Selection of Effective Moss Control Agents for Polytrichum commune and Marchantia polymorpha in Pinus densiflora Container Seedlings
Abstract
1. Introduction
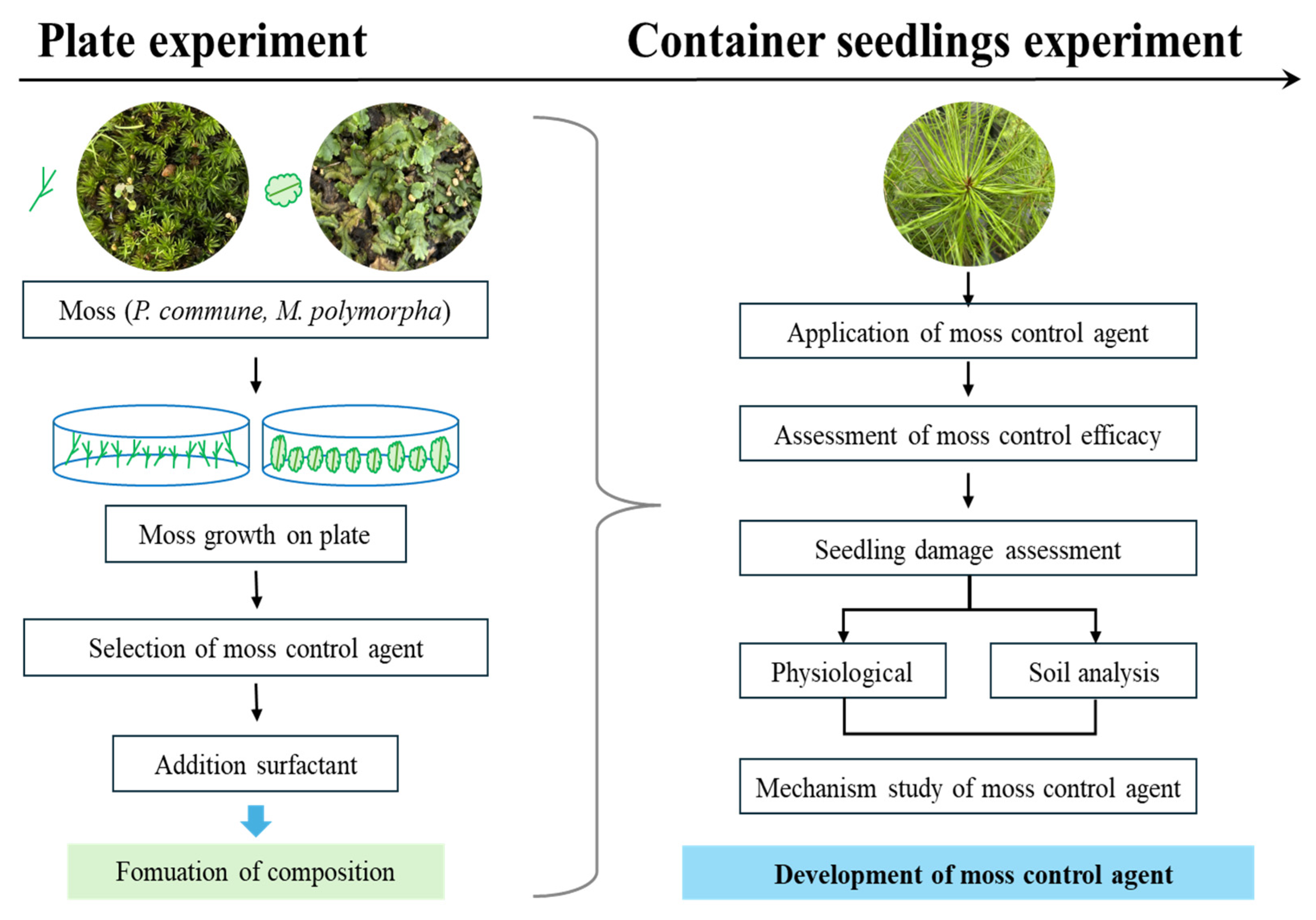
2. Materials and Methods
2.1. Moss and Seedling Materials
2.2. Preparation of Moss Control Agents
2.3. Preparation of Plant Leaf Extracts
2.4. Application of Moss Control Agents and Evaluation of Treatment Efficacy
2.5. Evaluation of Surfactant Effect
2.6. Moss Control Trials on Container Seedlings
2.7. Evaluation of Phytotoxicity in Seedlings Following Moss Control Treatment
2.8. Seedling Physiological and Growth Responses
2.9. Analysis of Soil Physicochemical Properties
2.10. Microscopic Analysis of Moss Control Mechanism
2.11. Statistical Analysis
3. Results
3.1. Effect on Moss Control Agents on Moss Control
3.2. Effect of Surfactant Addition on Moss Control
3.3. Moss Control Effect in P. densiflora Container Seedlings
3.4. Physiological Changes and Seedling Damage, Growth of Moss Control Agent-Treated Seedlings
3.5. Changes in Soil Physicochemical Properties
3.6. Elucidation of the Moss Control Mechanism of Terpenoid-Based Compounds
4. Discussion
4.1. Screening and Evaluation of Effective Moss Control Agents
4.2. The Role of Surfactants in Moss Control
4.3. Moss Control in Container Seedlings
4.4. Effect of Moss Control Agent on Container Seedlings
4.5. Effect of Moss Control Agents on Soil Physicochemical Properties
4.6. Moss Control Efficacy and Phytotoxicity
4.7. Moss Control Mechanism of Terpenoid-Based Compounds
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| TA | Terpinyl acetate |
| HEO | Hinoki essential oil |
| Li | Limonene |
| PA | Pelargonic acid |
| BS | Baking soda |
| PLE | Pine (Pinus densiflora) leaf extract |
| QLE | Quercus glauca leaf extract |
| RLE | Rumex crispus leaf extract |
| ELI | Electrolyte leakage index |
| SDP | Seedling damage point |
| SG | Shoot growth |
| RCG | Root collar diameter growth |
| MCR | Moss control rate |
| EC | Electrical conductivity |
| ICP | Inductively coupled plasma spectroscopy |
| NC | Negative control |
References
- Shaw, J.; Renzaglia, K. Phylogeny and Diversification of Bryophytes 1. Am. J. Bot. 2004, 91, 1557–1581. [Google Scholar] [CrossRef] [PubMed]
- Christenhusz, M.J.M.; Byng, J.W. The Number of Known Plant Species in the World and Its Annual Increase. Phytotaxa 2016, 261, 201–217. [Google Scholar] [CrossRef]
- Fausey, J.C. Controlling Liverwort and Moss Now and in the Future. HortTechnology 2003, 13, 35–38. [Google Scholar] [CrossRef]
