Abstract
Severe fluid loss in fractured, depleted reservoirs usually defeat conventional water-based drilling fluids (WBDFs), and rigid lost-circulation materials (LCMs) struggle to form durable, conformal seals. We report an eco-oriented colloidal gas aphron (CGA) fluid built from a nanostructured corn biopolymer (NCBP) and a biodegradable peanut-oil-derived surfactant, benchmarked against a reference fluid (RF) and aphron-only baselines (aphron based fluid, ABF). NCBP, produced by ball milling, was confirmed nanostructured by x-ray diffraction (XRD), Fourier transform infrared spectroscopy (FTIR), electron and atomic microscopies. Performance was evaluated from 25 to 90 °C for rheology, aphron stability and filtration at low temperature and low pressure (LTLP) of 100 psi and 25 °C, with post-test mud cake imaging. The optimized formulation, NCBP-2, showed stronger shear-thinning and higher gel strengths with heat, sustained stable and uniform aphrons for at least 120 min with foam persistence beyond 24 h, and delivered 3.0 mL filtrate with a 0.8 mm mud cake. These outcomes correspond to 60% less filtrate and approximately 73% thinner mud cakes than RF (7.5 mL; 3.0 mm), and about 14% and 33% improvements over the best ABF (3.5 mL; 1.2 mm). Micrographs revealed denser, finer-pored mud cakes, consistent with a mechanism in which deformable aphrons bridge micro-fractures while nano-scale polymeric fillers tighten the mud cake network. The results demonstrate decisive loss-control gains with temperature-tolerant rheology, supporting bio-based CGA fluids for depleted and fractured formations.
1. Introduction
Colloidal Gas Aphrons (CGA), initially termed microfoams by Sebba [1], are characterized by small bubble sizes (25–100 µm) and a dual-shell structure, setting them apart from conventional foams [2]. Brookey was the first to utilize CGAs in drilling fluids, blending surfactants and polymers to create encapsulated microbubbles [3]. Aphrons constitute 10 to 15% of drilling fluid under atmospheric pressure and are generated through high-speed mixing, eliminating the need for high-pressure equipment. CGA fluids effectively mitigate fluid invasion in porous media through two mechanisms: faster movement of aphrons during mud influx and increased low-shear-rate viscosity, halting penetration [4]. Their soft, non-adhesive seal simplifies cleanup and maintains high return permeability, proving advantageous in depleted or fractured formations [5,6]. Aphrons present a solution to drilling challenges in mature oil fields by minimizing fluid losses and allowing for safer drilling without underbalanced conditions [7,8,9]. Additionally, they have been shown to improve drilling speed and establish effective seals, making them valuable in high-angle and horizontal wells across various worldwide applications.
Across global basins, CGA (aphron-based) drilling fluids have repeatedly shown effectiveness in depleted, fractured, and low-pressure formations by controlling losses and enabling safe overbalanced or near-balanced drilling where conventional approaches, including underbalanced drilling, were unsuccessful [9,10,11,12,13,14,15]. Building on these field outcomes, current research is increasingly directed toward improving the environmental profile of CGA systems through the use of plant-derived surfactants and other biodegradable additives that can still generate stable aphron structures [16,17,18,19,20,21,22]. In parallel, nanoparticles such as silica or nano-clays have been incorporated to strengthen the aphron shell and polymer network, improving bubble stability, increasing low shear viscosity, and enhancing pore or microfracture sealing, with reported resistance to thermal and mechanical stresses relevant to downhole conditions [18,19,23,24,25].
Table 1 brings together three lines of work on CGA drilling fluids: eco-friendly, non-eco-friendly, and semi-eco-friendly systems. Eco-friendly papers use plant-based surfactants (such as saponins and Olea europaea extracts) with natural polymers like xanthan or starch. They repeatedly show stable microbubbles, shear-thinning flow, and lower API fluid loss, and they often improve performance further with gentle solid aids (e.g., fumed/colloidal silica). Some starch-amino-acid systems also keep good properties at higher temperatures. Non-eco-friendly studies, built around conventional surfactants (SDS/SDBS or cationic analogs), provide the baseline mechanics: more polymer usually shrinks and stabilizes bubbles; more surfactants can increase bubble size and raise apparent viscosity. These fluids are well described by standard rheology models (Power Law or Herschel–Bulkley), and stability often peaks near 90 °C before filtration control weakens at higher temperatures. Semi-eco-friendly work sits in between. It mixes natural/modified polymers with either conventional surfactants or nanoparticles. By tuning particle wettability and building polymer-particle networks, these studies raise low-shear viscosity, strengthen bubble shells, and cut filtrate, and some show long half-life at elevated temperatures. However, most still rely on SDS/SDBS or inorganic nanoparticles, so they are only partially green.

Table 1.
Summary of CGA formulations from the literature categorized as eco-, semi-eco, and non-eco-friendly, with materials, concentrations, and key outcomes.
The performance metrics selected in this study are aligned with the governing CGA loss-control mechanisms and are reported in a field-relevant manner. Aphron size distribution is directly linked to bridging and sealing efficiency in loss zones. Yield-stress and shear-thinning rheology control transport, suspension, and the low-shear-rate viscosity response that suppresses further invasion. Therefore, these indicators are evaluated together with LTLP filtration and mud-cake characteristics to connect microstructure to practical fluid-loss control [4,16,17,22,23,24,25,26,27,28]. Because temperature can influence both interfacial stabilization and polymer-network strength, the 25 to 90 °C window is used to assess whether the aphron population and rheological profile remain stable as thermal thinning becomes more pronounced and to maintain consistency with the temperature ranges commonly discussed for CGA systems in the literature [17,22,23,24,25,26,27,29]. To facilitate translation of the laboratory findings into formulation decisions for depleted and fractured formations, results are presented using drilling-fluid design parameters and standard LTLP filtration response supported by mud-cake observations, while all formulations are prepared and evaluated using a consistent workflow to improve comparability and reproducibility across systems [4,7,8,9,11,30,31].
Across all streams, two ideas are consistent: CGA fluids behave as yield-stress, shear-thinning materials [19,20,21,22,23,25,26,32,33], and finer, denser bubble populations with modest interfacial reinforcement reduce filtrate [13,16,17,18,21,22,23,26,27,29,33,34,35,36]. Despite recent progress, there are still few studies that combine a biosurfactant with a nanobiopolymer and compare the results to conventional fluids for rheology, aphron size, and filtration. The present study addresses this gap by combining a peanut-oil-derived surfactant with a nanostructured corn biopolymer, and by evaluating aphron size distribution and rheology within a practical 25 to 90 °C window. The aim and main advantages are a fully bio-based CGA design without synthetic surfactants or inorganic nanoparticles, and a mechanism-based evaluation linking aphron size, rheology, and LTLP filtration to practical fluid-loss control.
2. Materials and Methods
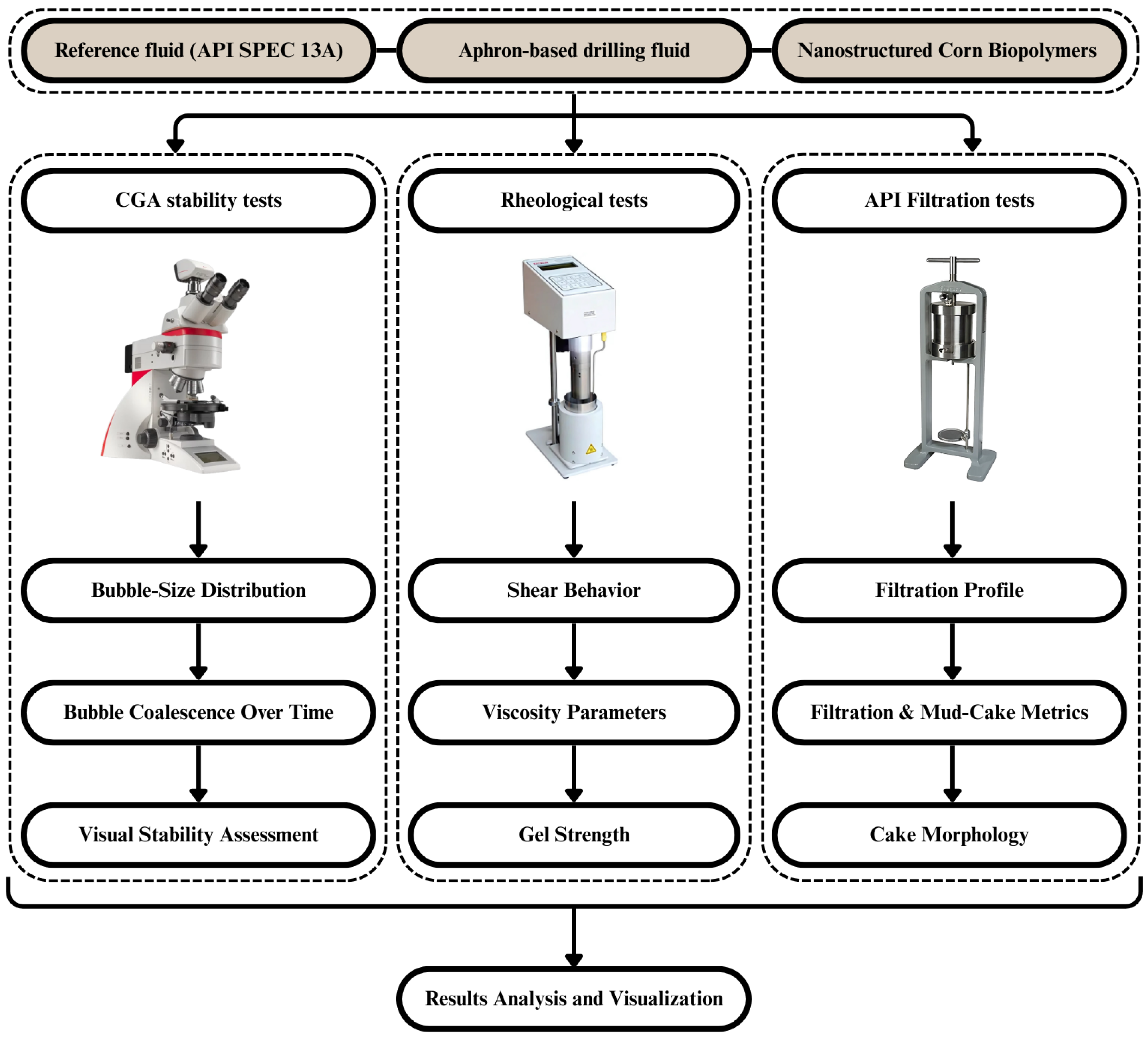
Figure 1 represents the experimental process for the production of CGA-based sustainable drilling fluids. A bentonite reference fluid was prepared with a peanut-oil-derived natural surfactant and a nanostructured corn biopolymer (NCBP). The formulations were tested over various concentrations and at different temperatures (25 °C to 90 °C). Aphron stability was evaluated with visual monitoring and microscopic imaging techniques using a Leica DM2700 P polarized light microscope (Leica Microsystems CMS GmbH, Wetzlar, Germany). Rheological properties were measured with a Grace M3600 viscometer (Grace Instrument Company, Houston, TX, USA), and API filtration tests evaluated fluid loss and mud cake quality. The effects of additives on performance were evaluated by processing the results. These procedures were performed in laboratories to study the performance of the CGA drilling fluids prepared for testing in terms of aphron stability and for rheological, filtration performance, in addition to other aspects.
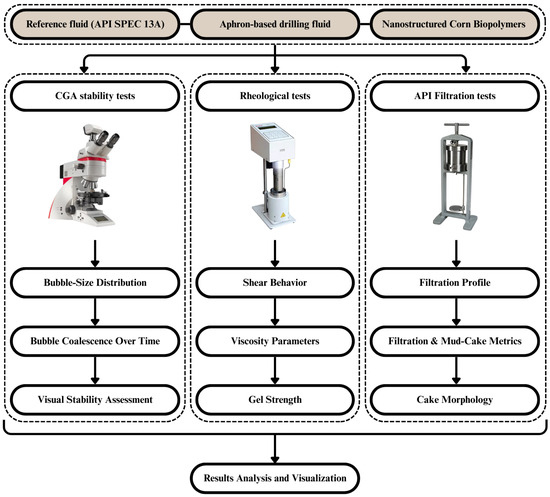
Figure 1.
Flowchart of experimental procedure design and evaluation of CGA-based drilling fluids.
The tests obtained are the essential evidence as to the influence with which the addition of natural surfactants and nanostructured corn biopolymer enhanced the important drilling fluid behaviors under controlled environments. The reference fluid (RF), aphron-based fluids (ABFs), and nanobiopolymer-enhanced fluids (NCBPs) were compared in the three significant test types such as CGA stability, rheology and API filtration (see Figure 2). Rheological recording was performed at 25 °C, 50 °C, 70 °C and 90 °C to obtain temperature-dependent performance of the fluids. The key parameters included bubble-size distribution, apparent and plastic viscosity, gel strength, filtrate volume, and mud-cake morphology. Such a comprehensive testing setup allows quantitative evaluation and visual assessment of formula for an integrated approach to better evaluate the formulation effects in performance enhancement.
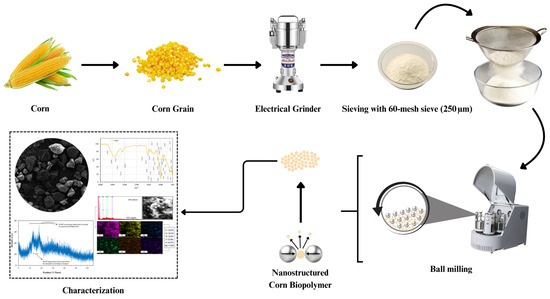
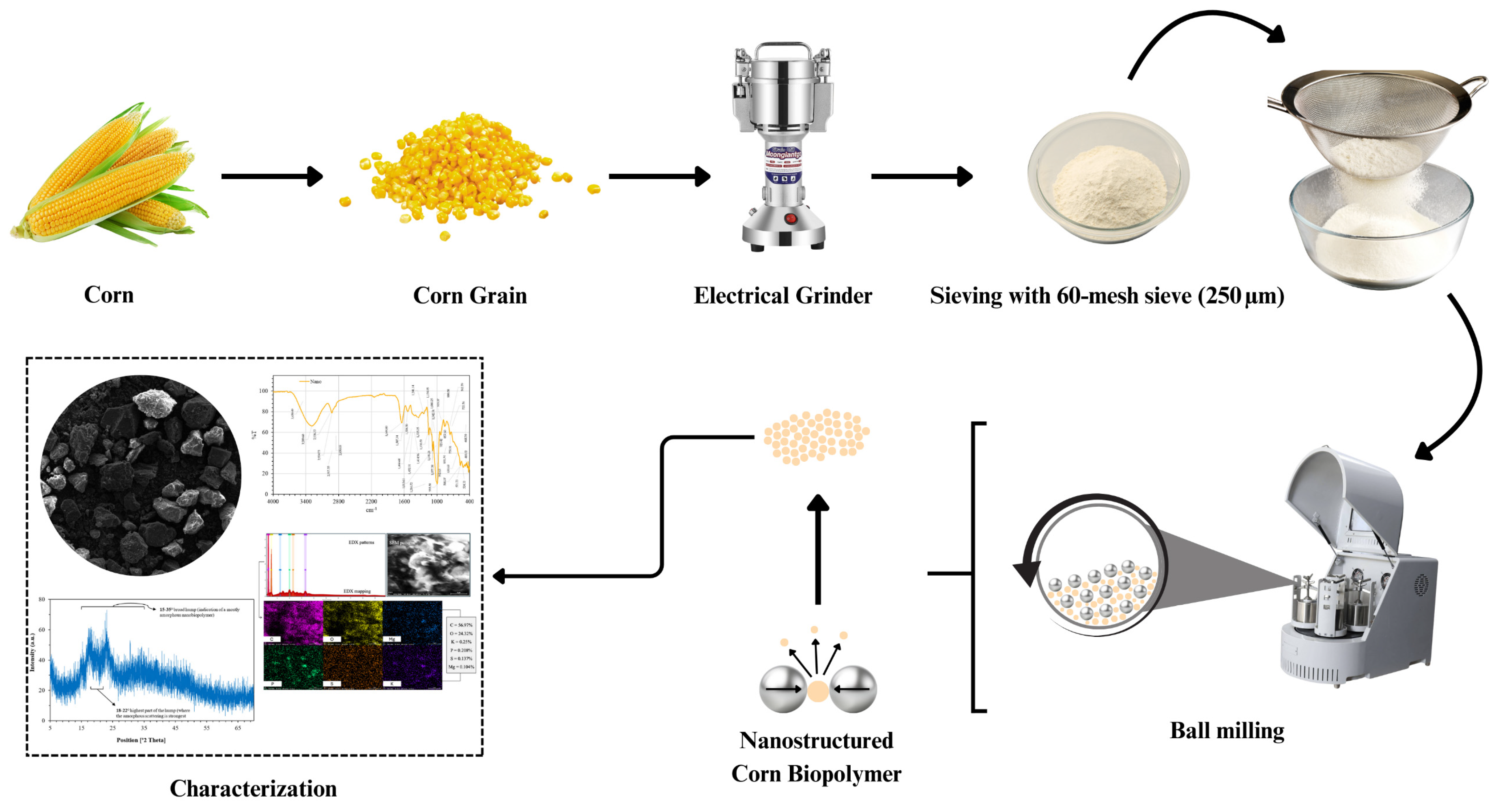
Figure 2.
Schematic representation of the preparation and characterization workflow for nanostructured corn biopolymer, including grinding, sieving, and ball milling.
2.1. Materials
The CGA-based drilling fluids were formulated using distilled water, soda ash, sodium hydroxide (NaOH), bentonite, xanthan gum (XG), a peanut-oil-derived surfactant, and nanostructured corn biopolymer. A reference fluid (RF) with no surfactant and no nanomaterial was prepared for baseline testing. Three aphron-based fluids were prepared with increasing surfactant concentrations, while three nanostructured corn biopolymer-enhanced fluids contained a fixed surfactant level and varying nano concentrations. All samples shared the same base additives and were prepared in 200–350 mL volumes (Table 2).

Table 2.
Formulation Details of Reference, Aphron-Based, and Nanostructured Corn Biopolymer-Enhanced CGA Fluids.
2.2. Preparation of Nanostructured Corn Biopolymer and Natural Surfactant
The nanostructured corn biopolymer was prepared through a top-down mechanical process involving sequential grinding, sieving, and ball-milling steps. As illustrated in Figure 2, whole corn kernels were first dried and mechanically grounded using an electric grinder to produce a coarse powder. The material was then passed through a 60-mesh (250 µm) sieve to remove oversized particles and collect the fine fraction. To further reduce particle size to the nanoscale, the sieved powder was subjected to high-energy ball milling, where repeated collisions and shear forces facilitated particle fragmentation and nano structuring. After the synthesis, the material underwent comprehensive physicochemical characterization. Elemental composition was determined via Energy-Dispersive X-ray Spectroscopy (EDX), while surface morphology was observed using Scanning Electron Microscopy (SEM). Functional groups were identified through Fourier-Transform Infrared (FTIR) Spectroscopy across the 400–4000 cm−1 range. Thermal behavior was analyzed using Thermogravimetric Analysis (TGA) under nitrogen atmosphere, heating from 0 to 700 °C at a rate of 10 °C/min. Particle-size distribution in aqueous dispersion was measured by Dynamic Light Scattering (DLS), and detailed nanoscale features were visualized using Transmission Electron Microscopy (TEM).
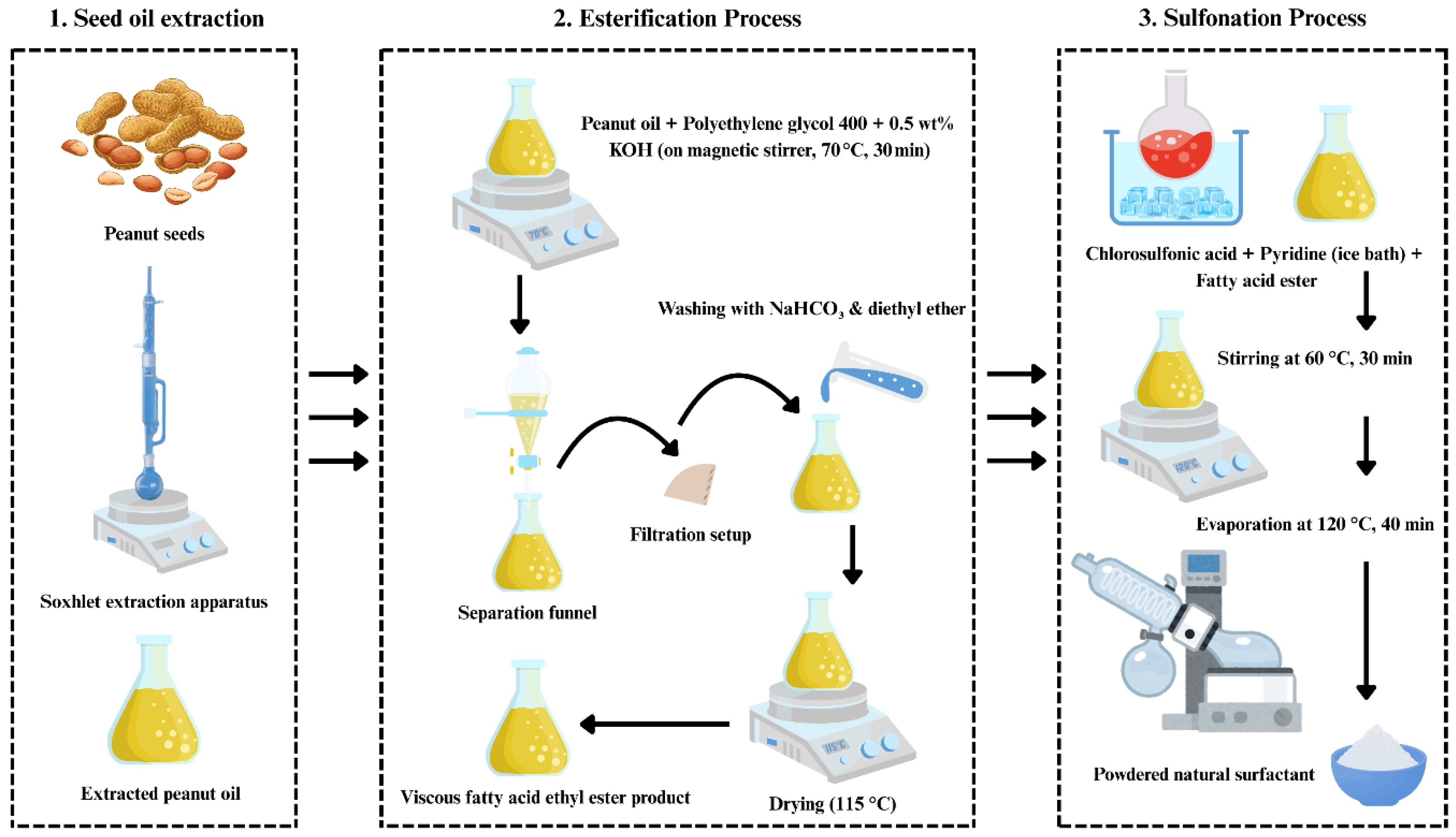
Together, these techniques confirmed the successful formation of a nanostructured, chemically active, and thermally stable corn-based biopolymer. In addition, the natural surfactant was synthesized in two steps (Figure 3). First, peanut oil was esterified with PEG-400 using 0.5 wt.% KOH at 70 °C for 30 min; the upper ester phase was separated, washed with NaHCO3/diethyl ether, and vacuum evaporated at 115 °C. Second, the ester was sulfonated by adding chlorosulfonic acid to pyridine under ice cooling, then stirred at 60 °C for 30 min; the mixture was quenched with Na2CO3/NaHCO3, extracted with n-butanol, and the aqueous phase was vacuum evaporated at 120 °C to yield sodium ethyl-ester sulfonate. Its critical micelle concentration (CMC) is 2000 ppm, the same as SDS, a widely used synthetic surfactant in CGA systems [37,38].
