Abstract
In this study, a novel silver(I) complex [Ag(HL1)2]NO3 (AgHL1) with coumarin derivative (3E)-3-(1-{[(pyridin-2-yl)methyl]amino}ethylidene)-3,4-dihydro-2H-benzopyran-2,4-dione (HL1) was prepared. The compounds HL1 and AgHL1 were characterized by IR and NMR spectroscopy, elemental analysis, and single crystal X-ray structural analysis. Specifically, the single crystal X-ray analysis determined the structures of both compounds HL1 and AgHL1 in their solid state, while NMR spectroscopy was used for structural determination in a solution. The HL1 proved to be a monodentate ligand and is coordinated to the Ag(I) atom through a nitrogen atom from the 2-picolylamine fragment. In the complex AgHL1, two molecules of neutral HL1 are coordinated forming a nearly linear N-Ag-N arrangement. An uncoordinated nitrate anion balances the positive charge of the complex cation. NMR spectroscopy also confirmed the stability of AgHL1 in DMSO-d6 for 3 days. In vitro cytotoxicity of HL1 and AgHL1 was performed over two cancerous cell lines A549 and HT-29 and their selectivity was verified on a healthy CCD-18Co cell line. AgHL1 exhibited low anticancer nonselective activity while the ligand was inactive. Also, the complex shows better antimicrobial activity than the positive controls on the Pseudomonas aeruginosa standard and clinical strain as well as on the tested molds.
1. Introduction
Metal-based drugs have been successfully used in the treatment of various diseases [1]. The discovery of cisplatin represented a milestone in the treatment of cancer and led to a deeper investigation of the therapeutic potential of metallodrugs. Nowadays, platinum compounds are effective in the treatment of various types of cancer [2]. Ferrochloroquine is used as an antimalarial agent [3], gold compounds treat rheumatoid arthritis, bismuth is known for antiulcer drugs [4], and compounds of gadolinium are used as MRI contrast agents [5]. Silver is one of the metal ions that are used clinically, too. The use of silver as an antimicrobial agent has been known since 1000 BC [6]. Silver nitrate and silver sulfadiazine were widely used in the treatment of wounds [7,8]. Nowadays, silver sulfadiazine is the most frequently used prophylactic agent in burn patients. It exhibits in vitro activity against microorganisms including S. aureus, E. coli, Klebsiella species, Pseudomonas aeruginosa, Proteus species, and Candida albicans [9]. However, there are some limitations in the usage of these compounds. The effect of these drugs is not long-term, so there is a risk of reinfection of the wound. Also, the development of resistance of some organisms is one of the limitations [10]. In addition to antimicrobial activity, silver complexes play an important role as antitumor agents with various mechanisms including DNA binding and cleavage, topoisomerase enzyme inhibition, generation of ROS, and induction of apoptosis [11].
To overcome problems like resistance, poor solubility, and severe side effects, a new strategy, seems to be, designing metal complexes with ligands that already have some biological activity. Therefore, N-heterocyclic ligands have been used in preparing anticancer agents [12]. Another widely used ligand is coumarin and its derivatives. Many derivatives of coumarin are being investigated as possible drugs with strong pharmacological activity, low toxicity, and side effects or with reduced resistance to these substances [13]. Out of the many studied effects, there can be mentioned anticoagulant [14], antimicrobial [15], anticancer [16], anti-inflammatory [17], or anti-HIV effect [18].
Thati et al. [19] investigated the antiproliferative effect of 4-hydroxy-3-nitro-coumarin and the mixed-ligand silver(I) complex of 4-oxy-3-nitro-coumarin-bis(phenanthroline) on four human-derived cell lines. The results showed a decrease in the proliferation of neoplastic cell lines of renal cancer A-498 and hepatic carcinoma HepG2, while non-neoplastic cell line Chang appeared to be less sensitive to the effect of the complex. Furthermore, regarding the results of IC50 values, the investigated complex was almost four times more potent than cisplatin, using HepG2 cell line. In another study, Mujahid et al. [20] prepared a series of silver(I) complexes with coumarin-based ligands and one of their phenanthroline adducts. The cytotoxicity of compounds was also studied against HepG2 and A-498 cancer cell lines. Tests showed that complexes were more cytotoxic than clinically used chemotherapeutic mitoxantrone. Also, it seems that complexes have quite different mechanisms of action to many metal-based therapeutics, due to excellent superoxide dismutase activity instead of interaction with DNA.
Considering the above-stated information, the main objective of the present manuscript is to investigate the antimicrobial and anticancer activity of a novel silver(I) complex [Ag(HL1)2]NO3 (AgHL1) with the ligand (3E)-3-(1-{[(pyridin-2-yl)methyl]amino}ethylidene)-3,4-dihydro-2H-benzopyran-2,4-dione (HL1). The compound HL1 has been previously synthesized by Brawley et al. [21] who studied its myosin inhibiting activity; however, its antimicrobial and anticancer properties as well as its crystal structure remain unexplored. Furthermore, our study compares the biological activities of the ligand HL1 itself with that of the complex AgHL1.
Given that the ligand has been synthesized before, our subsequent research phases focused on preparing halogen-modified HL1 (F, Cl, and Br) and the formation of complexes with different metal ions with the aim of enabling an exploration of the correlation between structural modifications and the biological activity of the modified ligand HL1 and diverse metal-containing complexes. However, all our attempts to synthesize Ag complexes with these ligands failed.
Therefore, in this manuscript, we only expound on the synthesis of the complex AgHL1 and its characterization (structure and stability). The structure of HL1 and AgHL1 in the solid state is investigated by single crystal X-ray structure analysis. The NMR spectroscopy is used to determine the structure of the ligand HL1 and complex AgHL1 in the solution and in the investigation of the stability of the complex AgHL1.
Additionally, their anticancer activity was assessed using two cancerous cell lines: A549 (lung carcinoma) and HT-29 (colorectal carcinoma), and their selectivity was confirmed on a healthy cell line, CCD-18Co (normal colon tissue cell line). According to statistical data from 2020, colorectal and lung carcinoma were the predominant cancer types diagnosed in males in Slovakia. In females, colorectal carcinoma was the second most prevalent, superseded only by breast cancer. Consequently, the selection of cell lines for testing was informed by these statistical insights [22]. Moreover, it is widely acknowledged that silver(I) and its compounds possess antimicrobial activity [23]. Therefore, the antimicrobial potency of both compounds was tested against 15 microorganisms, encompassing bacteria and fungi, with results benchmarked against the standards doxycycline and fluconazole, respectively, and AgNO3.
2. Results and Discussion
2.1. Synthesis
In this work we prepared the novel silver(I) complex [Ag(HL1)2]NO3 (AgHL1) with coumarin derivative (3E)-3-(1-{[(pyridin-2-yl)methyl]amino}ethylidene)-3,4-dihydro-2H-benzopyran-2,4-dione (HL1). The ligand was synthesized through a two-step process. Initially, acetylation of 4-hydroxy-2H-chromen-2-one was carried out using acetic acid in the presence of phosphorus oxychloride [24]. Subsequently, a condensation reaction between 3-acetyl-4-hydroxy-2H-chromen-2-one and 2-picolylamine was taken in methanol to yield the final product, in accordance with the established literature [21,25]. The reaction was performed under reflux conditions, following which the reaction mixture was cooled in a refrigerator for approximately 5 h. A white solid product was then isolated by filtration and air dried. To obtain crystals for X-ray analysis, the ligand was recrystallized from methanol at 24 °C and the identity of crystalline and powder was proved by comparison of their IR spectra. The ligand was then fully characterized by NMR spectroscopy, elemental analysis, and X-ray analysis. The purity of the ligand (from the reflux) was checked by NMR spectroscopy, so the ligand was used in the synthesis of the complex without another purification.
The complex AgHL1 was synthesized by mixing an aqueous solution of AgNO3 and an ethanolic solution of HL1 in a molar ratio of 1:2. As the ligand is not completely soluble in ethanol at room temperature, the solution of the ligand was heated up to about 50 °C and stirred on a magnetic stirrer until complete dissolution of the ligand. Then, aqueous solution of the AgNO3 was added. The beaker was wrapped in aluminum foil to prevent light from entering. The reaction mixture was cooled and allowed to crystallize in the dark at a temperature of 24 °C for approximately 2 weeks to give the beige AgHL1 crystals. Crystals were stored in the dark at room temperature.
As mentioned in the introduction, halogen derivatives of the ligand HL1 were prepared [26]; however, the attempt to synthesize Ag complexes with these ligands failed.
2.2. IR and NMR Spectra
The IR spectra (Figure S5) of the ligand and the complex are similar and comprise stretching vibrations at 3000–2800 (C-H aromatic and aliphatic), 1610–1560 (C=C aromatic), and 1400–1300 cm−1 (C-O). However, there are some differences in the spectra. There is a slight shift up of the lactone C=O (coumarin) band from 1689 (HL1) to 1698 cm−1 (AgHL1). C-N vibrations of pyridine ring change from 1335 and 1301 cm−1 (HL1) to 1337, 1359 and 1287 cm−1 (AgHL1), indicating coordination through nitrogen atom originating in the pyridine ring of the ligand, which is confirmed by X-ray analysis. Also, pyridine ring deformation bands are slightly shifted from 779 and 753 cm−1 (HL1) to 776 and 751 cm−1 (AgHL1). In addition, in the IR spectrum of the complex there is a new band at 431 cm−1, which could represent Ag-N interaction.
The ligand HL1 and the complex AgHL1 were fully characterized by NMR spectroscopy. The 1H and 13C NMR (Figure S1) assignments were determined using splitting patterns, chemical shifts, and 2D NMR data from COSY, HSQC, and HMBC (Figure S2) spectra. The 1H and 13C NMR data for the ligand HL1 and the complex AgHL1 in DMSO-d6 are summarized in Table 1. DMSO-d6 was chosen as the solvent due to the poor solubility of HL1 and AgHL1 in non-coordinating solvents. The analysis of NMR data for both samples demonstrates a minimal effect of the coordinated Ag atom on the ligand’s proton and carbon chemical shifts [27] (Table 1). The interaction between the silver atom and the ligand may not have been sufficiently strong to induce significant alterations in the ligand’s electronic environment, as evidenced by cases where the NMR chemical shifts in ligands in complexes closely approximate those observed in their unbound state [28,29]. However, the X-ray crystal structure analysis confirmed the structure of the complex AgHL1. Furthermore, the 15N-HMBC NMR verified the coordination of N1′ atom to the central atom. The chemical shift for the N1′ atom is −71.8 ppm in the unbound ligand and −72.9 ppm in the AgHL1 complex (Figures S3 and S4).

Table 1.
1H and 13C NMR chemical shifts (δ, ppm) of the ligand HL1 and the complex AgHL1 together with structure of HL1 showing atom numbering used for NMR assignment.
Notably, in 1H NMR spectra of both the free ligand and the complex show a downfield shift for the proton of the NH11 group upon formation of an H-bond with the oxygen atom of the carbonyl group C4=O. This shift to higher frequency, indicative of H-bond formation, results from a decrease in diamagnetic shielding due to a reduced electron density near the hydrogen [30]. The formation of the H-bond was further confirmed by X-ray crystal structure analysis.
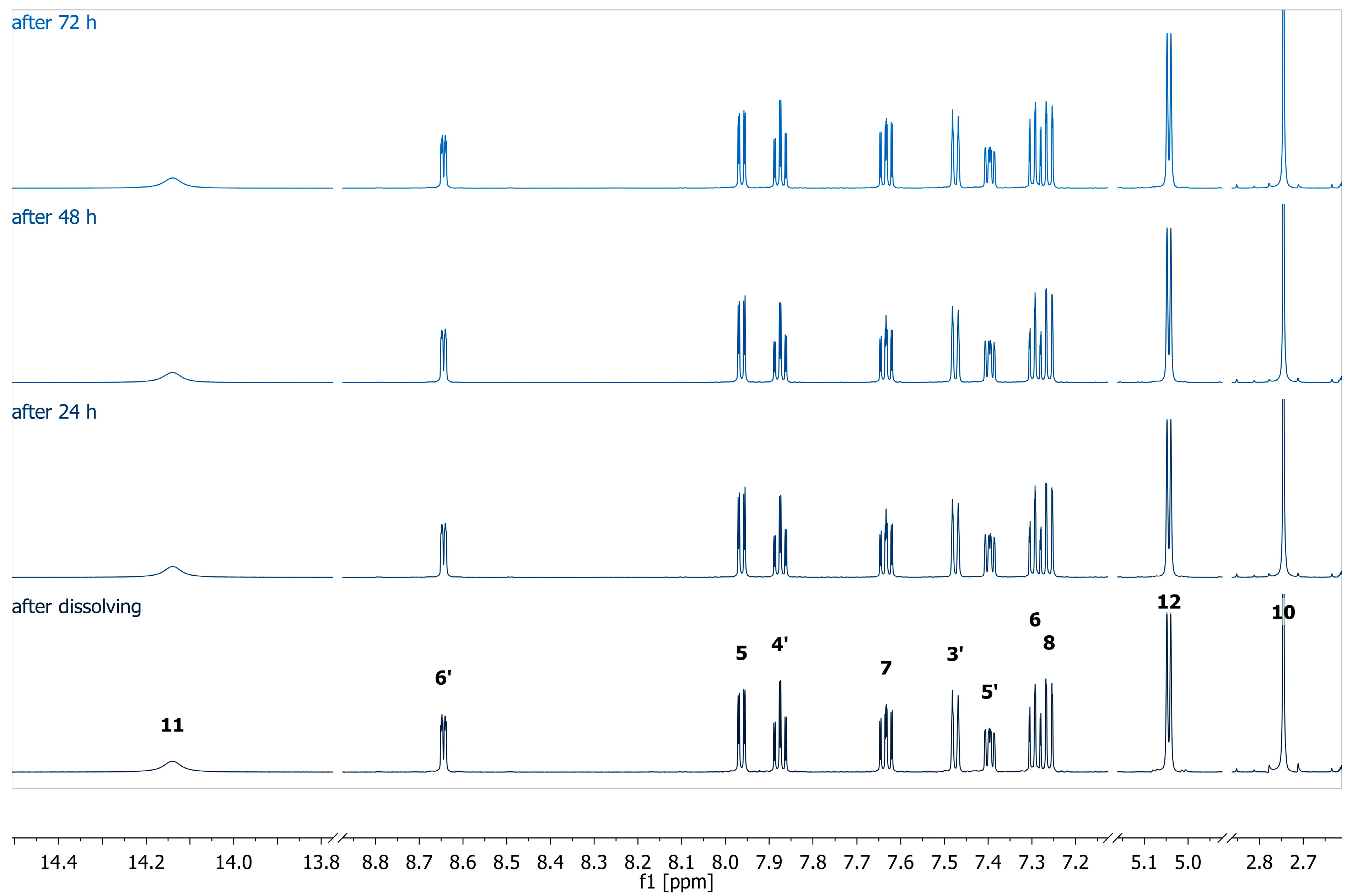
The stability of the complex was studied by measuring 1H NMR at different time intervals (Figure 1). The 1H NMR spectra exhibited sharp and well-resolved signals, indicating stable and consistent coordination of ligand donor atoms to the silver center at 298 K.
