Abstract
Today’s yeast total biomass and viability measurements during the brewing process are dependent on offline methods such as methylene blue or florescence dye-based staining, and/or the usage of flow cytometric measurements. Additionally, microscopic cell counting methods decelerate an easy and quick prediction of yeast viability. These processes are time consuming and result in a time-delayed response signal, which not only reduces the knowledge of the performance of the yeast itself, but also impacts the quality of the final product. Novel approaches in process monitoring during the aerobic and anaerobic fermentation of Saccharomyces cerevisiae are not only limited to classical pH, dO2 and off-gas analysis, but they also use different in situ and online sensors based on different physical principles to determine the biomass, product quality and cell death. Within this contribution, electrochemical impedance spectroscopy (EIS) was used to monitor the biomass produced in aerobic and anaerobic batch cultivation approaches, simulating the propagation and fermentation unit operation of industrial brewing processes. Increases in the double-layer capacitance (CDL), determined at frequencies below 1 kHz, were proportional to the increase of biomass in the batch, which was monitored in the online and inline mode. A good correlation of CDL with the cell density was found. In order to prove the robustness and flexibility of this novel method, different state-of-the-art biomass measurements (dry cell weight—DCW and optical density—OD) were performed for comparison. Because measurements in this frequency range are largely determined by the double-layer region between the electrode and media, rather minor interferences with process parameters (aeration and stirring) were to be expected. It is shown that impedance spectroscopy at low frequencies is not only a powerful tool for the monitoring of viable yeast cell concentrations during operation, but it is also perfectly suited to determining physiological states of the cells, and may facilitate biomass monitoring in the brewing and yeast-propagating industry drastically.
1. Introduction
Microbial cultivations play a key role in many different fields, such as in food, drug and bulk chemical production, as well as in waste-to-value concepts [1]. Process monitoring, such as pH, dissolved oxygen (dO2) and off-gas analysis, is state of the art in today’s industrial cultivations for guaranteeing product quality and safety. However, the most important parameter in bioprocesses, the biomass, can only be determined using offline methods or complex soft-sensor applications [2]. These control systems are often dependent on inline/online/at-line detection systems, such as high-performance liquid chromatography (HPLC) for metabolites, off-gas balance, and/or dielectric spectroscopy measurements. The use of accurate and reliable biomass measurement systems [3,4], especially of viable cell concentrations (VCCs), enables proper process control tools, which lead subsequently to more robust and reliable bioprocesses. The VCC is measured using offline measurement principles including marker proteins or fluorescence probes, such as flow cytometry or confocal microscopy [5,6]. Because these control and analytical tools are cost intensive, classical bulk food products—such as yeast and beer—are produced in rather uncontrolled environments. Not only the complex raw material, but especially growth conditions of the yeast (propagation and fermentation) are of high importance for the quality of the final product. The implementation of online vitality measurements in the brewing industry has historically been hindered by affordable, simple, robust and reproducible tests [7].
In general, online and inline biomass measurement approaches are rather scarce and are based on physical measurement principles. One principle generally applied is high-frequency alternating current (AC) impedance spectroscopy with high field amplitudes, used on the basis of the ß-dispersion [8,9]. Cells with an integer cell membrane affect the relative permittivity between two electrodes and, therefore, this signal is used for the estimation of VCCs. A detailed description of the measurement principles can be found in [10,11,12,13].
The model organism for the application of AC measurements in the ß-dispersion range is yeast, being a very important expression host for recombinant proteins [14,15,16]. Additionally, approaches towards more complex expression systems, such as filamentous fungi and Chinese hamster ovary (CHO) cells, are already performed [17,18,19]. These measurements show a strong dependence upon physical process parameters (such as aeration and stirring—causing gas bubbles, temperature shifts and pH gradients), and are furthermore highly affected by changes in the media’s composition during cultivation.
However, not only high-frequency impedance spectroscopy in the ß-range can be used for the determination of biomass, but changes of the electrical double layer by the adsorption/desorption of cells at the electrode surface (detectable at low frequencies in the mHz range; α-dispersion) can also provide valuable information. Besides the cell type itself (cell wall/membrane compositions, size and shape), many physical parameters, especially in the media (pH and ion concentrations), can influence the potential distribution in the double layer [20,21]. Furthermore, the given method via α-dispersion detection is capable of detecting even very small numbers of bacteria in soil, food and feces-polluted water using interdigitated microelectrode designs [22,23,24,25,26,27]. These studies were only performed at a very small scale and with a low cell concentration. In general, a threshold in the measurement was present at low cell concentrations. Exceeding this limitation, over time, very stable signals were achieved in these studies. Besides direct measurements in the broth, a modified electrode system in an interdigitated design can be used [28,29,30]. First approaches towards process monitoring were shown by Kim et al. [31], who worked with an inline sensor used in the lower frequency range between 40 Hz and 10 kHz for the real-time monitoring of biomass. Kim et al. showed the feasibility for measuring changes in the double-layer capacitance (CDL), but no analysis of the CDL itself was performed; only discrete extracted values for distinct frequency values were used. Recent studies on Escherichia. coli showed reasonable results for VCC determination not only in the batch phase, but also in the fed-batch approaches, leading to far higher cell densities [32].
In this study, impedance measurements in the α-dispersion range were performed during the batch-based cultivation of Saccharomyces cerevisiae aimed for usage in brewing applications. Different state-of-the-art methods were applied for determination of the corresponding total biomass—dry cell weight (DCW) and optical density (OD610) offline. Flow cytometry (FCM) in combination with the fluorescence dye (bis-(1,3-dibutylbarbituricacid)trimethineoxonol) (DiBAC) was used for a cell physiology evaluation to account for changes in the viability during cultivation. With this knowledge, we were able to correlate the total biomass to the extracted CDL.
A prototype inline probe was designed and built for easy plug-in measurements of the biomass. Online and new inline probes were tested using defined media with glucose and maltose in different concentrations and with malt extract as the complex base material in brewing.
2. Materials and Methods
2.1. Expression Host and Cultivation
All cultivations were performed using the S. cerevisiae strain, supplied by Brauerei GUSSWERK (Salzburg, Austria). For the preculture, 500 mL of sterile Delft medium was inoculated from frozen stocks (1.5 mL; −80 °C) and incubated in a 2500 mL High-Yield shake flask for 20 h (230 rpm; 28 °C). Batch cultivations were performed in a stainless-steel Sartorius Biostat Cplus bioreactor (Sartorius, Göttingen, Germany) with a 10 L working volume, and in an Infors Techfors-S bioreactor (Infors HT; Bottmingen, Switzerland) with a 20 L working volume. Aerobic batches were cultivated using 1000 to 1400 rpm stirrer speeds with an aeration of 2 vvm. Anaerobic batches were cultivated at 600 rpm and with a 2 to 4 L/min N2 flow. The composition of the defined Delft medium used was as follows: 7.5 g/L (NH4)2SO4, 14.4 g/L KH2PO4, 0.5 g/L MgSO4·7H2O, 2 mL of trace metal stock, 1 mL of vitamins, 50 µL of polypropylenglycol (PPG) as Antifoam, and maltose and glucose in different concentrations as a carbon source. For the malt extract-based fermentation, a preculture with Delft media was cultivated, which was afterwards inoculated into the malt extract solution (150 g/L malt extract in deionized water; Weyermann, Bavarian Pilsner, Bamberg, Germany).
2.2. Analytical Procedures
For the DCW measurements, 1 mL of the cultivation broth was centrifuged at about 9000 g, subsequently washed with 0.9% NaCl solution, and centrifuged again. After drying the cells at 105 °C for 48 h, the pellet was evaluated gravimetrically. DCW measurements were performed in five replicates and the mean error for DCW was about 3%. Offline OD610 measurements were performed in duplicates in a UV/VIS photometer, Genisys 20 (Thermo Scientific, Waltham, MA, USA).
Verification of the cell viability in defined medium samples was performed using FCM measurements. After the addition of DiBAC (Thermo Scientific, Waltham, MA, USA), the diluted cultivation broth was measured using a CyFlow Cube 8 flow cytometer (Sysmex-Partec, Bornbach, Germany). DiBAC is sensitive to the plasma membrane potential, and therefore a distinction between viable and non-viable cells can be achieved. Detailed information on the viability assay can be found elsewhere [33]. The overall errors for this method were in the range of 0.5% to 1%.
Sugar concentrations in the fermentation broth were determined using a Supelco C-610H HPLC column (Supelco, Bellefonte, PA, USA) on an Ultimate 300 HPLC system (Thermo Scientific, Waltham, MA, USA) using 0.1% H3PO4 as a running buffer at 0.5 mL/min. Ethanol concentrations were determined using an Aminex HPLC column (Biorad, Hercules, CA, USA) on an Agilent 1100 System (Agilent Systems, Santa Clara, CA, USA) with 40 mM H2SO4 as a running buffer at 0.6 mL/min.
Cultivation off-gas was analyzed by gas sensors: IR for CO2 and ZrO2-based for O2 (Blue Sens Gas analytics, Herten, Germany).
