Abstract
This study investigated the effect of Callistemon citrinus flower extract (C.E.) on the structural and functional properties of chitosan-based bioplastics for food packaging applications. Increasing C.E. concentration produced changes in colour and opacity, with reduced L values and higher a values at 5% C.E., resulting in a reddish appearance. Film opacity increased from 2.49% ± 0.10 for control to 13.60% ± 0.72 for film with 5% C.E., reducing transparency. The addition of 5% C.E. improved oxygen barrier properties, reducing O2 permeability compared to the control that was similar to Low-density polyethylene (LDPE). A qualitative antimicrobial screening showed that 5% C.E. effectively inhibited S. aureus (16 mm) and L. monocytogenes (12 mm), while no inhibition was detected against E. coli. Cheddar cheese packaged in films with C.E. exhibited higher weight loss (11.63% at 28 days) than LDPE (2.90%), whereas moisture retention remained moderate (76.62% for the film with 5% C.E.). Oxidative stability improved with lower lipid oxidation (K232 = 2.90 at 28 days) in cheese wrapped in C.E. films. Consumer evaluation indicated a positive perception, with 73% of participants willing to purchase products in C.E.-packaged films. Overall, C.E. enhanced antioxidant and antibacterial benefits, but further optimization may be required to improve moisture retention.
1. Introduction
The growing global demand for sustainable and eco-friendly materials has catalyzed innovation in the field of food packaging. Traditional petroleum-based plastics, widely used in the food industry, have been long associated with environmental challenges such as pollution and non-biodegradability. These concerns, together with increasing awareness of food waste, have driven the exploration of alternative materials [1]. Among these, bioplastics have emerged as a promising solution, offering the potential to reduce the environmental footprint of food packaging while maintaining or enhancing food protection [2]. Food packaging plays a vital role in ensuring food safety, extending shelf life and minimizing waste. To be effective, packaging must provide a barrier against moisture, gases and microbial contamination while remaining environmentally sustainable. Chitosan, a biopolymer obtained through the deacetylation of naturally occurring chitin, has gained significant attention due to its excellent film-forming ability, biodegradability and inherent antimicrobial activity. Its versatility and capacity to form flexible and transparent films makes it particularly suitable for food packaging applications, where it can act as a barrier to oxygen and water vapour, thereby helping to preserve the quality and safety of food products [3].
Recent advancements in bioplastic technology have focused on enhancing the functional properties of chitosan films by incorporating natural additives or bioactive compounds [4]. Several studies have demonstrated the effectiveness of chitosan-based films enriched with floral or polyphenolic extracts in improving the preservation of hard cheeses, often showing enhanced oxidative stability and microbial protection [4,5,6,7]. The flower extract of Callistemon citrinus, commonly known as the bottlebrush plant, is rich in bioactive compounds, including flavonoids, phenolic acids, and essential oils. These compounds possess strong antioxidant and antimicrobial properties (Laganà et al., 2020 [8]) making the extract particularly attractive for food packaging applications. By integrating Callistemon citrinus flower extract into chitosan films, we have developed packaging materials that in addition to protecting food from environmental and microbial factors, also potentially extend the shelf life of packaged products. The incorporation of Callistemon citrinus flower extract into chitosan-based films offers multiple benefits. The natural antimicrobial properties of the extract can help inhibit the growth of foodborne pathogens, reducing the risk of contamination and spoilage [9]. Additionally, its antioxidant activity can mitigate oxidative processes that lead to the degradation of fats and other sensitive components in food [10]. These functionalities are particularly advantageous for the storage of products like cheddar cheese, which are susceptible to quality deterioration over time. Cheddar cheese, a high-value dairy product, requires packaging that can maintain its texture, flavour, and nutritional value while preventing contamination and moisture loss. The use of bioplastics enriched with natural bioactive compounds might represent an innovative approach to addressing these challenges [11]. Moreover, the integration of Callistemon citrinus extract aligns with the principles of green chemistry and sustainability [12]. The extract is derived from a renewable plant source, and its use in combination with biodegradable chitosan supports the development of environmentally friendly packaging materials [13,14]. This approach not only reduces reliance on synthetic additives and plastics but also leverages the multifunctional benefits of natural compounds. Such innovations contribute to a circular economy model in which materials are designed to be reused, recycled, or biodegraded, minimizing their impact on the environment.
In this study, the development and characterization of chitosan-based films enriched with Callistemon citrinus flower extract are explored, with a particular focus on their application for cheddar cheese packaging. The goal was to evaluate the physicochemical, barrier, and functional properties of these films and to assess their effectiveness in preserving cheese quality and extending shelf life. This research contributes to the growing body of knowledge on bioplastic food packaging and highlights the potential of combining natural polymers and plant-derived bioactive compounds to create sustainable, functional materials for the food industry. Through testing and optimization of bioplastic formulations, we aspire to contribute to the development of sustainable and effective food packaging solutions that prioritize both food safety and environmental sustainability.
2. Materials and Methods
2.1. Materials
Low-molecular weight (50,000–190,000 Da) chitosan was supplied by Sigma Aldrich (St. Louis, MO, USA) and glycerol from Carlo Erba (Milan, Italy). Nutrient agar, phosphate-buffered saline (PBS), phenolphthalein, sodium hydroxide and ethanol were from Sigma-Aldrich (St. Louis, MO, USA). McFarland equivalence turbidity standards and ethyl acetate were obtained from Thermo Fisher Scientific (Waltham, MA, USA). Grated cheddar cheese was purchased from a local grocery store in Reading (UK). Callistemon citrinus extract was provided by the University of Messina. Its extraction procedure, total phenolic content, antioxidant capacity, and phytochemical profile (including HPLC-MS analysis) have been fully characterized in our previous work [13]. The same batch of extract was used for all experiments to ensure reproducibility. Low-density polyethylene (LDPE) was obtained from a local supermarket.
2.2. Production of Bioplastics
Film-forming solutions (FFSs) were prepared by dissolving a 2% w/w low-molecular weight chitosan stock solution chitosan in 1% (v/v) acetic acid to obtain a 2% (w/w) polymer solution. The dispersion was stirred at 600 rpm for 4 h at room temperature to ensure complete hydration of the polymer. The pH of the solution was then adjusted to 2.5 using 1 M acetic acid to promote full protonation of chitosan and facilitate dissolution. Once a clear and homogeneous chitosan solution was obtained, it was incorporated at 30% (w/v) relative to the chitosan content and mixed for an additional 20 min. No phase separation was observed following glycerol addition. The acidic environment ensured good compatibility between chitosan and glycerol, resulting in a stable and homogeneous FFS. Subsequently, concentrations of C.E. (1.25, 2.5, and 5% w/w), previously dissolved in water using ultrasound, were added to the chitosan–glycerol solution. The mixture was stirred continuously for 20 min and cast onto a Coatmaster surface and left to air dry at room temperature for 16 h. Once dried, the A4-sized films were carefully peeled off from the surface (Figure 1). In all experiments, a control film was prepared using the same chitosan–glycerol formulation described above but without the addition of Callistemon citrinus extract. This control served as the reference material for evaluating the effect of the extract on the structural, optical, barrier, and antimicrobial properties of the films.

Figure 1.
Chitosan-based bioplastics with 30% of glycerol without and with different concentrations of C.E. (a) Control film; (b) film with 1.25% C.E.; (c) film with 2.5% C.E.; (d) film with 5% C.E. w/w of chitosan.
2.3. Colour, Opacity and Transparency Parameters of Films
The colour parameters (CIE-Lab scale) of the films were determined using a colorimeter (Chroma Metre CR-400, Konica Minolta, Tokyo, Japan). Luminosity (L), red–green (a), and yellow–blue (b) parameters were calculated. L value, also known as lightness, ranges from 0 (black) to 100 (white) [15]. The transparency and opacity of the films, which indicated the amount of light that can pass through the material, were measured with the BIORAD SmartSpec 3000 spectrophotometer (Bio-Rad Laboratories, Hercules, CA, USA) [16]. The films were cut in strips of 3 cm of length and 1 cm of width, and the thickness of each strip was measured in five points. The opacity was measured for each sample by the following equation [17]:
where A600 is the absorbance at 600 nm, and the thickness is the average of 5 points in mm.
Opacity (%) = [A600/thickness] × 100
The transparency was calculated as follows:
where T% is the percent transmittance of light at 600 nm and thickness is the average of five points in mm. All measurements were performed in triplicate.