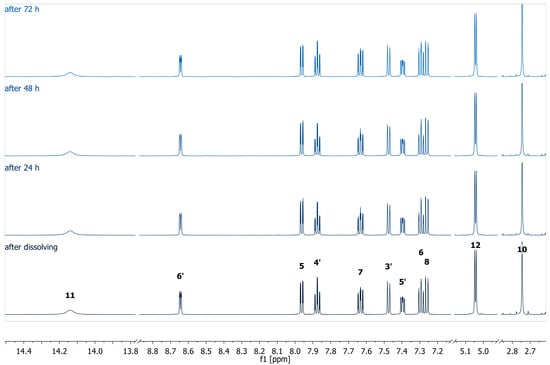
Figure 1.
1H (600 MHz, DMSO-d6) spectra of the complex AgHL1 measured at different time periods.
2.3. X-Ray Structure Analysis
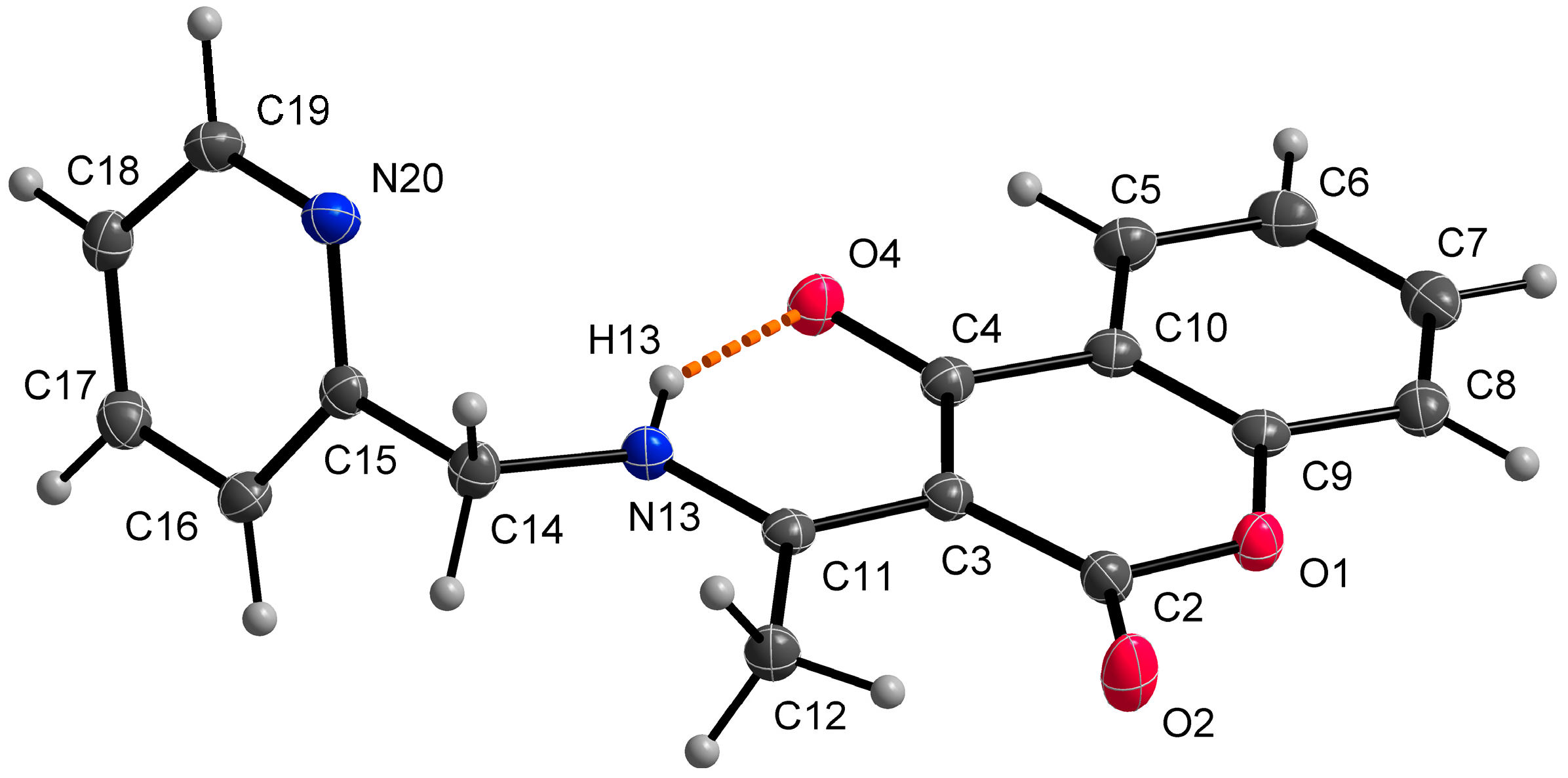
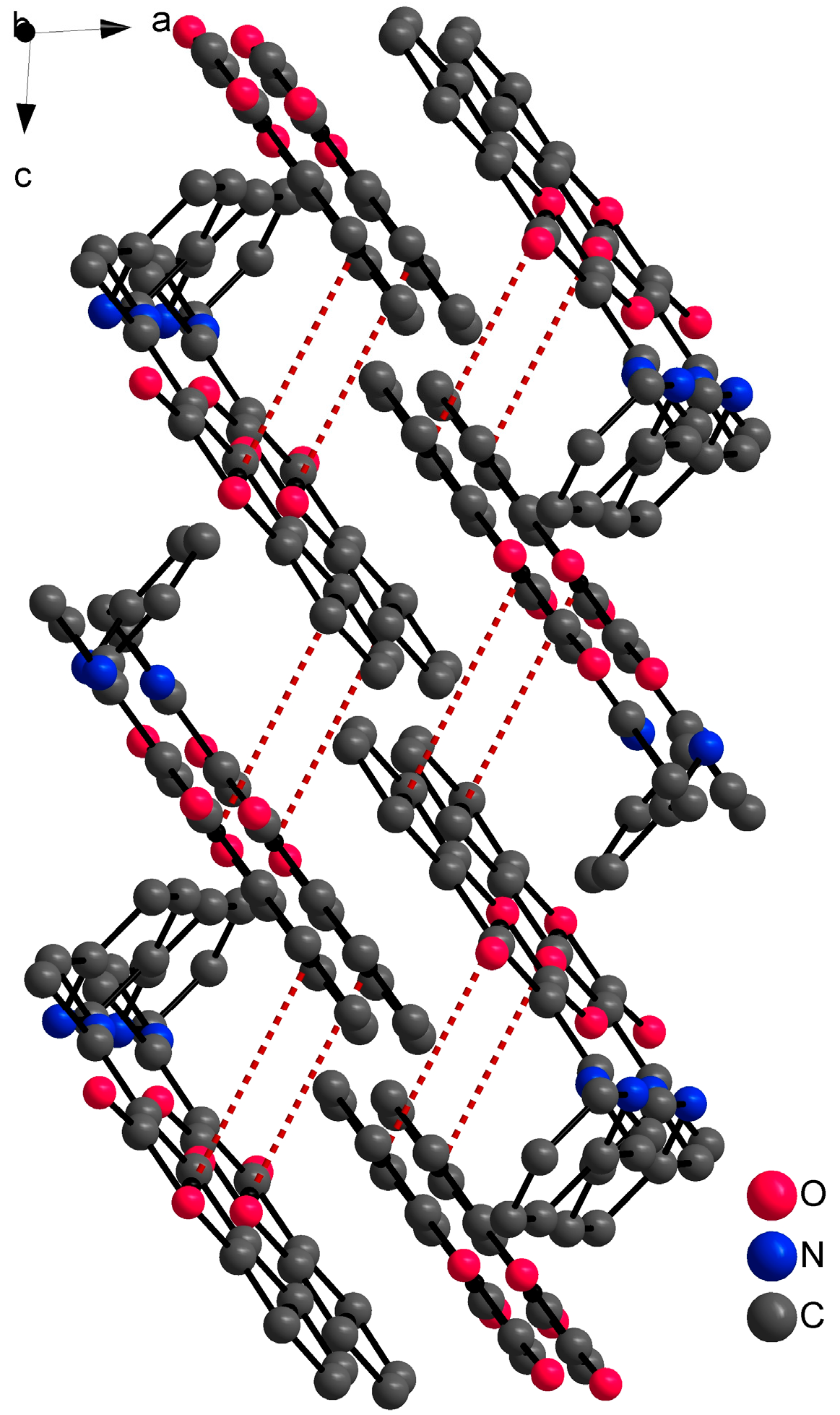
Single crystal X-ray structural analysis showed that ligand HL1 crystalizes in a monoclinic space group P21/c. The molecule of the ligand consists of bicyclic coumarin fragment (benzopyran-2,4-dione) and (pyridin-2-yl)methyl which are linked by aminoethylidene chain (Figure 2). The dihedral angle between coumarin and pyridine rings is 75.22(2)°. The presence of an intramolecular N13-H13⋯O4 hydrogen bond, with H13⋯O4 = 1.74 Å, N13⋯O4 = 2.5372(13) Å and N13-H13⋯O4 = 147°, causes the formation of a six-membered ring and the molecule is in keto-amine tautomeric form. Due to the formation of O4=C4-C3=C11-N13-H13 conjugated bond ring system, the flattening of the C4-C3 (1.4392(17) Å) and the C3=C11 (1.4413(17) Å) (Table 2) bonds is observed, although the bonds are formally single and double, respectively. The hydrogen bond mentioned above can be classified as a resonance-assisted hydrogen bond caused by π-electron delocalization [31]. Benzopyran-2,4-dione rings are connected by π-π interactions while a 3D structure is formed (Figure 3). The distance between centroids of the benzopyran-2,4-dione ring is 3.9407(7) Å and the angle γ between the vector joining the two centroids and the normal to the ring is 26.4°.
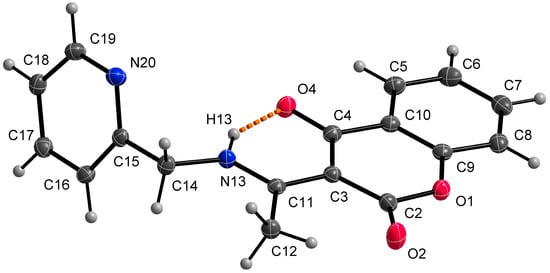
Figure 2.
Molecular structure of HL1, displacement ellipsoids are drawn at 50% probability. The orange dashed line describes a hydrogen bond.

Table 2.
Selected bond lengths and angles in HL1 [Å, °].
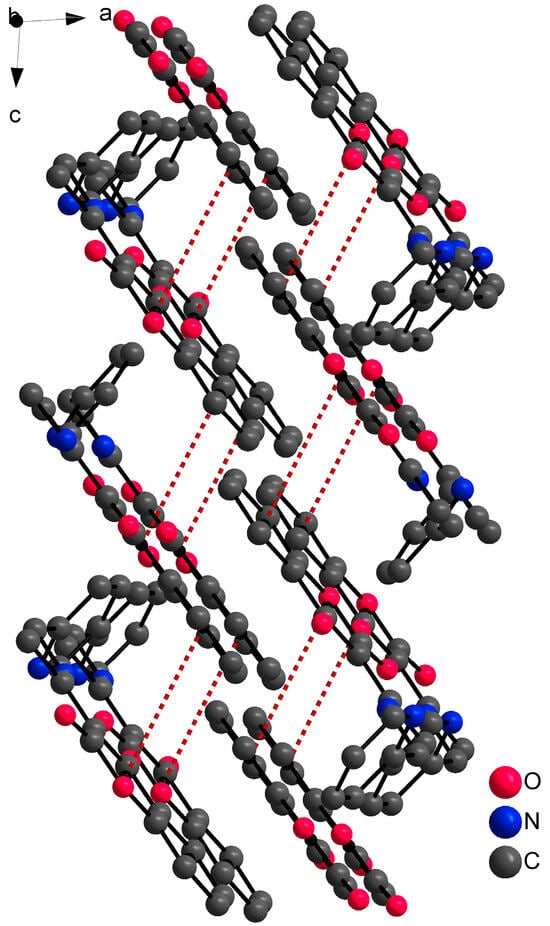
Figure 3.
