Genotoxicity of Silver Nanoparticles
Abstract
1. Introduction
1.1. Silver Nanoparticles Production
1.2. AgNPs Characteristics
1.3. Uses of AgNPs
1.4. Genotoxicity Evaluation
2. Objective
3. Search Strategy
- The article contains information on in vitro or in vivo genotoxicity testing of AgNPs.
- The genotoxicity assay(s) included in the article is (are) well established and validated genotoxicity assays (i.e., those included in the EFSA, ISO or ICH guidelines on genotoxicity testing).
- Exclusion criteria: The article deals with environmental and ecotoxicity studies
- The article deals with gene expression evaluations.
4. Results and Discussion
4.1. In Vitro Studies
4.2. In Vivo Studies
5. General Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Second Regulatory Review on Nanomaterials; The European Commission Communication from the Commission to the European Parliament, the Council and the European Economic and Social Committee: Brussels, Belgium, 2012.
- Rauscher, H.; Rasmussen, K.; Sokull-Klüttgen, B. Regulatory Aspects of Nanomaterials in the EU. Chem. Ing. Tech. 2017, 89, 224–231. [Google Scholar] [CrossRef]
- Biological Evaluation of Medical Devices, Part 22 Guidance on Nanomaterials (ISO/TR 10993-32); International Organization of Standarization: Geneva, Switzerland, 2017.
- Commisssion Reccomendation of 18 October 2011 on the Definition of Nanomaterial (2011/696/EU); The European Commission: Brussels, Belgium, 2011; Volume 275, pp. 38–40.
- Types and Uses of Nanomaterials, Including Safety Aspects. Accompanying the Second Regulatory Review on Nanomaterials; The European Commission: Brussels, Belgium, 2012.
- Zhang, X.; Liu, Z.; Shen, W.; Gurunathan, S. Silver Nanoparticles: Synthesis, Characterization, Properties, Applications, and Therapeutic Approaches. Int. J. Mol. Sci. 2016, 17, 1534. [Google Scholar] [CrossRef]
- Calderón-Jiménez, B.; Johnson, M.E.; Montoro Bustos, A.R.; Murphy, K.E.; Winchester, M.R.; Vega Baudrit, J.R. Silver Nanoparticles: Technological Advances, Societal Impacts, and Metrological Challenges. Front. Chem. 2017, 5, 6. [Google Scholar] [CrossRef]
- Khan, A.M.; Korzeniowska, B.; Gorshkov, V.; Tahir, M.; Schrøder, H.; Skytte, L.; Rasmussen, K.L.; Khandige, S.; Møller-Jensen, J.; Kjeldsen, F. Silver nanoparticle-induced expression of proteins related to oxidative stress and neurodegeneration in an in vitro human blood-brain barrier model. Nanotoxicology 2019, 13, 221–239. [Google Scholar] [CrossRef]
- Nowack, B.; Krug, H.F.; Height, M. 120 years of nanosilver history: Implications for policy makers. Environ. Sci. Technol. 2011, 45, 1177–1183. [Google Scholar] [CrossRef]
- Pulit-Prociak, J.; Banach, M. Silver nanoparticles—A material of the future…? Open Chem. 2016, 14, 76–91. [Google Scholar] [CrossRef]
- Consumer Products Inventory. Available online: https://www.nanotechproject.org/cpi/ (accessed on 24 November 2019).
- Hassanen, E.I.; Khalaf, A.A.; Tohamy, A.F.; Mohammed, E.R.; Farroh, K.Y. Toxicopathological and immunological studies on different concentrations of chitosan-coated silver nanoparticles in rats. Int. J. Nanomed. 2019, 14, 4723–4739. [Google Scholar] [CrossRef]
- McGillicuddy, E.; Murray, I.; Kavanagh, S.; Morrison, L.; Fogarty, A.; Cormican, M.; Dockery, P.; Prendergast, M.; Rowan, N.; Morris, D. Silver nanoparticles in the environment: Sources, detection and ecotoxicology. Sci. Total Environ. 2017, 575, 231–246. [Google Scholar] [CrossRef]
- BEUC-The European Consumer Organisation. Available online: https://www.beuc.eu/safety/nanotechnology (accessed on 24 November 2019).
- The Nanodatabase. Available online: http://nanodb.dk/ (accessed on 24 November 2019).
- SAFENANO. Available online: https://www.safenano.org/ (accessed on 24 November 2019).
- OECD—Safety of manufactured nanomaterials. Available online: http://www.oecd.org/chemicalsafety/nanosafety/ (accessed on 24 November 2019).
- nanoHUB—Simulation, Education, and Community for Nanotechnology. Available online: http://nanohub.org/ (accessed on 24 November 2019).
- Oberdörster, G.; Oberdörster, E.; Oberdörster, J. Nanotoxicology: An Emerging Discipline Evolving from Studies of Ultrafine Particles. Environ. Health Perspect. 2005, 113, 823–839. [Google Scholar] [CrossRef]
- Chen, X.; Schluesener, H.J. Nanosilver: A nanoproduct in medical application. Toxicol. Lett. 2008, 176, 1–12. [Google Scholar] [CrossRef]
- Ahmed, S.; Ahmad, M.; Swami, B.L.; Ikram, S. A review on plants extract mediated synthesis of silver nanoparticles for antimicrobial applications: A green expertise. J. Adv. Res. 2016, 7, 17–28. [Google Scholar] [CrossRef]
