Lipschütz Ulcer and SARS-CoV-2: What We Currently Know?
Abstract
:1. Introduction
1.1. Major Criteria
- Acute onset of at least one painful vulvar ulcer.
- Need to exclude infectious and non-infectious causes.
1.2. Minor Criteria
- Localized ulcer at the level of the vestibule or labia minora.
- Abstention from sexual intercourse in the last 3 months or patient who has never had sexual intercourse.
- Flu-like symptoms.
- Development of systemic infection at least 2–4 weeks preceding ulcer development [5].
2. Materials and Methods
3. Results
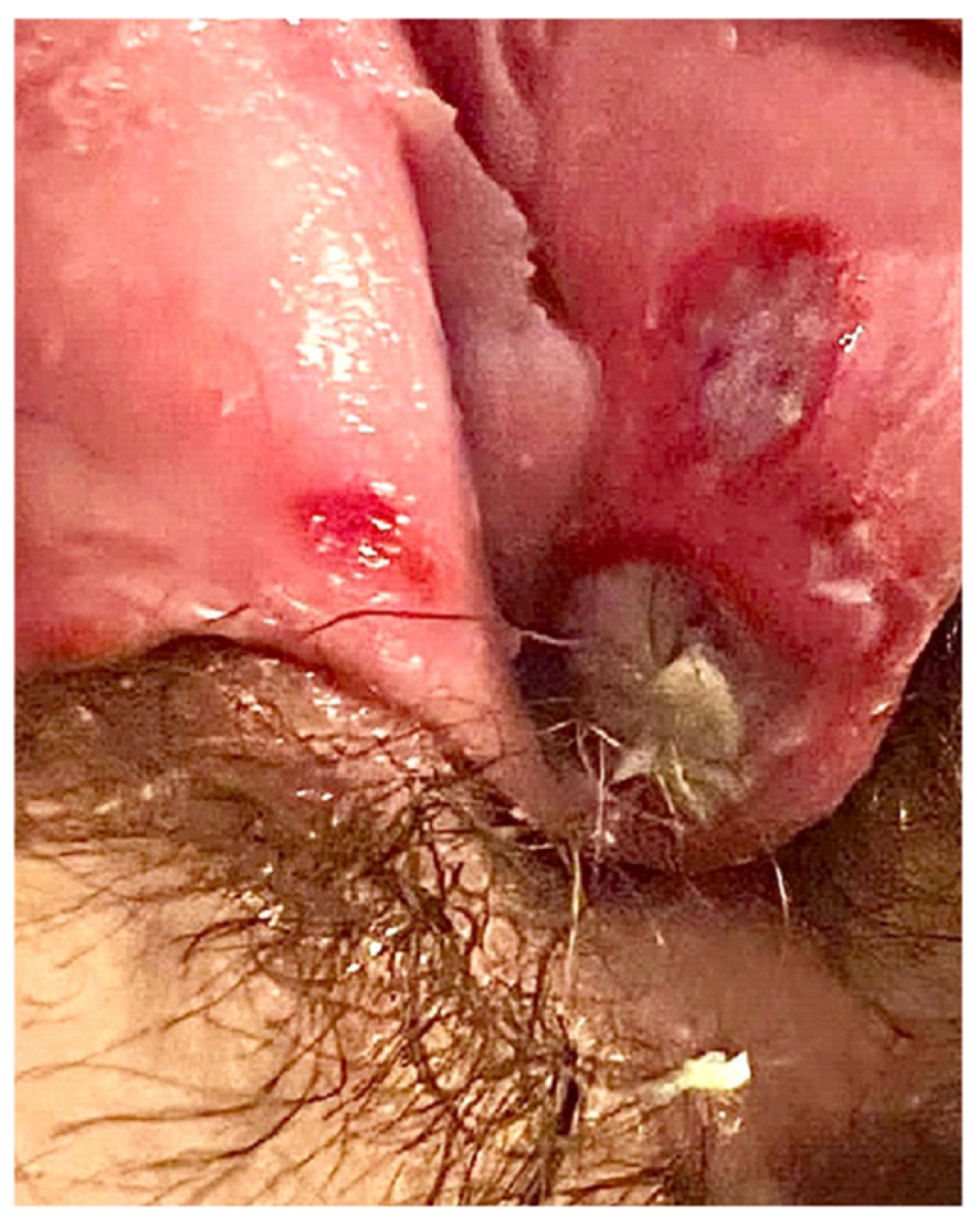
