Simulation and Validation of Green Hydrogen for the Production of Renewable Diesel: Case Study in La Guajira, Colombia
Abstract
1. Introduction
2. Methodology
2.1. Equipment Recognition
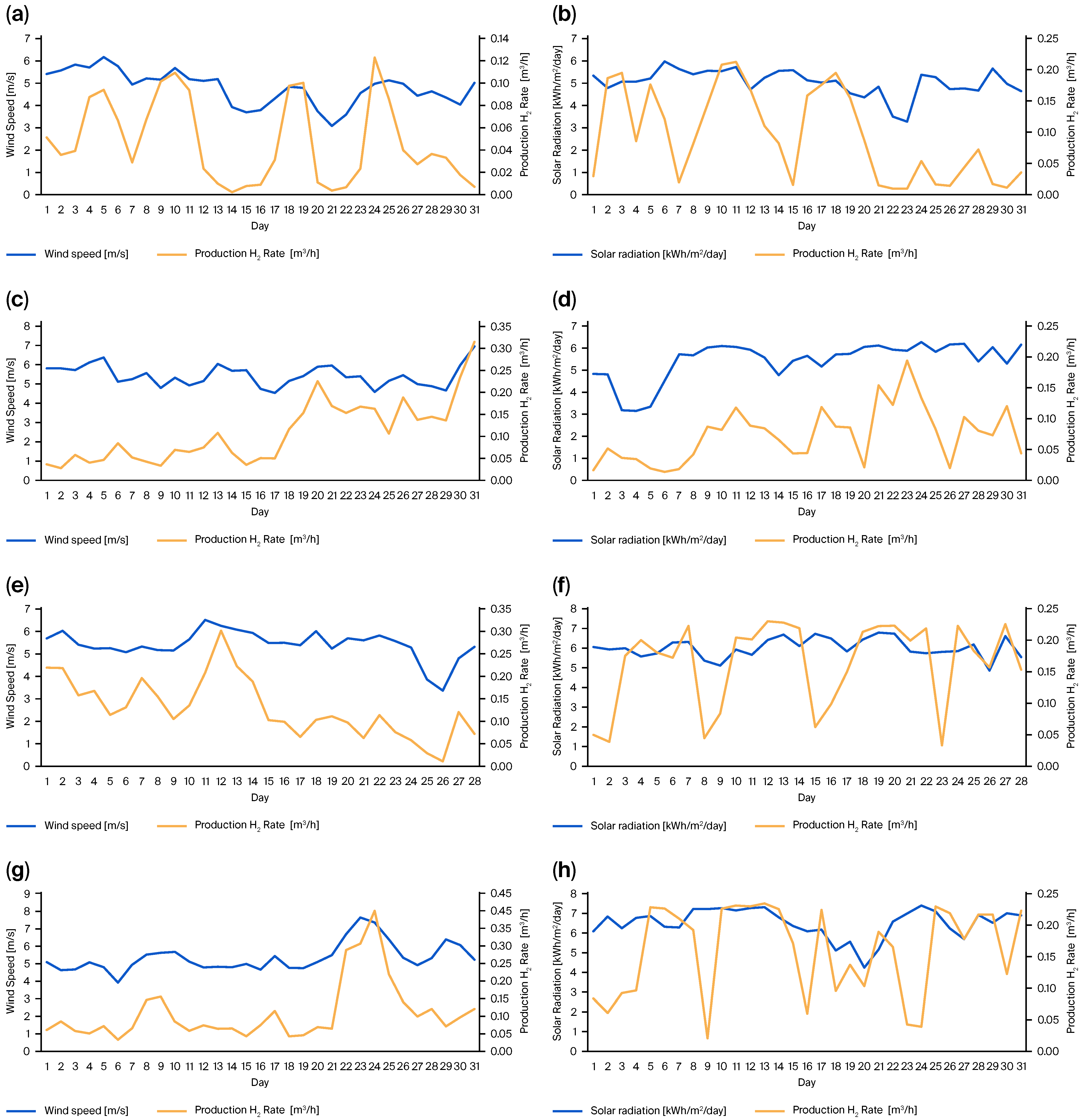
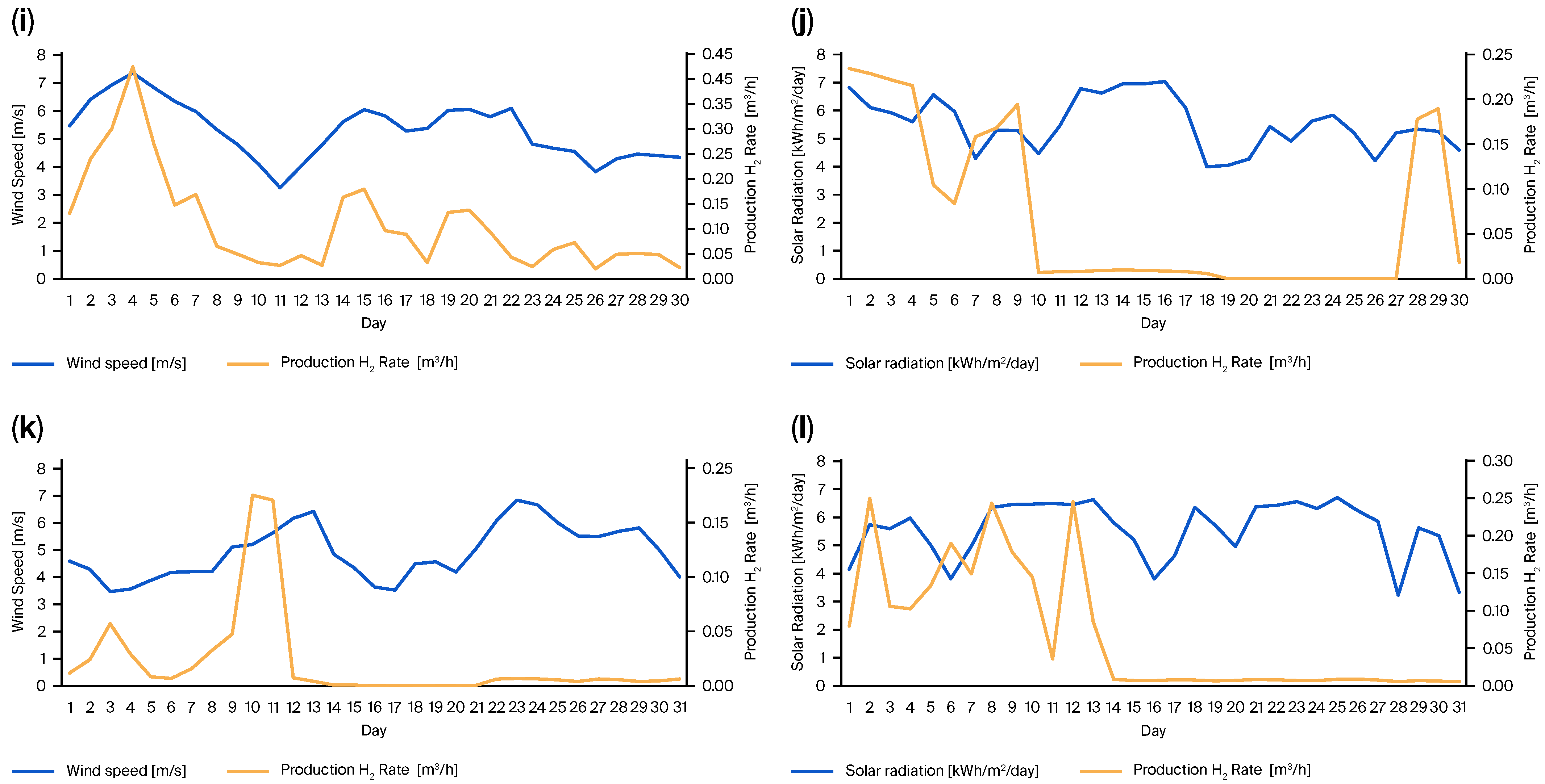
2.2. Experimental Validation of Green Hydrogen Production
2.3. General Model of Palm Oil Triglycerides
2.4. Stoichiometric Balance of Requirements in DR Production
2.4.1. Triglyceride Saturation
2.4.2. Triglyceride Cleavage (Hydrogenolysis)
2.4.3. Formation of n-Paraffins (Deoxygenation of Fatty Acids)
Decarboxylation (DCX)
Decarbonylation (DCN)
Hydrodeoxygenation (HDO)
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chia, S.; Nomanbhay, S.; Ong, M.; Shamsuddin, A.; Chew, K.; Show, P. Renewable diesel as fossil fuel substitution in Malaysia: A review. Fuel 2022, 314, 123137. [Google Scholar] [CrossRef]
- Ajeeb, W.; Gomes, D.; Neto, R.; Baptista, P. Life cycle analysis of hydrotreated vegetable oils production based on green hydrogen and used cooking oils. Fuel 2025, 390, 134749. [Google Scholar] [CrossRef]
- Alles, K.; Demirel, Y. Measuring risk of renewable diesel production processes using a multi-criteria decision strategy. Chemosphere 2024, 354, 141695. [Google Scholar] [CrossRef]
- Lin, J.; Nurazaq, W.; Wang, W.; Lee, C.; Poon, H.; Gan, S.; Duong, V.; Prapainainar, P. Unsteady Spray Dynamics of Hydro-Processed Renewable Diesel: Influence of Thermo-Physical Properties under High-Pressure Injection. J. Energy Inst. 2025, 122, 102222. [Google Scholar] [CrossRef]
- Bezergianni, S.; Dimitriadis, A. Comparison between different types of renewable diesel. Renew. Sustain. Energy Rev. 2013, 21, 110–116. [Google Scholar] [CrossRef]
- Kordouli, E.; Lycourghiotis, S.; Bourikas, K.; Lycourghiotis, A.; Kordulis, C. Renewable diesel synthesis by hydro-processing in green solvents. Curr. Opin. Green Sustain. Chem. 2024, 48, 100936. [Google Scholar] [CrossRef]
