Abstract
Lomatium is a genus of 98 species, widely distributed in western North America. This work presents a chemometric analysis of the essential oils of seven species of Lomatium (L. anomalum, L. dissectum var. dissectum, L. multifidum, L. nudicaule, L. packardiae, L. papilioniferum, and L. triternatum var. triternatum) from the intermountain western United States (Oregon and Idaho). The essential oils were obtained by hydrodistillation and analyzed by gas chromatographic methods. Lomatium packardiae essential oil can be characterized as limonene-rich, L. anomalum is a species rich in sabinene and α-pinene, and L. multifidum essential oils were rich in myrcene, while L. dissectum var. dissectum essential oils were dominated by octyl acetate and decyl acetate, L. papilioniferum essential oils from western Idaho had high p-cymene and 2-methyl-5-(1,2,2-trimethylcyclopentyl)phenol concentrations, while those from Oregon had relatively high β-phellandrene and sedanenolide levels. The essential oils of L. triternatum var. triternatum were too variable to confidently assign a chemical type. The major components in the L. nudicaule essential oils were β-phellandrene (16.0–45.7%), (Z)-ligustilide (5.6–47.1%), (E)-β-ocimene (3.3–9.9%), and δ-3-carene (0.2–12.6%). The enantiomeric distributions of α-pinene, camphene, sabinene, β-pinene, limonene, and linalool were also utilized to discriminate between the Lomatium taxa. There are not enough consistent data to properly characterize L. triternatum var. triternatum or the Oregon L. papilioniferum essential oils. Additional research is needed to confidently describe the chemotype(s) of these species.
Keywords:
anomalum; dissectum; grayi; nudicaule; packardiae; papilioniferum; triternatum; chemotaxonomy; enantiomers 1. Introduction
The genus Lomatium Raf. (Apiaceae) comprises around 98 species, which are distributed in western North America [1]. The genus is part of one of the largest plant radiations in North America, the Perennial Endemic North American Apiaceae (PENA) clade [2,3]. Several species of Lomatium have been used by Native Americans of the Pacific Northwest as medicines as well as food [4]. As part of our continuing interest in essential oils from aromatic and medicinal plants in the intermountain western United States, the purpose of this work is to examine the essential oil compositions, including the enantiomeric distributions of chiral terpenoids, of Lomatium species growing in eastern Oregon and western Idaho.
The Lomatium triternatum (nineleaf biscuitroot) complex has a widespread distribution from British Columbia, south into northern California, and east to Montana, Wyoming, Colorado, and New Mexico [5,6]. It is a perennial herb (ca. 20–80 cm tall) growing from a taproot. The leaves are basal with petioles 8–20 cm long, leaves divided 1–3 times. The inflorescence is a loose flat umbel of yellow flowers on stalks 3–10 cm long. The seeds are flat with five ribs and thin wings on the sides [5,7,8]. The taxonomy of the L. triternatum is complex, is not well delineated, and is in flux [2,3,9,10]. These include, but are not necessarily limited to, Lomatium triternatum (Pursh) J.M. Coult. & Rose (which includes the infraspecific taxa L. triternatum var. triternatum, Lomatium triternatum f. lancifolium (H. St. John) H. St. John, Lomatium triternatum subsp. platycarpum (Torr.) Cronquist, Lomatium triternatum var. brevifolium (J.M. Coult. & Rose) Mathias, and Lomatium triternatum var. macrocarpum (J.M. Coult. & Rose) Mathias), Lomatium anomalum Jones ex J.M. Coult. & Rose, and Lomatium packardiae Cronquist [1]. As far as we are aware, there are no reports on the essential oils of L. triternatum. The purpose of this research is to examine the hypothesis that the volatile phytochemistry of the different taxa of L. triternatum will delineate the members of the complex.
Lomatium grayi (J.M. Coult. & Rose) J.M. Coult. & Rose (Gray’s biscuitroot) is a large (up to 60 cm tall) perennial herb with a branched basal stem structure and finely divided leaves with a pungent odor. The inflorescence is an umbel with numerous yellow flowers [11]. The native range of L. grayi is east of the Cascades in southern British Columbia, Washington and Oregon, northern Nevada, western Idaho, Utah, western Wyoming, western Colorado, and northwestern New Mexico [12]. However, the Lomatium grayi complex is morphologically diverse across its range. Alexander and co-workers have proposed splitting L. grayi into four species based on morphometric analysis [13]. These include Lomatium papilioniferum J.A. Alexander & Whaley (distributed east of the Cascades in southern British Columbia, Washington, Oregon, northern Nevada, and western Idaho), Lomatium klickitatense J.A. Alexander & Whaley (found in Klickitat County, Washington, and surrounding areas), Lomatium depauperatum (M.E. Jones) J.A. Alexander & Whaley (syn. Lomatium grayi var. depauperatum (M.E. Jones) Mathias) (ranges in western Utah and eastern Nevada), and Lomatium grayi (in eastern Idaho, eastern Utah, southwestern Wyoming, and western Colorado). In Idaho, L. papilioniferum is found in western and central Idaho while L. grayi is found only in southeastern Idaho.
Lomatium dissectum (fernleaf biscuitroot) is a perennial herb. The inflorescence is an umbel of numerous small maroon red flowers; the leaves are ternate-pinnately dissected, 15–35 cm wide with a 3–30 cm petiole. The fruit is oblong-ovate to elliptic, 12–16 mm long, with thick lateral wings. The plant occurs in western North America, northern California, north into Washington, and east into Idaho [14,15]. Lomatium dissectum (Nutt.) Mathias & Constance var. dissectum, Lomatium dissectum var. multifidum (Nutt.) Mathias & Constance, and Lomatium dissectum var. eatonii (J.M. Coult. & Rose) Cronquist had been treated as varieties of L. dissectum. However, they are currently treated as separate species, Lomatium dissectum (Nutt.) Mathias & Constance and Lomatium multifidum (Nutt.) R.P. McNeill & Darrach (syn. Lomatium dissectum var. multifidum (Nutt.) Mathias & Constance and Lomatium dissectum var. eatonii (J.M. Coult. & Rose) Cronquist) [14].
Lomatium multifidum is a perennial herb, growing up to 1.2 m tall. The leaves are triangular-ovate to round and ternate-pinnately dissected. The inflorescence is an umbel of numerous small yellow flowers; the fruit is dorsally compressed, with the lateral wings usually well developed. The plant occurs in arid regions of the western United States (Arizona, California, Colorado, Idaho, Montana, Nevada, Oregon, Utah, Washington, and Wyoming) and into southwestern Canada (British Columbia, Alberta, and Saskatchewan) [16,17].
Lomatium nudicaule (Nutt.) J.M. Coult. & Rose (barestem biscuitroot) is a perennial forb with a stout taproot. The plant can reach a height of 20–45 cm; the leaves are compound ternate to biternate, leaflets are oval, 2–5 cm long; the inflorescence is an umbel with yellow flowers; fruits are 8–12 mm long, 2–5 mm wide, with 0.5 mm wide wings. The plant ranges from southern British Columbia, south through Washington and Oregon and into northern California, and east into Idaho Nevada and northwestern Utah [5,18]. There have been no previous reports on the essential oil of L. nudicaule.
In this work, we present the essential oil compositions of L. anomalum (Figure 1), L. dissectum (Figure 2), L. multifidum (Figure 3), L. nudicaule (Figure 4), L. packardiae (Figure 5), L. papilioniferum (Figure 6), and L. triternatum var. triternatum (Figure 7). The purpose of this study is to characterize the volatile components of understudied Lomatium species, including enantiomeric distributions of chiral terpenoid components.

Figure 1.
Lomatium anomalum Jones ex J.M. Coult. & Rose. (A) Several plants at time of collection (2 June 2022, photograph by K. Swor). (B) Photograph of plants at time of collection (30 May 2024, photograph by K. Swor). (C) Scan of pressed plant.

Figure 2.
Lomatium dissectum (Nutt.) Mathias & Constance. (A) Several plants (photograph by W.N. Setzer). (B) Closeup of the inflorescence (photograph by K. Swor). (C) Scan of the pressed plant.

Figure 3.
Lomatium multifidum (Nutt.) R.P. McNeill & Darrach. (A) Flowering stage (photograph by K. Swor). (B) Fruiting stage (photograph by W.N. Setzer). (C) Scan of the pressed plant.

Figure 4.
Lomatium nudicaule (Nutt.) J.M. Coult. & Rose. (A) Photograph of the plant at the time of collection (21 May 2024, photograph by W.N. Setzer). (B) Closeup of the fruits (© Paul Schlichter, with permission [19]). (C) Scan of the pressed plant.

Figure 5.
Lomatium packardiae Cronquist. (A) Photograph of the plant (W.N. Setzer). (B) Scan of the pressed plant material.

Figure 6.
Lomatium papilioniferum J.A. Alexander & Whaley. (A) Flowering stage (photo by K. Swor). (B) Fruiting stage (photo by W.N. Setzer). (C) Scan of pressed plant material.

Figure 7.
Lomatium triternatum (Pursh) J.M. Coult. & Rose var. triternatum. (A) Photograph of the plant (K. Swor). (B) Scan of the pressed plant material.
2. Results and Discussion
There have been several investigations on Lomatium essential oils reported in the literature. A summary of the major components is listed in Table 1.

