Abstract
Two zinc(II) hexaborate(2−) complexes, [Zn{κ3O-B6O7(OH)6}(κ3N-dien)]·0.5H2O (dien = NH(CH2CH2NH2)2) (1) and (NH4)2[Zn{κ2O-B6O7(OH)6}2(H2O)2]·2H2O (2), and a zinc(II) dodecaborate(6−) complex, (1,3-pnH2)3[(κ1N-H3N{CH2}3NH2)Zn{κ3O-B12O18(OH)6}]2·14H2O (1,3-pn = 1,3-diaminopropane) (3), have been synthesized and characterized by single-crystal XRD studies. The complexes crystallized through self-assembly processes, from aqueous solutions containing 10:1 ratios of B(OH)3 and appropriate Zn(II) amine complex: [Zn(dien)2](OH)2, [Zn(NH3)4](OH)2, and [Zn(pn)3](OH)2. The hexaborate(2−) anions in 1 and 2 are coordinated to octahedral Zn(II) centres as tridentate (1) or bidentate ligands (2) and the dodecaborate(6−) ligand in 3 is tridentate to a tetrahedral Zn(II) centre.
1. Introduction
There are more than two hundred known borate (polyborate) minerals, and many more known synthetic polyborates [1,2,3]. Borates are generally comprised of cationic moieties partnered with anionic units containing boron, oxygen, and in many cases hydroxyl hydrogen. Oxidoborates (or hydroxyoxidoborates) are the more appropriate terms, but the term borate (or polyborate) has been used for many years and will be used in this manuscript. Borates are a class of compounds with rich structural diversity [4,5,6,7], and have been synthesized by solvothermal methods or from aqueous solution by the addition of B(OH)3 to a solution containing the appropriate templating cation [7]. Polyborate salts obtained from aqueous solution usually contain discrete, isolated or insular hydroxyl anions, whilst polyborate salts prepared via solvothermal methods are often more condensed and contain anionic polymeric 1-D chains, 2-D layers or 3-D networks with a variety of framework building blocks [1,7]. Salts formed from aqueous solution often contain the pentaborate(1−) [B5O6(OH)4]− anion since this anion is structurally well suited to forming crystalline supramolecular lattices, which are held together by strong H-bond interactions [8,9,10,11]. We have developed a strategy to overcome pentaborate(1−) salt formation by utilizing more highly charged (> (+1)) metal complex cations with ligands having the potential to form multiple H-bond interactions to template crystallization from aqueous solution of polyborate salts of unusual structures. In this context we have isolated two novel polyborate anions: [B7O9(OH)6]3− [12] and [B8O10(OH)6]2− [13]. We have also recently started to investigate Zn(II)/polyborate chemistry and have been able to isolate an insular bi-Zn(II) complex containing a rare dodecaborate(6−) anion [14] and two polymeric 1-D coordination chains with hexaborate(2−) ligands bridging Zn(II) centres [15]. There are a number of other structural reports on polyborate/Zn(II) chemistry [16,17,18,19,20,21,22,23], including the industrially important Zn[B3O4(OH)3] [24].
In this manuscript we describe the synthesis and XRD structures of two new Zn(II)/hexaborate(2−) complexes: [Zn{κ3O-B6O7(OH)6}(κ3N-dien)]·0.5H2O (dien = NH(CH2CH2NH2)2) (1) and (NH4)2[Zn{κ2O-B6O7(OH)6}2(H2O)2]·2H2O (2). We also report a Zn(II)/dodecaborate(6−) complex (1,3-pnH2)3[(κ1N-H3N{CH2}3NH2)Zn{κ3O-B12O18(OH)6}]2·14H2O (1,3-pn = 1,3-diaminopropane) (3). All three complexes are insular and the hexaborate(2−) ligand is tridentate in 1, whereas in 2 it is bidentate to octahedral Zn(II) centres. The dodecaborate(6−) ligand in 3 is tridentate to a tetrahedral Zn(II) centre. The structures of these two anions are drawn schematically in Figure 1.
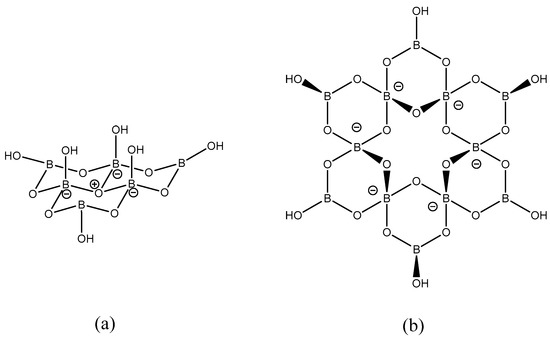
Figure 1.
The (a) hexaborate(2−) anion, [B6O7(OH)6]2−, observed in 1 and 2; and (b) dodecaborate(6−) anion, [B12O18(OH)6]6−, observed in 3. These diagrams show the location of formal Lewis charges.
2. Results and Discussion
2.1. Synthesis and Characterization
Compounds 1, 2 and 3 were prepared in moderate yield through crystallization from aqueous solution initially containing B(OH)3 and [Zn(dien)2](OH)2, [Zn(NH3)4](OH)2 or [Zn(pn)3](OH)2 for 1, 2 and 3, respectively. The hydroxide salts were prepared in situ from the corresponding sulphate salts by the addition of Ba(OH)2 and removal of precipitated BaSO4 (Scheme 1).
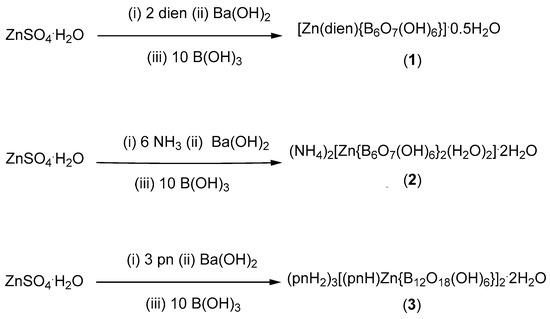
Scheme 1.
