A Thermodynamic Approach for the Prediction of Oiling Out Boundaries from Solubility Data
Abstract
1. Introduction
2. Theory and Model Development
2.1. Fluid Phase Thermodynamics
2.1.1. Free Energy of the Reference Fluid
2.1.2. First Perturbation Term
2.1.3. Second Perturbation Term
2.1.4. Chemical Potential and Pressure
2.2. Solid Phase Thermodynamics
2.3. Gelation Boundary
2.4. Model Implementation
2.4.1. Obtaining Molecular Interaction Parameters
2.4.2. Practical Considerations
2.4.3. Calculation of Other Phase Boundaries
3. Experimental Section — Methods
3.1. Pyraclostrobin – Isopropanol/Cyclohexane
3.2. Compound Z – Methanol/Water
3.3. Idebenone – Hexane/Methylene Chloride
3.4. C₃₅H₄₁Cl₂N₃O₂ – Ethanol/Water
3.5. Vanillin – 1-Propanol/Water
3.6. Data Extraction and Processing
4. Results
5. Discussion
5.1. Relevance of the Spinodal and Gelation Lines to Process Development
5.2. Limitations of the Current Modeling Approach
6. Concluding Remarks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lewis, A.; Seckler, M.; Kramer, H.; van Rosmalen, G. Industrial Crystallization: Fundamentals and Applications; Cambridge University Press: Cambridge, UK, 2015; p. 346. [Google Scholar]
- Tung, H.-H.; Paul, E.L.; Midler, M.; McCauley, J.A. Crystallization of Organic Compounds: An Industrial Perspective, 1st ed.; John Wiley & Sons: Hoboken, NJ, USA, 2009; p. 304. [Google Scholar]
- de Albuquerque, I.; Mazzotti, M. Influence of Liquid-Liquid Phase Separation on the Crystallization of L-Menthol from Water. Chem. Eng. Technol. 2017, 40, 1339–1346. [Google Scholar] [CrossRef]
- Li, K.; Wu, S.; Xu, S.; Du, S.; Zhao, K.; Lin, L.; Yang, P.; Yu, B.; Hou, B.; Gong, J. Oiling out and Polymorphism Control of Pyraclostrobin in Cooling Crystallization. Ind. Eng. Chem. Res. 2016, 55, 11631–11637. [Google Scholar] [CrossRef]
- Lu, J.; Li, Y.-P.; Wang, J.; Ren, G.-B.; Rohani, S.; Ching, C.-B. Crystallization of an Active Pharmaceutical Ingredient That Oils Out. Sep. Purif. Technol. 2012, 96, 1–6. [Google Scholar] [CrossRef]
- Lu, J.; Li, Y.-P.; Wang, J.; Li, Z.; Rohani, S.; Ching, C.-B. Study on the Oiling-out and Crystallization for the Purification of Idebenone. Org. Process Res. Dev. 2012, 16, 442–446. [Google Scholar] [CrossRef]
- Deneau, E.; Steele, G. An In-Line Study of Oiling Out and Crystallization. Org. Process Res. Dev. 2005, 9, 943–950. [Google Scholar] [CrossRef]
- Bhamidi, V.; Skrzypczak-Jankun, E.; Schall, C.A. Dependence of Nucleation Kinetics and Crystal Morphology of a Model Protein System on Ionic Strength. J. Cryst. Growth 2001, 232, 77–85. [Google Scholar] [CrossRef]
- Bonnett, P.E.; Carpenter, K.J.; Dawson, S.; Davey, R.J. Solution Crystallisation via a Submerged Liquid–Liquid Phase Boundary: Oiling Out. Chem. Commun. 2003, 6, 698–699. [Google Scholar] [CrossRef]
- Heijna, M.C.R.; Theelen, M.J.; van Enckevort, W.J.P.; Vlieg, E. Spherulitic Growth of Hen Egg-White Lysozyme Crystals. J. Phys. Chem. B 2007, 111, 1567–1573. [Google Scholar] [CrossRef]