- Sidhu, M.K.; Lopez, R.G.; Chaudhari, S.; Saha, D. A Review of Common Liverwort Control Practices in Container Nurseries and Greenhouse Operations. HortTechnology 2020, 30, 471–479. [Google Scholar] [CrossRef]
- Khadduri, N. Using Essential Oils to Control Moss and Liverwort in Containers. In National Proceedings: Forest and Conservation Nursery Associations—2010; Riley, L.E., Haase, D.L., Pinto, J.R., Eds.; Proc. RMRS-P-65; USDA Forest Service, Rocky Mountain Research Station: Fort Collins, CO, USA, 2011; pp. 133–138. [Google Scholar]
- Altland, J.E.; Wehtje, G.; Sibley, J.; Miller, M.E.; Gilliam, C.H.; Krause, C. Differential Response of Liverwort (Marchantia polymorpha) Tissue to POST-Applied Quinoclamine. Weed Technol. 2011, 25, 580–585. [Google Scholar] [CrossRef]
- Dumroese, R.K.; Pinto, J.R.; Jacobs, D.F.; Davis, A.S.; Horiuchi, B. Subirrigation Reduces Water Use, Nitrogen Loss, and Moss Growth in a Container Nursery. Nativ. Plants J. 2006, 7, 253–261. [Google Scholar] [CrossRef]
- Särkkä, L.E.; Tahvonen, R. Control of Liverwort (Marchantia polymorpha L.) Growth in Nursery Plants with Mulches of Sphagnum Moss and Blackcurrant Stem Pieces. Agric. Food Sci. 2020, 29, 250–256. [Google Scholar] [CrossRef]
- Kim, J.J.; Lee, K.J.; Song, K.S.; Cha, Y.G.; Choi, K.S.; Chung, Y.S.; Lee, J.H.; Yoon, T.S. Chemical Control of Mosses in Container Nursery. Res. Plant Dis. 2010, 16, 331–335. [Google Scholar] [CrossRef]
- Newby, A.; Altland, J.E.; Gilliam, C.H.; Wehtje, G. Postemergence Liverwort Control in Container-Grown Nursery Crops. J. Environ. Hortic. 2006, 24, 230–236. [Google Scholar] [CrossRef]
- Yu, X.Y.; Ying, G.G.; Kookana, R.S. Reduced Plant Uptake of Pesticides with Biochar Additions to Soil. Chemosphere 2009, 76, 665–671. [Google Scholar] [CrossRef]
- Steppeler, F.; Iwan, D.; Wojaczyńska, E.; Wojaczyński, J. Chiral Thioureas—Preparation and Significance in Asymmetric Synthesis and Medicinal Chemistry. Molecules 2020, 25, 401. [Google Scholar] [CrossRef]
- Komaki, N.; Watanabe, T.; Ogasawara, A.; Sato, N.; Mikami, T.; Matsumoto, T. Antifungal Mechanism of Hinokitiol against Candida albicans. Biol. Pharm. Bull. 2008, 31, 735–737. [Google Scholar] [CrossRef] [PubMed]
- Bajpai, V.K.; Baek, K.H.; Kang, S.C. Control of Salmonella in Foods by Using Essential Oils: A Review. Food Res. Int. 2012, 45, 722–734. [Google Scholar] [CrossRef]
