Impact of Plant Growth Promoting Rhizobacteria in the Orchestration of Lycopersicon esculentum Mill. Resistance to Plant Parasitic Nematodes: A Metabolomic Approach to Evaluate Defense Responses Under Field Conditions
Abstract
1. Introduction
2. Materials and Methods
2.1. Biocontrol Agents
2.2. Root-knot Nematode (Meloidogyne incognita) Culture
2.3. Raising of Plants
2.4. Growth Attributes
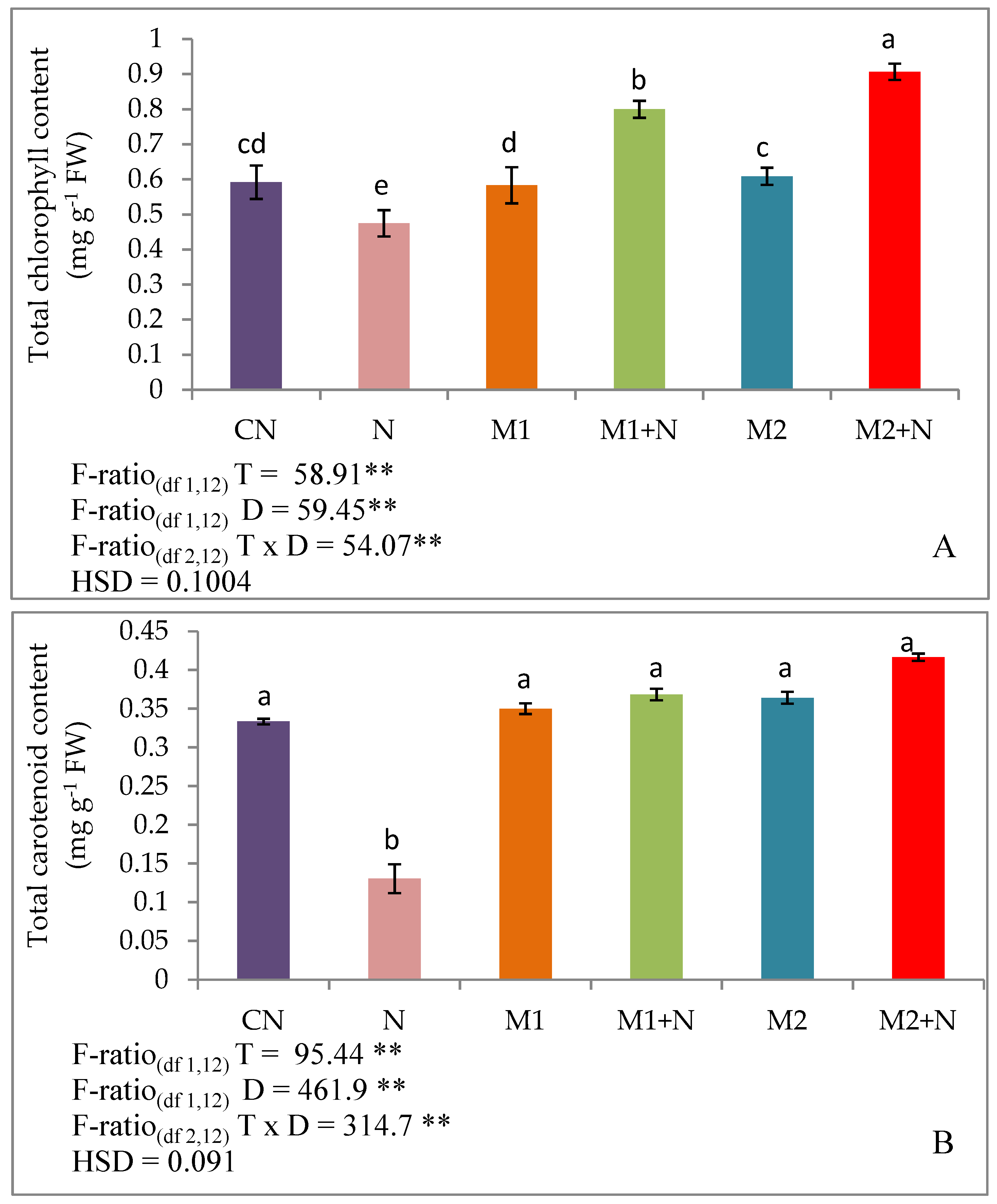
2.5. Photosynthetic Pigments
2.5.1. Total Chlorophyll and Carotenoid Content
2.5.2. Total Xanthophyll Content
2.5.3. Gaseous Exchange Parameters
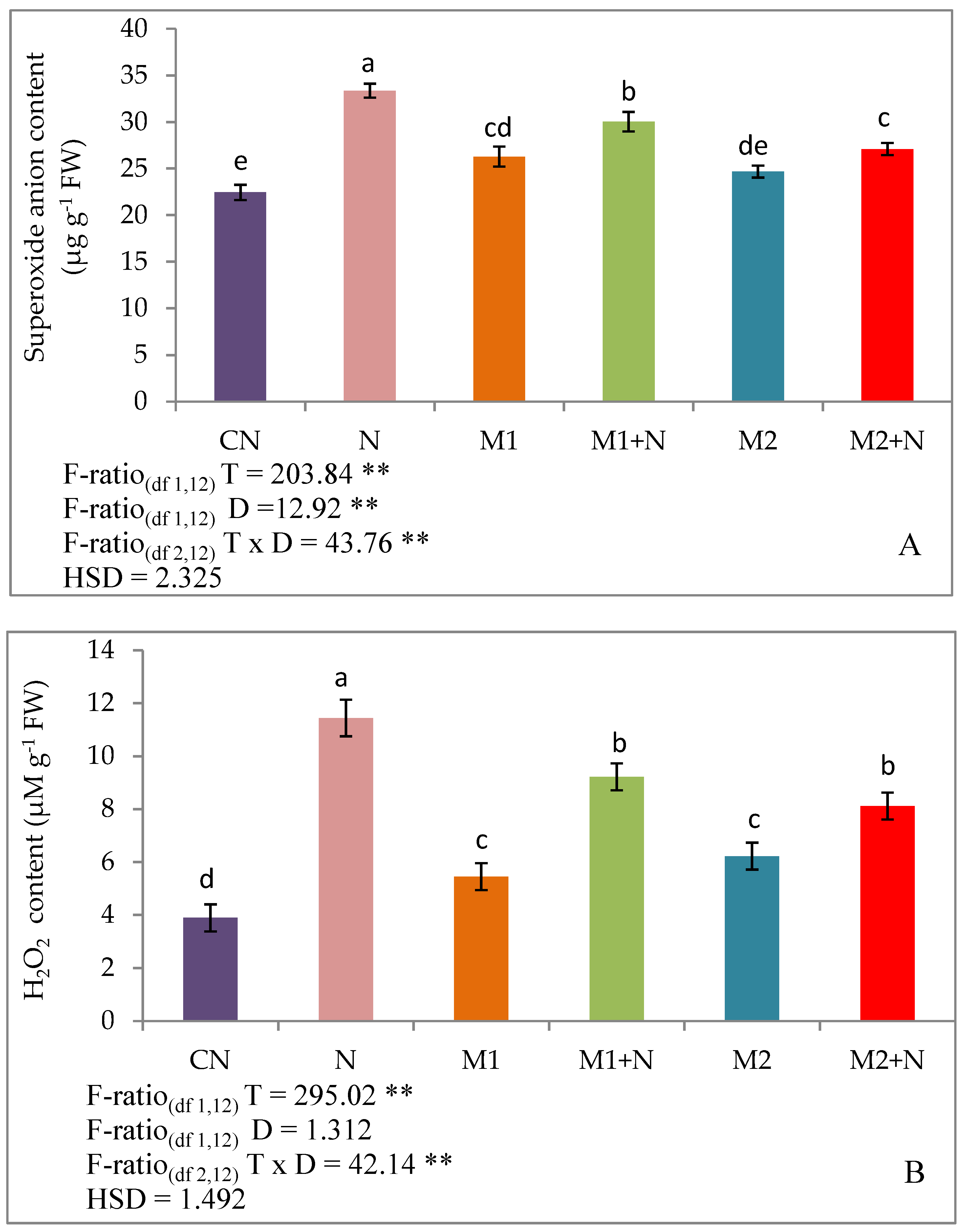
2.6. Oxidative Burst
2.6.1. Superoxide Anion Content
2.6.2. Hydrogen Peroxide Content
2.6.3. Malondialdehyde Content
2.7. Antioxidative Enzymes
2.7.1. Superoxide dismutase Activity (EC.1.15.1.1)
2.7.2. Guaiacol peroxidase Activity (EC. 1.11.1.7)
2.7.3. Catalase Activity (EC.1.11.1.6)
2.7.4. Glutathione Peroxidase Activity (EC. 1.11.1.9)
2.7.5. Ascorbate Peroxidase Activity (EC.1.11.1.11)
2.7.6. Dehydroascorbate Reductase Activity (EC. 1.8.5.1)
2.7.7. Glutathione-S-Transferase Activity (EC.2.5.1.18)
2.7.8. Glutathione Reductase Activity (EC.1.6.4.2)
2.7.9. Polyphenol oxidase Activity (EC. 1.14.18.1)
2.8. Non-Enzymatic Antioxidants
2.8.1. Glutathione Content
2.8.2. Ascorbic Acid Content
2.8.3. Tocopherol Content
2.9. Phenolic Compounds
2.9.1. Total Phenols
2.9.2. Total Flavonoids
2.9.3. Total Anthocyanins
2.10. Osmoprotectants
2.10.1. Total Osmolytes
2.10.2. Total Carbohydrates
2.10.3. Total Reducing Sugars
2.10.4. Trehalose Content
2.10.5. Glycine Betaine Content
2.10.6. Proline Content
2.10.7. Free Amino Acid Content
2.11. Organic Acid Profiling
2.12. Statistical Analysis
3. Results
3.1. Effect of Biocontrol Agents on Growth Parameters of PPN Infected L. esculentum Plants
3.2. Effect of Biocontrol Agents on Photosynthetic Pigments of PPNPPN Infected L. esculentumPlants
3.3. Effect of Biocontrol Agents on Gas Exchange Parameters of PPN Infected L. esculentum Plants
3.4. Effect of Biocontrol Agents on Oxidative Damage in PPN Infected L. esculentum Plants
3.5. Effect of Biocontrol Agents on Antioxidative Enzymes Activities of PPN Infected L. esculentum Plants
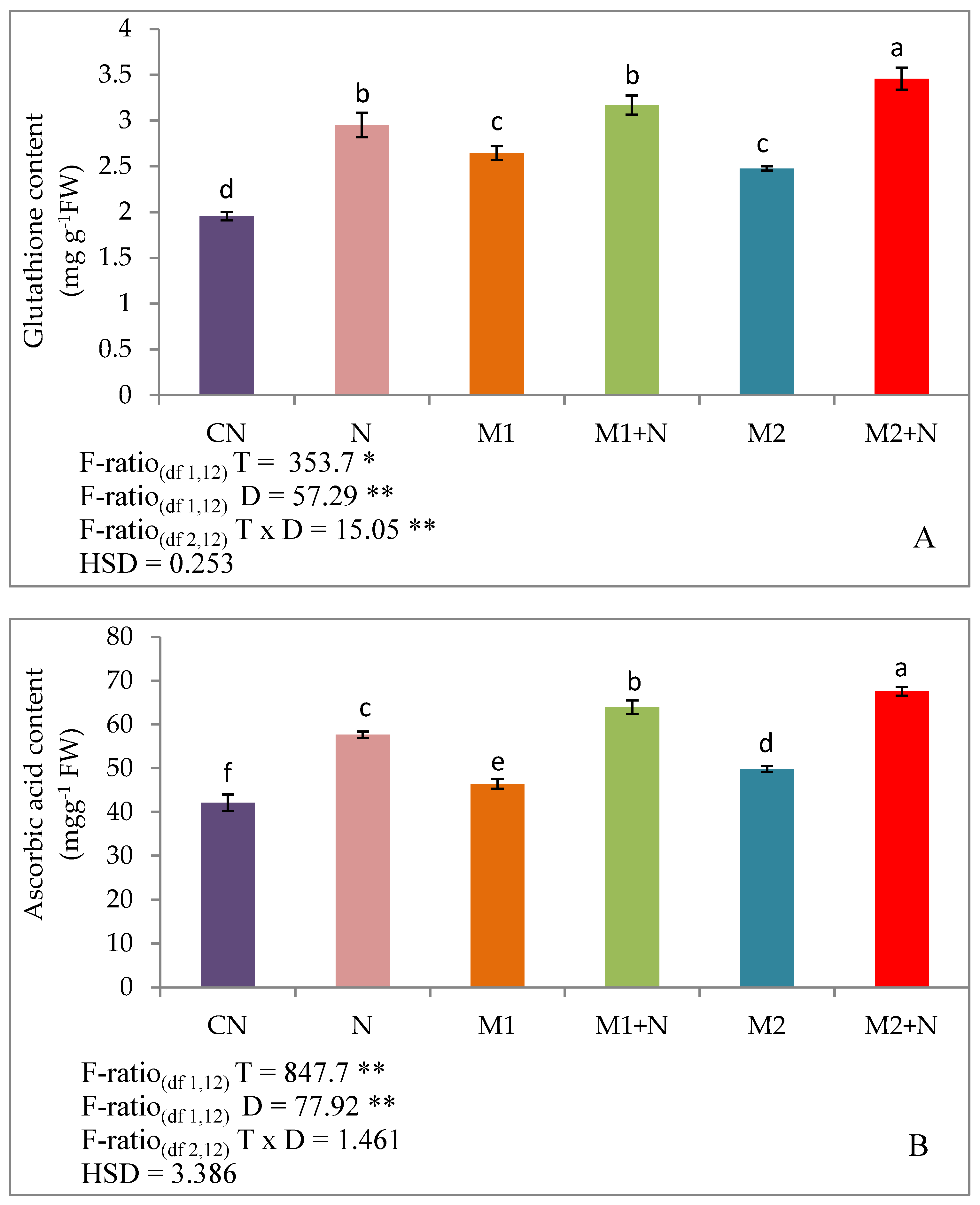
3.6. Effect of Biocontrol Agents on Non-Enzymatic Antioxidants of PPNInfected L. esculentum Plants
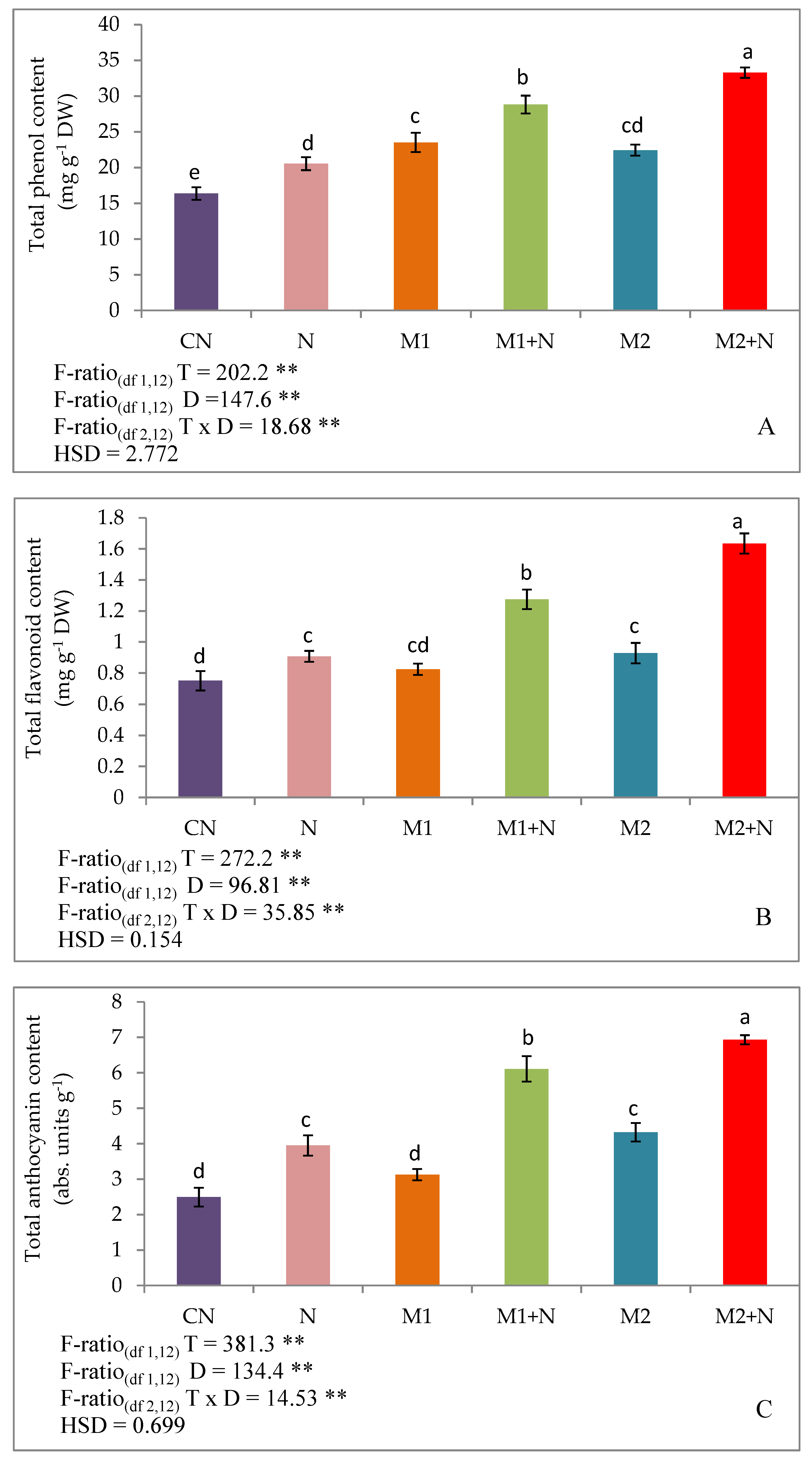
3.7. Phenolic Compounds
Effect of Biocontrol Agents on Total Phenols, Flavonoids and Anthocyanins in PPN Infected L. esculentum Plants
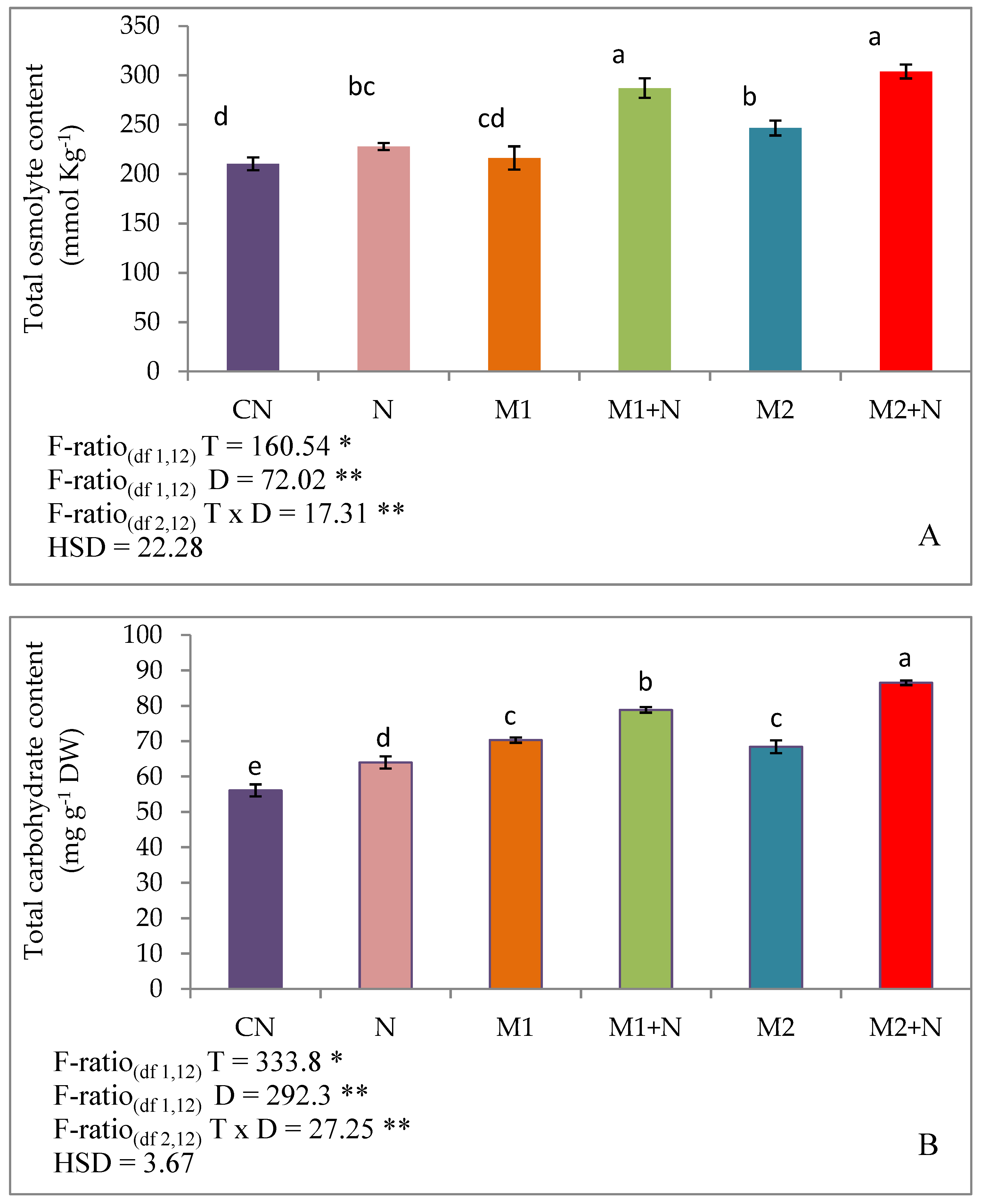
3.8. Osmoprotectants
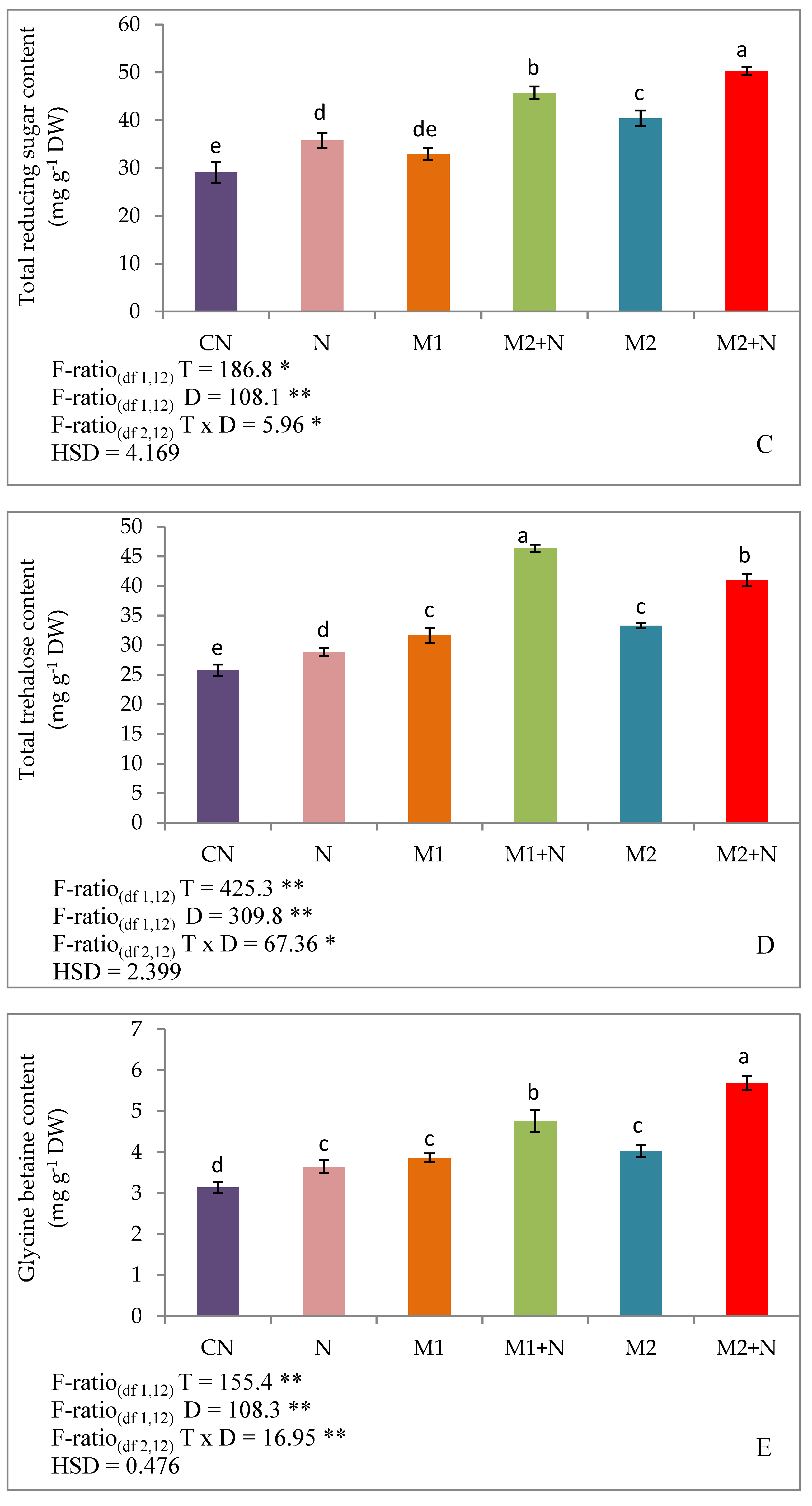
3.8.1. Effect of Biocontrol Agents on Total Osmolytes, Carbohydrates, Reducing Sugars in PPN Infected L. esculentum Plants
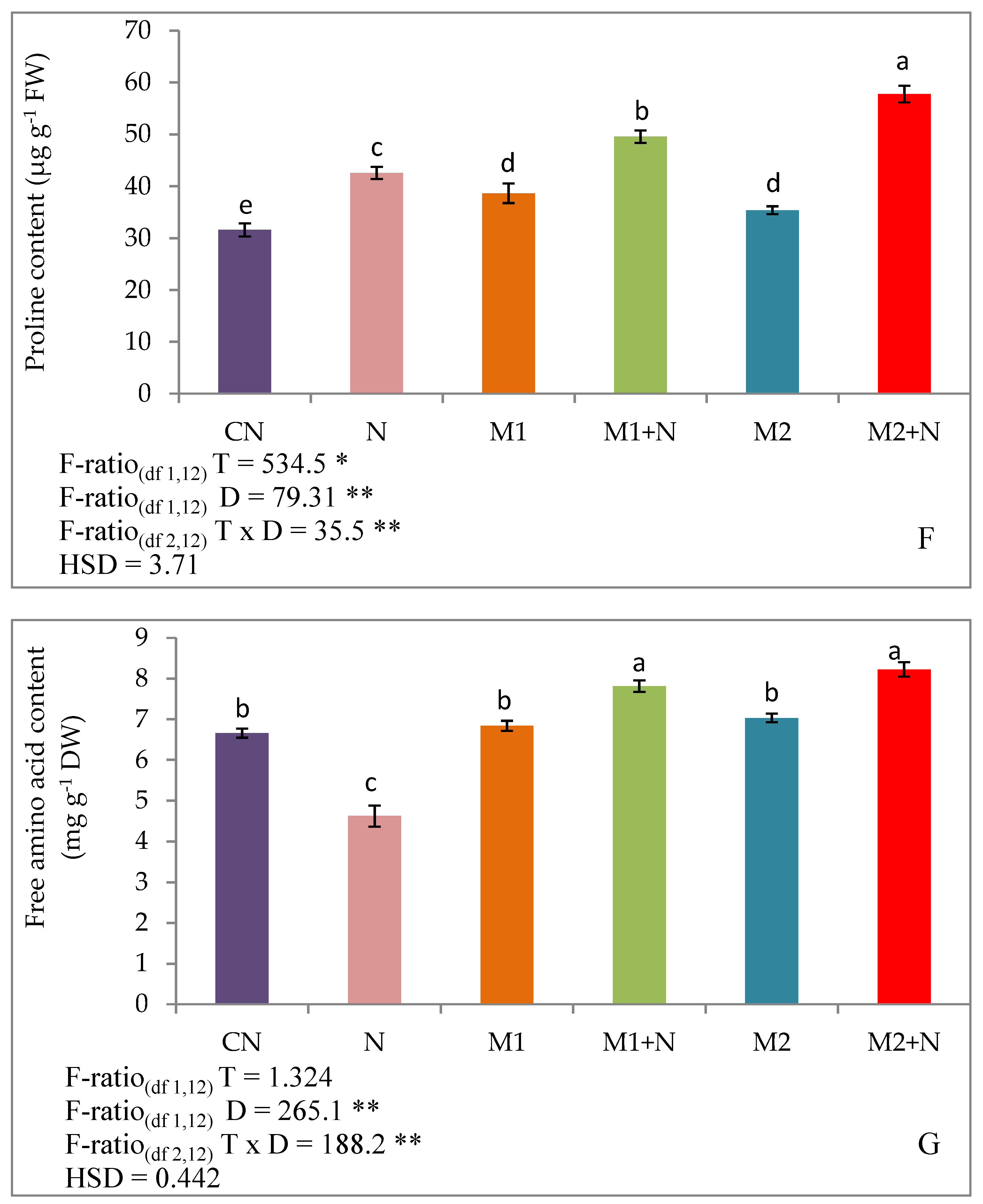
3.8.2. Effect of Biocontrol Agents on Trehalose, Glycine Betaine, Proline and Free Amino Acid in PPN Infected L. esculentum Plants
3.8.3. Effect of biocontrol Agents on Organic Acid Profiling in PPN Infected L. esculentum Plants
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sudhakar, P.; Purushotham, K. Evaluation of F1 hybrids of tomato (Solanum lycopersicum L.). J. Res. ANGRAU 2009, 37, 77–81. [Google Scholar]
- Meena, O.P.; Bahadur, V.; Jagtap, A.; Saini, P. Genetic analysis of agronomic and biochemical variables among different tomato (Solanum lycopersicum L.) accessions. J. Appl. Nat. Sci. 2015, 7, 806–816. [Google Scholar] [CrossRef]
- Afifah, E.N.; Murti, R.H.; Nuringtyas, T.R. Metabolomics Approach for The Analysis of Resistance of Four Tomato Genotypes (Solanum lycopersicum L.) to Root-Knot Nematodes (Meloidogyne incognita). Open Life Sci. 2019, 14, 141–149. [Google Scholar] [CrossRef]
- Coyne, D.L.; Cortada, L.; Dalzell, J.J.; Claudius-Cole, A.O.; Haukeland, S.; Luambano, N.; Talwana, H. Plant-Parasitic Nematodes and Food Security in Sub-Saharan Africa. Annu. Rev. Phytopathol. 2018, 56, 381–403. [Google Scholar] [CrossRef]