Transparency (T%) = log (%T600)/thickness
2.4. Film Permeability Towards Oxygen
Oxygen permeability (O2P) of the control and 5% C.E. was measured according to Giosafatto et al. 2014, [18] and Mirpoor et al. 2024 [19] to evaluate their ability to act as a barrier against oxygen. Intermediate concentrations (1.25% and 2.5%) were not analyzed because preliminary screening showed no measurable differences compared to the control, whereas the 5% C.E. film represented the formulation with the highest functional impact. As reference material, low-density polyethylene (LDPE) was used. The measurements were performed in duplicate for each film (50% RH, 25 °C) as recommended for the Total Perm apparatus (ExtraSolution s.r.l., Pisa, Italy) according to a Standard Method (ASTM D3985–05, 2010 [20]), which provides highly reproducible readings with minimal instrumental variability. The two replicates showed negligible deviation, confirming the robustness of the measurement.
2.5. Antimicrobial Activity of C.E.
The antimicrobial properties of FFSs were assessed against representative Gram-positive and Gram-negative foodborne pathogens. Listeria monocytogenes 1043S and Staphylococcus aureus NCTC 8532 were used as Gram-positive strains, while Escherichia coli ATCC 25922 and Pseudomonas aeruginosa NCTC 10322 represented Gram-negative bacteria. Each strain was cultured on nutrient agar at 37 °C for 18 h, and a single colony was suspended in PBS. The turbidity was adjusted to match a 0.5 McFarland standard (≈1.5 × 108 CFU/mL) [21]. Antimicrobial activity was evaluated using a disc diffusion assay following CLSI guidelines [22]. Sterile paper discs (10 mm) were immersed in the corresponding FFSs, dried under sterile conditions, and placed on agar plates previously inoculated with the bacterial suspensions. Mueller–Hinton agar was used for all strains except L. monocytogenes, which was plated on Tryptic Soy agar. After incubation at 37 °C for 18–24 h, inhibition zones were measured, subtracting the disc diameter [23]. The disc diffusion assay was performed as a qualitative screening test to confirm the antimicrobial activity of the previously characterized extract [13]. The assay was not intended to provide quantitative inhibition data; therefore, antibiotic and solvent controls were not included.
2.6. Application of the Films for Food Packaging
Approximately 10 g of grated cheddar cheese, purchased from a local grocery store in Reading (UK), were packed in each film, following the casting of the FFS. LDPE served as the commercial reference packaging. All the samples were stored at 4 °C for 28 days. Samples were analyzed for physicochemical changes, including weight loss, moisture content, water activity, pH, titratable acidity, lipid oxidation and colour at predetermined intervals (days 0, 7, 14, 21, 28) [24].
2.7. Chemical Analysis of Grated Cheddar Cheese
2.7.1. Weight Loss (WL), Moisture Content (MC) and Water Activity (WA)
The weight loss (WL) of cheddar cheese during storage was determined by recording the mass of each cheese sample at every storage interval and expressing the change as a percentage of the initial weight. Moisture content (MC) was measured using the oven-drying method: samples were dried at 105 °C for 24 h, and moisture was calculated from the difference between initial and final weight. Water activity (WA) was measured at 20 °C using a water activity metre (Hygrolab Rotronic, Process Sensing Technology, Bassersdorf, Switzerland) [24].
2.7.2. pH and Titratable Acidity
Cheese samples (4 g) were homogenized with 20 mL of distilled water using a T25 digital Ultra-Turrax (IKA, Staufen, Germany) at 10,000 rpm for 30 s [25]. The homogenate was centrifuged at 8000 g for 10 min, and the pH of the supernatant was measured using a pH metre (Orion, Boston, MA, USA). Titratable acidity was determined by titrating 10 mL of the supernatant with 0.1 M NaOH solution until reaching the end point of titration at pH 8.3, using phenolphthalein as an indicator. Results were reported as the percentage of lactic acid and calculated using the following equation:
where V is the amount of 0.1 N NaOH consumed in titration step (mL); m is the amount of the cheese sample (g); 0.009 is the milli-equivalent weight of lactic acid (g).
2.7.3. Lipid Extraction and Oxidative Stability
Lipids were extracted following a solvent-based method using an ethyl acetate–ethanol mixture (2:1 v/v) [26]. Two extraction cycles were performed, and the combined filtrates were evaporated under reduced pressure at 40 °C. The dried lipid fraction was weighed to calculate extraction yield, according to the following equation. All analyses were conducted in triplicate and each for 7 days for all the samples.
Yield (%) = (total extracted weight/cheese weight) × 100
Oxidative stability was assessed by determining the K232 and K270 values [27], which are commonly used indicators to assess lipid oxidation in food products. These parameters are widely used indicators of primary and secondary lipid oxidation, respectively, and are commonly applied in the quality evaluation of edible oils. Specifically, K232 reflects the formation of conjugated dienes generated during the early stages of oxidation, whereas K270 is associated with conjugated trienes and secondary oxidation products such as aldehydes and ketones. Briefly, 10 mg of oil were dissolved with 5 mL of hexane and the absorbance at 232 and 270 nm was then measured using the same UV/Vis spectrophotometer and the K232 and K270 values were calculated as follows:
where Eλ is the absorbance of the solution at 232 and 270 nm, [c] is the concentration of oil in g/100 mL, and l is the path length of the quartz cuvette.
Kλ = Eλ/([c] × l)
2.7.4. Visual Assessment and Colour Parameters of Cheese
Visual assessment of the cheese samples was carried out by taking digital images of the samples at different storage times using a lightbox with LED lights placed on both sides of the box. For colour measurement, the cheese sample (30 mm × 30 mm) was placed on a surface, and the CIELAB colour parameters (L-lightness, a-redness, and b-yellowness) on the sample surface were monitored using a LabScan XE colorimeter (HunterLab, Reston, VA, USA). The colour change (ΔE) between the cheese samples at day 0 and storage times was expressed (Deng et al., 2017 [28]) according to the following equation:
2.8. Panel Test
Consumers (n = 50) were recruited through an electronic announcement within the University community and consumer liking, and a preference test was conducted (Lignou et al., 2021 [29]). The panel consisted of 71% females and 29% males with a mean age of 31 years old, ranging from 18 to 68 years old. Participants evaluated the visual appearance of bioplastics. Each consumer was presented with two samples: a transparent control film and the film with 5% of C.E. that had a reddish colour. Consumers rated the samples for appearance, transparency and perceived quality using a five-point scale with extremes of 1 (dislike extremely) and 5 (like extremely). After the evaluation of each film, participants were also asked whether they would consider purchasing a product packaged in that material, based on their prior experience and expectations. The evaluation involved only non-invasive visual assessment, with no collection of personal or sensitive data and no physical or psychological intervention. In accordance with institutional guidelines, this activity is exempt from formal ethical approval. All participants were adults (≥18 years) and provided informed consent prior to participation.
2.9. Statistical Analysis
All statistical analyses were carried out using SPSS software (version 29, SPSS Inc., Chicago, IL, USA). For longitudinal measurements collected over storage time (weight loss, moisture content, water activity, pH, titratable acidity, lipid oxidation, and colour parameters), a two-way ANOVA model was applied with packaging type and storage time as fixed factors, including their interaction. When significant main or interaction effects were observed (p < 0.05), Duncan’s multiple range test was used for post hoc comparisons. All measurements were performed in triplicate.
3. Results
3.1. Colour and Opacity Properties of Film Functionalized with C.E.
The results in Table 1 indicated significant changes in the colour properties (L, a, b) of the bioplastic as the concentration of C.E. increased. The control film (prepared without C.E.) had a high lightness value. As the concentration of C.E. increased, the L value decreased, with the film becoming progressively darker. As C.E. is added, the a value increased, indicating a shift towards a red hue. For the film with 5% C.E., the a value was 31.28 ± 0.71, representing a strong red coloration. The control film had a yellow tint, and this yellowish hue became more pronounced with the addition of C.E. These results showed that the addition of C.E. significantly affects the colour of the bioplastic films, making them reddish as the concentration of C.E. increased.

Table 1.
Colorimetric (L, a, b), opacity and transparency value of films with different concentrations of C.E.