- Bhamidi, V.; Dumoleijn, K.; Guha, D.; Kirk, S.K.; De Bruyn, A.; Pymer, A.K. From Experiments and Models to Business Decisions: A Scale-up Study on the Reactive Crystallization of a Crop Protection Agent. Org. Process Res. Dev. 2019, 23, 342–354. [Google Scholar] [CrossRef]
- Duffy, D.; Cremin, N.; Napier, M.; Robinson, S.; Barrett, M.; Hao, H.; Glennon, B. In situ Monitoring, Control and Optimization of a Liquid–Liquid Phase Separation Crystallization. Chem. Eng. Sci. 2013, 77, 112–121. [Google Scholar] [CrossRef]
- Barker, J.A.; Henderson, D. Perturbation Theory and Equation of State for Fluids. II. A Successful Theory of Liquids. J. Chem. Phys. 1967, 47, 4714–4721. [Google Scholar] [CrossRef]
- Barker, J.A.; Henderson, D. Perturbation Theory and Equation of State for Fluids: The Square-Well Potential. J. Chem. Phys. 1967, 47, 2856–2861. [Google Scholar] [CrossRef]
- Weeks, J.D.; Chandler, D.; Andersen, H.C. Role of Repulsive Forces in Determining the Equilibrium Structure of Simple Liquids. J. Chem. Phys. 1971, 54, 5237–5247. [Google Scholar] [CrossRef]
- Verlet, L.; Weis, J.-J. Equilibrium Theory of Simple Liquids. Phys. Rev. A 1972, 5, 939–952. [Google Scholar] [CrossRef]
- Verlet, L.; Weis, J.-J. Perturbation Theory for the Thermodynamic Properties of Simple Liquids. Mol. Phys. 1972, 24, 1013–1024. [Google Scholar] [CrossRef]
- Barker, J.A.; Henderson, D. What is “Liquid”? Understanding the States of Matter. Rev. Mod. Phys. 1976, 48, 587–671. [Google Scholar] [CrossRef]
- Kang, H.S.; Lee, C.S.; Ree, T.; Ree, F.H. A Perturbation Theory of Classical Equilibrium Fluids. J. Chem. Phys. 1985, 82, 414–423. [Google Scholar] [CrossRef]
- Kang, H.S.; Ree, T.; Ree, F.H. A Perturbation Theory of Classical Solids. J. Chem. Phys. 1986, 84, 4547–4557. [Google Scholar] [CrossRef]
- Tang, Y. On the First-order Mean Spherical Approximation. J. Chem. Phys. 2003, 118, 4140–4148. [Google Scholar] [CrossRef]
- Tuinier, R.; Fleer, G.J. Critical Endpoint and Analytical Phase Diagram of Attractive Hard-Core Yukawa Spheres. J. Phys. Chem. B 2006, 110, 20540–20545. [Google Scholar] [CrossRef]
- Löwen, H. Density Functional Theory of Inhomogeneous Classical Fluids: Recent Developments and new Perspectives. J. Phys. Condens. Matter 2002, 14, 11897–11905. [Google Scholar] [CrossRef]
- Rosenfeld, Y. Structure and Effective Interactions in Multi-Component Hard-Sphere Liquids: The Fundamental-Measure Density Functional Approach. J. Phys. Condens. Matter 2002, 14, 9141–9152. [Google Scholar] [CrossRef]
- Roth, R.; Evans, R.; Lang, A.; Kahl, G. Fundamental Measure Theory for Hard-sphere Mixtures Revisited: The White Bear Version. J. Phys. Condens. Matter 2002, 14, 12063–12078. [Google Scholar] [CrossRef]
- Bannerman, M.N.; Lue, L.; Woodcock, L.V. Thermodynamic Pressures for Hard Spheres and Closed-Virial Equation-of-State. J. Chem. Phys. 2010, 132, 084507. [Google Scholar] [CrossRef] [PubMed]