2.3. Impedance Measurements
Physical analysis of VCCs in state-of-the-art capacitance probes, which rely on β-dispersion (107–104 Hz), show a high dependence on process parameters (e.g., stirring, temperature, pH, salt and substrate concentration, etc.) and the cultivation phase (exponential growth phase, starvation phase, etc.) [12,33]. We focused the measurement on a different physical phenomenon (α-dispersion), which yields valuable information mainly regarding the biomass concentration. The “α-dispersion effect”, at frequencies below 10 kHz, which is most likely a result of deformation of ionic species around the cell membranes, was used for these measurements. The dielectric response was therefore proportional to the ionic charge gathered around the membrane of adsorbed cells on the electrode [20,21]. Impedance measurements were recorded in the range of 106 to 10−1 Hz with amplitudes of 100 to 250 mV using the Alpha-A high-resolution dielectric analyzer (Novocontrol, Montabaur, Germany). Because measurements in this frequency range are largely determined by the double-layer region between the electrode and the media, rather minor interferences with the process parameters (aeration and stirring) were to be expected. Online flow cells showed the benefit of a laminar flow through the cell and minor turbulence, but they generally had the problems of differences in the process state (side stream) and of performing sterilization procedures. Inline probes should overcome these problems, but they may be strongly affected by the process parameters. Details on the fitting procedure and data evaluation are given in [32].
2.4. Inline Probe Construction
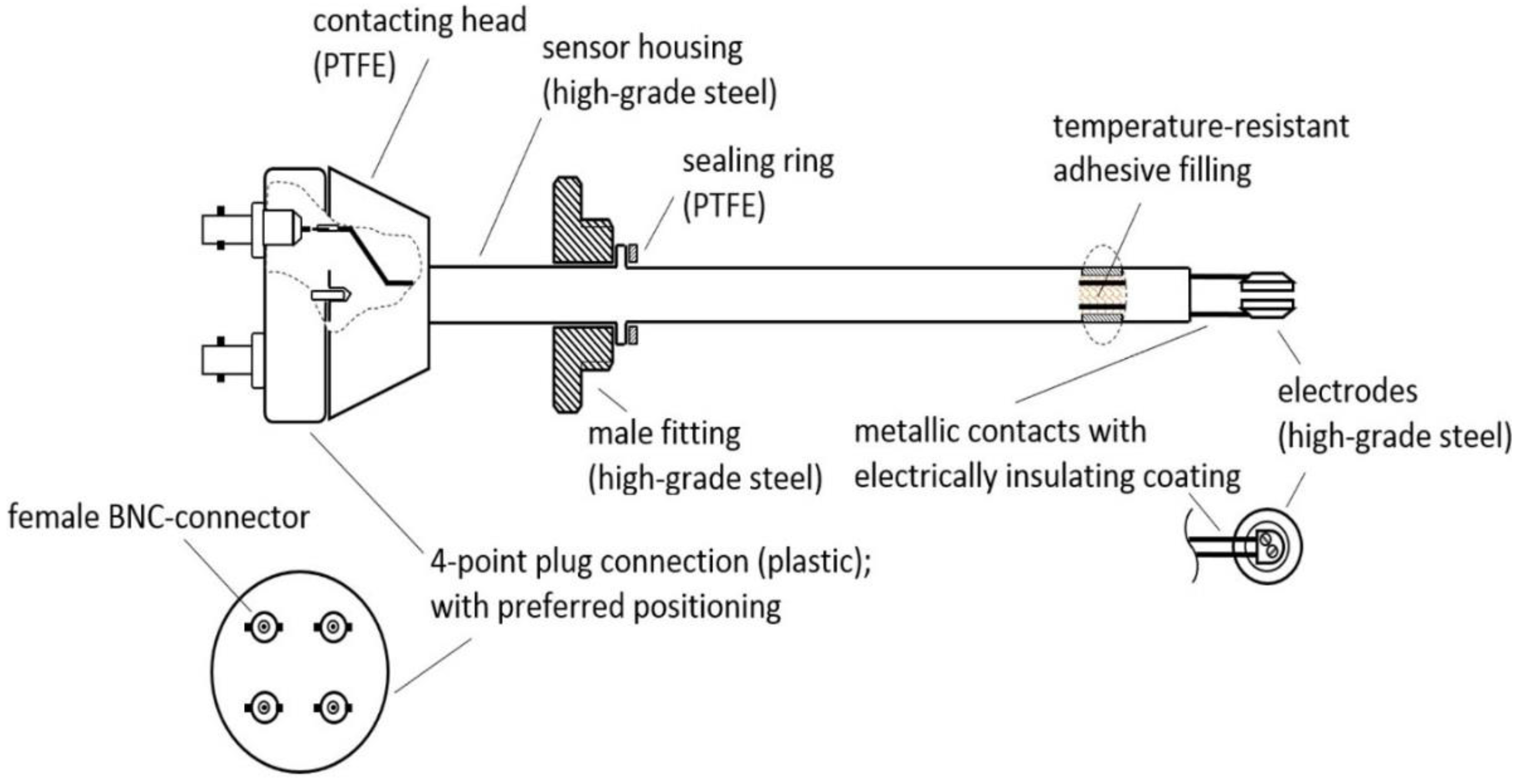
As online probes are not directly situated inside the reactor but are often supplied by a side stream of the fermentation broth, changes in the metabolism in this time interval may be highly possible, but less disruption of the signal is also observed by the stirring and aeration of the system. Furthermore, online probes always pose the danger of contaminating the process, as the sterile barrier is not kept inside the fermenter. Therefore, for sterile processes without constant streams of broth, the assembly of an inline probe prototype used a commonly used 25 mm B. Braun safety port with O-ring (Ingold connector). Materials were chosen to be permanently stable at 130 °C, and could easily sustain in situ autoclavation procedures. The physical analysis of VCCs was monitored and investigated by the inline probe sketched in Figure 1.
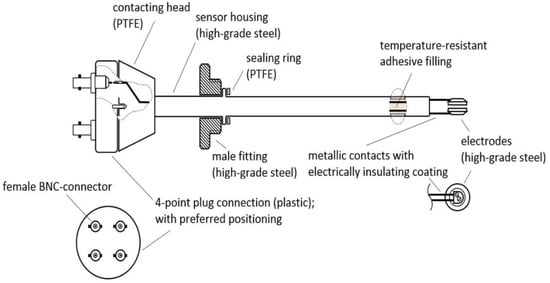
Figure 1.
Sketch of the inline probe prototype indicating used materials and wiring. Connection to the impedance analyzer was performed using a four-point BNC (Bayonet Neill–Concelman) connector.
The body as well as the electrodes of the inline probe consist of high-grade steel, that is, austenitic stainless steel, which is approximately 140 mm in length and at least 12 mm in diameter. Each electrode has a diameter of 10 mm. The gap between the electrodes is approximately 2 mm.
3. Results and Discussion
3.1. Aerobic and Anaerobic Batch Cultivations in Defined Media Monitored in Online Mode
Yeast, as a well-known host for diauxic growth, was cultivated aerobically and anaerobically using different carbon sources, preliminarily present in malted barley and wheat grain. A batch-based design was used in both cultivations. Growth rates of corresponding cultivations in Figure 1 are given in Table 1. The specific growth rate describes the increase of biomass in a given time interval normalized to the biomass inside the reactor (dx/dt*1/x(t), with x being the biomass).

Table 1.
Specific growth rate µ of batch phases determined by offline dry cell weight (DCW) measurements (given in Figure 1).
3.1.1. Raw Data and General Considerations
The measured impedance raw data were analyzed by a resistance RDL in parallel to a non-ideal capacitance (constant phase element) CPEDL (parameter Q, n). These elements most likely originate from the double-layer region close to the electrode and can be expressed by Equation (1):
where ω is the arc frequency and i is the imaginary number; n and Q are obtained from a fit to experimental data. In principle, these parameters can be used to calculate the CDL according to CDL = (RDL1−n × QDL)1/n.