The results in Table 1 clearly show that the opacity of the films increased as the concentration of C.E. increased. The control film, without C.E., had a low opacity of 2.49% ± 0.10. As the concentration of C.E. increased, the opacity of the films also increased significantly (p < 0.05), reaching a maximum value of 13.60% ± 0.72. The progressive darkening of the films with increasing C.E. concentration could be attributed to the high content of phenolic compounds and flavonoids naturally present in Callistemon citrinus flowers. These molecules exhibit intrinsic chromophoric properties, absorbing light in the visible range and imparting a reddish coloration to the polymer matrix. The increase in the a and b values is therefore consistent with the chemical profile of the extract and reflects its uniform incorporation within the chitosan network. The incorporation of C.E. led to a significant reduction in transparency compared to the control film, with no significant differences among the three concentrations tested. This indicates that even low levels of extract were sufficient to decrease light transmittance, likely due to the presence of light-absorbing compounds within the polymer matrix. Phenolic compounds could interact with chitosan chains through hydrogen bonding, creating localized regions with different refractive indices. These heterogeneities reduce light transmittance and contribute to the overall opacity of the films. Similar behaviour has been reported for other plant-derived additives incorporated into polysaccharide matrices, where pigment-rich extracts significantly alter optical clarity ([17,30]). From a functional perspective, the reduced transparency may be advantageous for certain food packaging applications. Lower light transmission could protect photosensitive foods from oxidative degradation, particularly dairy products where lipid oxidation is light-induced. However, the esthetic impact of reddish coloration must be considered depending on the target product and consumer expectations. Overall, the observed optical modifications reflect both the chemical nature of the extract and its interactions with the polymer matrix, providing insight into how natural additives can tailor the visual and functional properties of biopolymer films.
3.2. Oxygen Permeability (O2P) of Films
The results in Table 2 showed the oxygen permeability (O2P) of different samples. The LDPE (low-density polyethylene) film had an O2P of 3.82 ± 0.03 cm3 mm m−2 d−1 kPa−1, which did not differ significantly from the control chitosan film. This indicates that both materials displayed comparable oxygen barrier performance under the tested conditions. In contrast, the film with 5% C.E. showed a markedly lower O2P value, indicating that the addition of C.E. greatly enhanced the film’s barrier properties against oxygen. This reduction suggests that the incorporation of C.E. contributed to a more tortuous diffusion pathway within the polymer matrix, thereby limiting oxygen transmission through the film.

Table 2.
Oxygen permeability (O2P of films with and without CE. LDPE as a reference material).
Water vapour permeability (WVP) measurements were not included in this study, as they were comprehensively reported in our earlier publication on the same films [13,14]. In that work, the WVP of 5% C.E.-functionalized films was reported as 13.64 ± 0.52 g·mm/m2·day·kPa, confirming its substantially higher WVP compared to LDPE (0.06 ± 0.01 g·mm/m2·day·kPa), consistent with the hydrophilic character of chitosan and the extract. Therefore, the present study focuses on oxygen permeability as the key barrier parameter relevant to oxidative stability in cheese packaging. Overall, these findings highlight the potential of using flower extract to improve the oxygen barrier properties of chitosan-based films, making them more suitable for applications where reduced oxygen permeability is desired, such as in food packaging to extend shelf life ([31,32]).
3.3. Antibacterial Activity of Extract-Containing FFSs
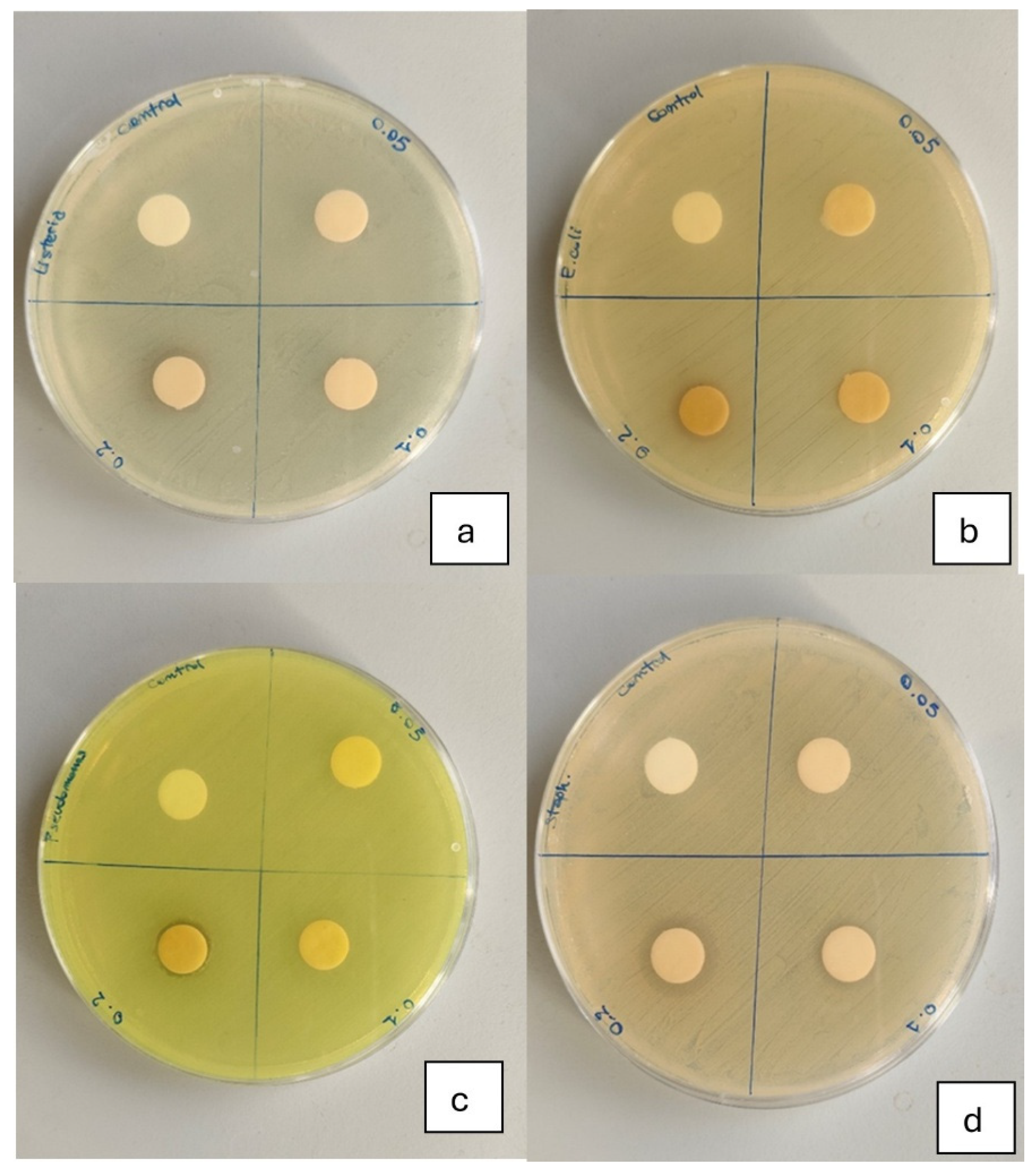
The antibacterial activity of the FFSs is presented in Figure 2 and summarized in Table 3. No inhibition zones were observed for the control FFS or for any formulation against E. coli. The FFS enriched with 5% C.E. showed the strongest antibacterial effect, with inhibition zones of 16 mm for S. aureus, 12 mm for L. monocytogenes, and 12 mm for P. aeruginosa (Table 3). The 2.5% C.E. formulation also showed measurable activity against S. aureus (11 mm), although significantly lower than the 5% C.E. sample (p < 0.05). These results confirm that C.E. possesses selective antibacterial properties, with a pronounced effect on Gram-positive bacteria, particularly S. aureus, in line with previous findings [33,34]. Conversely, no inhibition effect was detected against E. coli, highlighting the strain-dependent nature of C.E.’s antimicrobial action, which is also reported by Seyydnejad et al. [35]. The study supports the potential of C.E. as a natural antibacterial agent for active food packaging applications. It is important to note that this assay represents a qualitative screening of antimicrobial potential rather than a quantitative evaluation.
