The Alcohol–High-Density Lipoprotein Athero-Protective Axis
Abstract
1. Introduction
2. Correlating HDL, Alcohol Ingestion, and ASCVD
3. Alcohol Metabolism
4. Chronic Alcohol Consumption Enhances MCE
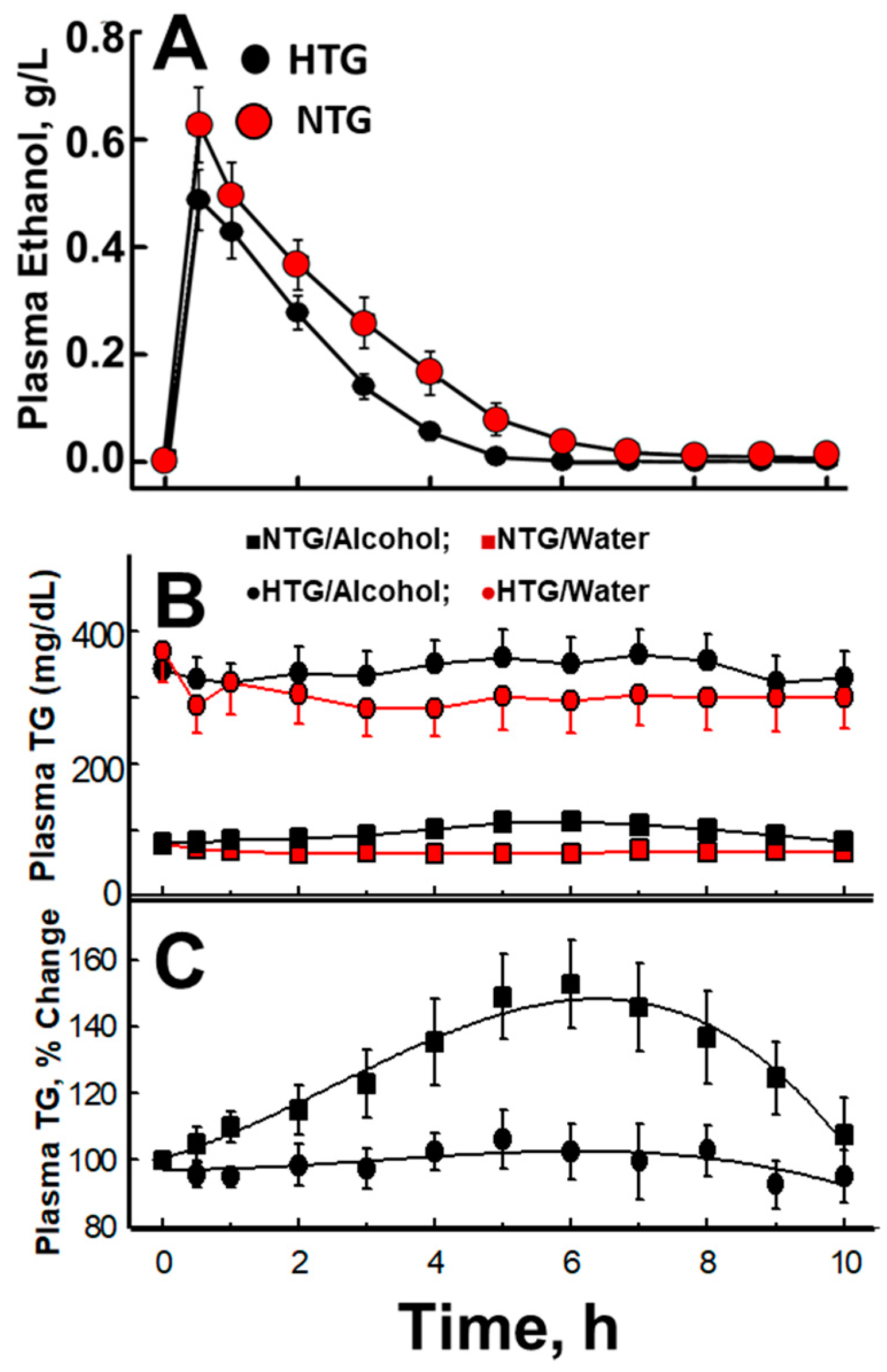
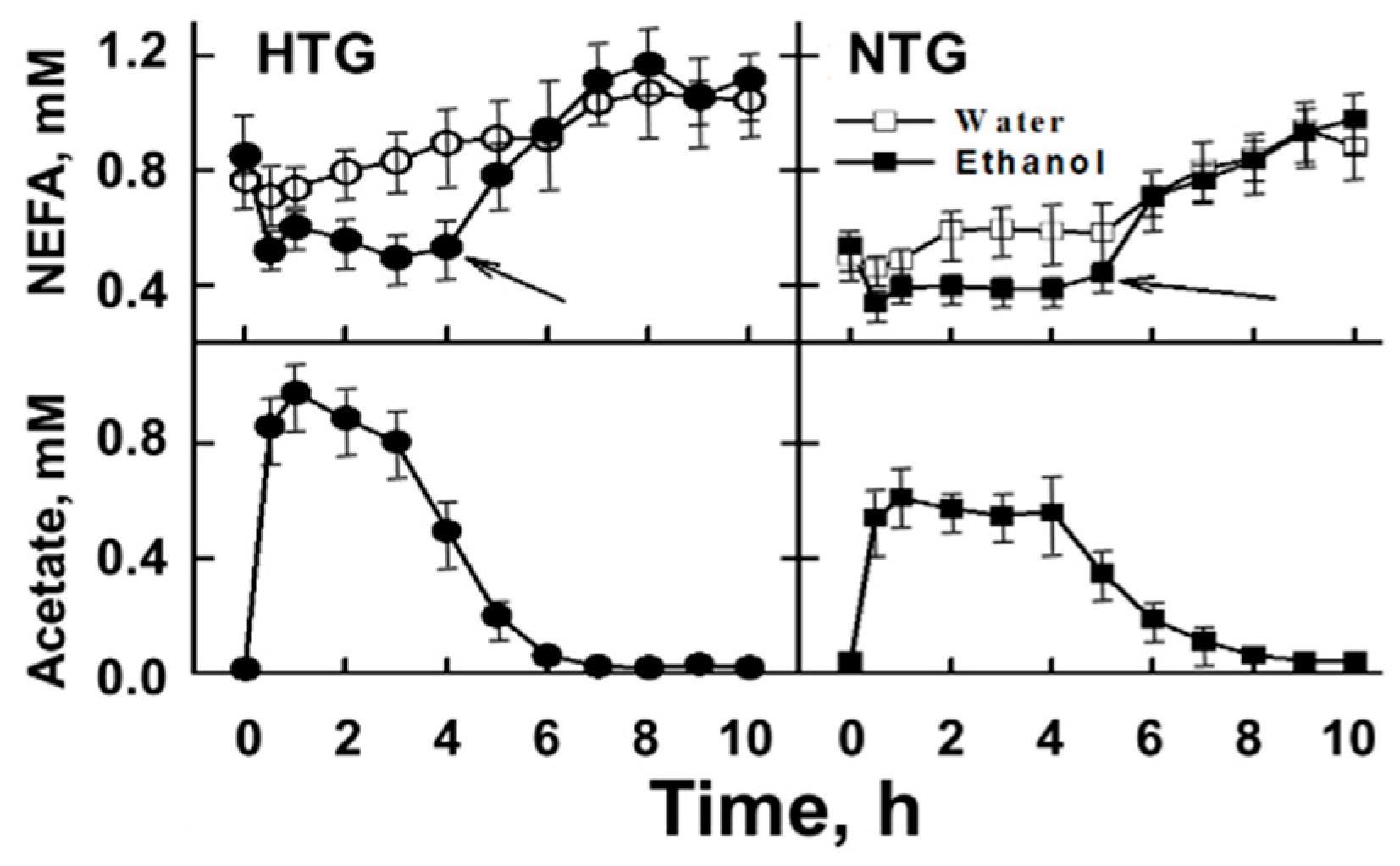
5. Acute Effects of Alcohol Consumption
6. The Role of Acetate in Alcohol-Mediated Effects on Plasma Lipid Metabolism
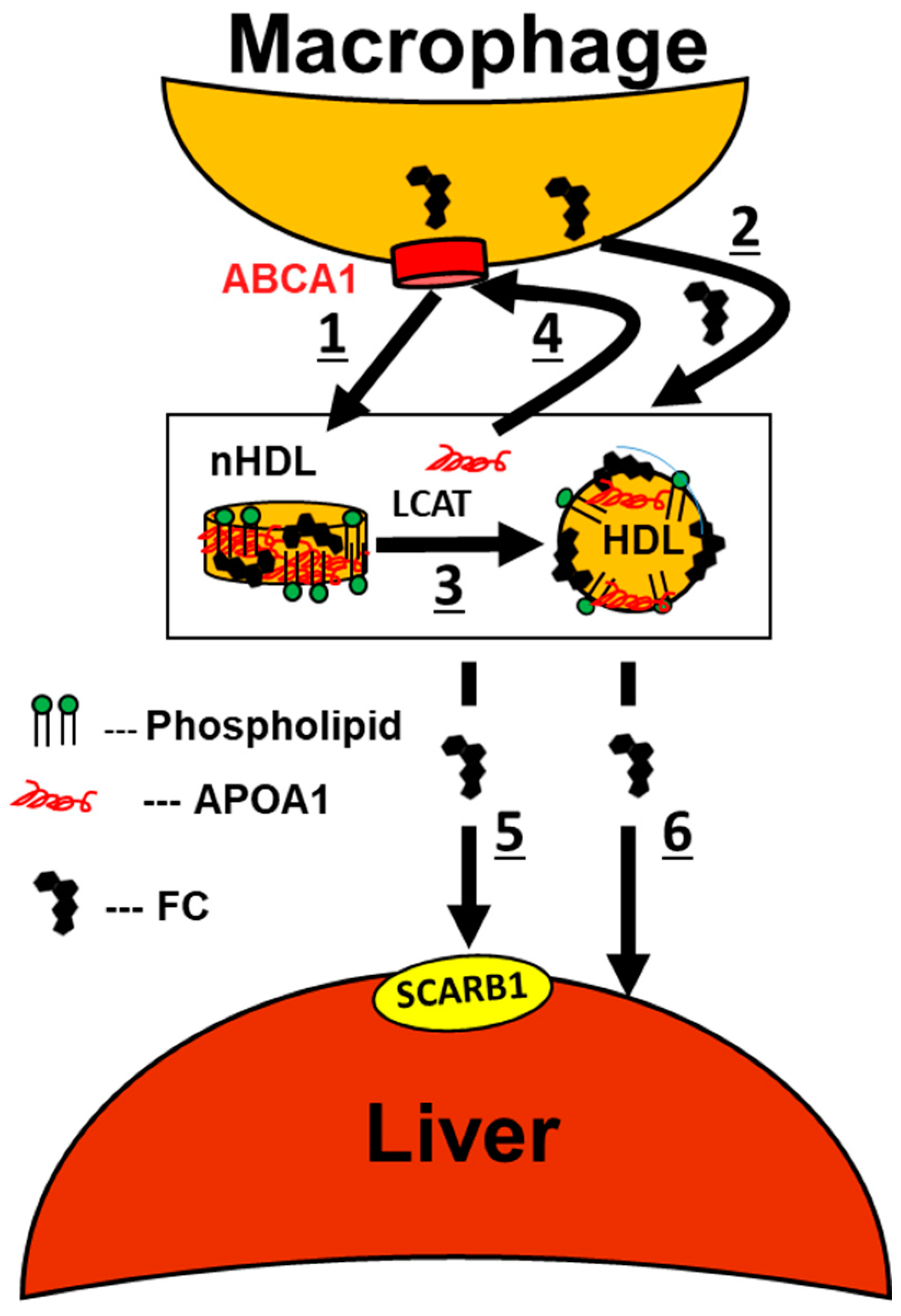
7. Mechanisms Underlying the Alcohol–HDL Athero-Protective Axis
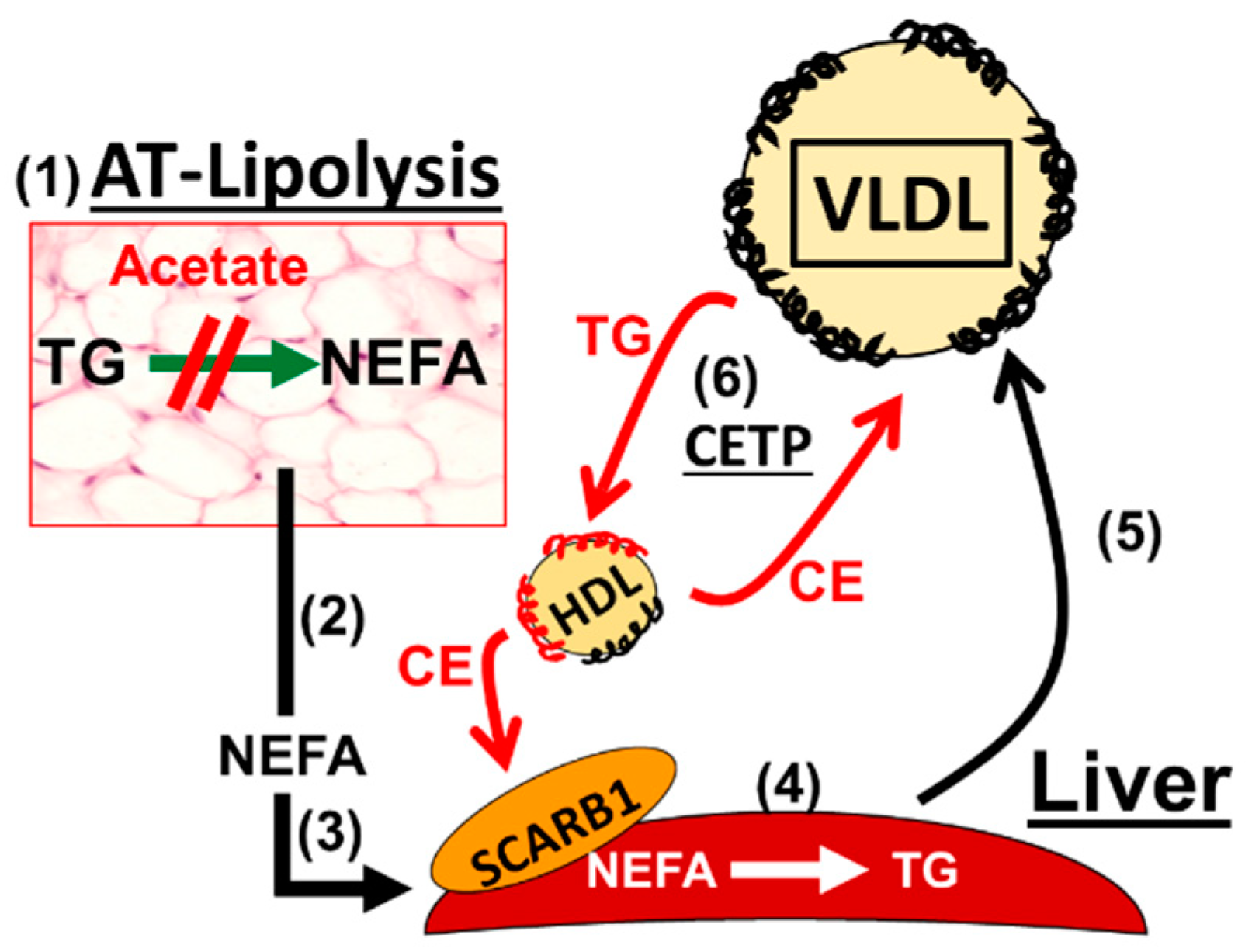
8. A Mechanistic Model Links Alcohol Ingestion to a Cardio-Protective Plasma Lipoprotein Profile
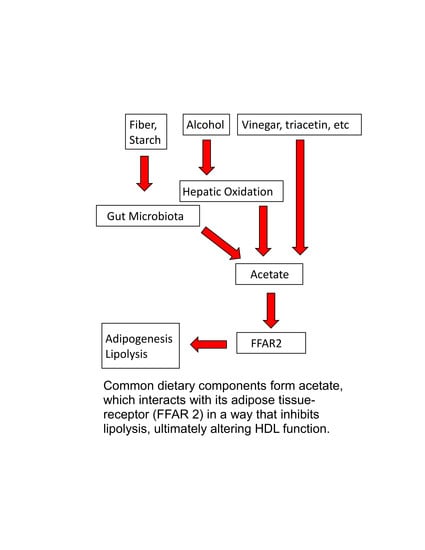
9. Acetate in Foods
10. Acetate as ASCVD Therapy
11. Open Questions
- Does acetate ingestion molar-equivalently enhance the postprandial lipemia seen with alcohol only?
- Whereas the lipemia induced by both alcohol and fat consumed separately increases with the magnitude of fasting plasma TG levels, is a similar relationship seen when acetate is consumed with fat-containing food and, if so, are the effects of acetate and fat co-ingestion synergistic or additive?
- Is the occurrence of obesity-linked diabetes, pancreatitis, and ASCVD among persons with mutation-associated deficiencies in alcohol-metabolizing enzymes different from that of those carrying the metabolically competent alleles?
- What if any role does the FFAR2 have in the etiology of ASCVD in consumers vs. non-consumers of alcohol and acetate or its precursors?
- Do molar-equivalent amounts of acetate and alcohol provide similar cardio-protective effects?
Author Contributions
Funding
Conflicts of Interest
References
- Gofman, J.W.; Young, W.; Tandy, R. Ischemic Heart Disease, Atherosclerosis, and Longevity. Circulation 1966, 34, 679–697. [Google Scholar] [CrossRef] [PubMed]
- Williams, P.T.; Feldman, D.E. Prospective study of coronary heart disease vs. HDL2, HDL3, and other lipoproteins in Gofman’s Livermore Cohort. Atherosclerosis 2011, 214, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Wilson, P.W.; Abbott, R.D.; Castelli, W.P. High density lipoprotein cholesterol and mortality. The Framingham Heart Study. Arteriosclerosis 1988, 8, 737–741. [Google Scholar] [CrossRef]
- Frick, M.H.; Elo, O.; Haapa, K.; Heinonen, O.P.; Heinsalmi, P.; Helo, P.; Huttunen, J.K.; Kaitaniemi, P.; Koskinen, P.; Manninen, V.; et al. Helsinki Heart Study: Primary-Prevention Trial with Gemfibrozil in Middle-Aged Men with Dyslipidemia. New Engl. J. Med. 1987, 317, 1237–1245. [Google Scholar] [CrossRef] [PubMed]
- Manninen, V.; Elo, M.O.; Frick, M.H.; Haapa, K.; Heinonen, O.P.; Heinsalmi, P.; Helo, P.; Huttunen, J.K.; Kaitaniemi, P.; Koskinen, P.; et al. Lipid Alterations and Decline in the Incidence of Coronary Heart Disease in the Helsinki Heart Study. JAMA 1988, 260, 641. [Google Scholar] [CrossRef] [PubMed]
- Robins, S.; Collins, D.; Wittes, J. Relation of gemfibrozil treatment and lipid levels with major coronary events. VA-HIT: A randomized controlled trial. ACC Curr. J. Rev. 2001, 10, 23–24. [Google Scholar] [CrossRef]
- Cuchel, M.; Rader, D. Macrophage Reverse Cholesterol Transport. Circulation 2006, 113, 2548–2555. [Google Scholar] [CrossRef]
- Zhong, S.; Sharp, D.S.; Grove, J.S.; Bruce, C.; Yano, K.; Curb, J.D.; Tall, A.R. Increased coronary heart disease in Japanese-American men with mutation in the cholesteryl ester transfer protein gene despite increased HDL levels. J. Clin. Investig. 1996, 97, 2917–2923. [Google Scholar] [CrossRef]
