Abstract
The extraction of plant essences and volatile organic compounds has been performed using various methods throughout history. The production of essential oils is a significant industry. One notable ornamental flower in the Philippines is Jasminum sambac (L.), also known as Arabian Jasmine or Sampaguita, which is highly fragrant and used in various cosmetics, food, and medicine. Researchers developed a method to produce quality J. sambac (L.) concrete using the Supercritical Fluid Extraction (SFE). Among the parameters explored, it was noted that no drying method had more pleasant odors, while other drying methods had varying effects on the extract scent. A temperature of 35 °C produces fragrant and sweet concrete, and temperatures above 40 °C result in burnt-smelling extract. Higher pressure enhanced the aroma and yield. The drying method also affected the output. Plucking petals before drying resulted in low-quality outcomes. Using a blow dryer damages the petals. A combination of low temperature, moderate pressure, and no drying method produced the best aromas. However, the process requires winterization to remove waxes in the samples, which will decrease the yield.
1. Introduction
The extraction of plant essences and extracts has been practiced since ancient times for applications in personal care, medicine, and cosmetics. These biomolecules have been extensively studied over the years due to their diverse benefits. Plant-based volatile organic compounds, often derived from essential oils, are particularly valued for their strong aromatic properties. These compounds include phenols, alcohols, esters, ketones, aldehydes, and heterocyclic compounds [1]. Additionally, certain plant extracts, such as pesticides, have been utilized in non-cosmetic applications due to their bioactive properties [2].
Highly valued for their numerous volatile compounds, plant-based volatile organic compounds have been extracted using various methods that have evolved and improved over centuries. Traditional techniques such as boiling, crushing, and distilling—commonly called conventional methods—have long been employed in the extraction process. These methods remain relevant today and are often used with modern approaches, such as Supercritical Fluid Extraction (SFE) [3]. For instance, flowers and herbs are harvested and processed to extract their aromatic compounds and sweet scents. Essential oils derived from flowering plants are widely utilized in the perfume industry to create a diverse range of fragrances for cosmetic purposes [4].
The production of essential oils from plants is in high demand in the Philippine economy. The country ranks 94th in exporting plant extracts and essential oils worth USD 819,000. Additionally, in the same year, essential oils were ranked 457th out of roughly 1200 products imported by the Philippines [5].
One of the most notable ornamental flowers in the Philippines is Jasminum sambac (L.) (J. sambac (L.)), commonly referred to as Arabian jasmine, jasmine, or Sampaguita. It is a highly fragrant and ornamental plant that is widely cultivated in Southeast Asian countries for aromatherapy, food, and skin care products [6,7]. Its cultural significance and importance are the factors for the income and support of Sampaguita farms; however, during the time of the pandemic, jasmine farms in Bulacan, Pampanga, Pangasinan, Laguna, and Albay plummeted, which poses an issue to the livelihood of the farmers [8,9].
Extracting essential oils from Jasminum sambac (L.) in ancient times involved steam distillation or solvent extraction, which could result in the degradation of the essential oils, and used hazardous solvents. In contrast, SFE has the potential to provide a much more efficient and environmentally friendly method of extracting essential oils from J. sambac (L.) [10]. Supercritical Fluid Extraction (SFE) is a modern extraction technique that has gained popularity in extracting essential oils from various plant materials. The method involves using a supercritical fluid, a substance used to maintain the temperature and pressure above its critical point.
Traditional methods such as steam distillation and solvent soaking have the advantage of being well-understood and relatively simple. These methods are capable of capturing aromatic compounds that are too delicate for harsh treatment. However, these methods often suffer from low yield, high labor/time input, possible thermal degradation or solvent residue, and high cost [10]. Modern methods like SFE, subcritical solvent systems, or assisted techniques like microwave or ultrasound extraction offer higher selectivity, better retention of delicate, heat-sensitive volatile compounds, fewer or no toxic residues, shorter extraction time, and often higher yield. Their disadvantages are usually high capital cost, greater technical complexity, need for specialized equipment, potentially more energy input, and sometimes the regulatory/safety issues involved with using exotic solvents or high pressures [10].
The Jasminum sambac (L.) market has declined significantly despite its versatile applications across various industries, including cosmetics, food, and pharmaceuticals. This decline may be attributed to several factors, including competition from synthetic alternatives, fluctuations in supply and demand, and challenges in extraction methods that affect product quality [11,12]. Furthermore, there is an issue surrounding Sampaguita farmers in the Philippines. The majority of the Sampaguita farmers are women [13]. Jasminum sambac farming has emerged as a vital livelihood for many families, particularly in regions like Pampanga and Laguna, where women play a crucial role in production and marketing.
The use of GC–MS (Gas Chromatography–Mass Spectrometry) and FTIR (Fourier Transform Infrared Spectrometry) analyses provides essential support in evaluating and optimizing the extraction of Sampaguita essential oils. GC–MS allows for the identification and quantification of individual volatile compounds such as linalool, benzyl acetate, and indole, thereby determining which extraction conditions best preserve the characteristic floral aroma profile [13]. In contrast, FTIR offers a rapid, non-destructive method to confirm the presence of functional groups and assess the overall chemical integrity of the extract. When applied together, these techniques complement each other by providing both compound-specific and structural information, ensuring that the selected extraction parameters maintain both yield and quality. This combined analytical approach strengthens the reliability of the findings and guides the selection of conditions that maximize aroma fidelity and minimize degradation [13].
The samples extracted are waxy when extracted. Jasmine flowers naturally contain waxes along with essential oils. During SFE, especially under certain conditions, these waxes can co-extract with the essential oil, leading to a thicker or waxy product. This is common in jasmine extraction and is why “jasmine concrete”, a waxy-buttery product, is often obtained before refining it into “jasmine absolute” through additional processing steps like ethanol washing and filtration [14].
Multiple methods allow the separation of waxes, often performed by pre-treatment and post-processing. Pre-treatment for jasmine flowers can help reduce wax content and optimize the extraction process, such as cryogenic freezing, ethanol soaking, and microwave-assisted pre-treatment.
Additionally, the potential uses of Supercritical Fluid Extraction (SFE) in obtaining the extract of the Sampaguita are to utilize it economically by producing a pleasingly fragrant product. This product will help to enhance the global market competitiveness of the essential oils imported. Consequently, this study allows us to see the difference in drying methods and to analyze the results of changing the parameters of extraction, namely temperature, pressure, and time, by finding the optimal parameters of SFE for Sampaguita, and, in turn, providing an alternative market for Sampaguita farmers.
This study was limited by time and budget constraints, which required the researchers to narrow the extraction temperature range to 35–45 °C. Consequently, the extreme operating conditions of 100 and 300 bar pressure, as well as 27 and 77 °C temperature, were not tested.
2. Materials and Methods
2.1. Extraction Parameters
In Supercritical Fluid Extraction, the SE-2000 and PM-10000C, TST Co., Ltd. (Taiwan Supercritical Technology Co., Ltd., Changhua, Taiwan), have three variables that allow users to adjust: temperature, pressure, and time. An RSM (response surface methodology) approach utilizing two independent variables will be used to optimize the yield of jasmine extract. An ANOVA analysis and regression coefficient calculations were performed on the data to yield a response surface that determines the optimal parameters for the extraction. The RSM parameters returned by the software are as follows (see Table 1).

Table 1.
RSM parameters.
The RSM parameters are references to studies performed to optimize Jasminum sambac (L.) extraction. The survey by Rassem, H. et al. explores the extraction of jasmine extract from flowers using supercritical CO2 [13]. This study is about extracting essential oil from jasmine flowers using supercritical CO2 and it examines the effects of extraction parameters such as pressure (100–300 bar) and temperature (300–350 K) on oil recovery [13].
2.2. J. sambac Samples and Preparation
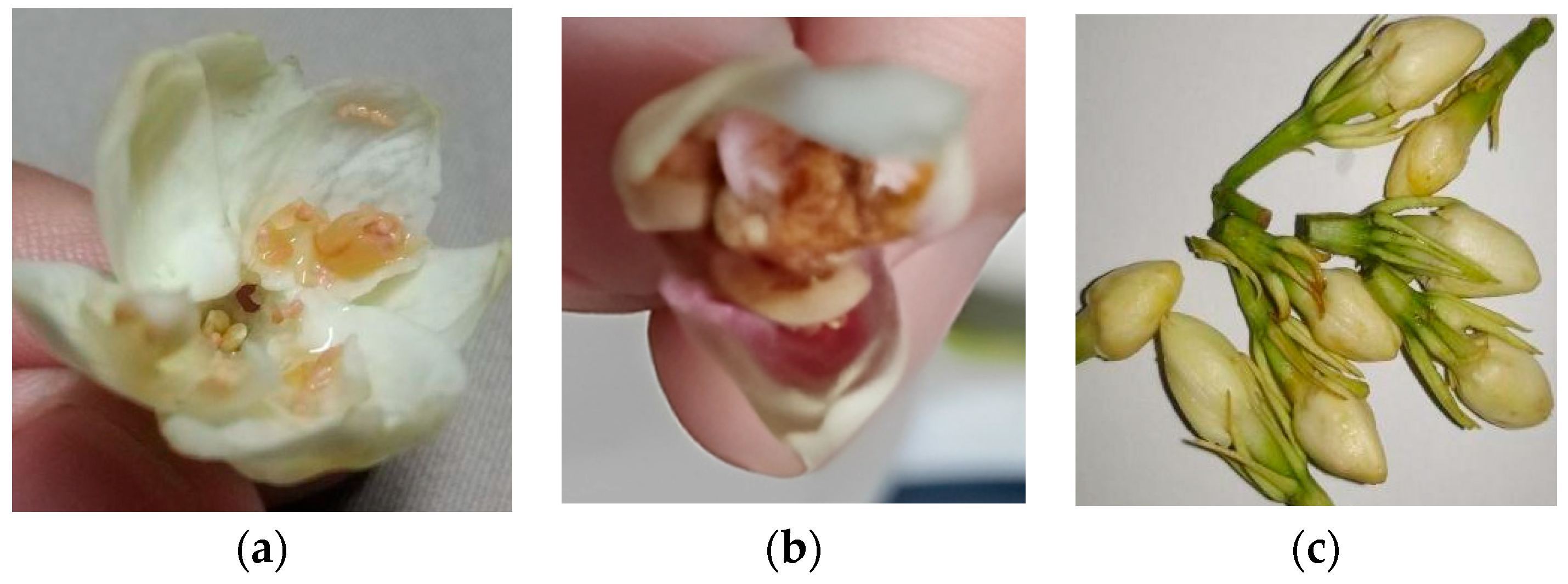
Sampaguita buds and open flowers were sourced from Laguna farms. The farmers were instructed to place the samples on ice or in a refrigerator right after collection. Storage and transport were at 0 °C to 4 °C. The J. sambac flowers, once received, were prepared according to the drying methods agreed upon. It was also noted that the flower and petal quality were checked when they were being plucked. Notable petals that are not fit for extraction are those seen in Figure 1, which are generally flowers that contain brown marks, are an off-colored white with brown specks, and with visible bugs and/or worms. Otherwise, the flowers were to be treated based on the agreed drying method.
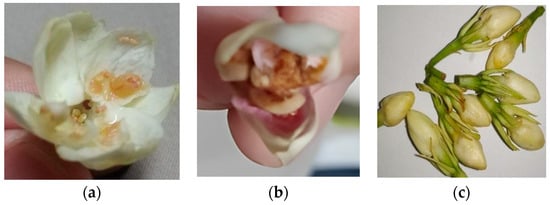
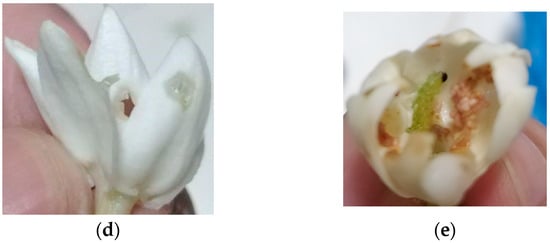
Figure 1.
Flowers (a) with brown residues, (b,c) which were off-colored/differently colored, (d) with bug bites, and (e) with worms and/or bugs are not suitable for SFE.
2.3. Drying Methods
The drying methods agreed upon are the methods that are located in Table 2, which are the simpler drying methods readily available to the researchers for use. Due to the limited time and budget, they could not conduct several trials for each. In other cases, the flowers were cleaned and separated from the stalks, and the size was reduced to maximize the surface area for contact during the Supercritical Fluid Extraction.

Table 2.
Drying methods and their description (A: after plucking; B: before plucking).
The drying method as a parameter allows the researchers to identify which preparation would allow samples to have a pleasant, unpleasant, or neutral odor when pairing with the three specific SFE parameters, namely temperature, pressure, and time.
2.4. J. sambac Extraction
The extraction procedure using the SFE machine is followed, and the parameters of the RSM values are used. Carbon dioxide is the fluid used for extraction. The petals are first weighed and noted before being extracted. After the J. sambac samples are collected at the output of the machine using plastic sterile centrifuge tubes, the sample extract mass is weighed. The tubes are labeled with the date, time, duration, temperature, and pressure of extraction, as well as the mass of petals and mass of extract. The extracts are placed in a freezer at 0 °C to 4 °C.
2.5. GC–MS and FTIR Analysis of Extract and Sensory Evaluation
The extract of the flowers is first tested for its scent, whether it is pleasant, unpleasant, or neutral. After grouping the samples based on scent, the extracts were collected in an Eppendorf tube based on the scent. There were two tubes for each scent, thus having paired samples per fragrance characterization. These samples in the Eppendorf tubes were stored in the freezer at 0 °C to 4 °C before being sent to the Spectroscopy Laboratory De La Salle University (DLSU)-Central Instrumentation Facility at LTI Spine Road, Laguna Blvd, Biñan, Laguna, Philippines, for analysis of samples through Gas Chromatography–Mass Spectrometry (GC–MS) and Fourier Transform Infrared Spectroscopy (FTIR) to identify and differentiate the compounds in the pleasant, unpleasant, and neutral odor samples. The machines used were Agilent 8890 GC system coupled with a 5977B Mass Selective Detector (MSD) for GC–MS and Agilent Cary 630 FTIR-ATR with manufacturer of Agilent Technologies (Santa Clara, CA, USA) for FTIR. This analysis includes the area under the curve, which indicates the amount identified in the sample, which can be used to determine the sample’s intensity.
The FTIR information given to the researchers by DLSU-Central Instrumentation Facility was only the results of a library search for waves that closely resemble the samples that were given to them, so they can only be used for identification.
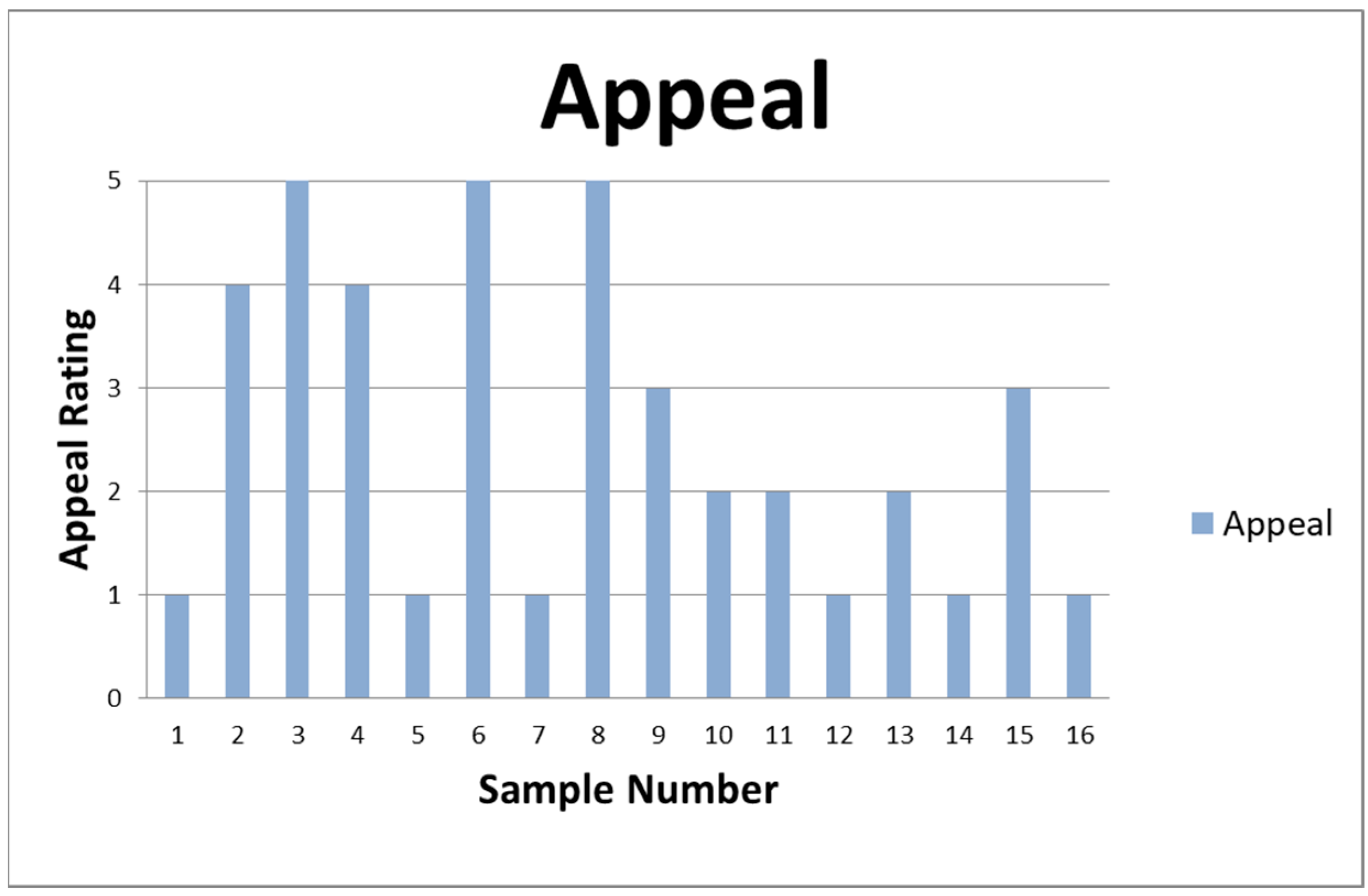
Afterward, the data were arranged by determining the yield according to the drying parameters and the odor with the extraction parameters, and the extracts were summarized with a sensory evaluation. The sensory evaluation summarizes all that is mentioned in the tables as an “Appeal”. This includes a 5-point scale, where 1 is Foul Odor, 2 is Neutral Scent, 3 is Mixed Results, 4 is Pleasant Odor, and 5 is Sweet Odor. The 5-point scale is determined by randomly selecting three participants after the extraction process. Their opinions about the odor are noted, and the commonalities between the three are selected based on the 5-point scale (see Figure 2).
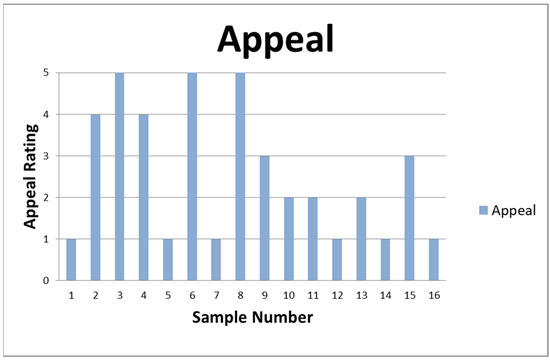
Figure 2.
Appeal rating of the samples of the sensory evaluation.
2.6. Literature Review of Compounds from GC–MS and FTIR Analysis for Odor Characterization
After obtaining the results of GC–MS and FTIR from the Spectroscopy Laboratory of DLSU, the compounds in the list are given a literature review and search on their odor to determine the prominent scent and its contribution to the quality of extraction.
3. Results and Discussion
3.1. Effects of the Drying Method
In studying the effect of the three factors, temperature, pressure, and time, the effect of these on the extraction of J. sambac was measured by the odor of the extract, its appearance, and the yield obtained.
Extraction of the jasmine concrete varies in appearance depending on the drying method used and the parameters set for extraction. As seen in Table 3, most of the extracts look yellowish with a waxy appearance. In contrast to companies selling oils, jasmine oils are often liquid with a deep brownish yellow [15].

