Abstract
This study investigates the impact of ethanol as a co-solvent in hydrothermal carbonization (HTC) of sewage sludge, a process referred to here as ethanothermal or solvothermal carbonization. Experiments were conducted at 180 °C, 200 °C, 220 °C, and 240 °C, comparing two sets of conditions: one using water (S/W) and the other using ethanol (S/E) as the reaction medium. The focus was placed on the composition of the aqueous phase, particularly the formation of volatile fatty acids (VFAs). Ethanol-assisted experiments consistently produced more alkaline process water (pH 7.6–8.2) compared to water-based runs. COD values in S/W samples ranged from 9358 mg/L to 19,756 mg/L, indicating significant organic loading. Hydrochar derived from the ethanol experiments exhibited higher energy content, with a peak high heating value (HHV) of 21.9 MJ/kg at 240 °C, compared to 19.9 MJ/kg in S/W samples. VFA concentrations were also enhanced under ethanothermal conditions, especially at lower temperatures: formic acid (30.4–34.8 mg/L), acetic acid (8.7–9.6 mg/L), and propionic acid (10.8–14.6 mg/L). These results demonstrate ethanol’s potential to enhance both the yield and quality of liquid and solid products in HTC of sewage sludge.
1. Introduction
Sewage sludge, a major byproduct of urban wastewater treatment plants, is generated primarily through the processes of primary and secondary sedimentation [1,2]. Globally, the volume of sludge produced is substantial, with many countries generating millions of tons annually, reaching as high as 45 million tons per year [1]. The high moisture content of sewage sludge, which in some cases exceeds 98%, poses a significant challenge for its efficient management, as it renders conventional handling methods highly energy intensive [3]. Chemically, sewage sludge comprises a complex matrix of organic and inorganic constituents, including toxic organic compounds, heavy metals, pathogenic microorganisms, and other microbiological contaminants [4]. Its composition includes proteins, lipids, fibrous and non-fibrous carbohydrates, and ash [5]. In terms of dry mass, proteins account for approximately 40 wt%, lipids range from 10 to 25 wt%, carbohydrates represent about 14 wt%, and ash can reach up to 30–50 wt% [6]. This complex and potentially hazardous composition necessitates careful handling and the development of effective treatment strategies [7].
Conventional sludge management practices include landfilling, incineration, and composting [8], with landfilling and incineration being the most commonly applied methods, particularly in North America [9]. However, these methods introduce environmental concerns and generate secondary waste streams requiring further management [8]. Landfilling occupies large tracts of land and poses risks of soil and groundwater contamination, while incineration emits air pollutants and entails high operational costs [10]. Anaerobic digestion is often employed as an alternative for biogas production, yet the process is time-consuming and typically results in a residual waste stream rich in moisture and carbon [11]. Furthermore, neither anaerobic digestion nor composting effectively mitigates the presence of heavy metals [12]. Overall, these traditional approaches are often associated with high economic and environmental costs [13]. In addition to generating valuable carbonaceous and aqueous products, hydrothermal carbonization can also serve as a pretreatment strategy that enhances the downstream processing of biomass (such as pyrolysis, gasification, or combustion) by reducing moisture content, improving fuel properties, and stabilizing reactive intermediates.
Hydrothermal carbonization (HTC) has emerged as a promising and energy-efficient technology for the treatment of wet biomass, including sewage sludge [14]. HTC is a thermochemical process that converts biomass under subcritical water conditions, typically at temperatures ranging from 180 to 250 °C [15]. It enables the direct processing of high-moisture feedstocks without the need for energy-intensive drying [16]. The main products of HTC are hydrochar—a carbon-rich solid with properties similar to those of charcoal—and an aqueous phase containing a wide array of low-molecular-weight organic compounds [17], such as phenolic derivatives resulting from carbohydrate degradation [18]. The HTC process initiates with hydrolysis and progresses through dehydration and decarboxylation reactions, ultimately leading to the formation of hydrochar and process water enriched with solubilized organics [19].
In recent studies, the incorporation of organic solvents as reaction media during HTC has gained attention. These solvents, which typically lack salts, help reduce corrosion during processing [20], and also inhibit lignin repolymerization, limit the formation of hydrophobic residues [21]. Polar organic solvents further suppress the formation of nitrogen-containing byproducts, thereby improving nitrogen distribution and overall product yield [17]. Ethanol, in particular, acts as a hydrogen donor, promoting hydrolysis and enhancing the hydrogen content in the system [22]. As noted by Masoumi et al., 2021 [23], polar solvents facilitate deoxygenation by accelerating hydrogenation pathways. Moreover, solvent-assisted HTC has been reported to improve the quality of the carbonaceous solid product relative to conventional HTC, often resulting in reduced residence times and a higher degree of dehydration [24]. As mentioned by Zhang et al., 2025 [25] the interaction of organic solvents with water significantly enhances dehydration and decarboxylation reactions. Also, Zhang et al., 2023 [26] noticed that the presence of alcohols catalyzes depolymerization during the HTC process.
The purpose of this research is to study the influence of different solvents on the HTC process. More specifically, in order to investigate and obtain a comprehensive picture of the effect of solvents on the process, a series of extensive experiments took place. For this purpose, sewage sludge was used as raw material and experiments were conducted with water and ethanol to compose the reaction medium. The research includes analyses of the liquid and solid products of the process, placing greater emphasis on the organic products of the aqueous phase and, in particular, VFAs regarding the effect that these two different solvents have on them.
This study primarily investigates how different solvents affect the formation and composition of organic compounds in the liquid phase of the HTC process. This was done as a wide range of research focuses on the effect of organic solvents on hydrochar. Most analyses are based on the quality of the solid product of the process. They focus on upgrading hydrochar to products such as nano-carbon material and graphic materials, without investigating the quality of process water. This creates a research gap in relation to products in the liquid phase of HTC. Therefore, information on high-value organic products such as VFAs and phenols is not rich in terms of HTC experiments using solvents.
2. Materials and Methods
The experiments were carried out on the island of Lesvos, using sewage sludge as raw material. The sludge came from the Lesvos Waste-Water Treatment Plant (WWTP), which treats urban wastewater in the wider region. With the collection of sewage sludge and before its use in experiments, some qualitative analyses were carried out.
2.1. Analysis of Raw Material
The experimental protocol began with the drying of sewage sludge. Specifically, 4 g of sludge was placed in an oven at 105 °C for 24 h in order to calculate the total solids (TSs) and moisture of the raw material. Then, the material was placed in a muffle furnace and further heated to 550 °C for 2 h to calculate the volatile solids (VSs). After this, the pH was measured using a De Brune Instruments Consort C932 (Wichelen, Belgium) pH-meter while the COD concentration was measured using the potassium dichromate method, which is reported more analytically below, as it was applied to the liquid products of HTC.
2.2. Experimental Process
The HTC process took place in a Parr 4577A (Moline, IL, USA) hydrothermal reactor of 1 L capacity. Two series of experiments were carried out under temperatures of 180 °C, 200 °C, 220 °C, and 240 °C. The first phase involved experiments with sludge and water (S/W) and the second experiments with sludge and ethanol (S/E). The quantity of mixtures and temperature and pressure conditions are evident in Table 1. The residence time for all experiments was 2 h. After the end of the HTC process and the cooling of the reactor, the collection of products was done. Besides centrifugation and Buchner funnel filtration, the hydrochar and the liquid phase were separated, weighed, and analyzed further.

Table 1.
HTC experimental layout.
2.3. Analysis Procedure
In a more general description, the analyses performed in the liquid phase of the process were measurements of pH, COD concentration, total phenols, and VFAs. On the other hand, in hydrochar, the high calorific value was measured. The analysis started by measuring the pH of all liquid samples. A De Bruyne Instruments Consort C932 pH-meter was used, which was preceded by calibration with pH at 4 and pH at 7 for greater accuracy [27]. It should be noted that mass yields were calculated on a wet basis and reflect total output distribution. The method of potassium dichromate was used to calculate the concentration of COD. Ιt was initially diluted with water in all samples. In S/W samples, a 1/100 dilution was made, as a first step of the analysis. For the analysis, a mixture was made that included 2.8 mL of silver sulfate, 1.2 mL of potassium dichromate, and 2 mL of diluted sample. This procedure was done for all samples of S/W. Then, all mixtures were placed in a Hach (Loveland, CO, USA) 45600 COD reactor at 250 °C for 2 h. After that, the concentration of COD was measured in a Hach (Loveland, CO, USA) DR/2400 spectrophotometer, according to the APHA method, 1998 [28]. The concentration of COD was measured only for S/W samples.
The concentration of phenols was measured through the Folin–Ciocalteu method. Initially, dilution of 1/100 with water was performed in all samples. Then, a mixture was made containing 6 mL of water, 1 mL of diluted sample, 0.5 mL of phenol reagent, 1.5 mL of sodium carbonate, and 1 mL of water. The above procedure was done for all samples. The mixtures were then placed in a shaded place for 2 h, followed by the measurement of the total phenol concentration in a Hach DR/2400 spectrophotometer [28].