ZDL = 1/(RDL−1 + (iω)n × QDL)
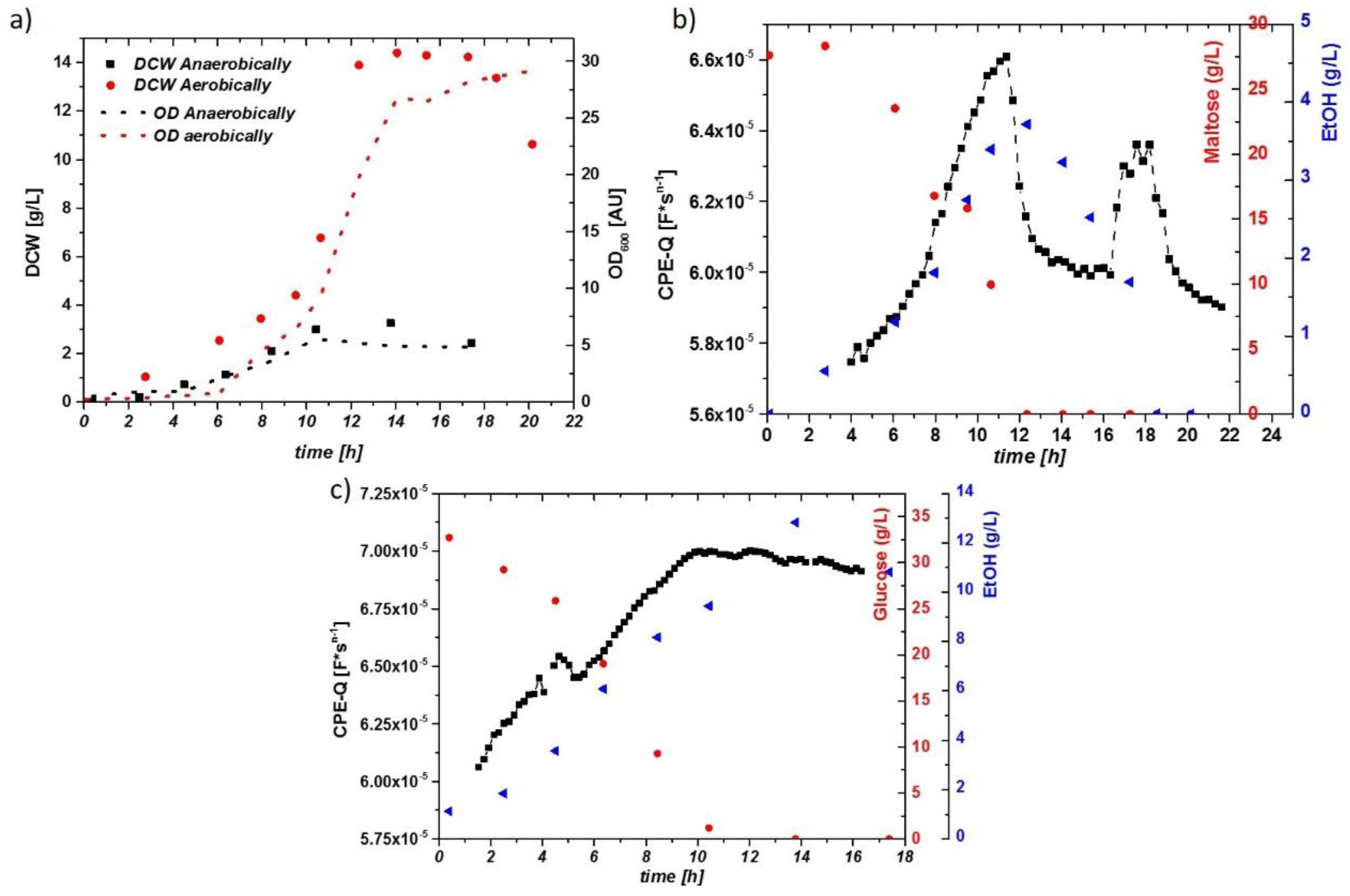
The aerobic growth of yeast results in partial aerobic metabolism and partial fermentation, well known as the Crabtree effect. During anaerobic growth, sugars are solely fermented to ethanol. The corresponding DCW and OD of two cultivations are given in Figure 2a. During growth on a high concentration of glucose, the respiratory capacity was generally too low, and ethanol was produced simultaneously. The sugar decrease and ethanol production are given in Figure 2b, which includes the Q value of the online impedance probe. Upon sugar depletion in the fermentation broth at t = 12 h, a strong decrease in the impedance signal is observed, which corresponds to the growth on ethanol. Anaerobic growth on glucose yielded much higher ethanol concentrations (about 1.5 vol % in this run), as shown in Figure 2c. The impedance signal increased over time with a maximum upon the complete glucose depletion. Because ethanol cannot be metabolized anaerobically and accumulates in the supernatant, no change in cell metabolism, but rather a shift from exponential growth into the stationary phase, was expected. Therefore, no steep drop in the impedance signal, but a smooth decrease over several hours, was observed.
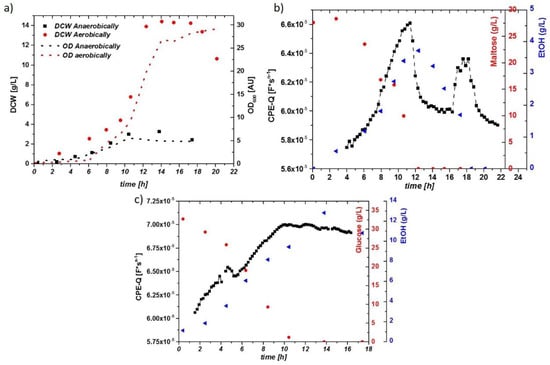
Figure 2.
(a) Time courses of the dry cell weight (DCW; g/L) and optical density (OD; AU) for the aerobically and anaerobically cultivated S. cerevisiae. Corresponding µ values are given in Table 1. (b) Impedance signal (constant phase element-Q: CPE-Q) and maltose/ethanol concentrations over time during the aerobic cultivation. A drop in the impedance signal is visible after depletion of maltose (change in metabolism). A further small increase upon ethanol uptake can be observed, until the second carbon source was depleted. (c) Impedance Signal (here, CPE-Q) and glucose/ethanol concentrations during the anaerobic cultivation. No sudden decrease of the impedance signal is spotted after depletion of glucose in the media; rather, a constant decrease in the signal can be observed.
3.1.2. Aerobic Cultivations
The double-layer resistance (RDL) could not be fitted accurately [32] (especially for the inline probe) as a result of the high overall fitting error, as already observed for cultivations with E. coli. Furthermore, n values, received from fittings with CPE elements, showed deviations dependent on the cultivation state (aerobic/anaerobic) and especially on the type of probe (inline/online). These changes in the n values made a comparison of runs difficult. For a better description of the different metabolic states, the fitting procedure was modified. For easier comparability of the performed runs, n was fixed to be 1 in the following data analysis, reflecting the idealized capacitance (Cideal) of the sample.
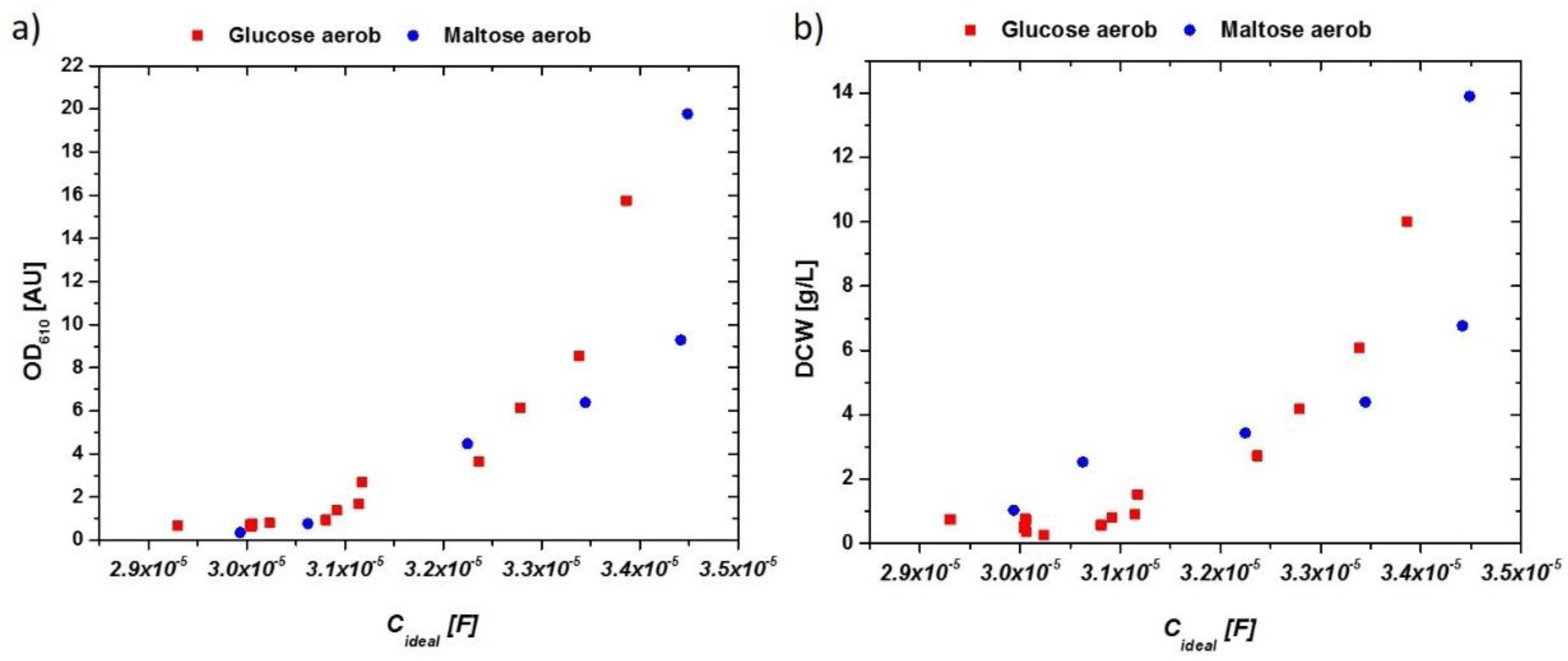
Measured values for biomass determination (OD610 and DCW) were correlated to the received idealized impedance signal referred to as Cideal. The corresponding data for aerobic cultivations on glucose as well as on maltose are given in Figure 3a for OD and in Figure 3b for the DCW measurement.
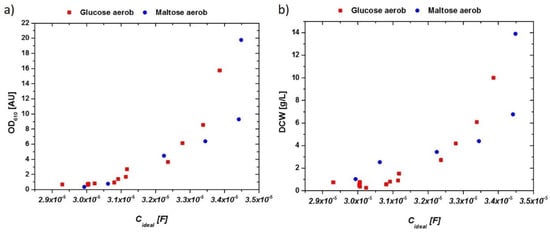
Figure 3.
(a) OD610 vs. extracted capacitance of the impedance signal in aerobic cultivations with glucose and maltose. (b) DCW vs. extracted capacitance of the impedance signal in aerobic cultivations. Very similar responses are obtained for both cultivations, irrespective of the used C-source.
