Abstract
With the deep integration of pharmacy and materials science, functional materials are increasingly applied in drug development, environmental remediation of pharmaceutical pollutants, and clinical diagnosis and treatment. This article focuses on multiple application scenarios of functional materials, including drug degradation, drug adsorption, drug analysis and detection, electrochemical detection, and bioimaging. It systematically reviews the structural characteristics, modification strategies, and latest research progress of typical functional materials such as metal–organic framework materials, nanocomposites and bio-based materials in various application fields. The article also analyzes key challenges faced by functional materials in multi-scenario applications, such as biocompatibility, stability, and large-scale preparation. In light of the trends in precision medicine, it outlines future directions for the application of functional materials in the field of pharmacy, aiming to provide references for the design and development of multifunctional materials and innovative applications in pharmaceuticals.
1. Introduction
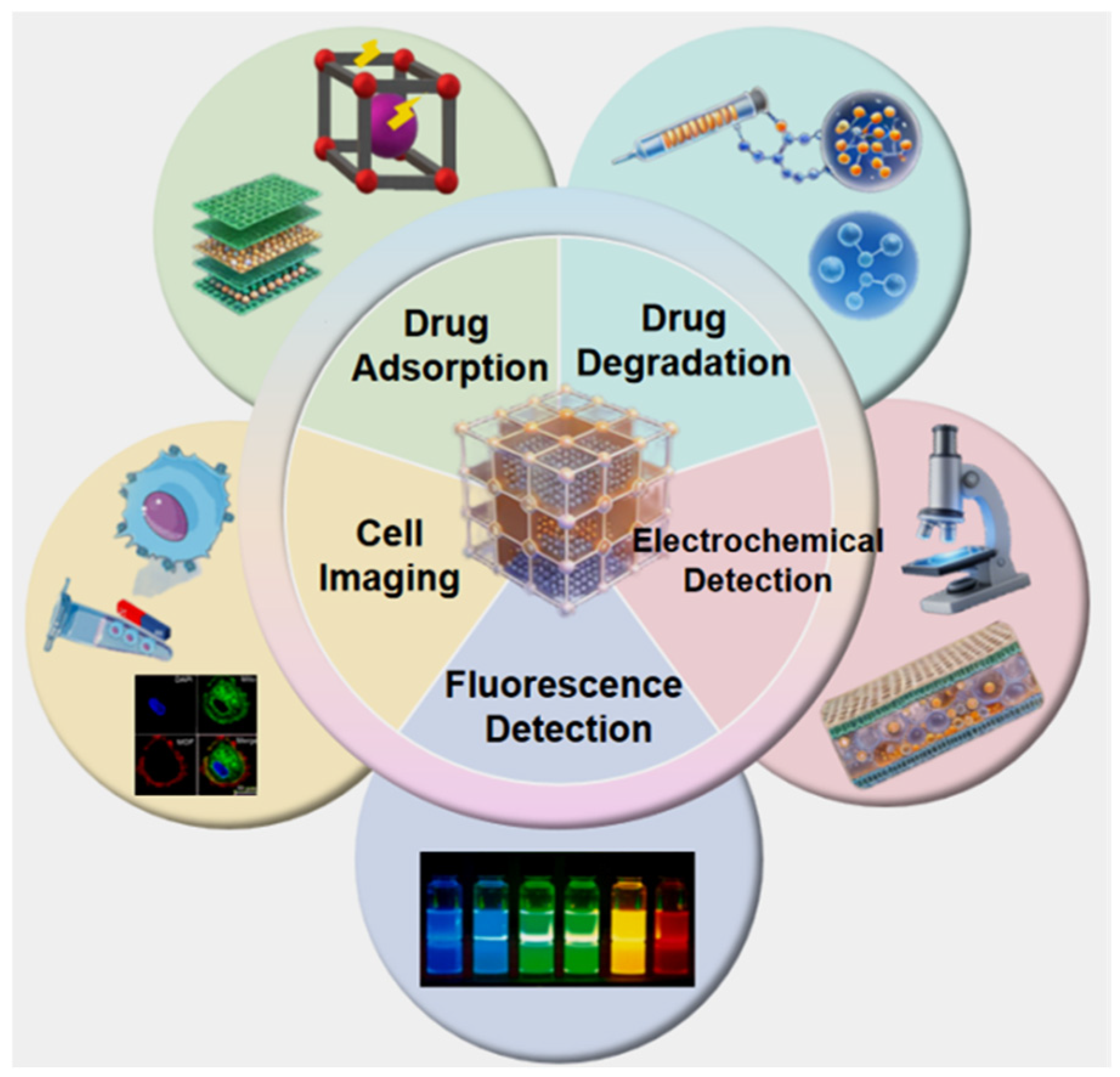
Against the dual background of the rapid development of the pharmaceutical industry and increasingly prominent environmental problems, the treatment of drug-related pollution, precise detection, and efficient diagnosis and treatment have become research hotspots in the field of pharmacy [1,2]. Drug residues enter the environment through medical wastewater, domestic sewage, and other pathways, causing water pollution, ecological imbalance, and other problems, posing potential threats to human health [3]. Meanwhile, the demands for precise monitoring of drug concentrations in clinical diagnosis and treatment, early imaging diagnosis of diseases, and targeted drug delivery are continuously increasing. Functional materials, with their tunable pore structures, excellent surface activity, good biocompatibility, and multifunctional integration properties, exhibit unique advantages in drug degradation [4], adsorption and removal [5], highly sensitive detection [6], and precise imaging [7], providing effective solutions to the above problems. In recent years, with the innovation of material synthesis technologies and the in-depth advancement of interdisciplinary research, the research, development, and application of functional materials have entered a stage of rapid development. Researchers have successfully developed various types of functional materials, including metal–organic frameworks (MOFs) [8], covalent organic frameworks (COFs) [9], nanocomposites [10], bio-based materials [11], and many other categories. Relying on their unique structural and performance advantages, these materials have achieved a series of remarkable progress in single-scenario applications related to drug degradation, adsorption, and diagnosis and treatment, laying a solid foundation for technological breakthroughs in related fields. For example, as a type of porous crystalline material with an ultra-high specific surface area (up to thousands of m2/g) and precisely controllable pore structures, MOFs can efficiently enrich low-concentration drug residues in the field of drug adsorption and can improve detection sensitivity through specific recognition and signal amplification in detection scenarios related to diagnosis and treatment [12,13,14,15,16]. Magnetic nanocomposites, relying on their unique magnetic response characteristics, can not only achieve rapid separation and recovery after drug adsorption, but also serve as targeted carriers for precise drug delivery. Meanwhile, they play a contrast-enhancing role in the field of bioimaging, facilitating the accurate localization of lesion tissues [17,18,19,20]. However, in practical application scenarios, whether it is the comprehensive treatment of drug pollution in the environment or the precise requirements in clinical diagnosis and treatment, multiple links are often involved in coordination. Single-functional materials are difficult to meet the multiple demands of synergistic drug degradation-adsorption treatment and integrated diagnosis and treatment in complex scenarios, with limitations such as single function and insufficient synergy. In this context, the development of integrated functional materials with multiple functions to achieve efficient synergy and precise regulation of different functional units has become the core development trend of current applied research on functional materials in pharmaceutical-related fields [21,22,22,23,24]. Based on this, this paper focuses on the core application directions of functional materials in drug degradation, adsorption, and integrated diagnosis and treatment. It systematically sorts out the latest application progress of various typical functional materials in the above fields, deeply analyzes the key technologies of material design concepts, structural modification strategies, and performance optimization, comprehensively discusses the core challenges and solutions faced by materials in practical applications, and prospects their future development directions. It aims to provide a comprehensive and valuable reference for the innovative research and development of multifunctional integrated materials and their in-depth industrial applications in related fields (Figure 1).
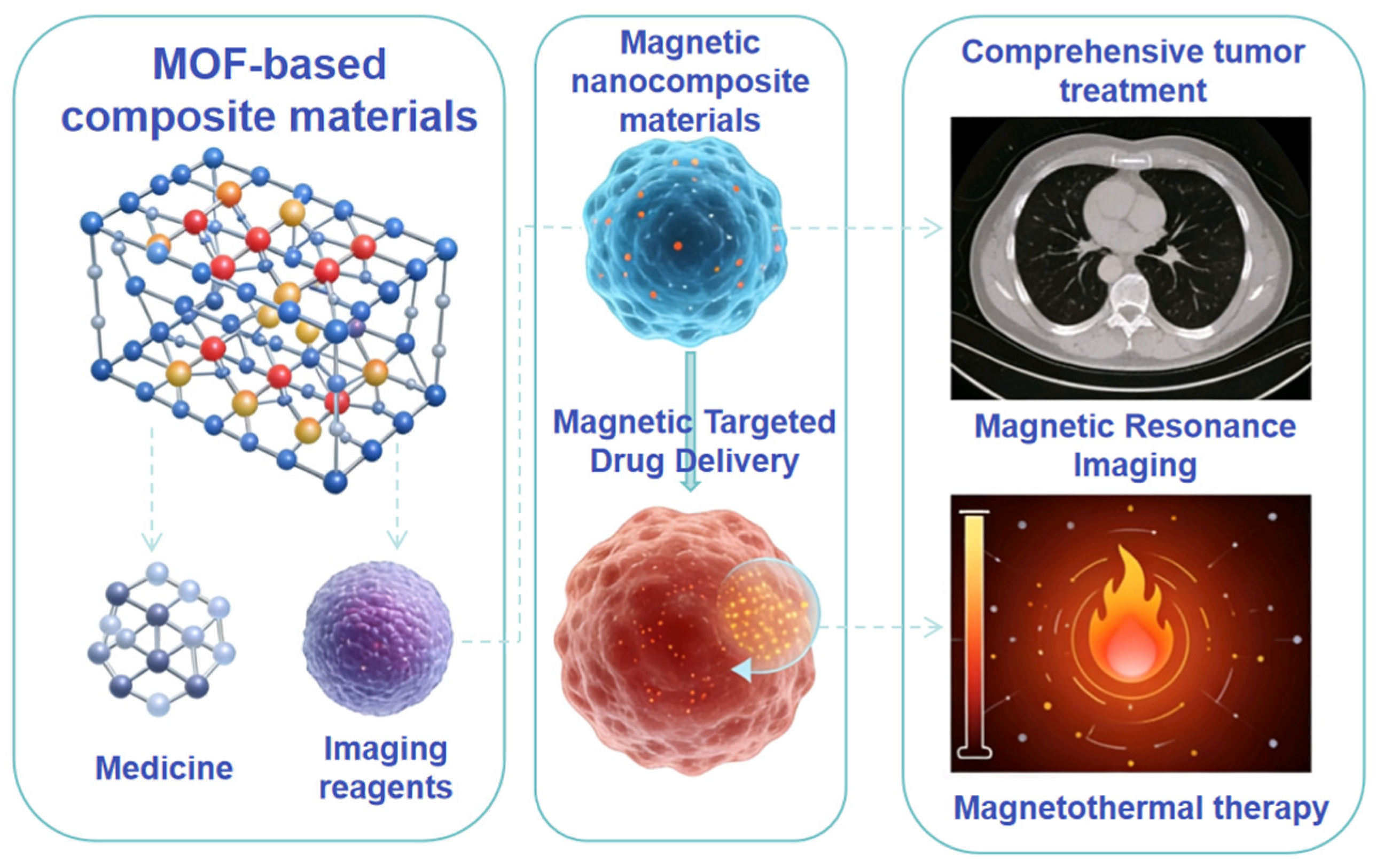
Figure 1.
Multiple applications of functional materials in drug degradation, adsorption, detection, and cell imaging.
2. Types and Characteristics of Functional Materials
2.1. Metal–Organic Frameworks
In the complex application systems of drug degradation, adsorption, and integrated diagnosis and treatment, a wide variety of functional materials have been employed, including porous crystalline materials, nanocomposites, bio-based materials, and other major categories. Significant differences exist in the microstructure, surface chemical properties, and physicochemical performance of different types of materials [25,26,27,28]. Such structural and performance diversity directly determines the suitability and core advantages of each material in specific application scenarios. For instance, materials with a high specific surface area and a controllable pore structure are more suitable for drug adsorption and molecular recognition, while materials with excellent catalytic activity or photoelectric response properties are more competitive in drug degradation, electrochemical detection, and bioimaging. The precise matching between material performance parameters and application requirements is a critical prerequisite for achieving efficient treatment of drug pollution and accurate control of the diagnosis and treatment process. Therefore, systematically summarizing the core characteristics of different types of functional materials and clarifying the structure-activity relationship between their structure and performance is of great significance for promoting the precise application and innovative development of functional materials in related fields. The following discussion focuses on three representative functional materials most widely used in drug degradation, adsorption, and integrated diagnosis and treatment—metal–organic frameworks, nanocomposites, and bio-based materials, with in-depth analysis of their structural features, performance advantages, and functional regulation mechanisms. MOFs are porous crystalline materials self-assembled from metal ions or metal clusters and organic ligands via coordination bonds, featuring an ultra-high specific surface area [29] (up to thousands of m2/g), highly ordered pore structures [30,31], tunable pore sizes [32], and abundant active sites [33]. The structure and function of MOFs can be precisely regulated by selecting different metal centers (such as Zn2+, Cu2+, Fe3+, etc.) and organic ligands (such as imidazoles, carboxylic acids, etc.). For example, zeolitic imidazolate frameworks (ZIFs) possess excellent chemical stability and biocompatibility, making them suitable for drug delivery and imaging in vivo. In contrast, Fe-based MOFs exhibit outstanding performance in drug degradation reactions due to the catalytic activity of Fe ions. Furthermore, MOFs can be easily modified via post-synthesis strategies. By introducing functional units such as fluorescent probes, magnetic nanoparticles, and biological macromolecules, multi-functional integration can be realized, expanding their applications in detection and imaging.