Table 1.
Major components of Lomatium essential oils reported in the literature.
2.1. Lomatium anomalum (L. triternatum Complex)
Three samples of L. anomalum were collected near Grangeville, western Idaho. Hydrodistillation gave colorless or pale yellow essential oils in yields of 1.57–1.68%. The gas chromatographic results are summarized in Table 2. The essential oils were dominated by sabinene (48.0–49.9%) and α-pinene (21.9–37.6%).

Table 2.
Essential oil composition (%) of Lomatium anomalum Jones ex J.M. Coult. & Rose.
2.2. Lomatium packardiae (L. triternatum Complex)
Four samples of L. packardiae were collected, two from the Arrowrock Reservoir area (Idaho) and two from the Midvale area (Idaho). The essential oil yields ranged from 1.04% to 1.92%. The essential oil compositions are summarized in Table 3. The major components in the essential oils of L. packardiae were limonene (48.6–72.2%), (Z)-ligustilide (12.3–19.1%), and β-phellandrene (4.4–6.2%).

Table 3.
Essential oil composition (%) of Lomatium packardiae Cronguist.
2.3. Lomatium triternatum var. triternatum (L. triternatum Complex)
Three individual samples of L. triternatum triternatum were collected near Prairie, Idaho. The chemical compositions of the L. triternatum triternatum essential oils are summarized in Table 4. Although the three samples were collected from the same location on the same day, there was remarkable variation in the essential oil compositions. For example, monoterpene hydrocarbons ranged from a high of 62.2% in sample Ltt#1 to a low of 13.8% in sample Ltt#2, while oxygenated monoterpenoids were highest in Ltt#2 (39.1%) but lowest in Ltt#3 (2.8%). These are reflected in β-phellandrene concentrations (48.5% and 29.4% in Ltt#1 and Ltt#3, respectively, but only 1.7% in Ltt#2) and myrcene concentrations (12.7% and 14.1% in Ltt#1 and Ltt#3, respectively, but 2.9% in Ltt#2). On the other hand, the cryptone concentration was highest in Ltt#2 (17.9%) compared to either Ltt#1 or Ltt#3 (3.7% and 0.8%). It is not clear what effects may have resulted in these vast differences.

Table 4.
Essential oil composition (%) of Lomatium triternatum (Pursh) J.M. Coult. & Rose var. triternatum.
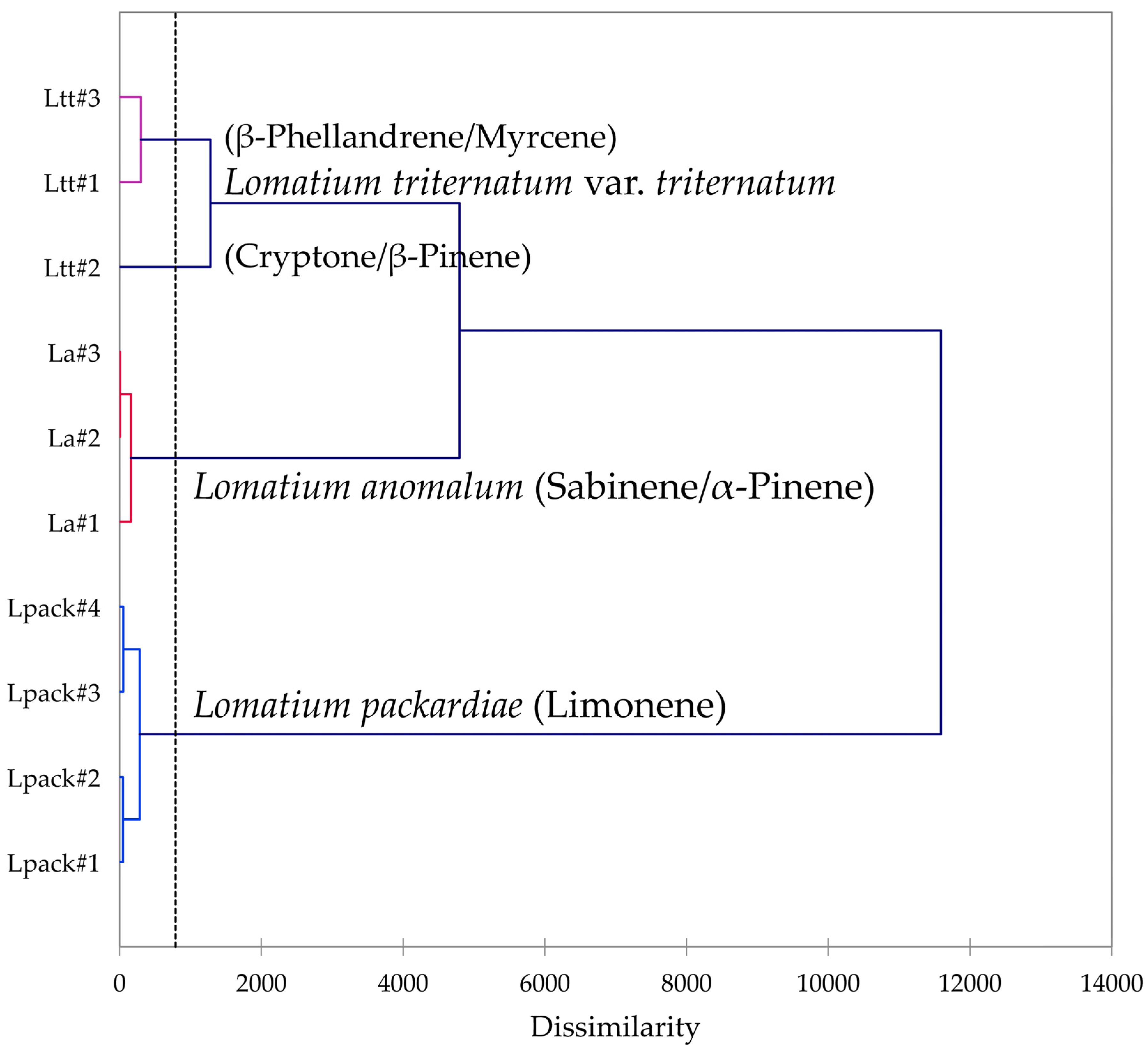
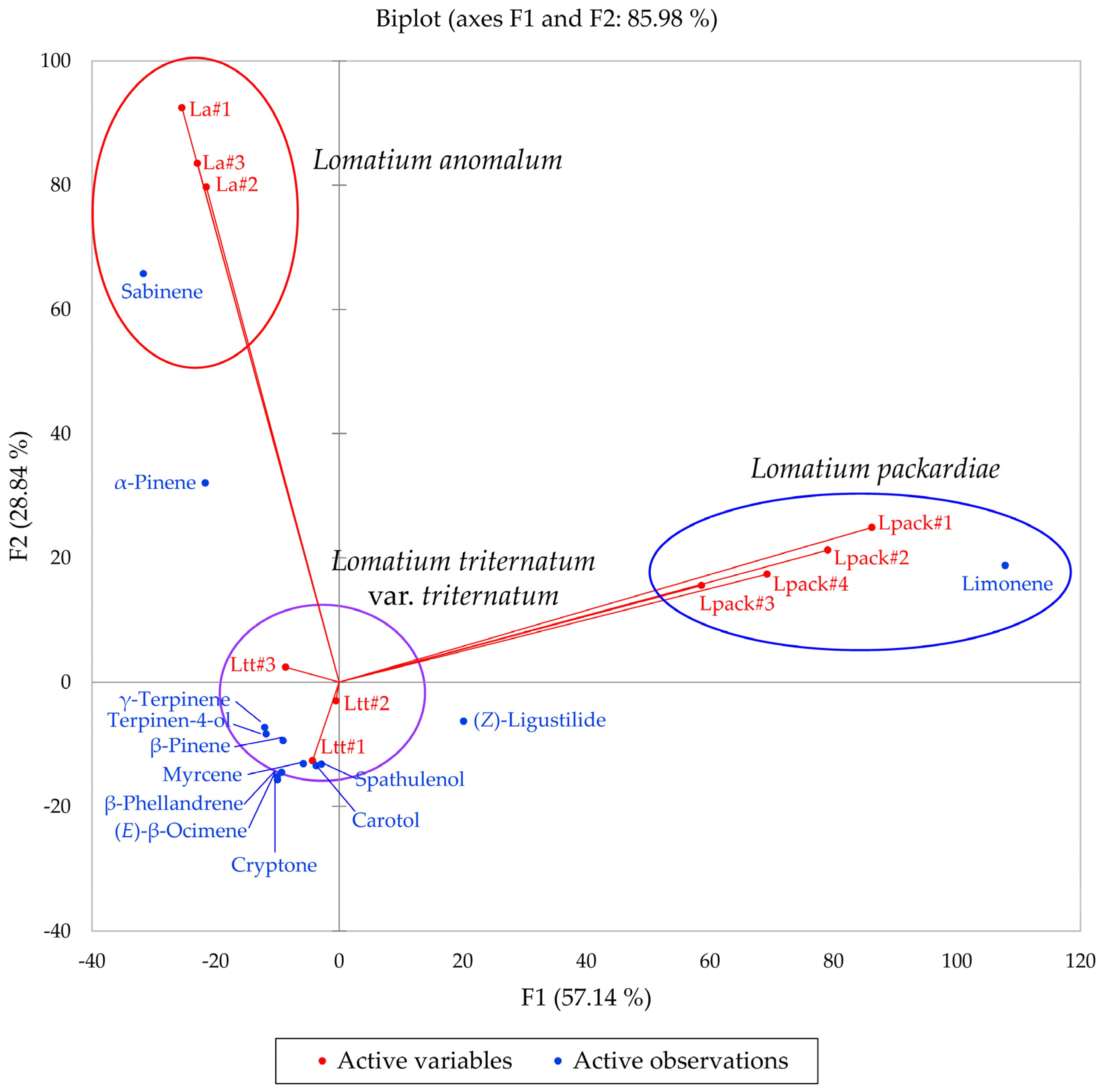
Multivariate analyses were performed using the essential oil compositions of L. anomalum, L. packardiae, and L. triternatum, three members of the L. triternatum complex, in order to visualize the chemical relationships between the three taxa. A hierarchical cluster analysis (HCA, Figure 8) confirms the large degree of dissimilarity between L. anomalum, L. packardiae, and L. triternatum. The HCA clearly separates the three taxa, the limonene-rich L. packardiae, the sabinene/α-pinene L. anomalum, and the L. triternatum group. The L. triternatum group is further subdivided in a β-phellandrene/myrcene type and a cryptone/β-pinene type. A principal component analysis (PCA, Figure 9) corroborates the groupings and their chemical correlations.
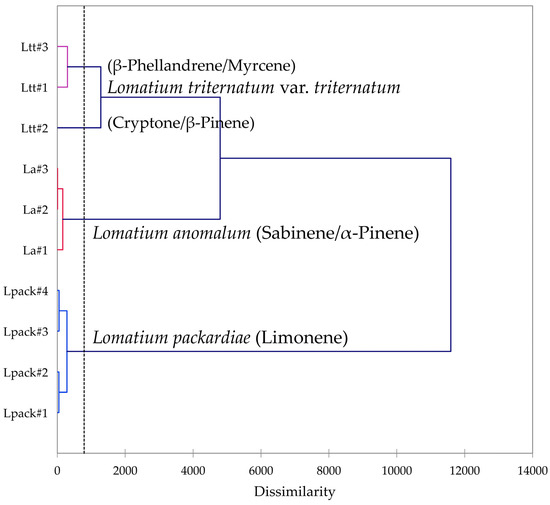
Figure 8.
Dendrogram obtained by hierarchical cluster analysis (HCA) of essential oil compositions (major essential oil components) of members of the Lomatium triternatum complex.
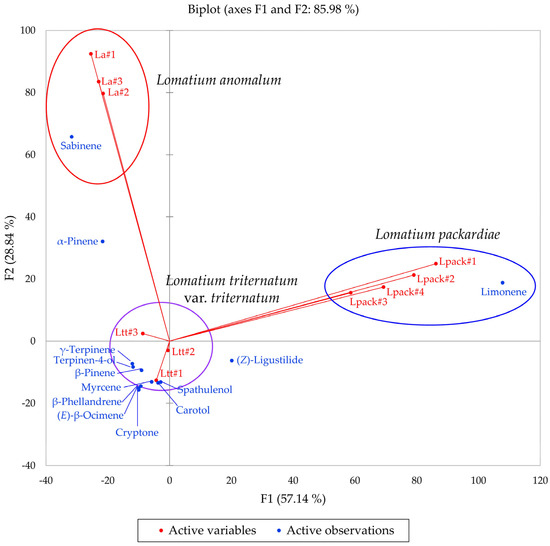
Figure 9.
The bidimensional plot of the first two components (F1 and F2) from principal component analysis (PCA) of members of the Lomatium triternatum complex, based on major components in their essential oils. La = Lomatium anomalum, Lpack = Lomatium packardiae, Ltt = Lomatium triternatum var. triternatum.
2.4. Lomatium dissectum (Lomatium dissectum Complex)
Five different individual plants were collected near Grangeville, Idaho. Hydrodistillation of the samples gave colorless essential oils in yields ranging from 1.94% to 2.74%. The chemical compositions of the essential oils are compiled in Table 5. Interestingly, terpenoids were found in very small quantities in L. dissectum essential oils. Fatty-acid-derived compounds, however, were the major components, including octyl acetate (37.8–48.4%), decyl acetate (33.9–45.8%), and decanol (9.8–18.4%). These results show some qualitative similarities to that reported by Bairamian and co-workers [22] on a sample from northern California. However, quantitatively, the samples are very different. The California sample had 5.3% octyl acetate, 3.2% decyl acetate, and 1.2% decanol, but a large concentration of palmitic acid (15.3%), which was found in only trace quantities in the samples from Idaho.