- Pérez-Rangel, N.; Coronado, C.; Ancheyta, J. Approaches to conditioning of vegetable oil feedstock for hydrotreating to produce renewable diesel. Fuel 2025, 383, 133897. [Google Scholar] [CrossRef]
- Mehra, K.; Goel, V.; Kumar, R. An integrated multi-attribute decision framework for sustainability assessment of renewable diesel fuel production pathways. Energy Convers. Manag. 2024, 309, 118461. [Google Scholar] [CrossRef]
- Hsu, K.; Wang, W.; Liu, Y. Experimental studies and techno-economic analysis of hydro-processed renewable diesel production in Taiwan. Energy 2018, 164, 99–111. [Google Scholar] [CrossRef]
- Das, A.K.; Sahu, S.K.; Panda, A.K. Current status and prospects of alternate liquid transportation fuels in compression ignition engines: A critical review. Renew. Sustain. Energy Rev. 2022, 161, 112358. [Google Scholar] [CrossRef]
- Pratama, J.; Rahmawati, Z.; Widyanto, A.; Gunawan, T.; Abdullah, W.; Jamari, N.; Hamzah, A.; Fansuri, H. Advancements in green diesel production for energy sustainability: A comprehensive bibliometric analysis. RSC Adv. 2024, 14, 36040–36062. [Google Scholar] [CrossRef] [PubMed]
- Julio, A.; Milessi, T.; Batlle, E.; Lora, E.; Maya, D.; Palacio, J. Techno-economic and environmental potential of Renewable Diesel as complementation for diesel and biodiesel in Brazil: A comprehensive review and perspectives. J. Clean. Prod. 2022, 371, 133431. [Google Scholar] [CrossRef]
- Yang, Z.; Shah, K.; Pilon-McCullough, C.; Faragher, R.; Azmi, P.; Hollebone, B.; Fieldhouse, B.; Yang, C.; Dey, D.; Lambert, P.; et al. Characterization of renewable diesel, petroleum diesel and renewable diesel/biodiesel/petroleum diesel blends. Renew. Energy 2024, 224, 120151. [Google Scholar] [CrossRef]
- UNE. Automotive Fuels. Paraffinic Diesel Fuel from Synthesis or Hydrotreatment. Requirements and Test Methods. 2024. Available online: https://www.une.org/encuentra-tu-norma/busca-tu-norma/norma?c=N0072509 (accessed on 5 November 2025).
