XGBoost and Artificial Neural Networks as Surrogate Models for Vapor–Liquid Equilibrium in PC-SAFT
Abstract
1. Introduction
2. Method
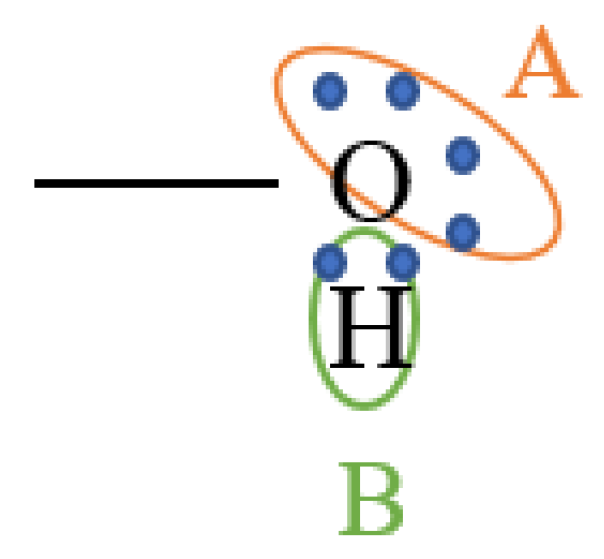
2.1. PC-SAFT
2.2. XGBoost
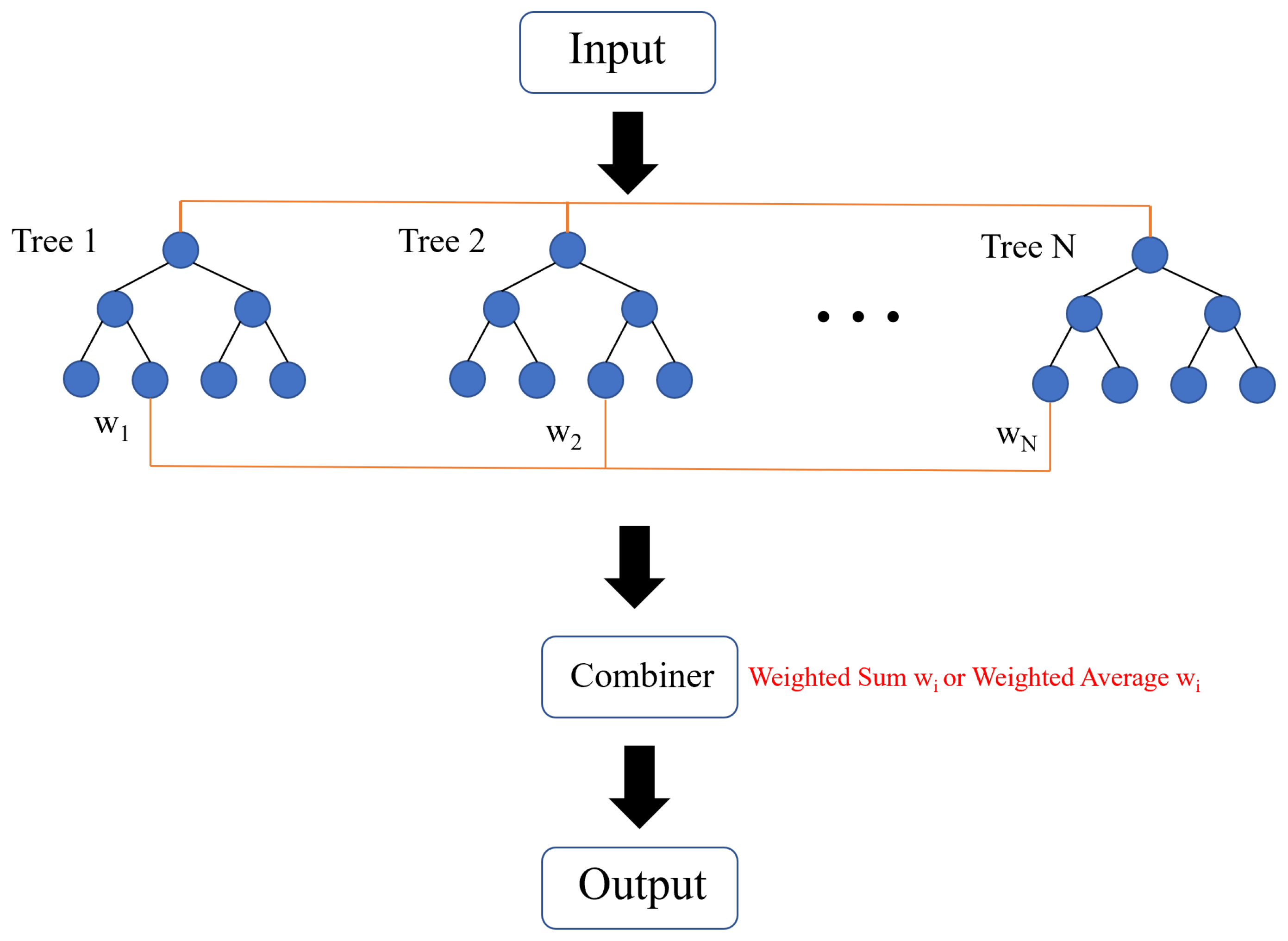
2.2.1. Tree Ensemble Model
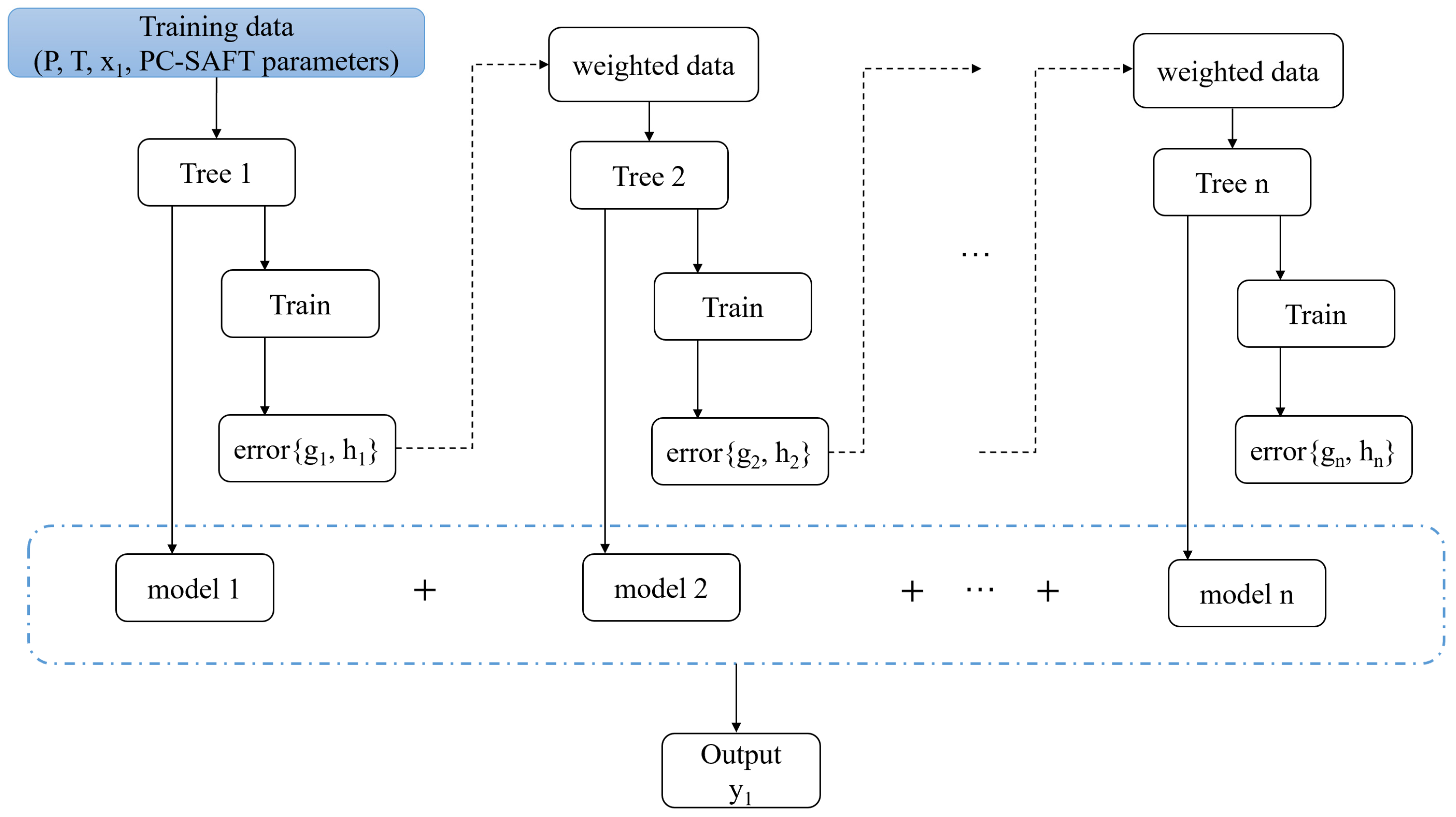
2.2.2. Gradient Tree Boosting
2.3. Artificial Neural Networks
2.4. XGBoost-ANN
2.5. Thermodynamic Consistency
3. Results and Discussion
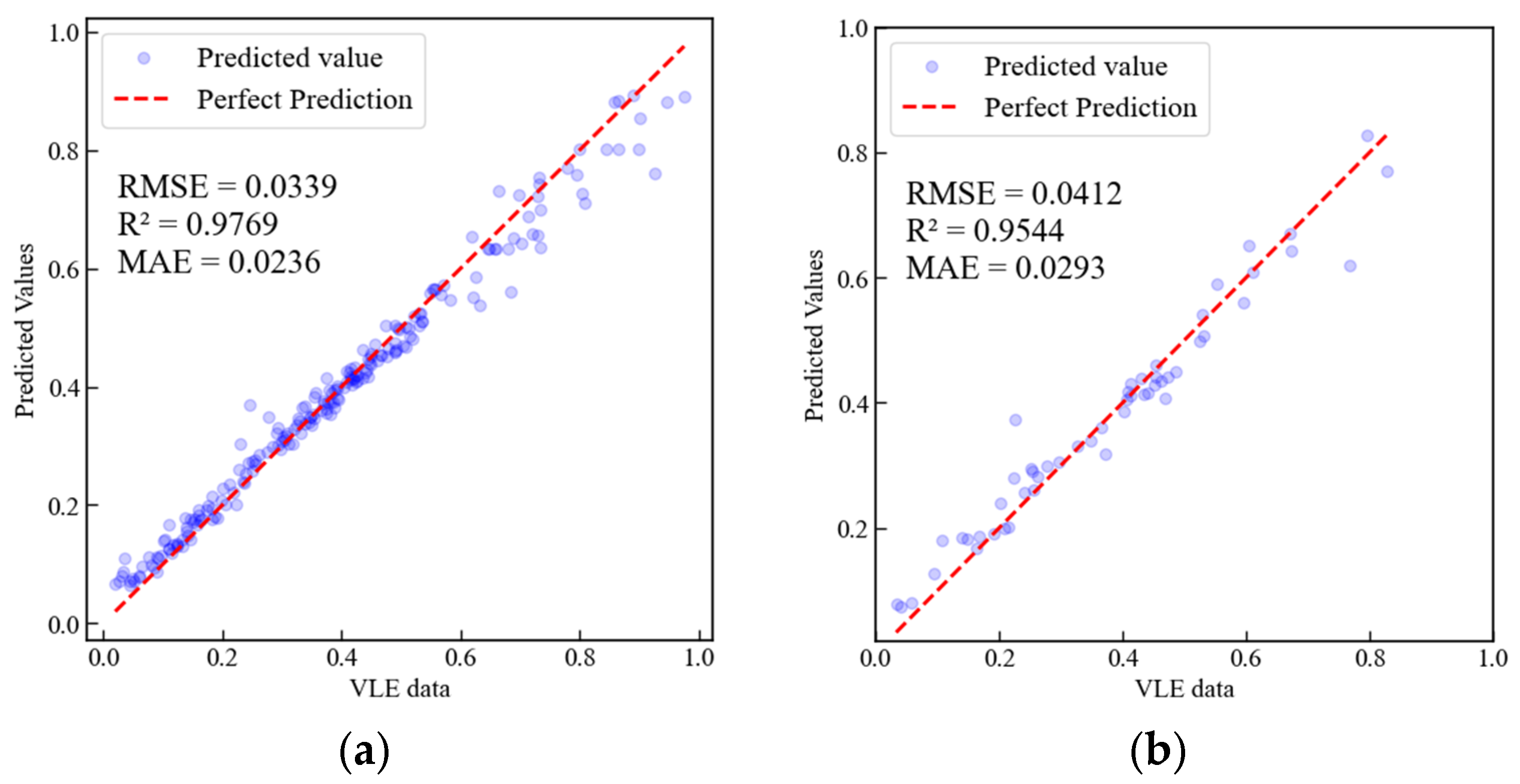
3.1. Water + Methanol
3.2. Water + Ethanol
3.3. Water + 1-Propanol
3.4. Water + 2-Propanol
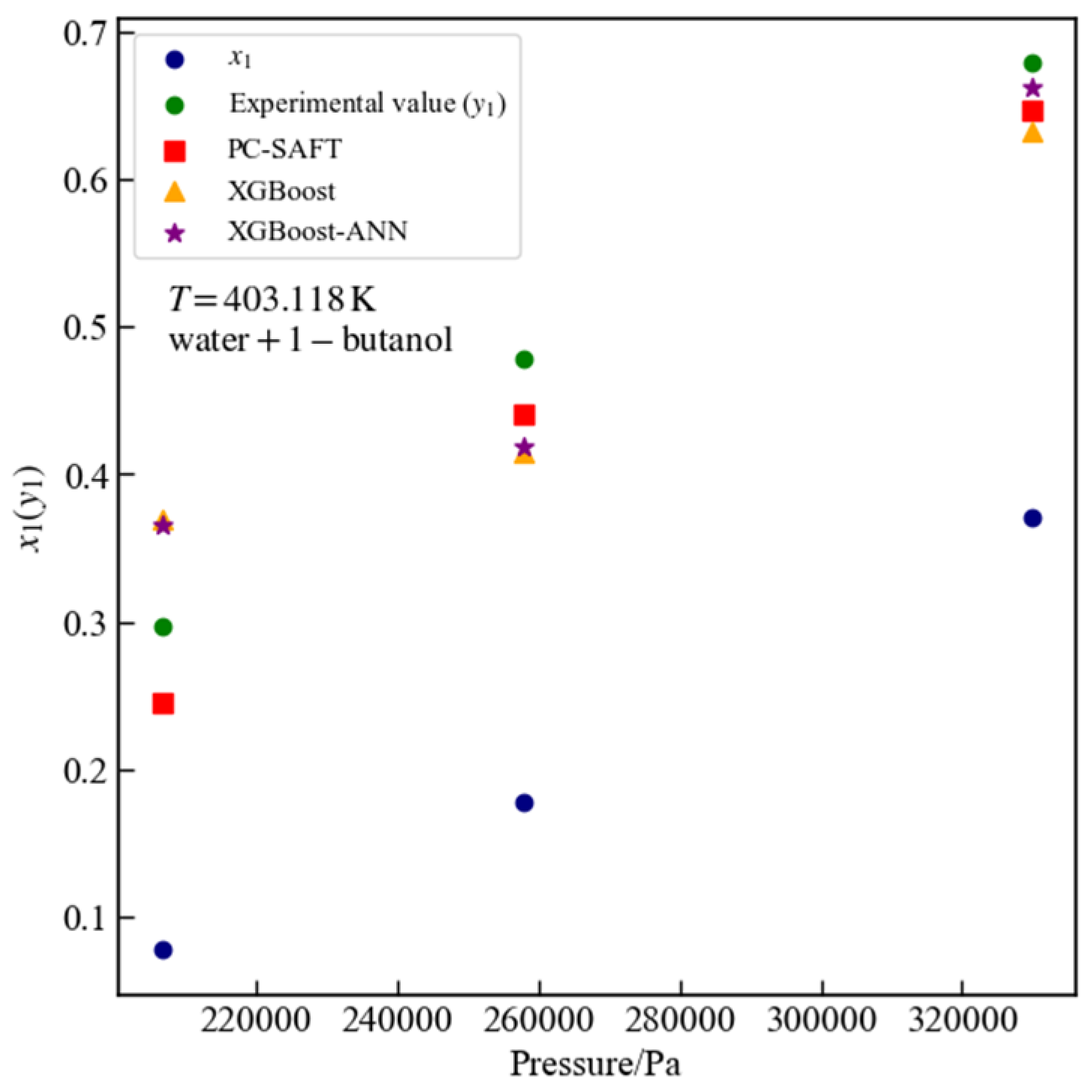
3.5. Water + 1-Butanol
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Nomenclature
| %AAD | average absolute percentage deviation |
| the association contribution term | |
| the dispersion contribution term | |
| the hard-chain reference contribution term | |
| ANN | Artificial Neural Network |
| Ap | pressure integral term |
| the integral of the fugacity coefficient | |
| the residual Helmholtz energy | |
| EoS | Equation of State |
| ft | additional perturbation function |
| the fugacity of the component i | |
| kB | Boltzmann constant |
| kij | binary interaction parameters |
| L | the objective function of XGBoost |
| LeavesN | the number of leaves in the tree |
| MAE | mean absolute error |
| mi | the number of segments |
| MSE | mean square error |
| NAi, NBi | the number of association sites |
| nc | the number of components |
| P | pressure |
| PINN | Physics-Informed Neural Network |
| RMSE | root mean square error |
| T | temperature |
| VE | reduced molar volume |
| VLE | vapor–liquid equilibrium |
| wi | the score associated with the ith leaf |
| yi | true value |
| predict value | |
| Z | compression factor |
| segment dimeter | |
| depth of pair potential, K | |
| associative volume | |
| associative energy | |
| penalty weight | |
| the residual chemical potential | |
| the chemical potential of component i | |
| the fugacity coefficient of the component i | |
| packing fraction | |
| penalize function | |
| penalization parameters | |
| activity coefficient of component i |
References
- Gross, J.; Sadowski, G. Perturbed-Chain SAFT: An Equation of State Based on a Perturbation Theory for Chain Molecules. Ind. Eng. Chem. Res. 2001, 40, 1244–1260. [Google Scholar] [CrossRef]
- Soave, G. Equilibrium constants from a modified Redlich-Kwong equation of state. Chem. Eng. Sci. 1972, 27, 1197–1203. [Google Scholar] [CrossRef]