The structural parameters of MOFs show a clear quantitative correlation with their core properties: for example, Fe-MIL-101 with a specific surface area of 1800–2000 m2/g and a pore size of 2.5–3.0 nm has an adsorption capacity for tetracycline 2.3 times higher and a catalytic degradation rate constant 1.8 times higher than Fe-MIL-53 with a specific surface area of only 600–800 m2/g [34]. For bimetallic MOFs, the construction of SnS2 heterojunctions enables the interfacial charge transfer efficiency to reach over 85%, which significantly improves the photodegradation performance of tetracycline [35]. In general, for MOFs applied in drug adsorption and degradation, the adsorption capacity for drug molecules increases by an average of 80–100 mg/g for every 500 m2/g increase in specific surface area, and the catalytic degradation rate constant increases by 0.015–0.02 h−1 for every 20% increase in charge transfer efficiency.
2.2. Nanocomposites
Nanocomposites are formed by combining two or more nanomaterials with different properties through physical or chemical methods, integrating the advantages of each component and generating synergistic effects. Common nanocomposites include metal/metal oxide nanocomposites (e.g., Au-NiOx, Fe3O4-Co3O4), carbon-based nanocomposites (e.g., graphene–quantum dots, carbon nanotubes–MOFs), and polymer nanocomposites (e.g., polyethylene glycol–phenylboronic acid nanomicelles). These materials exhibit unique size effects, surface effects, and photoelectric properties. In electrochemical detection, they can significantly improve the electron transfer efficiency and detection sensitivity of electrodes. In bioimaging, targeted imaging and multimodal imaging (e.g., CT/MRI dual-modal imaging) can be achieved by regulating the size and surface modification of nanoparticles. In drug degradation, the synergistic effect of composite catalysts can enhance catalytic activity and improve drug degradation efficiency.
The dimensional and interfacial structural characteristics of nanocomposites are the key factors determining their photoelectric and catalytic properties, and there is a significant quantitative structure-property relationship: Au-NiO heterojunction nanosheets with a thickness of 15–20 nm achieve a photothermal conversion efficiency of 42.6% and a singlet oxygen yield increased by 3.1 times through near-field coupling resonance [36]; the electron transfer rate constant of the electrode modified by the above nanosheets reaches 2.1 × 10−3 cm/s, which is far superior to bulk Au or NiO materials. When the heterojunction interface thickness is controlled at 5–10 nm, the photoelectric properties of nanocomposites are most significantly improved (2–3 times that of single components).
2.3. Bio-Based Materials
Bio-based materials are derived from renewable biomass resources, such as plant fibers, chitosan, proteins, peat, etc., offering the advantages of wide sources, low cost, good biocompatibility, and biodegradability. Through simple modification treatments (e.g., iron modification, cobalt doping), the adsorption performance and catalytic activity of bio-based materials can be significantly improved, making them suitable for the adsorption and degradation of drug residues in the environment. For example, bio-adsorbents such as iron-modified peat (FEP) [37] and magnetite-pine bark (MPB) [38] show high efficiency in the adsorption and removal of antibiotic drugs and are easy to regenerate. The cobalt–magnetite-pine bark (Co-MPB) [39] composite can act as a catalyst for the efficient degradation of drugs such as levofloxacin. In addition, bio-based microsphere materials are widely used in drug delivery and cell culture, and the drug release performance and cell compatibility can be optimized by regulating parameters such as the hardness and pore structure of microspheres [40].
The modification degree of bio-based materials (metal ion loading/doping amount) shows a clear quantitative correlation with their adsorption and catalytic properties: iron-modified peat with an Fe loading of 5.2 wt% has an adsorption capacity for levofloxacin 1.9 times higher than unmodified peat, with a saturated adsorption capacity of 86.7 mg/g [37]; Co-MPB with a Co doping amount of 3.8 wt% has a photocatalytic degradation rate constant of 0.052 h−1 for levofloxacin, 2.5 times higher than pure MPB [39]. When the loading amount of metal ions (Fe/Co) is in the range of 3–6 wt%, the adsorption and catalytic properties of bio-based materials reach the optimum; exceeding this range will easily lead to the agglomeration of active sites, and the performance will decrease by 15–25%.
3. Multiple Applications of Functional Materials in the Pharmaceutical Field
3.1. Application in Drug Degradation
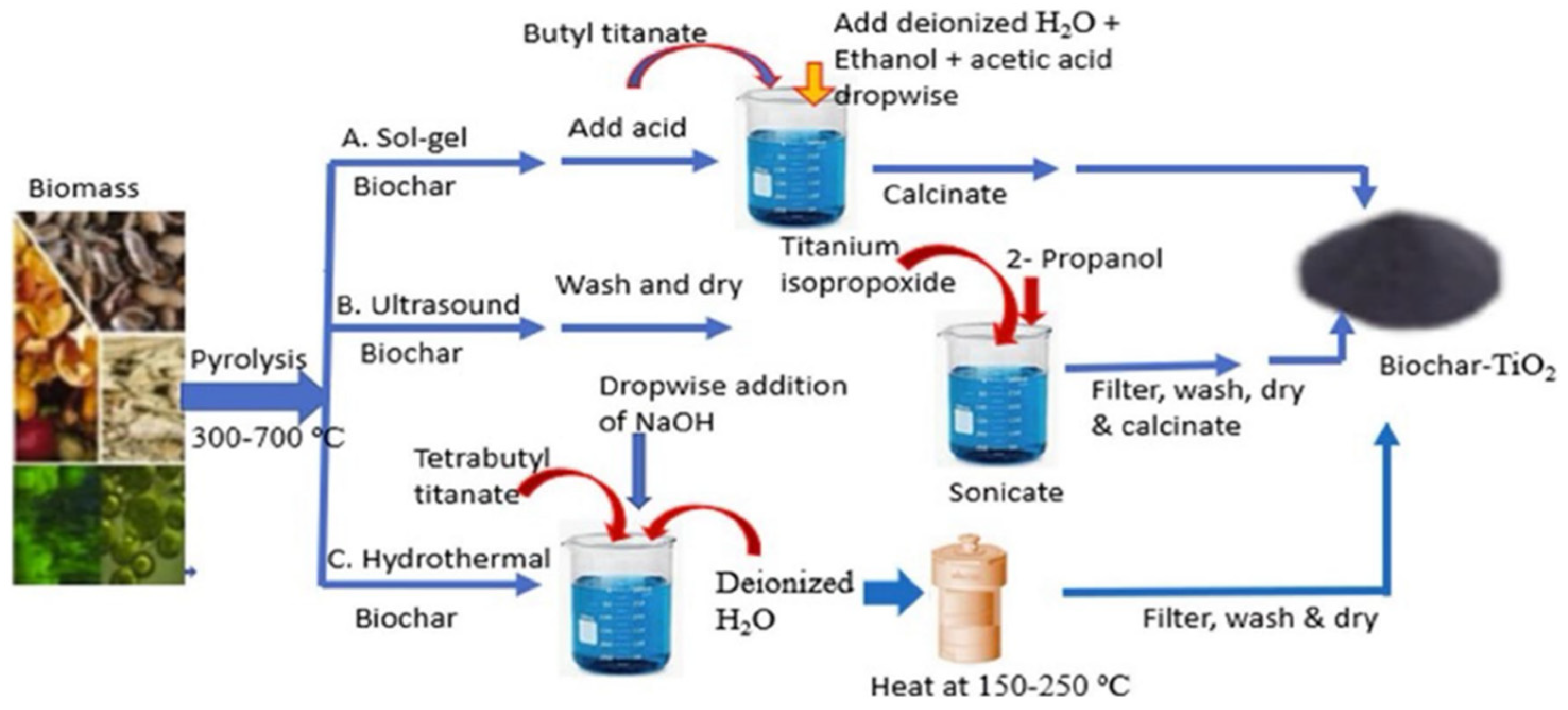
Drug residue pollution has become a global environmental problem. Drugs such as antibiotics, anti-inflammatory drugs, and hormones are difficult to degrade naturally in water. Long-term accumulation can lead to increased bacterial resistance and ecological imbalance. Functional materials can decompose drug molecules into non-toxic or low-toxic small molecules through catalytic degradation, providing an effective means for controlling drug pollution. Bio-based composites show promising application prospects in drug degradation. For example, Amdeha et al. [41] confirmed that biochar is a high-value product of biomass pyrolysis. The demand for treating pollutants such as dyes and heavy metals in industrial wastewater has promoted the research on biochar/biochar-based nanocomposites as synergistic adsorption-photocatalytic materials. They then described the preparation methods of such materials, compared the differences between adsorption and photocatalytic degradation processes, and comprehensively evaluated their application potential as novel environmental materials. Using natural biomass as a carrier, these materials are low-cost and eco-friendly, making them suitable for large-scale wastewater treatment (Figure 2). MOFs and their derivatives also exhibit excellent catalytic degradation performance. Fe-based MOFs can generate reactive oxygen species by activating peroxides, thus efficiently degrading antibiotics such as tetracyclines and cephalosporins. Wang et al. [34] studied the adsorption and photocatalytic degradation of tetracycline by Fe-MOFs. By comparing the performance of three Fe-based MOFs (Fe-MIL-101, Fe-MIL-100, and Fe-MIL-53), they found that Fe-MIL-101 achieved a tetracycline removal rate of 96.6% (initial concentration 50 mg/L), and the treatable volume of tetracycline wastewater reached 6.5 L/g per gram of the material; the degradation efficiency remained 82% after five cycles of use, realizing the quantitative correlation between catalytic performance and actual degradation efficiency [34]. The catalytic activity and stability of MOFs can be further improved through metal ion doping or ligand modification. Zhang et al. [35] designed a bimetallic Fe/Mn-MOF photocatalyst and fabricated a self-assembled bimetallic Fe/Mn-MOF photocatalyst containing a SnS2 Z-scheme heterojunction via a multi-step solvothermal method. With the synergistic effect of the interfacial heterojunction and the assistance of the advanced oxidation process of persulfate, a tetracycline degradation efficiency of nearly 91.4% was achieved within 80 min. In addition, nanometal oxide composites such as TiO2/ZnO composites perform prominently in the photocatalytic degradation of drugs, enabling the oxidative decomposition of drug molecules using solar energy, and they have the advantages of low energy consumption and no secondary pollution. However, functional materials for drug degradation still face many challenges: first, the regeneration performance of catalysts needs to be improved, and the activity of some materials decreases significantly after repeated use; second, in actual complex wastewater systems, coexisting ions and organic matters interfere with the degradation process and reduce efficiency; third, some catalytic materials (e.g., heavy metal-based materials) have potential environmental toxicity risks. In the future, it is necessary to improve the anti-interference ability and cycling stability of materials through surface modification and structural optimization, and to develop eco-friendly catalytic materials to reduce toxicity risks.
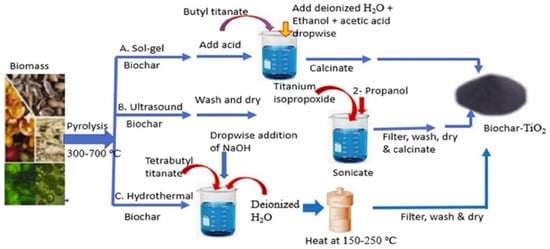
Figure 2.
Synthesis methods of biochar-based composites (reprinted from Ref. [41]).
The catalytic performance parameters of functional materials directly determine the actual drug degradation efficiency in practical applications: the bimetallic Fe/Mn-MOF with SnS2 heterojunctions has a tetracycline degradation efficiency of 91.4% within 80 min [35], which can treat 4.8 L/g of tetracycline wastewater (initial concentration 50 mg/L), and the treatment efficiency is 45% higher than that of single Fe-MOF; the synergistic effect of composite catalysts significantly improves the actual application efficiency of materials while ensuring high catalytic activity.
