Nitrogen and Bromide Co-Doped Hydroxyapatite Thin Films with Antimicrobial Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
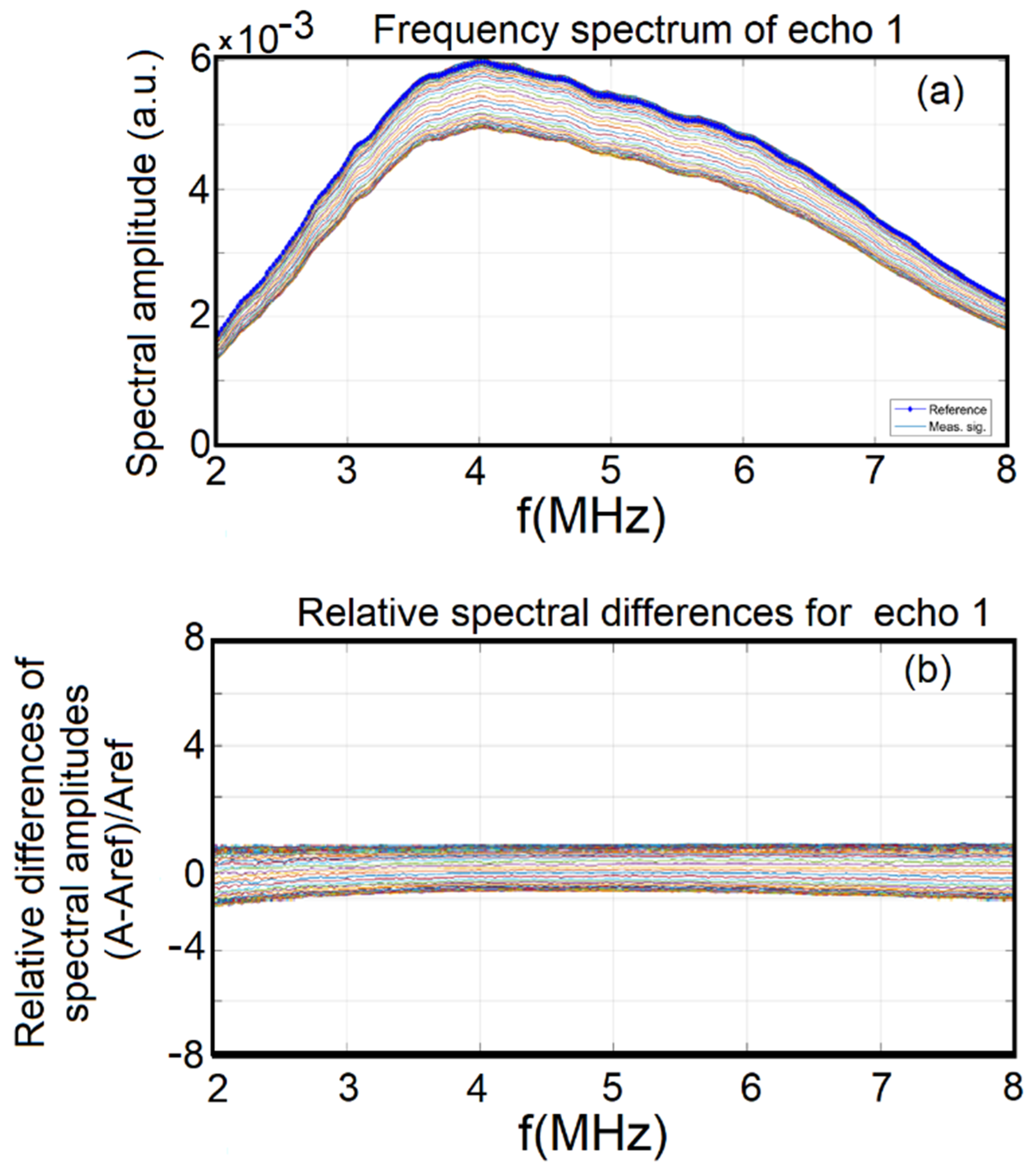
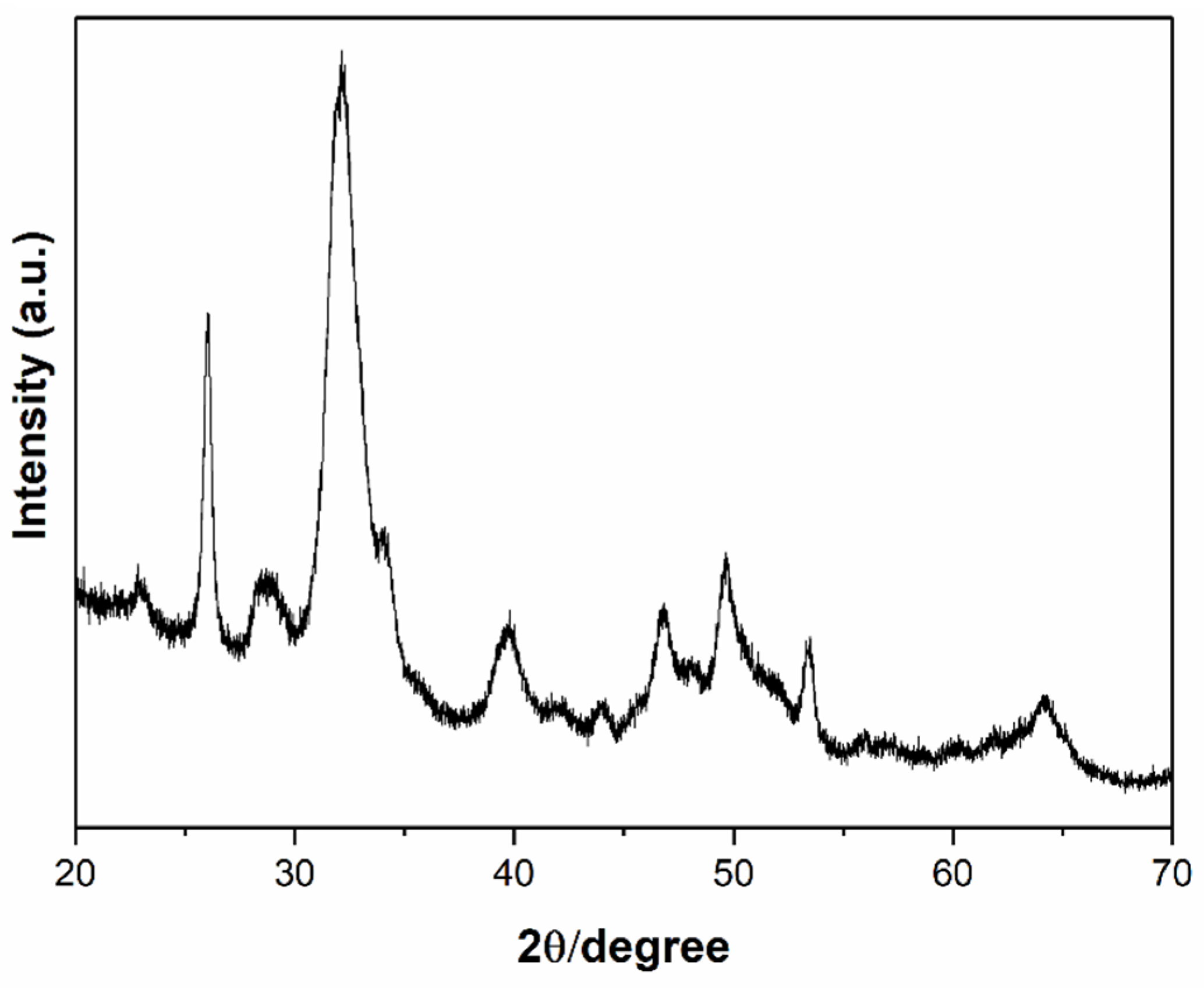
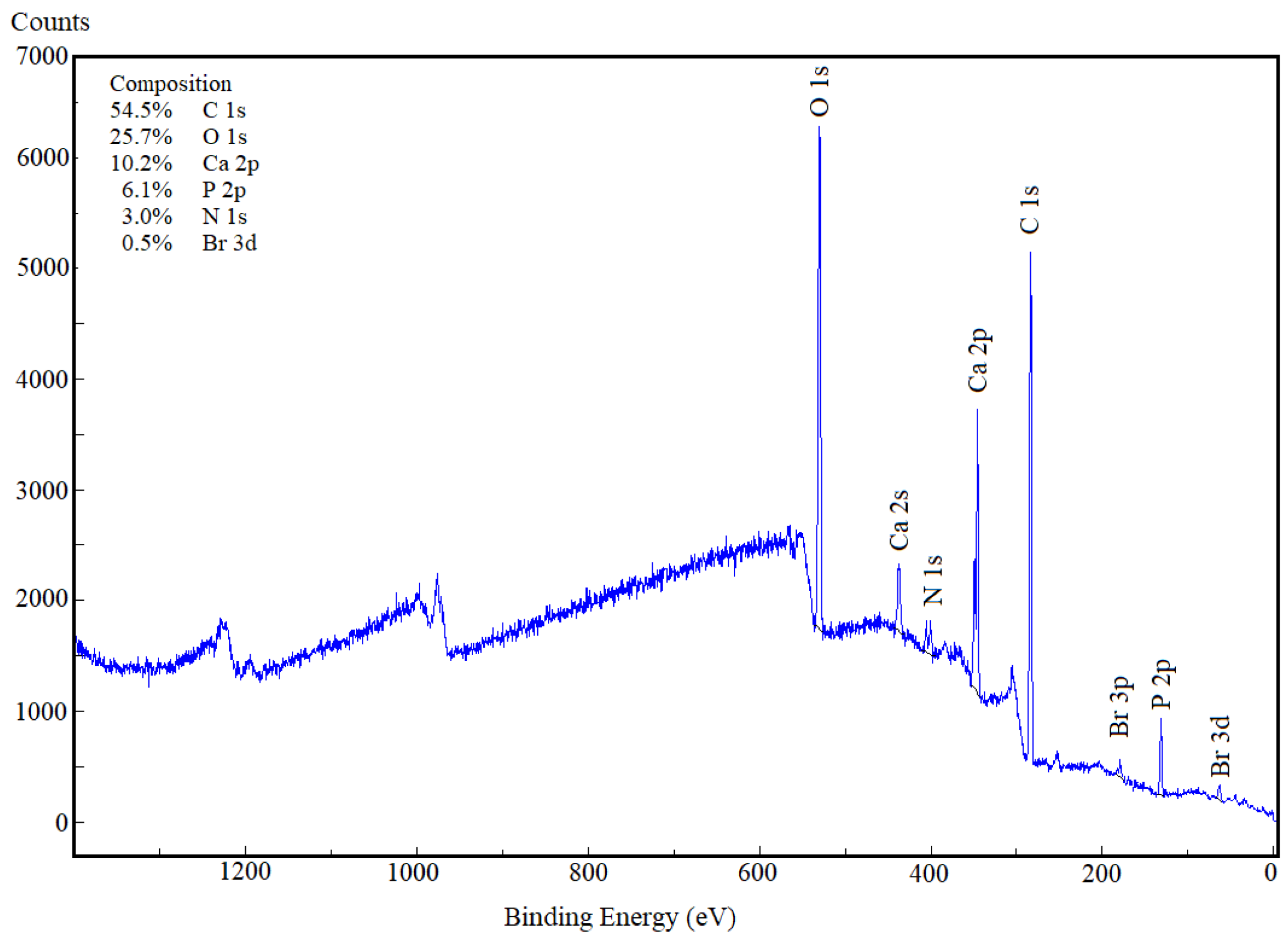
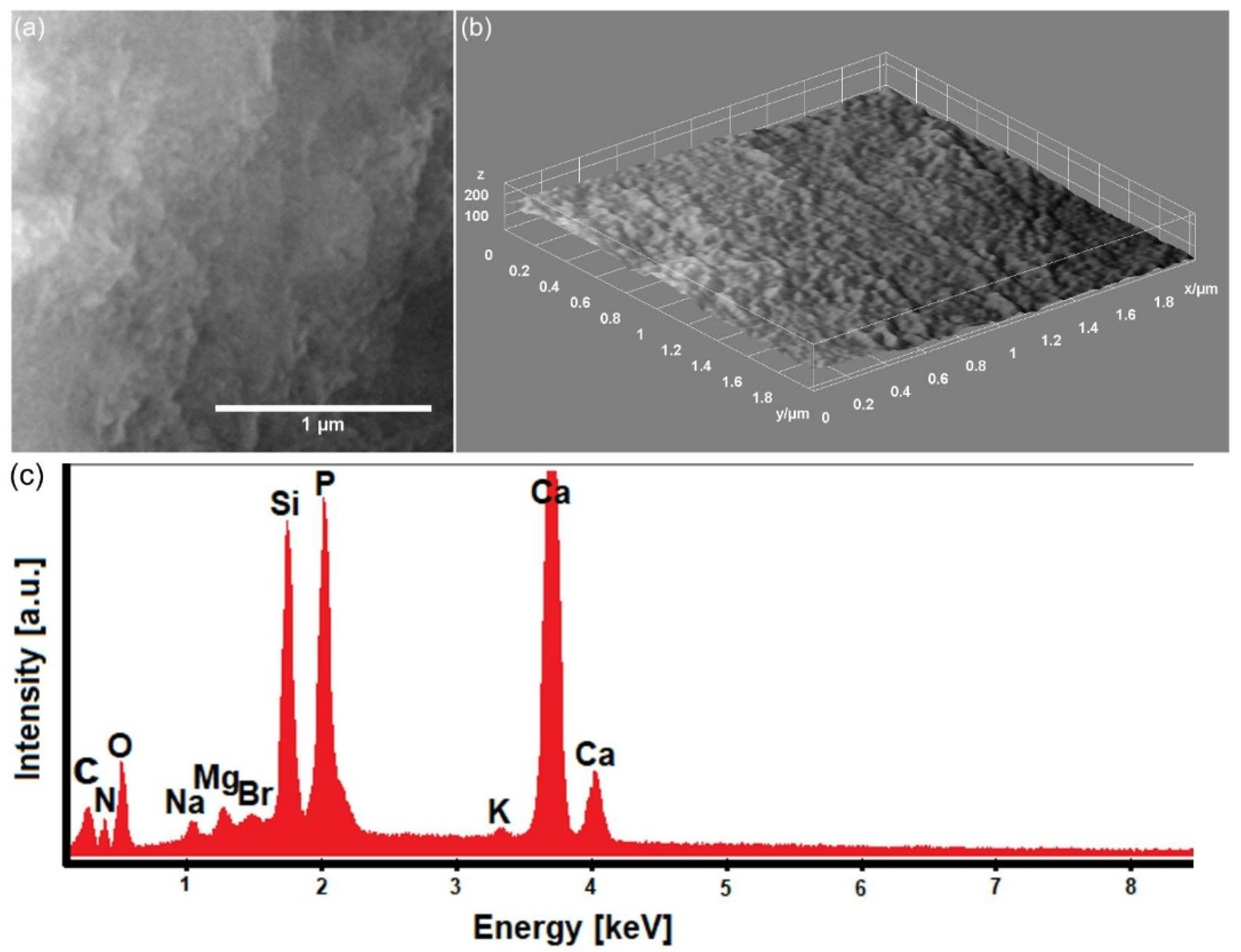
2.2. Characterization Methods
2.3. In Vitro Antimicrobial Assay
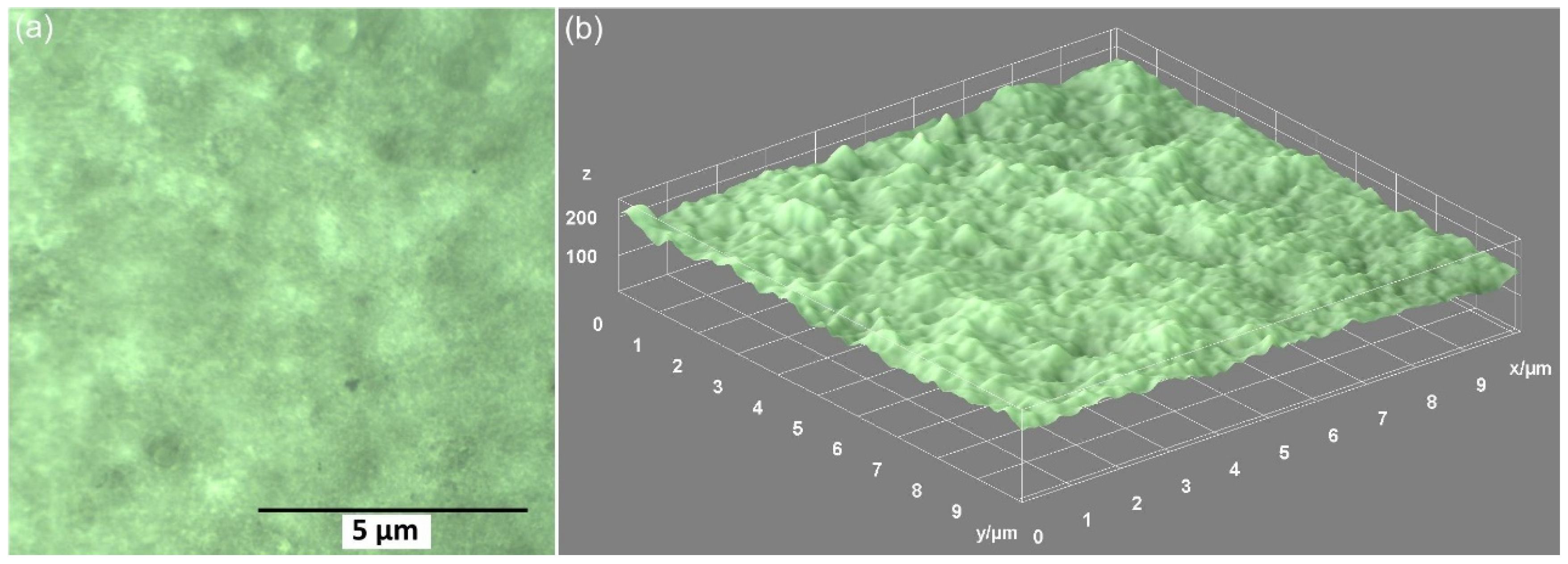
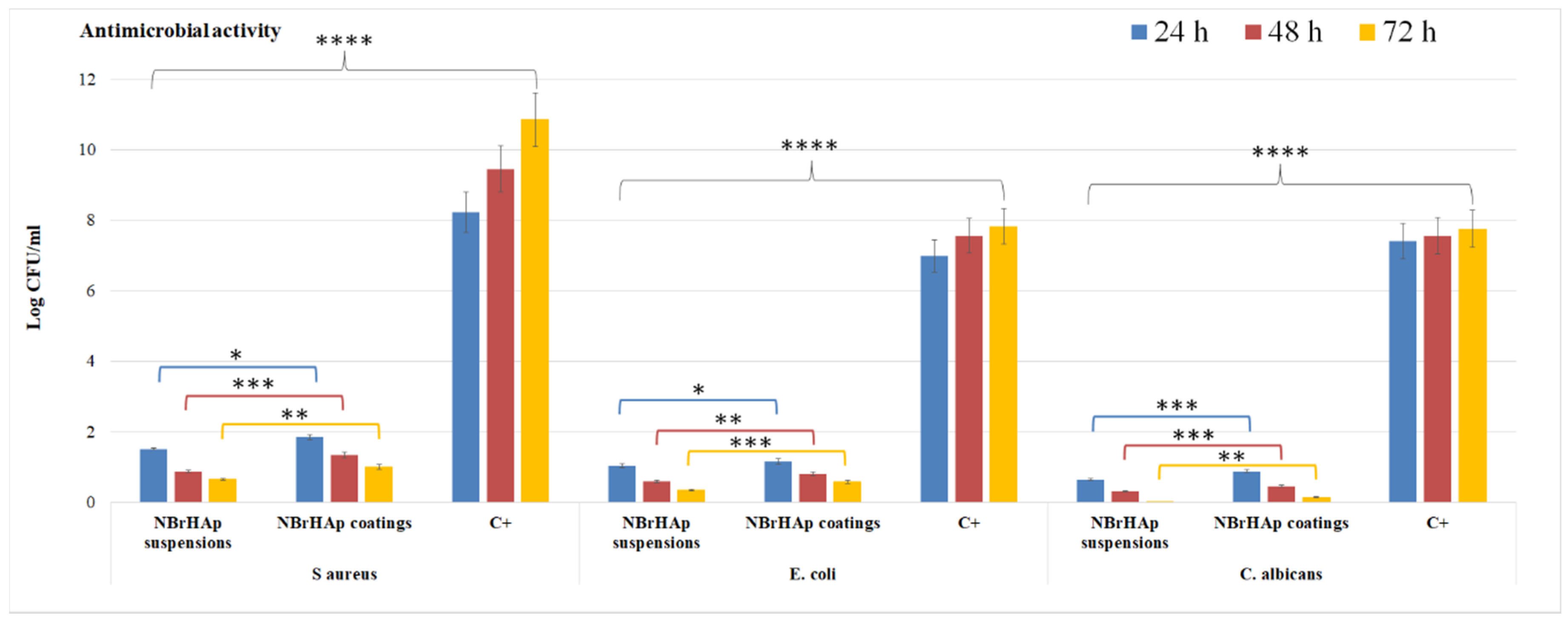
3. Results
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Arami, H.; Mohajerani, M.; Mazloumi, M.; Khalifehzadeh, R.; Lak, A.; Sadrnezhaad, S.K. Rapid formation of hydroxyapatite nanostrips via microwave irradiation. J. Alloys Compd. 2009, 469, 391–394. [Google Scholar] [CrossRef]
- Pu’ad, N.M.; Haq, R.A.; Noh, H.M.; Abdullah, H.Z.; Idris, M.I.; Lee, T.C. Synthesis method of hydroxyapatite: A review. Mater. Today Proc. 2020, 29, 233–239. [Google Scholar] [CrossRef]