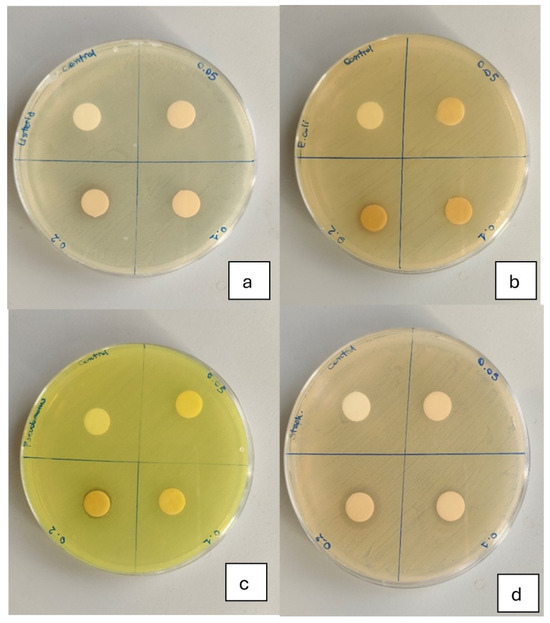
Figure 2.
Inhibition of bacterial growth by different concentrations of C.E. against (a) Listeria monocytogenes, (b) Escherichia coli, (c) Pseudomonas aeruginosa and (d) Staphylococcus aureus. Each Petri dish is divided into four quadrants. From left to right, the quadrants contain discs impregnated with increasing concentrations of C.E.: the first quadrant corresponds to the control without extract, followed by samples containing 1.25%, 2.5%, and 5% C.E., respectively. The inhibition zones around the discs indicate the antimicrobial activity at each tested concentration.

Table 3.
Inhibition zone diameters (mm) of FFSs containing different concentrations of C.E. against selected bacterial strains.
The antibacterial results indicate that the FFSs enriched with C.E. were effective only against Gram-positive bacteria, a trend that reflects the intrinsic antimicrobial profile of C.E. Phenolic compounds and flavonoids—major constituents of Callistemon citrinus extracts—are known to interact preferentially with the thick peptidoglycan layer of Gram-positive bacteria, disrupting membrane integrity and inducing leakage of intracellular components [36,37]. In contrast, Gram-negative bacteria possess an outer membrane rich in lipopolysaccharides, which acts as an additional permeability barrier and reduces the susceptibility to plant-derived phenolics [38]. This structural difference explains the absence of inhibition against E. coli and the pronounced activity observed against S. aureus and L. monocytogenes. From an application standpoint, this selective antibacterial spectrum does not limit the suitability of C.E. for active food packaging. In dairy products such as cheddar cheese, Gram-positive bacteria—including Listeria monocytogenes, Staphylococcus aureus, and spoilage lactic acid bacteria—represent the most relevant microbiological hazards. Therefore, the ability of C.E.-functionalized films to inhibit Gram-positive strains is particularly advantageous for this type of food matrix. The results support the potential of C.E. as a natural antimicrobial agent capable of enhancing the microbiological stability of cheese during storage, even though its activity does not extend to Gram-negative bacteria [39].
3.4. Food Packaging Applications
Since no microbiological enumeration was performed on cheese samples, the antimicrobial effect of the films in the food matrix can only be inferred as potential. Based on this consideration, the control film and film with 5% of C.E. were selected for food packaging applications, as the 5% C.E. formulation exhibited the highest antimicrobial potential in the qualitative screening assay and demonstrated superior technological performance, including improved mechanical and barrier properties [13]. Specifically, the film with 5% C.E. exhibited the highest heat seal strength, which is crucial for maintaining packaging integrity during storage, as previously demonstrated by Avitabile et al., 2024 [13]. Additionally, the film with 5% C.E. demonstrated good moisture barrier properties compared to the control, contributing to the preservation of product quality and shelf life, which are key requirements in food packaging application. These functional attributes, combined with its visually acceptable appearance as confirmed by consumer evaluation in this study (see below), justify the chosen selection for further testing. The need for such features for food packaging has also been reported by Costa et al. (2018) [40], which highlighted the importance of mechanical strength and barrier properties in maintaining cheese safety and quality. The visual assessment of the samples was carried out by taking digital images of the grated cheddar cheese at each storage time using the lightbox with LED lights placed on either sides of the box as a light source (Figure 3). This method ensured uniform and controlled lighting conditions for accurate visual comparison. Microbiological assessment during cheese storage was limited to visual inspection of surface spoilage under controlled conditions. Quantitative microbial counts were not performed, as the aim of this study was to evaluate packaging performance rather than to characterize microbial dynamics.

Figure 3.
Grated cheddar cheese sealed in LDPE, chitosan-based film with 30% glycerol as control and chitosan-based film +5% C.E. samples (from left to right) on day 0, 7, 14, 21 and 28.
As shown in Figure 3, the visual assessment did not reveal noticeable changes in appearance or evidence of visible microbial growth in any of the samples throughout the 28-day storage period. This qualitative observation is consissent with the analytical results reported in Section 3.5.3 and Section 3.5.4, where colour paramters and oxidation indices confirmed the stability of the cheese during storage. Together, these findings support the potential of the selected films, particularly the active film containing C.E., to preserve product quality over time. The absence of visible spoilage is especially relevant when compared to findings from studies on cheese storage, where microbial spoilage, such as mould growth or surface discoloration, often becomes apparent under suboptimal packaging conditions as reported by Marcuzzo et al. (2012) [41]. The maintained visual quality observed in this study further demonstrated the protective efficacy of the selected films and supports their potential application in commercial cheese packaging, where product appearance strongly influences consumer acceptance. Although the consumer panel exhibited a gender imbalance (71% female, 29% male), no gender-dependent differences were observed in liking scores or purchase intention. The reddish coloration of the C.E.-containing films did not appear to elicit divergent responses between genders. As the purpose of the test was exploratory and aimed at assessing general visual acceptance rather than demographic effects, the observed imbalance is not expected to have influenced the overall interpretation of the results.
3.5. Chemical Analysis of Grated Cheddar Cheese
3.5.1. Weight Loss, Moisture Content and Water Activity
The WL percentage is a critical indicator of the cheese stability under different packaging conditions [42]. The results in Table 4 showed a gradual increase in WL of cheese stored in LDPE, from 0.39% on day 7 to 2.90% by day 28. This indicated that LDPE packaging effectively minimized weight loss, suggesting that it provides a barrier against environmental factors that contribute to product degradation. The control film had a significantly higher WL after one week and increased to 11.63% by day 28. Similarly, a study by Kanmani and Rhim (2014) [43] exploring the integration of extract into films demonstrated that although the extract imparted antimicrobial properties, it did not markedly improve the film’s barrier properties against moisture. This suggested that without proper packaging, products are more susceptible to moisture loss and other degrading factors. Although the film with 5% C.E. initially performed better than the control, its higher weight loss indicates that the addition of C.E. does not significantly enhance weight retention compared to LDPE. The MC is essential for understanding product quality and shelf life, as it influences microbial growth, texture, and sensory properties. The MC of cheese in LDPE remained relatively stable around 65% throughout the storage period. This stability suggests that LDPE effectively retained moisture, maintaining product quality over time. In contrast, the control film exhibited an increasing moisture content, as shown by Giannakas et al. (2022) [44]. This trend may indicate a moisture absorption issue of the control packaging, likely due to the hydrophilic nature of chitosan and the presence of glycerol as a plasticizer, which increases the film’s affinity for water molecules and its permeability to moisture. The film with 5% C.E. initially had a higher MC which further increased to 76.62% after 4 weeks. While the incorporation of C.E. may have slightly improved moisture retention compared to the control film, it was still less effective than LDPE. This could be attributed to the potential hygroscopic properties of C.E. components. WA, critical for microbial growth, decreased in both the control and 5% C.E. films over time, suggesting limited effect on moisture retention compared to LDPE [45]. The combination of high WL and relatively stable MC/WA values reflects the difference between whole-sample mass loss and subsample moisture determination. WL quantifies the total mass lost from the packaged cheese portion, whereas MC and aw are measured on independently collected analytical subsamples, which retain the intrinsic moisture characteristic of cheddar cheese. Therefore, the two parameters are not expected to correlate directly.

Table 4.
Weight loss, moisture content and water activity of cheese samples packaged by LDPE, control and film with 5% C.E. stored at 4 °C for 28 days.
Although the physicochemical parameters of the cheese (WL, MC, WA) did not differ significantly between the control film and the 5% C.E. film, the addition of C.E. provided a functional benefit that is not reflected in these barrier properties. The primary contribution of C.E. is its antimicrobial activity, which is clearly demonstrated by the inhibition zones observed against S. aureus, L. monocytogenes, and P. aeruginosa. This selective antibacterial effect is particularly relevant for cheese, where Gram-positive pathogens and spoilage organisms represent the main microbiological risks during storage. Therefore, even if the incorporation of C.E. does not significantly modify moisture loss or water activity, it enhances the active protection of the product by suppressing microbial growth—an advantage that cannot be achieved with the control film or LDPE. This highlights the practical value of C.E. as a natural antimicrobial agent for active food packaging applications [46,47]. In summary, the use of LDPE packaging significantly minimized WL and maintained MC more effectively than the control and 5% C.E. films over 28 days. The control exhibited the highest WL and less stable MC. The addition of 5% C.E. showed some promise in retaining moisture but did not outperform like LDPE. However, its antimicrobial functionality provides an added value that compensates for the lack of improvement in passive barrier properties.