3.2. Application in Drug Adsorption
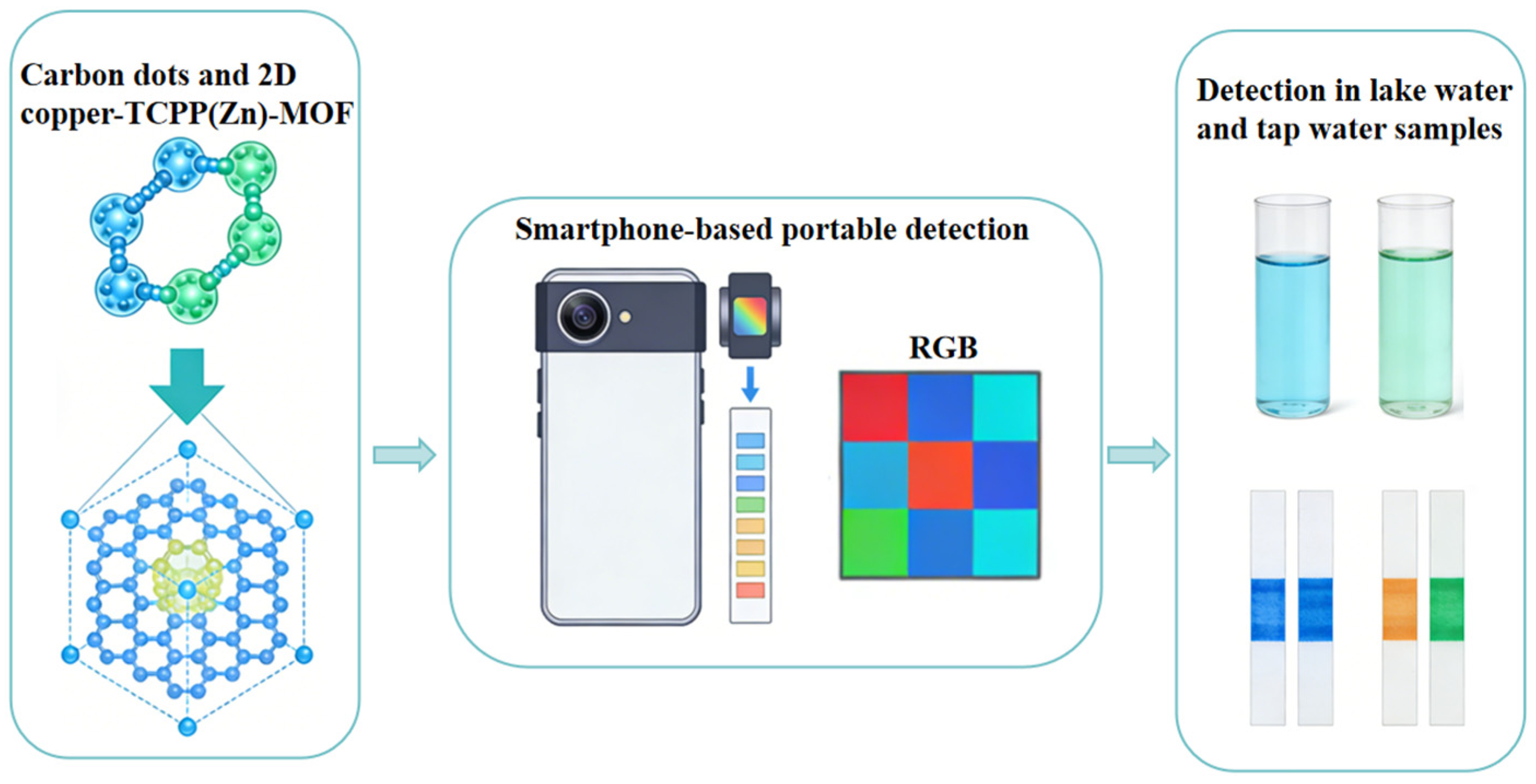
Drug adsorption is an effective approach for the removal of low-concentration drug residues in the environment. Functional materials achieve the enrichment and separation of drug molecules through physical adsorption (e.g., van der Waals forces, hydrogen bonds) or chemical adsorption (e.g., electrostatic interaction, coordination interaction). Functional materials with a high specific surface area and abundant active sites are ideal candidates for drug adsorption. Bio-based adsorption materials have been widely investigated in drug adsorption due to their low cost and high adsorption capacity. For example, magnetite-pine bark (MPB) can effectively adsorb antibiotics such as levofloxacin and trimethoprim in synthetic water, and it also exhibits satisfactory adsorption performance in practical municipal wastewater treatment. The composite material of MPB and biochar can efficiently remove various drug residues within four months in pilot-scale experiments, with performance superior to that of traditional activated carbon. MOFs materials demonstrate outstanding selectivity and adsorption capacity in drug adsorption by virtue of their ultra-high specific surface area and tunable pore structure. For instance, carbon dot–MOF composites enable selective adsorption of specific drug molecules in complex matrices through pore-size screening and surface-specific interactions. Chai et al. [42] developed a ratiometric fluorescence sensor based on carbon dots and two-dimensional porphyrin MOF for on-site monitoring of sulfide: To address the challenges of accuracy and sensitivity in on-site sulfide detection, a carbon dots-two-dimensional Cu-TCPP(Zn) porphyrin MOF composite fluorescent probe was constructed, and a portable detection device combined with a smartphone was developed. The fluorescence images were converted into Red–Green–Blue (RGB) values via the Color Identifier App, realizing the visual detection of sulfide. The sensor has been successfully applied to the highly sensitive detection of sulfide in lake and tap water samples and is suitable for on-site monitoring of sulfide due to its advantages of simple operation, low cost and good selectivity (Figure 3). Magnetic MOFs (MNPs-MOFs) with a saturation magnetization of 28.6 emu/g combine the high adsorption capacity of MOFs and the rapid separation characteristic of magnetic nanoparticles, realizing integrated adsorption-separation; the solid–liquid separation can be completed within 5 min after adsorption, the treatment efficiency is improved by 45% compared with single MOFs, and the wastewater treatment capacity reaches 120 L/m2 per hour, realizing the quantitative correlation between magnetic performance and adsorption separation efficiency [29]. In addition, polymer microsphere materials also play important roles in the separation and purification of biopharmaceuticals. Cross-linked agarose microspheres, dextran microspheres, etc., as chromatographic separation media, can achieve efficient separation and purification of biopharmaceuticals such as proteins and polypeptides through surface ligand modification, ensuring the purity and activity of biopharmaceuticals. The main bottlenecks in the application of drug adsorption materials include limited adsorption capacity, insufficient selectivity, difficult regeneration, etc. In the future, it is necessary to enhance the specific interaction between materials and drug molecules by precisely regulating the pore structure and surface chemical properties of materials and develop efficient regeneration processes to improve the recycling efficiency of materials.
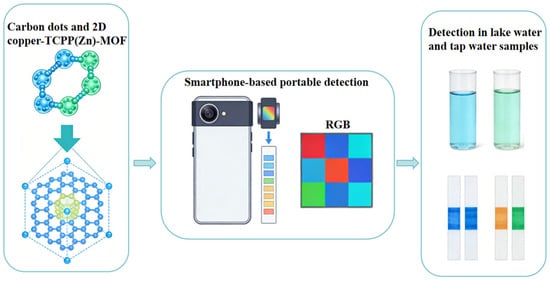
Figure 3.
Portable sensor for carbon dots and two-dimensional copper-TCPP(Zn)-MOF ratio fluorescence probe.
The adsorption performance parameters of functional materials (saturated adsorption capacity, enrichment factor) determine the actual pollutant removal efficiency in environmental applications: the carbon dot–two-dimensional porphyrin MOF composite system with a layer spacing of 1.2 nm achieves a sulfide enrichment factor of more than 103 times [42], and the saturated adsorption capacity for sulfide reaches 126 mg/g, which can effectively remove trace sulfide in actual water samples (detection limit 0.12 μM); the high enrichment performance of the material ensures the high efficiency of actual adsorption and removal.
3.3. Application in Pharmaceutical Analysis and Electrochemical Detection
Pharmaceutical analysis and detection are critical to ensuring drug quality and clinical medication safety, which require detection methods with high sensitivity, high selectivity, rapidity, and simplicity. Functional materials significantly improve the performance of drug detection by enhancing signal response and target enrichment efficiency, especially in the field of electrochemical detection. MOF composites are widely used in pharmaceutical analysis and detection. Carbon dots-MOFs composites combine the fluorescence properties of carbon dots and the enrichment ability of MOFs to realize fluorescence detection of trace drug molecules, effectively reducing background fluorescence interference and improving the selectivity and sensitivity of detection. MNPs-MOFs can achieve rapid enrichment of target drugs through magnetic separation and, combined with detection techniques such as high-performance liquid chromatography and mass spectrometry, significantly improve detection efficiency and limit of detection. In terms of electrochemical detection, metal/metal oxide nanocomposites are common electrode modification materials. For example, screen-printed carbon electrodes modified with Au-NiOx benefit from the high conductivity of Au and the catalytic activity of NiOx. Fang et al. [36] developed Au-NiO heterostructure nanosheets for photodynamic/photothermal synergistic antibacterial therapy and imaging tracking. The Au-NiO nanosheets were prepared via hydrothermal and laser-assisted methods, in which the charge transfer and resonance energy transfer of O-Au-O bonds enhanced the plasmonic effect and improved the photothermal conversion efficiency. As fluorescent probes, these nanosheets enabled targeted and early detection of bacteria, with limits of detection as low as 145.6 cfu/mL and 157.7 cfu/mL for Staphylococcus aureus and Pseudomonas aeruginosa, respectively, which is 3 orders of magnitude higher than the detection sensitivity of unmodified electrodes, and the detection recovery rate of actual samples reaches 95.2–102.3% with a relative standard deviation of less than 3.5% [36].
Meanwhile, the combination of photothermal and photodynamic therapy achieved a bactericidal rate of over 99% against both bacteria under sunlight. Furthermore, the nanosheets were composited with hydrogel to prepare nanodressings, realizing in situ monitoring and inactivation of bacteria at skin wound sites. With low cytotoxicity, this material provides an integrated platform for bacterial theranostics featuring targeting, imaging, and inactivation. In addition, phenylboronic acid-polyethylene glycol-long-chain alkyl C18 polymer [43] can be used to construct electrochemical sensors. Based on the specific binding between phenylboronic acid and glucose, accurate detection of glucose can be realized, which is suitable for clinical monitoring of diabetes. The combination of 3D printing technology and microfluidic chips provides a new platform for pharmaceutical analysis and detection. For example, an integrated microreactor prepared based on photocurable 3D printing technology [44] can be seamlessly integrated with a liquid chromatography-mass spectrometry (LC-MS) system for in vitro drug metabolism analysis. The system features simple operation and high stability, with relative standard deviations of chromatographic peak area and retention time of only 2.53% and 0.91%, respectively. Under the same sample matrix and detection conditions, the limits of detection (LOD) of the functional material-based detection methods discussed in this paper are of the same order of magnitude as those of HPLC-MS/MS technology. For some sensing systems constructed based on the target enrichment and signal amplification properties of materials, the LOD is reduced by 1–2 orders of magnitude compared with HPLC-MS/MS, demonstrating superior sensitivity for trace detection. The two detection technologies form an effective performance complementarity: HPLC-MS/MS holds a core advantage in the simultaneous qualitative and quantitative analysis of multiple analytes and the accurate analysis of complex matrices, while functional material-based detection methods feature simple operation, rapid response and portability, making them more suitable for on-site rapid detection and real-time monitoring scenarios. They can be used in synergy according to actual detection requirements. Current challenges for functional materials used in drug detection include: Biological fluids interfere with the signal stability and repeatability of electrochemical sensors in multiple aspects: biological macromolecules such as proteins and polypeptides are prone to non-specific adsorption on the electrode surface, forming an adsorption film that hinders electron transfer and causes signal attenuation and baseline drift; electrolyte ions and small molecule metabolites in body fluids alter the physicochemical properties of the system and undergo competitive reactions with detection targets, resulting in signal fluctuations; substances such as dissolved oxygen and reactive oxygen species can also corrode the electrode modification layer, damage the structure of the sensing interface, and reduce detection repeatability. To address the above interferences, strategies such as hydrophilic modification of electrode materials, introduction of specific recognition functional units, and construction of nanocomposite modification layers can be adopted to inhibit non-specific adsorption, reduce interference from coexisting substances, and enhance the structural stability of electrodes, thereby improving the signal performance of sensors in biological fluids [29,36]; susceptibility to interference in complex biological matrices, leading to reduced accuracy and reliability; poor biocompatibility of some detection materials, making them difficult to apply in in vivo detection; and insufficient miniaturization and portability of detection equipment. In the future, it is necessary to develop anti-fouling and biocompatible functional materials, combined with micro-nano processing technology and artificial intelligence algorithms, to realize in situ, real-time, and accurate detection of drugs.