- ASTM. Specification for Diesel Fuel. (ASTM International, 2021). Available online: https://www.astm.org (accessed on 5 November 2025).
- Gómez-Doménech, D.; Herrero, L.; Ballesteros, R.; Lapuerta, M. Perspective on the use and benefits of a fossil-free advanced diesel fuel: An effective low-emission alternative to electrification. Biomass Bioenergy 2025, 200, 107965. [Google Scholar] [CrossRef]
- Pérez, W.; Marín, J.; Río, J.; Peña, J.; Rios, L. Upgrading of palm oil renewable diesel through hydroisomerization and formulation of an optimal blend. Fuel 2017, 209, 442–448. [Google Scholar] [CrossRef]
- Happonen, M.; Lähde, T.; Messing, M.; Sarjovaara, T.; Larmi, M.; Wallenberg, L.; Virtanen, A.; Keskinen, J. The comparison of particle oxidation and surface structure of diesel soot particles between fossil fuel and novel renewable diesel fuel. Fuel 2010, 89, 4008–4013. [Google Scholar] [CrossRef]
- Serrano, L.; Santana, B.; Pires, N.; Correia, C. Performance, Emissions, and Efficiency of Biodiesel versus Hydrotreated Vegetable Oils (HVO), Considering Different Driving Cycles Sensitivity Analysis (NEDC and WLTP). Fuels 2021, 2, 448–470. [Google Scholar] [CrossRef]
- Mariano, J.; Emmett, E. What to Know About Renewable Diesel and Biodiesel. Baker Institute for Public Policy. 2023. Available online: https://www.bakerinstitute.org/research/what-know-about-renewable-diesel-and-biodiesel (accessed on 5 November 2025).
- Lunardi, P.; Julio, P.; Bolson, V.; Mayer, F.; Castilhos, F. Comparative techno-economic assessment of renewable diesel production integrated with alternative hydrogen supply. Int. J. Hydrogen Energy 2024, 89, 820–835. [Google Scholar] [CrossRef]
- Hussain, M.; Biradar, C. Production of hydroprocessed renewable diesel from Jatropha oil and evaluation of its properties. Mater. Today Proc. 2023, 72, 1420–1425. [Google Scholar] [CrossRef]
- Edeh, I.; Raheem, A. Renewable diesel Production: A Review. Petro Chem. Indus. Intern. 2023, 6, 93–105. [Google Scholar]
- Jong, S.; Antonissen, K.; Hoefnagels, R.; Lonza, L.; Wang, M.; Faaij, A.; Junginger, M. Life-cycle analysis of greenhouse gas emissions from renewable jet fuel production. Biotechnol. Biofuels 2017, 10, 64. [Google Scholar] [CrossRef]
- Tirado, A.; Alvarez-Majmutov, A.; Ancheyta, J. Modeling and simulation of a multi-bed industrial reactor for renewable diesel hydroprocessing. Renew. Energy 2022, 186, 173–182. [Google Scholar] [CrossRef]
