Abstract
Gas pressure changes during the process of coal mine gas drainage and CBM recovery. It is of great importance to understand the influence of sorption pressure on gas diffusion; however, the topic remains controversial in past studies. In this study, four samples with different coal ranks were collected and diffusion experiments were conducted under different pressures through the adsorption and desorption processes. Three widely used models, i.e., the unipore diffusion (UD) model, the bidisperse diffusion (BD) model and the dispersive diffusion (DD) model, were adopted to compare the applicability and to calculate the diffusion coefficients. Results show that for all coal ranks, the BD model and DD model can match the experimental results better than the UD model. Concerning the fast diffusion coefficient Dae of the BD model, three samples display a decreasing trend with increasing gas pressure while the other sample shows a V-type trend. The slow diffusion coefficient Die of BD model increases with gas pressure for all samples, while the ratio β is an intrinsic character of coal and remains constant. For the DD model, the characteristic rate parameter kΦ does not change sharply and the stretching parameter α increases with gas pressure. Both Dae and Die are in proportion to kΦ, which reflect the diffusion rate of gas in the coal. The impacts of pore characteristic on gas diffusion were also analyzed. Although pore size distributions and specific surface areas are different in the four coal samples, correlations are not apparent between pore characteristic and diffusion coefficients.
1. Introduction
During the process of coal mine gas drainage and CBM recovery, the gas flow process can be divided into two stages. First, driven by the concentration gradient force, the gas adsorbed on the surface of coal matrix desorbs and then diffuses into the fracture/cleat system of coal. Second, the dissociative state gas permeates to the surface well or the underground borehole driven by the pressure gradient force. Therefore, two key factors that affect net gas movement result are the gas diffusion coefficient and the gas permeability in the fracture. The diffusion coefficient represents the essential parameter of diffusibility and related studies show that it could be affected by temperature [1,2], moisture [3], pressure [4,5], gas type [6,7,8], sample size [6,9,10], and coal sample features [11,12]. It should be noted that the coal seam gas pressure is in the dynamic condition during gas extraction. Hence, it is of great significance to understand the impact of pressure on the gas diffusion coefficient. Several research papers on this topic have been conducted but arguments can be found on how gas pressure impacts the diffusion coefficients.
For example, some scholars believe that the diffusion coefficient is in direct proportion to gas pressure. Charrière et al. [2] used CH4 and CO2 to conduct the adsorption kinetics experiments when the pressure is equal to 0.1 MPa and 5 MPa respectively. They found that the diffusion coefficient increases with gas pressure. Pan et al. [3] performed CH4 adsorption/desorption diffusion test within 0~4 MPa pressures range, and results show a direct ratio between diffusion coefficient and gas pressure. Jian et al. [13] carried out the desorption experiments within 0~4.68 MPa pressure range and the conclusion remains the same. However, some scholars reckon that the diffusion coefficient decreases with the increase in pressure. Cui et al. [8] found that the diffusion coefficient of CO2 reduces when gas pressure is smaller than 3.6 MPa. Staib et al. [4] conducted the adsorption kinetics experiments and analyzed the results using the BD model. It was found that the diffusion coefficient Da lowers when the pressure increases. Shi et al. [14] tested the influence of CO2 injection on microporous diffusion coefficient after the adsorption of CH4 was balanced. Findings show that the increasing injection pressure of CO2 would cause the reduction of micropore diffusion coefficient. There are also a few scholars who concluded that gas pressure has small effects on the diffusion coefficient. Nandi et al. [15] conducted CH4 adsorption/desorption experiments on bituminous and anthracite coals and they did not find an apparent relationship between pressure and gas sorption rate. To summarize, the research outcomes are listed in Table 1.

Table 1.
Summary of diffusion coefficient changing trend with the increase in pressure.
By reviewing the previous studies, it can be concluded that the effect of pressure on gas diffusion coefficient remains controversial till now. It is difficult to compare the research outputs horizontally because of the diversified calculation models and experimental methods, such as experimental apparatus, gas pressure and gas type. Moreover, most of the coal samples used in the studies was bituminous coal, because gas diffusibility and diffusion coefficient in coal are closely correlated with the types of coals. It is unknown whether the results stand for coal with different metamorphic grades.
In the present study, we aim at investigating the influence of sorption pressure on gas diffusion and examining which previous finding is more convincible. To guarantee the comparability of the results, all the experiments are carried out under similar gas pressure range. Both adsorption and desorption kinetics test are conducted. Three widely used diffusion models are adopted to analyze the results and eliminate the possible differences induced by diffusion models. Four coal samples with different ranks are collected from typical mining areas in China, the test results are cross-compared to understand does the coal rank have impacts on the relationship between gas pressure and diffusion coefficients.
2. The Diffusion Models
Three widely used diffusion models, i.e., the unipore diffusion (UD) model, the bidisperse diffusion (BD) model and the dispersive diffusion (DD) model, will be used in this study. The expressions and the assumptions are introduced as follows.
2.1. The Unipore Diffusion Model
The UD [13,16,17] model assumes that the coal particle has only one type of pore and the gas diffuses under the concentration gradient between exterior and interior of the coal particle. The UD model is illustrated in Figure 1. Both UD model and BD model follow the following assumptions: (a) the diffusion system is isothermal; (b) the geometric shape of the particle coal is the standard sphere; (c) coal and pore system are isotropic and homogeneous; (d) the pores are incompressible; (e) gas follows the linear adsorption rule; (f) gas diffusion in pores is in line with Fick’s Second Law. It can be expressed as [18],
mt in the equation refers to the total gas adsorption/desorption quantity at time t, m∞ is the total quantity after the gas adsorption/desorption is balanced, r represents the radius of spherical coal particle, D refers to the diffusion coefficient (m2/s) and the value of is defined as the effective diffusion coefficient De (1/s).
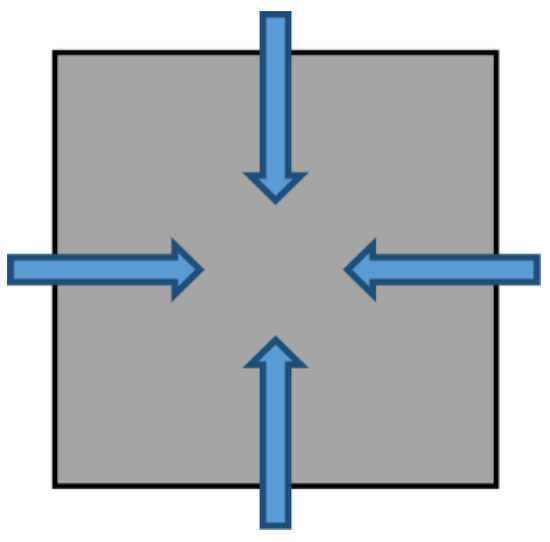
Figure 1.
Concepts of gas diffusion under unipore diffusion (UD) Model [19].
2.2. The Bidisperse Diffusion Model
The BD model [5,8,9,14,20,21] assumes that the coal particle includes independent macropore and micropore systems, which are represented by Da and Di respectively. The gas diffusion under the two systems are driven by the concentration gradients between exterior and interior of the coal particle. The BD model is illustrated in Figure 2. The simplified BD model includes the fast macropore diffusion stage and the slow micropore diffusion stage [5,22].
Figure 2.
Concepts of gas diffusion under bidisperse diffusion (BD) Model [19].
Concerning the fast macropore diffusion stage, the diffusion model is denoted as,
ma in the equation refers to the total gas adsorption/desorption quantity at time t in the macropore, ra and Da represent the radius of macropore spherical coal particle and macropore diffusion coefficient (m2/s), respectively. The value of is defined as the effective diffusion coefficient Dae (1/s).
Concerning the lower micropore diffusion stage, the diffusion model is denoted as,
mi in the equation refers to the total gas adsorption/desorption quantity in the micropore at time t, ri and Di represent the radius of micropore spherical coal particle and micropore diffusion coefficient (m2/s), respectively. The value of is defined as the effective diffusion coefficient Die (1/s).
The BD model is expressed as,
is the ratio of macropore adsorption/desorption to the total adsorption/desorption.
2.3. The Dispersive Diffusion Model
In recent years, the dispersive diffusion model was developed and it assumes that a distribution of characteristic times for diffusion. The diffusion is dispersed and represents the wide distribution of diffusion feature time. Therefore, theoretically, the DD model can avoid the simplification of pore structure and reflect the real physical experimental process. The DD model is expressed as,
mt in the equation refers to the total gas adsorption/desorption quantity, m∞ is the total quantity after the gas adsorption/desorption is balanced, kΦ is the characteristic rate parameter, α is the stretching parameter (0 < α < 1). The research of Staib et al. [23] shows that α is an intrinsic property of coal and is greatly influenced by the coal pore characteristic.
3. Diffusion Experiments
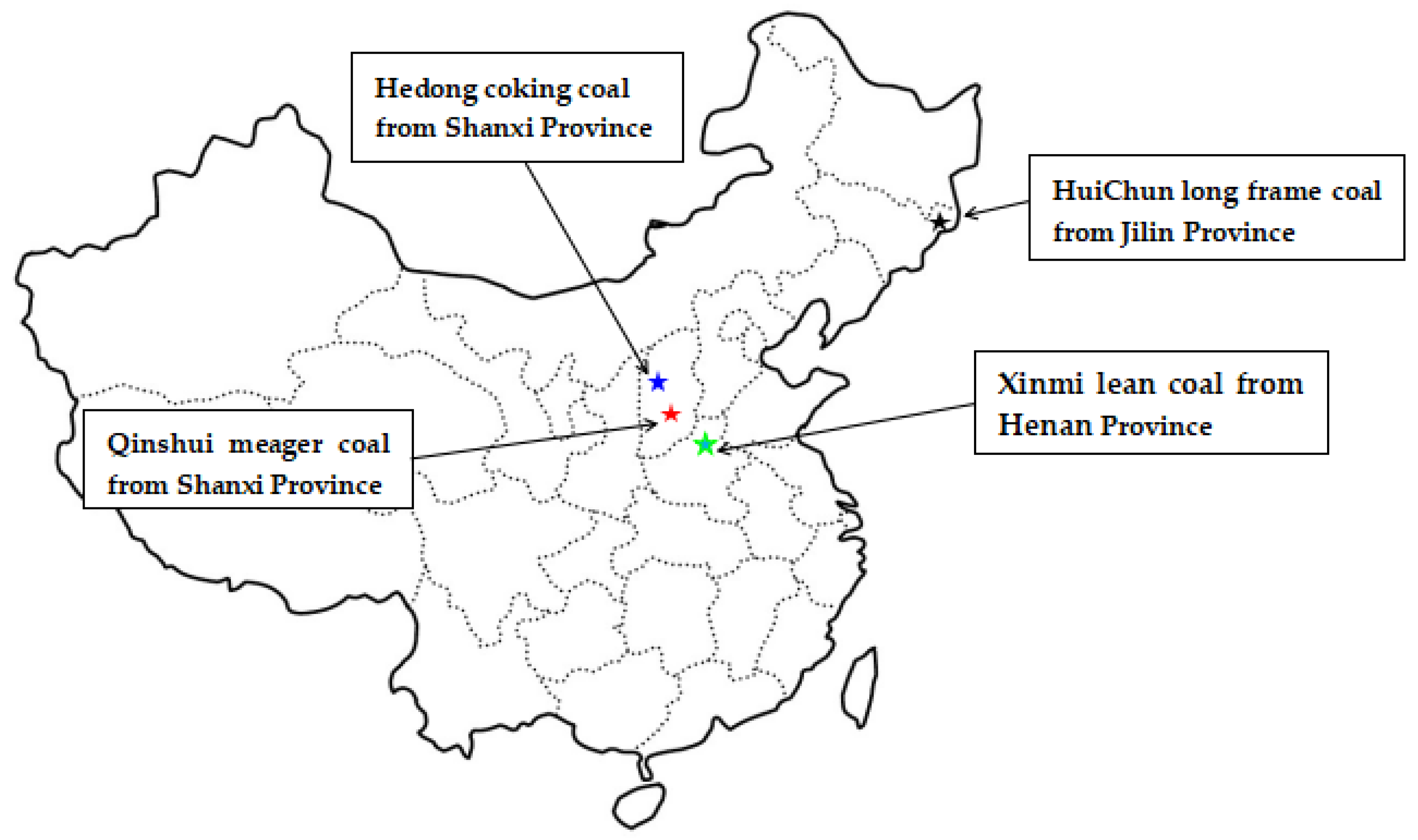
To carry out the diffusion experiments and analyze the impact of pressure on the methane diffusion, the iSorb HP (Quantachrome) instrument was used. The set maximum adsorption pressure is 6 MPa, the coal sample quality is 40 g and the experimental temperature is 315 K. Coal samples were collected from the HuiChun long frame coal at Jilin Province, Hedong coking coal at Shanxi Province, Xinmi lean coal at Henan Province, and Qinshui meager coal at Shanxi Province (Figure 3), with the four coal samples are denoted as HC, HD, XM and QS, respectively. The coal samples were ground into 0.2~0.25 mm particle samples and the prepared coal particles were dried under the 100 °C vacuum state for 24 h to remove moisture. The proximate analysis results are shown in Table 2.