3.4. Application in Bioimaging
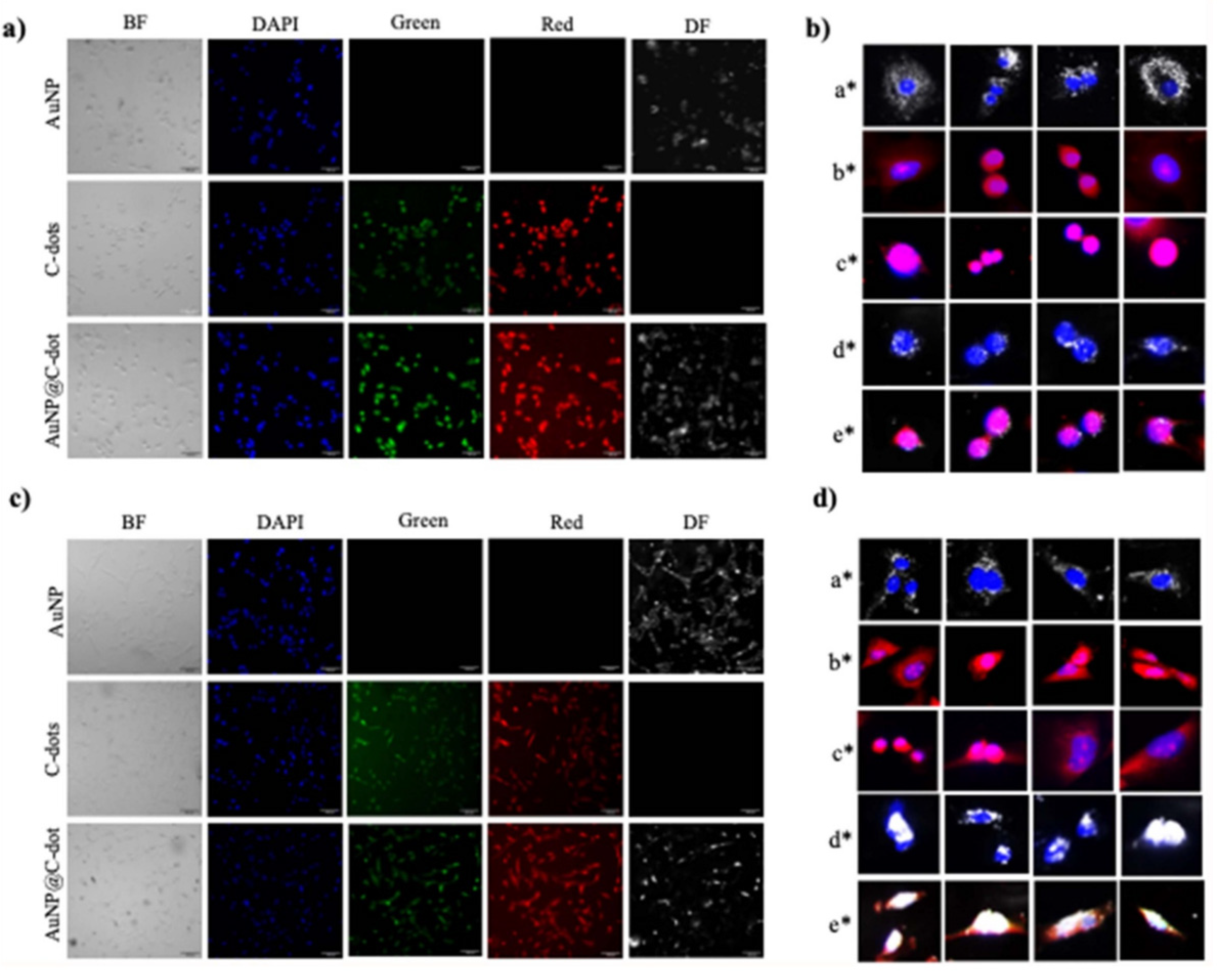
Bioimaging technology is an important tool for early disease diagnosis and the study of drug action mechanisms. As imaging contrast agents, functional materials can significantly improve imaging resolution and contrast, enabling accurate localization and visualization of lesion tissues. Nanomaterials are important carriers for bioimaging contrast agents. Iron oxide nanoparticles [45] exhibit superparamagnetism and are used for cellular imaging of tissues such as the liver and spleen. Gold nanoparticles (AuNPs) [46] are suitable for CT imaging due to their excellent X-ray attenuation coefficient (Figure 4). Through surface modification with targeting ligands, they can achieve specific imaging of tumor tissues, improving the accuracy of tumor diagnosis. MOF materials [47], with their tunable structure and function, can serve as carriers for multimodal imaging contrast agents, realizing MRI/CT, fluorescence/MRI and other multimodal imaging, providing more comprehensive information for accurate disease diagnosis. For example, magnetic MOF composites possess both magnetic response and fluorescence properties, enabling simultaneous MRI and fluorescence imaging to improve imaging sensitivity and specificity. In addition, polymer nanocarriers (such as dendrimers and nanomicelles) can achieve integration of drug delivery and imaging by encapsulating imaging reagents, allowing for real-time monitoring of drug distribution and release in vivo. The development of green nanotechnology has laid a foundation for the clinical application of bioimaging materials. Nanoparticles synthesized using plant extracts (such as mannose-modified gold nanoparticles) or bio-derived nanomaterials (such as magnetosomes) exhibit good biocompatibility and targeting ability, reducing in vivo toxicity and improving imaging safety. Clear research conclusions have been formed on the core optical and magnetic performance parameters of such bioimaging functional materials: the r2 relaxivity of superparamagnetic iron oxide nanoparticles is 100~200 mM−1·s−1, and the r1 relaxivity of ultra-small superparamagnetic iron oxide nanoparticles is about 5~15 mM−1·s−1; the CT attenuation coefficient of 5~20 nm gold nanoparticles reaches 5~10 HU·mM−1, which is significantly better than clinical iodine-based contrast agents; the fluorescence quantum yield of carbon dot–two-dimensional porphyrinic MOF composite fluorescent probes is 30~60%, and that of MOF-based two-photon imaging probes can also reach 25~40%. These parameters provide core performance support for the bioimaging application of materials [8,42,45,46,47]. For example, RGD-labeled magnetosomes with an r2 relaxivity of 156 mM−1·s−1 can achieve specific enrichment within 24 h in U87 glioma models, with a 3-fold increase in MRI contrast and a tumor/normal tissue enrichment ratio of 8.2:1, and the imaging signal-to-noise ratio is increased by 2.8 times [48]. The main challenges currently faced by functional materials for bioimaging include unclear long-term in vivo toxicity, insufficient imaging stability, and limited targeting ability. In the future, emphasis should be placed on developing degradable and biocompatible multifunctional imaging materials, combined with artificial intelligence algorithms to optimize image analysis, so as to achieve early and accurate diagnosis of diseases and real-time monitoring of therapeutic effects.
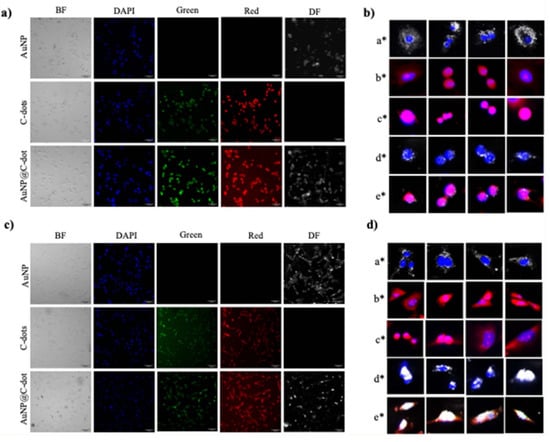
Figure 4.
Intracellular delivery of nanoparticles to A549 (a)/(b) and BEAS-2B (c)/(d) cells. Their localization in cells was visualized using red, green, bright field (BF) and dark-field (DF) channels. Nanoparticle accumulation is indicated by the overlap of dark/red signals with the DAPI-stained nucleus at the single-cell level. Overlaps: (a∗) DAPI & DF of AuNP, (b∗) red & DAPI of C-dots, (c∗) red & DAPI of AuNP@C-dot, (d∗) DAPI & DF of AuNP@C-dot, (e∗) red & DAPI & DF of AuNP@C-dot. Scale bars: 50 μm (a,c), 20 μm (b,d). (reprinted from Ref. [46]).
3.5. Multi-Functional Integrated Applications
With the development of precision medicine, functional materials featuring integrated diagnosis and treatment have become a research hotspot. Such materials can simultaneously achieve multiple functions including drug delivery, imaging diagnosis, and therapeutic efficacy monitoring, significantly improving therapeutic outcomes while reducing side effects. For example, MOF-based composites can load both drugs and imaging agents [49]. Folic acid-modified upconversion nanoscale MOFs with a drug loading of 18.5 wt% can realize targeted drug delivery and imaging localization of lesion tissues; the drug enrichment amount in tumor sites reaches 12.6 μg/g, which is 3.2 times higher than that of non-targeted materials, and the tumor inhibition rate reaches 90.6% after drug release. Magnetic nanocomposites can integrate magnetic-targeted drug delivery, MRI imaging, and magnetothermal therapy, providing a novel strategy for the comprehensive treatment of tumors. In addition, functional materials have also realized multi-functional integration in drug metabolism research. The integration of 3D-printed microreactors with LC-MS systems enables the combination of drug metabolism reactions and metabolite detection, simplifying operational procedures and improving detection efficiency, thus providing a high-performance platform for the study of drug metabolism mechanisms. The development of multi-functional integrated materials is still in the initial stage, facing challenges such as complex material structures, poor functional synergy, and difficulties in clinical translation. MOF-based nanomaterials exhibit distinct and regulable in vivo pharmacokinetic characteristics, organ enrichment distribution and metabolic clearance pathways: their pharmacokinetics are regulated by particle size and surface modification, with MOF nanoparticles of 20~100 nm modified by hydrophilic polymers such as polyethylene glycol having an in vivo circulation half-life of up to 6~24 h, and the blood drug concentration change conforming to the two-compartment model; in terms of organ distribution, unmodified MOFs are mainly enriched in the liver and spleen, while targeted ligand modification or particle size optimization enables specific enrichment in tumor and inflammatory tissues with a tumor/normal tissue enrichment ratio of 5~10 times; metabolic clearance is mainly dominated by renal urinary excretion after framework degradation, and undegraded particles are excreted with feces after phagocytosis by the reticuloendothelial system, with Fe/Mn-based MOFs and other biocompatible metal-based MOFs showing better in vivo metabolic safety [8,12,49]. In the future, precise material design is required to realize the synergistic effect of each functional unit. Meanwhile, it is necessary to strengthen the biosafety evaluation of materials to promote their clinical translation and application (Figure 5).
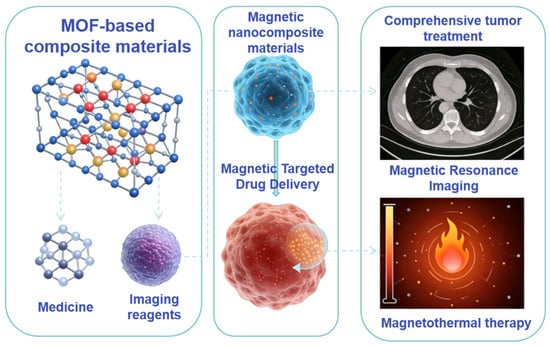
Figure 5.
MOF-based magnetic nanocomposites for integrated magnetic targeted drug delivery, MRI and magnetothermal therapy in comprehensive tumor treatment.
A systematic quantitative comparison of the core performance and efficiency parameters of typical functional materials is presented in Table 1, which clearly reflects the performance differences and application advantages of various materials in different pharmaceutical scenarios and provides a direct reference for the precise design and selection of functional materials.

Table 1.
Quantitative comparison of core performance/efficiency parameters of typical functional materials.
4. Key Challenges in the Application of Functional Materials
4.1. Biocompatibility and Safety
Biocompatibility is a fundamental prerequisite for the application of functional materials in in vivo diagnosis and therapy [50,51,52]. Certain functional materials may induce toxicity in vivo, leading to inflammation, organ damage, and other adverse effects [53,54,55]. For instance, heavy metal-based nanomaterials such as CdTe quantum dots and Pb-based MOFs can release toxic metal ions in vivo, causing damage to organs including the liver and kidneys [56]. The degradation products of some MOFs may also pose potential risks to living organisms. For example, Song et al. [57] reported that iron ion aggregates released during the degradation of oxidation-responsive poly (dimethylferrocene) aggregates can induce oxidative stress in vivo. This iron ion-mediated oxidative stress shares mechanistic similarities with the biological effects triggered by the degradation products of certain iron-based MOFs. Moreover, the size and surface properties of nanomaterials significantly influence their biocompatibility. Nanoparticles with small dimensions (<10 nm) can readily enter cells and induce cytotoxicity, while hydrophobic surfaces tend to promote nonspecific protein adsorption [58,59].