- Mathur, P.; Jha, S.; Ramteke, S.; Jain, N.K. Pharmaceutical aspects of silver nanoparticles. Artif. Cells Nanomed. Biotechnol. 2018, 46, 115–126. [Google Scholar] [CrossRef]
- Schneider, G. Antimicrobial silver nanoparticles – regulatory situation in the European Union. Mater. Today Proc. 2017, 4, S200–S207. [Google Scholar] [CrossRef]
- Abdal Dayem, A.; Hossain, M.; Lee, S.; Kim, K.; Saha, S.; Yang, G.; Choi, H.; Cho, S. The Role of Reactive Oxygen Species (ROS) in the Biological Activities of Metallic Nanoparticles. Int. J. Mol. Sci. 2017, 18, 120. [Google Scholar] [CrossRef]
- Sau, T.K.; Rogach, A.L.; Jäckel, F.; Klar, T.A.; Feldmann, J. Properties and Applications of Colloidal Nonspherical Noble Metal Nanoparticles. Adv. Mater. 2010, 22, 1805–1825. [Google Scholar] [CrossRef]
- Herron, N.; Thorn, D.L. Nanoparticles: Uses and Relationships to Molecular Cluster Compounds. Adv. Mater. 1998, 10, 1173–1184. [Google Scholar] [CrossRef]
- Mayer, A.B.R. Colloidal metal nanoparticles dispersed in amphiphilic polymers. Polym. Adv. Technol. 2001, 12, 96–106. [Google Scholar] [CrossRef]
- El Badawy, A.M.; Luxton, T.P.; Silva, R.G.; Scheckel, K.G.; Suidan, M.T.; Tolaymat, T.M. Impact of Environmental Conditions (pH, Ionic Strength, and Electrolyte Type) on the Surface Charge and Aggregation of Silver Nanoparticles Suspensions. Environ. Sci. Technol. 2010, 44, 1260–1266. [Google Scholar] [CrossRef]
- Li, J.; Tang, M.; Xue, Y. Review of the effects of silver nanoparticle exposure on gut bacteria. J. Appl. Toxicol. 2019, 39, 27–37. [Google Scholar] [CrossRef]
- Huynh, K.A.; Chen, K.L. Aggregation Kinetics of Citrate and Polyvinylpyrrolidone Coated Silver Nanoparticles in Monovalent and Divalent Electrolyte Solutions. Environ. Sci. Technol. 2011, 45, 5564–5571. [Google Scholar] [CrossRef]
- Bastos, V.; Duarte, I.F.; Santos, C.; Oliveira, H. Genotoxicity of citrate-coated silver nanoparticles to human keratinocytes assessed by the comet assay and cytokinesis blocked micronucleus assay. Environ. Sci. Pollut. Res. 2017, 24, 5039–5048. [Google Scholar] [CrossRef]
- Nallanthighal, S.; Chan, C.; Bharali, D.J.; Mousa, S.A.; Vásquez, E.; Reliene, R. Particle coatings but not silver ions mediate genotoxicity of ingested silver nanoparticles in a mouse model. NanoImpact 2017, 5, 92–100. [Google Scholar] [CrossRef]
- Ahlberg, S.; Antonopulos, A.; Diendorf, J.; Dringen, R.; Epple, M.; Flöck, R.; Goedecke, W.; Graf, C.; Haberl, N.; Helmlinger, J.; et al. PVP-coated, negatively charged silver nanoparticles: A multi-center study of their physicochemical characteristics, cell culture and in vivo experiments. Beilstein J. Nanotechnol. 2014, 5, 1944–1965. [Google Scholar] [CrossRef]
- Silver, S.; Phung, L.T.; Silver, G. Silver as biocides in burn and wound dressings and bacterial resistance to silver compounds. J. Ind. Microbiol. Biotechnol. 2006, 33, 627–634. [Google Scholar] [CrossRef]
- Fortescue-Brickdale, J.M. Collargol: A review of some of its clinical applications, with experiments on its antiseptic action. Bristol. Med. Chir. J. 1903, 21, 337–344. [Google Scholar]
- Manes, M. Silver Impregnated Carbon. U.S. Patent 3,374,608A, 1968. [Google Scholar]
- Hofer, U. The cost of antimicrobial resistance. Nat. Rev. Microbiol. 2019, 17, 3. [Google Scholar] [CrossRef]
- Kaur, A.; Preet, S.; Kumar, V.; Kumar, R.; Kumar, R. Synergetic effect of vancomycin loaded silver nanoparticles for enhanced antibacterial activity. Colloid. Surf. B-Biointerfaces 2019, 176, 62–69. [Google Scholar] [CrossRef]
- Huma, Z.; Gupta, A.; Javed, I.; Das, R.; Hussain, S.Z.; Mumtaz, S.; Hussain, I.; Rotello, V.M. Cationic Silver Nanoclusters as Potent Antimicrobials against Multidrug-Resistant Bacteria. ACS Omega 2018, 3, 16721–16727. [Google Scholar] [CrossRef]
- Franci, G.; Falanga, A.; Galdiero, S.; Palomba, L.; Rai, M.; Morelli, G.; Galdiero, M. Silver Nanoparticles as Potential Antibacterial Agents. Molecules 2015, 20, 8856–8874. [Google Scholar] [CrossRef]
- Wen, H.; Dan, M.; Yang, Y.; Lyu, J.; Shao, A.; Cheng, X.; Chen, L.; Xu, L. Acute toxicity and genotoxicity of silver nanoparticle in rats. PLoS ONE 2017, 12, e0185554. [Google Scholar] [CrossRef]
- Yang, L.; Kuang, H.; Zhang, W.; Aguilar, Z.P.; Wei, H.; Xu, H. Comparisons of the biodistribution and toxicological examinations after repeated intravenous administration of silver and gold nanoparticles in mice. Sci. Rep. 2017, 7, 3303. [Google Scholar] [CrossRef]
- Hebeish, A.; El-Rafie, M.H.; EL-Sheikh, M.A.; Seleem, A.A.; El-Naggar, M.E. Antimicrobial wound dressing and anti-inflammatory efficacy of silver nanoparticles. Int. J. Biol. Macromol. 2014, 65, 509–515. [Google Scholar] [CrossRef]
- Wei, L.; Lu, J.; Xu, H.; Patel, A.; Chen, Z.-S.; Chen, G. Silver nanoparticles: Synthesis, properties, and therapeutic applications. Drug Discov. Today 2015, 20, 595–601. [Google Scholar] [CrossRef]