Table 3.
Summary of the SFE extract.
The highest yield of 0.0655% was with the drying method of Overnight Air Drying (A), and the lowest of 0.0040% was with the drying method of 3-day Air Drying (A). With each drying method presented, most of the colors have a yellow, waxy appearance. However, most extracts with drying methods of Overnight Air Dry (A) and (B) have appearances that are yellowish with red or brown tints. Blow Drying (A) and (B) consistently show a yellowish waxy appearance.
Thus, the color of the extracts can vary based on the drying technique. Jasmine crude oil extracted using the Overnight Air Dry method often displays yellowish hues with red or brown tints, while that from blow drying consistently maintains a yellowish waxy appearance. The consistency of the waxy appearance across various drying methods suggests that the drying process plays a crucial role in determining the final product’s visual characteristics.
3.2. SFE Extracts
The samples were exposed to higher temperatures, and the resulting extracts remained solid even at room temperature. This phase difference is a result of the extraction of SFE, which allows waxes to be part of the product, hence the solid appearance even at room temperature [16,17]. In contrast, these waxes are often co-extracted products that require another separation process, such as winterization [18].
The temperature of 35 °C resulted in the extracted Sampaguita concrete being fragrant and sweet. An extraction temperature of 40 °C does give a pleasant fragrance, but not as sweet. The resulting product of 35 and 40 °C was yellowish-green in color and quite waxy in texture. However, samples that were subjected to temperatures above 40 °C yielded a reddish-orange color. These samples also tend to smell burnt instead of having the sweet smell. However, it is also seen that higher temperatures allow for a higher yield of extraction. It was also noted that those petals that are not dried are often more pleasant-smelling.
3.3. Sensory Evaluation
Odor quality can be used as a screening method before sending the samples to the DLSU-Central Instrumentation Facility.
Temperature can significantly impact the aroma of the extracted jasmine concrete. Moderate temperatures, such as 35 °C, are often ideal for preserving the delicate and desirable aromatic compounds. Higher temperatures, above 40 °C, may cause the degradation or transformation of these compounds, leading to changes in the scent and taste. In some cases, excessive heat can even form undesirable compounds, such as those responsible for a burnt or off flavor.
Higher extraction temperatures generally result in a higher yield. This is because increased temperature enhances the solubility of the aromatic compounds in the extraction solvent or supercritical fluid. However, excessively high temperatures can also lead to the co-extraction of unwanted compounds, such as waxes and pigments, which may alter the crude extract’s appearance and texture [19].
3.4. Compound Identification Through FTIR and GC–MS Analysis
3.4.1. Pleasant-Smelling Samples
FTIR Analysis
In the FTIR analysis, the first pleasant odor sample (Sample 6) shows that the sample has a very close similarity to PARVAN 3150, F.N. 3502*REFINED PARAFFIN WAX, commonly refined paraffin wax (RPW). The percentage quality of the sample to RPW is 92.425%. This compound is a straight chain of hydrocarbons that is odorless and colorless with a waxy appearance and has a melting point of 23 to 67 °C [20]. According to the Editors in Britannica, RPW allows ease of extraction of volatile organic compounds fit for perfumes and other plant extract applications. This quality also explains why the sample has a floral quality that is pleasant to smell [20].
However, the second pleasant-smelling sample (Sample 2) that was sent had a quality of POLY(ETHYLENE). Polyethylene is odorless in nature. While the FTIR result cannot explain the specific reason why a non-aromatic compound has the quality of an aromatic compound (the flower extract), it could be more textural than it is aromatic. The “waxiness” of the aromatic samples is due to the waxes mixing with the oils and extracted compounds of the J. sambac petals.
The FTIR analysis of the research by Rassem et al. [21], relating to Jasminum samba extracts, has very similar peak regions, which further confirms the FTIR analysis. This is also true with the other FTIR bands and peaks of other samples [22].
GC–MS
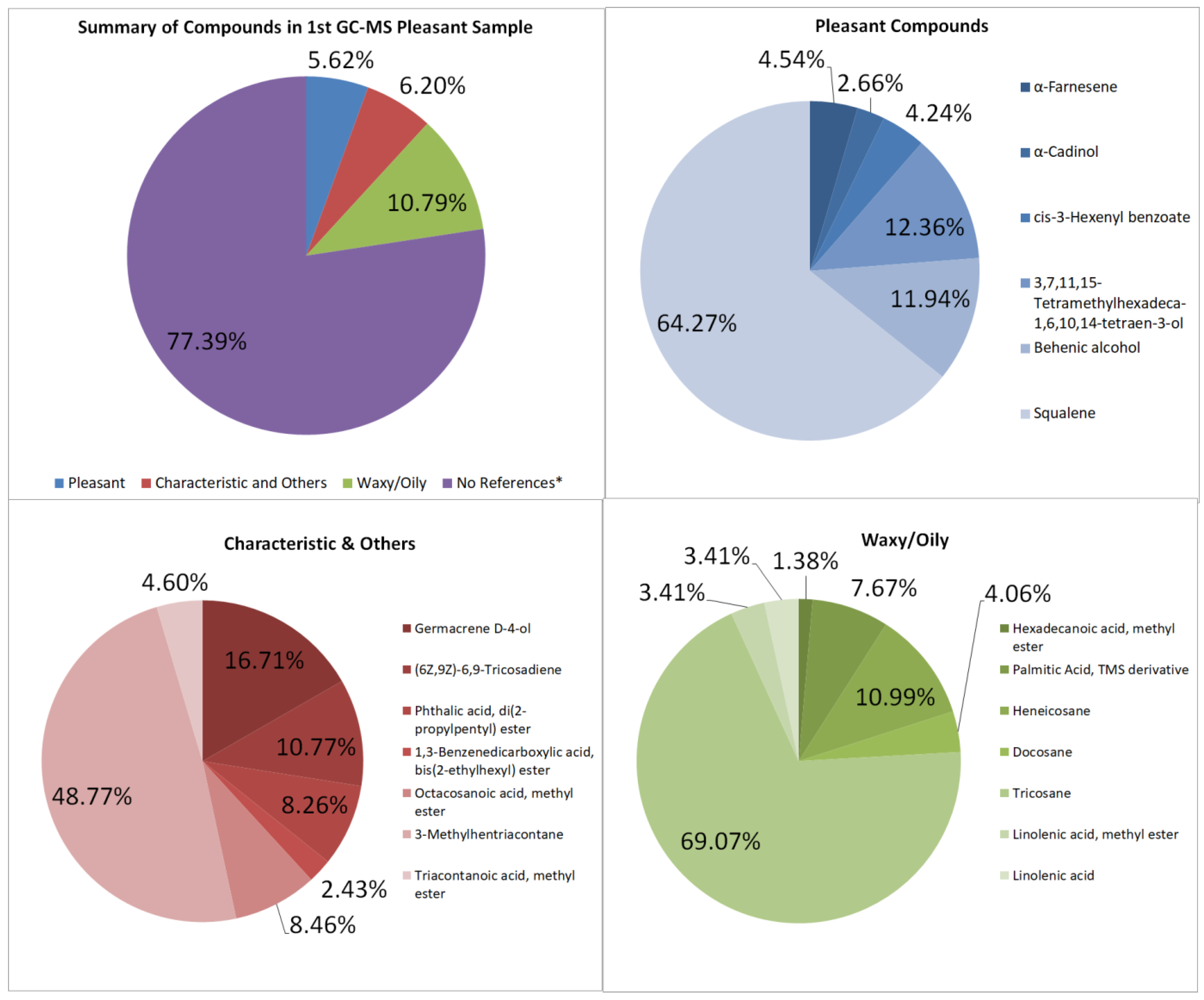
The GC–MS of the pleasant-smelling samples is summarized with its compounds and peak area seen in Table A1 and Table A2, and Figure 3. Some compounds from the extracted oil are also found in the study of Akram, A., et al. [23]; the extract from Jasminum sambac contains benzyl alcohol, benzyldehyde, citral, 2-Phenyl ethyl acetate, linalool, geraniol, eugenol, farnesol, citrinyl acetate, nerol, geranyl acetate, and citronellol. The first sample totals 2,028,713,630 units of area in the GC–MS graph. No unpleasant odor compounds were detected, and there are six pleasant-smelling compounds totaling 114,003,297.8, which is about 5.62% of the overall area of the GC–MS spectra. The floral aroma is contributed to by α-Farnesene, α-Cadinol, cis-3-Hexenyl benzoate, and 3,7,11,15-Tetramethylhexadeca-1,6,10,14-tetraen-3-ol [24,25]. The main volatile components of jasmine are α-farnesene, (Z)-3-hexenyl benzoate, linalool, benzyl alcohol, benzyl acetate, methyl anthranilate, and indole [17]. Chemicals like 1,3-Benzenedicarboxylic acid, bis(2-ethylhexyl) ester, Squalene, and Phthalic acid, di(2-propylpentyl) ester have characteristic odors; these contribute to 6.20%. Although these compounds are said to have a specific scent, it was not explicitly stated whether the odors are pleasant or unpleasant upon research.
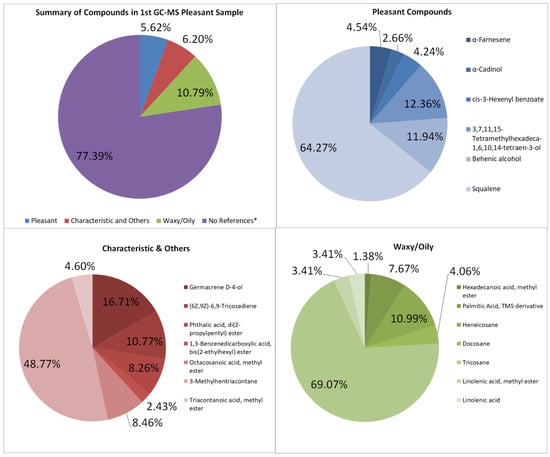
Figure 3.
Summary and listed GC–MS compounds of first pleasant sample with listed percentages for each classified compound (No references* are compounds that cannot be found references for its odor).
In addition to the similar compounds in the study, there are more likely to be the pleasant-smelling compounds that are most dominant according to the study, and thus will likely be present in all the other samples [26].
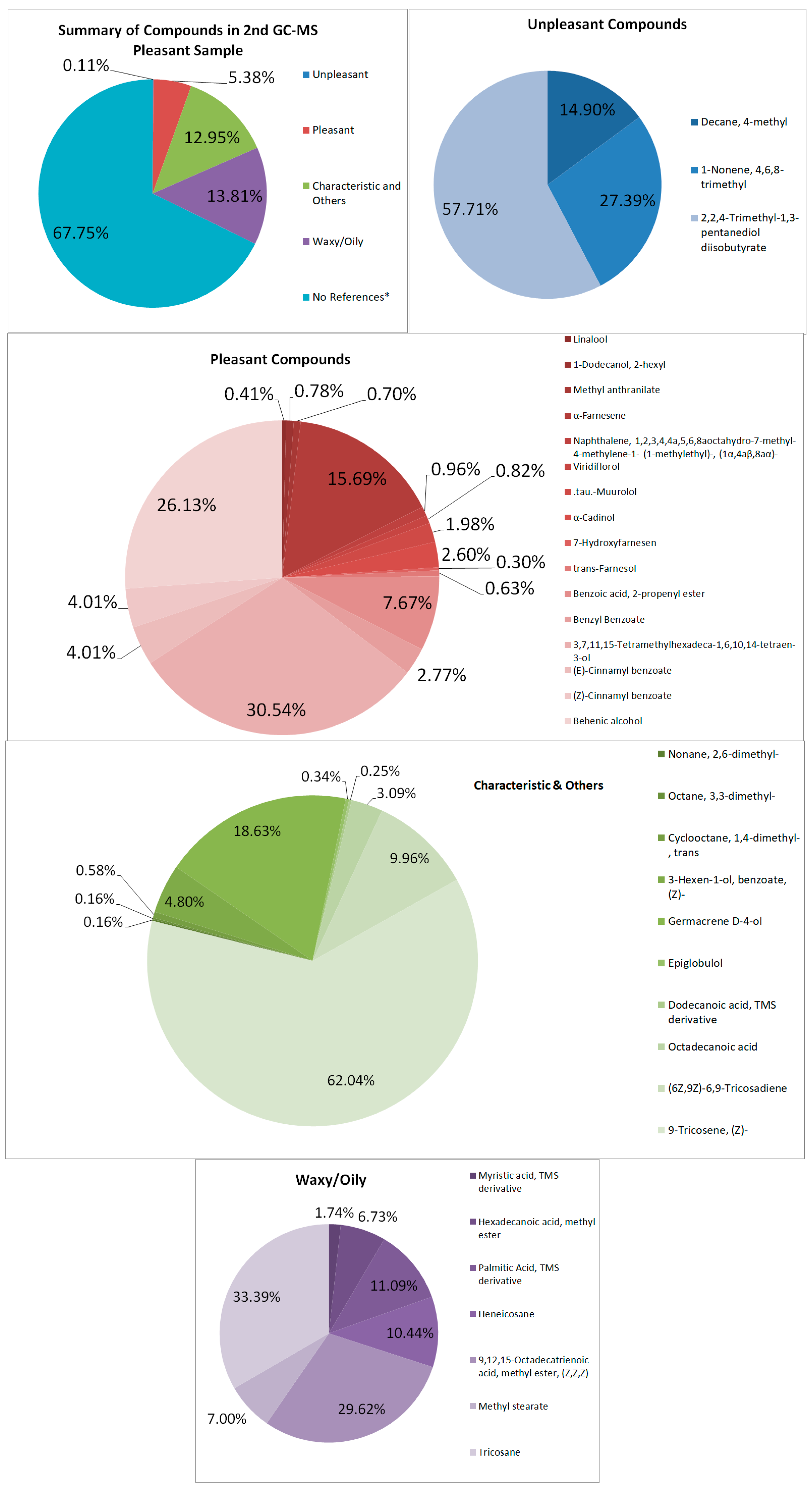
In the second sample (see Figure 4 and Table 2), compounds that are suspected to aid in the strong quality of the sample are Nonane, 2,6-dimethyl, 1-Dodecanol, 2-hexyl, and methyl anthranilate [27,28,29]. Pleasant odor compounds contributed to 5.38% of the total area of the spectrum. Similarly, some are found in the first sample, such as the floral aroma. However, some compounds are qualitatively bad, such as 1-Nonene, 4,6,8-trimethyl, and Decane, 4-methyl, which may impart undesirable qualities, which may contribute to the quality of the sample, though it was not prominent [30,31,32]. Octadecyl octyl ether, which is stated to have odor-masking properties, may mask either the pleasant or unpleasant-smelling sample.
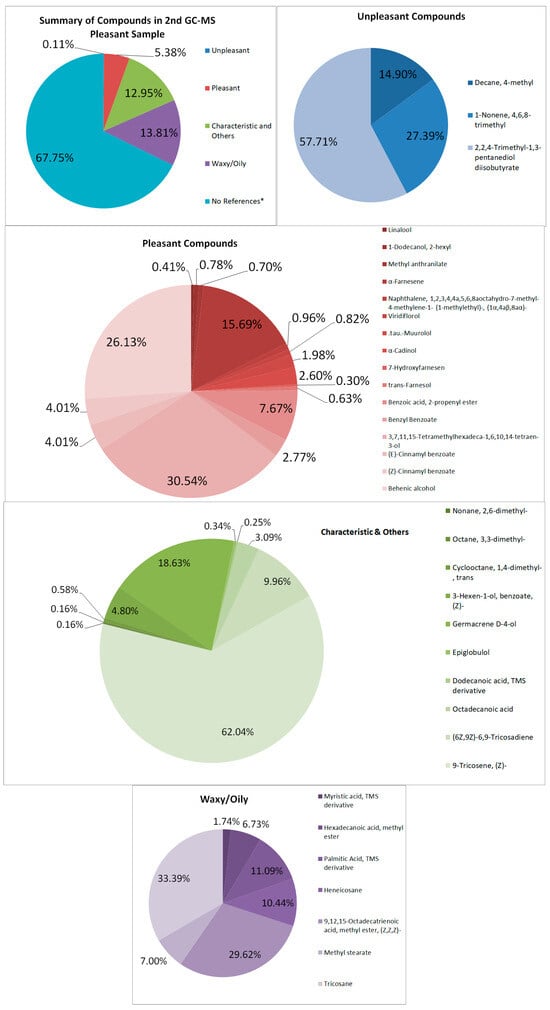
Figure 4.
Summary and listed GC–MS compounds of second pleasant sample with listed percentages for each classified compound (No references* are compounds that cannot be found references for its odor).
The waxy components may have contributed to the waxy quality and appearance of the samples, contributing to 10.79% and 13.81% of the first and second pleasant-smelling samples, respectively. This is due to the lack of winterization processes for removing wax from the extract.
3.4.2. Unpleasant-Smelling Samples
FTIR
This unpleasant-smelling sample (Sample 1) has an FTIR quality which is most comparable to the properties of paraffin wax, the same as the first pleasant-smelling sample. The second unpleasant-smelling sample (Sample 5) has the same quality as polyethylene. As mentioned earlier, in some cases, these qualities analyzed via a library search of FTIR are more texturally related than scent-related. The second unpleasant-smelling sample, however, did have the second highest quality, ethylene propylene diene terpolymer. This specific compound, according to the reference below, is a low-odor EPDM (ethylene propylene diene monomer) that has a distinct, unpleasant odor. This odor becomes much more potent at higher temperatures, which may be one of the reasons for the “odd smell” description in the sensory evaluation.
GC–MS
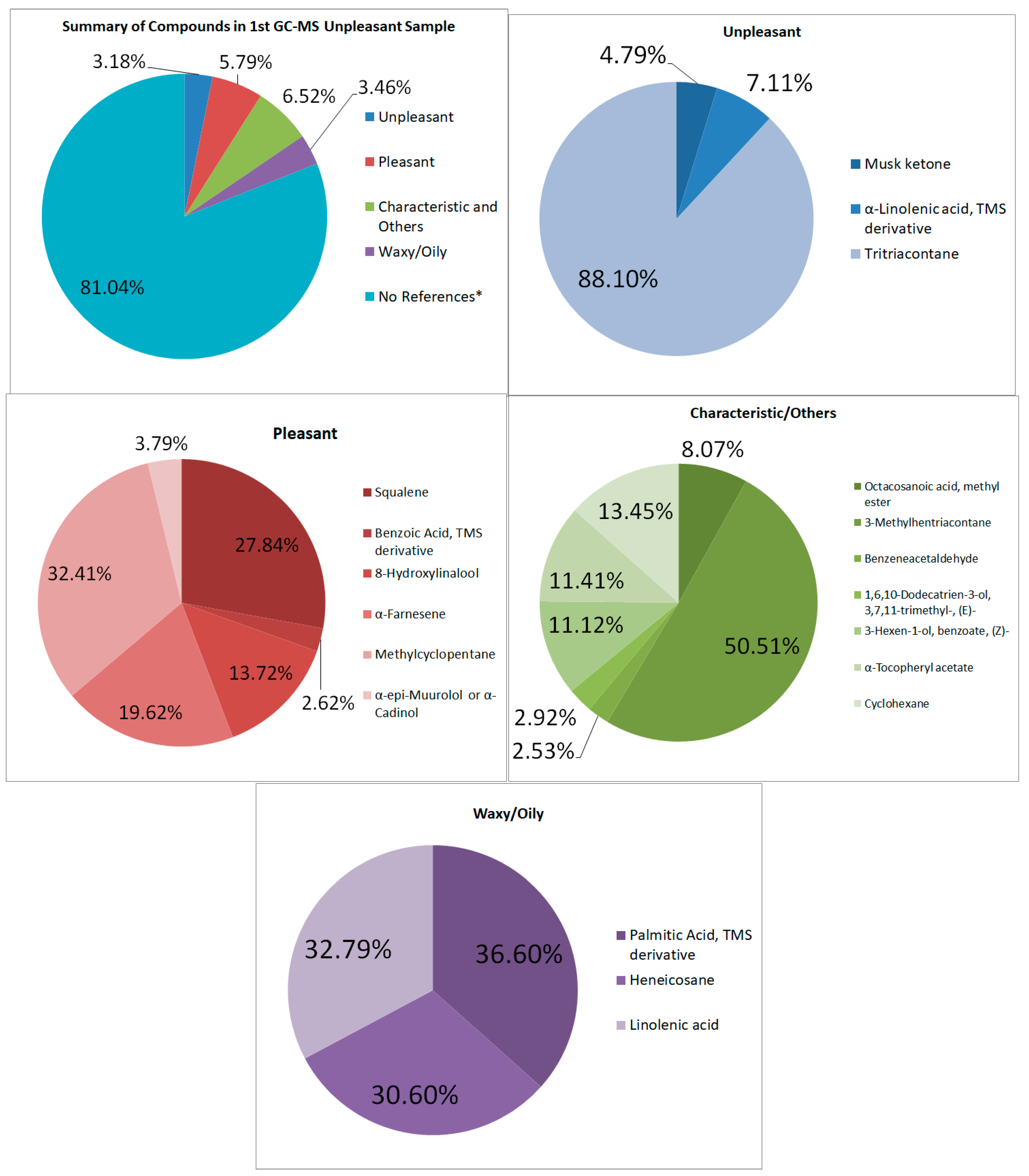
As for the first set of unpleasant-smelling samples (See Figure 5), there was about 3.18% of the total area having unpleasant odor compounds; however, a total of 5.79% were pleasant-smelling compounds. Most of these are characterized by a fruity, herbal, and sweet odor. Other compounds that were detected in GC–MS are some that have “characteristic” odors that do not have a lot of description in the related literature but are unique to the compound itself. These compounds include Squalene, α-Tocopheryl acetate, and α-Tocopheryl acetate [33,34].
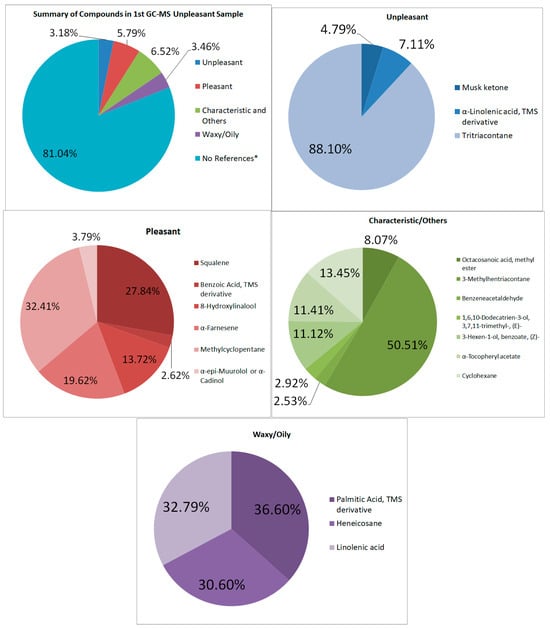
Figure 5.
Summary and listed GC–MS compounds of first unpleasant sample with listed percentages for each classified compound (No references* are compounds that cannot be found references for its odor).
The foul-smelling compounds in the GC–MS are fewer than the pleasant-smelling compounds found in the sample, totaling 3.18%. The musk ketone is one of the more intense aromas, according to references. It is a “highly tenacious musky aroma with a discreet animal note” [35].
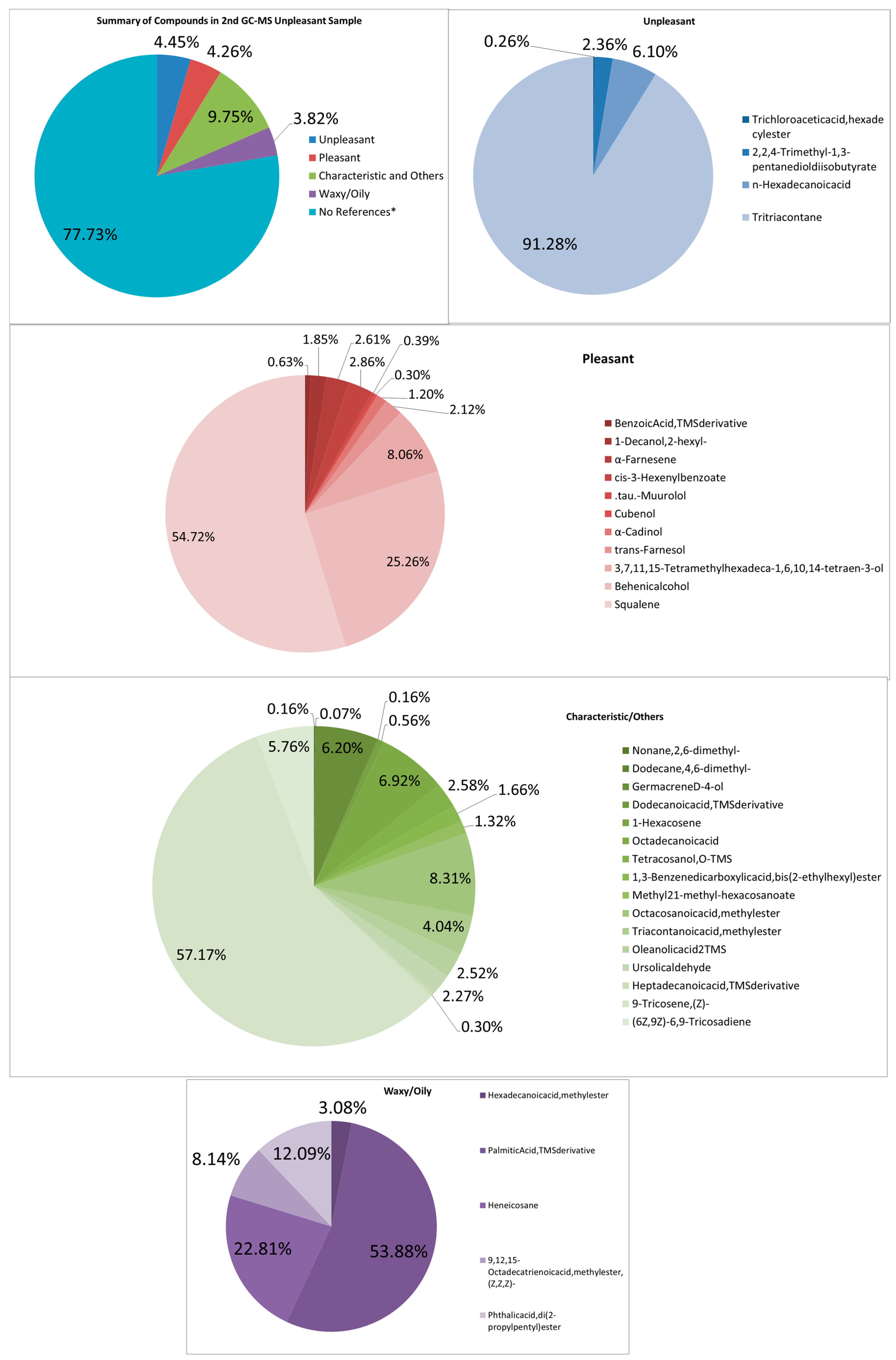
In a similar case with the second unpleasant-smelling sample (See Figure 6), it has more compounds than all the other samples. Many more pheromone components of the same kind of compound are seen. Other compounds such as Nonane, 2,6-dimethyl-, Cubenol, Octacosanoic acid, and methyl ester are most unique. The intensity of the aroma can be from other pungent and musty odors, such as Trichloroacetic acid, hexadecyl ester, and 2,2,4-Trimethyl-1,3-pentanediol diisobutyrate, n-Hexadecanoic acid. The combination of these odors also seems to contribute to the odd and foul smell.
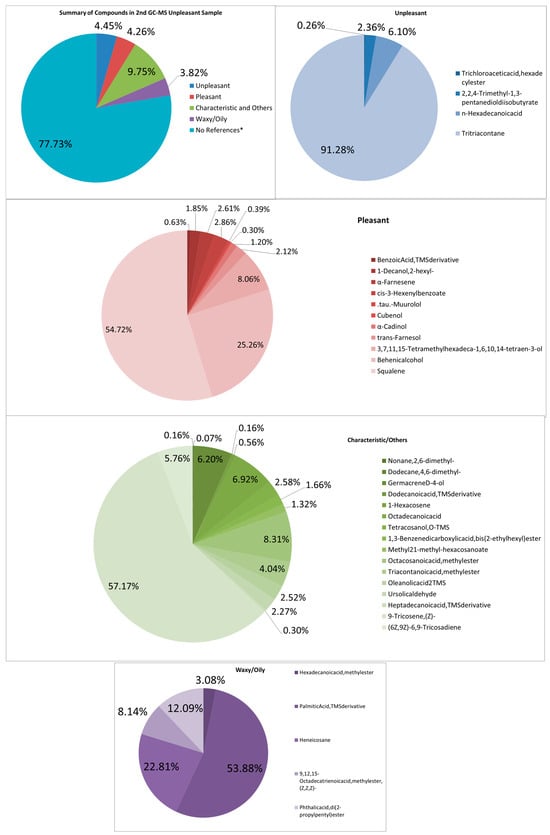
Figure 6.
Summary and listed GC–MS compounds of second unpleasant sample with listed percentages for each classified compound (No references* are compounds that cannot be found references for its odor).
3.4.3. Neutral Samples
FTIR
Samples with barely any detectable aromas or no scent are categorized here. The two samples fit perfectly with the polyethylene similarity and quality from the FTIR analysis. Both samples (Samples 9 and 10, respectively) in this category were detected to have a faint scent that dissipates quickly when smelled. It was difficult to tell if they had a scent or not. Some say their faint smell was detected, though they did not notice their specific scent. This scent might be related to EPDM in both samples.
GC–MS
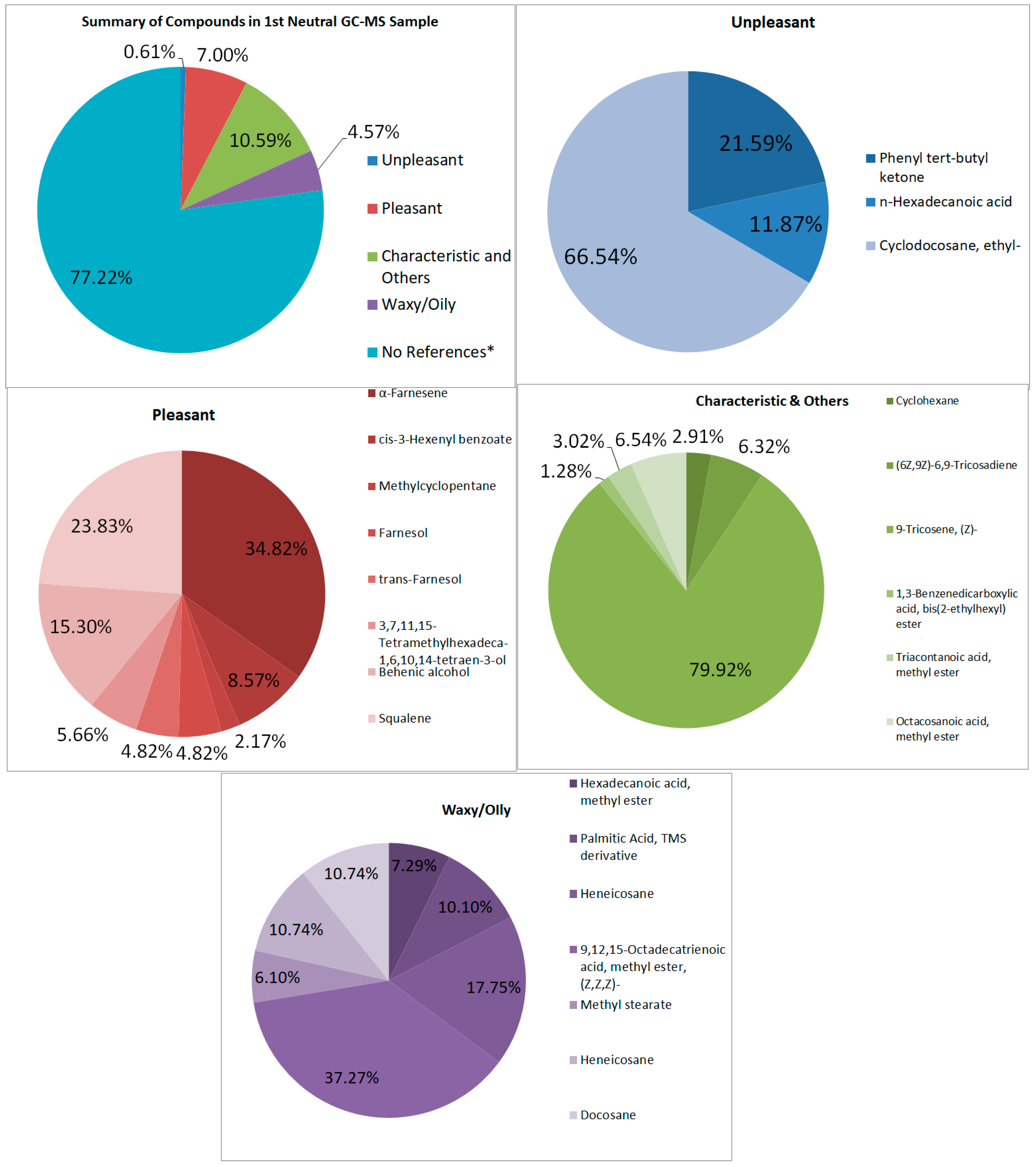
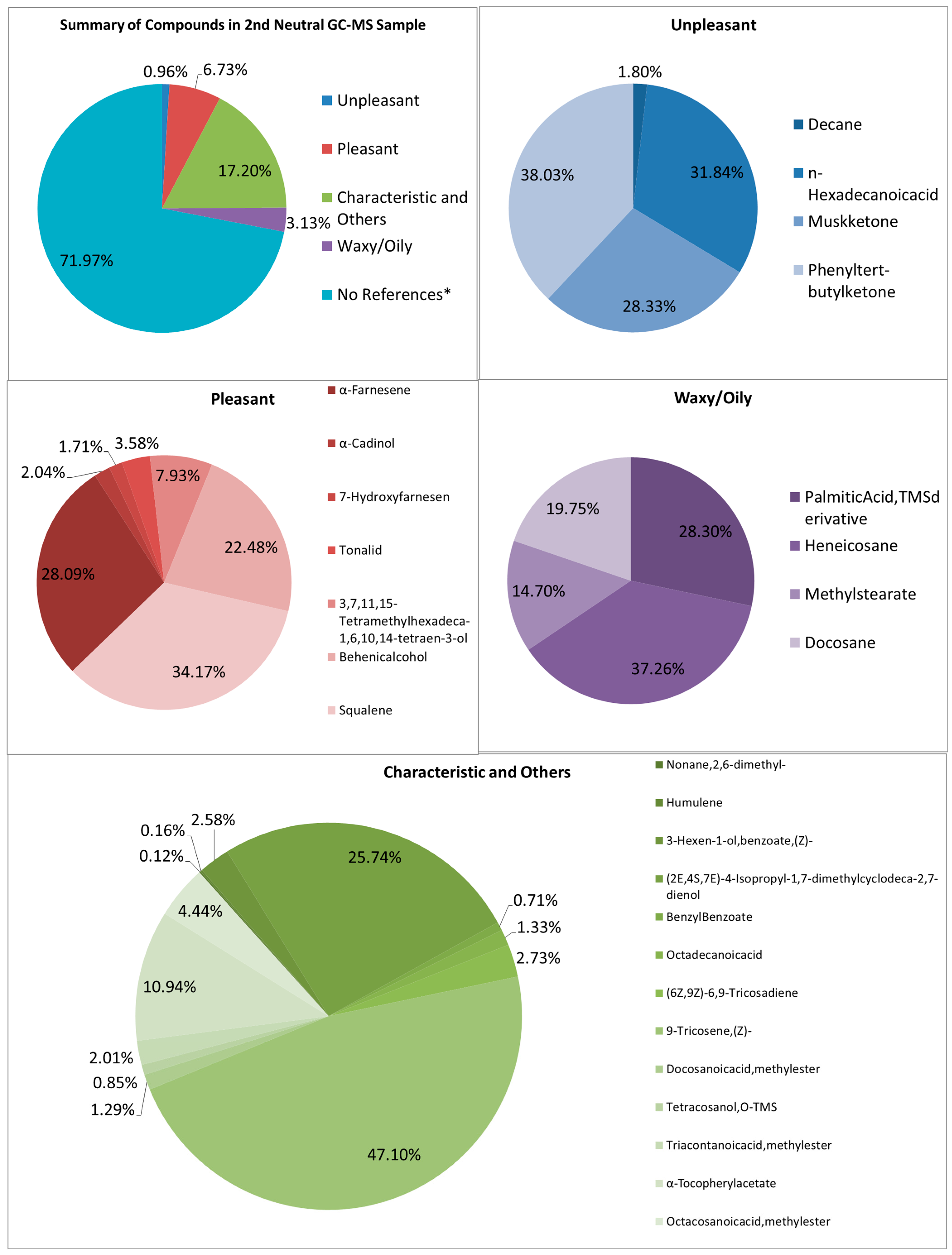
Looking at both samples (See Figure 7 and Figure 8), multiple samples have the “odorless” scent profile. Very faint scents of pleasant and unpleasant smells exist in the first and second samples, respectively. It is also difficult to tell why these scents were barely detectable, though the graphs show large signals. The percentage of pleasant to unpleasant is around 7.003% and 0.608% for the first neutral sample, and 0.963% and 6.732% for the second neutral sample, respectively. Both show higher percentages in the characteristic odor, which may be the ones overpowering the other compounds.
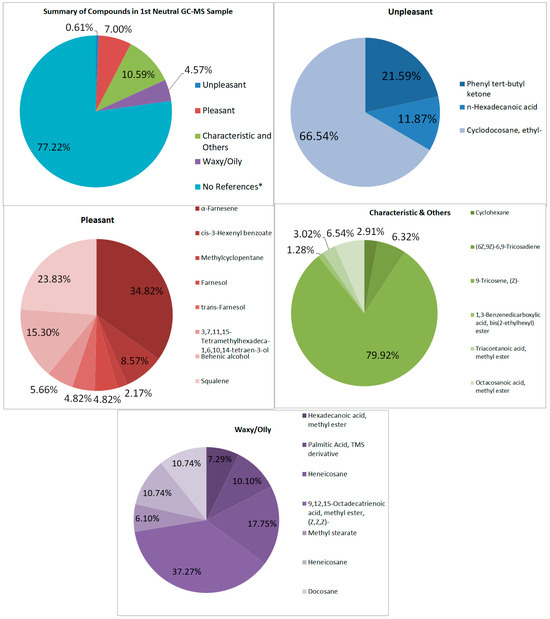
Figure 7.
Summary and listed GC–MS compounds of first neutral sample with listed percentages for each classification of compound (No references* are compounds that cannot be found references for its odor).
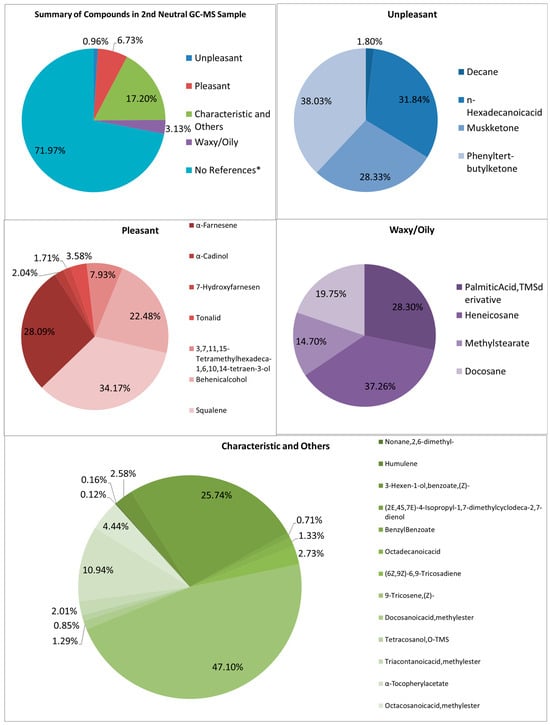
Figure 8.
Summary and listed GC–MS compounds of second neutral sample with listed percentages for each classification of compound (No references* are compounds that cannot be found references for its odor).
The FTIR and GC–MS analyses showed strong signals, indicating the presence of chemical compounds despite the weak olfactory perception. This is because not all volatile organic compounds have low odor thresholds; some compounds may be present in relatively high concentrations but contribute little to human scent detection because they lack strong odor activity or are suppressed by other components [36,37]. In FTIR, the strong polyethylene and EPDM similarities suggest that the samples may contain non-volatile or weakly volatile constituents, which generate clear spectral signals but do not easily volatilize to produce a noticeable aroma. Similarly, the GC–MS results show distinct compound peaks with notable percentages for both pleasant and unpleasant odorants, yet the overall scent remained faint. This may be due to matrix effects or competitive interactions, where higher-percentage compounds dominate but have inherently low odor impact, masking or suppressing the perception of minor but more odor-active volatiles. Additionally, differences in volatility, molecular weight, and odor thresholds contribute to why the graphs show significant signals while the human nose detects little to no smell [38,39]. Therefore, the neutral category highlights the important distinction between instrumental detection of chemical composition and human sensory perception, underscoring that strong analytical signals do not always correlate with strong olfactory intensity.
3.5. Statistical Significance of Yield to Parameters
With the compounds known through GC–MS and FTIR analysis, the relationship between yield and parameters is crucial in confirming the credibility of the extraction optimization. Using multiple linear regression, comparing the relationship between extract yield, temperature, pressure, and time, the R-squared value is 0.34899, which translates to a moderate relationship between the three parameters and the yield; however, considering the p-value, temperature is most statistically significant, having the value of 0.03413, compared to pressure and time. Typically, raising the temperature boosts extraction yield by increasing the diffusion rate and solubility of soluble phytochemicals in the extraction solvent. It also helps the solvent penetrate plant tissues more easily by reducing its viscosity and surface tension, which benefits the interaction between the solvent, solutes, and plant materials [40], which confirms the dependence of increased temperature in extraction. However, increasing the temperature results in more unwanted compounds found in unpleasant samples. Additionally, pressure, according to Hartati et al. [38], is still a factor that increases the yield, though tested more under room temperature conditions, which means that data from other studies suggest that pressure still contributes to extraction yield but needs to be explored more thoroughly.
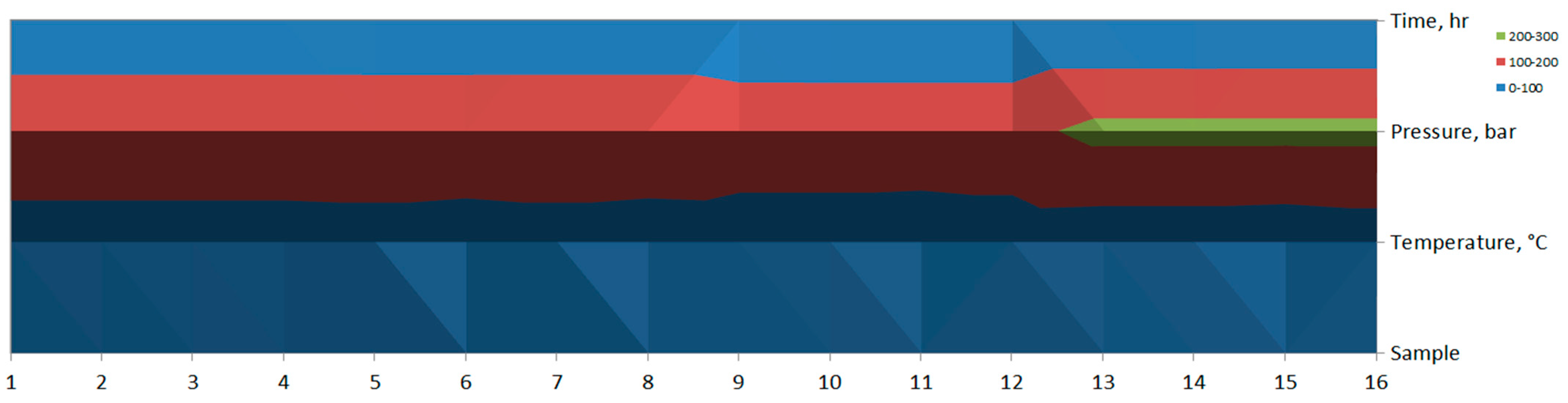
In the contour plot (see Figure 9), the changes in color bands suggest a relationship between temperature, pressure, and time across different samples. There seems to be a significant region at higher pressure (green, 200–300 bar) occurring around Sample 14 and Sample 15.
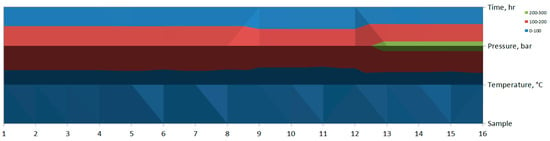
Figure 9.
Contour plot of ANOVA analysis.
In a post hoc analysis with a Bonferroni correction of 0.0833333, the two-tailed p-value of the T Test with Two-Sample Assuming Unequal Variances of 3.04 × 10−17, 3.02 × 10−17, 1.05 × 10−12, 2.01 × 10−16, 6.96 × 10−17, and 3.51 × 10−17 is significantly smaller than 0.0833333 (See Table 4 and Table 5 for full regression and ANOVA). Therefore, all comparisons would be considered statistically significant when using the Bonferroni correction threshold of 0.0833333.