- Tari, N.E.; Motlagh, M.M.K.; Sohrabi, B. Synthesis of hydroxyapatite particles in catanionic mixed surfactants template. Mater. Chem. Phys. 2011, 131, 132–135. [Google Scholar] [CrossRef]
- Predoi, S.-A.; Ciobanu, C.S.; Motelica-Heino, M.; Chifiriuc, M.C.; Badea, M.L.; Iconaru, S.L. Preparation of Porous Hydroxyapatite Using Cetyl Trimethyl Ammonium Bromide as Surfactant for the Removal of Lead Ions from Aquatic Solutions. Polymers 2021, 13, 1617. [Google Scholar] [CrossRef]
- Namiki, R.; Suyama, T.; Izawa, C.; Ikeda-Fukazawa, T.; Honda, M.; Watanabe, T.; Aizawa, M. Chemical State of Nitrogen in Nitrogen-Doped Hydroxyapatite Ceramics with Enhanced Bioactivity. Key Eng. Mater. 2016, 720, 215–218. [Google Scholar] [CrossRef]
- Pramanik, S.; Agarwal, A.K.; Rai, K.N.; Garg, A. Development of high strength hydroxyapatite by solid-state-sintering process. Ceram. Int. 2007, 33, 419–426. [Google Scholar] [CrossRef]
- Fathi, M.H.; Mohammadi Zahrani, E. Mechanical alloying synthesis and bioactivity evaluation of nanocrystalline fluoridated hydroxyapatite. J. Cryst. Growth 2009, 311, 1392–1403. [Google Scholar] [CrossRef]
- Ciobanu, C.S.; Iconaru, S.L.; Massuyeau, F.; Constantin, L.V.; Costescu, A.; Predoi, D. Synthesis, structure, and luminescent properties of europium-doped hydroxyapatite nanocrystalline powders. J. Nanomater. 2012, 2012, 61. [Google Scholar] [CrossRef]
- Fihri, A.; Len, C.; Varma, R.S.; Solhy, A. Hydroxyapatite: A review of syntheses, structure and applications in heterogeneous catalysis. Coord. Chem. Rev. 2017, 347, 48–76. [Google Scholar] [CrossRef]
- Liu, D.M.; Troczynski, T.; Tseng, W.J. Water-based sol–gel synthesis of hydroxyapatite: Process development. Biomaterials 2001, 22, 1721–1730. [Google Scholar] [CrossRef]
- Widiyastuti, W.; Setiawan, A.; Winardi, S.; Nurtono, T.; Setyawan, H. Particle formation of hydroxyapatite precursor containing two components in a spray pyrolysis process. Front. Chem. Sci. Eng. 2014, 8, 104–113. [Google Scholar] [CrossRef]
- Safavi, M.S.; Walsh, F.C.; Surmeneva, M.A.; Surmenev, R.A.; Khalil-Allafi, J. Electrodeposited hydroxyapatite-based biocoatings: Recent progress and future challenges. Coatings 2021, 11, 110. [Google Scholar] [CrossRef]
- Arcos, D.; Vallet-Regí, M. Substituted hydroxyapatite coatings of bone implants. J. Mater. Chem. B 2020, 8, 1781–1800. [Google Scholar] [CrossRef]
- What Chemical Elements are Found in the Human Body? Available online: news-medical.net (accessed on 20 October 2021).