- Giesz, P.; Mackiewicz, E.; Nejman, A.; Celichowski, G.; Cieślak, M. Investigation on functionalization of cotton and viscose fabrics with AgNWs. Cellulose 2017, 24, 409–422. [Google Scholar] [CrossRef]
- Wijnhoven, S.W.P.; Peijnenburg, W.J.G.M.; Herberts, C.A.; Hagens, W.I.; Oomen, A.G.; Heugens, E.H.W.; Roszek, B.; Bisschops, J.; Gosens, I.; Van De Meent, D.; et al. Nano-silver—A review of available data and knowledge gaps in human and environmental risk assessment. Nanotoxicology 2009, 3, 109–138. [Google Scholar] [CrossRef]
- Xu, L.; Li, X.; Takemura, T.; Hanagata, N.; Wu, G.; Chou, L. Genotoxicity and molecular response of silver nanoparticle (NP)-based hydrogel. J. Nanobiotechnol. 2012, 10, 16. [Google Scholar] [CrossRef]
- Li, W.; Sun, T.; Zhou, S.; Ma, Y.; Shi, Q.; Xie, X.; Huang, X.-M. A comparative analysis of antibacterial activity, dynamics, and effects of silver ions and silver nanoparticles against four bacterial strains. Int. Biodeterior. Biodegradation 2017, 123, 304–310. [Google Scholar] [CrossRef]
- Ardestani, M.S.; Fordoei, A.S.; Abdoli, A.; Ahangari Cohan, R.; Bahramali, G.; Sadat, S.M.; Siadat, S.D.; Moloudian, H.; Nassiri Koopaei, N.; Bolhasani, A.; et al. Nanosilver based anionic linear globular dendrimer with a special significant antiretroviral activity. J. Mater. Sci. Mater. Med. 2015, 26, 179. [Google Scholar] [CrossRef]
- Villeret, B.; Dieu, A.; Straube, M.; Solhonne, B.; Miklavc, P.; Hamadi, S.; Le Borgne, R.; Mailleux, A.; Norel, X.; Aerts, J.; et al. Silver Nanoparticles Impair Retinoic Acid-Inducible Gene I-Mediated Mitochondrial Antiviral Immunity by Blocking the Autophagic Flux in Lung Epithelial Cells. ACS Nano 2018, 12, 1188–1202. [Google Scholar] [CrossRef]
- Tolaymat, T.M.; El Badawy, A.M.; Genaidy, A.; Scheckel, K.G.; Luxton, T.P.; Suidan, M. An evidence-based environmental perspective of manufactured silver nanoparticle in syntheses and applications: A systematic review and critical appraisal of peer-reviewed scientific papers. Sci. Total Environ. 2010, 408, 999–1006. [Google Scholar] [CrossRef]
- Roszak, J.; Domeradzka-Gajda, K.; Smok-Pieniążek, A.; Kozajda, A.; Spryszyńska, S.; Grobelny, J.; Tomaszewska, E.; Ranoszek-Soliwoda, K.; Cieślak, M.; Puchowicz, D.; et al. Genotoxic effects in transformed and non-transformed human breast cell lines after exposure to silver nanoparticles in combination with aluminium chloride, butylparaben or di-n-butylphthalate. Toxicol. Vitr. 2017, 45, 181–193. [Google Scholar] [CrossRef] [PubMed]
- Vila, L.; García-Rodríguez, A.; Cortés, C.; Marcos, R.; Hernández, A. Assessing the effects of silver nanoparticles on monolayers of differentiated Caco-2 cells, as a model of intestinal barrier. Food Chem. Toxicol. 2018, 116, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Shaalan, M.I.; El-Mahdy, M.M.; Theiner, S.; El-Matbouli, M.; Saleh, M. In vitro assessment of the antimicrobial activity of silver and zinc oxide nanoparticles against fish pathogens. Acta Vet. Scand. 2017, 59, 49. [Google Scholar] [CrossRef] [PubMed]
- Laux, P.; Riebeling, C.; Booth, A.M.; Brain, J.D.; Brunner, J.; Cerrillo, C.; Creutzenberg, O.; Estrela-Lopis, I.; Gebel, T.; Johanson, G.; et al. Biokinetics of nanomaterials: The role of biopersistence. NanoImpact 2017, 6, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, S.; Joshi, L. Assessment of Exposure to Metallic Nanoparticles (NPs), Focusing on Silver (AgNPs) on Marine and Freshwater Model Organisms at a Cellular and Genetic Level; Environmental Protection Agency: Galway, Ireland, 2015; Report No. 150.
- Boudreau, M.D.; Imam, M.S.; Paredes, A.M.; Bryant, M.S.; Cunningham, C.K.; Felton, R.P.; Jones, M.Y.; Davis, K.J.; Olson, G.R. Differential Effects of Silver Nanoparticles and Silver Ions on Tissue Accumulation, Distribution, and Toxicity in the Sprague Dawley Rat Following Daily Oral Gavage Administration for 13 Weeks. Toxicol. Sci. 2016, 150, 131–160. [Google Scholar] [CrossRef] [PubMed]
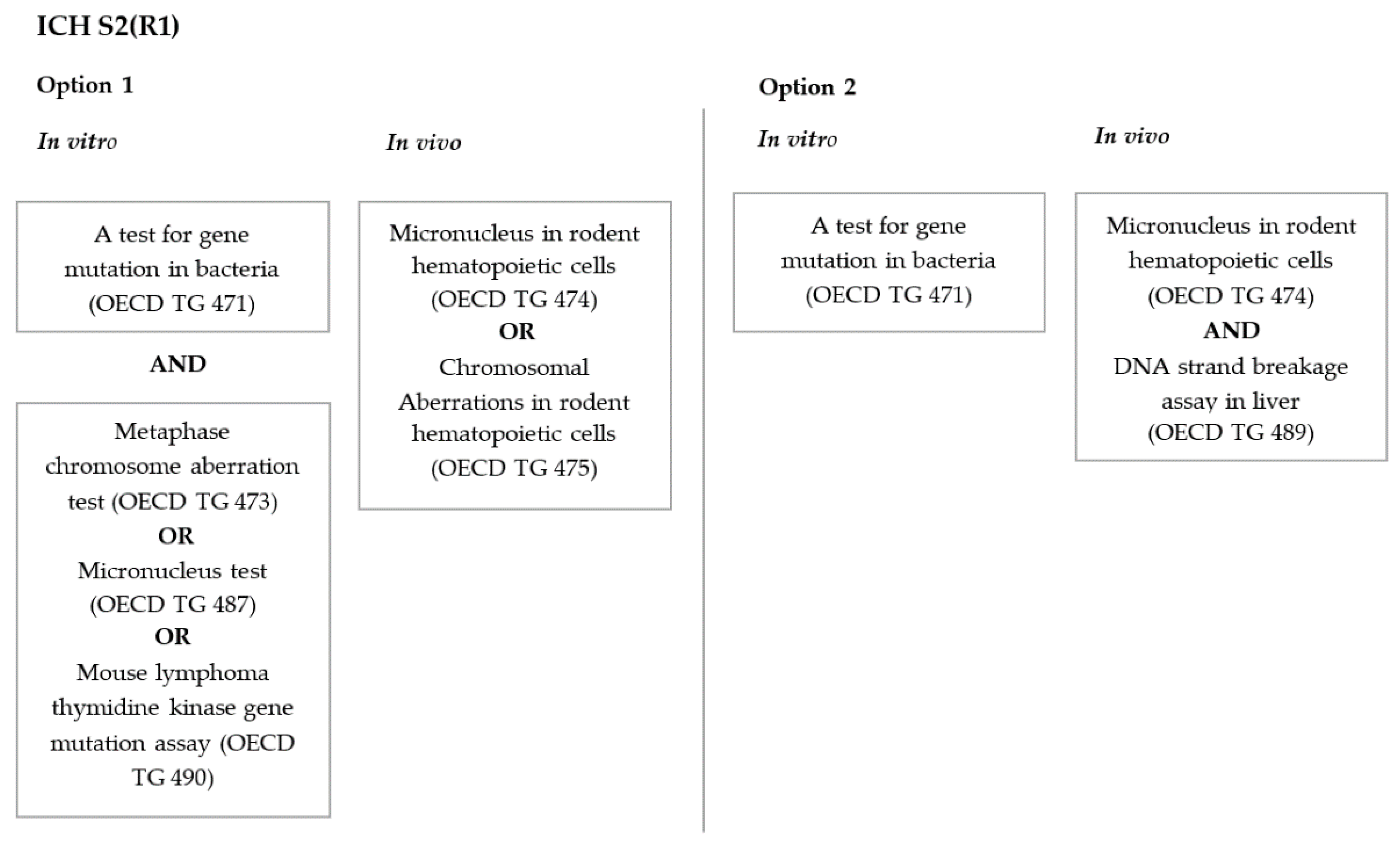
- Guidance on Genotoxicity Testing and Data Interpretation for Pharmaceuticals Intended for Human Use, S2 (R1); European Medicine Agency: Amsterdam, The Netherlands, 2011.
- Kim, H.R.; Park, Y.J.; Shin, D.Y.; Oh, S.M.; Chung, K.H. Appropriate In Vitro Methods for Genotoxicity Testing of Silver Nanoparticles. Environ. Health Toxicol. 2013, 28, 1–8. [Google Scholar] [CrossRef]
- Report of the OECD Expert Meeting Series on the Safety of Manufactured Nanomaterials No. 43 JT03367819; Organisation of Economic Co-operation and Development (OECD) Publishing: Paris, France, 2014; Volume 43, pp. 1–37.