Table 5.
Essential oil compositions (%) of Lomatium dissectum (Nutt.) Mathias & Constance.
2.5. Lomatium multifidum (Lomatium dissectum Complex)
A total of 12 samples of L. multifidum were collected from locations in eastern Oregon and western Idaho. The essential oils obtained were colorless to yellow with yields ranging from 1.60% to 6.15%. The chemical compositions of the L. multifidum essential oils are shown in Table 6. A total of 206 compounds were identified in the essential oils of L. multifidum, which accounted for 87.9% to 99.3% of the total compositions. There was some variation in the compositions of the essential oils. The major components were myrcene (12.5–54.1%), (E)-β-ocimene (0.3–37.4%), limonene (0.7–14.0%), α-bisabolol (0.0–26.3%), and β-phellandrene (trace-21.3%). In contrast, a sample of L. multifidum (reported as Lomatium dissectum var. multifidum) from southern California showed 6.0% myrcene, 1.0% (E)-β-ocimene, 3.3% limonene + β-phellandrene, and 0.1% α-bisabolol [22].

Table 6.
Chemical compositions (%) of the essential oils of Lomatium multifidum (Nutt.) R.P. McNeill & Darrach.
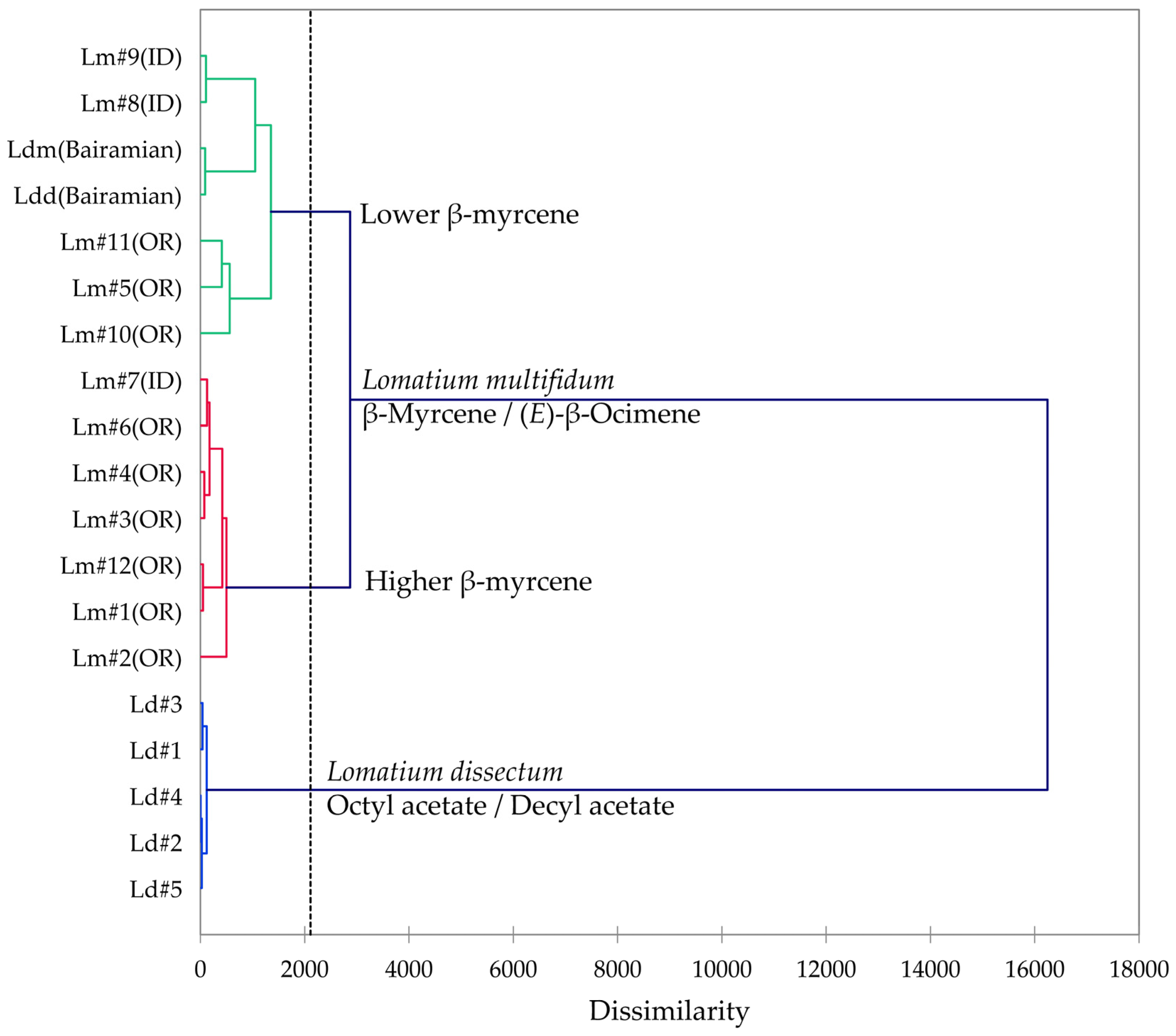
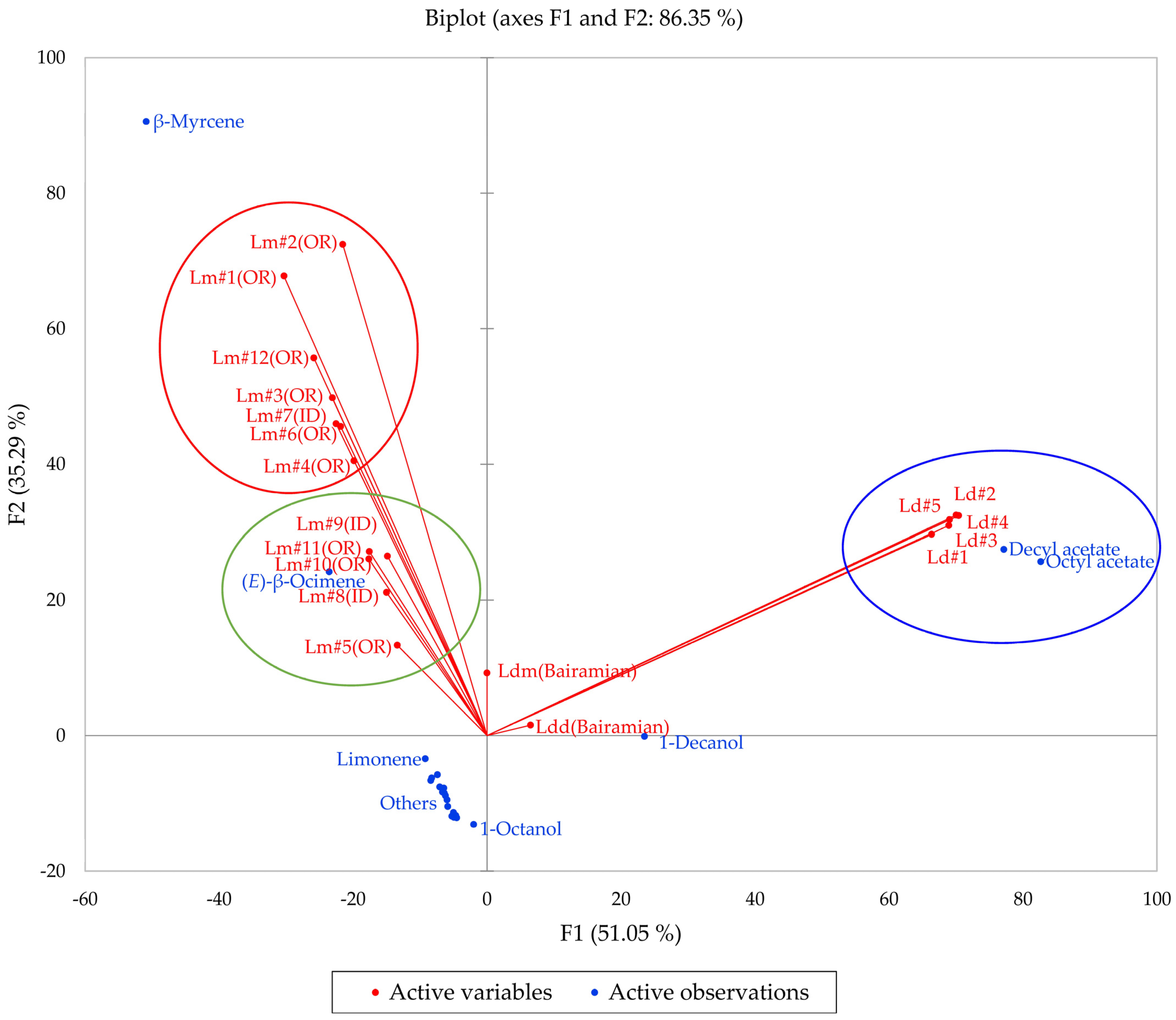
Multivariate analyses (HCA and PCA) were carried out in order to visualize the chemical differences and associations in the essential oils of the two members of the L. dissectum complex (L. dissectum and L. multifidum). The HCA dendrogram and the PCA biplot are shown in Figure 10 and Figure 11, respectively. The HCA shows two major clusters: (1) a cluster made up of L. dissectum samples, dominated by octyl acetate and decyl acetate, and (2) a cluster with β-myrcene and (E)-β-ocimene as defining components and populated by L. multifidum samples. The L. dissectum and L. multifidum samples from Bairamian and co-workers [22] were included in the HCA for comparison. The L. multifidum cluster can be subdivided further depending on the concentrations of β-myrcene. The PCA biplot also shows three groupings: (1) the L. dissectum group, (2) the L. multifidum high β-myrcene group, and (3) the L. multifidum less β-myrcene group. The two samples from Bairamian and co-workers (L. dissectum var. multifidum and L. dissectum var. dissectum) are separated from the other groups.
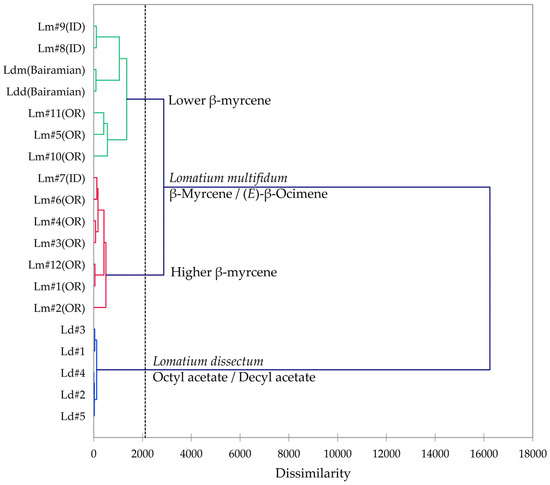
Figure 10.