- Somasekhar, N.; Prasad, J.S. Plant—Nematode Interactions: Consequences of Climate Change. In Crop Stress and Its Management: Perspectives and Strategies; Springer: Cham, The Netherlands, 2011; pp. 547–564. [Google Scholar] [CrossRef]
- Sikora, R.A.; Coyne, D.; Hallmann, J.; Timper, P. Plant Parasitic Nematodes in Subtropical and Tropical Agriculture; Cabi: Beijing, China, 2018. [Google Scholar]
- Ibrahim, H.M.M.; Ahmad, E.M.; Martínez-Medina, A.; Aly, M.A.M. Effective approaches to study the plant-root knot nematode interaction. Plant Physiol. Biochem. 2019, 141, 332–342. [Google Scholar] [CrossRef] [PubMed]
- Davis, E. Getting to the roots of parasitism by nematodes. Trends Parasitol. 2004, 20, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Phulera, S. Plant Parasitic Nematodes: The Hidden Enemies of Farmers; Excel India Publishers: New Delhi, India, 2015; Volume 68. [Google Scholar]
- Quentin, M.; Abad, P.; Favery, B. Plant parasitic nematode effectors target host defense and nuclear functions to establish feeding cells. Front. Plant Sci. 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Eves-vandenAkker, S.; Birch, P.R.J. Opening the Effector Protein Toolbox for Plant–Parasitic Cyst Nematode Interactions. Mol. Plant 2016, 9, 1451–1453. [Google Scholar] [CrossRef]
- Jacquet, M.; Bongiovanni, M.; Martinez, M.; Verschave, P.; Wajnberg, E.; Castagnone-Sereno, P. Variation in resistance to the root-knot nematode Meloidogyne incognita in tomato genotypes bearing the Mi gene. Plant Pathol. 2005, 54, 93–99. [Google Scholar] [CrossRef]
- Golinowski, W.; Grundler, F.M.W.; Sobczak, M. Changes in the structure of Arabidopsis thaliana during female development of the plant-parasitic nematode Heterodera schachtii. Protoplasma 1996, 194, 103–116. [Google Scholar] [CrossRef]
- Kyndt, T.; Vieira, P.; Gheysen, G.; de Almeida-Engler, J. Nematode feeding sites: Unique organs in plant roots. Planta 2013, 238, 807–818. [Google Scholar] [CrossRef] [PubMed]
- Haydock, P.P.J.; Woods, S.R.; Grove, I.G.; Hare, M.C. Chemical control of nematodes. In Plant Nematology; CABI: Wallingford, UK, 2013; pp. 459–479. [Google Scholar] [CrossRef]
- Compant, S.; Clément, C.; Sessitsch, A. Plant growth-promoting bacteria in the rhizo- and endosphere of plants: Their role, colonization, mechanisms involved and prospects for utilization. Soil Biol. Biochem. 2010, 42, 669–678. [Google Scholar] [CrossRef]
- Collange, B.; Navarrete, M.; Peyre, G.; Mateille, T.; Tchamitchian, M. Root-knot nematode (Meloidogyne) management in vegetable crop production: The challenge of an agronomic system analysis. Crop Prot. 2011, 30, 1251–1262. [Google Scholar] [CrossRef]
- Paul, D.; Lade, H. Plant-growth-promoting rhizobacteria to improve crop growth in saline soils: A review. Agron. Sustain. Dev. 2014, 34, 737–752. [Google Scholar] [CrossRef]
- Viljoen, J.J.F.; Labuschagne, N.; Fourie, H.; Sikora, R.A. Biological control of the root-knot nematode Meloidogyne incognita on tomatoes and carrots by plant growth-promoting rhizobacteria. Trop. Plant Pathol. 2019, 44, 284–291. [Google Scholar] [CrossRef]
- Borah, B.; Ahmed, R.; Hussain, M.; Phukon, P.; Wann, S.B.; Sarmah, D.K.; Bhau, B.S. Suppression of root-knot disease in Pogostemoncablin caused by Meloidogyne incognita in a rhizobacteria mediated activation of phenylpropanoid pathway. Biol. Control 2018, 119, 43–50. [Google Scholar] [CrossRef]
- Glick, B.R. Bacteria with ACC deaminase can promote plant growth and help to feed the world. Microbiol. Res. 2014, 169, 30–39. [Google Scholar] [CrossRef]
- Cetintas, R.; Kusek, M.; Fateh, S.A. Effect of some plant growth-promoting rhizobacteria strains on root-knot nematode, Meloidogyne incognita, on tomatoes. Egypt. J. Biol. Pest Control 2018, 28, 7. [Google Scholar] [CrossRef]
- Li, X.; Hu, H.-J.; Li, J.-Y.; Wang, C.; Chen, S.-L.; Yan, S.-Z. Effects of the Endophytic Bacteria Bacillus cereus BCM2 on Tomato Root Exudates and Meloidogyne incognita Infection. Plant Dis. 2019, 103, 1551–1558. [Google Scholar] [CrossRef]