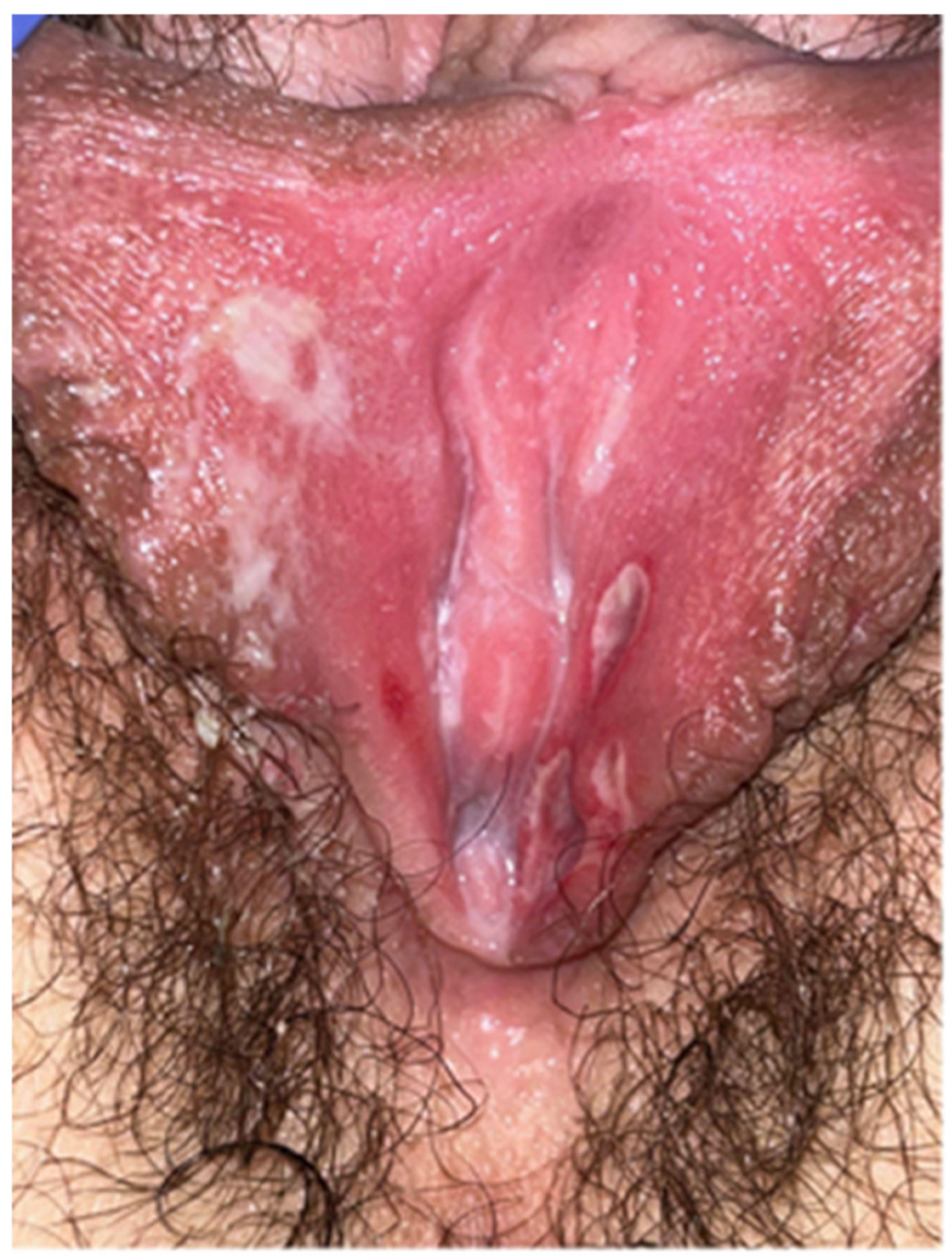
3.1. Case Report
3.2. Literature Review
- The ulcers predominantly affect adolescents; in fact, there are only two cases of older patients, aged 29 and 35 years old.
- The most common clinical manifestations are represented by flu-like symptoms and vulvar pain.
- If the ulcer is linked to the vaccine, it usually manifests within 48–72 h following the vaccination.
- As per protocol, all other possible infectious causes were excluded in each patient by means of serological and culture investigations.
- The resolution occurs spontaneously within 2–3 weeks.