Dendrogram obtained by hierarchical cluster analysis (HCA) of essential oil compositions (major essential oil components) of members of the Lomatium dissectum complex. Lm (OR) = Lomatium multifidum from eastern Oregon, Lm (ID) = Lomatium multifidum from western Idaho, Ld = Lomatium dissectum (from western Idaho), Ldd (Bairamian) = Lomatium dissectum var. dissectum from reference [22], Ldm (Baiaramian) = Lomatium dissectum var. multifidum from reference [22].
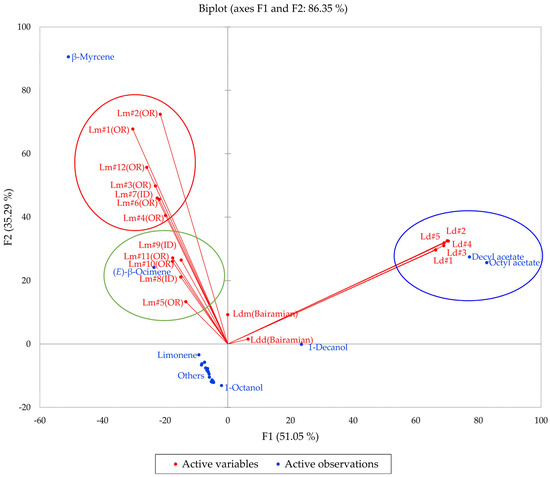
Figure 11.
The bidimensional plot of the first two components (F1 and F2) from principal component analysis (PCA) of members of the Lomatium dissectum complex, based on major components in their essential oils. Lm (OR) = Lomatium multifidum from eastern Oregon, Lm (ID) = Lomatium multifidum from western Idaho, Ld = Lomatium dissectum (from western Idaho), Ldd (Bairamian) = Lomatium dissectum var. dissectum from reference [22], Ldm (Baiaramian) = Lomatium dissectum var. multifidum from reference [22].
2.6. Lomatium nudicaule
Seven samples of L. nudicaule were collected from three sites in western Idaho. The colorless to pale yellow essential oils were obtained in yields of 0.15% to 3.01%. The essential oil compositions are presented in Table 7. A total of 109 compounds were identified in the essential oils, accounting for 90.4% to 98.7% of the total compositions. The major components in the L. nudicaule essential oils were β-phellandrene (16.0–45.7%), (Z)-ligustilide (5.6–47.1%), (E)-β-ocimene (3.3–9.9%), δ-3-carene (0.2–12.6%), myrcene (0.7–6.1%), cryptone (0.3–7.7%), and germacrene B (0.2–9.3%).

Table 7.
Chemical compositions (%) of the essential oils of Lomatium nudicaule (Nutt.) J.M. Coult. & Rose.
2.7. Lomatium papilioniferum (Lomatium grayi Complex)
A total of eight samples of L. papilioniferum were collected from north-central Oregon, along the Columbia River (four samples), and from western Idaho (four samples). The plants gave colorless to yellow essential oils (0.20–3.33% yield). The essential oil compositions showed notable differences between the Oregon samples and the Idaho samples (Table 8). Essential oils from both collection locations were generally rich in p-cymene (3.1–47.8% and 20.4–22.9%) and γ-terpinene (0.1–30.9% and 7.3–15.1%) for the Oregon and Idaho samples, respectively. However, sedanenolide (1.5–10.8%), myrcene (3.1–27.5%), and (E)-β-ocimene (0.7–7.2%) were relatively abundant in the Oregon samples but were either not observed (sedanenolide) or found in only small quantities (myrcene and (E)-β-ocimene) in the Idaho samples. Conversely, 2-methyl-5-(1,2,2-trimethylcyclopentyl)phenol (24.9–31.5%) and cuparene (3.5–6.0%) were abundant in the Idaho samples but not observed in the Oregon samples.