- Haase, C.L.; Tybjaerg-Hansen, A.; Grande, P.; Frikke-Schmidt, R. Genetically Elevated Apolipoprotein A-I, High-Density Lipoprotein Cholesterol Levels, and Risk of Ischemic Heart Disease. J. Clin. Endocrinol. Metab. 2010, 95, 500–510. [Google Scholar] [CrossRef]
- Voight, B.F.; Peloso, G.M.; Orho-Melander, M.; Frikke-Schmidt, R.; Barbalić, M.; Jensen, M.K.; Hindy, G.; Holm, H.; Ding, E.L.; Johnson, T.; et al. Plasma HDL cholesterol and risk of myocardial infarction: A mendelian randomisation study. Lancet 2012, 380, 572–580. [Google Scholar] [CrossRef]
- Schwartz, G.G.; Olsson, A.G.; Abt, M.; Ballantyne, C.M.; Barter, P.J.; Brumm, J.; Chaitman, B.R.; Holme, I.; Kallend, D.; Leiter, L.A.; et al. Effects of Dalcetrapib in Patients with a Recent Acute Coronary Syndrome. New Engl. J. Med. 2012, 367, 2089–2099. [Google Scholar] [CrossRef] [PubMed]
- Barter, P.J.; Caulfield, M.J.; Eriksson, M.; Grundy, S.M.; Kastelein, J.J.; Komajda, M.; López-Sendón, J.; Mosca, L.; Tardif, J.-C.; Waters, D.D.; et al. Effects of Torcetrapib in Patients at High Risk for Coronary Events. New Engl. J. Med. 2007, 357, 2109–2122. [Google Scholar] [CrossRef] [PubMed]
- Boden, W.E.; Probstfield, J.L.; Anderson, T.; Chaitman, B.R.; Desvignes-Nickens, P.; Koprowicz, K.; McBride, R.; Teo, K.; Weintraub, W. Niacin in patients with low HDL cholesterol levels receiving intensive statin therapy. N. Engl. J. Med. 2011, 365, 2255–2267. [Google Scholar]
- Santander, N.G.; Contreras-Duarte, S.; Awad, M.F.; Lizama, C.; Passalacqua, I.; Rigotti, A.; Busso, D. Developmental abnormalities in mouse embryos lacking the HDL receptor SR-BI. Hum. Mol. Genet. 2013, 22, 2551. [Google Scholar] [CrossRef][Green Version]
- Anonymous. The coronary drug project. Findings leading to further modifications of its protocol with respect to dextrothyroxine. The coronary drug project research group. JAMA 1972, 220, 996–1008. [Google Scholar]
- Maclure, M. Demonstration of Deductive Meta-Analysis: Ethanol Intake and Risk of Myocardial Infarction. Epidemiol. Rev. 1993, 15, 328–351. [Google Scholar] [CrossRef]
- Gaziano, J.; Buring, J.E. Alcohol intake, lipids and risks of myocardial infarction. Ciba Foundation Symposium—Bilharziasis 1998, 216, 86–110. [Google Scholar] [CrossRef]
- Toth, P.P.; Barter, P.J.; Rosenson, R.S.; Boden, W.E.; Chapman, M.J.; Cuchel, M.; D’Agostino, R.B.; Davidson, M.H.; Davidson, W.S.; Heinecke, J.W.; et al. High-density lipoproteins: A consensus statement from the National Lipid Association. J. Clin. Lipidol. 2013, 7, 484–525. [Google Scholar] [CrossRef]
- Rosenson, R.S.; Brewer, H.B.; Davidson, W.S.; Fayad, Z.A.; Fuster, V.; Goldstein, J.; Hellerstein, M.; Jiang, X.-C.; Phillips, M.; Rader, D.J.; et al. Cholesterol efflux and atheroprotection: Advancing the concept of reverse cholesterol transport. Circulation 2012, 125, 1905–1919. [Google Scholar] [CrossRef]
- Rosenson, R.S.; Brewer, H.B.; Ansell, B.J.; Barter, P.; Chapman, M.J.; Heinecke, J.W.; Kontush, A.; Tall, A.R.; Webb, N.R. Dysfunctional HDL and atherosclerotic cardiovascular disease. Nat. Rev. Cardiol. 2015, 13, 48–60. [Google Scholar] [CrossRef]
- Rohatgi, A.; Khera, A.; Berry, J.D.; Givens, E.G.; Ayers, C.R.; Wedin, K.E.; Neeland, I.J.; Yuhanna, I.S.; Rader, D.R.; De Lemos, J.A.; et al. HDL cholesterol efflux capacity and incident cardiovascular events. New Engl. J. Med. 2014, 371, 2383–2393. [Google Scholar] [CrossRef] [PubMed]