- Douvartzides, S.; Charisiou, N.; Papageridis, K.; Goula, M. Green diesel: Biomass feedstocks, production technologies, catalytic research, fuel properties and performance in compression ignition internal combustion engines. Energies 2019, 12, 809. [Google Scholar] [CrossRef]
- Sotelo-Boyás, R.; Liu, Y.; Minowa, T. Renewable diesel production from the hydrotreating of rapeseed oil with Pt/zeolite and NiMo/Al2O3 catalysts. Ind. Eng. Chem. Res. 2011, 50, 2791–2799. [Google Scholar] [CrossRef]
- Bezergianni, S.; Dimitriadis, A.; Kalogianni, A.; Pilavachi, P. Hydrotreating of waste cooking oil for biodiesel production. Part I: Effect of temperature on product yields and heteroatom removal. Bioresour. Technol. 2010, 101, 6651–6656. [Google Scholar] [CrossRef] [PubMed]
- Ajith, B.; Patel, G.; Der, O.; Selvan, C.; Samuel, O.; Annadurai, S.; Thajudeen, K.; Yadav, K. Microwave-assisted transesterification of hybrid Garcinia gummi-gutta and Garcinia indica oils: Optimization using RSM and meta-heuristic algorithms for high-yield biodiesel production. Biomass Bioenergy 2025, 202, 108223. [Google Scholar] [CrossRef]
- Zhu, C.D.; Samuel, O.D.; Patel, M.; Der, O.; Abbas, M.; Hussain, F.; Ting, T.T. Enhancing CI engine performance and emission control using a hybrid RSM–Rao algorithm for ZnO-doped castor–neem biodiesel blends. Case Stud. Therm. Eng. 2025, 74, 106841. [Google Scholar] [CrossRef]
- Kalnes, T.; Shonnard, D.; Marker, T. Green Diesel: A Second Generation Biofuel. Int. J. Chem. React. Eng. 2007, 5, 1–11. [Google Scholar] [CrossRef]
- Concas, G.; Cocco, D.; Lecis, L.; Petrollese, M. Life Cycle Analysis of a Hydrogen Valley with multiple end-users. J. Phys. Conf. Ser. 2022, 2385, 012035. [Google Scholar] [CrossRef]
- Wang, Z.; Zhao, F.; Dong, B.; Wang, D.; Ji, Y.; Cai, W.; Han, F. Life cycle framework construction and quantitative assessment for the hydrogen fuelled ships: A case study. Ocean Eng. 2023, 281, 114740. [Google Scholar] [CrossRef]
- Sadeghi, S.; Ghandehariun, S.; Rosen, M. Comparative economic and life cycle assessment of solar-based hydrogen production for oil and gas industries. Energy 2020, 208, 118347. [Google Scholar] [CrossRef]
- Lorenzi, G.; Baptista, P.; Venezia, B.; Silva, C.; Santarelli, M. Use of waste vegetable oil for hydrotreated vegetable oil production with high-temperature electrolysis as hydrogen source. Fuel 2020, 278, 117991. [Google Scholar] [CrossRef]
- Atallah, M.; Elsayed, A.; Alqahtani, M.; Shaheen, A. Hybrid renewable energy systems for seawater-based green hydrogen in Egyptian coastal zones: A case study. Unconv. Resour. 2025, 8, 100239. [Google Scholar] [CrossRef]