Table 8.
Chemical compositions (%) of Lomatium papilioniferum J.A. Alexander & Whaley from northern Oregon and western Idaho.
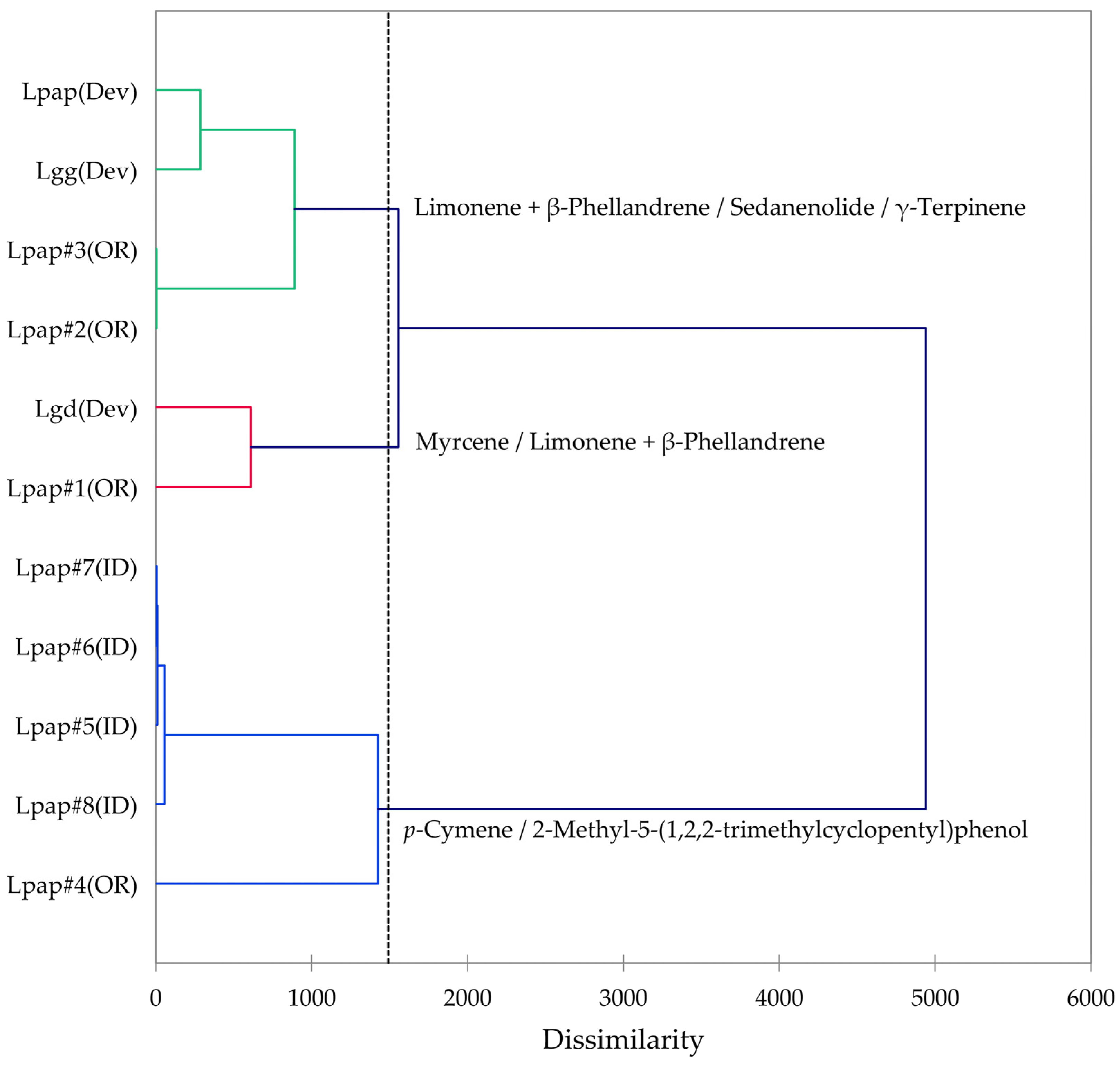
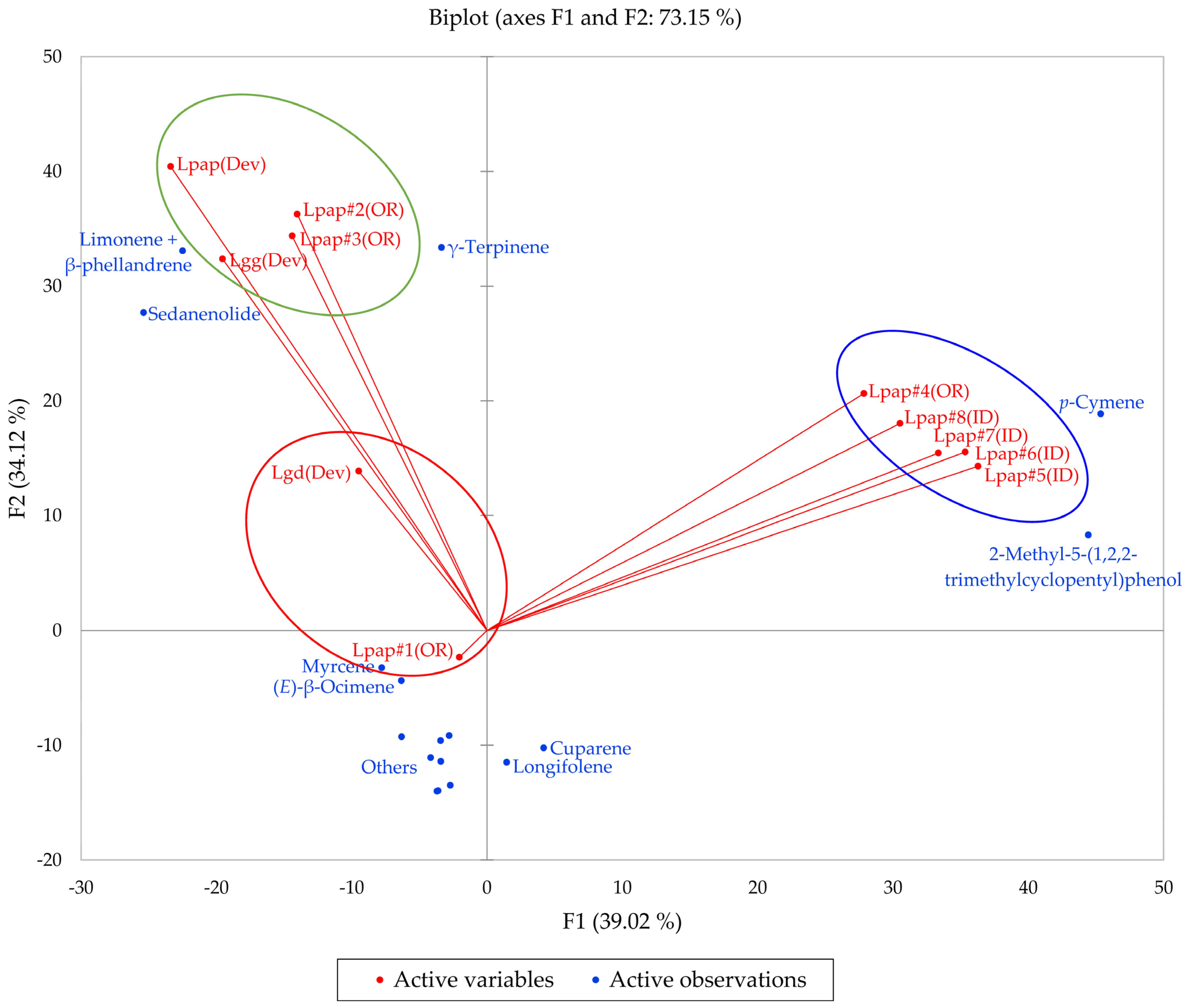
Based on the morphological characteristics as well as the geographical ranges suggested by Alexander et al. [13], the L. grayi samples in this work were identified as L. papilioniferum. Dev and co-workers [24] analyzed three taxa of the L. grayi complex, L. grayi var. grayi, L. grayi var. depauparatum, and L. grayi “new variety”, which is presumably L. papilioniferum based on the location of the collection site (northern Nevada). In order to compare the chemical compositions of the L. grayi complex (in this work and [24]), both HCA and PCA were carried out (Figure 12 and Figure 13).
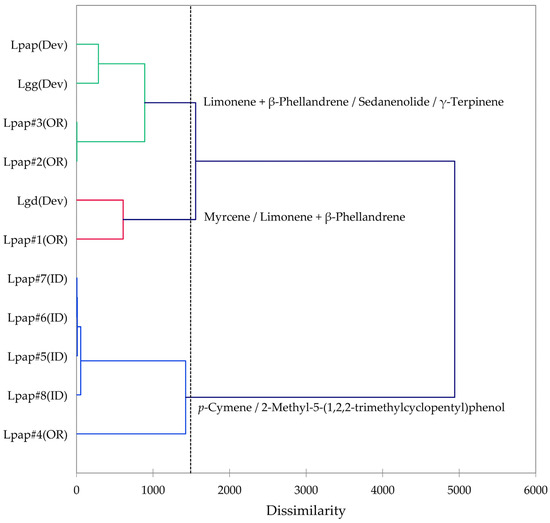
Figure 12.
Dendrogram obtained by hierarchical cluster analysis (HCA) of essential oil compositions (major essential oil components) of members of the Lomatium grayi complex. Lpap (OR) = Lomatium papilioniferum from northern Oregon, Lpap (ID) = Lomatium papilioniferum from western Idaho, Lpap(Dev) = Lomatium “new species” (L. papilioniferum) from reference [24], Lgg(Dev) = Lomatium grayi var. grayi from reference [24], Lgd(Dev) = Lomatium grayi var. depauparatum from reference [24].
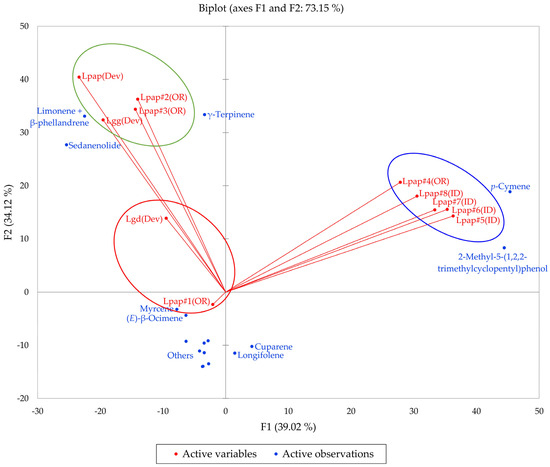
Figure 13.
The bidimensional plot of the first two components (F1 and F2) from principal component analysis (PCA) of members of the Lomatium grayi complex, based on major components in their essential oils. Lpap (OR) = Lomatium papilioniferum from northern Oregon, Lpap (ID) = Lomatium papilioniferum from western Idaho, Lpap(Dev) = Lomatium “new species” (L. papilioniferum) from reference [24], Lgg(Dev) = Lomatium grayi var. grayi from reference [24], Lgd(Dev) = Lomatium grayi var. depauparatum from reference [24].
The HCA shows two major groupings (samples #1, #2, and #3 from Oregon and the three L. grayi samples from Dev et al. [24]). This group can be further divided into two groups, a limonene + β-phellandrene/sedanenolide/γ-terpinene group and a myrcene/limonene + β-phellandrene group. The second major group, with a very different chemical profile, is dominated by p-cymene and 2-methyl-5-(1,2,2-trimethylcyclopentyl)phenol. It is not clear what factors are responsible for the chemical differences observed between the Oregon L. papilioniferum samples; these were collected on the same day (17 April 2023) from the same location (along the Columbia River in north-central Oregon). The four L. papilioniferum samples from Idaho, collected on the same day (21 May 2024) from the same location (western Idaho), showed very similar chemical profiles. The PCA verifies the HCA. There is a group that correlates strongly with limonene + β-phellandrene, γ-terpinene, and sedanenolide (Lpap#2, and #3, Lpap(Dev), and Lgg(Dev)), a group that correlates strongly with p-cymene, and two individual samples (Lgd(Dev and Lpap#1)). The p-cymene group may constitute a discrete chemotype of L. papilioniferum, while the volatile phytochemical profiles displayed by the Oregon samples are complicated and unresolved.
2.8. Analysis of Variance
Analysis of variance (ANOVA) examinations were carried out to identify statistically significant differences in percentages of essential oil components (Table 9). Analyses of the essential oil compositions of L. anomalum, L. packardiae, and L. triternatum var. triternatum allow for discrimination between the members of the L. triternatum complex. Lomatium packardiae essential oils contain a significantly higher concentration of limonene (60.9% ± 10.1%) than the other essential oils, including L. anomalum (1.2% ± 0.5%) or L. triternatum triternatum (2.5% ± 2.0%). (Z)-Ligustilide concentrations were significantly higher in L. packardiae (16.2% ± 3.0%) than either L. anomalum (0.4% ± 0.4%) or L. triternatum triternatum (not observed). On the other hand, L. anomalum essential oils had significantly higher concentrations of both sabinene (48.7% ± 1.0%) and α-pinene (27.7% ± 8.6%) than the other Lomatium essential oils. Lomatium triternatum var. triternatum, on the other hand, cannot be defined chemically with the data available; there was too much variation in the essential oil compositions.