Part of the 3D structure of HL1 viewed along the ac plane with π-π interactions (red dashed lines). Hydrogen atoms are omitted because of clarity.
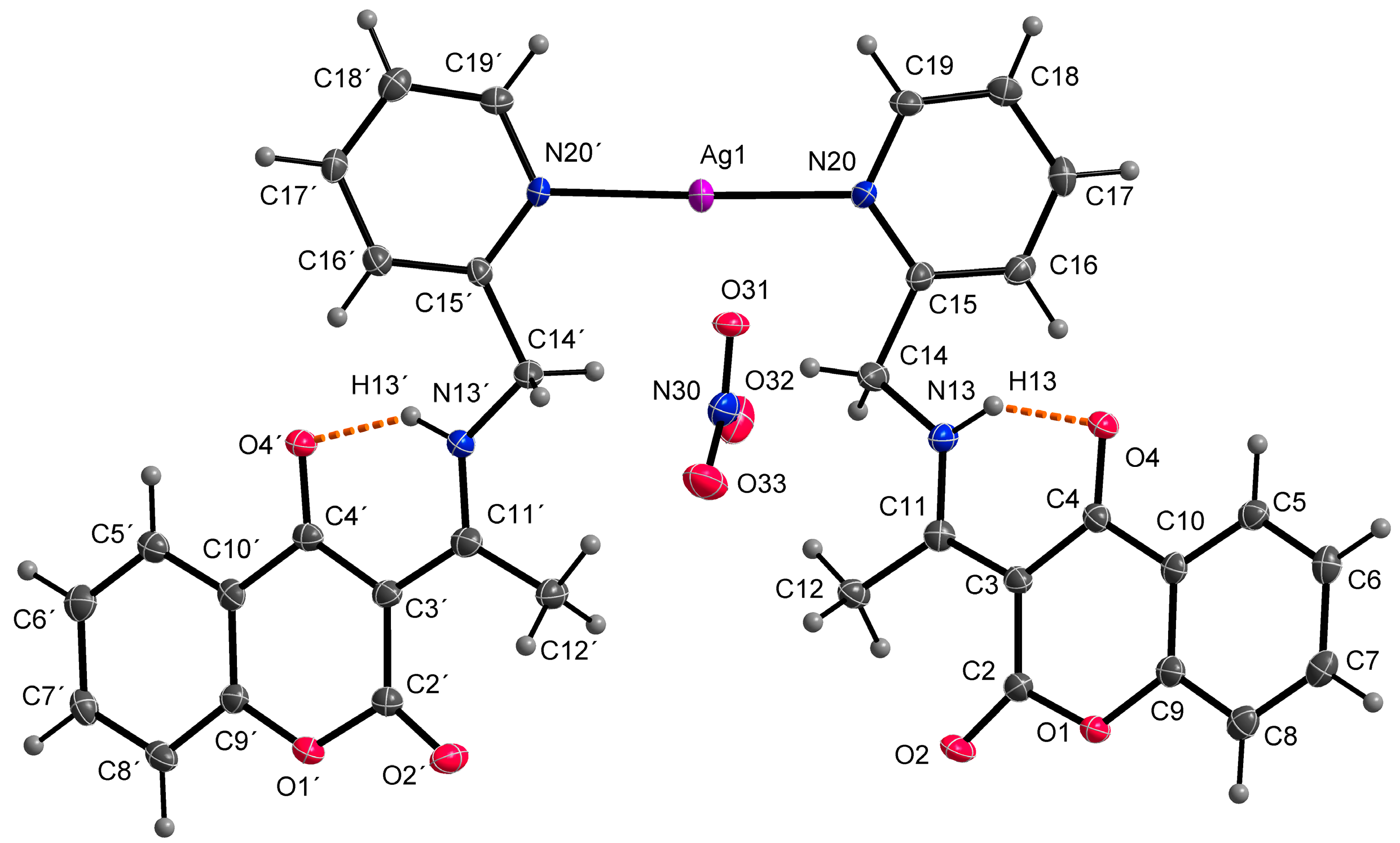
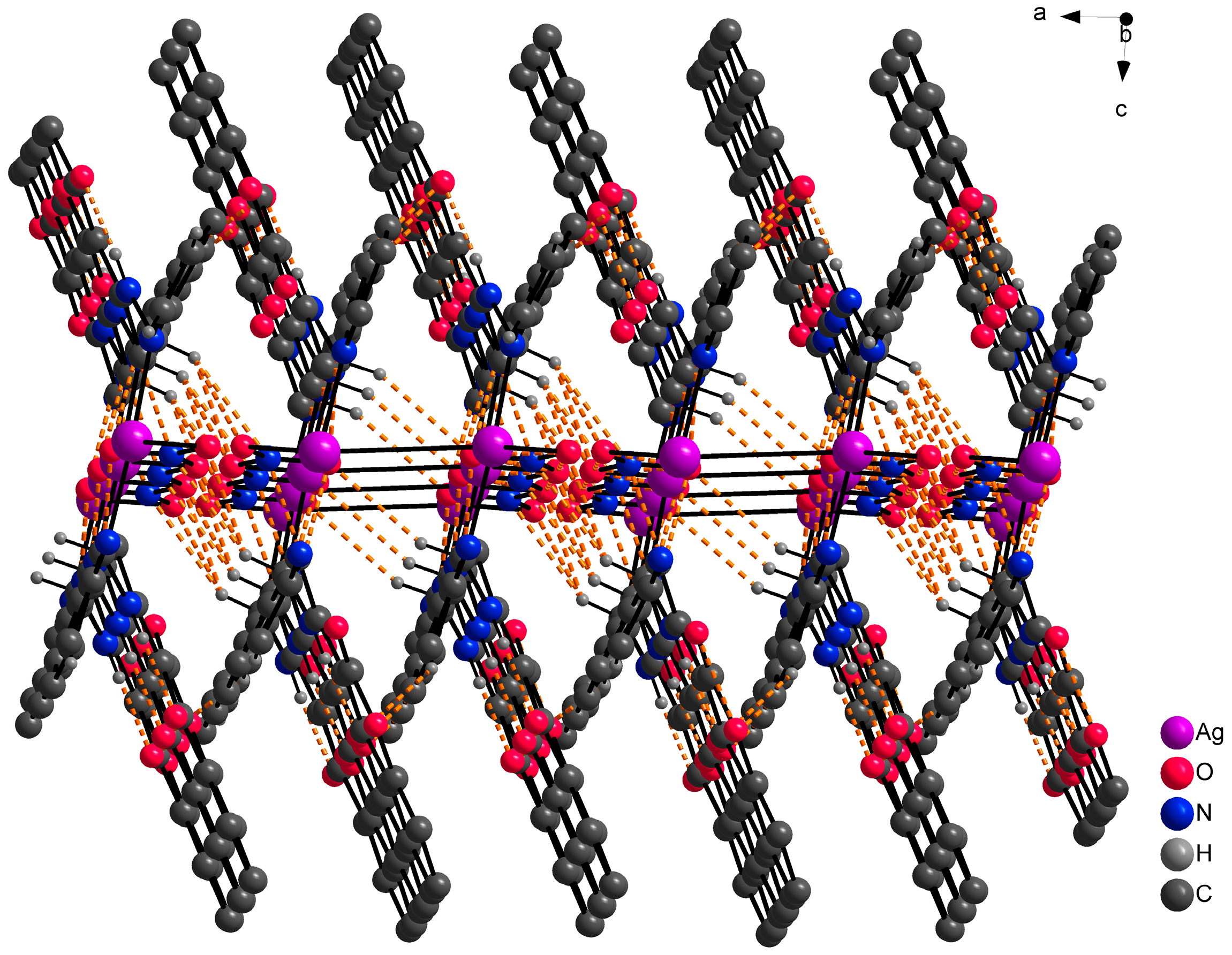
At the outset, it was hypothesized that the ligand HL1 would demonstrate a tridentate bonding mode, involving donor atoms N13, N20, and O4, analogous to complexes with similar coumarin derivatives as previously documented [32,33]. However, the single crystal X-ray analysis of the AgHL1 complex revealed that this assumption was incorrect. In subsequent discussion, we elaborate on the binding mode of the ligand HL1 to the central atom and the structure of the complex AgHL1. The complex crystallizes in a triclinic space group P-1. Two molecules of HL1 are bound to the central Ag1 atom only via N20 (and N20’) atoms, with an average Ag1-N20 bond distance of 2.170(4) Å and the N20-Ag1-N20’ bond angle being 176.69(17)° (Table 3), thus the ligand is monodentate with the donor N20 atom originating from the (pyridin-2-yl)methyl fragment. This aligns with the usual range for Ag-N(pyridyl) bonds, spanning from 1.869 to 2.987 Å [34]. Additionally, Wang et al. [35] reported a complex where the central silver atom is bonded to two nitrogen atoms derived from pyridyl groups. In that complex, the Ag-N bond length is 2.158(4) Å, closely paralleling our findings. However, the N-Ag-N angle in their case is perfectly linear at 180°. As a consequence, atom N13 in AgHL1 remains protonated and, thus, the ligand is electroneutral. The positive charge of the formed complex cation is balanced by the uncoordinated nitrate anion. The shape of the coordination polyhedron is linear (Figure 4). Selected bond lengths and angles are given in Table 3. Due to intermolecular hydrogen bonds and weak interactions (Table 4), the AgHL1 complex creates a 3D structure shown in Figure 5.

Table 3.
Selected bonds and angles in AgHL1 [Å, °].
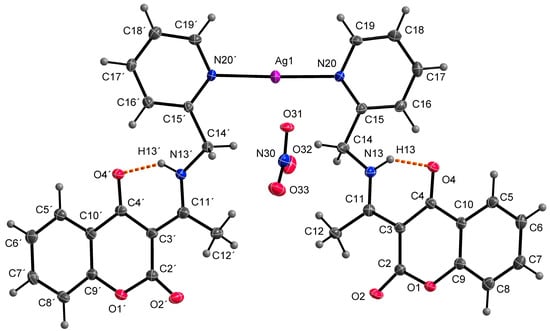
Figure 4.
Molecular structure of AgHL1, displacement ellipsoids are drawn at 50% probability. Orange dashed lines describe hydrogen bonds.

Table 4.
Hydrogen bonds in AgHL1 [Å, °].
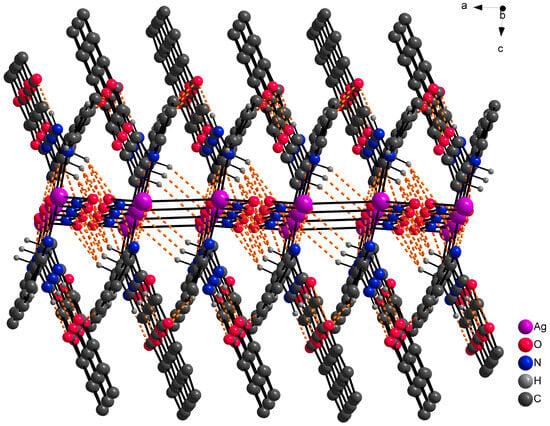
Figure 5.
Part of the 3D structure of AgHL1 viewed along the ac plane with hydrogen bond interactions (orange dashed lines). Hydrogen atoms not involved in hydrogen bond interactions are omitted because of clarity.
2.4. In Vitro Antitumor Activity
2.4.1. Effect of HL1 and AgHL1 on Metabolic Activity of A549, HT-29 and CCD-18Co Cell Lines and Determination of IC50 Values
To determine the potential inhibitory effect of HL1 and AgHL1 on the metabolic activity of A549 lung and HT-29 colorectal carcinoma cell lines, an MTT assay was performed 24 and 48 h after the exposure of the cells to tested compounds (1–100 μM) and 1% DMSO. Moreover, the metabolic activity of CCD-18Co healthy colon fibroblasts was also analyzed to verify the selectivity of tested complexes to cancer cells.