The late stage during the end of the batch cultivation showed deviations in the impedance signal as a result of metabolic changes in the system (compared to Figure 2b). Deviations between glucose and maltose may have been a result of a different sugar transportation through the membrane. Because maltose uptake is mediated by a proton-mediated symporter, a change in the counterion-cloud and therefore changes in the overall impedance would be very likely [34]. Furthermore, changes in the overall membrane structure, producing maltose-transporting proteins (maltose permease), not present in glucose-grown cells, may have changed the magnitude of the impedance signal in these cultivations. However, the impedance signal, especially at the end of the aerobic batch-phase, held valuable information on the present metabolism of S. cerevisiae.
3.1.3. Anaerobic Cultivations
Ethanol production may have had further effects on the impedance signal, holding information on the physiological state of the system. Sugar concentrations of up to 200 g/L showed no effect on the magnitude of the impedance signal [32]. However, growth conditions of S. cerevisiae may have impacted on the impedance signal. To test the impact of growth conditions, different anaerobic cultivations (ANA) were performed, according to Table 2. Oxygen from air was eliminated with a flow of nitrogen at 4 L/min through the fermenter. Gas analysis of the off-gas confirmed the absence of oxygen throughout the entire batch.

Table 2.
Starting sugar concentrations in different anaerobic batch runs (ANA) extracted by high-pressure liquid chromatography (HPLC) measurements.
Raw data for cultivations with a different sugar concentration (compared to Figure 2c), including maltose and glucose grown anaerobically, showed high ethanol concentrations, which reached a maximum of about 3 vol % in these cultivations (ANA2).
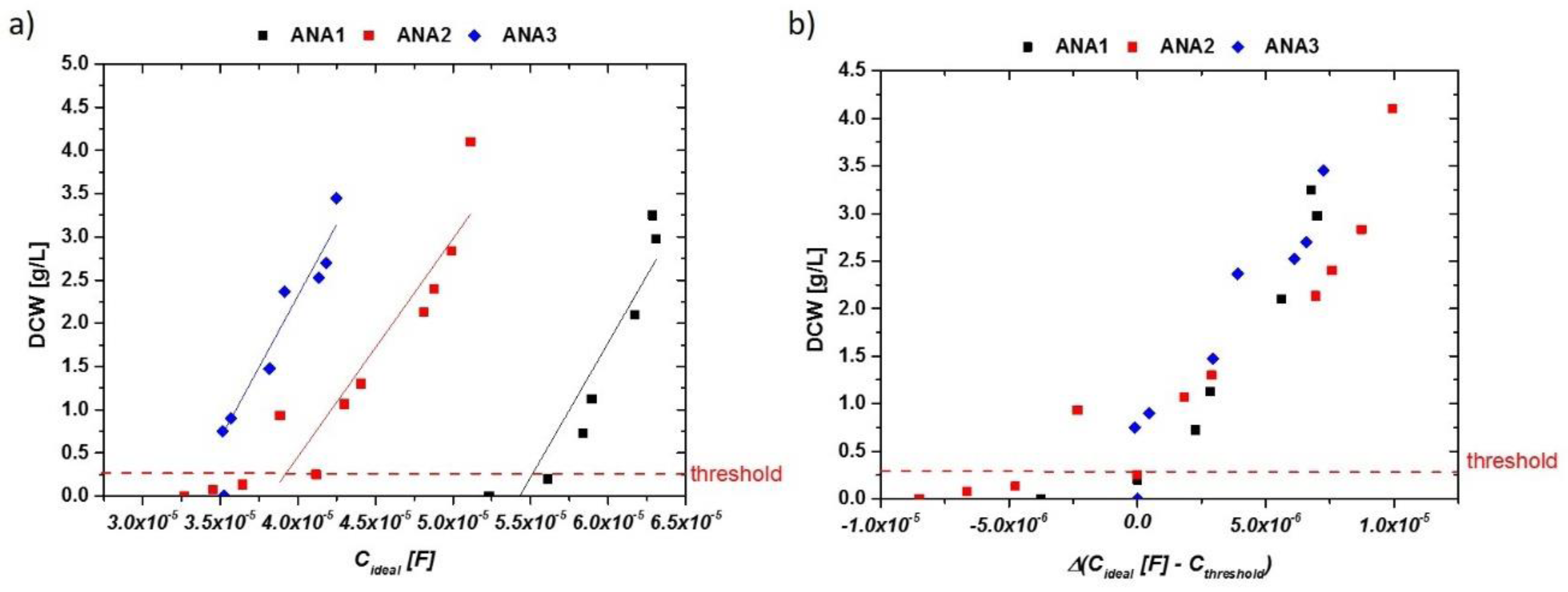
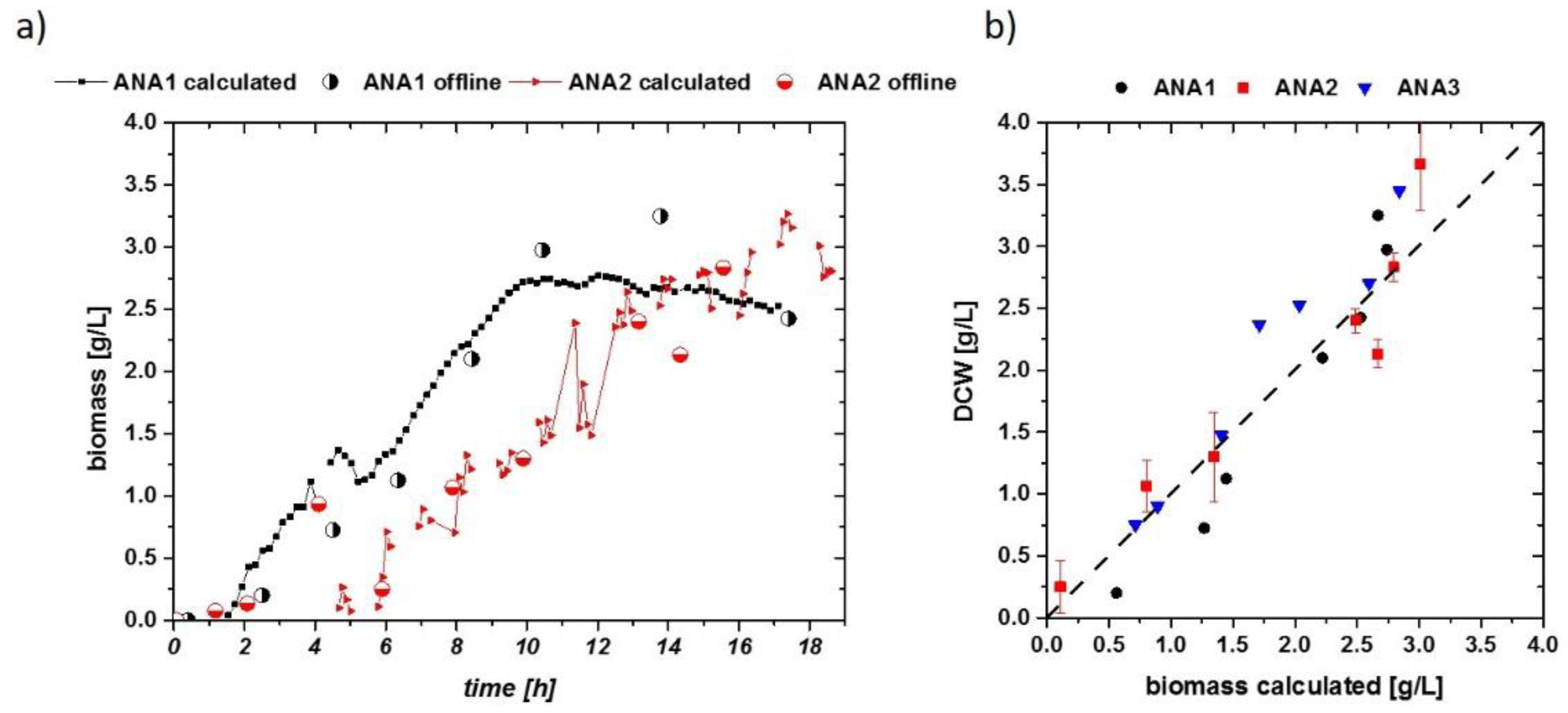
Anaerobic growth could be well described, except for very early time points, when cell densities were below the threshold of about 0.3 g/L. For inline OD measurements, very similar results could be obtained (not shown). Generally, the fits for aerobic and anaerobic cultivations are used to estimate biomass in real-time for the aerobic and anaerobic runs. As a very good linear description can be obtained in anaerobic runs, the real-time estimation of biomass is straightforward, as shown in Figure 4a,b, after normalization to the threshold of the impedance signal.
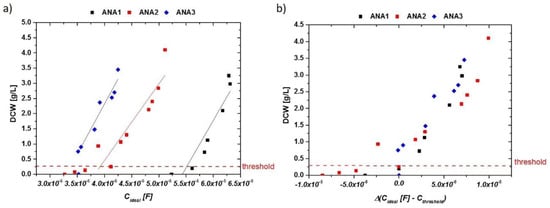
Figure 4.
(a) Idealized capacitances of anaerobic runs exhibiting sugar concentrations of up to 100 g/L in the fermentation broth (mixture of maltose and glucose). Ethanol concentrations reached 3 vol %. Different absolute values could be found in these cultivations, but the increase of the impedance signal with DCW is very similar. (b) DCW vs. delta of impedance signal (ideal capacity) in the anaerobic runs. Normalization to the threshold value of about 0.3 g/L resulted in very reproducible signals for very different fermentation runs.