- Henderson, D.; Scalise, O.H.; Smith, W.R. Monte Carlo Calculations of the Equation of State of the Square-well Fluid as a Function of Well Width. J. Chem. Phys. 1980, 72, 2431–2438. [Google Scholar] [CrossRef]
- ten Wolde, P.R.; Frenkel, D. Enhancement of Protein Crystal Nucleation by Critical Density Fluctuations. Science 1997, 277, 1975–1977. [Google Scholar] [CrossRef] [PubMed]
- Valadez-Pérez, N.E.; Benavides, A.L.; Schöll-Paschinger, E.; Castañeda-Priego, R. Phase Behavior of Colloids and Proteins in Aqueous Suspensions: Theory and Computer Simulations. J. Chem. Phys. 2012, 137, 084905. [Google Scholar] [CrossRef]
- Vega, L.; de Miguel, E.; Rull, L.F.; Jackson, G.; McLure, I.A. Phase Equilibria and Critical Behavior of Square-well Fluids of Variable Width by Gibbs Ensemble Monte Carlo Simulation. J. Chem. Phys. 1992, 96, 2296–2305. [Google Scholar] [CrossRef]
- Debenedetti, P.G. Metastable Liquids: Concepts and Principles, 1st ed.; Princeton University Press: Princeton, NJ, USA, 1996. [Google Scholar]
- Anderson, V.J.; Lekkerkerker, H.N.W. Insights into Phase Transition Kinetics from Colloid Science. Nature 2002, 416, 811–815. [Google Scholar] [CrossRef]
- Asherie, N.; Lomakin, A.; Benedek, G.B. Phase Diagram of Colloidal Solutions. Phys. Rev. Lett. 1996, 77, 4832–4835. [Google Scholar] [CrossRef]
- Lomakin, A.; Asherie, N.; Benedek, G.B. Monte Carlo Study of Phase Separation in Aqueous Protein Solutions. J. Chem. Phys. 1996, 104, 1646–1656. [Google Scholar] [CrossRef]
- Rosenbaum, D.F.; Kulkarni, A.; Ramakrishnan, S.; Zukoski, C.F. Protein Interactions and Phase Behavior: Sensitivity to the Form of the Pair Potential. J. Chem. Phys. 1999, 111, 9882–9890. [Google Scholar] [CrossRef]
- Kulkarni, A.M.; Zukoski, C.F. Nanoparticle Crystal Nucleation: Influence of Solution Conditions. Langmuir 2002, 18, 3090–3099. [Google Scholar] [CrossRef]
- Gil-Villegas, A.; Galindo, A.; Whitehead, P.J.; Mills, S.J.; Jackson, G.; Burgess, A.N. Statistical Associating Fluid Theory for Chain Molecules with Attractive Potentials of Variable Rrange. J. Chem. Phys. 1997, 106, 4168–4186. [Google Scholar] [CrossRef]
- Patel, B.H.; Docherty, H.; Varga, S.; Galindo, A.; Maitland, G.C. Generalized Equation of State for Square-well Potentials of Variable Range. Mol. Phys. 2005, 103, 129–139. [Google Scholar] [CrossRef]
- Hard Sphere Model. Available online: http://www.sklogwiki.org/SklogWiki/index.php/Hard_sphere_ model (accessed on 7 April 2019).
- Carnahan-Starling Equtaion of State. Available online: http://www.sklogwiki.org/SklogWiki/index.php/Carnahan-Starling_equation_of_state (accessed on 7 April 2019).
- Carnahan, N.F.; Starling, K.E. Equation of State for Nonattracting Rigid Spheres. J. Chem. Phys. 1969, 51, 635–636. [Google Scholar] [CrossRef]
- Mean Value Theorem. Available online: https://en.wikipedia.org/wiki/Mean_value_theorem (accessed on 7 April 2019).
