Rheological, Thermal and Mechanical Properties of Blown Film Based on Starch and Clay Nanocomposites
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Particle Size Analysis by DLS
2.3. Determination of the Rheometric Conditions of the Mixture
2.4. Production of Thermoplastic Starch (TPS) with Nanoclays and Pelletization
2.5. Production of Blown Plastic Films
2.6. Characterization
2.6.1. FTIR Analysis
2.6.2. Rheological Analysis
2.6.3. Thermogravimetric Analysis (TGA)
2.6.4. Differential Scanning Calorimetry (DSC) Analysis
2.6.5. Determination of Tensile Strength and Strain
2.6.6. Determination of Tear Strength
2.6.7. SEM
2.6.8. Contact Angle
2.6.9. Determination of Water Vapor Permeability
2.6.10. Statistical Analysis
3. Results and Discussion
3.1. Dynamic Light Scattering (DLS) Analysis
3.2. Torque Rheometry
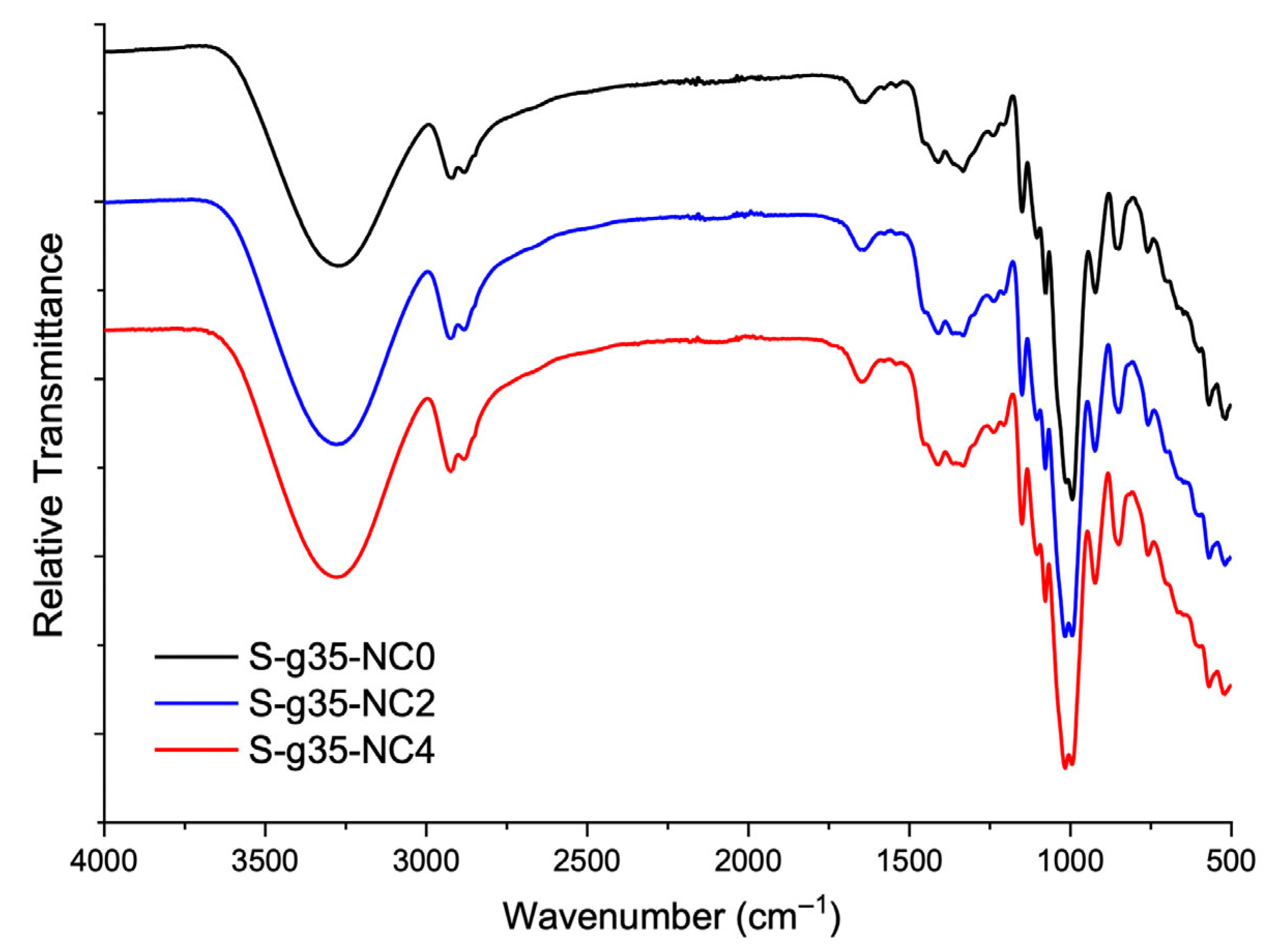
3.3. FTIR
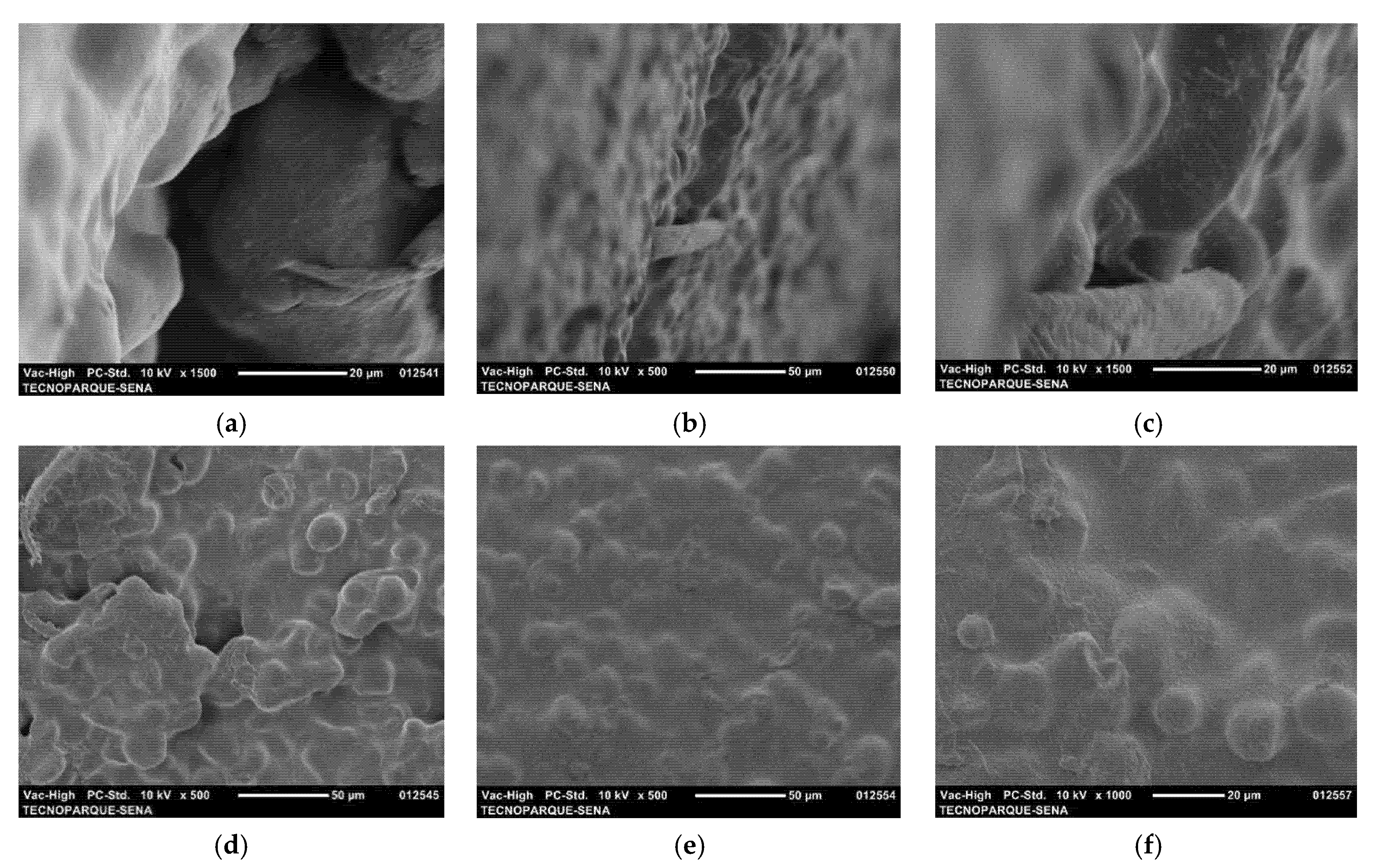