- Tezer, T. Multi-objective optimization of hybrid renewable energy systems with green hydrogen integration and hybrid storage strategies. Int. J. Hydrogen Energy 2025, 142, 1249–1271. [Google Scholar] [CrossRef]
- Nasser, M.; Al-Sharafi, A.; Al-Buraiki, A.; Yilbas, B.; Khairy, M. Hydrogen production via using hybrid renewable energy and waste fuels derived systems incorporating heat recovery and carbon capture measures. Appl. Energy 2025, 401, 126746. [Google Scholar] [CrossRef]
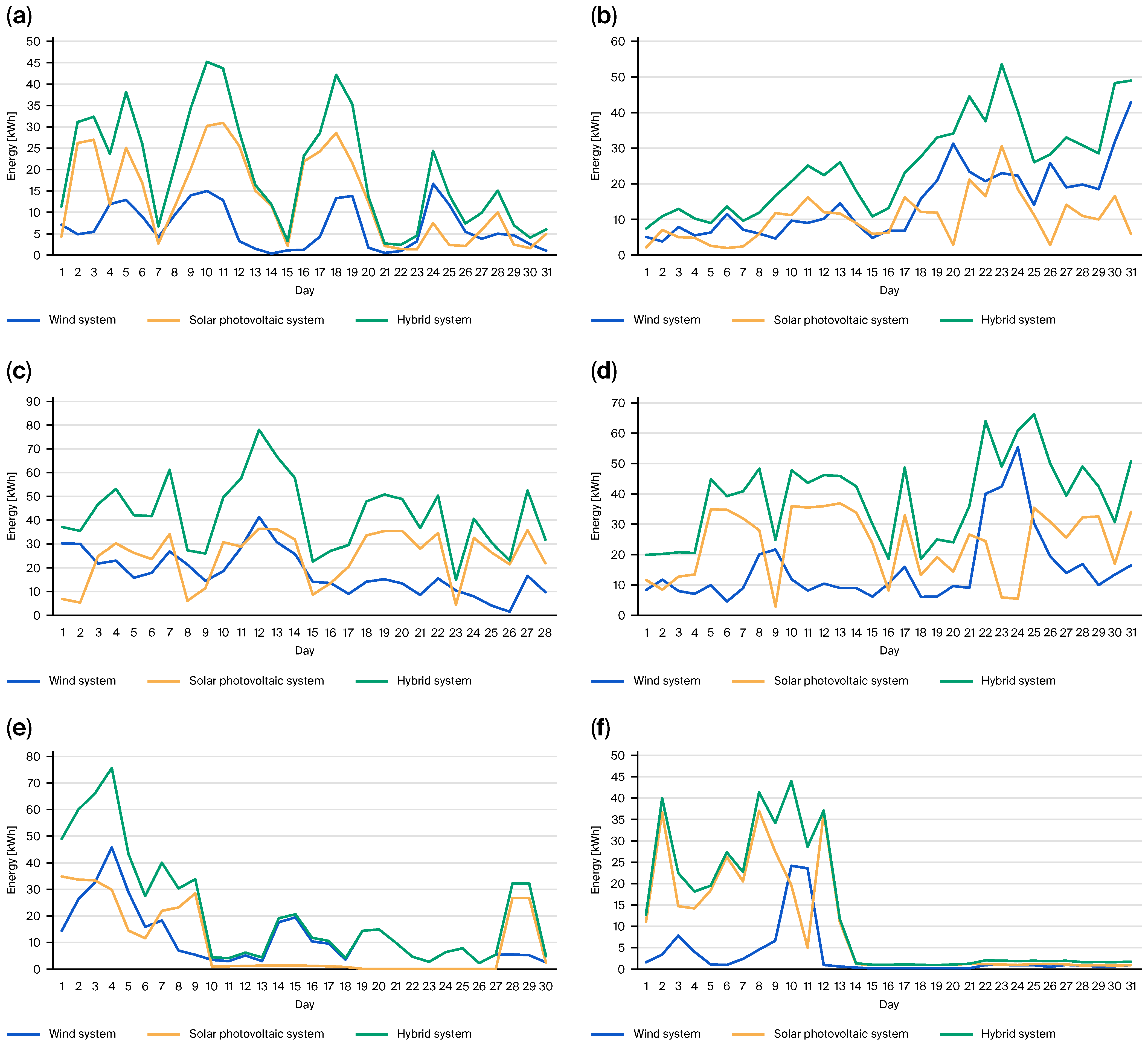
- Cruz, L.; Serrano-Florez, D.; Bastidas-Barranco, M. Analysis of the Availability Curve of the 15 kW Wind–Solar Hybrid Microplant Associated with the Demand of the Power-to-Gas (PtG) Pilot Plant Located at University of La Guajira. Processes 2024, 12, 1903. [Google Scholar] [CrossRef]
- Perpiñán, L.; Serrano, D.; Bastidas, M. Simulación del sistema power to gas ubicado en la universidad de la guajira mediante el software homer pro. Rev. Ambient. Agua Aire Suelo 2022, 13, 49–64. [Google Scholar] [CrossRef]
- Barranco, M.; Florez, D.; Arrieta, A.; Uribe, C.; Granda, A.; Ramirez, M. Desarrollo de sistemas Power to Gas basados en fuentes de energía renovables no convencionales del departamento de La Guajira. In Alianza Séneca: Impulsando La Transformación Y La Sostenibilidad Energética De Colombia; Ministerio de Ciencia Tecnología e Innovación: Bogotá, Colombia, 2023; pp. 1–661. [Google Scholar]
- Castañeda, M.; Cano, A.; Jurado, F.; Sánchez, H.; Fernández, L. Sizing optimization, dynamic modeling and energy management strategies of a stand-alone PV/hydrogen/battery-based hybrid system. Int. J. Hydrogen Energy 2013, 38, 3830–3845. [Google Scholar] [CrossRef]
- Rangel, R.; Acosta, D.; Corredor, M.; Garcia-Freites, S.; Sanjuán, M. Modelación de un Sistema de Producción e Inyección de Hidrógeno Verde en Redes de Transporte de Gas Natural para Evaluar el Impacto de la Dinámica del Sistema en la Operación de Promigas; Asociación Colombiana de Ingenieros de Petróleos (ACIPET): Bogota, Colombia, 2023; pp. 1–13. [Google Scholar]
- Li, C.; Zhu, X.; Cao, G.; Sui, S.; Hu, M. Dynamic modeling and sizing optimization of stand-alone photovoltaic power systems using hybrid energy storage technology. Renew. Energy 2009, 34, 815–826. [Google Scholar] [CrossRef]
- Shahidi, F. Bailey’s Industrial Oil And Fat Products; John Wiley & Sons: Hoboken, NJ, USA, 2005. [Google Scholar]
- Gunstone, F.; Harwood, J.; Dijkstra, A. The Lipid Handbook with CD-ROM; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar]
