Abstract
Novel initiation systems for the production of poly(methyl methacrylate) (PMMA) bone cements based on low-toxicity tertiary amine initiators and hydroxyapatite nanoparticles were investigated. Bicomponent systems formed by a solid component containing PMMA and benzoyl peroxide (BPO) and a liquid component containing methylmethacrylate and low-toxicity aliphatic (dimethylamino-ethoxy-ethane) (DMEE) and aromatic (dimethylamino-benzaldehyde) (DMAB) tertiary amines were tested at two amine concentrations (0.75 and 3.75 wt%) and compared with the standard tertiary amine dimethyl-p-toluidine (DMT). The components were mixed in a 2:1 ratio (solid/liquid) for 60 s. Nanocomposites were prepared using nano-hydroxyapatite inserted into the PMMA-based polymer matrix at various concentrations between 1.0 and 3.75 wt%, aiming to increase the biocompatibility of bone cements applied in cranioplasty. The concentration of tertiary amines directly affects the reaction rate, and increasing the concentration accelerates the curing reaction. Thermal analyses (DSC and TGA) revealed that the produced polymers did not show significant changes in glass transition temperature (113 °C and 115 °C), nor in onset (150–158 °C) or peak degradation temperatures (353 °C and 355 °C). The reaction with the aliphatic amine proved to be slow, as no polymerization occurred within the time period stipulated in the study. However, drastic changes did take place when the cure occurred in the presence of nano-hydroxyapatite. The cure with DMAB (3.75 wt%) that presented an exothermic peak at 37 min (53 °C) showed a peak at 16 min (70 °C), and the cure with DMEE (3.75 wt%) a peak at 11 min (62.5 °C) after the addition of nano-hydroxyapatite. In conclusion, addition of nano-hydroxyapatite significantly influenced both the time and the temperature of cure reaction in all amines studied, expanding the possibility of using new initiators in polymerization systems for cranioplasty flaps.
1. Introduction
Cranioplasty is a common technique used to repair bone defects in the skull resulting from fractures and/or traumatic cranial deformities. In addition to alleviating neurological symptoms, it also helps mitigate psychosocial symptoms [1]. However, it may result in complications due to the potential for bone resorption and infections [2,3].
Self-curing bone cements based on PMMA have been widely used in cranioplasties. The chemical polymerization reaction of acrylic monomers is initiated by benzoyl peroxide and a tertiary amine, which allows the reaction to occur at room temperature [4,5].
Currently, the standard tertiary amine used in radical polymerization systems at room temperature is N,N-dimethyl-p-toluidine (DMT), which can efficiently promote the reaction in a short period of time at room temperature [6,7]. However, studies have proven that DMT has neurotoxic potential. Other disadvantages include high polymerization temperatures, which can cause difficulties for doctors [8,9,10].
Over the years, various tertiary amines for use in the polymerization synthesis of PMMA-based bone cement have been studied [11,12]. Furthermore, the scientific community has been actively searching for new, more biocompatible tertiary amines for biomedical applications [13,14,15,16,17].
In addition to replacing DMT with more biocompatible initiators, a wide range of PMMA-based composites has also been continuously investigated to improve the biocompatibility of PMMA-based bone cements used in cranioplasties. In this context, hydroxyapatite (HA) has emerged as a promising bioactive particle capable of improving the biocompatibility of the material, promoting a greater osteoblastic response and, consequently, directly reinforcing osseointegration [18].
This study investigated the synthesis of PMMA-based bone cements, proposing two new amines, dimethylamino-ethoxy-ethane (DMEE) and dimethylamino-benzaldehyde (DMAB). Both amines have lower toxicity than the standard amine. The effect of inserting hydroxyapatite nanoparticles was also investigated, in an attempt to improve the biocompatibility of bone cements for application in cranioplasty.
2. Materials and Methods
2.1. Materials
Preparation of polymers and nanocomposite was carried out using methyl methacrylate (methyl 2-methylprop-2-enoate) (MMA) (purity > 99%), poly(methyl methacrylate) (purity > 99%), dibenzoyl peroxide A75 (BPO) (LUPEROX®, Pennsylvania, PA, USA), dimethyl-p-toluidine (N,N-dimethyl-4-methylaniline) (purity > 99% − lethal dose = 139 mg/kg), dimethylamino-ethoxy-ethano (2-[2-(dimethylamino)ethoxy]ethan-1-ol) (purity > 99% − lethal dose = 2460 mg/kg), dimethylamino-benzaldehyde (4-(dimethylamino)benzaldehyde) (purity >99% − lethal dose = 800 mg/kg), and chloroform (trichloromethane), all of which were supplied by Sigma-Aldrich (Saint Louis, MO, USA). Methyl methacrylate was purified by distillation. Nano-hydroxyapatite was synthesized from (NH)2 HPO4 and Ca(NO3)2 at 90 °C and pH 9 according to methodology previously described [19].
2.2. Preparation of Polymers and Nanocomposite
Polymers and nanocomposites (bone cement) were synthesized from a physical mixture of the two components, powder (P) and liquid (L), with a P:L ratio of 2:1 (w/w), using a glass stick for 60 s. The powder component consisted of PMMA (10 g) and the initiator BPO (0.40 g). The liquid component contained two compounds: the MMA monomer (4.5 mL) and the tertiary amine co-initiator (0.75 wt% = 0.0040 mL and 3.75 wt% = 0.02 mL). For synthesis of the nanocomposites, a hydroxyapatite-based nanoparticle (1 wt% = 0.1 g; 2.5 wt% = 0.25 g and 3.75 wt% = 0.375 g) was dispersed through a tip sonicator in the liquid component. Nanoparticle dispersion in the PMMA matrix was achieved by ultrasonication using a probe sonicator operating at 20% amplitude. The total sonication time was 60 s, applied in two cycles of 30 s, with a 10 s interval between cycles to minimize thermal effects. The polymerization occurred at room temperature.
2.3. Characterization of HA Nanoparticle
The morphology of nano-hydroxyapatite was characterized by transmission electron microscopy using a high-resolution transmission electron microscope (TEM)—JEOL 2100F 200 kV 2 (JEOL Ltd., Tokyo, Japan).
Fourier-transform infrared spectroscopy (FTIR) analyses of nano-hydroxyapatite powder were performed using a Shimadzu IRPrestige-21 spectrometer. FTIR analyses were performed in the range of 4000–400 cm−1, using KBr pellets.
X-ray diffraction (XRD) measurements of nano-hydroxyapatite powder were carried out using a PANalytical X-Per PRO instrument (Tokyo, Japan) using CuKα radiation (λ = 0.15405 nm), operated at 40 kV, 40 mA, an angular range of 10° < 2θ < 80° with 0.05° steps, and a counting time of 160 s/step.
2.4. Characterization of Polymers and Nanocomposite
Nuclear magnetic resonance 1H-NMR spectra were recorded at 25 °C using a Varian Mercury VX-300 NMR spectrometer (Palo Alto, CA, USA) and CDCl3 as solvent. Analysis parameters included 16 scans, a 10 s relaxation delay, and an acquisition time of 2.5 s, with an experimental error of 5%.
Gel permeation chromatography (GPC) was performed on a Shimadzu LC 20 system. A Shim-pack GPC-803 (Kyoto, Japan), 5 μm Phenogel columns, and a refractive index detector were used, with dimethylacetamide as solvent at a flow rate of 0.8 mL/min. The samples were solubilized at 80 °C for 24 h and filtered through a 0.45 µm PTFE filter. The analyses were performed with columns conditioned at 80 °C, using a solution concentration of 0.3% (w/v) and an injection volume of 20 µL. A calibration curve was constructed with monodisperse PMMA standards. All samples were completely dissolved.
Thermal properties were determined by DSC using a Hitachi 7020 calorimeter (Tokyo, Japan) with a 10 mg sample. In the first run, heating to 250 °C was carried out, followed by cooling at a rate of 50 °C/min, to erase the thermal history of the material. In the second run, heating and cooling at a rate of 10 °C/min were carried out to determine the glass transition temperature (Tg). Both runs were performed under flow N2. The estimated error for each sample in 3 injections was 2%.
Thermogravimetric analyses (TGA) were performed with 10 mg of sample, using a TA TGA Q500 thermogravimeter (New Castle, DE, USA). Measurements were conducted under a N2 atmosphere at a heating rate of 10 °C/min up to 700 °C, with a gas flow rate of 20 mL/min.