3.4. SEM of Nanoclays and Starch
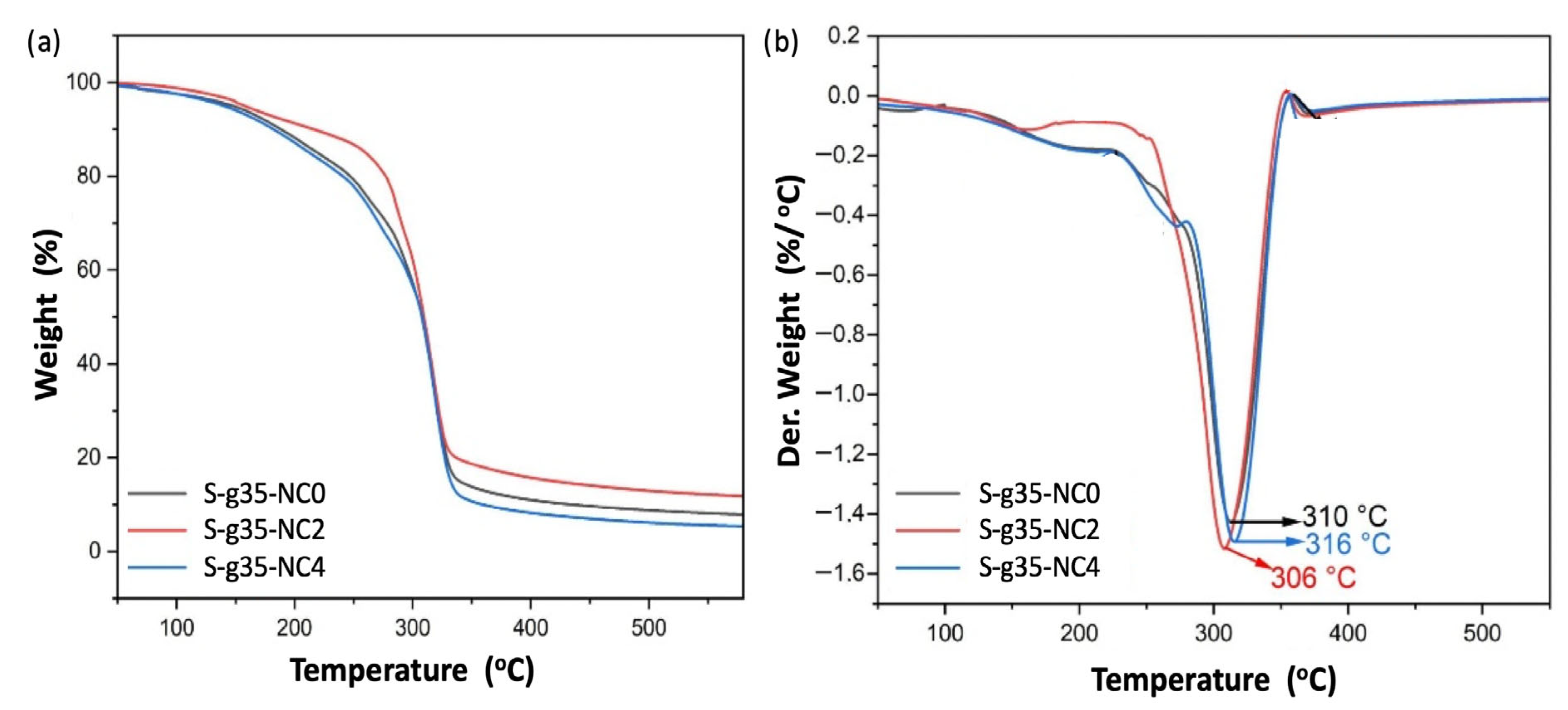
3.5. DSC and TGA
3.6. Characterization of Thermoplastic Starch (TPS) by DSC and TGA
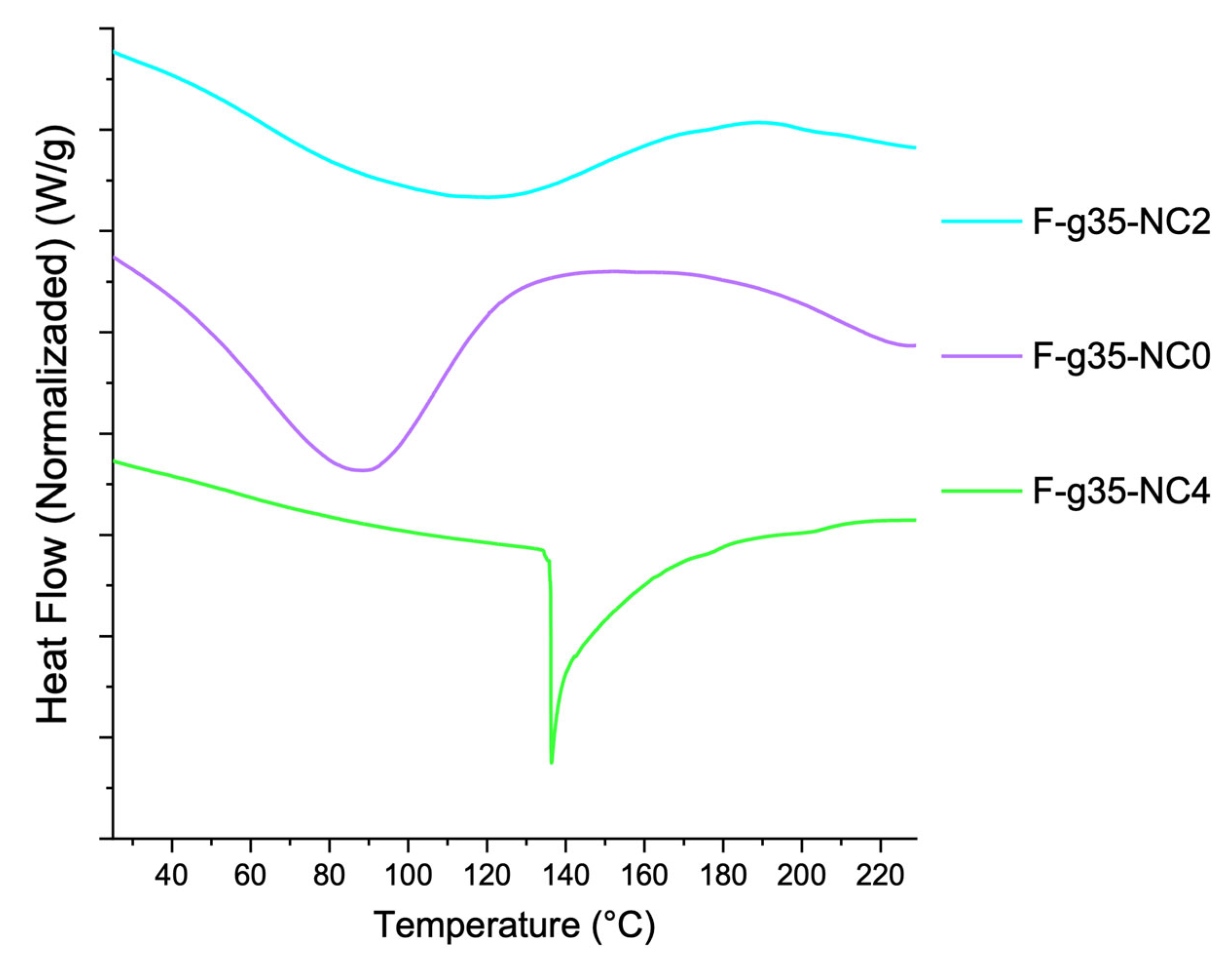
3.7. DSC Thermograms of the Films
3.8. Rheological Analysis of the TPS and the Obtained Films
3.9. Contact Angle
3.10. Determination of Mechanical and Barrier Properties
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| DSC | Differential Scanning Calorimetry |
| TGA | Thermogravimetric Analysis |
| FTIR | Fourier Transform Infrared Spectroscopy |
| NC | Nanoclay |
| TGA | Thermoplastic Starch |
| DLS | Dynamic light scattering |
| SEM | Scanning electron microscope |
References
- Plastics Europe. Plastics—The Facts 2024. Available online: https://plasticseurope.org/knowledge-hub/the-circular-economy-for-plastics-a-european-analysis-2024/ (accessed on 17 October 2024).