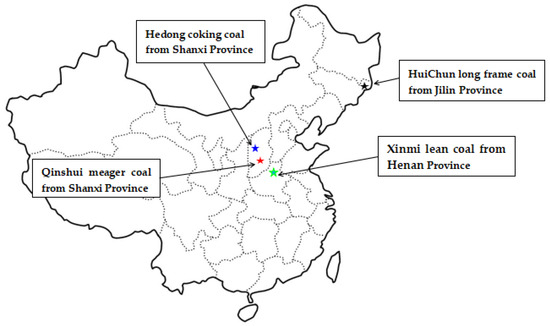
Figure 3.
The diagram of coal samples collection places.

Table 2.
Proximate analysis results.
Manometric method is used in the experiments and the methane isothermal adsorption and diffusion kinetics are tested [24]. The gas state equation that implies the void volume of reference tank and coal samples tank is,
Four series data were recorded by pressure sensor in the experiments; (a) gas pressure in the reference tank before the gas is injected into the coal samples tank, Pm1; (b) gas pressure in the reference tank after the gas is injected into the coal samples tank, Pm2; (c) gas pressure in the coal samples tank before the reference tank gas is injected, Pc1; (d) gas pressure in the coal samples tank after the reference tank gas is injected, Pc2. Pm1, Pm2 and Pc1 are constant while Pc2 is flexible.
The adsorption gas quantity at the i pressure step and time t in the adsorption diffusion process is,
Therefore, the adsorption diffusion ratio of each pressure step is,
∞ in the equation represents the required time when the i pressure step is balanced.
While the total adsorption quantity at the pressure balance point is,
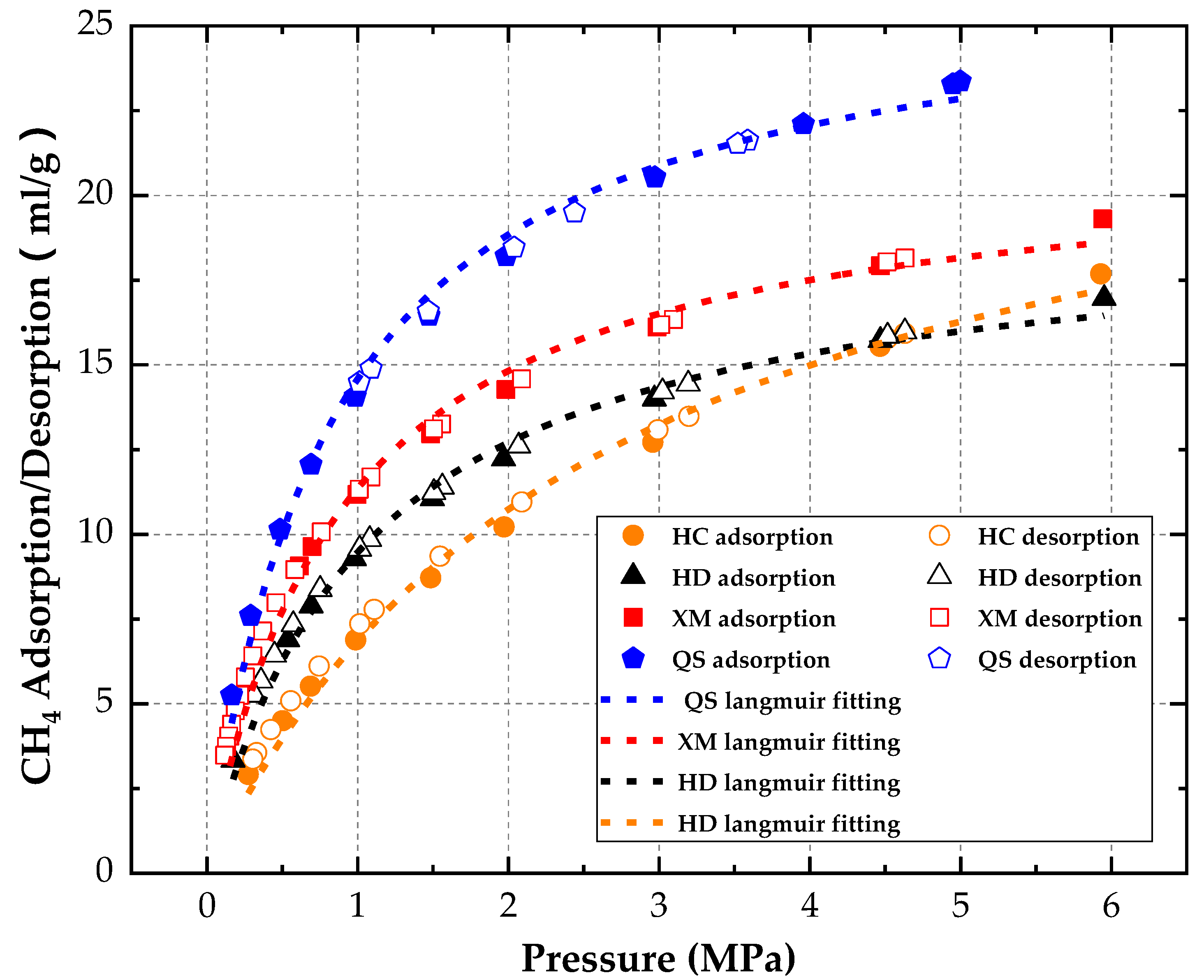
Therefore, is equal to the gas adsorption quantity at the i pressure step and time t divided by the gas adsorption quantity when the i pressure step is balanced. Formula (9) is used to calculate the adsorption gas quantity at each balanced gas point and the adsorption/desorption isothermal lines [19] of coal samples are shown in Figure 4.
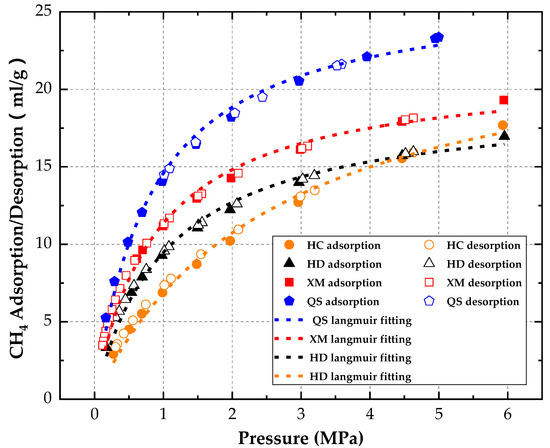
Figure 4.
Adsorption-desorption isotherm of coal samples.
Langmuir model (Equation (10)) is used to fit the adsorption and desorption data of CH4 and the correlation R2 is listed in Table 3. It can be seen that the adsorption and desorption characteristics of CH4 are represented well by the Langmuir model. The adsorption characteristic parameters are calculated in Table 3.
a, b in the equation are the adsorption characteristic parameters. a represents the Langmuir adsorption quantity and b refers to the Langmuir adsorption pressure. X is the adsorbed gas quantity and P refers to the gas pressure.

Table 3.
Adsorption characteristic parameters of coal samples.
Overall, similar to the previous findings [25,26], no apparent hysteresis phenomenon is found in the absorption/desorption process, in other words, the absorption/desorption process of CH4 can be reversed.
4. Analysis and Discussion
4.1. Model Applicability Analysis
Based on the Equation (8), the diffusion ratio can be calculated at any moment. The approximate method was used to fit the experimental results when applying the UD and BD models. In the process of fitting the UD and BD models, findings show that the calculation results are adequately convergent when the infinite series n is expanded to the fifth term. This can ensure the accuracy of model fitting results and further reduces the calculation difficulties. Therefore, all data was processed by setting n expand to five as the standard for calculation.