3.5.2. pH and Titratable Acidity of Cheese
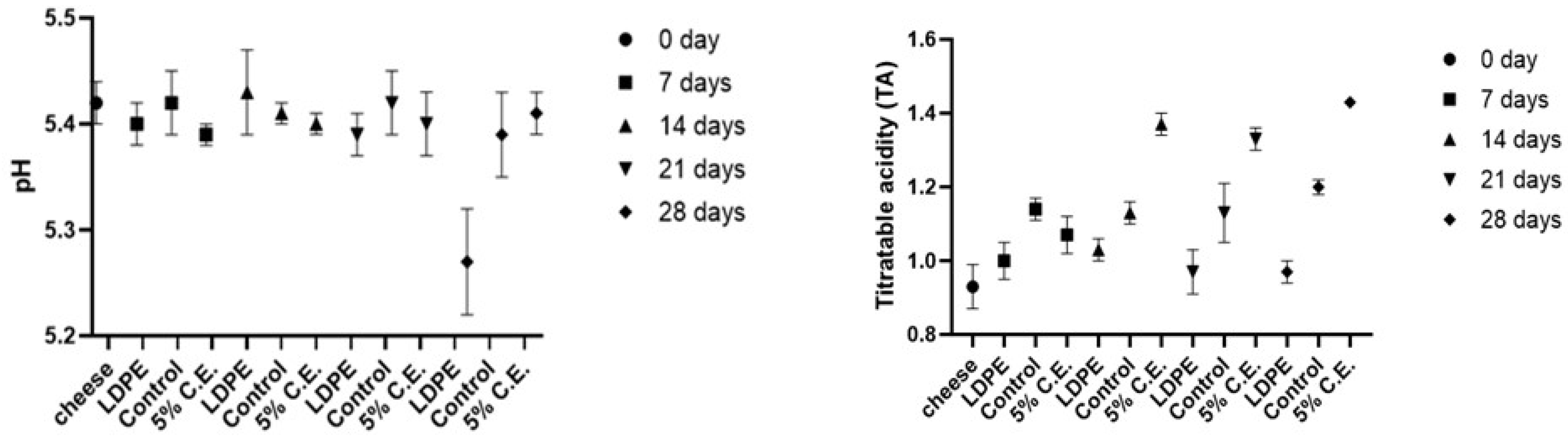
The plot (Figure 4) shows the variation in pH and titratable acidity (TA) of cheddar cheese over a period of 28 days for three different samples: LDPE, control and film with 5% C.E. The TA is expressed as a percentage of lactic acid in the cheese.
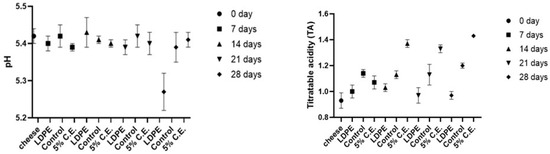
Figure 4.
The pH and titratable acidity of LDPE, control film and film with 5% C.E. stored at 4 °C for 28 days.
The pH values in Figure 4 for all samples remained relatively stable over the 28-day period, with most samples maintaining a pH of 5.4. In this case, the pH did not drop significantly due to the capacity of the cheese matrix, which helps maintain a stable pH as reported in the literature [48]. The TA for all samples was measured starting from cheese value of 0.93% lactic acid at day 0. LDPE exhibited a gradual increase in acidity, while the control film showed a steady rise in TA, reaching 1.20% lactic acid by day 28. The 5% C.E. sample also demonstrated an upward trend in acidity, with notable increase. The data in Figure 4 suggested that while the pH remains stable for the samples, the TA increased over time, indicating potential fermentation processes. The increase in TA over time likely reflected ongoing microbial activity, which can lead to the production of organic acids. In cheese, it is generally attributed to lactic acid fermentation driven by lactic acid bacteria, commonly involved in the fermentation process during cheese maturation. Additionally, the specific formulation of packaging and storage conditions may have influenced microbial activity and pH stabilization.
3.5.3. Oxidative State of Cheese Oil
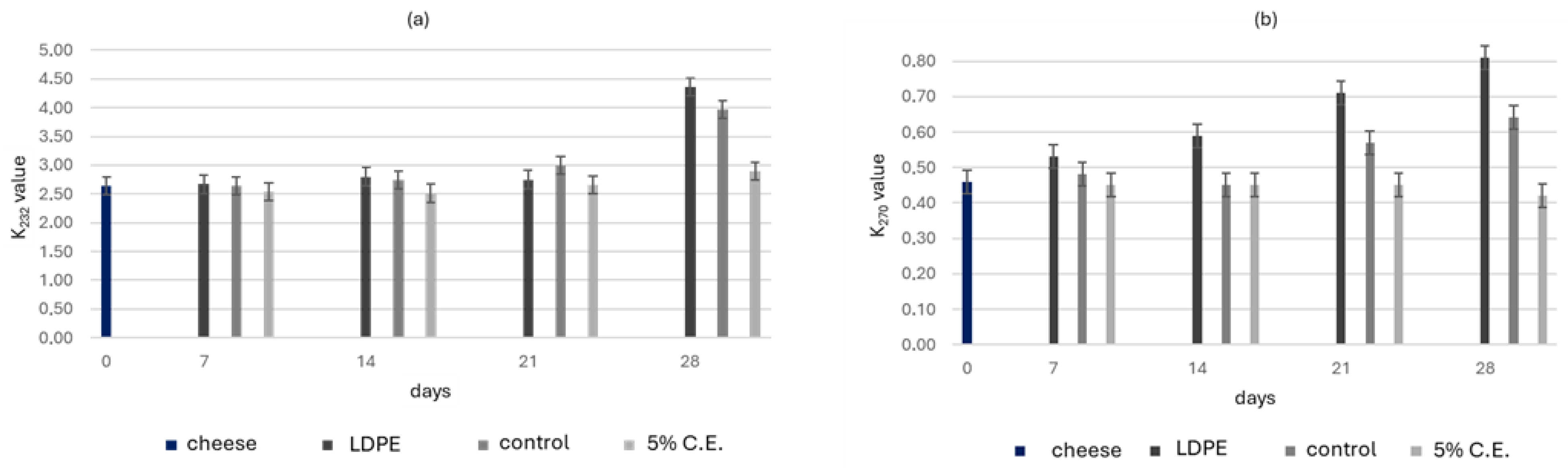
The lipid extraction yield from the cheddar cheese was approximately 54%. To assess the oxidative stability during storage, K232 and K270 values were measured over a 28-day period at 4 °C (Figure 5). The K232 value reflects the formation of conjugated dienes, which are primary oxidation products, while the K270 value indicates the presence of secondary oxidation products, such as aldehydes and ketones, which are typically associated with more advanced stages of lipid degradation.
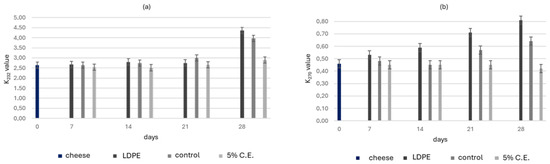
Figure 5.
(a) K232 and (b) K270 of the LDPE, control and film with 5% C.E. samples stored at 4 °C for 28 days. Values were presented as mean ± SD.
At day 0, the K232 value was 2.64, and the K270 value was 0.46 for all cheese samples. After 7 days, K232 slightly increased for the LDPE film, while the control film stayed stable. The 5% C.E. film showed the best performance, reducing K232 to 2.51. In terms of secondary oxidation, the LDPE film had an increased K270, while the control decreased to 0.44; the 5% C.E. film maintained the lowest K270 value at 0.40, suggesting oxidation inhibition. At the end of the storage period, the LDPE and control films showed significant increases in both K232 and K270, indicating high oxidation (Figure 5). The 5% C.E. film maintained the lowest K232 and K270 values, demonstrating its strong antioxidant effect and confirming its ability to inhibit both primary and secondary oxidation throughout the storage period.