- Nazzaro, F.; Fratianni, F.; Coppola, R.; Feo, V.D. Essential Oils and Antifungal Activity. Pharmaceuticals 2017, 10, 86. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Kim, J.G. Evaluation of Herbicidal Activity of Vinegar and Wood Vinegar against Weed Species. Weed Turf. Science 2018, 7, 140–146. [Google Scholar] [CrossRef]
- Dudai, N.; Larkov, O.; Putievsky, E.; Ravid, U.; Lewinsohn, E. Essential Oils as Allelochemicals and Their Potential Use as Bioherbicides. J. Chem. Ecol. 1999, 25, 1079–1089. [Google Scholar] [CrossRef]
- Loddo, D.; Jagarapu, K.K.; Strati, E.; Trespidi, G.; Nikolić, N.; Masin, R.; Berti, A.; Otto, S. Assessing Herbicide Efficacy of Pelargonic Acid on Several Weed Species. Agronomy 2023, 13, 1511. [Google Scholar] [CrossRef]
- Lin, H.; Zhou, H.; Xu, L.; Zhu, H.; Huang, H. Effect of Surfactant Concentration on the Spreading Properties of Pesticide Droplets on Eucalyptus Leaves. Biosyst. Eng. 2016, 143, 42–49. [Google Scholar] [CrossRef]
- Yoo, J.Y.; Cho, K.Y.; Kim, J.H. Review of the Study on the Surfactant-Induced Foliar Uptake of Pesticide. Korean J. Pestic. Sci. 2002, 6, 16–24. [Google Scholar]
- Do, Q.D.; Angkawijaya, A.E.; Tran-Nguyen, P.L.; Huynh, L.H.; Soetaredjo, F.E.; Ismadji, S.; Ju, Y.H. Effect of extraction solvent on total phenol content, total flavonoid content, and antioxidant activity of Limnophila aromatica. J. Food Drug Anal. 2014, 22, 296–302. [Google Scholar] [CrossRef]
- Kumar, S.; Pandey, A.K. Chemistry and biological activities of flavonoids: An overview. Sci. World J. 2013, 2013, 162750. [Google Scholar] [CrossRef] [PubMed]
- Altemimi, A.; Lakhssassi, N.; Baharlouei, A.; Watson, D.G.; Lightfoot, D.A. Phytochemicals: Extraction, isolation, and identification of bioactive compounds from plant extracts. Plants 2017, 6, 42. [Google Scholar] [CrossRef] [PubMed]
- Park, D.-J.; Kim, D.-H.; Yong, S.-H.; Kim, S.-A.; Park, K.-B.; Cha, S.-A.; Lee, J.-H.; Choi, M.-S. Establishment of Efficient Method for Evaluation of Heat Stress Tolerance of Herbaceous Plant Species and Selection of Heat-Tolerant Plants. Horticulturae 2024, 10, 1290. [Google Scholar] [CrossRef]
- Campolo, O.; Giunti, G.; Laigle, M.; Michel, T.; Palmeri, V. Essential oil-based nano-emulsions: Effect of different surfactants, sonication and plant species on physicochemical characteristics. Ind. Crops Prod. 2020, 157, 112935. [Google Scholar] [CrossRef]
- Chutia, M.; Bhuyan, P.D.; Pathak, M.G.; Sarma, T.C.; Boruah, P. Antifungal Activity and Chemical Composition of Citrus reticulata Blanco Essential Oil against Phytopathogenic Fungi. LWT-Food Sci. Technol. 2009, 42, 777–780. [Google Scholar] [CrossRef]