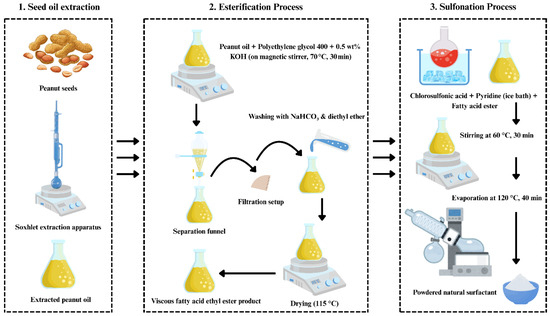
Figure 3.
Stepwise synthesis of the natural surfactant from peanut oil via seed-oil extraction, esterification, and sulfonation processes.
2.3. Formulation of the CGA Drilling Fluid
The CGA drilling fluids were prepared by sequentially adding soda ash, NaOH, and bentonite to distilled water, followed by stirring at 600 rpm for 30 min and static aging for 20 h, in accordance with API Specification 13A, Nineteenth Edition, 2019 [39]. In nano-based formulations, nanostructured corn biopolymer was added after the alkalis. Xanthan gum was then incorporated and mixed at 12,000 rpm for 15 min, followed by the addition of the surfactant, which was homogenized at 12,000 rpm for 3 min to generate the aphron structure. Rheological and filtration properties were evaluated using a Grace M3600 viscometer and a standard Fann Series 300 API Filter Press (Fann Instrument Company, Houston, TX, USA). Morphological features were examined using a Leica DM2700 P polarized light microscope.
2.4. CGA Stability Tests
ABF2 was chosen, as it was the most appropriate surfactant concentration among its formulations, and thus allowed close observation for aphron stability evaluation. It produced a finer and more stable aphron population, with higher bubble count and a better size distribution with less coalescence. It also maintained the desired yield-stress and shear-thinning behavior and showed lower LTLP filtrate than the other surfactant concentrations. It was prepared using 200 mL of distilled water, after which the total volume expanded to approximately 750 mL due to aphron generation. Foam collapse and phase separation were monitored over time under static conditions to measure aphron stability. Bubble size distribution and structural integrity were also examined by performing microscopic analysis. Images at different time intervals and surfactant concentrations were taken to examine how formulation affected aphron morphology and the tendency for bubble coalescence.
2.5. Rheological Tests
Values for rheological behavior of CGA drilling fluids were determined using a Grace M3600 viscometer at 25 °C, 50 °C, 70 °C, and 90 °C. Standard rotational speeds were measured at 600 and 300 rpm to calculate plastic viscosity (PV), yield point (YP), and apparent viscosity (AV) under API RP 13B-1 conditions [40]. Gel strengths for 10 s and 10 min were also collected for establishing the stability of the fluid. Alongside the standard values, additional measurements were recorded at 200, 100, 60, 30, 6, and 3 rpm to achieve a more detailed assessment of the shear stress/viscosity/shear rate relationship. These longer data points allowed for the derivation of detailed flow curves and subsequent explanation of the non-Newtonian fluid behavior of fluids. Comparisons of reference fluid, aphron-based fluids, and nanostructured corn biopolymer-enhanced fluids were carried out to assess the impact of peanut-oil-derived surfactant and nano concentrations upon rheological performance on the respective formulations of the two fluids at different temperature and shearing conditions.
2.6. API Filtration Test
CGA drilling fluids filtration performance was tested using the Fann Series 300 API Filter Press at a differential pressure of 100 psi for 30 min, according to API RP 13B-1. All fluid samples were tested at ambient temperature using Fann special hardened filter paper (3.5 in, 9 cm diameter). The 100-psi differential pressure was supplied from a nitrogen cylinder using controlled pressure regulators to maintain stable test pressure. During the process, the filtrate volume was measured at 5 s intervals for filtration rate profile and early-stage instability detection. At the close of the 30 min duration, total filtrate volume was determined. The thickness and visual properties of the resulting mud cake were also checked and recorded. These results were applied to demonstrate the efficacy of aphron structures and nanostructured corn biopolymer additives for controlling fluid loss as well as improving mud cake quality. To establish the influence of formulation variables on filtration performance, a comparative analysis was done by the reference fluid, aphron-based fluids, and nanostructured corn biopolymer-enhanced fluids.
3. Results and Discussion
3.1. Characterization of the Nanobiopolymer and Natural Surfactant
3.1.1. XRD and EDX Analyses of Nanostructured Corn Biopolymer
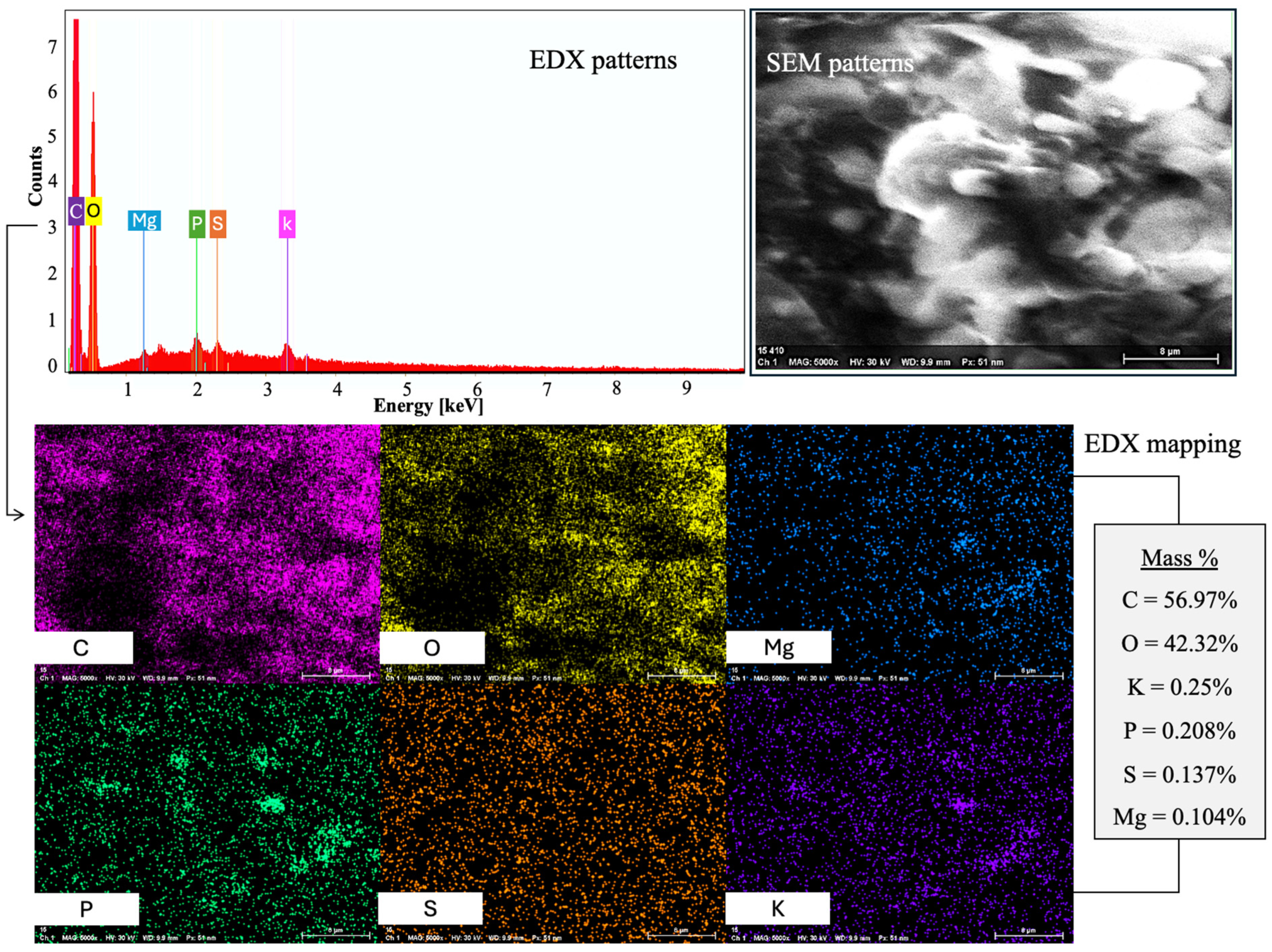
Energy-Dispersive X-ray Spectroscopy (EDX) was applied to ascertain the elemental composition of the nanostructured corn biopolymer. Results showed that the material is mainly composed of carbon (56.98%) and oxygen (42.32%), which supports its organic biopolymeric character. Minor elements including potassium (0.25%), phosphorus (0.21%), and sulfur (0.14%) were also detected, which could be due to innate biological residues or processing agents. The high levels of carbon and oxygen correspond to the expected composition of polysaccharide-based biopolymers made from corn. The atomic percentage also shows a tendency for carbon (64.01%) to be dominant over oxygen (35.70%), consistent with its carbohydrate-rich nature. Phosphorus and sulfur residues in trace amounts may also suggest residual phytochemicals or enzymatic interaction from corn grain processing [28]. The spectrum and elemental breakdown corresponding to this analysis is shown in Figure 4.
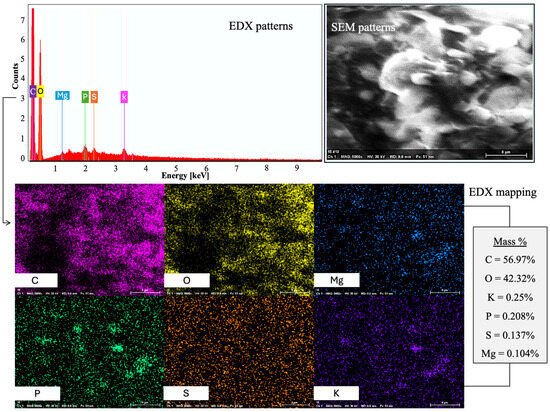
Figure 4.
The NCBP’s FESEM-EDX reveals a C-O-rich matrix with scattered Mg, P, S, and K.
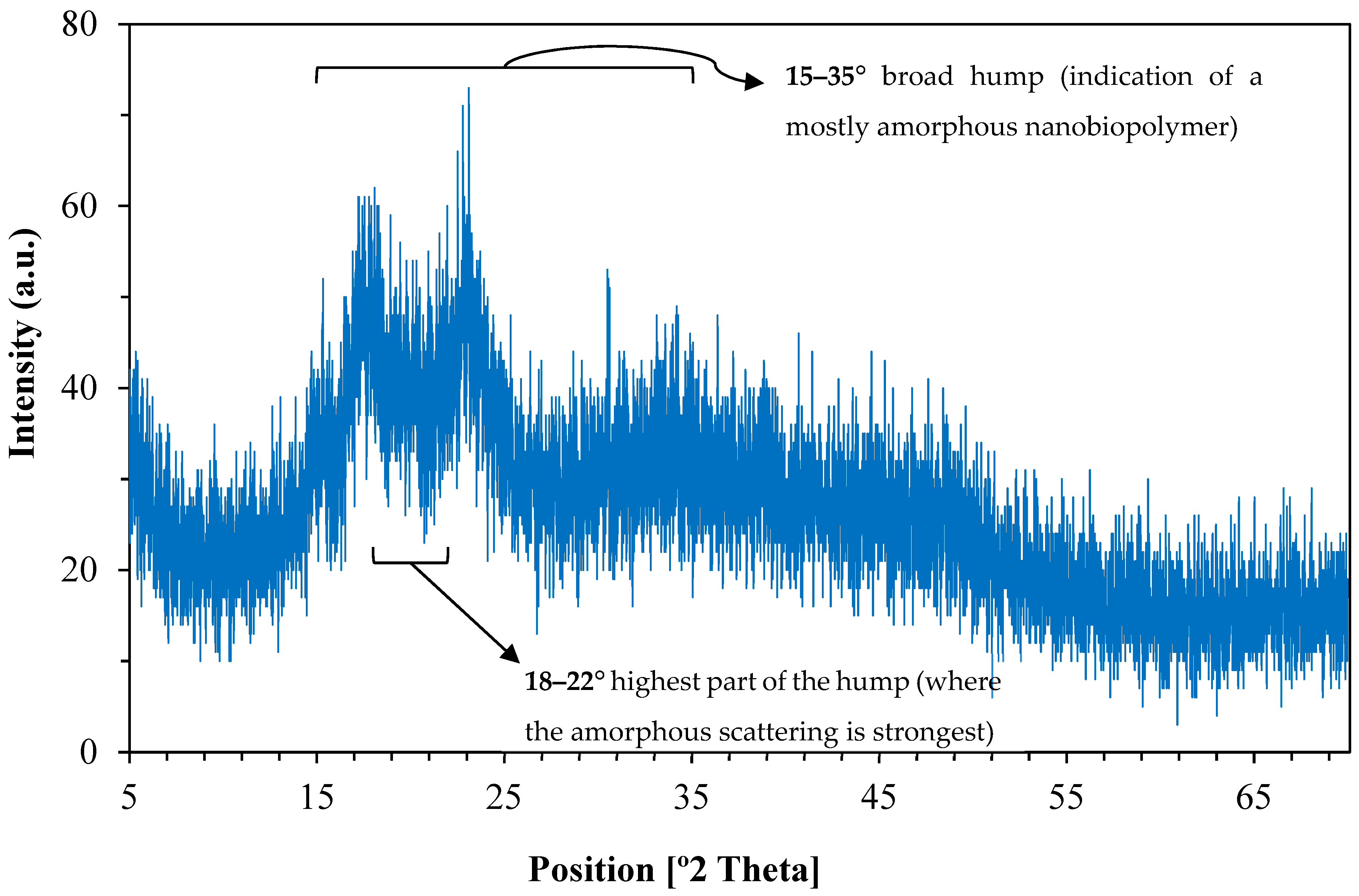
The X-ray diffraction (XRD) pattern of the synthesized nanostructured corn biopolymer presented in Figure 5 shows a broad hump centered around 2θ = 18–22°, suggesting an amorphous or poli-crystalline behavior [41]. With no sharp diffraction peaks, it can be inferred that there is no long-range crystalline order, which is common in biopolymers and indicates the formation of nanoparticles from ball milling [42,43]. The gradual decline in intensity beyond 30° also confirms the disorderly molecular structure characteristic of biopolymeric nanostructures. This amorphous nature can be advantageous in some drilling fluid applications, for example fluid loss control and stabilization, as they achieve enhanced dispersion and interaction with other components [44].
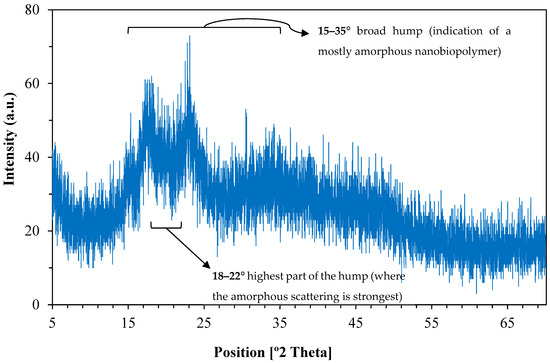
Figure 5.
XRD pattern of the nanostructured corn biopolymer shows a broad amorphous peak (18–22°) indicating low crystallinity.
3.1.2. FTIR Spectrum
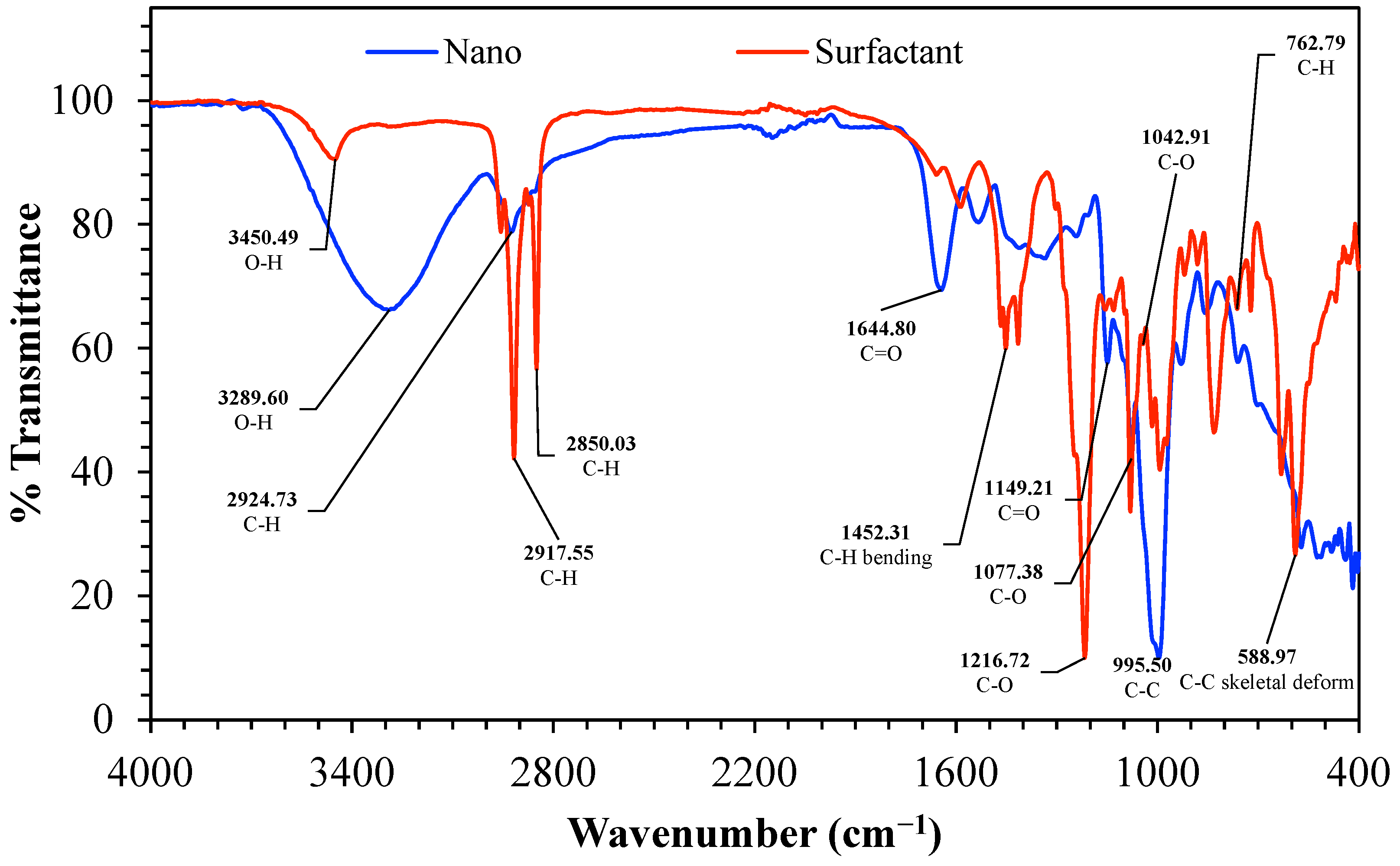
FTIR spectra provided in Figure 6 contrast the nanostructured corn biopolymer (Nano, blue) and the peanut-oil-derived surfactant (Surfactant, red) and confirm the anticipated functionalities in both materials. The Nano spectrum consists mainly of a broad O-H stretching envelope in the region 3600–3000 cm−1, with strong peaks at 3450.49 and 3289.60 cm−1, consistent with hydroxyl-rich polysaccharide backbones and high hydrogen-bonding content [45]. The aliphatic C-H stretching in the same spectrum is weaker and occurs at about 2924.73 cm−1, in accordance with the residual hydrocarbon parts in the biopolymer mixture [46]. The surfactant spectrum features stronger aliphatic features like these with clearest C-H stretching bands at 2917.55 and 2850.03 cm−1 representing the long fatty chain contribution typical of oil derived molecules.
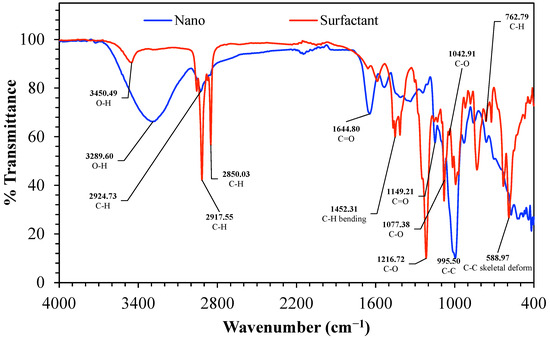
Figure 6.
FTIR spectrum of the nanostructured corn biopolymer, indicative of polysaccharide-based biopolymer structure. and the peanut-oil-derived surfactant, highlighting hydroxyl, aliphatic, carbonyl, and ester functionalities relevant to surfactant performance.
For the mid-wavenumber region, the band at 1644.80 cm−1 assigned to C=O describes carbonyl containing groups, and the 1452.31 cm−1 band represents C-H bending, thus providing evidence for the surfactant fraction containing ester or another related carbonyl chemistry. The region of the fingerprint also distinguishes the two materials; 1216.72, 1149.21, 1077.38, and 1042.91 cm−1 exhibit prominent C-O stretching bands (consistent with ether or ester C-O linkages and for the Nano, polysaccharide C-O-C and C-O vibrations) [45,47]. Other low wavenumber signals (C-C, C-H, and C-C skeletal deformation), around 995.50 cm−1, 762.79 cm−1, and 588.97 cm−1 respectively, indicate backbone and skeletal vibrations in the underlying organic lattice [48]. Overall, the Nano shows a broad hydroxyl-rich signature consistent with a polysaccharide-based nanobiopolymer. In contrast, the surfactant shows stronger aliphatic, carbonyl, and C-O features, supporting its role as an oil-derived amphiphilic ingredient for interfacial stabilization and improved packing or sealing in the formulated system.