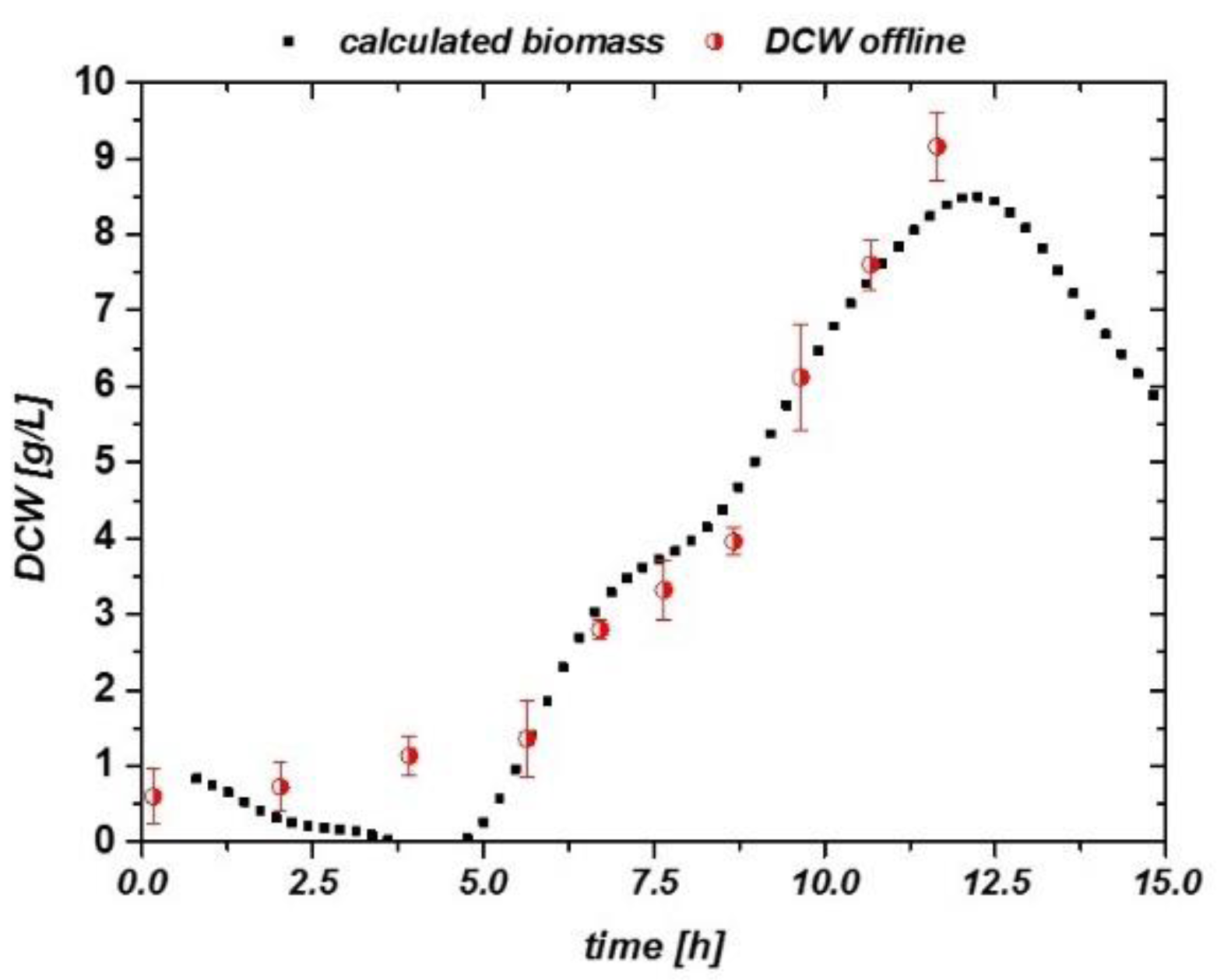
Using these results, DCW (half-filled circles) values are well described by the impedance signal over the process time (Figure 5a). A general quality of the fit is given in Figure 5b. The calculated DCW versus the measured DCW is situated close to the first median. Values not situated along the first median indicate the overall error in the fitting routine, compared to a residual analysis. As cell densities were very low during these cultivations, errors during the DCW measurement were about 10% of the actual mean value (highlighted for ANA2 in Figure 5b). When comparing the accuracy and threshold to those of E. coli cultivations, S. cerevisiae cultivations had a very good reproducibility for aerobic and anaerobic growth using the online impedance probe, even in the low-biomass concentration regime. A threshold of about 0.3 g/L DCW also gives a strong benefit for monitoring highly dynamic systems.
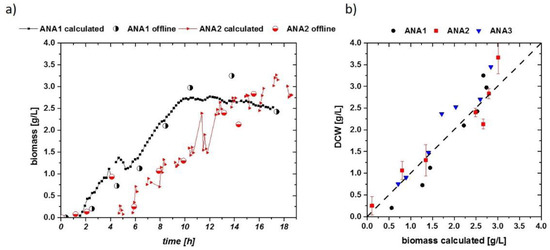
Figure 5.
(a) Biomass calculated from the impedance signal fit extracted in Figure 4a, including offline-measured DCW values as circles. Flow cytometric measurements confirmed that no dead population is visible; thus, DCW can be compared to the viable cell concentration (VCC) in these runs. (b) Residual analysis of the three anaerobic runs. Despite the low cell densities, a very accurate correlation can be found in all cultivations. Error bars are exemplarily plotted for the ANA2 cultivation.
3.2. Aerobic and Anaerobic Cultivations in Defined Media Using the New Inline Probe
The impedance signal in the online mode could be used to estimate the viable cell concentrations in the aerobic and anaerobic cultivations. The newly built inline probe prototype was measured alternating to the online probe in two cultivation runs (one aerobic and the other anaerobic). Impedance raw data of the aerobic run are given in Figure 6a. Two very distinct features are visible. At higher frequencies, a shift to negative differential resistances is visible. Similar phenomena have already been observed in E. coli fermentations, even before inoculation of the system [32]. Further, a reduction of the capacity by an order of magnitude is observed between the online probe and the inline probe, compared to Figure 6a.
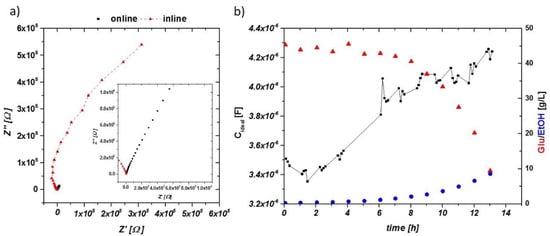
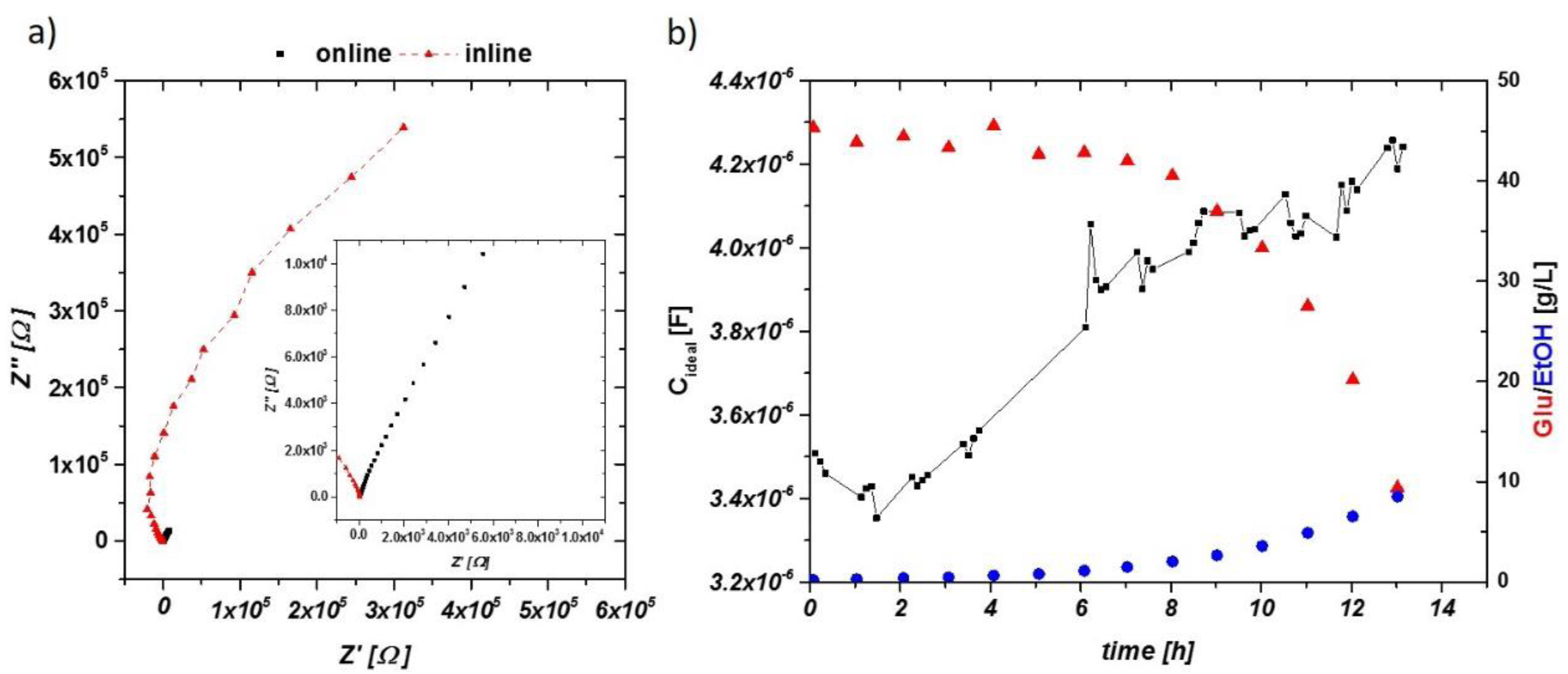
Figure 6.