The analysis also focused on the concentration of VFAs. The process began by centrifuging the liquid for 10 min to avoid any solids and then filtering. Then, equal amounts of isopropanol and process water were mixed and placed in an ultra-sonic bath at 40 °C for 15 min. After, the mixture was introduced to a distilled column to evaporate the organic solvent, leaving behind the VFAs. At the end of the distillation, the liquid was filtered with 0.45 μm filters. This process was done for all samples. The efficiency of the extraction method was estimated using the CRM46975 Supelco Volatile Free Acid Mix as a standard, with the recovery efficiency estimated at 59.6 ± 0.2%. The measurement was done on a Shimadzu Nexis 2030 GC-BID and with an Agilent J&W HP-FFAP column. The method used had the following parameters: a 1 μL sample injection volume, an injection temperature of 160 °C, an oven temperature program ranging from 80 °C to 230 °C, a flow rate of 59 mL/min, and a BID detector temperature of 280 °C.
The final stage of the analyses involved the measurement of the high calorific value in all hydrochar samples. The measurement was done on a Parr 6400 calorimeter preceded by calibration with benzoic acid. Then, 0.3 g of hydrochar from each sample was placed for measurement. All samples were measured three times for greater accuracy.
2.4. Limitations of the Study
This work centers on the effect that only water and ethanol have on the HTC process. This is a limitation of the range of solvents that could be used in the experiments. The lack of ethanol-only solvent blank experiments is a methodological limitation, as it may confound the interpretation of volatile fatty acid (VFA) and COD data in the ethanol-assisted HTC runs, and this will be the focus of future upcoming studies. In addition, more qualitative analyses could be performed on the solid product of HTC to obtain a larger picture of the effect of solvents on its quality. Also, we did not investigate the quality of the gaseous products. All the above are topics that will be addressed in future experiments in the context of a wider range of solvents, temperatures, and analyses.
3. Results and Discussion
3.1. Analysis of Sewage Sludge
Before the start of the experimental process, the sewage sludge underwent some qualitative analyses in terms of TS, VS, moisture, pH, and COD. After drying, the total solids were 2.7%, volatile solids were 0.8%, and humidity was 96.5%. The percentage of moisture appeared to be very high, which was also reported by Wang et al., 2022 [29] who noted a moisture percentage in sewage sludge over 80%, while Roslan et al., 2023 [30] reported 83.7% for their own experiments. In a similar case, the TSs were calculated to be in the range of 4.5% according to Blach and Engelhart, 2021 [31]. Regarding the pH of the sewage sludge, this appeared alkaline with a value of 7.5, while the organic load from the measurement of the COD concentrations was 5824 mg/L. In other studies, the pH was estimated at 7.8, and thus alkaline [32], while COD was estimated at approximately 2175 mg/L [33].
3.2. Mass Yield After Experimental Process
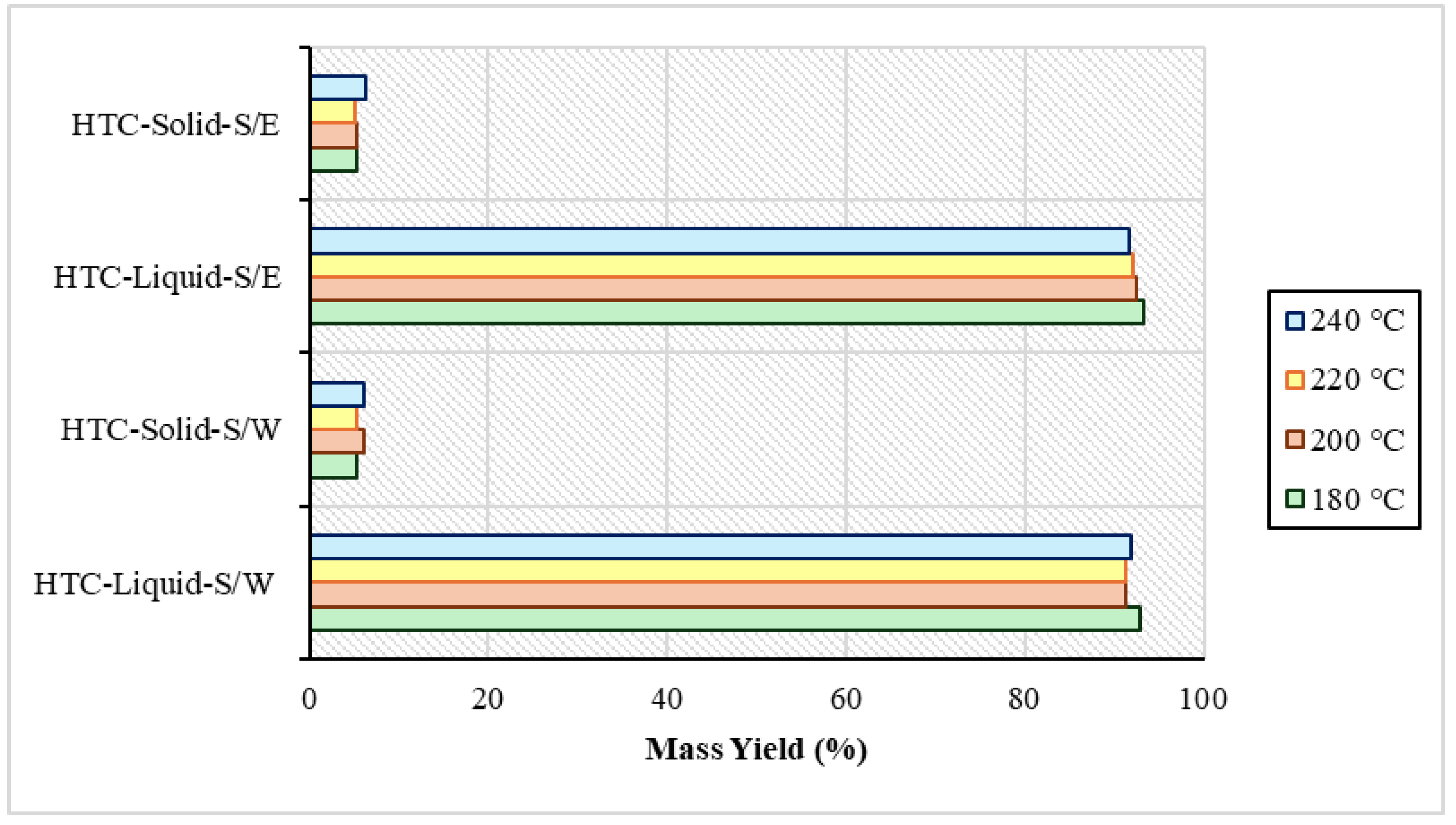
After the end of the product separations, weighing was performed to obtain a clear picture of the quantity of the products resulting from the process. It was observed in both the S/W and S/E experiments that process water had the highest percentage in the yield with a range of 91–93%. Hydrochar appeared at a lower rate ranging from 5% to 6.3%. This difference was due to the low percentage of total solids in the raw material, as mentioned above. The mass yield of the experiments is distinguished in Figure 1 below. In Xu et al.’s study, in 2022 [34] due to the catalytic amplification of the reactions of hydrolysis, dehydration, and thermodynamic decomposition of organic matter in the presence of solvents, the hydrochar yield was reduced and the dissolution of organic matter was promoted. Also, in other hydrothermal carbonization experiments with the presence of organic solvents using Brewer’s grains, isopropanol showed a better performance in the production of liquid product compared to ethanol, which also had in these experiments a yield very close to water at 25% and 23%, respectively. Also, in the conversion efficiency of the solid, ethanol reached 77% and water 72%, rates which are very close [35].
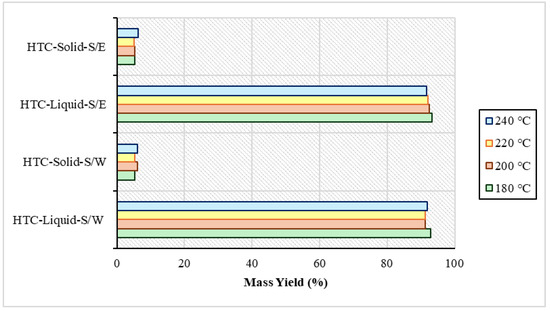
Figure 1.
Mass yield after sewage sludge and water (S/W) experiments and sewage sludge and ethanol (S/E) experiments.