- Hardy, A.; Benford, D.; Halldorsson, T.; Jeger, M.J.; Knutsen, H.K.; More, S.; Naegeli, H.; Noteborn, H.; Ockleford, C.; Ricci, A.; et al. Guidance on risk assessment of the application of nanoscience and nanotechnologies in the food and feed chain: Part 1, human and animal health. EFSA J. 2018, 16, 5327. [Google Scholar]
- Test No. 476: In Vitro Mammalian Cell Gene Mutation Test; Organisation of Economic Co-operation and Development (OECD) Publishing: Paris, France, 1997.
- OECD GUIDELINE FOR THE TESTING OF CHEMICALS—In Vitro Mammalian Cell Gene Mutation Tests Using the Thymidine Kinase Gene (TG 490); Organisation of Economic Co-operation and Development (OECD) Publishing: Paris, France, 2015.
- OECD GUIDELINE FOR THE TESTING OF CHEMICALS—In Vitro Mammalian Cell Micronucleus Test (TG 487); Organisation of Economic Co-operation and Development (OECD) Publishing: Paris, France, 2010.
- OECD GUIDELINE FOR THE TESTING OF CHEMICALS—Mammalian Erythrocyte Micronucleus Test (TG 474); Organisation of Economic Co-operation and Development (OECD) Publishing: Paris, France, 2016.
- OECD GUIDELINE FOR THE TESTING OF CHEMICALS—In Vivo Mammalian Alkaline Comet Assay (TG 489); Organisation of Economic Co-operation and Development (OECD) Publishing: Paris, France, 2016.
- OECD GUIDELINE FOR THE TESTING OF CHEMICALS—Transgenic Rodent Somatic and Germ Cell Gene Mutation Assays (TG 488); Organisation of Economic Co-operation and Development (OECD) Publishing: Paris, France, 2011.
- Bolea, E.; Jiménez-Lamana, J.; Laborda, F.; Abad-Álvaro, I.; Bladé, C.; Arola, L.; Castillo, J.R. Detection and characterization of silver nanoparticles and dissolved species of silver in culture medium and cells by AsFlFFF-UV-Vis-ICPMS: Application to nanotoxicity tests. Analyst 2014, 139, 914–922. [Google Scholar] [CrossRef]
- Home PubMed-NCBI. Available online: https://www.ncbi.nlm.nih.gov/pubmed (accessed on 24 November 2019).
- OECD GUIDELINE FOR THE TESTING OF CHEMICALS—In Vitro Mammalian Chromosomal Aberration Test (TG 473); Organisation of Economic Co-operation and Development (OECD) Publishing: Paris, France, 2016.
- OECD GUIDELINE FOR THE TESTING OF CHEMICALS—Mammalian Bone Marrow Chromosomal Aberration Test (TG 475); Organisation of Economic Co-operation and Development (OECD) Publishing: Paris, France, 1997.
- Guo, X.; Li, Y.; Yan, J.; Ingle, T.; Jones, M.Y.; Mei, N.; Boudreau, M.D.; Cunningham, C.K.; Abbas, M.; Paredes, A.M.; et al. Size- and coating-dependent cytotoxicity and genotoxicity of silver nanoparticles evaluated using in vitro standard assays. Nanotoxicology 2016, 10, 1373–1384. [Google Scholar] [CrossRef]
- Sahu, S.C.; Njoroge, J.; Bryce, S.M.; Yourick, J.J.; Sprando, R.L. Comparative genotoxicity of nanosilver in human liver HepG2 and colon Caco2 cells evaluated by a flow cytometric in vitro micronucleus assay. J. Appl. Toxicol. 2014, 34, 1226–1234. [Google Scholar] [CrossRef]
- Ivask, A.; Voelcker, N.H.; Seabrook, S.A.; Hor, M.; Kirby, J.K.; Fenech, M.; Davis, T.P.; Ke, P.C. DNA Melting and Genotoxicity Induced by Silver Nanoparticles and Graphene. Chem. Res. Toxicol. 2015, 28, 1023–1035. [Google Scholar] [CrossRef]
- Sahu, S.C.; Roy, S.; Zheng, J.; Ihrie, J. Contribution of ionic silver to genotoxic potential of nanosilver in human liver HepG2 and colon Caco2 cells evaluated by the cytokinesis-block micronucleus assay. J. Appl. Toxicol. 2016, 36, 532–542. [Google Scholar] [CrossRef] [PubMed]
- Sahu, S.C.; Njoroge, J.; Bryce, S.M.; Zheng, J.; Ihrie, J. Flow cytometric evaluation of the contribution of ionic silver to genotoxic potential of nanosilver in human liver HepG2 and colon Caco2 cells. J. Appl. Toxicol. 2016, 36, 521–531. [Google Scholar] [CrossRef]
- Butler, K.S.; Peeler, D.J.; Casey, B.J.; Dair, B.J.; Elespuru, R.K. Silver nanoparticles: Correlating nanoparticle size and cellular uptake with genotoxicity. Mutagenesis 2015, 30, 577–591. [Google Scholar] [CrossRef] [PubMed]
- Lebedová, J.; Hedberg, Y.S.; Odnevall Wallinder, I.; Karlsson, H.L. Size-dependent genotoxicity of silver, gold and platinum nanoparticles studied using the mini-gel comet assay and micronucleus scoring with flow cytometry. Mutagenesis 2018, 33, 77–85. [Google Scholar] [CrossRef]
- Wang, J.; Che, B.; Zhang, L.W.; Dong, G.; Luo, Q.; Xin, L. Comparative genotoxicity of silver nanoparticles in human liver HepG2 and lung epithelial A549 cells. J. Appl. Toxicol. 2017, 37, 495–501. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Qin, T.; Ingle, T.; Yan, J.; He, W.; Yin, J.-J.; Chen, T. Differential genotoxicity mechanisms of silver nanoparticles and silver ions. Arch. Toxicol. 2017, 91, 509–519. [Google Scholar] [CrossRef] [PubMed]
- Souza, T.A.J.; Franchi, L.P.; Rosa, L.R.; da Veiga, M.A.M.S.; Takahashi, C.S. Cytotoxicity and genotoxicity of silver nanoparticles of different sizes in CHO-K1 and CHO-XRS5 cell lines. Mutat. Res. Toxicol. Environ. Mutagen. 2016, 795, 70–83. [Google Scholar] [CrossRef]