Table 4.
Regression statistics of yield vs. temperature vs. pressure vs. time.

Table 5.
ANOVA of yield vs. temperature vs. pressure vs. time.
4. Conclusions and Recommendations
The researchers explored methods to optimize the extraction of the quality oil of Jasminum sambac (L.) using the Supercritical Fluid Extraction method with the respective optimized range of parameters. The four key parameters affecting the scent of the oil are drying method, temperature, pressure, and time. Temperature and pressure had the most significant impact on the oil’s quality. At 35 °C, the oil is fragrant and sweet; at 40 °C, the oil has a pleasant fragrance but less sweetness; and temperatures above 40 °C result in burnt-smelling oil, with 45 °C being the point at which the oil persists in having unpleasant odor compounds from the GC–MS analysis.
Higher pressure also enhances the aroma and yield of the oil. Lower temperatures and pressures produce a floral and sweet quality but with a lower yield; however, generally, higher temperatures yield more extraction. On another note, extracts are not purely oil; they contain a mixture of wax which requires another process, namely winterization, which in turn will reduce the yield.
The drying method also affects the overall output. Plucking petals before drying often results in unpleasant-quality oils, while those plucked after drying consistently have pleasant aromas. Using a blow dryer for drying can damage the petals, leading to unpleasant-smelling oils. Overnight air drying gave mixed results, but no drying method resulted in better and more consistent aromas.
Author Contributions
Conceptualization, L.C. and L.T.; Methodology, A.J.S.M., A.M.A. and L.T.; Software, L.C.; Validation, A.M.A. and L.T.; Resources, L.T.; Writing—original draft, A.J.S.M.; Writing—review & editing, A.J.S.M., A.M.A. and L.T.; Supervision, A.M.A., L.C., D.M. and L.T.; Project administration, L.C., D.M. and L.T.; Funding acquisition, D.M. and L.T. All authors have read and agreed to the published version of the manuscript.
Funding
We acknowledge the CRADLE project Department Capital of Science and Technology for providing funds.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
Appendix A. List of Figures
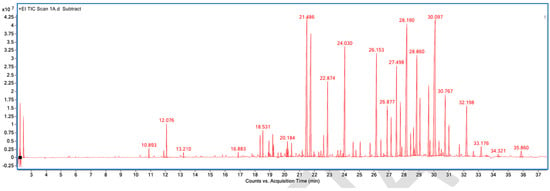
Figure A1.
GC–MS counts vs. acquisition time of the first pleasant-smelling sample.
Figure A1.
GC–MS counts vs. acquisition time of the first pleasant-smelling sample.
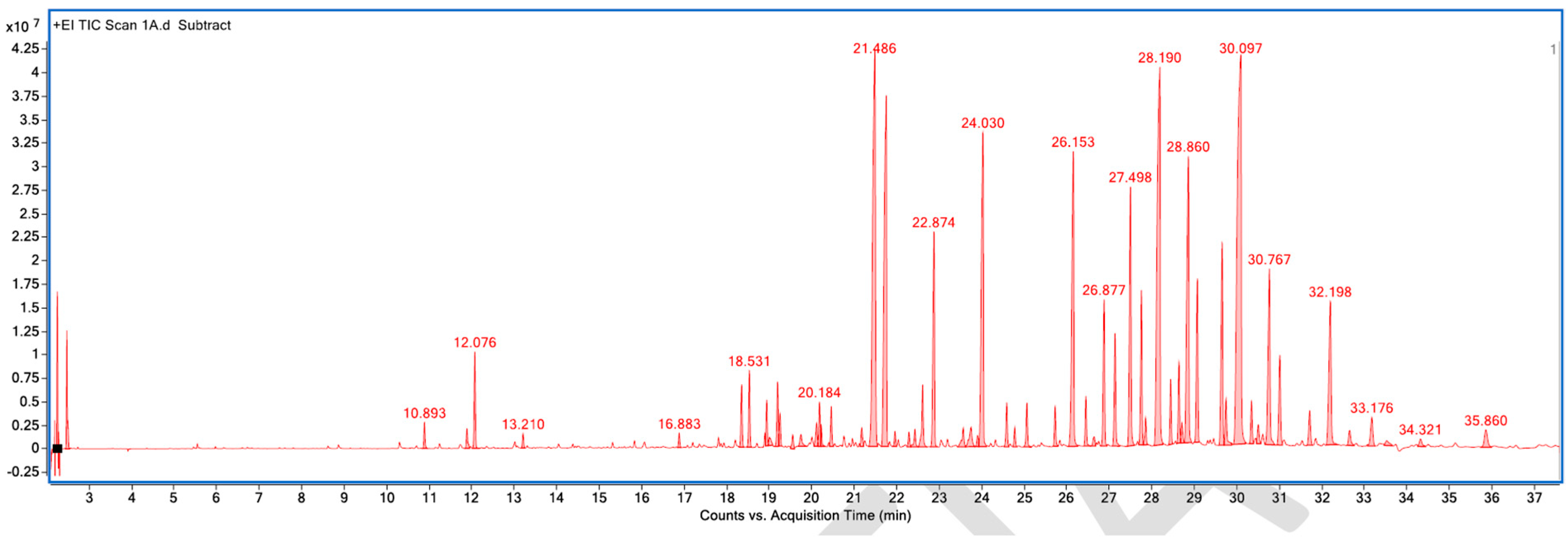
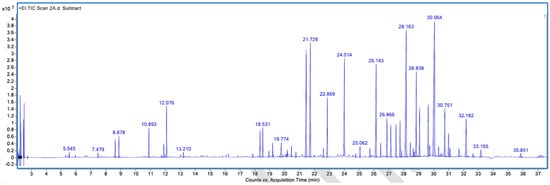
Figure A2.
GC–MS counts vs. acquisition time of the first unpleasant-smelling sample.