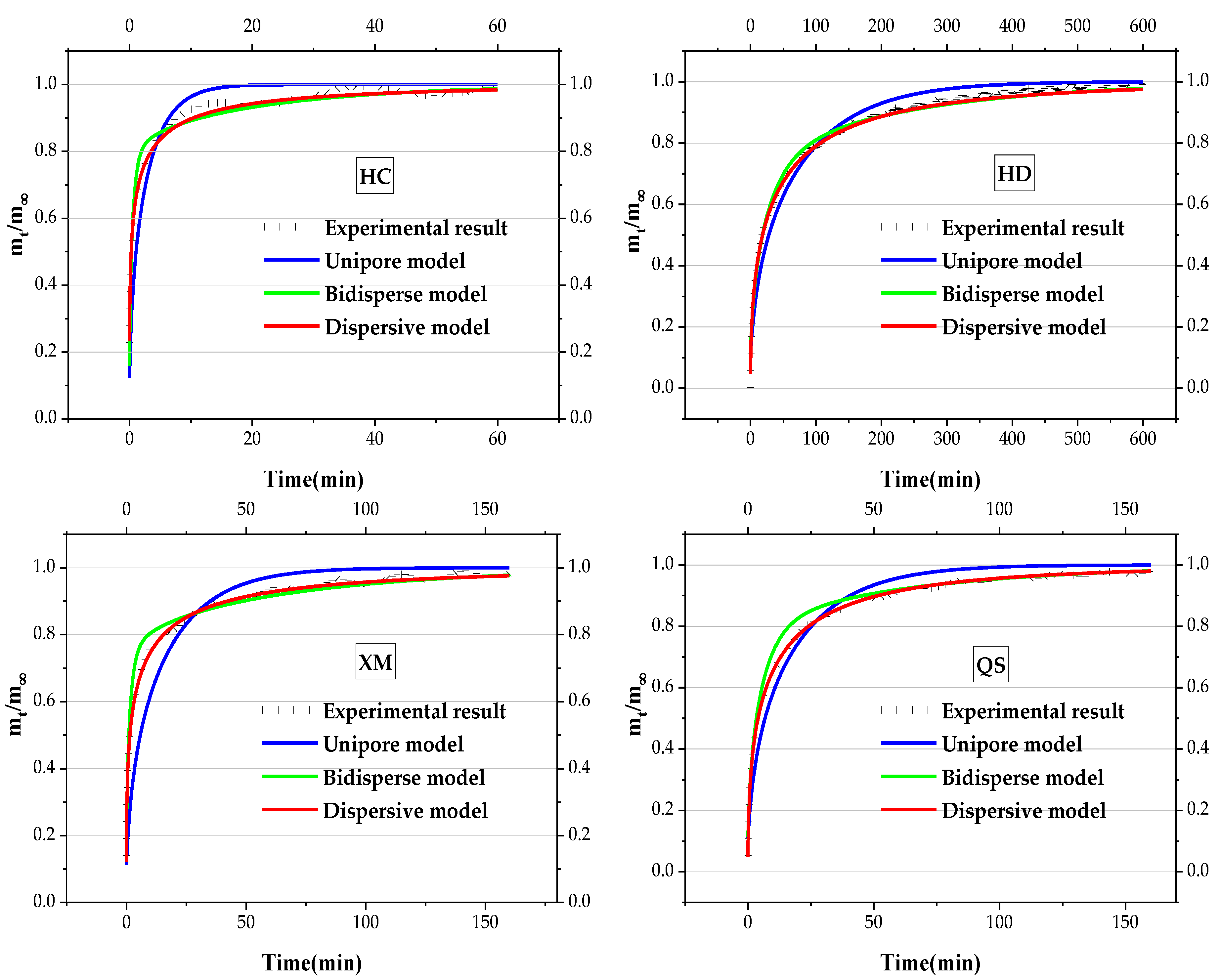
Taking the gas balance pressure increases from 0.7 MPa to 1 MPa as an example, the experimental results and the fitting diffusion lines of coal samples are shown in Figure 5.
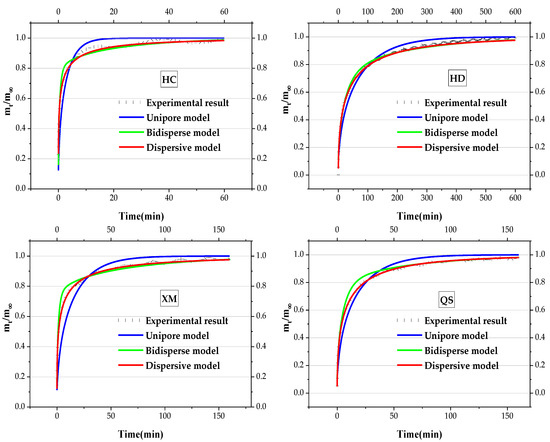
Figure 5.
Experimental results and fitting diffusion lines of coal samples.
It is shown in Figure 5 that the fitting line by the UD model is below the experimental line before some critical moment regardless of the coal samples, indicating that the fitting value is smaller than the actual value. After a certain time, the fitting line keeps above the experimental line and implies that fitting value is larger than the actual value. Therefore, the experimental results cannot be restored by the fitting line regardless of moderating the UD coefficient. The fitting effect of the BD model is superior to the UD model and the fitting line of the DD model is closer to the actual line. It suggests that the whole gas adsorption/desorption process cannot be accurately described by the UD model due to the complicated pore structures. The fitting line through the BD model includes the double structure of macropore and micropore and thus shows a higher coincidence degree with the experimental results. The DD model shows the best coincidence degree with the real experimental results. Therefore, the BD model and DD model are selected to calculate the gas diffusion characteristic parameter.
4.2. Analysis of Pressure’s Effect on the Gas Diffusion
(1) The BD Model
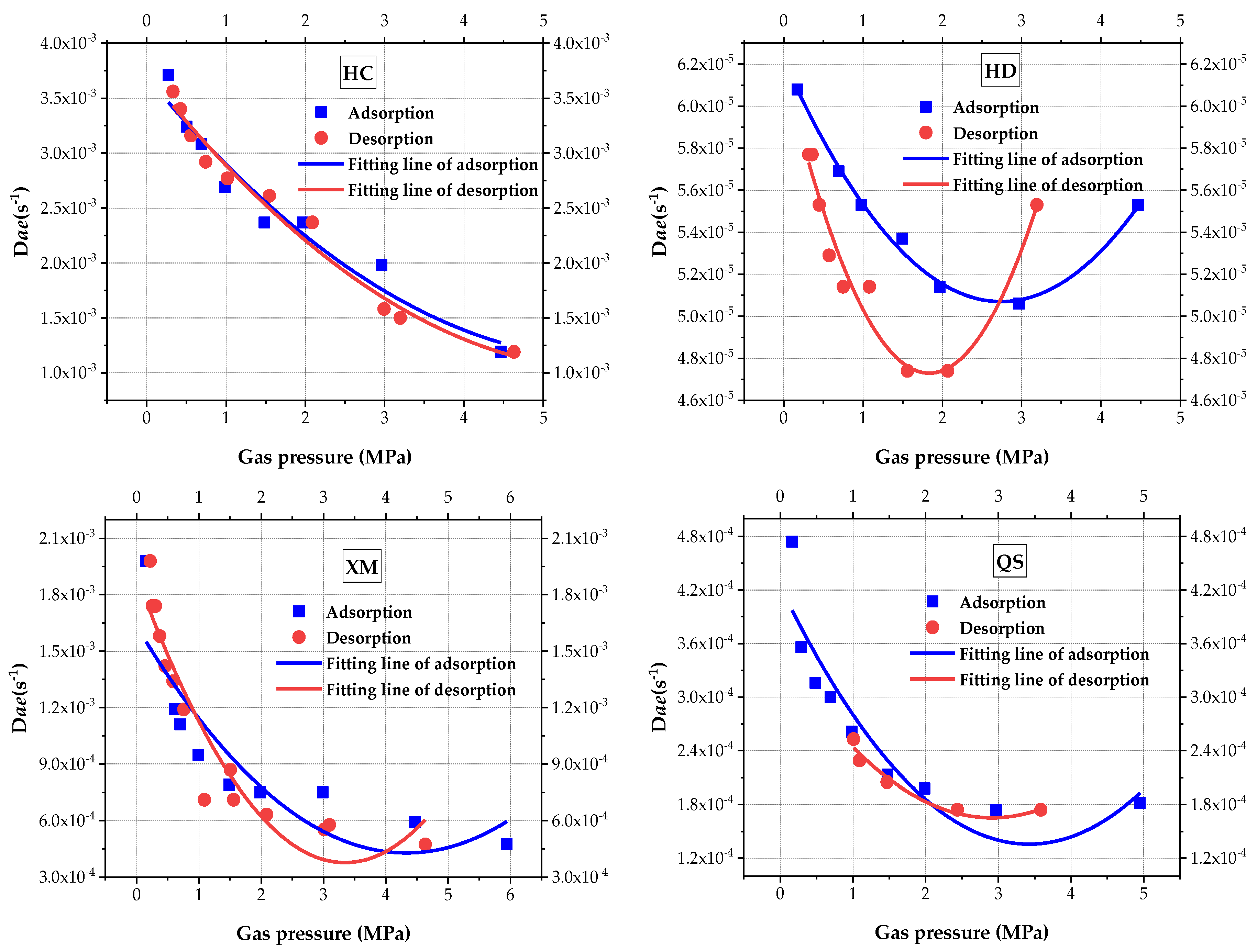
Equation (4) implies that the BD model includes three unknown parameters, including fast effective diffusion coefficient Dae, slow effective diffusion coefficient Die and the ratio of macropore adsorption/desorption to the total adsorption/desorption β. Using Equation (4) to calculate the BD characteristic parameters and analyze the impact of pressure on Dae (Figure 6) and quadratic polynomial is to fit the results, the fitting goodness and calculated coefficient are shown in Table 4. As can be seen from Figure 6, the macropore diffusion coefficient Dae decreases with the increase in pressure in three out of four sample coals (i.e, HC, XM and QS). Concerning the HD, Dae shows a V-shape trend, which first decreases and then increases as the increases in pressure. Figure 6 also shows that the impact law of pressure on Dae is better illustrated by the quadratic polynomial. When comparing the values of Dae, in both the adsorption and desorption processes, Dae(HC) > Dae(XM) > Dae(QS) > Dae(HD). The difference of Dae in the absorption versus the desorption process becomes larger from HC to QS. No significant increasing trend of HC, XM and QS is found when the pressure increases, It is suspected that the set maximum pressure is not in the threshold level.
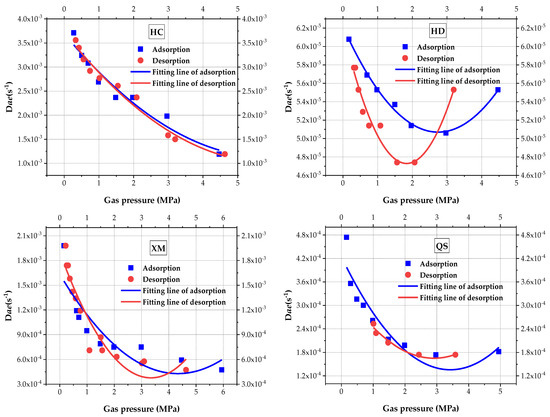
Figure 6.
The diagram of the variation of macro-diffusion coefficients with pressure.

Table 4.
Goodness of fit and diffusion coefficient.