- Peng, D.; Robinson, D.B. A rigorous method for predicting the critical properties of multicomponent systems from an equation of state. AIChE J. 1977, 23, 137–144. [Google Scholar] [CrossRef]
- McBride, K.; Sundmacher, K. Overview of Surrogate Modeling in Chemical Process Engineering. Chem. Ing. Tech. 2019, 91, 228–239. [Google Scholar] [CrossRef]
- Winz, J.; Engell, S. Data-efficient surrogate modeling of thermodynamic equilibria using Sobolev training, data augmentation and adaptive sampling. Chem. Eng. Sci. 2024, 299, 120461. [Google Scholar] [CrossRef]
- Rittig, J.G. Gibbs–Duhem-informed neural networks for binary activity coefficient prediction. Digit. Discov. 2023, 2, 1752–1767. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, T.; Sun, S.; Gao, X. Accelerating flash calculation through deep learning methods. J. Comput. Phys. 2019, 394, 153–165. [Google Scholar] [CrossRef]
- Ottaiano, G.Y.; Martins, T.D. Machine learning models for vapor-liquid equilibrium of binary mixtures: State of the art and future opportunities. Chem. Eng. Res. Des. 2024, 211, 66–77. [Google Scholar] [CrossRef]
- Wang, S.; Sobecki, N.; Ding, D.; Zhu, L.; Wu, Y.-S. Accelerating and stabilizing the vapor-liquid equilibrium (VLE) calculation in compositional simulation of unconventional reservoirs using deep learning based flash calculation. Fuel 2019, 253, 209–219. [Google Scholar] [CrossRef]
- Sun, J.; Xue, J.; Yang, G.; Li, J.; Zhang, W. Vapor–liquid phase equilibrium prediction for mixtures of binary systems using graph neural networks. AIChE J. 2025, 71, e18637. [Google Scholar] [CrossRef]
- Ihunde, T.A.; Olorode, O. Application of physics-informed neural networks to compositional modeling. J. Pet. Sci. Eng. 2022, 211, 110175. [Google Scholar] [CrossRef]
- Raissi, M.; Perdikaris, P.; Karniadakis, G.E. Physics-informed neural networks: A deep learning framework for solving forward and inverse problems involving nonlinear partial differential equations. J. Comput. Phys. 2019, 378, 686–707. [Google Scholar] [CrossRef]
- Xiang, Z.; Peng, W.; Liu, X.; Yao, W. Self-adaptive loss balanced Physics-informed neural networks. Neurocomputing 2022, 496, 11–34. [Google Scholar] [CrossRef]
- Li, S.; Feng, X. Dynamic Weight Strategy of Physics-Informed Neural Networks for the 2D Navier–Stokes Equations. Entropy 2022, 24, 1254. [Google Scholar] [CrossRef]