Figure A2.
GC–MS counts vs. acquisition time of the first unpleasant-smelling sample.
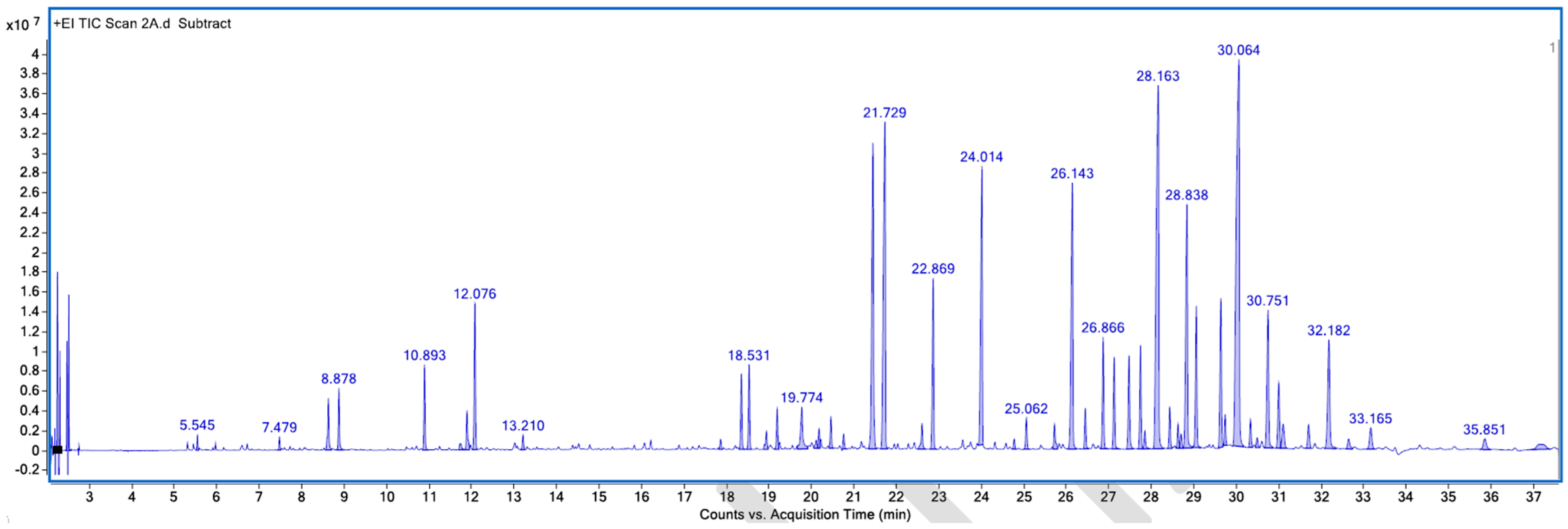
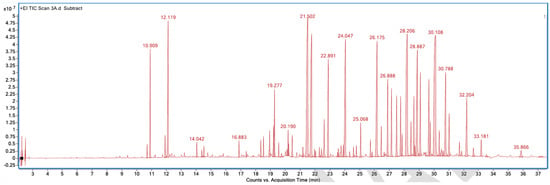
Figure A3.
GC–MS counts vs. acquisition time of the first neutral scent sample.
Figure A3.
GC–MS counts vs. acquisition time of the first neutral scent sample.
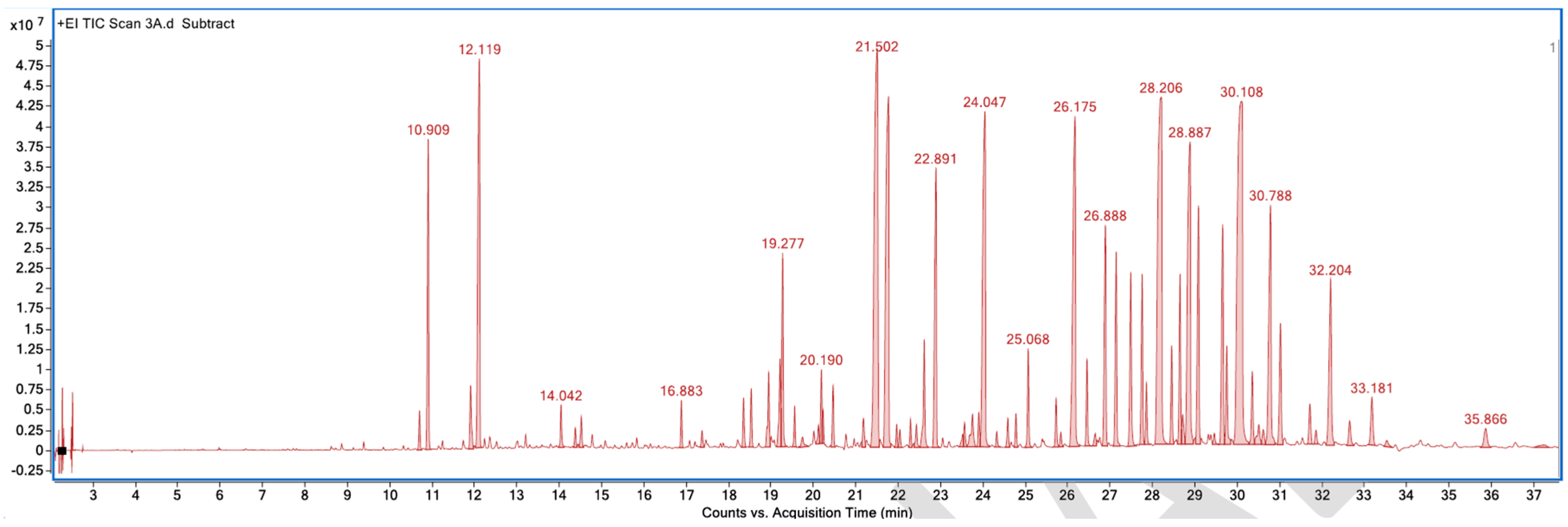
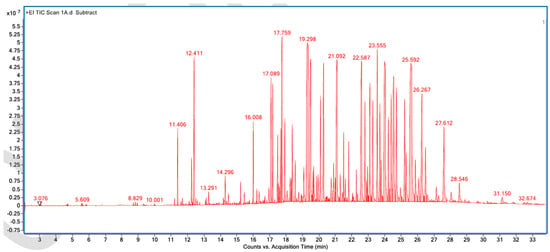
Figure A4.
GC–MS counts vs. acquisition time of the second pleasant sample.
Figure A4.
GC–MS counts vs. acquisition time of the second pleasant sample.
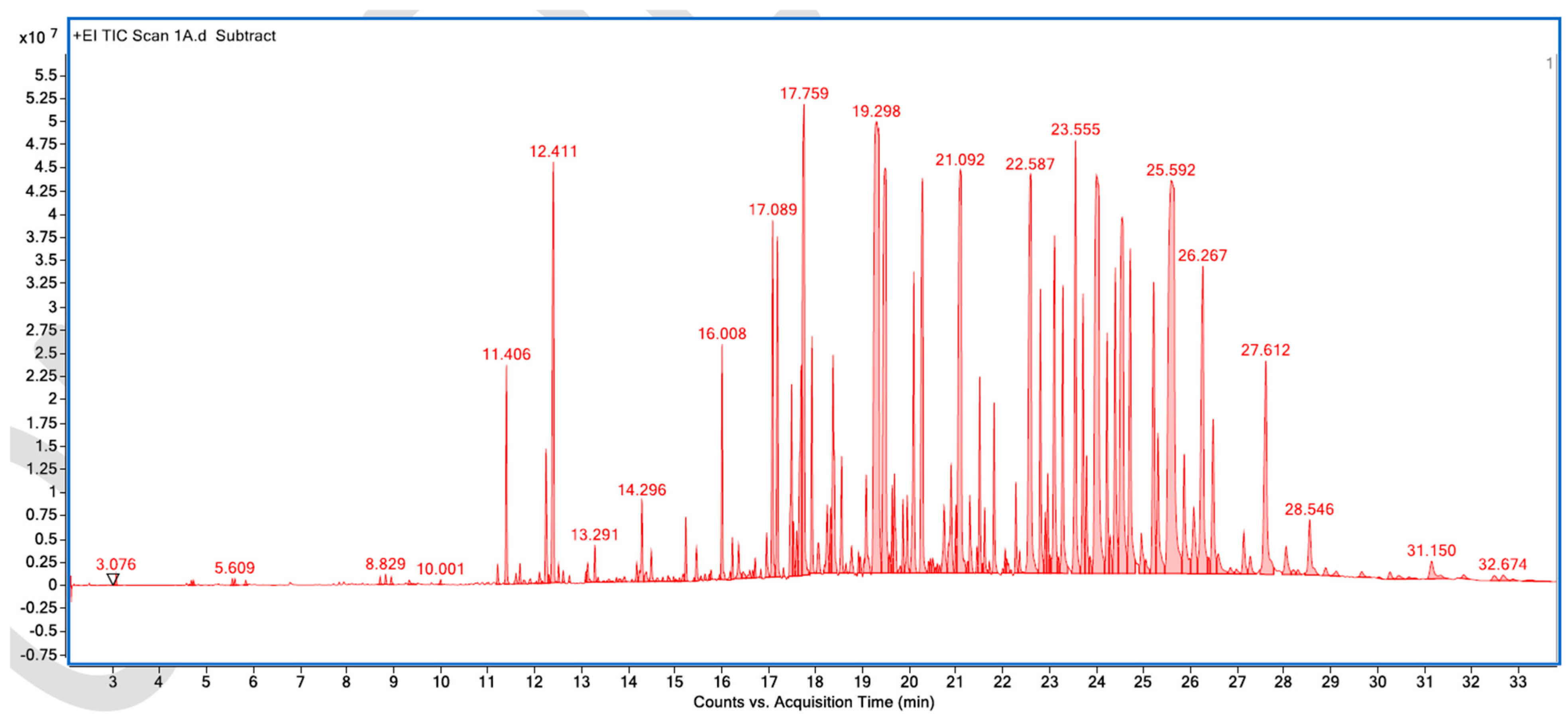
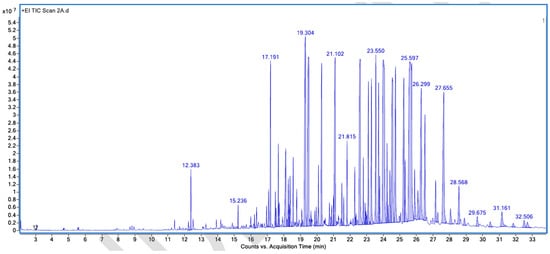
Figure A5.
GC–MS counts vs. acquisition time of the first unpleasant-smelling sample.
Figure A5.
GC–MS counts vs. acquisition time of the first unpleasant-smelling sample.
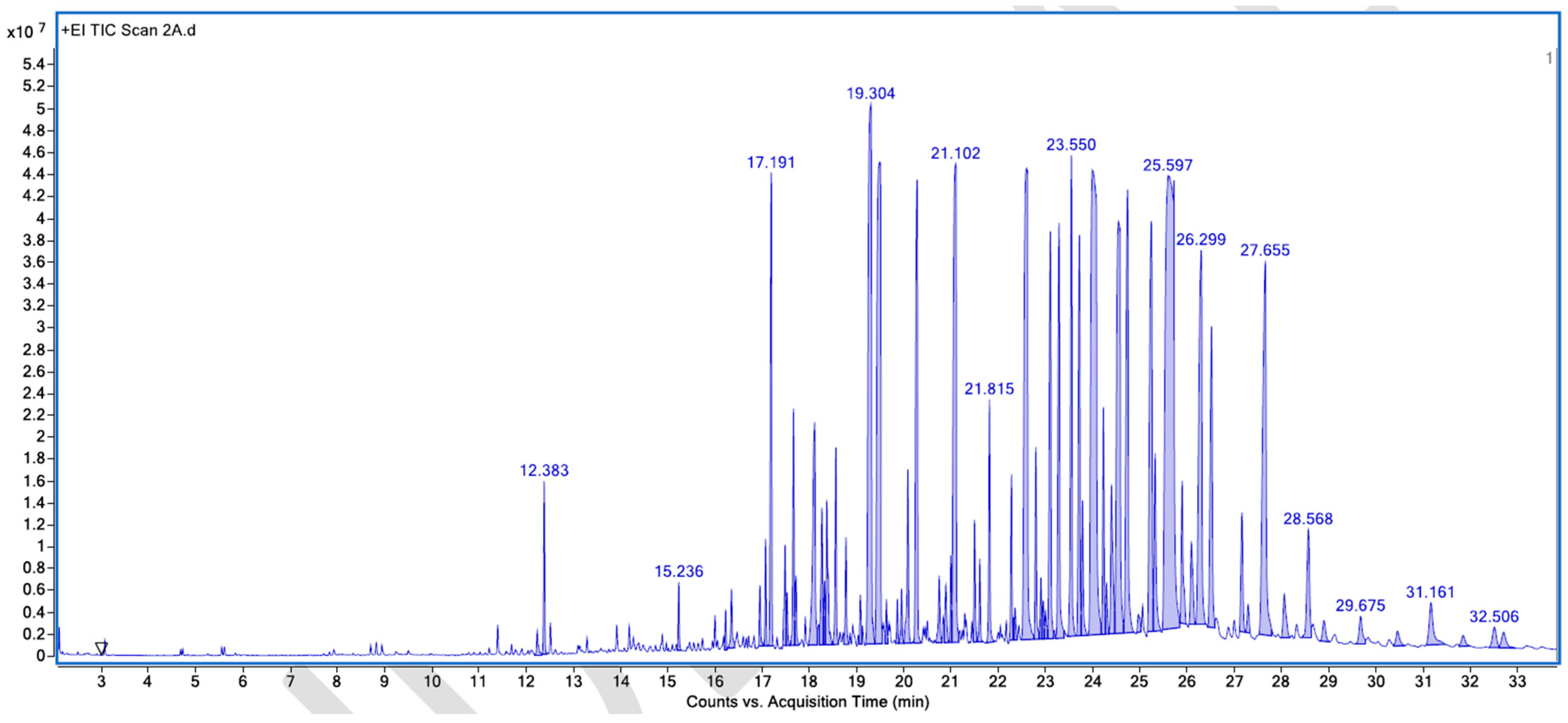
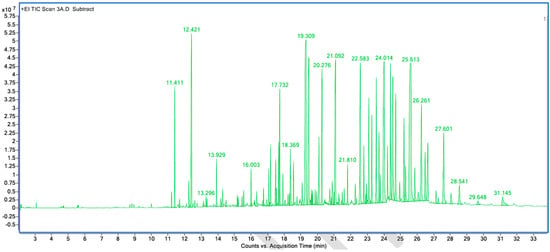
Figure A6.
GC–MS counts vs. acquisition time of the second neutral scent sample.
Figure A6.
GC–MS counts vs. acquisition time of the second neutral scent sample.
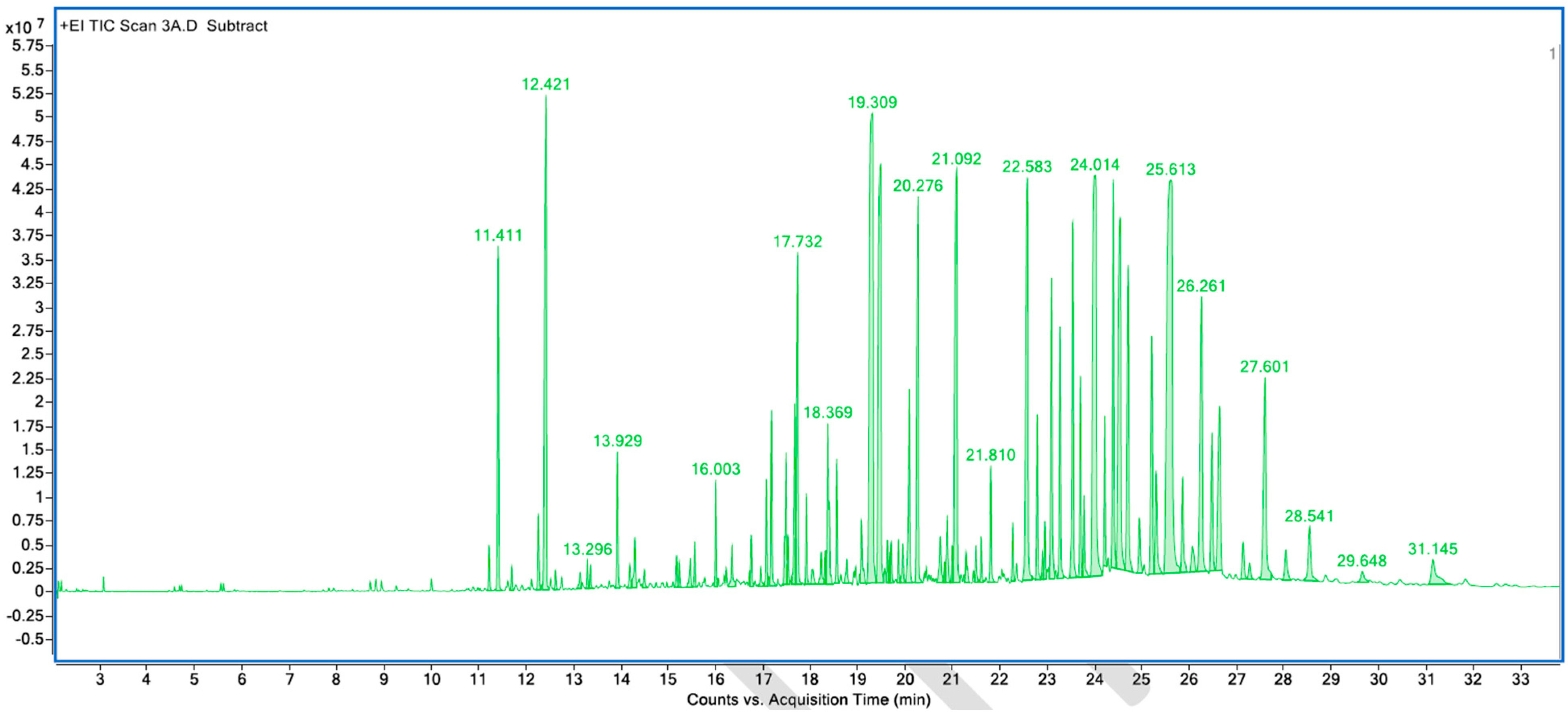
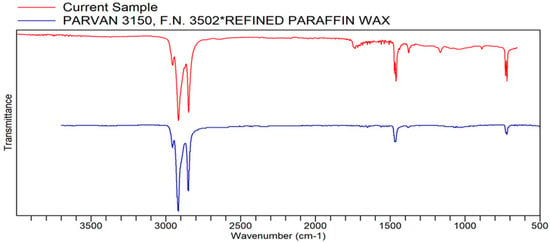
Figure A7.
FTIR transmittance–wavenumber graph of first pleasant-smelling sample.
Figure A7.
FTIR transmittance–wavenumber graph of first pleasant-smelling sample.
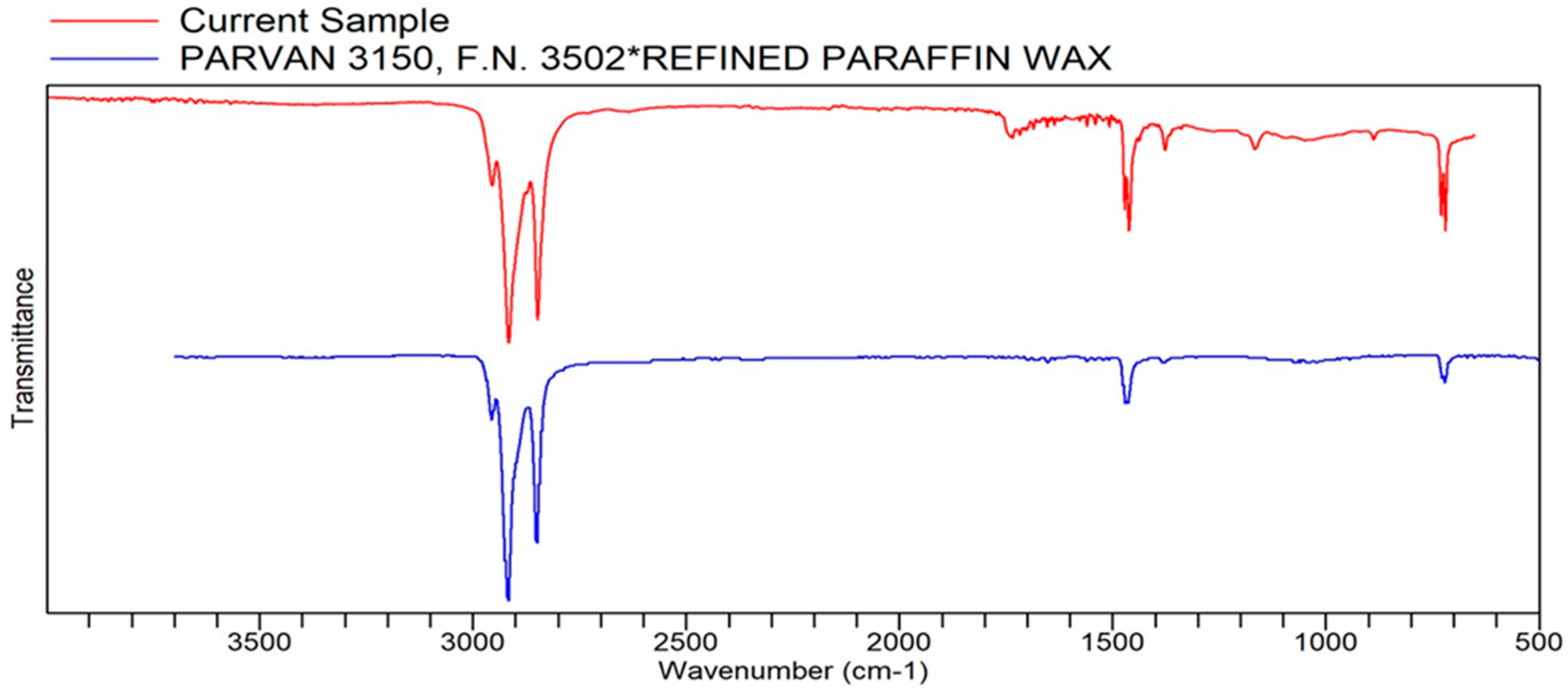
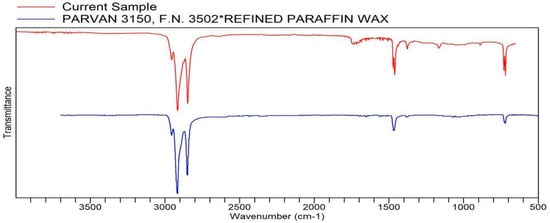
Figure A8.
FTIR transmittance–wavenumber graph of first unpleasant-smelling sample.
Figure A8.
FTIR transmittance–wavenumber graph of first unpleasant-smelling sample.
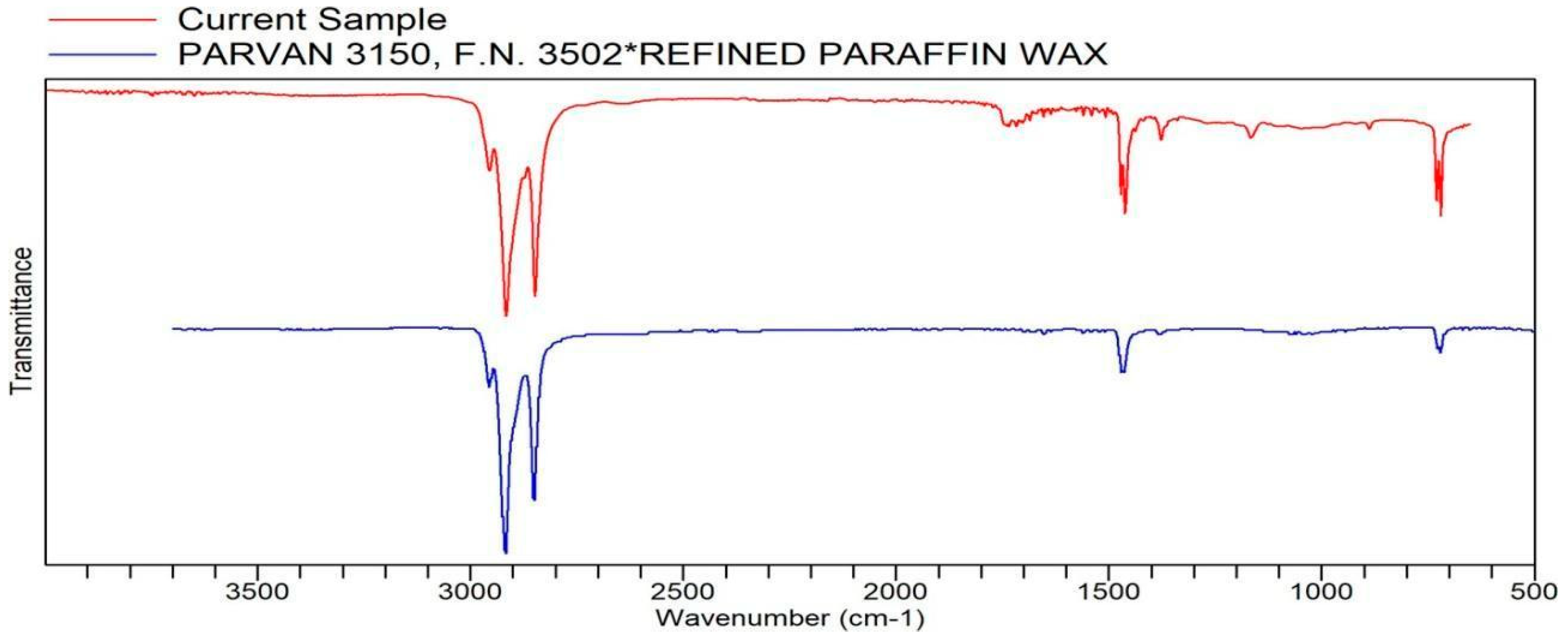
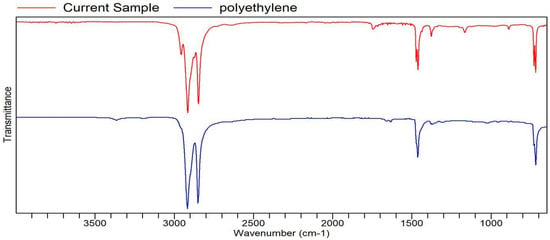
Figure A9.
FTIR transmittance–wavenumber graph of first neutral smell sample.
Figure A9.
FTIR transmittance–wavenumber graph of first neutral smell sample.
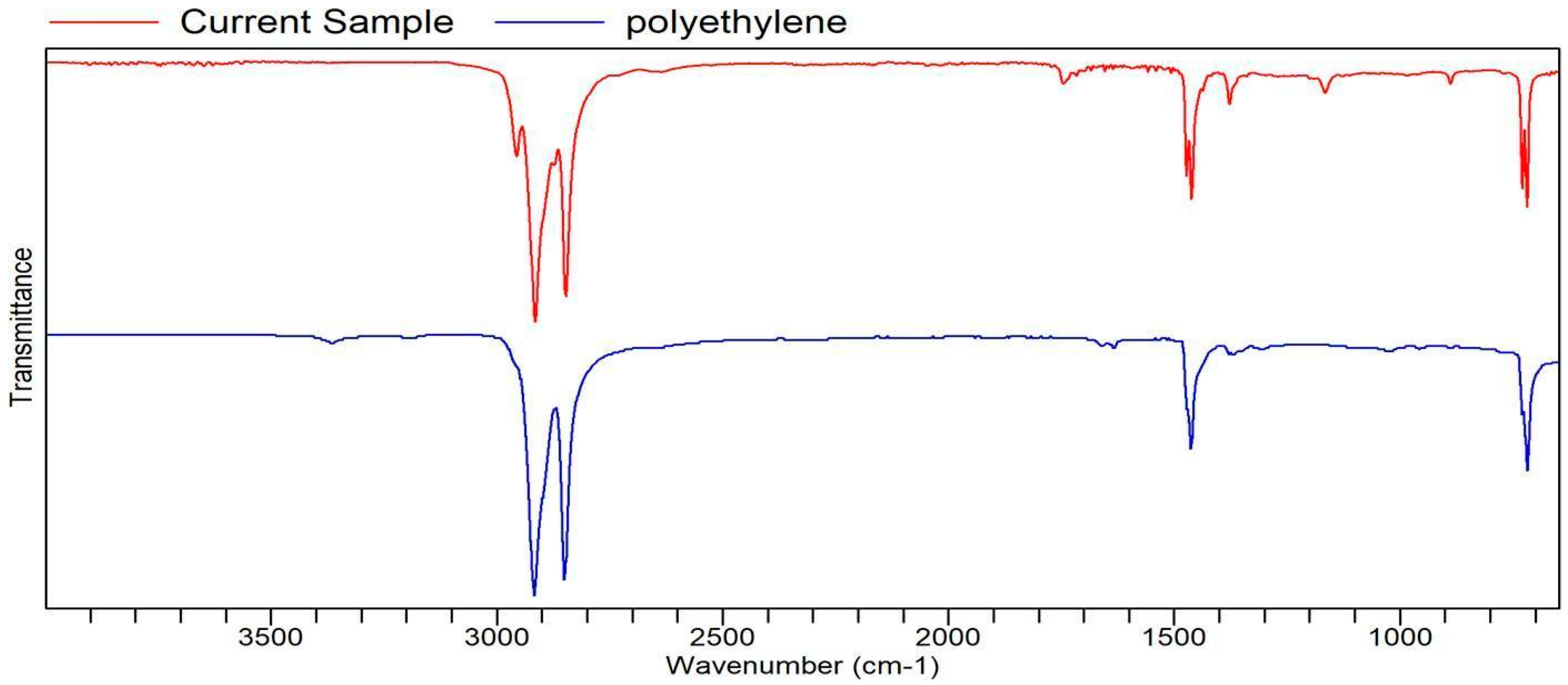
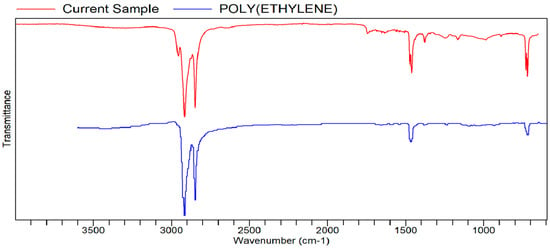
Figure A10.
FTIR transmittance–wavenumber graph of second pleasant-smelling sample.
Figure A10.
FTIR transmittance–wavenumber graph of second pleasant-smelling sample.
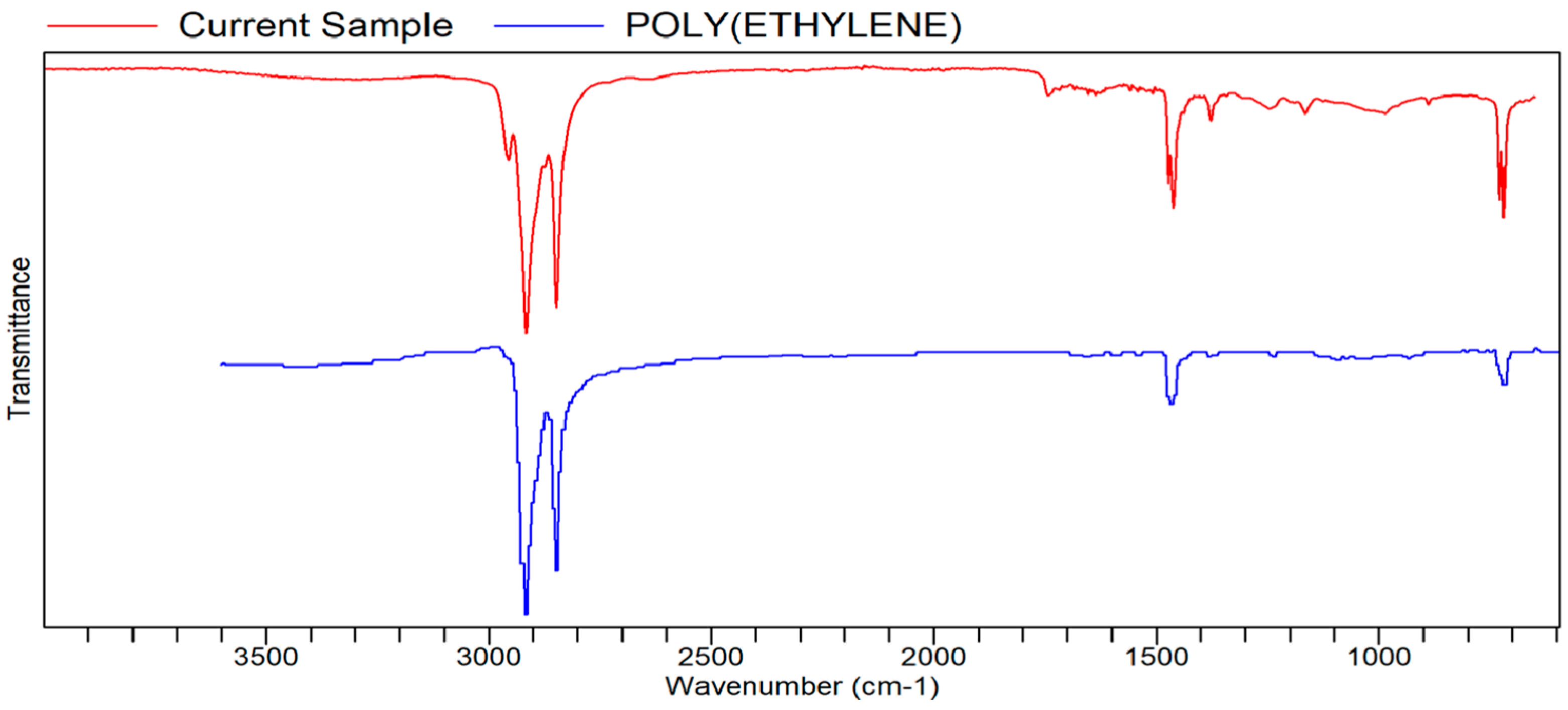
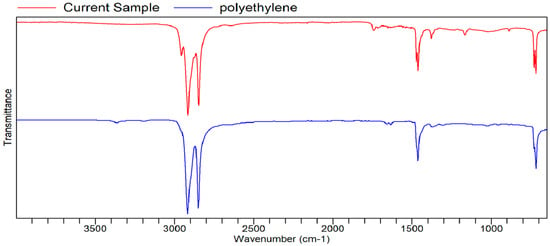
Figure A11.
FTIR transmittance–wavenumber graph of second unpleasant-smelling sample.
Figure A11.
FTIR transmittance–wavenumber graph of second unpleasant-smelling sample.
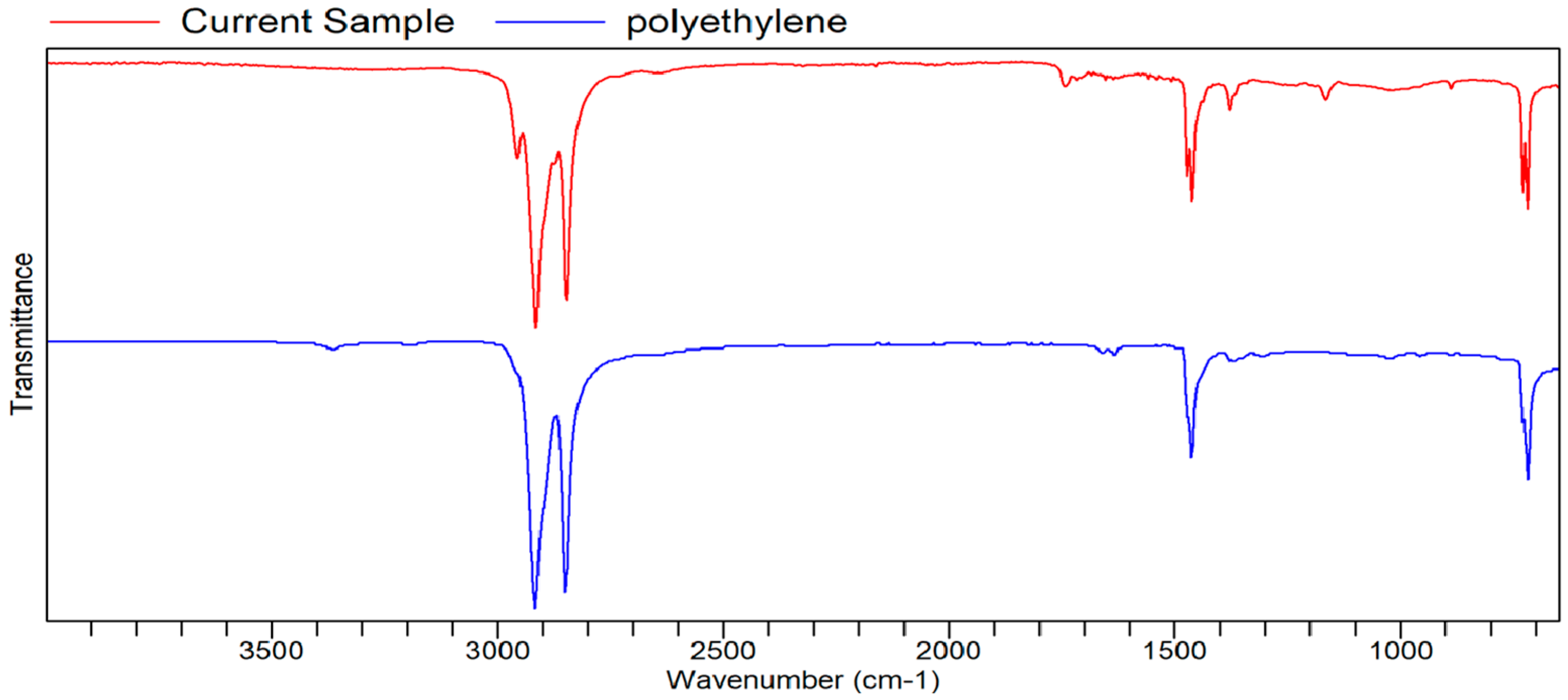
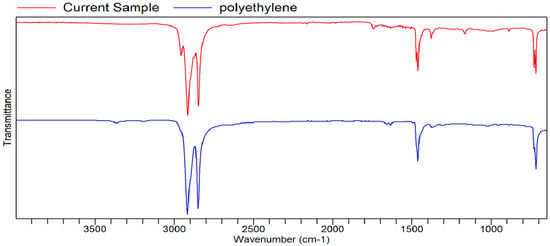
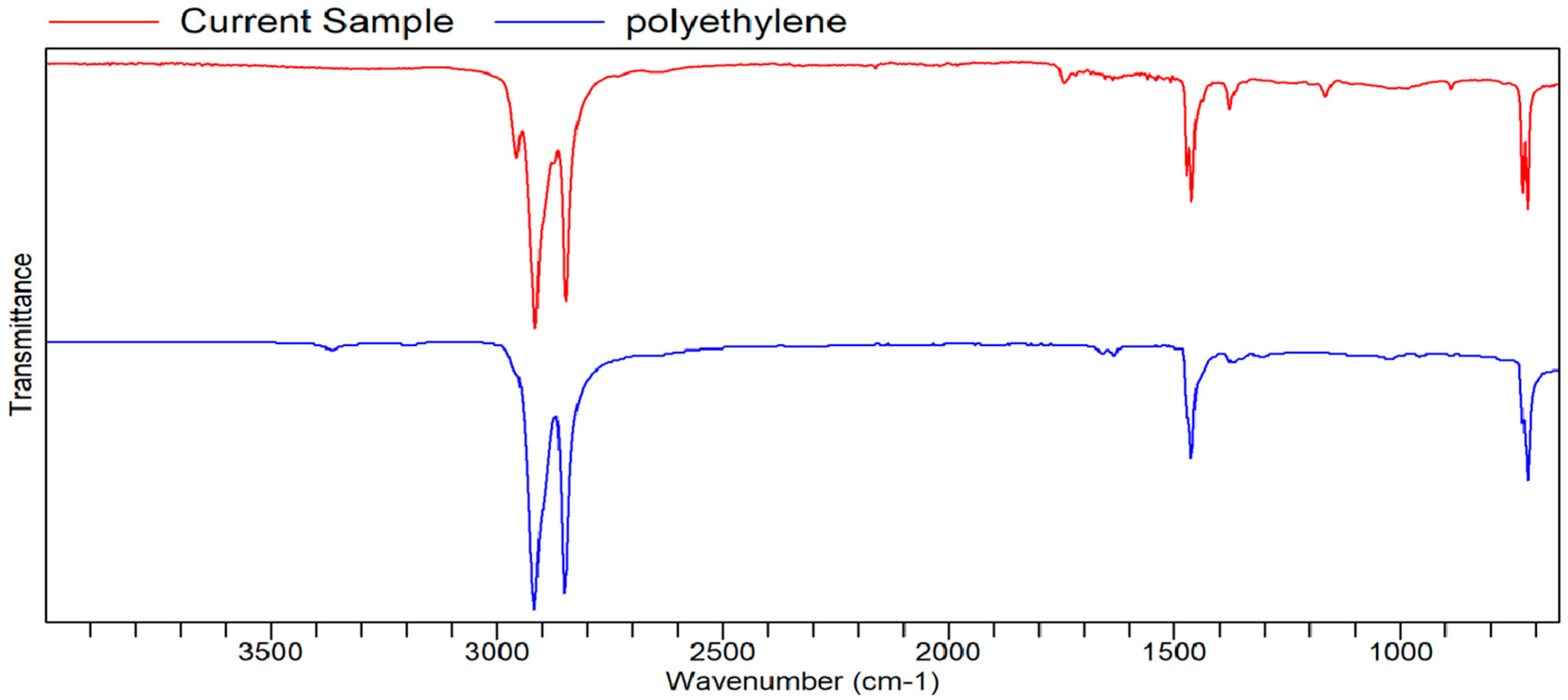
Figure A12.
FTIR transmittance–wavenumber graph of second neutral smell sample.
Figure A12.
FTIR transmittance–wavenumber graph of second neutral smell sample.
Appendix B. List of Tables