- Lucantonio, S.; Giuliano, A.; Rossi, L.; Gallucci, K. Green Diesel Production via Deoxygenation Process: A Review. Energies 2023, 16, 844. [Google Scholar] [CrossRef]
- Samikannu, A.; Mani, M.; Konwar, L.; Mäki-Arvela, P.; Virtanen, P.; Mikkola, J. Hydrodeoxygenation of Triglycerides into Renewable Diesel. Biorefining Renew. Diesel Prod. Mark. 2025, 89, 96–137. [Google Scholar] [CrossRef]
- Carvajal-Romo, G.; Valderrama-Mendoza, M.; Rodríguez-Urrego, D.; Rodríguez-Urrego, L. Assessment of solar and wind energy potential in La Guajira, Colombia: Current status, and future prospects. Sustain. Energy Technol. Assess. 2019, 36, 100531. [Google Scholar] [CrossRef]
- Camargo, E.; Becerra, J.; Silva-Ortega, J. Caracterización de los potenciales de Energía Solar y Eólica para la integración de Proyectos sostenibles en Comunidades Indígenas en La Guajira Colombia. Espacios 2017, 38, 11. [Google Scholar]

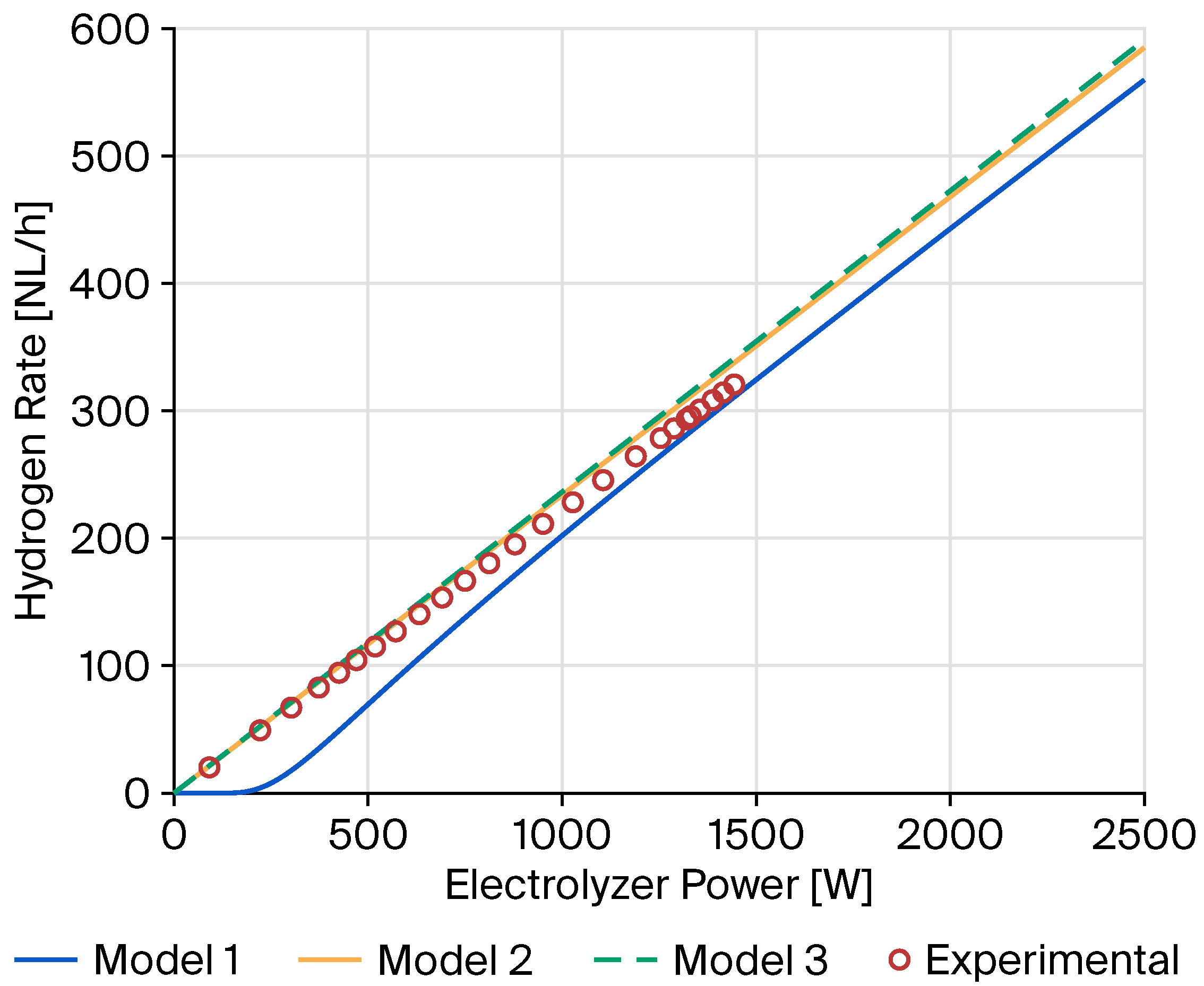
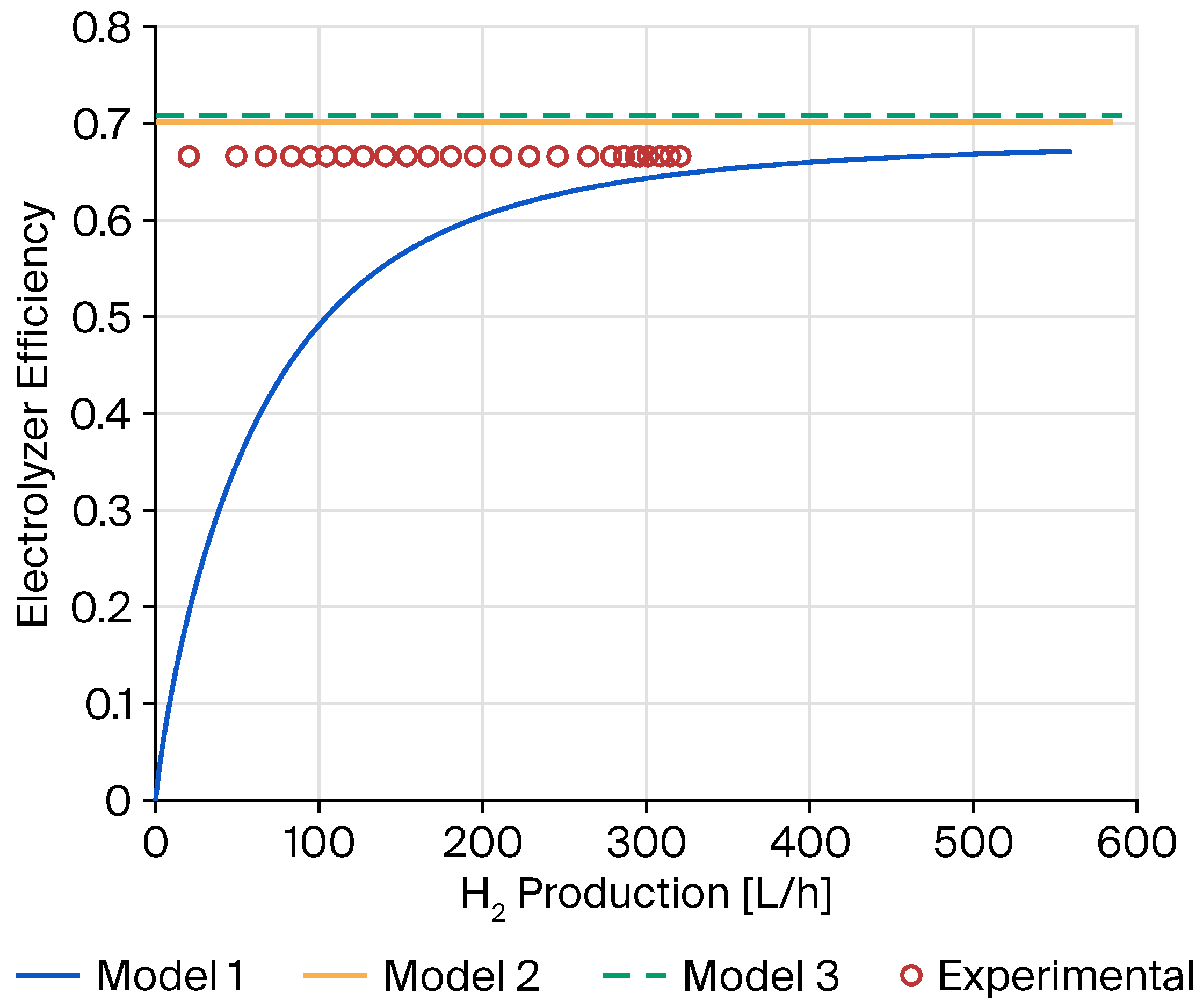
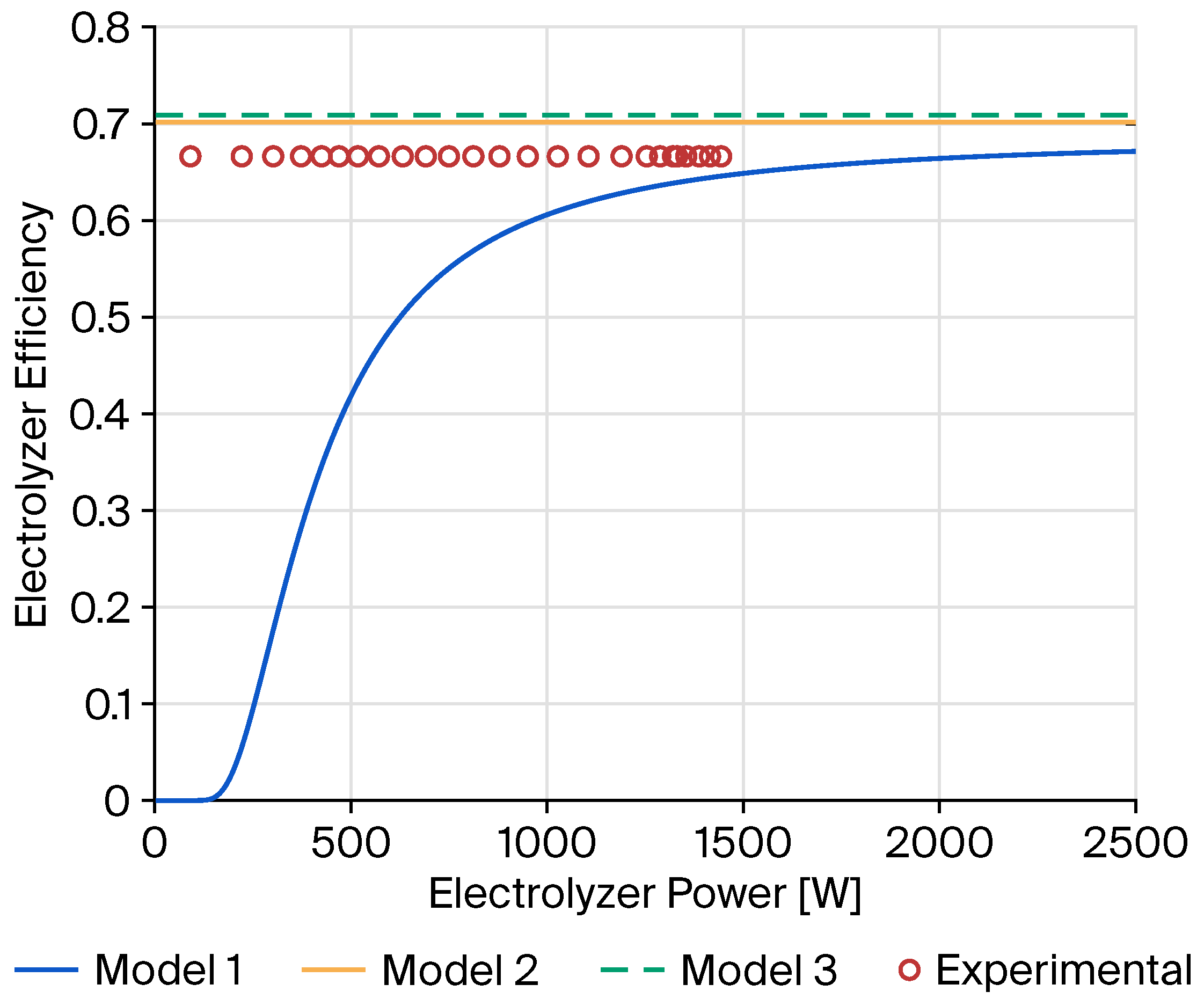
| Error Metric | Model 1 | Model 2 | Model 3 |
|---|---|---|---|
| Mean Squared Error (MSE) | 835.9485 | 130.0476 | 187.3681 |
| R2; (Coefficient of Determination) | 0.9023 | 0.9848 | 0.9781 |
| Triglyceride (TAG) | Composition (Fatty Acids) | General Formula | Molar Mass (g/mol) | Comment |
|---|---|---|---|---|
| POP (Palmitoyl– Oleoyl–Palmitoyl) | C16:0–C18:1–C16:0 | 807–808 | Most representative model for crude palm oil (predominates due to high palmitate and oleate content). | |
| POO (Palmitoyl– Oleoyl–Oleoyl) | C16:0–C18:1–C18:1 | 831–832 | Highly abundant in the liquid fraction (palm olein). | |
| POL (Palmitoyl– Oleoyl–Linoleoyl) | C16:0–C18:1–C18:2 | 833–834 | Present at lower levels; contributes unsaturations (fluidity). | |
| PLP (Palmitoyl– Linoleoyl–Palmitoyl) | C16:0–C18:2–C16:0 | 809–810 | Provides a balance between saturation and unsaturation. | |
| SOS (Stearoyl– Oleoyl–Stearoyl) | C18:0–C18:1–C18:0 | 891–892 | More typical of cocoa butter fats, but occurs in some palm oil fractions. |
| Triglyceride (TAG) | Palm Oil Mass (g) | RD Mass (g) | Propane Mass (g) | ||
|---|---|---|---|---|---|
| DCX | DCN | HDO | |||
| POP | 16,452.96 | 13,130.73 | 13,130.73 | 13,961.29 | 870.11 |
| POO | 15,181.10 | 12,243.97 | 12,243.97 | 12,987.10 | 778.52 |
| POL | 13,703.27 | 11,077.87 | 11,077.87 | 11,750.23 | 704.37 |
| PLP | 14,685.68 | 11,748.55 | 11,748.55 | 12,491.68 | 778.52 |
| SOS | 17,560.37 | 14,238.14 | 14,238.14 | 15,068.70 | 870.11 |