Nonspecific protein adsorption on material surfaces may lead to immune-mediated clearance, thereby reducing circulation time and bioavailability in vivo [60,61]. The adsorption of serum proteins onto biomaterial surfaces acts as a key trigger for macrophage recognition, phagocytosis, and rapid systemic clearance, posing a major obstacle to the clinical translation of nanomaterials. Jang et al. [62] demonstrated that protein nanoparticles functionalized with polyethylene glycol (PEG) and conjugated with an epidermal growth factor receptor (EGFR)-targeting affibody effectively evade serum protein-mediated nonspecific clearance and macrophage phagocytosis while maintaining targeted retention, resulting in a significantly prolonged in vivo circulation half-life. These findings indicate that PEG modification confers excellent resistance to protein adsorption, thereby helping to avoid macrophage phagocytosis and extend systemic circulation. This strategy offers a promising approach to reconcile the trade-off between nonspecific clearance and targeting efficiency in nanomaterials and presents a novel nanocarrier candidate for precision diagnosis and therapy of EGFR-related diseases.
Although surface modification with biocompatible materials such as PEG and chitosan can reduce nonspecific protein adsorption, the modification procedures are often complex and may compromise material functionality [63,64,65,66]. In addition, the immunogenicity of certain materials remains a critical concern affecting biocompatibility. Foreign body responses elicited by materials in vivo can trigger inflammation and impair therapeutic performance. Thus, enhancing the biocompatibility of functional materials while minimizing their in vivo toxicity remains a key challenge for their successful clinical translation [67].
4.2. Stability and Durability
The stability of functional materials in complex environments (such as in vivo body fluids and wastewater systems) directly determines their application performance. For example, MOFs are prone to structural collapse in aqueous environments, resulting in decreased adsorption and catalytic performance [68,69,70,71,72]. The metal ion leaching level of Fe/Mn-based and other metal-based MOFs is closely related to the system pH, ionic strength and the coordination bond stability of the materials themselves. Under the conventional application conditions of neutral aqueous environments and physiological fluids (pH 7.0~7.4), the metal ion leaching amount of such MOFs is mostly at the μg/L level. However, the coordination bonds are prone to dissociation under acidic conditions, leading to a slight increase in leaching level. The modification strategies mentioned in this paper, such as metal ion doping, ligand modification and heterojunction construction, can effectively inhibit metal ion leaching and enhance the structural stability of the materials. To maintain the structural integrity of MOFs in aqueous and biological environments, their water resistance and biocompatibility can be enhanced via strategies including ligand functional group modification to strengthen the coordination bond affinity, introduction of highly stable metal clusters for framework construction, hydrophilic polymer coating, and composite formation with inorganic nanomaterials. Meanwhile, crystal facet engineering can be employed to optimize the crystal structure, reduce framework swelling and dissociation in aqueous phases, and thus achieve the structural stability of MOFs in aqueous and biological environments [8,35].
Nanomaterials may aggregate in vivo or in water, impairing their imaging and drug delivery properties. The aggregation of nanomaterials is mainly caused by van der Waals forces and electrostatic interaction between particles. The surface modification of nanomaterials can reduce their aggregation, but the modification layer is easy to fall off in complex environments, leading to the reaggregation of particles. In the applications of drug degradation and adsorption, insufficient long-term durability and significant activity attenuation after repeated use increase the application cost. For example, the active sites on the surface of bio-based materials are easy to lose after repeated use, leading to the decline of adsorption and catalytic performance. The catalytic activity of some nanocomposites will decrease due to the leaching of active components and the blockage of active sites. Therefore, enhancing the structural stability and performance durability of materials in complex environments is an important guarantee for the large-scale application of functional materials [73,74,75].
4.3. Large-Scale Preparation and Cost Control
At present, the preparation of most functional materials remains at the laboratory small-scale stage [76]. The complex synthesis processes and high costs make it difficult to meet the large-scale demands of practical applications [77]. For instance, the synthesis of MOFs usually requires harsh reaction conditions (such as high temperature, high pressure and organic solvents) with relatively expensive raw materials. The small-scale synthesis of MOFs can ensure the uniformity of material structure and performance, but the batch-to-batch performance variation is large in large-scale synthesis, which affects the application effect. The preparation of nanocomposites often suffers from problems such as uneven dispersion and large batch-to-batch performance variations. The uniform dispersion of different components is the key to the preparation of nanocomposites, but in large-scale preparation, it is difficult to control the dispersion state of components, leading to the decline of material performance [78,79].
Although bio-based materials have abundant sources, the optimization and large-scale production of their modification processes still face challenges. The modification of bio-based materials usually requires multiple steps, and the reaction conditions are difficult to control in large-scale production, leading to the uneven modification of materials and the decline of performance. In addition, the regeneration and recycling of materials are also important factors affecting the application cost. The high regeneration cost and low cycle times of some materials limit their large-scale application in environmental treatment. Therefore, developing simple, efficient, low-cost and large-scale preparation technologies is a critical bottleneck for the industrial application of functional materials.
4.4. Clinical Translation and Regulatory Approval
The clinical application of functional materials in the pharmaceutical field requires strict regulatory approval, involving biosafety evaluation, efficacy verification, formulation of quality control standards, and other procedures [79,80,81,82,83]. At present, most multifunctional materials are still in the basic research stage, lacking systematic preclinical research and clinical trial data. The preclinical research of functional materials needs to carry out a series of experiments such as in vivo toxicity, pharmacokinetics and therapeutic effect, which takes a long time and is high-cost. In addition, the inconsistent quality control standards make it difficult to meet the requirements of regulatory approval. The preparation process of functional materials is complex, and the batch-to-batch performance variation is large, which makes it difficult to formulate unified quality control standards [84].
Furthermore, the long-term in vivo safety and therapeutic effects of materials still need further verification [85]. Most of the current research is limited to short-term in vivo experiments, and the long-term accumulation toxicity and chronic side effects of materials are not clear enough [86]. The clinical translation of functional materials also faces the problem of poor clinical transformation ability of laboratory achievements. The disconnection between laboratory research and clinical application makes it difficult for many excellent research achievements to be transformed into clinical products. In addition, the high research and development cost and long cycle of functional materials also limit the investment of enterprises, which affects the clinical translation process. Therefore, the clinical translation and regulatory approval of functional materials are important challenges restricting their clinical application.
5. Future Perspectives
5.1. Precise Design of Multifunctional Integrated Materials
Looking to the future, the precise design of multifunctional integrated materials should be guided by specific application requirements [87]. Through strategies such as multi-component synergistic assembly and precise surface modification, the efficient integration of functions—including degradation, adsorption, detection, imaging, and drug delivery—can be achieved, thereby enhancing the overall application performance of these materials [88]. The essence of precise design lies in realizing synergistic effects among different functional units while preserving the independent performance of each function [89]. For instance, the development of theranostic integrated materials capable of targeted imaging, intelligent drug release, and therapeutic efficacy monitoring will provide essential support for precision medicine. Intelligent drug release systems in particular can enable on-demand drug release in response to the microenvironment of diseased tissues (e.g., pH, reactive oxygen species, temperature), thereby reducing off-target effects and systemic toxicity.
The design of environmentally friendly materials that integrate high-efficiency adsorption with catalytic degradation holds great promise for the advanced treatment of pharmaceutical pollution. Adsorption-degradation integrated materials can first enrich low-concentration drug residues via adsorption and subsequently degrade the adsorbed molecules through catalytic processes, enabling efficient removal of drug contaminants. Furthermore, the precise design of materials must also account for the practical conditions of their intended application environments—such as the development of anti-interference materials for use in complex water matrices or biocompatible materials for in vivo applications. Integrating computational simulation with material design offers the potential for predictive material engineering, improving design efficiency and reducing research and development costs.
5.2. Green Synthesis and Sustainable Development
Green synthesis technology is poised to become a key development direction for functional materials in the pharmaceutical field, driven by the need to address challenges such as high preparation costs, environmental pollution, and limited biocompatibility [90,91,92]. The core principle of green synthesis lies in minimizing the environmental impact of material preparation while maintaining material performance, thereby supporting the sustainable development of materials research and application.
First, renewable biomass resources are expected to serve as primary feedstocks for the preparation of green functional materials, replacing conventional non-renewable chemical raw materials. Biomass sources—including plant fibers, chitosan, peat, and fruit waste—offer advantages such as abundant availability, low cost, and biodegradability. Through simple physical or chemical modification, these materials can be converted into bio-based adsorbents, catalytic materials, and drug carriers. For example, employing lignin and cellulose derived from agricultural and forestry waste as organic ligands to synthesize bio-based metal–organic frameworks (MOFs) can reduce production costs and enhance material biodegradability, thereby mitigating secondary environmental pollution caused by material accumulation. Ghazaleh Ashrafi et al. [91] addressed key challenges in environmental pollution control by systematically reviewing biosynthetic pathways utilizing biological waste and naturally derived nanomaterials. Their work highlighted the application mechanisms and environmental benefits of these materials in degrading waterborne dyes, removing nitrate-based metal pollutants, and constructing eco-friendly composites. Furthermore, the use of plant extracts as substitutes for toxic chemical reducing agents and stabilizers in nanomaterial synthesis enables the green preparation of metal nanoparticles and nanocomposites while simultaneously improving their biocompatibility.
Second, the development of mild and efficient synthetic processes will gradually replace traditional harsh preparation conditions. Conventional methods for synthesizing functional materials such as MOFs and nanocomposites often require high temperature, high pressure, organic solvents, and prolonged reaction times, resulting in substantial energy consumption and environmental pollution [93]. In contrast, emerging techniques—including room-temperature solvent-free synthesis, microwave-assisted synthesis, and ultrasonic synthesis—offer advantages such as low energy consumption, short reaction times, and reduced environmental impact, rendering them suitable for large-scale production. Bag et al. [94] employed microwave-assisted solvothermal methods to synthesize a series of structurally analogous lanthanide metal–organic frameworks (Ln-MOFs) on a large scale, achieving rapid synthesis within five minutes. Compared with conventional solvothermal approaches, this method markedly improved efficiency, with a maximum yield of 2 g—significantly surpassing the typical 10 mg scale of traditional methods. For instance, microwave-assisted synthesis enables the rapid preparation of Fe-based MOFs at room temperature, yielding materials with higher specific surface area and more active sites than those produced via conventional hydrothermal synthesis. In addition, replacing traditional toxic organic solvents with green alternatives such as water, ethanol, or ionic liquids can further reduce the environmental footprint of the synthesis process.
Third, the design of degradable and recyclable functional materials will be increasingly emphasized to enable closed-loop material utilization. For environmental functional materials employed in drug degradation and adsorption, developing recyclable materials with high cycling stability can reduce application costs and prevent secondary pollution. For example, modifying bio-based materials with magnetic nanoparticles facilitates rapid separation and recovery from aqueous environments, thereby improving recycling efficiency. Chen et al. [95] addressed this by incorporating magnetic nickel carbon black as a loading agent in flow-electrode electrochemical deionization technology, optimizing the preparation of ammonia-rich solutions while simultaneously resolving the challenge of electrode solid–liquid separation. For biomedical functional materials intended for in vivo diagnosis and therapy, designing degradable materials that can be safely metabolized by the body offers a solution to the issue of long-term accumulation toxicity. For instance, degradable polymer nanomicelles and MOFs modified with biodegradable ligands can break down into non-toxic small molecules in vivo and be excreted through metabolic pathways, significantly enhancing the biosafety of these materials.
5.3. Interdisciplinary Integration and Technological Innovation
The innovative advancement of functional materials in the pharmaceutical field relies on the cross-integration of multiple disciplines, including materials science, pharmacy, medicine, computer science, micro-nano processing, and artificial intelligence [96]. Such interdisciplinary convergence fosters new research paradigms and technical methodologies, helping to overcome the current bottlenecks in the development of functional materials and driving the continuous upgrading and iteration of both material performance and application technologies.
The integration of artificial intelligence (AI) and materials science is an important trend in the precise design and high-throughput screening of functional materials [97]. Traditional material design relies on experimental trial and error, which has the disadvantages of long cycles, high cost and low efficiency. AI algorithms such as machine learning and deep learning can be used to establish the structure–performance correlation model of functional materials by learning a large number of existing experimental data and theoretical calculation data [98]. Based on this model, the performance of new materials can be predicted, and the optimal material composition and preparation process can be designed, which significantly improves the efficiency of material development [99]. For example, machine learning can be used to screen the metal centers and organic ligands of MOF materials, and design MOF materials with high catalytic activity and good stability for drug degradation [100]. In addition, AI can also be combined with bioimaging and drug detection technology to realize the intelligent analysis of imaging data and detection data, improve the accuracy of disease diagnosis and drug detection [101].