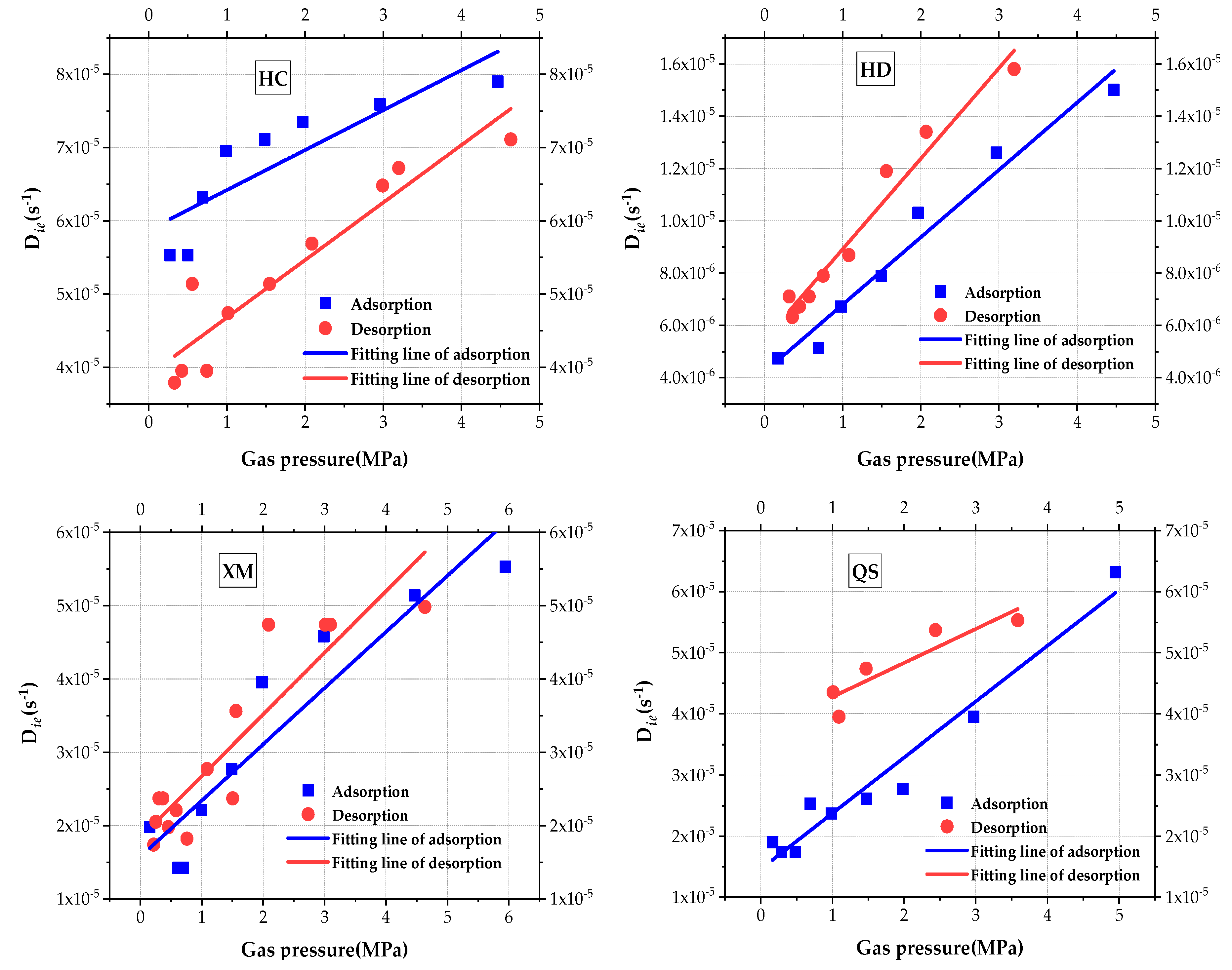
The impact of gas pressure on Die is analyzed and is shown in Figure 7. Linear regression is used to fit the results and, results of the fitting goodness and calculated coefficient are given in Table 5. It can be clearly seen that the slow efficient diffusion coefficient Die increases with the increase in pressure for all four samples. The impact law of pressure on Die is better explained by the linear regression. When comparing the values of Die, the order is Die(HC) > Die(XM) > Die(QS) > Die(HD) in the adsorption process and Die(HC) > Die(QS) > Die(XM) > Die(HD) in the desorption process.
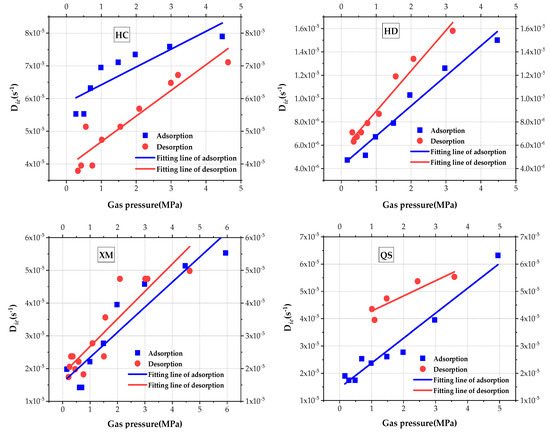
Figure 7.
The diagram of variation of micro-diffusion coefficient with pressure.

Table 5.
Goodness of fit and diffusion coefficient.
The calculation results show that β is 0.74~0.76 for HC, 0.58~0.6 for HD, 0.67~0.69 for XM and 0.69~0.7 for QS, respectively, implying that the diffusion characteristic parameter β keeps constant in the adsorption/desorption process. This further indicates that β which represents the intrinsic property would not show a significant fluctuation with the change in pressure.
(2) The DD Model
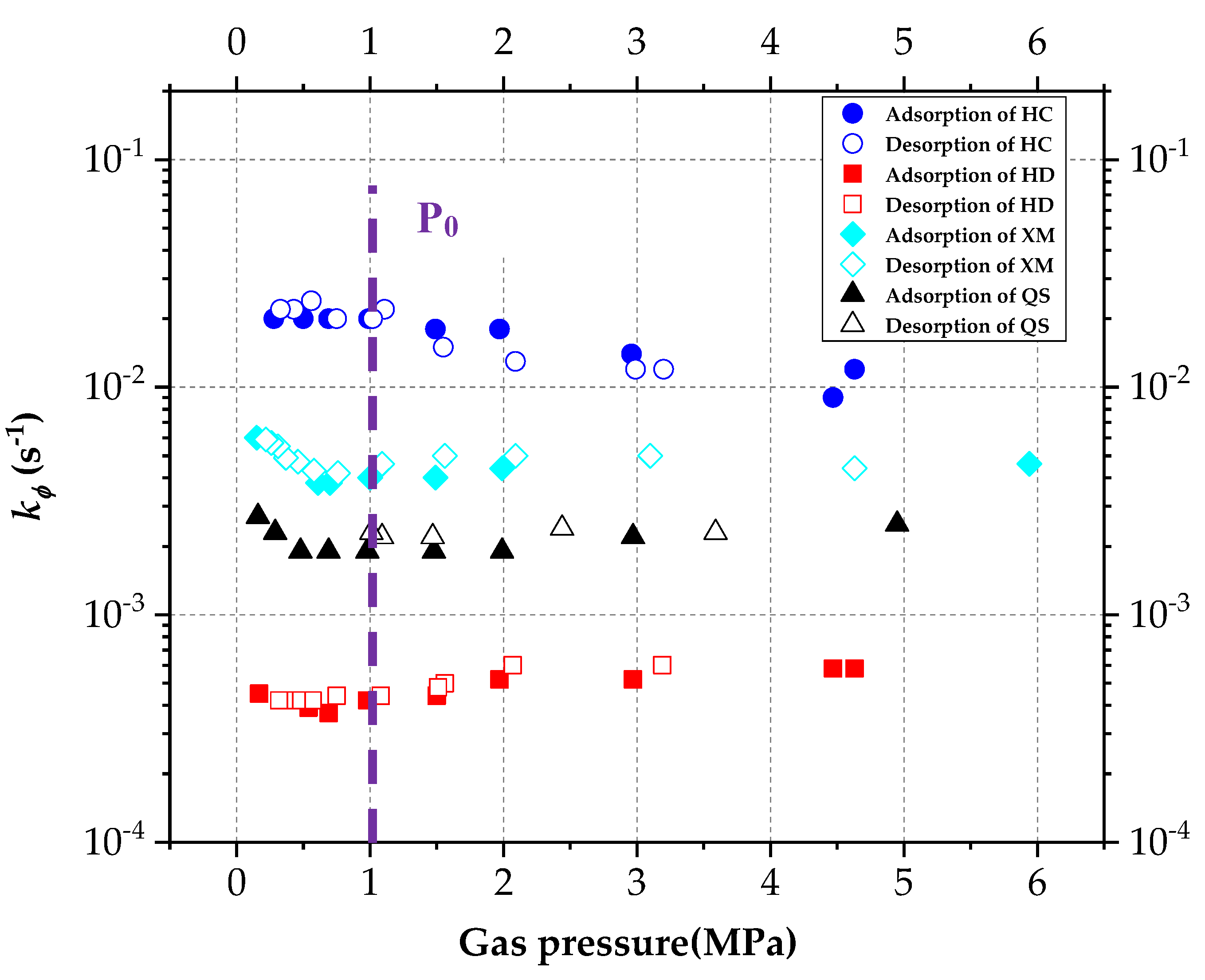
The DD model includes two unknown characteristic parameters, the characteristic rate parameter kΦ and the stretching parameter α. The influencing law of pressure on the kΦ and α are re-analyzed, and calculated based on the gas diffusion experimental results and Equation (5). The results are shown in Figure 8 and Figure 9, respectively.
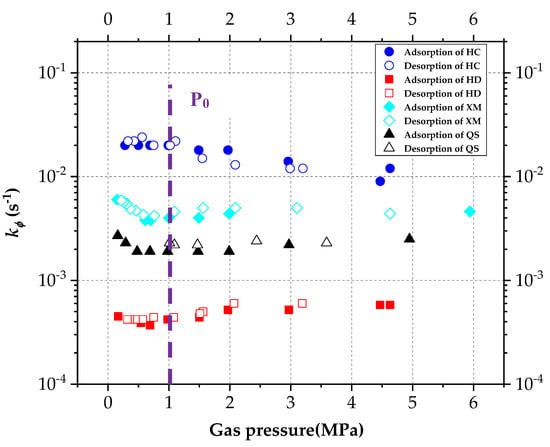
Figure 8.
The diagram of variation of characteristic rate parameter kΦ with pressure.
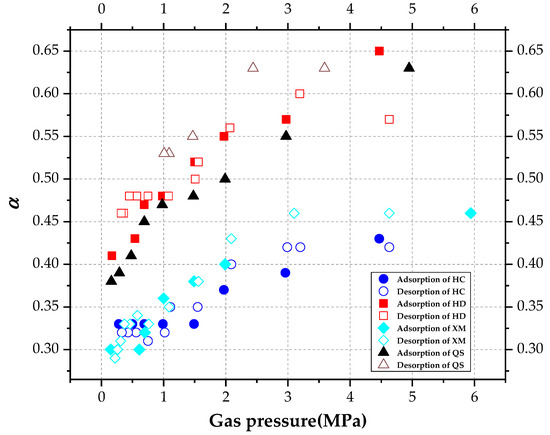
Figure 9.