- Difonzo, F.V.; Lopez, L.; Pellegrino, S.F. Physics informed neural networks for an inverse problem in peridynamic models. Eng. Comput. 2024, 5, 1–10. [Google Scholar] [CrossRef]
- Hakim, M.; Behmardikalantari, G.; Abedini Najafabadi, H.; Pazuki, G.; Vosoughi, A.; Vossoughi, M. Prediction of liquid–liquid equilibrium behavior for aliphatic+aromatic+ionic liquid using two different neural network-based models. Fluid Phase Equilibria 2015, 394, 140–147. [Google Scholar] [CrossRef]
- Petersen, R.; Fredenslund, A.; Rasmussen, P. Artificial neural networks as a predictive tool for vapor-liquid equilibrium. Comput. Chem. Eng. 1994, 18, S63–S67. [Google Scholar] [CrossRef]
- Ghanadzadeh, H.; Ahmadifar, H. Estimation of (vapour+liquid) equilibrium of binary systems (tert-butanol+2-ethyl-1-hexanol) and (n-butanol+2-ethyl-1-hexanol) using an artificial neural network. J. Chem. Thermodyn. 2008, 40, 1152–1156. [Google Scholar] [CrossRef]
- Urata, S.; Takada, A.; Murata, J.; Hiaki, T.; Sekiya, A. Prediction of vapor–liquid equilibrium for binary systems containing HFEs by using artificial neural network. Fluid Phase Equilibria 2002, 199, 63–78. [Google Scholar] [CrossRef]
- Yamamoto, H.; Tochigi, K. Prediction of vapor–liquid equilibria using reconstruction—Learning neural network method. Fluid Phase Equilibria 2007, 257, 169–172. [Google Scholar] [CrossRef]
- Reyhani, S.Z.; Ghanadzadeh, H.; Puigjaner, L.; Recances, F. Estimation of Liquid−Liquid Equilibrium for a Quaternary System Using the GMDH Algorithm. Ind. Eng. Chem. Res. 2009, 48, 2129–2134. [Google Scholar] [CrossRef]
- Moghadam, M.; Asgharzadeh, S. On the application of artificial neural network for modeling liquid-liquid equilibrium. J. Mol. Liq. 2016, 220, 339–345. [Google Scholar] [CrossRef]
- Chen, T.; Guestrin, C. XGBoost: A Scalable Tree Boosting System. In Proceedings of the KDD’16: The 22nd ACM SIGKDD International Conference on Knowledge Discovery and Data Mining, San Francisco, CA, USA, 13–17 August 2016; pp. 785–794. [Google Scholar] [CrossRef]
- Rehner, P.; Bauer, G.; Gross, J. FeOs: An Open-Source Framework for Equations of State and Classical Density Functional Theory. Ind. Eng. Chem. Res. 2023, 62, 5347–5357. [Google Scholar] [CrossRef]
- Kontogeorgis, G.M. Thermodynamic Models for Industrial Applications: From Classical and Advanced Mixing Rules to Association theories; John Wiley & Sons: Hoboken, NJ, USA, 2009. [Google Scholar]