Similar values of K232 have been reported in a study by Bai et al. [27] which observed K232 values reaching up to 4.5 in hazelnut kernels, supporting the consistency of the oxidative behaviour observed in the present study. The slight increase in K270 values detected during storage may be attribuited to the presence of natural antioxidant compounds released from the active the films. Such compounds are known to inhibit the formation of the secondary oxidation product, thereby contributing to lipid stability. This mechanism has been described in dairy products by Samet–Bali et al. [49] who demonstrated that phenolific compounds played a key role limiting the increase in K270 values. Similarly, Shadidi et al. [50] highlighted the role of natural antioxidants in slowing lipid oxidation and preserving the quality of products [51,52]. In agreement with these findings, the results in Figure 5 indicate that the antioxidant properties of C.E. effectively reduced both primary (K232) and secondary (K270) oxidation in cheddar cheese.
3.5.4. Colour Parameters of Cheese
The data presented in Table 5 depicts the colorimetric properties of the cheddar cheese stored in the film types (LDPE, control, and +5% C.E.) measured over a 28-day period. The parameters measured include lightness (L), chromaticity values (a and b), and colour difference (ΔE) compared to the initial measurements taken on day 0. These parameters provide insights into how the colour and appearance of the cheese sealed in the films change over time [53]. The results showed that LDPE maintained better colour stability compared to control and +5% C.E. over the 28-day period, despite some fluctuations. The control films demonstrated significant degradation in colour, as evidenced by decreasing L* values and high ΔE, indicating a loss of lightness. The film + 5% C.E. exhibited less pronounced degradation compared to the control, although they still showed changes over time. The addition of C.E. helped to moderate colour change, but the films overall still showed noticeable degradation. Additionally, the role of storage conditions in preserving cheese colour has been highlighted by Ghasemian et al. [32], who reported that cheeses stored at 5 °C showed almost no changes in a and b values over time, indicating good colour stability under refrigerated conditions. These findings align with our results, suggesting that appropriate storage temperature and packaging are crucial factors in maintaining the visual quality of cheese during storage.

Table 5.
The colour parameters (L, a, b and ΔE) of the LPDE, control and 5% C.E. films stored at 4 °C for 28 days.
It is worth noting that the antioxidant properties of the chitosan-based film functionalized with C.E. used in this study have already been extensively characterized in our previous work, where both TPC and DPPH analyses confirmed the strong radical-scavenging capacity of the incorporated extract [13]. These previously reported findings support the behaviour observed during cheese storage in the present study, indicating that the intrinsic antioxidant activity of the films contributes to moderating oxidative changes and colour degradation over time.
3.6. Panel Test
The impact of C.E. on the appearance and quality of bioplastics was assessed through a panel test [54]. Panellists rated the visual appearance of the control film with a score of 4 (34.1%), while the bioplastic containing 5% C.E. received a score of 5 (38.6%), indicating a slightly higher preference for the film with the flower extract. Regarding the perceived safety of C.E. used in bioplastics, panellists provided a score of 4 (38.6%), suggesting a generally positive perception. The reddish colour of the bioplastics containing 5% of C.E. was viewed favourably, with panellists expressing a preference for its use in a final product. However, when considering the overall perceived quality of the product, particularly in the context of its production from renewable resources, the evaluation score remained above 3 (25.6%). The majority (72.7%) of panellists indicated that they would consider purchasing a product stored in packaging with 5% of C.E. When asked about willingness to buy such a product in terms of whether bioplastic was slightly more expensive than conventional plastic, 52% responded affirmatively, while 48% answered “maybe,” reflecting some hesitancy toward cost implications [55]. These findings suggested that C.E. incorporation in bioplastics positively influences visual appeal and consumer acceptance, while maintaining a generally favourable perception of safety and sustainability [56].
4. Conclusions
The findings of this study highlight the potential of incorporating C.E. into chitosan-based films for food packaging applications. The addition of C.E. significantly influenced the optical properties of the films, producing a reddish coloration that intensified with increasing concentration of C.E. The enhanced opacity suggests that C.E. may be advantageous for packaging applications where reduced transparency is desirable, particularly for light-sensitive foods. The films maintained sufficient structural stability to be handled and heat-sealed during packaging operations, and their mechanical properties have been previously characterized in detail for the same formulations [13]. The oxygen barrier properties of the films were notably improved with 5% of C.E., which exhibited a marked reduction in oxygen permeability compared to LDPE. This behaviour aligns with the previously reported permeability profile of this formulation and suggests that C.E.-enhanced films may contribute to limiting oxidative deterioration in packaged foods.
The antimicrobial properties of C.E. were confirmed, with significant inhibitory effects against Staphylococcus aureus and Listeria monocytogenes, while the lack of activity against Escherichia coli indicated that additional antibacterial agents may be required to broaden the spectrum of protection. The antimicrobial evaluation in this study was qualitative and intended as a preliminary confirmation of the activity of the C.E. extract, which has been previously characterized in detail [13]. While no visible spoilage was observed on cheese samples, we acknowledge that quantitative microbiological counts would provide a more comprehensive assessment, and this represents an area for future investigation. The antimicrobial and antioxidant effects observed in this study are in line with the phytochemical composition previously reported for this extract, which includes a well-defined profile of phenolic constituents [13].
Food packaging evaluations showed that although C.E. films performed better than the control, they did not match the protective capacity of LDPE, particularly in terms of weight loss prevention and moisture retention. The combination of higher WL and relatively stable MC values reflects the difference between whole-sample mass loss and sub-sample moisture determination. This behaviour is consistent with the water vapour permeability previously reported for this films [13]. Neverthless, the films demonstrated promising antioxidative properties, reducing lipid oxidation and maintaining improved chemical stability over 28 days, indicating their suitability for foods prone to oxidative degradation. Although the C.E.-functionalized chitosan films demonstrated superior oxygen barrier performance and effectively reduced oxidative deterioration, they were less efficient than LDPE in controlling moisture transfer and weight loss. This limitation is particularly relevant for industrial cheese packaging, where humidity management is a key determinant of product quality. To overcome this limitation, future formulations could incorporate hydrophobic components such as lipids or waxes, which are known to reduce water vapour transmission in biopolymer matrices. Alternatively, multilayer or laminate structures combining chitosan with more moisture-resistant biopolymers, or the addition of nanofillers (e.g., nanoclays, cellulose nanocrystals), may significantly enhance the water vapour barrier while preserving the functional benefits of the extract. Consumer perception analysis revealed a positive reception response towards bioplastics enhanced with C.E., with most participants expressing willingness to purchase products packaged in these materals, reflecting growing consumer interest in sustainable and biodegradable packaging solutions. Overall, this study highlighted the potential of C.E.-enhanced chitosan films as an eco-friendly alternative to conventional packaging. Future research should focus on optimizing the film composition to improve moisture retention and mechanical performance, ensuring competitive functionality with conventional plastic materials. Additionally, studies including quantitative microbiological analyses are required to confirm the antimicrobial performance of C.E.-functionalized films directly in food systems.
Author Contributions
Conceptualization, S.F.M.; methodology, M.A. and S.F.M.; software, M.A. and S.F.M.; validation, C.V.L.G., D.C. and S.F.M.; formal analysis, M.A. and S.F.M.; investigation, M.A. and S.F.M.; resources, C.V.L.G. and D.C.; data curation, C.V.L.G. and S.F.M.; writing—original draft preparation, M.A., writing—review and editing, C.V.L.G., D.C. and S.F.M.; visualization, C.V.L.G. and S.F.M.; supervision, C.V.L.G. and S.F.M.; project administration, C.V.L.G., D.C. and S.F.M.; funding acquisition, C.V.L.G. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by the project PRIN 2022 PNRR P2022973R3 “Sustainable Melanin Production by Lignocellulose Agricultural Byproducts for Food Safety Applications (MeLA)” (D. D. n. 1289 of 4 August 2023) and funded by the European Union–NextGenerationEU.
