] Case-control | There was no statistically significant difference in BOP among individuals in all groups | Smoke does not improve BoP |
| BoP | ALHarth, 2018 [59] Prospective |
At baseline, BOP was significantly higher in NS than CS and E-Cigs (p < 0.01). In E-Cigs and NS, there was no significant difference in BOP at 3 months’ (p > 0.05) and 6 months’ (p > 0.05) follow-up | Cigarette smoke improves Bop |
| Bop | Fangxi Xu, 2021 [66] Case-control | BoP similarly increased over time in all three groups | BoP changes in all groups |
| BoP | BinShabaib, 2019 [54] Case-control | BOP was manifested more among NS than in CS (p < 0.05) and E-Cigs (p < 0.05). No differences were among E-Cigs and NS | BoP was more often manifested among never smokers |
| PI | Mokeem, 2018 [50] Case-control | Percentage of sites with plaque were significantly higher among CS and WS compared with E-Cigs (p < 0.05) and NS (p < 0.05). Percentage of sites with plaque were significantly higher among E-Cigs compared to NS (p < 0.05). There was no statistically significant difference in PI among CS and WS and E-Cigs | Smoke increases plaque accumulation. |
| PI | Ibraheem, 2020 [58] Case-control | PI (p < 0.01) was significantly higher among CS, WS and E-Cigs than NS | Smoke gets worse PI |
| PI | Aldakheel, 2020 [57] Case-control | The scores of PI (p < 0.001) were significantly higher between CS, E-Cigs, and NS with periodontitis compared with NS without periodontitis. There was no statistically significant difference in PI among CS, E-Cigs, and NS with periodontitis | Smoke gets worse periodontitis |
| PI | ALHarthi, 2018 [59] Prospective | In CS, there was no statistically significant difference in mean PI at 6 months’ follow-up compared with baseline and 3 months’ follow-up. In E-Cigs and NS, there was no significant difference in PI at 3 months’ (p > 0.05) and 6 months’ (p > 0.05) follow-up | Vaping does not influence PI |
| PI | BinShabaib, 2019 [54] Case-control | PI (p < 0.05), was significantly higher among CS than NS. No differences were among E-Cigs and NS | Smoke gets worse plaque index |
| PI | Al-Hamoudi, 2020 [61] Cross-sectional | At baseline, there were no differences in PI, among E-Cigs and NS. At the 3-month follow-up, there were no significant differences in PI, in ES compared to baseline, while there were significant reductions in PI, among NS | PI is better among NS after 3-month follow-up |
| PI | Vohra, 2020 [55] Case-control | PD (p < 0.05) was increased in CS than E-Cigs and NS | Cigarettes smoke gets worse plaque index |
| PI | Karaaslan, 2020 [52] Case-control | No significative differences were found between the groups for PI | Smoke does not influence PI |
| PD | Vohra, 2020 [55] Case-control | PD (p < 0.05) was increased in CS than E-Cigs and NS. | Cigarette smoke gets worse PD |
| PD | Javed, 2017 [56] Cohort | PD ≥ 4 mm (p < 0.01) were significantly higher in CS and in E-Cigs compared with NS | Smoke influences PD |
| PD | Ibraheem, [58] Case-control 2020 | PD (p < 0.01) was significantly higher among CS, WS, and E-Cigs than NS | Smoke gets worse PD |
| PD | Aldakheel, 2020 [57] Case-control | The scores of PD (p < 0.001) were significantly higher among CS, E-Cigs, and NS with periodontitis compared with NS without periodontitis. There was no statistically significant difference in, PD, among CS, E-Cigs, and NS with periodontitis. | Smoke gets worse periodontitis |
| PD | ALHarthi, 2018 [59] Prospective | In CS, there was no statistically significant difference in PD at 6 months’ follow-up compared with baseline and 3 months’ follow-up. In E-Cigs and NS, there was no significant difference in PD at 3 months’ and 6 months’ follow-up | Vaping does not influence PD |