Synthesis of Zn(II) hexaborate(2−) and dodecaborate(6−) complexes (dien = NH(CH2CH2NH2)2, pn = 1,3-diaminopropane).
Compounds 1, 2 and 3 are formed through self-assembly processes. B(OH)3, when dissolved in aqueous solution at moderate to high pH, exists not as boric acid but as a dynamic combinatorial library (DCL) [25,26] of a variety of polyborate anions which are in rapid equilibria [27,28]. Likewise, Zn(II) complexes are labile [29], and a DCL of Zn(II)/amine species are also present in the solution. The products crystallize from solution maximizing energetically favourable solid-state interactions, including coordination bonds, Coulombic attractions, H-bonding and steric effects [30,31].
Compounds 1, 2 and 3 were characterized spectroscopically (NMR and IR), by thermal DSC/TGA analysis and by single-crystal XRD studies (Section 2.2). They all gave satisfactory bulk elemental analysis.
The thermal TGA/DSC data obtained for 1–3 (see Supplementary Materials) were consistent with the structures determined by single-crystal X-ray diffraction studies (see below) and can be interpreted by multi-step decomposition processes. For 1 this involved loss of interstitial water (<190 °C), further loss of water with cross-condensation of hexaborate(2−) ligands (190–380 °C) and finally oxidation and/or evaporation of the organic dien ligand (380–650 °C) to leave an anhydrous zinc borate ZnB6O10 (= ZnO·3B2O3) as a glassy residue. Glassy solids with masses consistent with ZnB12O19 (= ZnO·6B2O3) were obtained as the final residues for both 2 and 3 since the initial starting Zn/B ratio was 1:12. The thermal decomposition of 3 followed a similar pattern to 1. Compound 2 had a TGA trace consistent with loss of initial interstitial water (<110 °C), loss of ammonia (110–250 °C), and final condensation of hexaborate(2−) anions (250–500 °C). Similar thermal behaviour has been observed in other metal polyborate species [12,13,24,32,33,34,35], including 1-D zinc hexaborate(1−) coordination polymers [Zn(en){B6O7(OH)6}·2H2O and [Zn(pn){B6O7(OH)6}]·1.5H2O [15]. Magnetic susceptibility χm data for 1–3 were ~ −200 × 10−6 cm3·mol−1 and typical for diamagnetic zinc(II) complexes.
IR spectra can be used to characterize polyborate species since characteristic B–O stretches are generally strong and often diagnostic [36]. Hexaborate(2−) ions, which are never “isolated” and usually found coordinated tridentate to metal centres, have been reported to show such bands at ~953(m) cm−1 and 808(s) cm−1. Compound 1 displayed bands at 950(m), 861(m) and 806(s) whilst 2 showed bands at 953(m), 904 (s) and 857(m). Thus, the strong band usually observed at 808 cm−1 was absent in 2 and replaced by a strong band at 904 cm−1. This may be a reflection on the unusual centrosymmetric bidentate hexaborate(2−) coordination mode observed in 2. The IR spectrum of 3 showed peaks at 1047(s), 952(m), 902(s) and 855(m), and there were corresponding absorptions in the reported spectrum of [(H3NCH2CH2NH2)Zn{B12O18(OH)6}Zn(en)(NH2CH2CH2NH3)]·8H2O [14], which also contains a coordinated dodecaborate(6−) ion. Possible diagnostic absorption bands for this anion have not been described before.
Compounds 1–3 were all insoluble in organic solvents but “dissolved” with decomposition in aqueous solution. 1H, 11B spectra of these solutions were obtained in D2O, as were the 13C spectra of 1 and 3. The 1H and 13C spectra showed peaks consistent with the organics present and the 1H spectra additionally displayed at H2O/exchangeable hydrogen peak (H2O, NH, BOH) at ~4.8 ppm. 11B spectra of 1–3 all showed a single signal at a + 17.4, +15.9 and +14.0 ppm, respectively. These signals are all downfield of those calculated [10] (at infinite dilution) for the boron/charge ratio of three (+13.8) for a hexaborate(2−) system, and two (+11.0) for the dodecaborate(6−) ions. This assumes fast B(OH)3/[B(OH)4]− exchange [27,28] and is also associated with the pH of the solution. The influence of the zinc(II) ions may also be important here by reducing the effective charge at boron.
2.2. X-ray Diffraction Studies
The structures of 1, 2 and 3 were determined by single-crystal XRD methods. Crystal data are given in the experimental section and all XRD data are available as Supplementary Materials.
Compounds 1 and 2 both contained the hexaborate(2−) anion coordinated to a Zn(II) centre and the structures of 1 and 2, showing their atomic numbering schemes, are shown in Figure 2 and Figure 3, respectively. The anionic complex in 2 was centrosymmetric with the asymmetric unit comprising of half the anion with the zinc(2+) ion on the inversion centre. Compound 1 was a neutral zinc(II) complex with 0.5 waters of crystallization. The neutral Zn(II) complex, [Zn{B6O7(OH)6}(dien)], contained a tridentate (κ3N) dien ligand and a tridentate (κ3O) hexaborate(2−) ligand. Compound 1 was disordered with two heavy atoms (O10, C4) of the ligand, and associated hydrogen atoms, split in a 1:1 ratio. One position also had an associated water of crystallization (O21). Compound 2 was a salt comprised of [NH4]+ cations, [Zn{B6O7(OH)6}2(H2O)2]2− anions and interstitial H2O molecules. Both hexaborate(2−) ligands in 2 were bidentate (κ2O) and the coordinated H2O molecules were trans. The Zn–O (hexaborate) distances in 2 {2.0692(9) Å (O11) and 2.1208(9) Å (O12)} were within the range of distances observed for 1 {2.0612(11)–2.1864(10) Å} despite the change in coordination mode of the hexaborate(2−) ligand. The Zn–O (H2O) distance in 2 was 2.1292(9) Å (O21), and the three Zn–N (dien) distances in 1 ranged from 2.1283(14)–2.1473(15) Å. The angles about the Zn(II) centres were 82.56(5)–100.26(5)° and 166.45(5)–175.22(5)° for 1, and 87.90(3)–92.10(3)° and 180.00° for 2. These angles and distances were consistent with previous reported octahedral complexes of Zn(II) with O and N donor ligands [37]. Bond lengths (B–O) and OBO and BOB bond angles associated with the hexaborate(2−) ligands in both 1 and 2 were very similar. For example, bond lengths to the central pyramidal O+ (1.5154(18)–1.5231(18) Å, 1; 1.5053(15)–1.5247(16) Å, 2) > other bond lengths to four coordinate borons (1.4407(19)–1.4791(19)Å, 1; 1.4413(18)–1.4889(15) Å, 2) > bond-lengths to three coordinate borons (1.362(2)–1.418(4) Å, 1; 1.3570(17)–1.3793(17) Å, 2) and consistent with distances and angles previously reported specifically for hexaborate(2−) complexes [15,32,38,39] and related polyborate systems [8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,32,33,34,35,36,38,39,40].