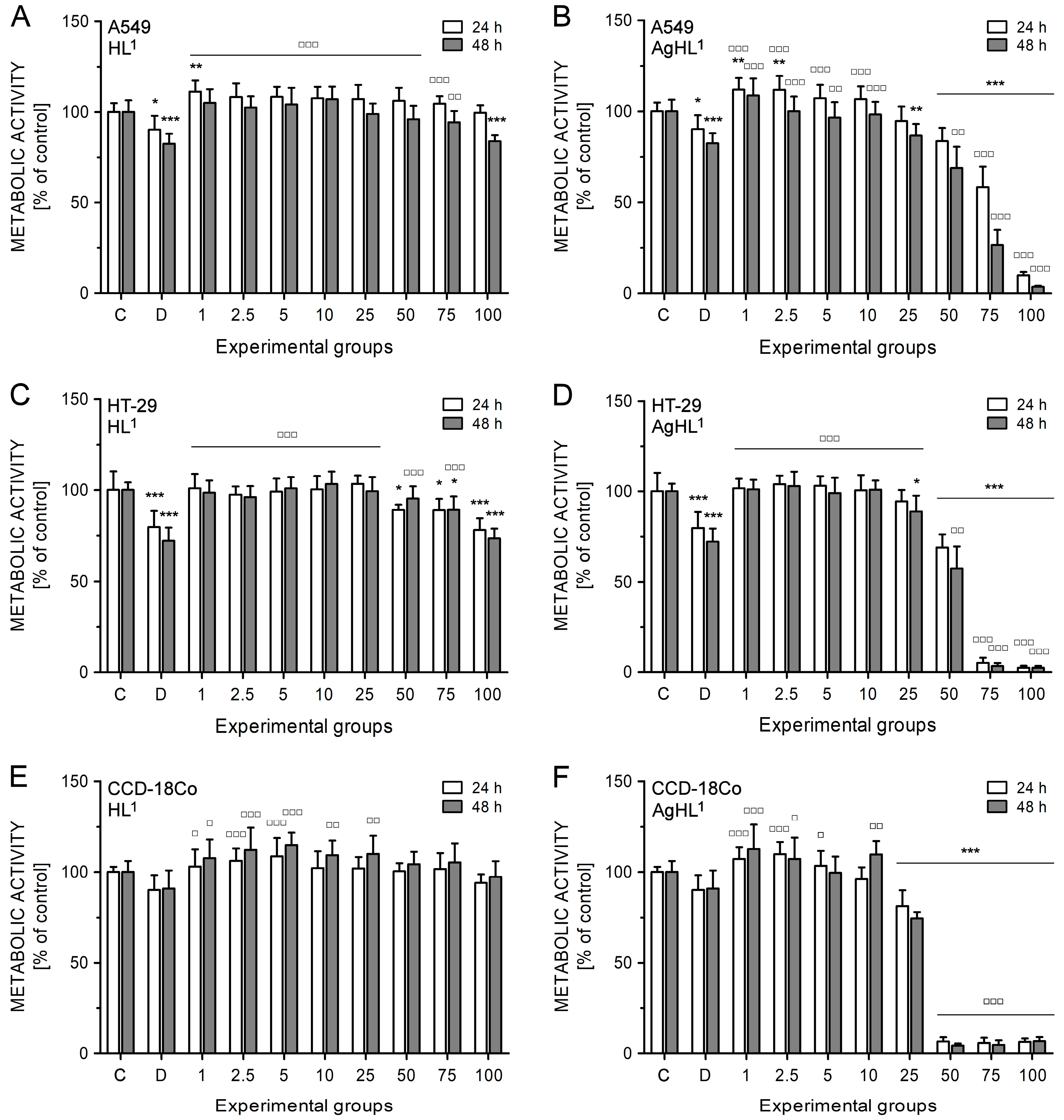
HL1 did not inhibit the metabolic activity of A549 (Figure 6A) and HT-29 cell lines (Figure 6C). A slight and significant decrease in metabolic activity was evident in the case of higher concentrations of HL1 (100 μM in A549 cells; 50, 75, and 100 μM in HT-29 cells); however, it was more likely caused by DMSO as a diluent of tested compounds. The drop of metabolic activity caused by 100 μM HL1 in A549 (83.77 ± 3.36% after 48 h) (Figure 6A) and HT-29 cell lines (78.12 ± 6.47% after 24 h; 73.43 ± 5.43% after 48 h) (Figure 6C) corresponds to the metabolic activity reduced by 1% DMSO (82.39 ± 5.53% in A549 cell line after 48 h; 79.71 ± 8.90% in HT-29 cell line after 24 h; 72.05 ± 7.38% in HT-29 cell line after 48 h). HL1 did not even inhibit the metabolic activity of CCD-18Co fibroblasts (Figure 6E).
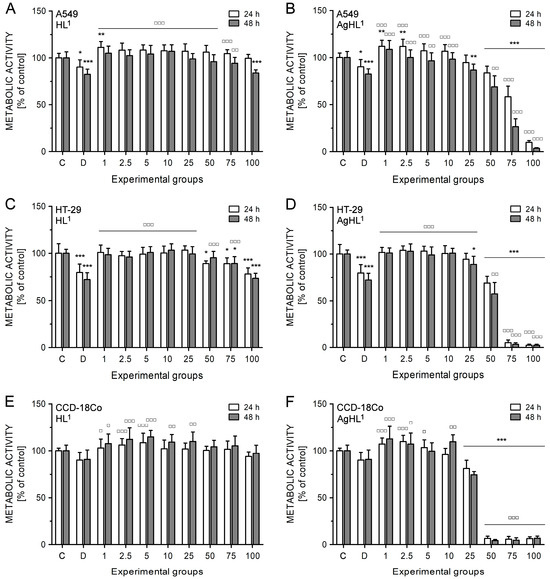
Figure 6.
The effect of HL1 (left (A,C,E)) and AgHL1 (right (B,D,F)) on metabolic activity of A549 (top (A,B)), HT-29 (middle (C,D)), and CCD-18Co cell lines (bottom (E,F)). The metabolic activity of the cells was estimated by MTT assay. The analyses were performed 24 and 48 h after the treatment of the cells with HL1 and AgHL1 at concentrations of 1–100 μM (all tested concentrations are shown on the x-axis in μM) and with 1% DMSO (marked as D on the x-axis). The results were evaluated as percentages of the untreated control (marked as C on the x-axis) and are expressed as the mean values ± SD of at least three independent experiments. The groups treated with 1% DMSO, HL1, or AgHL1 were compared with the untreated control (* p < 0.05, ** p < 0.01, *** p < 0.001) and the groups treated with HL1 or AgHL1 were also compared with the group treated with 1% DMSO (□ p < 0.05, □□ p < 0.01, □□□ p < 0.001).
On the other hand, AgHL1 showed a time- and dose-dependent inhibitory effect on the metabolic activity of both cancer cell lines (Figure 6B,D). In the case of A549 cell line, 1 and 2.5 μM AgHL1 slightly but significantly increased the metabolic activity after 24 h above the untreated control levels (>100%). However, significant inhibition in metabolic activity was observed in the experimental groups treated with 50 μM (83.59 ± 7.29% after 24 h; 68.88 ± 11.60% after 48 h), 75 μM (58.33 ± 11.32% after 24 h; 26.58 ± 8.36% after 48 h) and 100 μM AgHL1 (9.83 ± 1.90% after 24 h; 3.60 ± 0.59% after 48 h) (Figure 6B). Moreover, the inhibitory effect of AgHL1 against HT-29 cell line was even more pronounced (50 μM group: 68.89 ± 7.28% after 24 h and 57.45 ± 11.93% after 48 h; 75 μM group: 5.11 ± 2.83% after 24 h and 3.34 ± 1.72% after 48 h; 100 μM group: 2.43 ± 1.12% after 24 h and 2.45 ± 1.02% after 48 h) (Figure 6D). However, a strong inhibitory effect on the metabolic activity was also observed in the case of CCD-18Co fibroblasts (25 μM group: 81.24 ± 8.65% after 24 h and 74.56 ± 3.39% after 48 h; 50 μM group: 6.69 ± 2.32% after 24 h and 4.39 ± 1.12% after 48 h; 75 μM group: 5.80 ± 2.97% after 24 h and 4.82 ± 2.46% after 48 h; 100 μM group: 6.47 ± 1.86% after 24 h and 6.81 ± 2.35% after 48 h) (Figure 6F). The obtained results suggest no selectivity of AgHL1 towards cancer cell lines.
Based on the results of the MTT assay, IC50 values for HL1 and AgHL1 for both carcinoma cell lines were evaluated (Table 5). As HL1 was not effective against A549 and HT-29 cell lines, its IC50 values could not be defined from the tested range of concentrations (>100 μM). In the case of AgHL1, lower IC50 values were obtained for HT-29 cell line indicating slightly higher sensitivity of HT-29 colorectal carcinoma cell line to AgHL1 compared to A549 lung carcinoma cell line. Moreover, IC50 values for HL1 and AgHL1 were also determined in CCD-18Co healthy fibroblasts (Table 5) to verify the selective anticancer effect of tested complexes. HL1 had no inhibitory effect on CCD-18Co fibroblasts (IC50 > 100 μM); however, IC50 values for AgHL1 were the lowest in this cell line (32.44 ± 0.60 μM after 24 h and 29.09 ± 1.14 μM after 48 h). These results suggest stronger inhibitory effects of AgHL1 on CCD-18Co healthy fibroblasts than on HT-29 and A549 carcinoma cell lines.

Table 5.
Half maximal inhibitory concentrations (IC50) of HL1 and AgHL1 in A549, HT-29, and CCD-18Co cell lines. Estimated IC50 values (μM) were derived from metabolic activity values of at least three independent experiments by OriginPro 8.5.0 SR1.
The obtained results indicate that, unlike HL1, AgHL1 has inhibitory potential towards the metabolic activity of cancer cell lines. However, the decrease in metabolic activity of the cells may either be the consequence of the cytotoxic and/or cytostatic action of the tested compound on the cancer cells or may just reflect the inhibitory potential of the compound towards cellular metabolism. Thus, more specific analyses will be needed to elucidate the effect of AgHL1 on A549 and HT-29 cell lines. On the other hand, AgHL1 also showed an inhibitory effect on CCD-18Co healthy fibroblasts, indicating no selectivity of this complex towards cancer cell lines.
Additionally, IC50 values for AgNO3 were assessed. As shown in Table 5, the complex AgHL1 exhibited slightly higher activity than AgNO3 on the A549 cell line. Conversely, on the HT-29 cell line, AgNO3 demonstrated greater cytotoxicity compared to the complex AgHL1. This increased effectiveness can be attributed to the rapid and nonspecific release of Ag+ ions from AgNO3. Owing to its high aqueous solubility, silver nitrate undergoes rapid dissociation, leading to the immediate release of Ag+ ions. These cations exhibit broad reactivity, engaging in non-specific interactions with diverse biomolecular targets, thereby contributing to pronounced cytotoxicity [36]. On the other hand, the formation of Ag–N bonds in complexes results in a more controlled and gradual release of Ag+, enhancing their interaction with cellular targets such as membranes, enzymes, and nucleic acids [37].
2.4.2. Effect of HL1 and AgHL1 on Proliferation of A549 and HT-29 Carcinoma Cell Lines
To verify the potential cytostatic action of AgHL1, a real-time analysis of cell proliferation was performed. The impact of HL1 and AgHL1 on the proliferation of A549 and HT-29 cell lines was determined by the monitoring of the confluence of seeded and treated cells using the IncuCyteTM ZOOM system (IncuCyteTM ZOOM 2016B) every 2 h for a total duration of 72 h.
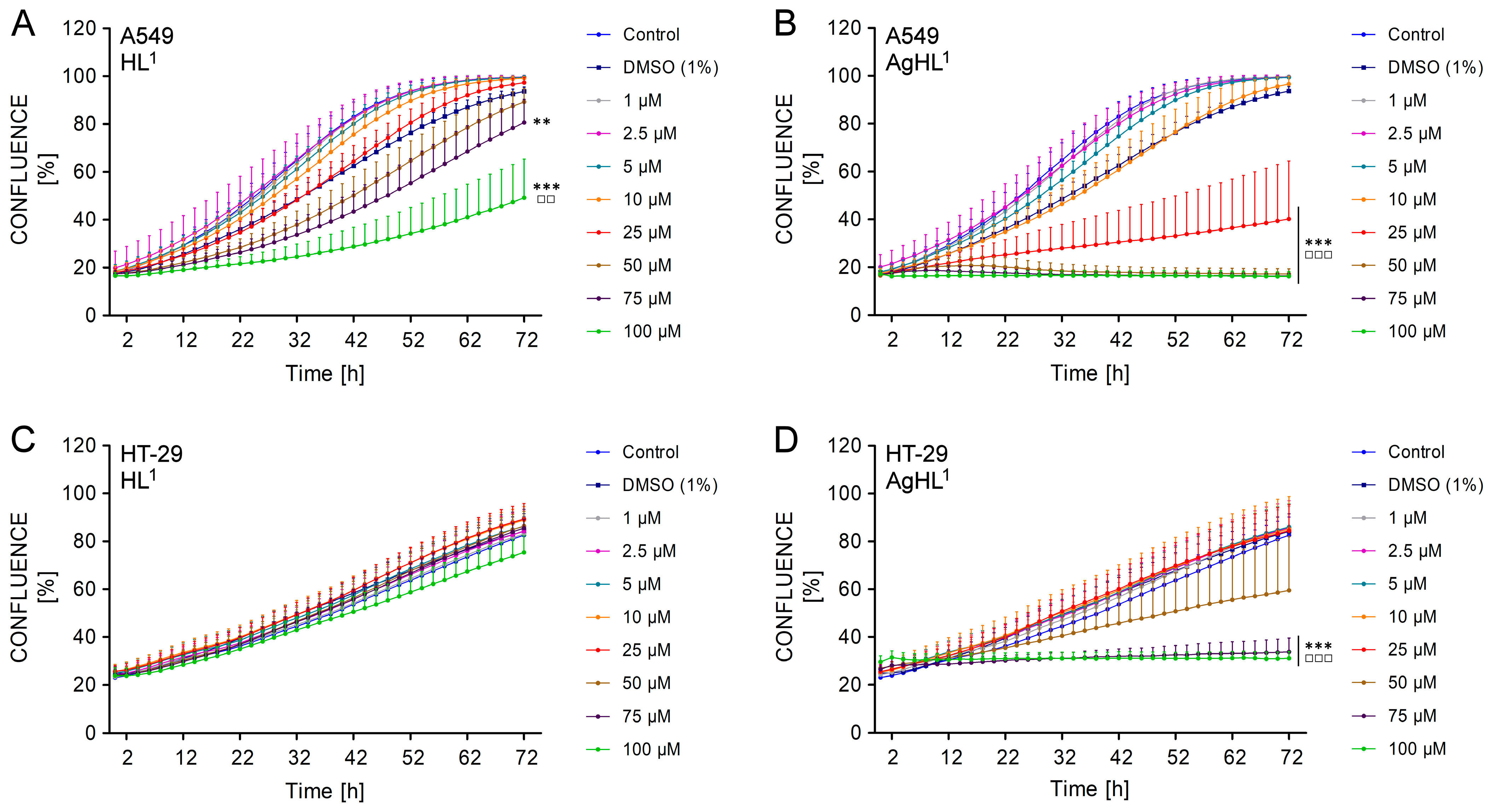
The obtained results are in agreement with the results of the MTT assay. HL1 did not inhibit the proliferation of the HT-29 cell line, as the confluence was continuously increasing in all treated experimental groups equally to untreated control throughout the whole duration of monitoring (from approx. 24% at 0 h to approx. 80% in all groups at 72 h) (Figure 7C). In the case of the A549 cell line, 75 μM and 100 μM HL1 slightly but significantly delayed the rate of cell growth, as the percentage of confluence was lower compared to the untreated control group (80.65 ± 9.14% in 75 μM group and 49.18 ± 15.25% in 100 μM group vs. 99.59 ± 0.51% in control) (Figure 7A). However, an increasing percentage of confluence in time suggests that A549 cell line maintained their proliferative activity even after HL1 treatment.