Table 9.
Comparison of Lomatium essential oil components by analysis of variance (ANOVA) followed by Tukey’s post hoc test.
In the Lomatium dissectum complex, it is easy to distinguish L. dissectum from L. multifidum. Lomatium dissectum essential oils were dominated by octyl acetate (42.6% ± 3.4%) and decyl acetate (40.4% ± 4.8%), which were detected in only minute, if at all, quantities in the other Lomatium essential oils. In contrast, L. multifidum had significantly higher myrcene concentrations (30.7% ± 13.2%) in its essential oils.
The volatile phytochemistry of L. papilioniferum seems to depend on geographical location. Collections from both Idaho and Oregon showed relatively high concentrations of p-cymene and γ-terpinene. β-Phellandrene was significantly higher in the Oregon samples (13.4% ± 11.9%) than the Idaho samples (trace amounts only), and sedanenolide concentrations were significantly greater in L. papilioniferum from Oregon (6.18% ± 5.26), which was not observed in any of the Idaho samples. Conversely, the L. papilioniferum essential oils from Idaho were dominated by 2-methyl-5-(1,2,2-trimethylcyclopentyl)phenol (29.0% ± 2.9%), which was virtually absent in the other Lomatium essential oils.
2.9. Enantiomeric Distributions
Enantioselective GC-MS was carried out on the Lomatium essential oil samples to examine the distribution of chiral terpenoid components. The enantiomeric distributions are summarized in Table 10. There is variation in the enantiomeric distributions, both between species and within species. In order to assess the differences between the species and sampling sites, the enantiomeric distributions of (+)-α-pinene, (−)-camphene, (+)-sabinene, (+)-β-pinene, (+)-limonene, and (+)-linalool were analyzed by an ANOVA followed by Tukey’s test using Minitab® 18 (Minitab Inc., State College, PA, USA). Differences at p < 0.05 were considered to be statistically significant. (Table 11).

Table 10.
Enantiomeric distribution of chiral terpenoid components of Lomatium species.

Table 11.
Enantiomer percentages and significance for chiral monoterpenoids in Lomatium species based on ANOVA/Tukey.
The three taxa in the Lomatium triternatum complex (L. anomalum, L. packardiae, and L. triternatum var. triternatum) are distinguished by significantly different α-pinene, sabinene, β-pinene, and limonene enantiomeric distributions. The (+)-α-pinene and (+)-sabinene levels are significantly greater in L. anomalum than in L. packardiae or L. triternatum triternatum.
Furthermore, (+)-β-pinene is significantly lower in L. triternatum triternatum than either L. anomalum or L. packardiae, and (+)-limonene is much greater in L. packardiae than L. anomalum or L. triternatum triternatum. There are significant differences in the limonene enantiomeric distributions between the Oregon L. papilioniferum samples and the Idaho L. papilioniferum samples. Likewise, (+)-limonene is significantly greater in L. dissectum compared with L. multifidum.
3. Materials and Methods
3.1. Plant Collection and Identification
The L. anomalum, L. packardiae, and L. triternatum plant samples were identified by W.N. Setzer using published botanical descriptions [2,3,10] and comparison with herbarium samples from the New York Botanical Garden [31,32,33] and the Intermountain Region Herbarium Network [34]. Lomatium papilioniferum was identified by W.N. Setzer using published botanical descriptions [13] and by comparison with herbarium samples from the New York Botanical Garden [35]. Lomatium dissectum and L. multifidum were identified by W.N. Setzer using published botanical descriptions [14] and verified by comparison with herbarium samples [36,37]. Lomatium nudicaule was identified in the field by W.N. Setzer using a field guide [5] and verified using published botanical descriptions [38,39,40] and herbarium samples from the New York Botanical Garden [41]. Voucher specimens of each species were deposited with the herbarium of the University of Alabama in Huntsville, and voucher numbers are presented in Table 12.