- Hartmann, A.; Rothballer, M.; Hense, B.A.; SchrÃder, P. Bacterial quorum sensing compounds are important modulators of microbe-plant interactions. Front. Plant Sci. 2014, 5. [Google Scholar] [CrossRef]
- Reinhold-Hurek, B.; Hurek, T. Living inside plants: Bacterial endophytes. Curr. Opin. Plant Biol. 2011, 14, 435–443. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Rahman, A.F.; Shaheen, H.A.; Abd El-Aziz, R.M.; Ibrahim, D.S.S. Influence of hydrogen cyanide-producing rhizobacteria in controlling the crown gall and root-knot nematode, Meloidogyne incognita. Egyptian J. Biol. Pest Control 2019, 29. [Google Scholar] [CrossRef]
- Marin-Bruzos, M.; Grayston, S.J. Biological Control of Nematodes by Plant Growth Promoting Rhizobacteria: Secondary Metabolites Involved and Potential Applications. In Secondary Metabolites of Plant Growth Promoting Rhizomicroorganisms; Springer: Singapore, 2019; pp. 253–264. [Google Scholar] [CrossRef]
- Korejo, F.; Ali, S.A.; Humayun, F.; Rahman, A.; Sultana, V.; Ara, J.; Ehteshamul-Haque, S. Management of root rotting fungi and root knot nematode with endophytic fluorescent Pseudomonas associated with Salvadora species. Pak. J. Bot. 2019, 51. [Google Scholar] [CrossRef]
- Terefe, M.; Tefera, T.; Sakhuja, P.K. Effect of a formulation of Bacillus firmus on root-knot nematode Meloidogyne incognita infestation and the growth of tomato plants in the greenhouse and nursery. J. Invertebr. Pathol. 2009, 100, 94–99. [Google Scholar] [CrossRef] [PubMed]
- Mehmood, U.; Inam-ul-Haq, M.; Saeed, M.; Altaf, A.; Azam, F. A Brief Review on Plant Growth Promoting Rhizobacteria (PGPR): A Key Role in Plant Growth Promotion. Plant Prot. 2018, 2, 77–82. [Google Scholar]
- Alquéres, S.; Meneses, C.; Rouws, L.; Rothballer, M.; Baldani, I.; Schmid, M.; Hartmann, A. The Bacterial Superoxide Dismutase and Glutathione Reductase Are Crucial for Endophytic Colonization of Rice Roots by Gluconacetobacter diazotrophicus PAL5. Mol. PlantMicrobe Interact. 2013, 26, 937–945. [Google Scholar] [CrossRef]
- Khanna, K.; Jamwal, V.L.; Kohli, S.K.; Gandhi, S.G.; Ohri, P.; Bhardwaj, R.; Wijaya, L.; Alyemeni, M.N.; Ahmad, P. Role of plant growth promoting Bacteria (PGPRs) as biocontrol agents of Meloidogyne incognita through improved plant defense of Lycopersicon esculentum. Plant Soil 2019, 436, 325–345. [Google Scholar] [CrossRef]
- Taylor, D.P.; Netscher, C. An improved technique for preparing perineal patterns of Meloidogyne spp. Nematologica 1974, 20, 268–269. [Google Scholar]
- Arnon, D.I. Copper enzymes in isolated chloroplasts, polyphenoxidase in Beta vulgaris. Plant Physiol. 1949, 24, 1–15. [Google Scholar] [CrossRef]
- Maclachlan, S.; Zalik, S. Plastid structure, chlorophyll concentration and free amino acid composition of a chlorophyll mutant of barley. Can. J. Bot. 1963, 41, 1053–1062. [Google Scholar] [CrossRef]
- Lawrence, J. Determination of total xanthophyll and marigold oleoresin. J. Assoc. Off. Anal. Chem. 1990, 2, 970–975. [Google Scholar]
- Wu, G.L.; Cui, J.; Tao, L.; Yang, H. Fluroxypyr triggers oxidative damage by producing superoxide and hydrogen peroxide in rice (Oryza sativa). Ecotoxicology 2009, 19, 124–132. [Google Scholar] [CrossRef] [PubMed]
- Velikova, V.; Yordanov, I.; Edreva, A. Oxidative stress and some antioxidant systems in acid rain-treated bean plants. Plant Sci. 2000, 151, 59–66. [Google Scholar] [CrossRef]
- Heath, R.L.; Packer, L. Photoperoxidation in isolated chloroplasts. Arch. Biochem. Biophys. 1968, 125, 189–198. [Google Scholar] [CrossRef]
- Kono, Y. Generation of superoxide radical during autoxidation of hydroxylamine and an assay for superoxide dismutase. Arch. Biochem. Biophys. 1978, 186, 189–195. [Google Scholar] [CrossRef]
- Pütter, J. Peroxidases. In Methods of Enzymatic Analysis; Elsevier: Amsterdam, The Netherlands, 1974; pp. 685–690. [Google Scholar] [CrossRef]