- Singh, P.; Pandey, V.K.; Singh, R.; Singh, K.; Dash, K.K.; Malik, S. Unveiling the potential of starch-blended biodegradable polymers for substantializing the eco-friendly innovations. J. Agric. Food Res. 2024, 15, 101065. [Google Scholar] [CrossRef]
- Gaur, A.; Singhal, N.; Singh, R.; Jain, R.; Singh, N.; Pant, G.; Karnwal, A.; Malik, T. Microplastics to Metabolomics: Understanding the Environmental and Health Implications of Plastic Pollution. Polym. Test. 2025, 150, 108918. [Google Scholar] [CrossRef]
- Rahardiyan, D.; Moko, E.M.; Tan, J.S.; Lee, C.K. Thermoplastic starch (TPS) bioplastic, the green solution for single-use petroleum plastic food packaging—A review. Enzym. Microb. Technol. 2023, 168, 110260. [Google Scholar] [CrossRef]
- Cataño, F.A.; Moreno-Serna, V.; Cament, A.; Loyo, C.; Yáñez-S., M.; Ortiz, J.A.; Zapata, P.A. Green composites based on thermoplastic starch reinforced with micro- and nano-cellulose by melt blending—A review. Int. J. Biol. Macromol. 2023, 248, 125939. [Google Scholar] [CrossRef]
- Rivadeneira-Velasco, K.E.; Utreras-Silva, C.A.; Díaz-Barrios, A.; Sommer-Márquez, A.E.; Tafur, J.P.; Michell, R.M. Green Nanocomposites Based on Thermoplastic Starch: A Review. Polymers 2021, 13, 3227. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhou, M.; Cheng, G.; Cheng, F.; Lin, Y.; Zhu, P.X. Fabrication and characterization of starch-based nanocomposites reinforced with montmorillonite and cellulose nanofibers. Carbohydr. Polym. 2019, 210, 429–436. [Google Scholar] [CrossRef]
- Burgess, M.; Holmes, H.; Sharmina, M.; Shaver, M.P. The future of UK plastics recycling: One bin to rule them all. Resour. Conserv. Recycl. 2021, 164, 105191. [Google Scholar] [CrossRef]
- Moshood, T.D.; Nawanir, G.; Mahmud, F.; Mohamad, F.; Ahmad, M.H.; AbdulGhani, A. Sustainability of biodegradable plastics: New problem or solution to solve the global plastic pollution? Curr. Res. Green Sustain. Chem. 2022, 5, 100273. [Google Scholar] [CrossRef]
- Olosho, A.I.; Alam, M.S.; Sukumaran Nair, K.; Ambade, A.V.; Adekola, F.A. Nonedible Thevetia peruviana Oil for the Synthesis of Biobased Thermosets and Vitrimers with Tunable Mechanical Properties. ACS Appl. Polym. Mater. 2024, 6, 2695–2708. [Google Scholar] [CrossRef]
- Yuan, N.; Xu, L.; Zhang, L.; Ye, H.; Zhao, J.; Liu, Z.; Rong, J. Superior hybrid hydrogels of polyacrylamide enhanced by bacterial cellulose nanofiber clusters. Mater. Sci. Eng. C 2016, 67, 221–230. [Google Scholar] [CrossRef]
- Bioeconomía Para una Colombia Potencia viva y Diversa: Hacia una Sociedad Impulasada por el Conocimiento. Ministerio de Ciencia, Tecnología e Innovación. Available online: https://minciencias.gov.co/sites/default/files/upload/paginas/bioeconomia_para_un_crecimiento_sostenible-qm_print.pdf (accessed on 18 October 2024).
- Abdolbaghi, S.; Pourmahdian, S.; Saadat, Y. Preparation of poly(acrylamide)/nanoclay organic-inorganic hybrid nanoparticles with average size of ∼250 nm via inverse Pickering emulsion polymerization. Colloid Polym. Sci. 2014, 292, 1091–1097. [Google Scholar] [CrossRef]
- Garcia, M.C.; Franco, C.M.L.; Júnior, M.S.S.; Caliari, M. Structural characteristics and gelatinization properties of sour cassava starch. J. Therm. Anal. Calorim. 2016, 123, 919–926. [Google Scholar] [CrossRef]
- Huang, H.-J.; Huang, S.-Y.; Wang, T.-H.; Lin, T.-Y.; Huang, N.-C.; Shih, O.; Jeng, U.-S.; Chu, C.-Y.; Chiang, W.-H. Clay nanosheets simultaneously intercalated and stabilized by PEGylated chitosan as drug delivery vehicles for cancer chemotherapy. Carbohydr. Polym. 2023, 302, 120390. [Google Scholar] [CrossRef]
- Jie, X.; Lin, C.; Qian, C.; He, G.; Feng, Y.; Yin, X. Preparation and properties of thermoplastic starch under the synergism of ultrasonic and elongational rheology. Int. J. Biol. Macromol. 2024, 274, 133155. [Google Scholar] [CrossRef]
- Zhang, S.; Li, X.; Gao, B.; Zhang, S. Reactive extrusion fabrication of thermoplastic starch with Ca2+ heterodentate coordination structure for harvesting multiple-reusable PBAT/TPS films. Carbohydr. Polym. 2024, 339, 122240. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F. Composition, structure, physicochemical properties, and modifications of cassava starch. Carbohydr. Polym. 2015, 122, 456–480. [Google Scholar] [CrossRef]
- Mina, J. Caracterización físico-mecánica de un almidón termoplástico (TPS) de yuca y análisis interfacial con fibras de Fique. Biotecnol. Sect. Agripecuario Agroindustrial 2012, 10, 99–109. [Google Scholar]
- Moreno-Sader, K.; García-Padilla, A.; Realpe, A.; Acevedo-Morantes, M.; Soares, J.B.P. Removal of Heavy Metal Water Pollutants (Co2+ and Ni2+) Using Polyacrylamide/Sodium Montmorillonite (PAM/Na-MMT) Nanocomposites. ACS Omega 2019, 4, 10834–10844. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, A.M.; Aziz, S.B.; Brza, M.A.; Saeed, S.R.; Al-Asbahi, B.A.; Sadiq, N.M.; Ahmed, A.A.A.; Murad, A.R. Glycerol as an efficient plasticizer to increase the DC conductivity and improve the ion transport parameters in biopolymer based electrolytes: XRD, FTIR and EIS studies. Arab. J. Chem. 2022, 15, 103791. [Google Scholar] [CrossRef]
- Enriquez, M.G.; Velasco, R.; Fernández, A. Caracterización de almidones de yuca nativos y modificados para la elaboración de empaques biodegradables. Biotecnol. Sect. Agropecu. Agroindustrial 2013, 11, 21–30. Available online: https://revistas.unicauca.edu.co/index.php/biotecnologia/article/view/1222 (accessed on 23 May 2025).