3.1.3. TGA and DSC Thermal Analyses
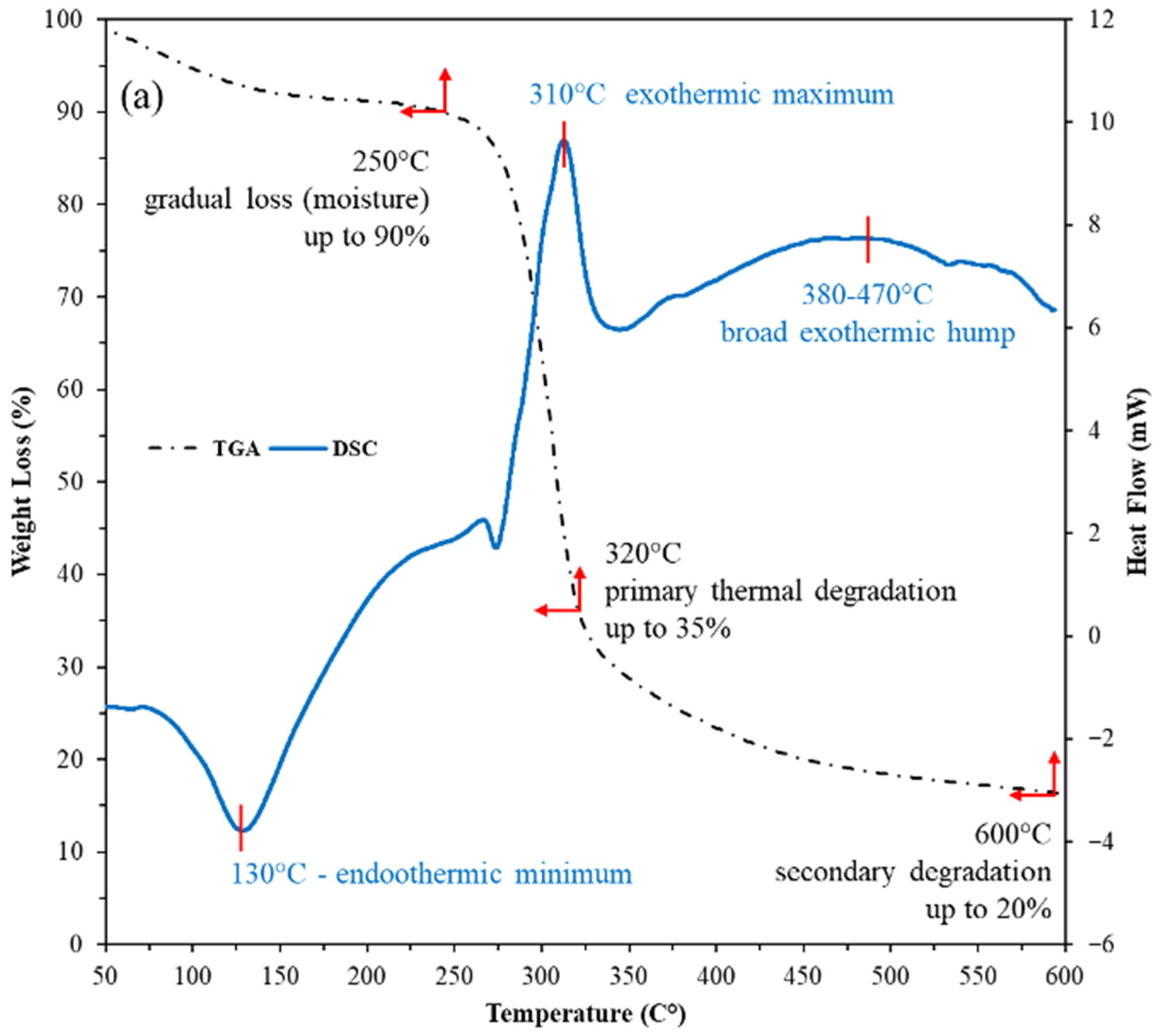
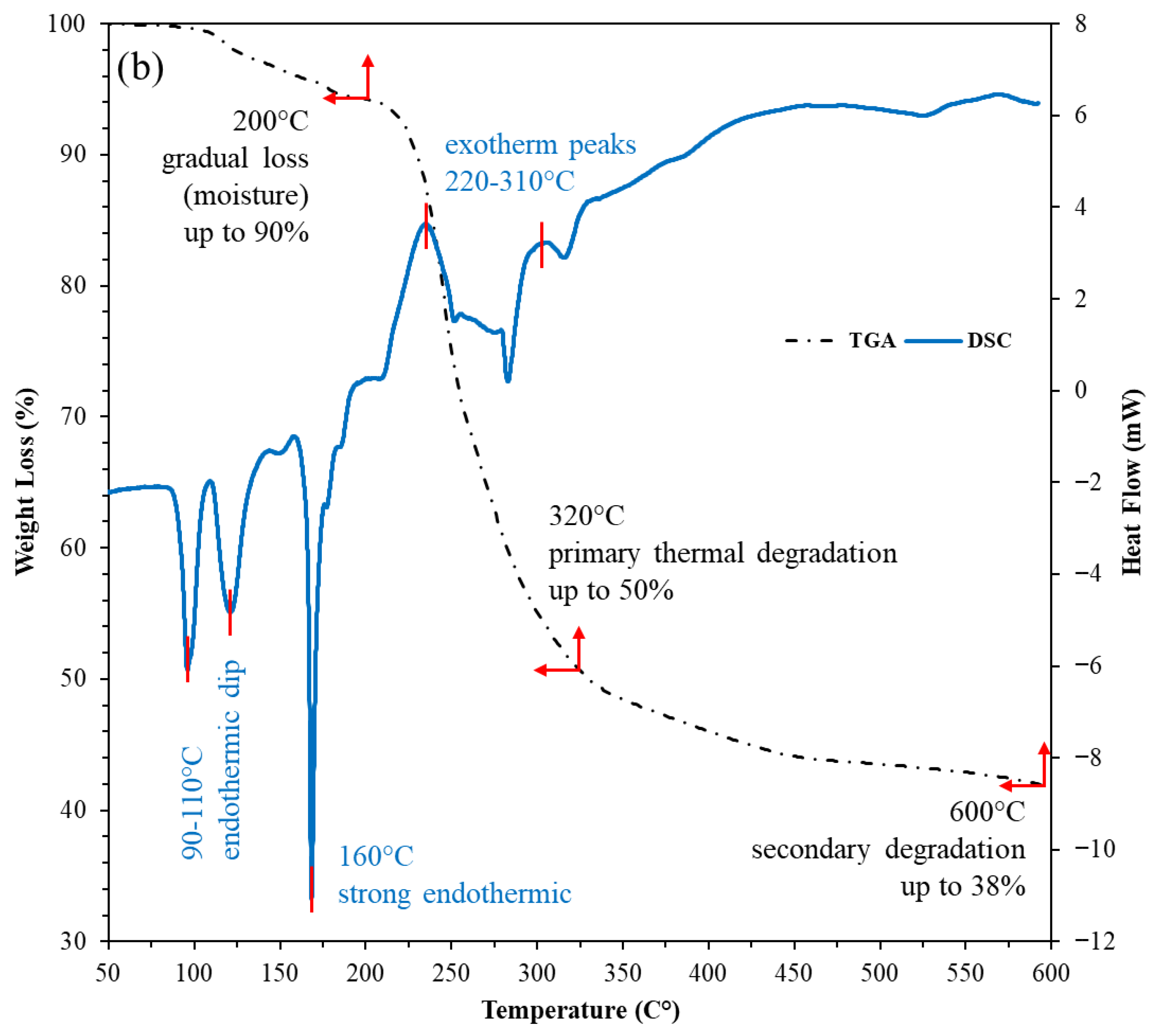
The combined TGA-DSC profiles show both the nanostructured corn biopolymer (NCBP) and the peanut-oil-derived surfactant are thermally robust within drilling/reservoir windows (Figure 7). The NCBP exhibits only minor mass loss below 250 °C (surface moisture/light volatiles), with its major degradation not initiating until 280–350 °C (TGA). Complementary DSC reveals a broad low-temperature exothermic event between 90 and 187 °C with a peak at 131 °C (ΔH = 136 J g−1), consistent with structural relaxation/reorganization and release of bound species. This is followed by a strong endothermic transition from 285 to 332 °C with a peak near 311 °C (ΔH = −120 J g−1). These features align with the onset of main chain softening just before the principal TGA step, corroborating stability across typical circulating temperatures is less than 200 °C (see Figure 7a). The surfactant shows multiple DSC features that map cleanly to its stepwise thermal behavior. Two low-temperature exotherms occur at 92–132 °C (peaks 96 and 121 °C; ΔH = 18 and 23 J g−1), suggesting ordering/crystal perfection of alkyl domains; a sharp exotherm at 166–173 °C (peak 168 °C; ΔH = 39 J g−1) indicates further ordering/crystallization; an ensuing endotherm spans 211–251 °C (peak 234 °C; ΔH = −33.7 J g−1), consistent with melting/phase transformation; and two higher-temperature, smaller exotherms at 279–290 °C (peak 283 °C; ΔH = 16.8 J g−1) and 307–325 °C (peak 316 °C; ΔH = 4.7 J g−1) [49]. These higher-T runs align with the TGA step starting around 280–350 °C (Figure 7b). Taken together, these transitions are still solidly above the drilling range conditions, indicating sufficient thermal headroom for field application, as most of the HPHT or ultra-HPHT wells’ reservoir temperature is around 175 to 220 °C [50].
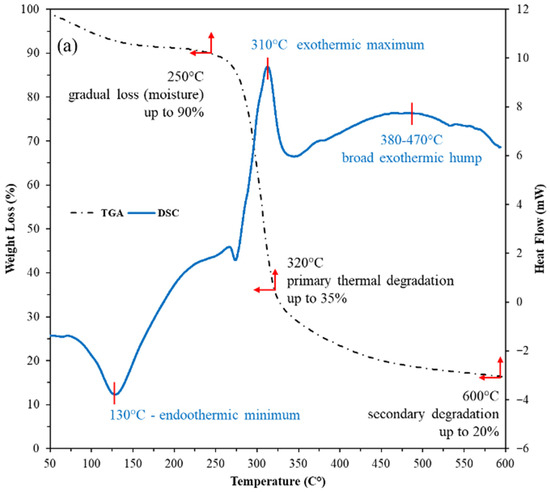
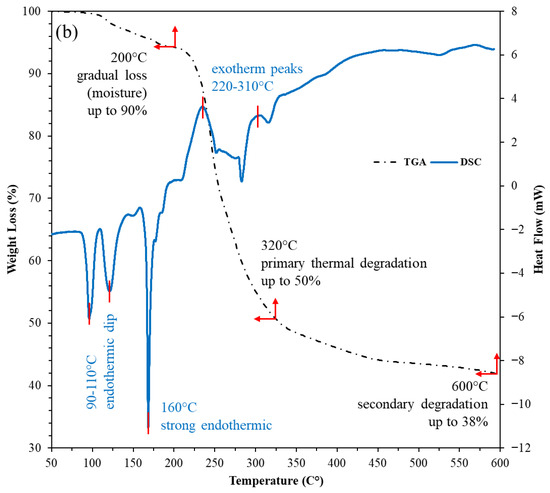
Figure 7.
TGA-DSC thermograms of; (a) nanostructured corn biopolymer, and (b) peanut-oil-derived surfactant, confirming stability well above borehole and reservoir temperatures.
3.1.4. Morphological and Particle-Size Scattering Analyses (AFM, SEM, TEM, and DLS) of Nanostructured Corn Biopolymer
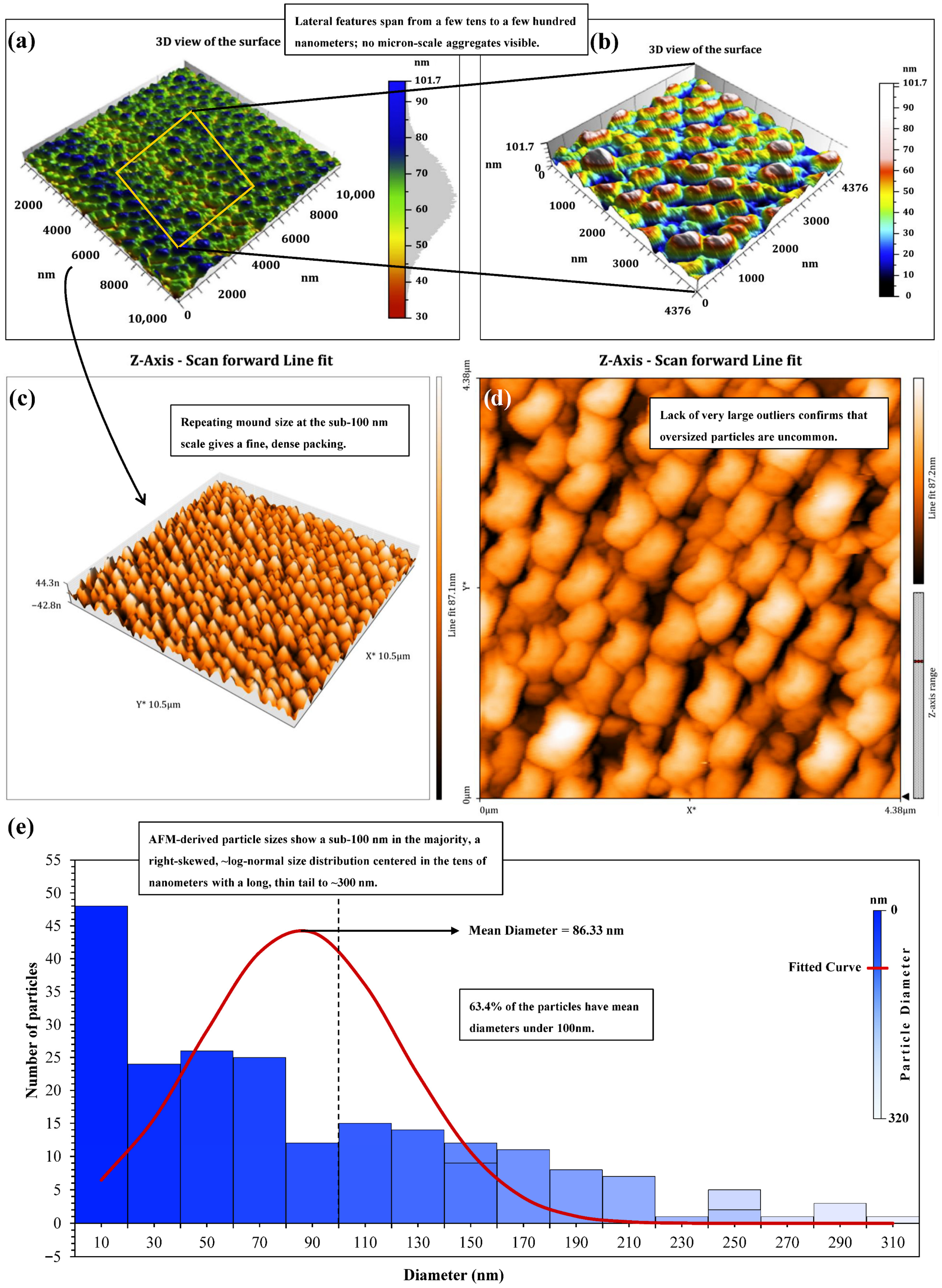
A total of four complementary techniques were utilized to study the structural properties of the synthesized nanostructured corn biopolymer: Atomic Force Microscopy (AFM), Transmission Electron Microscopy (TEM), Scanning Electron Microscopy (SEM), and Dynamic Light Scattering (DLS). Collectively, these techniques furnish qualitative as well as quantitative details of particle morphology, surface structures, and size distribution. AFM was applied to investigate surface topography and roughness on the nanoscale, providing a 3D visualization of the biopolymer’s structure. SEM and TEM techniques offered detailed information on particle morphology, with SEM affording a wider surface picture, while TEM was used for a more detailed view on internal and fine structure characteristics. DLS, on the other hand, recorded the hydrodynamic diameter of particles in suspension, factoring in hydration shells and potential aggregation. The AFM panels at multiscale reflect a well-balanced nanoscale network and not scattered micro-aggregates. The 10 × 10 µm scale view (Figure 8a) includes uniformly distributed nano-mounds that define the lateral extent (typically in the tens to few-hundred nm) of the field. The boxed zone is further studied at higher resolution (Figure 8b–d). After line-fit correction to remove stage tilt, the 3D zoom (Figure 8c) shows a closely packed, repetitive relief without a preferred in-plane alignment. The accompanying 2D height map (Figure 8d) confirms abundant, relatively equal-sized nano-mounds with small valleys. Threshold-based particle segmentation on a 4.376 × 4.376 µm window generated 213 particles comprising 14.74% of the imaged area corresponding to a particle areal density of 1.1 × 107 mm−2, so many features but no conformation to continuous ridges.
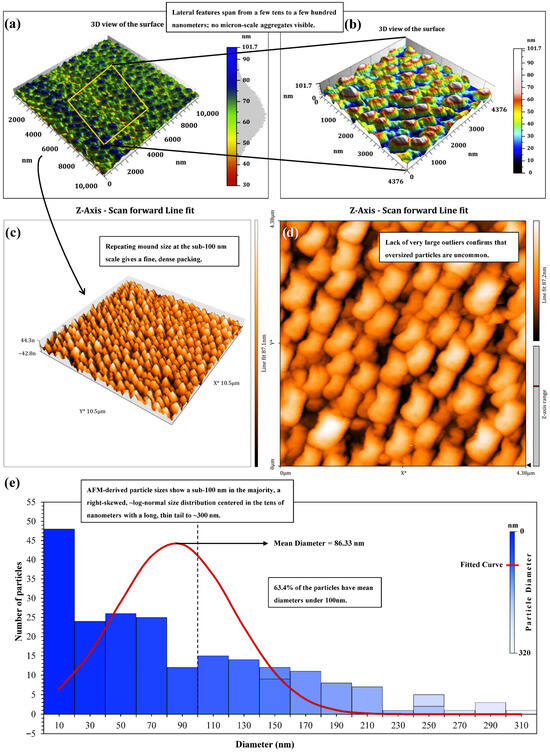
Figure 8.
AFM morphology and particle-size statistics of the nanostructured corn biopolymer (NCBP); (a) 10 × 10 µm overview; (b) 3D zoom of a 4.38 × 4.38 µm area; (c) Line-fit-corrected 3D render (tilt removed); (d) 2D height map of the same region (bright = peaks, dark = valleys), and (e) AFM-derived size histogram.
The AFM size histogram derived is shown in Figure 8e and covers mean diameters from 6 to 303 nm, with a weighted mean of 86.33 nm and a distinctively right-skewed, at approximately log-normal behavior. Most particles, 173 of them, are in the small class (<150 nm); 32 are Medium (150–230 nm), and eight are Large (>230 nm). Overall, Sub-100 nm objects predominate more than all detections (63.4%). For example, the sparse, high-diameter tail out to 300 nm suggests that oversized outlier is rare and does not influence the statistics. By thus matching together the plan-view texture with the histogram, it is therefore consistent; a dense but non-coalesced group of nano-grains that lies within 85–90 nm of center and has almost no polydispersity at the top. This morphology suggests a high interfacial area per unit volume and multiple nanoscale contact points. The particle population should diffuse easily, stabilize colloids efficiently, and contribute predictably to the flow and filtration dynamics of CGA drilling fluids while avoiding large, defect-forming agglomerates [51,52].
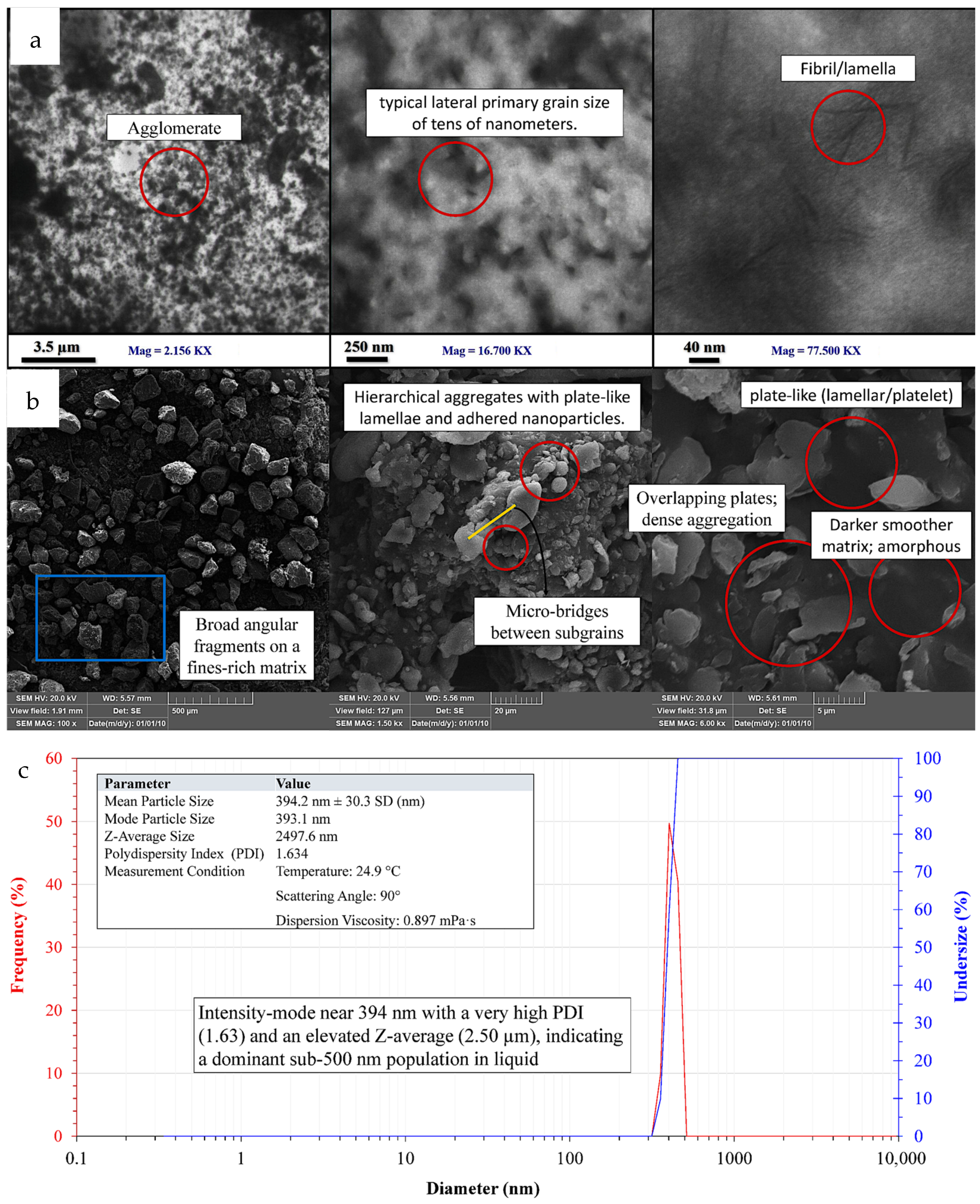
TEM images (Figure 9a) show a nanoscale, polymer-derived shape made up of thin plate-like lamellae and short fibrillar segments that look like they are partially stacked and sometimes overlap. These individual features tend to have lateral dimensions ranging from tens to low-hundreds of nanometers. In some fields, there are tight clusters where plates meet edge-to-edge or face-to-face. The differences in contrast suggest that there are areas of higher density within the platelets and lighter gaps between the plates [53]. It is consistent with the premise that semicrystalline polysaccharide domains are combined with lower-density material. This scene is unchanged with diverse magnifications. Few single lamellae are not very far apart, but most fields exhibit local aggregates which maintain the plate-like shape but form a thicker multi-plate stack.