(a) Impedance raw data in the Nyquist plot for an aerobic cultivation. Black squares represent the signal from the online probe—enlarged in the inlay—and red triangles, the inline probe at similar time stages. Capacitance of the inline probe is one order of magnitude lower (as a result of smaller electrode areas). (b) The dependence of the impedance signal (not smoothed), glucose consumption and ethanol production in an aerobic cultivation using the inline probe.
The capacitance of our almost-ideal plate capacitor design, as built in the inline and online probe, is proportional to
where C is the capacity (F), d the distance between the plates, ε is the dielectric constant (εR·ε0), and A is the area of the electrode. For an electrode with half the diameter, the capacitance signal should decrease by a factor of 4. Stirring and aeration of the system may have effects on the used electrode area, and may reduce the measured capacitance even more.
C = ε·d/A
The extracted idealized capacitance of an aerobic run measured using the inline probe is given in Figure 6b, including process values of glucose consumption and ethanol production. Gaps in the time scale were caused by alternating measurements of the inline and online probes during the cultivation. After 1 h, an increasing signal in the capacitance was found, in accordance with measurements using the online probe. However, higher fluctuations in the signal were visible, making a smoothing of the raw capacitance signal beneficial. Smoothing was performed in the aerobic fermentation using the OriginPro 9 (Northampton, MA, USA) five-point fast fourier transformation (FFT) smoothing procedure.
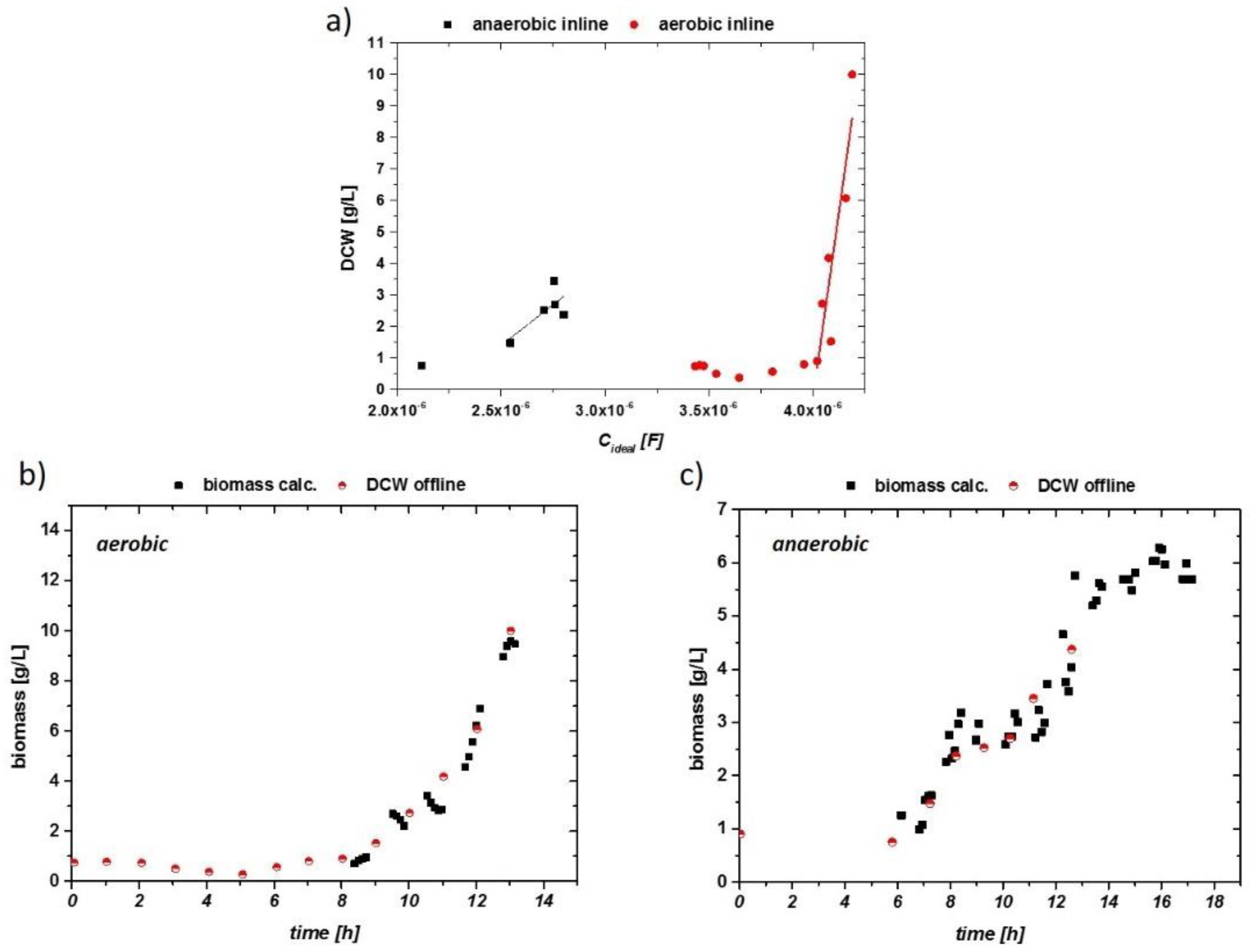
The smoothed signals—especially for the aerobic run—were then used for fitting the biomass data. The corresponding results are given in Figure 7a. Clearly, a higher threshold for accurate data acquisition was found for the inline probe, it having the lower limit of 1 g/L DCW biomass (compared to about 0.3 g/L for the online probe). Exceeding the threshold, a good linear trend can generally be seen for the aerobic and anaerobic fermentation in both samples.
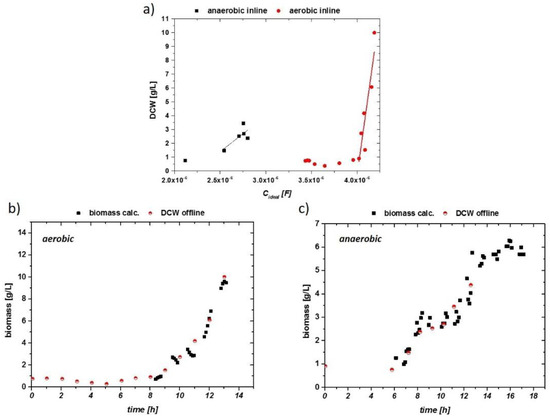
Figure 7.
(a) Impedance signal vs. offline DCW for aerobic (circles) and anaerobic (squares) cultivations with linear fits beyond a threshold of 1 g/l DCW. (b) Impedance signal of aerobic run (smoothed), calculated via a linear fit into a viable biomass. This is correlated to the offline DCW. (c) Impedance signal vs. DCW of the anaerobic run.
The correlations of the calculated biomass compared to the offline biomass are given in Figure 7b,c. Early stages without pronounced growth could not be monitored during the cultivation. The exponential growth could be described accurately with the inline probe, despite high aeration and stirring rates. Promising results were also obtained for the anaerobic cultivation, despite higher fluctuations in the signal. For defined minimal media, the measurement with the inline probe showed reproducible, stable results in the used systems (Sartorius and Techfors with 10 to 20 L as the maximum volume of the fermenter).
Larger systems in the brewing industry, including longer residual times in the side stream, may affect the online signal and change the absolute impedance signal and slopes of the DCW versus capacitance curves. Furthermore, larger tank reactors may include inhomogeneities within the system, which have effects on the signal and have to be taken into account. As residual times in the different bypasses may result in high variations, online probes therefore should be calibrated within the measurement system. Signals of the assembled inline probe are affected by process conditions such as stirring and aeration, which results in higher fluctuations of the signal in general. An increase in the electrode area may have a beneficial impact on the stability of the signal, as absolute capacitance values are one order of magnitude lower, compared to the online probe.
3.3. Aerobic Growth of Yeast on Complex Malt Extract Medium
Defined media have the advantage of good reproducibility and easy analytics, such as OD for biomass determination and HPLC for sugar/ethanol analytics, and are therefore perfectly suited for the first development steps. However, because defined media are scarce in their use in industrial processes for yeast production, malt extract for the production of pilsner beer was used for this cultivation run. Complex media such as malt extract and molasses often have the drawback that OD measurements generally show a very high blank adsorption (especially in the IR range), and cannot easily be used online in those cultivations for the determination of the total biomass.