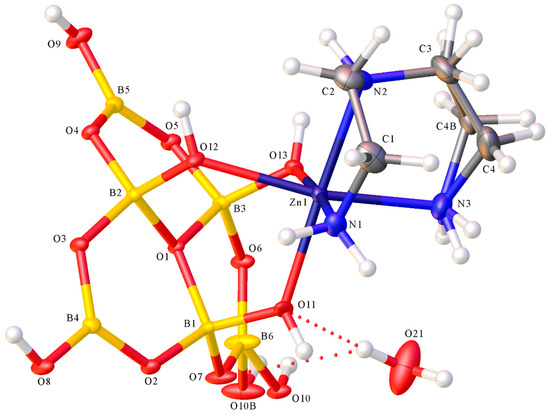
Figure 2.
Molecular structure of [Zn{κ3O-B6O7(OH)6}(κ3N-dien)]·0.5H2O (dien = NH(CH2CH2NH2)2) (1) showing atomic labelling.
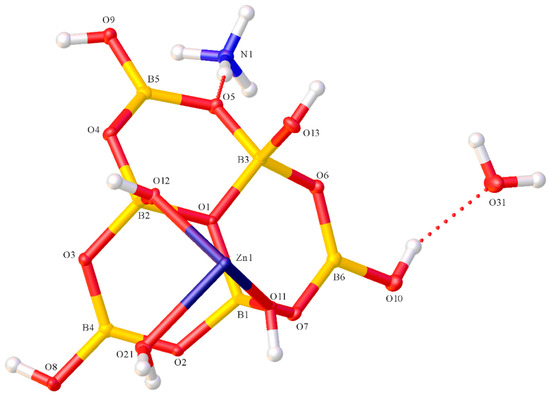
Figure 3.
Molecular structure of the asymmetric unit of (NH4)2[Zn{κ2O-B6O7(OH)6}2(H2O)2]·2H2O (2), showing atomic labelling.
H-bonding interactions are commonly observed in most polyborate solid-state structures. They were observed at many locations in the solid-state structures of 1 and 2 and must be partly responsible for the self-assembly of these structures from their constituents. Compound 1 showed H-bond interactions between the neutral complexes as well as these complexes and the water of crystallization. Compound 2 showed H-bond cation/anion and anion/H2O interactions. The energetically favourable reciprocal R22(8) (Etter [41] nomenclature) O8H8→O3*, O8*H8*→O3) linked hexaborate(2−) units in 1. There were also unusual R22(6) (O9H9→O12*H12*→O4) and R22(8) (N2H2→O8* and O13H13→O2*) arrangements between neighbouring hexaborate units in 1; the latter ring included Zn(1). Compound 2 also had two energetically favourable reciprocal R22(8) interactions between neighbouring hexaborate(2−) units (O13H13→O6*, O13*H13*→O6 and O8H8→O3*,O8*H8*→O3). There was also an unusual intramolecular H-bond in 2 between the coordinated H2O molecule and the hexaborate(2−) ligand (O21H21A→O13) as part of an intramolecular R11(8) system incorporating the Zn1 centre (Figure 4). The coordinated H2O also H-bonded to a neighbouring hexaborate O21H21B→O2*. O13 is the hexaborate hydroxyl oxygen atom that fulfilled the role as third coordination donor atom in 1 and in other tridentate hexaborate complexes. In this particular local environment of 2, the energetics of forming this H-bond and the H2O–Zn coordination bond must outweigh the energetics of a simple borate O–Zn coordinate bond. O13H13 also H-bonded to a neighbouring hexaborate (O13H13→O6*). Full details of these H-bond interactions are given in the Supplementary Materials.
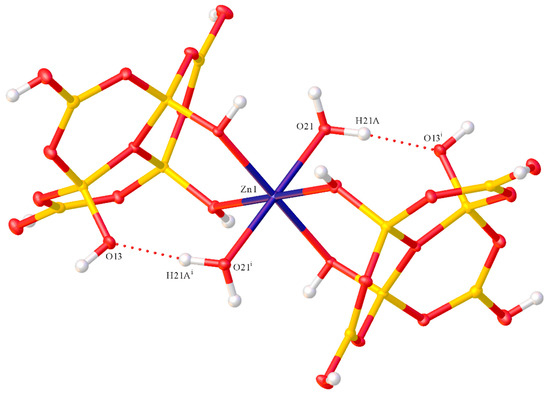
Figure 4.
The intramolecular O21H21A→O13 H-bond interaction in 2. [d(O21–H21) 0.87 Å, d(H21–O13) 1.79 Å; d(O21···O13) 2.6446(13) Å; angle O21H21O13, 169.7°] which is part of two R11(8) rings, incorporating Zn–O coordinate bonds (symmetry i = 2 − x, 1 − y, 2 − z).