- Tao, F.-M.; Song, Y.; Mason, E.A. Derivative of the Hard-sphere Radial Distribution Function at Contact. Phys. Rev. A 1992, 46, 8007–8008. [Google Scholar] [CrossRef]
- He, G.; Tan, R.B.H.; Kenis, P.J.A.; Zukoski, C.F. Generalized Phase Behavior of Small Molecules and Nanoparticles. J. Phys. Chem. B 2007, 111, 12494–12499. [Google Scholar] [CrossRef]
- He, G.; Tan, R.B.H.; Kenis, P.J.A.; Zukoski, C.F. Metastable States of Small-Molecule Solutions. J. Phys. Chem. B 2007, 111, 14121–14129. [Google Scholar] [CrossRef]
- Bergenholtz, J.; Fuchs, M.; Voigtmann, T. Colloidal Gelation and Non-ergodicity Transitions. J. Phys. Condens. Matter 2000, 12, 6575. [Google Scholar] [CrossRef][Green Version]
- Lafferrère, L.; Hoff, C.; Veesler, S. Polymorphism and Liquid-Liquid Demixing in Supersaturated Drug Solution. Eng. Life Sci. 2003, 3, 127–131. [Google Scholar] [CrossRef]
- Veesler, S.; Lafferrère, L.; Garcia, E.; Hoff, C. Phase Transitions in Supersaturated Drug Solution. Org. Process Res. Dev. 2003, 7, 983–989. [Google Scholar] [CrossRef]
- Lafferrère, L.; Hoff, C.; Veesler, S. In Situ Monitoring of the Impact of Liquid−Liquid Phase Separation on Drug Crystallization by Seeding. Cryst. Growth Des. 2004, 4, 1175–1180. [Google Scholar] [CrossRef]
- Lafferrère, L.; Hoff, C.; Veesler, S. Study of Liquid–Liquid Demixing from Drug Solution. J. Cryst. Growth 2004, 269, 550–557. [Google Scholar] [CrossRef]
- Veesler, S.; Revalor, E.; Bottini, O.; Hoff, C. Crystallization in the Presence of a Liquid−Liquid Phase Separation. Org. Process Res. Dev. 2006, 10, 841–845. [Google Scholar] [CrossRef]
- Zhao, H.; Xie, C.; Xu, Z.; Wang, Y.; Bian, L.; Chen, Z.; Hao, H. Solution Crystallization of Vanillin in the Presence of a Liquid–Liquid Phase Separation. Ind. Eng. Chem. Res. 2012, 51, 14646–14652. [Google Scholar] [CrossRef]
- Engauge Digitizer. Available online: https://markummitchell.github.io/engauge-digitizer/ (accessed on 7 April 2019).
- Design Institute for Physcial Properties (DIPPR) Database. Available online: https://www.aiche.org/dippr (accessed on 7 April 2019).
- Lever Rule. Available online: https://en.wikipedia.org/wiki/Lever_rule (accessed on 7 April 2019).