- Aebi, H. Catalase in vitro. In Methods Enzymology; Colowick, S., Kaplan, N., Eds.; Elsevier: Amsterdam, The Netherlands, 1984; Volume 105, pp. 121–126. [Google Scholar]
- Flohé, L.; Günzler, W.A. Assays of glutathione peroxidase. In Methods Enzymology; Elsevier: Amsterdam, The Netherlands, 1984; pp. 114–120. [Google Scholar] [CrossRef]
- Nakano, Y.; Asada, K. Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol. 1981, 22, 867–880. [Google Scholar]
- Dalton, D.A.; Russell, S.A.; Hanus, F.J.; Pascoe, G.A.; Evans, H.J. Enzymatic reactions of ascorbate and glutathione that prevent peroxide damage in soybean root nodules. Proc. Natl. Acad. Sci. USA 1986, 83, 3811–3815. [Google Scholar] [CrossRef]
- Habig, W.H.; Jakoby, W.B. Assays for differentiation of glutathione S-Transferases. In Methods Enzymology; Elsevier: Amsterdam, The Netherlands, 1981; pp. 398–405. [Google Scholar] [CrossRef]
- Carlberg, I.; Mannervik, B. Purification and characterization of the flavoenzyme glutathione reductase from rat liver. J. Biol. Chem. 1975, 250, 5475–5480. [Google Scholar]
- Kumar, K.; Khan, P. Peroxidase and polyphenol oxidase in excised ragi (Eleusine corocana cv PR 202) leaves during senescence. Indian J. Exp. Biol. 1982, 20, 412–416. [Google Scholar]
- Noctor, G.; Foyer, C.H. Ascorbate and glutathione: Keeping active oxygen under control. Annu. Rev. Plant Biol. 1998, 49, 249–279. [Google Scholar] [CrossRef]
- Roe, J.H.; Kuether, C.A. The determination of ascorbic acid in whole blood and urine through the 2, 4-dinitrophenylhydrazine derivavative of dehydroascorbic acid. J. Biol. Chem. 1943, 147, 399–407. [Google Scholar]
- Martinek, R.G. Method for the determination of vitamin E (total tocopherols) in serum. Clin. Chem. 1964, 10, 1078–1086. [Google Scholar] [PubMed]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Zhishen, J.; Mengcheng, T.; Jianming, W. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 1999, 64, 555–559. [Google Scholar] [CrossRef]
- Mancinelli, A.L. Photoregulation of Anthocyanin Synthesis: VIII. Effect of Light Pretreatments. Plant Physiol. 1984, 75, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Hedge, J.; Hofreiter, B.; Whistler, R. Carbohydrate Chemistry; Academic Press: New York, NY, USA, 1962; Volume 17. [Google Scholar]
- Miller, G.L. Use of Dinitrosalicylic Acid Reagent for Determination of Reducing Sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Trevelyan, W.E.; Harrison, J.S. Studies on yeast metabolism. 5. The trehalose content of baker’s yeast during anaerobic fermentation. Biochem. J. 1956, 62, 177–183. [Google Scholar] [CrossRef]
- Grieve, C.M.; Grattan, S.R. Rapid assay for determination of water soluble quaternary ammonium compounds. Plant Soil 1983, 70, 303–307. [Google Scholar] [CrossRef]
- Bates, L.S.; Waldren, R.P.; Teare, I.D. Rapid determination of free proline for water-stress studies. Plant Soil 1973, 39, 205–207. [Google Scholar] [CrossRef]
- Lee, Y.P.; Takahashi, T. An improved colorimetric determination of amino acids with the use of ninhydrin. Anal. Biochem. 1966, 14, 71–77. [Google Scholar] [CrossRef]
- Chen, M.-C.; Wang, M.-K.; Chiu, C.-Y.; Huang, P.-M.; King, H.-B. Determination of low molecular weight dicarboxylic acids and organic functional groups in rhizosphere and bulk soils of Tsuga and Yushania in a temperate rain forest. Plant Soil 2001, 231, 37–44. [Google Scholar] [CrossRef]
- Gupta, R.; Singh, A.; Srivastava, M.; Shanker, K.; Pandey, R. Plant-microbe interactions endorse growth by uplifting microbial community structure of Bacopa monnieri rhizosphere under nematode stress. Microbiol. Res. 2019, 218, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Čepulytė, R.; Danquah, W.B.; Bruening, G.; Williamson, V.M. Potent Attractant for Root-Knot Nematodes in Exudates from Seedling Root Tips of Two Host Species. Sci. Rep. 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Abd-El-Khair, H.; El-Nagdi, W.M.A.; Youssef, M.M.A.; Abd-Elgawad, M.M.M.; Dawood, M.G. Protective effect of Bacillus subtilis, B. pumilus, and Pseudomonas fluorescens isolates against root knot nematode Meloidogyne incognita on cowpea. Bull. Natl. Res. Centre 2019, 43. [Google Scholar] [CrossRef]