3.3. pH Values of Liquid Products
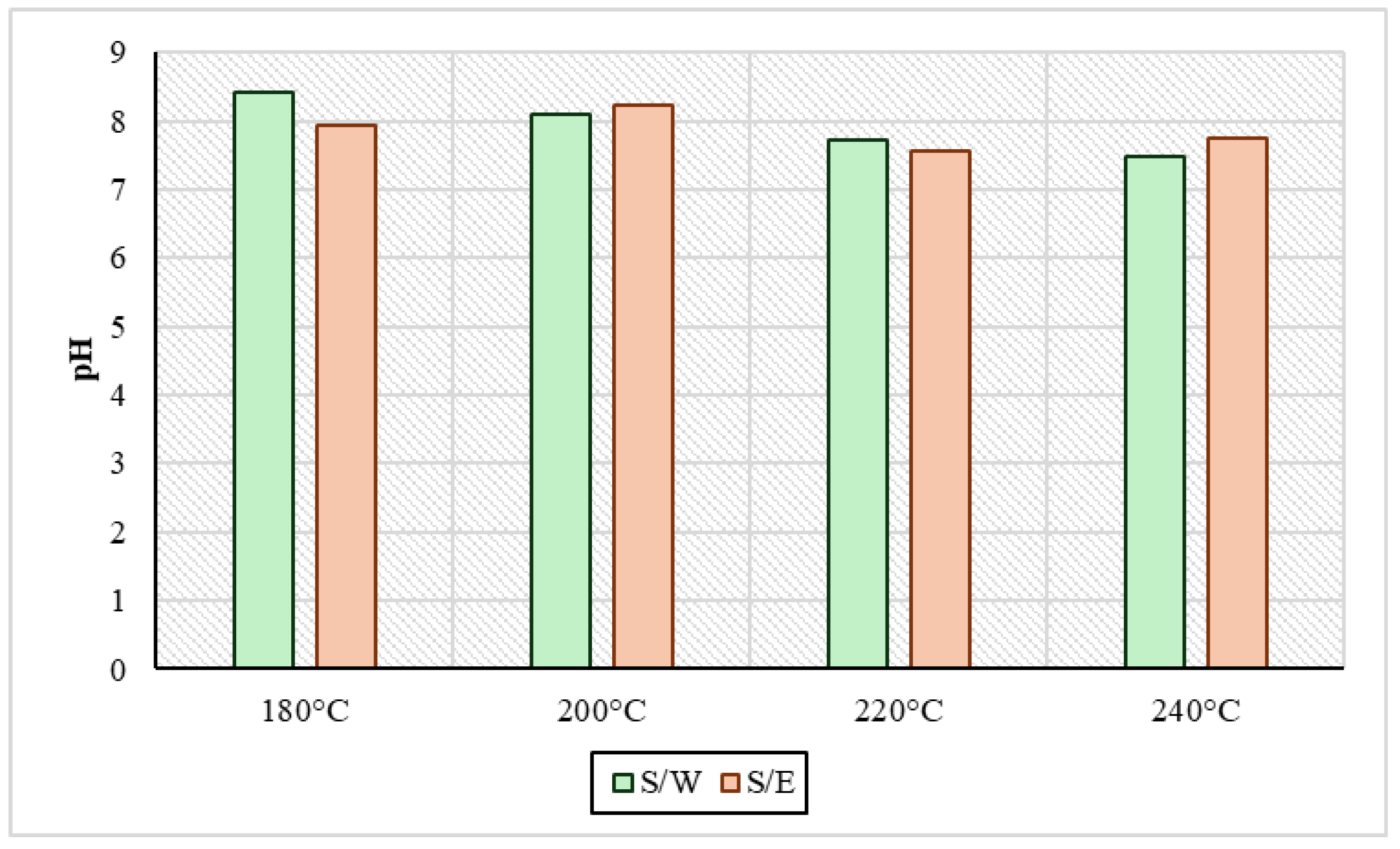
Regarding the pH values of the liquid products from HTC are presented in Figure 2. It was observed in all cases that they ranged in a mild alkaline range with values from 7.6 to 8.4. In the samples produced by reaction medium, the pH of the water S/W dropped with temperature increase; specifically, in the sample at 180 °C, the pH value was 8.4, and at 240 °C, it ended up being 7.5. On the other hand, in HTC liquid products obtained with the presence of ethanol S/E in the process, the pH values appeared to be systematically and consistently more alkaline than those of the S/W samples at all temperatures. As reported by Wilk et al., 2019 [36] who did experiments of the HTC of sewage sludge, at 200 °C for 4 h, a strong alkaline pH is an indicator of the presence of volatile organic compounds in process water. Also, through HTC experiments at 200 °C with sewage sludge, a pH close to 7 was measured according to Xu et al., 2020 [37]. In addition, in Wang et al.’s study, 2019 [12] there was a certain point in HTC in a temperature range from 210 °C to 220 °C where the pH rose and left the range of alkalinity due to the high production of alkaline groups such as ammonia. As mentioned above, it was observed that ethanol in the medium meant that the process remained more stable at alkaline pH, contributing to the production of volatile organic acids.
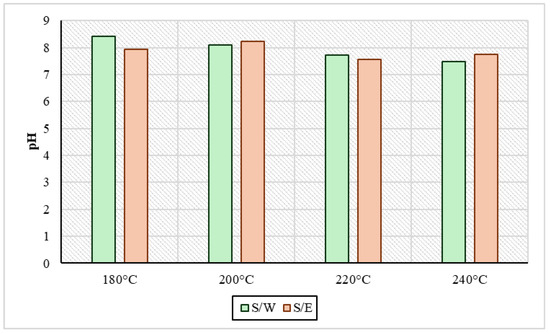
Figure 2.
pH values in process water for sewage sludge and water samples (S/W) and also for sewage sludge and ethanol samples (S/E) for every experimental temperature condition.
3.4. Total Phenolic Content
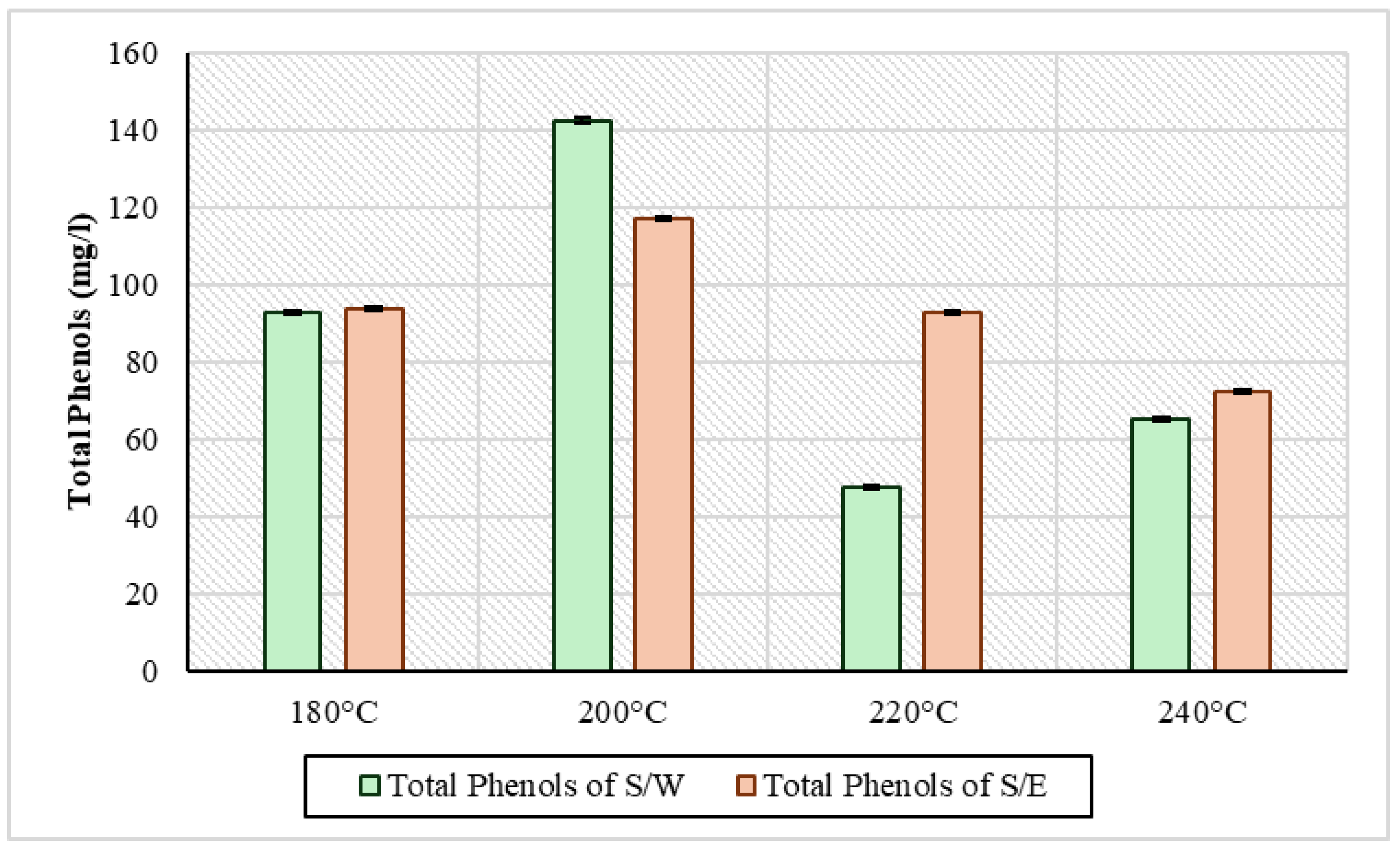
Total phenols, as they are presented in Figure 3, had almost the same image in the two series of experiments. In general, in the first two temperatures, there was an increase in concentration, namely from 180 °C to 200 °C, and then a decrease to 240 °C. Looking at the S/W samples, the concentration of total phenols was 92.8 mg/L at 180 °C, and this increased to the highest concentration at 200 °C at 142 mg/L. A decrease was then observed to 220 °C at 47.8 mg/L and a slight increase to 65.2 mg/L at 240 °C. This variation may be due to the fact that phenolic compounds tend to condense into the solid product of HTC, forming secondary hydrochar and with increasing temperature breaking up part of it, releasing a small amount of phenols into process water [38]. Generally, however, high-temperature conditions destroy phenols, causing a reduction to higher conditions [39]. This behavior was observed in the S/E experiments. The concentrations started at 93.8 mg/L at 180 °C, increased to 117.2 mg/L at 200 °C, and then with the increase in temperature, there was a gradual decrease in total phenols to 92.8 mg/L and 72.4 mg/L at 220 °C and 240 °C, respectively. According to Czerwińska et al., 2024 [40] in their HTC experiments using sewage sludge at 200 °C with a residence time of 2 h, the concentration of phenols was 127 mg/L. In another series of sewage sludge HTC experiments, at temperatures of 190 °C, 205 °C, and 220 °C, the concentration of phenols increased from 361 mg/L to 391 mg/L at 205 °C and with the increase in temperature to 384 mg/L [41].