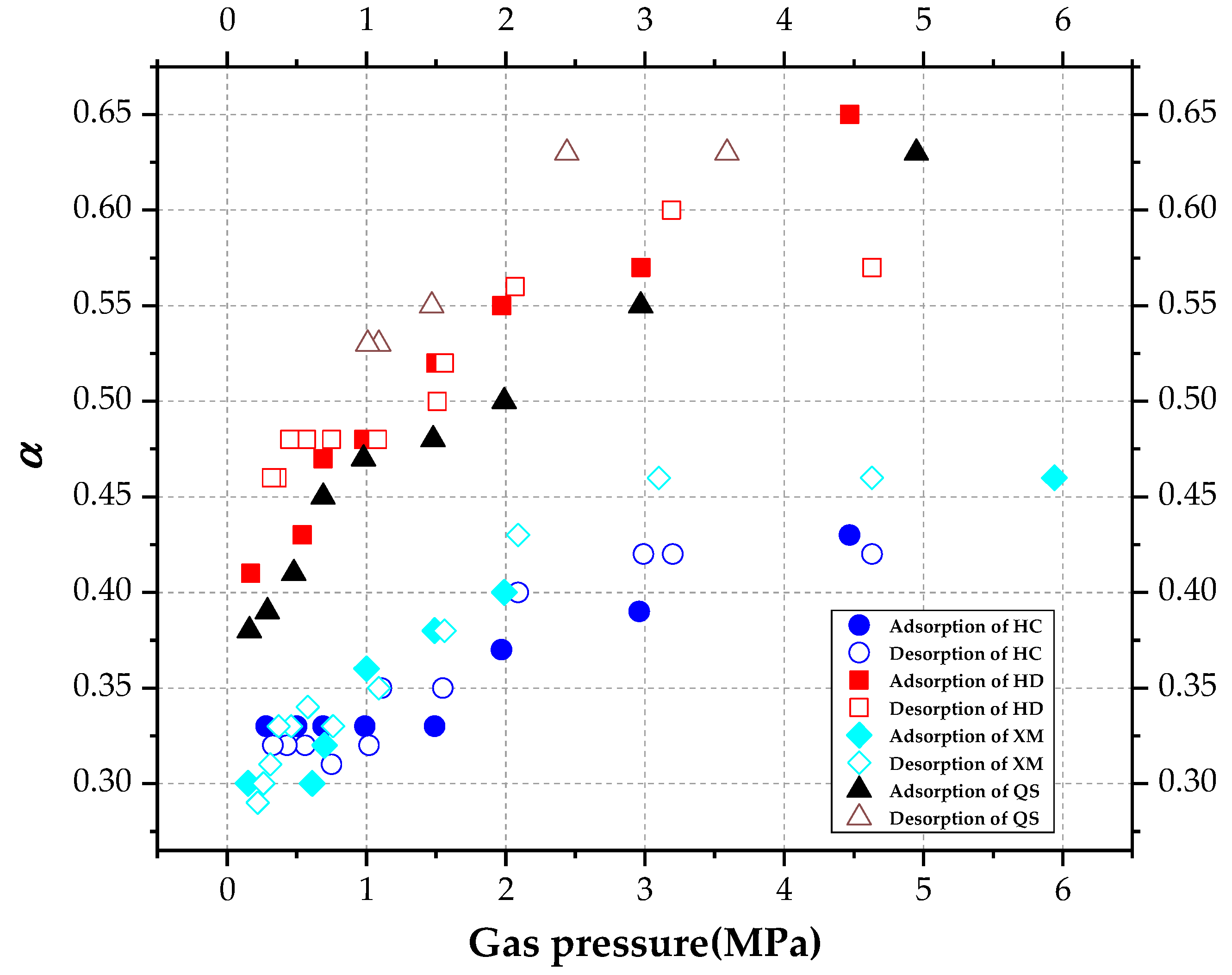
The diagram of variation of stretching parameter α with pressure.
Gregory Staib et al. [23,27] found that kΦ decreases with the increase in pressure which ranges from 0~3 MPa in their studies. In terms of the vitrinite-rich coal samples, α increases with gas pressure while for the inertinite-rich coal samples, no significant changing trend is found for α. Figure 8 shows that in our study, kΦ keeps unchanged in the pressure fluctuation process. Concerning XM and QS, kΦ slightly decreases with the increase in pressure when the pressure is less than P0, but it keeps constant while the pressure is larger than P0.
As shown in Figure 9, α increases with pressure. The mean values of α were calculated in Table 6. The mean value of α is ordered as α(HD) > α(QS) > α(XM) > α(HC) in the absorption process, while the order is α(QS) > α(HD) > α(HC) > α(HM) in the desorption process.

Table 6.
Stretching parameter α.
(3) Analysis of the correlation of diffusion characteristics parameters
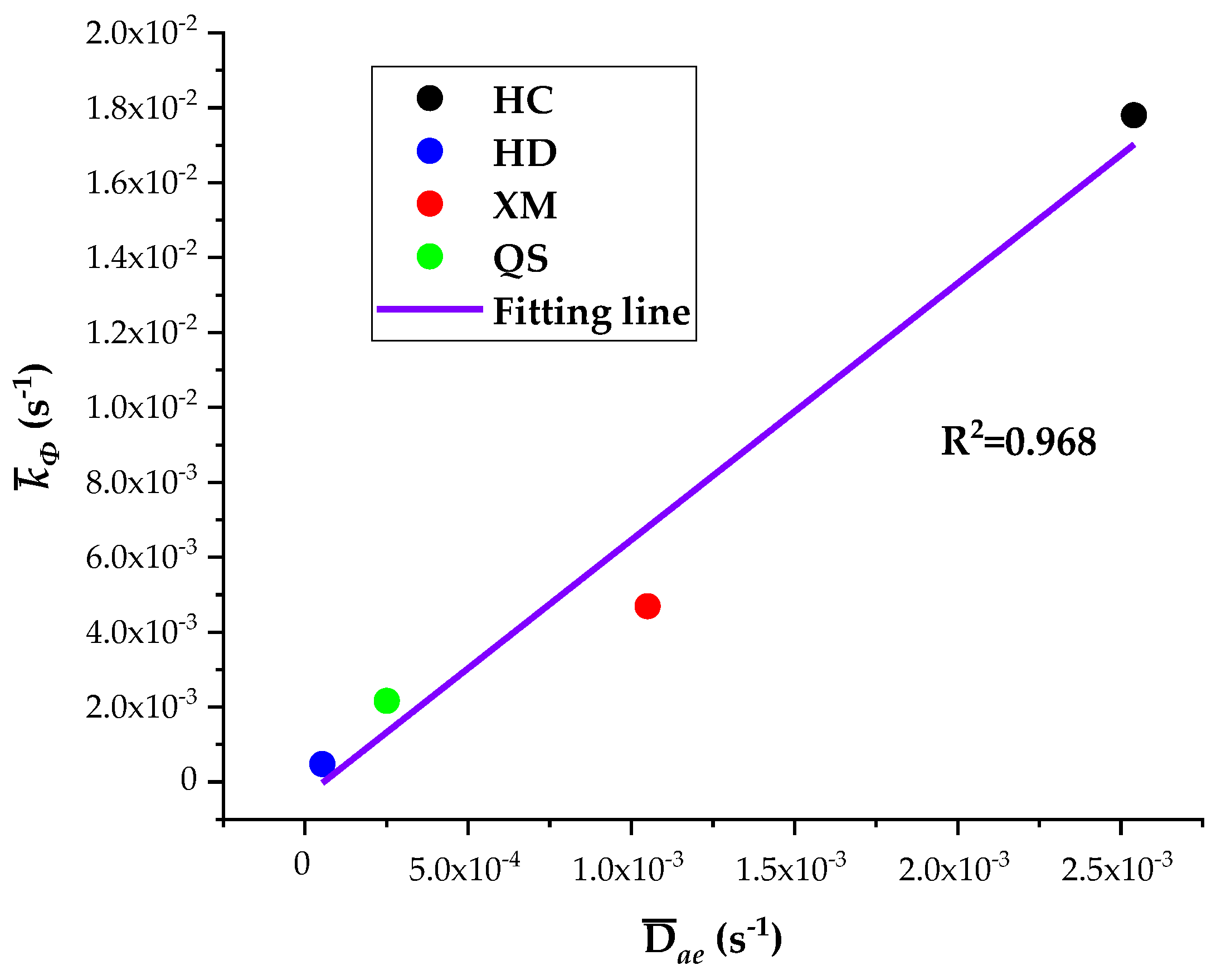
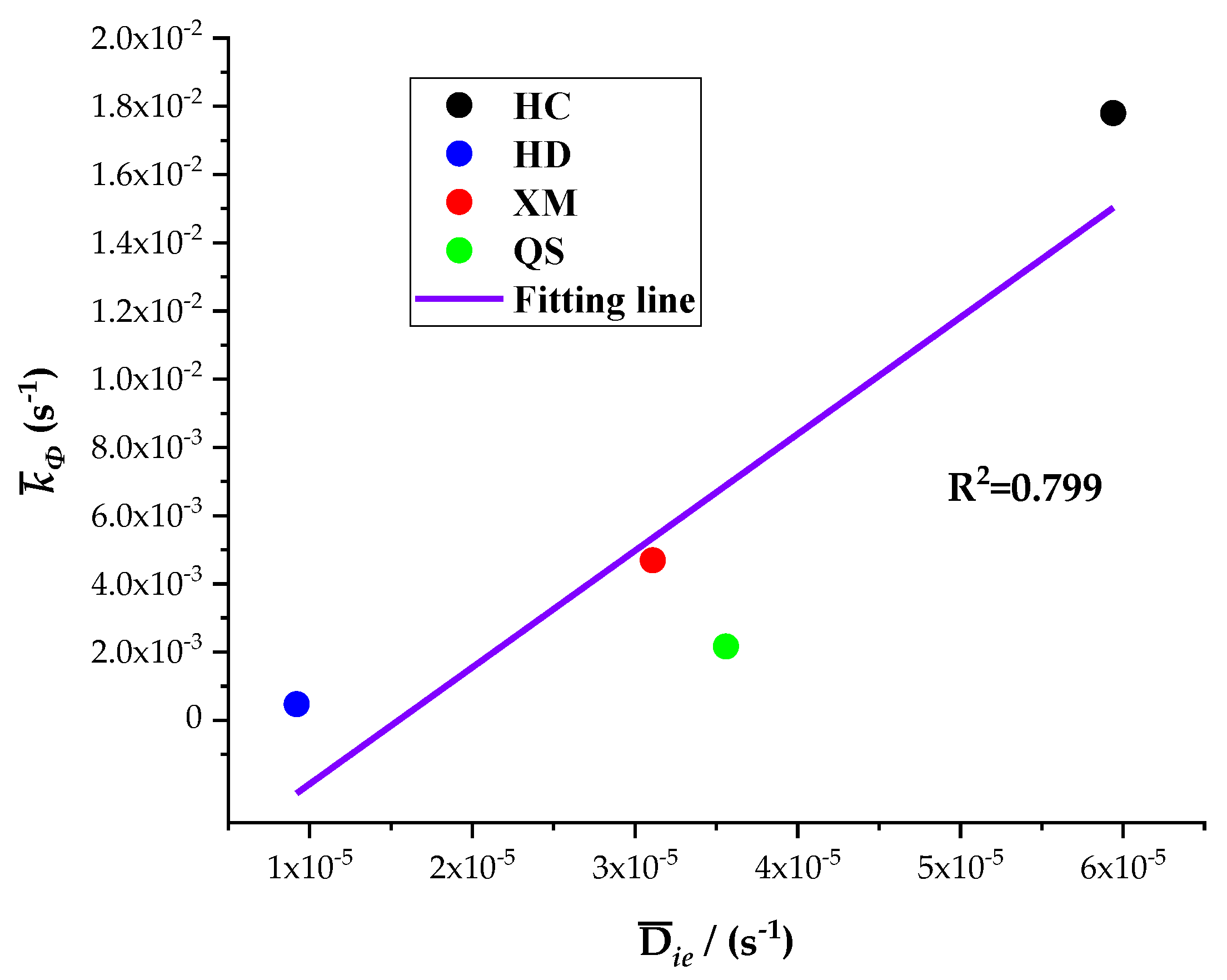
The five diffusion characteristics parameters of the BD and DD models are treated by the homogenization procedure and the results are shown in Table 7. It can be seen that Dae, Die and kΦ are the largest for HC, in the middle for XM and QS, and the smallest for HD. The linear regression results of kΦ on Dae and kΦ on Die are shown in Figure 10 and Figure 11, respectively.