- Shimamura, M. Marchantia polymorpha: Taxonomy, Phylogeny and Morphology of a Model System. Plant Cell Physiol. 2016, 57, 230–256. [Google Scholar] [CrossRef]
- Turek, C.; Stintzing, F.C. Stability of essential oils: A review. Compr. Rev. Food Sci. Food Saf. 2013, 12, 40–53. [Google Scholar] [CrossRef]
- Mishler, B.D.; Churchill, S.P. Transition to a Land Flora: Phylogenetic Relationships of the Green Algae and Bryophytes. Cladistics 1985, 1, 305–328. [Google Scholar] [CrossRef]
- Al-Thahab, D.L.; Al-Jibouri, M.M. Allelopathic Properties of Essential Oils: Potential Role in Weed Management. Allelopath. J. 2016, 38, 1–20. [Google Scholar]
- Lama, A.D.; Klemola, T.; Tyystjärvi, E.; Niemelä, P.; Vuorisalo, T. Physiological and Compensatory Growth Responses of Jatropha curcas (L.) Seedlings to Simulated Herbivory and Drought Stress. S. Afr. J. Bot. 2019, 121, 486–493. [Google Scholar] [CrossRef]
- Altland, J.E.; Wehtje, G.; Gilliam, C.H.; Miller, M.E. Liverwort (Marchantia polymorpha) Control with Quinoclamine. Weed Technol. 2007, 21, 483–488. [Google Scholar] [CrossRef]
- Crandall-Stotler, B.; Stotler, R.E.; Long, D.G. Phylogeny and Classification of the Marchantiophyta. Edinb. J. Bot. 2009, 66, 155–198. [Google Scholar] [CrossRef]
- Anderson, R.G.; Deb, D.; Fedkenheuer, K.; McDowell, T. Herbicidal Effects and Non-Target Impacts of Pelargonic Acid on Crop and Weed Species. Weed Technol. 2018, 32, 559–566. [Google Scholar] [CrossRef]
- Egorov, A.; Bubnov, A.; Pavluchenkova, L.; Partolina, A.; Postnikov, A. Applying Chemical Control to Suppress Liverwort (Marchantia polymorpha L.) and Other Mosses When Growing Containerized Seedlings of Pine and Spruce. Balt. For. 2021, 27, id288. [Google Scholar] [CrossRef]
- Webber, C.L.; Taylor, M.J.; Shrefler, J.W. Weed Control in Yellow Squash Using Sequential Postdirected Applications of Pelargonic Acid. HortTechnology 2014, 24, 25–29. [Google Scholar] [CrossRef]
- Morita, Y.; Sakagami, Y.; Okabe, T.; Ohe, T.; Inamori, Y.; Ishida, N. The Mechanism of the Bactericidal Activity of Hinokitiol. Biocontrol Sci. 2007, 12, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Angelini, L.G.; Carpanese, G.; Cioni, P.L.; Morelli, I.; Macchia, M.; Flamini, G. Essential Oils from Mediterranean Lamiaceae as Weed Germination Inhibitors. J. Agric. Food Chem. 2003, 51, 6158–6164. [Google Scholar] [CrossRef] [PubMed]
- Anderson, C.T.; Carroll, A.; Akhmetova, L.; Somerville, C. Real-Time Imaging of Cellulose Reorientation during Cell Wall Expansion in Arabidopsis Roots. Plant Physiol. 2010, 152, 787–796. [Google Scholar] [CrossRef] [PubMed]