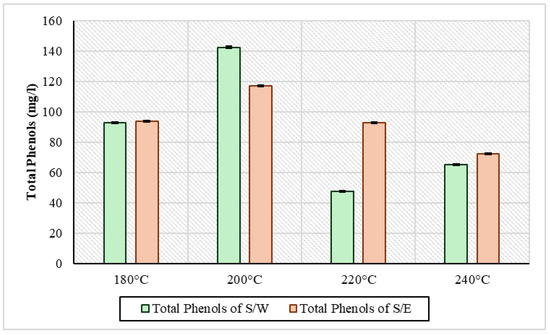
Figure 3.
Total phenolic content in liquid phase of HTC for all samples in every temperature condition.
3.5. COD Concentration
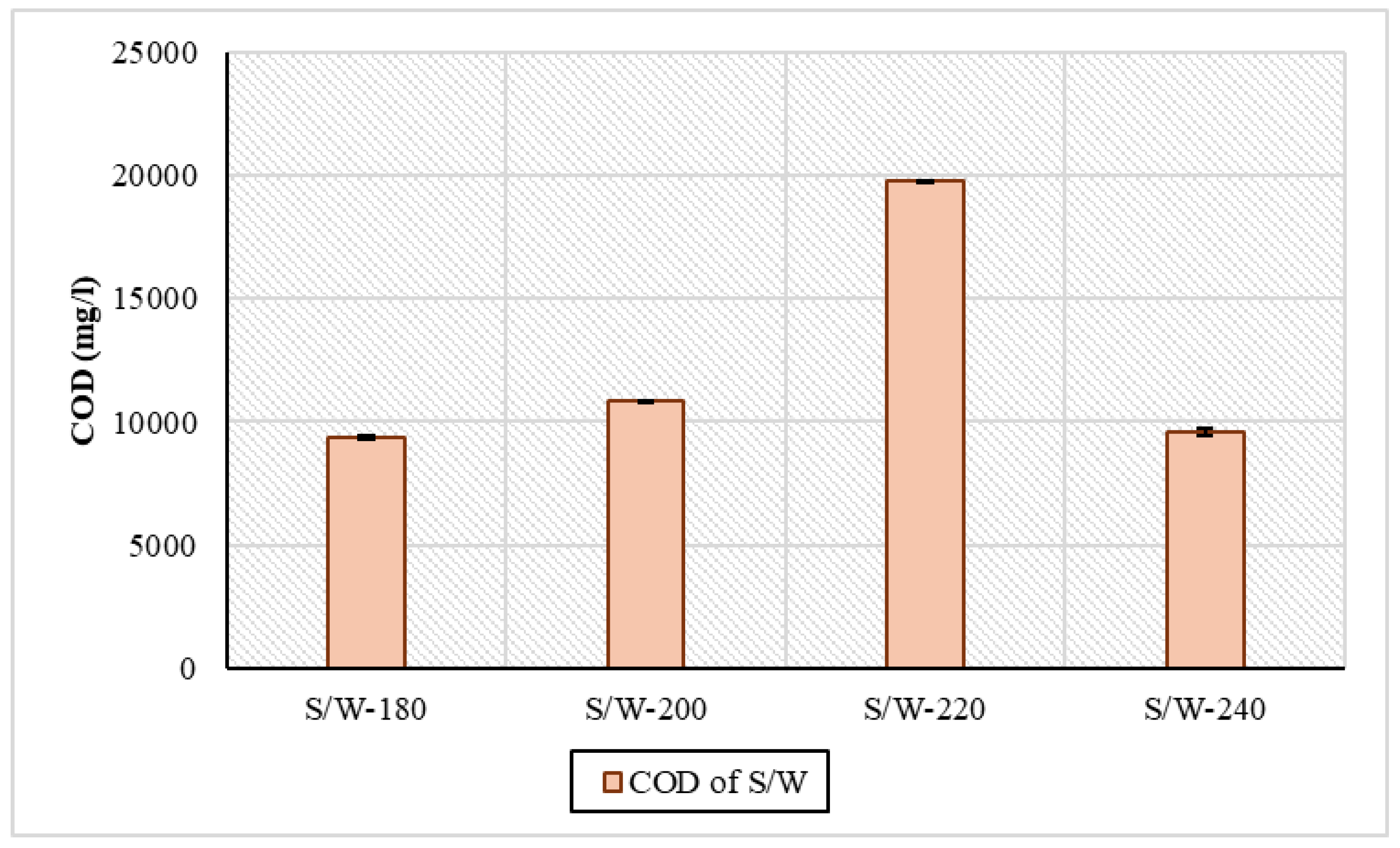
There appeared to be large differences in the organic load between the two phases of the experiments. In S/W samples, the concentration of COD ranged from 9358 mg/L to 19,756 mg/L. More specifically, from 180 °C to 220 °C, there was an increase in COD, while at 240 °C, there was a drop, from 19,756 mg/L to 9594 mg/L. These fluctuations in concentration are the result of the dissolution and decomposition of organic matter in the process [42]. In similar HTC experiments with sewage sludge at 210 °C for 4 h, the concentration of COD was 7000 mg/L according to Stutzenstein et al., 2018 [43]. In another series of HTC experiments with digested sewage sludge, a similar variation in concentration within the temperature ranges was shown: at temperatures of 160 °C, 220 °C, and 250 °C and a residence time of 30 min, they were 12,642 mg/L, 12,992 mg/L, and 12,164 mg/L [44]. Due to the absence of an ethanol removal step or ethanol-only blanks, COD measurements were performed and reported in Figure 4 only for the S/W (sewage sludge and water) samples to avoid misinterpretation of organic load contributions.
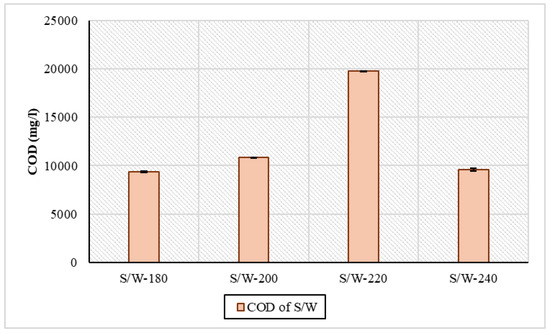
Figure 4.
Organic load through COD concentration in process water for sewage sludge and water samples.