Table 12.
Collection and hydrodistillation details for Lomatium species.
3.2. Hydrodistillation
The fresh plant materials were stored frozen (–20 °C) until distillation. For each sample, the fresh/frozen aerial parts were chopped and hydrodistilled using a Likens-Nickerson apparatus [42,43,44] with continuous extraction of the distillate for four hours. The chopped plant material was placed in a 1000-mL flask with enough distilled water to cover the material. Dichloromethane (25 mL) was used in the receiving flask. Evaporation of the dichloromethane gave the essential oils, summarized in Table 12.
3.3. Gas Chromatographic Analysis
The essential oils of the aerial parts of L. anomalum, L. dissectum, L. multifidum, L. nudicaule, L. packardiae, L. papilioniferum, and L. triternatum var. triternatum were analyzed by gas chromatography–mass spectrometry (GC-MS), gas chromatography coupled with flame ionization detection (GC-FID), and chiral GC-MS as previously described [45]. Instrumental details are provided as supplementary material (Supplementary Table S1). Retention indices (RIs) were determined using a homologous series of n-alkanes using the linear formula of van den Dool and Kratz [46]. The essential oil components were identified by comparing their retention index values (within ten RI units) and their mass spectral fragmentation patterns (>80% similarity) with those reported in the Adams [47], FFNSC3 [48], NIST20 [49], and Satyal [50] databases. The compound percentages were calculated from raw peak areas without standardization. The individual enantiomers were determined using enantioselective GC-MS by comparison of MS fragmentation and RI values with authentic samples (Sigma-Aldrich, Milwaukee, WI, USA), which were compiled in our in-house database. Percentages of each enantiomer were calculated from raw peak integration.
3.4. Statistical Analyses
An agglomerative hierarchical cluster analysis (HCA) and principal component analysis (PCA) were carried out using XLSTAT v. 2018.1.1.62926 (Addinsoft, Paris, France). The HCA and PCA analyses were carried out using the percentages of the most abundant components (Lomatium triternatum complex: limonene, sabinene, β-phellandrene, α-pinene, (Z)-ligustilide, myrcene, β-pinene, cryptone, (E)-β-ocimene, carotol, γ-terpinene, terpinen-4-ol, and spathulenol; Lomatium dissectum complex: β-myrcene, decyl acetate, octyl acetate, (E)-β-ocimene, 1-decanol, limonene, α-bisabolol, β-phellandrene, unidentified (RI 1959), 2-methyloct-(3E)-en-5-yne, longifolene, (Z)-β-ocimene, p-cymene, camphene, bornyl acetate, α-eudesmol, γ-terpinene, terpinolene, 1-octanol, γ-eudesmol, globulol, agarospyryl acetate, and viridiflorene; Lomatium grayi complex: p-cymene, γ-terpinene, limonene + β-phellandrene, 2-methyl-5-(1,2,2-trimethylcyclopentyl)phenol, sedanenolide, myrcene, (E)-β-ocimene, (Z)-β-ocimene, cuparene, 3-butylphthalide, piperitone, longifolene, humulol, terpinolene, α-pinene, (E)-nerolidol, and decyl acetate) from this study in addition to compositions previously reported. Dissimilarity was used to determine clusters considering Euclidean distance, and Ward’s method was used to define agglomeration. The PCA, type correlation, was carried out to verify the chemical associations (clusters) from the HCA analysis. An analysis of variance was conducted by a one-way ANOVA followed by the Tukey test [51] using Minitab® 18 (Minitab Inc., State College, PA, USA). Differences at p < 0.05 were considered to be statistically significant.
4. Conclusions
In this work, the essential oils of seven species of Lomatium (L. anomalum, L. dissectum, L. multifidum, L. nudicaule, L. packardiae, L. papilioniferum, and L. triternatum var. triternatum) from the intermountain western United States were obtained and analyzed by gas chromatographic methods. This work complements previously published essential oil analyses of Lomatium species. In addition, the enantiomeric distributions of chiral terpenoid components in this work serve to further characterize the Lomatium species. The three species in the Lomatium triternatum complex can be distinguished by their essential oil compositions. Lomatium packardiae essential oil can be characterized as a limonene-rich essential oil, and L. anomalum is a species rich in sabinene and α-pinene. The essential oils of L. dissectum and L. multifidum, members of the Lomatium dissectum complex, are readily discriminated based on essential oil composition. Lomatium multifidum essential oils were rich in myrcene while L. dissectum essential oils were dominated by octyl acetate and decyl acetate. Lomatium papilioniferum essential oils from western Idaho are readily characterized by high p-cymene and 2-methyl-5-(1,2,2-trimethylcyclopentyl)phenol concentrations. North-central Oregon L. papilioniferum essential oils were variable but may be tentatively classified as high in β-phellandrene and sedanenolide. There are not enough consistent data to properly characterize the chemotype(s) of L. triternatum var. triternatum. Because of the variation observed in the Oregon L. papilioniferum essential oils, additional collection and analyses are needed to confidently describe the chemotype(s) of that species, as well as the L. grayi complex in general. Additional sampling from other geographical locations would be helpful. The life cycle and timing of the sampling could affect the composition; additional sampling throughout the phenological stages of each species would provide important additional information.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/plants14020186/s1: Table S1: Instrument details for the gas chromatographic analyses of Lomatium species.
Author Contributions
Conceptualization, W.N.S.; methodology, W.N.S. and P.S.; software, P.S.; validation, W.N.S., formal analysis, W.N.S., A.P. and P.S.; investigation, W.N.S., A.P., P.S. and K.S.; resources, W.N.S. and P.S.; data curation, W.N.S.; writing—original draft preparation, W.N.S.; writing—review and editing, W.N.S., A.P., P.S., K.S. and C.C.S.; project administration, W.N.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Data Availability Statement
All data are available in this report. Additional information is available from the corresponding author upon reasonable request.
Acknowledgments
This work was carried out as part of the activities of the Aromatic Plant Research Center (APRC, https://aromaticplant.org/) (accessed on 9 January 2025). We thank Joyce Kelly R. da Silva (Universidade Federal do Pará, Belém, Brazil) for helpful discussions.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- World Flora Online. Lomatium Raf. Available online: https://www.worldfloraonline.org/taxon/wfo-4000022127 (accessed on 26 May 2024).
- Stevens, M.; Mansfield, D.H.; Smith, J.F.; Feist, M.A.E. Resolving the anomaly of Lomatium anomalum: Discovery of a new species in southwestern Idaho (U.S.A.), Lomatium andrusianum (Apiaceae). J. Bot. Res. Inst. Tex. 2018, 12, 1–15. [Google Scholar] [CrossRef]
- Ottenlips, M.V.; Mansfield, D.H.; Buerki, S.; Feist, M.A.E.; Downie, S.R.; Dodsworth, S.; Forest, F.; Plunkett, G.M.; Smith, J.F. Resolving species boundaries in a recent radiation with the Angiosperms353 probe set: The Lomatium packardiae/L. anomalum clade of the L. triternatum (Apiaceae) complex. Am. J. Bot. 2021, 108, 1217–1233. [Google Scholar] [CrossRef] [PubMed]
- Moerman, D.E. Native American Ethnobotany; Timber Press, Inc.: Portland, OR, USA, 1998; ISBN 978-0-88192-453-4. [Google Scholar]
- Turner, M.; Gustafson, P. Wildflowers of the Pacific Northwest; Timber Press, Inc.: Portland, OR, USA, 2006; ISBN 978-0-88192-745-0. [Google Scholar]
- Kartesz, J.T. The Biota of North America Program (BONAP). Available online: https://bonap.net/MapGallery/County/Lomatium%20triternatum.png (accessed on 9 June 2024).
- Tilley, D.; St. John, L.; Ogle, D.; Shaw, N. Plant Guide for Nineleaf Biscuitroot (Lomatium triternatum); United States Department of Agriculture, Natural Resources Conservation Service, Idaho Plant Materials Center: Aberdeen, ID, USA, 2010. [Google Scholar]
- Gucker, C.L.; Shaw, N.L. Nineleaf Biscuitroot: Lomatium triternatum (Pursh) J.M. Coult. & Rose. In Western Forbs: Biology, Ecology, and Use in Restoration; Gucker, C.L., Shaw, N.L., Eds.; Great Basin Fire Science Exchange: Reno, NV, USA, 2020; p. 21. [Google Scholar]
- Lesica, P.; Kittelson, P.M. Morphological and ecological segregation of two sympatric Lomatium triternatum (Apiaceae) varieties in Montana. Madroño 2023, 60, 211–216. [Google Scholar] [CrossRef]
- Smith, J.F.; Mansfield, D.H.; Stevens, M.; Sosa, E.; Feist, M.A.E.; Downie, S.R.; Plunkett, G.M.; Darrach, M. Try Tri again? Resolving species boundaries in the Lomatium triternatum (Apiaceae) complex. J. Syst. Evol. 2018, 56, 218–230. [Google Scholar] [CrossRef]