- Since the treatment is purely symptomatic, in most cases the therapy consisted of a local anesthetic like lidocaine for topical use and oral NSAIDs to help control pain, sitz baths for greater intimate hygiene, and, in some cases, the use of topical corticosteroid. The use of the latter was observed in more complex cases and, above all, in cases in which the patient was particularly suffering for which there was a need for more aggressive treatment.
| Authors and Year | Median Age | Prior Sexual Activity | Symptoms | Oral Ulcer | Infectious Cause (HSV, CMV, EBV, HCV, HBV Mycoplasma pneumoniae, Toxoplasma gondii) | Previous Vaccination for COVID-19 | Previous or Concomitant SARS-CoV-2 | Treatment | Remission |
|---|---|---|---|---|---|---|---|---|---|
| Alberelli et al., 2021 [16] | 10 | No | Fever, vulvar pain | No | Negative. Toxoplasma not performed | Undeclared | Yes | Gentamicin cream and oral amoxicillin, ibuprofen as needed for pain | 2 weeks |
| Christl et al., 2021 [17] | 19 | Yes | Flu-like syndrome, vulvar pain | Yes | Not performed | Undeclared | Yes | Lidocaine for topical use, ibuprofen orally for pain control for 2 weeks, dexamethasone after for 1 week | 3 weeks |
| Drucker et al., 2022 [25] | 14 | Undeclared | Vulvar pain after the vaccine | No | HSV, CMV, EBV negative. Others not performed | Yes (Pfizer) | Negative | Lidocaine for topical use and sitz baths | 10 days |
| Frederiks et al., 2022 [23] | 12 | No | Constipation and vulvar pain 3 days after the vaccine | No | Negative. HBV, HCV not performed | Yes (Pfizer) | Not performed | Oral analgesia, laxatives, medium strength topical corticosteroid ointment | 10 days |
| González-Romero et al., 2022 [26] | 24 | No | Flu-like symptoms and vulvar pain after the vaccine | No | HSV, HBV, HCV, Mycoplasma negative. Toxoplasma, CMV, EBV not performed | Yes (AstraZeneca) | Not performed | Prednisone (30 mg) and oral analgesics | 3 weeks |
| Hsu et al., 2022 pz1 [24] | 12 | No | Dysuria, hematuria, flu-like symptoms 2 days after the vaccine | No | HSV and EBV negative. Others not performed | Yes (Pfizer) | Negative | Topical clobetasol 0.05%, topical lidocaine 2%, as needed oral acetaminophen and ibuprofen, and sitz baths | 2 weeks |
| Hsu et al., 2022 pz2 [24] | 14 | No | Vulvar pain caused by ulcers that arose 3 days after the vaccine | No | HSV and EBV negative. Others not performed | Yes (Pfizer) | Negative | Topical clobetasol 0.05%, topical lidocaine 2%, as needed oral acetaminophen and ibuprofen, and sitz baths | 4 weeks |
| Hsu et al., 2022 pz3 [24] | 29 | No | Flu-like symptoms 23 h after the vaccine | Yes | HSV, CMV, EBV negative. Others not performed | Yes (Moderna) | Negative | Topical clobetasol 0.005% | 2–4 weeks |
| Lucero Sangster-Carrasco et al., 2023 [27] | 33 | Yes | Vulvar pain and malaise 3 days after the vaccine | Yes | HSV, CMV, EBV, Toxoplasma negative. Mycoplasma, HCV, HBV not performed | Yes (AstraZeneca) | Undeclared | Oral antibiotics and oral NSAIDs | 2 weeks |
| Jacyntho et al., 2022 [18] | 35 | No | Progressive severe vulvar pain | No | Negative | Undeclared | Yes | Oral NSAIDs and oral corticosteroid | 1 week |
| Morais et al., 2022 [19] | 17 | Yes | Flu-like symptoms, itching, dysuria | No | HSV, mycoplasma CMV, EBV negative. Toxoplasma, HCV, HBV not performed | Undeclared | Yes | Oral NSAIDs and lidocaine for topical use | 2 weeks |