Table A1.
GC–MS summary of significant compounds and area of the first pleasant-smelling sample.
Table A1.
GC–MS summary of significant compounds and area of the first pleasant-smelling sample.
| Notes | Area | Percent (%) | |
|---|---|---|---|
| Unpleasant-Smelling Compounds | |||
| N/A | 0 | 0 | |
| Total | 0 | 0 | |
| Pleasant-Smelling Compounds | |||
| α-Farnesene | sweet, woody, and fruity smell | 5,180,291.48 | 0.2553 |
| α-Cadinol | floral, fresh, and fruity odor of tea flowers | 3,027,782.37 | 0.1492 |
| cis-3-Hexenyl benzoate | green, herbaceous, floral, and woody odor | 4,828,245.4 | 0.2380 |
| 3,7,11,15-Tetramethylhexadeca-1,6,10,14-tetraen-3-ol | floral | 14,087,864.88 | 0.6944 |
| Behenic alcohol | mild, pleasant, aromatic | 10,400,228.42 | 0.5127 |
| 3,209,425.88 | 0.1582 | ||
| Squalene | “faint agreeable odor” | 73,269,459.38 | 3.6116 |
| Total | 114,003,297.8 | 5.6195 | |
| Characteristic and Others | |||
| Germacrene D-4-ol | characteristic odor | 21,033,012.76 | 1.0368 |
| (6Z,9Z)-6,9-Tricosadiene | pheromone component | 5,128,708.79 | 0.2528 |
| 3,224,764.08 | 0.1590 | ||
| 5,199,288.16 | 0.2563 | ||
| Phthalic acid, di(2-propylpentyl) ester | “characteristic odor” | 10,398,735.97 | 0.5126 |
| 1,3-Benzenedicarboxylic acid, bis(2-ethylhexyl) ester | “slight odor” | 3,058,139.39 | 0.1507 |
| Octacosanoic acid, methyl ester | characteristic | 10,648,040.61 | 0.5249 |
| 3-Methylhentriacontane | pheromone component | 61,385,160.33 | 3.0258 |
| Triacontanoic acid, methyl ester | “characteristic odor” | 5,788,126.16 | 0.2853 |
| Total | 125,863,976.3 | 6.2041 | |
| Waxy or Oily Compounds | |||
| Hexadecanoic acid, methyl ester | weak “waxy” odor | 3,011,745.59 | 0.1485 |
| Palmitic Acid, TMS derivative, | slight waxiness | 16,795,154.37 | 0.8279 |
| Heneicosane | waxy | 15,165,101.58 | 0.7475 |
| waxy | 8,895,049.09 | 0.4385 | |
| Docosane | minimal waxiness, nearly odorless | 8,895,049.09 | 0.4385 |
| Tricosane | waxy | 151,194,085 | 7.4527 |
| Linolenic acid, methyl ester | oily, fatty, sometimes woodsy | 7,466,982.14 | 0.3681 |
| Linolenic acid | oily, fatty, sometimes woodsy | 7,466,982.14 | 0.3681 |
| Total | 218,890,149 | 10.7896 |