| PD | BinShabaib, 2019 [54] Case-control | PD (p < 0.05) was significantly higher among individuals in CS than NS. No differences were among E-Cigs and NS | Smoke gets worse PD |
| PD | Al-amoudi, 2020 [61] Cross-sectional | At baseline, there were no differences in PD, among E-Cigs and NS. At the 3-month follow-up, there were no significant differences in PD in E-Cigs compared to baseline values, but there was a statistically significant reductions in PD among NS | SRP improves PD in NS |
| PD | Mokeem, 2018 [50] Case-control | There was no statistically significant difference in PD among E-Cigs c and NS PD was significantly higher among CS and WS compared with E-Cigs and NS. | Vaping does not influence PD |
| PD | Karaaslan, 2020 [52] Case-control | No significative differences were found between the groups for PD | Smoke does not influence PD |
| PD | Fangxi Xu, 2021 Case-control | PD similarly increased over time in all three groups | PD increased in all three groups |
| GI | Al-amoudii, 2020 [61] Cross-sectional | At baseline, GI was significantly higher in NS than E-Cigs; at the 3-month follow-up, there were no significant differences in GI, in E-Cigs compared to baseline, while there were statistically significant reductions in GI among NS. | Vaping improves gingival conditions |
| GI | Karaaslan, 2020 [52] Case-control | GI was significantly higher in E-Cigs and EX-CS than CS and GI was significantly lower in group E-Cigs than EX-CS | Smoke improves GI |
| GI | Aldakheel, 2020 [57] Case-control | GI (p < 0.001) was significantly higher among CS, E-Cigs, and NS with periodontitis compared with NS without periodontitis. There was no statistically significant difference in GI, among CS, E-Cigs, and NS with periodontitis | GI is worst among subjects with periodontitis |
| MBL | Vohra, 2020 [55] Case-control | There was no statistically significant difference in MBL among CS, E-Cigs and NS | Smoke does not influence MBL among the groups |
| MBL | Ibraheem, 2020 [58] Case-control | MBL (p < 0.01) was significantly higher among CS, WS, E-Cigs than NS | Smoke gets worse bone loss |
| MBL | Aldakheel, 2020 [57] Case-control | The scores mesial (p < 0.001) and distal (p < 0.001) MBL were significantly higher among CS, E-Cigs, and NS with periodontitis compared with NS without periodontitis. There was no statistically significant difference in mesial and distal MBL among CS, E-Cigs, and NS with periodontitis | Smoke gets worse periodontitis |
| MBL | BinShabaib, 2019 [54] Case-control | MBL was significantly higher in CS (p < 0.01) and E-Cigs (p < 0.01) than NS No differences were among E-Cigs and NS | Smoke gets worse bone loss |
| MBL | Al-Hamoudi, 2020 [61] Cross-sectional | At baseline, there were no differences in MBL among E-Cigs and NS. At the 3-month follow-up, there were no statistically significant differences in MBL between the two groups | Vaping does not influence MBL |
| MBL | Mokeem, 2018 [50] Case-control | MBL were significantly higher among CS and WS compared to E-Cigs and NS There was no difference in MBL among E-Cigs and NS | Smoke gets worse bone loss |
| MBL | Javed, 2017 [56] Cohort | There was no difference in MBL among the groups | No differences among the groups |
| MT | Javed, 2017 [56] Cohort | There was no difference in MT among the groups | Smoke does not influence MT |
| MT | Vohra, 2020 [55] Case-control | There was no statistically significant difference in MT among CS, E-Cigs, NS | Smoke does not influence MT |