3.6. Concentration of VFAs
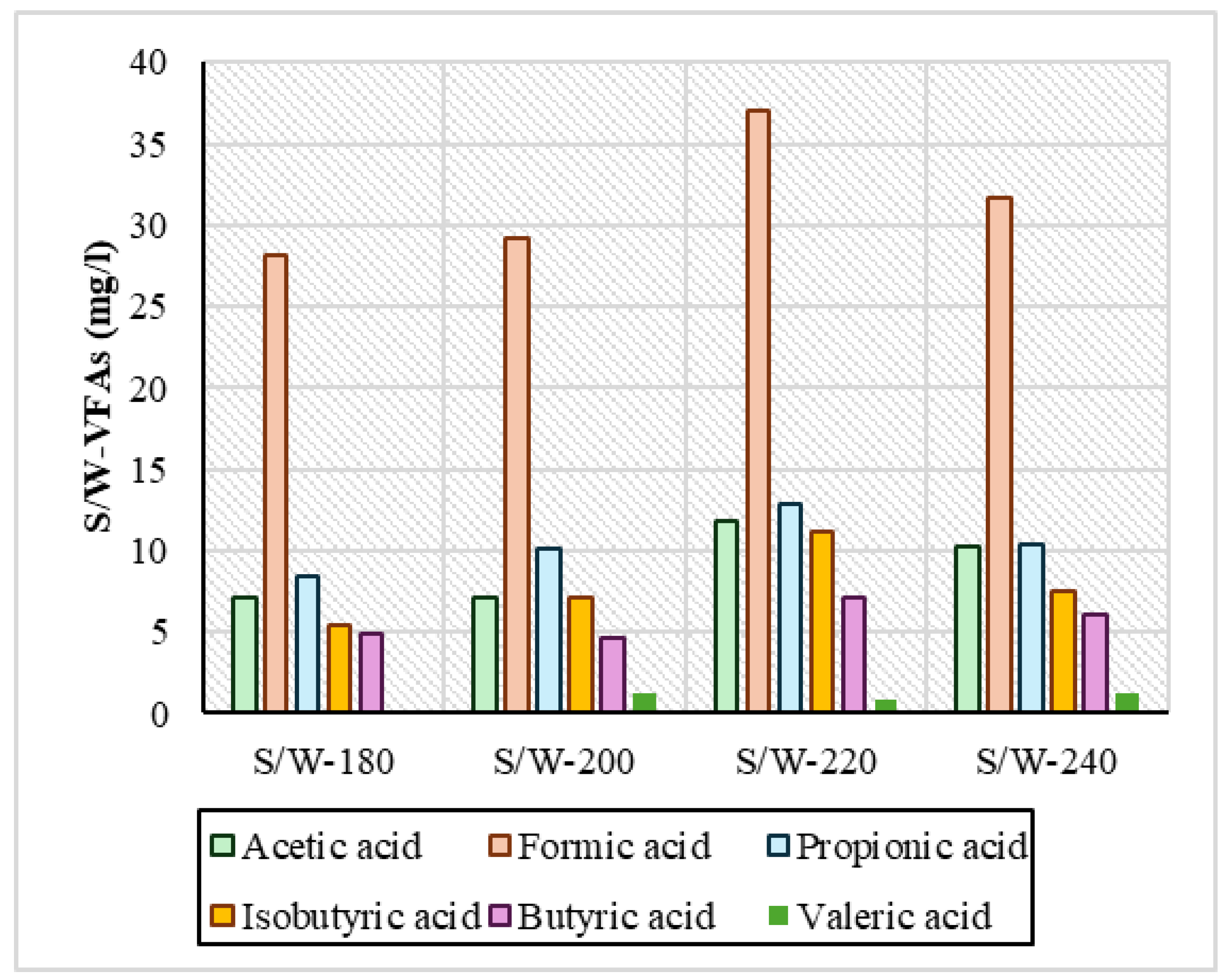
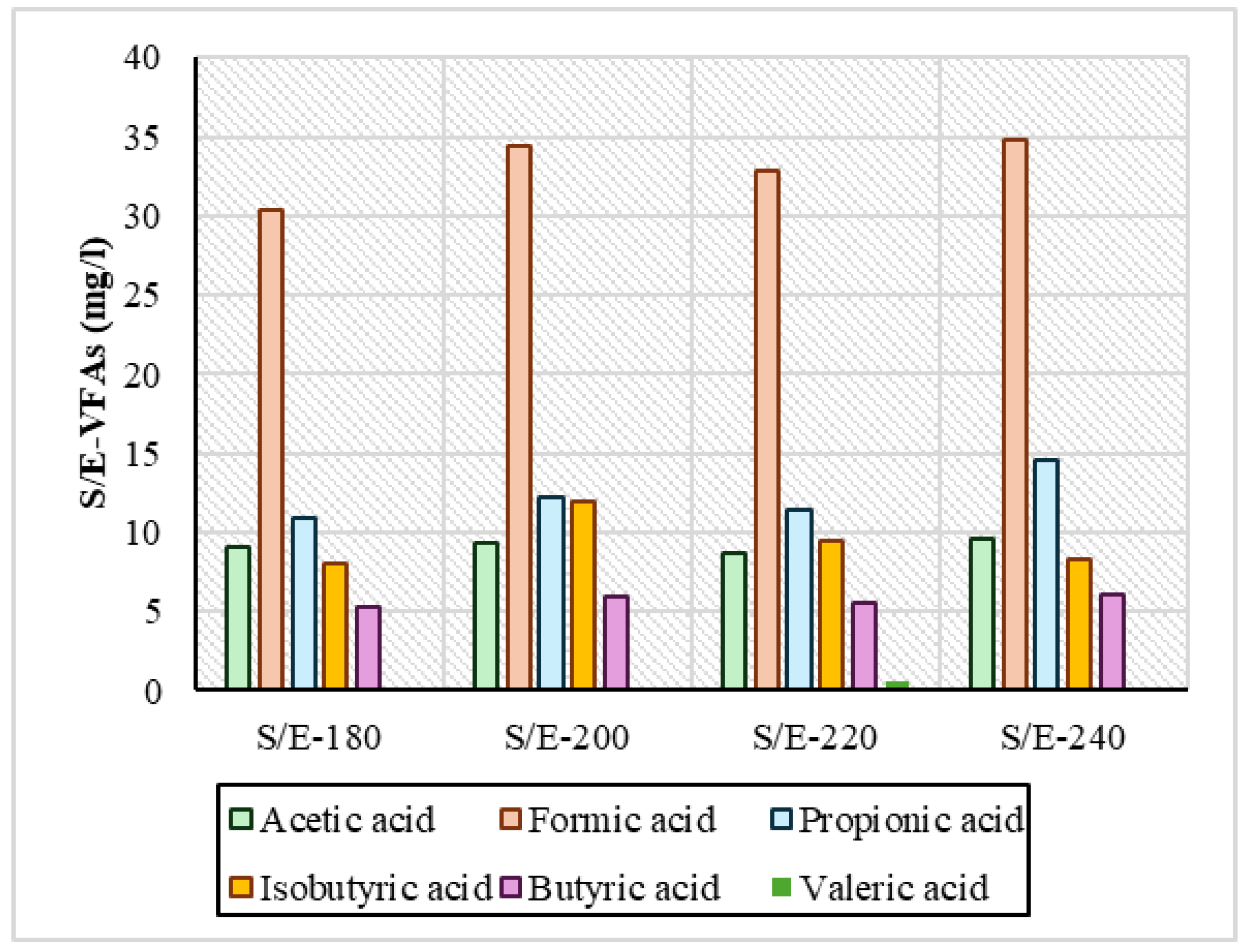
Regarding VFAs, the measurements are presented in Figure 5 and Figure 6 and showed that in both series of experiments, acetic, formic, propionic, and isobutyric acids were produced in larger quantities. In the reactive ethanol experiments, it was observed that across the temperature range, it had a consistently higher concentration of VFA compared to the S/W experiments. Specifically regarding formic acid at all temperatures, in the S/E samples, its concentration ranged above 30 mg/L in a range from 30.4 mg/L to 34.8 mg/L, and in propionic acid it ranged above 10 mg/L from 10.8 mg/L to 14.6 mg/L. For the S/W samples, formic acid had a concentration of 28.1 mg/L at 220 °C, and the highest concentration was 37.1 mg/L. In general, however, the values in the remaining temperatures were lower and had variations relative to the S/E samples. Isobutyric and butyric acids were shown to be low and with similar concentrations in both series of experiments. In HTC with increasing temperature, there was an increase in the concentration of VFAs in process water [45], which is evident in all samples. The same was reported by Yu et al., 2018 [4]: after HTC of digested sludge at 160 °C, 200 °C, and 240 °C, the concentration of VFAs was steadily increasing. In the samples mainly in S/W, the variations observed may be due to the fact that in low-temperature ranges, when the temperature increases, enzymatic hydrolysis reactions are favored, causing a small conversion of VFAs to phenolic compounds through many dehydration and condensation reactions, contributing to the eventual reduction in VFAs, as reported by Zhang et al., 2024 [46]. In addition, in a study of hydrothermal carbonization at 250 °C that used brewing cereals and alcoholic solvents such as ethanol, methanol, and isopropanol, the high formation of oxygen-enriched functional groups of mainly carboxylates became apparent, as reported by Hossain et al., 2023 [47]. A critical point that needs to be addressed is the actual role of ethanol in the production of VFAs. Recent studies [48,49] have shown that ethanol under hydrothermal/subcritical conditions can produce measurable volatile acids and other organics (especially acetic acid, and potentially minor formic or propionic acid depending on the conditions). In the context of HTC experiments, any additional COD or VFA measured in the liquid could partly originate from the ethanol itself rather than the feedstock. Without a solvent-only blank run, one cannot distinguish how much of the observed organic product load (e.g., VFAs in process water) is contributed by the solvent. Therefore, future studies should include ethanol-only control experiments to quantify background VFA/oxidizable organic generation. The literature above strongly supports that ethanol is not an inert medium at HTC conditions—it undergoes thermochemical reforming into short-chain acids and related organics, warranting blank corrections in order for accurate interpretations to be made.