- Chen, X.; Yao, W.; Gao, F.; Zheng, D.; Wang, Q.; Cao, J.; Tan, H.; Zhang, Y. Physicochemical Properties Comparative Analysis of Corn Starch and Cassava Starch, and Comparative Analysis as Adhesive. J. Renew. Mater. 2021, 9, 979–992. [Google Scholar] [CrossRef]
- Elkhalifah, A.E.I.; Maitra, S.; Azmi Bustam, M.; Murugesan, T. Thermogravimetric analysis of different molar mass ammonium cations intercalated different cationic forms of montmorillonite. J. Therm. Anal. Calorim. 2012, 110, 765–771. [Google Scholar] [CrossRef]
- Olopade, B.K.; Nwinyi, O.C.; Adekoya, J.A.; Lawal, I.A.; Abiodun, O.A.; Oranusi, S.U.; Njobeh, P.B. Thermogravimetric Analysis of Modified Montmorillonite Clay for Mycotoxin Decontamination in Cereal Grains. Sci. World J. 2020, 2020, 1–5. [Google Scholar] [CrossRef]
- Caicedo, C.; Pulgarin, H.L.C. Study of the Physical and Mechanical Properties of Thermoplastic Starch/Poly(Lactic Acid) Blends Modified with Acid Agents. Processes 2021, 9, 578. [Google Scholar] [CrossRef]
- ASTM E1252-98; Standard Practice for General Techniques for Obtaining Infrared Spectra for Qualitative Analysis. ASTM International; Book of Standards. Volume 03.06. Available online: https://www.astm.org/e1252-98r21.html (accessed on 1 August 2025).
- ASTM D3418; Standard Test Method for Transition Temperatures and Enthalpies of Fusion and Crystallization of Polymers by Differential Scanning Calorimetry. ASTM International: Philadelphia, PA, USA, 2015.
- ASTM D1922; Test Method for Propagation Tear Resistance of Plastic Film and Thin Sheeting by Pendulum Method. ASTM International: West Conshohocken, PA, USA, 2020.
- ASTM F1249-20; Standard Test Method for Water Vapor Transmission Rate Through Plastic Film and Sheeting Using a Modulated Infrared Sensor. ASTM International: West Conshohocken, PA, USA, 2020.