- Cosgrove, D.J. Reconstructing Our Models of Cellulose and Primary Cell Wall Assembly. Curr. Opin. Plant Biol. 2014, 22, 122–131. [Google Scholar] [CrossRef]










| ELI | SDP | SG | RCG | MCR | |
|---|---|---|---|---|---|
| ELI | 1 | ||||
| SDP | 0.743 ** | 1 | |||
| SG | −0.594 * | −0.970 ** | 1 | ||
| RCG | −0.746 * | −0.878 ** | 0.847 * | 1 | |
| MCR | 0.525 | 0.817 ** | −0.766 ** | −0.695 * | 1 |
| Treatment | pH | Organisms (g/kg) | P (mg/kg) | Exchangeable Cation cmol+/kg | EC (dS/m) | ||
|---|---|---|---|---|---|---|---|
| K | Ca | Mg | |||||
| Control | 7.7 | 0 | 8 | 0.74 | 6.6 | 5.1 | 0.1 |
| Surfactant control (5%) | 7.1 | 0 | 4 | 0.67 | 5.9 | 4.7 | 0.1 |
| Baking soda | 7.1 | 0 | 6 | 0.66 | 6.4 | 4.4 | 0.1 |
| Pelargonic acid | 6.8 | 0 | 6 | 0.67 | 5.9 | 3.8 | 0.1 |
| Terpinyl acetate | 6.9 | 0 | 7 | 0.66 | 7.6 | 5.1 | 0.1 |
| Limonene | 6.9 | 0 | 7 | 0.56 | 6.3 | 4.2 | 0.1 |
| Hinoki essential oil | 6.9 | 0 | 7 | 0.56 | 6.3 | 4.2 | 0.1 |
| pine leaf extract | 6.7 | 0 | 9 | 0.56 | 6.3 | 4.3 | 0.2 |
| Quinoclamine product (9%) | 6.8 | 0 | 5 | 0.56 | 6.7 | 4.5 | 0.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, S.-H.; Lee, J.-H.; Yong, S.-H.; Kim, S.-A.; Kim, D.-H.; Park, K.-B.; Cha, S.-A.; Jung, J.; Kim, H.-S.; Choi, M.-S. Selection of Effective Moss Control Agents for Polytrichum commune and Marchantia polymorpha in Pinus densiflora Container Seedlings. Plants 2025, 14, 3417. https://doi.org/10.3390/plants14223417
Han S-H, Lee J-H, Yong S-H, Kim S-A, Kim D-H, Park K-B, Cha S-A, Jung J, Kim H-S, Choi M-S. Selection of Effective Moss Control Agents for Polytrichum commune and Marchantia polymorpha in Pinus densiflora Container Seedlings. Plants. 2025; 14(22):3417. https://doi.org/10.3390/plants14223417
Chicago/Turabian StyleHan, Seung-Hyun, Ji-Hyeon Lee, Seong-Hyeon Yong, Seon-A Kim, Do-Hyun Kim, Kwan-Been Park, Seung-A Cha, Jenna Jung, Hyun-Seop Kim, and Myung-Suk Choi. 2025. "Selection of Effective Moss Control Agents for Polytrichum commune and Marchantia polymorpha in Pinus densiflora Container Seedlings" Plants 14, no. 22: 3417. https://doi.org/10.3390/plants14223417
APA StyleHan, S.-H., Lee, J.-H., Yong, S.-H., Kim, S.-A., Kim, D.-H., Park, K.-B., Cha, S.-A., Jung, J., Kim, H.-S., & Choi, M.-S. (2025). Selection of Effective Moss Control Agents for Polytrichum commune and Marchantia polymorpha in Pinus densiflora Container Seedlings. Plants, 14(22), 3417. https://doi.org/10.3390/plants14223417