- Jiang, X.; Foldbjerg, R.; Miclaus, T.; Wang, L.; Singh, R.; Hayashi, Y.; Sutherland, D.; Chen, C.; Autrup, H.; Beer, C. Multi-platform genotoxicity analysis of silver nanoparticles in the model cell line CHO-K1. Toxicol. Lett. 2013, 222, 55–63. [Google Scholar] [CrossRef]
- Che, B.; Luo, Q.; Zhai, B.; Fan, G.; Liu, Z.; Cheng, K.; Xin, L. Cytotoxicity and genotoxicity of nanosilver in stable GADD45α promoter-driven luciferase reporter HepG2 and A549 cells. Environ. Toxicol. 2017, 32, 2203–2211. [Google Scholar] [CrossRef]
- Wang, X.; Li, T.; Su, X.; Li, J.; Li, W.; Gan, J.; Wu, T.; Kong, L.; Zhang, T.; Tang, M.; et al. Genotoxic effects of silver nanoparticles with/without coating in human liver HepG2 cells and in mice. J. Appl. Toxicol. 2019, 39, 908–918. [Google Scholar] [CrossRef] [PubMed]
- Choo, W.; Moon, B.; Song, S.; Oh, S.M. Morphological transformation induced by silver nanoparticles in a Balb/c 3T3 A31-1-1 mouse cell model to evaluate in vitro carcinogenic potential. Environ. Health Toxicol. 2017, 32, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Azqueta, A.; Dusinska, M. The use of the comet assay for the evaluation of the genotoxicity of nanomaterials. Front. Genet. 2015, 6, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Ávalos, A.; Haza, A.I.; Morales, P. Manufactured silver nanoparticles of different sizes induced DNA strand breaks and oxidative DNA damage in hepatoma and leukaemia cells and in dermal and pulmonary fibroblasts. Folia Biol. 2015, 61, 33–42. [Google Scholar]
- Martinez Paino, I.M.; Zucolotto, V. Poly(vinyl alcohol)-coated silver nanoparticles: Activation of neutrophils and nanotoxicology effects in human hepatocarcinoma and mononuclear cells. Environ. Toxicol. Pharmacol. 2015, 39, 614–621. [Google Scholar] [CrossRef] [PubMed]
- Brkić Ahmed, L.; Milić, M.; Pongrac, I.M.; Marjanović, A.M.; Mlinarić, H.; Pavičić, I.; Gajović, S.; Vinković Vrček, I. Impact of surface functionalization on the uptake mechanism and toxicity effects of silver nanoparticles in HepG2 cells. Food Chem. Toxicol. 2017, 107, 349–361. [Google Scholar] [CrossRef]
- Farah, M.A.; Ali, M.A.; Chen, S.-M.; Li, Y.; Al-Hemaid, F.M.; Abou-Tarboush, F.M.; Al-Anazi, K.M.; Lee, J. Silver nanoparticles synthesized from Adenium obesum leaf extract induced DNA damage, apoptosis and autophagy via generation of reactive oxygen species. Colloids Surf. B-Biointerfaces 2016, 141, 158–169. [Google Scholar] [CrossRef]
- Gliga, A.R.; Skoglund, S.; Odnevall Wallinder, I.; Fadeel, B.; Karlsson, H.L. Size-dependent cytotoxicity of silver nanoparticles in human lung cells: The role of cellular uptake, agglomeration and Ag release. Part. Fibre Toxicol. 2014, 11, 11. [Google Scholar] [CrossRef]
- Milić, M.; Leitinger, G.; Pavičić, I.; Zebić Avdičević, M.; Dobrović, S.; Goessler, W.; Vinković Vrček, I. Cellular uptake and toxicity effects of silver nanoparticles in mammalian kidney cells. J. Appl. Toxicol. 2015, 35, 581–592. [Google Scholar] [CrossRef]
- Jiravova, J.; Tomankova, K.B.; Harvanova, M.; Malina, L.; Malohlava, J.; Luhova, L.; Panacek, A.; Manisova, B.; Kolarova, H. The effect of silver nanoparticles and silver ions on mammalian and plant cells in vitro. Food Chem. Toxicol. 2016, 96, 50–61. [Google Scholar] [CrossRef]
- Juarez-Moreno, K.; Gonzalez, E.B.; Girón-Vazquez, N.; Chávez-Santoscoy, R.A.; Mota-Morales, J.D.; Perez-Mozqueda, L.L.; Garcia-Garcia, M.R.; Pestryakov, A.; Bogdanchikova, N. Comparison of cytotoxicity and genotoxicity effects of silver nanoparticles on human cervix and breast cancer cell lines. Hum. Exp. Toxicol. 2017, 36, 931–948. [Google Scholar] [CrossRef] [PubMed]
- Castiglioni, S.; Caspani, C.; Cazzaniga, A.; Maier, J.A. Short-and long-term effects of silver nanoparticles on human microvascular endothelial cells. World J Biol Chem 2014, 5, 457–464. [Google Scholar] [CrossRef] [PubMed]
- Castro-Gamboa, S.; Garcia-Garcia, M.R.; Piñon-Zarate, G.; Rojas-Lemus, M.; Jarquin-Yañez, K.; Angel Herrera-Enriquez, M.; Fortoul, T.I.; Toledano-Magaña, Y.; Garcia-Iglesias, T.; Pestryakov, A.; et al. Toxicity of silver nanoparticles in mouse bone marrow-derived dendritic cells: Implications for phenotype. J. Immunotoxicol. 2019, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kukut Hatipoglu, M.; Keleştemur, S.; Altunbek, M.; Culha, M. Source of cytotoxicity in a colloidal silver nanoparticle suspension. Nanotechnology 2015, 26, 195103. [Google Scholar] [CrossRef] [PubMed]
- Franchi, L.P.; Matsubara, E.Y.; Soenen, S.J.; Takahashi, C.S.; Manshian, B.B.; de Souza, T.A.J.; Rosolen, J.M. Cyto- and genotoxic effects of metallic nanoparticles in untransformed human fibroblast. Toxicol. Vitr. 2015, 29, 1319–1331. [Google Scholar] [CrossRef]
- Asare, N.; Duale, N.; Slagsvold, H.H.; Lindeman, B.; Olsen, A.K.; Gromadzka-Ostrowska, J.; Meczynska-Wielgosz, S.; Kruszewski, M.; Brunborg, G.; Instanes, C. Genotoxicity and gene expression modulation of silver and titanium dioxide nanoparticles in mice. Nanotoxicology 2016, 10, 312–321. [Google Scholar] [CrossRef]