- Khera, A.V.; Cuchel, M.; De La Llera-Moya, M.; Rodrigues, A.; Burke, M.F.; Jafri, K.; French, B.C.; Phillips, J.A.; Mucksavage, M.L.; Wilensky, R.L.; et al. Cholesterol efflux capacity, high-density lipoprotein function, and atherosclerosis. New Engl. J. Med. 2011, 364, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Saleheen, D.; Scott, R.; Javad, S.; Zhao, W.; Rodrigues, A.; Picataggi, A.; Lukmanova, D.; Mucksavage, M.L.; Luben, R.N.; Billheimer, J.; et al. Association of HDL cholesterol efflux capacity with incident coronary heart disease events: A prospective case-control study. Lancet Diabetes Endocrinol. 2015, 3, 507–513. [Google Scholar] [CrossRef]
- Soria-Florido, M.T.; Castañer, O.; Lassale, C.; Estruch, R.; Salas-Salvadó, J.; Martínez-González, M.Á.; Corella, D.; Ros, E.; Arós, F.; Elosua, R.; et al. Dysfunctional High-Density Lipoproteins Are Associated With a Greater Incidence of Acute Coronary Syndrome in a Population at High Cardiovascular Risk. Circulation 2020, 141, 444–453. [Google Scholar] [CrossRef]
- Swertfeger, D.; Rebholz, S.; Li, H.; Shah, A.S.; Davidson, W.S.; Lu, L.J. Feasibility of a plasma bioassay to assess oxidative protection of low-density lipoproteins by high-density lipoproteins. J. Clin. Lipidol. 2018, 12, 1539–1548. [Google Scholar] [CrossRef]
- Yvan-Charvet, L.; Wang, N.; Tall, A.R. Role of HDL, ABCA1, and ABCG1 Transporters in Cholesterol Efflux and Immune Responses. Arter. Thromb. Vasc. Boil. 2010, 30, 139–143. [Google Scholar] [CrossRef]
- Zimmer, S.; Grebe, A.; Bakke, S.S.; Bode, N.; Halvorsen, B.; Ulas, T.; Skjelland, M.; De Nardo, D.; Labzin, L.; Kerksiek, A.; et al. Cyclodextrin promotes atherosclerosis regression via macrophage reprogramming. Sci. Transl. Med. 2016, 8, 333ra50. [Google Scholar] [CrossRef]
- Li, Y.; Schwabe, R.F.; DeVries-Seimon, T.; Yao, P.M.; Gerbod-Giannone, M.-C.; Tall, A.R.; Davis, R.J.; Flavell, R.; Brenner, D.A.; Tabas, I. Free Cholesterol-loaded Macrophages Are an Abundant Source of Tumor Necrosis Factor-α and Interleukin-6. J. Boil. Chem. 2005, 280, 21763–21772. [Google Scholar] [CrossRef]
- Fotakis, P.; Kothari, V.; Thomas, D.G.; Westerterp, M.; Molusky, M.M.; Altin, E.; Abramowicz, S.; Wang, N.; He, Y.; Heinecke, J.W.; et al. Anti-Inflammatory Effects of HDL (High-Density Lipoprotein) in Macrophages Predominate Over Proinflammatory Effects in Atherosclerotic Plaques. Arter. Thromb. Vasc. Boil. 2019, 39, 253. [Google Scholar] [CrossRef]
- Tchoua, U.; Rosales, C.; Tang, D.; Gillard, B.K.; Vaughan, A.; Lin, H.Y.; Courtney, H.S.; Pownall, H.J. Serum Opacity Factor Enhances HDL-Mediated Cholesterol Efflux, Esterification and Anti Inflammatory Effects. Lipids 2010, 45, 1117–1126. [Google Scholar] [CrossRef]
- Ross, R.; A Glomset, J. Atherosclerosis and the arterial smooth muscle cell: Proliferation of smooth muscle is a key event in the genesis of the lesions of atherosclerosis. Science 1973, 180, 1332–1339. [Google Scholar] [CrossRef] [PubMed]
- Gillard, B.K.; Bassett, G.R.; Gotto, A.M.; Rosales, C.; Pownall, H.J. Scavenger receptor B1 (SR-B1) profoundly excludes high density lipoprotein (HDL) apolipoprotein AII as it nibbles HDL-cholesteryl ester. J. Boil. Chem. 2017, 292, 8864–8873. [Google Scholar] [CrossRef] [PubMed]
- Glass, C.; Pittman, R.C.; Weinstein, D.B.; Steinberg, D. Dissociation of tissue uptake of cholesterol ester from that of apoprotein A-I of rat plasma high density lipoprotein: Selective delivery of cholesterol ester to liver, adrenal, and gonad. Proc. Natl. Acad. Sci. USA 1983, 80, 5435–5439. [Google Scholar] [CrossRef] [PubMed]
- Acton, S.; Rigotti, A.; Landschulz, K.T.; Xu, S.; Hobbs, H.H.; Krieger, M. Identification of Scavenger Receptor SR-BI as a High Density Lipoprotein Receptor. Science 1996, 271, 518–520. [Google Scholar] [CrossRef]