Compound 3 was an ionic compound comprised of [H3N(CH2)3NH3]2+ cations and [(H3N(CH2)3NH2)ZnB12O18(OH)6]3− anions, with the anions containing the dodecaborate(6−) ligand coordinated κ3O to a tetrahedral Zn(II) centre which also had a monoprotonated monodentate κ1N-H3N(CH2)3NH2 ligand. There were also seven waters of crystallization per Zn(II) centre. A diagram of the structure is shown in Figure 5.
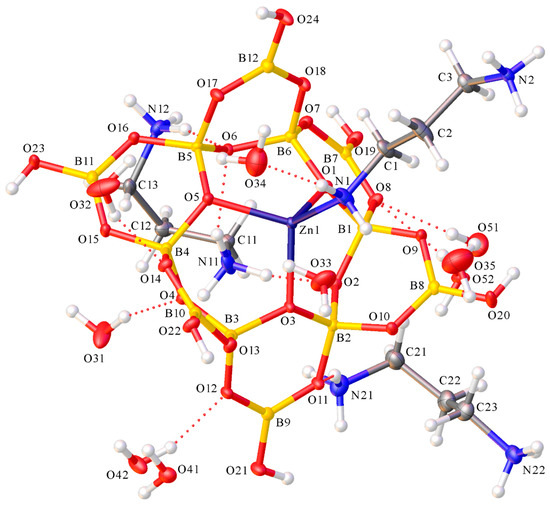
Figure 5.
Diagram of (1,3-pnH2)3[(κ1N-H3N{CH2}3NH2)Zn{κ3O-B12O18(OH)6}]2·14H2O (1,3-pn = 1,3-diaminopropane) (3) showing atomic labelling.
The Zn–O (dodecaborate) distances in 3 {1.9592(18) Å (O3)–1.9717(18) Å (O1)} were shorter than those observed for 1 or 2, reflecting tetrahedral vs. octahedral coordination geometries. The Zn1N1 distance was 2.006(2) Å, and internuclear angles about Zn1 ranged from 103.43(7)–117.86(9)°. These data are very similar to those of the closely related di-Zn(II) complex [(NH3CH2CH2NH2)Zn{B12O18(OH)6}Zn(en)(NH2CH2CH2NH3)] [14] that features 1,2-diaminoethane. The dodecaborate(6−) anion (Figure 1b) is comprised of six boroxole rings fused so as to produce a larger central {B6O6} ring, with each boron atom within this ring carrying a formal negative charge due to their four-coordinate nature. This anion was first reported in 1990 in the structure of Ag6[B12O18(OH)6]·3H2O [42]. The dodecaborate(6−) anion in 3 is closely related to the deprotonated structures found in Na8[B12O20(OH)4] [43] and Zn6[B12O24] [44]. The central ring oxygen atoms alternate up and down on different sides of the central ring and are ideally set-up to bind tridentate to a metal centre. The dodecaborate(6−) anion has been previously observed to coordinate in a tridentate mode in the following compounds: [(NH3CH2CH2NH2)Zn{B12O18(OH)6}–Zn(en)(NH2CH2CH2NH3)] [14], Na2Cs4Ba2[B12O18(OH)6](OH)4 [45], K7[(BO3)Mn{B121O18(OH)6}]·H2O [46] and K7[(BO3)Zn{B12O18(OH)6}]·H2O [47].
The six four-coordinate boron atoms had B–O distances ranging between 1.441(3)–1.506(3) Å and their O–B–O angles ranged from 106.3(2)–112.1(2)°. The remaining six borons of the anion were three-coordinate and had significantly shorter B–O distances {1.351(3)–1.386(3) Å} and larger O–B–O angles {115.4(2)–123.1(2)°}. These bond lengths are similar to those observed for [(NH3CH2CH2NH2)Zn(B12O18(OH)6}Zn(en)(NH2CH2CH2NH3)] [14], other similarly fused boroxole systems [14,45,46,47] and the hexaborate(2−) complexes 1 and 2.
The hydroxyl hydrogen atom, the amino hydrogen atoms of the protonated 1,3-diaminopropane ions and ligands and the waters of crystallization form numerous H-bond interactions and they were presumably responsible—at least in part—for this remarkable self-assembly from mono-boron species. There are numerous cation/anion H-bond interactions, and three of the six potential dodecaborate hydroxyl interactions are R22(8): O20H20→O15*, O23H23→O9* and O24H24→O17*, with only the latter reciprocal. “Simple” inter-borate H-bonds originate from O19H19 and O22H22 whilst O21H21 has a non-borate interaction and H-bonds to an H20 (O31). This configuration contrasts with that of [(NH3CH2CH2NH2)Zn{B12O18(OH)6}Zn(en)(NH2CH2CH2NH3)], where all six were involved in R22(8) interactions. However, a structural motif that is similar to that found in [(NH3CH2CH2NH2)Zn{B12O18(OH)6}Zn(en)(NH2CH2CH2NH3)] is that amino hydrogen atoms of the uncoordinated nitrogen (N2) of the H3N(CH2)3NH2 ligand H-bond and link with dodecaborate(6−) units of adjacent complexes. Full details of these H-bond interactions are in the Supplementary Materials.
3. Experimental
3.1. General
All chemicals were obtained from commercial sources. Combustion analysis (CHN) were obtained from OEA laboratories Ltd. in Callington, Cornwall, UK. NMR spectra were obtained on a Bruker Avance spectrometer (Bruker, Coventry, UK) (in D2O) operating at 400.1 MHz (1H), 100.6 MHz (13C) or 128.4 MHz (11B) with data reported as δ (ppm) with positive chemical shifts to a high frequency of tetramethylsilane (TMS) (1H, 13C) and BF3·OEt2 (11B). FTIR spectra were obtained on a PerkinElmer 100 FTIR spectrometer (PerkinElmer, Seer Green, UK) as KBr pellets. TGA/DSC analyses were undertaken in air on an SDT Q600 V4.1 Build 59 instrument (New Castle, DE, USA), using Al2O3 crucibles between 10–800 °C with a ramp temperature rate of 10 °C·min−1.