| Popatia et al., 2022 [28] | 12 | No | Fever 1 day after the vaccine | No | HSV negative. Others not performed | Yes (Pfizer) | Negative | Lidocaine 2% jelly, triamcinolone 0.1%, acetaminophen and ibuprofen for pain | 10 days |
| Sartor et al., 2023 pz1 [21] | 17 | Undeclared | Vulvar pain 2 days after the vaccine | Undeclared | HSV negative. Others not performed | Yes | Undeclared | Methylprednisolone taper, amoxicillin, lidocaine for topical use, NSAIDs, sitz baths | 20 days |
| Sartor et al., 2023 pz2 [21] | 14 | Undeclared | Vulvar pain 2 days the vaccine | Undeclared | HSV negative. Others not performed | Yes | Undeclared | Lidocaine for topical use, NSAIDs | 25 days |
| Sartor et al., 2023 pz3 [21] | 13 | Undeclared | Vulvar pain 2 days the vaccine | Undeclared | HSV negative. Others not performed | Yes | Undeclared | Oral prednisone, cephalexin, lidocaine for topical use, NSAIDs, sitz baths | 14 days |
| Sartor 2023 pz4 [21] | 12 | Undeclared | Vulvar pain 2 days the vaccine | Undeclared | CMV, EBV negative. Others not performed | Yes | Undeclared | Oral steroids | 21 days |
| Sartor 2023 pz5 [21] | 14 | Undeclared | Vulvar pain 2 days after the vaccine | Undeclared | Not performed | Yes | Undeclared | Undeclared | 21 days |
| Sartor 2023 pz6 [21] | 15 | Undeclared | Vulvar pain 2 days after the vaccine | Undeclared | EBV negative. Others not performed | Yes | Undeclared | Undeclared | 21 days |
| Sartor 2023 pz7 [21] | 16 | Undeclared | Vulvar pain 2 days after the vaccine | Undeclared | Not performed | Yes | Undeclared | Zinc oxide, NSAID, lidocaine for topical use | 10 days |
| Sartor 2023 pz8 [21] | 16 | Undeclared | Vulvar pain 2 days after the vaccine | Undeclared | Not performed | Yes | Undeclared | Zinc oxide, NSAID, lidocaine for topical use | 10 days |
| Schmitt 2023 [20] | 15 | No | Nausea, headache, fever of 38.3 °C, vulvar pain | No | HSV, CMV, EBV negative. Others not performed | No | Yes | Alternating paracetamol or ibuprofen and cold compresses | 14 days |
| Scott 2022 [22] | 15 | No | Vaginal pain and sores, 3 days after the vaccine | No | Not performed | Yes (Pfizer) | Not performed | Sitz baths, oral pain relievers, topical anesthetics, topical steroids | 1 week |
| Wojcicki 2022 [9] | 16 | No | Vaginal pain, myalgia, and fatigue 2 days after the vaccine | Yes | HSV, CMV, EBV negative. Others not performed | Yes (Pfizer) | Negative | Topical lidocaine, ibuprofen and paracetamol for pain control, topical Clobetasol, norethindrone (5 mg) for menstrual suppression while ulcers heal | 14 days |
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cheng, S.X.; Chapman, M.S.; Margesson, L.J.; Birenbaum, D. Genital ulcers caused by Epstein- Barr virus. J. Am. Acad. Dermatol. 2004, 51, 824–826. [Google Scholar] [CrossRef]
- Delgado-García, S.; Palacios-Marqués, A.; Martínez-Escoriza, J.C.; Martín-Bayón, T.A. Acute genital ulcers. BMJ Case Rep. 2014, 2014, bcr2013202504. [Google Scholar] [CrossRef] [PubMed]
- Mourinha, V.; Costa, S.; Urzal, C.; Guerreiro, F. Lipschütz ulcers: Uncommon diagnosis of vulvar ulcerations. Case Rep. 2016, 2016, bcr2015214338. [Google Scholar] [CrossRef] [PubMed]
- Vieira-Baptista, P.; Lima-Silva, J.; Beires, J.; Martinez-de-Oliveira, J. Lipschütz ulcers: Should we rethink this? An analysis of 33 cases. Eur. J. Obstet. Gynecol. Reprod. Biol. 2016, 198, 149–152. [Google Scholar] [CrossRef]