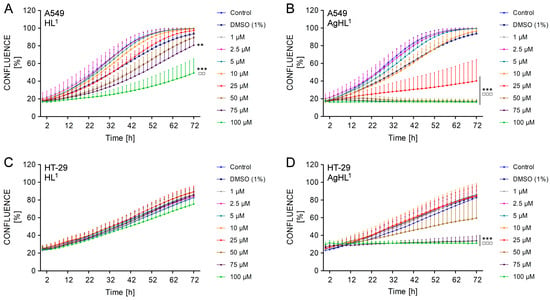
Figure 7.
The effect of HL1 (left (A,C)) and AgHL1 (right (B,D)) on proliferation of A549 (top (A,B)) and HT-29 cell lines (bottom (C,D)). The proliferation of the cells is presented as a percentage of confluence (the percentage of the cell-occupied area of the culture well surface) and was monitored by IncuCyteTM ZOOM (Essen Bioscience) every 2 h after the treatment of the cells with HL1 and AgHL1 (1–100 μM) and with 1% DMSO for a total duration of 72 h. The results are expressed as the mean values ± SD of three independent experiments. The groups treated with 1% DMSO, HL1 or AgHL1 were compared with the untreated control (** p < 0.01, *** p < 0.001) and the groups treated with HL1 or AgHL1 were also compared with the group treated with 1% DMSO (□□ p < 0.01; □□□ p < 0.001).
On the other side, higher concentrations of AgHL1 inhibited the proliferation of A549 and HT-29 carcinoma cell lines, as the confluence of both cell lines was not increasing in time. The confluence of HT-29 cell line treated with 75 μM and 100 μM AgHL1 remained almost constant (26.52 ± 2.68% at 0 h vs. 33.80 ± 5.43% at 72 h in 75 μM group; 29.61 ± 2.40% at 0 h vs. 31.09 ± 2.69% at 72 h in 100 μM group) (Figure 7D). In the case of the A549 cell line, the confluence even slightly decreased after the treatment with 50, 75, and 100 μM AgHL1 below the percentage of confluence at 0 h (17.44 ± 1.71% at 0 h vs. 17.21 ± 2.00% at 72 h in 50 μM group; 17.10 ± 0.95% at 0 h vs. 16.32 ± 1.05% at 72 h in 75 μM group; 17.21 ± 1.59% at 0 h vs. 16.27 ± 1.87% at 72 h in 100 μM group) (Figure 7B). Moreover, 25 μM AgHL1 in the A549 cell line (Figure 7B) and 50 μM AgHL1 in the HT-29 cell line (Figure 7D) also significantly delayed the rate of cell proliferation, as the percentage of confluence was lower compared to the untreated control group.
The obtained results suggest that AgHL1 was more effective against A549 and HT-29 carcinoma cell lines than HL1. Unlike HL1, AgHL1 inhibited not only the metabolic activity but also the proliferation of A549 and HT-29 cell lines.
So far, the antitumor properties of Ag(I) complexes of coumarin derivatives have been poorly studied. However, some evidence of the antitumor effects of other metal complexes, especially palladium complexes with a coumarin-derived ligand, may indicate their potential use in the treatment of tumors. A palladium(II) complex of 3-(1-(2-hydroxyethylamino)ethylidene)chroman-2,4-dione was shown to be effective against L929 mouse fibrosarcoma, B16 mouse melanoma, and U251 human glioma cell lines [25]. Similarly, in another study, palladium(II) complex of 3-(1-(3-hydroxypropylamino)ethylidene)chroman-2,4-dione decreased the viability of B16 and U251 cell lines, while the ligand showed only moderate cytotoxic activity [32]. In both cases, the IC50 values of these complexes were lower than the corresponding IC50 values for cisplatin treatment, suggesting that the complexes could be therapeutically effective at low concentrations with overall lower systemic toxicity after administration to the patient. The cytotoxic activity of palladium(II) complex of 3-(1-aminoethylidene)-2H-chromene-2,4(3H)-dione prepared by Budzisz et al. [38] was also more potent than carboplatin treatment in HeLa (cervix epithelial carcinoma), K562 (chronic myelogenous leukemia), and A549 cell lines. However, none of these studies verified the selectivity of the complexes towards cancer cells. In the current study, the Ag(I) complex of coumarin derivative, AgHL1, inhibited not only the metabolic activity but also the proliferation of A549 and HT-29 cell lines. Compared with the above-mentioned complexes, the relatively high IC50 values for AgHL1 in A549 cells (79.34 μM at 24 h and 59.66 μM at 48 h) appear to be insufficient to compete the cytotoxic effect of the chemotherapeutic agent, mitoxantrone (37.2 μM at 24 h and 4.46 μM at 48 h), demonstrated in the same cancer cell line [39]. Conversely, in the case of HT-29 cell line, AgHL1 (IC50 values 55,81 µM at 24 h and 52,86 µM at 48 h) could be more potent than documented cisplatin treatment (IC50 values 181,1 µM at 24 h and 104,5 µM at 48 h) [40]. Despite the demonstrated cytotoxic and even cytostatic activity, AgHL1 also showed an inhibitory effect on CCD-18Co healthy fibroblasts, indicating no selectivity of this complex towards cancer cells.
2.5. In Vitro Antimicrobial Activity
The results of in vitro testing of antimicrobial activities of the synthesized ligand HL1, corresponding complex AgHL1, positive controls doxycycline and fluconazole are shown in Table 6. Since Ag+ ions are antimicrobially active, as stated below, experiments were also conducted using the AgNO3 control to assess AgHL1 efficacy. The intensity of antimicrobial action varied depending on the type of substances.

Table 6.
Antimicrobial activity of HL1 and AgHL1, and positive controls (MIC and MMC values for substances are given as µg∙mL−1).
The complex has significantly better activity than the ligand, nevertheless, the activity of AgNO3 is comparable to the activity of AgHL1. MIC values for the ligand were obtained from 250 to >1000 μg∙mL−1 and for the complex from 7.81 to 500 μg∙mL−1. There is no difference in the effect of the tested substances on Gram-positive and Gram-negative bacteria and fungi. The complex shows better activity than the positive controls doxycycline on the Pseudomonas aeruginosa standard and clinical strain as well as on the tested molds.
Similar results were also obtained by the authors of other studies where the Ag complexes were significantly more antimicrobial active in relation to ligands derived from coumarin [41,42,43,44,45,46,47].
Sahoo et al. gave an overview of the current knowledge about coumarin derivatives as promising antibacterial agents, including complexes with silver [48]. Silver complexes with phenathroline adducts exhibited good antibacterial activity [42]. The compound with coumarin-bearing naphthalene-benzimidazole was a good antibacterial agent against S. aureus and E. faecalis in comparison to positive controls [43]. Similarly, silver salts of butyl, pentyl, and hexyl substituted imidazolium of 6-methyl coumarin had shown good antibacterial action against E. coli and P. aeruginosa in comparison to the standard [44,45,46,47]. Also, a sol–gel matrix with Ag-coumarin complexes may provide non-toxic surfaces with antibacterial properties [49].
The minimum inhibitory concentrations (MICs) of silver ions were 0.5 μg·mL−1 against E. coli, and 1 μg·mL−1 against P. aeruginosa, and S. aureus [50]. In the case [51] MICs for silver ions were 1.69 μg·mL−1 against S. aureus, and 0.84 μg·mL−1 against P. aeruginosa. In contrast to these values the MIC and MBC of silver nanoparticles against S. aureus was found to be 625 μg·mL−1 [52].
It is believed that the antibacterial activity of silver-based complexes is strictly related to their solubility and stability in water, lipophilicity, redox ability, and release rate of silver ions [53]. Silver ions (Ag+) preferentially target the cell membrane, inducing bacteriolysis by inhibiting peptidoglycan elongation in Staphylococcus aureus and compromising the integrity of the outer membrane in Escherichia coli [54]. This process culminates in DNA damage and the subsequent loss of replication capabilities [55]. Staphylococci exhibit universal sensitivity to Ag+ and cannot create resistant strains, whereas E. coli rapidly acquires resistance through mutations that alter Ag+ efflux mechanisms and outer membrane porins [56]. The antimicrobial efficacy of Ag+ is influenced by interactions with other ions; magnesium ions (Mg2+) diminish its effectiveness by inhibiting cellular uptake of Ag+, whereas zinc ions (Zn2+) marginally enhance its activity by synergistically augmenting cell death and reducing ferrihydrite reduction [57]. Upon entry into a bacterial cell, silver accumulates, resulting in cell death. Such bacteria serve as an effective reservoir for prolonged release of silver and prolonged antimicrobial activity that can last up to 40 days [58], which further encourages its practical use.
3. Materials and Methods
3.1. Materials and Chemicals
Reagents were purchased from the following commercial sources and used as obtained: phosphorus oxychloride (Merck, Darmstadt, Germany, 99%), 4-hydroxycoumarin (4-hydroxy-2H-chromen-2-one) (Sigma Aldrich, Co., St. Louis, MO, USA, 98%), 2-picolylamine (Sigma Aldrich, Co., St. Louis, MO, USA, 99%), methanol (Centralchem, Bratislava, Slovakia, 99.5%), acetone (Centralchem, Bratislava, Slovakia, 99.5%), silver(I) nitrate (Sigma Aldrich, Co., St. Louis, MO, USA, 99%), and acetic acid (Honeywell, Seelze, Germany, 99.8%).
3.2. Syntheses
3.2.1. Synthesis of HL1
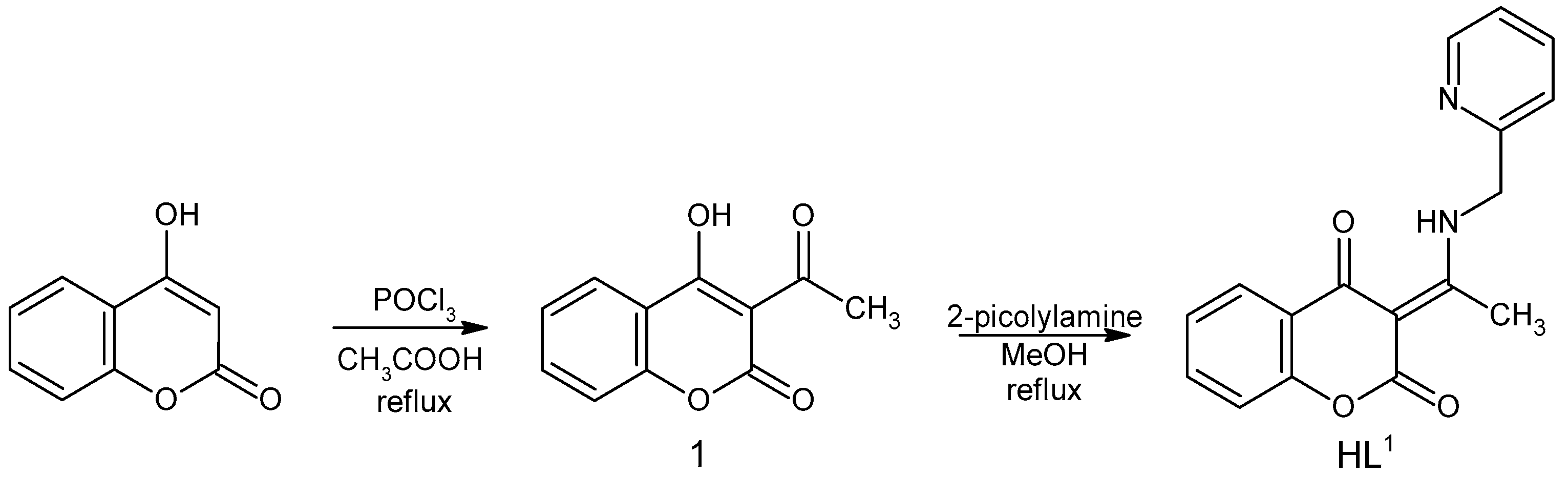
Starting compound 3-acetyl-4-hydroxy-2H-chromen-2-one (1) was prepared by acetylation of 4-hydroxy-2H-chromen-2-one (Scheme 1) [24]. To a solution of 4-hydroxy-2H-chromen-2-one (3 g, 18.6 mmol) in acetic acid (16 mL), phosphorus oxychloride POCl3 (5.6 mL) was added dropwise. The mixture was then refluxed for 45 min. After cooling in the refrigerator, a slightly yellow precipitate of 1 was filtered under suction, dried on air, and used without another purification. Yield: 54%.