| MT | ALHarthi, 2018 [59] Prospective | There was no difference in the numbers of MT in all groups | Smoke does not influence MT |
| MT | BinShabaib, 2019 [54] Case-control | No differences among the groups | Smoke does not influence MT |
| Periodontal Inflammatory Parameter | Author, Year Reference Study Design | Main Result(s) | Considerations |
|---|---|---|---|
| IL-1b | Mokeem, 2018 [50] Case-control | There was no difference in IL-1β levels among E-Cigs and NS IL-1β (p < 0.01) levels were significantly higher among CS, WS than E-Cigs and NS | Cigarette and pipe smoke increase gingival inflammation |
| IL-1b | BinShabaib, 2019 [54] Case-control | The concentration of IL-1β was significantly higher in the GCF samples of CS (p < 0.05) than E-Cigs and NS. No differences were among E-Cigs and NS | Cigarettes smoke increase inflammation |
| IL-6 | Bin Shabaib, 2019 [54] Case-control | The concentration of IL-6 was significantly higher in the GCF samples of CS (p < 0.05) than E-Cigs and NS. No differences were among E-Cigs and NS | Cigarettes smoke increase inflammation |
| IL-6 | Mokeem, 2018 [50] Case-control | IL-6 (p < 0.01) levels were significantly higher among CS, WS than E-Cigs and NS | Cigarette smoke increases gingival inflammation |
| TNF-a | Bin Shabaib,2019 [54] | The concentration of TNF-α was significantly higher in the GCF samples of CS (p < 0.05) than E-Cigs and NS. No differences were among E-Cigs and NS | Cigarettes smoke increase inflammation |
| TNF-a | Karaaslan, 2020 [52] Case-control | TNF-a level of Group CS (4.20 +/− 0.14) was significantly higher than E-Cigs | Smoke increases gingival inflammation |
| COTININE | Mokeem, 2018 [50] Case-control | Cotinine levels were significantly higher among CS (p < 0.001) and WS (p < 0.001) and E-Cigs (p < 0.001) than NS | Smoke increases gingival inflammation |
| COTININE | Fangxi Xu, 2021 [66] Case-control | Salivary cotinine levels was highest among CS | CS have highest salivary cotinine levels |
| IL-8 | Karaslaan, 2020 [52] Case-control | IL-8 level of CS (70.47 +/− 2.76) was significantly lower than in E-Cigs and FS | Smoke improves il-8 levels |
| MMP-8 | BinShabaib, 2019 [54] Case-control | The concentrations of MMP-8 were significantly higher in the GCF samples of CS (p < 0.05) than E-Cigs and NS. No differences were among E-Cigs and NS | Cigarettes smoke increase inflammation |
| IFN-y | BinShabaib, 2019 [54] Case-control | The concentration of I FN-γ was significantly higher in the GCF samples of CS (p < 0.05) than ES and NS. No differences were among E-Cigs and NS | Cigarettes smoke increase inflammation |
| CPI | Jeong, 2020 [26] | Periodontal disease was more prevalent in E-Cigs and CS than NS | E-Cigs and CS were each significantly associated with increased periodontal disease rates. This study suggests that vaping may not be a safe alternative to smoking |
| IL-4 | Al-Hamoudi, 2020 [61] Cross-sectional | At baseline, there were no differences, and GCF IL-4 among E-Cigs and NS. At the 3-month follow-up, GCF IL-4 levels were significantly elevated in ES and in NS (p < 0.05) compared to baseline. After3-month, GCF IL-4, levels were significantly higher in NS (p < 0.05) than in E-Cigs | Levels of GCF IL-4 increased after SRP in E-Cigs and NS with CP; however, the anti-inflammatory effect of SRP was more profound in NS than in E-Cigs |