- Smoak, K.A.; Aloor, J.J.; Madenspacher, J.; Merrick, B.A.; Collins, J.B.; Zhu, X.; Cavigiolio, G.; Oda, M.N.; Parks, J.S.; Fessler, M.B. Myeloid Differentiation Primary Response Protein 88 Couples Reverse Cholesterol Transport to Inflammation. Cell Metab. 2010, 11, 493–502. [Google Scholar] [CrossRef]
- Van der Vorst, E.P.C.; Theodorou, K.; Wu, Y.; Hoeksema, M.A.; Goossens, P.; Bursill, C.A.; Aliyev, T.; Huitema, L.F.A.; Tas, S.W.; Wolfs, I.M.J.; et al. High-Density Lipoproteins Exert Pro-inflammatory Effects on Macrophages via Passive Cholesterol Depletion and PKC-NF-κB/STAT1-IRF1 Signaling. Cell Metab. 2017, 25, 197–207. [Google Scholar] [CrossRef]
- Li, X.-M.; Tang, W.H.W.; Mosior, M.K.; Huang, Y.; Wu, Y.; Matter, W.; Gao, V.; Schmitt, D.; DiDonato, J.A.; Fisher, E.A.; et al. Paradoxical association of enhanced cholesterol efflux with increased incident cardiovascular risks. Arter. Thromb. Vasc. Boil. 2013, 33, 1696–1705. [Google Scholar] [CrossRef] [PubMed]
- Mutharasan, R.K.; Thaxton, C.S.; Berry, J.; Daviglus, M.L.; Yuan, C.; Sun, J.; Ayers, C.; Lloyd-Jones, D.; Wilkins, J.T. HDL efflux capacity, HDL particle size, and high-risk carotid atherosclerosis in a cohort of asymptomatic older adults: The Chicago Healthy Aging Study. J. Lipid Res. 2017, 58, 600–606. [Google Scholar] [CrossRef]
- Aeddula, N.R.; Trivedi, N.; Pathireddy, S.; De Vries, R.; Groen, A.K.; Dullaart, R.P.F.; Asleh, R.; Levy, A.P.; Blum, S.; Frohlich, J.; et al. Cholesterol Efflux Capacity and Atherosclerosis. New Engl. J. Med. 2011, 364, 1472–1475. [Google Scholar] [CrossRef]
- Josefs, T.; Wouters, K.; Tietge, U.J.; Annema, W.; Dullaart, R.P.; Vaisar, T.; Arts, I.C.; Van Der Kallen, C.J.; Stehouwer, C.D.; Schalkwijk, C.G.; et al. High-density lipoprotein cholesterol efflux capacity is not associated with atherosclerosis and prevalence of cardiovascular outcome: The CODAM study. J. Clin. Lipidol. 2020, 14, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Brenn, T. The Tromso heart study: Alcoholic beverages and coronary risk factors. J. Epidemiol. Community Heal. 1986, 40, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Cuvelier, I.; Steinmetz, J.; Mikstacki, T.; Siest, G. Variations in total phospholipids and high-density lipoprotein phospholipids in plasma from a general population: Reference intervals and influence of xenobiotics. Clin. Chem. 1985, 31, 763–766. [Google Scholar] [CrossRef]
- Clevidence, B.A.; Reichman, M.E.; Judd, J.T.; Muesing, R.A.; Schatzkin, A.; Schaefer, E.J.; Li, Z.; Jenner, J.; Brown, C.C.; Sunkin, M. Effects of alcohol consumption on lipoproteins of premenopausal women. A controlled diet study. Arter. Thromb. Vasc. Boil. 1995, 15, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Hendriks, H.F.J.; Veenstra, J.; Van Tol, A.; Groener, J.E.M.; Schaafsma, G. Modearte Doses of Alcoholic Beverages with Dinner and Postprandial High Density Lipoprotein Composition. Alcohol Alcohol. 1998, 33, 403–410. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Baer, D.J.; Judd, J.T.; Clevidence, B.A.; Muesing, R.A.; Campbell, W.S.; Brown, E.D.; Taylor, P.R. Moderate alcohol consumption lowers risk factors for cardiovascular disease in postmenopausal women fed a controlled diet. Am. J. Clin. Nutr. 2002, 75, 593–599. [Google Scholar] [CrossRef]
- Hartung, G.H.; Lawrence, S.J.; Reeves, R.S.; Foreyt, J.P. Effect of alcohol and exercise on postprandial lipemia and triglyceride clearance in men. Atherosclerosis 1993, 100, 33–40. [Google Scholar] [CrossRef]