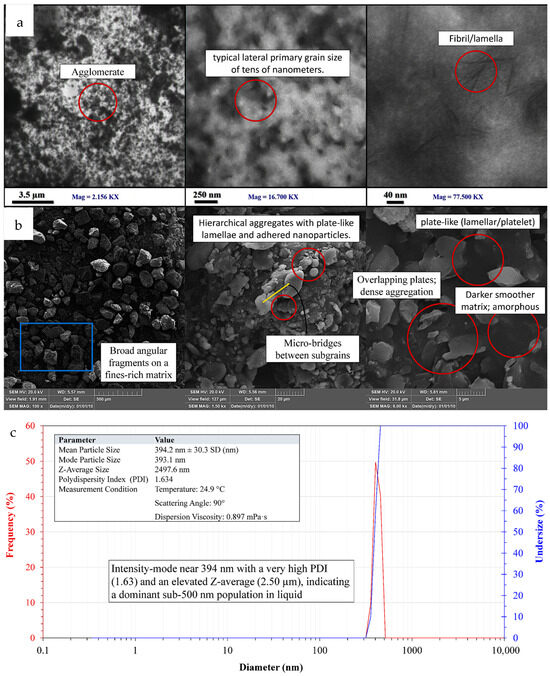
Figure 9.
TEM, SEM, and DLS of the nanostructured corn biopolymer showing platelet-like particles that aggregate into dense clusters; (a) TEM reveals fibrillar/lamellar features, (b) SEM shows hierarchical plate stacks with fines, and (c) DLS reports a main peak near 394 nm with high PDI and an elevated Z-average.
The SEM micrographs (Figure 9b) build on this by demonstrating similar platelet-dominant habits in the dry state at larger lateral scales. The platelets are densely packed and irregular with sharp edge surfaces. These platelets tend to tile, overlap, and form clusters which look like flakes. This dense packing shows that the particles are strongly sticking together probably because hydrogen bonds are forming between the hydroxyl-rich biopolymer moieties as they dry [54]. The background looks smoother and darker compared to the brighter platelets. This indicates that there may exist an amorphous or less-ordered phase that links the bright lamellae and forms contiguous domains [55]. TEM and SEM demonstrate that lamellar is the dominant morphology allowing mesoscale aggregates to assemble.
DLS data (Figure 9c) indicates that when dispersion of the same material is in liquid, similar behavior occurs. They also exhibit a peak with a high PDI near 394 nm. The large peak and high PDI indicate that the sizes are dispersed, and that soft agglomerates and possibly a hydration shell are both contributing to the increase in the hydrodynamic size picked up by DLS [56,57]. Due to very strong preference for large scatterers under intensity weighting, even a small number of micro-scale clusters can still move the peak around and increase the Z-average. It is not surprising that the features of TEM are smaller than 150 nm and DLS has larger hydrodynamic sizes. This is because imaging measures dry projected size in the primary and few-layer aggregates with immediate spatial resolution, while DLS measures how particles and clusters move as hydrated aggregates and how they scatter light in the dispersion. Microscopy reveals that the nanostructured corn biopolymer builds thin, plate-like blocks designed to stack and pack together closely. This is the reason for broad and right-skewed DLS profile and sticking-together particles. All these observations lead to a lamellar, semicrystalline biopolymer that is nanoscale at the unit of concentration of its primary units but polydisperse in suspension due to aggregation. All of its formations and connections are perfectly compatible with those of a polysaccharide-based substance.
3.1.5. 1H NMR and GC-Based Analysis of Surfactant
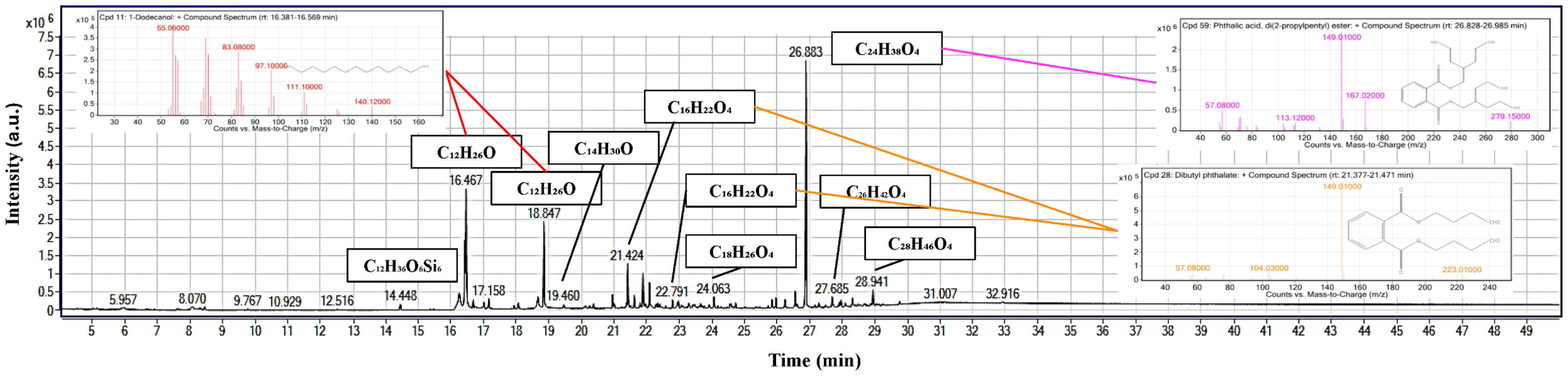
Peanut-oil surfactant, analyzed by GC-MS (Figure 10), is enriched in long-chain alcohols, fatty-acid derivatives, and antioxidant compounds, which are of particular interest for drilling fluids and aphron technology [58,59,60]. The most abundant long-chain alcohols were 1-Dodecanol (=18% of TIC area) and 1-tetradecanol, which reduce surface tension and stabilize the gas–liquid interfaces of colloidal gas aphrons [61,62], thus directly aiding the persistence of bubbles and fluid-loss control. Among those mentioned above, bis(2-ethylhexyl) phthalate (=17%) along with dibutyl and didecyl phthalates are some of the phthalate esters that can act as plasticizers and facilitate film flexibility and viscosity stabilization of aphron-based drilling fluids [63]. Identification of terephthalate esters suggests a lubricity-type contribution through friction-reduction behavior and supports thermal robustness, while the fatty-acid derivative fraction is the most defensible contributor to emulsification in drilling-fluid systems [64]. The identification of a phenolic antioxidant derivative (4-methyl-2,4-bis(p-hydroxyphenyl)pent-1-ene, 2TMS derivative, =16%) finally points out the contribution of oxidative stabilizers which can sustain the chemical integrity of the surfactant system in the harshest borehole environment [60]. Together, the profiles of these compounds suggest that the surfactant mixture has a synergistic quality that gives a good surface activity, emulsion stability, lubricity, and oxidative resistance that is crucial for maintaining aphron drilling fluids even in fractured and high-temperature reservoirs [36,65].
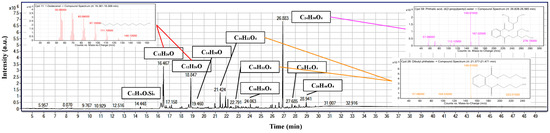
Figure 10.
GC-MS TIC of the peanut-oil-derived surfactant with key peaks annotated and representative EI spectra insets, highlighting putative alkane and fatty-acid-ester features and later phthalate-type signals.
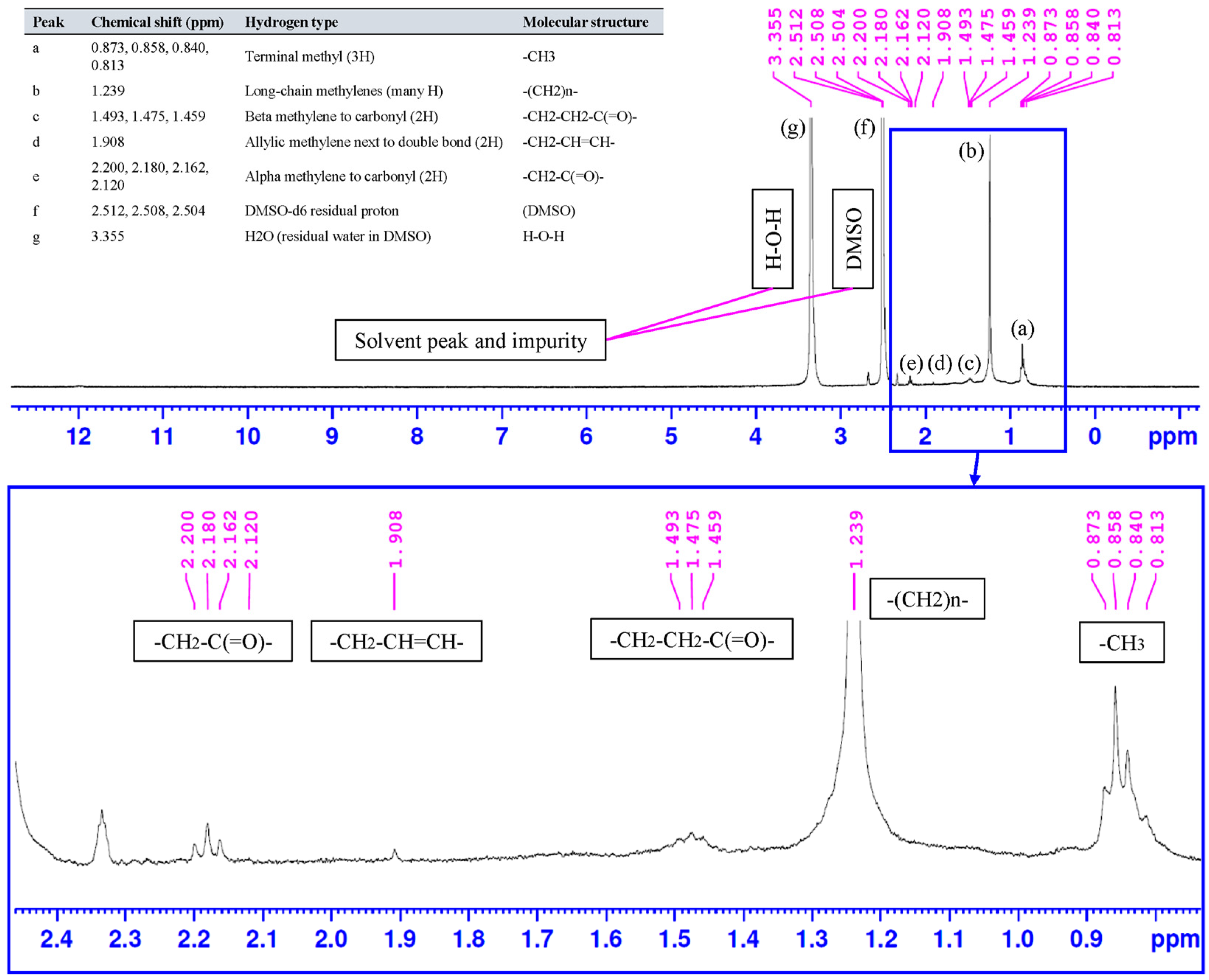
The 1H NMR spectrum of the synthesized peanut-oil-derived surfactant (Figure 11) shows the characteristic proton pattern of a long-chain fatty ester, with the main resonances concentrated in the aliphatic region. The terminal methyl group appears as a set of signals at 0.873 to 0.813 ppm (peak a). The intense envelope at 1.239 ppm (peak b) corresponds to the bulk methylene repeat units of the hydrocarbon chain, (CH2)n, confirming the dominance of long alkyl chains [66]. Signals at 1.493 to 1.459 ppm (peak c) are assigned to the beta methylene protons relative to the carbonyl group (-CH2-CH2-C(=O)-) [67]. The resonance at 1.908 ppm (peak d) is attributed to allylic methylene protons adjacent to unsaturated bonds (-CH2-CH=CH-) [68], indicating the retention of unsaturation typical of peanut-oil-derived fatty chains. The downfield cluster between 2.200 and 2.120 ppm (peak e) is assigned to alpha methylene protons next to the ester carbonyl (-CH2-C(=O)-), further supporting ester formation following the PEG-400 esterification step [69]. The solvent related signals at 2.512 to 2.504 ppm (peak f, residual DMSO-d6) and at 3.355 ppm (peak g, residual water in DMSO) are also observed [70].
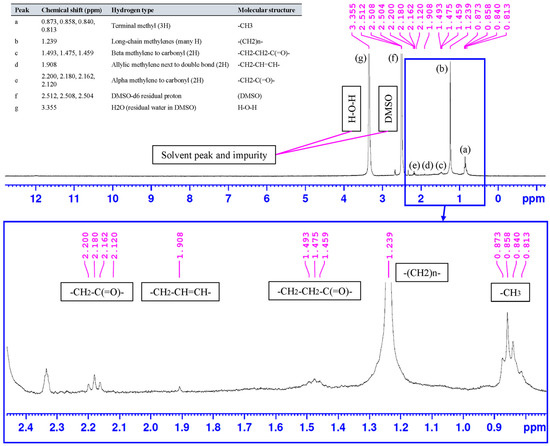
Figure 11.
1H NMR spectrum of the synthesized peanut-oil-derived surfactant (DMSO-d6) with peak assignments.
The recorded spectral window is limited and does not fully capture the region above approximately 3.33 ppm, where polyether and sulfate-adjacent methylene resonances are typically expected for PEG-based sulfated surfactants. However, the presence of these functionalities is supported by FTIR (Figure 6), which exhibits strong C-O stretching bands in the 1200 to 1000 cm−1 region consistent with PEG ether linkages and ester C-O vibrations, together with the expected aliphatic C-H stretching bands near 2920 and 2850 cm−1 [71]. Taken together, the NMR assignments confirm the fatty chain ester backbone, while the complementary FTIR features support the successful incorporation of oxygenated head-group functionalities that fall outside the captured NMR range.
3.2. Stability of the CGA
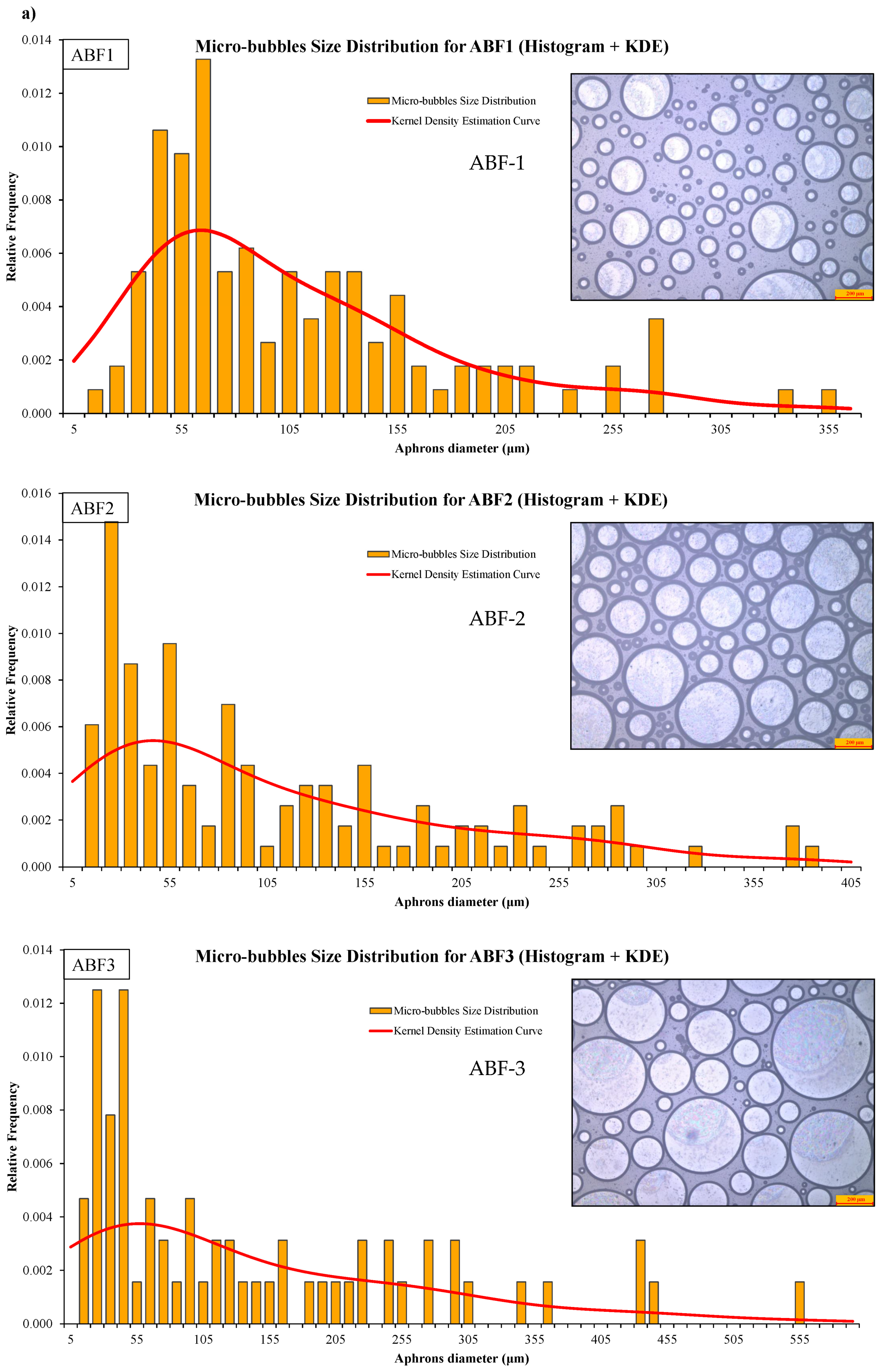
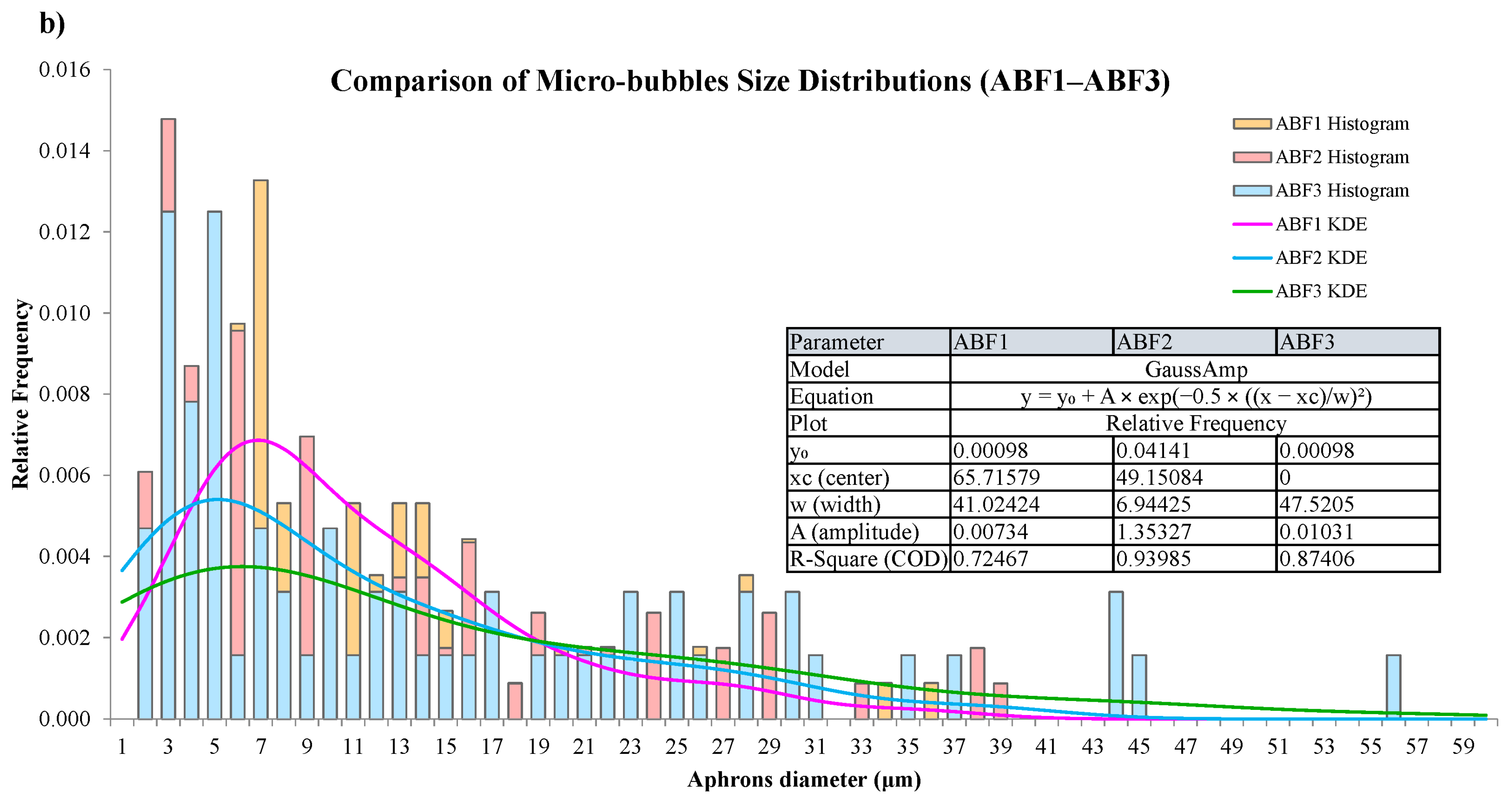
CGA fluid stability assessment was performed using bubble count, diameter distribution, time-dependent visual inspection, and statistics. Microscopic images of the CGA formed using the ABF1, ABF2, and ABF3 formulations and histograms of the size distribution of CGA structures are shown in Figure 12a. ABF2 had a narrower, more uniform, densely packed bubble population, with a narrower size range and more microbubbles with sizes <100 µm. This reinforces its choice as the best formulation. The bubble size distributions of all the aphron-based fluids were summarized in Figure 12b and kernel density estimation (KDE) was applied to estimate bubble sizes. ABF2 had a higher peak concentration and better size distributions, suggesting enhanced stability and dispersion. Fitted Gaussian model parameters of individual samples are also shown in Figure 12b. ABF2 demonstrated the smallest width (w = 6.94), maximum amplitude, and best fit (R2 = 0.94), thereby confirming its consistency of behavior with the strong and efficient generation of small and stable bubbles [72].