- Zanini, N.C.; Ferreira, R.R.; Barbosa, R.F.S.; de Souza, A.G.; Camani, P.H.; Oliveira, S.A.; Mulinari, D.R.; Rosa, D.S. Two different routes to prepare porous biodegradable composite membranes containing nanoclay. J. Appl. Polym. Sci. 2023, 140, e54630. [Google Scholar] [CrossRef]
- Bangar, S.P.; Whiteside, W.S.; Ashogbon, A.O.; Kumar, M. Recent advances in thermoplastic starches for food packaging: A review. Food Packag. Shelf Life 2021, 30, 100743. [Google Scholar] [CrossRef]
- Bernardo, C.N.; Kling, I.C.S.; Ferreira, W.H.; Andrade, C.T.; Simao, R.A. Starch films containing starch nanoparticles as produced in a single step green route. Ind. Crops Prod. 2022, 177, 114481. [Google Scholar] [CrossRef]
- Calambas, H.L.; Fonseca, A.; Adames, D.; Aguirre-Loredo, Y.; Caicedo, C. Physical-Mechanical Behavior and Water-Barrier Properties of Biopolymers-Clay Nanocomposites. Molecules 2021, 26, 6734. [Google Scholar] [CrossRef]
- Dang, K.M.; Yoksan, R.; Pollet, E.; Avérous, L. Morphology and properties of thermoplastic starch blended with biodegradable polyester and filled with halloysite nanoclay. Carbohydr. Polym. 2020, 242, 116392. [Google Scholar] [CrossRef] [PubMed]
- Müller, C.M.O.; Laurindo, J.B.; Yamashita, F. Composites of thermoplastic starch and nanoclays produced by extrusion and thermopressing. Carbohydr. Polym. 2012, 89, 504–510. [Google Scholar] [CrossRef] [PubMed]
- Gil, E.; Mesa, M. Efectos de la Modificación de Arcilla con Ácido Cítrico en la Morfología y Retrogradación de Almidón Termoplástico (TPS) Plastificado con Glicerol. Bachelor’s Thesis, Universidad de los Andes, Bogotá, Colombia, 2013. [Google Scholar]
- Gutiérrez, T.J. Starch-based food packaging films processed by reactive extrusion/thermo-molding using chromium octanoate-loaded zeolite A as a potential triple-action mesoporous material (reinforcing filler/food-grade antimicrobial organocatalytic nanoreactor). Food Packag. Shelf Life 2022, 34, 100974. [Google Scholar] [CrossRef]
- Guzmán, M.; Murillo, E.A. Structural, thermal, rheological, morphological and mechanical properties of thermoplastic starch obtained by using hyperbranched polyester polyol as plasticizing agent. DYNA 2018, 85, 178–186. [Google Scholar] [CrossRef]
- Wu, H.; Hou, A.; Hu, X.; Lu, X.; Qu, J.-P. Effect of elongational rheology on plasticization and properties of thermoplastic starch prepared by biaxial eccentric rotor extruder. Ind. Crops Prod. 2022, 176, 114323. [Google Scholar] [CrossRef]
- Giridhar, G.; Manepalli, R.K.N.R.; Apparao, G. Contact Angle Measurement Techniques for Nanomaterials. In Thermal and Rheological Measurement Techniques for Nanomaterials Characterization; Elsevier: Amsterdam, The Netherlands, 2017; pp. 173–195. [Google Scholar] [CrossRef]
- Gutiérrez, T.J.; Ollier, R.; Alvarez, V.A. Surface Properties of Thermoplastic Starch Materials Reinforced with Natural Fillers. In Functional Biopolymers; Springer: Berlin/Heidelberg, Germany, 2018; pp. 131–158. [Google Scholar] [CrossRef]
- Noshirvani, N.; Hong, W.; Ghanbarzadeh, B.; Fasihi, H.; Montazami, R. Study of cellulose nanocrystal doped starch-polyvinyl alcohol bionanocomposite films. Int. J. Biol. Macromol. 2018, 107, 2065–2074. [Google Scholar] [CrossRef] [PubMed]
- Aguirre-Loredo, R.Y.; Fonseca-García, A.; Calambas, H.L.; Salazar-Arango, A.; Caicedo, C. Improvements of thermal and mechanical properties of achira starch/chitosan/clay nanocomposite films. Heliyon 2023, 9, e16782. [Google Scholar] [CrossRef]
- Medina, L. Evaluación de los Parámetros de Procesamiento y Formulación de Involucrados en el Desarrollo de una Lamina TPS Para Termoformado. Bachelor’s Thesis, Universidad de los Andes, Bogotá, Colombia, 2007. [Google Scholar]