- Ali, S.S.; Vidhale, N. Bacterial siderophore and their application: A review. Int. J. Curr. Microbiol. Appl. Sci. 2013, 2, 303–312. [Google Scholar]
- Tong-Jian, X.; Fang, C.; Chao, G.; Qing-Yun, Z.; Qi-Rong, S.; Wei, R. Bacillus cereus X5 enhanced bio-organic fertilizers effectively control root-knot nematodes (Meloidogyne sp.). Pedosphere 2013, 23, 160–168. [Google Scholar]
- Zakaria, H.M.; Kassab, A.S.; Shamseldean, M.M.; Oraby, M.M.; El-Mourshedy, M.M.F. Controlling the root-knot nematode, Meloidogyne incognita in cucumber plants using some soil bioagents and some amendments under simulated field conditions. Ann. Agric. Sci. 2013, 58, 77–82. [Google Scholar] [CrossRef][Green Version]
- Banora, M.Y.; Almaghrabi, O.A.A. Differential response of some nematode-resistant and susceptible tomato genotypes to Meloidogyne javanica infection. J. Plant Prot. Res. 2019, 59, 113–123. [Google Scholar]
- Strajnar, P.; Širca, S.; Urek, G.; Šircelj, H.; Železnik, P.; Vodnik, D. Effect of Meloidogyne ethiopica parasitism on water management and physiological stress in tomato. Eur. J. Plant Pathol. 2012, 132, 49–57. [Google Scholar] [CrossRef]
- Gálvez, A.; del Amor, F.M.; Ros, C.; López-Marín, J. New traits to identify physiological responses induced by different rootstocks after root-knot nematode inoculation (Meloidogyne incognita) in sweet pepper. Crop Prot. 2019, 119, 126–133. [Google Scholar] [CrossRef]
- Nunes da Silva, M.; Pintado, M.E.; Sarmento, B.; Stamford, N.P.; Vasconcelos, M.W. A biofertilizer with diazotrophic bacteria and a filamentous fungus increases Pinus pinaster tolerance to the pinewood nematode (Bursaphelenchus xylophilus). Biol. Control 2019, 132, 72–80. [Google Scholar] [CrossRef]
- Goulart, R.D.R.; Terra, W.C.; Salgado, S.M.D.L.; Alves, J.D.; Campos, V.P.; Fatobene, B.J.D.R.; Marchiori, P.E.R.; de Souza, S.R.; Oliveira, R.D.A.D.L. Meloidogyne paranaensis and M. exigua alter coffee physiology. Nematology 2018, 21, 459–467. [Google Scholar] [CrossRef]
- Mazurkiewicz-Zapałowicz, K.; Wróbel, J.; Janowicz, K. Interaction Between Golden Nematode Globodera Rostochiensis (Woll.), Behrens and Soil Saprophytic Fungi—Its Influence on The Processes of Gaseous Exchange and Crop Yield of Potato (Solanum Tuberosum L.). Arch. Phytopathol. Plant Prot. 2002, 35, 279–286. [Google Scholar] [CrossRef]
- Golinowski, W.; Grymaszewska, G. Reakcja tkanek korzenia na porażenie nicieniami z rodzaju Heterodera. Kosmos 1991, 40, 319–329. [Google Scholar]
- Mioranza, T.M.; Inagaki, A.M.; Müller, M.A.; Stangarlin, J.R.; Guimarães, V.F.; Klein, J.; Kuhn, O.J. Gas exchange and photosynthetic light response curves in nematode-infected tomato plants treated with Thuya occidentalis. Aust. J. Crop. Sci. 2018, 12, 583–591. [Google Scholar] [CrossRef]
- Thakur, R.K.; Dhirta, B.; Shirkot, P. Studies on Effect of Gold Nanoparticles on Meloidogyne Incognita and Tomato Plants Growth and Development; Cold Spring Harbor Laboratory: Cold Spring Harbor, NY, USA, 2018. [Google Scholar] [CrossRef]
- Gupta, R.; Singh, A.; Ajayakumar, P.V.; Pandey, R. Microbial interference mitigates Meloidogyne incognita mediated oxidative stress and augments bacoside content in Bacopa monnieri L. Microbiol. Res. 2017, 199, 67–78. [Google Scholar] [CrossRef] [PubMed]
- Chavan, S.B.; Deshpande, M.V. Chitinolytic enzymes: An appraisal as a product of commercial potential. Biotechnol. Prog. 2013, 29, 833–846. [Google Scholar] [CrossRef]
- Singh, S.P.; Gupta, R.; Gaur, R.; Srivastava, A.K. Streptomyces spp. alleviate Rhizoctonia solani mediated oxidative stress in Solanum lycopersicon. Ann. Appl. Biol. 2015, 168, 232–242. [Google Scholar] [CrossRef]