- de Albuquerque, I.; Mazzotti, M. Crystallization Process Design Using Thermodynamics to Avoid Oiling Out in a Mixture of Vanillin and Water. Cryst. Growth Des. 2014, 14, 5617–5625. [Google Scholar] [CrossRef]
- Codan, L.; Bäbler, M.U.; Mazzotti, M. Phase Diagram of a Chiral Substance Exhibiting Oiling Out in Cyclohexane. Cryst. Growth Des. 2010, 10, 4005–4013. [Google Scholar] [CrossRef]
- Du, Y.; Wang, H.; Du, S.; Wang, Y.; Huang, C.; Qin, Y.; Gong, J. The Liquid–Liquid Phase Separation and Crystallization of Vanillin in 1-Propanol/Water Solution. Fluid Ph. Equilib. 2016, 409, 84–91. [Google Scholar] [CrossRef]
- Kiesow, K.; Tumakaka, F.; Sadowski, G. Experimental Investigation and Prediction of Oiling out During Crystallization Process. J. Cryst. Growth 2008, 310, 4163–4168. [Google Scholar] [CrossRef]
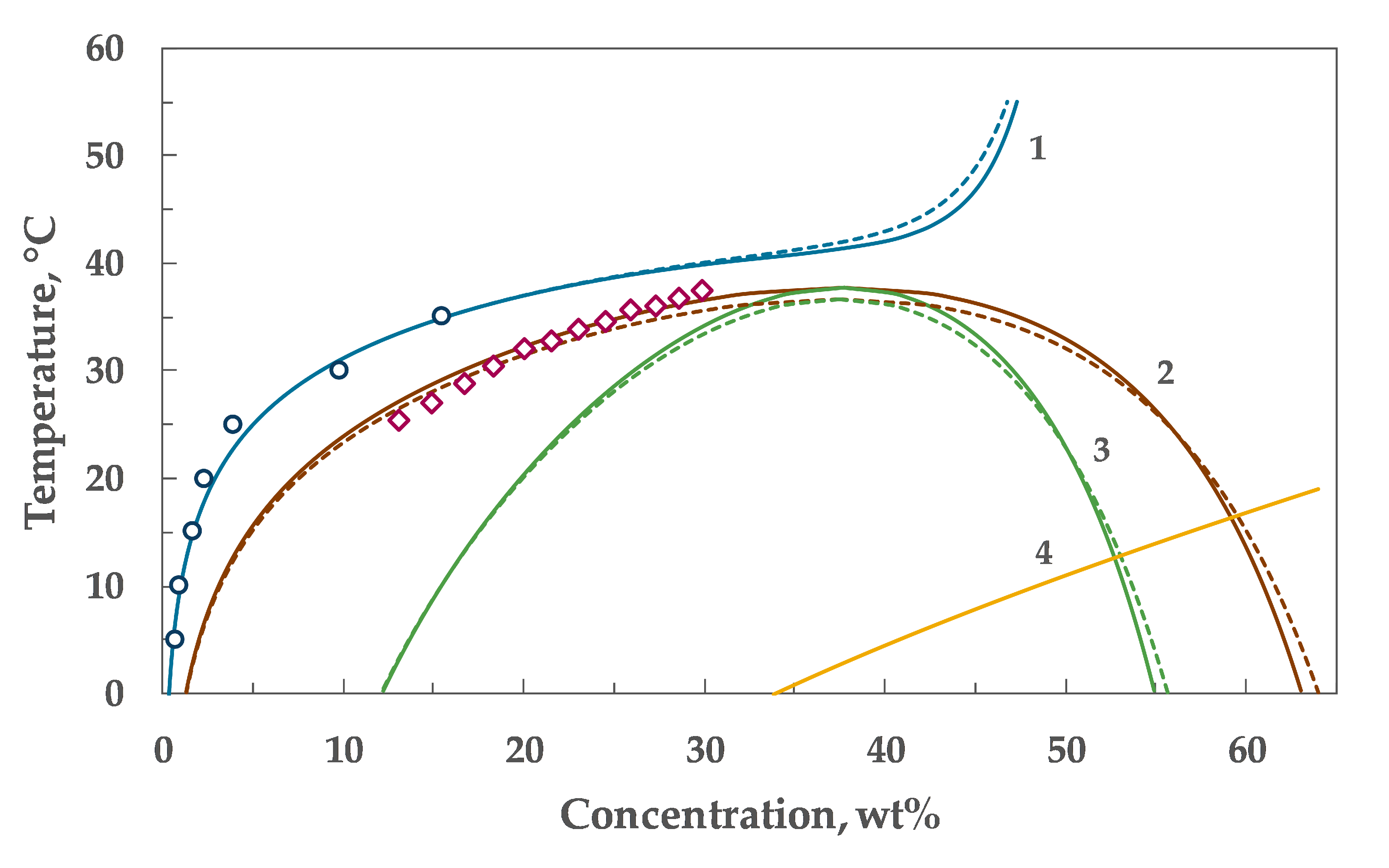
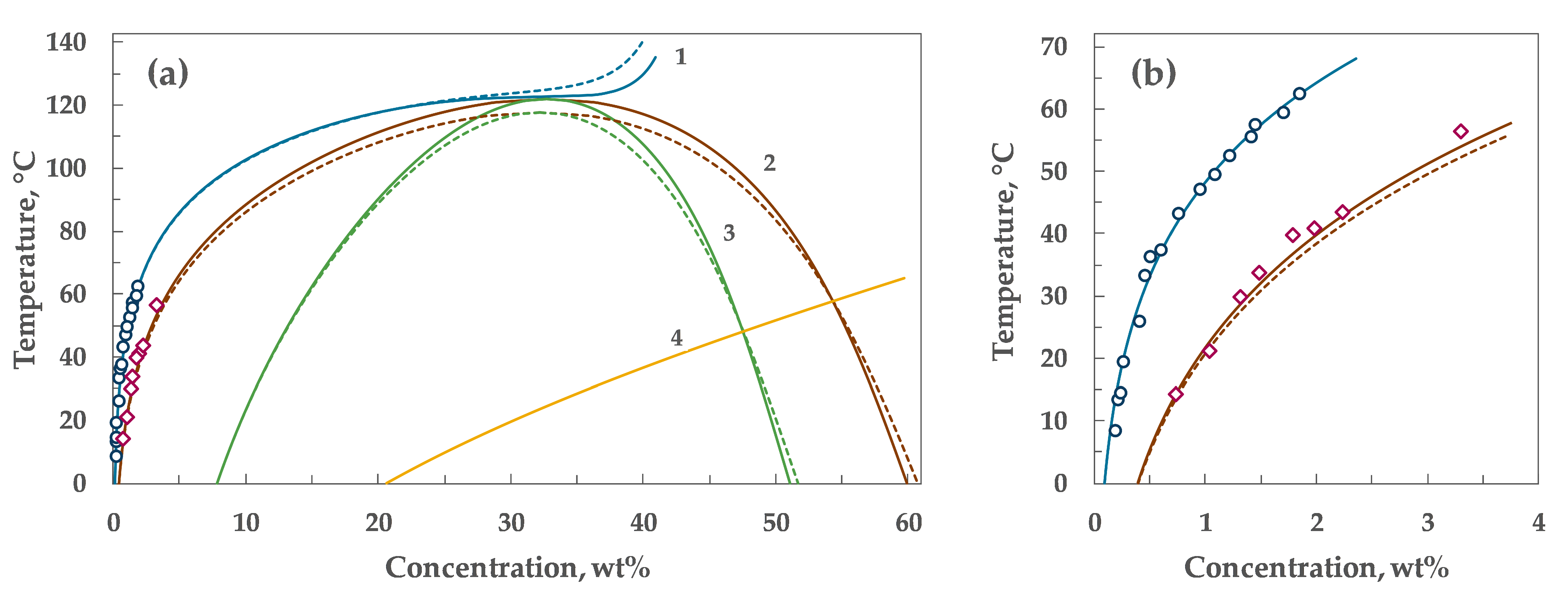
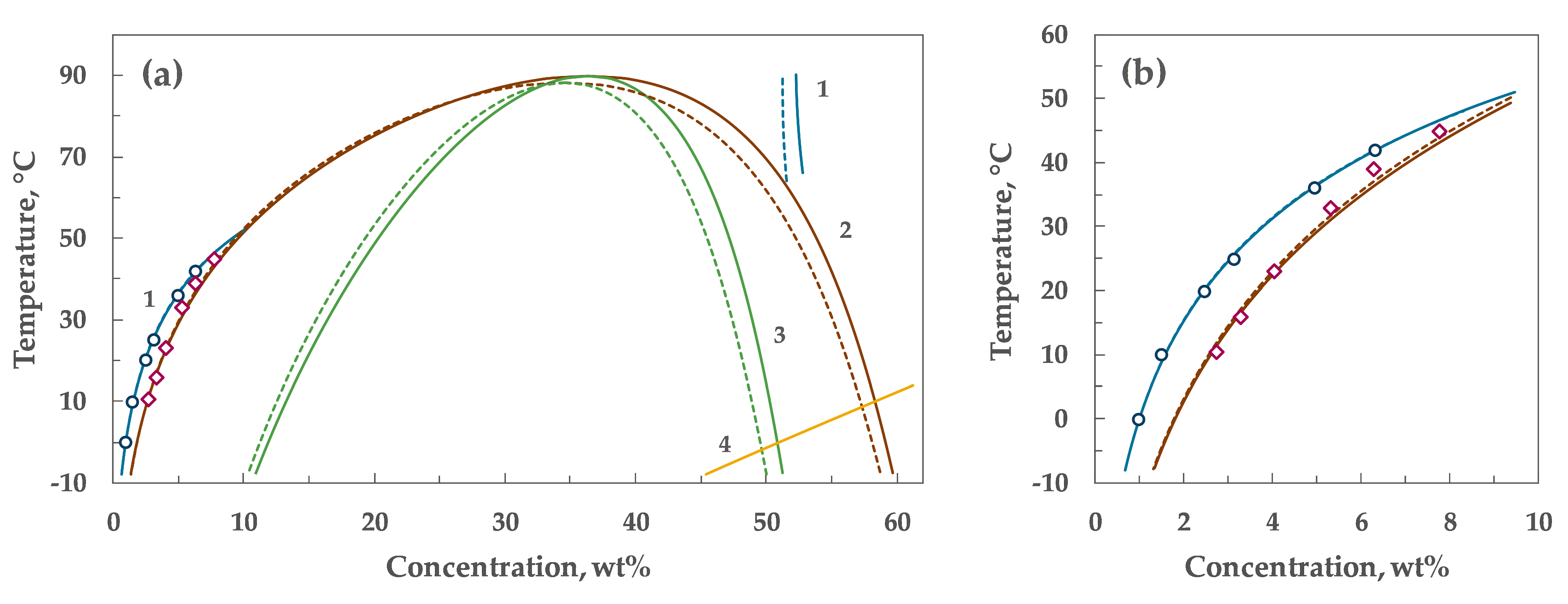
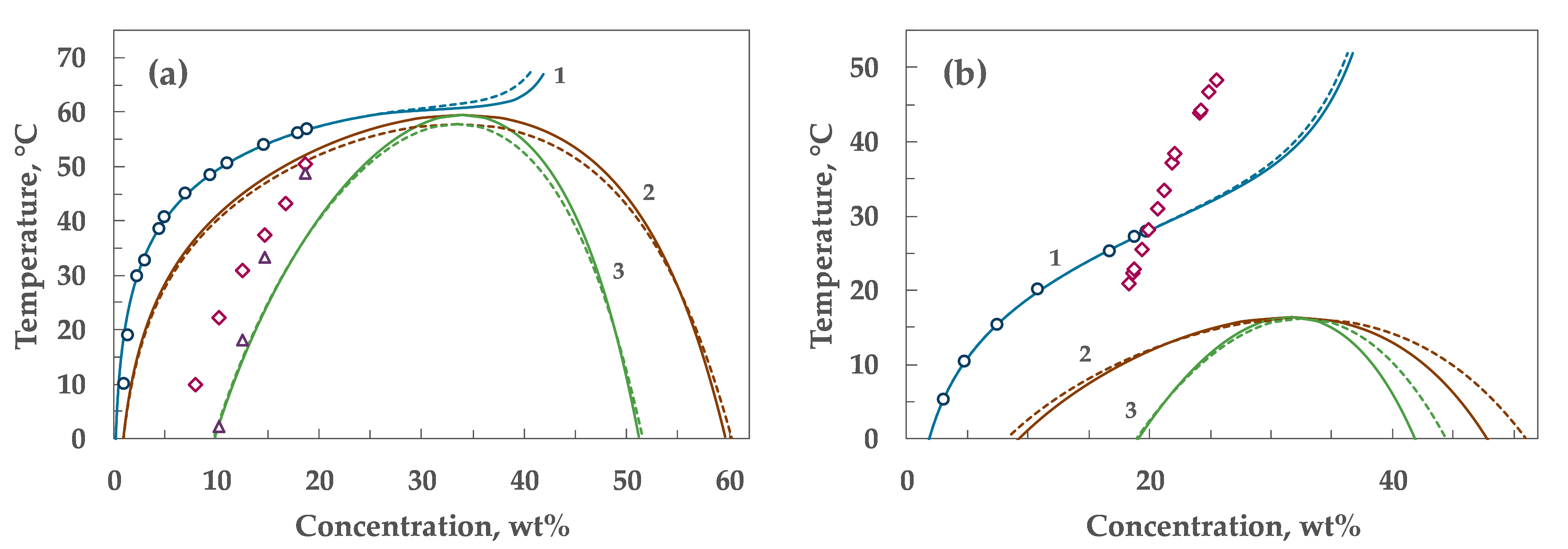
| α₀ | α₁ | λ | Tc, °C | Wc, wt% |
| Equation (11) | ||||
| 1399.4 | −3.3992 | 1.2866 | 36.63 | 37.45 |
| Equation (12) | ||||
| 1399.4 | −3.3985 | 1.2861 | 37.72 | 37.63 |
| α₀ | α₁ | λ | Tc, °C | Wc, wt% |
| Equation (11) | ||||
| 713.83 | −0.7412 | 1.3006 | 117.57 | 32.24 |
| Equation (12) | ||||
| 713.86 | −0.7413 | 1.3005 | 121.74 | 32.66 |
| α₀ | α₁ | λ | Tc, °C | Wc, wt% |