- Izutani, N.; Imazato, S.; Nakajo, K.; Takahashi, N.; Takahashi, Y.; Ebisu, S.; Russell, R.R.B. Effects of the antibacterial monomer 12-methacryloyloxydodecylpyridinium bromide (MDPB) on bacterial viability and metabolism. Eur. J. Oral Sci. 2011, 119, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Galarraga-Vinueza, M.E.; Passoni, B.; Benfatti, C.A.M.; Mesquita-Guimarães, J.; Henriques, B.; Magini, R.S.; Fredel, M.C.; Meerbeek, B.V.; Teughels, W.; Souza, J.C.M. Inhibition of multi-species oral biofilm by bromide doped bioactive glass. J. Biomed. Mater. Res. A 2017, 105, 1994–2003. [Google Scholar] [CrossRef] [PubMed]
- Bagherzadeh, R.; Montazer, M.; Latifi, M.; Sheikhzadeh, M.; Sattari, M. Evaluation of comfort properties of polyester knitted spacer fabrics finished with water repellent and antimicrobial agents. Fibers Polym. 2007, 8, 386–392. [Google Scholar] [CrossRef]
- Nakata, K.; Tsuchido, T.; Matsumura, Y. Antimicrobial cationic surfactant, cetyltrimethylammonium bromide, induces superoxide stress in Escherichia coli cells. J. Appl. Microbiol. 2011, 110, 568–579. [Google Scholar] [CrossRef]
- Criado, A.; Lavela, P.; Ortiz, G.F.; Tirado, J.L.; Gzouli, S.; Edfouf, Z.; Pérez-Vicente, C. CTAB-Assisted Synthesis of C@Na3V2(PO4)2F3 With Optimized Morphology for Application as Cathode Material for Na-Ion Batteries. Front. Phys. 2019, 7, 207. [Google Scholar] [CrossRef]
- Smith, D.K.; Korgel, B.A. The importance of the CTAB surfactant on the colloidal seed-mediated synthesis of gold nanorods. Langmuir 2008, 24, 644–649. [Google Scholar] [CrossRef]
- Cao, D.; Jin, X.; Gan, L.; Wang, T.; Chen, Z. Removal of phosphate using iron oxide nanoparticles synthesized by eucalyptus leaf extract in the presence of CTAB surfactant. Chemosphere 2016, 159, 23–31. [Google Scholar] [CrossRef]
- Clarke, J.D. Cetyltrimethyl Ammonium Bromide (CTAB) DNA Miniprep for Plant DNA Isolation. Cold Spring Harb. Protoc. 2009, 4, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Yu, Q.; Zhang, B.; Ma, F.; Jia, C.; Xiao, C.; Zhang, B.; Xing, L.; Li, M. Novel mechanisms of surfactants against Candida albicans growth and morphogenesis. Chem. Biol. Interact. 2015, 227, 1–6. [Google Scholar] [CrossRef]
- Abduraimova, A.; Molkenova, A.; Duisembekova, A.; Mulikova, T.; Kanayeva, D.; Atabaev, T.S. Cetyltrimethylammonium Bromide (CTAB)-Loaded SiO2–Ag Mesoporous Nanocomposite as an Efficient Antibacterial Agent. Nanomaterials 2021, 11, 477. [Google Scholar] [CrossRef] [PubMed]
- McDonnell, G.E. Antisepsis, Disinfection, and Sterilization; ASM Press: Washington, DC, USA, 2007. [Google Scholar]
- Oliveira, W.F.; Silva, P.M.S.; Silva, R.C.S.; Silva, G.M.M.; Machado, G.; Coelho, L.C.B.B.; Correia, M.T.S. Staphylococcus aureus and Staphylococcus epidermidis infections on implants. J. Hosp. Infect. 2018, 98, 111–117. [Google Scholar] [CrossRef]
- Crémet, L.; Broquet, A.; Brulin, B.; Jacqueline, C.; Dauvergne, S.; Brion, R.; Asehnoune, K.; Corvec, S.; Heymann, D.; Caroff, N. Pathogenic potential of Escherichia coli clinical strains from orthopedic implant infections towards human osteoblastic cells. Pathog. Dis. 2015, 73, ftv065. [Google Scholar] [CrossRef]
- Passarelli, P.C.; De Leonardis, M.; Piccirillo, G.B.; Desantis, V.; Papa, R.; Rella, E.; Mastandrea Bonaviri, G.N.; Papi, P.; Pompa, G.; Pasquantonio, G.; et al. The Effectiveness of Chlorhexidine and Air Polishing System in the Treatment of Candida albicans Infected Dental Implants: An Experimental In Vitro Study. Antibiotics 2020, 9, 179. [Google Scholar] [CrossRef]
- Ciobanu, C.S.; Massuyeau, F.; Constantin, L.V.; Predoi, D. Structural and physical properties of antibacterial Ag-doped nano-hydroxyapatite synthesized at 100 C. Nanoscale Res. Lett. 2011, 6, 1–8. [Google Scholar] [CrossRef]
- Predoi, D.; Iconaru, S.L.; Predoi, M.V. Dextran-coated zinc-doped hydroxyapatite for biomedical applications. Polymers 2019, 11, 886. [Google Scholar] [CrossRef] [PubMed]
- Predoi, D.; Iconaru, S.L.; Predoi, M.V. Bioceramic Layers with Antifungal Properties. Coatings 2018, 8, 276. [Google Scholar] [CrossRef]
- Danilchenko, S.N.; Kukharenko, O.G.; Moseke, C.; Protsenko, I.Y.; Sukhodub, L.F.; Sulkio-Cleff, B. Determination of the bone mineral crystallite size and lattice strain from diffraction line broadening. Cryst. Res. Technol. 2002, 37, 1234–1240. [Google Scholar] [CrossRef]
- Barrett, C.S.; Cohen, J.B.; FaberJ, J.; Jenkins, R.; Leyden, D.E.; Russ, J.C.; Predecki, P.K. Advances in X-ray Analysis; Plenum Press: New York, NY, USA, 1986; Volume 29. [Google Scholar]
- Visser, J.W. A fully automatic program for finding the unit cell from powder data. J. Appl. Cryst. 1969, 2, 89–95. [Google Scholar] [CrossRef]
- Predoi, D.; Iconaru, S.L.; Deniaud, A.; Chevallet, M.; Michaud-Soret, I.; Buton, N.; Prodan, A.M. Textural, Structural and Biological Evaluation of Hydroxyapatite Doped with Zinc at Low Concentrations. Materials 2017, 10, 229. [Google Scholar] [CrossRef]
- ImageJ Website. Available online: http://imagej.nih.gov/ij (accessed on 1 October 2021).