- Hartung, G.H.; Foreyt, J.P.; Mitchell, R.E.; Reeves, R.S.; Gotto, A.M. Effect of Alcohol Intake on High-Density Lipoprotein Cholesterol Levels in Runners and Inactive Men. JAMA 1983, 249, 747. [Google Scholar] [CrossRef]
- Kabagambe, E.K.; Baylin, A.; Ruiz-Narvaez, E.; Rimm, E.B.; Campos, H. Alcohol intake, drinking patterns, and risk of nonfatal acute myocardial infarction in Costa Rica. Am. J. Clin. Nutr. 2005, 82, 1336–1345. [Google Scholar] [CrossRef]
- Mukamal, K.J.; Jensen, M.K.; Grønbæk, M.; Stampfer, M.J.; Manson, J.E.; Pischon, T.; Rimm, E.B. Drinking Frequency, Mediating Biomarkers, and Risk of Myocardial Infarction in Women and Men. Circulation 2005, 112, 1406–1413. [Google Scholar] [CrossRef]
- Beulens, J.W.J.; Rimm, E.B.; Ascherio, A.; Spiegelman, N.; Hendriks, H.F.J.; Mukamal, K.J. Alcohol Consumption and Risk for Coronary Heart Disease among Men with Hypertension. Ann. Intern. Med. 2007, 146, 10. [Google Scholar] [CrossRef]
- Mukamal, K.J.; Chiuve, S.E.; Rimm, E.B. Alcohol Consumption and Risk for Coronary Heart Disease in Men with Healthy Lifestyles. Arch. Intern. Med. 2006, 166, 2145. [Google Scholar] [CrossRef] [PubMed]
- Yusuf, S.; Hawken, S.; Ounpuu, S.; Dans, T.; Avezum, A.; Lanas, F.; McQueen, M.; Budaj, A.; Pais, P.; Varigos, J.; et al. Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the INTERHEART study): Case-control study. Lancet 2004, 364, 937–952. [Google Scholar] [CrossRef]
- Goldberg, I.J.; Mosca, L.; Piano, M.R.; Fisher, E.A. Wine and Your Heart. A Science Advisory for Healthcare Professionals from the Nutrition Committee, Council on Epidemiology and Prevention, and Council on Cardiovascular Nursing of the American Heart Association. Circulation 2001, 103, 472–475. [Google Scholar] [CrossRef] [PubMed]
- Linn, S.; Carroll, M.; Johnson, C.; Fulwood, R.; Kalsbeek, W.; Briefel, R. High-density lipoprotein cholesterol and alcohol consumption in US white and black adults: Data from NHANES II. Am. J. Public Heal. 1993, 83, 811–816. [Google Scholar] [CrossRef] [PubMed]
- Silva, E.R.D.O.E.; Foster, D.; Harper, M.M.; Seidman, C.E.; Smith, J.D.; Breslow, J.L.; Brinton, E.A. Alcohol consumption raises HDL cholesterol levels by increasing the transport rate of apolipoproteins A-I and A-II. Circulation 2000, 102, 2347–2352. [Google Scholar] [CrossRef]
- Thun, M.J.; Monaco, J.H.; Peto, R.; Heath, C.W.; Doll, R.; Lopez, A.; Henley, S.J. Alcohol Consumption and Mortality among Middle-Aged and Elderly U.S. Adults. New Engl. J. Med. 1997, 337, 1705–1714. [Google Scholar] [CrossRef]
- Gordon, T.; Kannel, W.B. Drinking and Mortality: The Framingham Study. Am. J. Epidemiology 1984, 120, 97–107. [Google Scholar] [CrossRef]
- Magnus, P.; Bakke, E.; Hoff, D.A.; Høiseth, G.; Graff-Iversen, S.; Knudsen, G.P.S.; Myhre, R.; Normann, P.T.; Næss, Ø.; Tambs, K.; et al. Controlling for High-Density Lipoprotein Cholesterol Does Not Affect the Magnitude of the Relationship Between Alcohol and Coronary Heart Disease. Circulation 2011, 124, 2296–2302. [Google Scholar] [CrossRef]
- Marques-Vidal, P.; Bochud, M.; Paccaud, F.; Waterworth, D.; Bergmann, S.; Preisig, M.; Waeber, G.; Vollenweider, P. No interaction between alcohol consumption and HDL-related genes on HDL cholesterol levels. Atherosclerosis 2010, 211, 551–557. [Google Scholar] [CrossRef]
- Baraona, E.; Lieber, C.S. Alcohol and lipids. Recent Dev. Alcohol. 1998, 14, 97–134. [Google Scholar]
- World Health Organization. United States of America, Alcohol Consumption. 2016. Available online: https://www.who.int/substance_abuse/publications/global_alcohol_report/profiles/usa.pdf (accessed on 26 June 2018).