Micro-nano processing technology and 3D printing technology provide new technical support for the personalized preparation and precise application of functional materials [102,103]. 3D printing technology can realize the personalized preparation of drug delivery carriers and bioimaging probes according to the individual characteristics of patients and the structural characteristics of lesion tissues, which is in line with the development demand of precision medicine. For example, 3D-printed hydrogel scaffolds loaded with functional nanomaterials can realize the targeted delivery of drugs to tumor tissues and the real-time monitoring of therapeutic effects. Kumar et al. [104] prepared oxygen-and drug-loaded periodic mesoporous organosilicon nanoparticles and constructed a 3D-printable nano-composite hydrogel precursor based on gelatin methacrylate to fabricate 3D scaffolds. Using rutin as a hydrophobic drug model, the material was demonstrated to enhance cell viability under both normoxic and hypoxic conditions, enabling sustained long-term delivery of drugs and oxygen. The interaction between fibroblasts and the hydrogel scaffold was tested to evaluate its impact on cell viability. Rheological and mechanical property characterization confirmed the hydrogel’s injectability and excellent 3D printing performance, validating its potential application in constructing artificial tissue structures. Micro-nano processing technology can prepare micro-nano scale functional material devices with high precision and high integration, such as microfluidic chips combined with electrochemical sensors, which can realize the rapid and on-site detection of trace drug molecules and biological markers. The combination of 3D printing and microfluidic technology can prepare integrated microreactors for in vitro drug metabolism analysis, which simplifies the experimental operation and improves the efficiency of drug research and development.
The cross-disciplinary integration of materials science and clinical medicine holds significant promise for advancing the clinical translation of functional materials. Strengthening the collaboration between materials researchers and clinical practitioners ensures that the development of functional materials is more closely aligned with real-world clinical needs, thereby addressing key challenges in their practical application. For instance, by considering the distinct clinical characteristics of various tumor types, it is possible to design integrated theranostic materials tailored to specific imaging modalities and drug release profiles. Moreover, clinical medicine offers a wealth of biological models and clinical data that facilitate in-depth investigations into the in vivo behavior and therapeutic efficacy of these materials. Similarly, the convergence of materials science and environmental science can drive the development of functional materials aimed at controlling pharmaceutical pollution. By accounting for the specific characteristics of real-world environmental contamination, materials can be tailored for the treatment of diverse types of pharmaceutical wastewater.
5.4. Clinical Translation and Improved Regulatory Systems
Accelerating the clinical translation of functional materials and strengthening the corresponding regulatory frameworks are critical to advancing their practical application in the pharmaceutical field, particularly in clinical diagnosis and therapy. To address the current bottlenecks in clinical translation—including insufficient preclinical research, inconsistent quality control standards, and underdeveloped regulatory systems—a series of targeted measures must be implemented to facilitate the successful transition of functional materials from bench to bedside.
First, strengthen the systematic preclinical research of functional materials and accumulate sufficient biosafety and efficacy data. On the basis of in vitro experiments, carry out long-term in vivo animal experiments to study the pharmacokinetics, tissue distribution, metabolic pathway, long-term toxicity and immunogenicity of materials in the body. For diagnosis- and treatment-integrated materials, it is necessary to carry out strict efficacy evaluation experiments to verify the therapeutic effect and safety of materials in different animal models. In addition, the establishment of standardized preclinical research platforms can realize the unified detection and evaluation of the performance of functional materials and improve the reliability and comparability of preclinical research data.
Second, establish unified quality control standards and evaluation systems for functional materials. The complex preparation process and large batch-to-batch performance variation in functional materials are the main obstacles to the formulation of quality control standards. It is necessary to combine the characteristics of different types of functional materials, formulate unified quality control indicators and detection methods from the aspects of raw material selection, preparation process, product performance and storage conditions. For example, for MOF materials, the key quality control indicators such as specific surface area, pore size distribution, active site content and structural stability should be clearly defined. For nanocomposites, the particle size, dispersion, component ratio and surface modification effect should be strictly controlled. The establishment of a standardized evaluation system can realize the scientific and objective evaluation of the performance and application potential of functional materials and provide a basis for the regulatory approval and industrial production of materials.
Third, promote the deep cooperation of industry–university–research and accelerate the technological transformation of functional materials. The disconnection between laboratory research and industrial production is an important reason for the slow clinical translation of functional materials. Strengthening the cooperation between universities, research institutions and enterprises can combine the research advantages of universities and research institutions with the production and market advantages of enterprises and realize the seamless connection from laboratory research to industrial production. Enterprises can participate in the research and development of functional materials in the early stage and carry out pilot test and industrialization research according to the market demand, which can reduce the research and development risk and accelerate the technological transformation. In addition, the government can provide policy and financial support for the research and development and industrialization of functional materials, such as setting up special research funds and supporting the construction of industrialization bases.
Fourth, improve the regulatory approval system for functional materials and optimize the approval process. According to the characteristics of functional materials, especially multifunctional integrated materials, formulate targeted regulatory approval guidelines and classify and manage materials according to their application fields and risk levels. For low-risk environmental functional materials, simplify the approval process and focus on the evaluation of material performance and environmental safety. For high-risk biomedical functional materials, strict regulatory approval standards should be formulated, and the whole process of material research, development, production and application should be supervised. In addition, strengthening international cooperation in regulatory approval and recognizing the mutual recognition of preclinical research data and approval results can accelerate the global clinical translation and application of functional materials.
6. Conclusions
Functional materials represented by metal–organic frameworks, nanocomposites and bio-based materials have shown broad application prospects in the pharmaceutical field including drug degradation, adsorption, pharmaceutical analysis and detection, bioimaging and integrated diagnosis and treatment. These materials rely on their unique structural characteristics and performance advantages to provide effective solutions for the control of environmental pharmaceutical pollution and the realization of precise clinical diagnosis and treatment and have become an important research hotspot in the cross field of materials science, pharmacy, environmental science and clinical medicine.
This paper systematically summarizes the structural characteristics, modification strategies and latest application progress of typical functional materials in various pharmaceutical-related fields and carries out quantitative comparison and critical analysis of the performance of different materials through structured tables. The structure–performance correlation of functional materials is deeply discussed, and the existing research limitations and research gaps in each application field are clearly pointed out. On this basis, the key challenges faced by the practical application and clinical translation of functional materials are comprehensively analyzed from four aspects: biocompatibility and safety, stability and durability, large-scale preparation and cost control, clinical translation and regulatory approval. In view of the current research status and existing problems of functional materials, the future research directions are prospected from four aspects: precise design of multifunctional integrated materials, green synthesis and sustainable development, interdisciplinary integration and technological innovation, clinical translation and improvement of regulatory systems. The precise design of materials is the core to improve the comprehensive performance of functional materials; green synthesis technology is the inevitable requirement to realize the sustainable development of materials; interdisciplinary integration is the important driving force for the innovative development of materials; and the improvement of clinical translation and regulatory systems is the key to promoting the practical application of materials. The four aspects are interrelated and mutually promoting, jointly pointing out the development path for in-depth research and industrial application of functional materials in the pharmaceutical field.
With the continuous innovation of material synthesis technology and the in-depth cross-integration of multiple disciplines, functional materials will be developed in the direction of high performance, multi-function, greenization and personalization. It is believed that the key challenges faced by the application of functional materials will be gradually solved through the joint efforts of researchers in various fields, and functional materials will play a more important role in the fields of environmental protection and precision medicine, providing strong material support for the sustainable development of the pharmaceutical industry and the improvement of clinical diagnoses and treatment levels.
Author Contributions
Conceptualization, X.C. (Xiaoyi Chen), W.W. and Y.L.; Writing—original draft, Y.W.; Writing—review and editing, X.T.; Validation, S.H. and X.C. (Xi Cao); Funding acquisition, Y.L. and W.W. All authors have read and agreed to the published version of the manuscript.
Funding
His work has been supported by the Department of Scientific Research project in Heilong Jiang Province (No. LH2022B022), 2025 Research Startup Fund Project for Doctoral Recipients (No. JMSUBZ2025-09) and “Research and development team of northern unique medicinal resources”, Jiamusi University “East Pole” academic team (team no. DJXSTD202403).
Data Availability Statement
The data and self-drawn figures supporting the findings of this study are not publicly available due to the involvement of privacy-related content. All relevant data are available from the corresponding author upon reasonable request.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Miettinen, M.; Khan, S.A. Pharmaceutical pollution: A weakly regulated global environmental risk. Rev. Eur. Comp. Int. Environ. Law 2022, 31, 75–88. [Google Scholar] [CrossRef]
- Zampelli, S.; Verna, R. Environmental Pollution from Pharmaceuticals. Life 2025, 15, 1341. [Google Scholar] [CrossRef]