- Tilley, D.; St. John, L.; Ogle, D.; Shaw, N. Plant Guide for Gray’s Biscuitroot (Lomatium grayi); United States Department of Agriculture, Natural Resources Conservation Service, Idaho Plant Materials Center: Aberdeen, ID, USA, 2011. [Google Scholar]
- Kartesz, J.T. The Biota of North America Program (BONAP). Available online: https://bonap.net/MapGallery/County/Lomatium%20grayi.png (accessed on 2 June 2024).
- Alexander, J.A.; Whaley, W.; Blain, N. The Lomatium grayi complex (Apiaceae) of the western United States: A taxonomic revision based on morphometic, essential oil composition, and larva-host coevolution studies. J. Bot. Res. Inst. Tex. 2018, 12, 387–444. [Google Scholar] [CrossRef]
- Feist, M.A.E.; Smith, J.F.; Mansfield, D.H.; Darrach, M.; McNeill, R.P.; Downie, S.R.; Plunkett, G.M.; Wilson, B.L. New combinations in Lomatium (Apiaceae, subfamily Apioideae). Phytotaxa 2017, 316, 95–98. [Google Scholar] [CrossRef]
- Constance, L.; Wetherwax, M. Lomatium dissectum. Available online: https://ucjeps.berkeley.edu/eflora/eflora_display.php?tid=31405 (accessed on 1 June 2024).
- Intermountain Region Herbarium Network. Lomatium multifidum. Available online: https://www.intermountainbiota.org/portal/collections/list.php (accessed on 29 May 2024).
- World Flora Online. Lomatium multifidum (Nutt.) R.P. McNeill & Darrach. Available online: http://www.worldfloraonline.org/taxon/wfo-0001423732 (accessed on 29 May 2024).
- Kartesz, J.T. The Biota of North America Program (BONAP). Available online: https://bonap.net/MapGallery/County/Lomatium%20nudicaule.png (accessed on 6 June 2024).
- Schlichter, P. Lomatium nudicaule. Available online: http://science.halleyhosting.com/nature/gorge/5petal/pars/lomatium/nudicaule/nudicaule10-18-2015.jpg (accessed on 6 June 2024).
- Beauchamp, P.S.; Chea, E.; Dimaano, J.G.; Dev, V.; Ly, B.; Miranda, A.E.; Whaley, W.H. Essential oil composition of six Lomatium species attractive to Indra swallowtail butterfly (Papilio indra): Principal component analysis against essential oil composition of Lomatium dissectum var. multifidum. J. Essent. Oil Res. 2009, 21, 535–542. [Google Scholar] [CrossRef]
- Asuming, W.A.; Beauchamp, P.S.; Descalzo, J.T.; Dev, B.C.; Dev, V.; Frost, S.; Ma, C.W. Essential oil composition of four Lomatium Raf. species and their chemotaxonomy. Biochem. Syst. Ecol. 2005, 33, 17–26. [Google Scholar] [CrossRef]
- Bairamian, S.; Beauchamp, P.S.; Descalzo, J.T.; Dev, B.C.; Dev, V.; Frost, S.C.; Nguyen, C.V. California Lomatiums part III. Composition of the hydrodistilled oils from two varieties of Lomatium dissectum. Isolation of a new hydrocarbon. J. Essent. Oil Res. 2004, 16, 461–468. [Google Scholar] [CrossRef]
- Beauchamp, P.S.; Dev, B.C.; Dev, V. California Lomatiums, part VI. Composition of the essential oils of Lomatium foeniculaceum ssp. fimbriatu. J. Essent. Oil Res. 2006, 18, 666–669. [Google Scholar] [CrossRef]
- Dev, V.; Ly, B.; Miranda, A.E.; Whaley, W. Lomatium grayi and Indra swallowtail butterfly. Composition of the essential oils of three varieties of Lomatium grayi (J. M. Coult et Rose) J. M. Coult et Rose. J. Essent. Oil Res. 2007, 19, 244–248. [Google Scholar] [CrossRef]
- Beauchamp, P.S.; Dev, V.; Tran, H.D.; Whaley, W.H. California Lomatiums, part VIII. Analysis of essential oils of Lomatium marginatum (Benth.) Coult. & Rose var. purpureum Jepson. isolation of (Z)-β-lomatene, a new sesquiterpene hydrocarbon. J. Essent. Oil Res. 2011, 23, 112–118. [Google Scholar] [CrossRef]
- Beauchamp, P.S.; Dev, V.; Kittisanthanon, K.; Ly, B. California Lomatiums, part X. Comparison of composition of the hydrodistilled oils from two subspecies of Lomatium mohavense. Nat. Prod. Res. 2011, 25, 1347–1351. [Google Scholar] [CrossRef] [PubMed]
- Beauchamp, P.S.; Dev, B.C.; Dev, V.; Midland, S.L.; Sims, J.J. California Lomatiums, part VII. Analysis of the essential oils of Lomatium nevadense (Watson) J. Coulter et Rose var. parishii (J. Coulter et Rose) Jepson. Isolation of trans-dauc-8-en-11-ol, a new sesquiterpene alcohol and naturally occurring 2′,3′,3′-trimethyl-2′,3′-dihydroangelicin. J. Essent. Oil Res. 2007, 19, 117–124. [Google Scholar] [CrossRef]
- Beauchamp, P.S.; Descalzo, J.T.; Dev, B.C.; Dev, V.; Nguyen, C.V.; Midland, S.L.; Sims, J.J.; Tham, F.S. California Lomatiums, part IV: Composition of the essential oils of Lomatium rigidum (M.E. Jones) Jepson. Structures of two new funebrene epimers and a tridecatriene. J. Essent. Oil Res. 2004, 16, 571–578. [Google Scholar] [CrossRef]
- Dev, V.; Whaley, W.H.; Bailey, S.R.; Chea, E.; Dimaano, J.G.; Jogani, D.K.; Ly, B.; Eggett, D. Essential oil composition of nine Apiaceae species from western United States that attract the female Indra Swallowtail butterfly (Papilio indra). Biochem. Syst. Ecol. 2010, 38, 538–547. [Google Scholar] [CrossRef]
- Bedrossian, A.; Beauchamp, P.E.; Dev, V.; Kwan, S.; Munevar-Mendoza, E.; Okoreeh, E.K.; Moore, P.E. Composition of the essential oil of Lomatium torreyi. J. Essent. Oil Res. 1998, 10, 473–477. [Google Scholar] [CrossRef]
- New York Botanical Garden. Starr Virtual Herbarium. Available online: https://sweetgum.nybg.org/science/vh/specimen-list/?SummaryData=Lomatium+anomalum (accessed on 9 June 2024).
- New York Botanical Garden. Starr Virtual Herbarium. Available online: https://sweetgum.nybg.org/science/vh/specimen-list/?SummaryData=Lomatium+packardiae (accessed on 9 June 2024).
- New York Botanical Garden. Starr Virtual Herbarium. Available online: https://sweetgum.nybg.org/science/vh/specimen-list/?SummaryData=Lomatium+triternatum (accessed on 20 May 2024).
- Intermountain Region Herbarium Network. Lomatium triternatum. Available online: https://www.intermountainbiota.org/portal/collections/list.php (accessed on 20 May 2024).
- New York Botanical Garden. Starr Virtual Herbarium. Available online: https://sweetgum.nybg.org/science/vh/specimen-list/?SummaryData=Lomatium+papilioniferum (accessed on 2 June 2024).
- New York Botanical Garden. Starr Virtual Herbarium. Available online: https://sweetgum.nybg.org/science/vh/specimen-list/?SummaryData=Lomatium+dissectum (accessed on 1 June 2024).
- New York Botanical Garden. Starr Virtual Herbarium. Available online: https://sweetgum.nybg.org/science/vh/specimen-list/?SummaryData=Lomatium+multifidum (accessed on 1 June 2024).
- Mathias, M.E. A revision of the genus Lomatium. Ann. Mo. Bot. Gard. 1938, 25, 225–297. [Google Scholar] [CrossRef]
- Gucker, C.L.; Shaw, N.L. Barestem biscuitroot: Lomatium nudicaule (Pursh) J.M. Coult. & Rose. In Western Forbs: Biology, Ecology, and Use in Restoration; Gucker, C.L., Shaw, N.L., Eds.; Great Basin Fire Science Exchange: Reno, NV, USA, 2021; p. 17. [Google Scholar]
- Tilley, D.; St. John, L. Plant Guide for Barestem Biscuitroot (Lomatium nudicaule); United States Department of Agriculture, Natural Resources Conservation Service, Idaho Plant Materials Center: Aberdeen, ID, USA, 2012. [Google Scholar]
- New York Botanical Garden. Starr Virtual Herbarium. Available online: https://sweetgum.nybg.org/science/vh/specimen-list/?SummaryData=Lomatium+nudicaule (accessed on 8 June 2024).
- Likens, S.T.; Nickerson, G.B. Detection of certain hop oil constituents in brewing products. Proc. Annu. Meet. Am. Soc. Brew. Chem. 1964, 22, 5–13. [Google Scholar] [CrossRef]
- Au-Yeung, C.Y.; MacLeod, A.J. A comparison of the efficiency of the Likens and Nickerson extractor for aqueous, lipid/aqueous, and lipid samples. J. Agric. Food Chem. 1981, 29, 502–505. [Google Scholar] [CrossRef]
- Bouseta, A.; Collin, S. Optimized Likens-Nickerson methodology for quantifying honey flavors. J. Agric. Food Chem. 1995, 43, 1890–1897. [Google Scholar] [CrossRef]
- Poudel, A.; Dosoky, N.S.; Satyal, P.; Swor, K.; Setzer, W.N. Essential oil composition of Grindelia squarrosa from southern Idaho. Molecules 2023, 28, 3854. [Google Scholar] [CrossRef] [PubMed]
- van den Dool, H.; Kratz, P.D. A generalization of the retention index system including linear temperature programmed gas-liquid partition chromatography. J. Chromatogr. A 1963, 11, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry, 4th ed.; Allured Publishing: Carol Stream, IL, USA, 2007; ISBN 978-1-932633-21-4. [Google Scholar]
- Mondello, L. FFNSC 3; Shimadzu Scientific Instruments: Columbia, MD, USA, 2016. [Google Scholar]
- NIST20; National Institute of Standards and Technology: Gaithersburg, MD, USA, 2020.
- Satyal, P. Development of GC-MS Database of Essential Oil Components by the Analysis of Natural Essential Oils and Synthetic Compounds and Discovery of Biologically Active Novel Chemotypes in Essential Oils. Ph.D. Dissertation, University of Alabama in Huntsville, Huntsville, AL, USA, 2015. [Google Scholar]
- Zar, J.H. Biostatistical Analysis, 3rd ed.; Prentice Hall: Upper Saddle River, NJ, USA, 1974; ISBN 0-13-084542-6. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).