3. Results and Discussion
3.1. Morphology of HA Nanoparticle
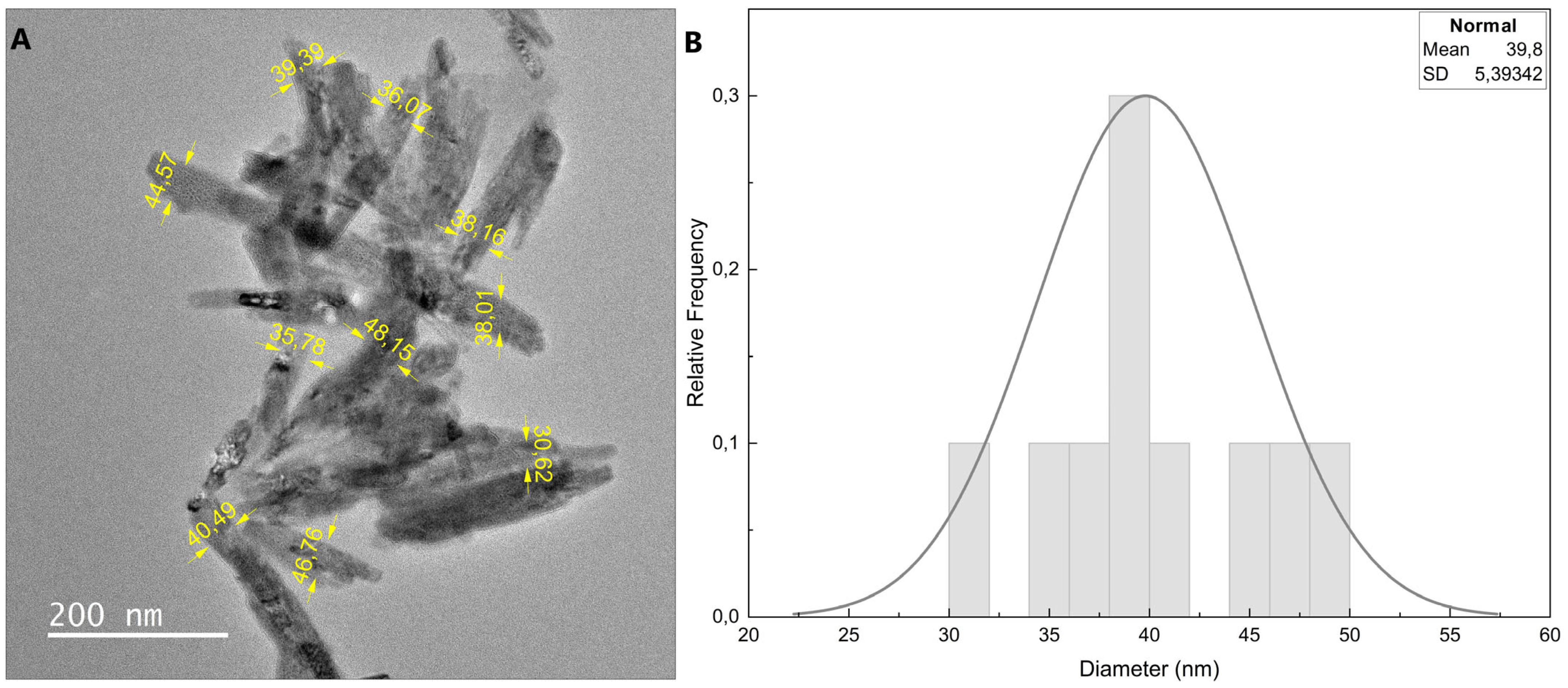
The structure and morphology of the nano-hydroxyapatite was investigated by transmission electron microscopy (TEM), Fourier-transform infrared spectroscopy (FTIR), and X-ray diffraction (XRD). TEM images of HA nanoparticles showed that they were rod-shaped particles with an average length of 230.5 ± 29.1 nm. The average diameter of the nanoparticles was 39.5 ± 5.39 nm, as shown in Figure 1.
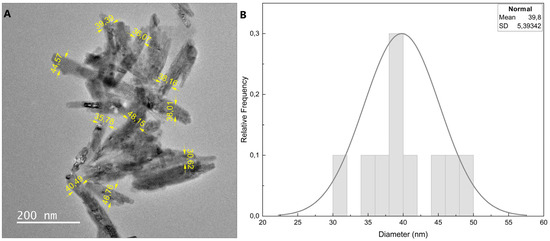
Figure 1.
(A) TEM images of hydroxyapatite nanoparticle; (B) diameter distribution of HA nanoparticles.
Functional groups associated with hydroxyapatite were identified by FTIR spectroscopy, analyzing the absorbance spectra for neat nano-hydroxyapatite in the region between 500 and 4000 cm−1. All specific absorption bands expected for the nano-hydroxyapatite structure were observed; these included an ion stretching vibration around 3572 cm−1, attributed to the presence of the hydroxyl (OH) groups, and vibrations of phosphate (PO4)3− groups at 1086 cm−1 and 629 cm−1. (Supplementary Material Figure S1).
The structural analysis of nano-hydroxyapatite was performed using X-ray powder. The X-ray diffraction showed sharp peaks, indicating significant crystallinity. Peak positions were in good agreement with the literature, and it was possible to observe that nano-hydroxyapatite exhibited several diffraction peaks, at 2θ values of 26.10, 29.20, 32.00, 32.10, 34.30, 40.00, 46.90, 49.70, and 53.30, corresponding to the respective crystal planes of (002), (210), (211), (112), (300), (202), (222), (213), and (004). (Supplementary Material Figure S2). These diffraction peaks can be attributed to hexagonal crystal structures according to the JCPDS standard card no. 01-0721243 [20,21]. The crystallite size (L) was calculated using the Scherrer equation:
where λ is the wavelength of the X-ray radiation (1.54 Å), θ is the angle of diffraction of the peak, K is the factor (0.89), and β is the total width at half maximum (FWHM) of the diffraction peak. The crystallite size according to the Scherrer equation was 8 nm. The average crystallite size (8 nm) estimated by the Scherrer equation was significantly smaller than the average nanoparticle size (230 nm × 40 nm) visualized by TEM, indicating that the hydroxyapatite nanoparticles exhibited a polycrystalline character.
3.2. Thermal Profile for Synthesis of Bone Cement and Nanocomposite
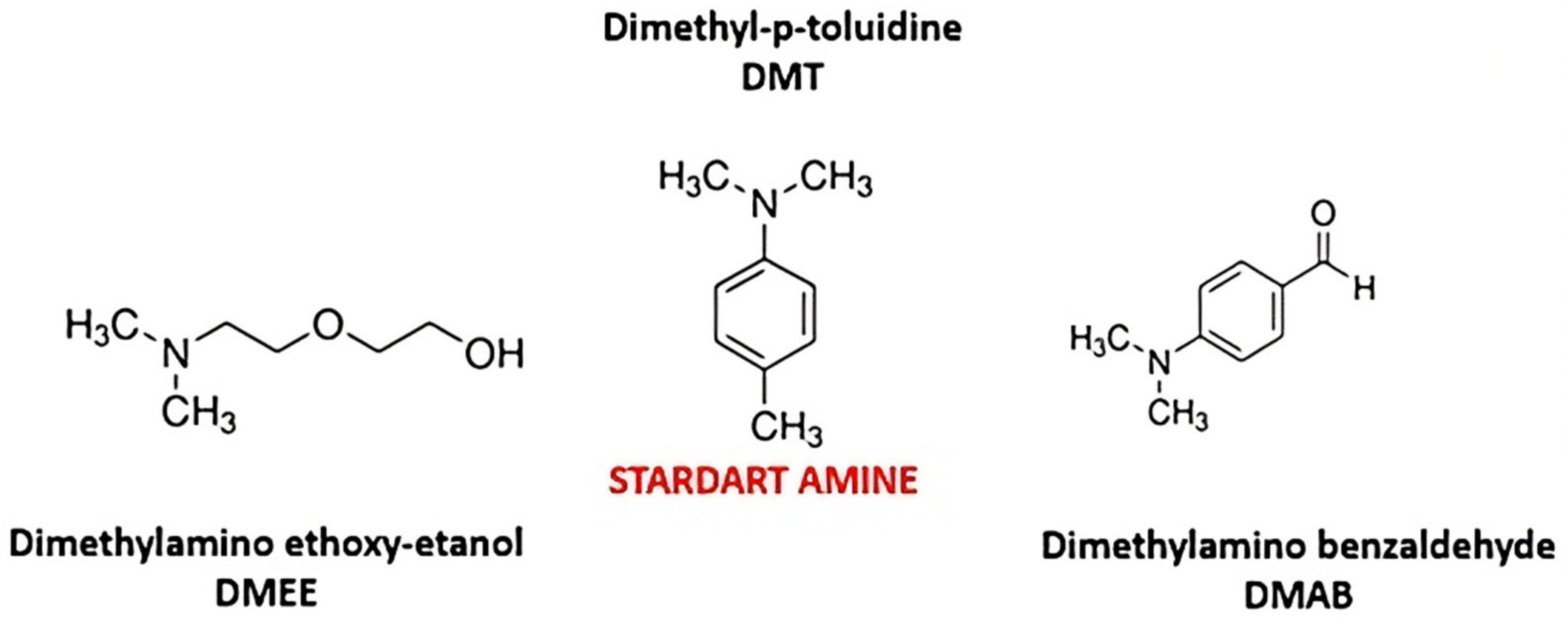
Two different tertiary amine structures, one aliphatic (DMEE) and another aromatic (DMAB) (Figure 2), were evaluated in this work. Both molecules have lower toxicity compared to the standard amine (DMT). The thermal profiles of bone cements with different tertiary amine concentrations were analyzed to understand their influence on polymerization. The influence of HA nanoparticles on composition at different concentrations was also analyzed. Temperature was measured using a thermocouple directly introduced in the bulk reaction. After mixing the two components of the system for 60 s, the temperature was monitored for 60 min.