3.2. Synthesis, Spectroscopic, Analytical and Crystallographic data for 1
A solution of NH(CH2CH2NH2)2 (dien) (2.16 mL, 20 mmol) in H2O (5 mL) was added to a solution of ZnSO4·H2O (1.79 g, 10 mmol) in H2O (10 mL). The reaction mixture was stirred at room temperature for 60 min before the addition of Ba(OH)2·8H2O (3.15 g, 10 mmol) in H2O (25 mL). This mixture was rapidly stirred for a further 30 min. The white precipitate of BaSO4 was removed by filtration and B(OH)3 (6.18 g, 10 mmol) dissolved in H2O (50 mL) was added to the filtrate, which was further stirred at room temperature for 3 h. The volume of this solution was reduced to 20 mL by gentle evaporation in a warm water bath. The concentrated solution was left for 10 days in NMR tubes for crystallization and yielded colourless crystals of [Zn(dien){B6O7(OH)6}]·0.46H2O (1) (1.9 g, 42%). Mp ≥ 300 °C. Anal. Calc.: C = 10.5%, H = 4.4%, N = 9.2%. Found: C = 10.7%, H = 4.1%, N = 9.3%. NMR. 1H/ppm: 2.5 (m, 8H), 4.8 (s, 37H, NH2, H2O, OH). 13C/ppm: 38.10. 11B/ppm: 17.4. IR (KBr/cm−1): 3549(s), 3384(s), 1642(m), 1442(s), 1427(s), 1362(s), 1249(m), 1193(s) 1108(s), 1028(s), 951(m), 861(m), 808(m). TGA: 100–190 °C, loss of 0.46 interstitial H2O 2.5 (1.8 calc.); 190–380 °C, condensation of polyborate with loss of three further H2O 15.2% (13.7% calc.); 380–650 °C, oxidation of dien 38.5% (36.3% calc.); residual ZnB6O10 61.5% (63.4% calc.). Magnetic susceptibility: χm = −210 × 10−6 cm3·mol−1.
Crystal data: C4H19.91B6N3O13.5Zn, Mr = 456.46, monoclinic, C2/c (No. 15), a = 26.0212(3) Å, b = 9.15620(10) Å, c = 13.6318(2) Å, β = 99.5800(10)°, α = γ = 90°, V = 3202.55(7) Å3, T = 100(2) K, Z = 8, Z’ = 1, μ(Mo Kα) = 1.613 mm−1, 18390 reflections measured, 3651 unique (Rint = 0.0241) which were used in all calculations. The final wR2 was 0.0666 (all data) and R1 was 0.0240 (I > 2σ(I)).
3.3. Synthesis, Spectroscopic, Analytical and Crystallographic Data for 2
A solution of NH3 (35%, 2.4 mL, 36 mmol) was added dropwise to a solution of ZnSO4·H2O (1.08 g, 6 mmol) in H2O (15 mL). The addition of Ba(OH)2·8H2O (1.89 g, 6 mmol) in H2O (35 mL) followed by rapid stirring for 15 min resulted in a precipitate of BaSO4 which was removed by filtration. B(OH)3 (3.71 g, 60 mmol) dissolved in H2O (30 mL) was added to the filtrate which was further stirred at room temperature for 30 min. The volume of this solution was reduced to 5 mL by gentle evaporation on a warm water bath and the concentrated solution was left for 3 days in NMR tubes for crystallization and yielded colourless crystals of [NH4]2[Zn{B6O7(OH)6}2(H2O)2]·2H2O (2) (2.1 g, 48%). Mp ≥ 300 °C. Anal. Calc.: H = 3.8%, N = 3.8%. Found: H = 4.0%, N = 3.7%. NMR: 11B/ppm: 15.9. IR (KBr/cm−1): 3212(s), 1400(s), 1357(s), 1048(s), 953(m), 904(m), 857(m). TGA: 100–110 °C, loss of 4 interstitial/coordinated H2O 10.2% (9.9% calc.); 110–250 °C, loss of 2 NH3 15.5% (14.8% calc.); 250–500 °C, condensation of polyborate with loss of six further H2O 31.1 (29.6 calc.); residual ZnB6O19 68.9% (68.2% calc.). Magnetic susceptibility: χm = −290 × 10−6 cm3·mol−1.
Crystal data: B12H28N2O30Zn, Mr = 731.33, triclinic, P−1 (No. 2), a = 7.4831(2) Å, b = 7.8551(2) Å, c = 11.0111(3) Å, α = 108.065(2)°, β = 95.020(2)°, γ = 90.118(2)°, V = 612.68(3) Å3, T = 100(2) K, Z = 1, Z’ = 0.5, μ(Mo Kα) = 1.138 mm−1, 16475 reflections measured, 2799 unique (Rint = 0.0314) which were used in all calculations. The final wR2 was 0.0559 (all data) and R1 was 0.0212 (I > 2σ(I)).
3.4. Synthesis, Spectroscopic, Analytical and Crystallographic Data for 3
A solution of NH2CH2CH2CH2NH2 (1,3-pn) (2.52 mL, 30 mmol) in H2O (10 mL) was added to a solution of ZnSO4·H2O (1.79 g, 10 mmol) in H2O (10 mL). The reaction mixture was stirred at room temperature for 60 min before the addition of Ba(OH)2·8H2O (3.15 g, 10 mmol) in H2O (25 mL). This mixture was rapidly stirred for a further 30 min. The white precipitate of BaSO4 was removed by filtration and B(OH)3 (6.18 g, 10 mmol) dissolved in H2O (50 mL) was added to the filtrate, which was further stirred at room temperature for 30 min. The volume of this solution was reduced to 5 mL by gentle evaporation in a warm water bath. The product was collected by filtration and carefully washed with cold H2O followed by CH3COCH3, and then dried at 40 °C for 1 h to yield colourless crystals of [H3N(CH2)3NH3]3[(H3N(CH2)3NH2)ZnB12O18(OH)6]2·14H2O (3) (4.1g, 46%). Mp ≥ 300 °C. Anal. Calc.: C = 10.0%, H = 5.9%, N = 7.8%. Found: C = 9.7%, H = 5.2%, N = 7.8%. NMR. 1H/ppm: 1.93 (p, 10H, CH2), 3.01 (t, 20H, CH2) 4.8 (s, 68H, NH2, H2O, OH). 13C/ppm: 26.9, 37.6. 11B/ppm: 14.0. IR (KBr/cm−1): 3405(s), 3263(s), 1644(m), 1532(m), 1352(s), 1151(m) 1047(s), 952(m), 902(s), 855(m). TGA: 100–190 °C, loss of 14 interstitial H2O 14.1% (13.9% calc.); 190–350 °C, condensation of polyborate with loss of six further H2O 6.9 (6.0 calc.); 350–800 °C, oxidation of organics 22.8% (22.0% calc.); residual Zn2B24O38 56.6% (55.4% calc.). p-XRD: d-spacing (Å)/(% rel. int.): 9.98(36), 9.44 (100), 8.50 (54), 8.08 (35), 6.93 (43). Magnetic susceptibility: χm = −180 × 10−6 cm3·mol−1.