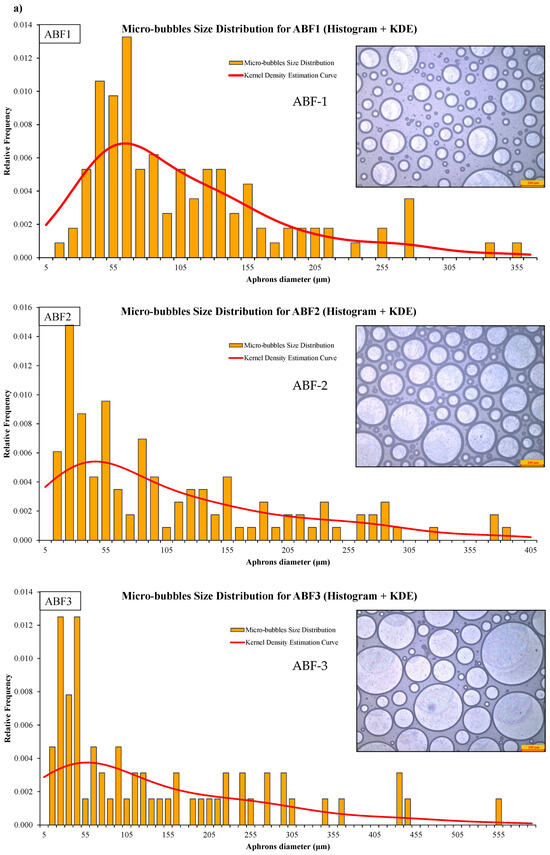
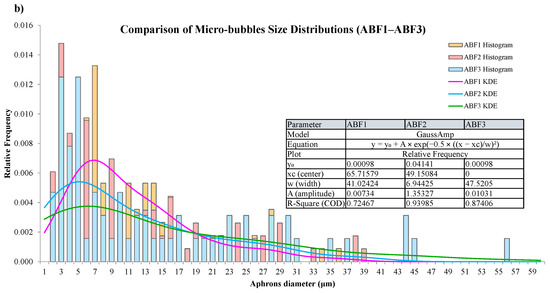
Figure 12.
Microscopic images and size-distribution histograms of aphrons in (a) ABF1, ABF2, and ABF3, and (b) comparison of aphron size distributions across ABF1, ABF2, and ABF3 using kernel-density estimation (KDE).
The microscopic evolution of ABF2 over 120 min is shown in Figure 13a. As time progressed, bubbles gradually coalesced and enlarged, resulting in a wider size distri-bution and a noticeable decline in bubble count [73]. Figure 13b shows the reduction in volume of ABF2 throughout 48 h. The initial volume of 750 mL steadily declined as aphrons began to coalesce and collapse, yet foam continued to persist significantly be-yond 24 h, indicating good temporal stability [74].


Figure 13.
(a) Tracking of aphron size and morphology in ABF2 over a 120 min period, and (b) Visual monitoring of CGA fluid volume stability (ABF2) over 48 h under static conditions.
3.3. Rheological Behavior of the CGA
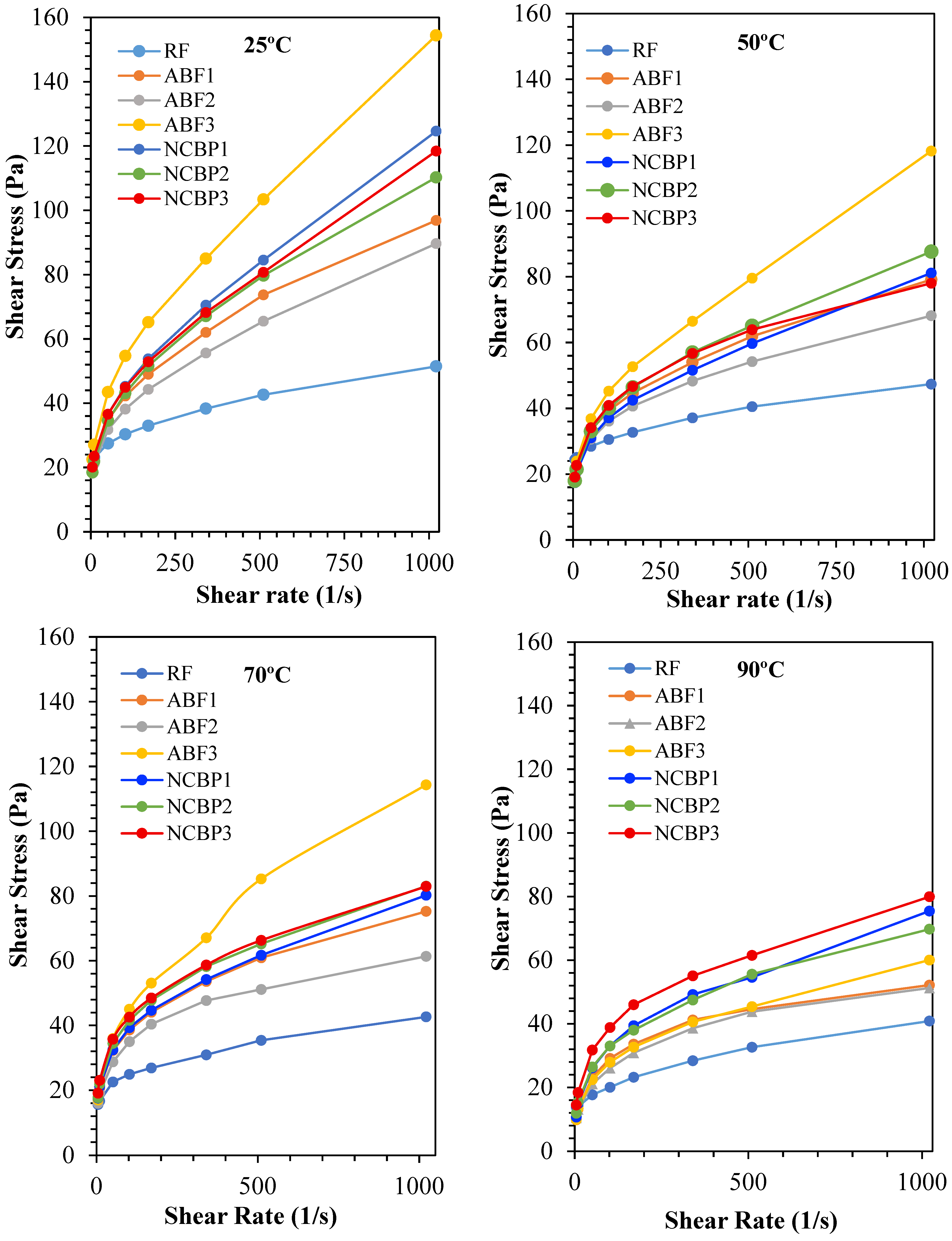
The shear stress and shear rate characteristic properties of the tested CGA-based drilling fluids were studied at 25 °C, 50 °C, 70 °C, and 90 °C to provide the properties to assess the rheological stability of the different fluid types under the increasing temperature. As anticipated, all fluids showed shear-thinning behavior, in which shear stress increases with shear rate but at a decreasing rate. From other tests performed at 25 °C, high surfactant content was associated with a higher shear stress in ABF3 followed by lower stress in ABF2 which had a more favorable structure than in ABF1 as shown in Figure 14. NCBP2 showed lower shear stress compared to NCBP1 and NCBP3, which suggested an optimum nano-loading level for maintaining flowability without over-thickening among nanostructured corn biopolymer enhanced fluids. This indicates that 1 wt.% NCBP provides sufficient microstructural reinforcement while preserving fluid flowability and aphron deformability. At 2 wt.% NCBP, the system can become over-structured, increasing low-shear resistance and limiting aphron mobility, which reduces the net benefit despite higher structural build-up.
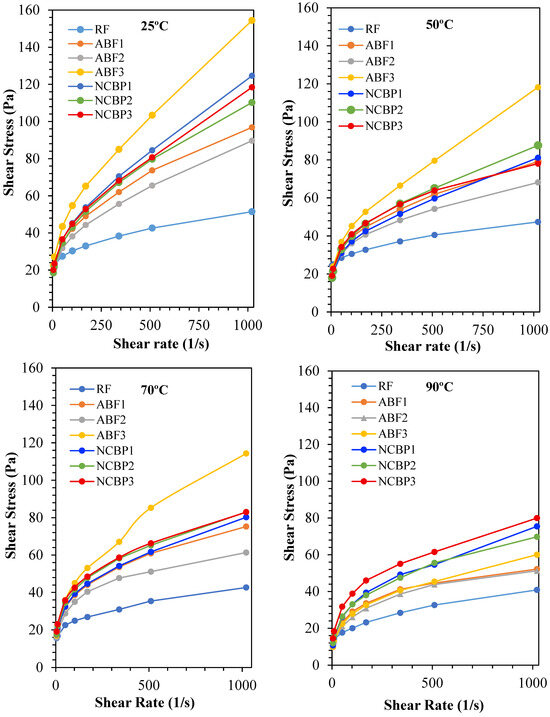
Figure 14.
Shear stress vs. shear rate profiles of all drilling fluid formulations at 25 °C, 50 °C, 70 °C, and 90 °C.
Figure 14 confirms the expected non-Newtonian drilling-fluid behavior. The shear stress increases nonlinearly with shear rate, indicating shear-thinning behavior, where the apparent viscosity (τ/γ̇) decreases as shear rate increases. At low shear rates, higher stress levels and yield-stress-like behavior indicate a stronger internal structure that supports suspension and helps reduce invasion in loss zones. At higher shear rates, the lower apparent viscosity improves pumping and circulation. The formulation differences in Figure 14 mainly reflect variations in low-shear structure strength and the degree of shear thinning.
Thermal softening was seen in all samples at 50 °C. Although ABF3 showed the highest stress profile, NCBP2 started to outperform NCBP1 and ABF1 in maintaining structural integrity, indicating that moderate nanobiopolymer loading offered the property of increased temperature stability. At 70 °C, where NCBP2 maintained a consistently superior performance compared to ABF2 and reached shear stress values of ABF1, thus demonstrating the balanced performance of NCBP2 with rheological and stability parameters is consistent. The differences were much clearer at 90 °C, where the stress in ABF3 dropped significantly and NCBP2 and NCBP3 proved to be the most thermally stable formulations, maintaining higher shear stress profiles than all ABF samples (see Figure 14). Overall, the rheogram confirms that NCBP2 outperforms the rest in thermally rheological balanced performance, enabling the satisfactory viscosity and resistance to stress in the system with higher temperature. These results confirm the synergistic effect of nanostructured corn biopolymers and surfactants to improve the high-temperature suitability of CGA-based fluids.
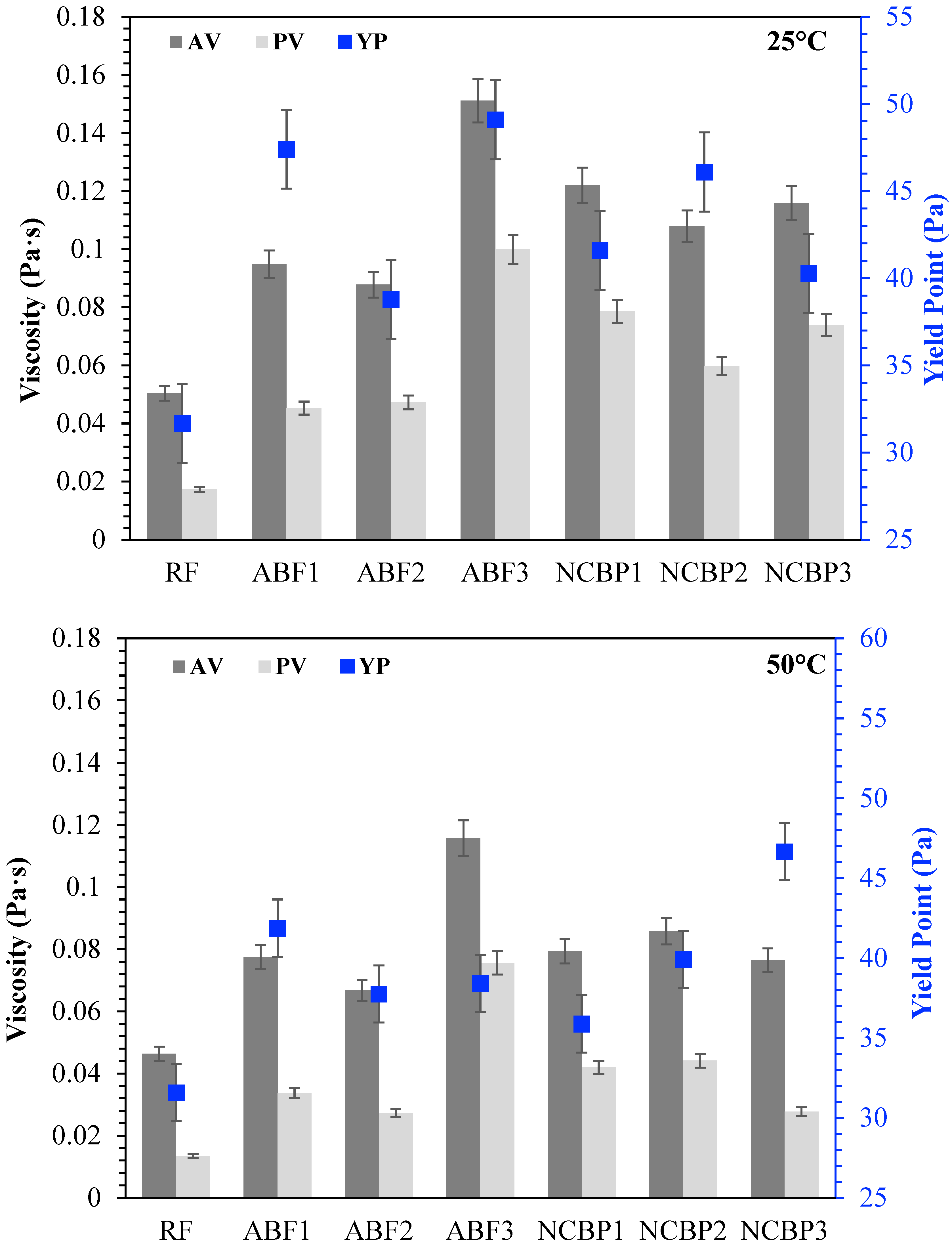
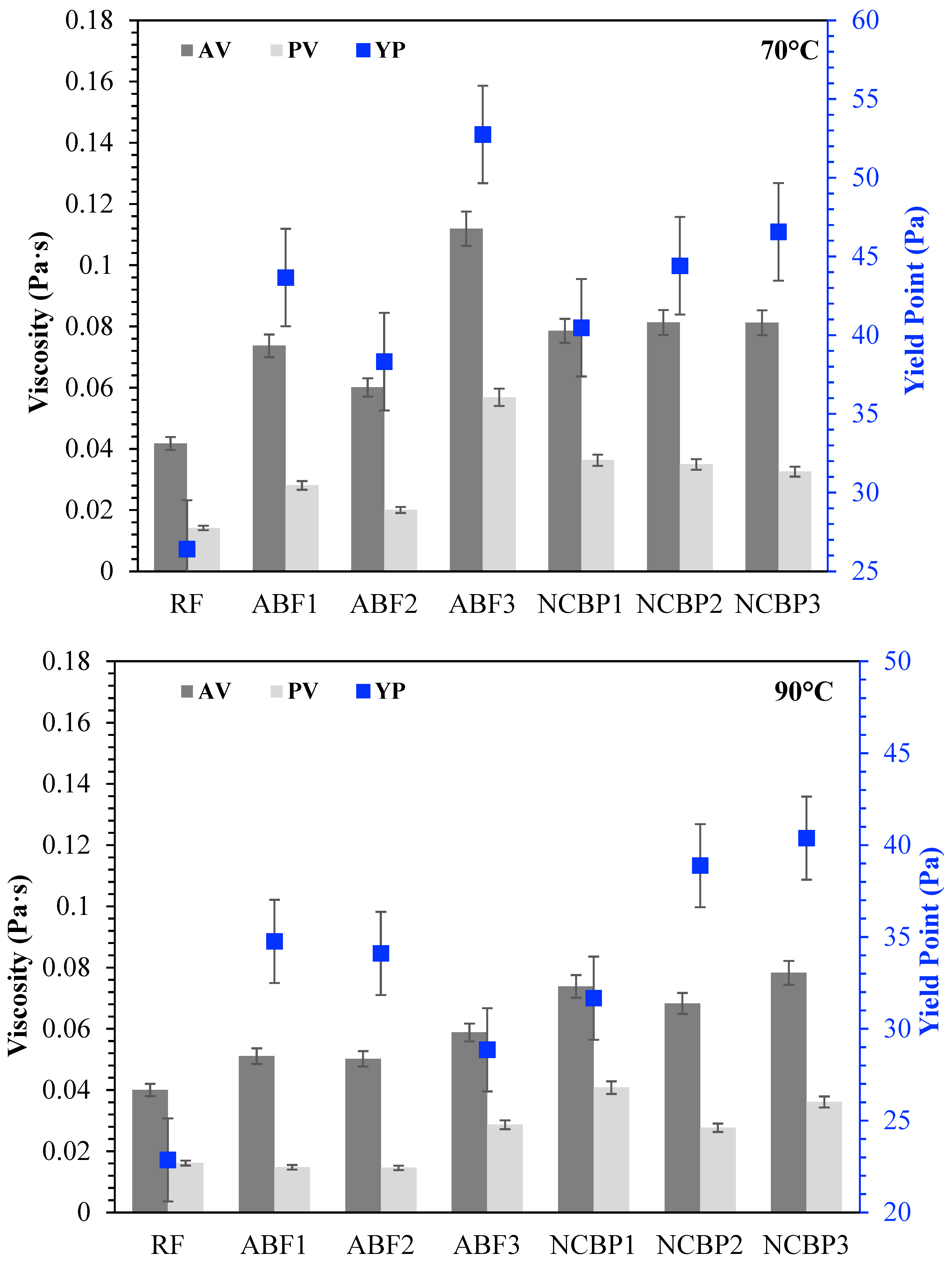
The rheological behavior of CGA drilling fluids was further investigated for comparison of apparent viscosity (AV), plastic viscosity (PV) and yield point (YP) at 25 °C, 50 °C, 70 °C and 90 °C, shown in Figure 15. At 25 °C, each fluid exhibited unique rheological characteristics. In aphron-based fluids, ABF3 showed the best AV and PV as a result of high surfactant concentration that accelerated foam density and structural complexity. ABF1 and ABF2 came next, though ABF2 had slightly lower viscosities, which was expected considering the more balanced bubble distribution and optimized stability. The fact that YP was maximized in ABF3 indicates that although the fluid was more resistant to flow, the high internal structure of the fluid may be unsuitable for thermal or long-term flow stability. When the temperature rose to 50 °C, all ABF samples showed low viscosity, characteristic of thermal softening. ABF3 shows, however, to have the highest AV and PV consistent with its better structural resistance. YP values declined marginally in both ABF1 and ABF2; however, the YP of ABF3 was relatively high, suggesting that its internal structure remained relatively intact. The difference between ABF3 and the ABFs was small at 70 °C. AV and PV values for the ABF1 and ABF2 decreased with greater severity and ABF3 maintained moderate rheological integrity but started to decrease in YP. Hence, ABF3’s viscosity is relatively more sensitive to heat although at an early stage it was relatively strong. All ABFs exhibited large decreases in AV, PV, and YP, particularly ABF1 and ABF2, at 90 °C. While ABF3 maintained comparatively larger viscosity than ABF1 and ABF2, its initial performance advantage decreased greatly, indicating that high initial viscosity did not mean superior performance at higher temperatures. In comparison, NCBP2 and NCBP3 exhibited excellent thermal stability, with higher overall AV and YP values across the tested temperatures, including 90 °C. This indicates that adding nanostructured corn biopolymer at moderate-to-high concentrations supports aphron structure retention, improves gel strength, and reduces the thermal thinning observed in surfactant-only systems.
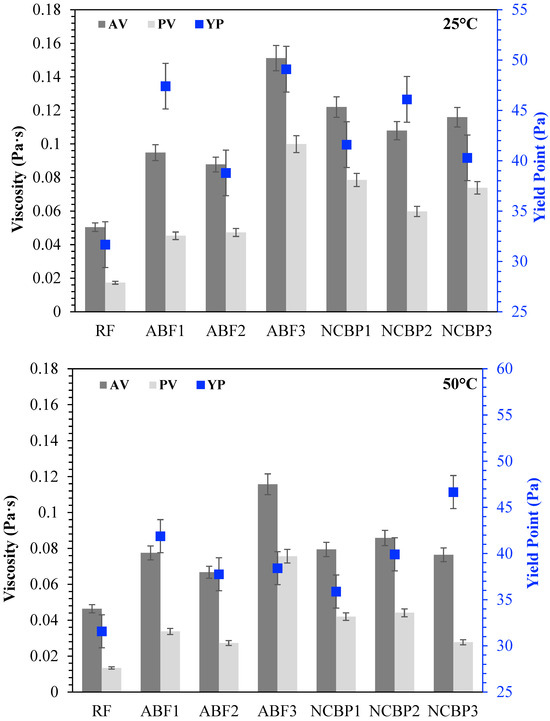
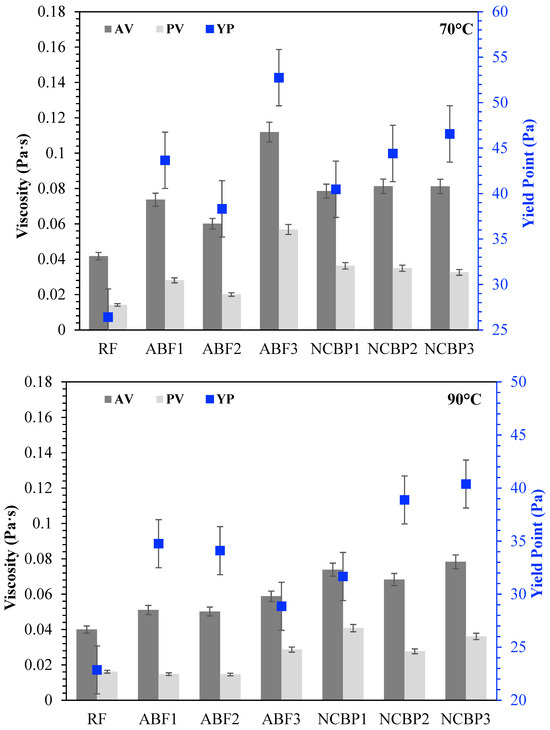
Figure 15.