Table 7.
Average gas diffusion parameters of experimental coal samples.
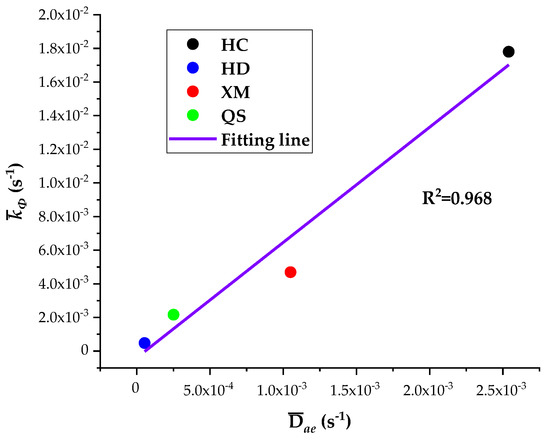
Figure 10.
The linear regression of kΦ on Dae.
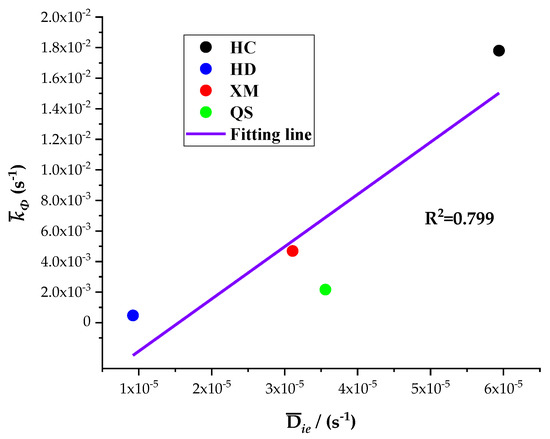
Figure 11.
The linear regression of kΦ on Die.
The results show a good linear correlation of Dae, Die and kΦ in our experimental results, and the goodness of fit is the best for Dae and kΦ. It suggests that both the diffusion coefficients Dae and Die and characteristic rate parameter kΦ are suitable for describing the coal gas diffusion rate. The analysis above suggests that the fast diffusion coefficient Dae decreases with the increase in pressure while the slow diffusion coefficient Die increases with the increase in pressure. kΦ keeps fixed and thus may be considered as a combined effect of Dae and Die.
4.3. Analysis of the Relationship between Pore Structure Characteristics and Gas Diffusion
By analyzing and summarizing the impact, law of CH4 diffusion under different pressures, we found the diversity of gas diffusion coefficients in both absorption and desorption process for different coal samples. Because the coal pore structures might directly affect the diffusion process of gas [28], experiments on the low temperature nitrogen absorption and mercury penetration were conducted to test the characteristics of coal pore structure.
The Quadrasorb instrument is used for the low temperature nitrogen absorption experiment and the PoreMaster60 mercury porosimeter instrument is applied for the mercury penetration. coal samples particles with 1~3 mm in size are prepared and dried. The low temperature nitrogen absorption method is suitable for testing the distribution of coal micropore ranging from 0~25 nm, which determines the coal specific surface area [28]. Because the mercury penetration method is not accurate in testing the micropore, it is only suitable for analyzing the pores which are bigger than 25 nm. Therefore, in this study, the computation of pore volume is calculated by the low temperature nitrogen absorption method when the pore size ranges from 0~25 nm and by the mercury penetration method if the pore size is bigger than 25 nm. The specific surface area and pore volume are given in Table 8 and Table 9, respectively.

Table 8.
Specific surface area of coal samples.

Table 9.
Pore volume of coal samples.
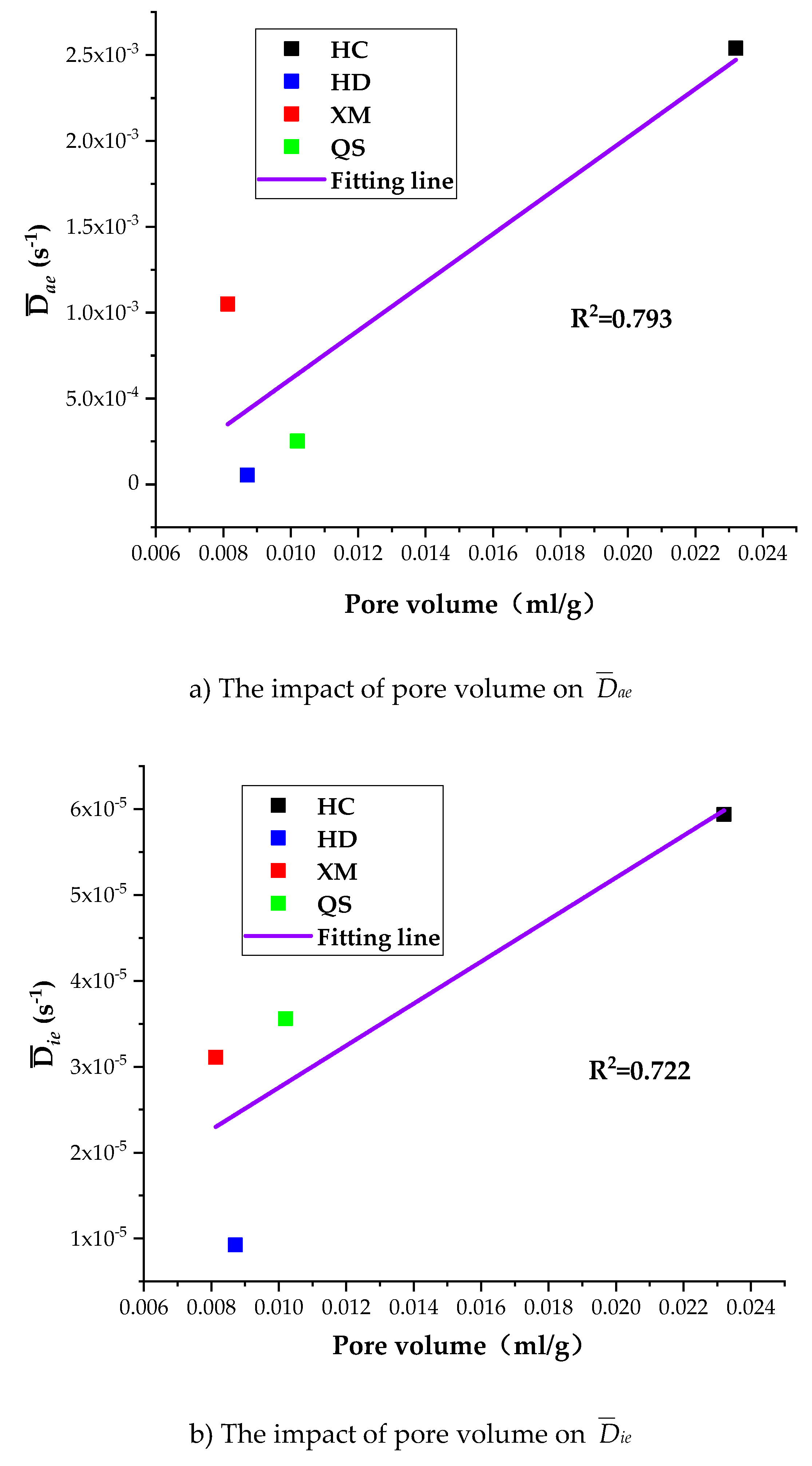
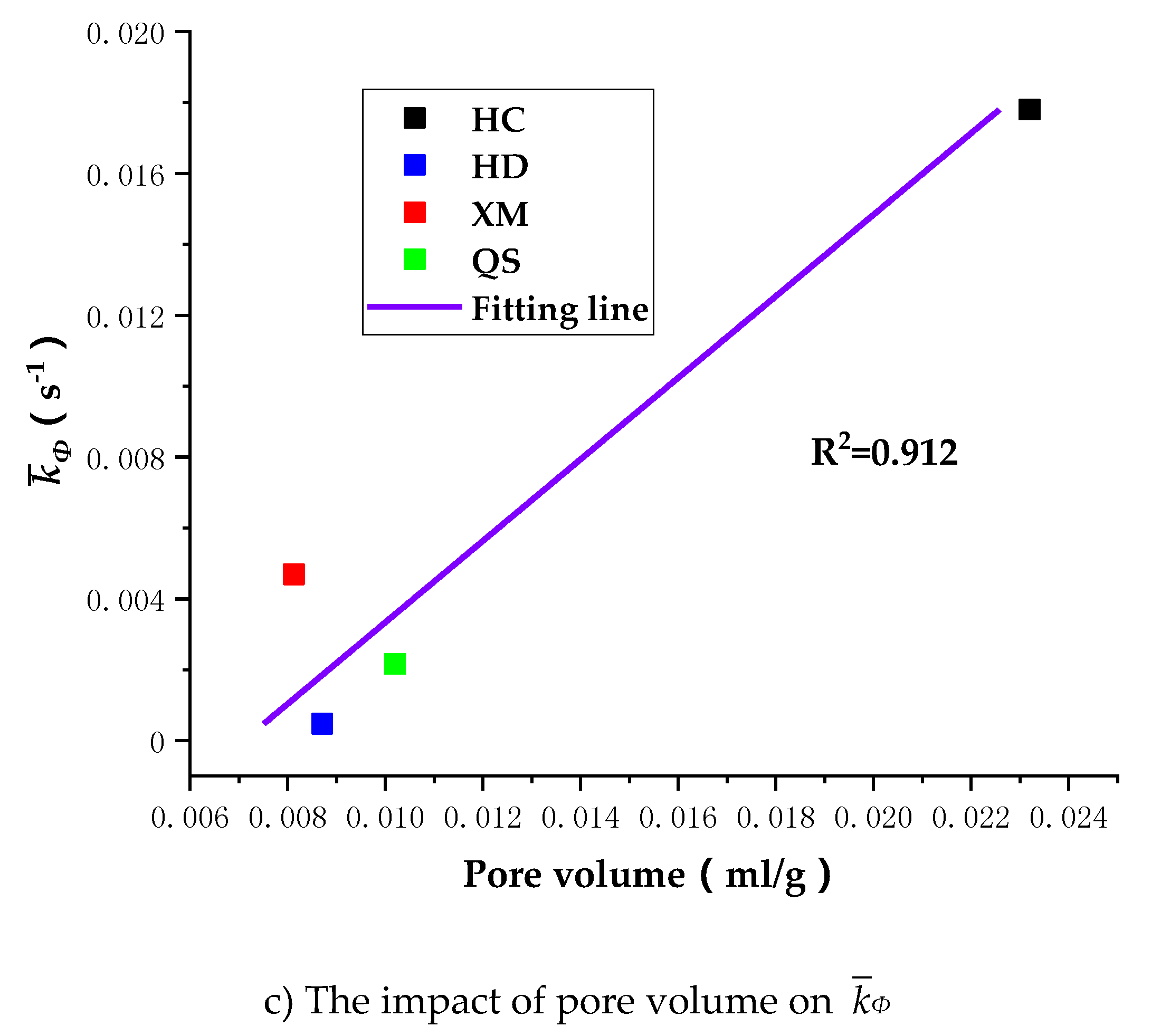
To further understand the impact of coal pore structure characteristics on the gas diffusion, we run the linear regressions of Dae, Die and kΦ on pore volume. As shown in Figure 12, the correlation between the pore volume and the diffusion coefficients is, largest for kΦ (R2 = 0.912), middle for Dae (R2 = 0.793) and smallest for Die (R2 = 0.722).