- Li, Y.; Bhalli, J.A.; Ding, W.; Yan, J.; Pearce, M.G.; Sadiq, R.; Cunningham, C.K.; Jones, M.Y.; Monroe, W.A.; Howard, P.C.; et al. Cytotoxicity and genotoxicity assessment of silver nanoparticles in mouse. Nanotoxicology 2014, 8, 36–45. [Google Scholar] [CrossRef]
- Patlolla, A.K.; Hackett, D.; Tchounwou, P.B. Genotoxicity study of silver nanoparticles in bone marrow cells of Sprague–Dawley rats. Food Chem. Toxicol. 2015, 85, 52–60. [Google Scholar] [CrossRef]
- Nallanthighal, S.; Chan, C.; Murray, T.M.; Mosier, A.P.; Cady, N.C.; Reliene, R. Differential effects of silver nanoparticles on DNA damage and DNA repair gene expression in Ogg1-deficient and wild type mice. Nanotoxicology 2017, 11, 996–1011. [Google Scholar] [CrossRef]
- Dobrzyńska, M.M.; Gajowik, A.; Radzikowska, J.; Lankoff, A.; Dušinská, M.; Kruszewski, M. Genotoxicity of silver and titanium dioxide nanoparticles in bone marrow cells of rats in vivo. Toxicology 2014, 315, 86–91. [Google Scholar] [CrossRef]
- El Mahdy, M.M.; Eldin, T.A.S.; Aly, H.S.; Mohammed, F.F.; Shaalan, M.I. Evaluation of hepatotoxic and genotoxic potential of silver nanoparticles in albino rats. Exp. Toxicol. Pathol. 2015, 67, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Kant Awasthi, K.; Verma, R.; Awasthi, A.; Awasthi, K.; Soni, I.J.; John, P. In Vivo Genotoxic Assessment Of Silver Nanoparticles In Liver Cells Of Swiss Albino Mice Using Comet Assay. Adv. Mater. Lett. 2015, 6, 187–193. [Google Scholar] [CrossRef]
- Kim, Y.J.; Rahman, M.M.; Lee, S.M.; Kim, J.M.; Park, K.; Kang, J.-H.; Seo, Y.R. Assessment of in vivo genotoxicity of citrated-coated silver nanoparticles via transcriptomic analysis of rabbit liver tissue. Int. J. Nanomed. 2019, 14, 393–405. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.D.C.; Azevedo, L.F.; de Souza Rocha, C.C.; Carneiro, M.F.H.; Venancio, V.P.; de Almeida, M.R.; Antunes, L.M.G.; de Carvalho Hott, R.; Rodrigues, J.L.; Ogunjimi, A.T.; et al. Evaluation of distribution, redox parameters, and genotoxicity in Wistar rats co-exposed to silver and titanium dioxide nanoparticles. J. Toxicol. Environ. Heal. Part A 2017, 80, 1156–1165. [Google Scholar] [CrossRef]
- Van der Zande, M.; Vandebriel, R.J.; Van Doren, E.; Kramer, E.; Herrera Rivera, Z.; Serrano-Rojero, C.S.; Gremmer, E.R.; Mast, J.; Peters, R.J.B.; Hollman, P.C.H.; et al. Distribution, Elimination, and Toxicity of Silver Nanoparticles and Silver Ions in Rats after 28-Day Oral Exposure. ACS Nano 2012, 6, 7427–7442. [Google Scholar] [CrossRef] [PubMed]
- Dakal, T.C.; Kumar, A.; Majumdar, R.S.; Yadav, V. Mechanistic Basis of Antimicrobial Actions of Silver Nanoparticles. Front. Microbiol. 2016, 7, 1–17. [Google Scholar] [CrossRef]
- Burdușel, A.-C.; Gherasim, O.; Grumezescu, A.M.; Mogoantă, L.; Ficai, A.; Andronescu, E. Biomedical Applications of Silver Nanoparticles: An Up-to-Date Overview. Nanomaterials 2018, 8, 681. [Google Scholar] [CrossRef]
- Kim, S.-H.; Lee, H.; Ryu, D.; Choi, S.; Lee, D. Antibacterial Activity of Silver-Nanoparticles Against Staphylococcus Aureus Antibacterial Activity of Silver-nanoparticles Against Staphylococcus aureus and Escherichia coli. Korean J. Microbiol. Biotechnol. 2011, 39, 77–85. [Google Scholar]
- McShan, D.; Ray, P.C.; Yu, H. Molecular toxicity mechanism of nanosilver. J. Food Drug Anal. 2014, 22, 116–127. [Google Scholar] [CrossRef]
- AshaRani, P.V.; Mun, G.L.K.; Hande, M.P.; Valiyaveettil, S. Cytotoxicity and genotoxicity of silver nanoparticles in human cells. ACS Nano 2009, 3, 279–290. [Google Scholar] [CrossRef]
- Mahmoudi, M.; Lynch, I.; Ejtehadi, M.R.; Monopoli, M.P.; Bombelli, F.B.; Laurent, S. Protein−Nanoparticle Interactions: Opportunities and Challenges. Chem. Rev. 2011, 111, 5610–5637. [Google Scholar] [CrossRef] [PubMed]
- Tee, J.K.; Ong, C.N.; Bay, B.H.; Ho, H.K.; Leong, D.T. Oxidative stress by inorganic nanoparticles. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2016, 8, 414–438. [Google Scholar] [CrossRef]
- Salim, E.; Abdel-Halim, K.; Abu-Risha, S.; Abdel-Latif, A. Induction of 8-hydroxydeoxyguanosine and ultrastructure alterations by silver nanoparticles attributing to placental transfer in pregnant rats and fetuses. Hum. Exp. Toxicol. 2019, 38, 734–745. [Google Scholar] [CrossRef] [PubMed]
- Kain, J.; Karlsson, H.L.; Moller, L. DNA damage induced by micro- and nanoparticles--interaction with FPG influences the detection of DNA oxidation in the comet assay. Mutagenesis 2012, 27, 491–500. [Google Scholar] [CrossRef] [PubMed]
- Magdolenova, Z.; Collins, A.; Kumar, A.; Dhawan, A.; Stone, V.; Dusinska, M. Mechanisms of genotoxicity. A review of in vitro and in vivo studies with engineered nanoparticles. Nanotoxicology 2014, 8, 233–278. [Google Scholar] [CrossRef] [PubMed]
- Sotiriou, G.A.; Teleki, A.; Camenzind, A.; Krumeich, F.; Meyer, A.; Panke, S.; Pratsinis, S.E. Nanosilver on nanostructured silica: Antibacterial activity and Ag surface area. Chem. Eng. J. 2011, 170, 547–554. [Google Scholar] [CrossRef] [PubMed]