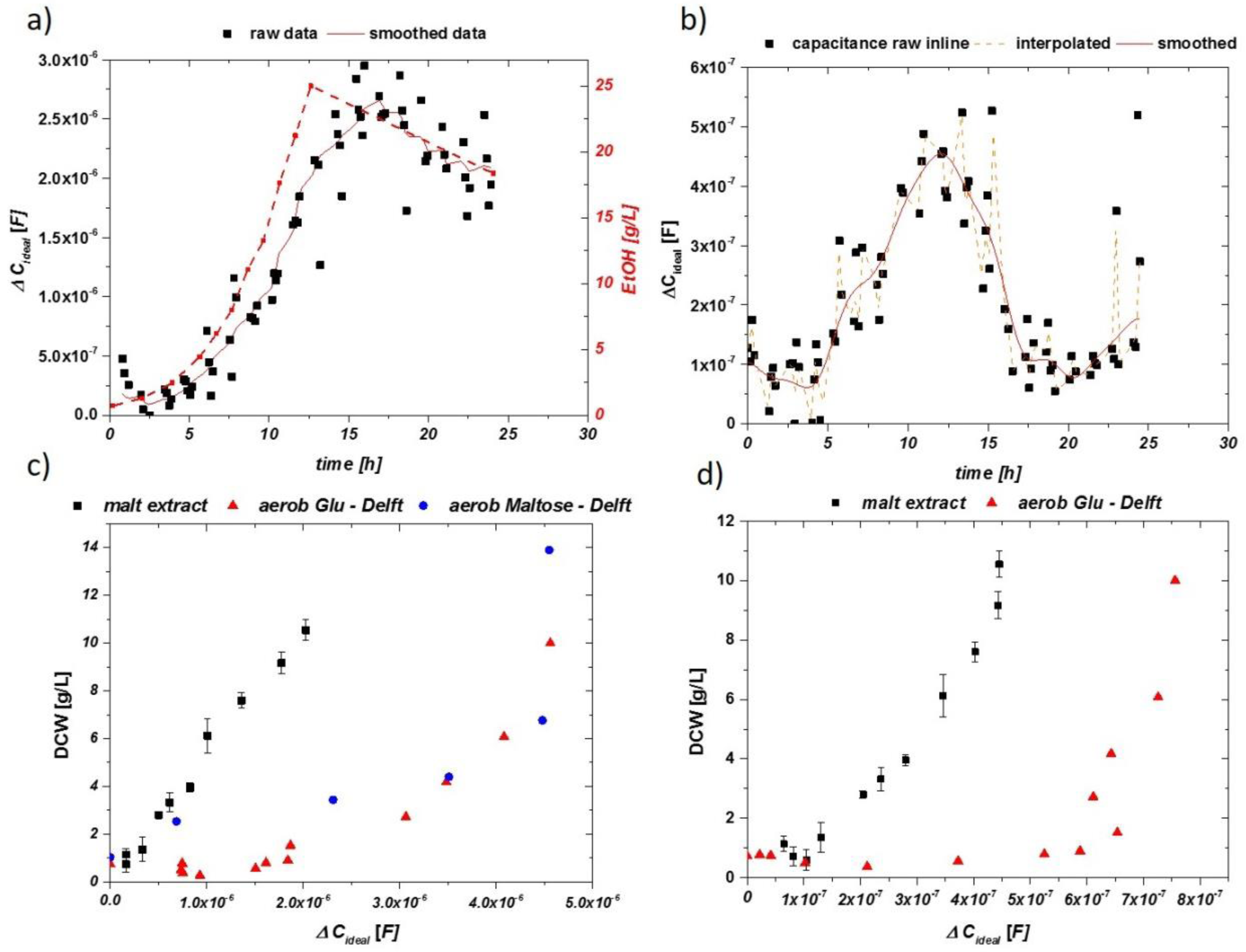
The performed cultivation was analyzed by HPLC and off-gas analytics to determine the end of the batch phase. HPLC data for sugar and ethanol are given in supplementary Figure S1. A mixture of different mono- and poly-saccharides was consumed during fermentation. This led to 10.6 g/L DCW and 3.2 vol % ethanol after the batch phase, at about t = 16 h. Online and inline impedance measurements were performed in the alternating mode for one cultivation. Raw data for the online impedance signal is given in Figure 8a, including information on the ethanol concentration during the cultivation. A steep increase is followed by a shoulder at about 13 h of the cultivation time, which may indicate a change in the sugar metabolism at the end of the batch phase. At process time t = 16 h, a decrease of the impedance signal is visible, generally observed for growth on ethanol. However, the decrease in the signal is rather smooth, compared to the distinct drop in defined media (Figure 2b). Interpolation of the signal and five-point FFT smoothing was performed to reduce the noise in the signal. The same procedure was performed for the inline impedance signal, given in Figure 8b. For the inline signal, a distinct decrease of the capacitance can be spotted after the end of the batch phase, which is accompanied by an increase of ethanol growth at a later process time.
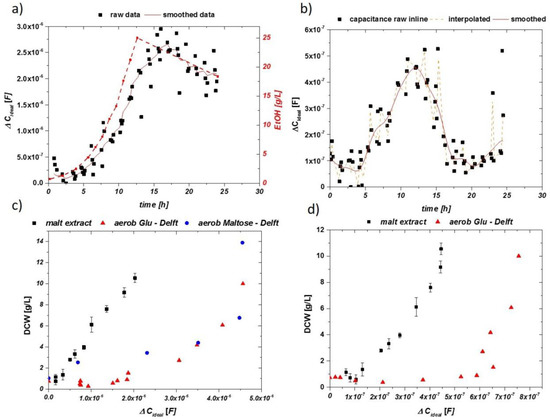
Figure 8.
(a) Impedance signal over the cultivation time for the online probe using only malt extract as growth media. The line (orange) shows the interpolation procedure. As for related aerobic cultivation, a drop in the impedance is observed after consumption of sugars. (b) Impedance signal raw data, interpolated and smoothed for the inline probe. (c) Normalized impedance signal vs. DCW for the online probe using malt extract compared to defined media. (d) Normalized impedance signal vs. DCW for the inline probe using malt extract and defined media with glucose.
The obtained smoothed and interpolated data are compared to the DCW taken and plotted in Figure 8c for the online probe and in Figure 8d for the inline probe. The slopes of the signals are very similar to extracted values for defined media samples (red/blue dots). However, an obvious shift in the signal intensity can be observed for both probes.
A linear fitting was applied to the impedance versus DCW plot in Figure 8c, and DCW was calculated through the impedance signal and compared to the offline measured signal in Figure 9. Beyond the given threshold of 1 g/L, a good description of the process could be achieved by the usage of the inline probe.
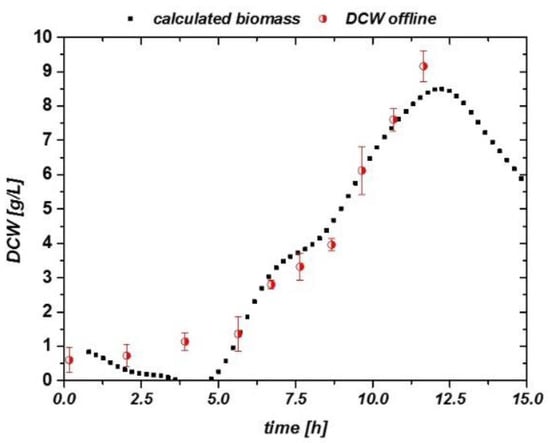
Figure 9.
Impedance signal vs. DCW for the inline probe in complex medium: malt extract. A good description is found beyond 1 g/L DCW in this experiment.
On the basis of these first measurements in complex media, impedance spectroscopy at frequencies in the kHz to mHz range seems to be a promising tool for online process monitoring in yeast production processes, and possibly even in anaerobic refining processes in brewing applications. The present decrease in the signal upon complete sugar consumption is a powerful feature in terms of a stop criterion in these cultivations. An optimization of the signal-to-noise ratio, especially for the inline probe, would be beneficial for increasing the accuracy of the biomass estimation. However, tower-type reactors rather than stirred tank reactors are generally used in brewing applications because of aroma compound reasons [35]. Hence, the stirring and aeration rates used for the development were much harsher compared to those used in the industry, and therefore a much more stable signal is to be expected. Calibration of the probe may be performed once within the used system and growth media, and it should remain stable for forthcoming measurements.
4. Conclusions
New online and inline probes based on EIS at low frequencies for the measurement of VCCs for S. cerevisiae were tested. First, cultivations were monitored using a formerly developed online probe for pharmaceutical E. coli fed-batch cultivations. Batch cultivations on defined media for aerobic and anaerobic growth showed stable results, irrespective of the carbon source or concentrations. A newly assembled inline probe was tested in aerobic and anaerobic cultivations in defined media and was compared to the online probe. A good description of the biomass growth during the process was achieved. Besides the determination of the biomass during the cultivation, physiological states could be determined, depending on the respiratory condition of the cells. This measurement setup for biomass is highly beneficial, especially in complex media such as malt extract or molasses, as optical online methods cannot be used in such optically dense media. The developed system therefore shows high potential for monitoring cell growth and harvest time points for yeast- and beer-producing industries.
Supplementary Materials
The following are available online at www.mdpi.com/2227-9040/5/3/24/s1. Figure S1: HPLC data of the used pilsner malt extract at different time stages. Increase of ethanol is applicable due to late retention time in the chromatogram. Glucose and Galactose are first, afterwards the disaccharides are consumed by the cells. After consumption of all easy accessible sugars, growth on ethanol starts leading to a decrease in ethanol concentration overnight.
Acknowledgments
The authors thank the Christian Doppler Gesellschaft (CDG) for funding.