- Giannakas, A.; Grigoriadi, K.; Leontiou, A.; Barkoula, N.-M.; Ladavos, A. Preparation, characterization, mechanical and barrier properties investigation of chitosan–clay nanocomposites. Carbohydr. Polym. 2014, 108, 103–111. [Google Scholar] [CrossRef]
- Vaezi, K.; Asadpour, G.; Sharifi, S.H. Bio nanocomposites based on cationic starch reinforced with montmorillonite and cellulose nanocrystals: Fundamental properties and biodegradability study. Int. J. Biol. Macromol. 2020, 146, 374–386. [Google Scholar] [CrossRef]
- Nouri, A.; Yaraki, M.T.; Ghorbanpour, M.; Agarwal, S.; Gupta, V.K. Enhanced Antibacterial effect of chitosan film using Montmorillonite/CuO nanocomposite. Int. J. Biol. Macromol. 2018, 109, 1219–1231. [Google Scholar] [CrossRef]
- Punia Bangar, S.; Whiteside, W.S.; Chaudhary, V.; Parambil Akhila, P.; Sunooj, K.V. Recent functionality developments in Montmorillonite as a nanofiller in food packaging. Trends Food Sci. Technol. 2023, 140, 104148. [Google Scholar] [CrossRef]
- Krishnaswamy, R.; Sukhadia, A. Orientation characteristics of LLDPE blown films and their implications on Elmendorf tear performance. Polymer 2000, 41, 9205–9217. [Google Scholar] [CrossRef]







| Sample | Gly (%) | Nanoclay (%) | |||
|---|---|---|---|---|---|
| 35 | 40 | 0 | 2 | 4 | |
| S-g35-NC0 | x | x | |||
| S-g35-NC2 | x | x | |||
| S-g35-NC4 | x | x | |||
| S-g40-NC0 | x | x | |||
| S-g40-NC2 | x | x | |||
| S-g40-NC4 | x | x | |||
| TS1 (°C) | TS2 (°C) | TS3 (°C) | TS4 (°C) | TS5 (°C) | TS6 (°C) | TS7 (°C) | TS8 (°C) | TS9 (°C) | TS10 (°C) | TS-D1 (°C) |
|---|---|---|---|---|---|---|---|---|---|---|
| 70 | 70 | 70 | 75 | 80 | 85 | 90 | 95 | 100 | 105 | 110 |
| Sample | Maximum Torque [N·m] |
|---|---|
| S-g35-NC0 | 8.5 |
| S-g35-NC2 | 7.6 |
| S-g35-NC4 | 8.2 |
| S-g40-NC0 | 6.5 |
| S-g40-NC2 | 5.4 |
| S-g40-NC4 | 6.1 |
| Films | Average Contact Angle 0 s | Average Contact Angle 10 s | Average Contact Angle 60 s |
|---|---|---|---|
| P |  |  |  |
| 52.00° ± 1.37 a | 56.48° ± 3.33 a | 50.66° ± 6.64 a | |
| PNA 2% |  |  |  |
| 73.90° ± 3.22 b | 55.21° ± 4.62 a | 46.12° ± 4.87 a | |
| PNA 4% |  |  |  |
| 89.93° ± 8.78 c | 58.17° ± 1.59 a | 51.13° ± 2.61 a |
| Films | Tensile Strength | Percentage of Deformation | Water Vapor Transmission Rate | Tear Resistance |
|---|---|---|---|---|
| (MPa) | (%) | (g/m2·day) | (kg) | |
| F-g35-NC0 | 0.48 ± 0.10 a | 6.80 ± 1.01 a | 0.170 ± 0.095 a | 0.090 ± 0.007 a |
| F-g35-NC2 | 0.20 ± 0.03 b | 44.57 ± 3.67 b | 0.054 ± 0.007 b | 0.680 ± 0.019 b |
| F-g35-NC4 | 0.23 ± 0.02 b | 66.90 ± 4.85 c | 0.030 ± 0.011 c | 0.740 ± 0.009 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Criollo Guevara, H.T.; Ocoró Caicedo, L.V.; Rios Acevedo, J.J.; Guancha Chalapud, M.A.; Caicedo, C. Rheological, Thermal and Mechanical Properties of Blown Film Based on Starch and Clay Nanocomposites. Processes 2026, 14, 276. https://doi.org/10.3390/pr14020276
Criollo Guevara HT, Ocoró Caicedo LV, Rios Acevedo JJ, Guancha Chalapud MA, Caicedo C. Rheological, Thermal and Mechanical Properties of Blown Film Based on Starch and Clay Nanocomposites. Processes. 2026; 14(2):276. https://doi.org/10.3390/pr14020276
Chicago/Turabian StyleCriollo Guevara, Heidy Tatiana, Lis Vanesa Ocoró Caicedo, Jhon Jairo Rios Acevedo, Marcelo Alexander Guancha Chalapud, and Carolina Caicedo. 2026. "Rheological, Thermal and Mechanical Properties of Blown Film Based on Starch and Clay Nanocomposites" Processes 14, no. 2: 276. https://doi.org/10.3390/pr14020276
APA StyleCriollo Guevara, H. T., Ocoró Caicedo, L. V., Rios Acevedo, J. J., Guancha Chalapud, M. A., & Caicedo, C. (2026). Rheological, Thermal and Mechanical Properties of Blown Film Based on Starch and Clay Nanocomposites. Processes, 14(2), 276. https://doi.org/10.3390/pr14020276