Table A2.
GC–MS summary of significant compounds and area of the second pleasant-smelling sample.
Table A2.
GC–MS summary of significant compounds and area of the second pleasant-smelling sample.
| Notes | Area | Percent (%) | |
|---|---|---|---|
| Unpleasant-Smelling Compounds | |||
| Decane, 4-methyl | pungent, acrid | 776,589.03 | 0.0170 |
| 1-Nonene, 4,6,8-trimethyl | unpleasant-smelling | 1,427,263.6 | 0.0312 |
| 2,2,4-Trimethyl-1,3-pentanediol diisobutyrate | musty odor | 3,007,628.3 | 0.0658 |
| Total | 5,211,480.93 | 0.1140 | |
| Pleasant-Smelling Compounds | |||
| Linalool | floral, citric, fresh, and sweet | 1,001,785.3 | 0.0219 |
| 1-Dodecanol, 2-hexyl | with a mild sweet odor | 1,914,146.1 | 0.0419 |
| Methyl anthranilate | grape-like smell | 1,730,390.1 | 0.0379 |
| α-Farnesene | sweet, woody, and fruity smell. | 38,575,207 | 0.8438 |
| Naphthalene, 1,2,3,4,4a,5,6,8aoctahydro-7-methyl-4-methylene-1-(1-methylethyl)-,(1α,4aβ,8aα)- | piquant | 2,371,798.2 | 0.0519 |
| Viridiflorol | sweet, green, herbal, fruity, tropical, minty odor. | 2,024,211 | 0.0443 |
| .tau.-Muurolol | herbal type odor; slight spicy | 4,878,448 | 0.1067 |
| α-Cadinol | floral, fresh, and fruity odor of tea flowers | 6,393,944.6 | 0.1399 |
| 7-Hydroxyfarnesen | citrus, flowery | 741,259.32 | 0.0162 |
| trans-Farnesol | floral scent | 1,549,129.3 | 0.0339 |
| Benzoic acid, 2-propenyl ester | fruity | 18,850,949 | 0.4123 |
| Benzyl Benzoate | weak, sweet-balsamic odor | 6,799,447.1 | 0.1487 |
| 3,7,11,15-Tetramethylhexadeca-1,6,10,14-tetraen-3-ol | floral | 75,092,203 | 1.6426 |
| (E)-Cinnamyl benzoate | sweet smell | 9,855,007.8 | 0.2156 |
| (Z)-Cinnamyl benzoate | sweet smell | 9,855,007.8 | 0.2156 |
| Behenic alcohol | mild, pleasant, aromatic | 64,254,257 | 1.4055 |
| Total | 245,887,190.6 | 5.3785 | |
| Characteristic and Others | |||
| Nonane, 2,6-dimethyl- | camphoreous | 931,876.01 | 0.0204 |
| Octane, 3,3-dimethyl- | VOC | 931,876.01 | 0.0204 |
| Cyclooctane, 1,4-dimethyl-, trans | VOC | 1,004,860 | 0.0220 |
| 1,222,376.3 | 0.0267 | ||
| Octadecyl octyl ether | octadecyls are odor maskers | 1,205,706.7 | 0.0264 |
| 3-Hexen-1-ol, benzoate, (Z)- | intense grassy-green odor | 28,400,595 | 0.6212 |
| Germacrene D-4-ol | characteristic odor | 110,319,891 | 2.4131 |
| Epiglobulol | characteristic | 2,024,211 | 0.0443 |
| Dodecanoic acid, TMS derivative | faint odor of bay oil | 1,479,934.6 | 0.0324 |
| Octadecanoic acid | “mild odor” | 18,301,603 | 0.4003 |
| (6Z,9Z)-6,9-Tricosadiene | pheromone component | 27,983,459 | 0.6121 |
| 14,381,094 | 0.3146 | ||
| 16,577,987 | 0.3626 | ||
| 9-Tricosene, (Z)- | pheromone component | 367,301,953 | 8.0343 |
| Total | 592,067,422.6 | 12.9508 | |
| Waxy Compounds | |||
| Myristic acid, TMS derivative | odorless, but some blends are oily | 10,978,338 | 0.2401 |
| Hexadecanoic acid, methyl ester | weak “waxy” odor | 42,472,332 | 0.9290 |
| Palmitic Acid, TMS derivative | slight waxiness | 70,031,808 | 1.5319 |
| Heneicosane | waxy | 65,893,071 | 1.4413 |
| 24,646,580 | 0.5391 | ||
| 9,12,15-Octadecatrienoic acid, methyl ester, (Z,Z,Z)- | oily | 162,366,628 | 3.5516 |
| Methyl stearate | waxy | 44,168,233 | 0.9661 |
| Tricosane | waxy | 210,780,830 | 4.6106 |
| Total | 631,337,820 | 13.8098 |