| Equation (11) | ||||
| 512.93 | −0.4744 | 1.3704 | 88.16 | 34.59 |
| Equation (12) | ||||
| 522.23 | −0.4830 | 1.3521 | 89.81 | 36.37 |
| α₀ | α₁ | λ | Tc, °C | Wc, wt% |
| C₃₅H₄₁Cl₂N₃O₂ – ethanol/water | ||||
| Equation (11) | ||||
| 995.26 | −1.9216 | 1.3002 | 57.68 | 33.44 |
| Equation (12) | ||||
| 1001.3 | −1.9373 | 1.2976 | 59.40 | 33.97 |
| Vanillin – 1-propanol/water | ||||
| Equation (11) | ||||
| 1056.0 | −2.3813 | 1.2284 | 16.15 | 32.49 |
| Equation (12) | ||||
| 1073.0 | −2.4226 | 1.2188 | 16.36 | 31.62 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bhamidi, V.; Abolins, B.P. A Thermodynamic Approach for the Prediction of Oiling Out Boundaries from Solubility Data. Processes 2019, 7, 577. https://doi.org/10.3390/pr7090577
Bhamidi V, Abolins BP. A Thermodynamic Approach for the Prediction of Oiling Out Boundaries from Solubility Data. Processes. 2019; 7(9):577. https://doi.org/10.3390/pr7090577
Chicago/Turabian StyleBhamidi, Venkateswarlu, and Brendan P. Abolins. 2019. "A Thermodynamic Approach for the Prediction of Oiling Out Boundaries from Solubility Data" Processes 7, no. 9: 577. https://doi.org/10.3390/pr7090577
APA StyleBhamidi, V., & Abolins, B. P. (2019). A Thermodynamic Approach for the Prediction of Oiling Out Boundaries from Solubility Data. Processes, 7(9), 577. https://doi.org/10.3390/pr7090577