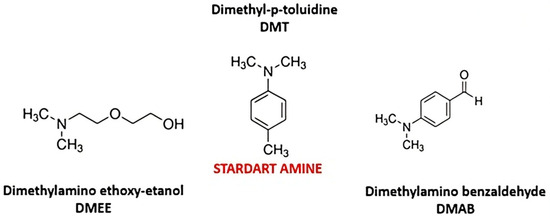
Figure 2.
Tertiary aliphatic and aromatic amines evaluated as co-initiators of bone cements.
First, the influence of the standard tertiary amine (DMT) concentration on the polymerization reaction was investigated using three different concentrations: 0.75 wt% (standard), 1.50 wt%, and 3.75 wt%. The results demonstrated that the higher the amine concentration, the faster and more exothermic the cure/polymerization reaction. (Supplementary Material Figure S3). The reaction enthalpies were calculated by DSC, and these confirmed the described trend. It must be highlighted that the reaction with 3.75 wt% DMT occurred so quickly that when the analysis was initiated, the reaction had already started, making it difficult to obtain an accurate analysis of the reaction enthalpy, resulting in an enthalpy evaluation which was lower than the true reaction enthalpy.
Subsequently, the amines proposed in this work (DMAB and DMEE) were tested using the standard concentration (0.75 wt%). Neither of the proposed amines polymerized during the experimental time of 60 min.
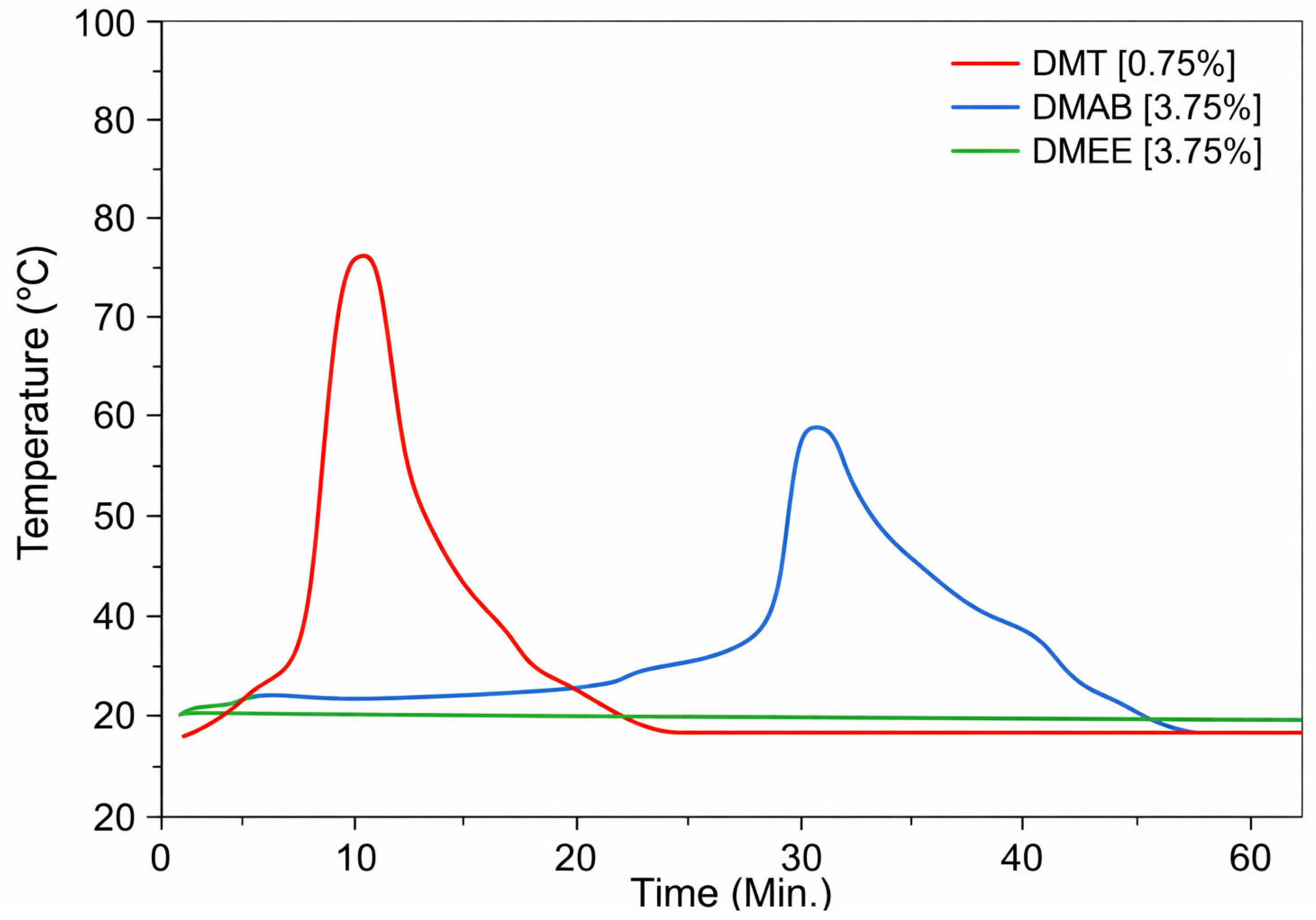
After increasing the amine concentration from 0.75 wt% to 3.75 wt%, only the DMAB was able to polymerize, presenting an exothermic peak of 53 °C in 37 min at a concentration of 3.75 wt% (Figure 3). Using 2.25 wt% of DMAB, the reaction was slower and less exothermic. (Supplementary Material Figure S4). The aliphatic amine continued to show no activity during the time interval proposed in the study. The aliphatic amine proved to be extremely slow-acting, taking approximately 7 days to consume all of the monomer. This issue was confirmed by FTIR analysis by monitoring monomer consumption along the reaction time, using the absorption band at 1650 cm−1 of C=C which was attributed to the monomer. (Supplementary Material Figures S5 and S6).
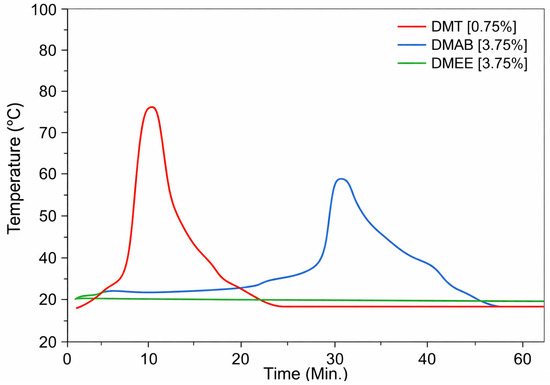
Figure 3.
Thermal profile of PMMA-based bone cement prepared by radical polymerization of systems initiated by DMT, DMAB, and DMEE in standard concentrations.