- de Oliveira Souza, H.; dos Santos Costa, R.; Quadra, G.R.; dos Santos Fernandez, M.A. Pharmaceutical pollution and sustainable development goals: Going the right way? Sustain. Chem. Pharm. 2021, 21, 100428. [Google Scholar] [CrossRef]
- Hu, J.; Chen, Y.; Li, Y.; Zhou, Z.; Cheng, Y. A thermo-degradable hydrogel with light-tunable degradation and drug release. Biomaterials 2017, 112, 133–140. [Google Scholar] [CrossRef]
- Kryuchkova, M.; Batasheva, S.; Akhatova, F.; Babaev, V.; Buzyurova, D.; Vikulina, A.; Volodkin, D.; Fakhrullin, R.; Rozhina, E. Pharmaceuticals removal by adsorption with montmorillonite nanoclay. Int. J. Mol. Sci. 2021, 22, 9670. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, D.; Garg, T.; Sheoran, A.; Singhal, S. Exploring the potential of MIL-53 (Fe) as a fluorescent probe for the highly selective and sensitive detection of anti-tuberculosis drug—Isoniazid. J. Mol. Struct. 2024, 1297, 136899. [Google Scholar] [CrossRef]
- Witika, B.A.; Bassey, K.E.; Demana, P.H.; Siwe-Noundou, X.; Poka, M.S. Current advances in specialised niosomal drug delivery: Manufacture, characterization and drug delivery applications. Int. J. Mol. Sci. 2022, 23, 9668. [Google Scholar] [CrossRef]
- He, S.; Wu, L.; Li, X.; Sun, H.; Xiong, T.; Liu, J.; Huang, C.; Xu, H.; Sun, H.; Chen, W.; et al. Metal-organic frameworks for advanced drug delivery. Acta Pharm. Sin. B 2021, 11, 2362–2395. [Google Scholar] [CrossRef] [PubMed]
- Barati, M.; Raissi, H.; Ghahari, A.; Farzad, F. Efficient Covalent Organic Frameworks for Treating Pharmaceuticals in Water. Ind. Eng. Chem. Res. 2025, 64, 13196–13210. [Google Scholar] [CrossRef]
- Rowley, J.V.; Wall, P.A.; Yu, H.; Howard, M.J.; Baker, D.L.; Kulak, A.; Green, D.C.; Thornton, P.D. Triggered and monitored drug release from bifunctional hybrid nanocomposites. Polym. Chem. 2022, 13, 100–108. [Google Scholar] [CrossRef]
- Iacob, A.T.; Lupascu, F.G.; Apotrosoaei, M.; Vasincu, I.M.; Tauser, R.G.; Lupascu, D.; Giusca, S.E.; Caruntu, I.D.; Profire, L. Recent biomedical approaches for chitosan based materials as drug delivery nanocarriers. Pharmaceutics 2021, 13, 587. [Google Scholar] [CrossRef]
- Lawson, H.D.; Walton, S.P.; Chan, C. Metal–organic frameworks for drug delivery: A design perspective. ACS Appl. Mater. Interfaces 2021, 13, 7004–7020. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Xu, J.W.; Liu, W.C.; Yang, S.Z.; Luo, M.M.; Han, Y.Y.; Liu, J.Q.; Batten, S.R. Designed metal–organic framework based on metal–organic polyhedron: Drug delivery. Inorg. Chem. Commun. 2016, 71, 32–34. [Google Scholar] [CrossRef]
- Maranescu, B.; Visa, A. Applications of metal-organic frameworks as drug delivery systems. Int. J. Mol. Sci. 2022, 23, 4458. [Google Scholar] [CrossRef]
- Li, X.; Lachmanski, L.; Safi, S.; Sene, S.; Serre, C.; Greneche, J.M.; Zhang, J.; Gref, R. New insights into the degradation mechanism of metal-organic frameworks drug carriers. Sci. Rep. 2017, 7, 13142. [Google Scholar] [CrossRef]
- Livesey, T.C.; Mahmoud, L.A.; Katsikogianni, M.G.; Nayak, S. Metal–organic frameworks and their biodegradable composites for controlled delivery of antimicrobial drugs. Pharmaceutics 2023, 15, 274. [Google Scholar] [CrossRef] [PubMed]
- Zulfiqar, N.; Nadeem, R.; Musaimi, O.A. Photocatalytic degradation of antibiotics via exploitation of a magnetic nanocomposite: A green nanotechnology approach toward drug-contaminated wastewater reclamation. ACS Omega 2024, 9, 7986–8004. [Google Scholar] [CrossRef]
- Awfa, D.; Ateia, M.; Fujii, M.; Yoshimura, C. Novel magnetic carbon nanotube-TiO2 composites for solar light photocatalytic degradation of pharmaceuticals in the presence of natural organic matter. J. Water Process Eng. 2019, 31, 100836. [Google Scholar] [CrossRef]
- Justin, R.; Chen, B. Multifunctional chitosan–magnetic graphene quantum dot nanocomposites for the release of therapeutics from detachable and non-detachable biodegradable microneedle arrays. Interface Focus 2018, 8, 20170055. [Google Scholar]
- Hojjati-Najafabadi, A.; Mansoorianfar, M.; Liang, T.; Shahin, K.; Wen, Y.; Bahrami, A.; Karaman, C.; Zare, N.; Karimi-Maleh, H.; Vasseghian, Y. Magnetic-MXene-based nanocomposites for water and wastewater treatment: A review. J. Water Process Eng. 2022, 47, 102696. [Google Scholar]
- Hovhannisyan, V.; Siposova, K.; Musatov, A.; Chen, S.J. Development of multifunctional nanocomposites for controlled drug delivery and hyperthermia. Sci. Rep. 2021, 11, 5528. [Google Scholar] [CrossRef]
- Peltonen, L.; Hirvonen, J. Drug nanocrystals–versatile option for formulation of poorly soluble materials. Int. J. Pharm. 2018, 537, 73–83. [Google Scholar] [PubMed]
- Wei, S.; Li, Y.; Liang, H.; Yen, Y.; Lin, Y.; Chang, H. Photoluminescent carbon nanomaterials for sensing of illicit drugs: Focus. Anal. Sci. 2022, 38, 247–260. [Google Scholar] [CrossRef] [PubMed]
- Savaser, A.; Esim, O. Current Trends in Nanomaterials: Drug Delivery and their Sensitive Assays. Curr. Drug Deliv. 2021, 18, 102. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Lei, S. Dual-mode detection of illicit drugs based on ZIF-8/Ag core–shell nanomaterials. Anal. Methods 2025, 17, 6859–6865. [Google Scholar] [CrossRef] [PubMed]
- Amiri, M.; Imanzadeh, H.; Sefid-Sefidehkhan, Y. An overview on electrochemical sensors based on nanomaterials for the determination of drugs of abuse. Curr. Drug Deliv. 2021, 18, 162–183. [Google Scholar] [CrossRef]
- Chen, L.; Wu, B.; Yuan, L.X. Application Progress of Nanomaterial-Based Delivery Systems in the Diagnosis and Treatment of Atherosclerosis. J. Shanghai Jiao Tong Univ. (Med. Sci.) 2025, 45, 1687–1693. [Google Scholar]
- Mu, J. Construction of Fluorescence Enhancement Systems Based on Metal Nanoclusters and Their Applications in Sensing. Doctoral Dissertation, Jilin University, Changchun, China, 2025. [Google Scholar]
- Zhang, Z.; Han, W.; Qing, J.; Meng, T.; Zhou, W.; Xu, Z.; Chen, M.; Wen, L.; Cheng, Y.; Ding, L. Functionalized magnetic metal organic framework nanocomposites for high throughput automation extraction and sensitive detection of antipsychotic drugs in serum samples. J. Hazard. Mater. 2024, 465, 133189. [Google Scholar] [CrossRef]
- Mohan, B.; Kumari, R.; Singh, G.; Singh, K.; Pombeiro, A.J.; Yang, X.; Ren, P. Covalent organic frameworks (COFs) and metal–organic frameworks (MOFs) as electrochemical sensors for the efficient detection of pharmaceutical residues. Environ. Int. 2023, 175, 107928. [Google Scholar] [CrossRef]
- Lak, M.; Hassaninejad-Darzi, S.K. Synergistic effect of silver, reduced graphene oxide and metal-organic framework-5 nanocomposite for the simultaneous voltammetric sensing of darunavir and naproxen pharmaceutical analytes using chemometric methods. Sens. Bio-Sens. Res. 2025, 51, 100933. [Google Scholar] [CrossRef]
- Sakamoto, R.; Fukui, N.; Maeda, H.; Toyoda, R.; Takaishi, S.; Tanabe, T.; Komeda, J.; Amo-Ochoa, P.; Zamora, F.; Nishihara, H. Layered metal-organic frameworks and metal-organic nanosheets as functional materials. Coord. Chem. Rev. 2022, 472, 214787. [Google Scholar]
- Jin, E.; Lee, I.S.; Yang, D.C.; Moon, D.; Nam, J.; Cho, H.; Kang, E.; Lee, J.; Noh, H.J.; Min, S.K.; et al. Origamic metal-organic framework toward mechanical metamaterial. Nat. Commun. 2023, 14, 7938. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Jia, F.; Wang, H.; Chen, F.; Fang, Y.; Dong, W.; Zeng, G.; Li, X.; Yang, Q.; Yuan, X. Simultaneously efficient adsorption and photocatalytic degradation of tetracycline by Fe-based MOFs. J. Colloid Interface Sci. 2018, 519, 273–284. [Google Scholar] [CrossRef]
- Zhang, R.; Xue, Z.; Jia, K.; Zhu, Y.; Liang, H.; Li, Q.; Yuan, N. Critical trigger of self-assembled bimetallic Fe/Mn-MOF with SnS2 heterojunctions by persulfate activation for efficient tetracyclines photodegradation. Environ. Res. 2024, 263, 120060. [Google Scholar] [CrossRef]
- Fang, G.; Liu, X.; Wang, D.; Si, T.; Liu, Z. Near-field coupling resonance enhancement with AuNiO Heterostructure nanosheets for high-performance photodynamic/photothermal Hybrid antibacterial & imaging tracking. Biosens. Bioelectron. 2025, 271, 117034. [Google Scholar]
- Uwayezu, J.N.; Kumpiene, J.; Carabante, I. Efficient removal of PFAS in groundwater and landfill leachate using iron-modified peat residues and electrochemical oxidation. J. Environ. Chem. Eng. 2025, 13, 118833. [Google Scholar] [CrossRef]
- Mohammadzadeh, M.; Leiviskä, T. Iron-modified peat and magnetite-pine bark biosorbents for levofloxacin and trimethoprim removal from synthetic water and various pharmaceuticals from real wastewater. Ind. Crops Prod. 2023, 195, 116491. [Google Scholar] [CrossRef]
- Fablet, L.; Choueikani, F.; Pédrot, M.; Marsac, R. What are the effects of environmental factors on Co speciation at the magnetite surface? Environ. Sci. Nano 2024, 11, 2036–2048. [Google Scholar] [CrossRef]
- Kaity, S.; Ghosh, A. Facile preparation of acrylamide grafted locust bean gum-poly (vinyl alcohol) interpenetrating polymer network microspheres for controlled oral drug delivery. J. Drug Deliv. Sci. Technol. 2016, 33, 1–12. [Google Scholar] [CrossRef]
- Amdeha, E. Biochar-based nanocomposites for industrial wastewater treatment via adsorption and photocatalytic degradation and the parameters affecting these processes. Biomass Convers. Biorefinery 2024, 14, 23293–23318. [Google Scholar] [CrossRef]
- Chai, H.; Ma, Y.; Yuan, Z.; Li, Y.; Liu, G.; Chen, L.; Tian, Y.; Tan, W.; Ma, J.; Zhang, G. A ratiometric fluorescence sensor based on carbon dots and two-dimensional porphyrinic MOFs for on-site monitoring of sulfide. Microchem. J. 2023, 185, 108288. [Google Scholar] [CrossRef]
- Zhao, Y.; Liu, C.; Chen, H.; Zhou, H.; Yu, S.; Mi, D.; Yue, S.; Qiao, W. Synthesis of asymmetrically dihydrophobic chain poly (ethylene glycol) lipids for long circulation and membrane fusion. J. Surfactants Deterg. 2022, 25, 643–654. [Google Scholar] [CrossRef]
- Mi, S.; Du, Z.; Xu, Y.; Sun, W. The crossing and integration between microfluidic technology and 3D printing for organ-on-chips. J. Mater. Chem. B 2018, 6, 6191–6206. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Cha, R.; Long, K.; Liu, Y.; Dong, Y.; Zhang, Y.; Wang, X.; Jiang, X. Functionalized Iron Oxide Nanoparticles for Both Dual-Modal Imaging and Erythropoiesis. ACS Appl. Mater. Interfaces 2024, 16, 68905–68917. [Google Scholar] [CrossRef] [PubMed]
- Kavuranpala, T.; Saydullaeva, I.; Özçelik, S. Developing gold nanoparticles decorated with carbon-dots for multiplexed cellular imaging. Nanotechnology 2025, 36, 445102. [Google Scholar] [CrossRef]
- Yang, C.; Wang, K.; Li, Z.; Mo, L.; Lin, W. A two-photon metal-organic framework nanoprobe with catalytic hairpin assembly for amplified MicroRNA imaging in living cells and tissues. Sens. Actuators B Chem. 2022, 359, 131593. [Google Scholar] [CrossRef]
- Tsutsumi, H.; Shirai, T.; Ohkusa, H.; Mihara, H. Gold nanoparticles conjugated with glycopeptides for lectin detection and imaging on cell surface. Protein Pept. Lett. 2018, 25, 84–89. [Google Scholar] [CrossRef]
- Chowdhuri, A.R.; Laha, D.; Pal, S.; Karmakar, P.; Sahu, S.K. One-pot synthesis of folic acid encapsulated upconversion nanoscale metal organic frameworks for targeting, imaging and pH responsive drug release. Dalton Trans. 2016, 45, 18120–18132. [Google Scholar] [CrossRef]
- Li, Y.; Tao, T.; Xiong, Y.; Guo, W.; Liang, Y. Multifunctional PLGA nanosystems: Enabling integrated diagnostic and therapeutic strategies. Front. Pharmacol. 2025, 16, 1670397. [Google Scholar] [CrossRef]
- Shawcross, J.; Bakhai, A.; Ansaripour, A.; Armstrong, J.; Lewis, D.; Agg, P.; Godoy, R.; Blunn, G. In vivo biocompatibility and pacing function study of silver ion-based antimicrobial surface technology applied to cardiac pacemakers. Open Heart 2017, 4, e000357. [Google Scholar] [CrossRef]
- Yang, M.Y.; Wang, Y.H.; Kuo, P.Y.; Yang, Y.C.; Hsieh, W.Y.; Liu, S.Y.; Huang, Y.F.; Yen, C.M.; Tang, C.M.; Liu, B.S.; et al. In vitro and In Vivo evaluation of graphene oxide–gold nanocomposites for enhanced biocompatibility and functional performance in biomaterial application. Colloids Surf. A Physicochem. Eng. Aspects 2025, 725, 137721. [Google Scholar] [CrossRef]