Table A3.
GC–MS summary of significant compounds and area of the first unpleasant-smelling sample.
Table A3.
GC–MS summary of significant compounds and area of the first unpleasant-smelling sample.
| Notes | Area | Percent (%) | |
|---|---|---|---|
| Unpleasant-Smelling Compounds | |||
| Musk ketone | highly tenacious musky aroma with a discreet animal note | 2,144,687.96 | 0.1525 |
| α-Linolenic acid, TMS derivative | some references say it is fishy/oily; some say it is odorless | 3,179,637.28 | 0.2260 |
| Tritriacontane | range in odor from odorless to a fuel-like odor | 39,412,448.51 | 2.8017 |
| Total | 44,736,773.75 | 3.1802 | |
| Pleasant-Smelling Compounds | |||
| Squalene | “faint agreeable odor” | 22,692,839.01 | 1.6132 |
| Benzoic Acid, TMS derivative | faint, pleasant odor | 2,137,985.27 | 0.1520 |
| 8-Hydroxylinalool | citrus-like, sweet, soapy, and lemon-like | 11,186,775.44 | 0.7952 |
| α-Farnesene | sweet, woody, and fruity smell | 15,990,851.96 | 1.1368 |
| Methylcyclopentane | sweet | 22,305,310.15 | 1.5856 |
| 4,107,283.42 | 0.2920 | ||
| α-epi-Muurolol | herbal | 3,086,232.01 | 0.2194 |
| α-Cadinol | floral, fresh, and fruity odor of tea flowers | ||
| Total | 81,507,277.26 | 5.7942 | |
| Characteristics and Others | |||
| Octacosanoic acid, methyl ester | characteristic | 6,529,256.25 | 0.4641 |
| 3-Methylhentriacontane | pheromone component | 40,871,050.72 | 2.9054 |
| Benzeneacetaldehyde | grassy | 2,046,250.26 | 0.1455 |
| 1,6,10-Dodecatrien-3-ol, 3,7,11-trimethyl-, (E)- | fresh bark odor | 2,365,617.83 | 0.1682 |
| 3-Hexen-1-ol, benzoate, (Z)- | intense grassy-green odor | 9,000,686.39 | 0.6398 |
| α-Tocopheryl acetate | characteristic | 9,231,315.14 | 0.6562 |
| Cyclohexane | sweet, pungent odor | 10,880,395.17 | 0.7735 |
| 10,797,874.96 | 0.7676 | ||
| Total | 91,722,446.72 | 6.5203 | |
| Waxy Compounds | |||
| Palmitic Acid, TMS derivative | slight waxiness | 17,838,641.56 | 1.2681 |
| Heneicosane | waxy | 8,440,458.89 | 0.6000 |
| 6,474,638.67 | 0.4603 | ||
| Linolenic acid | 15,983,006.75 | 1.1362 | |
| Total | 48,736,745.87 | 3.4646 |

Table A4.
GC–MS summary of significant compounds and area of the second unpleasant-smelling sample.
Table A4.
GC–MS summary of significant compounds and area of the second unpleasant-smelling sample.
| Notes | Area | Percent (%) | |
|---|---|---|---|
| Unpleasant-Smelling Compounds | |||
| Trichloroaceticacid, hexadecylester | pungent, sharp | 511,976.24 | 0.0116 |
| 2,2,4-Trimethyl-1,3-pentanedioldiisobutyrate | musty odor | 4,627,215.6 | 0.1050 |
| n-Hexadecanoicacid | ‘rancid’ and ‘pungent’ odors | 11,956,959 | 0.2713 |
| Tritriacontane | range in odor from odorless to a fuel-like odor | 178,875,511 | 4.0587 |
| Total | 195,971,661.8 | 4.4466 | |
| Pleasant-Smelling Compounds | |||
| BenzoicAcid,TMSderivative | faint, pleasant odor | 1,186,255.2 | 0.0269 |
| 1-Decanol,2-hexyl- | mild, sweet odor | 1,256,663.2 | 0.0285 |
| α-Farnesene | sweet, woody, and fruity smell | 4,894,050.6 | 0.1110 |
| 1-Decanol,2-hexyl- | mild, sweet odor | 951,428.3 | 0.0216 |
| cis-3-Hexenylbenzoate | green, herbaceous, floral, and woody odor | 5,362,932.1 | 0.1217 |
| .tau.-Muurolol | herbal type odor; slight spicy | 734,662.17 | 0.0167 |
| Cubenol | spicy and herbal | 567,901.47 | 0.0129 |
| α-Cadinol | floral, fresh, and fruity odor of tea flowers | 2,255,064 | 0.0512 |
| trans-Farnesol | floral scent | 3,979,945.1 | 0.0903 |
| 3,7,11,15-Tetramethylhexadeca-1,6,10,14-tetraen-3-ol | floral | 15,117,986 | 0.3430 |
| 1-Decanol,2-hexyl- | mild, sweet odor | 1,256,663.2 | 0.0285 |
| Behenicalcohol | mild, pleasant, aromatic | 34,546,007 | 0.7838 |
| 12,859,135 | 0.2918 | ||
| Squalene | “faint agreeable odor” | 102,663,839 | 2.3294 |
| Total | 187,632,532.3 | 4.2574 | |
| Characteristic and Others | |||
| Nonane,2,6-dimethyl- | camphoreous | 676,510.19 | 0.0153 |
| Dodecane,4,6-dimethyl- | strong sweet-corn-like aroma | 291,572.1 | 0.0066 |
| GermacreneD-4-ol | characteristic odor | 26,646,212 | 0.6046 |
| Dodecanoicacid,TMSderivative | faint odor of bay oil | 684,929.83 | 0.0155 |
| 1-Hexacosene | mild | 2,414,809.6 | 0.0548 |
| Octadecanoicacid | “mild odor” | 29,731,365 | 0.6746 |
| Tetracosanol,O-TMS | characteristic | 11,102,856 | 0.2519 |
| 1,3-Benzenedicarboxylicacid,bis(2-ethylhexyl)ester | “slight odor” | 7,125,228.9 | 0.1617 |
| Methyl21-methyl-hexacosanoate | fruity-type odor | 5,661,947.4 | 0.1285 |
| Octacosanoicacid,methylester | characteristic | 35,703,227 | 0.8101 |
| Triacontanoicacid,methylester | “characteristic odor” | 17,341,539 | 0.3935 |
| Oleanolicacid2TMS | characteristic | 10,807,484 | 0.2452 |
| Ursolicaldehyde | characteristic | 9,729,875 | 0.2208 |
| Heptadecanoicacid,TMSderivative | pheromone component | 1,297,625.3 | 0.0294 |
| 9-Tricosene,(Z)- | pheromone component | 4,600,337.6 | 0.1044 |
| 240,988,707 | 5.4680 | ||
| (6Z,9Z)-6,9-Tricosadiene | pheromone component | 8,844,206.6 | 0.2007 |
| 6,574,194.6 | 0.1492 | ||
| 9,318,822.5 | 0.2114 | ||
| Total | 429,541,449.6 | 9.7462 | |
| Waxy Odors | |||
| Hexadecanoicacid,methylester | weak “waxy” odor | 5,185,499.6 | 0.1177 |
| PalmiticAcid,TMSderivative | slight waxiness | 1,617,083.9 | 0.0367 |
| 89,000,228 | 2.0194 | ||
| Heneicosane | waxy | 38,354,308 | 0.8703 |
| 9,12,15-Octadecatrienoicacid,methylester,(Z,Z,Z)- | oily | 13,691,718 | 0.3107 |
| Phthalicacid,di(2-propylpentyl)ester | “characteristic odor” | 20,331,662 | 0.4613 |
| Total | 168,180,499.5 | 3.8160 |

Table A5.
GC–MS summary of significant compounds and area of the first neutral odor sample.
Table A5.
GC–MS summary of significant compounds and area of the first neutral odor sample.
| Notes | Area | Percent (%) | |
|---|---|---|---|
| Unpleasant-Smelling Compounds | |||
| Phenyl tert-butyl ketone | unpleasant | 9,004,534.49 | 0.2623 |
| n-Hexadecanoic acid | ‘rancid’ and ‘pungent’ odors | 4,951,027.32 | 0.1442 |
| Cyclodocosane, ethyl- | cyclodocosane is musty | 6,897,525.67 | 0.2010 |
| Total | 20,853,087.48 | 0.6075 | |
| Pleasant-Smelling Compounds | |||
| α-Farnesene | sweet, woody, and fruity smell | 83,701,909.86 | 2.4385 |
| cis-3-Hexenyl benzoate | green, herbaceous, floral, and woody odor | 20,593,293.36 | 0.6000 |
| Methylcyclopentane | sweet | 5,205,381.56 | 0.1517 |
| Farnesol | mild, light-woody, linden, floral | 11,598,405.13 | 0.3379 |
| trans-Farnesol | floral scent | 11,598,405.13 | 0.3379 |
| 3,7,11,15-Tetramethylhexadeca-1,6,10,14-tetraen-3-ol | floral | 13,608,158.22 | 0.3965 |
| Behenic alcohol | mild, pleasant, aromatic | 19,208,391.39 | 0.5596 |
| 7,212,273.25 | 0.2101 | ||
| 10,366,741.04 | 0.3020 | ||
| Squalene | “faint agreeable odor” | 57,289,446.95 | 1.6691 |
| Total | 240,382,405.9 | 7.0032 | |
| Characteristic and Others | |||
| Cyclohexane | sweet, pungent odor | 5,205,381.56 | 0.1517 |
| 5,389,937.01 | 0.1570 | ||
| (6Z,9Z)-6,9-Tricosadiene | pheromone component | 9,921,740.66 | 0.2891 |
| 9-Tricosene, (Z)- | pheromone component | 290,560,215.4 | 8.4651 |
| (6Z,9Z)-6,9-Tricosadiene | pheromone component | 5,689,718.8 | 0.1658 |
| (6Z,9Z)-6,9-Tricosadiene | pheromone component | 7,379,949.17 | 0.2150 |
| 1,3-Benzenedicarboxylic acid, bis(2-ethylhexyl) ester | “slight odor” | 4,646,143.26 | 0.1354 |
| Triacontanoic acid, methyl ester | “characteristic odor | 10,986,908.05 | 0.3201 |
| Octacosanoic acid, methyl ester | characteristic | 23,773,091.44 | 0.6926 |
| Total | 363,553,085.4 | 10.5916 | |
| Waxy Odors | |||
| Hexadecanoic acid, methyl ester | weak “waxy” odor | 11,445,635.5 | 0.3335 |
| Palmitic Acid, TMS derivative | slight waxiness | 15,861,788.16 | 0.4621 |
| Heneicosane | waxy | 27,873,257.53 | 0.8121 |
| 9,12,15-Octadecatrienoic acid, methyl ester, (Z,Z,Z)- | oily | 58,522,173 | 1.7050 |
| Methyl stearate | waxy | 9,570,314.44 | 0.2788 |
| Heneicosane | waxy | 16,865,047.04 | 0.4913 |
| Docosane | minimal waxiness, nearly odorless | 16,865,047.04 | 0.4913 |
| Total | 157,003,262.7 | 4.5741 |