| IL-9 | Al-Hamoudi, 2020 [61] Cross-sectional | At baseline, there were no differences in IL-9, among E-Cigs and NS. At the 3-month follow-up, GCF IL-9 levels were significantly elevated in E-Cigs and in NS (p < 0.05) compared to baseline. After 3 months, GCF IL-9 levels were significantly higher in NS (p < 0.05) than in E-Cigs | Levels of GCF IL-9 increased after SRP in E-Cigs and NS with CP; however, the anti-inflammatory effect of SRP was more profound in NS than in E-Cigs |
| IL-10 | Al-Hamoudi, 2020 [61] Cross-sectional | At baseline, there were no differences in IL-10 among E-Cigs and NS. At the 3-month follow-up, GCFIL-10 levels were significantly elevated in E-Cigs and in NS (p < 0.05) compared to baseline.After3-month, GCF IL-10, was significantly higher in NS (p < 0.05) than in E-Cigs | Levels of GCF IL-10 increased after SRP in E-Cigs and NS with CP; however, the anti-inflammatory effect of SRP was more profound in NS than in E-Cigs |
| IL-13 | Al-Hamoudi, 2020 [61] Cross-sectional | At baseline, there were no differences in IL-13 among E-Cigs and NS. At the 3-month follow-up, GCF IL-13 levels were significantly elevated in E-Cigs and in NS (p < 0.05) compared to baseline.After3-month, GCF IL-13 levels were significantly higher in NS (p < 0.05) than in E-Cigs | Levels of GCF IL-13 increased after SRP in E-Cigs and NS with CP; however, the anti-inflammatory effect of SRP was more profound in NS than in E-Cigs |
| CO | Fangxi Xu, 2021 [66] Case-control | CO levels was highest among CS | CS have highest CO levels |
| GSH-PX AND 8-OHdG | Karaaslan, 2020 [52] Case-control | Although the GSH-Px level of Group II was higher than Group I, this difference was not statistically significant, but the mean GSH-Px level of Group III was significantly higher than in Groups I and II. There was no significant association among the groups | CS and E-Cigs had the same unfavorable effects on the markers of oxidative stress and inflammatory cytokines |
| RANKL | Ibraheem, 2020 [58] Case-control | The RANKL levels were significantly higher among CS (14.9 ± 8.2 pg/mL) (p < 0.001) and WS (12.6 ± 8.8 pg/mL) (p < 0.01) and E-Cigs (11.5 ± 8.4 pg/mL) (p < 0.01) than NS (3.5 ± 0.7 pg/mL). There was no significant difference in RANKL among CS, WS and ES | CS and WS and E-Cigs are associated with an increased expression of RANKL in the GCF |
| OPG | Ibraheem, 2020 [58] Case-control | The OPG levels were significantly higher among CS (95.9 ± 7.2 pg/mL) (p < 0.001) and WS (86.6 ± 5.8 pg/mL) (p < 0.01) and E-Cigs (77.5 ± 3.4 pg/mL) (p < 0.05) than NS (21.5 ± 10.7 pg/mL) | CS and WS and E-Cigs are associated with an increased expression of OPG in the GCF |
| Clinical and Radiographic Peri-Implant Parameters | Author, Year Reference Study Design | Main Result(s) | Considerations |
|---|---|---|---|
| BoP | ArRejaie, 2018 [60] Case-control | Peri-implant BOP was significantly higher in NS compared with CS and E-Cigs (p < 0.01) | Smoke improves Bop |
| BoP | Al-Aali, 2018 [51] Case-control | BOP was statistically significantly higher in NS compared to E-Cigs (p < 0.01) | Vaping smoke improves Bop |
| BoP | Sinha, 2020 [63] Case-control | BOP was significantly higher in NS than E-Cigs | Vaping smoke improves Bop |
| BoP | Alqahtani, 2019 [62] Cross-sectional | BoP was higher in NS compared with CS (p < 0.05),WS (p < 0.05) and E-Cigs (p < 0.05) | Smoke improves Bop |
| BoP | Al Deeb, 2020 [64] Case-control | BOP was statistically significantly higher in NS than other groups | Smoke improves Bop |
| BoP | Alqahtani, 2019 [62] Cross-sectional | BOP in CS, WS, and E-Cigs showed statistical differences (p < 0.01) respect to NS | Smoke improves Bop |
| BoP | Al-Aali, 2018 [51] Case-control | BOP was statistically significantly higher in NS compared to E-Cigs (p < 0.01) | Vaping smoke improves Bop |