- Gwyddion. Available online: http://gwyddion.net/ (accessed on 1 October 2021).
- Predoi, D.; Iconaru, S.L.; Predoi, M.V.; Motelica-Heino, M.; Buton, N.; Megier, C. Obtaining and Characterizing Thin Layers of Magnesium Doped Hydroxyapatite by Dip Coating Procedure. Coatings 2020, 10, 510. [Google Scholar] [CrossRef]
- Predoi, D.; Iconaru, S.L.; Predoi, M.V. Fabrication of Silver- and Zinc-Doped Hydroxyapatite Coatings for Enhancing Antimicrobial Effect. Coatings 2020, 10, 905. [Google Scholar] [CrossRef]
- Iconaru, S.L.; Prodan, A.M.; Turculet, C.S.; Beuran, M.; Ghita, R.V.; Costescu, A.; Groza, A.; Chifiriuc, M.C.; Chapon, P.; Gaiaschi, S.; et al. Enamel Based Composite Layers Deposited on Titanium Substrate with Antifungal Activity. J. Spectrosc. 2016, 2016, 4361051. [Google Scholar] [CrossRef]
- Bodycomb. Available online: http://www.horiba.com/us/particle (accessed on 17 November 2021).
- Fissan, H.; Ristig, S.; Kaminski, H.; Asbacha, C.; Epple, M. Comparison of different characterization methods for nanoparticle dispersions before and after aerosolization. Anal. Methods 2014, 6, 7324. [Google Scholar] [CrossRef]
- Zhao, J.; Dong, X.; Bian, M.; Zhao, J.; Zhang, Y.; Sun, Y.; Chen, J.H.; Wang, X.H. Solution combustion method for synthesis of nanostructured hydroxyapatite, fluorapatite and chlorapatite. Appl. Surf. Sci. 2014, 314, 1026–1033. [Google Scholar] [CrossRef]
- Rusu, V.M.; Ng, C.H.; Wilke, M.; Tiersch, B.; Fratzl, P.; Peter, M.G. Size controlled hydroxyapatite nanoparticles as self-organised organic-inorganic composite materials. Biomaterials 2005, 26, 5414–5426. [Google Scholar] [CrossRef] [PubMed]
- Bokuniaeva, A.O.; Vorokh, A.S. Estimation of particle size using the Debye equation and the Scherrer formula for polyphasic TiO2 powder. J. Phys. Conf. Ser. 2019, 1410, 012057. [Google Scholar] [CrossRef]
- Klug, H.P.; Alexander, L.E. X-ray Diffraction Procedures for Polycrystallite and Amorphous Materials, 2nd ed.; Wiley: New York, NY, USA, 1974. [Google Scholar]
- Salarian, M.; Solati-Hashjin, M.; Shafiei, S.S.; Goudarzi, A.; Salarian, R.; Nemati, A. Surfactant-assisted synthesis and characterization of hydroxyapatite nanorods under hydrothermal conditions. Mater. Sci.-Pol. 2009, 1, 27. [Google Scholar]
- Kaneko, N.; Suzuki, Y.; Umeda, R.; Namiki, R.; Izawa, C.; Fukazawa, T.I.; Honda, M.; Takei, T.; Watanabe, T.; Aizawa, M. Development of nitrogen-doped hydroxyapatite ceramics. J. Asian Ceram. Soc. 2020, 8, 130–137. [Google Scholar] [CrossRef]
- Ismail, R.A.; Salim, E.T.; Hamoudi, W.K. Characterization of nanostructured hydroxyapatite prepared by Nd: YAG laser deposition. Mater. Sci. Eng. C 2013, 33, 47–52. [Google Scholar] [CrossRef]
- Available online: https://www.who.int/antimicrobial-resistance/publications/surveillancereport/en/ (accessed on 15 October 2021).
- Ventola, C.L. The antibiotic resistance crisis: Part 1: Causes and threats. Pharm. Ther. 2015, 40, 277–283. [Google Scholar]
- World Health Organization. Global Action Plan on Antimicrobial Resistance. 2021. Available online: cdc.gov/drugresistance/biggest-threats.html (accessed on 25 February 2021).