Apparent viscosity (AV), plastic viscosity (PV), and yield point (YP) of all drilling fluid formulations at 25 °C, 50 °C, 70 °C, and 90 °C.
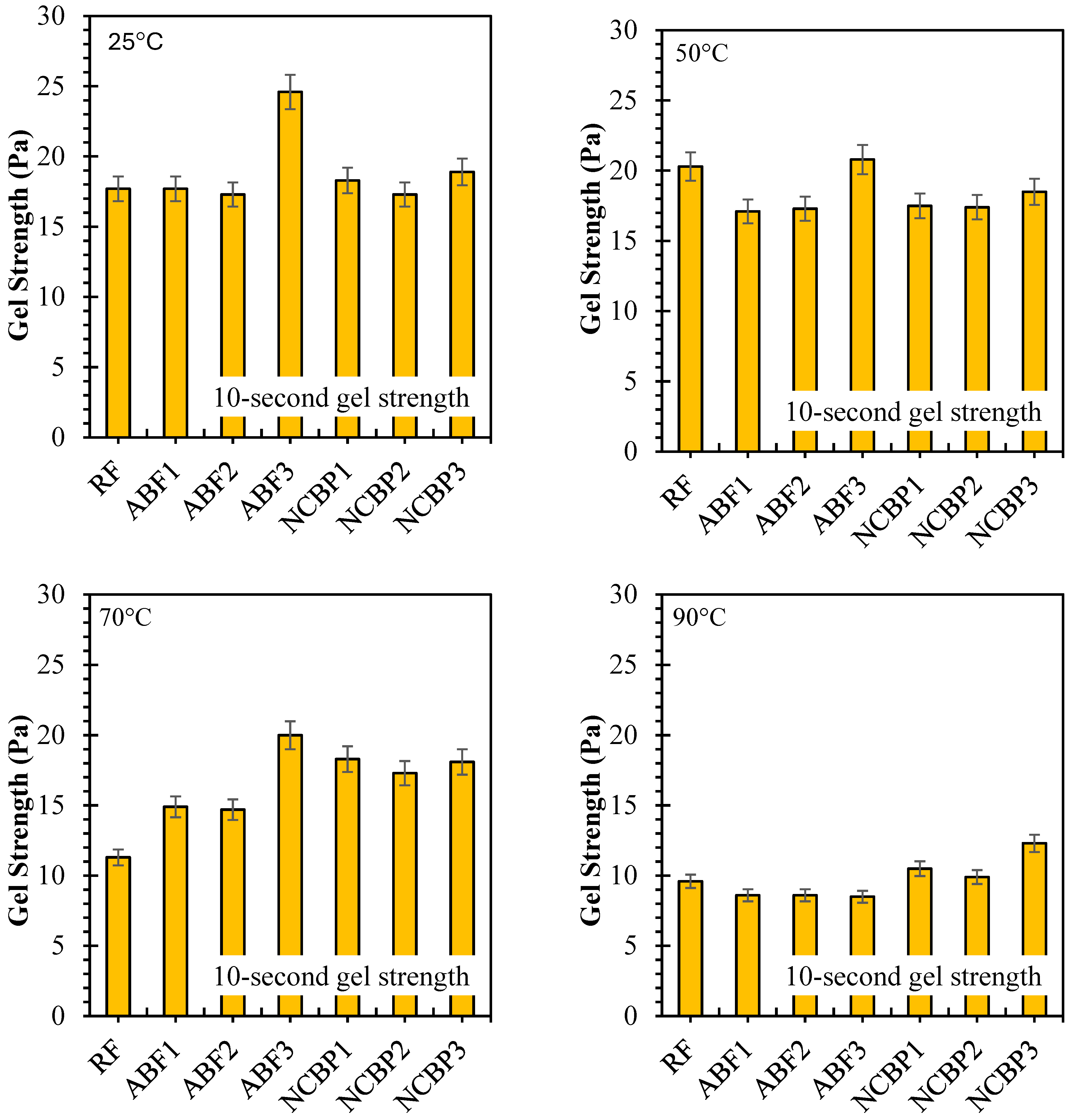
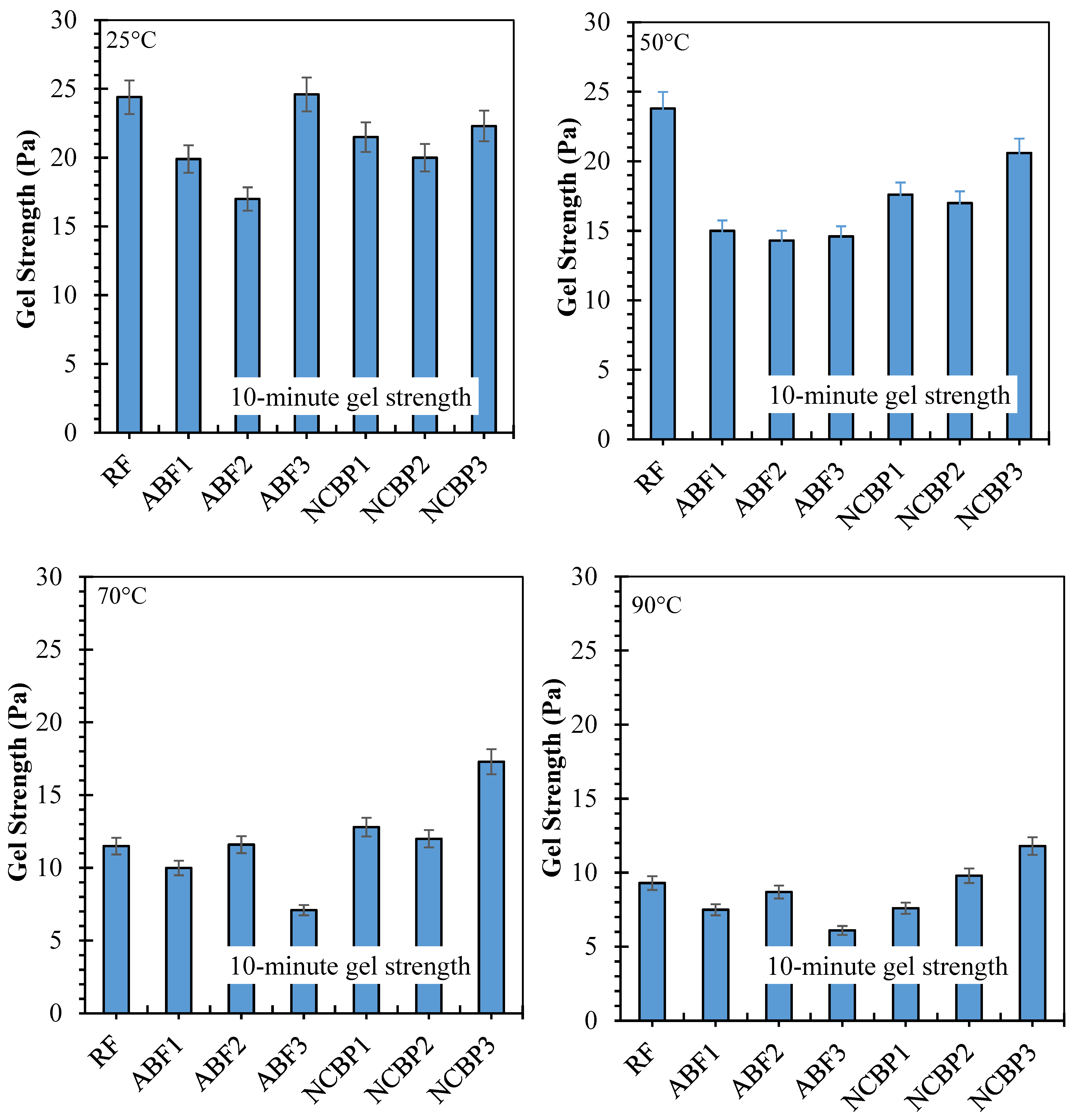
10 s and 10 min gel strengths have been reported for seven formulations from 25 °C to 90 °C, respectively, in Figure 16 showing solids-suspension capacity under static conditions and thermal stress. Strong gels formed at 25 °C were obtained across the fluids which was observed for ABF3 (24.6 Pa at 10 s and 10 min), indicating that in high surfactant content a dense foam network was established. Next up was NCBP3 (18.9 Pa at 10 s; 22.3 Pa at 10 min), further supporting the structural contribution made by the nanostructured corn biopolymer. RF also showed high initial gels that reached 17.7 Pa and 24.4 Pa probably due to an undisturbed bentonite structure. Gel strengths decreased in all systems with increasing temperature, with the most pronounced reductions at 70 °C and 90 °C. For ABF3, values decreased to 9.6 Pa (10 s) and 7.5 Pa (10 min) at 90 °C, retaining 39.0% and 30.5% of the room temperature values. NCBP3 had the highest gels at 90 °C (12.3 Pa:10 s and 11.8 Pa:10 min) for a retention of 65.1% and 52.9%, respectively. On the other hand, NCBP1 and NCBP2 were better than the surfactant-only ABFs at high temperature. Nanobiopolymer-based fluids such as NCBP3 overall maintained a better thermal stability over both short-term and long-term gel strengths, indicating the ability of nanostructured corn biopolymer to reinforce the microstructure and maintain gels for longer than aphron-only or conventional bentonite systems at high temperatures.
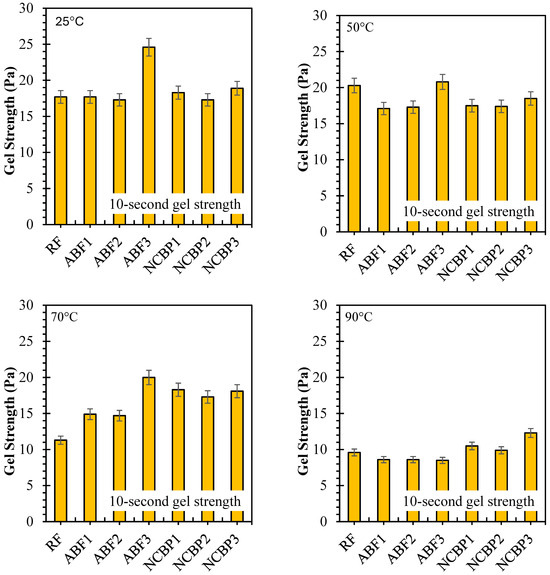
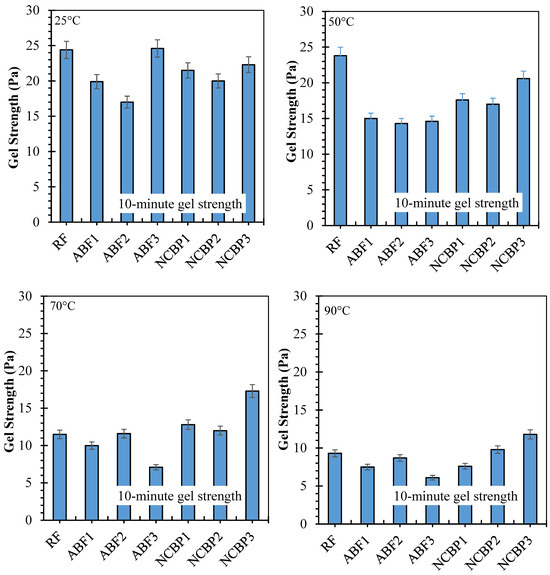
Figure 16.
10 s and 10 min gel strength values of all drilling fluid formulations measured at 25 °C, 50 °C, 70 °C, and 90 °C.
3.4. Filtration Performance and Fluid Loss Control
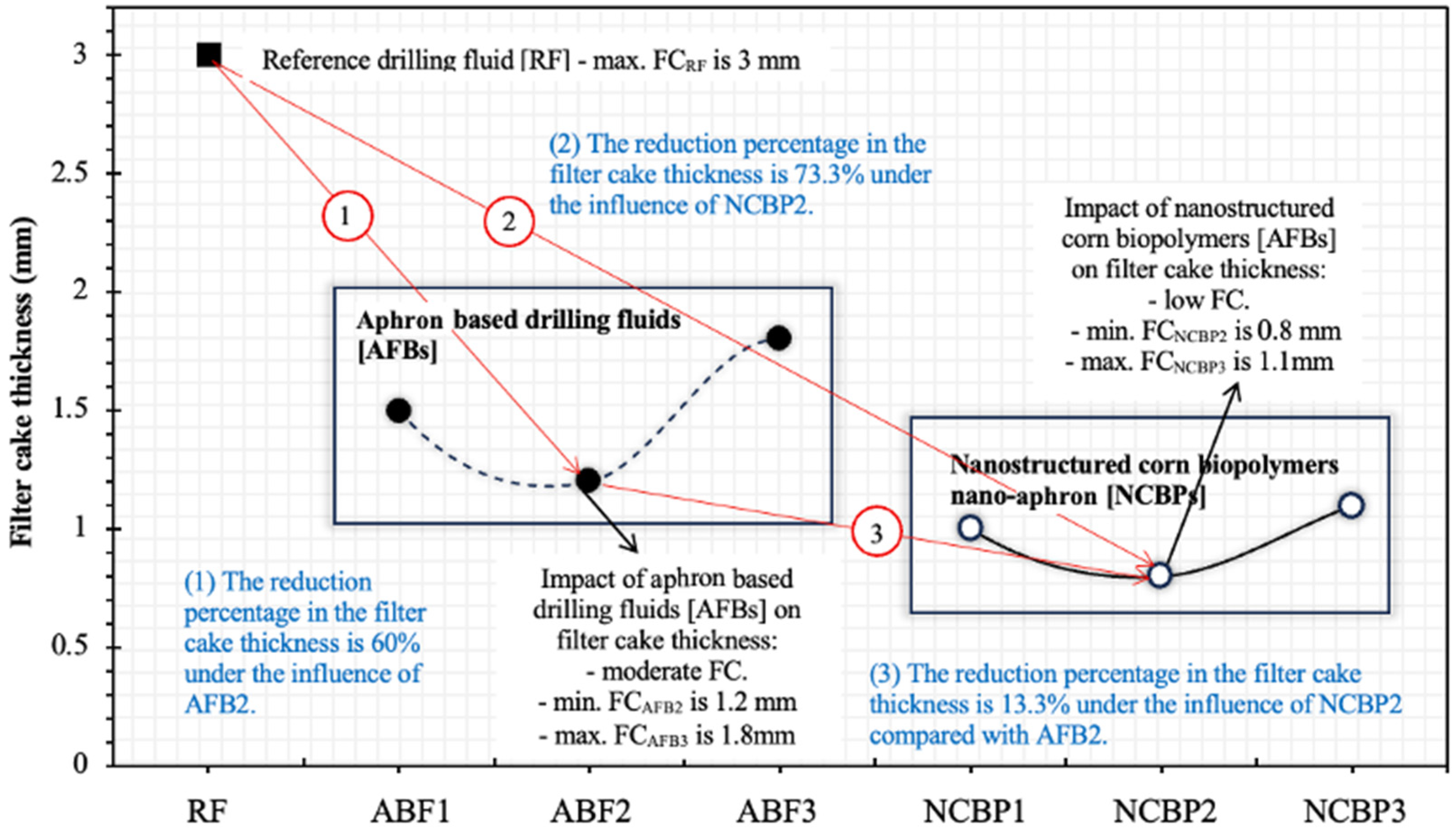
Figure 17 illustrates the variation in filter cake thickness for the reference fluid (RF), aphron-based drilling fluids (ABFs) and nanostructured corn biopolymer-enhanced aphron fluids (NCBPs). The results clearly demonstrate the progressive improvement in filtration control as the formulation evolves from a conventional bentonite-based system to aphron-based fluids and finally to nano-reinforced aphron systems. The reference fluid (RF) produced the thickest filter cake of 3.0 mm, reflecting the limited sealing efficiency of conventional water-based drilling fluids in the absence of deformable or nanoscale plugging agents. Such thick and relatively rigid cakes are typical of bentonite-dominated systems and are undesirable due to increased differential sticking risk and formation damage.
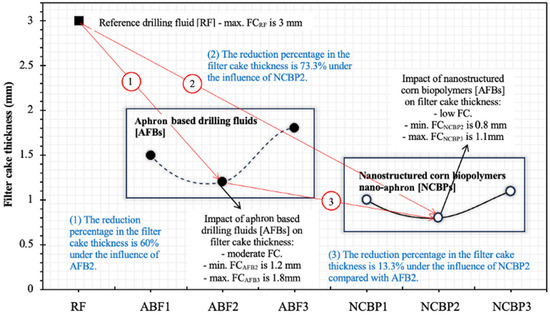
Figure 17.
Measured thicknesses of the obtained filter cakes for the reference drilling fluid (RF), aphron drilling fluid (ABF1, ABF2 and ABF3), and nanostructured corn biopolymer drilling fluid (NCBP1, NCBP2 and NCBP3).
The introduction of aphrons in the ABF formulations significantly reduced filter-cake thickness, confirming the effectiveness of aphron-based drilling fluids in loss control. Among the ABFs, ABF2 exhibited the best performance, forming a filter cake of approximately 1.2 mm, corresponding to a 60% reduction relative to the reference fluid (see Figure 17). This improvement is attributed to the ability of aphrons to act as soft, deformable bridging elements that rapidly accumulate at the fluid-formation interface, sealing pore throats and microfractures. However, the performance of ABFs was not monotonic with surfactant concentration. While ABF1 and ABF2 showed improved sealing, ABF3 resulted in a thicker cake (1.8 mm), indicating that excessive surfactant content can destabilize aphron size distribution, promote coalescence, and weaken the integrity of the sealing structure.
A further and more pronounced reduction in filter-cake thickness was achieved with the incorporation of nanostructured corn biopolymers (NCBPs). Compared with ABF2, the addition of nanobiopolymer led to an additional 13.3% reduction in cake thickness, with NCBP2 producing the thinnest and most compact cake (0.8 mm). Overall, this corresponds to a 73.3% reduction relative to the reference fluid, highlighting the strong synergistic effect between aphrons and nanostructured biopolymers. The superior performance of NCBP2 can be explained by a combined sealing mechanism. While aphrons provide macro- and micro-scale deformable bridging, the nanostructured corn biopolymer acts as a fine filler that occupies interstitial voids within the aphron network and between bentonite platelets. This dual-scale sealing mechanism results in a denser, less permeable filter cake with reduced thickness. In contrast, higher nanobiopolymer loading (NCBP3) led to a slight increase in cake thickness (1.1 mm), likely due to nanoparticle agglomeration and excessive viscosity, which can hinder optimal particle packing and aphron mobility. In other words, beyond the optimum (1 wt.%), excessive NCBP can promote a thicker, less favorably packed cake and reduce aphron transport to the sealing interface, which offsets the expected filtration gains.
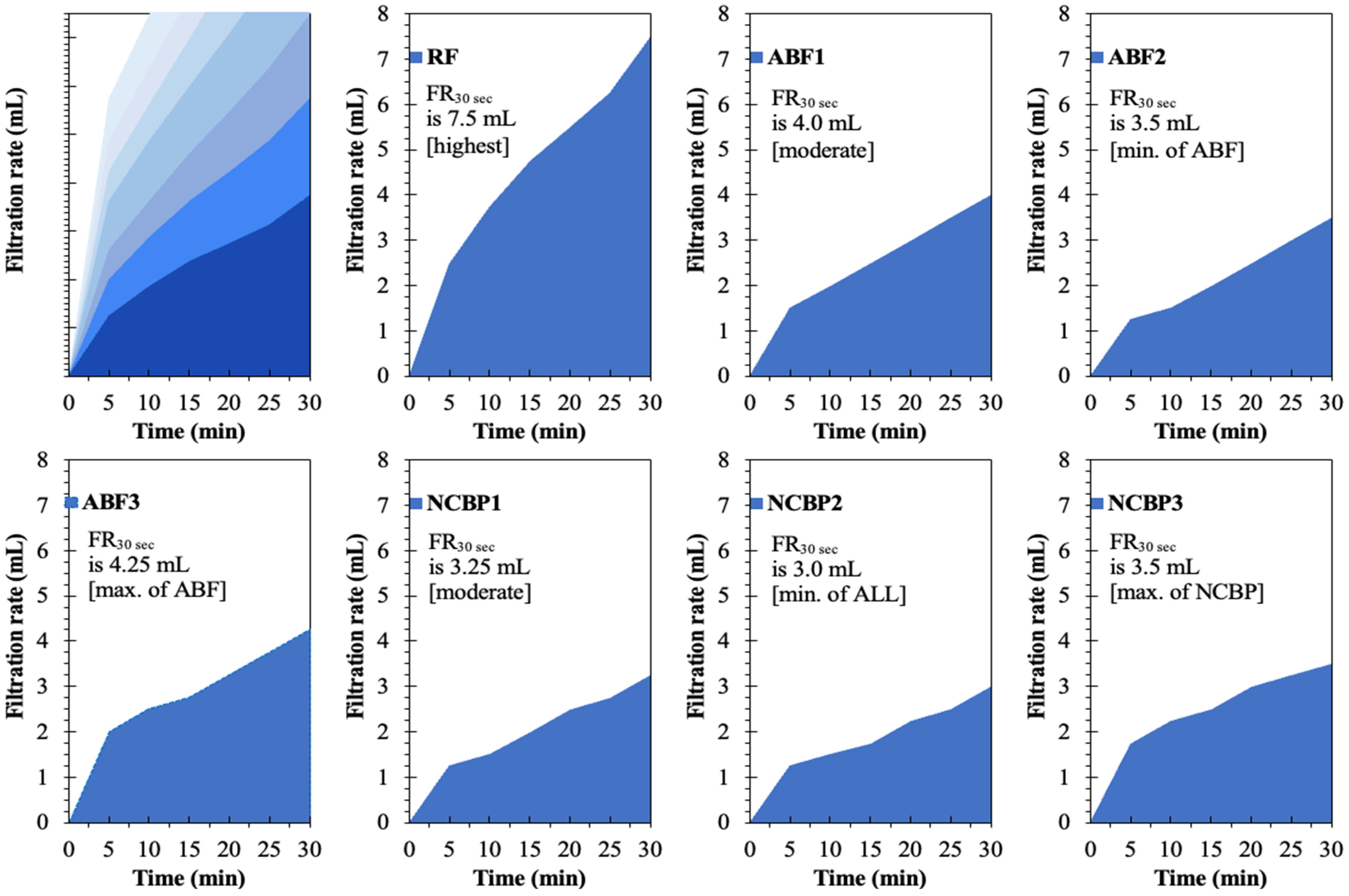
Figure 18 presents the time-dependent filtration behavior of the reference fluid (RF), aphron-based drilling fluids (ABFs), and nanostructured corn biopolymer-enhanced aphron fluids (NCBPs) under API (LTLP) conditions. The filtration curves clearly demonstrate the progressive improvement in fluid-loss control achieved through aphron generation and subsequent nanobiopolymer reinforcement. The reference fluid (RF) exhibits the highest filtration rate throughout the test, reaching a total filtrate volume of 7.5 mL after 30 min, which is the maximum among all formulations. The steep initial slope within the first few minutes indicates rapid filtrate invasion before a stable filter cake is formed. This behavior is characteristic of conventional bentonite-based fluids, where rigid particles require longer times to bridge pore throats and establish an effective seal. In contrast, the aphron-based fluids show a marked reduction in both early-time and cumulative filtration. Among the ABFs, ABF2 demonstrates the lowest filtrate volume (3.5 mL), representing the best performance within the aphron-only systems. This reduction reflects the ability of aphrons to rapidly migrate to the filtration interface and form a deformable, low-permeability barrier that limits fluid invasion from the early stages of filtration. ABF1 and ABF3 display higher filtrate volumes (4.0 and 4.25 mL, respectively), indicating that aphron performance is strongly dependent on surfactant concentration. Excessive surfactant loading in ABF3 likely promotes aphron coalescence and size heterogeneity, which weakens sealing efficiency and increases filtrate flow.