| BoP | AlJasser, 2021 [65] | The prevalence of BOP was observed in the three groups as 72%, (CS) 76.5% (E-Cigs) and 88.9% (NS) at baseline | Smoke improves Bop |
| PI | Alqahtani, 2019 [62] Cross-sectional | PI was significantly higher among individuals CS (p < 0.05), WS (p < 0.05), and E-Cigs (p < 0.05) compared with NS | Smoke gets worse plaque index |
| PI | ArRejaie, 2018 [60] Case-control | PI (p < 0.01 was significantly higher in CS and E-Cigs than NS. There were differences statistically significantly among CS and E-Cigs | Cigarettes smoke gets worse plaque index |
| PI | Al-Aali, 2018 [51] Case-control | PI showed no significant difference between NS and E-Cigs | Vaping does not influence PI |
| PI | Sinha, 2020 [63] Case-control | PI showed no significant difference between NS and E-Cigs | Vaping does not influence PI |
| PI | Alqahtani, 2019 [62] Cross-sectional | PI (p < 0.05) was significantly higher in CS, WS and E-Cigs than NS | Smoke gets worse plaque index |
| PI | Al Deeb, 2020 [64] Case-control | PI was higher in CS and E-Cigs than NS at baseline. Statistically significant reduction in PI was observed on baseline and at 12 weeks in all groups | Smoke gets worse plaque index |
| PI | AlJasser, 2021 [65] Case-control | PI of 100% of NS had changed to ‘0′ and 35% change in cigarettes and 30% change in E-Cigs which is statistically significant (P = 0.016) | PI was higher in NS than other groups |
| PD | ArRejaie, 2018 [60] Case-control | PD was significantly higher in CS and E-Cigs than NS | Smoke gets worse PD |
| PD | Al-Aali, 2018 [51] Case-control | PD was statistically significantly higher in E-Cigs than NS | Vaping gets worse PD |
| PD | Sinha, 2020 [63] Case-control | PD was significantly higher in E-Cigs than NS | Vaping gets worse PD |
| PD | Alqahtani, 2019 [62] Cross-sectional | PD was significantly higher among CS, WS and E-Cigs compared with NS. Among smokers, CS and WS showed significantly higher PD compared with E-Cigs | Smoke gets worse PD |
| PD | Alqahtani, 2019 [62] Cross-sectional | PD (p < 0.05) was significantly higher in CS, WS and E-Cigs than NS | Smoke gets worse PD |
| PD | Al Deeb, 2020 [64] Case-control | PD was higher in CS and E-Cigs than NS at baseline. Statistically significant reduction in PD parameter was observed on baseline and at 12 weeks in all groups | Smoke gets worse PD |
| PD | AlJasser, 2021 [65] Case-control | PD have shown statistically significant change across the three groups over the four-time intervals of observation (P = 0.024) | Smoke gets worse PD |
| MBL | Al-Aali, 2018 [51] Case-control | Peri-implant bone loss was statistically significantly higher in E-Cigs than NS | Smoke increases periodontal inflammation |
| MBL | ArRejaie, 2018 [60] Case-control | MBL was significantly higher in CS compared with E-Cigs and NS (p < 0.01). There were differences statistically significantly among CS and E-Cigs | Cigarettes smoke gets worse bone loss |
| MBL | Sinha, 2020 [63] Case-control | MBL was significantly higher in E-Cigs than NS | smoke gets worse bone loss |
| MBL | Alqahtani, 2019 [62] Cross-sectional | MBL (p < 0.01) was significantly higher among CS, WS and E-Cigs compared with NS. CS and WS showed significantly higher Peri-implant BL compared with E-Cigs (p < 0.05) | Cigarettes smoke gets worse bone loss |
| PD | ArRejaie, 2018 [60] Case-control | PD was significantly higher in CS and E-Cigs than NS | Smoke gets worse PD |
| Inflammatory Periodontal Parameter | Author, Year Reference Study Design | Main Result(s) | Considerations |
|---|---|---|---|