- Singh, A.; Jain, A.; Sarma, B.K.; Upadhyay, R.S.; Singh, H.B. Rhizosphere microbes facilitate redox homeostasis in Cicer arietinum against biotic stress. Ann. Appl. Biol. 2013, 163, 33–46. [Google Scholar] [CrossRef]
- Vos, C.; Schouteden, N.; van Tuinen, D.; Chatagnier, O.; Elsen, A.; De Waele, D.; Panis, B.; Gianinazzi-Pearson, V. Mycorrhiza-induced resistance against the root–knot nematode Meloidogyne incognita involves priming of defense gene responses in tomato. Soil Biol. Biochem. 2013, 60, 45–54. [Google Scholar] [CrossRef]
- Bajestani, M.S.; Moghadam, E.M.; Aghnoum, R.; Rohani, H. Genotypic and Biochemical variation in the response of barley to the Root-knot nematode (Meloidogyne javanica) at seedling stage. Pakistan J. Phytopathol. 2019, 31, 07–17. [Google Scholar] [CrossRef]
- Moghbeli, E.; Nemati, S.H.; Aroiee, H.; Olfati, J.-A. Evaluation of Resistance, Enzymatic Response, and Phenolic Compounds in Roots of F1 Cucumber Hybrids to Fusarium oxysporum F. Sp. Radicis-Cucumerinum. J. Hortic. Res. 2017, 25, 117–124. [Google Scholar] [CrossRef]
- Sung, Y.W.; Lee, I.H.; Shim, D.; Lee, K.-L.; Nam, K.J.; Yang, J.-W.; Lee, J.J.; Kwak, S.-S.; Kim, Y.-H. Transcriptomic changes in sweetpotato peroxidases in response to infection with the root-knot nematode Meloidogyne incognita. Mol. Biol. Rep. 2019, 46, 4555–4564. [Google Scholar] [CrossRef] [PubMed]
- Sahebani, N.; Hadavi, N. Induction of H2O2 and related enzymes in tomato roots infected with root knot nematode (M.javanica) by several chemical and microbial elicitors. Biocontrol Sci. Technol. 2009, 19, 301–313. [Google Scholar] [CrossRef]
- Labudda, M. Ascorbate-glutathione pathway as an important player in redox regulation in nematode-infested plants: What we have learned so far. Physiol. Mol. Plant Pathol. 2018, 103, 47–53. [Google Scholar] [CrossRef]
- Smirnoff, N.; Wheeler, G.L. Ascorbic Acid in Plants: Biosynthesis and Function. Crit. Rev. Plant Sci. 2000, 19, 267–290. [Google Scholar] [CrossRef]
- Kesba, H.H.; El-Beltagi, H.S. Biochemical changes in grape rootstocks resulted from humic acid treatments in relation to nematode infection. Asian Pac. J. Trop. Biomed. 2012, 2, 287–293. [Google Scholar] [CrossRef]
- Molinari, S.; Leonetti, P. Molecular Signaling Involved in Immune System Activation Against Root-Knot Nematodes by Bio-Control Agents in Tomato Plants; Cold Spring Harbor Laboratory: Cold Spring Harbor, NY, USA, 2019. [Google Scholar] [CrossRef]
- Singh, U.B.; Singh, S.; Khan, W.; Malviya, D.; Sahu, P.K.; Chaurasia, R.; Sharma, S.K.; Saxena, A.K. Drechslerella dactyloides and Dactylaria brochopaga mediated induction of defense related mediator molecules in tomato plants pre-challenged with Meloidogyne incognita. Ind. Phytopathol. 2019, 72, 309–320. [Google Scholar] [CrossRef]
- Clarke, C.R.; Vinatzer, B.A. Characterizing the Immune-Eliciting Activity of Putative Microbe-Associated Molecular Patterns in Tomato. In Methods in Molecular Biology; Springer: New York, NY, USA, 2017; pp. 249–261. [Google Scholar] [CrossRef]
- Galeng-Lawilao, J.; Kumar, A.; Cabasan, M.T.N.; De Waele, D. Comparison of the penetration, development and reproduction of Meloidogyne graminicola, and analysis of lignin and total phenolic content in partially resistant and resistant recombinant inbred lines of Oryza sativa. Trop. Plant Pathol. 2018, 44, 171–182. [Google Scholar] [CrossRef]
- Chin, S. The Role of Flavonoids in the Interaction Between the Plant, Medicago truncatula and the Nematode, Meloidogyne Javanica; Australian National University: Canberra, Australia, 2019. [Google Scholar]
- Labudda, M.; Różańska, E.; Czarnocka, W.; Sobczak, M.; Dzik, J.M. Systemic changes in photosynthesis and reactive oxygen species homeostasis in shoots of Arabidopsis thaliana infected with the beet cyst nematode Heterodera schachtii. Mol. Plant Pathol. 2018, 19, 1690–1704. [Google Scholar] [CrossRef]