- Paszke, A.; Gross, S.; Massa, F.; Lerer, A.; Bradbury, J.; Chanan, G.; Killeen, T.; Lin, Z.; Gimelshein, N.; Antiga, L.; et al. PyTorch: An Imperative Style, High-Performance Deep Learning Library. arXiv 2019, arXiv:1912.01703. [Google Scholar] [CrossRef]
- Arce, P.F.; Vieira, N.F.; Igarashi, E.M.S. Thermodynamic Modeling and Simulation of Biodiesel Systems at Supercritical Conditions. Ind. Eng. Chem. Res. 2018, 57, 751–767. [Google Scholar] [CrossRef]
- Smith, J.M.; Van Ness, H.C.; Abbott, M.M. Introduction to Chemical Engineering Thermodynamics, 6th ed.; McGraw-Hill: New York, NY, USA, 2014. [Google Scholar]
- Valderrama, J.O.; Zavaleta, J. Thermodynamic consistency test for high pressure gas–solid solubility data of binary mixtures using genetic algorithms. J. Supercrit. Fluids 2006, 39, 20–29. [Google Scholar] [CrossRef]
- Saali, A.; Shokouhi, M.; Sakhaeinia, H.; Kazemi, N. Thermodynamic Consistency Test of Vapor–liquid Equilibrium Data of Binary Systems Including Carbon Dioxide (CO2) and Ionic Liquids Using the Generic Redlich–Kwong Equation of State. J. Solut. Chem. 2020, 49, 383–404. [Google Scholar] [CrossRef]
- Alvarez, V.H.; Aznar, M. Application of a Thermodynamic Consistency Test to Binary Mixtures Containing an Ionic Liquid. Open Thermodyn. J. 2008, 2, 25–38. [Google Scholar] [CrossRef][Green Version]
- Pedregosa, F.; Varoquaux, G.; Gramfort, A.; Michel, V.; Thirion, B.; Grisel, O.; Blondel, M.; Prettenhofer, P.; Weiss, R.; Dubourg, V.; et al. Scikit-learn: Machine Learning in Python. J. Mach. Learn. Res. 2011, 12, 2825–2830. [Google Scholar]
- Akiba, T.; Sano, S.; Yanase, T.; Ohta, T.; Koyama, M. Optuna: A Next-generation Hyperparameter Optimization Framework. arXiv 2019, arXiv:1907.10902. [Google Scholar] [CrossRef]
- Gross, J.; Sadowski, G. Application of the perturbed-chain SAFT equation of state to associating systems. Ind. Eng. Chem. Res. 2002, 41, 5510–5515. [Google Scholar] [CrossRef]
- Huang, S.H.; Radosz, M. Equation of state for small, large, polydisperse, and associating molecules. Ind. Eng. Chem. Res. 1990, 29, 2284–2294. [Google Scholar] [CrossRef]
- Hall, D.J.; Mash, C.J.; Pemberton, R.C. NPL Report Chemistry; National Physical Laboratory: Middlesex, UK, 1979.
- McGlashan, M.L.; Williamson, A.G. Isothermal Liquid-Vapor Equilibriums for System Methanol-Water. J. Chem. Eng. Data 1976, 21, 196–199. [Google Scholar] [CrossRef]