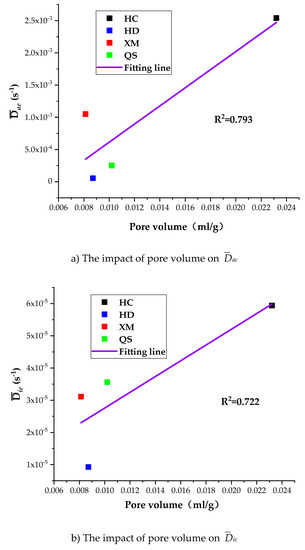
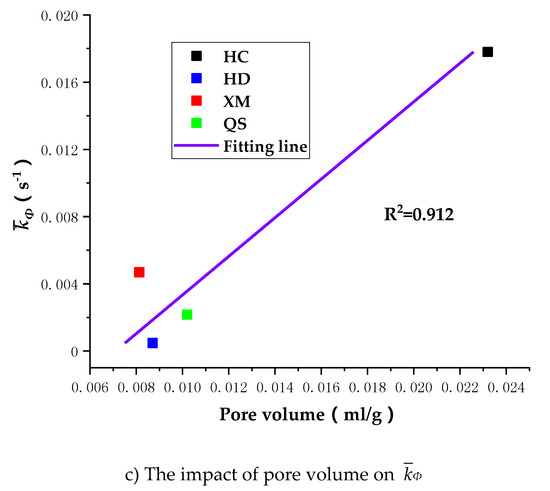
Figure 12.
The impact of pore volume on Dae, Die and kΦ.
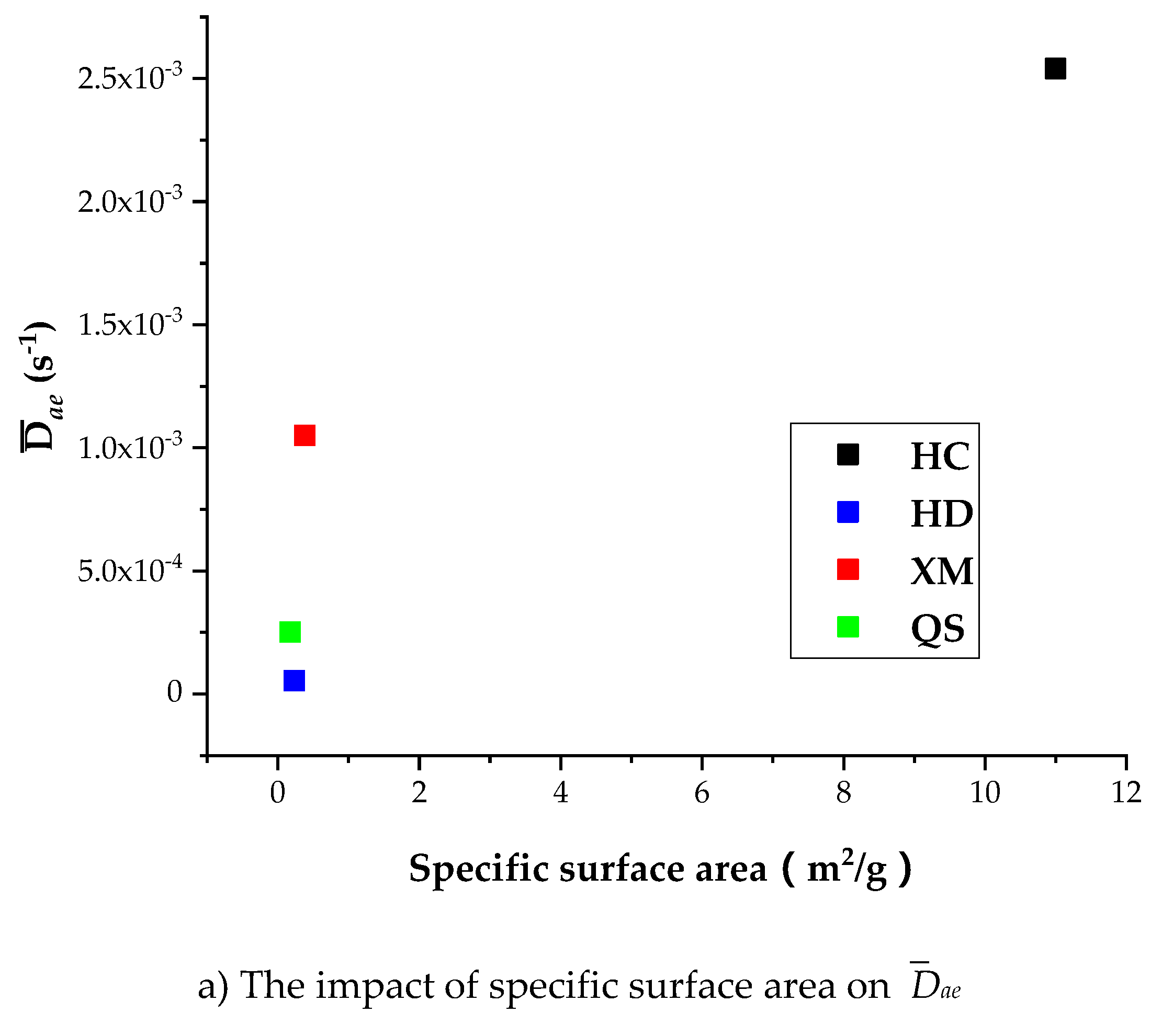
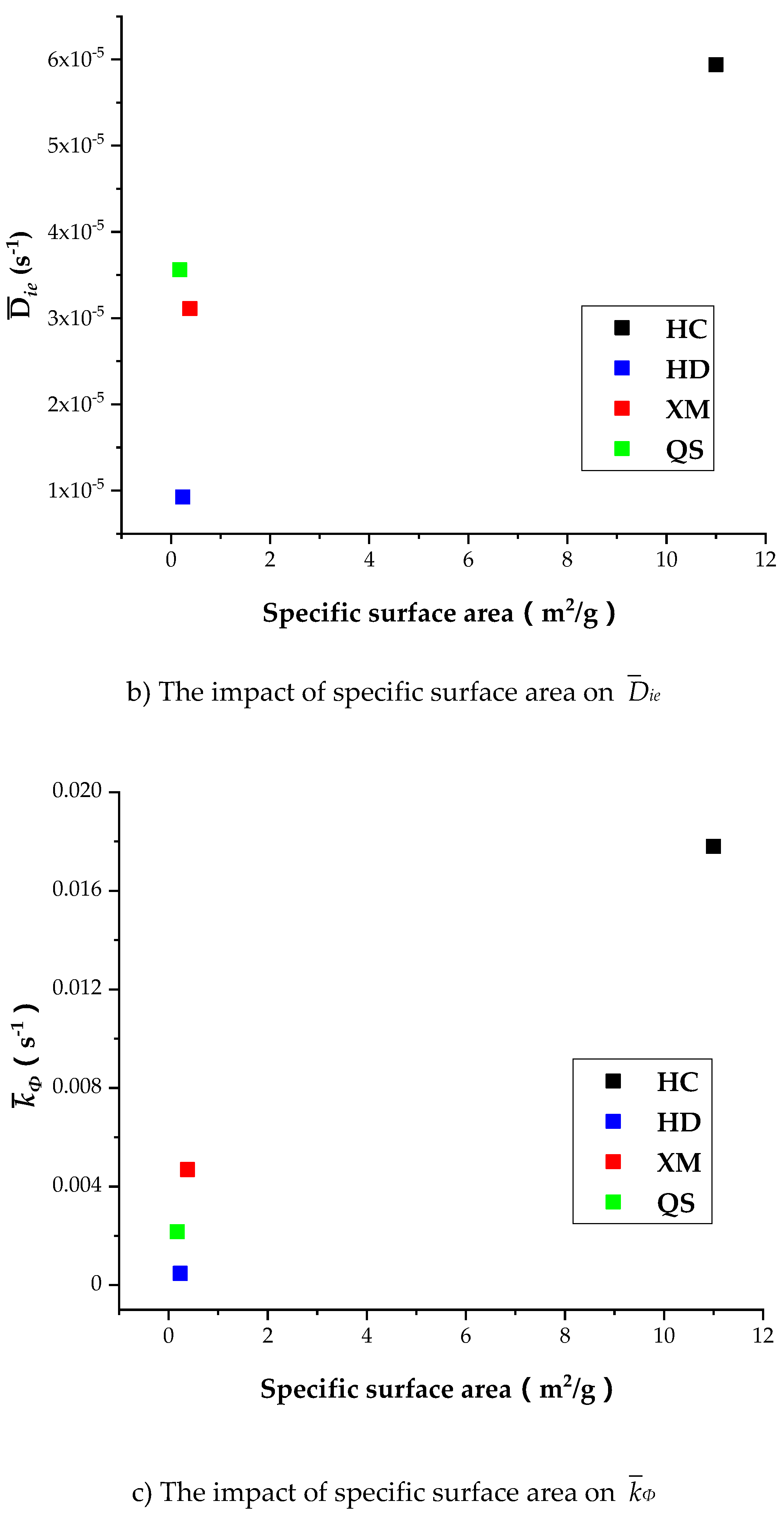
Table 8 shows that the specific surface area is larger in HC relatively to other coal samples, indicating that the porosity in HC is well developed than other coal samples. It is shown in Table 7 that Dae, Die and kΦ of CH4 is the largest in HC, suggesting the porosity development level is correlated with the diffusion rate. However, Figure 13a shows that Dae of HD, QS and XM significantly increases when Dae is smaller than 1.6 × 10−11 while the specific surface area keeps unchanged. Figure 13b,c show that the impact of specific surface area on Die and kΦ is small in all coal samples excluding HC. It is worth to mention that our experimental results can only be considered as reference due to the small number of coal samples. The impact of coal structure on the diffusion parameters requires further study. In conclusion, the fluctuation of diffusion coefficients with respect to the gas pressure is correlated to the variation of pore characteristics, but the reason is still mysterious due to lack of evidence.