- Rageh, M.M.; El-Gebaly, R.H.; Afifi, M.M. Antitumor activity of silver nanoparticles in Ehrlich carcinoma-bearing mice. Naunyn. Schmiedebergs. Arch. Pharmacol. 2018, 391, 1421–1430. [Google Scholar] [CrossRef]

| NP Size (nm) | Coating | Cells | H | µg/mL | Result | Ref. |
|---|---|---|---|---|---|---|
| 20 | Citrate | L5178Y | 4 | 1–60 | + | [72] |
| PVP | + | |||||
| 50 | Citrate | + | ||||
| PVP | + | |||||
| 100 | Citrate | + | ||||
| PVP | + |
| NP Size (nm) | Coating | Cells | H | μg/mL | Result | Ref. |
|---|---|---|---|---|---|---|
| 5 | HBEC | 48 | 1–20 | - | [78] | |
| TK6 | 28 | 1–1.5 | + | [80] | ||
| 10 | CHO-K1 | 24 | 0.025–2.5 | + | [81] | |
| CHO-XRS5 | - | |||||
| JURKAT E61 | 24 | 1–25 | + | [77] | ||
| TPH-1 | + | |||||
| 15 | MCF-10A | 24 | 5.9–23.5 | - | [52] | |
| MCF-7 | 48 | 4.1-16.3 | - | |||
| MDA-MB-231 | 1.2–4.9 | - | ||||
| CHO-K1 | 24 | 1–5–10 | + | [82] | ||
| 20–50 | HepG2 | 48 | 12.5–200 | + | [83] | |
| A549 | + | |||||
| HepG2 | 24 | 12.5–200 | + | [79] | ||
| A549 | + | |||||
| 20 | JURKAT | 24 | 1–25 | + | [77] | |
| TPH-1 | + | |||||
| HepG2 (s) | 48 | 5–15 | - | [73] | ||
| CACO2(s) | - | |||||
| HepG2 (a) | 1–10 | + | ||||
| CACO2 (a) | - | |||||
| HepG2 | 24 | 20–160 | + | [84] | ||
| PVP | + | |||||
| Citrate | Lymphocytes | 44 | 0.2–25 | + | [74] | |
| bPEI | + | |||||
| Citrate | JURKAT | 24 | 0.1–25 | + | ||
| bPEI | + | |||||
| Citrate | WIL2-NS | + | ||||
| bPEI | + | |||||
| Citrate | L5178Y | 4 | 1.25–4 | + | [72] | |
| TK6 | 2.5–15 | + | ||||
| PVP | L5178Y | 1.25–8 | + | |||
| TK6 | 2.5–30 | + | ||||
| 30 | Citrate | HaCat | 24 | 10–40 | + | [31] |
| 48 | + | |||||
| 45 | MCF-10A | 24 | 5.9–23.5 | - | [52] | |
| MCF-7 | 48 | 4.1–16.3 | - | |||
| MDA-MB-231 | 1.2–4.9 | - | ||||
| 50 | HBEC | 48 | 1–20 | - | [78] | |
| HepG2 | 4 | 10–100 | - | [75] | ||
| 24 | 2.5–25 | - | ||||
| CACO2 | 4 | 10–100 | - | |||
| 24 | 2.5–25 | - | ||||
| HepG2 | 40–44 | 1–20 | + | [76] | ||
| CACO2 | - | |||||
| JURKAT | 24 | 1–50 | + | [77] | ||
| TPH-1 | + | |||||
| Citrate | L5178Y | 4 | 1.25–20 | + | [72] | |
| TK6 | 2.5–120 | - | ||||
| PVP | L5178Y | 1.25–30 | + | |||
| TK6 | 2.5–140 | - | ||||
| 90 | Balb3T3 A31-1-1 | 24 | 0.17–10.60 | + | [85] | |
| 100 | CHO-K1 | 24 | 0.025–2.5 | - | [81] | |
| CHO-XRS5 | + | |||||
| JURKAT | 24 | 1–50 | + | [77] | ||
| TPH-1 | + | |||||
| Citrate | L5178Y | 4 | 1.25–35 | + | [72] | |
| TK6 | 2.5–400 | + | ||||
| PVP | L5178Y | 1.25–50 | + | |||
| TK6 | 2.5–400 | + |
| NP Size (nm) | Coating | Cells | H | l | SC | Fpg | Endo-III | OGG-1 | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| 5 | HBEC | 48 | 1–20 | + | [78] | ||||
| PEI + PVP | HepG2 | 24 | 0.1–1.6 | + | + | - | [87] | ||
| HL-60 | + | + | + | ||||||
| NHDF | + | + | + | ||||||
| HPF | + | + | + | ||||||
| 5–15 | PVA | Blood cells | 4 | 1–50 (μM *) | + | [88] | |||
| HepG2 | + | ||||||||
| 5–20 | AOT | HepG2 | 24 | 1–10 | + | [89] | |||
| CTAB | + | ||||||||
| PVP | + | ||||||||
| BSA | + | ||||||||
| PLL | + | ||||||||
| 10–30 | MCF-7 | 24 | 5–150 | + | [90] | ||||
| 10 | CACO2 | 24 | 1–50 | - | + | [53] | |||
| CHO-K1 | 24 | 0.025–2.5 | + | [81] | |||||
| CHO-XRS5 | + | ||||||||
| JURKAT E6-1 | 24 | 2.5–20 | + | [77] | |||||
| TPH-1 | 1–25 | + | |||||||
| PVP | BEAS-2B | 4 | 10 | - | [91] | ||||
| 24 | + | ||||||||
| Citrate | 4 | - | |||||||
| 24 | + | ||||||||
| 13–60 | Citrate | PK15 | 24 | 1–75 | + | [92] | |||
| 48 | + | ||||||||
| 15 | MCF-10A | 24 | 5.9–23.5 | + | - | [52] | |||
| MCF-7 | 4.1–16.3 | + | - | ||||||
| MDA-MB-231 | 1.2–4.9 | - | - | ||||||
| 20–50 | HepG2 | 48 | 12.5–200 | + | [83] | ||||
| A549 | + | ||||||||
| HepG2 | 24 | 12.5–200 | + | ||||||
| A549 | + | ||||||||
| 20 | JURKAT E6-1 | 24 | 5–25 | + | [77] | ||||
| TPH-1 | 5–40 | + | |||||||
| HepG2 | 20–160 | + | [84] | ||||||
| PVP | + | ||||||||
| 27 | NIH3T3 | 6 | 30.1–90.1 | + | [93] | ||||
| SVK14 | 25.4–76.1 | - | |||||||
| 30 | Citrate | HaCat | 24 | 10–40 | + | [31] | |||
| 48 | + | ||||||||
| 35 | PVP | Hela | 12 | 1.25–10 | + | [94] | |||
| 24 | + | ||||||||
| MDA-MB-231 | 12 | + | |||||||
| 24 | + | ||||||||
| MCF-7 | 12 | + | |||||||
| 24 | + | ||||||||
| HMEC | 24 h | 2.5–5 | + | [95] | |||||
| ECFC | + | ||||||||
| BMDC | 12 | 0.03–1 | + | [96] | |||||
| 40 | HepG2 | 24 | 0.1–6.7 | + | + | + | [87] | ||
| HL-60 | + | + | + | ||||||
| NHDF | + | + | + | ||||||
| HPF | + | - | - | ||||||
| Citrate | BEAS-2B | 4 | 10 | - | [91] | ||||
| 24 | + | ||||||||
| 45 | MCF-10A | 24 | 5.9–23.5 | - | - | [52] | |||
| MCF-7 | - | - | |||||||
| MDA-MB-231 | 2–8.1 | - | - | ||||||
| 50 | HBEC | 48 | 1–20 | + | [78] | ||||
| JURKAT E6-1 | 24 | 10–50 | - | [77] | |||||
| TPH-1 | - | ||||||||
| BEAS-2B | 4 | 10 | - | [91] | |||||
| 24 | + | ||||||||
| 56.4 | A549 | 24 | 10–50 | + | [97] | ||||
| 48 | + | ||||||||
| 60 | HEK293T | 24 | 10–40 | + | [82] | ||||
| 60–105 | A549 | 24 | 25 | - | [97] | ||||
| 75 | Citrate | BEAS-2B | 4 | 10 | - | [91] | |||
| 24 | + | ||||||||