- Sadoghi, B.; Stary, G.; Wolf, P.; Komericki, P. Ulcus vulvae acutum Lipschütz: A systematic literature review and a diagnostic and therapeutic algorithm. J. Eur. Acad. Dermatol. Venereol. 2020, 34, 1432–1439. [Google Scholar] [CrossRef] [PubMed]
- Moise, A.; Nervo, P.; Doyen, J.; Kridelka, F.; Maquet, J.; Vandenbossche, G. Ulcer of Lipschutz, a rare and unknown cause of genital ulceration Facts. Views Vision ObGyn 2018, 10, 55–57. [Google Scholar]
- Huppert, J.S. Lipschutz ulcers: Evaluation and management of acute genital ulcers in women. Dermatol. Ther. 2010, 23, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Sárdy, M.; Wollenberg, A.; Niedermeier, A.; Flaig, M. Genital ulcers associated with Epstein-Barr virus infection (ulcus vulvae acutum). Acta Derm. Venereol. 2011, 91, 55–59. [Google Scholar] [CrossRef] [PubMed]
- Wojcicki, A.V.; O’Brien, K.L. Vulvar aphthous ulcer in an adolescent after Pfizer-BioNTech (BNT162b2) COVID-19 vaccination. J. Pediatr. Adolesc. Gynecol. 2022, 35, 167–170. [Google Scholar] [CrossRef]
- Barrett, M.M.; Sangüeza, M.; Werner, B.; Kutzner, H.; Carlson, J.A. Lymphocytic arteritis in Epstein-Barr virus vulvar ulceration (Lipschütz disease): A report of 7 cases. Am. J. Dermatopathol. 2015, 37, 691–698. [Google Scholar] [CrossRef]
- Brinca, A.; Canelas, M.M.; Carvalho, M.J.; Vieira, R.; Figueiredo, A. Lipschütz ulcer (ulcus vulvae acutum): A rare cause of genital lesion. An. Bras. Dermatol. 2012, 87, 622–624. [Google Scholar] [CrossRef] [PubMed]
- Truchuelo, M.T.; Vano-Galván, S.; Alcántara, J.; Pérez, B.; Jaén, P. Lipschütz ulcers in twin sisters. Pediatr. Dermatol. 2012, 29, 370–372. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.; Drake, S.M.; Dedicoat, M.; Wood, M.J. Genital ulcers associated with acute Epstein- Barr virus infection. Sex Transm. Infect 1998, 74, 296–297. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. (Eds.) Cochrane Handbook for Systematic Reviews of Interventions Version 6.3 (Updated February 2022); Cochrane: London, UK, 2022; Available online: www.training.cochrane.org/handbook (accessed on 9 July 2023).
- Higgins, J.P.T.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. (Eds.) Cochrane Handbook for Systematic Reviews of Interventions, 2nd ed.; John Wiley&Sons: Chichester, UK, 2019. [Google Scholar]
- Alberelli, M.C.; Panariello, A.; Olivi, M.; Borghi, A.; Zedde, P.; Corazza, M. Ulcus vulvae acutum and SARS-CoV-2: An aetiological role? J. Eur. Acad. Dermatol. Venereol. 2021, 35, e647–e649. [Google Scholar] [CrossRef]
- Christl, J.; Alaniz, V.I.; Appiah, L.; Buyers, E.; Scott, S.; Huguelet, P.S. Vulvar Aphthous Ulcer in an Adolescent With COVID-19. J. Pediatr. Adolesc. Gynecol. 2021, 34, 418–420. [Google Scholar] [CrossRef]
- Jacyntho, C.M.A.; Lacerda, M.I.; Carvalho, M.S.R.; Ramos, M.R.M.S.; Vieira-Baptista, P.; Bandeira, S.H.A.D. COVID-19 related acute genital ulcer: A case report. Einstein 2022, 20, eRC6541. [Google Scholar] [CrossRef]
- Morais, M.L.; Moura, M.; Moreira, A. Lipschütz’s ulcers in an adolescent with SARS-CoV2 infection. Clin. Case Rep. 2022, 10, e6503. [Google Scholar] [CrossRef]
- Schmitt, T.M.; Devries, J.; Ohns, M.J. Lipschutz Ulcers in an Adolescent After SARS-CoV-2 Infection. J. Pediatr. Health Care 2023, 37, 63–66. [Google Scholar] [CrossRef]
- Sartor, R.A.; Lawson, A.; Moncada-Madrazo, M.; Altchek, C.; Vash-Margita, A.; Cron, J. Vulvar Aphthous Ulcers in Perimenarchal Adolescents after COVID-19 Vaccination: A Multicenter Case Series. J. Pediatr. Adolesc. Gynecol. 2023, 36, 268–272. [Google Scholar] [CrossRef]