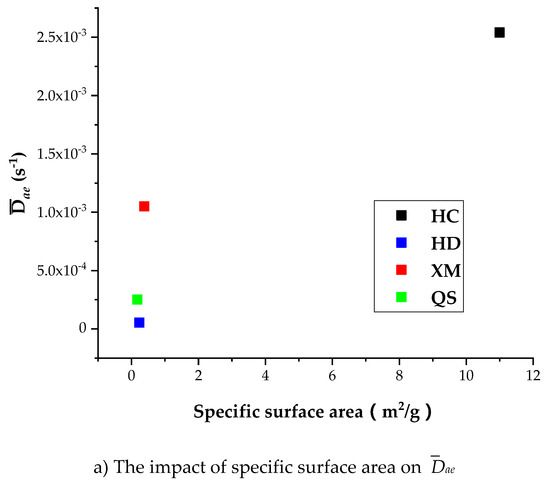
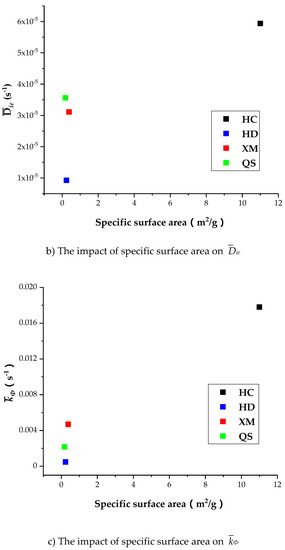
Figure 13.
The impact of specific surface area on Dae, Die, and kΦ.
4.4. Discussion on the Influence of Inconstant Diffusion Coefficients on CBM Recovery
Previous studies have demonstrated that the BD diffusion cannot be overlooked and replaced by UD diffusion if diffusion is a constraint of gas production, especially for the coal seam with relatively large cleat spacing [24]. In this study, we found the BD and DD models are more accurate in describing the diffusion process, while pressure has apparent influence on the diffusion coefficients. From this point of view, the inconstant diffusion coefficients will have impacts on the CBM recovery rate. In terms of BD coefficients, most samples show an increase of fast diffusion coefficient Dae but a decrease of slow diffusion coefficient Die during the drop of coal seam pressure. The increase or decrease of diffusion coefficient will certainly accelerate or hinder gas flow, but these two effects might be compromised for the BD model and the ultimate effect depends on the net value of these two effects. For the DD model, kΦ keeps at a stable level, this phenomenon proves the above speculation as kΦ can be seen as a combination of Dae and Die. However, the stretching parameter α decreases during pressure dropping, which indicates the CBM recovery rate will be reduced due to the change of diffusion coefficient.
5. Conclusions
- (1)
- Compared with the UD model, the BD and DD models are more accurate in describing the whole gas adsorption/desorption process.
- (2)
- The fast efficient diffusion coefficient Dae decreases with the increase in pressure for three out of four coal samples (i.e, HC, XM and QS) while it shows a V-shape with the increasing pressure for HD. The slow efficient diffusion coefficient Die is positively correlated with the pressure for all coal samples. The diffusion characteristic parameter β keeps constant in the adsorption and desorption process for all coal samples.
- (3)
- kΦ keeps fixed when the pressure changes and the stretching parameter α increases with the increase in pressure.
- (4)
- Both the effective diffusion coefficient Dae and Die and the characteristic rate parameter kΦ can be used to describe the gas diffusion rate. The impact of pore volume on Dae, Die and kΦ differs in the four coal samples while Dae, Die and kΦ are slightly affected by the specific surface area. The influence of pore structure characteristics on gas diffusion ability still requires further study.
Author Contributions
Conceptualization, G.W. and X.Y.; methodology, G.W.; validation, J.Z.; formal analysis, X.Y. and G.W.; data curation, G.W.; writing—original draft preparation, X.Y. and G.W.; writing—review and editing, G.W., J.Z. and T.R.; visualization, X.Y. and G.W.; supervision, J.Z. and T.R.
Funding
This work was supported by National Natural Science Foundation of China (51604153), National Science and Technology Major Project (2016ZX05045-004-006), National key research and development Project (2018YFB0605601).
Acknowledgments
We sincerely thank assistant professor Chunling Xia from Queen Mary University of London for improving the language of this paper.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Zhang, Y.; Xing, W.L.; Liu, S.Y.; Liu, Y.; Yang, M.J.; Zhao, J.F.; Song, Y.C. Pure methane, carbon dioxide, and nitrogen adsorption on anthracite from China over a wide range of pressures and temperatures: Experiments and modeling. RSC Adv. 2015, 5, 52612–52623. [Google Scholar] [CrossRef]
- Charrière, D.; Pokryszka, D.; Behra, P. Effect of pressure and temperature on diffusion of CO2 and CH4 into coal from the Lorraine basin (France). Int. J. Coal Geol. 2010, 81, 373–380. [Google Scholar] [CrossRef]
- Pan, Z.J.; Connell, L.D.; Camilleri, M.; Connelly, L. Effects of matrix moisture on gas diffusion and flow in coal. Fuel 2010, 89, 3207–3217. [Google Scholar] [CrossRef]
- Staib, G.; Richard, S.; Gray, E.M.A. A pressure and concentration dependence of CO2 diffusion in two Australian bituminous coals. Int. J. Coal Geol. 2013, 116, 106–116. [Google Scholar] [CrossRef]
- Clarkson, C.R.; Bustin, R.M. The effect of pore structure and gas pressure upon the transport properties of coal: A laboratory and modelling study. 2. Adsorption rate modelling. Fuel 1999, 78, 1345–1362. [Google Scholar] [CrossRef]
- Han, F.S.; Busch, A.; Krooss, B.M.; Liu, Z.Y.; Yang, J.L. CH4 and CO2 sorption isotherms and kinetics for different size fractions of two coals. Fuel 2013, 108, 137–142. [Google Scholar] [CrossRef]
- Li, D.Y.; Liu, Q.F.; Weniger, P.; Gensterblum, Y.; Busch, A.; Krooss, B.M. High-pressure sorption isotherms and sorption kinetics of CH4 and CO2 on coals. Fuel 2010, 89, 569–580. [Google Scholar] [CrossRef]
- Cui, X.J.; Bustin, R.M.; Dipple, G. Selective transport of CO2, CH4, and N2 in coals: Insights from modeling of experimental gas adsorption data. Fuel 2004, 83, 293–303. [Google Scholar] [CrossRef]
- Busch, A.; Gensterblum, Y.; Krooss, B.M.; Littke, R. Methane and carbon dioxide adsorption–diffusion experiments on coal: Upscaling and modelling. Int. J. Coal Geol. 2004, 60, 151–168. [Google Scholar] [CrossRef]
- Zhang, J. Experimental study and modeling for CO2 diffusion in coals with different particle sizes: Based on gas absorption (imbibition) and pore structure. Energy Fuels 2016, 30, 531–543. [Google Scholar] [CrossRef]
- Karacan, C.Ö. Heterogeneous sorption and swelling in a confined and stressed coal during CO2 injection. Energy Fuels 2003, 17, 1595–1608. [Google Scholar] [CrossRef]
- Crosdale, P.J.; Beamish, B.B.; Valix, M. Coalbed methane sorption related to coal composition. Int. J. Coal Geol. 1998, 35, 147–158. [Google Scholar] [CrossRef]
- Jian, X.; Guan, P.; Zhang, W. Carbon dioxide sorption and diffusion in coals: Experimental investigation and modeling. Sci. China Earth Sci. 2012, 55, 633–643. [Google Scholar] [CrossRef]
- Shi, J.Q.; Durucan, S. A bidisperse pore diffusion model for methane displacement desorption in coal by CO2 injection. Fuel 2003, 82, 1219–1229. [Google Scholar] [CrossRef]
- Nandi, S.P.; Walker, P.L. Activated diffusion of methane from coals at elevated pressures. Fuel 1975, 54, 81–86. [Google Scholar] [CrossRef]
- Švábová, M.; Weishauptová, Z.; Prˇibyl, O. The effect of moisture on the sorption process of CO2 on coal. Fuel 2012, 92, 187–196. [Google Scholar] [CrossRef]
- Pone, J.D.N.; Halleck, P.M.; Mathews, J.P. Sorption capacity and sorption kinetic measurements of CO2 and CH4 in confined and unconfined bituminous coal. Energy Fuels 2009, 23, 4688–4695. [Google Scholar] [CrossRef]
- Crank, J. The Mathematics of Diffusion, 2nd ed.; Oxford University Press: New York, NY, USA, 1975; ISBN 0198533446. [Google Scholar]
- Wang, G.D.; Ren, T.; Qi, Q.X.; Lin, J.; Liu, Q.Q.; Zhang, J. Determining the diffusion coefficient of gas diffusion in coal: Development of numerical solution. Fuel 2017, 196, 47–58. [Google Scholar] [CrossRef]
- Siemons, N.; Wolf, K.H.A.A.; Bruining, J. Interpretation of carbon dioxide diffusion behavior in coals. Int. J. Coal Geol. 2007, 72, 315–324. [Google Scholar] [CrossRef]
- Wang, G.D.; Ren, T.; Qi, Q.X.; Zhang, L.; Liu, Q.Q. Prediction of Coalbed Methane (CBM) Production Considering Bidisperse Diffusion: Model Development, Experimental Test, and Numerical Simulation. Energy Fuels 2017, 31, 5785–5797. [Google Scholar] [CrossRef]
- Ruckenstein, E.; Vaidyanathan, A.S.; Youngquist, G.R. Sorption by solids with bidisperse pore structures. Chem. Eng. Sci. 1971, 26, 1305–1318. [Google Scholar] [CrossRef]
- Staib, G.; Sakurovs, R.; Gray, E.M.A. Dispersive diffusion of gases in coals. Part I: Model development. Fuel 2015, 143, 612–619. [Google Scholar] [CrossRef]
- Busch, A.; Gensterblum, Y. CBM and CO2-ECBM related sorption processes in coal: A review. Int. J. Coal Geol. 2011, 87, 49–71. [Google Scholar] [CrossRef]
- Goodman, A.L.; Busch, A.; Duffy, G.J.; Fitzgerald, J.E.; Gasem, K.A.M.; Gensterblum, Y.; Krooss, B.M.; Levy, J.; Ozdemir, E.; Pan, Z.; et al. An Inter-laboratory comparison of CO2 isotherms measured on Argonne Premium Coal samples. Energy Fuels 2004, 18, 1175–1182. [Google Scholar] [CrossRef]
- Wang, G.D. Adsorption and Desorption Hysteresis of Coal Seam Gas and Its Influence on Gas Permeability [D]; China University of Mining and Technology: Beijing, China, 2015. (In Chinese) [Google Scholar]
- Staib, G.; Sakurovs, R.; Gray, E.M.A. Dispersive diffusion of gases in coals. Part II: An assessment of previously proposed physical mechanisms of diffusion in coal. Fuel 2015, 143, 620–629. [Google Scholar] [CrossRef]
- Liu, H.H.; Mou, J.H.; Cheng, Y.P. Impact of pore structure on gas adsorption and diffusion dynamics for long-flame coal. Nat. Gas Sci. Eng. 2015, 22, 203–221. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).