- de Kraker, M.E.; Stewardson, A.J.; Harbarth, S. Will 10 million people die a year due to antimicrobial resistance by 2050? PLoS Med. 2016, 13, e1002184. [Google Scholar] [CrossRef] [PubMed]
- Cohen, M.E.; Salmasian, H.; Li, J.; Liu, J.; Zachariah, P.; Wright, J.D.; Freedberg, D.E. Surgical antibiotic prophylaxis and risk for postoperative antibiotic-resistant infections. J. Am. Coll. Surg. 2017, 225, 631–638.e3. [Google Scholar] [CrossRef]
- DeNegre, A.A.; Ndeffo Mbah, M.L.; Myers, K.; Fefferman, N.H. Emergence of antibiotic resistance in immunocompromised host populations: A case study of emerging antibiotic resistant tuberculosis in AIDS patients. PLoS ONE 2019, 14, e0212969. [Google Scholar] [CrossRef]
- Bartoletti, M.; Giannella, M.; Tedeschi, S.; Viale, P. Multidrug-resistant bacterial infections in solid organ transplant candidates and recipients. Infect. Dis. Clin. N. Am. 2018, 32, 551–580. [Google Scholar] [CrossRef]
- Ravi, S.; Zhu, M.; Luey, C.; Young, S.W. Antibiotic resistance in early periprosthetic joint infection. ANZ J. Surg. 2016, 86, 1014–1018. [Google Scholar] [CrossRef]
- Ma, J.; Jemal, A.; Fedewa, S.A.; Islami, F.; Lichtenfeld, J.L.; Wender, R.C.; Cullen, K.J.; Brawley, O.W. The American Cancer Society 2035 challenge goal on cancer mortality reduction. CA Cancer J. Clin. 2019, 69, 351–362. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Sun, P.; Wu, H.; Bai, N.; Wang, R.; Zhu, W.; Zhang, J.; Liu, F. Inactivation of Staphylo coccus aureus and Enterococcus faecalis by a direct-current, cold atmospheric-pressure air plasma microjet. J. Biomed. Mater. Res. 2010, 24, 264–269. [Google Scholar] [CrossRef]
- Gehanno, J.F.; Louvel, A.; Nouvellon, M.; Caillard, J.-F.; Pestel-caron, M. Aerial dispersal of meticillin-resistant Staphylococcus aureus in hospital rooms by infected or colonised patients. J. Hosp. Infect. 2009, 71, 256–262. [Google Scholar] [CrossRef]
- Gopal, J.; Narayana, J.L.; Wu, H.-F. TiO2 nanoparticle assisted mass spectrometry as biosensor of Staphylococcus aureus, key pathogen in nosocomial infections from air, skin surface and human nasal passage. Biosens. Bioelectron. 2011, 27, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Farias, E.A.O.; Dionisio, N.A.; Quelemes, P.V.; Leal, S.H.; Matos, J.M.E.; Silva Filho, E.C.; Bechtold, I.H.; Leite, J.R.S.; Eiras, C. Development and characterization of multilayer films of polyaniline, titanium dioxide and CTAB for potential antimicrobial applications. Mater. Sci. Eng. C 2014, 35, 449–454. [Google Scholar] [CrossRef]
- Gheorghe, I.; Tatu, A.L.; Lupu, I.; Thamer, O.; Cotar, A.I.; Pircalabioru, G.G.; Popa, M.; Cristea, V.C.; Lazar, V.; Chifiriuc, M.C. Molecular characterization of virulence and resistance features in Staphylococcus aureus clinical strains isolated from cutaneous lesions in patients with drug adverse reactions. Rom. Biotechnol. Lett. 2017, 22, 12321–12327. [Google Scholar]
- Available online: https://www.cdc.gov/ecoli/general/index.html (accessed on 24 October 2021).
- Salim, M.M.; Malek, N.A.N.N.; Ramli, N.I.A.; Hanim, S.A.M.; Hamdan, S. Antibacterial activity of CTAB-modified zeolite NaY with different CTAB loading. Malays. J. Fund. Appl. Sci. 2014, 10. [Google Scholar] [CrossRef]
- Vincent, J.L.; Anaissie, E.; Bruining, H.; Demajo, W.; El-Ebiary, M.; Haber, J.; Hiramatsu, Y.; Nitenberg, G.; Nyström, P.O.; Pittet, D.; et al. Epidemiology, diagnosis and treatment of systemic Candida infection in surgical patients under intensive care. Intensive Care Med. 1998, 24, 206–216. [Google Scholar] [CrossRef] [PubMed]
- Klepser, M.E. Candida resistance and its clinical relevance. Pharmacotherapy 2006, 26, 68–75. [Google Scholar] [CrossRef]
- Campoccia, D.; Montanaro, L.; Arciola, C.R. The significance of infection related to orthopedic devices and issues of antibiotic resistance. Biomaterials 2006, 27, 2331–2339. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Pardo, D.; Pigrau, C.; Lora-Tamayo, J.; Soriano, A.; del Toro, M.D.; Cobo, J.; Palomino, J.; Euba, G.; Riera, M.; Sánchez-Somolinos, M.; et al. Gram-negative prosthetic joint infection: Outcome of a debridement, antibiotics and implant retention approach. A large multicentre study. Clin. Microbiol. Infect. 2014, 20, O911–O919. [Google Scholar] [CrossRef]
- García-Oltra, E.; García-Ramiro, S.; Martínez, J.C.; Tibau, R.; Bori, G.; Bosch, J.; Mensa, J.; Soriano, A. Prosthetic joint infection by Candida spp. Rev. Esp. Quimioter. 2011, 24, 37–41. [Google Scholar]
- Kucharíková, S.; Gerits, E.; De Brucker, K.; Braem, A.; Ceh, K.; Majdič, G.; Španič, T.; Pogorevc, E.; Verstraeten, N.; Tournu, H.; et al. Covalent immobilization of antimicrobial agents on titanium prevents Staphylococcus aureus and Candida albicans colonization and biofilm formation. J. Antimicrob. Chemother. 2016, 71, 936–945. [Google Scholar] [CrossRef] [PubMed]
- Sendi, P.; Frei, R.; Maurer, T.B.; Trampuz, A.; Zimmerli, W.; Graber, P. Escherichia coli variants in periprosthetic joint infection: Diagnostic challenges with sessile bacteria and sonication. J. Clin. Microbiol. 2010, 48, 1720–1725. [Google Scholar] [CrossRef]
- Pircalabioru, G.G.; Chifiriuc, M.C. Nanoparticulate drug-delivery systems for fighting microbial biofilms: From bench to bedside. Future Microbiol. 2020, 15, 679–698. [Google Scholar] [CrossRef]
- Lorowitz, W.; Saxton, E.; Sondossi, M.; Nakaoka, K. Integrating statistics with a microbiology laboratory activity. J. Microbiol. Biol. Educ. 2005, 6, 14–19. [Google Scholar] [CrossRef]
- Ivanova, A.; Surmeneva, M.A.; Grubova, I.Y.; Sharonova, A.A.; Pichugin, V.F.; Chaikina, M.V.; Buck, V.; Prymak, O.; Epple, M.; Surmenev, R.A. Influence of the substrate bias on the stoichiometry and structure of RF-magnetron sputter-deposited silver-containing calcium phosphate coatings. Materwiss. Werksttech. 2013, 44, 218–225. [Google Scholar] [CrossRef]