The temperature peak observed in polymerization reactions using the standard tertiary amine (DMT) and the aromatic amine DMAB is related to the initiation and propagation stages of the polymerization reaction. After mixing the cement components, the tertiary amine decomposes the benzoyl peroxide initiator, generating free radicals that interact with the methacrylate monomer, generating other radicals that subsequently exothermic react with other monomer molecules until the polymer is formed, releasing energy and causing a temperature increase [22].
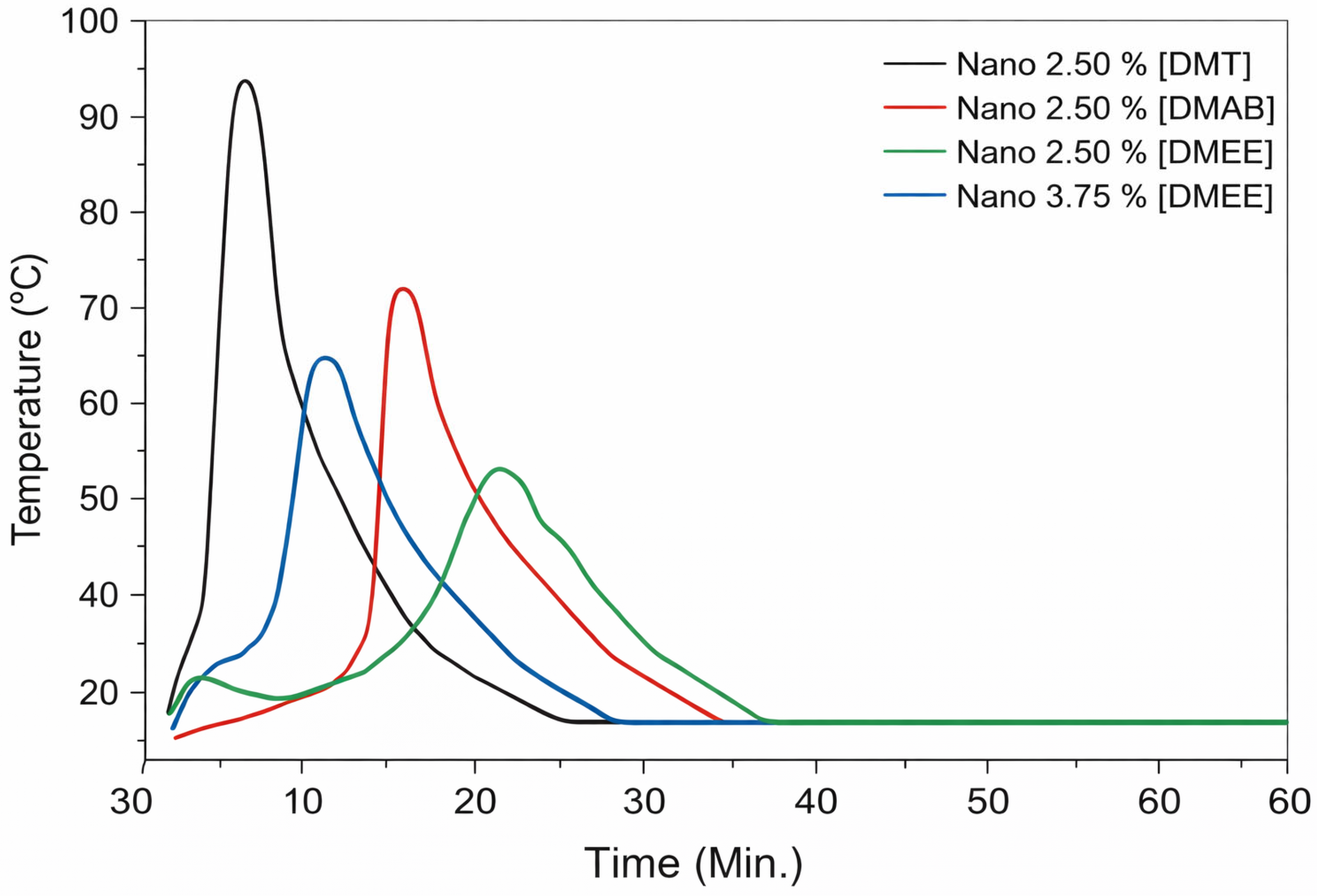
In this work, PMMA-nano-hydroxyapatite nanocomposites with different nanoparticle concentrations were also prepared by polymerizing the monomer in presence of the nanofiller. The reactions were monitored using a thermocouple for 60 min. In general, the addition of nano-hydroxyapatite into this polymerization system increases the speed of the reaction, and the higher the nano-hydroxyapatite concentration, the faster the reaction and the greater the amount of heat released. Curiously, however, it seems that a saturation effect occurs, so that above a certain high nano-hydroxyapatite concentration (5% and 7.5 wt%) no change in the reaction rate is observed, a phenomenon associated with difficulty in filler dispersion.
In reactions with the standard amine (DMT), reaction temperatures reached 78 °C in 8 min and 94 °C in 6 min in reactions with 1 wt% and 2.5 wt% nanoparticles, respectively. (Supplementary Material Figure S7). In syntheses with the amine (DMAB), reaction temperatures reached 53 °C in 36 min and 70 °C in 16 min in reactions containing 1% and 2.5 wt% of nanoparticles, respectively. (Supplementary Material Figure S8).
Considering that HA nanoparticles tend to accelerate the reaction, the aliphatic tertiary amine DMEE was also tested. With the addition of the nano-hydroxyapatite to polymerization systems co-initiated by DMEE, the reaction exhibited an exothermic peak within the study time limit, as shown in Figure 4. The reaction temperatures reached 51 °C in 22 min and 62.5 °C in 11 min, respectively, in the reactions with 2.5 wt% and 3.75 wt% HA nanoparticles.
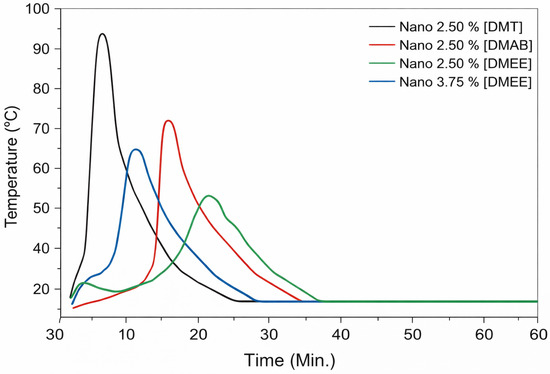
Figure 4.
Thermal profiles of polymerizations of MMA co-initiated by DMT, DMAB, and DMEE at different concentrations in PMMA-based bone cement.
The acceleration effect may be related to the fact that the hydroxyapatite nanoparticle interacted with the BPO/amine initiation system, facilitating electron transfer and generating a catalytic effect, consequently increasing the reaction rate and temperature.
The literature reports that the addition of hydroxypatite particles into MMA polymerization causes a reduction in the reaction temperature [23,24]. In the case of the nano-hydroxyapatite used in this work, the opposite effect was observed, i.e., an accelerating increase in reaction temperature. This demonstrates that tertiary amines like DMEE, previously considered inefficient due to their extremely slow action, are very effective in accelerating MMA polymerization reactions at room temperature when co-actuating with nano-hydroxyapatite. It is worth noting that there are no reports in the literature of a reaction acceleration effect caused by the addition of hydroxyapatite nanoparticles into PMMA-based bone cements.
It was also possible to observe that the polymerization reaction using the aliphatic amine DMEE and 3.75 wt% of nano-hydroxyapatite resulted in peaks in maximum temperature and reaction time similar to those in polymerization reactions with the standard tertiary amine.
3.3. Molecular Weights of Polymers and Nanocomposites
The molecular weights and molecular weight distributions of these PMMA-base bone cements were investigated by using gel permeation chromatography (GPC). The main findings are presented in Table 1.

Table 1.
Molecular weights, glass transitions, temperatures of thermal weight loss, and monomer conversions of PMMA-based bone cement prepared with DMT and DMB at different amine concentrations.
According to Table 1, the increase in the concentration of the standard tertiary amine DMT in the bone cement composition caused a decrease in both number average molecular weight (Mn) and weight average molecular weight (Mw). Bone cements synthesized with the amine DMAB presented lower Mn and Mw values when compared to the standard system (DMT, 0.75 wt%). Regarding molecular weight distribution, regardless of the tertiary amine and its concentration, all cements showed a slight increase in polydispersity (Mw/Mn) as the DMT concentration increased.