| IL-1 b | ArRejaie, 2018 [60] Case-control | IL-1 b levels were statistically significantly higher in CS than E-Cigs and NS IL-1-b levels were statistically significantly higher in E-Cigs than NS | Smoke increase inflammation |
| IL-1 b | Alqahtani, 2019 [62] Cross-sectional | IL-1β was significantly higher in CS, WS, and E-Cigs than NS. No statistical differences for cytokines were observed between CS and WS | Smoke increases gingival inflammation |
| IL-1 b | Al-Aali, 2018 [51] Case-control | IL-1b (p < 0.01) was statistically significantly higher in E-Cigs than NS | Smoke increases gingival inflammation |
| IL-1 b | Sinha, 2020 [63] Case-control | IL-1 b levels was significantly higher in E-Cigs than NS | Smoke increases gingival inflammation |
| IL-1 b | AlJasser, 2021 [65] Case-control | Comparison of mean IL-1β values showed statistically significant variation between the three groups in the four observation intervals (p < 0.0001) | Smoke increases inflammation |
| IL-6 | AlJasser, 2021 [65] Case-control | Comparison of mean IL-6 values showed a statistically significant change between the three groups in the four observation intervals (p < 0.0001) | Smoke increases inflammation |
| TNF-a | Sinha, 2020 [63] Case-control | TNF- α levels was significantly higher in E-Cigs than NS | Smoke increases inflammation |
| TNF-a | Al Deeb, 2020 [64] Case-control | TNF-a was higher in CS and E-Cigs than NS at baseline. A statistically significant reduction from baseline to 12 weeks was reported in the biomarker levels for all the study groups. | Smoke increases inflammation |
| TNF-a | Al-Aali, 2018 [51] Case-control] | TNF-a (p < 0.001) was statistically significantly higher in E-Cigs than NS | Smoke increases gingival inflammation |
| TNF-a | Alqahtani, 2019 [62] Cross-sectional | TNF-α was significantly higher in CS, WS, and E-Cigs than NS. No statistical differences for cytokines were observed among CS and WS | Smoke increases gingival inflammation |
| COTININE | Alqahtani, 2019 [62] Cross-sectional | PISF and cotinine levels were significantly higher in CS (p < 0.05) and WS (p < 0.05) and E-Cigs (p < 0.05) than NS. | Smoke increases gingival inflammation |
| PISF (peri-implant sulcular fluid) | Sinha, 2020 [63] Case-control | PISF concentrations were found relatively higher in E-Cigs than NS | Smoke increases gingival inflammation |
| PISF | Alqahtani, 2019 [62] Cross-sectional | PISF is significantly higher among smokers than NS | Smoke increases gingival inflammation |
| PISF | Alqahtani, 2019 [62] Cross-sectional | PISF is higher among smokers | Smoke increases gingival inflammation |
| PISF | Al-Aali, 2018 [51] Case-control | The PISF volume (p < 0.05) collected for E-Cigs was statistically significantly higher than NS | Smoke increases gingival inflammation |
| PISF | ArRejaie, 2018 [60] Case-control | The PISF volume (p < 0.01) collected for CS and vaping individuals was statistically significantly higher than NS | Smoke increases gingival inflammation |
| PISF | Al Deeb, 2020 [64] Case-control | A statistically significant reduction from baseline to 12 weeks was reported in the biomarker levels for all the study groups. | A statistically significant reduction was reported in the biomarker levels for all the study groups. |
| MMP-8 | Al Deeb, 2020 [64] Case-control | MMP-8 was higher in CS and ES than NS at baseline. A statistically significant reduction from baseline to 12 weeks was reported in the biomarker levels for all the study groups | Smoke increases inflammation |
| MMP-8 | AlJasser, 2021 [65] Case-control | The comparison of mean values of MMP-8, has shown statistically significant change across the three groups over the four intervals of observation (p < 0.0001) | Smoke increases inflammation |