- Ferguson, J.B.; Funnel, W.S. The Determination of Vapor and Liquid Compositions in Binary Systems. I. Methyl Alcohol-Water. J. Phys. Chem. 1929, 33, 1–8. [Google Scholar] [CrossRef]
- Yao, J.; Li, H.; Han, S. Vapor–Liquid Equilibrium Data for Methanol–Water–NaCl at 45 °C. Fluid Phase Equilibria 1999, 4, 15. [Google Scholar] [CrossRef]
- Fu, J.; Wang, K.; Hu, Y. Studies on vapor-liquid and liquid-liquidvapor equilibria for the ternary system methanol-methyl methacrylatewater (Ⅱ) ternary system. Chin. J. Chem. Eng. 1989, 4, 14. [Google Scholar]
- Broul, M.; Hlavaty, K.; Linek, J. Liquid-Vapour Equilibrium in Systems of Electrolytic Components: V the System Methanol + Water + Lithium Chloride at 60 c. Collect Czech Chem. Commun. 1969, 34, 28–34. [Google Scholar] [CrossRef]
- Schuberth, H. Schuberth H. Influence of Simple Salts on the Isothermal Vapor-Liquid Equilibrium Behavior of the Methanol-Water System. Z. Fuer Phys. Chem. 1974, 255, 165–179. [Google Scholar] [CrossRef]
- Bao, Z.; Liu, M.; Yang, J. Measurement and Correlation of Moderate Pressure Vapor-Liquid Equilibrium Data for Methanol-Water Binary System. J. Chem. Ind. Eng. China 1995, 46, 230. [Google Scholar]
- Griswold, J. Phase-Equilibria of the Acetone-Methanol-Water System from 100℃ into the Critical Region. Chem. Eng. Progr., Symp. Syr. 1952, 48, 18–34. [Google Scholar]
- Schröder, W. Messungen von Siedegleichgewichten Bei Überdruck. Chem. Ing. Tech. 1958, 30, 523–525. [Google Scholar] [CrossRef]
- Phutela, R.; Kooner, Z.; Fenby, D. Vapour Pressure Study of Deuterium Exchange Reactions in Water-Ethanol Systems: Equilibrium Constant Determination. Aust. J. Chem. 1979, 32, 2353. [Google Scholar] [CrossRef]
- Yamamoto, H.; Terano, T.; Nishi, Y.; Tokunaga, J. Vapor-Liquid Equilibria for Methanol + Ethanol + Calcium Chloride, + Ammonium Iodide, and + Sodium Iodide at 298.15 K. J. Chem. Eng. Data 1995, 40, 472–477. [Google Scholar] [CrossRef]
- Herraiz, J.; Shen, S.; Coronas, A. Vapor−Liquid Equilibria for Methanol + Poly(Ethylene Glycol) 250 Dimethyl Ether. J. Chem. Eng. Data 1998, 43, 191–195. [Google Scholar] [CrossRef]
- Vu, D.T.; Lira, C.T.; Asthana, N.S.; Kolah, A.K.; Miller, D.J. Vapor−Liquid Equilibria in the Systems Ethyl Lactate + Ethanol and Ethyl Lactate + Water. J. Chem. Eng. Data 2006, 51, 1220–1225. [Google Scholar] [CrossRef]
- Kurihara, K.; Minoura, T.; Takeda, K.; Kojima, K. Isothermal Vapor-Liquid Equilibria for Methanol + Ethanol + Water, Methanol + Water, and Ethanol + Water. J. Chem. Eng. Data 1995, 40, 679–684. [Google Scholar] [CrossRef]