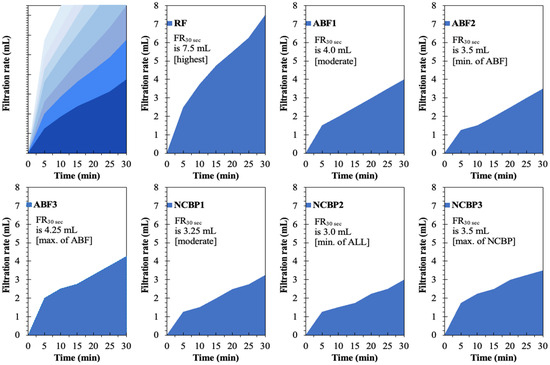
Figure 18.
Obtained filtration rates of the reference drilling fluid (RF), aphron drilling fluid (ABF1, ABF2 and ABF3), and nanostructured-corn-biopolymer drilling fluid (NCBP1, NCBP2 and NCBP3).
A further and more pronounced improvement is observed with the incorporation of nanostructured corn biopolymers. The NCBP formulations consistently exhibit lower filtration rates and flatter curves, indicating improved early time sealing and more stable filter cake development. NCBP2 achieves the lowest overall filtrate volume (3.0 mL) among all tested fluids, confirming its superior filtration control (see Figure 18). This corresponds to a reduction of approximately 60% relative to the reference fluid, and a measurable improvement compared with the best aphron-only formulation (ABF2). The enhanced performance of NCBP2 can be attributed to a synergistic sealing mechanism. While aphrons provide rapid, deformable bridging at the pore scale, the nanostructured corn biopolymer acts as a fine-scale filler that occupies micro-voids within the aphron network and between bentonite platelets. This dual-scale structure reduces filter-cake permeability, suppresses late-time filtrate flow, and results in a more stable filtration profile. In contrast, NCBP3 shows a slight increase in filtrate volume (3.5 mL), suggesting that excessive nanobiopolymer loading may lead to particle agglomeration or increased viscosity, which can hinder optimal packing and aphron transport to the filtration interface. This supports an optimum loading behavior where 1 wt.% achieves effective micro-void filling and tight cake formation without over-structuring the fluid.
Other nano-enhanced CGA systems reported in the literature often rely on interfacial particle adsorption and particle-network formation to stabilize bubbles and tighten the deposited structure, which can improve fluid-loss control but may introduce dispersion sensitivity, agglomeration risk, and higher solids loading [15,16,20,24,35,75]. In contrast, the present formulation achieves loss-control improvement primarily through a bio-based surfactant film and a bio-derived nanostructured polymer network that supports aphron stability and promotes a denser, less permeable cake. This shifts the design from inorganic-particle-dominated stabilization toward a renewable, bio-based reinforcement strategy.
In this study, filtration behavior was evaluated under standard LTLP API conditions, while temperature effects on rheology and aphron stability were assessed up to 90 °C. Although these results demonstrate clear improvements in fluid-loss control and mud-cake quality, additional verification under HTHP filtration conditions and through direct shale reactivity/inhibition tests would further strengthen the assessment of performance in harsher downhole environments and reactive shale formations. These complementary evaluations are recommended as an extension of the present work.
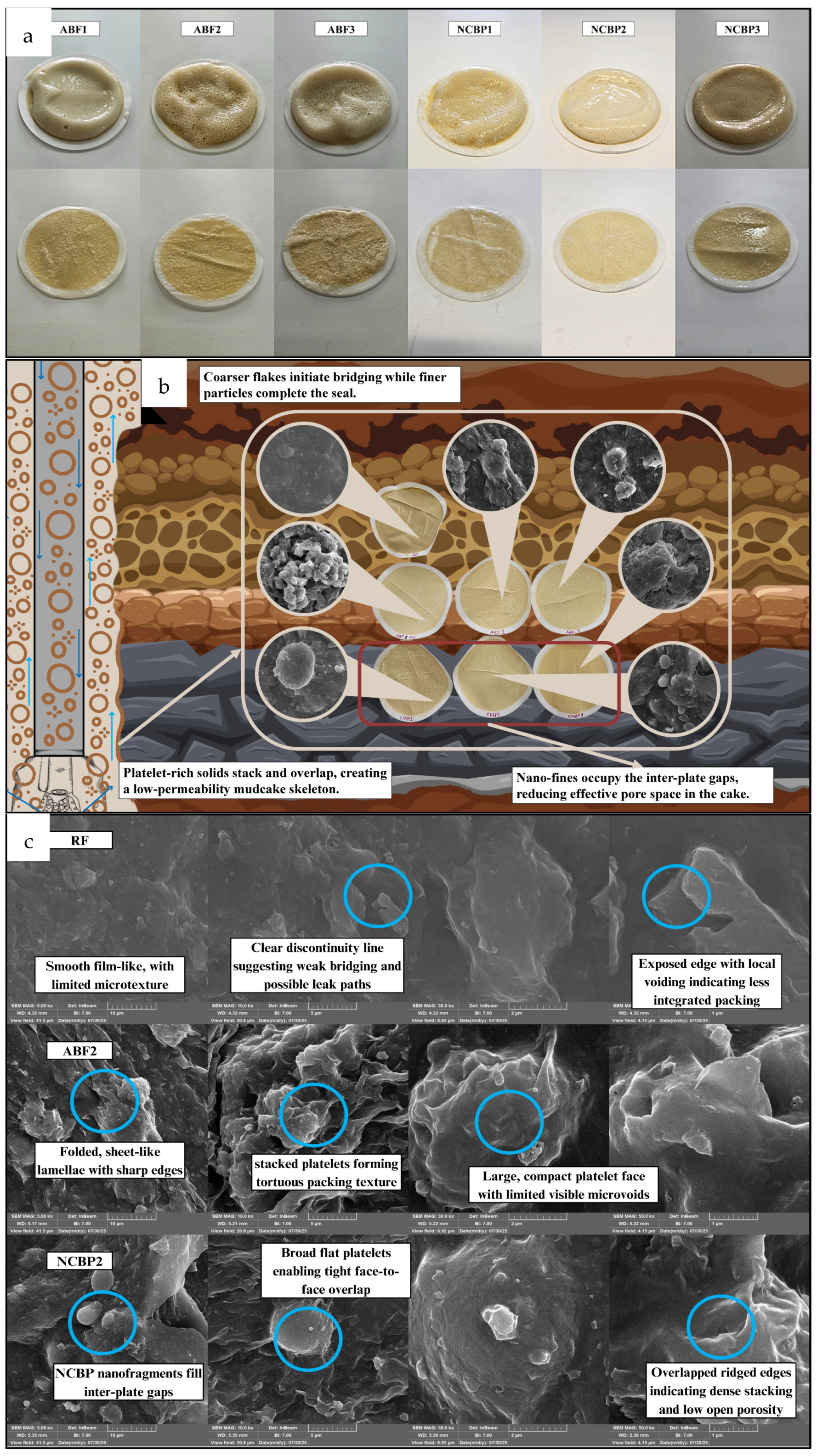
Figure 19 compares the mud-cake sealing behavior and explains why the ABF and NCBP systems reduce fluid loss compared with the reference fluid. As shown in Figure 19a, the aphron-based mud cakes photographed before and after washing reveal clear differences in mechanical integrity. RF formed a thin, brittle, uneven layer that was more prone to damage after washing, while the ABF and NCBP samples generally produced denser, more uniform mud cakes that remained intact, indicating stronger particle bonding and better sealing. The FESEM images in Figure 19b further clarify these differences in microstructure. RF shows a conventional plate-like packing typical of bentonite-based systems, but with features that can still allow connected micro-pathways when the stacking is not well locked. In the ABF systems, the surfaces appear rougher and more heterogeneous, and in ABF2 especially, spherical aphron domains are visible within the solid matrix. These deformable aphron structures can act as solid-like but highly deformable bridging elements that help plug voids and interrupt filtrate pathways, improving the barrier quality of the mud cake. The NCBP mud cakes exhibit denser, irregular flaky morphologies with larger compact sheets, and NCBP2 shows the tightest layer coverage with fewer open channels. This is consistent with multi-scale packing where nanoscale-corn-biopolymer fragments and polymer-rich domains help occupy gaps between stacked platelets, strengthening the network and reducing permeability. These trends are reinforced in the higher-magnification views in Figure 19c. RF appears more layered, with clearer discontinuities and weaker interparticle bridging. ABF2 shows aphron entrapment within the deposited structure, while NCBP2 forms a tightly interconnected flaky network with limited pore connectivity. Figure 19 shows that aphron stabilization and nanobiopolymer incorporation both restructure the mud cake from a simpler stacked-solid layer into a more effectively packed and better supported barrier. This explains the higher sealing efficiency and lower fluid loss of ABF and NCBP fluids relative to RF.
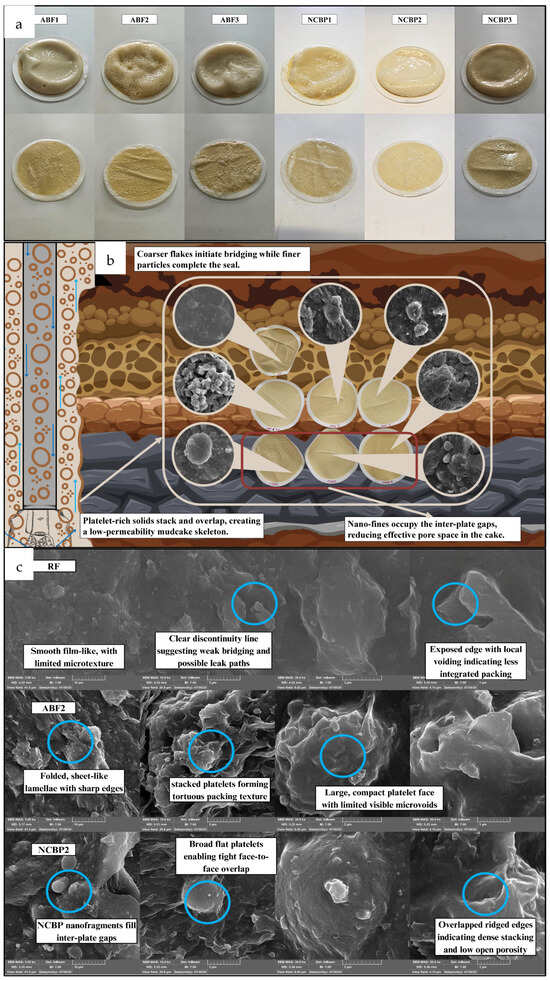
Figure 19.
(a) API aphron-based mud cakes before and after washing, showing surface sealing characteristics; (b) Visual and SEM images of all samples’ mud cakes, and (c) SEM images of RF, ABF2, and NCBP2 at different magnifications.
4. Mechanism and Environmental Impact
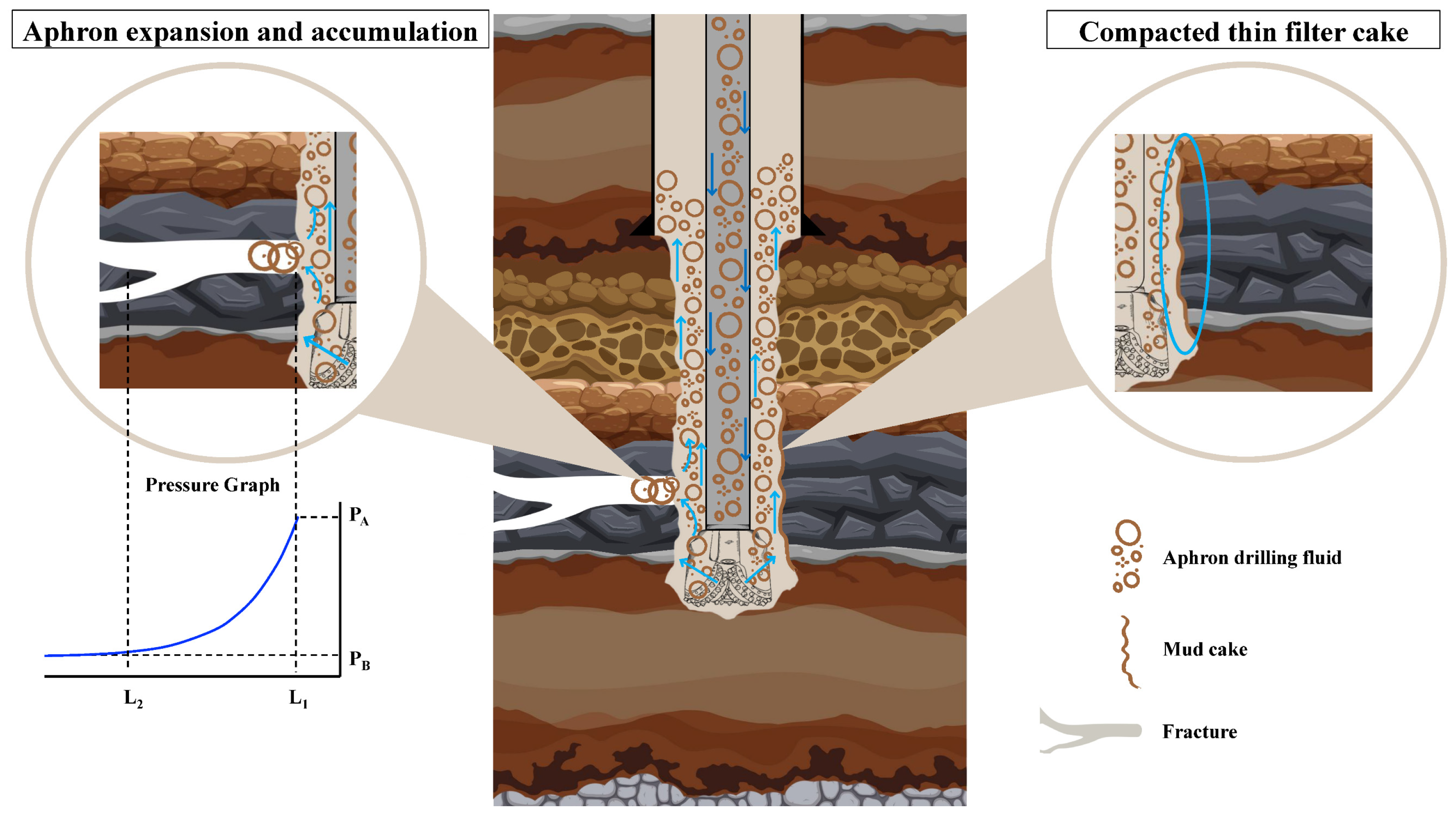
When a CGA drilling fluid reaches a fractured or highly permeable zone, the sealing process starts at the fracture mouth and builds inward. As shown in Figure 20, aphrons preferentially accumulate at narrow openings and constrictions because the local flow lines converge there and the bubbles behave as highly deformable “solid-like” [76]. Under differential pressure, they pack together, slightly expand and rearrange, and rapidly create a compressible plug that cuts the initial filtrate loss [77]. At the same time, the continuous phase carries plate-like and flaky solids toward the same restriction. Coarser flakes and platelets tend to bridge first and form a skeletal framework, then smaller fragments and nano-sized domains occupy the remaining inter-plate gaps, turning that framework into a tighter barrier with fewer connected flow channels [78]. This is where the formulation chemistry becomes critical; bentonite and xanthan gum provide a strong low-shear structure so the aphrons do not freely rise, merge, or drain, and they help the plug resist erosion once it forms [21]. The peanut-oil-derived anionic surfactant reduces interfacial tension and creates a more elastic, hydrated bubble film, while the nanostructured corn biopolymer reinforces that interfacial film and the surrounding liquid lamella, improving resistance to coalescence and drainage [24].
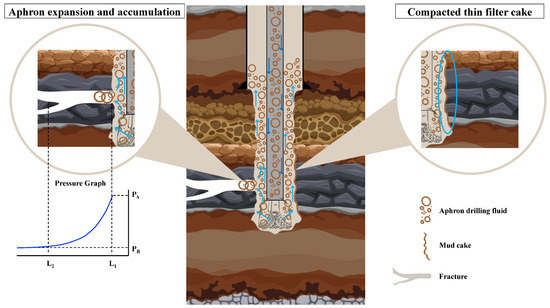
Figure 20.
Conceptual illustration of CGA fluid behavior in a fractured formation, showing aphron expansion and the formation of a thin, compact mud cake to seal fractures and minimize fluid invasion.
The practical outcome is a thin, compacted mud cake (right side of Figure 20) because the sealing is driven by tough-yet-flexible plus efficient particle packing, not by building a thick solids bed. This “thin but tight” mud-cake concept is consistent with the literature on sustainable and nano-enabled additives (cellulose nanocrystals, modified starches, and biomass-derived nanoparticles), where improved filtration control is repeatedly linked to multi-scale packing, pore-throat bridging, and gap-filling by finer fractions [78]. From an environmental perspective, the same design choices can reduce the overall footprint. Replacing part of the performance role of conventional synthetic polymers and high-dose solids with biodegradable, renewable biopolymers (corn-based) and bio-derived surfactants (peanut oil) supports lower-toxicity, water-based systems. It also aligns with the broader trend of using agricultural and biomass wastes (for example, cellulose from husks, modified starch, and plant-ash or bagasse-derived particles) as green drilling-fluid additives [79]. Mechanistically, better sealing also has a direct sustainability benefit because it can reduce lost circulation and filtrate invasion, which lowers the total fluid volume consumed and the amount of contaminated waste generated on surface, while also reducing the risk of formation damage and associated remediation [80].
5. Conclusions
A green CGA drilling fluid was thus formulated with a nanostructured corn biopolymer (NCBP) and a peanut-oil-derived anionic surfactant in this work. The resulting formulation displayed synergistic effects on stability, rheology and filtration. Key conclusions include:
- Synthesis as well as characterization of nanostructured corn biopolymer (NCBP) by AFM, which showed an average particle size 86.33 nm confirmed that this nanotechnology is suitable for the modification of drilling fluid;
- In the system a biodegradable surfactant developed by a two-step esterification and sulfonation process yielded a functional anionic agent, which stabilized CGA bubbles and dispersed them more efficiently;
- In microscopic and visual investigations, the presence of NCBP additives contributed to increasing the bubble film thickness and stability, decreasing the coalescence and improving aphron structural integrity;
- NCBP fluids showed superior shear-thinning properties and greater gelling strength from 25 °C to 90 °C. At 90 °C, NCBP3 retained 65.1% (10 s) and 52.9% (10 min) of its room-temperature gels as compared to 39.0% and 30.5% for the best surfactant-only ABF.
- NCBP2 produced the lowest filtrate (3.0 mL) and the thinnest mud cake (0.8 mm), which is −60.0% filtrate and −73.3% mud cake compared to reference fluid. Compared to the best aphron-only system (ABF2), NCBP2 further reduced filtrate by 14.3% and mud cake thickness by 33.3%;
- NCBP formulations produced more compact and uniform mud cakes, supporting wellbore stability and lowering invasion risk;
- The schematic illustration (Figure 20) visualizes how CGA fluids function in fractured formations: aphrons enlarge as they enter fractures, supported by bentonite, xanthan gum, and nanobiopolymers that collectively build a dense seal at the fracture mouth, reducing loss and invasion.
Before field deployment, further validation is recommended, including long-term stability during extended circulation and thermal exposure, and compatibility with formation fluids and salinity. Cost and scale-up feasibility, including material availability and repeatable surfactant synthesis, should also be assessed. The use of biodegradable and nanostructured additives in CGA systems represents a promising and environmentally conscious approach to drilling fluid design, especially for managing severe loss-circulation zones.