3.4. Thermal Characterization of PMMA-Base Bone Cements
Differential scanning calorimetry (DSC) and thermogravimetry (TGA) were used to evaluate the thermal characterization of PMMA-base bone cements.
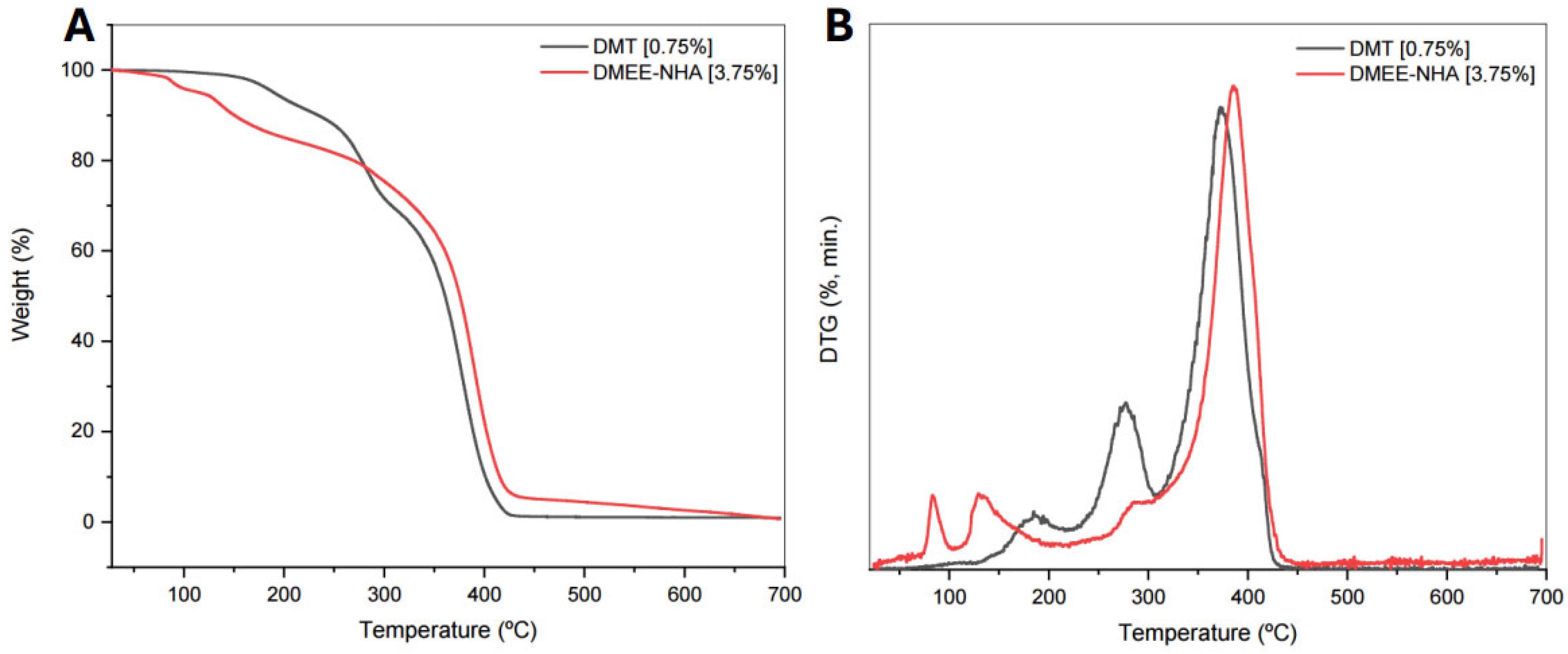
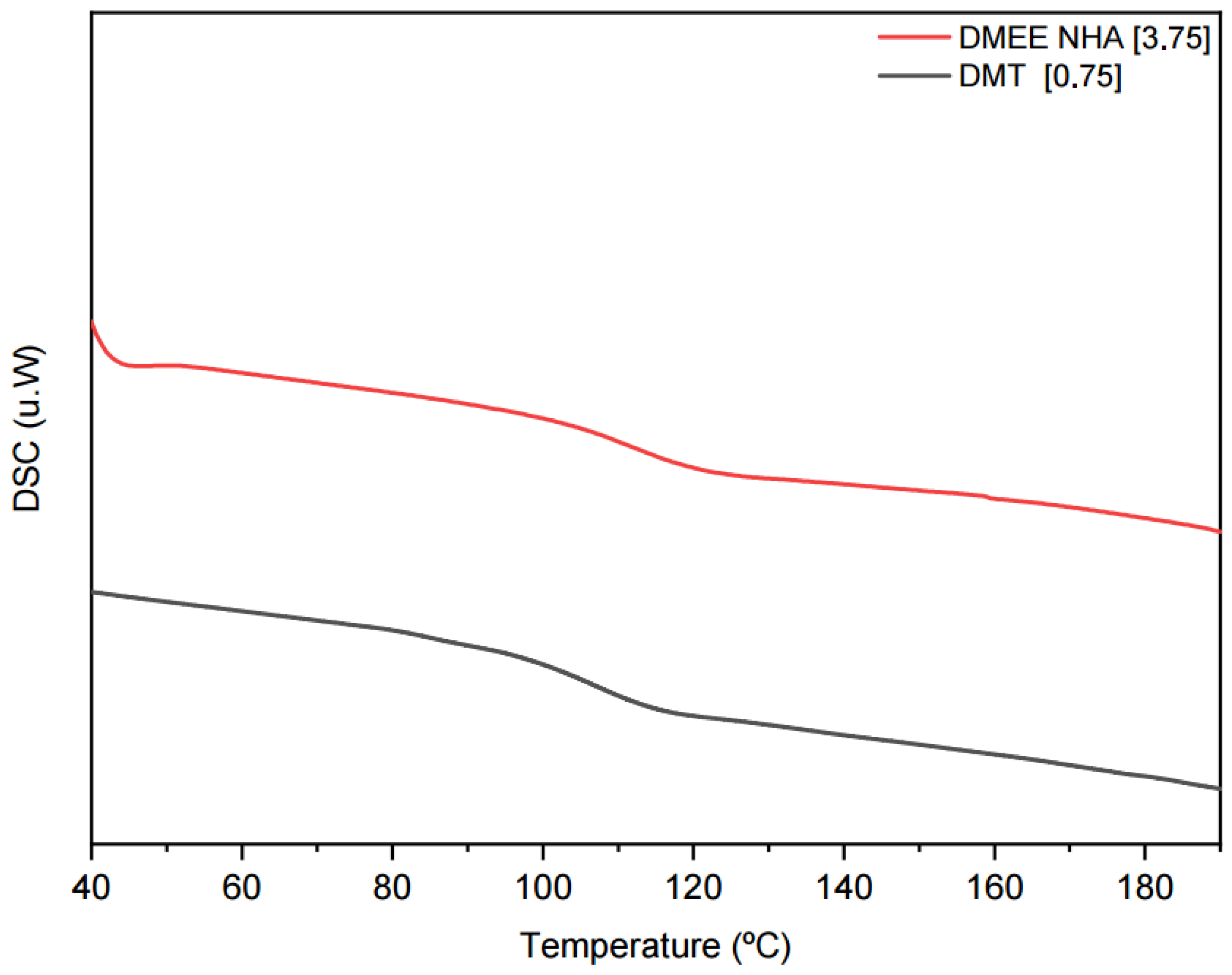
Table 1 and Table 2, and Figure 5 and Figure 6, present the DSC and TGA results for these bone cements, prepared in the absence or presence of nano-hydroxyapatite, and show the glass transition temperature (Tg), the initial weight loss temperature (Tonset), and the final weight loss temperatures (TOffset) for these materials.

Table 2.
Glass transition temperatures, thermal weight loss temperatures, and monomer conversions of PMMA- nano-hydroxyapatite bone cements prepared with DMT, DMB, and DMEE.
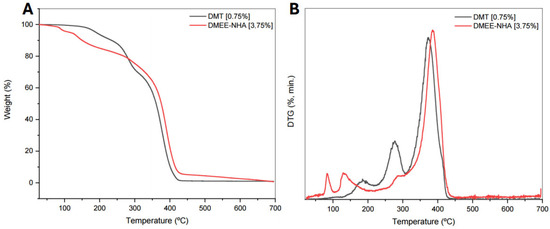
Figure 5.