- Kang, J.W.; Diky, V.; Chirico, R.D.; Magee, J.W.; Muzny, C.D.; Abdulagatov, I.; Kazakov, A.F.; Frenkel, M. Quality Assessment Algorithm for Vapor−Liquid Equilibrium Data. J. Chem. Eng. Data 2010, 55, 3631–3640. [Google Scholar] [CrossRef]
- Schuberth, H. Isothermic behavior of phase-equilibrium in the system N-alcohol-water-urea at 60-degrees Celsius. Z. Phys. Chem.-Leipz. 1980, 261, 777–790. [Google Scholar] [CrossRef]
- Arango, I.C.; Villa, A.L. Isothermal Vapor–Liquid and Vapor–Liquid–Liquid Equilibrium for the Ternary System Ethanol+water+diethyl Carbonate and Constituent Binary Systems at Different Temperatures. Fluid Phase Equilibria 2013, 339, 31–39. [Google Scholar] [CrossRef]
- Cristino, A.F.; Rosa, S.; Morgado, P.; Galindo, A.; Filipe, E.J.M.; Palavra, A.M.F.; Nieto De Castro, C.A. High-Temperature Vapour–Liquid Equilibrium for the Water–Alcohol Systems and Modeling with SAFT-VR: 1. Water–Ethanol. Fluid Phase Equilibria 2013, 341, 48–53. [Google Scholar] [CrossRef]
- Niesen, V.; Palavra, A.; Kidnay, A.J.; Yesavage, V.F. An Apparatus for Vapor—Liquid Equilibrium at Elevated Temperatures and Pressures and Selected Results for the Water—Ethanol and Methanol—Ethanol Systems. Fluid Phase Equilibria 1986, 31, 283–298. [Google Scholar] [CrossRef]
- Barr-David, F.; Dodge, B.F. Vapor-Liquid Equilibrium at High Pressures. The Systems Ethanol-Water and 2-Propanol-Water. J. Chem. Eng. Data 1959, 4, 107–121. [Google Scholar] [CrossRef]
- Murti, P.; Van Winkle, M. Vapor-Liquid Equilibria for Binary Systems of Methanol, Ethyl Alcohol, 1-Propanol, and 2-Propanol with Ethyl Acetate and 1-Propanol-Water. Ind. Eng. Chem. Chem. Eng. Data Ser. 1958, 3, 72–81. [Google Scholar] [CrossRef]
- Schreiber, E.; Schüttau, E.; Rant, D.; Schuberth, H. Die Beeinflußbarkeit des isothermen Phasengleichgewichtsverhaltens der Systeme 11-Propanol/Wasser und n-Butanol/Wasser durch einige Metallchloride. Z. Für Phys. Chem. 1971, 247, 23–40. [Google Scholar] [CrossRef]
- Woerpel, U.; Vohland, P.; Schuberth, H. The Effect of Urea on the Vapor-Liquid Equilibrium Behavior of n-Propanol/Water at 60 °C. Z. Phys. Chem. 1977, 258, 905–912. [Google Scholar]
- Wrewsky, M.Z. Composition and Vapor Pressure of Binary Mixtures. Phys. Chem. Stoechiom. Verwandschaftsl. 1913, 81, 1–29. [Google Scholar]
- Ratcliff, G.A.; Chao, K.C. Prediction of Thermodynamic Properties of Polar Mixtures by a Group Solution Model. Can. J. Chem. Eng. 1969, 47, 148–153. [Google Scholar] [CrossRef]