- Scott, S.M.; Alaniz, V.; Appiah, L.; Buyers, E.; Holton, C.; Huguelet, P. Vulvar Aphthous Ulcers in an Adolescent After Coronavirus Disease 2019 (COVID-19) Vaccination. Obstet. Gynecol. 2022, 140, 514–517. [Google Scholar] [CrossRef]
- Frederiks, A.J.; Foster, R.S.; Ricciardo, B. Lipschütz ulceration in a 12-year-old girl following second dose of Comirnaty (Pfizer) COVID-19 vaccine. Int. J. Womens Dermatol. 2022, 8, e066. [Google Scholar] [PubMed]
- Hsu, T.; Sink, J.R.; Alaniz, V.I.; Zheng, L.; Mancini, A.J. Acute Genital Ulceration After Severe Acute Respiratory Syndrome Coronavirus 2 Vaccination and Infection. J. Pediatr. 2022, 246, 271–273. [Google Scholar] [CrossRef] [PubMed]
- Drucker, A.; Corrao, K.; Gandy, M. Vulvar Aphthous Ulcer Following Pfizer-BioNTech COVID-19 Vaccine—A Case Report. J. Pediatr. Adolesc. Gynecol. 2022, 35, 165–166. [Google Scholar] [CrossRef] [PubMed]
- González-Romero, N.; Morillo Montañes, V.; Vicente Sánchez, I.; García García, M. Úlceras de Lipschütz tras la vacuna frente a la COVID-19 de AstraZeneca [Lipschütz Ulcers after the AstraZeneca COVID-19 Vaccine]. Actas. Dermosifiliogr. 2022, 113, S29–S31. [Google Scholar] [CrossRef]
- Sangster-Carrasco, L.; Paz-Temoche, R.; Coronado-Arroyo, J.; Concepción-Zavaleta, M.; Roseboom, P.; Concepción-Urteaga, L.; Zavaleta-Gutiérrez, F. Lipschütz acute vulvar ulcer related to COVID-19 vaccination: First case report in South America. Medwave 2023, 23, e2674. [Google Scholar] [CrossRef] [PubMed]
- Popatia, S.; Chiu, Y.E. Vulvar aphthous ulcer after COVID-19 vaccination. Pediatr. Dermatol. 2022, 39, 153–154. [Google Scholar] [CrossRef]
- Lande, M.B.; Mowry, J.A.; Houghton, D.C.; White, C.R., Jr.; Borzy, M.S. Immune complex disease associated with Epstein-Barr virus infectious mononucleosis. Pediatr. Nephrol. 1998, 12, 651–653. [Google Scholar] [CrossRef]
- Wands, J.R.; Perrotto, J.L.; Isselbacher, K.J. Circulating immune complexes and complement sequence activation in infectious mononucleosis. Am. J. Med. 1976, 60, 269–272. [Google Scholar] [CrossRef]
- Magro, C.; Mulvey, J.J.; Berlin, D.; Nuovo, G.; Salvatore, S.; Harp, J.; Baxter-Stoltzfus, A.; Laurence, J. Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID-19 infection: A report of five cases. Transl. Res. 2020, 220, 1–13. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Merlino, L.; Volpicelli, A.I.; Dominoni, M.; Pasquali, M.F.; D’Ovidio, G.; Gardella, B.; Senatori, R. Lipschütz Ulcer and SARS-CoV-2: What We Currently Know? Diseases 2023, 11, 121. https://doi.org/10.3390/diseases11030121
Merlino L, Volpicelli AI, Dominoni M, Pasquali MF, D’Ovidio G, Gardella B, Senatori R. Lipschütz Ulcer and SARS-CoV-2: What We Currently Know? Diseases. 2023; 11(3):121. https://doi.org/10.3390/diseases11030121
Chicago/Turabian StyleMerlino, Lucia, Agnese Immacolata Volpicelli, Mattia Dominoni, Marianna Francesca Pasquali, Giulia D’Ovidio, Barbara Gardella, and Roberto Senatori. 2023. "Lipschütz Ulcer and SARS-CoV-2: What We Currently Know?" Diseases 11, no. 3: 121. https://doi.org/10.3390/diseases11030121
APA StyleMerlino, L., Volpicelli, A. I., Dominoni, M., Pasquali, M. F., D’Ovidio, G., Gardella, B., & Senatori, R. (2023). Lipschütz Ulcer and SARS-CoV-2: What We Currently Know? Diseases, 11(3), 121. https://doi.org/10.3390/diseases11030121