- Eng, M.Y.; Luczak, S.E.; Wall, T.L. ALDH2, ADH1B, and ADH1C Genotypes in Asians: A Literature Review. Alcohol Res. Heal. J. Natl. Inst. Alcohol Abus. Alcohol. 2007, 30, 22–27. [Google Scholar]
- Ebtehaj, S.; Gruppen, E.G.; Bakker, S.J.; Dullaart, R.P.; Tietge, U.J. HDL (High-Density Lipoprotein) Cholesterol Efflux Capacity Is Associated With Incident Cardiovascular Disease in the General Population. Arter. Thromb. Vasc. Boil. 2019, 39, 1874–1883. [Google Scholar] [CrossRef] [PubMed]
- Khera, A.V.; Demler, O.V.; Adelman, S.J.; Collins, H.L.; Glynn, R.J.; Ridker, P.M.; Rader, D.J.; Mora, S. Cholesterol Efflux Capacity, High-Density Lipoprotein Particle Number, and Incident Cardiovascular Events: An Analysis From the JUPITER Trial (Justification for the Use of Statins in Prevention: An Intervention Trial Evaluating Rosuvastatin). Circulation 2017, 135, 2494–2504. [Google Scholar] [CrossRef] [PubMed]
- Sierksma, A.; Vermunt, S.H.F.; Lankhuizen, I.M.; Van Der Gaag, M.S.; Scheek, L.M.; Grobbee, D.E.; Van Tol, A.; Hendriks, H.F. Effect of Moderate Alcohol Consumption on Parameters of Reverse Cholesterol Transport in Postmenopausal Women. Alcohol. Clin. Exp. Res. 2004, 28, 662–666. [Google Scholar] [CrossRef]
- Lesná, I.K.; Suchánek, P.; Stávek, P.; Poledne, R. May alcohol-induced increase of HDL be considered as atheroprotective? Physiol. Res. 2009, 59, 407–413. [Google Scholar]
- Van Der Gaag, M.S.; Van Tol, A.; Vermunt, S.H.; Scheek, L.M.; Schaafsma, G.; Hendriks, H.F. Alcohol consumption stimulates early steps in reverse cholesterol transport. J. Lipid Res. 2001, 42, 2077–2083. [Google Scholar]
- Perret, B.; Ruidavets, J.-B.; Vieu, C.; Jaspard, B.; Cambou, J.-P.; Tercé, F.; Collet, X. Alcohol consumption is associated with enrichment of high-density lipoprotein particles in polyunsaturated lipids and increased cholesterol esterification rate. Alcohol. Clin. Exp. Res. 2002, 26, 1134–1140. [Google Scholar] [CrossRef]
- Senault, C.; Betoulle, D.; Luc, G.; Hauw, P.; Rigaud, D.; Fumeron, F. Beneficial effects of a moderate consumption of red wine on cellular cholesterol efflux in young men. Nutr. Metab. Cardiovasc. Dis. 2000, 10, 63–69. [Google Scholar]
- Davidson, W.S.; Gillotte, K.L.; Lund-Katz, S.; Johnson, W.J.; Rothblat, G.H.; Phillips, M. The Effect of High Density Lipoprotein Phospholipid Acyl Chain Composition on the Efflux of Cellular Free Cholesterol. J. Boil. Chem. 1995, 270, 5882–5890. [Google Scholar] [CrossRef]
- Akopian, D.; Kawashima, R.L.; Medh, J. Phosphatidylcholine-Mediated Aqueous Diffusion of Cellular Cholesterol Down-Regulates the ABCA1 Transporter in Human Skin Fibroblasts. Int. J. Biochem. Res. Rev. 2015, 5, 214–224. [Google Scholar] [CrossRef]
- Tchoua, U.; Gillard, B.K.; Pownall, H.J. HDL superphospholipidation enhances key steps in reverse cholesterol transport. Atherosclerosis 2009, 209, 430–435. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gillotte, K.L.; Davidson, W.S.; Lund-Katz, S.; Rothblat, G.H.; Phillips, M.C. Removal of cellular cholesterol by pre-beta-HDL involves plasma membrane microsolubilization. J. Lipid Res. 1998, 39, 1918–1928. [Google Scholar] [PubMed]
- Beulens, J.W.; Sierksma, A.; Van Tol, A.; Van Gent, T.; Fournier, N.; Paul, J.-L.; Hendriks, H.F. Moderate Alcohol Consumption Increases Cholesterol Reflux mediated by ABCA1. Alcohol. Clin. Exp. Res. 2004, 28, 7. [Google Scholar] [CrossRef]
- Chung, B.-H.; Franklin, F.; Cho, B.H.S.; Segrest, J.P.; Hart, K.; Darnell, B.E. Potencies of Lipoproteins in Fasting and Postprandial Plasma to Accept Additional Cholesterol Molecules Released From Cell Membranes. Arter. Thromb. Vasc. Boil. 1998, 18, 1217–1230. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wilson, D.E.; Schreibman, P.H.; Brewster, A.C.; Arky, R.A. The enhancement of alimentary lipemia by ethanol in man. J. Lab. Clin. Med. 1970, 75, 264–274. [Google Scholar] [PubMed]
- Pownall, H.J. Dietary ethanol is associated with reduced lipolysis of intestinally derived lipoproteins. J. Lipid Res. 1994, 35, 2105–2113. [Google Scholar] [PubMed]
- Julia, Z.; Duchene, E.; Fournier, N.; Bellanger, N.; Chapman, M.J.; Le Goff, W.; Guerin, M. Postprandial lipemia enhances the capacity of large HDL2 particles to mediate free cholesterol efflux via SR-BI and ABCG1 pathways in type IIB hyperlipidemia. J. Lipid Res. 2010, 51, 3350–3358. [Google Scholar] [CrossRef]
- Berryman, C.E.; Grieger, J.A.; West, S.G.; Chen, C.-Y.O.; Blumberg, J.B.; Rothblat, G.H.; Sankaranarayanan, S.; Kris-Etherton, P.M. Acute consumption of walnuts and walnut components differentially affect postprandial lipemia, endothelial function, oxidative stress, and cholesterol efflux in humans with mild hypercholesterolemia. J. Nutr. 2013, 143, 788–794. [Google Scholar] [CrossRef]
- Vasudevan, M.; Tchoua, U.; Gillard, B.K.; Jones, P.H.; Ballantyne, C.M.; Pownall, H.J. Modest diet-induced weight loss reduces macrophage cholesterol efflux to plasma of patients with metabolic syndrome. J. Clin. Lipidol. 2013, 7, 661–670. [Google Scholar] [CrossRef][Green Version]