Informed Consent Statement
This research does not involve any animal studies. The consumer evaluation consisted solely of non-invasive visual assessment, with no collection of personal or sensitive data and no interventions on participants. In accordance with institutional guidelines, this activity is exempt from formal ethical approval. All participants were adults (≥18 years) and provided informed consent.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Singh, N.; Ognunseitan, O.A.; Wong, M.H.; Tang, Y. Sustainable material alternatives to petrochemical plastics pollution: A review analysis. Sustain. Horiz. 2022, 2, 100016. [Google Scholar] [CrossRef]
- Cazón, P.; Vàzquez, M. Application of chitosan as food packaging materials. Sustain. Agric. Rev. 2019, 36, 81–123. [Google Scholar] [CrossRef]
- Wang, L.; Wang, Q.; Tong, J.; Zhou, J. Physicochemical properties of chitosan films incorporated with honeysuckle flower extract for active food packaging. J. Food Process Eng. 2015, 40, 12305. [Google Scholar] [CrossRef]
- Muñoz-Tebar, N.; Pèrez-Alvarez, J.A.; Fernàndez-Lòpez, J.; Viuda-Martos, M. Chitosan edible films and coating with added bioactive compounds: Antibacterial and antioxidant properties and their application for food products: A review. Polymers 2023, 15, 396. [Google Scholar] [CrossRef] [PubMed]
- Seyrekoğlu, F.; Efdal, E. Chitosan-Based Edible Films as Innovative Preservation Tools for Fermented and Dairy Products. Fermentation 2025, 11, 542. [Google Scholar] [CrossRef]
- Flórez, M.; Vàzquez, M.; Cazòn, P. Enhancing the quality of Havarti cheese: Chitosan films with nettle Urtica dioica L. extract as slice separators to retard lipid oxidation. LWT 2023, 189, 115504. [Google Scholar] [CrossRef]
- Khubiev, O.M.; Egorov, A.R.; Kirichuk, A.A.; Khrustalev, V.N.; Tshkhovrebov, A.G.; Kritchenkov, A.S. Chitosan-Based Antibacterial Films for Biomedical and Food Applications. Int. J. Mol. Sci. 2023, 24, 10738. [Google Scholar] [CrossRef]
- Laganà, G.; Barreca, D.; Smeriglio, A.; Germanò, M.P.; D’angelo, V.; Calderaro, A.; Bellocco, E.; Trombetta, D. Evaluation of Anthocyanin profile, antioxidant, cytoprotective and anti-angiogenic properties of Callistemon citrinus flowers. Plants 2020, 9, 1045. [Google Scholar] [CrossRef]
- Gonelimali, F.D.; Lin, J.; Miao, W.; Xuan, J.; Charles, F.; Chen, M.; Hatab, S.R. Antimicrobial properties and mechanism of action of some plant extracts against food pathogens and spoilage microorganism. Frontiers 2018, 9, 1639. [Google Scholar] [CrossRef]
- Sousa Vieira, I.R.; Azevedo de de Carvalho, A.P.; Conte-Junior, C.A. Recent advances in biobased and biodegradable polymer nanocomposites, nanoparticles, and natural antioxidants for antibacterial and antioxidant food packaging applications. Compr. Rev. Food Sci. Food Saf. 2022, 21, 3673–3716. [Google Scholar] [CrossRef]
- Periyasamy, T.; Asrafali, S.P.; Lee, J. Recent Advances in Functional Biopolymer Films with Antimicrobial and Antioxidant Properties for Enhanced Food Packaging. Polymers 2025, 17, 1257. [Google Scholar] [CrossRef]
- Soni, V.; Raizada, P.; Singh, P.; Coung, H.N.; Rangabhashiyam, S.; Saini, A.; Saini, R.V.; Van Le, Q.; Nadda, A.K.; Le, T.T.; et al. Sustainable and green trends in using plant extracts for the synthesis of biogenic metal nanoparticles toward environmental and pharmaceutical advances: A review. Environ. Res. 2021, 202, 111622. [Google Scholar] [CrossRef] [PubMed]
- Avitabile, M.; Mirpoor, S.F.; Esposito, S.; Merola, G.; Mariniello, L.; Patanè, G.T.; Barreca, D.; Giosafatto, C.V.L. Manufacture of bioplastics prepared from chitosan functionalized with Callistemon citrinus extract. Polymers 2024, 16, 2693. [Google Scholar] [CrossRef]
- Avitabile, M.; Aleksov, A.; Giosafatto, C.V.L.; Restaino, F.O.; Lesjak, M.; Živanović, N.; Mariniello, L.; Simin, N. Pectin-Based Bioplastics Functionalized with Polyphenols from Rose Oil Distillation Wastewater Exhibit Antioxidant Activity. Biomacromolecules 2024, 25, 7695–7703. [Google Scholar] [CrossRef]
- Mirpoor, S.F.; Patanè, G.T.; Corrado, I.; Giosafatto, C.V.L.; Ginestra, G.; Nostro, A.; Foti, A.; Gucciardi, P.G.; Mandalari, G.; Barreca, D.; et al. Functionalization of Polyhydroxyalkanoates (PHA)-Based Bioplastic with Phloretin for Active Food Packaging: Characterization of Its Mechanical, Antioxidant, and Antimicrobial Activities. Int. J. Mol. Sci. 2023, 24, 11628. [Google Scholar] [CrossRef]
- Guzman-Puyol, S.; Benìtez, J.J.; Heredia-Guerrero, J.A. Transparency of polymeric food packaging materials. Food Res. Int. 2022, 161, 111792. [Google Scholar] [CrossRef]
- Siripatrawan, U.; Harte, B.R. Physical properties and antioxidant activity of an active film from chitosan incorporated with green tea extract. Food Hydrocoll. 2010, 24, 770–775. [Google Scholar] [CrossRef]
- Giosafatto, C.V.L.; Di Pierro, P.; Gunning, A.P.; Mackie, A.; Porta, R.; Mariniello, L. Trehalose-containing hydrocolloid edible films prepared in the presence of transglutaminase. Biopolymers 2014, 101, 9. [Google Scholar] [CrossRef]
- Mirpoor, S.F.; Zannini, D.; Santagata, G.; Giosafatto, C.V.L. Cardoon seed oil cake proteins as substrate for microbial transglutaminase: Their application as matrix for bio-based packaging to extend the shelf-life of peanuts. Food Hydrocoll. 2024, 147, 109339. [Google Scholar] [CrossRef]
- ASTM D3985–05; Standard Test Method for Oxygen Gas Transmission Rate Through Plastic Film and Sheeting Using a Coulometric Sensor. ASTM International: West Conshohocken, PA, USA, 2010.