- Liu, J.; Li, S.; Fang, Y.; Zhu, Z. Boosting antibacterial activity with mesoporous silica nanoparticles supported silver nanoclusters. J. Colloid Interface Sci. 2019, 555, 470–479. [Google Scholar] [CrossRef]
- Huang, R.S.; Hou, B.F.; Li, H.T.; Fu, X.C.; Xie, C.G. Preparation of silver nanoparticles supported mesoporous silica microspheres with perpendicularly aligned mesopore channels and their antibacterial activities. RSC Adv. 2015, 5, 61184–61190. [Google Scholar] [CrossRef]
- McDonnell, G.; Russell, A.D. Antiseptics and disinfectants: Activity, action, and resistance. Clin. Microbiol. Rev. 1999, 12, 147–179. [Google Scholar] [CrossRef]
- Chauret, C.P. Sanitization. In Encyclopedia of Food Microbiology, 2nd ed.; Robinson, R.K., Ed.; Academic Press: Cambridge, MA, USA, 2014. [Google Scholar]
- McKeen, L. Introduction to Food Irradiation and Medical Sterilization. In The Effect of Sterilization on Plastics and Elastomers; William Andrew: Norwich, NY, USA, 2012; pp. 1–40. [Google Scholar] [CrossRef]
- Nayak, R.; Padhye, R. Antimicrobial finishes for textiles. In Functional Finishes for Textiles; Paul, R., Ed.; Woodhead Publishing: Sawston, UK, 2015. [Google Scholar]
- Gilbert, P.; Moore, L.E. Cationic antiseptics: Diversity of action under a common epithet. J. Appl. Microbiol. 2005, 99, 703–715. [Google Scholar] [CrossRef] [PubMed]
- Ahlstrom, B.; Thompson, R.A.; Edebo, L. The effect of hydrocarbon chain length, pH and temperature on the binding and bactericidal effect of amphiphilic betaine esters on Salmonella typhimurium. APMIS 1999, 107, 318–324. [Google Scholar] [CrossRef]
- Hamilton, W.A. The mechanism of the bacteriostatic action of tetrachlorosalicylanilide: A membrane-active antibacterial compound. J. Gen. Microbiol. 1968, 50, 441–458. [Google Scholar] [CrossRef]
- Ioannou, C.J.; Hanlon, G.W.; Denyer, S.P. Action of disinfectant quaternary ammonium compounds against Staphylococcus aureus. Antimicrob. Agents Chemother. 2007, 51, 296–306. [Google Scholar] [CrossRef]
- Marini, M.; Bondi, M.; Iseppi, R.; Toselli, M.; Pilati, F. Preparation and antibacterial activity of hybrid materials containing quaternary ammonium salts via sol-gel process. Eur. Polym. J. 2007, 43, 3621–3628. [Google Scholar] [CrossRef]
- Simoes, M.; Pereira, M.O.; Vieira, M.J. Effect of mechanical stress on biofilms challenged by different c chemicals. Water Res. 2005, 39, 5142–5152. [Google Scholar] [CrossRef]
- Araújo, P.A.; Lemos, M.; Mergulhão, F.; Melo, L.; Simões, M. The influence of interfering substances on the antimicrobial activity of selected quaternary ammonium compounds. Int. J. Food Sci. 2013, 2013, 237581. [Google Scholar] [CrossRef] [PubMed]
- Gerba, C.P. Quaternary Ammonium Biocides: Efficacy in Application. Appl. Environ. Microbiol. 2014, 81, 464–469. [Google Scholar] [CrossRef]
- Paulus, W. Directory of Microbicides for the Protection of Materials—A Handbook; Springer: London, UK, 1993. [Google Scholar]
- Salton, M.R.J. Lytic agents, cell permeability, and monolayer penetrability. J. Gen. Physiol. 1968, 52, 227–252. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, C.; Pereira, A.M.; Pereira, M.C.; Melo, L.F.; Simões, M. Physiological changes induced by the quaternary ammonium compound benzyldimethyldodecylammonium chloride on Pseudomonas fluorescens. J. Antimicrob. Chemother. 2011, 66, 1036–1043. [Google Scholar] [CrossRef]
- Sands, J.A. Virucidal activity of cetyltrimethylammonium bromide (CTAB) below the critical micelle concentration. FEMS Microbiol. Lett. 1986, 36, 261–263. [Google Scholar] [CrossRef][Green Version]
- Chatain-Ly, M.H.; Moussaoui, S.; Rigobello, V.; Demarigny, Y.; Vera, A. Antiviral effect of cationic compounds on bacteriophages. Front. Microbiol. 2013, 4, 46. [Google Scholar] [CrossRef] [PubMed]
- Hamouda, T.; Baker, J.R., Jr. Antimicrobial mechanism of action of surfactantlipid preparations in enteric Gram-negative bacilli. J. Appl. Microbiol. 2000, 89, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Hu, J.; Zhang, S.; Zhou, P.; Zhao, X.; Xu, H.; Zhao, X.; Yaseen, M.; Lu, J.R. Molecular mechanisms of antibacterial and antitumor actions of designed surfactant-like peptides. Biomaterials 2012, 33, 592–603. [Google Scholar] [CrossRef] [PubMed]
- Tuson, H.H.; Weibel, D.B. Bacteria–surface interactions. Soft Matter 2013, 9, 4368–4380. [Google Scholar] [CrossRef] [PubMed]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iconaru, S.L.; Ciobanu, C.S.; Predoi, D.; Motelica-Heino, M.; Negrilă, C.C.; Badea, M.L.; Predoi, M.V.; Chifiriuc, C.M.; Popa, M. Nitrogen and Bromide Co-Doped Hydroxyapatite Thin Films with Antimicrobial Properties. Coatings 2021, 11, 1505. https://doi.org/10.3390/coatings11121505
Iconaru SL, Ciobanu CS, Predoi D, Motelica-Heino M, Negrilă CC, Badea ML, Predoi MV, Chifiriuc CM, Popa M. Nitrogen and Bromide Co-Doped Hydroxyapatite Thin Films with Antimicrobial Properties. Coatings. 2021; 11(12):1505. https://doi.org/10.3390/coatings11121505
Chicago/Turabian StyleIconaru, Simona Liliana, Carmen Steluta Ciobanu, Daniela Predoi, Mikael Motelica-Heino, Constantin Cătălin Negrilă, Monica Luminita Badea, Mihai Valentin Predoi, Carmen Mariana Chifiriuc, and Marcela Popa. 2021. "Nitrogen and Bromide Co-Doped Hydroxyapatite Thin Films with Antimicrobial Properties" Coatings 11, no. 12: 1505. https://doi.org/10.3390/coatings11121505
APA StyleIconaru, S. L., Ciobanu, C. S., Predoi, D., Motelica-Heino, M., Negrilă, C. C., Badea, M. L., Predoi, M. V., Chifiriuc, C. M., & Popa, M. (2021). Nitrogen and Bromide Co-Doped Hydroxyapatite Thin Films with Antimicrobial Properties. Coatings, 11(12), 1505. https://doi.org/10.3390/coatings11121505