Crystal data: C7.5H49B12N5O31Zn, Mr = 900.60, triclinic, P−1 (No. 2), a = 9.3681(2) Å, b = 10.6910(2) Å, c = 19.2746(4) Å, α = 82.954(2)°, β = 76.156(2)°, γ = 68.655(2)°, V = 1744.44(7) Å3, T = 100(2) K, Z = 2, Z’ = 1, μ(Mo Kα) = 0.821 mm−1, 38,867 reflections measured, 7958 unique (Rint = 0.0389) which were used in all calculations. The final wR2 was 0.1053 (all data) and R1 was 0.0425 (I > 2σ(I)).
3.5. X-ray Crystallography
Single-crystal X-ray crystallography was undertaken at the Engineering and Physical Sciences Research Council (EPSRC) National Crystallography service at the University of Southampton, (Southampton, UK). Suitable crystals of 1, 2 and 3 were selected and mounted on a MITIGEN holder in perfluoroether oil on a Rigaku FRE+ equipped with HF Varimax confocal mirrors and an AFC12 goniometer and HG Saturn 724+ detector diffractometer. The crystals were kept at T = 100(2) K during data collection. Using Olex2 [48], the structures were solved with the ShelXT [49] structure solution program using the Intrinsic Phasing solution method. The models were then refined with ShelXL [50] using least squares minimisation. Cambridge Crystallographic Data Centre (CCDC) 1898912 (1), 1898913 (2), 1898914 (3) contain the supplementary crystallographic data for this paper. These data can be obtained free of charge via http://www.ccdc.cam.ac.uk/conts/retreiving.html (or from CCDC, 12 Union Road, Cambridge CB2 1EZ, UK; Fax: +44 1223 336033; email deposit@ccdc.ac.uk).
4. Conclusions
The strategy of using more highly charged cationic labile transition-metal complexes to template self-assembly (by crystallization) of polyborate anions from alkaline aqueous solutions originally containing B(OH)3 has resulted in the synthesis of three new zinc polyborate complexes in moderate yields (40–50%). These complexes contain either hexaborate(2−) or dodecaborate(6−) ligands and are stabilized by Zn–O coordinate bonds. The solid-state structures are further stabilized by multiple intramolecular and/or intermolecular H-bond interactions which are prevalent in polyborate structures.
Supplementary Materials
The following are available online at https://www.mdpi.com/2304-6740/7/4/44/s1. TGA and single-crystal XRD data. Cif and checkcif files.
Author Contributions
M.A.B. conceived the experiments; M.A.A. synthesized and characterized the complexes and grew the single crystals; P.N.H. and S.J.C. solved the crystal structures; M.A.B. wrote the paper with contributions from all co-authors.
Funding
This research received no external funding.
Acknowledgments
We thank the EPSRC for the use of the X-ray Crystallographic Service (NCS, Southampton, UK).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Schubert, D.M.; Knobler, C.B. Recent studies of polyborate anions. Phys. Chem. Glasses Eur. J. Glass Sci. Technol. B 2009, 50, 71–78. [Google Scholar]
- Grice, J.D.; Burns, P.C.; Hawthorne, F.C. Borate minerals II. A hierarchy of structures based upon the borate fundamental building block. Can. Mineral. 1999, 37, 731–762. [Google Scholar]
- Becker, P. A contribution to borate crystal chemistry: Rules for the occurrence of polyborate anion types. Z. Kristallogr. 2001, 216, 523–533. [Google Scholar] [CrossRef]
- Heller, G. A survey of structural types of borates and polyborates. Top. Curr. Chem. 1986, 131, 39–98. [Google Scholar]
- Belokoneva, E.L. Borate crystal chemistry in terms of the extended OD theory: Topology and symmetry analysis. Crystallogr. Rev. 2005, 11, 151–198. [Google Scholar] [CrossRef]
- Christ, C.L.; Clark, J.R. A crystal-chemical classification of borate structures with emphasis on hydrated borates. Phys. Chem. Miner. 1977, 2, 59–87. [Google Scholar] [CrossRef]
- Beckett, M.A. Recent Advances in crystalline hydrated borates with non-metal or transition-metal complex cations. Coord. Chem. Rev. 2016, 323, 2–14. [Google Scholar] [CrossRef]
- Wiebcke, M.; Freyhardt, C.C.; Felsche, J.; Engelhardt, G. Clathrates with three-dimensional host structures of hydrogen bonded pentaborate [B5O6(OH)4]− ions: Pentaborates with the cations NMe4+, NEt4+, NPhMe3+ and pipH+ (pipH+ = piperidinium). Z. Naturforsch. 1993, 48b, 978–985. [Google Scholar] [CrossRef]
- Visi, M.Z.; Knobler, C.B.; Owen, J.J.; Khan, M.I.; Schubert, D.M. Structures of self-assembled nonmetal borates derived from α,ω-diaminoalkanes. Cryst. Growth Des. 2006, 6, 538–545. [Google Scholar] [CrossRef]