- Van Tol, A.; Groener, J.E.M.; Scheek, L.M.; Van Gent, T.; Veenstra, J.; Van De Pol, H.; Hendriks, H.F.J.; Schaafsma, G. Induction of net mass lipid transfer reactions in plasma by wine consumption with dinner. Eur. J. Clin. Investig. 1995, 25, 390–395. [Google Scholar] [CrossRef]
- Gaubatz, J.W.; Gillard, B.K.; Rosales, C.; Pownall, H.J. Dietary Alcohol and Fat Differentially Affect Plasma Cholesteryl Ester Transfer Activity and Triglycerides in Normo- and Hypertriglyceridemic Subjects. Lipids 2020. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, S.; Watanabe, J.; Kawajiri, K. Genetic polymorphisms in the 5′-flanking region change transcriptional regulation of the human cytochrome P450IIE1 gene. J. Biochem. 1991, 110, 559–565. [Google Scholar] [CrossRef] [PubMed]
- Rimm, E.B.; Klatsky, A.; Grobbee, D.; Stampfer, M.J. Review of moderate alcohol consumption and reduced risk of coronary heart disease: Is the effect due to beer, wine, or spirits? BMJ 1996, 312, 731–736. [Google Scholar] [CrossRef]
- Barefoot, J.C.; Grønbæk, M.; Feaganes, J.R.; McPherson, R.S.; Williams, R.; Siegler, I.C. Alcoholic beverage preference, diet, and health habits in the UNC Alumni Heart Study. Am. J. Clin. Nutr. 2002, 76, 466–472. [Google Scholar] [CrossRef] [PubMed]
- Pownall, H.J.; Ballantyne, C.M.; Kimball, K.T.; Simpson, S.L.; Yeshurun, D.; Gotto, A.M. Effect of moderate alcohol consumption on hypertriglyceridemia: A study in the fasting state. Arch. Intern. Med. 1999, 159, 981–987. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, S.; Fushimi, T.; Kishi, M.; Irie, S.; Tsuji, S.; Hosokawa, N.; Kaga, T. Bioavailability of acetate from two vinegar supplements: Capsule and drink. J. Nutr. Sci. Vitaminol. 2010, 56, 266–269. [Google Scholar] [CrossRef][Green Version]
- Nilsson, N.O.; Belfrage, P. Effects of acetate, acetaldehyde, and ethanol on lipolysis in isolated rat adipocytes. J. Lipid Res. 1978, 19, 737–741. [Google Scholar]
- Nilsson, N.E.; Kotarsky, K.; Owman, C.; Olde, B. Identification of a free fatty acid receptor, FFA2R, expressed on leukocytes and activated by short-chain fatty acids. Biochem. Biophys. Res. Commun. 2003, 303, 1047–1052. [Google Scholar] [CrossRef]
- Hong, Y.-H.; Nishimura, Y.; Hishikawa, D.; Tsuzuki, H.; Miyahara, H.; Gotoh, C.; Choi, K.-C.; Feng, D.D.; Chen, C.; Lee, H.-G.; et al. Acetate and Propionate Short Chain Fatty Acids Stimulate Adipogenesis via GPCR43. Endocrinology 2005, 146, 5092–5099. [Google Scholar] [CrossRef]
- Ge, H.; Li, X.; Weiszmann, J.; Wang, P.; Baribault, H.; Chen, J.-L.; Tian, H.; Li, Y. Activation of G Protein-Coupled Receptor 43 in Adipocytes Leads to Inhibition of Lipolysis and Suppression of Plasma Free Fatty Acids. Endocrinology 2008, 149, 4519–4526. [Google Scholar] [CrossRef]
- Abramson, E.A.; Arky, R.A. Acute antilipolytic effects of ethyl alcohol and acetate in man. J. Lab. Clin. Med. 1968, 72, 105–117. [Google Scholar] [PubMed]
- Pownall, H.J.; Brauchi, D.; Kilinç, C.; Osmundsen, K.; Pao, Q.; Payton-Ross, C.; Gotto, A.M.; Ballantyne, C.M. Correlation of serum triglyceride and its reduction by omega-3 fatty acids with lipid transfer activity and the neutral lipid compositions of high-density and low-density lipoproteins. Atherosclerosis 1999, 143, 285–297. [Google Scholar] [CrossRef]
- Balasubramanyam, A.; Sekhar, R.V.; Jahoor, F.; Jones, P.H.; Pownall, H.J. Pathophysiology of dyslipidemia and increased cardiovascular risk in HIV lipodystrophy: A model of ‘systemic steatosis’. Curr. Opin. Lipidol. 2004, 15, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Hlebowicz, J.; Darwich, G.; Björgell, O.; Almér, L.-O. Effect of apple cider vinegar on delayed gastric emptying in patients with type 1 diabetes mellitus: A pilot study. BMC Gastroenterol. 2007, 7, 46. [Google Scholar] [CrossRef] [PubMed]
- Johnston, C.S.; Kim, C.M.; Buller, A.J. Vinegar improves insulin sensitivity to a high-carbohydrate meal in subjects with insulin resistance or type 2 diabetes. Diabetes Care 2004, 27, 281–282. [Google Scholar] [CrossRef] [PubMed]
- Johnston, C.S.; White, A.M.; Kent, S.M. Preliminary evidence that regular vinegar ingestion favorably influences hemoglobin A1c values in individuals with type 2 diabetes mellitus. Diabetes Res. Clin. Pr. 2009, 84, e15–e17. [Google Scholar] [CrossRef]
- Liatis, S.; Grammatikou, S.; Poulia, K.A.; Perrea, D.; Makrilakis, K.; Diakoumopoulou, E.; Katsilambros, N. Vinegar reduces postprandial hyperglycaemia in patients with type II diabetes when added to a high, but not to a low, glycaemic index meal. Eur. J. Clin. Nutr. 2010, 64, 727–732. [Google Scholar] [CrossRef]
- Balliett, M.; Burke, J.R. Changes in anthropometric measurements, body composition, blood pressure, lipid profile, and testosterone in patients participating in a low-energy dietary intervention. J. Chiropr. Med. 2013, 12, 3–14. [Google Scholar] [CrossRef][Green Version]
- Shishehbor, F.; Mansoori, A.; Sarkaki, A.R.; Jalali, M.T.; Latifi, S.M. Apple Cider Vinegar Attenuates Lipid Profile in Normal and Diabetic Rats. Pak. J. Boil. Sci. 2008, 11, 2634–2638. [Google Scholar] [CrossRef]
- Jasbi, P.; Baker, O.; Shi, X.; Gonzalez, L.A.; Wang, S.; Anderson, S.; Xi, B.; Gu, H.; Johnston, C.S. Daily red wine vinegar ingestion for eight weeks improves glucose homeostasis and affects the metabolome but does not reduce adiposity in adults. Food Funct. 2019, 10, 7343–7355. [Google Scholar] [CrossRef]