| MMP-9 | ArRejaie, 2018 [60] Case-control | MMP-9 (p = 0.0198) levels were statistically significantly higher in CS than E-Cigs and NS MMP-9 (p = 0.0198) levels were statistically significantly higher in E-Cigs than NS | Smoke increases inflammation |
| Study | Bias Due to Confounding | Bias in Selection of Participants | Bias in Measurement Classification of Interventions | Bias Due to Deviations from Intended Interventions | Bias Due to Missing Data | Bias in Measurement of Outcomes | Bias Due to Selection of the Reported Result |
|---|---|---|---|---|---|---|---|
| Mokeem [50] | Y/PY/ PN/N | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI |
| Al-Aali [51] | Y/PY/ PN/N | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI |
| Karaaslan [52] | Y/PY/ PN/N | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI |
| AlQahtani [53] | Y/PY/ PN/N | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI |
| BinShabaib [54] | Y/PY/ PN/N | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI |
| Vohra [55] | Y/PY/ PN/N | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI |
| Javed [56] | Y/PY/ PN/N | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI |
| Jeong [26] | Y/PY/ PN/N | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI |
| Aldakheel [57] | Y/PY/ PN/N | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI |
| Ibraheem [58] | Y/PY/ PN/N | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI |
| ALHarthi [59] | Y/PY/ PN/N | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI |
| ArRejaie [60] | Y/PY/ PN/N | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI |
| Al-Hamoudi [61] | Y/PY/ PN/N | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI |
| Alqahtani [62] | Y/PY/ PN/N | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI |
| Sinha [63] | Y/PY/ PN/N | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI |
| Al Deeb [64] | Y/PY/ PN/N | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI |
| AlJasser [65] | Y/PY/ PN/N | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI |
| Fangxi Xu [66] | Y/PY/ PN/N | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI | Y/PY/ PN/N/NI |
| Risk of bias judgements | CRITICAL | SERIOUS | LOW | LOW | LOW | MODERATE | LOW |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
D’Ambrosio, F.; Pisano, M.; Amato, A.; Iandolo, A.; Caggiano, M.; Martina, S. Periodontal and Peri-Implant Health Status in Traditional vs. Heat-Not-Burn Tobacco and Electronic Cigarettes Smokers: A Systematic Review. Dent. J. 2022, 10, 103. https://doi.org/10.3390/dj10060103
D’Ambrosio F, Pisano M, Amato A, Iandolo A, Caggiano M, Martina S. Periodontal and Peri-Implant Health Status in Traditional vs. Heat-Not-Burn Tobacco and Electronic Cigarettes Smokers: A Systematic Review. Dentistry Journal. 2022; 10(6):103. https://doi.org/10.3390/dj10060103
Chicago/Turabian StyleD’Ambrosio, Francesco, Massimo Pisano, Alessandra Amato, Alfredo Iandolo, Mario Caggiano, and Stefano Martina. 2022. "Periodontal and Peri-Implant Health Status in Traditional vs. Heat-Not-Burn Tobacco and Electronic Cigarettes Smokers: A Systematic Review" Dentistry Journal 10, no. 6: 103. https://doi.org/10.3390/dj10060103
APA StyleD’Ambrosio, F., Pisano, M., Amato, A., Iandolo, A., Caggiano, M., & Martina, S. (2022). Periodontal and Peri-Implant Health Status in Traditional vs. Heat-Not-Burn Tobacco and Electronic Cigarettes Smokers: A Systematic Review. Dentistry Journal, 10(6), 103. https://doi.org/10.3390/dj10060103