- Beckett, M.A.; Coles, S.J.; Davies, R.A.; Horton, P.N.; Jones, C.L. Pentaborate(1−) salts templated by substituted pyrrolidinium cations: Synthesis, structural characterization, and modelling of solid-state H-bond interactions by DFT calculations. Dalton Trans. 2015, 44, 7032–7040. [Google Scholar] [CrossRef] [PubMed]
- Beckett, M.A.; Bland, C.C.; Horton, P.N.; Hursthouse, M.B.; Varma, K.S. Supramolecular structures containing “isolated” pentaborate anions and non-metal cations: Crystal structures of [Me3NCH2CH2OH][B5O6(OH)4] and [4-Mepy,4-MepyH][B5O6(OH)4]. J. Organomet. Chem. 2007, 692, 2832–2838. [Google Scholar] [CrossRef]
- Altahan, M.A.; Beckett, M.A.; Coles, C.J.; Horton, P.N. A new polyborate anion [B7O9(OH)6]3−: Self-assembly, XRD and thermal properties of s-fac-[Co(en)3][B7O9(OH)6]·9H2O. Inorg. Chem. Commun. 2015, 59, 95–98. [Google Scholar] [CrossRef]
- Altahan, M.A.; Beckett, M.A.; Coles, C.J.; Horton, P.N. A new decaoxidooctaborate(2−) anion, [B8O10(OH)6]2−: Synthesis and characterization of [Co(en)3][B5O6(OH)4][B8O10(OH)6]·5H2O (en = 1,2-diaminoethane). Inorg. Chem. 2015, 54, 412–414. [Google Scholar] [CrossRef]
- Altahan, M.A.; Beckett, M.A.; Coles, C.J.; Horton, P.N. Transition-metal complexes with oxidoborates. Synthesis and XRD characterization of [H3NCH2CH2NH2)Zn{κ3O,O’,O’’-B12O18(OH)6-κ1O’’’} Zn(en)(NH2CH2CH2NH3)]·8H2O: A neutral bimetallic zwiterionic polyborate system containing the “isolated” dodecaborate(6−) anion. Pure Appl. Chem. 2018, 90, 625–632. [Google Scholar] [CrossRef]
- Altahan, M.A.; Beckett, M.A.; Coles, C.J.; Horton, P.N. Two 1-D Coordination Polymers containing Zinc(II) Hexaborates: [Zn(en){B6O7(OH)6}]·2H2O (en = 1,2-diaminoethane) and [Zn(pn){B6O7(OH)6}]·1.5H2O (pn = (+/−) 1,2-diaminopropane). Crystals 2018, 8, 470. [Google Scholar] [CrossRef]
- Wang, G.-M.; Sun, Y.-Q.; Yang, G.-Y. Synthesis and crystal structures of two new pentaborates. J. Solid State Chem. 2005, 178, 729–735. [Google Scholar] [CrossRef]
- He, Y.; Yang, J.; Xi, C.-Y.; Chen, J.-S. Solvothermal synthesis and crystal structure of Zn(en)3B5O7(OH)3. Chem. Res. Chin. Univ. 2006, 22, 271–273. [Google Scholar] [CrossRef]
- Jiang, H.; Yang, B.-F.; Wang, G.-M. [Zn(dap)3][Zn(dap)B5O8(OH)2]2: A novel organic-inorganic hybrid chain-like zincoborate made up of [B5O8(OH)2]3− and [Zn(dap)]2+ linkers. J. Clust. Sci. 2017, 28, 1421–1429. [Google Scholar] [CrossRef]
- Wei, L.; Sun, A.-H.; Xue, Z.-Z.; Pan, J.; Wang, G.-M.; Wang, Y.-X.; Wang, Z.-H. Hydrothermal synthesis and structural characterization of a new hybrid zinc borate, [Zn(dap)2][B4O6(OH)2]. J. Clust. Sci. 2017, 28, 1453–1462. [Google Scholar] [CrossRef]
- Zhao, P.; Cheng, L.; Yang, G.Y. Synthesis and characterization of a new organic-inorganic hybrid borate [Zn(dab)0.5(dab’)0.5(B4O6(OH)2]·H2O. Inorg. Chem. Commun. 2012, 20, 138–141. [Google Scholar] [CrossRef]
- Paul, A.V.; Sachidananda, K.; Natarajan, S. [B4O9H2] cyclic borate units as the building unit in a family of zinc borate structures. Cryst. Growth Des. 2010, 10, 456–464. [Google Scholar] [CrossRef]
- Pan, R.; Chen, C.-A.; Yang, B.-F. Two new octaborates constructed of two different sub-clusters and supported by metal complexes. J. Clust. Sci. 2017, 28, 1237–1248. [Google Scholar] [CrossRef]
- Zhao, P.; Lin, Z.-E.; Wei, Q.; Cheng, L.; Yang, G.-Y. A pillared-layered zincoborate with an anionic network containing unprecedented zinc oxide chains. Chem. Commun. 2014, 50, 3592–3594. [Google Scholar] [CrossRef]
- Schubert, D.M.; Alam, F.; Visi, M.Z.; Knobler, C.B. Structural characterization and chemistry of an industrially important zinc borate, Zn[B3O4(OH)3]. Chem Mater. 2003, 15, 866–871. [Google Scholar] [CrossRef]
- Sola, J.; Lafuente, M.; Atcher, J.; Alfonso, I. Constitutional self-selection from dynamic combinatorial libraries in aqueous solution through supramolecular interactions. Chem. Commun. 2014, 50, 4564–4566. [Google Scholar] [CrossRef] [PubMed]
- Corbett, P.T.; Leclaire, J.; Vial, L.; West, K.R.; Wietor, J.-L.; Sanders, J.K.M.; Otto, S. Dynamic combinatorial chemistry. Chem. Rev. 2006, 106, 3652–3711. [Google Scholar] [CrossRef] [PubMed]
- Salentine, G. High-field 11B NMR of alkali borate. Aqueous polyborate equilibria. Inorg. Chem. 1983, 22, 3920–3924. [Google Scholar] [CrossRef]
- Anderson, J.L.; Eyring, E.M.; Whittaker, M.P. Temperature jump rate studies of polyborate formation in aqueous boric acid. J. Phys. Chem. 1964, 68, 1128–1132. [Google Scholar] [CrossRef]