Author Contributions
C. Slouka, J. Kopp and M. Strahammer performed cultivation. G. Brunauer assembled the online and inline probes and helped during cultivation. J. Fricke, J. Fleig and C. Herwig helped with data evaluation and gave valuable input for the manuscript. C. Slouka, G. Brunauer and J. Fricke prepared the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviation
| dO2 | dissolved oxygen |
| CDL | double layer capacitance |
| EIS | electrochemical impedance spectroscopy |
| DCW | dry cell weight |
| OD | optical density |
| VCC | viable cell concentration |
| AC | alternating current |
| CHO | Chinese hamster ovary (cells) |
| DiBAC | (bis-(1,3-dibutylbarbituricacid)trimethineoxonol) |
| HPLC | high-pressure liquid chromatography |
| BNC | Bayonet Neill Concelman |
| Z | general impedance |
| R | resistance |
| ω | arc frequency |
| CPE | constant phase element |
| ε | dielectric constant |
References
- Gavrilescu, M.; Chisti, Y. Biotechnology—A sustainable alternative for chemical industry. Biotechnol. Adv. 2005, 23, 471–499. [Google Scholar] [CrossRef] [PubMed]
- Sagmeister, P.; Wechselberger, P.; Jazini, M.; Meitz, A.; Langemann, T.; Herwig, C. Soft sensor assisted dynamic bioprocess control: Efficient tools for bioprocess development. Chem. Eng. Sci. 2013, 96, 190–198. [Google Scholar] [CrossRef]
- Clarke, D.; Blake-Coleman, B.; Carr, R.; Calder, M.; Atkinson, T. Monitoring reactor biomass. Trends Biotechnol. 1986, 4, 173–178. [Google Scholar] [CrossRef]
- Kiviharju, K.; Salonen, K.; Moilanen, U.; Eerikäinen, T. Biomass measurement online: The performance of in situ measurements and software sensors. J. Ind. Microbiol. Biotechnol. 2008, 35, 657–665. [Google Scholar] [CrossRef] [PubMed]
- Davey, H.M.; Kell, D.B. Flow cytometry and cell sorting of heterogeneous microbial populations: The importance of single-cell analyses. Microbiol. Rev. 1996, 60, 641–696. [Google Scholar] [PubMed]
- Veal, D.; Deere, D.; Ferrari, B.; Piper, J.; Attfield, P. Fluorescence staining and flow cytometry for monitoring microbial cells. J. Immunol. Methods 2000, 243, 191–210. [Google Scholar] [CrossRef]
- Lodolo, E.J.; Kock, J.L.; Axcell, B.C.; Brooks, M. The yeast Saccharomyces cerevisiae–the main character in beer brewing. FEMS Yeast Res. 2008, 8, 1018–1036. [Google Scholar] [CrossRef] [PubMed]
- Schwan, H.P. Electrical and acoustic properties of biological materials and biomedical applications. IEEE Trans. Biomed. Eng. 1984, 31, 872–878. [Google Scholar] [CrossRef] [PubMed]
- Schwan, H.P.; Foster, K.R. RF-field interactions with biological systems: Electrical properties and biophysical mechanisms. Proc. IEEE 1980, 68, 104–113. [Google Scholar] [CrossRef]
- Yardley, J.E.; Kell, D.B.; Barrett, J.; Davey, C.L. On-line, real-time measurements of cellular biomass using dielectric spectroscopy. Biotechnol. Genet. Eng. Rev. 2000, 17, 3–36. [Google Scholar] [CrossRef] [PubMed]
- Dabros, M.; Dennewald, D.; Currie, D.J.; Lee, M.H.; Todd, R.W.; Marison, I.W.; von Stockar, U. Cole–Cole, linear and multivariate modeling of capacitance data for on-line monitoring of biomass. Bioprocess Biosyst. Eng. 2009, 32, 161–173. [Google Scholar] [CrossRef] [PubMed]
- Soley, A.; Lecina, M.; Gámez, X.; Cairo, J.; Riu, P.; Rosell, X.; Bragos, R.; Godia, F. On-line monitoring of yeast cell growth by impedance spectroscopy. J. Biotechnol. 2005, 118, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Carvell, J.P.; Dowd, J.E. On-line measurements and control of viable cell density in cell culture manufacturing processes using radio-frequency impedance. Cytotechnology 2006, 50, 35–48. [Google Scholar] [CrossRef] [PubMed]
- Gerngross, T.U. Advances in the production of human therapeutic proteins in yeasts and filamentous fungi. Nat. Biotechnol. 2004, 22, 1409–1414. [Google Scholar] [CrossRef] [PubMed]
- Buckholz, R.G.; Gleeson, M.A.G. Yeast Systems for the Commercial Production of Heterologous Proteins. Nat. Biotechol. 1991, 9, 1067–1072. [Google Scholar] [CrossRef]
- Cereghino, G.P.L.; Cregg, J.M. Applications of yeast in biotechnology: Protein production and genetic analysis. Curr. Opin. Biotechnol. 1999, 10, 422–427. [Google Scholar] [CrossRef]
- Konakovsky, V.; Yagtu, A.C.; Clemens, C.; Müller, M.M.; Berger, M.; Schlatter, S.; Herwig, C. Universal Capacitance Model for Real-Time Biomass in Cell Culture. Sensors 2015, 15, 22128–22150. [Google Scholar] [CrossRef] [PubMed]
- Ehgartner, D.; Sagmeister, P.; Herwig, C.; Wechselberger, P. A novel real-time method to estimate volumetric mass biodensity based on the combination of dielectric spectroscopy and soft-sensors. J. Chem. Technol. Biotechnol. 2015, 90, 262–272. [Google Scholar] [CrossRef]
- Ferreira, A.P.; Vieira, L.M.; Cardoso, J.P.; Menezes, J.C. Evaluation of a new annular capacitance probe for biomass monitoring in industrial pilot-scale fermentations. J. Biotechnol. 2005, 116, 403–409. [Google Scholar] [CrossRef] [PubMed]
- Poortinga, A.T.; Bos, R.; Norde, W.; Busscher, H.J. Electric double layer interactions in bacterial adhesion to surfaces. Surf. Sci. Rep. 2002, 47, 1–32. [Google Scholar] [CrossRef]
- Bot, C.; Prodan, C. Probing the membrane potential of living cells by dielectric spectroscopy. Eur. Biophys. J. 2009, 38, 1049–1059. [Google Scholar] [CrossRef] [PubMed]
- Radke, S.M.; Alocilja, E.C. Design and fabrication of a microimpedance biosensor for bacterial detection. IEEE Sens. J. 2004, 4, 434–440. [Google Scholar] [CrossRef]
- Yang, L.; Li, Y.; Griffis, C.L.; Johnson, M.G. Interdigitated microelectrode (IME) impedance sensor for the detection of viable Salmonella typhimurium. Biosens. Bioelectron. 2004, 19, 1139–1147. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Li, H.; Zhang, F.; Li, X.; Wang, L.; Chen, Y. Online impedance monitoring of yeast cell culture behaviors. Microelectron. Eng. 2011, 88, 1711–1713. [Google Scholar] [CrossRef]
- Lei, K.F. Review on impedance detection of cellular responses in micro/nano environment. Micromachines 2014, 5, 1–12. [Google Scholar] [CrossRef]
- Yang, L.; Ruan, C.; Li, Y. Detection of viable Salmonella typhimurium by impedance measurement of electrode capacitance and medium resistance. Biosens. Bioelectron. 2003, 19, 495–502. [Google Scholar] [CrossRef]
- Gonzalez, J.; Santana, A.F.; Mirza-Rosca, J. Effect of bacterial biofilm on 316 SS corrosion in natural seawater by EIS. Corros. Sci. 1998, 40, 2141–2154. [Google Scholar] [CrossRef]
- Bayoudh, S.; Othmane, A.; Ponsonnet, L.; Ouada, H.B. Electrical detection and characterization of bacterial adhesion using electrochemical impedance spectroscopy-based flow chamber. Colloids Surf. A 2008, 318, 291–300. [Google Scholar] [CrossRef]
- Wu, J.; Ben, Y.; Chang, H.-C. Particle detection by electrical impedance spectroscopy with asymmetric-polarization AC electroosmotic trapping. Microfluid. Nanofluid. 2005, 1, 161–167. [Google Scholar] [CrossRef]
- K'Owino, I.O.; Sadik, O.A. Impedance spectroscopy: A powerful tool for rapid biomolecular screening and cell culture monitoring. Electroanalysis 2005, 17, 2101–2113. [Google Scholar] [CrossRef]
- Kim, Y.-H.; Park, J.-S.; Jung, H.-I. An impedimetric biosensor for real-time monitoring of bacterial growth in a microbial fermentor. Sens. Actuators B 2009, 138, 270–277. [Google Scholar] [CrossRef]
- Slouka, C.; Wurm, D.J.; Brunauer, G.; Welzl-Wachter, A.; Spadiut, O.; Fleig, J.; Herwig, C. A Novel Application for Low Frequency Electrochemical Impedance Spectroscopy as an Online Process Monitoring Tool for Viable Cell Concentrations. Sensors 2016, 16, 1900. [Google Scholar] [CrossRef] [PubMed]
- Langemann, T.; Mayr, U.B.; Meitz, A.; Lubitz, W.; Herwig, C. Multi-parameter flow cytometry as a process analytical technology (PAT) approach for the assessment of bacterial ghost production. Appl. Microbiol. Biotechnol. 2016, 100, 409–418. [Google Scholar] [CrossRef] [PubMed]
- Weusthuis, R.A.; Pronk, J.T.; Van Den Broek, P.; Van Dijken, J. Chemostat cultivation as a tool for studies on sugar transport in yeasts. Microbiol. Rev. 1994, 58, 616–630. [Google Scholar] [PubMed]
- Okabe, M.; Katoh, M.; Furugoori, F.; Yoshida, M.; Mitsui, S. Growth and fermentation characteristics of bottom brewer’s yeast under mechanical stirring. J. Ferment. Bioeng. 1992, 73, 148–152. [Google Scholar] [CrossRef]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).