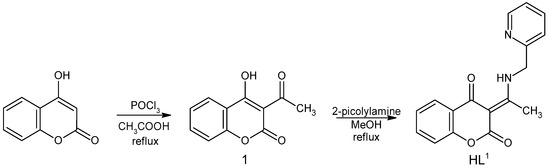
Scheme 1.
Reaction scheme of the HL1 synthesis.
Ligand HL1 was prepared by the condensation reaction of 1 with 2-picolylamine (Scheme 1) based on reactions described previously [21]. The mixture of 1 (0.205 g, 1 mmol) and 2-picolylamine (0.108 g, 1 mmol) in 25 mL methanol was refluxed for about 90 min. The reaction was monitored by TLC, using mobile phase toluene: acetone = 7:3 (v/v). After the reaction was complete, the clear solution was placed in a freezer to give a white product, which was filtered under suction and dried on air. Yield: 60%.
HL1—Calc. for C17H14N2O3 (294.30 g∙mol−1): C, 69.38; H, 4.79; N, 9.52%. Found: C, 69.44; H, 5.13; N, 9.53%.
IR (ATR, cm−1): 1689(m), 1606(m), 1586(m), 1567(m), 1460(m), 1434(w), 1335(m), 1302(m), 1225(w), 951(w), 898(m), 779(w), 753(s), 438(m) (Figure S5).
1H NMR (600 MHz, DMSO-d6): δ 14.14 (s, 1H, H11), 8.64 (ddd, J = 4.8, 1.8, 0.9 Hz, 1H, H6′), 7.96 (dd, J = 7.8, 1.7 Hz, 1H, H5), 7.87 (td, J = 7.7, 1.8 Hz, 1H, H4′), 7.63 (ddd, J = 8.2, 7.2, 1.7 Hz, 1H, H7), 7.47 (d, J = 7.9 Hz, 1H, H3′), 7.39 (ddd, J = 7.5, 4.8, 1.1 Hz, 1H, H5’), 7.29 (ddd, J = 8.1, 7.3, 1.0 Hz, 1H, H6), 7.26 (dd, J = 8.2, 1.1 Hz, 1H, H8) 5.04 (d, J = 5.1 Hz, 2H, H12), 2.75 (s, 3H, H10) ppm.
13C NMR (150 MHz, DMSO-d6): δ 179.5 (C4), 176.2 (C9), 162.0 (C2), 154.6 (C2’), 153.1 (C8a), 149.3 (C6’), 137.3 (C4’), 134.0 (C7), 125.7 (C5), 123.7 (C6), 123.1 (C5’), 122.2 (C3’), 120.3 (C4a), 116.2 (C8), 96.4 (C3), 48.7 (C12), 19.0 (C10) ppm.
3.2.2. Synthesis of [Ag(HL1)2]NO3 (AgHL1)
The complex [Ag(HL1)2]NO3 (AgHL1) was prepared by mixing 30 mL of warm (50 °C) ethanolic solution of the ligand HL1 (88.2 mg, 0.3 mmol) and 10 mL aqueous solution of AgNO3 (25.4 mg, 0.15 mmol) under strong stirring (molar ratio of AgNO3: HL1 = 1:2) in the beaker wrapped in aluminum foil. The formed solution was slowly cooled down and crystalized in the dark at temperature of 24 °C for about 2 weeks. Beige crystals of AgHL1 were then filtered off and dried on air. Yield: 73%.
[Ag(HL1)2]NO3 (AgHL1)—Calc. for C34H26AgN5O9 (758.48 g∙mol−1): C, 53.84; H, 3.72; N, 9.23%. Found: C, 53.71; H, 3.79; N, 9.13%.
IR (ATR, cm−1): 1698(s), 1607(m), 1568(m), 1465(m), 1337(m), 1287(s), 1231(w), 900(w), 751(s), 431(m) (Figure S5).
1H NMR (600 MHz, DMSO-d6): δ 14.14 (s, 1H, H11), 8.64 (ddd, J = 4.9, 1.8, 1.0 Hz, 1H, H6′), 7.96 (dd, J = 7.8, 1.5 Hz, 1H, H5), 7.87 (td, J = 7.8, 1.8 Hz, 1H, H4′), 7.63 (ddd, J = 8.2, 7.2, 1.7 Hz, 1H, H7), 7.48 (dt, J = 7.8, 1.0 Hz, 1H, H3′), 7.40 (ddd, J = 7.6, 4.8, 1.0 Hz, 1H, H5′), 7.29 (ddd, J = 8.1, 7.2, 1.1 Hz, 1H, H6), 7.26 (dd, J = 8.2, 1.0 Hz, 1H, C8), 5.04 (d, J = 5.2 Hz, 2H, H12), 2.75 (s, 3H, H10) ppm (Figure S1).
13C NMR (150 MHz, DMSO-d6): δ 179.5 (C4), 176.2 (C9), 162.0 (C2), 154.6 (C2′), 153.1 (C8a), 149.4 (C6′), 137.4 (C4′), 134.0 (C7), 125.7 (C5), 123.6 (C6), 123.1 (C5′), 122.2 (C3′), 120.3 (C4a), 116.2 (C8), 96.4 (C3), 48.8 (C12), 19.0 (C10) ppm (Figure S1).
3.3. Physical Measurements
Elemental analyses of C, H, and N were obtained on CHNOS Elemental Analyzer vario MICRO from Elementar Analysensysteme GmbH, Langenselbold, Germany.
The infrared spectra of the prepared compounds were recorded on a Nicolet 6700 FT-IR spectrophotometer from Thermo Scientific equipped with a diamond crystal Smart Orbit™ in the range 4000–400 cm−1. To process the results, the program OMNICTM from Thermo Scientific, Waltham, MA, USA, was used.
NMR spectra were recorded on a Varian VNMRS (599.87 MHz for 1H, 150.84 MHz for 13C, and 60.80 MHz for 15N) spectrometer with a 5 mm inverse-detection H-X probe equipped with a z-gradient coil at 298.15 K. All the pulse programs were taken from the Varian sequence library. Chemical shifts (δ in ppm) are given from internal solvent and the partially deuterated residual DMSO-d6 39.5 ppm for 13C; DMSO-d5 2.5 ppm for 1H. External nitromethane (0.0 ppm) was used for 15N references. NMR spectra were processed and analyzed in MestReNova v. 15.0.1 (Mestrelab Research, Santiago de Compostela, Spain).
3.4. X-Ray Data Collection and Structure Refinement
The data collections for HL1 and AgHL1 were carried out on an XtaLAB Synergy diffractometer equipped with a HyPix detector. CrysAlisPro software, Version 1.0.43 [59] was used for data collection and cell refinement, data reduction, and absorption correction. The structures were solved by SHELXT [60] and refined by subsequent Fourier syntheses using SHELXL-2018 [61], implemented in WinGX program suit [62]. A geometric analysis was performed using SHELXL-2018, PLATON [63] was used to analyze π-π interaction, while DIAMOND [64] was used for molecular graphics. Carbon-bonded hydrogen atoms were placed in calculated positions and refined riding on their parent C atoms. Nitrogen-bonded hydrogen atoms were found in the different Fourier maps and then refined riding on their parent N atoms. A summary of crystal data and structure refinement for the ligand HL1 and the complex AgHL1 are presented in Table S1.
3.5. In Vitro Antitumor Activity
3.5.1. Cell Lines and Growth Conditions
A human lung adenocarcinoma cell line A549, human colorectal adenocarcinoma cell line HT-29, and human colon fibroblasts CCD-18Co were used in experiments and were purchased from the American Type Culture Collection (ATCC, Rockville, MD, USA). CCD-18Co cell line was derived from normal colon tissue; thus, it is used as a normal non-transformed control in cancer studies. A549 and HT-29 cells were grown in complete RPMI-1640 medium (Sigma-Aldrich, St. Louis, MO, USA) and CCD-18Co cells were grown in MEM medium (Biosera, Nuaillé, France) in an incubator at 37 °C, 95% humidity and 5% CO2 atmosphere. All cultivation media were supplemented with 10% fetal bovine serum (FBS; Biosera), 7.5% NaHCO3, and antibiotics (1% antibiotic–antimycotic 100× and 50 μg∙mL−1 gentamicin, Biosera).
3.5.2. Reagents and Experimental Design
HL1 and AgHL1 were dissolved in dimethyl sulfoxide (DMSO) at a final concentration of 10 mM and stored at room temperature in the dark. For the experiments, the working solutions of HL1 and AgHL1 were always freshly prepared immediately before the addition to the cell culture.
For the MTT assay and cell proliferation assay, the cells were seeded in 96-well plates (TPP, Trasadingen, Switzerland) in a density of 6.700 cells per well (A549), 10.000 cells per well (HT-29) or 5000 cells per well (CCD-18Co), and were left to settle for 24 h before treatment. Afterward, the cells were treated with different concentrations of HL1 and AgHL1 ranging from 1 μM to 100 μM and with 1% DMSO (the highest final concentration of the diluent corresponding to DMSO concentration in the experimental groups treated with 100 μM HL1 or AgHL1).
3.5.3. MTT Assay and IC50 Values Evaluation
MTT assay was performed to determine the changes in the metabolic activity of A549, HT-29, and CCD-18Co cell lines treated with HL1 and AgHL1 and to calculate the half-maximal inhibitory concentrations (IC50) of HL1 and AgHL1.
MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; Sigma-Aldrich) was added to the cells (final concentration 0.5 mg∙mL−1) 24 and 48 h after the treatment with HL1, AgHL1, and 1% DMSO. MTT is yellow water-soluble tetrazolium salt that is reduced in metabolically active cells by mitochondrial dehydrogenases to purple water-insoluble formazan crystals. This metabolic conversion was stopped after 4 h incubation at 37 °C and the formazan crystals were dissolved by the addition of sodium dodecyl sulfate (SDS; SERVA Electrophoresis GmbH, Heidelberg, Germany) at a final concentration of 3.3%. The absorbance (λ = 584 nm) was measured using a BMG FLUOstar Optima (BMG Labtechnologies GmbH, Offenburg, Germany). Results were evaluated as percentages of the absorbance of the untreated control.
The IC50 values of HL1 and AgHL1 were extrapolated from a dose–response fit to the metabolic activity data using OriginPro 8.5.0 SR1 (OriginLab Corp., Northampton, MA, USA) and are expressed as mean IC50 values ± standard deviation (SD) from at least three independent experiments.
3.5.4. Cell Proliferation Assay
To determine the potential antiproliferative (cytostatic) effect of HL1 and AgHL1 on A549 and HT-29 cell lines, a real-time label-free proliferation assay was performed using the IncuCyteTM ZOOM live cell imaging system (Essen BioScience, Ann Arbor, MI, USA).
The 96-well plates were placed into an IncuCyteTM ZOOM system within a regular cell culture incubator (37 °C, 95% humidity, 5% CO2) immediately after the treatment of the cells with HL1, AgHL1, and 1% DMSO. Proliferation of the cells was monitored by analyzing the percentage of confluence (the cell occupied area—the percentage of the culture well surface area that is covered by a layer of cells) using the IncuCyteTM ZOOM automated software every 2 h for a total duration of 72 h. Results were evaluated as percentages of the confluence for untreated control and each experimental group.
3.5.5. Statistical Analysis
The results are expressed as the mean values ± standard deviation (SD) of at least three independent experiments. The data were analyzed using one-way ANOVA with Tukey’s post-test and the significance levels are indicated in the legend for each particular figure.
3.6. In Vitro Antimicrobial Activity
3.6.1. Test Substances, Microorganisms, and Microbial Suspension
DMSO was purchased from Acros Organics (Geel, Belgium). Resazurin was obtained from Alfa Aesar GmbH & Co. (KG, Karlsruhe, Germany). An antibiotic, doxycycline (Galenika A.D., Belgrade, Serbia), was dissolved in a nutrient liquid medium, a Mueller–Hinton broth (Torlak, Belgrade, Serbia), while an antimycotic, fluconazole (Pfizer Inc., New York, NY, USA) was dissolved in Tryptone soya broth (Torlak, Belgrade, Serbia). The tested compounds were dissolved in DMSO and then diluted into a nutrient liquid medium to achieve a concentration of 10%.
The list of tested microorganisms is presented in Table 6. All clinical isolates were a generous gift from the Institute of Public Health, Kragujevac. The other microorganisms were provided from a collection held by the Microbiology Laboratory, Faculty of Science, University of Kragujevac.
Bacterial and yeast suspensions were prepared by the direct colony method. The turbidity of the initial suspension was adjusted using a densitometer (DEN-1, BioSan, Latvia). Initial bacterial suspensions contain about 108 colony forming units (CFU)·mL−1 for bacteria and the suspension of yeast contains 106 CFU per mL. A total of 1:100 dilutions of initial suspension were additionally prepared into sterile 0.85% saline [65]. The suspensions of fungal spores were prepared by gentle stripping of spores from agar slants with growing aspergilli. The resulting suspensions were 1:1000 diluted in a sterile 0.85% saline.