Curves for (A) TG and (B) DTG of bane cement prepared with DMT 0.75 wt% and nanocomposite with 3.75 wt% DMEE and hydroxyapatite.
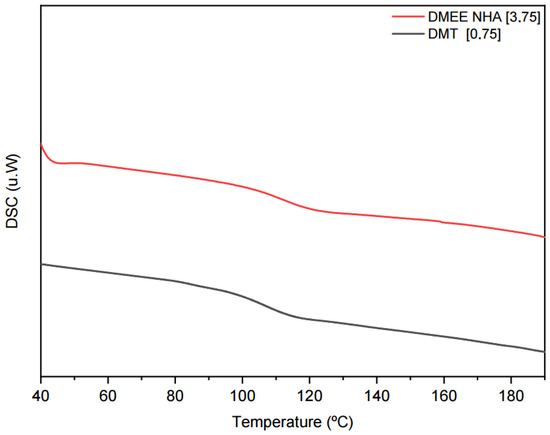
Figure 6.
DSC curves of bone cements prepared with 0.75 wt% DMT and nanocomposite with 3.75 wt% DMEE and hydroxyapatite.
Based on the data, it seems that there are no significant changes in the Tg of the bone cements with variation in amine concentration in the formulations, regardless of the tertiary amine used, because Tg varied between 115 °C and 106 °C. Nanocomposites synthesized with DMEE presented the lowest values.
TGA data showed small variations in Tonset and Toffset with the use of different amines and nano-hydroxyapatite concentrations. Both temperatures tended to decrease with increasing concentrations of tertiary amine and nano-hydroxyapatite. When correlating the amines used in the syntheses, we noted that the bone cements produced with DMAB and DMEE presented Tonset and Toffset values which were slightly lower than those obtained with the standard amine. In nanocomposites synthesized by DMEE, the decrease in glass transition temperature and initial degradation temperature may be related to the high content of unreacted monomers in the composition.
3.5. Residual Monomer in PMMA-Based Bone Cements
Residual monomer in PMMA-based bone cements was evaluated by proton nuclear magnetic resonance (1H NMR), with the aim of determining the content of MMA residue after the reaction time considered in this study (60 min). The residual monomer obtained from H NMR was calculated using the sum of the integrals corresponding to the monomers divided by the sum of the integrals of the values corresponding to the polymers and monomers [25].
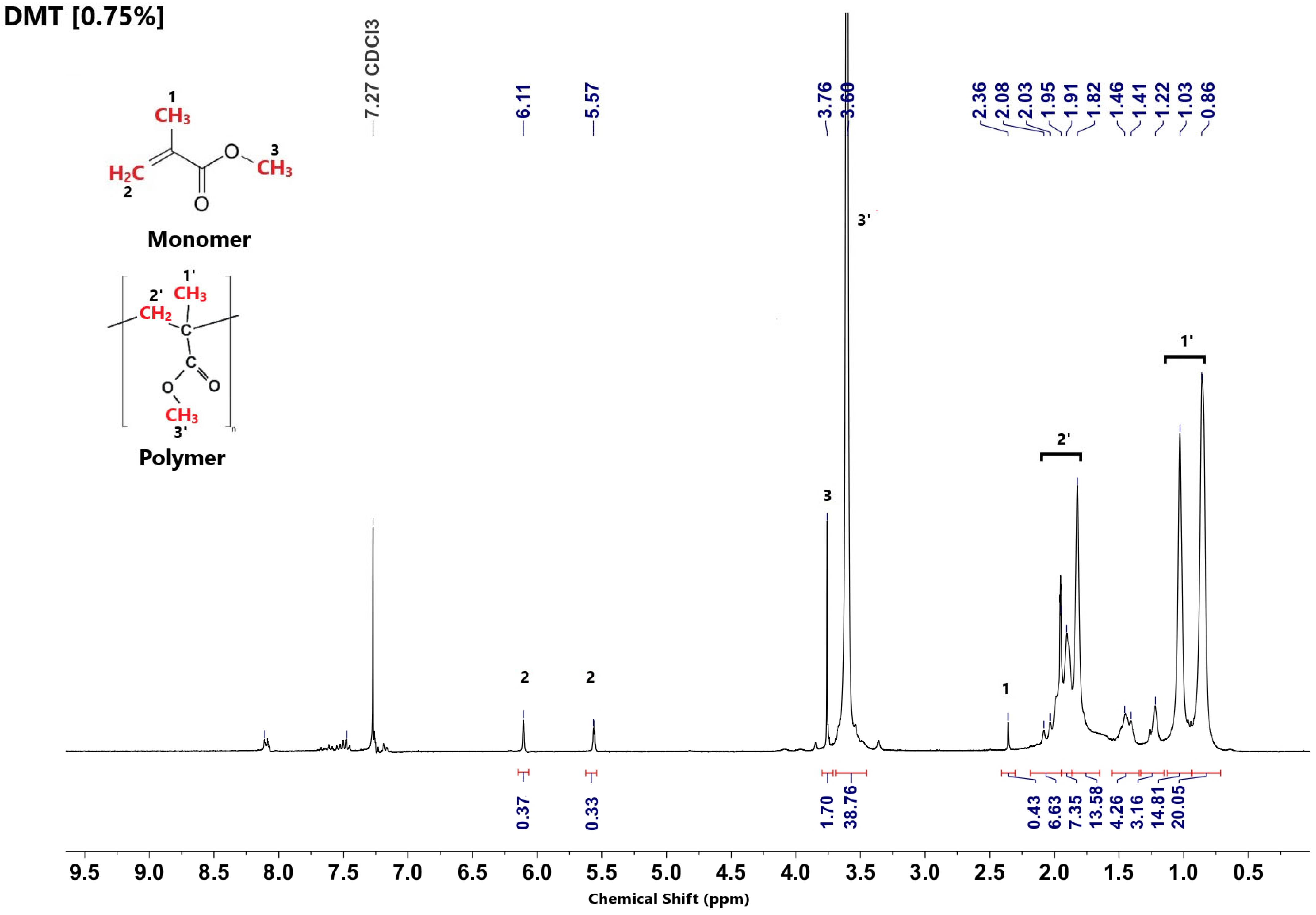
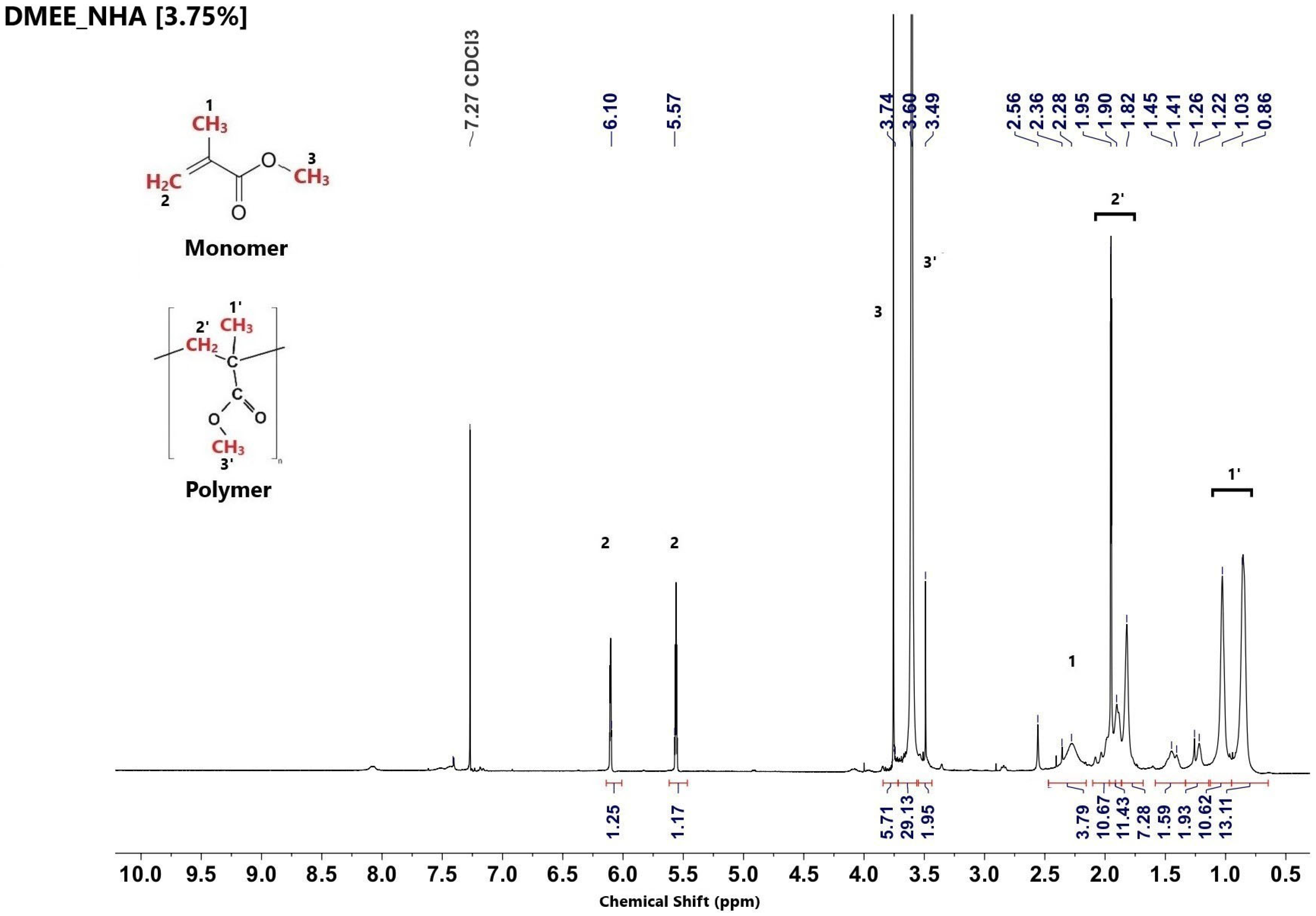
The signals at chemical shift δ = 2.20 ppm and δ = 3.78 ppm can be clearly attributed to the CH3 and O-CH3 groups of MMA, respectively. Those at δ = 5.58 ppm and δ = 6.10 ppm can be ascribed to the acrylic methylene (CH2) group. Furthermore, the signals in the range of δ = 0.82–1.20 ppm correspond to the resonance of the CH3 group of MMA units in the polymer chains, and the sharp and very intense signal at δ = 3.55 ppm is attributed to the O-CH3 group of methacrylate units incorporated into polymer chains. The ratio between the integrated intensities is directly related to the molar ratio between MMA and the number of MMA units incorporated in the polymer chains. The characteristic peak of the solvent (CHCl3) is located at δ = 7.27 ppm [25].