| Triglyceride (TAG) | Palm Oil Mass (g) | RD Mass (g) | Propane Mass (g) | ||
|---|---|---|---|---|---|
| DCX | DCN | HDO | |||
| POP | 21,713.52 | 17,329.06 | 17,329.06 | 18,425.17 | 1148.31 |
| POO | 20,035.01 | 16,158.77 | 16,158.77 | 17,139.51 | 1027.44 |
| POL | 18,084.66 | 14,619.84 | 14,619.84 | 15,507.17 | 929.59 |
| PLP | 19,381.18 | 15,504.95 | 15,504.95 | 16,485.68 | 1027.44 |
| SOS | 23,175.01 | 18,790.55 | 18,790.55 | 19,886.66 | 1148.31 |
| Triglyceride (TAG) | Palm Oil Mass (g) | RD Mass (g) | Propane Mass (g) | ||
|---|---|---|---|---|---|
| DCX | DCN | HDO | |||
| POP | 28,183.05 | 22,492.24 | 22,492.24 | 23,914.94 | 1490.45 |
| POO | 26,004.42 | 20,973.26 | 20,973.26 | 22,246.21 | 1333.56 |
| POL | 23,472.97 | 18,975.81 | 18,975.81 | 20,127.52 | 1206.55 |
| PLP | 25,155.79 | 20,124.63 | 20,124.63 | 21,397.58 | 1333.56 |
| SOS | 30,079.98 | 24,389.17 | 24,389.17 | 25,811.88 | 1490.45 |
| Triglyceride (TAG) | Palm Oil Mass (g) | RD Mass (g) | Propane Mass (g) | ||
|---|---|---|---|---|---|
| DCX | DCN | HDO | |||
| POP | 31,035.52 | 24,768.73 | 24,768.73 | 26,335.43 | 1641.30 |
| POO | 28,636.39 | 23,096.02 | 23,096.02 | 24,497.80 | 1468.53 |
| POL | 25,848.72 | 20,896.40 | 20,896.40 | 22,164.67 | 1328.67 |
| PLP | 27,701.87 | 22,161.49 | 22,161.49 | 23,563.28 | 1468.53 |
| SOS | 33,124.45 | 26,857.66 | 26,857.66 | 28,424.36 | 1641.30 |
| Triglyceride (TAG) | Palm Oil Mass (g) | RD Mass (g) | Propane Mass (g) | ||
|---|---|---|---|---|---|
| DCX | DCN | HDO | |||
| POP | 16,836.69 | 13,436.98 | 13,436.98 | 14,286.91 | 890.40 |
| POO | 15,535.17 | 12,529.53 | 12,529.53 | 13,290.00 | 796.68 |
| POL | 14,022.87 | 11,336.24 | 11,336.24 | 12,024.28 | 720.80 |
| PLP | 15,028.20 | 12,022.56 | 12,022.56 | 12,783.02 | 796.68 |
| SOS | 17,969.93 | 14,570.22 | 14,570.22 | 15,420.14 | 890.40 |
| Triglyceride (TAG) | Palm Oil Mass (g) | RD Mass (g) | Propane Mass (g) | ||
|---|---|---|---|---|---|
| DCX | DCN | HDO | |||
| POP | 10,657.48 | 8505.49 | 8505.49 | 9043.49 | 563.62 |
| POO | 9833.63 | 7931.09 | 7931.09 | 8412.45 | 504.29 |
| POL | 8876.35 | 7175.74 | 7175.74 | 7611.27 | 456.26 |
| PLP | 9512.72 | 7610.18 | 7610.18 | 8091.54 | 504.29 |
| SOS | 11,374.81 | 9222.82 | 9222.82 | 9760.82 | 563.62 |
| Triglyceride (TAG) | Mass (kg) | Total Mass (kg) | Palm Oil Mass (kg) | RD Mass (kg) | Propane Mass (kg) | Mass (kg) | CO Mass (kg) | Water Mass (kg) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Saturation | Fatty Acid Decomposition | DCX | DCN | HDO | DCX | DCN | HDO | DCX | DCN | DCN | HDO | ||||