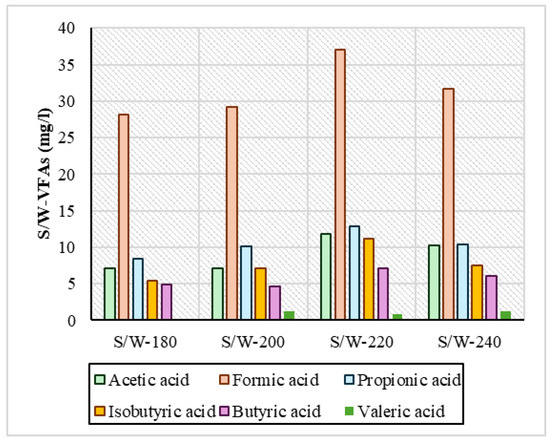
Figure 5.
VFA concentration in process water for the experiments of sewage sludge and water (S/W).
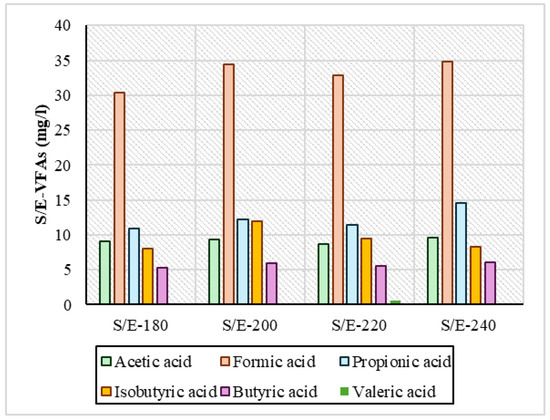
Figure 6.
VFA concentration in samples from sewage sludge and ethanol (S/E) experiments.
3.7. HHV of Hydrochar
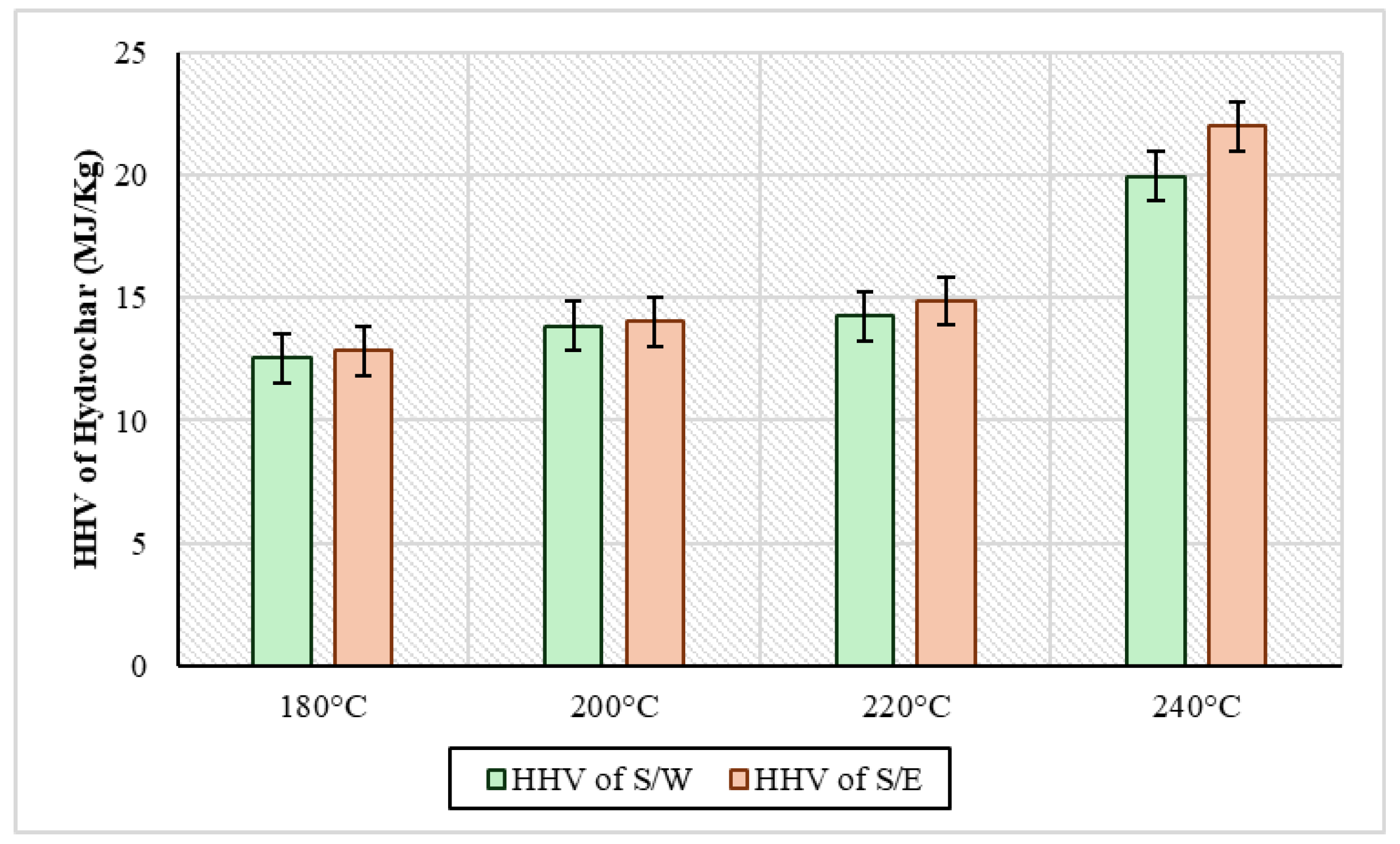
The high calorific value was measured for all solid samples and the results are presented in Figure 7. In a more general context, the analysis showed that in S/W samples, the HHV ranged from 12.5 MJ/kg to 19.9 MJ/kg, while in S/E samples, it ranged from 12.8 MJ/kg to 21.9 MJ/kg. In particular, it was observed in both cases that HHV had a tendency to increase with increasing temperature. Also, at all temperatures, the hydrochar from the S/E experiments appeared to have a consistently higher calorific value compared to the hydrochar of the S/W experiments. In the case of the experiments of Youn et al., 2023 [50] by adding ethanol to kenaf hydrothermal carburetion at temperatures of 200 °C and 225 °C and a residence time of 30 min, HHV was 16.7 kJ/g and 15.9 kJ/g, respectively. On the other hand, in the same experiments, in the absence of ethanol, hydrochar had an HHV of 18.3 kJ/g at 200 °C and 19.2 kJ/g at 225 °C. In addition, Kousar et al., 2025 [51] conducted HTC experiments at 200 degrees for 10 h where they tested a wide range of organic solvents, using cooked rice as the raw material. Analyses of HHV on hydrochar showed that in HTC without an organic solvent, the hydrochar had a value of 25.4 MJ/kg, in HTC with aniline it was 23.7 MJ/kg, with xylene it was 26.7 MJ/kg, with chlorobenzene it was 25.8 MJ/kg, and with glycol it was 27.7 MJ/kg. Similarly, in the above case, when several different organic solvents were used, it was shown that the majority of organic solvents contributed to the higher HHVs relative to water as a reaction medium of the process. In general, however, the HHVs being low compared to those of other types of biomasses is due to the fact that sewage sludge is a material that with an increase in temperature during hydrothermal carbonization, this increases the ash content of the hydrochar formed, indicating the transfer of more organic matter to the liquid piece [52].
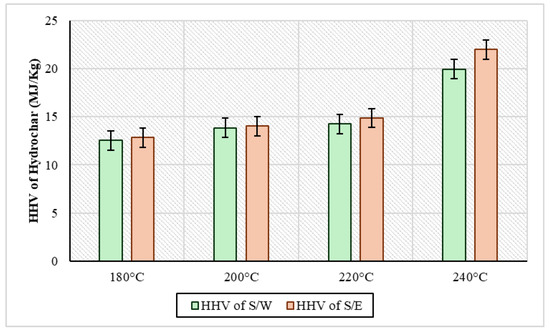
Figure 7.
HHV of the solid product of HTC with sewage sludge and water (S/W) and sewage sludge and ethanol (S/W) for every experimental temperature.