Figure 7 and Figure 8 show 1H NMR spectra for the bone cement synthesized from the standard amine and the aliphatic amine DMEE, respectively. Other results can be found in the Supplementary Materials (Supplementary Material Figures S9–S12). The residual monomer contents of these bone cements are available in Table 1 and Table 2. Regarding the polymers synthesized with the standard amine, the residual monomer content varied between 2.61 and 2.88 mol% with different amine concentrations (0.75 and 3.75 wt%). The polymers synthesized with the amine DMAB presented slightly higher residual monomer content when compared to the standard amine (DMT).
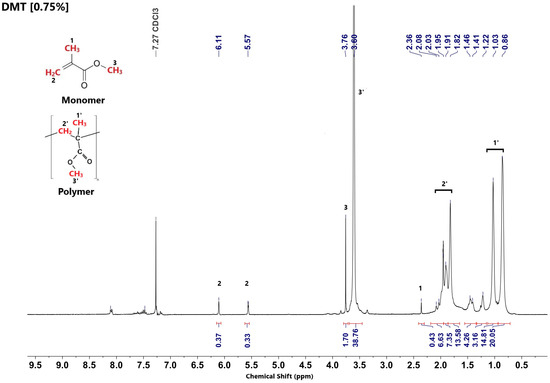
Figure 7.
1H NMR spectrum of bone cement prepared in the presence of the standard amine DMT.
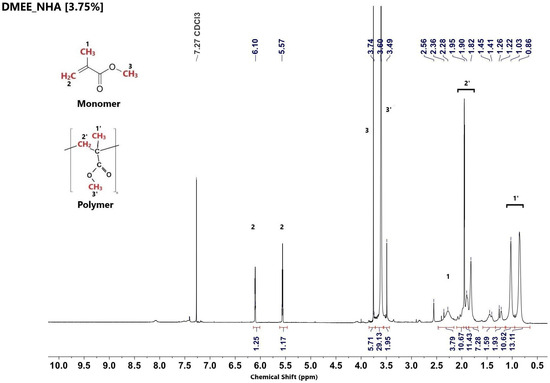
Figure 8.
1H NMR spectrum of nanocomposite prepared in the presence of the aliphatic amine DMEE and 3.75 wt% of hydroxyapatite.
At all concentrations, PMMA nanocomposites synthesized with the aliphatic amine DMEE presented a high content of unreacted monomer, when compared with nanocomposites synthesized from aromatic amines (DMT and DMAB). This can be attributed to their lower radical efficiency and to kinetic limitations during the polymerization process. In biomedical applications, residual monomers can interfere with the osseointegration process.
4. Conclusions
Standard polymerization reactions indicated that the higher the amine concentration in the composition, the faster the reaction occurs and the greater the heat release. The aromatic amine DMAB induces polymerization only at high concentrations (3.75 wt%). Nanocomposites synthesized with DMEE give rise to bone cements with lower glass transition temperatures, and lower initial and final degradation temperatures, when compared to the standard formulation (DMT). All PMMA-based bone cements also showed a molecular distribution of the polymer which exhibited only small variations compared to the standards. Microstructural analyses demonstrated that bone cements synthesized with the aliphatic amine DMEE presented a relevant percentage of residual monomers when compared to aromatic amines (DMT and DMAB). The inclusion of hydroxyapatite nanoparticles can accelerate polymerization reactions.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/pr14020213/s1. Figure S1: FTIR spectrum of nano-hydroxyapatite used in the preparation of PMMA nanocomposites bone cement.; Figure S2. The X-ray diffraction of nano-hydroxyapatite used in the preparation of PMMA nanocomposites bone cement.; Figure S3. Thermal profile of polymerizations of MMA co-initiated by the standard tertiary amine DMT at different concentrations in PMMA-based bone cement.; Figure S4. Thermal profile of polymerizations of MMA co-initiated by DMT and DMAB at different concentrations in PMMA-based bone cement.; Figure S5. FTIR spectra of PMMA-based bone cement: Monomer consumption in the polymerization of MMA using DMEE at different reaction times.; Figure S6. FTIR spectrum of MMA monomer.; Figure S7. Thermal profile of polymerizations of MMA co-initiated by DMT at different NanoHA concentrations in PMMA/nano-hydroxyapatite based bone cement.; Figure S8. Thermal profile of polymerizations of MMA co-initiated by DMAB at different nano-hydroxyapatite concentrations in PMMA/nano-hydroxyapatite based bone cement. Figure S9. 1H NMR spectrum of cement bone prepared with 3.75 wt% of DMT.; Figure S10. 1H NMR spectrum of cement bone prepared with 3.75 wt% of DMAB.; Figure S11. 1H NMR spectrum of cement bone prepared with 2.5 wt% of nano-hydroxyapatite and DMT.; Figure S12. 1H NMR spectrum of cement bone prepared with 2.5 wt% of nano-hydroxyapatite and DMAB.
Author Contributions
Conceptualization, M.L.D. and A.M.R.; Data curation, M.A.C.; Formal analysis, M.A.C. and A.M.R.; Investigation, M.A.C.; Resources, M.L.D.; Writing—original draft, M.A.C.; Writing—review and editing, M.L.D.; Supervision, M.L.D.; Project administration, M.L.D.; Funding acquisition, M.L.D. All authors have read and agreed to the published version of the manuscript.
Funding
Agencies: Conselho Nacional de Desenvolvimento Científico e Tecnológico—CNPq (Grants 307364/2018-6 and 310337/2023-2) and Fundação Carlos Chagas de Amparo à Pesquisa do Rio de Janeiro—FAPERJ (Grant 200.359/2023).
Data Availability Statement
The original contributions presented in this study are included in the article/Supplementary Materials. For more information, please contact the corresponding author.
Acknowledgments
The authors thank the following Brazilian agencies for their financial support: Conselho Nacional de Desenvolvimento Científico e Tecnológico—CNPq (Grants 307364/2018-6 and 310337/2023-2) and Fundação Carlos Chagas de Amparo à Pesquisa do Rio de Janeiro—FAPERJ (Grant 200.359/2023).