| POP | 0.3 | 0.9 | 0.00 | 0.9 | 1.351 | 2.552 | 124.879 | 99.663 | 99.663 | 105.967 | 6.604 | 19.813 | 12.608 | 8.105 | 16.210 |
| POO | 0.537 | 0.806 | 0.00 | 0.806 | 1.209 | 2.552 | 115.226 | 92.933 | 92.933 | 98.573 | 5.909 | 17.727 | 11.281 | 7.252 | 14.504 |
| POL | 0.729 | 0.729 | 0.00 | 0.729 | 1.094 | 2.552 | 104.009 | 84.082 | 84.082 | 89.185 | 5.346 | 16.039 | 10.206 | 6.561 | 13.123 |
| PLP | 0.537 | 0.806 | 0.00 | 0.806 | 1.209 | 2.552 | 111.465 | 89.172 | 89.172 | 94.813 | 5.909 | 17.727 | 11.281 | 7.252 | 14.504 |
| SOS | 0.3 | 0.9 | 0.00 | 0.9 | 1.351 | 2.552 | 133.284 | 108.068 | 108.068 | 114.372 | 6.604 | 19.813 | 12.608 | 8.105 | 16.210 |
| Triglyceride (TAG) | Mass (kg) | Total Mass (kg) | RD Production (Gallons) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Saturation | Fatty Acid Decomposition | DCN | HDO | DCX | DCN | HDO | Propane Mass (kg) | ||
| POP | 2.40 | 9.62 | 16.83 | 20.43 | 32.45 | 247.87 | 247.87 | 263.55 | 52.88 |
| POO | 4.66 | 11.66 | 18.65 | 22.14 | 38.46 | 250.50 | 250.50 | 265.70 | 51.28 |
| POL | 7.01 | 14.02 | 21.03 | 24.53 | 45.56 | 251.08 | 251.08 | 266.32 | 51.40 |
| PLP | 4.82 | 12.05 | 19.28 | 22.89 | 39.76 | 248.47 | 248.47 | 264.19 | 53.01 |
| SOS | 2.25 | 9.01 | 15.77 | 19.14 | 30.41 | 251.83 | 251.83 | 266.52 | 49.55 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lagos Herrera, A.; Herrera Arroyave, J.; Serrano-Florez, D.; Bastidas-Barranco, M. Simulation and Validation of Green Hydrogen for the Production of Renewable Diesel: Case Study in La Guajira, Colombia. Processes 2025, 13, 3913. https://doi.org/10.3390/pr13123913
Lagos Herrera A, Herrera Arroyave J, Serrano-Florez D, Bastidas-Barranco M. Simulation and Validation of Green Hydrogen for the Production of Renewable Diesel: Case Study in La Guajira, Colombia. Processes. 2025; 13(12):3913. https://doi.org/10.3390/pr13123913
Chicago/Turabian StyleLagos Herrera, Adriana, Jose Herrera Arroyave, Dario Serrano-Florez, and Marlon Bastidas-Barranco. 2025. "Simulation and Validation of Green Hydrogen for the Production of Renewable Diesel: Case Study in La Guajira, Colombia" Processes 13, no. 12: 3913. https://doi.org/10.3390/pr13123913
APA StyleLagos Herrera, A., Herrera Arroyave, J., Serrano-Florez, D., & Bastidas-Barranco, M. (2025). Simulation and Validation of Green Hydrogen for the Production of Renewable Diesel: Case Study in La Guajira, Colombia. Processes, 13(12), 3913. https://doi.org/10.3390/pr13123913