Author Contributions
Conceptualization, R.D. and J.A.; methodology, R.D.; investigation, R.D.; resources, J.A.; data curation, R.D.; writing—original draft, R.D.; writing—review and editing, J.A. and H.O.; supervision, J.A. and H.O.; project administration, H.O. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
All data supporting the findings of this study are included within the article, further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| CGA | Colloidal Gas Aphron |
| ABF | Aphron-Based Fluid |
| NCBP | Nanostructured Corn Biopolymer |
| RF | Reference Fluid |
| WBDF | Water-Based Drilling Fluid |
| LCM | Lost Circulation Material |
| API | American Petroleum Institute |
| API RP 13B-1 | API Recommended Practice for Field Testing Water-Based Drilling Fluids |
| LTLP | Low Temperature Low Pressure |
| AV | Apparent Viscosity |
| PV | Plastic Viscosity |
| YP | Yield Point |
| LSRV | Low Shear Rate Viscosity |
| CMC | Critical Micelle Concentration |
References
- Sebba, F. Microfoams—An unexploited colloid system. J. Colloid Interface Sci. 1971, 35, 643–646. [Google Scholar] [CrossRef]
- Sebba, F. Separations Using Aphrons. Sep. Purif. Methods 1985, 14, 127–148. [Google Scholar] [CrossRef]
- Brookey, T. “Micro-Bubbles”: New Aphron Drill-In Fluid Technique Reduces Formation Damage in Horizontal Wells. In Proceedings of the SPE Formation Damage Control Conference, Lafayette, LA, USA, 18–19 February 1998. [Google Scholar]
- Growcock, F.B.; Belkin, A.; Fosdick, M.; Irving, M.; O’Connor, B.; Brookey, T. Recent Advances in Aphron Drilling-Fluid Technology. SPE Drill. Complet. 2007, 22, 74–80. [Google Scholar] [CrossRef]
- Pasdar, M.; Kamari, E.; Kazemzadeh, E.; Ghazanfari, M.H.; Soleymani, M. Investigating fluid invasion control by Colloidal Gas Aphron (CGA) based fluids in micromodel systems. J. Nat. Gas Sci. Eng. 2019, 66, 1–10. [Google Scholar] [CrossRef]
- Mehrjoo, H.; Kazemzadeh, Y.; Ismail, I.; Gaganis, V. A Comprehensive Review of Colloidal gas aphrons applications in the Oil Industry. J. Pet. Explor. Prod. Technol. 2025, 15, 44. [Google Scholar] [CrossRef]
- Kinchen, D.; Peavy, M.A.; Brookey, T.; Rhodes, D. Case history: Drilling techniques used in successful redevelopment of low pressure H2S gas carbonate formation. In Proceedings of the Drilling Conference, Amsterdam, The Netherlands, 27 February–1 March 2001. [Google Scholar]
- Gaurina-Međimurec, N.; Pašić, B. Aphron-based drilling fluids: Solution for low pressure reservoirs. Rud.-Geološko-Naft. Zb. 2009, 21, 65–72. [Google Scholar]
- Montilva, J.; Ivan, C.; Friedheim, J.; Bayter, R. Aphron Drilling Fluid: Field Lessons from Successful Application in Drilling Depleted Reservoirs in Lake Maracaibo. In Proceedings of the Offshore Technology Conference, Houston, TX, 6–9 May 2002. [Google Scholar] [CrossRef]
- Molaei, A.; Waters, K.E. Aphron applications—A review of recent and current research. Adv. Colloid Interface Sci. 2015, 216, 36–54. [Google Scholar] [CrossRef]
- Rea, A.B.; Paiuk, B.P.; Miguel Climaco, J.; Vallejo, M.; Leon, E.; Inojosa, J. Application of Aphrons Technology in Drilling Depleted Mature Fields. In Proceedings of the SPE Latin American and Caribbean Petroleum Engineering Conference, Port of Spain, Trinidad and Tobago, 27–30 April 2003. [Google Scholar]
- Growcock, F.B.; Simon, G.A.; Rea, A.B.; Leonard, R.S.; Noello, E.; Castellan, R. Alternative Aphron-Based Drilling Fluid. In Proceedings of the IADC/SPE Drilling Conference, Dallas, TX, USA, 2–4 March 2004. [Google Scholar]
- Arabloo, M.; Shahri, M.P.; Zamani, M. Characterization of Colloidal Gas Aphron-Fluids Produced from a New Plant-Based Surfactant. J. Dispers. Sci. Technol. 2013, 34, 669–678. [Google Scholar] [CrossRef]
- Schaneman, B.D.; Jones, T.; Rea, A.B. Aphrons Technology—A Solution. In Proceedings of the AADE 2003 National Technology Conference: Practical Solutions for Drilling Challenges, Houston, TX, USA, 1 April 2003; Available online: https://www.aade.org/application/files/4915/7304/4596/AADE-03-NTCE-41-Schaneman.pdf (accessed on 10 October 2025).
- Tabzar, A.; Ziaee, H.; Arabloo, M.; Ghazanfari, M.H. Physicochemical properties of nano-enhanced colloidal gas aphron (NCGA)-based fluids. Eur. Phys. J. Plus 2020, 135, 312. [Google Scholar] [CrossRef]
- Tabzar, A.; Ghazanfari, M.H. Pore-scale analysis of filtration loss control by Colloidal Gas Aphron Nano-Fluids (CGANF) in heterogeneous porous media. Exp. Therm. Fluid Sci. 2016, 77, 327–336. [Google Scholar] [CrossRef]
- Ahmadi, M.A.; Galedarzadeh, M.; Shadizadeh, S.R. Colloidal gas aphron drilling fluid properties generated by natural surfactants: Experimental investigation. J. Nat. Gas Sci. Eng. 2015, 27, 1109–1117. [Google Scholar] [CrossRef]
- Zhu, W.; Zheng, X. High Temperature Sealing Performance of Novel Biodegradable Colloidal Gas Aphron (CGA) Drilling Fluid System. J. Jpn. Pet. Inst. 2021, 64, 331–339. [Google Scholar] [CrossRef]
- Zhu, W.; Zheng, X. Study on the Rheological Properties of Ultra-High Temperature CGA Drilling Fluids. In Proceedings of the 2nd International Conference on Green Energy, Environment and Sustainable Development (GEESD2021), Online, China, 26 June 2021. [Google Scholar]
- Hossein Hassani, A.; Hossein Ghazanfari, M. Impact of Hydrophobicity of SiO2 Nanoparticles on Enhancing Properties of Colloidal Gas Aphron Fluids: An Experimental Study. J. Energy Resour. Technol. 2017, 140, 012901. [Google Scholar] [CrossRef]
- Zhu, W.; Zheng, X. High temperature performance of bentonite-based colloidal gas aphron (CGA) drilling fluid: Stability, rheology and filtration. In Proceedings of the 8th Academic Conference of Geology Resource Management and Sustainable Development, Beijing, China, 19 December 2020; pp. 329–337. [Google Scholar]
- Zhu, W.; Zheng, X. Effective Modified Xanthan Gum Fluid Loss Agent for High-Temperature Water-Based Drilling Fluid and the Filtration Control Mechanism. ACS Omega 2021, 6, 23788–23801. [Google Scholar] [CrossRef]
- Zhu, W.; Zheng, X.; Li, G. Micro-bubbles size, rheological and filtration characteristics of Colloidal Gas Aphron (CGA) drilling fluids for high temperature well: Role of attapulgite. J. Pet. Sci. Eng. 2020, 186, 106683. [Google Scholar] [CrossRef]
- Zhu, W.; Wang, B.; Zheng, X. Preparation and Foam Stabilization Mechanism of an Ultrahigh-Temperature Colloidal Gas Aphron (CGA) System Based on Nano-SiO2. ACS Omega 2023, 8, 46091–46100. [Google Scholar] [CrossRef]
- Arabloo, M.; Pordel Shahri, M. Experimental studies on stability and viscoplastic modeling of colloidal gas aphron (CGA) based drilling fluids. J. Pet. Sci. Eng. 2014, 113, 8–22. [Google Scholar] [CrossRef]
- Bjorndalen, N.; Kuru, E. Physico-Chemical Characterization of Aphron-Based Drilling Fluids. J. Can. Pet. Technol. 2008, 47, PETSOC-08-11-15-CS. [Google Scholar] [CrossRef]
- Arabloo, M.; Pordel Shahri, M. Effect of surfactant and polymer on the characteristics of aphron-containing fluids. Can. J. Chem. Eng. 2016, 94, 1197–1201. [Google Scholar] [CrossRef]
- Chen, C.; Tong, F.; Sun, R.; Yang, J.; Pang, Z.; Liu, X. Plasma Effects on Properties and Structure of Corn Starch: Characterization and Analysis. Foods 2023, 12, 4042. [Google Scholar] [CrossRef]
- Hosseini, S.A.; Khamehchi, E.; Dabir, B.; Alizadeh, A.; Mansoori, Z. Experimental Investigation of Water Based Colloidal Gas Aphron Fluid Stability. Colloids Interfaces 2019, 3, 31. [Google Scholar] [CrossRef]
- Ramirez, F.; Greaves, R.; Montilva, J. Experience Using Microbubbles-Aphron Drilling Fluid in Mature Reservoirs of Lake Maracaibo. In Proceedings of the International Symposium and Exhibition on Formation Damage Control, Lafayette, LA, USA, 20–21 February 2002. [Google Scholar]
- White, C.C.; Chesters, A.P.; Ivan, C.D.; Maikranz, S.; Nouris, R. Aphron-based Drilling Fluid: Novel Technology for Drilling Depleted Formations in the North Sea. In Proceedings of the SPE/IADC Drilling Conference, Amsterdam, The Netherlands, 19–21 February 2003. [Google Scholar]
- Ziaee, H.; Arabloo, M.; Ghazanfari, M.H.; Rashtchian, D. Herschel–Bulkley rheological parameters of lightweight colloidal gas aphron (CGA) based fluids. Chem. Eng. Res. Des. 2015, 93, 21–29. [Google Scholar] [CrossRef]
- Zhu, W.; Zheng, X.; Li, G.; Liu, K. Impact of foaming agent on the performance of colloidal gas aphron drilling fluid for geothermal drilling. GRC Trans. 2019, 43, 1–10. [Google Scholar]
- Tabzar, A.; Arabloo, M.; Ghazanfari, M.H. Rheology, stability and filtration characteristics of Colloidal Gas Aphron fluids: Role of surfactant and polymer type. J. Nat. Gas Sci. Eng. 2015, 26, 895–906. [Google Scholar] [CrossRef]
- Tabzar, A.; Wood, D.A. Chapter 11—Gas-aphron drilling fluids enhanced with silica nanoparticles and biosurfactant. In Sustainable Natural Gas Drilling; Wood, D.A., Cai, J., Eds.; Elsevier: Amsterdam, The Netherlands, 2024; Volume 4, pp. 325–354. [Google Scholar]
- Zhu, W.; Zheng, X. Alkyl Glycine Surfactant: An Efficient High-Temperature Resistant and Biodegradable Foaming Agent for Colloidal Gas Aphron (CGA) Drilling Fluid. Pet. Chem. 2021, 61, 1305–1318. [Google Scholar] [CrossRef]
- Hama, S.M.; Manshad, A.K.; Ali, J.A. Experimental investigation of new derived anionic natural surfactant from peanut oil: Application for enhanced oil recovery. J. Mol. Liq. 2024, 395, 123876. [Google Scholar] [CrossRef]
- Riazi, M.; Hemmati-Sarapardeh, A.; Schaffie, M.; Zabihi, R.; Riazi, M. A comparative analysis of alpha olefin sulfonate and dodecyl sulfate in aphronic fluid containing xanthan gum in a wide range of temperatures. Sci. Rep. 2025, 15, 2507. [Google Scholar] [CrossRef]
- API. Drilling Fluids Materials. In API Specification 13A, 9th ed.; American Petroleum Institute: Washington, DC, USA, 2019. [Google Scholar]
- API RP 13B-1; Field Testing Water-Based Drilling Fluids. American Petroleum Institute: Washington, DC, USA, 2019.
- Lu, K.; He, X.; Burhani, D.; Hu, J.; Rudolf, P.; Maniar, D.; Folkersma, R.; Voet, V.S.D.; Loos, K. Self-Healing, Remoldable, and Conductive Starch-Based Dual Reversible Cross-Linking Hydrogels for Strain Sensors. ACS Appl. Mater. Interfaces 2025, 17, 38438–38450. [Google Scholar] [CrossRef]
- Hasan, M.; Khaldun, I.; Ulva, M.; Nisa, K.; Gani, A.; Habibati, H. Bio-nanocomposite films from sugar palm starch/chitosan nanoparticle enriched with butterfly pea anthocyanins. Case Stud. Chem. Environ. Eng. 2025, 11, 101174. [Google Scholar] [CrossRef]
- Huang, Z.-Q.; Xie, X.-l.; Chen, Y.; Lu, J.-p.; Tong, Z.-F. Ball-milling treatment effect on physicochemical properties and features for cassava and maize starches. Comptes Rendus Chim. 2008, 11, 73–79. [Google Scholar] [CrossRef]
- Al-Ziyadi, H.; Verma, A. Smart additives for fluid loss control in water-based drilling fluid: A review of recent developments. Pet. Res. 2025, 10, 882–893. [Google Scholar] [CrossRef]
- Masłowski, M.; Aleksieiev, A.; Miedzianowska, J.; Strzelec, K. Potential Application of Peppermint (Mentha piperita L.), German Chamomile (Matricaria chamomilla L.) and Yarrow (Achillea millefolium L.) as Active Fillers in Natural Rubber Biocomposites. Int. J. Mol. Sci. 2021, 22, 7530. [Google Scholar] [CrossRef] [PubMed]
- Belardi, I.; Marrocchi, A.; Alfeo, V.; Sileoni, V.; De Francesco, G.; Paolantoni, M.; Marconi, O. Sequential Extraction and Attenuated Total Reflection–Fourier Transform Infrared Spectroscopy Monitoring in the Biorefining of Brewer’s Spent Grain. Molecules 2023, 28, 7992. [Google Scholar] [CrossRef] [PubMed]
- Pasieczna-Patkowska, S.; Cichy, M.; Flieger, J. Application of Fourier Transform Infrared (FTIR) Spectroscopy in Characterization of Green Synthesized Nanoparticles. Molecules 2025, 30, 684. [Google Scholar] [CrossRef]
- Aussanasuwannakul, A.; Singkammo, S. Multiscale Characterization of Rice Starch Gelation and Retrogradation Modified by Soybean Residue (Okara) and Extracted Dietary Fiber Using Rheology, Synchrotron Wide-Angle X-Ray Scattering (WAXS), and Fourier Transform Infrared (FTIR) Spectroscopy. Foods 2025, 14, 1862. [Google Scholar] [CrossRef]
- Mao, Y.; Gong, J.; Zhu, M.; Ito, H. Crystal Transition Behavior and Thermal Properties of Thermal-Energy-Storage Copolymer Materials with an n-Behenyl Side-Chain. Polymers 2019, 11, 1512. [Google Scholar] [CrossRef]
- Gao, W.; Liu, K.; Su, Y.; Sheng, L.; Cao, C.; Dou, X.; Zhang, L. Active Cooling Method for Downhole Systems in High Temperature Environment. In Proceedings of the SPE Western Regional Meeting, San Jose, CA, USA, 23–26 April 2019. [Google Scholar]
- Ali, J.A.; Gailani, R.; Abdullah, A.D.; Jaf, P.T.; Simo, S.M.; Abdalqadir, M.; Faris, V.M. Performance evaluation of the nano-biodegradable drilling fluid using the greenly synthesized zinc nanorods and gundelia seed waste. Environ. Sci. Pollut. Res. 2024, 31, 51381–51400. [Google Scholar] [CrossRef]
- Gokapai, V.; Pothana, P.; Ling, K. Nanoparticles in Drilling Fluids: A Review of Types, Mechanisms, Applications, and Future Prospects. Eng 2024, 5, 2462–2495. [Google Scholar] [CrossRef]
- Ren, H.; Zhang, Z.; Li, Y.; Liu, S.; Zhang, X. A New Method for Preparing Cross-Sections of Polymer Composite Membranes for TEM Characterization by Substrate Stripping and Double-Orientation Embedding. Membranes 2025, 15, 288. [Google Scholar] [CrossRef]
- Zarski, A.; Kapusniak, K.; Ptak, S.; Rudlicka, M.; Coseri, S.; Kapusniak, J. Functionalization Methods of Starch and Its Derivatives: From Old Limitations to New Possibilities. Polymers 2024, 16, 597. [Google Scholar] [CrossRef]
- Stern, T. Transcrystalline Mechanism of Banded Spherulites Development in Melt-Crystallized Semicrystalline Polymers. Polymers 2024, 16, 2411. [Google Scholar] [CrossRef] [PubMed]
- Joudeh, N.; Linke, D. Nanoparticle classification, physicochemical properties, characterization, and applications: A comprehensive review for biologists. J. Nanobiotechnol. 2022, 20, 262. [Google Scholar] [CrossRef] [PubMed]
- Murgueitio-Herrera, E.; Carpio, P.; Bungacho, P.; Tapia, L.T.; Camacho, C.; Debut, A. Removal of Cadmium and Lead from Tires Discarded in the Open Sea with Multicomponent Nanoparticles from Sugarcane Bagasse. Nanomaterials 2025, 15, 1700. [Google Scholar] [CrossRef] [PubMed]
- Błaż, S.; Zima, G.; Jasiński, B.; Kremieniewski, M. Invert Drilling Fluids with High Internal Phase Content. Energies 2021, 14, 4532. [Google Scholar] [CrossRef]
- Huang, S.; Dong, T.; Jiang, G.; Yang, J.; Yang, X.; Wang, Q. Synthesis and Performance Evaluation of High-Temperature-Resistant Extreme-Pressure Lubricants for a Water-Based Drilling Fluid Gel System. Gels 2024, 10, 505. [Google Scholar] [CrossRef]
- Błaż, S.; Zima, G.; Jasiński, B.; Kremieniewski, M. Increasing the Thermal Resistance of Water-Based Mud for Drilling Geothermal Wells. Energies 2024, 17, 4537. [Google Scholar] [CrossRef]
- Wu, P.-C.; Lin, S.-Y.; Tsay, R.-Y. Phase transition phenomena of the adsorbed 1-dodecanol monolayer at the air–water interface. J. Chin. Inst. Chem. Eng. 2007, 38, 443–450. [Google Scholar] [CrossRef]
- Myrick, S.H.; Franses, E.I. Effect of Dispersed Tetradecanol Particles or Droplets on the Dynamic Surface Tension of Aqueous Tetradecanol Systems. Langmuir 1999, 15, 1556–1561. [Google Scholar] [CrossRef]
- Liu, Z. (Ed.) Introduction. In Green Catalytic Hydrogenation of Phthalate Plasticizers; Springer Nature: Singapore, 2022; pp. 1–31. [Google Scholar]
- Zamani, M.; Fasihi, M.; Rasouli, S. Enhancing the flexibility and thermal properties of polylactic acid/thermoplastic starch blends through selective plasticization. Carbohydr. Polym. Technol. Appl. 2025, 10, 100780. [Google Scholar] [CrossRef]
- Zhu, W.; Zheng, X.; Shi, J.; Wang, Y. A high-temperature resistant colloid gas aphron drilling fluid system prepared by using a novel graft copolymer xanthan gum-AA/AM/AMPS. J. Pet. Sci. Eng. 2021, 205, 108821. [Google Scholar] [CrossRef]
- Jena, U.; McCurdy, A.T.; Warren, A.; Summers, H.; Ledbetter, R.N.; Hoekman, S.K.; Seefeldt, L.C.; Quinn, J.C. Oleaginous yeast platform for producing biofuels via co-solvent hydrothermal liquefaction. Biotechnol. Biofuels 2015, 8, 167. [Google Scholar] [CrossRef]
- Zia, N.; Iqbal, Z.; Raza, A.; Zia, A.; Shafique, R.; Andleeb, S.; Walker, G.C. Glycol-Chitosan-Based Technetium-99m-Loaded Multifunctional Nanomicelles: Synthesis, Evaluation, and In Vivo Biodistribution. Nanomaterials 2022, 12, 2198. [Google Scholar] [CrossRef]
- Awogbemi, O.; Kallon, D.V.V.; Aigbodion, V.S.; Mzozoyana, V. Property Determination, FA Composition and NMR Characterization of Palm Oil, Used Palm Oil and Their Methyl Esters. Processes 2022, 10, 11. [Google Scholar] [CrossRef]
- Braga, E.; Damasceno, L.; Barros de Sousa Silva, C.; Silva, L.; Cavalcante, M.; Barreto, C.; Silva, S.; Murilo Tavares de Luna, F.; Bertini, L.; Nascimento, T.; et al. 1H NMR and UV-Vis as Analytical Techniques to Evaluate Biodiesel Conversion and Oxidative Stability. Fuels 2024, 5, 107–122. [Google Scholar] [CrossRef]
- Richard, A.M.; Tao, D.; LeClair, C.A.; Leister, W.; Tretyakov, K.V.; White, E.V.; Lewis, K.C.; Sefler, A.; Shinn, P.; Collins, B.J.; et al. Analytical Quality Evaluation of the Tox21 Compound Library. Chem. Res. Toxicol. 2025, 38, 15–41. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Huang, J.; Xiao, Q.; Hu, S.; Li, S.; Zheng, J.; Du, Z.; Peng, J.; Chen, H. Glu-Urea-Lys Scaffold Functionalized Superparamagnetic Iron Oxide Nanoparticles Targeting PSMA for In Vivo Molecular MRI of Prostate Cancer. Pharmaceutics 2022, 14, 2051. [Google Scholar] [CrossRef]
- Bello, A.; Ivanova, A.; Rodionov, A.; Aminev, T.; Mishin, A.; Bakulin, D.; Grishin, P.; Belovus, P.; Penigin, A.; Kyzyma, K.; et al. An Experimental Study of High-Pressure Microscopy and Enhanced Oil Recovery with Nanoparticle-Stabilised Foams in Carbonate Oil Reservoir. Energies 2023, 16, 5120. [Google Scholar] [CrossRef]
- Sakhaei, Z.; Ghorbani-Saadatabadi, N.; Escrochi, M.; Riazi, M. Mechanistic insight into the colloidal gas aphrons stability in the presence of petroleum hydrocarbons. Fuel 2024, 368, 131576. [Google Scholar] [CrossRef]
- Hadian Nasr, N.; Mahmood, S.M.; Akbari, S.; Hematpur, H. A comparison of foam stability at varying salinities and surfactant concentrations using bulk foam tests and sandpack flooding. J. Pet. Explor. Prod. Technol. 2020, 10, 271–282. [Google Scholar] [CrossRef]
- Hassani, A.H.; Ghazanfari, M.H. Improvement of non-aqueous colloidal gas aphron-based drilling fluids properties: Role of hydrophobic nanoparticles. J. Nat. Gas Sci. Eng. 2017, 42, 1–12. [Google Scholar] [CrossRef]
- Bjorndalen, N.; Alvarez, J.M.M.; Jossy, W.E.E.; Kuru, E. A Study of the Effects of Colloidal Gas Aphron Composition on Pore Blocking. SPE Drill. Complet. 2010, 26, 139–150. [Google Scholar] [CrossRef]
- Belkin, A.; Irving, M.; O’Connor, R.; Fosdick, M.; Hoff, T.; Growcock, F.B. How Aphron Drilling Fluids Work. In Proceedings of the SPE Annual Technical Conference and Exhibition, Dallas, TX, USA, 9–12 October 2005. [Google Scholar]
- Cheraghian, G. Nanoparticles in drilling fluid: A review of the state-of-the-art. J. Mater. Res. Technol. 2021, 13, 737–753. [Google Scholar] [CrossRef]
- Wang, X.; Lei, Q.; Luo, J.; Wang, P.; Xiao, P.; Ye, Y.; Wu, X.; Liu, Y.; Zhang, G. Application of Nanocellulose in Oilfield Chemistry. ACS Omega 2021, 6, 20833–20845. [Google Scholar] [CrossRef]
- Shivhare, S.; Kuru, E. A study of the pore-blocking ability and formation damage characteristics of oil-based colloidal gas aphron drilling fluids. J. Pet. Sci. Eng. 2014, 122, 257–265. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.