4. Conclusions
This study examined the influence of ethanol as a co-solvent in the hydrothermal carbonization (HTC) of sewage sludge, with particular emphasis on the composition of the liquid phase. Ethanol-assisted HTC (S/E) consistently resulted in more alkaline process water compared to conventional water-based HTC (S/W), with pH values ranging from 7.6 to 8.2 across all temperatures. Although COD was only measured in S/W samples to avoid interference from unremoved ethanol, the results indicated substantial organic loading in the aqueous phase. Volatile fatty acids (VFAs), including formic, acetic, and propionic acid, were detected in higher concentrations in S/E samples, particularly at lower temperatures. This suggests that ethanol may enhance the production or preservation of VFAs during HTC, while a fraction of formation may be due to the cracking of ethanol. The high heating value (HHV) of hydrochar also improved under ethanol conditions, with the highest value observed at 240 °C (21.9 MJ/kg) compared to 19.9 MJ/kg in the S/W experiments. These results demonstrate that ethanol as a reactive medium can positively influence both the solid and liquid products of HTC. However, the absence of gas-phase analysis and solvent blank controls limits the ability to fully account for ethanol’s transformation pathways. Future studies should include ethanol-only control runs and gas product characterization to achieve a complete mass and energy balance and to distinguish solvent-derived organics from biomass-derived ones. Overall, it can clearly be observed that the presence of ethanol as a co-solvent in the HTC process contributes to the upgrading of both the solid and the liquid product. This fact is evident from the high heating value of hydrochar in the presence of ethanol as well as the enrichment of the liquid product with high-value chemicals such as VFAs in the experiments using ethanol. Consequently, all the results make HTC in the presence of ethanol a promising process as a pretreatment strategy for moist biomass and also both suitable for waste treatment and efficient for the production of high-value-added materials.
Author Contributions
Conceptualization, S.V.; methodology, S.V. and S.M.; software, S.V. and D.L.; validation, S.V.; formal analysis, D.L. and S.V.; investigation, D.L. and S.V.; resources, S.V. and S.M.; data curation, D.L. and S.V.; writing—original draft preparation, D.L.; writing—review and editing, S.V.; visualization, S.V.; supervision, S.V. and S.M.; project administration, S.V. and S.M.; funding acquisition, S.V. and S.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research was partially funded by the project TRINEFLEX, which has received funding from the European Union’s Horizon Europe research and innovation program under Grant Agreement No. 101058174 “TrineFlex”. The research was partially funded by the project BioFairNet, which has received funding from the European Commission’s Horizon Europe research and innovation program under Grant Agreement No. 101181568.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors on request.
Acknowledgments
During the preparation of this manuscript, the authors used ChatGPT 5.1 for the purposes of correcting linguistic errors. The authors have reviewed and edited the output and take full responsibility for the content of this publication.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| COD | Chemical Oxygen Demand |
| °C | Degrees of Celsius |
| HHV | High Heating Value |
| HTC | Hydrothermal Carbonization |
| MJ/kg | Megajoule per kilogram |
| mg/L | Milligram per liter |
| TSs | Total Solids |
| VFAs | Volatile Fatty Acids |
| VSs | Volatile Solids |
References
- Wang, W.; Zhou, W.; Wang, Y.; Wang, M.; Wang, Q.; Hu, Y.; Wu, J.; Hu, J.; Luo, T.; Fan, L. Production of low-nitrogen and oxygen biocrude and coupling recovery of nitrogen and phosphorus from hydrothermal liquefaction of sewage sludge with MgCl2. Chem. Eng. J. 2025, 505, 159184. [Google Scholar] [CrossRef]
- Yang, Y.; Du, Z.; Cao, C.; Duan, P. Production of high value-added carbon material precursors via direct hydrothermal conversion of municipal sewage sludge coupled with air flotation separation: A review. Sci. Total Environ. 2025, 982, 179655. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.P.; Zhang, C.; Li, X.; Yu, S.H.; Tan, P.; Fang, Q.Y.; Chen, G. A two-step process for sewage sludge treatment: Hydrothermal treatment of sludge and catalytic hydrothermal gasification of its derived liquid. Fuel Process. Technol. 2018, 180, 67–74. [Google Scholar] [CrossRef]
- Yu, Y.; Lei, Z.; Yang, X.; Yang, X.; Huang, W.; Shimizu, K.; Zhang, Z. Hydrothermal carbonization of anaerobic granular sludge: Effect of process temperature on nutrients availability and energy gain from produced hydrochar. Appl. Energy 2018, 229, 88–95. [Google Scholar] [CrossRef]
- Qian, L.; Wang, S.; Savage, P.E. Hydrothermal liquefaction of sewage sludge under isothermal and fast conditions. Bioresour. Technol. 2017, 232, 27–34. [Google Scholar] [CrossRef]
- He, C.; Chen, C.L.; Giannis, A.; Yang, Y.; Wang, J.Y. Hydrothermal gasification of sewage sludge and model compounds for renewable hydrogen production: A review. Renew. Sustain. Energy Rev. 2014, 39, 1127–1142. [Google Scholar] [CrossRef]
- Gao, N.; Kamran, K.; Quan, C.; Williams, P.T. Thermochemical conversion of sewage sludge: A critical review. Prog. Energy Combust. Sci. 2020, 79, 100843. [Google Scholar] [CrossRef]
- Fan, Y.; Hornung, U.; Dahmen, N. Hydrothermal liquefaction of sewage sludge for biofuel application: A review on fundamentals, current challenges and strategies. Biomass Bioenergy 2022, 165, 106570. [Google Scholar] [CrossRef]
- Gude, V.G. Energy positive wastewater treatment and sludge management. Edorium J. Waste Manag. 2015, 1, 10–15. [Google Scholar]
- Fonts, I.; Gea, G.; Azuara, M.; Ábrego, J.; Arauzo, J. Sewage sludge pyrolysis for liquid production: A review. Renew. Sustain. Energy Rev. 2012, 16, 2781–2805. [Google Scholar] [CrossRef]
- Thomsen, L.B.S.; Carvalho, P.N.; Dos Passos, J.S.; Anastasakis, K.; Bester, K.; Biller, P. Hydrothermal liquefaction of sewage sludge; energy considerations and fate of micropollutants during pilot scale processing. Water Res. 2020, 183, 116101. [Google Scholar] [CrossRef]
- Wang, L.; Chang, Y.; Li, A. Hydrothermal carbonization for energy-efficient processing of sewage sludge: A review. Renew. Sustain. Energy Rev. 2019, 108, 423–440. [Google Scholar] [CrossRef]
- Huang, H.J.; Yuan, X.Z.; Li, B.T.; Xiao, Y.D.; Zeng, G.M. Thermochemical liquefaction characteristics of sewage sludge in different organic solvents. J. Anal. Appl. Pyrolysis 2014, 109, 176–184. [Google Scholar] [CrossRef]
- Colin, J.; Sarrion, A.; Diaz, E.; de la Rubia, M.A.; Mohedano, A.F. Ecotoxicity assessment of hydrochar from hydrothermal carbonization of biomass waste. Sustain. Chem. Pharm. 2025, 44, 101909. [Google Scholar] [CrossRef]
- Masoumi, S.; Borugadda, V.B.; Nanda, S.; Dalai, A.K. Hydrochar: A review on its production technologies and applications. Catalysts 2021, 11, 939. [Google Scholar] [CrossRef]
- Wang, C.; Lin, X.; Zhang, X.; Show, P.L. Research advances on production and application of algal biochar in environmental remediation. Environ. Pollut. 2024, 348, 123860. [Google Scholar] [CrossRef]
- Liakos, D.; Altiparmaki, G.; Kalampokidis, A.; Lekkas, D.F.; Vakalis, S. The role of hydrochar on the production of biogas and volatile fatty acids during anaerobic digestion of cheese whey wastewater. Sustain. Chem. Pharm. 2023, 35, 101153. [Google Scholar] [CrossRef]
- Toor, S.S.; Rosendahl, L.; Rudolf, A. Hydrothermal liquefaction of biomass: A review of subcritical water technologies. Energy 2011, 36, 2328–2342. [Google Scholar] [CrossRef]
- Román, S.; Nabais, J.M.V.; Laginhas, C.; Ledesma, B.; González, J.F. Hydrothermal carbonization as an effective way of densifying the energy content of biomass. Fuel Process. Technol. 2012, 103, 78–83. [Google Scholar] [CrossRef]
- Guvenatam, B. Catalytic Pathways for Lignin Depolymerization. Ph.D. Thesis, Eindhoven University of Technology, Eindhoven, The Netherlands, 2015. Available online: https://research.tue.nl/en/publications/08fcceb0-6ab6-45e9-ab13-69fc9f076fae (accessed on 15 November 2025).