3.6.2. Microdilution Method
Antimicrobial activity was tested by determining the minimum inhibitory concentration (MIC) and the minimum microbicidal concentration (MMC) using the microdilution method with resazurin [66]. The 96-well plates were prepared by dispensing 100 μL of nutrient broth, Mueller–Hinton broth for bacteria, and Tryptone soya broth for yeasts and molds, into each well. A 100 μL aliquot from the stock solution of the tested compound (with a concentration of 2000 μg∙mL−1) was added to the first row of the plate. Then, twofold serial dilutions were performed by using a multichannel pipette. The obtained concentration range was from 1000 to 7.8 μg∙mL−1. The method is described in detail in the reported paper [67].
The growth of bacteria and yeasts was monitored by adding resazurin, an indicator of microbial growth. Resazurin is a blue non-fluorescent dye that becomes pink and fluorescent when reduced to resorufin by oxidoreductases within viable cells. The inoculated plates were incubated at 37 °C for 24 h for bacteria, 28 °C for 48 h for yeasts, and 28 °C for 72 h for molds. MIC was defined as the lowest concentration of the tested substance that prevented resazurin color change from blue to pink. For molds, MIC values of the tested substance were determined as the lowest concentration that visibly inhibited mycelia growth. Minimum microbicidal concentration was determined by plating 10 mL of samples from wells, where no indicator color change and mycelia growth were recorded, on a nutrient agar medium. At the end of the incubation period, the lowest concentration with no growth (no colony) was defined as the minimum microbicidal concentration.
Doxycycline and fluconazole, dissolved in nutrient liquid medium, were used as positive controls. It was observed that 10% DMSO did not inhibit the growth of microorganisms. Each test included growth control and sterility control. All tests were performed in duplicate and MICs were constant.
4. Conclusions
A novel silver(I) complex [Ag(HL1)2]NO3 (AgHL1) with coumarin derivative (3E)-3-(1-{[(pyridin-2-yl)methyl]amino}ethylidene)-3,4-dihydro-2H-benzopyran-2,4-dione (HL1), has been synthesized and subjected to characterization. The molecular structure of AgHL1 and HL1 has been elucidated through single-crystal X-ray crystallography. In vitro antitumor efficacy assessments of the ligand, its complex and AgNO3 were conducted on A549 lung cancer and HT-29 colorectal carcinoma cell lines utilizing the MTT assay and cell proliferation analysis. Remarkably, while the ligand exhibited moderate activity, the AgHL1 complex displayed a pronounced, time- and dose-dependent inhibitory effect on the metabolic activities of both cancerous cell lines. Nevertheless, AgHL1 also induced inhibitory effects on CCD-18Co healthy fibroblasts, revealing an absence of selectivity for cancerous cells. Corroborating the MTT assay findings, AgHL1 impeded both the metabolic activity and proliferation in A549 and HT-29 cell lines. Moreover, an in vitro examination of antimicrobial efficacy was performed for both the ligand and AgHL1, evaluated against established positive controls doxycycline and fluconazole, and AgNO3. The complex and AgNO3 demonstrated a similar activity which was substantially higher compared to the ligand’s activity. There was no discrepancy observed in the effect of the substances tested against Gram-positive and Gram-negative bacteria as well as fungi. The complex exhibited enhanced efficacy compared to the positive controls against the Pseudomonas aeruginosa standard and clinical strains, in addition to the molds tested.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/inorganics13050164/s1, Figure S1. 1H (600 MHz, DMSO-d6) and 13C (150 MHz, DMSO-d6) spectra of the complex AgHL1; Figure S2. 1H,13C-HSQC and 1H,13C-HMBC spectra of the complex AgHL1; Figure S3. 15N-HMBCspectra of HL1; Figure S4. 15N-HMBCspectra of AgHL1; Figure S5. Comparison of the IR spectra of HL1 and AgHL1; Table S1. Crystal data and structure refinement of HL1 and AgHL1.
Author Contributions
Conceptualization, I.P. and J.K.; investigation, J.K., Z.J., V.D., M.V., M.L., K.Ć., I.R., R.J. and I.P.; resources, I.P., R.J. and I.R.; writing—original draft preparation, J.K., Z.J., M.V., I.R. and I.P.; writing—review and editing, J.K., Z.J., M.V., I.R. and I.P.; visualization, J.K., Z.J. and V.D.; supervision, I.P., I.R. and R.J.; project administration, I.P., I.R. and R.J.; funding acquisition, I.P., I.R. and R.J. All authors have read and agreed to the published version of the manuscript.
Funding
The financial support of Slovak grant agency VEGA 1/0126/23 is gratefully acknowledged. This work was further supported by the project Open Scientific Community for Modern Interdisciplinary Research in Medicine (OPENMED) under Grant ITMS2014+: 313011V455. This work was supported by the Serbian Ministry of Science, Technological development, and Innovations (Agreement No. 451-03-137/2025-03/200122 and 451-03-136/2025-03/200122).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The supplementary crystallographic data for HL1 and AgHL1 presented in the study are openly available at “http://www.ccdc.cam.ac.uk/structures” with deposition Numbers CCDC 2384413 and 2384414, provided by the Cambridge Crystallographic Data Centre.
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Mcquitty, R.J. Metal-Based Drugs. Sci. Prog. 2014, 97, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Peng, K.; Liang, B.-B.; Liu, W.; Mao, Z.-W. What Blocks More Anticancer Platinum Complexes from Experiment to Clinic: Major Problems and Potential Strategies from Drug Design Perspectives. Coord. Chem. Rev. 2021, 449, 214210. [Google Scholar] [CrossRef]
- Biot, C.; Delhaes, L.; N’Diaye, C.M.; Maciejewski, L.A.; Camus, D.; Dive, D.; Brocard, J.S. Synthesis and Antimalarial Activity in Vitro of Potential Metabolites of Ferrochloroquine and Related Compounds. Bioorg. Med. Chem. 1999, 7, 2843–2847. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Sadler, P.J. Metals in Medicine. Angew. Chem. Int. Ed. 1999, 38, 1512–1531. [Google Scholar] [CrossRef]
- Yan, G.-P.; Robinson, L.; Hogg, P. Magnetic Resonance Imaging Contrast Agents: Overview and Perspectives. Radiography 2007, 13, e5–e19. [Google Scholar] [CrossRef]
- Fraise, A.P. Historical Introduction. In Principles and Practice of Disinfection, Preservation & Sterilization; Fraise, A.P., Lambert, P.A., Maillard, J.-Y., Eds.; Blackwell Publishing Ltd.: Hoboken, NJ, USA, 2004; ISBN 1-4051-0199-7. [Google Scholar]
- Moyer, C.A. Treatment of Large Human Burns with 0.5% Silver Nitrate Solution. Arch. Surg. 1965, 90, 812. [Google Scholar] [CrossRef]
- Fox, C.L. Silver Sulfadiazine—A New Topical. Arch. Surg. 1968, 184–188. [Google Scholar] [CrossRef]
- Bessey, P.Q. Wound Care. In Total Burn Care; Herndon, D.N., Ed.; W.B. Saunders: Edinburgh, Scotland, 2007; pp. 127–135. ISBN 978-1-4160-3274-8. [Google Scholar]
- Dai, T.; Huang, Y.-Y.; Sharma, S.K.; Hashmi, J.T.; Kurup, D.B.; Hamblin, M.R. Topical Antimicrobials for Burn Wound Infections. Recent Pat. Anti-Infect. Drug Discov. 2010, 5, 124–151. [Google Scholar] [CrossRef]
- Raju, S.K.; Shridharshini, K.; Praveen, S.; Maruthamuthu, M.; Mohanapriya, K. Silver Complexes as Anticancer Agents: A Perspective Review. Ger. J. Pharm. Biomater. 2022, 1, 06–28. [Google Scholar] [CrossRef]
- Balcıoğlu, S.; Olgun Karataş, M.; Ateş, B.; Alıcı, B.; Özdemir, İ. Therapeutic Potential of Coumarin Bearing Metal Complexes: Where Are We Headed? Bioorg. Med. Chem. Lett. 2020, 30, 126805. [Google Scholar] [CrossRef]
- Matos, M.J.; Santana, L.; Uriarte, E.; Abreu, O.A.; Molina, E.; Yordi, E.G. Coumarins—An Important Class of Phytochemicals. In Phytochemicals-Isolation, Characterisation and Role in Human Health; Rao, A.V., Rao, L.G., Eds.; InTech: Tokyo, Japan, 2015; ISBN 978-953-51-2170-1. [Google Scholar]
- Rettie, A.E. The Pharmocogenomics of Warfarin: Closing in on Personalized Medicine. Mol. Interv. 2006, 6, 223–227. [Google Scholar] [CrossRef]
- Matos, M.; Vazquez-Rodriguez, S.; Santana, L.; Uriarte, E.; Fuentes-Edfuf, C.; Santos, Y.; Muñoz-Crego, A. Synthesis and Structure-Activity Relationships of Novel Amino/Nitro Substituted 3-Arylcoumarins as Antibacterial Agents. Molecules 2013, 18, 1394–1404. [Google Scholar] [CrossRef]
- Kapoor, S. The Anti-Neoplastic Effects of Coumarin: An Emerging Concept. Cytotechnology 2013, 65, 787–788. [Google Scholar] [CrossRef] [PubMed]
- Bansal, Y.; Sethi, P.; Bansal, G. Coumarin: A Potential Nucleus for Anti-Inflammatory Molecules. Med. Chem. Res. 2013, 22, 3049–3060. [Google Scholar] [CrossRef]
- Xu, Z.; Chen, Q.; Zhang, Y.; Liang, C. Coumarin-Based Derivatives with Potential Anti-HIV Activity. Fitoterapia 2021, 150, 104863. [Google Scholar] [CrossRef]
- Thati, B.; Noble, A.; Creaven, B.S.; Walsh, M.; McCann, M.; Devereux, M.; Kavanagh, K.; Egan, D.A. Role of Cell Cycle Events and Apoptosis in Mediating the Anti-Cancer Activity of a Silver(I) Complex of 4-Hydroxy-3-Nitro-Coumarin-Bis(Phenanthroline) in Human Malignant Cancer Cells. Eur. J. Pharmacol. 2009, 602, 203–214. [Google Scholar] [CrossRef] [PubMed]
- Mujahid, M.; Kia, A.F.-A.; Duff, B.; Egan, D.A.; Devereux, M.; McClean, S.; Walsh, M.; Trendafilova, N.; Georgieva, I.; Creaven, B.S. Spectroscopic Studies, DFT Calculations, and Cytotoxic Activity of Novel Silver(I) Complexes of Hydroxy Ortho-Substituted-Nitro-2H-Chromen-2-One Ligands and a Phenanthroline Adduct. J. Inorg. Biochem. 2015, 153, 103–113. [Google Scholar] [CrossRef] [PubMed]
- Brawley, J.; Etter, E.; Heredia, D.; Intasiri, A.; Nennecker, K.; Smith, J.; Welcome, B.M.; Brizendine, R.K.; Gould, T.W.; Bell, T.W.; et al. Synthesis and Evaluation of 4-Hydroxycoumarin Imines as Inhibitors of Class II Myosins. J. Med. Chem. 2020, 63, 11131–11148. [Google Scholar] [CrossRef]
- OECD. Onkologický Profil Krajiny: Slovenská Republika 2023. In EU Country Cancer Profiles; OECD Publishing: Paris, France, 2023; ISBN 9789264710344. [Google Scholar]
- Fromm, K.M. Silver Coordination Compounds with Antimicrobial Properties. Appl. Organomet. Chem. 2013, 27, 683–687. [Google Scholar] [CrossRef]
- Sukdolak, S.; Solujić, S.; Manojlović, N.; Vuković, N.; Krstić, L.J. Hantzsch Reaction of 3-(2-Bromoacetyl)-4-Hydroxy-Chromen-2-One. Synthesis of 3-(Thiazol-4-Yl)-4-Hydroxy Coumarines. J. Heterocycl. Chem. 2004, 41, 593–596. [Google Scholar] [CrossRef]
- Ilić, D.R.; Jevtić, V.V.; Radić, G.P.; Arsikin, K.; Ristić, B.; Harhaji-Trajković, L.; Vuković, N.; Sukdolak, S.; Klisurić, O.; Trajković, V.; et al. Synthesis, Characterization and Cytotoxicity of a New Palladium(II) Complex with a Coumarine-Derived Ligand. Eur. J. Med. Chem. 2014, 74, 502–508. [Google Scholar] [CrossRef] [PubMed]