- Mitrou, P.; Petsiou, E.; Papakonstantinou, E.; Maratou, E.; Lambadiari, V.; Dimitriadis, P.; Spanoudi, F.; A Raptis, S.; Dimitriadis, G. The role of acetic acid on glucose uptake and blood flow rates in the skeletal muscle in humans with impaired glucose tolerance. Eur. J. Clin. Nutr. 2015, 69, 734–739. [Google Scholar] [CrossRef] [PubMed]
- Johnston, C.S.; Steplewska, I.; Long, C.A.; Harris, L.N.; Ryals, R.H. Examination of the Antiglycemic Properties of Vinegar in Healthy Adults. Ann. Nutr. Metab. 2010, 56, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Bouderbala, H.; Kaddouri, H.; Maharrar, M.; Kheroua, O.; Saidi, D. P2-11: Anti obesogenic effects of apple cider vinegar in rats subjected to a high fat diet. Annales de Cardiologie et d’Angéiologie 2015, 64, S27. [Google Scholar] [CrossRef]
- Fushimi, T.; Suruga, K.; Oshima, Y.; Fukiharu, M.; Tsukamoto, Y.; Goda, T. Dietary acetic acid reduces serum cholesterol and triacylglycerols in rats fed a cholesterol-rich diet. Br. J. Nutr. 2006, 95, 916–924. [Google Scholar] [CrossRef] [PubMed]
- Johnston, C.S.; Gaas, C.A. Vinegar: Medicinal Uses and Antiglycemic Effect. MedGenMed Medscape Gen. Med. 2006, 8, 61. [Google Scholar]
- Halima, B.H.; Sonia, G.; Sarra, K.; Houda, B.J.; Fethi, B.S.; Abdallah, A. Apple Cider Vinegar Attenuates Oxidative Stress and Reduces the Risk of Obesity in High-Fat-Fed Male Wistar Rats. J. Med. Food 2018, 21, 70–80. [Google Scholar] [CrossRef]
- Pingitore, A.; Gonzalez-Abuin, N.; Ruz-Maldonado, I.; Huang, G.C.; Frost, G.; Persaud, S.J. Short chain fatty acids stimulate insulin secretion and reduce apoptosis in mouse and human islets in vitro: Role of free fatty acid receptor 2. Diabetes, Obes. Metab. 2018, 21, 330–339. [Google Scholar] [CrossRef]
- Pomare, E.W.; Branch, W.J.; Cummings, J.H. Carbohydrate fermentation in the human colon and its relation to acetate concentrations in venous blood. J. Clin. Investig. 1985, 75, 1448–1454. [Google Scholar] [CrossRef]
- Topping, D.L.; Clifton, P.M. Short-chain fatty acids and human colonic function: Roles of resistant starch and nonstarch polysaccharides. Physiol. Rev. 2001, 81, 1031–1064. [Google Scholar] [CrossRef]
- Frost, G.; Sleeth, M.L.; Sahuri-Arisoylu, M.; Lizarbe, B.; Cerdan, S.; Brody, L.; Anastasovska, J.; Ghourab, S.; Hankir, M.; Zhang, S.; et al. The short-chain fatty acid acetate reduces appetite via a central homeostatic mechanism. Nat. Commun. 2014, 5, 3611. [Google Scholar] [CrossRef]
- Lundquist, F.; Tygstrup, N.; Winkler, K.; Mellemgaard, K.; Munck-Petersen, S. Etanol Metabolism and Production of Free Acetate in the Human Liver. J. Clin. Investig. 1962, 41, 955–961. [Google Scholar] [CrossRef] [PubMed]
- Siler, S.Q.; Neese, R.A.; Hellerstein, M.K. De novo lipogenesis, lipid kinetics, and whole-body lipid balances in humans after acute alcohol consumption. Am. J. Clin. Nutr. 1999, 70, 928–936. [Google Scholar] [CrossRef] [PubMed]
- Liljeberg, H.; Björck, I. Delayed gastric emptying rate may explain improved glycaemia in healthy subjects to a starchy meal with added vinegar. Eur. J. Clin. Nutr. 1998, 52, 368–371. [Google Scholar] [CrossRef] [PubMed]
- Leeman, M.; Ostman, E.; Björck, I. Vinegar dressing and cold storage of potatoes lowers postprandial glycaemic and insulinaemic responses in healthy subjects. Eur. J. Clin. Nutr. 2005, 59, 1266–1271. [Google Scholar] [CrossRef]
- Sugiyama, M.; Tang, A.C.; Wakaki, Y.; Koyama, W. Glycemic index of single and mixed meal foods among common Japanese foods with white rice as a reference food. Eur. J. Clin. Nutr. 2003, 57, 743–752. [Google Scholar] [CrossRef]
- Ostman, E.; Elmståhl, H.G.L.; Björck, I.M.E. Inconsistency between glycemic and insulinemic responses to regular and fermented milk products. Am. J. Clin. Nutr. 2001, 74, 96–100. [Google Scholar] [CrossRef]
- Vu, C.N.; Ruiz-Esponda, R.; Yang, E.Y.; Chang, E.; Gillard, B.K.; Pownall, H.J.; Hoogeveen, R.; Coraza, I.; Balasubramanyam, A. Altered relationship of plasma triglycerides to HDL cholesterol in patients with HIV/HAART-associated dyslipidemia: Further evidence for a unique form of Metabolic Syndrome in HIV patients. Metabolism 2013, 62, 1014–1020. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rosales, C.; Gillard, B.K.; Gotto, A.M., Jr.; Pownall, H.J. The Alcohol–High-Density Lipoprotein Athero-Protective Axis. Biomolecules 2020, 10, 987. https://doi.org/10.3390/biom10070987
Rosales C, Gillard BK, Gotto AM Jr., Pownall HJ. The Alcohol–High-Density Lipoprotein Athero-Protective Axis. Biomolecules. 2020; 10(7):987. https://doi.org/10.3390/biom10070987
Chicago/Turabian StyleRosales, Corina, Baiba K. Gillard, Antonio M. Gotto, Jr., and Henry J. Pownall. 2020. "The Alcohol–High-Density Lipoprotein Athero-Protective Axis" Biomolecules 10, no. 7: 987. https://doi.org/10.3390/biom10070987
APA StyleRosales, C., Gillard, B. K., Gotto, A. M., Jr., & Pownall, H. J. (2020). The Alcohol–High-Density Lipoprotein Athero-Protective Axis. Biomolecules, 10(7), 987. https://doi.org/10.3390/biom10070987