- Weimer, P.; de Araújo Lock, G.; Amaral Antunes Nunes, K.; Cassanta Rossi, R.; Scherer Koester, L. Association effect of the phytocannabinoid beta-caryophyllene and indomethacin carried in topical nanoemulgels: An evaluation by in vivo anti-inflammatory model. Nat. Prod. Res. 2025, 39, 3335–3341. [Google Scholar]
- Li, Z.; Shi, M.; Li, N.; Xu, R. Application of functional biocompatible nanomaterials to improve curcumin bioavailability. Front. Chem. 2020, 8, 589957. [Google Scholar] [CrossRef]
- Elansary, M.; Belaiche, M.; Mouhib, Y.; Lemine, O.M.; Bentarhlia, N.; Bsoul, I. Novel biocompatible nanomaterial for biomedical application: Structural, morphological, magnetic, and in vivo toxicity investigations. Ceram. Int. 2023, 49, 4551–4570. [Google Scholar]
- Aftab, A.; Waseem, M. Remediation of carcinogen group I metal contaminants; insights into the application of functionalized carbon nanotubes. J. Mol. Liq. 2025, 434, 128064. [Google Scholar] [CrossRef]
- Song, C.; Chen, M.; Tan, J.; Xu, J.; Zhang, Y.; Zhang, G.; Hu, X.; Liu, S. Self-amplified cascade degradation and oxidative stress via rational pH regulation of oxidation-responsive poly (ferrocene) aggregates. J. Am. Chem. Soc. 2023, 145, 17755–17766. [Google Scholar]
- Ogawa, A.; Tahori, A.; Yano, M.; Hirobe, S.; Terada, S.; Kanematsu, H. Antibiofilm property and biocompatibility of siloxane-based polymer coatings applied to biomaterials. Materials 2023, 16, 7399. [Google Scholar] [CrossRef] [PubMed]
- Aminov, L.; Sindilar, E.V.; Pasca, A.S.; Antohi, C.; Decolli, Y.; Stamatin, O.; Costin, L.L.; Bulancea, B.P.; Francu, L.; Mihalas, E.; et al. In Vivo Evaluation of Biocompatibility of Three Biomaterials Used in Endodontics for Prosthetic Purposes in Complex Rehabilitation Treatment. Appl. Sci. 2021, 11, 6519. [Google Scholar] [CrossRef]
- Vincent, M.P.; Bobbala, S.; Karabin, N.B.; Frey, M.; Liu, Y.; Navidzadeh, J.O.; Stack, T.; Scott, E.A. Surface chemistry-mediated modulation of adsorbed albumin folding state specifies nanocarrier cleara. Nat. Commun. 2020, 12, 648. [Google Scholar] [CrossRef]
- Xiao, Y.; Zhou, Z.; Zuo, Y.; Wu, X.; Liu, Y.; Li, Y.; Gao, Y.; Zhang, X.; Wang, Y.; Hu, L.; et al. Layer-by-layer fabrication of alginate/polyethyleneimine multilayer on magnetic interface with enhanced efficiency in immuno-capturing circulating tumor cells. Anal. Chim. Acta 2024, 1312, 342778. [Google Scholar] [CrossRef] [PubMed]
- Jang, S.; Jun, H.; Eom, S.; Zhao, S.; Murthy, N.; Kang, S.; Kim, H. EGFR Affibody and PEG functionalized protein nanoparticles: Sustaining targeting and macrophage evasion. Int. J. Biol. Macromol. 2025, 307, 142167. [Google Scholar] [CrossRef]
- Bračič, M.; Mohan, T.; Griesser, T.; Stana-Kleinschek, K.; Strnad, S.; Fras-Zemljič, L. One-step noncovalent surface functionalization of PDMS with chitosan-based Bioparticles and their protein-repellent properties. Adv. Mater. Interfaces 2017, 4, 1700416. [Google Scholar] [CrossRef]
- Gökaltun, A.; Kang, Y.B.; Yarmush, M.L.; Usta, O.B.; Asatekin, A. Simple surface modification of poly (dimethylsiloxane) via surface segregating smart polymers for biomicrofluidics. Sci. Rep. 2019, 9, 7377. [Google Scholar] [CrossRef]
- Tian, R.; Luo, M.; Li, J. Spontaneous protein desorption from self-assembled monolayer (SAM)-coated gold nanoparticles. Phys. Chem. Chem. Phys. 2018, 20, 68–74. [Google Scholar] [CrossRef]
- Bryniarski, M.A.; Tuhin, M.T.H.; Shomin, C.D.; Nasrollahi, F.; Ko, E.C.; Soto, M.; Chung, K.; Poon-Andersen, C.; Primack, R.; Wong, D.; et al. Utility of cellular measurements of non-specific endocytosis to assess the target-independent clearance of monoclonal antibodies. J. Pharm. Sci. 2024, 113, 3100–3111. [Google Scholar] [CrossRef]
- Zhang, R.; Ma, Q.; Zheng, N.; Wang, R.; Visentin, S.; He, L.; Liu, S. Plant polyphenol-based injectable hydrogels: Advances and biomedical applications. Adv. Healthc. Mater. 2025, 14, 2500445. [Google Scholar] [CrossRef] [PubMed]
- Yadav, A.K.; Kenny, M.S.; Musa, E.N.; Walz, T.J.; Gładysiak, A.; Stylianou, K.C. Hydrolytic Stability of Metal–Organic Frameworks: An Experimental Study. Chem. Mater. 2025, 37, 9965–9984. [Google Scholar] [CrossRef]
- Vasudhevan, P.; Manikandan, V.; Iqbal, N.; Ullah, S.; Ma, H.; Singh, S.; Varshney, D.; Pu, S. Metal-Organic Frameworks for Wastewater Remediation: Sustainable Synthesis, Properties, and Applications. Chem. Rec. 2025, 25, e202500076. [Google Scholar] [CrossRef]
- Patil, P.D.; Suvarna, M.; Gargate, N.; Tiwary, A.; Palresha, D.; Tiwari, M.S.; Kagale, S.; Bhange, V.P.; Nadar, S. Recent advances in carbonous metal–organic frameworks (carbon-MOFs): Synthesis and environmental application. J. Ind. Eng. Chem. 2025, 153, 172–199. [Google Scholar] [CrossRef]
- Yu, W.; Li, A.; Lu, Z.; Jiang, H.; Jiang, Y.; Ni, H.; Wang, Y.; Chen, L. Coordination bond regulation in metal–organic framework composites: From precise synthesis to advanced catalysis and emerging adsorption/purification applications. Sep. Purif. Technol. 2025, 387, 136625. [Google Scholar] [CrossRef]
- Makableh, Y.F.; Al-Abed, R.; Aljaiuossi, G. Stability and optical enhancement of perovskite materials by nanocomposite PMMA sandwich structure in an open air environment. J. Mater. Sci. Mater. Electron. 2021, 32, 7106–7122. [Google Scholar] [CrossRef]
- Si, G.R.; He, T.; Kong, X.J.; Xie, L.H.; Li, J.R. Stable Metal–Organic Frameworks for Air and Water Pollution Control. Acc. Mater. Res. 2025, 6, 1006–1019. [Google Scholar] [CrossRef]
- Orellana, W. Ligand-dependent stability and ORR/OER activity of single-layer metal-organic frameworks. Electrochim. Acta 2026, 558, 148589. [Google Scholar] [CrossRef]
- Tang, H.; Chen, C.; Chen, Z.; Li, Z.; Guo, M.; Wang, Z.; Shao, Z.; Liu, Z.; Shen, Y.; Yue, Z.; et al. Phase and microstructure stability of Ba-containing proton conducting electrolyte materials under reducing environment of protonic ceramic cells-BaCeO3 and BaZrO3. Ceram. Int. 2025, 51, 63626–63638. [Google Scholar] [CrossRef]
- Seibel, F.I.; Brião, V.B.; Graciola, S.; Pontié, M.; Hannachi, A.; Shabani, M.; Alyoun, E.M.; Shaheed, M.H. Routes for reuse of aged pressure-driven membranes: Current status and future perspectives. Desalination 2025, 622, 119690. [Google Scholar] [CrossRef]
- Alcarazo, M. Dibenzothiophenium Salts: Practical Alternatives to Hypervalent I (III)-Based Reagents. Acc. Chem. Res. 2025, 58, 635–646. [Google Scholar] [CrossRef]
- Cowie, B.E.; Porley, V.; Robertson, N. Solar disinfection (SODIS) provides a much underexploited opportunity for researchers in photocatalytic water treatment (PWT). ACS Catal. 2020, 10, 11779–11782. [Google Scholar] [CrossRef]
- Pratsinis, S.E. Aerosol-based technologies in nanoscale manufacturing: From functional materials to devices through core chemical engineering. AIChE J. 2010, 56, 3028–3035. [Google Scholar] [CrossRef]
- Ranjan, R. Development of Complex Generics and Similar Biological Products: An Industrial Perspective of Reverse Engineering. AAPS PharmSciTech 2025, 26, 95. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Han, N.; Son, H.; Kim, S.J.; Kweon, S. Functional Peptide-Based Biomaterials for Pharmaceutical Application: Sequences, Mechanisms, and Optimization Strategies. J. Funct. Biomater. 2026, 17, 37. [Google Scholar] [CrossRef]
- Shrestha, S.; Bhattacharya, S. Versatile use of nanosponge in the pharmaceutical arena: A mini-review. Recent Pat. Nanotechnol. 2020, 14, 351–359. [Google Scholar] [CrossRef]
- Chen, W.L.; Wagner, J.; Heugel, N.; Sugar, J.; Lee, Y.W.; Conant, L.; Malloy, M.; Heffernan, J.; Quirk, B.; Zinos, A.; et al. Functional near-infrared spectroscopy and its clinical application in the field of neuroscience: Advances and future directions. Front. Neurosci. 2020, 14, 540710. [Google Scholar] [CrossRef]
- Qian, H.; Liu, J.; Wang, X.; Pei, W.; Fu, C.; Ma, M.; Huang, C. The state-of-the-art application of functional bacterial cellulose-based materials in biomedical fields. Carbohydr. Polym. 2023, 300, 120252. [Google Scholar] [CrossRef]
- Liu, X.; Qiao, Y.; Chen, H. Progress and challenges in the application of nanomaterials in the treatment of gastrointestinal tumors: From basic research to clinical translation. World J. Cancer Res. 2025, 15, 115. [Google Scholar] [CrossRef]
- Nahla, U.A.; Suleman, M.; Khan, I.; Saddique, M.N. Translating nanorobotic oncology: Promise, progress, and persistent barriers. Ann. Med. Surg. 2026, 88, 1136–1137. [Google Scholar] [CrossRef]
- Li, X.; Fu, L.; Chen, F.; Lv, Y.; Zhang, R.; Zhao, S.; Karimi-Maleh, H. Cyclodextrin-based architectures for electrochemical sensing: From molecular recognition to functional hybrids. Anal. Methods 2025, 17, 4300–4320. [Google Scholar] [CrossRef]
- Kumeria, T. Advances on porous nanomaterials for biomedical application (drug delivery, sensing, and tissue engineering). ACS Biomater. Sci. Eng. 2022, 8, 4025–4027. [Google Scholar] [CrossRef] [PubMed]
- Maji, T.K. Editorial for forum on applied supramolecular materials. ACS Appl. Mater. Interfaces 2023, 15, 25079–25081. [Google Scholar] [CrossRef] [PubMed]
- Parray, A.H.; Sohal, N.; Singh, P.; Kolte, J. Green synthesis of carbon quantum dots from biomass: A sustainable approach to nanotechnology. Biomass Bioenergy 2026, 204, 108447. [Google Scholar] [CrossRef]
- Ashrafi, G.; Nasrollahzadeh, M.; Jaleh, B.; Sajjadi, M.; Ghafuri, H. Biowaste-and nature-derived (nano) materials: Biosynthesis, stability and environmental applications. Adv. Colloid Interface Sci. 2022, 301, 102599. [Google Scholar]
- Zhang, S.; Cui, C.; Huang, S.; Wang, Z.; Wu, S. Application and mechanisms of biochar-immobilized enzymes in environmental remediation: A review. Biochar 2026, 8, 4. [Google Scholar] [CrossRef]
- Zhang, X.; Yan, M.; Chen, P.; Li, J.; Li, Y.; Li, H.; Liu, X.; Chen, Z.; Yang, H.; Wang, S.; et al. Emerging MOFs, COFs, and their derivatives for energy and environmental applications. Innovation 2025, 6, 100778. [Google Scholar] [PubMed]
- Bag, P.P.; Wang, X.S.; Cao, R. Microwave-assisted large scale synthesis of lanthanide metal–organic frameworks (Ln-MOFs), having a preferred conformation and photoluminescence properties. Dalton Trans. 2015, 44, 11954–11962. [Google Scholar] [CrossRef]
- Chen, T.; Xu, L.; Wei, S.; Tang, X.; Chen, H. Enhanced ammonia-rich solution production and electrode separation using magnetic nickel-loaded carbon black in flow-electrode electrochemical deionization (FEED). Desalination 2022, 544, 116152. [Google Scholar] [CrossRef]
- Ikram, H.; Al Rashid, A.; Koç, M. Additive manufacturing of smart polymeric composites: Literature review and future perspectives. Polym. Compos. 2022, 43, 6355–6380. [Google Scholar] [CrossRef]
- Zheng, Y. Artificial intelligence and machine learning for materials. Curr. Opin. Solid State Mater. Sci. 2025, 34, 101202. [Google Scholar] [CrossRef]
- Pan, X.; Xie, Y.; Li, C.; He, Y.; Zhang, Y.; Wang, Y.; Li, Z.; Peng, P.; Wang, J. Convergence of Computational Materials Science and AI for Next-Generation Energy Storage Materials. J. Electron. Mater. 2026, 55, 45–114. [Google Scholar] [CrossRef]
- Zhou, B.; Li, X.; Pan, Y.; He, B.; Gao, B. Artificial intelligence-assisted next-generation biomaterials: From design and preparation to medical applications. Colloids Surf. B Biointerfaces 2025, 255, 114970. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Song, P.; Li, G.; Han, Y.; Ren, X.; Bai, L.; Su, J. AI energized hydrogel design, optimization and application in biomedicine. Mater. Today Bio 2024, 25, 101014. [Google Scholar] [CrossRef]
- Osaro, E.; Karpinski, N.; Alornyo, S.; Ighalo, J.O. Artificial intelligence in materials science and chemistry: Past, present and future trajectories. Mater. Today Chem. 2025, 49, 103115. [Google Scholar] [CrossRef]
- Gao, L.; Zhang, Q.; Gu, M. Femtosecond laser micro/nano processing: From fundamental to applications. Int. J. Extrem. Manuf. 2025, 7, 022010. [Google Scholar]
- Duarte, L.C.; Figueredo, F.; Chagas, C.L.S.; Cortón, E.; Coltro, W.K.T. A review of the recent achievements and future trends on 3D printed microfluidic devices for bioanalytical applications. Anal. Chim. Acta 2024, 1299, 342429. [Google Scholar] [CrossRef]
- Kumar, R.; Kehr, N.S. 3D-printable oxygen-and drug-carrying nanocomposite hydrogels for enhanced cell viability. Nanomaterials 2022, 12, 1304. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.