- Kurjan, J.; Jendželovská, Z.; Smolková, R.; Volarevic, V.; Smolko, L.; Jendželovský, R.; Potočňák, I. Manuscript in Preparation; Pavol Jozef Šafárik University: Košice, Slovakia, 2025. [Google Scholar]
- Butera, V.; D’Anna, L.; Rubino, S.; Bonsignore, R.; Spinello, A.; Terenzi, A.; Barone, G. How the Metal Ion Affects the 1H NMR Chemical Shift Values of Schiff Base Metal Complexes: Rationalization by DFT Calculations. J. Phys. Chem. A 2023, 127, 9283–9290. [Google Scholar] [CrossRef]
- El-Naggar, M.A.; Sharaf, M.M.; Albering, J.H.; Abu-Youssef, M.A.M.; Kassem, T.S.; Soliman, S.M.; Badr, A.M.A. One Pot Synthesis of Two Potent Ag(I) Complexes with Quinoxaline Ligand, X-Ray Structure, Hirshfeld Analysis, Antimicrobial, and Antitumor Investigations. Sci. Rep. 2022, 12, 20881. [Google Scholar] [CrossRef] [PubMed]
- Sakharov, S.G.; Kovalev, V.V.; Gorbunova, Y.E.; Tokmakov, G.P.; Skabitskii, I.V.; Kokunov, Y.V. Structures of Silver Nitrate Complexes with Quinolines According to NMR Data. Russ. J. Coord. Chem. 2017, 43, 75–81. [Google Scholar] [CrossRef]
- Dalvit, C.; Veronesi, M.; Vulpetti, A. 1H and 19F NMR Chemical Shifts for Hydrogen Bond Strength Determination: Correlations between Experimental and Computed Values. J. Magn. Reson. 2022, 12–13, 100070. [Google Scholar] [CrossRef]
- Gilli, G.; Bellucci, F.; Ferretti, V.; Bertolasi, V. Evidence for Resonance-Assisted Hydrogen Bonding from Crystal-Structure Correlations on the Enol Form of the Beta-Diketone Fragment. J. Am. Chem. Soc. 1989, 111, 1023–1028. [Google Scholar] [CrossRef]
- Avdović, E.H.; Stojković, D.L.J.; Jevtić, V.V.; Kosić, M.; Ristić, B.; Harhaji-Trajković, L.; Vukić, M.; Vuković, N.; Marković, Z.S.; Potočňák, I.; et al. Synthesis, Characterization and Cytotoxicity of a New Palladium(II) Complex with a Coumarin-Derived Ligand 3-(1-(3-Hydroxypropylamino)Ethylidene)Chroman-2,4-Dione. Crystal Structure of the 3-(1-(3-Hydroxypropylamino)Ethylidene)-Chroman-2,4-Dione. Inorg. Chim. Acta 2017, 466, 188–196. [Google Scholar] [CrossRef]
- Usman, M.; Zaki, M.; Khan, R.A.; Alsalme, A.; Ahmad, M.; Tabassum, S. Coumarin Centered Copper(II) Complex with Appended-Imidazole as Cancer Chemotherapeutic Agents against Lung Cancer: Molecular Insight via DFT-Based Vibrational Analysis. RSC Adv. 2017, 7, 36056–36071. [Google Scholar] [CrossRef]
- Bruno, I.J.; Cole, J.C.; Edgington, P.R.; Kessler, M.; Macrae, C.F.; McCabe, P.; Pearson, J.; Taylor, R. New Software for Searching the Cambridge Structural Database and Visualizing Crystal Structures. Acta Crystallogr. Sect. B Struct. Sci. 2002, 58, 389–397. [Google Scholar] [CrossRef]
- Wang, P.-N.; Yeh, C.-W.; Tsou, C.-H.; Ho, Y.-W.; Lin, S.-C.; Suen, M.-C. Structural Diversity in the Self-Assembly of Ag(i) Complexes Containing 2,6-Dimethyl-3,5-Dicyano-4-(3-Pyridyl)-1,4-Dihydropyridine. New J. Chem. 2014, 38, 1079. [Google Scholar] [CrossRef]
- Ratte, H.T. Bioaccumulation and Toxicity of Silver Compounds: A Review. Environ. Toxicol. Chem. 1999, 18, 89–108. [Google Scholar] [CrossRef]
- Żyro, D.; Radko, L.; Śliwińska, A.; Chęcińska, L.; Kusz, J.; Korona-Głowniak, I.; Przekora, A.; Wójcik, M.; Posyniak, A.; Ochocki, J. Multifunctional Silver(I) Complexes with Metronidazole Drug Reveal Antimicrobial Properties and Antitumor Activity against Human Hepatoma and Colorectal Adenocarcinoma Cells. Cancers 2022, 14, 900. [Google Scholar] [CrossRef]
- Budzisz, E.; Keppler, B.K.; Giester, G.; Wozniczka, M.; Kufelnicki, A.; Nawrot, B. Synthesis, Crystal Structure and Biological Characterization of a Novel Palladium( II ) Complex with a Coumarin-Derived Ligand. Eur. J. Inorg. Chem. 2004, 2004, 4412–4419. [Google Scholar] [CrossRef]
- Buľková, V.; Vargová, J.; Babinčák, M.; Jendželovský, R.; Zdráhal, Z.; Roudnický, P.; Košuth, J.; Fedoročko, P. New Findings on the Action of Hypericin in Hypoxic Cancer Cells with a Focus on the Modulation of Side Population Cells. Biomed. Pharmacother. 2023, 163, 114829. [Google Scholar] [CrossRef]
- Leong, K.H.; Looi, C.Y.; Loong, X.-M.; Cheah, F.K.; Supratman, U.; Litaudon, M.; Mustafa, M.R.; Awang, K. Cycloart-24-Ene-26-Ol-3-One, a New Cycloartane Isolated from Leaves of Aglaia Exima Triggers Tumour Necrosis Factor-Receptor 1-Mediated Caspase-Dependent Apoptosis in Colon Cancer Cell Line. PLoS ONE 2016, 11, e0152652. [Google Scholar] [CrossRef]
- Creaven, B.S.; Egan, D.A.; Kavanagh, K.; McCann, M.; Noble, A.; Thati, B.; Walsh, M. Synthesis, Characterization and Antimicrobial Activity of a Series of Substituted Coumarin-3-Carboxylatosilver(I) Complexes. Inorg. Chim. Acta 2006, 359, 3976–3984. [Google Scholar] [CrossRef]
- Mujahid, M.; Trendafilova, N.; Arfa-Kia, A.F.; Rosair, G.; Kavanagh, K.; Devereux, M.; Walsh, M.; McClean, S.; Creaven, B.S.; Georgieva, I. Novel Silver(I) Complexes of Coumarin Oxyacetate Ligands and Their Phenanthroline Adducts: Biological Activity, Structural and Spectroscopic Characterisation. J. Inorg. Biochem. 2016, 163, 53–67. [Google Scholar] [CrossRef] [PubMed]
- Karataş, M.O.; Olgundeniz, B.; Günal, S.; Özdemir, İ.; Alıcı, B.; Çetinkaya, E. Synthesis, Characterization and Antimicrobial Activities of Novel Silver(I) Complexes with Coumarin Substituted N-Heterocyclic Carbene Ligands. Bioorg. Med. Chem. 2016, 24, 643–650. [Google Scholar] [CrossRef]
- Achar, G.; Ramya, V.C.; Upendranath, K.; Budagumpi, S. Coumarin-tethered (Benz)Imidazolium Salts and Their Silver(I) N-heterocyclic Carbene Complexes: Synthesis, Characterization, Crystal Structure and Antibacterial Studies. Appl. Organomet. Chem. 2017, 31, e3770. [Google Scholar] [CrossRef]
- Achar, G.; Shahini, C.R.; Patil, S.A.; Budagumpi, S. Synthesis, Structural Characterization, Crystal Structures and Antibacterial Potentials of Coumarin–Tethered N–Heterocyclic Carbene Silver(I) Complexes. J. Organomet. Chem. 2017, 833, 28–42. [Google Scholar] [CrossRef]
- Achar, G.; Uppendranath, K.; Ramya, V.C.; Biffis, A.; Keri, R.S.; Budagumpi, S. Synthesis, Characterization, Crystal Structure and Biological Studies of Silver(I) Complexes Derived from Coumarin-Tethered N-Heterocyclic Carbene Ligands. Polyhedron 2017, 123, 470–479. [Google Scholar] [CrossRef]
- Achar, G.; Agarwal, P.; Brinda, K.N.; Małecki, J.G.; Keri, R.S.; Budagumpi, S. Ether and Coumarin–Functionalized (Benz)Imidazolium Salts and Their Silver(I)–N–Heterocyclic Carbene Complexes: Synthesis, Characterization, Crystal Structures and Antimicrobial Studies. J. Organomet. Chem. 2018, 854, 64–75. [Google Scholar] [CrossRef]
- Ranjan Sahoo, C.; Sahoo, J.; Mahapatra, M.; Lenka, D.; Kumar Sahu, P.; Dehury, B.; Nath Padhy, R.; Kumar Paidesetty, S. Coumarin Derivatives as Promising Antibacterial Agent(s). Arab. J. Chem. 2021, 14, 102922. [Google Scholar] [CrossRef]
- Jaiswal, S.; Bhattacharya, K.; Sullivan, M.; Walsh, M.; Creaven, B.S.; Laffir, F.; Duffy, B.; McHale, P. Non-Cytotoxic Antibacterial Silver–Coumarin Complex Doped Sol–Gel Coatings. Colloids Surf. B 2013, 102, 412–419. [Google Scholar] [CrossRef]
- Li, W.-R.; Sun, T.-L.; Zhou, S.-L.; Ma, Y.-K.; Shi, Q.-S.; Xie, X.-B.; Huang, X.-M. A Comparative Analysis of Antibacterial Activity, Dynamics, and Effects of Silver Ions and Silver Nanoparticles against Four Bacterial Strains. Int. Biodeterior. Biodegrad. 2017, 123, 304–310. [Google Scholar] [CrossRef]
- Kvitek, L.; Panacek, A.; Prucek, R.; Soukupova, J.; Vanickova, M.; Kolar, M.; Zboril, R. Antibacterial Activity and Toxicity of Silver – Nanosilver versus Ionic Silver. J. Phys. Conf. Ser. 2011, 304, 012029. [Google Scholar] [CrossRef]
- Parvekar, P.; Palaskar, J.; Metgud, S.; Maria, R.; Dutta, S. The Minimum Inhibitory Concentration (MIC) and Minimum Bactericidal Concentration (MBC) of Silver Nanoparticles against Staphylococcus aureus. Biomater. Investig. Dent. 2020, 7, 105–109. [Google Scholar] [CrossRef]
- Claudel, M.; Schwarte, J.V.; Fromm, K.M. New Antimicrobial Strategies Based on Metal Complexes. Chemistry 2020, 2, 849–899. [Google Scholar] [CrossRef]
- Ishida, T. Antibacterial Mechanism of Ag+ Ions for Bacteriolyses of Bacterial Cell Walls via Peptidoglycan Autolysins, and DNA Damages. MOJ Toxicol. 2018, 4, 345–350. [Google Scholar] [CrossRef]
- Feng, Q.L.; Wu, J.; Chen, G.Q.; Cui, F.Z.; Kim, T.N.; Kim, J.O. A Mechanistic Study of the Antibacterial Effect of Silver Ions onEscherichia Coli andStaphylococcus Aureus. J. Biomed. Mater. Res. 2000, 52, 662–668. [Google Scholar] [CrossRef]
- Randall, C.P.; Oyama, L.B.; Bostock, J.M.; Chopra, I.; O’Neill, A.J. The Silver Cation (Ag+): Antistaphylococcal Activity, Mode of Action and Resistance Studies. J. Antimicrob. Chemother. 2013, 68, 131–138. [Google Scholar] [CrossRef]
- Ge, C.; Huang, M.; Huang, D.; Dang, F.; Huang, Y.; Ahmad, H.A.; Zhu, C.; Chen, N.; Wu, S.; Zhou, D. Effect of Metal Cations on Antimicrobial Activity and Compartmentalization of Silver in Shewanella Oneidensis MR-1 upon Exposure to Silver Ions. Sci. Total Environ. 2022, 838, 156401. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, D.S.; Abd El-Baky, R.M.; Sandle, T.; Mandour, S.A.; Ahmed, E.F. Antimicrobial Activity of Silver-Treated Bacteria against Other Multi-Drug Resistant Pathogens in Their Environment. Antibiotics 2020, 9, 181. [Google Scholar] [CrossRef]
- CrysAlisPRO, Version 1.0.43, Oxford Diffraction/Agilent Technologies UK Ltd.: Yarnton, UK, 2020.
- Sheldrick, G.M. SHELXT–Integrated Space-Group and Crystal-Structure Determination. Acta Crystallogr. Sect. A Found. Adv. 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. Crystal Structure Refinement with SHELXL. Acta Crystallogr. Sect. C Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Farrugia, L.J. WinGX and ORTEP for Windows: An Update. J. Appl. Crystallogr. 2012, 45, 849–854. [Google Scholar] [CrossRef]
- Spek, A.L. Structure Validation in Chemical Crystallography. Acta Crystallogr. Sect. D Biol. Crystallogr. 2009, 65, 148–155. [Google Scholar] [CrossRef]
- Brandenburg, K. DIAMOND, Version 3.2k ed; Crystal Impact GbR: Bonn, Germany, 2014. [Google Scholar]
- Andrews, J.M. Determination of Minimum Inhibitory Concentrations. J. Antimicrob. Chemother. 2001, 48 (Suppl. 1), 5–16. [Google Scholar] [CrossRef] [PubMed]
- Sarker, S.D.; Nahar, L.; Kumarasamy, Y. Microtitre Plate-Based Antibacterial Assay Incorporating Resazurin as an Indicator of Cell Growth, and Its Application in the in Vitro Antibacterial Screening of Phytochemicals. Methods 2007, 42, 321–324. [Google Scholar] [CrossRef]
- Radić, G.P.; Glođović, V.V.; Radojević, I.D.; Stefanović, O.D.; Čomić, L.R.; Ratković, Z.R.; Valkonen, A.; Rissanen, K.; Trifunović, S.R. Synthesis, Characterization and Antimicrobial Activity of Palladium(II) Complexes with Some Alkyl Derivates of Thiosalicylic Acids: Crystal Structure of the Bis(S-Benzyl-Thiosalicylate)–Palladium(II) Complex, [Pd(S-Bz-Thiosal)2]. Polyhedron 2012, 31, 69–76. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).