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Obaeed, H.; Hamdan, K. Reconstruction and evaluation of 3D Printing PMMA cranioplasty implants. Int. J. Interact. Des. Manuf. 2024, 18, 4233–4245. [Google Scholar] [CrossRef]
- Hamböck, M.; Hosmann, A.; Seemann, R.; Wolf, H.; Schachinger, F.; Hajdu, S.; Widhalm, W. The impact of implant material and patient age on the long-term outcome of secondary cranioplasty following decompressive craniectomy for severe traumatic brain injury. Acta Neurochir. 2020, 162, 745–753. [Google Scholar] [CrossRef]
- Giese, H.; Meyer, J.; Unterberg, A.; Beynon, C. Long-term complications and implant survival rates after cranioplastic surgery: A single-center study of 392 patients. Neurosurg. Rev. 2021, 44, 1755–1763. [Google Scholar] [CrossRef]
- Souza, L.S.; de Carvalho Santos, A.A.; Carvalho, J.; Bezerra, A.; Sales, A.; Robles, M.; Almeida, L.; Castro, M.; Freire, N.; Santos, B. Infectious risk of using biomaterials for cranioplasty: Polymethylmethacrylate vs. autologous bone. Res. Soc. Dev. 2022, 11, e0511830483. [Google Scholar] [CrossRef]
- Vijfeijken, S.; Münker, T.; Spijker, R.; Karssemakers, L.; Vandertop, W.; Becking, A.; Ubbink, D.T. Autologous bone is inferior to alloplastic cranioplasties: Safety of autograft and allograft materials for cranioplasties, a systematic review. World Neurosurg. 2018, 117, 443–452.e8. [Google Scholar] [CrossRef]
- Zoller, A.; Gigmes, D.; Guillaneuf, Y. A Simulation of radical polymerization of methyl methacrylate at room temperature using a tertiary amine/BPO initiating system. Polym. Chem. 2015, 6, 5719–5727. [Google Scholar] [CrossRef]
- Zoller, A.; Kockler, K.B.; Rollet, M.; Lefay, C.; Gigmes, D.; Barner-Kowollik, C.; Guillaneuf, Y. A complete kinetic study of a versatile functional monomer: Acetoacetoxyethyl methacrylate (AAEMA). Polym. Chem. 2016, 7, 5518–5525. [Google Scholar] [CrossRef]
- Golz, T.; Graham, C.; Busch, L.; Wulf, J.; Winder, R. Temperature elevation during simulated polymethylmethacrylate (PMMA) cranioplasty in a cadaver mode. J. Clin. Neurosci. 2010, 17, 617–622. [Google Scholar] [CrossRef] [PubMed]
- Dunnick, K.; Brix, A.; Sanders, M.; Travlos, S. N,N-Dimethyl-p-toluidine, a Component in Dental Materials, Causes Hematologic Toxic and Carcinogenic Responses in Rodent Model Systems. Toxicol. Pathol. 2014, 42, 603–615. [Google Scholar] [CrossRef] [PubMed]
- Las, E.; Verwilghen, D.; Mommaerts, M.Y. A systematic review of cranioplasty material toxicity in human subjects. J. Craniomaxillofacial Surg. 2021, 49, 34–46. [Google Scholar] [CrossRef]
- Lal, J.; Green, R. Effect of Amine Accelerators on the Polymerization of Methyl Methacrylate with Benzoyl Peroxide. J. Polym. Sci. 1955, 17, 403–409. [Google Scholar] [CrossRef]
- Brauer, G.; Steinberger, D.; Stansbury, J. Dependence of curing time, peak temperature and mechanical properties on the composition of bone cement. J. Biomed. Mater. Res. 1986, 20, 839–852. [Google Scholar] [CrossRef]
- Vazquez, B.; Elvira, C.; Levenfeld, B.; Pascual, B.; Goñi, I.; Gurruchaga, M.; Ginebra, M.P.; Gil, F.X.; Planell, J.A.; Liso, P.A.; et al. Application of tertiary amines with reduced toxicity to the curing process of acrylic bone cements. J. Biomed. Mater. Res. 1997, 34, 129–136. [Google Scholar] [CrossRef]
- Vázquez, B.; Deb, S.; Bonfield, W.; San Román, J. Characterization of new acrylic bone cements prepared with oleic acid derivatives. J. Biomed. Mater. Res. 2002, 63, 88–97. [Google Scholar] [CrossRef]
- Inami, C.; Shimizu, H.; Suzuki, S.; Haraguchi, N.; Itsuno, S. Study on the performance of methyl methacrylate polymerization: Comparison of partially oxidized tri-n-butylborane and benzoyl peroxide with aromatic tertiary amines. Dent. Mater. J. 2019, 38, 430–436. [Google Scholar] [CrossRef]
- Rybalko, V.; Pisarenko, E.; D’yachenko, P.; Timerkhanov, S.; Palamarchuk, A.; Korchmarek, A. The Influence of the Structure of Tertiary Amines on Low-Temperature Curing of a Highly Filled Acrylic Composite Initiated with Benzoyl Peroxide. Polym. Sci. 2019, 12, 231–235. [Google Scholar] [CrossRef]
- Russo, T.; De Santis, R.; Gloria, A.; Barbaro, K.; Altigeri, A.; Fadeeva, I.V.; Rau, J.V. Modification of PMMA Cements for Cranioplasty with Bioactive Glass and Copper Doped Tricalcium Phosphate Particles. Polymers 2020, 12, 37. [Google Scholar] [CrossRef]
- Zaszczyńska, A.; Kołbuk, D.; Gradys, A.; Sajkiewicz, P. Development of Poly(methyl methacrylate)/nano-hydroxyapatite (PMMA/nHA) Nanofibers for Tissue Engineering Regeneration Using an Electrospinning Technique. Polymers 2024, 16, 531. [Google Scholar] [CrossRef] [PubMed]
- Pedrosa Dos Anjos, S.; Mavropoulos, E.; Bernardo, P.; Granjeiro, J.; Rossi, A.; Dias, M. Structure and biological compatibility of Polycaprolactone/zinc-hydroxyapatite electrospun nanofibers for tissue regeneration. J. Bioact. Compat. Polym. 2021, 36, 314–333. [Google Scholar] [CrossRef]
- Leal, C.; Beserra, V.; Silva Filho, C.; Menezes, L.; Tabchoury, M.; Vale, C. Development of an Experimental Dentifrice with Hydroxyapatite Nanoparticles and High Fluoride Concentration to Manage Root Dentin Demineralization. Int. J. Nanomed. 2020, 15, 7469–7479. [Google Scholar] [CrossRef]
- Lett, J.A.; Sagadevan, S.; Prabhakar, J.J.; Hamizi, N.A.; Badruddin, I.A.; Johan, M.R.; Marlinda, A.R.; Abdul Wahab, Y.; Yunus Khan, T.M.; Kamangar, S. Drug Leaching Properties of Vancomycin Loaded Mesoporous Hydroxyapatite as Bone Substitutes. Processes 2019, 7, 826. [Google Scholar] [CrossRef]
- Aschilias, D.S.; Sideridou, I.D. Study of the effect of two bpo/amine initiation systems on the free-radical polymerization of mma used in dental resins and bone cements. J. Macromol. Sci. Part A Pure Appl. Chem. 2007, 39, 1435–1450. [Google Scholar] [CrossRef]
- Vallo, C.I.; Montemartini, P.E.; Fanovich, M.A.; López, J.M.P.; Cuadrado, T.R. Polymethylmethacrylate-based bone cement modified with hydroxyapatite. J. Biomed. Mater. Res. 1999, 48, 150–158. [Google Scholar] [CrossRef]
- Santos Pita, J.; Melo, P.; Nele, M.; Pinto, J. Effect of process variables on the preparation of artificial bone cements. Braz. J. Chem. Eng. 2012, 30, 865–876. [Google Scholar] [CrossRef]
- Pascual, B.; Vtiquez, B.; Gurruchaga, M.; Goni, I.; Ginebra, M.P.; Gil, F.; Planell, J.; Levenfeld, B.; San Romon, J. New aspects of the effect of size and size distribution on the setting parameters and mechanical properties of acrylic bone cements. Biomaterials 1996, 17, 509–516. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.