- Udovenko, V.V.; Mazanko, T.F. Liquid-Vapour Equilibrium in Propan-2-Ol-Water and Propan-2-Ol-Benzene Systems. Russ. J. Phys. Chem. USSR. 1967, 41, 863. [Google Scholar]
- Wu, H.S.; Hagewiesche, D.; Sandler, S.I. Vapor—Liquid Equilibria of 2-Propanol + Water + N,N-Dimethyl Formamide. Fluid Phase Equilibria 1988, 43, 77–89. [Google Scholar] [CrossRef]
- Lyzlova, R.V.; Zaiko, L.N.; Susarev, M.P. Experimental Investigation and Calculation of Liquid-Vapor Equilibrium in the Ternary System n-Butyl Alcohol + Isobutyl Alcohol + Water at 35 c. Zhurnal Prikl. Khimii 1979, 52, 551–555. [Google Scholar]
- Kharin, S.E.; Perelygin, V.M. Liquid-Vapor Phase Equilibrium in Ethanol-n-Butanol and Water-n-Butanol Systems. Izv. Vyssh. Ucheb. Zaved Khim. Khim. Tekhnol. 1969, 12, 424–428. [Google Scholar]








| Hyperparameter | The Range of Hyperparameters | Parameter Usage |
|---|---|---|
| n_estimators | [60, 400] | The number of decision trees |
| max_depth | [3, 10] | The maximum depth of each decision tree |
| learning_rate | [0.01, 0.06] | Learning rate |
| subsample | [0.5, 0.9] | The fraction of training data randomly sampled for training each tree |
| colsample_bytree | [0.6, 0.9] | The fraction of features randomly sampled for constructing each tree |
| colsample_bylevel | [0.7, 0.9] | The fraction of features sampled at each split within a tree |
| colsample_bynode | [0.7, 0.9] | The fraction of features sampled for each node split in a tree |
| reg_alpha | [0.1, 20] | The L1 regularization term on weights |
| reg_lambda | [0.1, 30] | The L2 regularization term on weights |
| min_child_weight | [2, 10] | The minimum sum of instance weights needed in a child node |
| gamma | [0.01, 1.0] | The minimum loss reduction required to make a split on a leaf node of the tree |
| max_delta_step | [1, 5] | The maximum weight change for each tree’s output |
| Hyperparameter | The Optimized Value |
|---|---|
| n_estimators | 81 |
| max_depth | 6 |
| learning_rate | 0.0592 |
| subsample | 0.7099 |
| colsample_bytree | 0.8816 |
| colsample_bylevel | 0.7113 |
| colsample_bynode | 0.8444 |
| reg_alpha | 0.1038 |
| reg_lambda | 0.1141 |
| min_child_weight | 3 |
| gamma | 0.0100 |
| max_delta_step | 1 |
| Hyperparameter | The Optimized Value |
|---|---|
| The number of layers | 4 |
| The number of neurons per layer | {64,52,60,8} |
| Batch size | 16 |