| 100 | CHO-K1 | 24 | 0.025–2.5 | + | [81] | ||||
| CHO-XRS5 | + | ||||||||
| PVP | GMO7492 | 24 | 0.01–10 | + | + | [98] | |||
| JURKAT E6-1 | 24 | 10–50 | - | [77] | |||||
| TPH-1 | - | ||||||||
| 105 | NIH3T3 | 6 | 1.3 | + | [99] | ||||
| SVK14 | 2.1 | + | |||||||
| BJ | 2.2 | + | |||||||
| 131.5 | NIH3T3 | 6 | 1.4 | + | |||||
| SVK14 | 2.2 | + | |||||||
| BJ | 2.3 | + |
| NPs Size (nm) | Coating | Animal Model | Tissue | Route | Duration | Dose (mg/kg Body Weight) | ST | Result | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| 5 | PVP | B6C3F1 mouse | Blood | i.v. | Single dose | 0.5–20 | 2 d | - | [100] |
| 10 | - | Sprague Dawley rat | Bone marrow | p.o. | 5 day | 5–100 | 1 d | + | [101] |
| 20 | Citrate | OGG1 −/− KO C57BL/6 mouse | Blood | p.o. | 3 day | 4 | 1 d | + | [102] |
| 7 day | + | ||||||||
| C57BL/6 mouse | 3 day | + | |||||||
| 7 day | + | ||||||||
| Citrate | C57BL/6 mouse | Blood | p.o. | 7 day | 4 | 1 d | + | [32] | |
| 1 w | + | ||||||||
| 2 w | + | ||||||||
| PVP | 1 d | - | |||||||
| 1 w | - | ||||||||
| 2 w | - | ||||||||
| Wistar rat | Bone marrow | i.v. | Single dose | 5 | 1 d | + | [103] | ||
| 1 w | + | ||||||||
| 4 w | - | ||||||||
| 10 | 1 d | + | |||||||
| 1 w | + | ||||||||
| 4 w | + | ||||||||
| ICR mouse | Bone marrow | p.o. | 28 day | 10–250 | 1 d | + | [84] | ||
| PVP | + | ||||||||
| 10-80 | Silicon | B6C3F1 mouse | Liver | i.v | Single dose | 25 | 2d | - | [100] |
| 3 day | - | ||||||||
| 200 | Wistar rat | Bone marrow | i.v. | Single dose | 5 | 1d | + | [103] | |
| 1w | + | ||||||||
| 4w | - | ||||||||
| 6.3-629 | Sprague Dawley rat | Bone marrow | i.v. | Single dose | 5 | 1d | - | [41] | |
| 15-100 | PVP | B6C3F1 mouse | Liver | i.v. | Single dose | 25 | 2d | - | [100] |
| - |
| Size (nm) | Coating | Animal Model | Tissue | Route | Duration | Dose (mg/kg Body Weight) | ST | Result | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| 10 | Sprague Dawley rat | Bone marrow | p.o. | 5 day | 5–100 | 1 d | + | [101] | |
| Albino rat | Bone marrow | i.p. | 28 day | 1–4 | 0 d | + | [104] | ||
| 6–629 | Sprague Dawley rat | Bone marrow | i.v. | Single dose | 5 | 1 d | + | [41] |
| NPs Size (nm) | Coating | Animal Model | Tissue | Route | Duration | Dose (mg/kg Body Weight) | ST | SC | Fpg | Endo-III | OGG-1 | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5 | Swiss albino mouse | Liver | p.o. | Single dose | 5–100 | 3 h | + | [105] | ||||
| 1 d | + | |||||||||||
| p.o.w. | 35 day | 10–20 | 3 h | + | ||||||||
| 8 | Citrate | New Zeland white rabbit | Liver | i.v. | Single dose | 0.5–5 | 7 d | + | [106] | |||
| 28 d | + | |||||||||||
| 10 | Sprague Dawley rat | Bone marrow | p.o. | 5 day | 5–100 | 0 | + | [101] | ||||
| 10-80 | Silicon | B6C3F1 mouse | Liver | i.v. | 3 day | 25 | 3 h | - | + | + | [100] | |
| 20 | Wistar rat | Bone marrow | i.v. | Single dose | 5 | 1 d | - | [103] | ||||
| 1 w | - | |||||||||||
| 4 w | - | |||||||||||
| 10 | 1 d | - | ||||||||||
| 1 w | - | |||||||||||
| 4 w | - | |||||||||||
| OGG1 −/− KO C57BL/6 mouse | Lung | i.v. | Single dose | 5 | 1 d | - | - | [99] | ||||
| 1 w | - | - | ||||||||||
| Liver | 1 d | - | - | |||||||||
| 1 w | - | - | ||||||||||
| Testis | 1 d | - | - | |||||||||
| 1 w | - | - | ||||||||||
| C57BL/6 mouse | Lung | 1 d | - | - | ||||||||
| 1 w | - | - | ||||||||||
| Liver | 1 d | - | - | |||||||||
| 1 w | - | - | ||||||||||
| Testis | 1 d | - | - | |||||||||
| 1 w | - | - | ||||||||||
| 90 | Wistar rat | Blood | p.o. | 45 day | 0.5 | 0 | - | [107] | ||||
| Liver | 0 | - | ||||||||||
| 15–100 | PVP | B6C3F1 mouse | Liver | i.v. | 3 day | 25 | 3 h | - | + | + | [100] | |
| 200 | Wistar rat | Bone marrow | i.v. | Single dose | 5 | 1 d | - | [106] | ||||
| 1 w | - | |||||||||||
| 4 w | - | |||||||||||
| OGG1 −/− KO C57BL/6 mouse | Lung | i.v. | Single dose | 5 | 1 d | - | - | [101] | ||||
| 1 w | + | - | ||||||||||
| Liver | 1 d | - | - | |||||||||
| 1 w | - | - | ||||||||||
| Testis | 1 d | - | - | |||||||||
| 1 w | - | - | ||||||||||
| C57BL/6 mouse | Lung | 1 d | - | - | ||||||||
| 1 w | - | + | ||||||||||
| Liver | 1 d | - | - | |||||||||
| 1 w | - | - | ||||||||||
| Testis | 1 d | - | - | |||||||||
| 1 w | - | + |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodriguez-Garraus, A.; Azqueta, A.; Vettorazzi, A.; López de Cerain, A. Genotoxicity of Silver Nanoparticles. Nanomaterials 2020, 10, 251. https://doi.org/10.3390/nano10020251
Rodriguez-Garraus A, Azqueta A, Vettorazzi A, López de Cerain A. Genotoxicity of Silver Nanoparticles. Nanomaterials. 2020; 10(2):251. https://doi.org/10.3390/nano10020251
Chicago/Turabian StyleRodriguez-Garraus, Adriana, Amaya Azqueta, Ariane Vettorazzi, and Adela López de Cerain. 2020. "Genotoxicity of Silver Nanoparticles" Nanomaterials 10, no. 2: 251. https://doi.org/10.3390/nano10020251
APA StyleRodriguez-Garraus, A., Azqueta, A., Vettorazzi, A., & López de Cerain, A. (2020). Genotoxicity of Silver Nanoparticles. Nanomaterials, 10(2), 251. https://doi.org/10.3390/nano10020251