- Franceschi, V.R.; Krokene, P.; Christiansen, E.; Krekling, T. Anatomical and chemical defenses of conifer bark against bark beetles and other pests. New Phytol. 2005, 167, 353–376. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, J.; ElAshry, A.E.N.; Anwar, S.; Erban, A.; Kopka, J.; Grundler, F. Metabolic profiling reveals local and systemic responses of host plants to nematode parasitism. Plant J. 2010. [Google Scholar] [CrossRef] [PubMed]
- Seo, Y.; Kim, Y.H. Control of Meloidogyne incognita Using Mixtures of Organic Acids. Plant Pathol. J. 2014, 30, 450–455. [Google Scholar] [CrossRef] [PubMed]
- Radwan, M.A. Comparative suppressive effect of some organic acids against Meloidogyne incognita infecting tomato. Pakistan J. Nematol. 2017, 35, 197–208. [Google Scholar] [CrossRef]
- Proença, D.N.; Schwab, S.; Vidal, M.S.; Baldani, J.I.; Xavier, G.R.; Morais, P.V. The nematicide Serratia plymuthica M24T3 colonizes Arabidopsis thaliana, stimulates plant growth, and presents plant beneficial potential. Braz. J. Microbiol. 2019, 50, 777–789. [Google Scholar] [CrossRef] [PubMed]
- Oh, M.; Han, J.W.; Lee, C.; Choi, G.J.; Kim, H. Nematicidal and Plant Growth-Promoting Activity of Enterobacter asburiae HK169: Genome Analysis Provides Insight into Its Biological Activities. J. Microbiol. Biotechnol. 2018, 28, 968–975. [Google Scholar] [CrossRef] [PubMed]
- Shemshura, O.N.; Bekmakhanova, N.E.; Mazunina, M.N.; Meyer, S.L.F.; Rice, C.P.; Masler, E.P. Isolation and identification of nematode-antagonistic compounds from the fungus Aspergillus candidus. FEMS Microbiol. Lett. 2016, 363, fnw026. [Google Scholar] [CrossRef][Green Version]
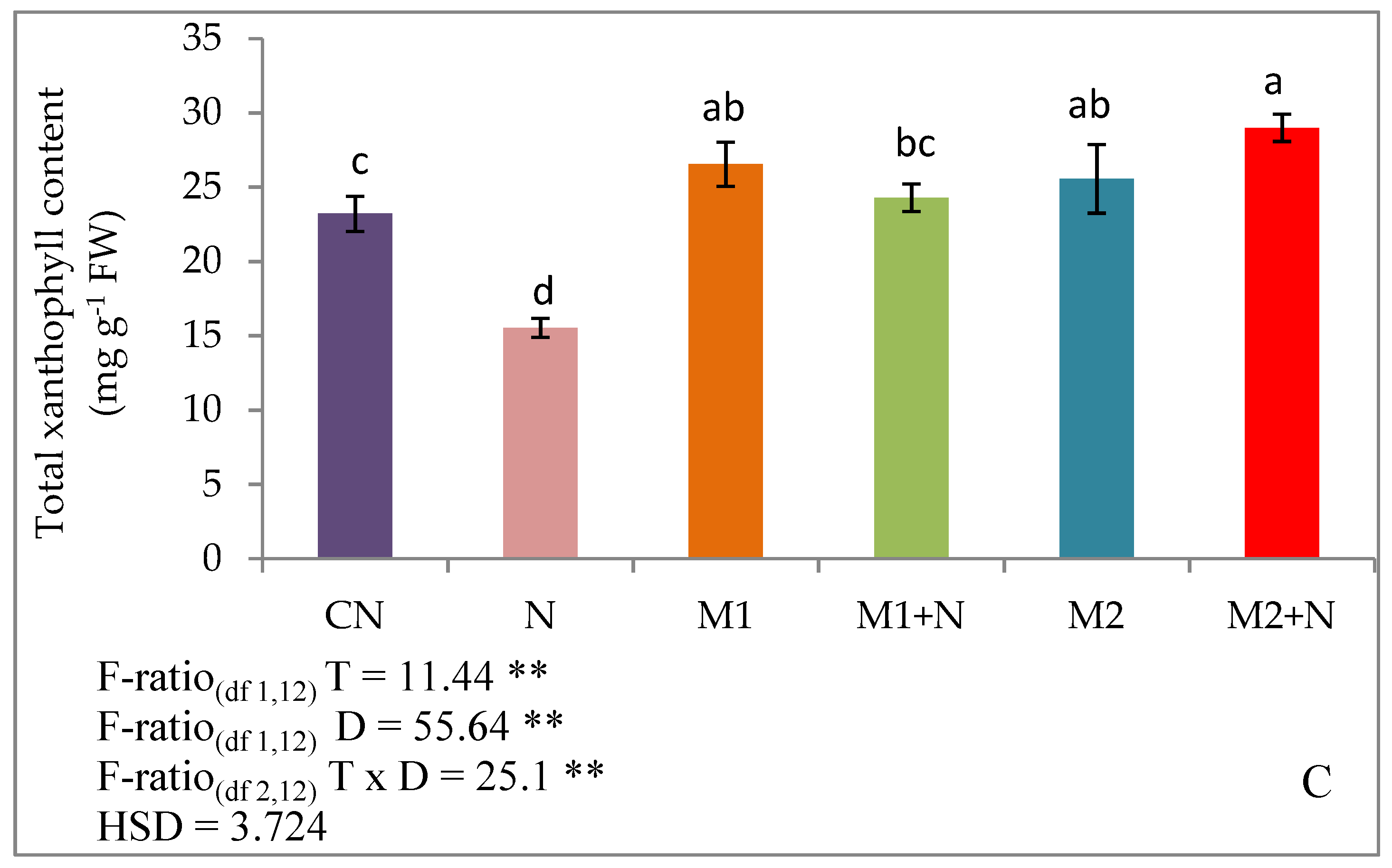
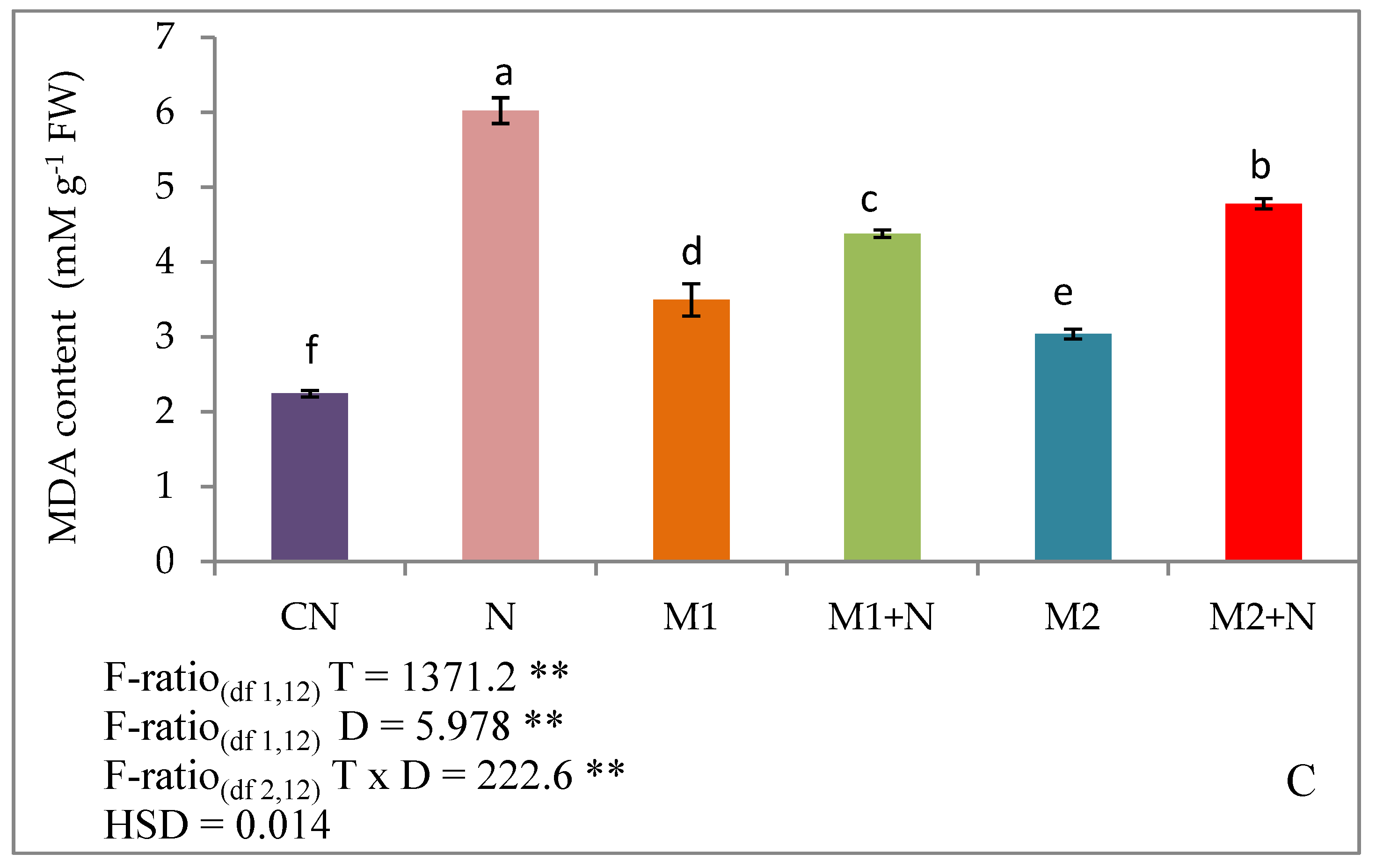
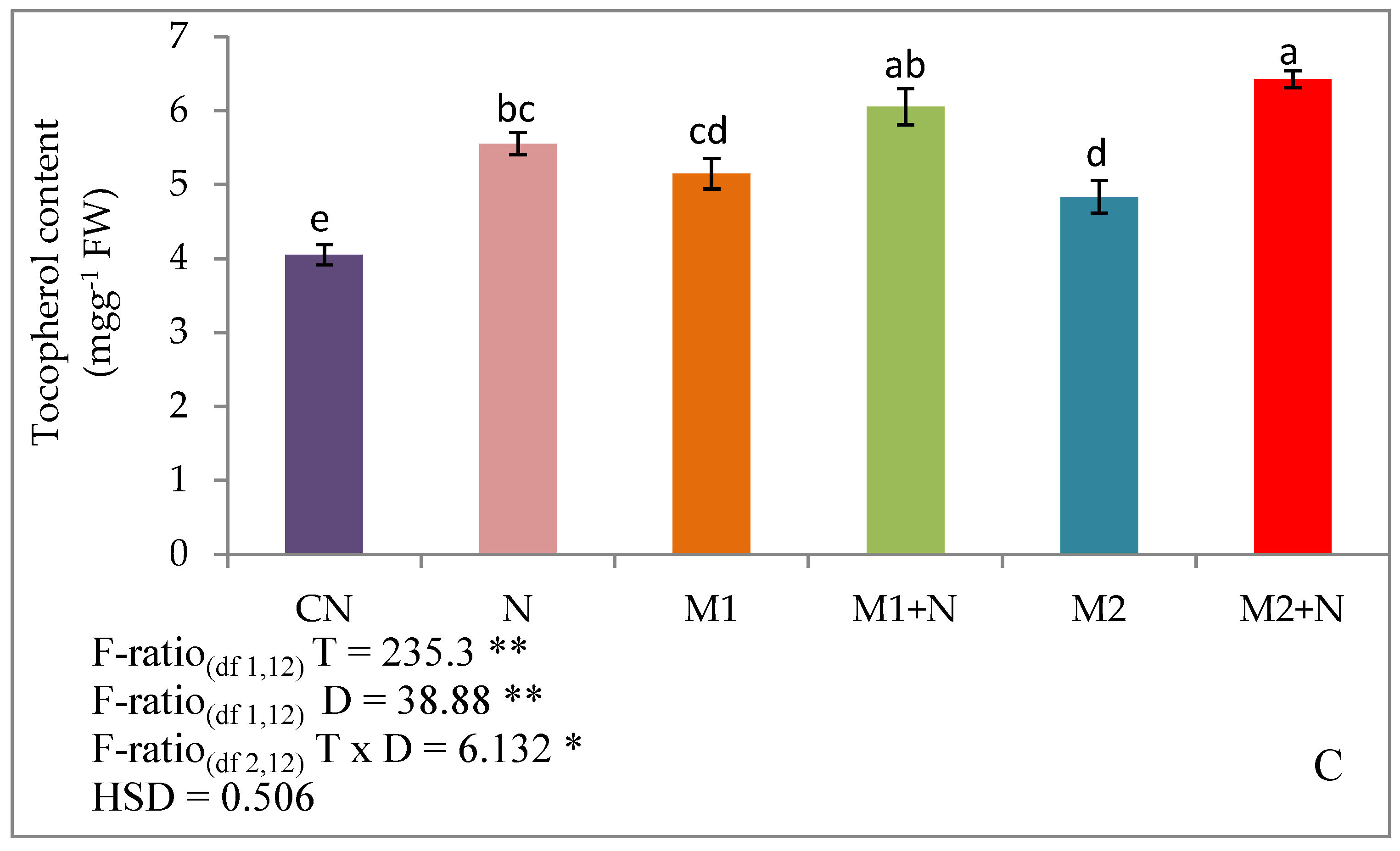
| Treatments | Root Length (cm) (Mean ± SD) | Shoot Length (cm) (Mean ± SD) | Fresh Weight (g/plant) (Mean ± SD) | Dry Weight (g/plant) (Mean ± SD) | No. of Galls (Mean ± SD) |
|---|---|---|---|---|---|
| CN | 15.78 ± 0.2505 b | 22.13 ± 0.4635 d | 4.69 ± 0.2495 a | 1.96 ± 0.1334 b,c | 0 |
| N | 8.96 ± 0.6502 d | 17.36 ± 0.6036 e | 3.04 ± 0.2502 c | 1.02 ± 0.1251 d | 27.33 ± 1.527 a |
| M1 | 17.42 ± 0.3651 b | 24.54 ± 0.8015 c | 4.71 ± 0.1182 a | 1.93 ± 0.0695 b,c | 0 |
| M1 + N | 15.53 ± 0.7946 b,c | 26.80 ± 1.1654 b | 4.05 ± 0.2501 b | 2.10 ± 0.1100 a,b | 19.66 ± 0.1.527 b |
| M2 | 19.50 ± 0.7379 a | 22.38 ± 0.4178 c,d | 4.21 ± 0.1608 a,b | 2.02 ± 0.1481 b | 0 |
| M2 + N | 13.59 ± 1.1119 c | 29.47 ± 1.0726 a | 4.55 ± 0.1574 a | 2.15 ± 0.099 a | 22 ± 1 b |
| F-ratio(df 1,12) T | 210.9 ** | 15.85 ** | 45.38 ** | 15.39 ** | 2520.5 ** |
| F-ratio(df 2,12) D | 68.13 ** | 110.6 ** | 12.27 ** | 46.41 ** | 27.54 ** |
| F-ratio(df 2,12) T × D | 20.65 * | 80.30 ** | 31.75 ** | 43.17 ** | 24.52 ** |
| HSD | 1.949 | 2.217 | 0.593 | 0.321 | 2.665 |
| Treatments | Net Photosynthetic Rate (µmol m2 s−1) (Mean ± SD) | Stomatal Conductance (mmol CO2 m2 s−1) (Mean ± SD) | Intracellular CO2 Rate (µmol mol1) (Mean ± SD) | Transpiration Rate (mmol m2 s−1) (Mean ± SD) |
|---|---|---|---|---|
| CN | 25.54 ± 0.9492 c | 0.474 ± 0.016 d | 436 ±8.426 b,c | 2.453 ± 0.115 b |
| N | 16.87 ± 0.3952 e | 0.324 ± 0.0075 e | 386.3 ± 6.760 d | 1.463 ± 0.1150 d |
| M1 | 28.33 ± 0.8304 b | 0.524 ± 0.0125 c | 455.6 ± 5.511 b | 2.75 ± 0.1178 a,b |
| M1+N | 22.44 ± 0.6165 d | 0.584 ± 0.0117 b | 419.7 ± 8.891 c | 1.903 ± 0.1078 c |
| M2 | 27.73 ± 0.535 b,c | 0.496 ± 0.0091 c,d | 442.3 ± 3.146 b | 2.97 ± 0.090 a |
| M2 + N | 31.55 ± 1.2690 a | 0.611 ± 0.0123 a | 480.9 ± 8.603 a | 2.13 ± 0.1418 c |
| F-ratio(df 1,12) T | 86.69 ** | 2.256 | 21.27 ** | 266.5 ** |
| F-ratio(df 2,12) D | 159.09 ** | 336.04 ** | 74.02 ** | 40.46 ** |
| F-ratio(df 2,12) T × D | 96.33 ** | 205.72 ** | 65.39 ** | 1.846 |
| HSD | 2.245 | 0.0326 | 19.702 | 0.3171 |
| Treatments | SOD (Mean ± SD) | POD (Mean ± SD) | CAT (Mean ± SD) | GPOX (Mean ± SD) | APOX (Mean ± SD) | DHAR (Mean ± SD) | GST (Mean ± SD) | GR (Mean ± SD) | PPO (Mean ± SD) |
|---|---|---|---|---|---|---|---|---|---|