Table A6.
GC–MS summary of significant compounds and area of the second neutral odor sample.
Table A6.
GC–MS summary of significant compounds and area of the second neutral odor sample.
| Notes | Area | Percent (%) | |
|---|---|---|---|
| Unpleasant-Smelling Compounds | |||
| Decane | gasoline-like odor | 604,738.63 | 0.0173 |
| n-Hexadecanoicacid | ‘rancid’ and ‘pungent’ odors | 10,695,786 | 0.3067 |
| Muskketone | highly tenacious musky aroma with a discreet animal note | 9,514,186.8 | 0.2728 |
| Phenyltert-butylketone | unpleasant | 12,772,890 | 0.3663 |
| Total | 33,587,601 | 0.9631 | |
| Pleasant-Smelling Compounds | |||
| α-Farnesene | sweet, woody, and fruity smell | 65,957,731 | 1.8913 |
| α-Cadinol | floral, fresh, and fruity odor of tea flowers | 4,801,024.4 | 0.1377 |
| 7-Hydroxyfarnesen | citrus, flowery | 4,021,592.9 | 0.1153 |
| Tonalid | sweet, amber, ambrette, fruity, musk | 8,403,166.6 | 0.2410 |
| 3,7,11,15-Tetramethylhexadeca-1,6,10,14-tetraen-3-ol | floral | 18,615,401 | 0.5338 |
| Behenicalcohol | mild, pleasant, aromatic | 46,458,265 | 1.3322 |
| Behenicalcohol | mild, pleasant, aromatic | 6,314,700.2 | 0.1811 |
| Squalene | “faint agreeable odor” | 80,224,892 | 2.3005 |
| Total | 234,796,773 | 6.7328 | |
| Characteristic and Others | |||
| Nonane,2,6-dimethyl- | camphoreous | 741,324.04 | 0.0213 |
| Humulene | earthy, woody, with spicy, herbal notes | 974,635.97 | 0.0279 |
| 3-Hexen-1-ol,benzoate,(Z)- | intense grassy-green odor | 15,485,274 | 0.4440 |
| (2E,4S,7E)-4-Isopropyl-1,7-dimethylcyclodeca-2,7-dienol | VOC | 154,419,161 | 4.4280 |
| BenzylBenzoate | weak, sweet-balsamic odor | 4,237,489 | 0.1215 |
| Octadecanoicacid | “mild odor” | 7,989,615.3 | 0.2291 |
| (6Z,9Z)-6,9-Tricosadiene | pheromone component | 16,355,475 | 0.4690 |
| 9-Tricosene,(Z)- | pheromone component | 282,558,255 | 8.1024 |
| Docosanoicacid,methylester | characteristic | 7,721,575.1 | 0.2214 |
| Tetracosanol,O-TMS | characteristic | 5,073,007 | 0.1455 |
| Triacontanoicacid,methylester | “characteristic odor” | 12,060,521 | 0.3458 |
| α-Tocopherylacetate | characteristic | 65,652,636 | 1.8826 |
| Octacosanoicacid,methylester | characteristic | 26,609,462 | 0.7630 |
| Total | 599,878,430 | 17.2015 | |
| Waxy Odors | |||
| PalmiticAcid,TMSderivative | slight waxiness | 30,867,835 | 0.8851 |
| Heneicosane | waxy | 40,643,834 | 1.1655 |
| Methylstearate | waxy | 16,035,763 | 0.4598 |
| Docosane | minimal waxiness, nearly odorless | 21,544,047 | 0.6178 |
| Total | 109,091,479 | 3.1282 |

Table A7.
FTIR library search results of the first pleasant-smelling sample.
Table A7.
FTIR library search results of the first pleasant-smelling sample.
| Quality | Library | Name |
|---|---|---|
| 0.92425 | Poly_D (68) | POLY(ETHYLENE) |
| 0.92256 | Agilent Polymer Handheld ATR Library (110) | Polyethylene, high density Approx Mw 125,000 Pellets CAS#25213-02-_2012-06-19T16-44-41 (Alfa Chemistry, New York, NY, USA) |
| 0.92256 | Agilent Polymer Handheld ATR Library (111) | Polyethylene, high density Approx Mw 125,000 Pellets CAS25213-02-9 (Alfa Chemistry, New York, NY, USA) |
| 0.92122 | Agilent Polymer Handheld ATR Library (106) | Polyethylene, chlorinated, Chlorine content 25 wt%, cast film CAS 64754-90-1 (Alfa Chemistry, New York, NY, USA) |
| 0.92041 | Agilent Polymer Handheld ATR Library (182) | Paraffin Wax and Polyvinyl Acetate Mixture (7% PVA Wax Coating) |

Table A8.
FTIR library search results of the second pleasant-smelling sample.
Table A8.
FTIR library search results of the second pleasant-smelling sample.
| Quality | Library | Name |
|---|---|---|
| 0.92251 | Intro_D (148) | PARVAN 3150, F.N. 3502*REFINED PARAFFIN WAX |
| 0.92145 | BioRad_Demo (47) | PARVAN 3150, F.N. 3502*REFINED PARAFFIN WAX |
| 0.92063 | Agilent Polymer Handheld ATR Library (106) | Polyethylene, chlorinated, Chlorine content 25 wt%, cast film CAS 64754-90-1 |
| 0.91934 | Poly_D (68) | POLY(ETHYLENE) |
| 0.91314 | Agilent Polymer Handheld ATR Library (182) | Paraffin Wax and Polyvinyl Acetate Mixture (7% PVA Wax Coating) |
| 0.90772 | Agilent Polymer Handheld ATR Library (110) | Polyethylene, high density Approx Mw 125,000 Pellets CAS#25213-02-_2012-06-19T16-44-41 |

Table A9.
FTIR library search results of the first unpleasant-smelling sample.
Table A9.
FTIR library search results of the first unpleasant-smelling sample.
| Quality | Library | Name |
|---|---|---|
| 0.91873 | Intro_D (148) | PARVAN 3150, F.N. 3502*REFINED PARAFFIN WAX |
| 0.91794 | BioRad_Demo (47) | PARVAN 3150, F.N. 3502*REFINED PARAFFIN WAX |
| 0.91770 | Agilent Polymer Handheld ATR Library (106) | Polyethylene, chlorinated, Chlorine content 25 wt%, cast film CAS 64754-90-1 |
| 0.91460 | Poly_D (68) | POLY(ETHYLENE) |
| 0.91025 | Agilent Polymer Handheld ATR Library (182) | Paraffin Wax and Polyvinyl Acetate Mixture (7% PVA Wax Coating) |
| 0.90567 | Agilent Polymer Handheld ATR Library (110) | Polyethylene, high density Approx Mw 125,000 Pellets CAS#25213-02-_2012-06-19T16-44-41 |

Table A10.
FTIR library search results of the second unpleasant-smelling sample.
Table A10.
FTIR library search results of the second unpleasant-smelling sample.
| Quality | Library | Name |
|---|---|---|
| 0.94096 | ATR Demo Library (41) | polyethylene |
| 0.93648 | Agilent Polymer Handheld ATR Library (23) | ETHYLENE PROPYLENE DIENE TERPOLYMER Ethylene 70% diene 4% Pellets CAS 25038-36-2 (Alfa Chemistry, New York, NY, USA) |
| 0.92137 | Poly_D (68) | POLY(ETHYLENE) |
| 0.91619 | Intro_D (148) | PARVAN 3150, F.N. 3502*REFINED PARAFFIN WAX |
| 0.91533 | BioRad_Demo (47) | PARVAN 3150, F.N. 3502*REFINED PARAFFIN WAX |
| 0.90404 | Agilent Polymer Handheld ATR Library (110) | Polyethylene, high density Approx Mw 125,000 Pellets CAS#25213-02-_2012-06-19T16-44-41 |

Table A11.
FTIR library search results of the first neutral smelling sample.
Table A11.
FTIR library search results of the first neutral smelling sample.
| Quality | Library | Name |
|---|---|---|
| 0.94139 | ATR Demo Library (41) | polyethylene |
| 0.94033 | Agilent Polymer Handheld ATR Library (23) | ETHYLENE PROPYLENE DIENE TERPOLYMER Ethylene 70% diene 4% Pellets CAS 25038-36-2 |
| 0.92766 | Agilent Polymer Handheld ATR Library (110) | Polyethylene, high density Approx Mw 125,000 Pellets CAS#25213-02-_2012-06-19T16-44-41 |
| 0.92766 | Agilent Polymer Handheld ATR Library (111) | Polyethylene, high density Approx Mw 125,000 Pellets CAS 25213-02-9 (Alfa Chemistry, New York, NY, USA) |
| 0.92558 | Intro_D (148) | PARVAN 3150, F.N. 3502*REFINED PARAFFIN WAX |

Table A12.
FTIR library search results of the second neutral smelling sample.
Table A12.
FTIR library search results of the second neutral smelling sample.
| Quality | Library | Name |
|---|---|---|
| 0.94393 | ATR Demo Library (41) | polyethylene |
| 0.94110 | Agilent Polymer Handheld ATR Library (23) | ETHYLENE PROPYLENE DIENE TERPOLYMER Ethylene 70% diene 4% Pellets CAS 25038-36-2 |
| 0.92479 | Poly_D (68) | POLY(ETHYLENE) |
| 0.92473 | Agilent Polymer Handheld ATR Library (110) | Polyethylene, high density Approx Mw 125,000 Pellets CAS#25213-02-_2012-06-19T16-44-41 |
| 0.92473 | Agilent Polymer Handheld ATR Library (111) | Polyethylene, high density Approx Mw 125,000 Pellets CAS 25213-02-9 |
References
- Darbre, P.D. Plant-based ingredients in personal care products. In Personal Care Products and Human Health; Elsevier: Amsterdam, The Netherlands, 2023; pp. 97–112. [Google Scholar] [CrossRef]
- Abdullahi, A.; Tijjani, A.; Abubakar, A.I.; Khairulmazmi, A.; Ismail, M.R. Plant biomolecule antimicrobials: An alternative control measures for food security and safety. In Herbal Biomolecules in Healthcare Applications; Elsevier: Amsterdam, The Netherlands, 2022; pp. 381–406. [Google Scholar] [CrossRef]
- Jasmine Oil—Jasmine Oil Manufacturer, Supplier, Wholesaler & Retailer, Kanpur, India. Available online: https://www.indiaaromaoils.com/jasmine-oil.html#:~:text=Jasmine%20oil%20has%20a%20medium (accessed on 20 February 2024).
- Pavela, R. History, presence and perspective of using plant extracts as commercial botanical insecticides and farm products for protection against insects—A review. Plant Prot. Sci. 2016, 52, 229–241. [Google Scholar] [CrossRef]
- Cimino, C.; Maurel, O.M.; Musumeci, T.; Bonaccorso, A.; Drago, F.; Souto, E.M.B.; Pignatello, R.; Carbone, C. Essential Oils: Pharmaceutical Applications and Encapsulation Strategies into Lipid-Based Delivery Systems. Pharmaceutics 2021, 13, 327. [Google Scholar] [CrossRef] [PubMed]
- Burger, P.; Plainfossé, H.; Brochet, X.; Chemat, F.; Fernandez, X. Extraction of Natural Fragrance Ingredients: History Overview and Future Trends. Chem. Biodivers. 2019, 16, e1900424. [Google Scholar] [CrossRef] [PubMed]
- Icamina, P. PH Lures Investors to Big Essential Oil Business. Available online: https://malaya.com.ph/news_business/ph-lures-investors-to-big-essential-oil-business/ (accessed on 20 February 2024).
- Sampaguita Livelihoods of Peri-Urban Metro Manila PDF Metro Manila Agriculture. Scribd. Available online: https://www.scribd.com/document/149692864/Sampaguita-Livelihoods-of-Peri-urban-Metro-Manila (accessed on 20 February 2024).
- National Museum of the Philippines. Philippine National Flower-Sampaguita—National Museum. 2021. Available online: https://www.nationalmuseum.gov.ph/2021/11/10/philippine-national-flower-Sampaguita/ (accessed on 20 February 2024).
- Uwineza, P.A.; Waśkiewicz, A. Recent Advances in Supercritical Fluid Extraction of Natural Bioactive Compounds from Natural Plant Materials. Molecules 2020, 25, 3847. [Google Scholar] [CrossRef] [PubMed]
- Flora, I.O. Lubao Mayor Eyes Boosting Local Sampaguita Production; SunStar Publishing Inc.: Cebu City, Philippines, 2023; Available online: https://www.sunstar.com.ph/pampanga/local-news/lubao-mayor-eyes-boosting-local-Sampaguita-production (accessed on 20 February 2024).
- Bacud, S.T.; Cardenas, V.R.; Velasco, L.R.I. The Sampaguita Livelihood System in Sta. Cruz, Laguna, Philippines: A Case of a Transformative Resilience Development. Procedia Econ. Financ. 2014, 18, 439–446. [Google Scholar] [CrossRef]
- Rassem, H.; Nour, A.; Yunus, R.; Zaki, Y.H.; Abdlrhman, H.S. Yield Optimization and Supercritical CO2 Extraction of Essential Oil from Jasmine Flower. Indones. J. Chem. 2019, 19, 479. [Google Scholar] [CrossRef]
- Braun, N.A. Jasminum grandiflorum: Influence of Flower Processing on Concrete and Absolute Composition. J. Essent. Oil Res. 2020, 15, 1934578X20960998. [Google Scholar]
- Arifan, F.; Adhy, S.; Broto, W.; Nuswantari, S.R. A Variation of Jasmine Essential Oil for a New Jasmine Aromatherapy Formulation. Ann. Trop. Med. Public Health 2021. [Google Scholar] [CrossRef]
- Capuzzo, A.; Maffei, M.; Occhipinti, A. Supercritical Fluid Extraction of Plant Flavors and Fragrances. Molecules 2013, 18, 7194–7238. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Xiong, Y.; An, H.; Li, J.; Li, Q.; Huang, J.; Liu, Z. Analysis of Volatile Components of Jasmine and Jasmine Tea during Scenting Process. Molecules 2022, 27, 479. [Google Scholar] [CrossRef] [PubMed]
- Extract Winterization Background & Basics. Aptia Engineering. 2022. Available online: https://aptiaengineering.com/2022/03/07/extract-winterization-background-and-basics/ (accessed on 20 February 2024).
- Nascimento, L.D.D.; Silva, S.G.; Cascaes, M.M.; Costa, K.S.D.; Figueiredo, P.L.B.; Costa, C.M.L.; de Aguiar Andrade, E.H.; de Faria, L.J.G. Drying Effects on Chemical Composition and Antioxidant Activity of Lippia thymoides Essential Oil, a Natural Source of Thymol. Molecules 2021, 26, 2621. [Google Scholar] [CrossRef] [PubMed]
- Menezes, J.; Athmaselvi, K.A. Report on Edible Films and Coatings. In Food Packaging and Preservation; Elsevier: Amsterdam, The Netherlands, 2018; pp. 177–212. [Google Scholar] [CrossRef]
- Paraffin Wax|Candle Making, Cosmetic Uses, Industrial Applications|Britannica. Available online: https://www.britannica.com/science/paraffin-wax (accessed on 17 September 2024).
- Rassem, H.H.; Nour, A.H.; Yunus, R.M. GC-MS analysis of bioactive constituents of jasmine flower. J. Chem. Eng. Ind. Biotechnol. 2018, 4, 52–59. [Google Scholar] [CrossRef]
- Alpha-Cadinol (CHEBI:132905). Available online: https://www.ebi.ac.uk/chebi/searchId.do?chebiId=CHEBI:132905 (accessed on 20 February 2024).
- Akram, A.; Younis, A.; Akhtar, G.; Ameer, K.; Farooq, A.; Hanif, M.A.; Saeed, M.; Lim, K.B. Comparative efficacy of various essential oil extraction techniques on oil yield and quality of Jasminum sambac L. Sci. Int. 2017, 5, 84–95. [Google Scholar] [CrossRef][Green Version]
- Cis-3 Hexenyl Benzoate. Available online: https://www.perfumersworld.com/view.php?pro_id=3GN00227 (accessed on 20 February 2024).
- Geranyl Linalool, 68931-30-6. Available online: http://www.thegoodscentscompany.com/data/rw1428051.html (accessed on 20 February 2024).
- Development from Jasminum sambac Flower Extracts of Products with Floral Fragrance and Multiple Physiological Activities—PMC. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8378956/ (accessed on 17 September 2024).
- 6-Dimethyl-3-oxatricyclo(4.2.1.0*2,4*)nonane. Available online: http://www.thegoodscentscompany.com/data/rw1061581.html (accessed on 20 February 2024).
- PubChem. 2-Hexyl-1-decanol. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/95337 (accessed on 20 February 2024).
- Scentspiracy. Methyl Anthranilate. Available online: https://www.scentspiracy.com/fragrance-ingredients/p/methyl-anthranilate#:~:text=What%20is%20Methyl%20Anthranilate%3F,Its%20molecular%20formula%20is%20C%E2%82%88H%E2%82%89NO%E2%82%82 (accessed on 20 February 2024).
- Zhang, D.; Sun, G.; Zhang, X. Analysis of a Gas Chromatography/Mass Spectrometry Thermal Desorption System with Simultaneous Sniffing for Determination of Off-Odor Compounds and Volatile Organic Compounds in Polypropylene Compo-sites. Sci. Adv. Mater. 2019, 11, 1594–1603. [Google Scholar] [CrossRef]
- PubChem. 4-Methyldecane. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/17835 (accessed on 20 February 2024).
- Panel, C.E.; Cosmetic Ingredient Review Expert Panel. Final Report on the Safety Assessment of Squalane and Squalene. J. Am. Coll. Toxicol. 1990, 1, 37–56. [Google Scholar] [CrossRef]
- α-Tocopheryl Acetate, 58-95-7. Available online: http://www.thegoodscentscompany.com/data/rw1372861.html (accessed on 20 February 2024).
- Musk Ketone (81-14-1)—Synthetic Ingredient for Perfumery. Scentspiracy. Available online: https://www.scentspiracy.com/fragrance-ingredients/p/musk-ketone (accessed on 20 February 2024).
- Grosch, W. Evaluation of the key odorants of foods by dilution experiments, aroma models and omission. Chem. Senses 2001, 26, 533–545. [Google Scholar] [CrossRef] [PubMed]
- Boelens, M.H.; van Gemert, L.J. Perceptual masking of odorants in binary mixtures. Chem. Senses 1993, 18, 355–367. [Google Scholar] [CrossRef]
- Hartati; Salleh, L.M.; Aziz, A.; Yunus, M.A.C. The Effect of Supercritical Fluid Extraction Parameters on the Swietenia Mahagoni Seed Oil Extraction and its Cytotoxic Properties. J. Teknol. Sci. Eng. 2014, 69, 51–53. [Google Scholar] [CrossRef][Green Version]
- van Harreveld, A.P. Odor concentration decay and stability in gas sampling bags. J. Air Waste Manag. Assoc. 2003, 53, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Salvador, A.; Chisvert, A. Analysis of Cosmetic Products. 2007. Available online: http://inci-dic.com/wp-content/uploads/2018/05/Analysis-of-Cosmetic-Products.pdf (accessed on 20 February 2024).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).