- Ouyang, X.; Huang, X.; Zhu, Y.; Qiu, X. Ethanol-enhanced liquefaction of lignin with formic acid as an in situ hydrogen donor. Energy Fuels 2015, 29, 5835–5840. [Google Scholar] [CrossRef]
- Pothoczki, S.; Pethes, I.; Pusztai, L.; Temleitner, L.; Ohara, K.; Bakó, I. Properties of hydrogen-bonded networks in ethanol–water liquid mixtures as a function of temperature: Diffraction experiments and computer simulations. J. Phys. Chem. B 2021, 125, 6272–6279. [Google Scholar] [CrossRef] [PubMed]
- Masoumi, S.; Boahene, P.E.; Dalai, A.K. Biocrude oil and hydrochar production and characterization obtained from hydrothermal liquefaction of microalgae in methanol-water system. Energy 2021, 217, 119344. [Google Scholar] [CrossRef]
- Flores–Oña, D.; Fullana, A. Carbon nanoparticles production using solvent assisted hydrothermal carbonization. Diam. Relat. Mater. 2020, 108, 107960. [Google Scholar] [CrossRef]
- Zhang, B.; Zhang, J.; Xia, A.; Tang, X.; Zhu, X.; Huang, Y.; Zhu, X.; Liao, Q. Co-production and upgrading of multiple products from hydrothermal carbonization of microalgae with organic solvent assistance. Bioresour. Technol. 2025, 429, 132514. [Google Scholar] [CrossRef]
- Zhang, Q.; Mu, K.; Han, J.; Qin, L.; Zhao, B.; Yi, L. Low nitrogen and high value hydrochar preparation through co-hydrothermal carbonization of sludge and saw dust with acid/alcohol assistance. Energy 2023, 278, 128012. [Google Scholar] [CrossRef]
- Sklavos, S.; Gatidou, G.; Stasinakis, A.S.; Haralambopoulos, D. Use of solar distillation for olive mill wastewater drying and recovery of polyphenolic compounds. J. Environ. Manag. 2015, 162, 46–52. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Wastewater, 20th ed.; American Public Health Association, Port City Press: Baltimore, MD, USA, 1998. [Google Scholar]
- Wang, R.; Lin, K.; Peng, P.; Lin, Z.; Zhao, Z.; Yin, Q.; Ge, L. Energy yield optimization of co-hydrothermal carbonization of sewage sludge and pinewood sawdust coupled with anaerobic digestion of the wastewater byproduct. Fuel 2022, 326, 125025. [Google Scholar] [CrossRef]
- Roslan, S.Z.; Zainudin, S.F.; Mohd Aris, A.; Chin, K.B.; Musa, M.; Mohamad Daud, A.R.; Syed Hassan, S.S.A. Hydrothermal carbonization of sewage sludge into solid biofuel: Influences of process conditions on the energetic properties of hydrochar. Energies 2023, 16, 2483. [Google Scholar] [CrossRef]
- Blach, T.; Engelhart, M. Optimizing the hydrothermal carbonization of sewage sludge—Response surface methodology and the effect of volatile solids. Water 2021, 13, 1225. [Google Scholar] [CrossRef]
- Tasca, A.L.; Stefanelli, E.; Raspolli Galletti, A.M.; Gori, R.; Mannarino, G.; Vitolo, S.; Puccini, M. Hydrothermal carbonization of sewage sludge: Analysis of process severity and solid content. Chem. Eng. Technol. 2020, 43, 2382–2392. [Google Scholar] [CrossRef]
- Altiparmaki, G.; Liakos, D.; Artikopoulos, A.; Vakalis, S. Hydrothermal Carbonization Treatment as a Pathway for Energy Utilization of Municipal Sludge and Agricultural Residues Through Co-Gasification. Processes 2025, 13, 2713. [Google Scholar] [CrossRef]
- Xu, Z.; Ma, X.; Liao, J.; Osman, S.M.; Wu, S.; Luque, R. Effects on the physicochemical properties of hydrochar originating from deep eutectic solvent (urea and ZnCl2)-assisted hydrothermal carbonization of sewage sludge. ACS Sustain. Chem. Eng. 2022, 10, 4258–4268. [Google Scholar] [CrossRef]
- Nasir, N.A.; Davies, G.; McGregor, J. Tailoring product characteristics in the carbonisation of brewers’ spent grain through solvent selection. Food Bioprod. Process. 2020, 120, 41–47. [Google Scholar] [CrossRef]
- Wilk, M.; Magdziarz, A.; Jayaraman, K.; Szymańska-Chargot, M.; Gökalp, I. Hydrothermal carbonization characteristics of sewage sludge and lignocellulosic biomass. A comparative study. Biomass Bioenergy 2019, 120, 166–175. [Google Scholar] [CrossRef]
- Xu, Z.X.; Song, H.; Li, P.J.; He, Z.X.; Wang, Q.; Wang, K.; Duan, P.G. Hydrothermal carbonization of sewage sludge: Effect of aqueous phase recycling. Chem. Eng. J. 2020, 387, 123410. [Google Scholar] [CrossRef]
- Ischia, G.; Sudibyo, H.; Miotello, A.; Tester, J.W.; Fiori, L.; Goldfarb, J.L. Identifying the transition from hydrothermal carbonization to liquefaction of biomass in a batch system. ACS Sustain. Chem. Eng. 2024, 12, 4539–4550. [Google Scholar] [CrossRef]
- Vakalis, S.; Georgiou, A.; Moustakas, K.; Fountoulakis, M. Assessing the effect of hydrothermal treatment on the volatile solids content and the biomethane potential of common reed (Phragmites australis). Bioresour. Technol. Rep. 2022, 17, 100923. [Google Scholar] [CrossRef]
- Czerwińska, K.; Śliz, M.; Wilk, M. Thermal disposal of post-processing water derived from the hydrothermal carbonization process of sewage sludge. Waste Biomass Valorization 2024, 15, 1671–1680. [Google Scholar] [CrossRef]
- Escala, M.; Zumbuhl, T.; Koller, C.; Junge, R.; Krebs, R. Hydrothermal carbonization as an energy-efficient alternative to established drying technologies for sewage sludge: A feasibility study on a laboratory scale. Energy Fuels 2013, 27, 454–460. [Google Scholar] [CrossRef]
- Langone, M.; Basso, D. Process waters from hydrothermal carbonization of sludge: Characteristics and possible valorization pathways. Int. J. Environ. Res. Public Health 2020, 17, 6618. [Google Scholar] [CrossRef]
- Stutzenstein, P.; Weiner, B.; Köhler, R.; Pfeifer, C.; Kopinke, F.D. Wet oxidation of process water from hydrothermal carbonization of biomass with nitrate as oxidant. Chem. Eng. J. 2018, 339, 1–6. [Google Scholar] [CrossRef]
- Aragón-Briceño, C.; Ross, A.B.; Camargo-Valero, M.A. Evaluation and comparison of product yields and bio-methane potential in sewage digestate following hydrothermal treatment. Appl. Energy 2017, 208, 1357–1369. [Google Scholar] [CrossRef]
- Huezo, L.; Vasco-Correa, J.; Shah, A. Hydrothermal carbonization of anaerobically digested sewage sludge for hydrochar production. Bioresour. Technol. Rep. 2021, 15, 100795. [Google Scholar] [CrossRef]
- Zhang, Z.; Yan, T.; Zhang, T.; Zhang, Z.; Wang, W.; Peng, H.; Li, D.; Zhu, Z. Volatile fatty acid release and metal ion concentration in hydrothermal carbonization liquid. J. Anal. Appl. Pyrolysis 2024, 183, 106815. [Google Scholar] [CrossRef]
- Hossain, N.; Nizamuddin, S.; Shah, K. Comparative study of solvothermal and catalytic solvothermal carbonization of rice husk for Fe (III), Zn (II), Cu (II), Pb (II) and Mn (II) adsorption, kinetics, surface chemistry and reaction mechanism. Environ. Sci. Water Res. Technol. 2023, 9, 1829–1848. [Google Scholar] [CrossRef]
- Wang, P.; Kim, M.S.; Bohutskyi, P.; dos Passos, J.S.; Collett, J.; Johnson, Z.; Subramaniam, S.; Kilgore, U.J.; Fox, S.P.; Cronin, D.J.; et al. Continuous wet air oxidation of the hydrothermal liquefaction aqueous product from various wet wastes. ACS Sustain. Resour. Manag. 2025, 2, 1562–1570. [Google Scholar] [CrossRef]
- Koido, K.; Ishida, Y.; Kumabe, K.; Matsumoto, K.; Hasegawa, T. Kinetics of ethanol oxidation in subcritical water. J. Supercrit. Fluids 2010, 55, 246–251. [Google Scholar] [CrossRef]
- Youn, H.S.; Kim, S.J.; Kim, G.H.; Um, B.H. Enhancing the characteristics of hydrochar via hydrothermal carbonization of Korean native kenaf: The effect of ethanol solvent concentration as co-solvent and reaction temperature. Fuel 2023, 331, 125738. [Google Scholar] [CrossRef]
- Kousar, S.; Javed, K.; Begum, B.; Naeem, M.M.; Zhang, S.; Hu, X. Organic solvent assisted hydrothermal processing of cooked rice to get insights into the evolution of chemical structure of hydrochar. Waste Manag. 2025, 205, 115007. [Google Scholar] [CrossRef]
- Dou, R.; Gao, F.; Tan, Y.; Xiong, H.-R.; Xu, Z.X.; Osman, S.M.; Zheng, L.-J.; Luque, R. Phosphorus species transformation and recovery in deep eutectic solvent-assisted hydrothermal carbonization of sewage sludge. Sustain. Chem. Pharm. 2024, 37, 101408. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.