| 1023.1298 |
| Model | Computation Time/s |
|---|---|
| PC-SAFT | 2.9185 |
| XGBoost | 0.0010 |
| XGBoost-ANN | 0.0030 |
| Temperature/K | XGBoost | XGBoost-ANN | ||
|---|---|---|---|---|
| MSE | MSE | |||
| 298.144 | 0.0001 | 0.9929 | 0.0002 | 0.9884 |
| 318.000 | 0.0002 | 0.9832 | 2.3384 × 10−5 | 0.9979 |
| 328.136 | 0.0002 | 0.9918 | 0.0003 | 0.9867 |
| 373.124 | 0.0044 | 0.9328 | 0.0025 | 0.9617 |
| 388.150 | 0.0001 | 0.9979 | 0.0003 | 0.9885 |
| 403.150 | 0.0028 | 0.9563 | 0.0017 | 0.9727 |
| Temperature/K | PC-SAFT | XGBoost | XGBoost-ANN | ||
|---|---|---|---|---|---|
| %AAD | %AAD | %AAD | |||
| 298.144 | 4.9033 | 8.3390 | 3.4357 | 6.184 | 1.4151 |
| 318.000 | 6.6105 | 11.2727 | 4.6622 | 6.4610 | 0.1495 |
| 328.136 | 5.3121 | 8.4368 | 3.1247 | 3.7656 | 1.5464 |
| 373.124 | 2.1875 | 6.4645 | 4.2773 | 5.9824 | 3.7952 |
| 388.150 | 1.6987 | 2.6912 | 0.9925 | 2.7253 | 1.0266 |
| 403.150 | 5.1908 | 7.2112 | 2.2004 | 9.6721 | 4.4814 |
| Temperature/K | XGBoost | XGBoost-ANN | ||
|---|---|---|---|---|
| MSE | MSE | |||
| 323.150 | 0.0003 | 0.9658 | 0.0001 | 0.9873 |
| 328.150 | 0.0006 | 0.9519 | 0.0003 | 0.9732 |
| 333.150 | 0.0002 | 0.9510 | 0.0001 | 0.9640 |
| 423.700 | 0.0007 | 0.9703 | 0.0023 | 0.9045 |
| Temperature/K | PC-SAFT | XGBoost | XGBoost-ANN | ||
|---|---|---|---|---|---|
| %AAD | %AAD | %AAD | |||
| 323.150 | 7.0501 | 7.7871 | 0.7380 | 5.4539 | 1.5962 |
| 328.150 | 6.0784 | 9.9871 | 3.9087 | 5.6462 | 0.4322 |
| 333.150 | 9.0990 | 9.2426 | 0.1437 | 7.2375 | 1.8614 |
| 423.700 | 1.4961 | 4.4587 | 2.9626 | 7.6918 | 6.1957 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pang, Y.; Ding, Z.; Li, Q. XGBoost and Artificial Neural Networks as Surrogate Models for Vapor–Liquid Equilibrium in PC-SAFT. Processes 2025, 13, 3918. https://doi.org/10.3390/pr13123918
Pang Y, Ding Z, Li Q. XGBoost and Artificial Neural Networks as Surrogate Models for Vapor–Liquid Equilibrium in PC-SAFT. Processes. 2025; 13(12):3918. https://doi.org/10.3390/pr13123918
Chicago/Turabian StylePang, Yiwen, Zhongwei Ding, and Qunsheng Li. 2025. "XGBoost and Artificial Neural Networks as Surrogate Models for Vapor–Liquid Equilibrium in PC-SAFT" Processes 13, no. 12: 3918. https://doi.org/10.3390/pr13123918
APA StylePang, Y., Ding, Z., & Li, Q. (2025). XGBoost and Artificial Neural Networks as Surrogate Models for Vapor–Liquid Equilibrium in PC-SAFT. Processes, 13(12), 3918. https://doi.org/10.3390/pr13123918