- Taube, H. Rates and mechanisms of substitutions in inorganic complexes in aqueous solution. Chem. Rev. 1952, 50, 69–126. [Google Scholar] [CrossRef]
- Dunitz, J.D.; Gavezzotti, A. Supramolecular synthons: Validation and ranking of intermolecular interaction energies. Cryst. Growth Des. 2012, 12, 5873–5877. [Google Scholar] [CrossRef]
- Desiraju, G.R. Supramolecular synthons in crystal engineering—A new organic synthesis. Angew. Chem. Int. Ed. Engl. 1995, 34, 2311–2327. [Google Scholar] [CrossRef]
- Altahan, M.A.; Beckett, M.A.; Coles, S.J.; Horton, P.N. Synthesis and characterization of polyborates template by cationic copper(II) complexes: Structural (XRD), spectroscopic, thermal (TGA/DSC) and magnetic properties. Polyhedron 2017, 135, 247–257. [Google Scholar] [CrossRef]
- Wang, G.-M.; Sun, Y.-Q.; Yang, G.-Y. Synthesis and crystal structures of three new borates templated by transition-metal complexes in situ. J. Solid State Chem. 2006, 179, 1545–1553. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, Y.; Zhu, J.; Liu, R.-B.; Xu, J.; Meng, C.-C. A new mixed ligand copper pentaborate with square-like, rectangular-like and ellipse-like channels formed via hydrogen bonds. Inorg. Chim. Acta 2011, 376, 401–407. [Google Scholar] [CrossRef]
- Liu, Z.-H.; Zhang, J.-J.; Zhang, W.-J. Synthesis, crystal structure and vibrational spectroscopy of a novel mixed ligands Ni(II) pentaborate [Ni(C4H10N2)(C2H8N2)2][B5O6(OH)4]2. Inorg. Chim. Acta 2006, 359, 519–524. [Google Scholar] [CrossRef]
- Li, J.; Xia, S.; Gao, S. FT-IR and Raman spectroscopic study of hydrated borates. Spectrochim. Acta 1995, 51A, 519–532. [Google Scholar] [CrossRef]
- Archibald, S.J. Zinc. In Comprehensive Coordination Chemistry II, 2nd ed.; McCleverty, J.A., Meyer, T.J., Eds.; Elsevier: Amsterdam, The Netherlands, 2003; Volume 6, pp. 1147–1251. [Google Scholar]
- Natarajan, S.; Klein, W.; Panthoefer, M.; Wuellen, L.V.; Jansen, M. Solution mediated synthesis and structure of the first anionic bis(hexaborato)-zincate prepared in the presence of organic base. Z. Anorg. Allg. Chem. 2003, 629, 959–962. [Google Scholar] [CrossRef]
- Jemai, N.; Rzaigui, S.; Akriche, S. Piperazine-1,4-diium bis(hexahydroxidoheptaoxidohexaborato-κ3O,O’,O’’) cobaltate(II) hexahydrate. Acta Cryst. 2014, E70, m167–m169. [Google Scholar] [CrossRef] [PubMed]
- Beckett, M.A.; Hibbs, D.E.; Hursthouse, M.B.; Malik, K.M.A.; Owen, P.; Varma, K.S. cyclo-Boratrisiloxane and cyclo-diboratetrasiloxane derivatives and their reactions with amines: Crystal and molecular structure of (p-BrC6H4BO)2(Ph2SiO)2. J. Organomet. Chem. 2000, 595, 241–247. [Google Scholar] [CrossRef]
- Etter, M.C. Encoding and decoding hydrogen-bond patterns of organic chemistry. Acc. Chem. Res. 1990, 23, 120–126. [Google Scholar] [CrossRef]
- Skakibaie-Moghadam, M.; Heller, G.; Timper, U. Die kristallstruktur von Ag6[B12O18(OH)6].3H2O einen neuen dokekaborat. Z. Kristallogr. 1990, 190, 85. [Google Scholar] [CrossRef]
- Menchetti, M.; Sabelli, C. A new borate polyanion in the structure of Na8[B12O20(OH)4]. Acta Cryst. 1979, B35, 2488–2493. [Google Scholar] [CrossRef]
- Choudhury, A.; Neeraj, S.; Natarajan, S.; Rao, C.N.R. An open-framework zincoborate formed by Zn6B12O24 clusters. J. Chem. Soc. Dalton Trans. 2002, 1535–1538. [Google Scholar] [CrossRef]
- Zhang, T.-J.; Pan, R.; He, H.; Yang, B.-F.; Yang, G.-Y. Solvothermal synthesis and structure of two new boranes containing [B7O9(OH)5]2− and [B12O18(OH)6]6− clusters. J. Clust. Sci. 2016, 27, 625. [Google Scholar] [CrossRef]
- Zhang, H.-X.; Zhang, J.; Zheng, S.T.; Yang, G.-Y. K7{(BO3)Mn[B12O18(OH)6]}·H2O: First manganese borate based on covalently linked B12O18(OH)6 clusters and BO3 units via Mn2+ cations. Inorg. Chem. Commun. 2004, 7, 781–783. [Google Scholar] [CrossRef]
- Rong, C.; Jiang, J.; Li, Q.-L. Synthesis and transitional metal borates K7{(BO3)Zn[B12O18(OH)6]}·H2O and quantum chemistry study. Chinese J. Inorg. Chem. 2012, 28, 2217–2222. [Google Scholar]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. Olex2: A complete structure solution, refinement and analysis program. J. Appl. Cryst. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Sheldrick, G.M. ShelXT-intergrated space-group and crystal structure determination. Acta Cryst. 2015, A71, 3–8. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with ShelXL. Acta Cryst. 2015, C27, 3–8. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).