| Control | 53.44 ± 3.506 d | 256.4 ± 6.612 f | 54.08 ± 1.859 d | 23.58 ± 0.8076 e | 389.1 ± 9.030 d | 136.8 ± 1.718 d | 21.73 ± 1.672 e | 210.5 ± 5.574 e | 71.13 ± 2.373 f |
| N | 71.97 ± 1.254 b | 402.8 ± 1.859 c | 70.94 ± 0.941 b | 33.65 ± 1.1611 c | 442.3 ± 11.220 c | 150.5 ± 6.151 c | 27.45 ± 1.831 c | 353.9 ± 3.156 b | 84.82 ± 3.642 e |
| M1 | 61.0 ± 1.787 c | 349.8 ± 6.733 e | 58.77 ± 3.792 c,d | 27.35 ± 0.8783 d | 651.0 ± 16.274 a | 167.1 ± 3.257 b | 32.03 ± 2.427 c | 286.7 ± 9.087 c | 96.28 ± 3.006 d |
| M1 + N | 84.45 ± 2.184 a | 524.2 ± 1.539 a | 78.62 ± 2.826 a | 36.0 ± 0.7885 b | 510.9 ± 3.854 b | 174.7 ± 2.368 a,b | 38.04 ± 1.908 b | 360.1 ± 4.650 a,b | 137.7 ± 4.165 a |
| M2 | 59.33 ± 3.729 c,d | 370.3 ± 1.270 d | 63.14 ± 2.310 c | 25.94 ± 0.3994 d | 401.9 ± 2.382 d | 156.7 ± 3.503 c | 31.41 ± 1.5008 c,d | 246.8 ± 13.211 d | 111.8 ± 3.856 c |
| M2 + N | 78.74 ± 1.727 ab | 454.6 ± 6.143 b | 76.50 ± 1.901 a,b | 40.1 ± 0.9736 a | 522.4 ± 8.446 b | 183.2 ± 2.587 a | 44.44 ± 2.130 a | 379.8 ± 3.659 a | 123.8 ± 5.584 b |
| F-ratio(df 1,12) T | 291.8 ** | 3667.7 ** | 193.4 ** | 720.3 ** | 3.199 | 90.11 ** | 81.78 ** | 11730.7 ** | 148.3 ** |
| F-ratio(df 2,12) D | 23.85 ** | 849.5 ** | 18.97 ** | 40.77 ** | 246.7 ** | 113.3 4** | 78.78 ** | 54.79 ** | 204.4 ** |
| F-ratio(df 2,12) T × D | 1.599 | 142.6 ** | 4.795 ** | 16.29 ** | 155.47 ** | 11.09 ** | 6.881 * | 37.20 ** | 26.91 ** |
| HSD | 6.967 | 12.972 | 6.686 | 2.375 | 36.445 | 9.758 | 5.308 | 19.95 | 10.69 |
| Treatments | Fumaric Acid (mg g−1 FW) (Mean ± SD) | Citric Acid (mg g−1 FW) (Mean ± SD) | Succinic Acid (mg g−1 FW) (Mean ± SD) | Malic Acid (mg g−1 FW) (Mean ± SD) |
|---|---|---|---|---|
| CN | 0.515 ± 0.0060 e | 4.337 ± 0.1838 e | 0.930 ± 0.0225 c | 3.409 ± 0.1758 f |
| N | 0.565 ± 0.0111 d | 6.162 ± 0.1476 cd | 1.206 ± 0.0338 b | 5.379 ± 0.2266 e |
| M1 | 0.649 ± 0.0225 c | 5.674 ± 0.1402 d | 0.985 ± 0.0121 c | 6.403 ± 0.1581 d |
| M1 + N | 0.718 ± 0.0193 b | 8.878 ± 0.2081 b | 1.351 ± 0.0327 a | 11.778 ± 0.4950 b |
| M2 | 0.611 ± 0.0106 c | 6.544 ± 0.1093 c | 0.953 ± 0.0106 c | 9.237 ± 0.1091 c |
| M2 + N | 0.780 ± 0.0198 a | 10.020 ± 0.3984 a | 1.413 ± 0.0353 a | 12.94 ± 0.3555 a |
| F-ratio(df 1,12) T | 160.7 ** | 750.6 ** | 859.22 ** | 746.3 ** |
| F-ratio(df 2,12) D | 173.2 ** | 297.2 ** | 33.144 ** | 866.5 ** |
| F-ratio(df 2,12) T × D | 23.83 ** | 24.24 ** | 17.907 ** | 53.17 ** |
| HSD | 0.0440 | 0.6022 | 0.0729 | 0.7842 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khanna, K.; Sharma, A.; Ohri, P.; Bhardwaj, R.; Abd_Allah, E.F.; Hashem, A.; Ahmad, P. Impact of Plant Growth Promoting Rhizobacteria in the Orchestration of Lycopersicon esculentum Mill. Resistance to Plant Parasitic Nematodes: A Metabolomic Approach to Evaluate Defense Responses Under Field Conditions. Biomolecules 2019, 9, 676. https://doi.org/10.3390/biom9110676
Khanna K, Sharma A, Ohri P, Bhardwaj R, Abd_Allah EF, Hashem A, Ahmad P. Impact of Plant Growth Promoting Rhizobacteria in the Orchestration of Lycopersicon esculentum Mill. Resistance to Plant Parasitic Nematodes: A Metabolomic Approach to Evaluate Defense Responses Under Field Conditions. Biomolecules. 2019; 9(11):676. https://doi.org/10.3390/biom9110676
Chicago/Turabian StyleKhanna, Kanika, Anket Sharma, Puja Ohri, Renu Bhardwaj, Elsayed F. Abd_Allah, Abeer Hashem, and Parvaiz Ahmad. 2019. "Impact of Plant Growth Promoting Rhizobacteria in the Orchestration of Lycopersicon esculentum Mill. Resistance to Plant Parasitic Nematodes: A Metabolomic Approach to Evaluate Defense Responses Under Field Conditions" Biomolecules 9, no. 11: 676. https://doi.org/10.3390/biom9110676
APA StyleKhanna, K., Sharma, A., Ohri, P., Bhardwaj, R., Abd_Allah, E. F., Hashem, A., & Ahmad, P. (2019). Impact of Plant Growth Promoting Rhizobacteria in the Orchestration of Lycopersicon esculentum Mill. Resistance to Plant Parasitic Nematodes: A Metabolomic Approach to Evaluate Defense Responses Under Field Conditions. Biomolecules, 9(11), 676. https://doi.org/10.3390/biom9110676