- Czajkowska, A.; Korsak, D.; Fiedoruk-Pogrebniak, M.; Koncki, R.; Strzelak, K. Turbidimetric flow analysis system for the investigation of microbial growth. Talanta 2024, 268, 125303. [Google Scholar] [CrossRef] [PubMed]
- Humphries, R.M.; Hindler, J.A.; Shaffer, K.; Campeau, S.A. Evaluation of Ciprofloxacin and Levofloxacin Disk Diffusion and Test using the 2019 Enterobacteriaceae CLSI breakpoints. J. Clin. Microbiol. 2019, 57, 3. [Google Scholar] [CrossRef]
- Trotta, F.; De Silvia, S.; Massironi, A.; Mirpoor, S.F.; Lignou, S.; Ghawi, S.K.; Charalampopoulos, D. Silver bionanocomposites as active food packaging: Recent advances future trends tackling the food waste crisis. Polymers 2023, 15, 4243. [Google Scholar] [CrossRef]
- Dag, D.; Jung, J.; Zhao, Y. Development and characterization of cellulose nanofiber reinforced hydroxypropyl methylcellulose films functionalized with propolis-loaded zein nanoparticles and its application for cheddar cheese storage. Int. J. Biol. Macromol. 2024, 261, 129790. [Google Scholar] [CrossRef]
- de Moraes, J.O.; Hilton, S.T.; Moraru, C.I. The effect of pulsed light and starch films with antimicrobials on Listeria innocua and the quality of sliced cheddar cheese during refrigerated storage. Food Control 2020, 112, 107134. [Google Scholar] [CrossRef]
- Lin, J.-H.; Liu, L.-Y.; Yang, M.-H.; Lee, M.-H. Ethyl Acetate/Ethyl Alcohol Mixtures as an Alternative to Folch Reagent for Extracting Animal Lipids. J. Agric. Food Chem. 2004, 52, 4984–4986. [Google Scholar] [CrossRef]
- Bai, X.; Dag, D.; Jung, J.; Zhao, Y. Investigation of antioxidant edible coting and modified atmosphere packaging for enhancing storability of hazelnut kernels. J. Agric. Food Res. 2024, 17, 101246. [Google Scholar] [CrossRef]
- Deng, Z.; Jung, J.; Zhao, Y. Development, characterization, and validation of chitosan absorbed cellulose nanofiber (CNF) films as water resistant and antibacterial food contact packaging. Lwt-Food Sci. Technol. 2017, 83, 132–140. [Google Scholar] [CrossRef]
- Lignou, S.; Oloyede, O.O. Consumer Acceptability and Sensory Profile of Sustainable Paper-Based Packaging. Foods 2021, 10, 990. [Google Scholar] [CrossRef] [PubMed]
- Kanatt, S.R.; Rao, M.S.; Chawla, S.P.; Sharma, A. Active chitosan–polyvinyl alcohol films with natural extracts. Food Hydrocoll. 2012, 29, 290–297. [Google Scholar] [CrossRef]
- Ressutte, J.B.; da Silva Saranti, T.F.; de Moura, M.R.; dos Santos Pozza, M.S.; da Silva Scapim, M.R.; Stafussa, A.O.; Madrona Scaramal, G. Citric acid incorporated in a chitosan film as an active packaging material to improve the quality and duration of matured cheese shelf life. J. Diary Res. 2022, 89, 201–207. [Google Scholar] [CrossRef]
- Ghasemian, S.O.; Ahmadi-Dastgerdi, A.; Abdollahi, A.; Tirtashi, F.E.; Zokaedi, M.; Fallah, N.; NajafAbadi, P.I.; Dolatyari, F. The Effect of Active Packaging Film Based on Chitosan Containing Rosemary (Rosmarinus officinalis L.) Extract on Cheese Shelf Life. J. Food Biochem. 2024, 1, 2108707. [Google Scholar] [CrossRef]
- Fayemi, P.O.; Öztürk, I.; Özcan, C.; Muguruma, M.; Yetim, H.; Sakata, R.; Ahhmed, A. Antimicrobial activity of extracts of Callistemon citrinus flowers and leaves against Listeria monocytogenes in beef burger. J. Food Meas. Charact. 2017, 11, 924–929. [Google Scholar] [CrossRef]
- Shehabeldine, A.M.; Ashour, R.M.; Okba, M.M.; Saber, F.R. Callistemon citrinus bioactive metabolites ad new inhibitors of methicillin-resistant Staphyloccocus aureus biofilm formation. J. Ethnopharmacol. 2020, 254, 112669. [Google Scholar] [CrossRef]
- Seyydnejad, S.M.; Niknejad, M.; Darabpoor, I.; Motamedi, H. Antibacterial Activity of Hydroalcoholic Extract of Callistemon citrinus and Albizia lebbeck. Am. J. Appl. Sci. 2010, 7, 13–16. [Google Scholar] [CrossRef]
- De Rossi, L.; Rocchetti, G.; Lucini, L.; Rebecchi, A. Antimicrobial Potential of Polyphenols: Mechanisms of Action and Microbial Responses—A Narrative Review. Antioxidants 2025, 14, 200. [Google Scholar] [CrossRef]
- Tan, Z.; Deng, J.; Ye, Q.; Zhang, Z. The Antibacterial Activity of Natural-derived Flavonoids. Curr. Top. Med. Chem. 2022, 22, 1009–1019. [Google Scholar] [CrossRef] [PubMed]
- Maher, C.; Hassan, K.A. The Gram-negative permeability barrier: Tipping the balance of the in and the out. Antimicrob. Chemother. 2023, 14, e01205-23. [Google Scholar] [CrossRef]
- Todorov, S.D.; Marinho de Almeida, B.; Lima, E.M.F.; Fabi, J.P.; Lajolo, F.M.; Aymoto Hassimotto, N.M. Phenolic Compounds and Bacteriocins: Mechanisms, Interactions, and Applications in Food Preservation and Safety. Mol. Nutr. Food Res. 2025, 69, e202400723. [Google Scholar] [CrossRef]
- Costa, M.J.; Maciel, L.C.; Teixeira, J.A.; Vicente, A.A.; Cerqueira, M.A. Use of edible films and coatings in cheese preservation: Opportunities and challenges. Food Res. Int. 2018, 107, 84–92. [Google Scholar] [CrossRef]
- Marcuzzo, E.; Peressini, D.; Sensidoni, A. Shelf Life of short ripened soft Cheese Stored under Various Packaging Conditions. J. Food Process. Preserv. 2012, 37, 1094–1102. [Google Scholar] [CrossRef]
- Mei, J.; Yuan, Y.; Wu, Y.; Li, Y. Characterization of edible starch-chitosan film and its application in the storage of Mongolian cheese. Int. J. Biol. Macromol. 2013, 57, 17–21. [Google Scholar] [CrossRef]
- Kanmani, P.; Rhim, J.-W. Antimicrobial and physical-mechanical properties of agar-based films incorporated with grapefruit seed extract. Carbohydr. Polym. 2014, 102, 708–716. [Google Scholar] [CrossRef] [PubMed]
- Giannakas, A.E.; Salam, C.E.; Moschovas, D.; Zaharioudakis, K.; Georgopoulos, S.; Asimakopoulos, G.; Aktypis, A.; Proestos, C.; Karakassides, A.; Avgeropoulos, A.; et al. The increse of soft cheese shelf-life packaged with edible films based on novel hybrid nanostructures. Gels 2022, 8, 539. [Google Scholar] [CrossRef] [PubMed]
- Tapia, M.S.; Alzamora, S.M.; Chirife, J. Effects of Water Activity (aw) on microbial stability as a hurdle in food preservation. In Water Activity in Foods; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2020. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, B.; Zhang, W.; Zhang, L.; Zhao, H.; Wang, S.; Huang, C. Recent Advances in Sustainable Antimicrobial Food Packaging: Insights into Release Mechanisms, Design Strategies, and Applications in the Food Industry. J. Agric. Food Chem. 2023, 71, 11806–11833. [Google Scholar] [CrossRef]
- Deshmukh, R.K.; Gaikwad, K.K. Natural antimicrobial and antioxidant compounds for active food packaging applications. Biomass Convers. Biorefin. 2024, 14, 4419–4440. [Google Scholar] [CrossRef]
- Laurienzo, P.; Malinconico, M.; Pizzano, R.; Manzo, C.; Piciocchi, N.; Sorrentino, A.; Volpe, M.G. Natural Polysaccharide-Based Gels for Dairy Food Preservation. J. Dairy Sci. 2006, 89, 2856–2864. [Google Scholar] [CrossRef] [PubMed]
- Samet-Bali, O.; Ammar, I.; Ennouri, M.; Attia, H. Physicochemical characteristics and storage stability of clarified butter fat “smen” produced from pasteurized and non-pasteurized milk. J. Pharm. Health Sci. 2017, 5, 195–205. [Google Scholar]
- Shadidi, F.; Zhong, Y. Lipid oxidation and improving the oxidative stability. Chem. Soc. Rev. 2010, 39, 4067–4079. [Google Scholar] [CrossRef]
- Momayezhaghighi, M.; Lante, A. Natural antioxidants recovered from food by-products as inhibitors of lipid oxidation. Croat. J. Food Sci. Technol. 2025, 17, 142–165. [Google Scholar] [CrossRef]
- Gutièrrez-del-Rio, I.; López-Ibáñez, S.; Magadán-Corpas, P.; Fernández-Calleja, L.; Pérez-Valero, A.; Tuñón-Granda, M.; Miguélez, E.M.; Villar, C.J.; Lombó, F. Terpenoids and Polyphenols as Natural Antioxidant Agents in Food Preservation. Antioxidants 2021, 10, 1264. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, Z.; Chen, Y.; Ma, X.; Xia, M. Chtiosan and procyanidin composite films with high antioxidant activity and pH responsivitiy for cheese packaging. Food Chem. 2021, 338, 128013. [Google Scholar] [CrossRef] [PubMed]
- Filho, W.L.; Barbir, J.; Abubakar, I.R.; Paço, A.; Stasiskiene, Z.; Hornbogen, M.; Fendt, M.T.C.; Voronova, V.; Klõga, M. Consumer attitudes and concerns with bioplastics use: An international study. PLoS ONE 2022, 17, e0266918. [Google Scholar] [CrossRef] [PubMed]
- Scarpi, D.; Russo, I.; Confente, I.; Hazen, B. Individual antecedents to consumer intention to switch to food waste bioplastic products: A configuration analysis. Ind. Mark. Manag. 2021, 93, 578–590. [Google Scholar] [CrossRef]
- Findrik, E.; Meixner, O. Drivers and barriers for consumers purchasing bioplastics–A systematic literature review. J. Clean. Prod. 2023, 410, 137311. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.